Introduction
Although cesarean scar pregnancy (CSP) is
considerably infrequent among patients with ectopic pregnancy, in
which the gestational sac (GS) is implanted into the myometrium at
the place of a previous cesarean section scar, it is one of the
most dangerous long-term complications following cesarean section
(1,2). The approximate incidence of CSP ranges
from 1/1,800 to 1/2,200 and the percentage of ectopic pregnancies
among women with a history of cesarean section accounts to 6.1% and
has exhibited an increasing tendency (3,4).
CSP has been classified as two different
pathophysiological types by Vial et al (5). The first type (CSP-I) exhibits a
certain success rate of viable birth, although the amniotic sac
implants into the previous cesarean section. The amniotic sac grows
towards the cervico-isthmic space and the uterine cavity with the
progression of pregnancy. A high risk of massive bleeding is
possible due to the placenta previa and placenta accrete (5,6). The
second type of CSP (CSP-II) is associated with a high risk of
bleeding and uterine rupture, in which the GS is deeply implanted
into the myometrium with the progression of pregnancy (5,6).
However, the CSP has been classified into three types according to
the association between the GS and uterine incision scar in 2016 in
the Expert opinion of Diagnosis and Treatment of Cesarean Scar
Pregnancy (7).
At present, to the best of our knowledge, there are
no unified standards available for the diagnosis and treatment of
CSP. The available treatments mainly include drug therapy, surgery,
uterine artery embolization, high-intensity focused ultrasound
(HIFU) ablation and combined therapy. At present, the removal of
the pregnancy tissue by surgery is the mainstay treatment.
Patients and methods
General patient information
The clinical data of 25 patients receiving timely
treatment with laparoscopic surgery at the Department of Gynecology
at Zheng Zhou Yi He Hospital (Zhengzhou, China) were collected
between January, 2017 and October, 2020. In addition, a total of 23
patients treated with suction curettage in operative hysteroscopy
were followed-up for 6 months. The following selection criteria
were used: i) A history of cesarean section delivery; ii) early
clinical manifestations of pregnancy; iii) a diagnosis of CSP
determined by ultrasound or confirmed by magnetic resonance imaging
(MRI) according to the recommended diagnostic criteria (8,9); iv)
stable vital signs and the desire to retain the uterus; v) a
gestational age <70 days. The clinical data of the patients who
received laparoscopic surgery are presented in Tables I and II. The patients were 22-40 years of age. A
total of 18 patients had one previous cesarean delivery and 7
patients had two previous cesarean deliveries. All these operations
were classified as lower uterine segment transverse incisions. The
missed menses of all patients ranged from 35 to 70 days. According
to the patient's condition, the surgical method was explained to
them, followed by a comprehensive discussion between the patient
and the surgeon explaining the pros and cons of the surgical
strategy. The patients then signed a surgery consent form as
routine clinical practice. The research protocol was approved by
the Ethics Committee of Zheng Zhou Yi He Hospital. The informed
consent to participate was waived off due to the retrospective
nature of the study.
 | Table IPatient indicators in laparoscopic
group. |
Table I
Patient indicators in laparoscopic
group.
| Patient no. | Age (years) | Time from last
menstruation (days) | Average diameter of
the gestation sac (mm) | No. of cesarean
sections | Period of bleeding
(days) | Initial β-HCG levels
(IU/l) |
|---|
| 1 | 27 | 60 | 36 | 1 | 25 | 1,556.8 |
| 2 | 24 | 70 | 15 | 2 | 30 | 1,348.2 |
| 3 | 39 | 39 | 11 | 2 | 7 | 15,638.9 |
| 4 | 27 | 43 | 18 | 1 | 3 | 38,237.1 |
| 5 | 30 | 50 | 28 | 1 | 19 | 84,059.9 |
| 6 | 26 | 54 | 30 | 1 | 23 | 24,377.0 |
| 7 | 26 | 41 | 16 | 1 | 10 | 1,248.3 |
| 8 | 28 | 39 | 14 | 1 | 9 | 5,632.7 |
| 9 | 31 | 42 | 17 | 1 | 24 | 33,471.1 |
| 10 | 32 | 45 | 22 | 2 | 11 | 6,869.6 |
| 11 | 27 | 48 | 27 | 1 | 8 | 4,956.8 |
| 12 | 25 | 55 | 33 | 1 | 21 | 22,613.2 |
| 13 | 30 | 47 | 23 | 1 | 13 | 53,791.4 |
| 14 | 32 | 51 | 29 | 2 | 19 | 9,467.9 |
| 15 | 27 | 54 | 32 | 1 | 20 | 892.0 |
| 16 | 31 | 49 | 30 | 1 | 10 | 8,653.2 |
| 17 | 29 | 46 | 23 | 1 | 9 | 9,201.8 |
| 18 | 25 | 52 | 31 | 2 | 12 | 11,030.0 |
| 19 | 36 | 48 | 29 | 1 | 16 | 4,250.4 |
| 20 | 30 | 56 | 34 | 1 | 13 | 3,485.2 |
| 21 | 24 | 43 | 20 | 1 | 8 | 12,092.3 |
| 22 | 28 | 50 | 28 | 2 | 18 | 6,241.0 |
| 23 | 27 | 55 | 34 | 1 | 11 | 2,893.3 |
| 24 | 29 | 46 | 25 | 1 | 6 | 8,362.4 |
| 25 | 33 | 59 | 36 | 2 | 20 | 22,013.4 |
 | Table IIPatient indicators in the
laparoscopic group following surgery. |
Table II
Patient indicators in the
laparoscopic group following surgery.
