Introduction
Testicular cancer is an uncommon neoplasm,
contributing to 1.5% of all malignancies in the male population
(1). This type of cancer is more
commonly found in young males (15-35 years of age); however, it can
also manifest in elderly individuals (2,3). Tumors
originating in the testicles are classified into germ-cell and
non-germ-cell tumors. The overwhelming majority (>95%) stem from
germ-cell lineage. Germ cell tumors encompass seminoma,
non-seminoma and exceedingly rare spermatocytic tumors (ScTs)
(4). The prevalence of germ cell
tumors is increasing globally, although notable regional variations
are evident. These tumors are exceptionally infrequent in Africa,
among Afro-American communities, and in Asia. Conversely, there
appears to be a greater occurrence of such tumors in Nordic
European nations, particularly in Denmark, Norway and Sweden
(4). ScT is a rare form of germ cell
tumor, comprising less than 1% of all cases. It is most commonly
diagnosed in males in their 60s and 70s. This tumor is slow-growing
and typically does not spread to other parts of the body. However,
it can still be life-threatening if not diagnosed and treated at an
early stage (4,5). It was previously classified as a type
of seminoma. However, it is currently recognized as a distinct
entity due to several important differences (5). The distinctive clinical characteristics
that differentiate ScTs from classical seminoma include
presentation at an elderly age, a lack of an undescended testicle
and a diminished inclination for metastasis (6). However, there is a minimal risk of
recurrence and metastasis, particularly among older patients
(7). The appropriate management of
individuals with ScTs has not yet been firmly established due to
their exceptionally low occurrence. Radical orchiectomy is
recommended by some experts, while others argue that testis-sparing
surgery may provide an adequate alternative (5,8).
The present study describes a rare case of a
spermatocytic tumor in an elderly male, including presentation,
investigations, management and follow-up. The present study aimed
