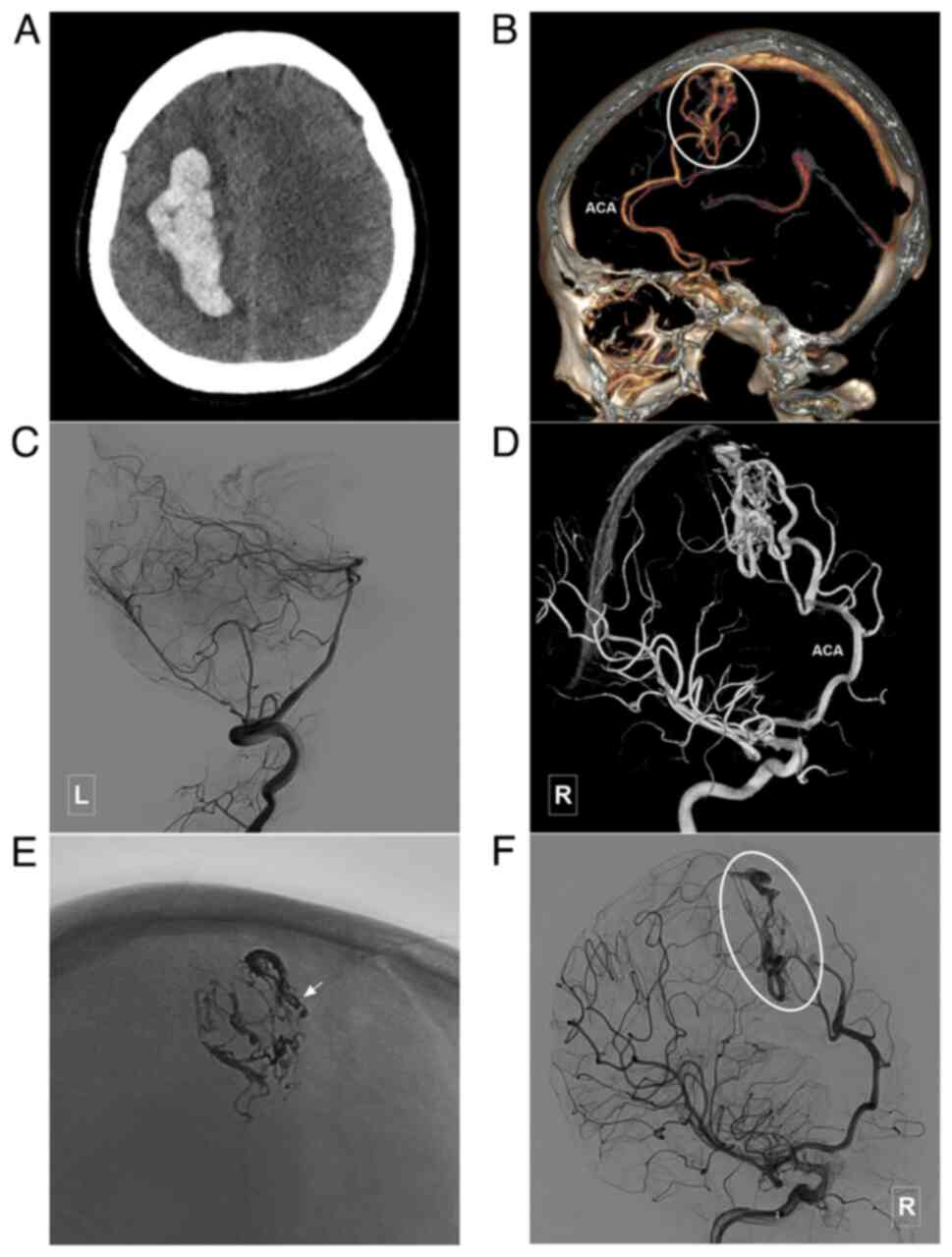
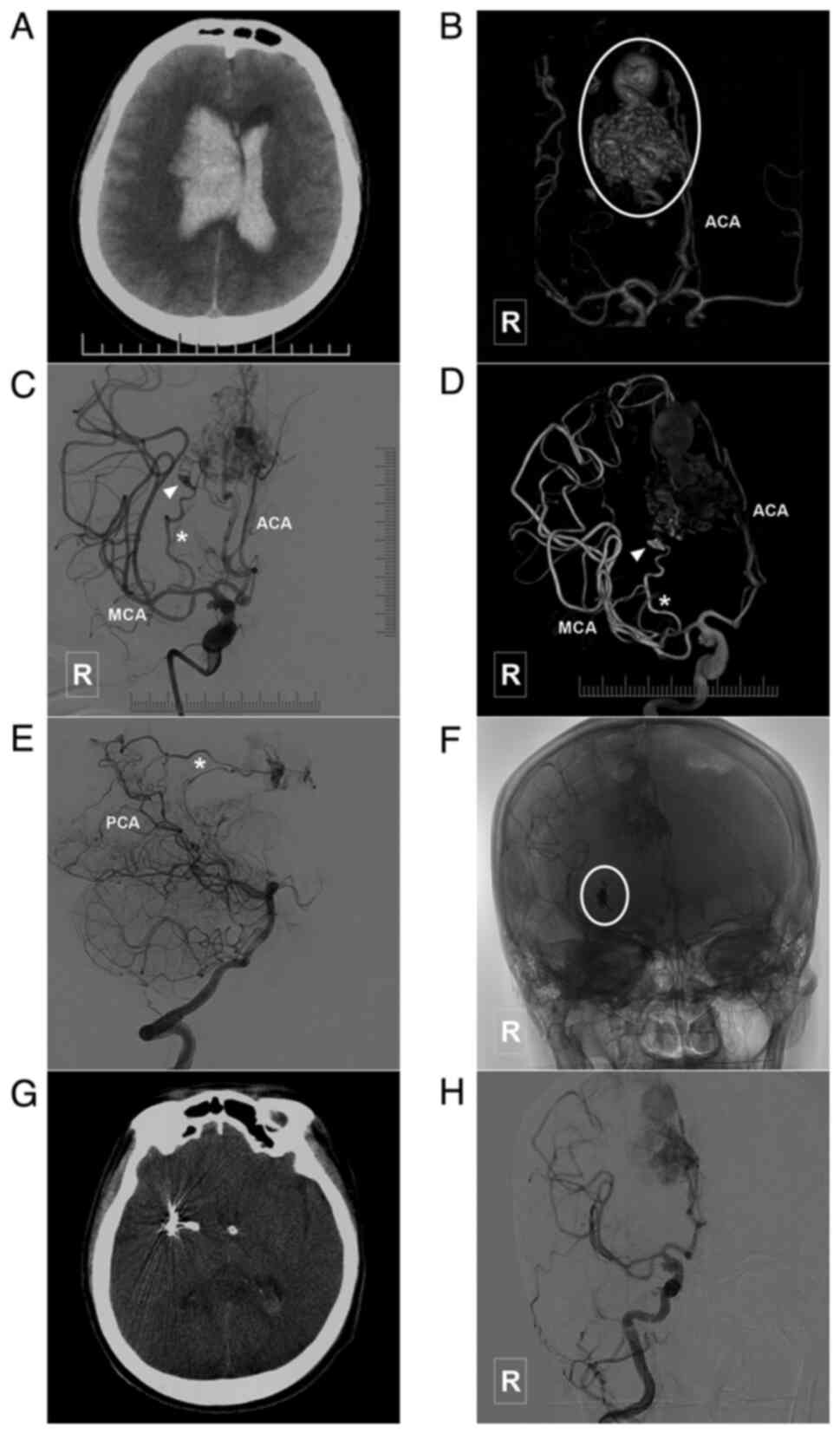
Introduction
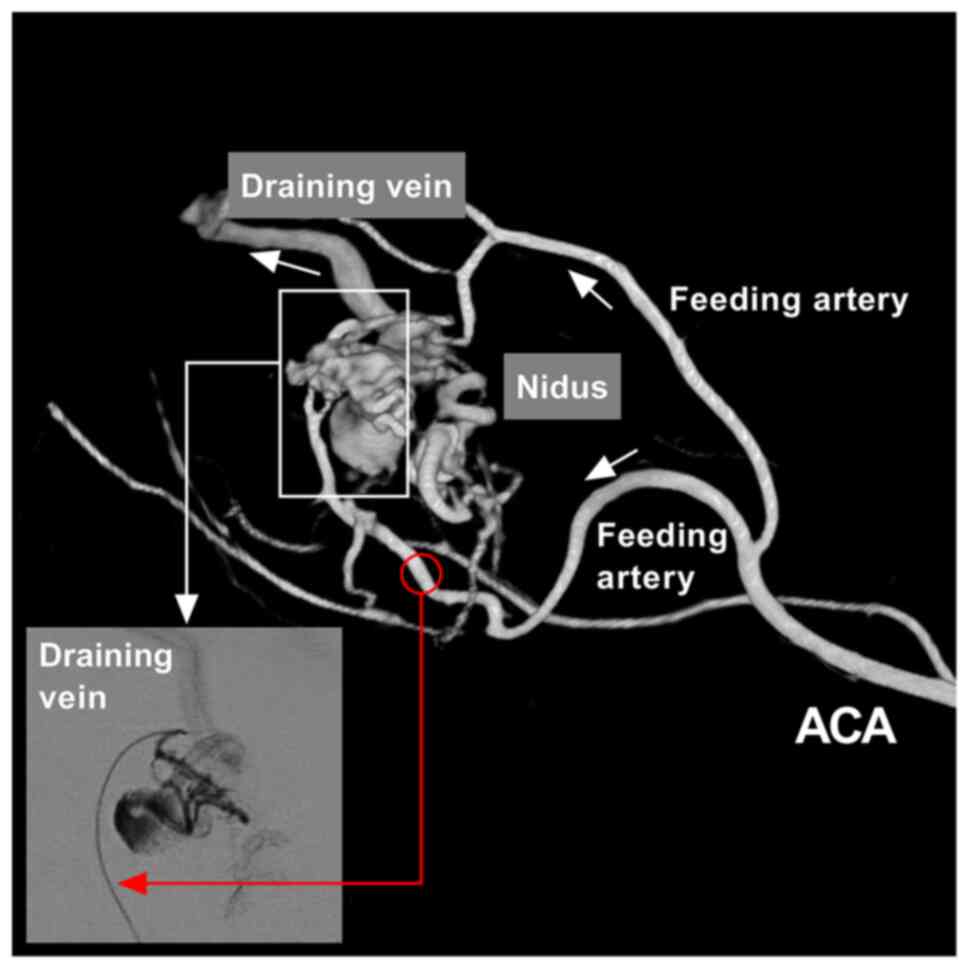
Brain arteriovenous malformation (BAVM) is a common
vascular disease. During the progression of BAVM, the feeding
artery may become progressively dilated, thinned and tortuous. The
nidus undergoes dynamic remodeling, during the process of which
thrombosis or rupture can occur. In addition, the draining vein can
become thickened and dilatated due to the continuous high pressure
of blood from the feeding arteries (1,2). The
typical angioarchitecture of BAVM is illustrated in Fig. 1. However, BAVMs at different sites
have different characteristics (3).
BAVMs can occur in the supplying area of the
anterior cerebral artery (ACA; thus termed ACA-BAVMs), which is
located at the para-midline region of the cerebral hemispheres and
is close to the medial veins of the frontal lobe. The posterior
part of the ACA is closely associated with the deep cerebral venous
system (4). ACA-BAVMs are
distributed above the corpus callosum and may be supplied by the
A2-A5 segments of the ACA (5).
Currently, endovascular treatment (EVT) is the
preferred treatment choice for BAVMs, both as a curative or
adjunctive treatment. However, to date, to the best of our
knowledge, there are only limited studies available evaluating the
safety and efficacy of EVT for ACA-BAVMs. Thus, the present study
retrospectively examined the safety of EVT for ACA-BAVMs in a total
of 60 patients. The results suggest that EVT may be a safe
treatment method for ACA-BAVMs.
Patients and methods
