Introduction
Lignac-de Toni-Debré-Fanconi syndrome [termed
Fanconi syndrome (FS)] is a proximal tubular defect that causes an
impaired reabsorption of the glomerular filtrate. Patients with
this condition can present with hypophosphatemia, renal glycosuria,
hypouricemia and aminoaciduria (1).
While it is mostly observed as a hereditary disease in childhood,
the acquired form is more common in adults (2). It is difficult to define the
epidemiology of FS, as it includes a wide variety of acquired,
inherited and exogenous factors unrelated to each other (2).
Phosphate depletion is the most critical clinical
aspect of FS as it leads to osteomalacia. Some patients present
with symptoms and signs related to hypophosphatemic osteomalacia
(HO) (3). These symptoms, including
proximal myopathy, muscle weakness, chronic arthritis sign, and
bone and back pain are non-specific (4). Thus, these patients present with these
symptoms and are misdiagnosed (5).
To the best of our knowledge, there are currently
only a limited number of publications available on the topic, and
the majority of these are related to drug-induced FS. In the study
conducted by Eguchi et al (6), the 25 cases of FS reported an
improvement in phosphate levels following the cessation or dose
reduction of adefovir-induced FS. The case reported in the study by
Park et al (7) involved a
patient diagnosed with idiopathic FS presenting with HO. The study
by Yamaguchi et al (8) also
reported a patient who presented with HO on the grounds of primary
biliary cirrhosis. Finally, in the study by Li et al
(9), there were nine cases that were
initially misdiagnosed; the majority of these presented with HO due
to drug-related FS.
The present study describes the case of a patient
who presented with joint pain and was diagnosed with idiopathic FS
with multiple osteoporotic fractures.
Case report
A 46-year-old male visited the outer center (Malatya
Public Hospital, Malatya, Turkey) complaining of pain in his hip.
The pain was also felt in the shoulders, arms and ankles. He
complained of a 1-year history of sustained walking difficulty. He
did not complain of any morning stiffness. The patient was
diagnosed with rheumatoid arthritis in the outer center (Malatya
Public Hospital) and was referred to our tertiary care hospital
(Inonu University Faculty of Medicine Turgut Ozal Medical
Center.
The patient was previously healthy, and no
additional disease was known. His family history was unremarkable,
and his vital signs were normal. According to the physical
examination, the hip joint and ankle were sensitive and became
painful with flexion or extension movement. There was no warmth,
redness, or swelling around these joints. Moreover, he was
stumbling.
The laboratory data revealed a creatinine level of
1.4 mg/dl, potassium level of 2.8 mmol/l (reference range, 3.5-5.5
mmol/l), calcium level of 7.8 mg/dl (reference range, 8.4-10.2
mg/dl), phosphorus level of 1.5 mg/dl (reference range, 2.7-4.3
mg/dl), uric acid level of 1.4 mg/dl (reference range, 3.5-4.2
mg/dl), serum glucose level of 90 mg/dl and urine glucose level of
716 mg (reference range, 1-35 mg; normoglycemic glycosuria), urine
protein level of 887 mg (reference range, 50-80 mg), metabolic
acidemia with a normal anion gap (pH 7.18; HCO3, 15.9 mmol/l), and
low serum levels of phosphorus and 1,25-dihydroxyvitamin D3 (15.0
pg/dl). In addition, the serum levels of parathyroid hormone (PTH)
were elevated (84.6 pg/ml; reference range, 14-72 pg/ml). Some of
these laboratory values are summarized in Table I.
 | Table ISome of the laboratory values of the
case described in the present study. |
Table I
Some of the laboratory values of the
case described in the present study.