to avoid citing predatory publications based on a well-known
predatory list (9).
Case report
Patient information
A 68-year-old male patient presented to the Urology
Clinic at Smart Health Tower, reporting a gradually enlarging,
painless right testicular mass for a period of 5 years. The case
had a history of right testicular blunt trauma 5 years prior. He
had ignored his condition until the mass grew to a size that began
to concern him.
Clinical findings
Upon a physical examination, it was found that he
had an enlarged, non-tender right testicular mass with no
associated palpable inguinal lymph nodes, and the left testis
appeared normal.
Diagnostic assessment
The levels of tumor markers, including
alpha-fetoprotein (AFP; 1.81 ng/ml; normal range, 0-40 ng/ml) and
human chorionic gonadotropin (hCG; 0.448 µIU/ml; normal range,
<3 µIU/ml) were within normal limits, while those of lactate
dehydrogenase were elevated to 400.3 U/l (normal range, 135-225
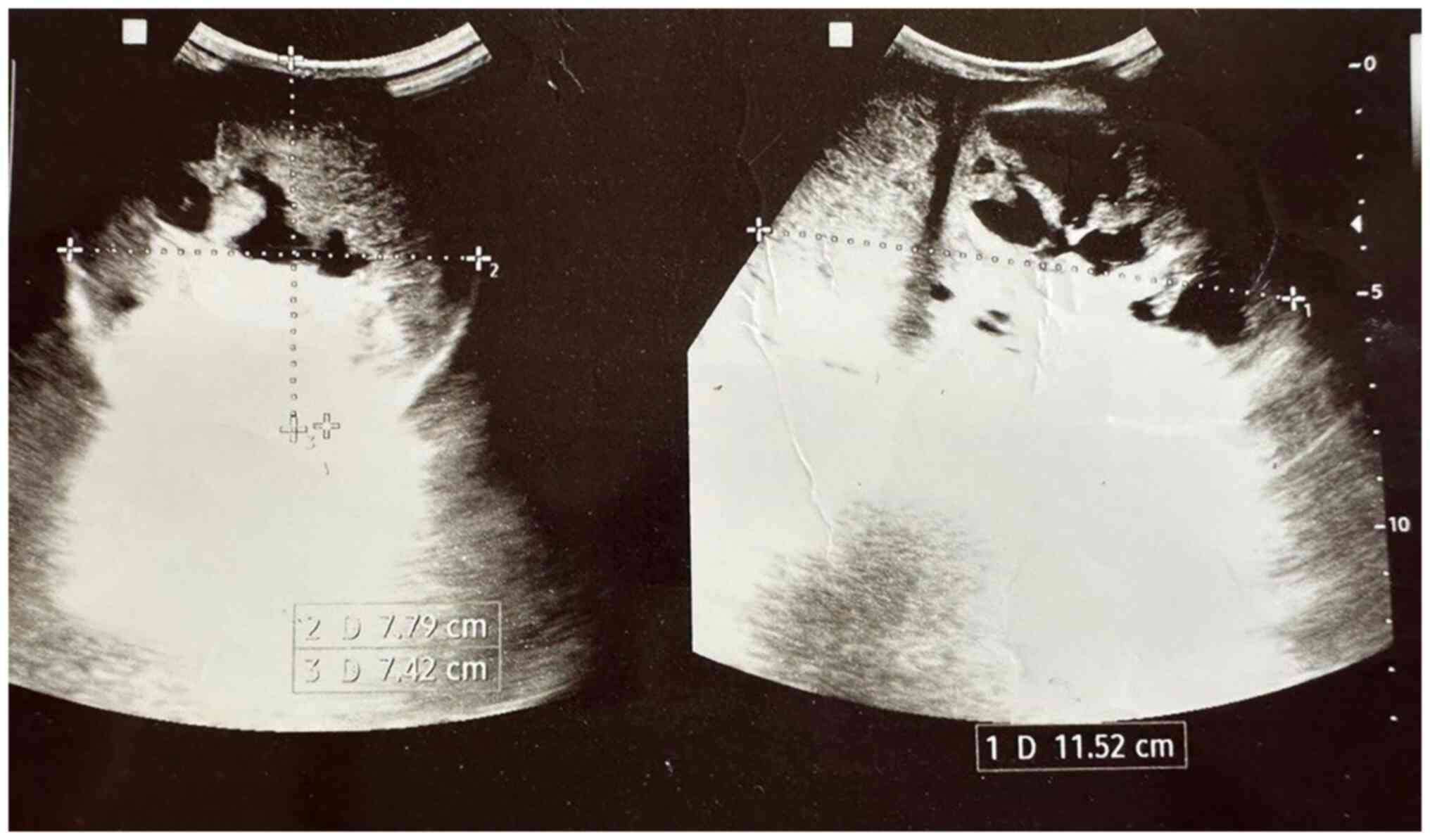
U/l). Scrotal ultrasound (US) imaging revealed a complex
heterogeneous mass almost replacing the right testis and measuring
11x8x7 cm (Fig. 1), while the left
testis appeared normal. No para-aortic lymph nodes were detected on
an abdominal US scan.
Therapeutic intervention
Under spinal anesthesia, a right radical
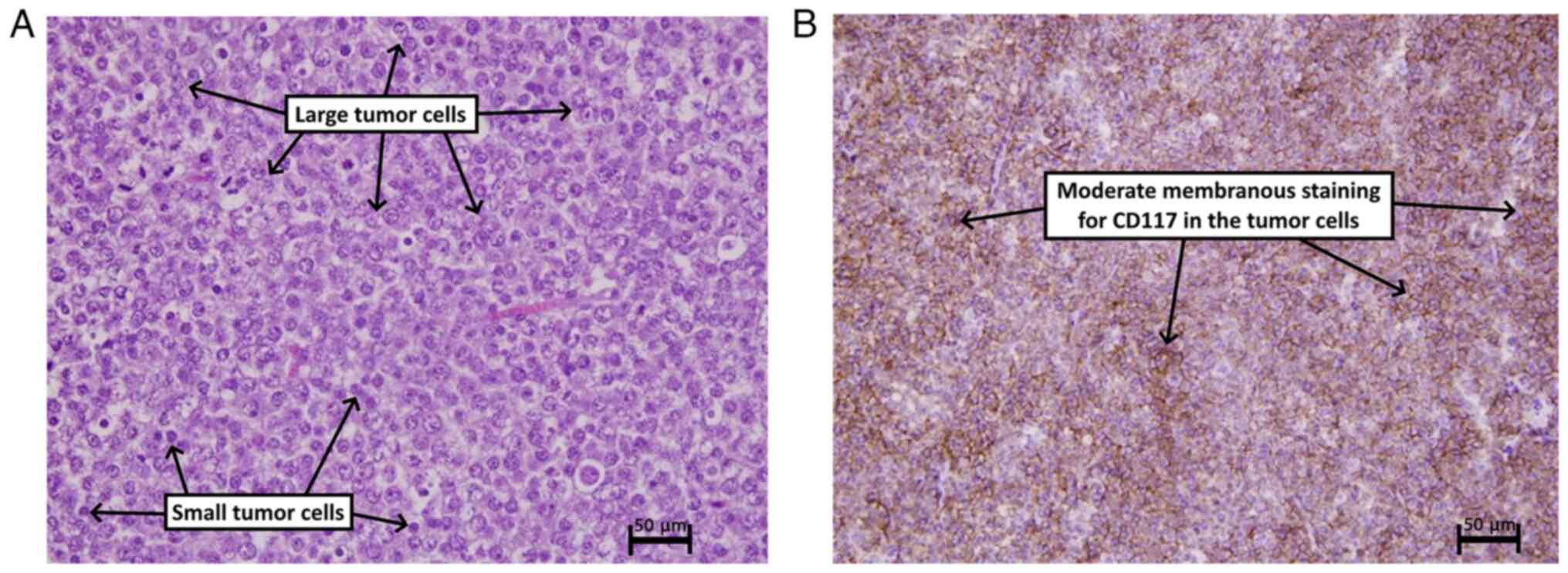
orchidectomy was performed. Histopathological and
immunohistochemical analysis revealed the presence of a
spermatocytic tumor in the testis (Fig.
2).
The histopathological examination was performed by
the authors' laboratory as follows: The sections (5-µm-thick) were
paraffin-embedded and fixed with 10% neutral buffered formalin at
room temperature for 24 h. They were then stained with hematoxylin
and eosin (Bio Optica Co.) for 1-2 min at room temperature, and
examined under a light microscope (Leica Microsystems GmbH).
For immunohistochemistry, the sections
(4-6-µm-thick) were cut from the paraffin blocks and transferred
onto glass slides with an electric charge. These slides were then