A total of 60 patients with ACA-BAVMs treated with
EVT from January, 2012 to January, 2020 were retrospectively
identified at the First Hospital of Jilin University, Changchun,
China, and analyzed. The present study was approved by the Ethics
Committee of the First Hospital of Jilin University (approval no.
2020-341). Written informed consent was obtained from the patients
or their legal relatives.
Inclusion criteria
The inclusion criteria were as follows: i) The BAVM
was located above the corpus callosum, at or close to the midline
of the ACA supplied area; ii) the ACA was the main (if not the
only) source of blood supply; iii) the patients had not undergone
any EVT, open surgery, or radiosurgery prior to admission to the
First Hospital of Jilin University.
ACA-BAVM classification
The ACA-BAVMs were classified according to the
anatomical association with the corpus callosum and ACA (Fig. 2). Type I BAVMs were defined as those
located at the first segment in Fig.
2, which refers to the area below and in front of the corpus
callosum genu (the supplying area of the A2 segment of the ACA).
Type II BAVMs were defined as those located at the second segment
in Fig. 2, which refers to the upper
area of the corpus callosum from the genu to the anterior portion
of corpus callosum body (the supplying area of the A3 segment of
the ACA). Type III BAVMs were defined as those located at the third
segment in Fig. 2, which refers to
the area from the anterior portion of corpus callosum body to the
splenium of the corpus callosum (the supplying area of the A4-5
segment of the ACA).