| Patient no. | β-HCG levels (IU/l)
at 3-4 days after surgery | Blood loss during
surgery (ml) | Time for β-HCG
levels to return to normal (days) | Post-operative
uterine thickness of scar (mm) | Menstruation
recovery time (days) |
|---|
| 1 | 48.4 | 1,000 | 15 | 2.5 | 40 |
| 2 | 38 | 800 | 15 | 3.1 | 38 |
| 3 | 1,583 | 200 | 21 | 4.0 | 45 |
| 4 | 947.7 | 50 | 14 | 2.8 | 35 |
| 5 | 6674 | 100 | 28 | 3.2 | 50 |
| 6 | 852.5 | 120 | 14 | 3.0 | 43 |
| 7 | 45 | 50 | 14 | 3.7 | 38 |
| 8 | 328.6 | 700 | 18 | 3.3 | 42 |
| 9 | 862.3 | 100 | 21 | 4.2 | 40 |
| 10 | 410 | 200 | 14 | 2.6 | 37 |
| 11 | 365.7 | 150 | 14 | 3.7 | 45 |
| 12 | 1,123 | 100 | 21 | 4.3 | 48 |
| 13 | 2,341.2 | 850 | 28 | 3.8 | 50 |
| 14 | 582.3 | 100 | 14 | 3.8 | 40 |
| 15 | 41.8 | 80 | 14 | 3.4 | 35 |
| 16 | 230 | 200 | 14 | 3.2 | 32 |
| 17 | 340 | 150 | 14 | 3.0 | 38 |
| 18 | 998.4 | 300 | 21 | 3.5 | 40 |
| 19 | 186.5 | 100 | 14 | 3.4 | 36 |
| 20 | 121.1 | 120 | 14 | 2.9 | 39 |
| 21 | 1,102.8 | 250 | 21 | 3.1 | 42 |
| 22 | 438.3 | 150 | 14 | 4.0 | 40 |
| 23 | 157.2 | 200 | 14 | 3.8 | 35 |
| 24 | 325.8 | 100 | 14 | 3.3 | 37 |
| 25 | 890.3 | 950 | 21 | 3.5 | 45 |
The following clinical findings were observed: i) A
total of 15 patients had a history of cesarean section and
menolipsis; ii) all patients presented with irregular vaginal
bleeding ranging from 3 to 30 days; iii) a total of 6 patients
(patients 2, 7, 11, 15, 20, and 23) had received prior curettage or
medical treatment in local hospitals 2 weeks to 1 month prior.
Patient 15 who was diagnosed with CSP in the local hospitals was
initially treated with 150 mg mifepristone, which was received
orally and with 600 µg misoprostol. After 1 week, the patient
received 50 mg methotrexate (MTX) intramuscularly as the first
treatment was not effective. Patients 2, 7, 20, and 23 had received
prior curettage. The levels of β-human chorionic gonadotropin
(β-HCG) were not reduced and the GS was still present as monitored
by ultrasonography. Patient 11 developed heavy bleeding due to the
blind curettage and was transferred immediately to our hospital.
The other 23 patients were diagnosed by ultrasound and received
suction curettage under hysteroscopy due to their relatively stable
conditions.
The following laboratory findings and specific
examinations were performed: i) The levels of β-HCG were assessed
prior to surgery, and ranged between 892 and 84059.9 IU/l; ii) the
characteristic ultrasound was an empty uterus and empty cervical
canal; iii) the MRI data of patients 1, 5, 12 and 14 were all
indicative of prompt GS, which was convex to the bladder.
Treatment
A total of 25 patients with CSP-II diagnosed by
ultrasound or MRI underwent laparoscopic surgery resection of the
scar with gestational tissue and wound repair to preserve the
uterus. The blood loss during the surgery was estimated to be
50-1,000 ml. The range of β-HCG concentration from the 3rd to the
4th day following surgery was reduced to 38-6,674 IU/l.