placed in an oven at 60˚C overnight. Antigen retrieval was carried
out using the Dako PT Link (Agilent Technologies, Inc.) by
immersing the sections in boiling water at 100˚C for 5 to 10 min.
Depending on the target antibody, a solution with either pH 6.0 or
9.0 was used. Following antigen retrieval, the slides were washed
for 15 min with a 20 ml buffer solution (0.05 mol/l Tris/HCl, 0.15
mol/l NaCl, 0.05% Tween-20, pH 7.6) at room temperature. To
facilitate this process, the slides were marked using the Dako Pen
(Agilent Technologies, Inc.). Endogenous peroxidase activity was
blocked by applying a 3% hydrogen peroxide solution. Subsequently,
primary antibodies were applied at room temperature followed by
incubation at room temperature (25˚C) for 80 min. Afterward, the
secondary antibody, which was horseradish peroxidase, was applied,
along with the chromogen (diaminobenzidine), both at room
temperature for 15 min. To achieve counterstaining, hematoxylin
Gill II was applied at room temperature for 30 sec. Finally, the
slides were allowed to dry, and coverslips were affixed.
Follow-up
The post-operative period was uneventful, and the
metastatic workup, including chest and abdominal computed
tomography scans, revealed no evidence of metastasis. The patient
was discharged the day after the surgery and ws instructed to
return for regular follow-up appointments.
Discussion
Continual discussions exist regarding the theory
that suggests precursor cells of ScTs arise during embryogenesis
(10). Certain scholars, however,
hold the view that ScTs emerge from fully matured cells such as
pachytene spermatocytes (5). In
addition, both the morphological and immunohistochemical attributes
of the tumor cells have indicated an origin from spermatogonial
stem cells (11). These tumors
originate from primary spermatocytes that have progressed to at
least the initiation stage of prophase meiosis (5). They are characterized by a distinctive
amplification of chromosome 9, which corresponds to the DMRT1 gene
and are consistently devoid of any association with other forms of
germ cell tumors (5).