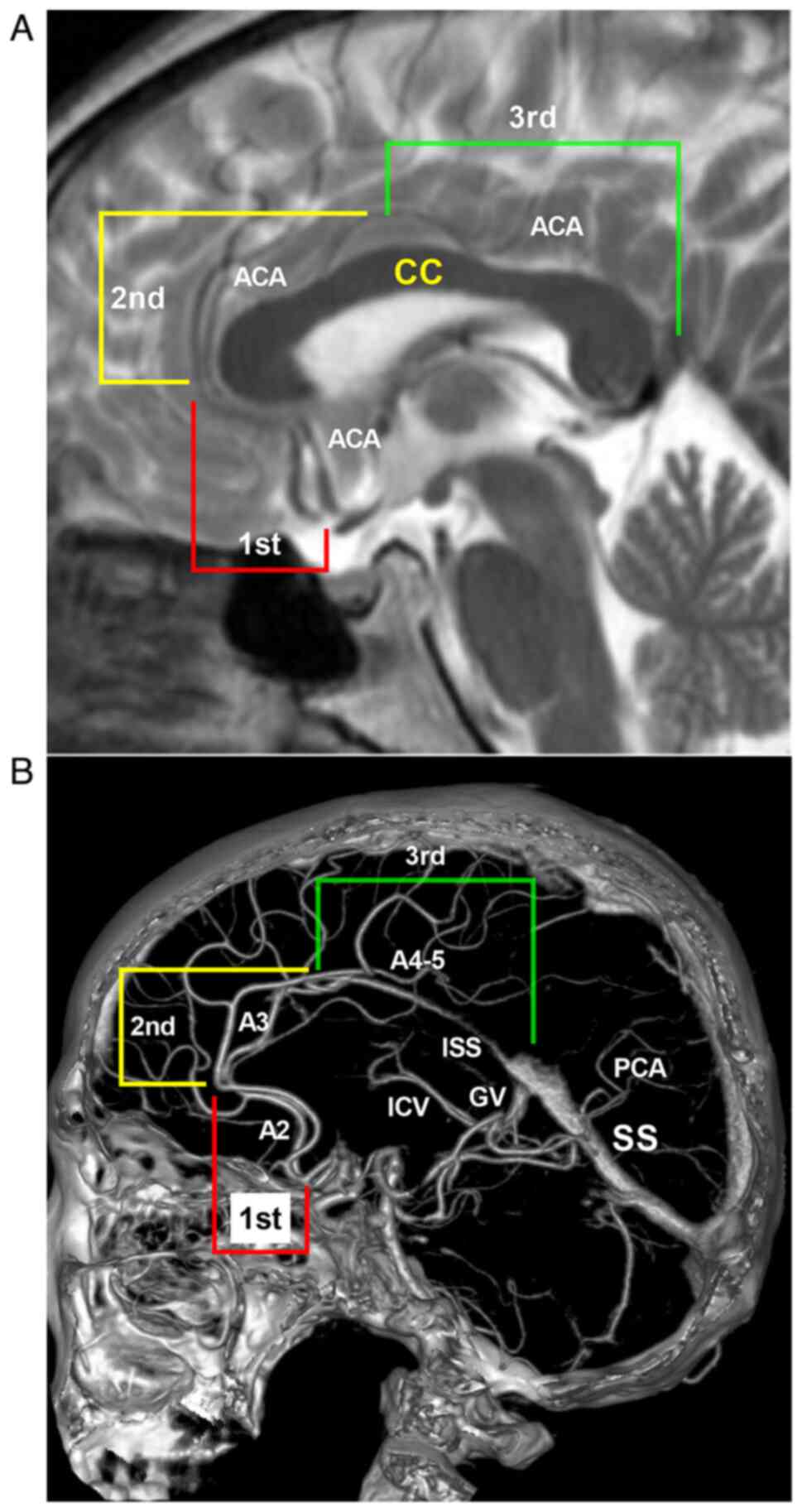 | Figure 2BAVM classification according to the
ACA and corpus callosum anatomy. (A) Head MRI illustrating the
different types of ACA-BAVMs along the various segments of the CC
and ACA. Type I ACA-BAVM locates at the first segment, that is, the
anteroinferior part of the CC genu. Type II ACA-BAVM locates at the
second segment, that is, from the genu to the anterior portion of
CC body. Type III ACA-BAVM locates at the third segment, that is,
from the anterior portion of CC body to the splenium of the CC. (B)
Location of the different types of ACA-BAVMs along the ACA on head
CTA (sagittal view). Type I BAVM is supplied by the A2 segment of
ACA. Type II BAVM is supplied by the A3 segment of ACA. Type III
BAVM is supplied by the A4-5 segment of ACA. ACA, anterior cerebral
artery; BAVM, brain arteriovenous malformation; CC, corpus
callosum; CTA, computed tomography angiography; GV, Galen vein;
ICV, the internal cerebral vein; ISS, inferior sagittal sinus; MRI,
magnetic resonance imaging; PCA, posterior cerebral artery; SS,
straight sinus. |
Scheme and strategy of EVT
All patients were treated under general anesthesia
via a transfemoral approach. Based on the angioarchitecture, the
main feeding artery was selected to perform the EVT. For the BAVM
nidus, a Marathon microcatheter (Lot no. B143380; product no.
105-5056; Medtronic) or Apollo microcatheter (Lot no. B207314;
product no. 105-5096-000; Medtronic) was used to access the nidus
of the BAVM to obtain the wedge position. Onyx liquid embolic
system (Lot no. B176414; product no. 105-7000-060; Medtronic) was
then casted. For flow-related aneurysms, an Echelon microcatheter
(Lot no. B229779; product no. 105-5091-150; Medtronic) was
preferred to perform coiling.
For thin feeding arteries originating at an acute
degree from the ACA in type I and II BAVMs, the micro guide wire
could be shaped into a ‘J’curve to pass the artery of origin. For
type II and III BAVMs with multiple feeding arteries, if
satisfactory EVT could not be achieved through the ACA, an
additional EVT was performed through other arteries, such as the
middle cerebral artery (MCA) and posterior cerebral artery
(PCA).
For the EVT of ruptured BAVMs, flow-related
aneurysms on the feeding artery or in the nidus of the BAVM were
given priority for treatment. In addition, for ruptured BAVMs, if
no weak structure was identified, the main purpose of the EVT was
to reduce the blood flow of BAVMs. For unruptured BAVMs, the main
purpose of the EVT was the targeted embolization of the dangerous
structures, such as aneurysms in the feeding artery or dilated
structures in the nidus. Conservative management would be
recommended for unruptured BAVMs without dangerous structures
(6).