| Parameter | Value | Reference range |
|---|
| Serum levels | | |
|
Creatinine | 1.4 mg/dl | 0.5-1.3 mg/dl |
|
Glucose | 90 mg/dl | 70-105 mg/dl |
|
Potassium | 2.8 mmol/l | 3.5-5.5 mmol/l |
|
Phosphorus | 1.5 mg/dl | 2.7-4.3 mg/dl |
|
Calcium | 7.8 mg/dl | 8.4-10.2 mg/dl |
|
Uric
acid | 1.4 mg/dl | 3.5-4.2 mg/dl |
|
Parathyroid
hormone | 84.6 pg/ml | 14-72 pg/ml |
|
1,25-Dihydroxyvitamin
D3 | 15.0 pg/dl | |
| 24-h urine
levels | | |
|
Glucose | 716 mg | 1-35 mg |
|
Protein | 887 mg | 50-80 mg |
|
Potassium | 9 mmol/l | 17-99 mmol/l |
|
Phosphorus | 13 mg | 5-189 mg |
|
Calcium | 5 mg | 0.2-9.4 mg |
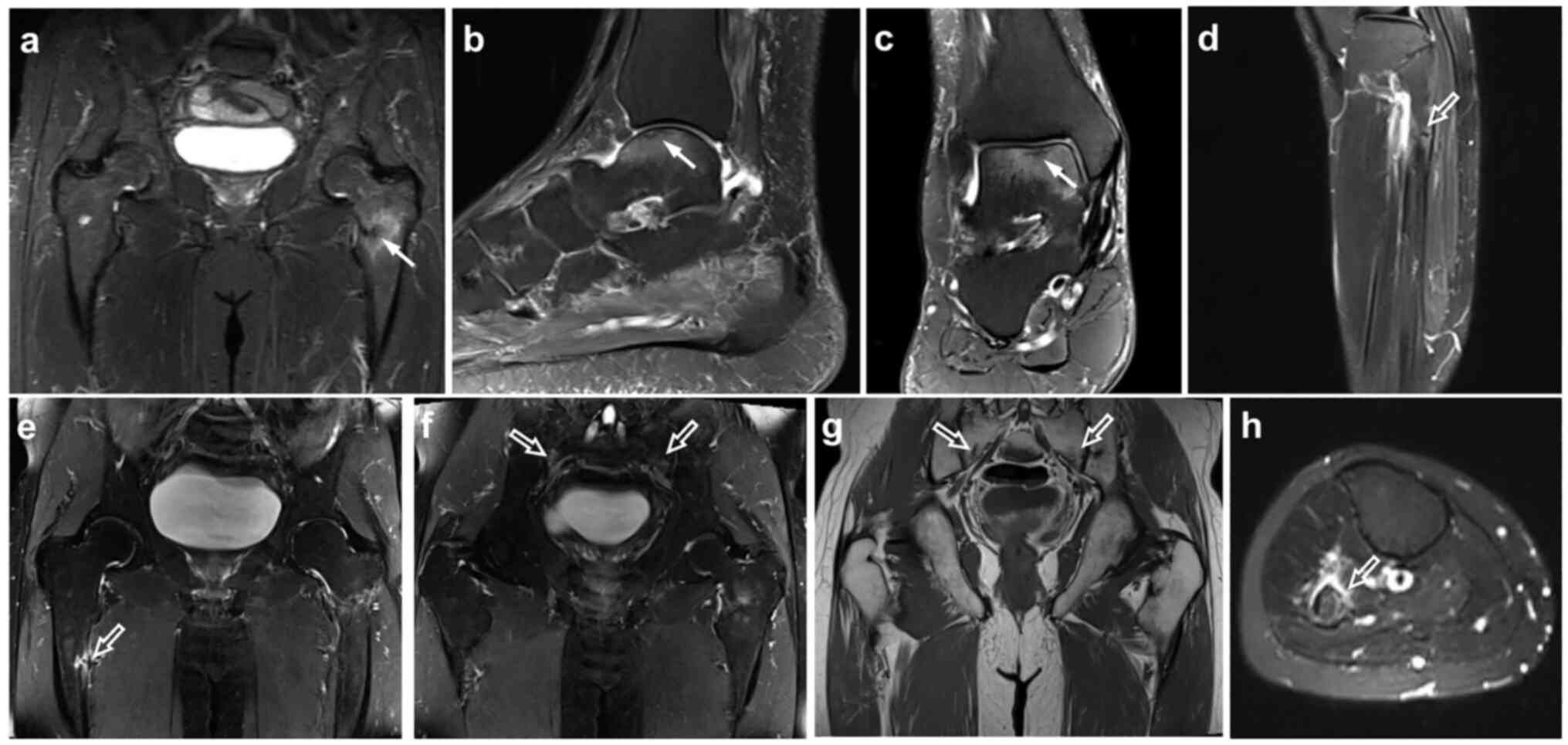
Bilateral proximal femur fractures and fractures of
the iliac and sacral bones and ankles were found via magnetic
resonance imaging (MRI) and on the basis of osteoporosis (Fig. 1). HO was diagnosed clinically on the
basis of laboratory analyses, and bone densitometry and MRI
findings. Thus, a bone biopsy was not performed. Blood and urine
biochemical data, and the radiology findings revealed severe
osteoporosis, thus indicating FS with proximal renal tubular
acidosis (RTA).