Pathological diagnosis identified as
placenta and villus
The surgical procedure was as follows: Laparoscopy
was performed under general anesthesia in the supine lithotomy
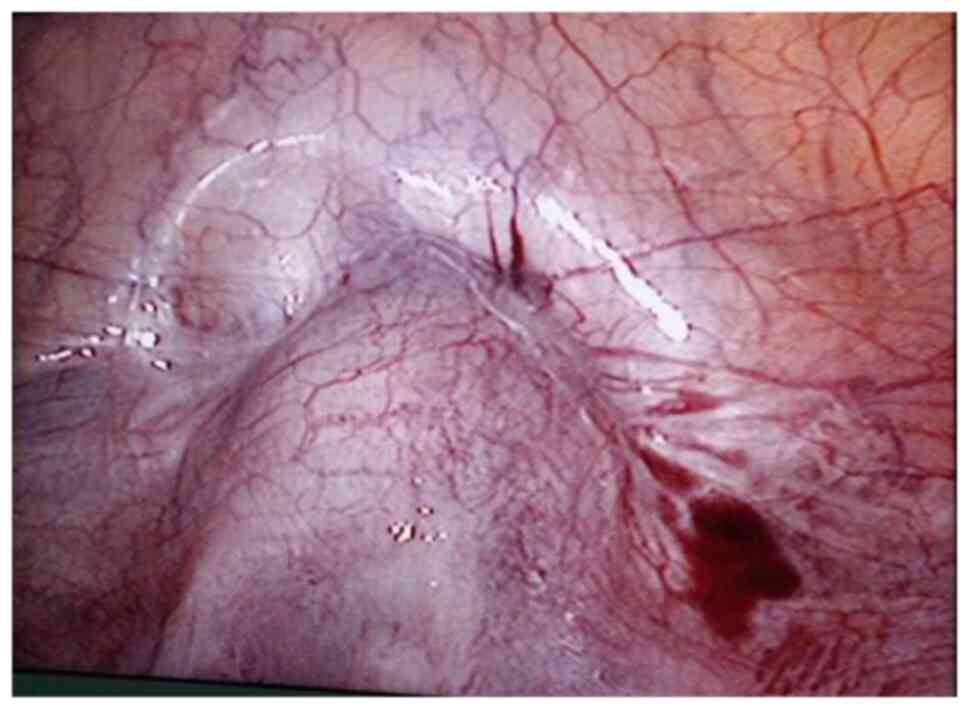
position. Laparoscopy displayed a bulging in the lower uterine
segment (Fig. 1). Subsequently, a 12
IU vasopressin solution was injected into the uterus. The bladder
peritoneum was incised to expose the pregnancy scar; a bulging was
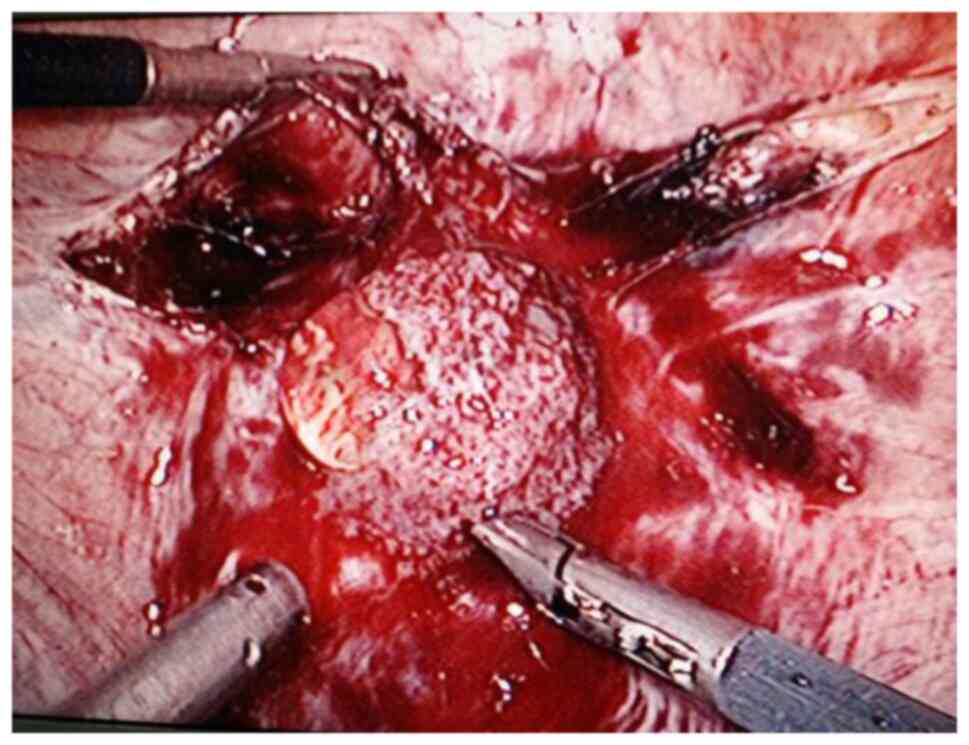
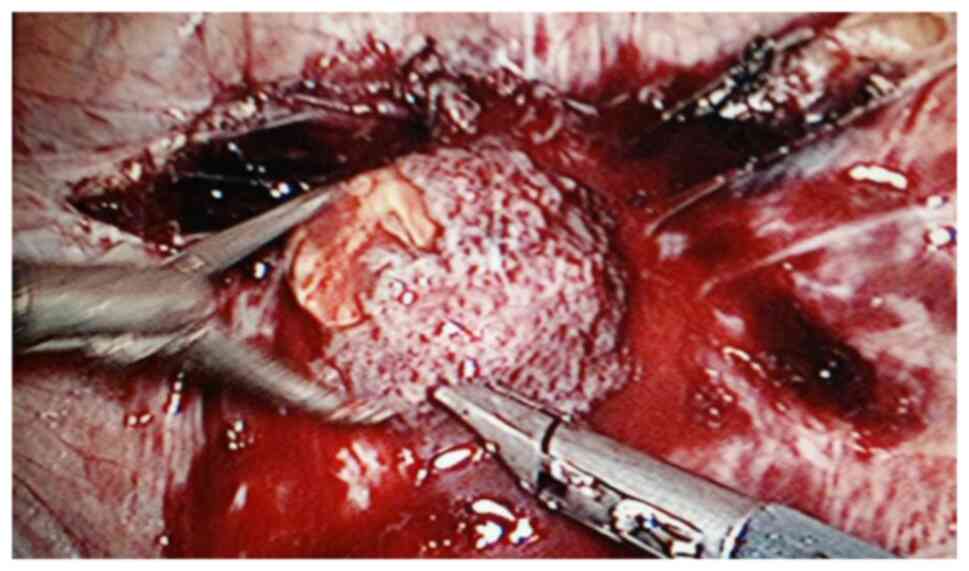
noted comprised of blood vessels and gestational tissues. The
gestational tissue was rapidly removed with grasping forceps,
scissors and an aspirator (Figs. 2
and 3). This procedure was performed
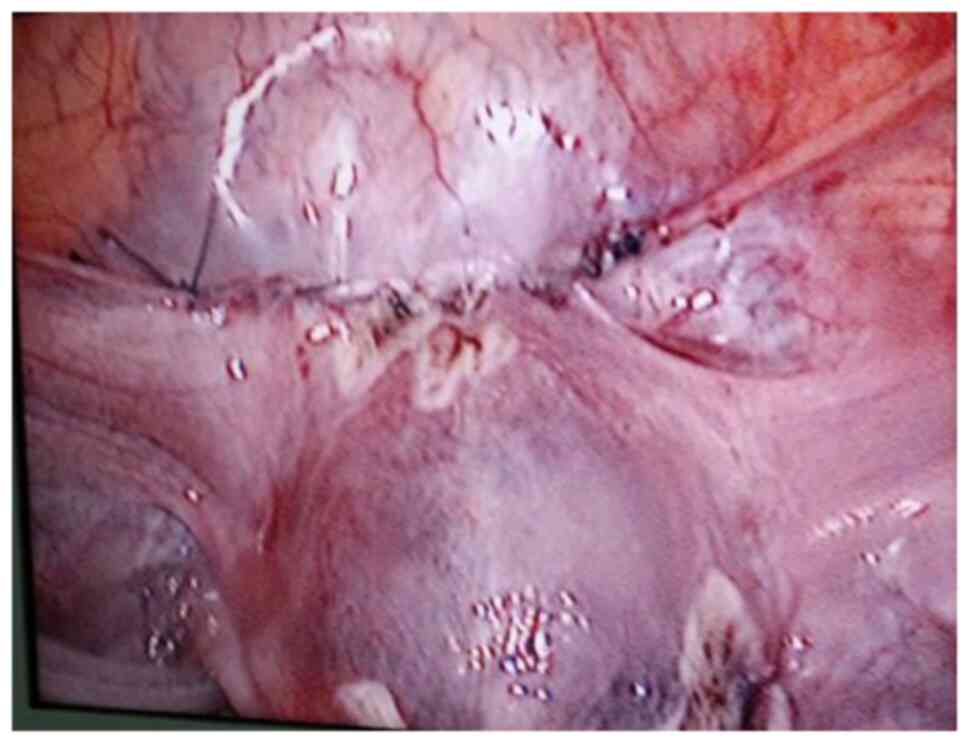
as quickly as possible. The myometrial scar was resected and
stanched by bipolar coagulation. Finaly, a continuous suture with
an absorbable suture (1-0) was used to close the uterine wound
(Fig. 4).
A total of 23 patients in the hysteroscopy group
underwent hysteroscopic removal of the pregnancy tissues and MTX
(50 mg/m2) was administered during surgery. A total of 4
of these patients did not agree to laparoscopic surgical treatment.
Uterine arterial embolization (UAE) was used as a pre-treatment to
reduce the risk of intraoperative bleeding.
Evaluation of therapeutic effects
The clinical effects of the two protocols were
determined based on the evaluation of the following indices:
Bleeding, recovery with preserve fertility, the absence of repeated
surgical intervention or embolization and the lack of any severe
complication. Serum β-HCG levels were measured every 3 to 7 days
following treatment until complete recovery. A transvaginal
ultrasound was performed every 4 weeks to evaluate the condition of
residual lesion and the thickness of the uterine scar.
Statistical analysis
The data were analyzed with SPSS 20.0 software (IBM
Corp.). The continuous data with a normal distribution are
presented as the mean ± standard deviation. Comparisons between the
two groups were analyzed using an unpaired t-test for continuous
variables. The significant level (a) was set at 0.05 and a P-value
<0.05 was considered to indicate a statistically significant
difference.