Outcome evaluation and follow-up
EVT-related complications, the length of hospital
stay and the Glasgow Outcome Scale (GOS) score (1, death; 2,
persistent vegetative state; 3, severe disability; 4, moderate
disability; 5, good recovery) at the time of discharge were all
recorded (7). The angiographic
follow-up and the modified Rankin Scale (mRS) score (0: no
symptoms; 1, no significant disability despite symptoms; 2, slight
disability, independent; 3, moderate disability, requires some
assistance, ambulatory; 4, moderately severe disability, requires
assistance, not ambulatory; 5, severe disability, bedridden,
incontinent, requires constant care) were also recorded (8).
Statistical analysis
GraphPad Software (8.02; GraphPad Software, Inc.)
was used for statistical analysis. Continuous variables are
expressed as then mean ± standard deviation. Ordinary one-way ANOVA
followed by Tukey's multiple comparisons test was used for the
comparison of multiple continuous variables. The Chi-squared test
was used to compare count data. A P-value <0.05 was considered
to indicate a statistically significant difference.
Results
General patient information
The patients were aged between 10 and 72 years (mean
age, 35.4±17.0 years), of whom 28 were female (46.7%, 28/60) and 32
were male (53.3%, 32/60). A total of 22 (36.7%, 22/60) patients had
unruptured ACA-BAVMs, of whom 21 patients complained of headaches
and 1 patient was admitted for epilepsy. In total, 38 (63.3%,
38/60) patients were admitted for intracranial hemorrhage,
including 3 patients with subarachnoid hemorrhage, 17 patients with
intracerebral hematoma (IH), 1 patient with IH and subdural
hematoma, 4 patients with intraventricular hemorrhage (IVH), and 13
patients with IH and IVH. Of the 38 patients with ruptured BAVMs,
18 patients were classified as Hunt-Hess grade I, 8 as grade II and
12 as grade III (9).
Imaging characteristics
The diameter of the ACA-BAVMs was 4.4±2.6 cm (range,
0.75-13.5 cm). The size was <3 cm in 19 cases (31.7%, 19/60),
3-6 cm in 28 cases (46.7%, 28/60) and >6 cm in 13 cases (21.6%,
13/60). In total, 16 cases (26.7%, 16/60) were classified as
Spetzler-Martin (SM) grade I, 20 cases (33.3%, 20/60) as SM grade
II, 21 cases (35%, 21/60) as SM grade III and 3 cases (5%, 3/60) as
SM grade IV (10).
A total of 6 cases (10%, 6/60) had flow-related
aneurysms on the feeding artery. In addition, in 42 cases (70%,
42/60), the ACA-BAVMs were drained by the superficial veins, in 14
cases (23.3%, 14/60) they were drained by the deep veins, and in 4
cases (6.7%, 4/60), they were drained by both the superficial and
deep veins.
ACA-BAVM types
Type I BAVMs were identified in 9 (15%, 9/60) cases.
Among these, five were supplied by the ACA, and four were supplied
by the ACA and MCA. Type II BAVMs were identified in 15 cases (25%,
15/60). Among these, nine were supplied by the branches from the
ACA, four were supplied by the ACA and MCA, and two were supplied
by the ACA, MCA and PCA. Type III BAVMs were identified in 36 cases
(60%, 36/60). Among these, 18 were supplied only by the ACA, 11
were supplied by the ACA and PCA, five were supplied by the ACA,
MCA and PCA, one was supplied by the ACA and MCA, and one was
supplied by the ACA, MCA, PCA and external carotid artery.
Results of EVT
Of the 54 (90%, 54/60) ACA-BAVMs without aneurysms,
48 (80%, 48/60) were embolized with Onyx via the ACA, two (3.3%,
2/60) via the ACA and MCA, two (3.3%, 2/60) via the ACA and PCA,
and one (1.7%, 1/60) via the MCA and PCA. In 1 case (1.7%, 1/60)
with a diffuse type I BAVM, reducing blood flow was achieved by the
coiling of the feeding artery.
Of the 6 (10%, 6/60) patients with aneurysms, 1
patient (1.7%, 1/60) with unruptured aneurysm underwent aneurysm
coiling and BAVM embolization with Onyx, 2 patients (3.3%, 2/60)
with unruptured aneurysms underwent aneurysm and BAVM embolization
with Onyx, and 3 patients (5%, 3/60) with ruptured aneurysms
underwent coiling only. For the BAVM nidus, immediate complete or
nearly complete embolization was achieved in 34 cases (56.7%,
34/60). Partial embolization was achieved in 22 (36.7%, 22/60)
cases. Four (6.7%, 4/60) cases were left untreated (in three cases,
after the flow-related aneurysms were embolized, the nidus was
treated at a later stage; in one case, the nidus was too diffuse to
be embolized, and only the feeding artery was coiled).
During the EVT process, there were 3 cases (5%,
3/60) of intraoperative bleeding, among whom, 2 cases had type I
ACA-BAVMs, and 1 case had type II ACA-BAVMs. When intraoperative
bleeding occurred, Onyx casting was continued until termination of
the bleeding was achieved. Following EVT, 1 patient died and 2
patients had a favorable recovery.
Outcome and follow-up
Outcome at discharge
In total, 1 patient (1.7%, 1/60) died from
intraoperative bleeding. At the time of discharge, 8 patients
(13.3%, 8/60) had a GOS score of 3, 3 patients (5%, 3/60) had a GOS
score of 4, and 48 patients (80%, 48/60) had a GOS score of 5. Of
the 59 patients who were discharged, the length of hospital stay
ranged from 2-58 days (range, 10.6±9.3 days).