Following the diagnosis, the patient was evaluated
to determine the etiology of FS. Anti-SSA and anti-SSB tests were
negative. The bone marrow biopsy performed following diagnosis was
compatible with the normocellular bone marrow; there was no
monoclonal component.
The patient was not on any medication in relation to
FS. A light microscopic examination of a kidney biopsy demonstrated
the cellular infiltration of the interstitium and proximal tubular
epithelium composed mainly of C3 without glomerular involvement.
Other common observations were moderate tubular degeneration and
regeneration, and tubular atrophy consistent with features of
TIN.
Idiopathic FS was considered for this patient.
Supportive treatments were thus commenced. Serum bicarbonate,
phosphorus, potassium and vitamin D were administered. He was
referred to the Inonu University Faculty of Medicine Orthopedic
Clinic and was not operated on for fractures. He was then
followed-up by the Inonu University Faculty of Medicine
Endocrinology Clinic for osteoporosis. It was recommended that he
continue the supplement therapy and the patient is still taking the
same supplements. His osteoporosis is stable and follow-up is being
continued. In addition, he was referred to the Inonu University
Faculty of Medicine Nephrology Clinic. The patient, who has stage 3
kidney disease, is still being followed-up at the nephrology
clinic.
In summary, the diagnosis of osteomalacia and
proximal tubular RTA remains a challenge for physicians due to its
non-specific joint manifestations. Severe osteomalacia and
osteoporosis are considered to be caused by tubulointerstitial
nephritis with FS, a rare, asymptomatic renal involvement.
Discussion
A total of 180 liters glomerular filtrate forms out
of the proximal tubule (PT) every day. Of this amount, 98% is
reabsorbed in the PT (10). However,
defects in the PT can lead to reabsorption deficiency, primarily of
bicarbonate and also of other solutes (such as phosphorus, amino
acid, glucose and uric acid). This condition characterizes FS
(11). Following the diagnosis of
FS, the etiology causing the defect in the PT needs to be
researched. Autosomal dominant hereditary diseases may be the
cause.
A major cause of FS in adults is the increased
excretion of monoclonal immunoglobulin light chains due to
monoclonal gammopathies that are otherwise latent. Sjögren's
syndrome, primary biliary cirrhosis, tyrosinemia, fructose
intolerance, galactosemia, Wilson's disease, Dent's disease, Lowe
syndrome and drug-induced cytopathic effects are also considered in
the etiology (10). The most toxic
agents for the PT are tenofovir, adefovir, ifosfamide, gentamicin,
acetazolamide, sodium bicarbonate, sodium valproate, fumaric acid
and deferasirox. Overall, any of these factors may be responsible
for FS, and even rare cases may be idiopathic (12). The diagnosis of FS is generally made
clinically with glycosuria, hypophosphatemia, aminoaciduria, normal
anion gap metabolic acidosis and proteinuria (12).
Complaints at referral regarding HO can be generally
non-specific, such as proximal myopathy, back pain, bone pain and
joint pain (4). As such complaints
are similar to those of rheumatologic patients, patients may be
misdiagnosed (5). The general
characteristics and clinical manifestation of patients with
hypophosphatemic osteomalacia are presented in Table II. In addition, some laboratory
values of cases are summarized in Table III. Li et al (9) considered HO and rheumatologic diseases,
such as rheumatoid arthritis and ankylosing spondylitis, in nine
patients who presented between 2011 and 2015. Following an etiology
search, HO and FS were then diagnosed (9). In another case involving FS diagnosis
on the grounds of primary cirrhosis, the patient first applied to
an orthopedic clinic with complaints of year-long bilateral knee
pain and walking difficulties. Later, her condition was diagnosed
by internal medicine as HO (8).