Results
No significant differences were noted between the
two groups as regards baseline characteristics, age, time from last
menstruation, the average diameter of the gestation sac, the number
of cesarean sections, the period of bleeding and initial human
chorionic gonadotropin (HCG) concentrations (all P>0.05,
Table III).
 | Table IIIComparison of general data of the
patients. |
Table III
Comparison of general data of the
patients.
| Parameter | Laparoscopic
group | Hysteroscopy
group | P-value |
|---|
| Number of cases
(n) | 25 | 23 | |
| Age (years) | 28.800±2.73 | 28.385±2.93 | 0.827 |
| Time from last
menstruation (days) | 49.200±1.27 | 48.000±1.31 | 0.650 |
| Average diameter of
the gestation sac (mm) | 23.400±7.90 | 25.077±7.33 | 0.113 |
| Number of cesarean
sections | 1.267±0.46 | 1.308±0.48 | 0.312 |
| Period of bleeding
(days) | 15.000±7.44 | 12.308±5.15 | 0.125 |
| Initial β-HCG level
(IU/l) |
20,277.393±23793.17 |
21,673.569±20,846.36 | 0.500 |
All 25 laparoscopy procedures were successful,
preserving the uterus without conversion to open laparotomy. The
pathological diagnosis was placenta and villus. Following a
follow-up period of 6 months, no apparent abnormalities were
reported. The time required for the β-HCG levels to reach a normal
level ranged from 2 to 4 weeks. The operation time ranged from 60
to 120 min. Intraoperative hemorrhage ranged from 50 to 1,000
ml.
The comparison between the laparoscopy and
hysteroscopy groups revealed statistically significant differences
(P<0.05, Table IV) as regards
the post-operative expression of HCG, the time required for HCG to
return to normal levels, the post-operative thickness of the
uterine scar and the menstruation recovery time. However, no
significant differences were noted in blood loss during the surgery
(P>0.05). A total of 4 patients in the hysteroscopy group were
treated with UAE to reduce the risk of intraoperative bleeding. The
surgery was successfully completed in all patients without any
surgical complications, such as hemorrhage. However, 2 patients had
residual lesions and 1 patient underwent laparoscopic surgery.
 | Table IVOutcomes of the different
treatments. |
Table IV
Outcomes of the different
treatments.
| Parameter | Laparoscopic
group | Hysteroscopy
group | P-value |
|---|
| After surgery β-HCG
level (IU/l; 3-4 days after surgery) |
1,082.900±1,675.99 |
2,675.385±1,605.72 | 0.017 |
| Blood loss during
operation (ml) | 306.667±339.13 | 336.154±230.91 | 0.79 |
| Time for the β-HCG
level to return to normal (days) | 17.667±5.04 | 27.462±8.79 | 0.001 |
| Post-operative
thickness of uterine scar (mm) | 3.413±0.55 | 2.154±0.36 | <0.001 |
| Recovery time of
menstruation (days) | 41.733±4.99 | 51.769±7.18 | <0.001 |
Discussion
CSP is a relatively infrequent type of ectopic