Outcome at follow-up. A total of 49 (81.7%,
49/60) patients could be contacted at the clinical follow-up, which
ranged from 12-78 months (mean, 31.1±20.5 months). Among these, 2
patients (4.1%, 2/49) died from non-cerebral disease. Of the
remaining 47 (95.9%, 47/49) patients, 39 (83%, 39/47) presented
with an mRS score of 0, 3 (6.4%, 3/47) with an mRS score of 1, 3
(6.4%, 3/47) with an mRS score of 2 and 2 (4.2%, 2/47) with an mRS
score of 3. In total, 20 (33.3%, 20/60) patients underwent an
angiographic follow-up, of whom 18 (90%, 18/20) had satisfactory
embolization and two (10%, 2/20) underwent additional EVT (after
EVT, they had no new neurological deficit). The main clinical
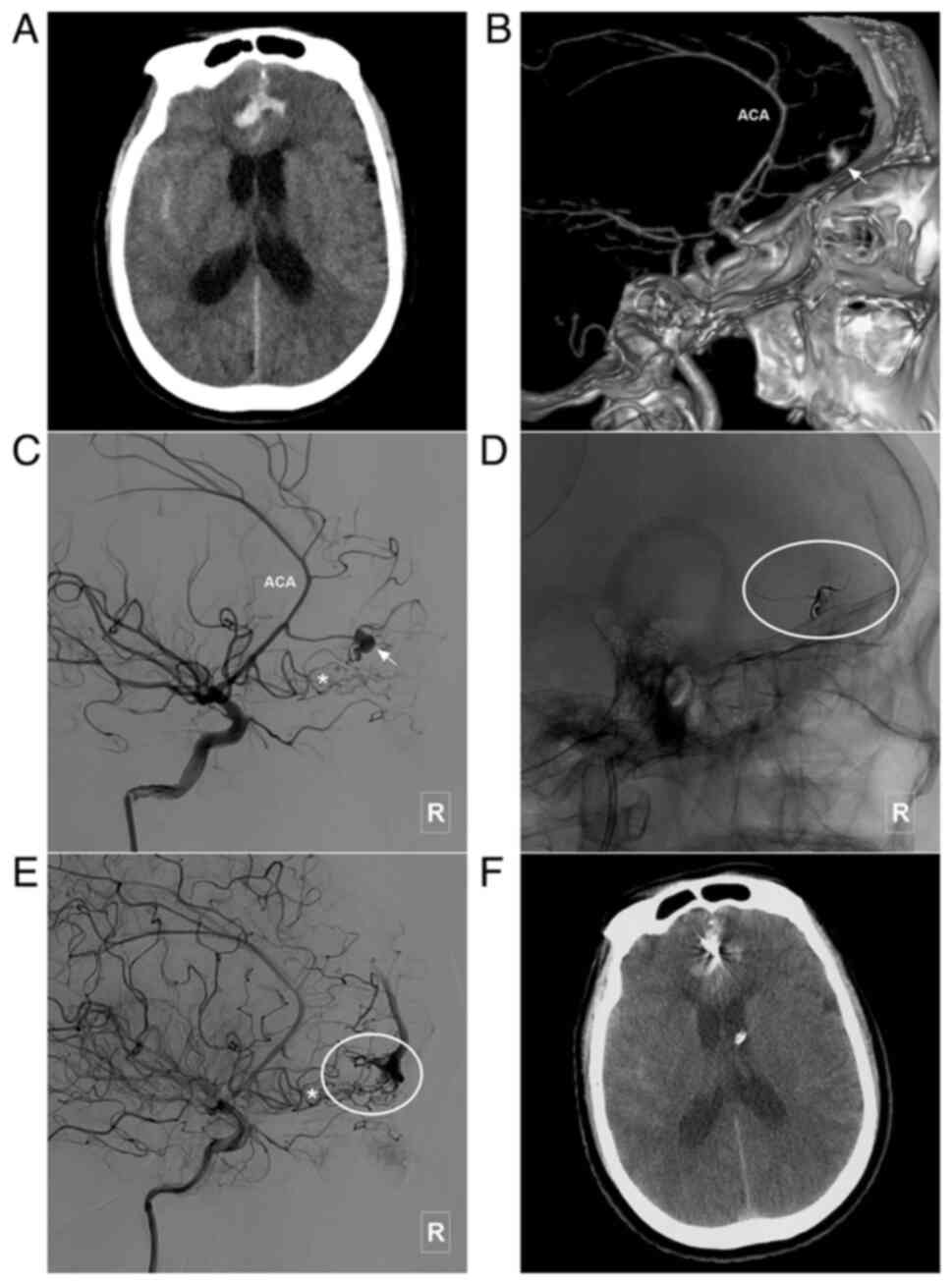
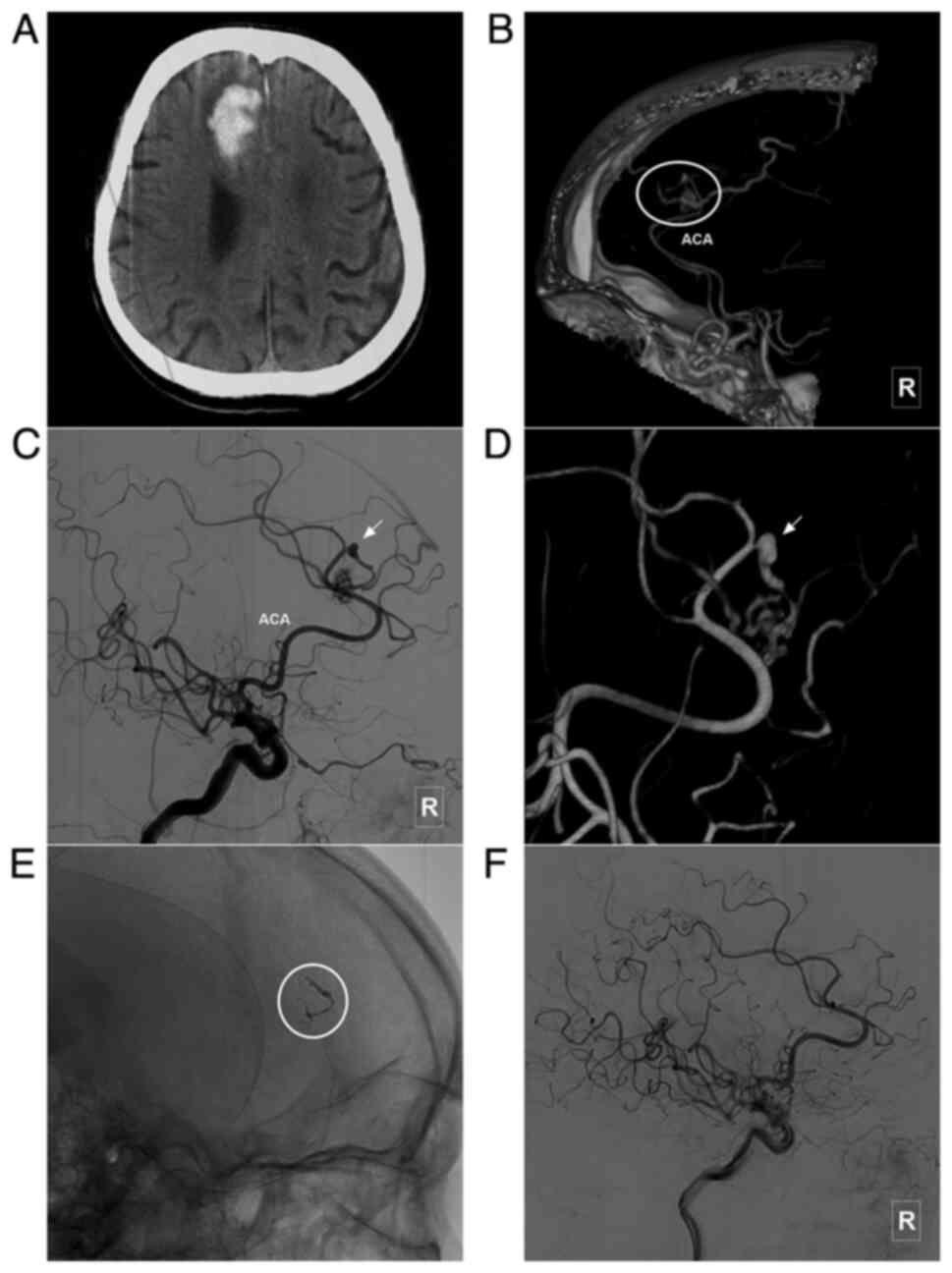
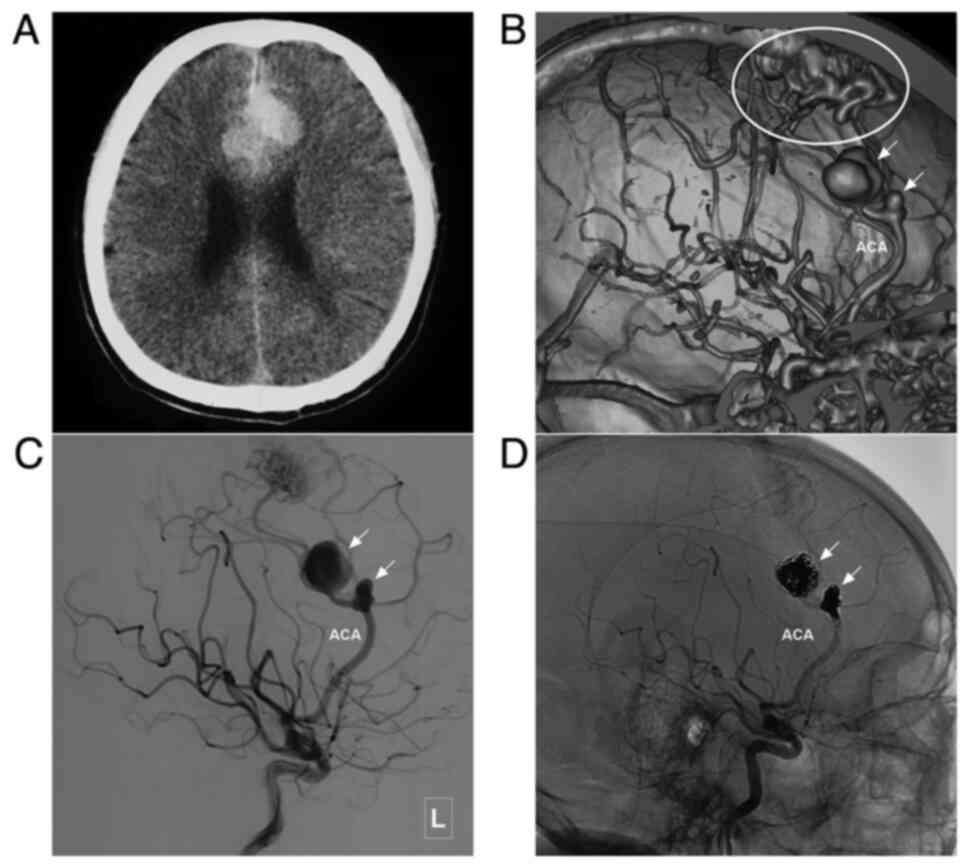
characteristics of the patients are presented in Table I. In addition, images of illustrative
cases are presented in Fig. 3,
Fig. 4, Fig. 5, Fig.
6, Fig. 7 and Fig. 8.
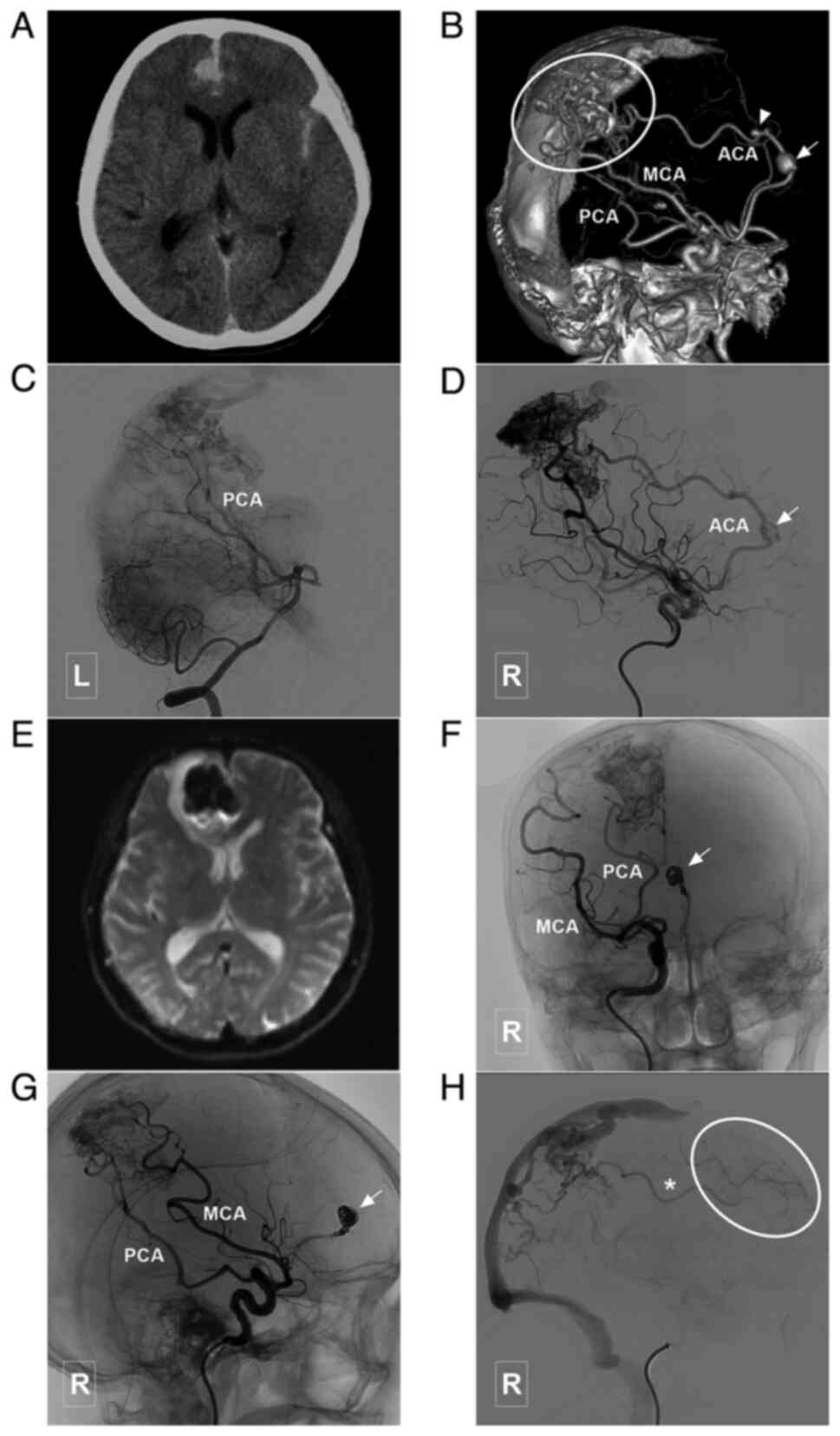 | Figure 7Typical case of a type III BAVM with
multiple flow-related aneurysms in the ACA trunk. (A) Head CT scam
illustrating a subarachnoid hemorrhage in the interhemispheric
fissure. (B) Head CTA illustrating a BAVM (encircled area) supplied
the ACA, MCA and PCA, and which drained to the sagittal sinus. The
ACA is the main feeding artery, with multiple flow-related
aneurysms (arrow and arrow head). (C) Angiogram of the left
vertebral artery shows that the posterior circulation is involved
in the blood supply of the BAVM. (D) A stent was used to assist in
coiling of the aneurysm (arrow) in the ACA. (E) MRI at 3 days
post-treatment reveals a new hemorrhage in the interhemispheric
fissure, which is considered to be caused by re-rupture of the
aneurysm. (F and G) Parent artery occlusion is performed to occlude
the aneurysm and the parent artery (arrow). (H) Angiogram of the
right internal carotid artery in venous phase illustrating
retrograde blood supply from the BAVM to the frontal lobe
(encircled area) through the ACA (asterisk). ACA, anterior cerebral