Thus, the awareness of HO is crucial. The present study aimed to
increase the awareness of physicians by discussing the mentioned
case and similar cases in the literature. Physicians have to be
suspicious of HO, whose prevalence is difficult to specify and not
stated in the literature. Likewise, medical history, physical
examination and drug use need to be thoroughly investigated.
Particularly in younger patients, the condition may be associated
with HO, given the presentation of unexplained bone and joint
pains.
 | Table IIGeneral characteristics clinical
manifestation of patients with hypophosphatemic osteomalacia. |
Table II
General characteristics clinical
manifestation of patients with hypophosphatemic osteomalacia.
| Clinical
manifestation | Ethnicity | Sex | Age, years | Etiology | Misdiagnosis | (Refs.) |
|---|
| Thoracic and back
pain | Chinese | F | 39 | Tumor | Lumbar disc
disease | (9) |
| Thoracic and back
pain | Chinese | M | 47 | Tumor | AS, osteoporosis | (9) |
| Thoracic and back
pain | Chinese | M | 43 | Drug-induced Fanconi
syndrome | AS, osteoporosis,
lumbar disc disease | (9) |
| Thoracic and back
pain | Chinese | M | 43 | Drug-induced Fanconi
syndrome | Chronic
arthritis | (9) |
| Thoracic and back
pain | Chinese | M | 34 | Chronic nephropathy
with acidosis | AS | (9) |
| Thoracic and back
pain | Chinese | M | 22 | Drug-induced Fanconi
syndrome | AS, chronic
arthritis | (9) |
| Thoracic and back
pain | Chinese | M | 50 | Drug-induced Fanconi
syndrome | AS, chronic
arthritis | (9) |
| Thoracic and back
pain | Chinese | F | 35 | Drug-induced Fanconi
syndrome | AS, osteoporosis | (9) |
| Thoracic and back
pain | Chinese | M | 55 | Drug-induced Fanconi
syndrome | Osteoporosis, lumbar
disc disease | (9) |
| Routine check-up | Korean | F | 52 | Idiopathic | Osteoporosis | (7) |
| Difficulty
walking | Japanese | F | 49 | Primary biliary
cirrhosis | Orthopedic
disorder | (8) |
| Difficulty
walking | Turkish | M | 46 | Idiopathic | RA | Present case |
 | Table IIISome laboratory values of cases of FS
in the literature. |
Table III
Some laboratory values of cases of FS
in the literature.
| Prognosis | Creatinine
(mg/dl) | Calcium (mg/dl) | Phosphate
(mg/dl) | PTH (pg/ml) | Vitamin D
(pg/dl) | (Refs.) |
|---|
| Improved | NA | 8.4 | 1.3 | 95 | NA | (9) |
| Improved | NA | 8.8 | 1.3 | 108.7 | NA | (9) |
| Improved | NA | 8.02 | 2.3 | 19.2 | NA | (9) |
| Improved | NA | 8.1 | 1.6 | 21.6 | NA | (9) |
| Denied treatment | NA | 8.4 | 1.6 | 17.8 | NA | (9) |
| Improved | NA | 8.5 | 1 | 50.9 | NA | (9) |
| Improved | NA | 8.9 | 2 | 28.8 | NA | (9) |
| Improved | NA | 8.8 | 2 | 21.4 | NA | (9) |
| Improved | NA | NA | NA | NA | NA | (9) |
| NA | 1,19 | 8.9 | 1.9 | 74.5 | 20 | (7) |
| Improved | 1,4 | 9.5 | 2.3 | NA | 11 | (8) |
| Improved | 1,4 | | 1.5 | 84 | 15 | Present case |
Case presentations are generally on the grounds of
osteoporosis, such as joint or bone pain and walking difficulties.