pregnancy. The cesarean section infiltrate grows into the
myometrium and even penetrates the uterine wall; therefore, in
early pregnancy, it can cause uterine hemorrhage, perforation and
even rupture. Due to its anatomy and pathology, it often causes
uncontrolled bleeding for the blind curettage and may require
hysterectomy, and endanger the lives of the patients. Therefore,
early detection and timely treatment are instrumental to preserve
fertility and avoid severe disease complications.
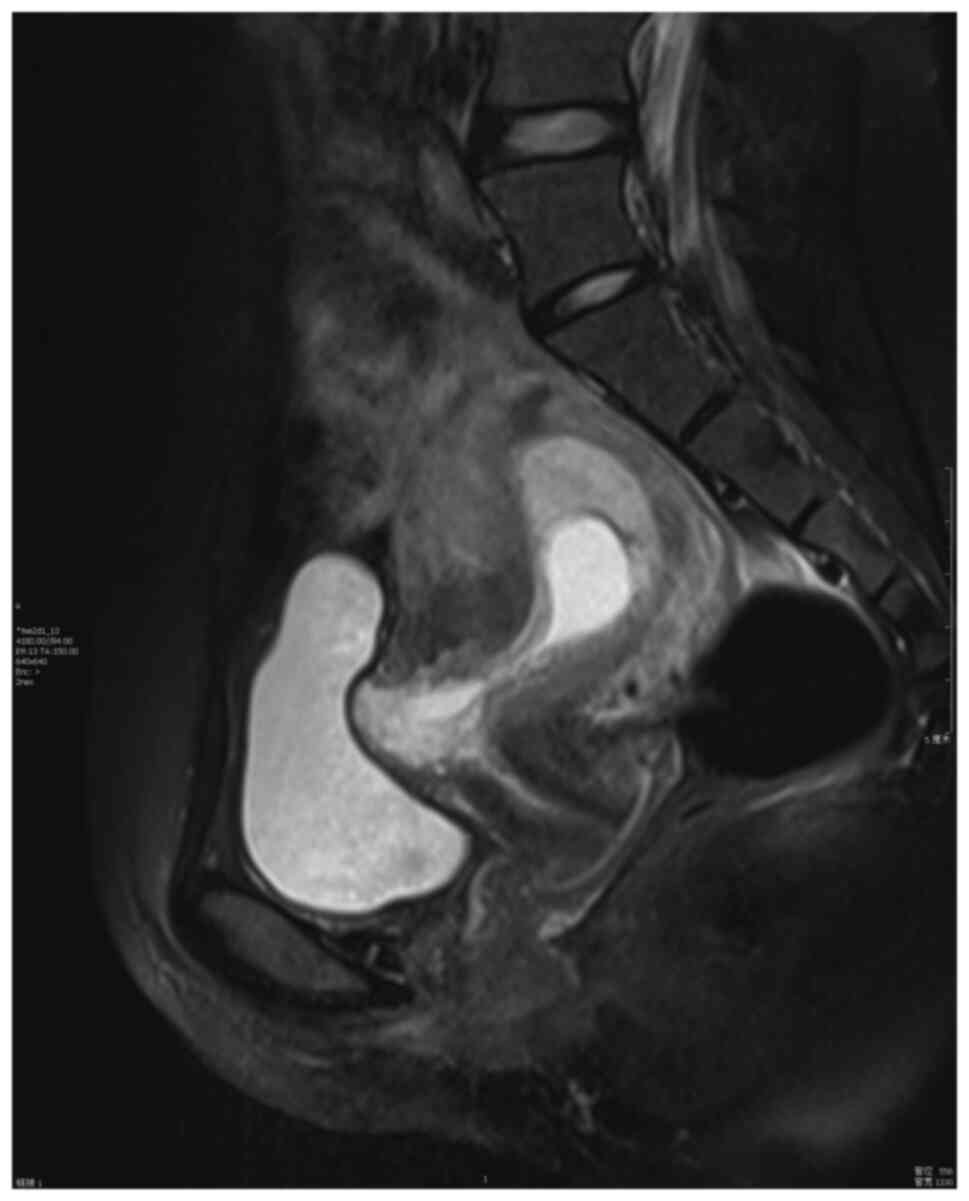
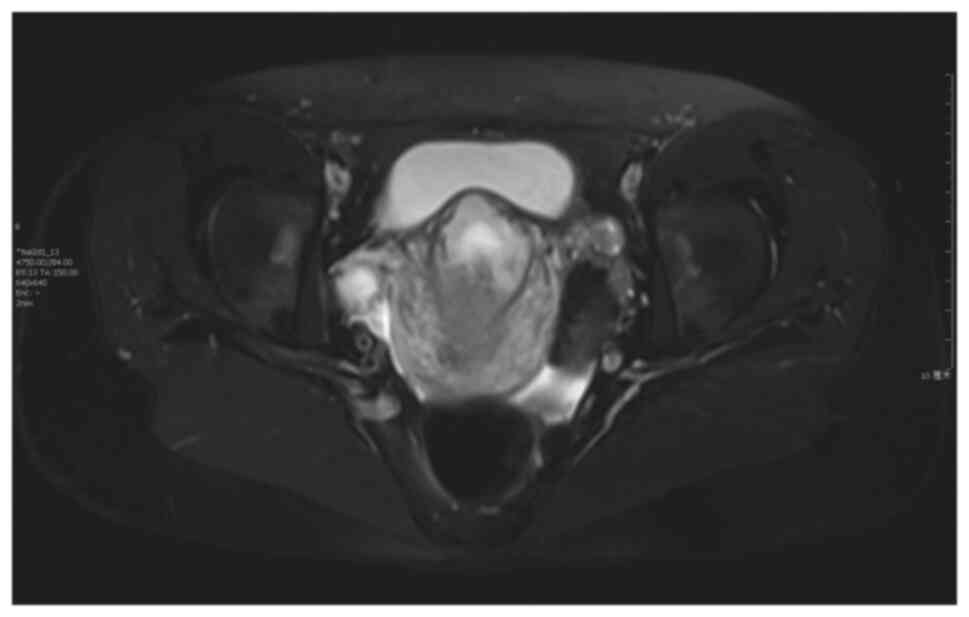
The imaging criteria for diagnosis (Figs. 5 and 6; patient 5) include the following: An
empty uterus and cervical canal; the development of the GS toward
the anterior wall of the isthmic portion; the loss of myometrial
anterior wall continuity on a sagittal plane of the uterus through
the GS; the absence of or diminished healthy myometrium between the
bladder and the sac; the monitoring of peri-trophoblastic vascular
flow with high velocity and low impedance surrounded by the sac
using Doppler examination (10,11). The
pathological features included trophoblast invasion and destructive
growth to the uterine wall. Since the opened blood vessels do not
possess systolic function and the scar tissue cannot contract, a
potential misdiagnosis leading to an artificial or spontaneous
abortion will result in massive hemorrhage (12).
The therapeutic strategies for CSP include drug
therapy, laparotomy surgery resection of gestational tissue,
hysteroscopic treatment, laparoscopic management, uterine artery
drug infusion and embolization, the transvaginal resection of
gestational nidus and repeated HIFU ablation.
The drugs used include mifepristone, MTX and
Radix trichosanthis. These can be used alone or in
combination. MTX and mifepristone are the main drugs of
conservative treatment for ectopic pregnancy. They are mainly
suitable for patients at an early gestational age, with a stable
condition, reduced bleeding and small lesions. Furthermore, this
type of treatment requires a prolonged follow-up period (the HCG
levels require a maximum duration of 4 months to return to normal)
(13).
Traditional laparotomy for the resection of the
lesion is mainly used in patients with critical condition and
severe bleeding. This type of treatment is associated with various
advantages, such as the ability to directly investigate straight
lesions and remove the nidus thoroughly. Patients with barren
requirements can have their scars directly resected with the
gestational tissue; however, the extent of trauma is greater. It
has been reported that open surgery is the optimal treatment option
for CSP, since it may be able to reduce the risk of recurrence of
this condition (14).