artery; BAVM, brain arteriovenous malformation; CT, computed
tomography; CTA, computed tomography angiography; L, left; MCA,
middle cerebral artery; MRI, magnetic resonance imaging; PCA,
posterior cerebral artery; R, right. |
 | Table IMain clinical data of the patients in
the present study. |
Table I
Main clinical data of the patients in
the present study.
| Characteristic | Data |
|---|
| Age (years) | |
|
Mean ±
SD | 35.4±17.0 |
|
Range | 10-72 |
| Female/male | 87.5% (28/32) |
| Presentation | |
|
Unruptured | 22 (36.7%,
22/60) |
|
Ruptured | 38 (63.3%,
38/60) |
| ACA-BAVM types | |
|
I | 9 (15%, 9/60) |
|
II | 15 (25%, 15/60) |
|
III | 36 (60%, 36/60) |
| Nidus size (cm) | |
|
Mean ±
SD | 4.4±2.6 |
|
Range | 0.75-13.5 |
| Spetzler-Martin
grade | |
|
1 | 16 (26.7%,
16/60) |
|
2 | 20 (33.3%,
20/60) |
|
3 | 21 (35%, 21/60) |
|
4 | 3 (5%, 3/60) |
| Associated
aneurysm | 10 (10%, 6/60) |
| Draining vein | |
|
Superficial | 42 (70%, 42/60) |
|
Deep | 14 (23.3%,
14/60) |
|
Both | 4 (6.7%, 4/60) |
| Embolization degree
of the nidus | |
|
Complete or
nearly complete | 34 (56.7%,
34/60) |
|
Partial | 22 (36.7%,
22/60) |
|
Intact | 4 (6.7%, 4/60) |
| Complications | |
|
Intraoperative
bleeding | 3 (5%, 3/60) |
| Glasgow Outcome Scale
at time of discharge | |
|
5 | 48 (80%, 48/60) |
|
4 | 3 (5%, 3/60) |
|
3 | 8 (13.3%, 8/60) |
|
2 | 0 |
|
1 | 1 (6.1%, 2/33) |
Results of statistical analysis
The results of statistical analysis are summarized
in Table II. No statistically
significant differences were noted in the diameter of the BAVMs,
IVH presentation and deep vein drainage among the three types of
ACA-BAVMs. Intraoperative hemorrhagic complications tended to occur
in type I and II BAVMs (P=0.0223). PCA involvement was more
commonly involved in type II and III BAVMs (P=0.0052).
 | Table IIResults of the statistical analysis of
the imaging characteristics of the ACA-BAVMs. |
Table II
Results of the statistical analysis of
the imaging characteristics of the ACA-BAVMs.
| Characteristics | Type I BAVMs
(n=9) | Type II BAVMs
(n=15) | Type III BAVMs