ScTs are rare, accounting for <1% of all
testicular cancers. These type of tumors predominantly affect older
males, typically in their 60s and 70s (7). However, previous studies have reported
the disease in young males aged <40 years and suggested for
clinicians and pathologists to be cognizant of the fact that ScTs
can manifest even in young patients. In a systematic review of 146
cases performed by Grogg et al (5), it was revealed that the majority of the
cases presented with testicular pain and/or enlargement. In
addition, the tumor in several cases was diagnosed after workup for
other concerns, a such as infertility, hydrocele, metastasis,
weight loss and back pain (5). They
also reported that the tumor caused no changes in the levels of
testosterone, estrogen or the clinical features of gynecomastia
(5). In the case presented herein,
the gradual enlargement of the painless testicular mass over a
5-year period is consistent with the indolent nature of ScTs. The
patient's history of blunt testicular trauma 5 years prior to
presentation raises the possibility of a causal relationship,
although the exact etiology of ScTs remains uncertain (4,7). It is
worth noting that the patient neglected his condition until the
mass grew to a size that began to concern him. This delay in
seeking medical attention underscores the importance of raising
awareness about testicular health and the significance of early
detection. In a previous case series study, Rabade et al
(8) reported that unilateral
testicular masses were evident in all patients. Instances of
bilateral involvement have been infrequently documented, primarily
manifesting as metachronous tumors. The range of tumor size has
been diverse, spanning from 2 to 20 cm, with an average measurement
of 7 cm (8). A clinical examination
plays a crucial role in the evaluation of testicular masses. In the
case in the present study, the non-tender, enlarged right
testicular mass (11 cm) with no associated palpable inguinal lymph
nodes raised the suspicion of a testicular tumor. The contralateral
testis appeared normal, which is consistent with the unilateral
nature of ScTs (1,3,4,7,8). The
levels of tumor markers, such as AFP and hCG were within the normal
range, which is consistent with the diagnosis of ScT (1,12). It
should be noted that these tumor markers are typically elevated in
other types of testicular cancers, such as yolk sac tumor and
choriocarcinoma (12).
In the case presented herein, scrotal US imaging
revealed a complex heterogeneous mass almost replacing the right
testis, confirming the presence of a sizable tumor. The absence of
para-aortic lymph nodes on abdominal ultrasound scan suggested
localized disease without metastasis (13,14).
Another study on managing 21 cases reported no metastasis (8). Although, other scholars have reported
14 cases of metastasis out of 146 cases, and ScTs with other
histological variants such as sarcoma and anaplastic tumors tend to
metastasize more than the pure ScTs (5,8). Despite
the absence of trials regarding the management of ScTs, however,
orchiectomy remains the standard treatment option based on the
reports and clinical series (5).
Testis sparing surgery is not recommended due to the difficulty of
differentiating ScTs from pure seminoma, in some cases even in
frozen section analysis (5). The
case in the present study underwent radical orchiectomy, which
allows for accurate staging and histopathological examination of
the excised specimen (5). The
differential diagnoses of ScTs in histopathological or
immunohistochemistry can be classical seminoma, embryonal cancer
and non-Hodgkin lymphoma. Hence, it is recommended for pathologists
to be firmly aware of the distinctive features of each disease
(8). In the case in the present
study, histopathological and immunohistochemical analyses confirmed
the diagnosis of a spermatocytic tumor. Post-operatively, the
patient had an uneventful recovery. Metastatic workup, including
chest and abdominal CT scans, revealed no evidence of metastasis.
Regular follow-up appointments were scheduled to monitor the
patient's condition and detect any potential recurrence or
metastasis. Long-term surveillance is crucial due to the risk of
late recurrence, even though these tumors tend to exhibit a slow
and indolent course (15,16).
In conclusion, the present case report underscores
the rarity and slow-growing nature of testicular ScTs, emphasizing
the significance of prompt identification and treatment. Clinical
examination, tumor markers, and imaging techniques are vital for
accurate diagnosis. Radical orchidectomy is crucial for ensuring
favorable outcomes, and long-term monitoring is necessary to detect
any potential recurrence or spread.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
RB was the major contributor to the conception of
the study, as well as in the literature search for related studies.
HOA and MBAA were involved in the literature review, in the writing
of the manuscript, and in the analysis and interpretation of the
patient's data. FHK, IA, RMA, KKM, SSF, AMA and RJR were involved
in the literature review, in the design of the study, in revision
of the manuscript and in the processing of the figures. RB and FHK
confirm the authenticity of all the raw data. All authors have read
and approved the final manuscript.
Ethics approval and consent to
participate
The patient provided written informed consent to
participate and publish any related data in the present study.