In the case presented herein, the patient was initially considered
to have arthritis, but an advanced search was subsequently
conducted. The MRI and laboratory test results were examined. The
existence of osteoporotic fractures in the MRI and proteinuria in
the laboratory findings, normal anion gap metabolic acidosis and
glycosuria were discussed in association with nephrology, and PT
deficiency was considered. Following the diagnosis of FS, a bone
marrow biopsy was performed for the purpose of etiological
research, and a normocellular bone marrow was revealed. The patient
had no additional drug use. Mouth and eye dryness were examined.
Schirmer's test yielded negative results, and Sjögren's syndrome
was not considered as the SSA and SSB antibody test results were
negative. The patient had no heavy metal exposition. He was
considered to suffer from idiopathic FS. Supportive treatment was
thus commenced. Serum bicarbonate, phosphorus, potassium, vitamin D
replacement was administered. He is still being followed-up by the
nephrology clinic due to his stage 3 kidney disease.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
ANC and SY were major contributors to the writing of
the manuscript, and also made substantial contributions to the
design of the study and to the interpretation of data for the
study. SY, IS and BE revised and edited the manuscript and also
advised on patient treatment. SY, IS, BE and ANC analyzed patient
data. ZÖ and ANC contributed to the conception of the study and
acquisition of the data, as well as in the analysis of data for the
study and provided the radiological images. All authors gave the
final approval of the version to be published and reviewed the
literature. ANC and SY confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
The patient provided written informed consent for
his clinical information to be used for the purposes of the present
study.
Patient consent for publication
The patient provided written informed consent for
the publication of his clinical data in the present case report
study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Klootwijk ED, Reichold M, Unwin RJ, Kleta
R, Warth R and Bockenhauer D: Renal Fanconi syndrome: Taking a
proximal look at the nephron. Nephrol Dial Transplant.
30:1456–1460. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dalmak S, Erek E, Serdengecti K, Okar I,
Ulku U and Basaran M: A case study of adult-onset hypophosphatemic
osteomalacia with idiopathic fanconi syndrome. Nephron. 72:121–122.
1996.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Norden AG, Scheinman SJ, Schodt-Lanckman
MM, Lapsley M, Nortier JL, Thakker RV, Unwin RJ and Wrong O:
Tubular proteinuria defined by a study of Dent's (CLCN5 mutation)
and other tubular diseases. Kidney Int. 57:240–249. 2000.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Reginato AJ, Falasca GF, Pappu R, McKnight
B and Agha A: Musculoskeletal manifestations of osteomalacia:
Report of 26 cases and literature review. Semin Arthritis Rheum.
28:287–304. 1999.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Jan de Beur SM: Tumor-induced
osteomalacia. JAMA. 294:1260–1267. 2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Eguchi H, Tsuruta M, Tani J, Kuwahara R
and Hiromatsu Y: Hypophosphatemic osteomalacia due to drug-induced
Fanconi syndrome associated with adefovir dipivoxil treatment for
hepatitis B. Intern Med. 53:233–237. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Park DJ, Jang KS and Kim GH: Adult
idiopathic renal fanconi syndrome: A case report. Electrolyte Blood
Press. 16:19–22. 2018.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yamaguchi S, Maruyama T, Wakino S,
Tokuyama H, Hashiguchi A, Tada S, Homma K and Monkawa T: A case of
severe osteomalacia caused by Tubulointerstitial nephritis with
Fanconi syndrome in asymptomotic primary biliary cirrhosis. BMC
Nephrol. 16(187)2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Li L, Wang SX, Wu HM, Luo DL, Dong GF,
Feng Y and Zhang X: Acquired hypophosphatemic osteomalacia is
easily misdiagnosed or neglected by rheumatologists: A report of 9
cases. Exp Ther Med. 15:5389–5393. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hall AM, Bass P and Unwın RJ: Drug-induced
renal Fanconi syndrome. QJM. 107:261–269. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Foreman JW: Fanconi Syndrome. Pediatr Clin
North Am. 66:159–167. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yaxley J and Pirrone C: Review of the
diagnostic evaluation of renal tubular acidosis. Ochsner J.
16:525–530. 2016.PubMed/NCBI
|