Hysteroscopic treatment has become the primary means
of diagnosis and treatment for CSP since the first report of
successful hysteroscopy to treat resection in 2005. Wang et
al (12) indicated that
hysteroscopy could clearly distinguish the GS and implantation area
of the vascular distribution in the embryonic sac and could be used
to guide or direct treatment. It may be an effective method for the
treatment of CSP. Moreover, it has the advantages of a shorter
operation time, reduced bleeding, rapid recovery, a shorter
hospitalization time, lower costs and the preservation of the
uterus. The typical patient with CSP-II may experience the risk of
hemorrhage and consequently the treatment is changed to laparotomy
or laparoscopic surgery.
Uterine artery chemoembolization is a hotspot
investigated in recent years for the treatment of CSP; its
advantages are the following: Firstly, focal perfusion MTX may
hinder the growth of trophoblast cells and lead to the inhibition
of embryonic growth; the increase in the local drug concentration
can terminate the bleeding of the bilateral uterine artery caused
by embolization, reduce the risk of peri-operative bleeding due to
curettage, allow the absorption of the thrombus following vascular
embolization, and post-operatively recover the unobstructed uterine
artery. The most important advantage is that the uterine functions
are not affected. UAE is often used as a pre-treatment.
Kang et al (15) first reported a case of CSP treated
successfully with transvaginal resection of the gestational nidus
in 2011. Due to its reduced complications, rapid post-operative
recovery and smaller trauma, gradually, it is widely used in
clinical practice, although it includes the shortcomings of a small
surgical field and difficult exposure.
The resection of the gestational tissue and repair
scar by laparoscopic surgery is suitable for the GS deep
implantation toward the myometrium and bulging from the uterine
serosal surface to abdominal cavity and bladder. Lee et al
(3) first reported a case of CSP
successfully treated with laparoscopy in 1999. Laparoscopic surgery
provides greater security; in the case of intraoperative bleeding,
the patient can undergo bilateral uterine artery ligation; however,
this requires advanced technology. The significant risks of CSP-II
include severe bleeding, perforation and rupture. During the blind
curettage used for the misdiagnosis of normal pregnancy, massive
hemorrhage and uterine rupture can occur, leading to an emergency
hysterectomy, which can endanger the lives of the patients.
Therefore, surgery should be the first choice for patients with
CSP-II. In the present study, all 25 operations were successfully
performed using laparoscopy with the surgical resection of the
gestational tissue and wound repair to preserve fertility without
conversion to open laparotomy. The key point during the operation
is to avoid injury to the bladder and ureter following the
separation of the bladder from the lower uterine segment.
Furthermore, the GS and uterine scar are completely excised as much
as possible (16). In the present
study, patients 1, 2, 8, 13 and 25 demonstrated a heavy blood loss
of almost 1,000 ml during the resection of the scar with the
gestational tissue. This was due to long-term bleeding following
the last menstruation (~1 month). A high amount of necrotic tissue
and obsolete hematoceles were observed in the GS; therefore, a
longer time would be required to resect the GS. Furthermore, the
operation included a limited time period to undergo bilateral
uterine artery ligation and reduce bleeding. Patients 6, 12, 14 and
23 were administered bilateral uterine artery ligation prior to the
resection of the scar, since the gestational mass size was >3
cm. Furthermore, the bleeding time of these 4 patients was >20
days. The blood loss during the surgery of these 4 patients was
markedly lower than that noted in patients 1, 2, 8, 13 and 25.
Therefore, the therapeutic approach to excise the uterine scar and
repair the uterine wound would depend on the size of the
gestational mass indicated by the ultrasound or the MRI scan.
However, considering the dense pre-operative adhesion formation,
the selection of transvaginal resection of the gestational nidus
may be optimal. This is due to the difficulty in separating the
bladder from the lower uterine segment and the possibility to
damage the bladder during laparoscopcy.
In conclusion, to reduce the incidence rate of CSP,
particular attention should be paid to examine the indication for
cesarean section in primiparae (17). The optimal therapeutic strategy for
CSP needs to ensure early detection and early treatment to reduce
bleeding and avoid the possibility of hysterectomy. Following the
diagnosis of CSP, the appropriate treatment needs to be selected by
clinicians according to the different types of patients, which
includes drug therapy, interventional therapy, surgical or combined
therapy. According to the results of the present study, which
included 25 patients, and the results reported by similar research
(18), laparoscopy is an effective
surgical method with limited associated trauma; it is also a safe
procedure devoid of complications and an efficient method with
which to completely remove the pregnancy tissue in a hysteroscopic
manner combined with laparoscopic surgery and the reversible
ligation of the uterine artery (18). The resection of the scar with the
gestational tissue and the wound repair caused by laparoscopy is
considered an effective method for the treatment of CSP-II. It can
remove the nidus directly and perform bilateral uterine artery
ligation to reduce bleeding; moreover, it removes the old scar and
can also increase the success rate of secondary pregnancy.
Currently, to the best of our knowledge, only a limited number of
reports have been published on the recurrence of laparoscopic
management for CSP-II, which may minimize the recurrent risk of CSP
through the resection of the old scar and may facilitate the new
wound repair by laparoscopy. However, the long-term complications
and safety require additional research studies and follow-up
observations.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials.