Patient consent for publication
The patient provided written informed consent to
participate and publish any related data and images in the present
study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Facchini G, Rossetti S, Berretta M,
Cavaliere C, D'Aniello C, Iovane G, Mollo G, Capasso M, Della Pepa
C, Pesce L, et al: Prognostic and predictive factors in testicular
cancer. Eur Rev Med Pharmacol Sci. 23(3885)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Mahmood ZH, Mohemed FM, Fatih BN, Qadir AA
and Abdalla SH: Cancer publications in one year (2022); a
cross-sectional study. Barw Med J. 1(no. 2)2023.
|
|
3
|
Williamson SR, Delahunt B, Magi-Galluzzi
C, Algaba F, Egevad L, Ulbright TM, Tickoo SK, Srigley JR, Epstein
JI and Berney DM: Members of the ISUP Testicular Tumour Panel. The
World Health Organization 2016 classification of testicular germ
cell tumours: A review and update from the international society of
urological pathology testis consultation panel. Histopathology.
70:335–346. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Secondino S, Rosti G, Tralongo AC, Nolè F,
Alaimo D, Carminati O, Naspro RLJ and Pedrazzoli P: Testicular
tumors in the ‘elderly’ population. Front Oncol.
12(972151)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Grogg JB, Schneider K, Bode PK, Wettstein
MS, Kranzbühler B, Eberli D, Sulser T, Beyer J, Hermanns T and
Fankhauser CD: A systematic review of treatment outcomes in
localised and metastatic spermatocytic tumors of the testis. J
Cancer Res Clin Oncol. 145:3037–3045. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Pendlebury S, Horwich A, Dearnaley DP,
Nicholls J and Fisher C: Spermatocytic seminoma: A
clinicopathological review of ten patients. Clin Oncol (R Coll
Radiol). 8:316–318. 1996.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Colecchia M and Bertolotti A:
Spermatocytic tumor. In: Encyclopedia of Pathology. Van Krieken J
(ed). Springer, Cham, pp1-3, 2019.
|
|
8
|
Rabade K, Panjwani PK, Menon S, Prakash G,
Pal M, Bakshi G and Desai S: Spermatocytic tumor of testis: A case
series of 26 cases elucidating unusual patterns with diagnostic and
treatment dilemmas. J Cancer Res Ther. 18 (Suppl 2):S449–S454.
2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Muhialdeen AS, Ahmed JO, Baba HO, Abdullah
IY, Hassan HA, Najar KA, Mikael TM, Mustafa MQ, Mohammed DA, Omer
DA, et al: Kscien's list; A new strategy to discourage predatory
journals and publishers (second version). Barw Med J. 1:30–32.
2023.
|
|
10
|
Menon S, Karpate A and Desai S:
Spermatocytic seminoma with rhabdomyosarcomatous differentiation: A
case report with a review of the literature. J Cancer Res Ther.
5:213–215. 2009.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Waheeb R and Hofmann MC: Human
spermatogonial stem cells: A possible origin for spermatocytic
seminoma. Int J Androl. 34:e296–e305. 2011.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Pedrazzoli P, Rosti G, Soresini E, Ciani S
and Secondino S: Serum tumour markers in germ cell tumours: From
diagnosis to cure. Crit Rev Oncol Hematol.
159(103224)2021.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Marko J, Wolfman DJ, Aubin AL and
Sesterhenn IA: Testicular seminoma and its mimics: From the
radiologic pathology archives. Radiographics. 37:1085–1098.
2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Xue N, Wang G, Zhang S and Lu Y: The value
of contrast-enhanced ultrasonography in differential diagnosis of
primary testicular germ cell tumors and non-germ cell tumors over
50 years old. Front Oncol. 13(1090823)2023.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ruf CG, Schmidt S, Kliesch S, Oing C,
Pfister D, Busch J, Heinzelbecker J, Winter C, Zengerling F, Albers
P, et al: Testicular germ cell tumours' clinical stage I:
Comparison of surveillance with adjuvant treatment strategies
regarding recurrence rates and overall survival-a systematic
review. World J Urol. 40:2889–2900. 2022.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Stoop H, van Gurp R, de Krijger R, Geurts
van Kessel A, Köberle B, Oosterhuis W and Looijenga L: Reactivity
of germ cell maturation stage-specific markers in spermatocytic
seminoma: Diagnostic and etiological implications. Lab Invest.
81:919–928. 2001.PubMed/NCBI View Article : Google Scholar
|