(n=36) | P-value |
|---|
| BAVM diameter
(cm) | 4.89±2.60 | 4.67±2.04 | 4.18±2.85 | 0.3830 |
| IVH presentation | 1/5a | 4/9a | 12/24a | 0.4708 |
| Deep vein
involvement | 2/9 | 5/15 | 11/36 | 0.8420 |
| Hemorrhagic
complication | 2/9 | 1/15 | 0/36 | 0.0223 |
| PCA involvement | 0/9 | 2/15 | 17/36 | 0.0052 |
Discussion
BAVM is a common congenital vascular disease that is
associated with the abnormal connection between arteries and veins,
and lacks an intervening capillary network, which can occur in any
part of the brain. The architecture depends on the location,
recruited arteries and draining veins, and the size of the BAVM
nidus (11).
The ACA lies between the bilateral cerebral
hemispheres. It runs an anteroposterior course along the lower part
of the cerebral falx and the upper part of the corpus callosum.
Under normal conditions, the ACA system is relatively independent
(12). In the case of chronic
persistent ischemia in the ACA supplying area, the ACA can
anastomose with the posterior choroidal arteries from the PCA or
with the pial branches of the PCA. This anastomosis is often
characterized by a direct anastomosis between the blood supply from
the PCA system and the distal trunk of the ACA (13). Generally speaking, the MCA only
compensates for the pial branches of ACA.
Some of the BAVMs can occur specifically in the
supplying area of ACA, exhibiting a variety of vascular
architectures (14). However, to
date, to the best of our knowledge, there is no detailed
classification of this specific type of BAVM. In the present study,
based on their anatomical association with surrounding structures,
the ACA-BAVMs were divided into three types.
In the present study on ACA-BAVMs, the arteries
distributed along the ACA differed. For example, there is a
potential collateral circulation between the posterior part of the
ACA and the PCA, and the anterior part of the ACA can only be
compensated by the pial branches from the MCA. BAVMs occurring in
the posterior part of the corpus callosum and ACA are closer to the
deep venous system (15).