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XML and LY provided the original idea for the study
and contributed to the design of the experiments. XML and NNW
conducted the experiments and collected the data. XML and XFX
analyzed the data and prepared the original draft of the
manuscript. LY and XFX confirm the authenticity of all the raw
data. All authors contributed to the interpretation and/or
discussion of the results, leading to the final version of the
manuscript, and all authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
The research protocol was approved by the Ethics
Committee of Zheng Zhou Yi He Hospital. The patients signed a
surgery consent form as routine clinical practice. The informed
consent to participate was waived off due to the retrospective
nature of the study.
Patient consent for publication
The patients provided written informed consent for
their data to be published.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Herman A, Weinraub Z, Avrech O, Maymon R,
Ron-El R and Bukovsky Y: Follow up and outcome of isthmic pregnancy
located in a previous caesarean section scar. Br J Obstet Gynaecol.
102:839–841. 1995.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Seow KM, Hwang JL, Tsai YL, Huang LW, Lin
YH and Hsieh BC: Subsequent pregnancy outcome after conservative
treatment of a previous cesarean scar pregnancy. Acta Obstet
Gynecol Scand. 83:1167–1172. 2004.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Lee CL, Wang CJ, Chao A, Yen CF and Soong
YK: Laparoscopic management of an ectopic pregnancy in a previous
caesarean section scar. Hum Reprod. 14:1234–1236. 1999.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rotas MA, Haberman S and Levgur M:
Cesarean scar ectopic pregnancies: Etiology, diagnosis, and
management. Obstet Gynecol. 107:1373–1381. 2006.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Vial Y, Petignat P and Hohlfeld P:
Pregnancy in a cesarean scar. Ultrasound Obstet Gynecol.
16:592–593. 2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Gurol-Urganci I, Bou-Antoun S, Lim CP,
Cromwell DA, Mahmood TA, Templeton A and van der Meulen JH: Impact
of Caesarean section on subsequent fertility: A systematic review
and meta-analysis. Hum Reprod. 28:1943–1952. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Family Planning Subgroup, Chinese Society
of Obstetrics and Gynocology, Chinese Medical Association. Expert
opinion of diagnosis and treatment of cesarean scar pregnancy
(2016). Zhonghua Fu Chan Ke Za Zhi. 51:568–572. 2016.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
8
|
Timor-Tritsch IE, Monteagudo A, Cali G,
Vintzileos A, Viscarello R, Al-Khan A, Zamudio S, Mayberry P,
Cordoba MM and Dar P: Cesarean scar pregnancy is a precursor of
morbidly adherent placenta. Ultrasound Obstet Gynecol. 44:346–353.
2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Timor-Tritsch IE, Monteagudo A, Cali G, El
Refaey H, Kaelin Agten A and Arslan AA: Easy sonographic
differential diagnosis between intrauterine pregnancy and cesarean
delivery scar pregnancy in the early first trimester. Am J Obstet
Gynecol. 215:225.e1–7. 2016.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Fylstra DL: Ectopic pregnancy within a
caesarean scar: A review. Obstet Gynecol Surv. 57:537–543.
2002.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Jurkovic D, Hillaby K, Woelfer B, Lawrence
A, Salim R and Elson CJ: First-trimester diagnosis and management
of pregnancies implanted into the lower uterine segment Cesarean
section scar. Ultrasound Obstet Gynecol. 21:220–227.
2003.PubMed/NCBI View
Article : Google Scholar
|
|
12
|
Wang YL, Su TH and Chen HS: Laparoscopic
management of an ectopic pregnancy in a lower segment cesarean
section scar: A review and case report. J Minim Invasive Gynecol.
12:73–79. 2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Doubilet PM, Benson CB, Frates MC and
Ginsburg E: Sonographically guided minimally invasive treatment of
unusual ectopic pregnancies. J Ultrasound Med. 23:359–370.
2004.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Fylstra DL, Pound-Chang T, Miller MG,
Cooper A and Miller KM: Ectopic pregnancy within a cesarean
delivery scar: A case report. Am J Obstet Gynecol. 187:302–304.
2002.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kang SY, Park BJ, Kim YW and Ro DY:
Surgical management of caesarean scar ectopic pregnancy:
Hysterectomy by transvaginal approach. Fertil Steril. 96:E25–E28.
2011.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang HY, Zhang J, Li YN, Wei W, Zhang DW,
Lu YQ and Zhang HF: Laparoscopic management or laparoscopy combined
with transvaginal management of type II cesarean scar pregnancy.
JSLS. 17:263–272. 2013.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Delbaere I, Cammu H, Martens E, Tency I,
Martens G and Temmerman M: Limiting the caesarean section rate in
low risk pregnancies is key to lowering the trend of increased
abdominal deliveries: An observational study. BMC Pregnancy
Childbirth. 12(3)2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Huang L, Zhao L and Shi H: Clinical
efficacy of combined hysteroscopic and laparoscopic surgery and
reversible ligation of the uterine artery for excision and repair
of uterine scar in patients with type II and III cesarean scar
pregnancy. Med Sci Monit. 26(e924076)2020.PubMed/NCBI View Article : Google Scholar
|