Therefore, statistical analysis was performed for
IVH presentation, deep vein involvement and PCA involvement among
the different types of ACA-BAVMs to determine whether the types of
ACA-BAVMs are associated with IVH presentation, deep vein
involvement and PCA. However, the results revealed no significant
differences in IVH presentation and deep vein involvement. This
suggests that ACA-BAVMs may have the similar imaging and clinical
characteristics. However, the statistical analysis revealed that
the PCA tended to provide blood flow to the distal ACA-BAVMs.
The principle of EVT for ACA-BAVMs is the same as
that for BAVMs in other areas, which mainly aims to manage
flow-related aneurysms and the nidus of BAVMs to reduce blood flow
to the draining venous system (6).
In the present study, 6 cases had with flow-related aneurysms, with
an incidence of 10%, which was lower than that observed in a
previous study (16). This type of
aneurysm is a definite risk factor of BAVMs and requires treatment
(Figs. 3, 4, 5,
7 and 8).
The efficacy and safety of EVT for ACA-BAVMs was
satisfactory, with a low complication rate of only 5% in the
present study. In the present study, 85% of the cases had a GOS
score of 4 or 5 at the time of discharge, and 89.3% of cases had an
mRS score of 0 and 1 during the follow-up period, which indicated
that EVT was a safe strategy for the treatment of ACA-BAVMs.
In the present study, complete or almost complete
embolization was achieved in 56.7% of the cases. The degree of
embolization is dependent on a number of factors, such as the size
and morphology of the nidus and diameter, and the tortuosity of
feeding. Moreover, the degree of embolization is not main treatment
goal; the main treatment goal is the management of the weak point
posing a risk. Thus, in the present study, the degree of AVM
embolization was not pursued.
Following ACA-BAVM classification, EVT becomes
strategic. Type I and II BAVMs are supplied by branches of the
proximal ACA, and these feeding arteries are commonly slim and
multiple. Microcatheter navigation close to the BAVM nidus is
difficult, which increases the difficulty in performing EVT and the
associated risks. In the present study, intraoperative bleeding
tended to occur in patients with type I and II BAVMs. As type II
and III BAVMs are supplied by the end of the ACA trunk, performing
EVT is relatively simple. However, when they are supplied by
multiple regional arteries, particularly from the PCA region, EVT
may then be a complex process. In some cases, the PCA has to be
used to perform the EVT as the PCA tends to be involved in type II
and III BAVMs.
The present study has some limitations which need to
be mentioned. One of these limitations is the low rate of
angiographic follow-up. The efficacy and safety of EVT were
presented as GOS and mRS scores at the time of discharge and
clinical follow-up, which proved that EVT was a safe treatment for
ACA-BAVMs. However, due to the very low rate of angiographic
follow-up, it is inappropriate to compare the pre- and
post-operative angiographic characteristics in this series of
patients.
In conclusion, ACA-BAVMs can be classified into
types I-III according to the position of the BAVMs along the ACA.
This classification is helpful. For type I and II BAVMs supplied by
slim and multiple branches of the proximal ACA, EVT is difficult to
perform, and intraoperative bleeding complications are common. For
type II and III BAVMs, as the PCA tends to be involved, in some
cases, the PCA has to be used to perform EVT. On the whole, the
present study demonstrated that EVT is a safe procedure for the
treatment of ACA-BAVMs.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
KH and JY designed the study and drafted the
manuscript. KH, YW and WL collected the data. KH and JY confirmed
the authenticity of all the raw data. YW and WL revised the
manuscript. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the First Hospital of Jilin University (approval no.
2020-341). Written informed consent was obtained from the patients
their legal relatives.
Patient consent for publication
The patients or their legal relatives provided
consent and agreed for their data (shown in the figures) to be
published.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lawton MT, Rutledge WC, Kim H, Stapf C,
Whitehead KJ, Li DY, Krings T, terBrugge K, Kondziolka D, Morgan
MK, et al: Brain arteriovenous malformations. Nat Rev Dis Primers.
1(15008)2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Solomon RA and Connolly ES Jr:
Arteriovenous malformations of the brain. N Engl J Med.
376:1859–1866. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ecker RD: Epistemology of brain
arteriovenous malformations. World Neurosurg. 89:697–698.
2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Batista LL and Azevedo HC: Anterior
cerebral artery. J Neurosurg. 101(717): author reply 717.
2004.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Picard L, Miyachi S, Braun M, Bracard S,
Per A and Marchal JC: Arteriovenous malformations of the corpus
callosum-radioanatomic study and effectiveness of intranidus
embolization. Neurol Med Chir (Tokyo). 36:851–859. 1996.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hou K, Xu K, Chen X, Ji T, Guo Y and Yu J:
Targeted endovascular treatment for ruptured brain arteriovenous
malformations. Neurosurg Rev. 43:1509–1518. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jennett B and Bond M: Assessment of
outcome after severe brain damage. Lancet. 1:480–484.
1975.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Rankin J: Cerebral vascular accidents in
patients over the age of 60. II. Prognosis. Scott Med J. 2:200–215.
1957.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hunt WE and Hess RM: Surgical risk as
related to time of intervention in the repair of intracranial
aneurysms. J Neurosurg. 28:14–20. 1968.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Spetzler RF and Martin NA: A proposed
grading system for arteriovenous malformations. J Neurosurg.
65:476–483. 1986.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li K, Guo Y, Qu L, Xu B, Xu K and Yu J:
Hybrid surgery for an arteriovenous malformation fed by an
accessory middle cerebral artery and drained by a developmental
venous anomaly: A case report and literature review. Exp Ther Med.
16:1994–2000. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Rhoton AL Jr: The supratentorial arteries.
Neurosurgery. 51 (Suppl 4):S53–S120. 2002.PubMed/NCBI
|
|
13
|
Hou K, Li G, Luan T, Xu K and Yu J: The
prospects and pitfalls in the endovascular treatment of moyamoya
disease-associated intracranial aneurysms. Neurosurg Rev.
44:261–271. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Pabaney AH, Ali R, Kole M and Malik GM:
Arteriovenous malformations of the corpus callosum: Pooled analysis
and systematic review of literature. Surg Neurol Int. 7 (Suppl
9):S228–S236. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
da Costa MDS, Santos BFO, de Almeida
Guardini FB and Chaddad-Neto F: Microsurgical treatment for
arteriovenous malformation of the corpus callosum and choroidal
fissure. Neurosurg Focus. 43(V12)2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Cagnazzo F, Brinjikji W and Lanzino G:
Arterial aneurysms associated with arteriovenous malformations of
the brain: Classification, incidence, risk of hemorrhage, and
treatment-a systematic review. Acta Neurochir (Wien).
158:2095–2104. 2016.PubMed/NCBI View Article : Google Scholar
|