Introduction
Traumatic neuroma is a rare, non-neoplastic lesion
that forms at the proximal end of a damaged nerve following trauma
or surgery, as a healing process involving the hyperplastic
proliferation of nerve fibers and connective tissue (1). Although it is a common pathology
following trauma and surgeries, it is rarely reported in clinical
practice (2). The lesion can develop
after any surgery, particularly an amputation (3). The lower extremities are the most
frequent site of occurrence, followed by the head and neck, radial
nerve and brachial plexus (1). A
biliary neuroma is a relatively rare benign tumor that can be
classified into two types: Primary and traumatic neuromas (4). On rare occasions, cholecystectomy has
been found to be associated with the etiology of this condition.
The traumatic neuroma may mimic malignant tumors due to its vague
clinical and imaging features (3).
Pathologies involving the biliary tree, such as infections,
inflammatory diseases, trauma, stones, or surgery, can be
considered as etiological factors for traumatic neuroma (5).
The present study reports a rare case of
post-laparoscopic cholecystectomy biliary tree traumatic neuroma
with reactive lymph nodes that was considered to be malignant
intraoperatively.
Case report
Patient information
A 47-year-old female patient presented to the GIT
Department at Smart Health Tower, with a history of laparoscopic
cholecystectomy for symptomatic gallstones, who had been affected
by upper abdominal pain and anorexia 4 weeks prior to presentation.
There was no vomiting or diarrhea. She had a negative past medical
history.
Clinical findings
Upon a physical examination, a yellow discoloration
of the sclera without pallor was observed. Upon palpation, the
abdomen was soft, non-distended and non-tender, and there was no
evidence of organomegaly. Bowel sounds were positive. A digital
rectal examination revealed normal-colored stool. The patient's
vital signs were normal.
Diagnostic assessment
The analyses of blood parameters, including white
blood cell count, serum creatinine, blood urea, C-reactive protein,
serum amylase, carcinoembryonic antigen and CA 19-9 levels,
revealed results within the normal range. The liver function
profile was elevated (total serum bilirubin was 4.2 mg/dl and
alkaline phosphatase was 250 IU/l). Magnetic resonance
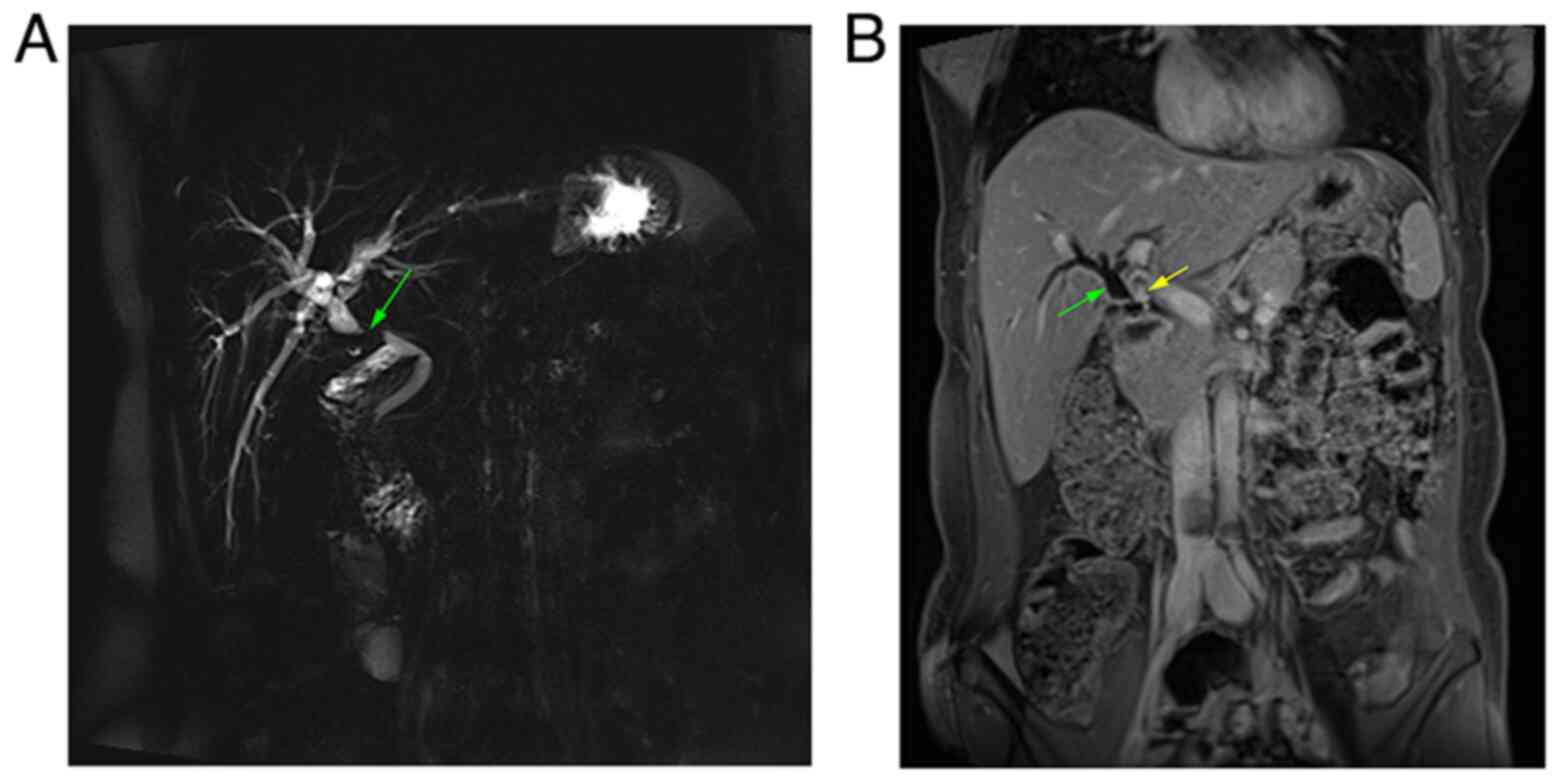
cholangiopancreatography (MRCP) revealed a dilated proximal bile
duct and mild dilatation of the intrahepatic biliary tree due to a
1-cm stricture, 16 mm distal to the confluence of the right and
left hepatic ducts (Fig. 1). Dynamic
liver magnetic resonance imaging (MRI) was performed to exclude
tumors, and it revealed the same finding of bile duct stricture due
to a clip on the bile duct. Based on this finding, the case was
diagnosed as a post-operative bile duct stricture. An endoscopic
retrograde cholangiopancreatography (ERCP) was initially planned to
be conducted.
Therapeutic intervention
The case was discussed by the multidisciplinary team
of Smart Health Tower, and by reviewing the imaging findings, the
team suspected thickening in the area of the stricture; therefore,
it was decided that surgery should be performed and not an ERCP.
Under general anesthesia, a laparotomy was made through donor
incisions. Intraoperatively, a hard bile duct mass of 2 cm in the
greatest dimension was observed with multiple enlarged lymph nodes
in the peri-hepatic region. It was suspected to be a case of bile
duct cancer. As a result, bile duct excision with proximal and
distal safe margins, as well as peri-hepatic lymphadenectomy, was
performed (14 lymph nodes were resected). A Roux-en-Y jejunostomy
was performed, and a drain was left in the subhepatic region. The
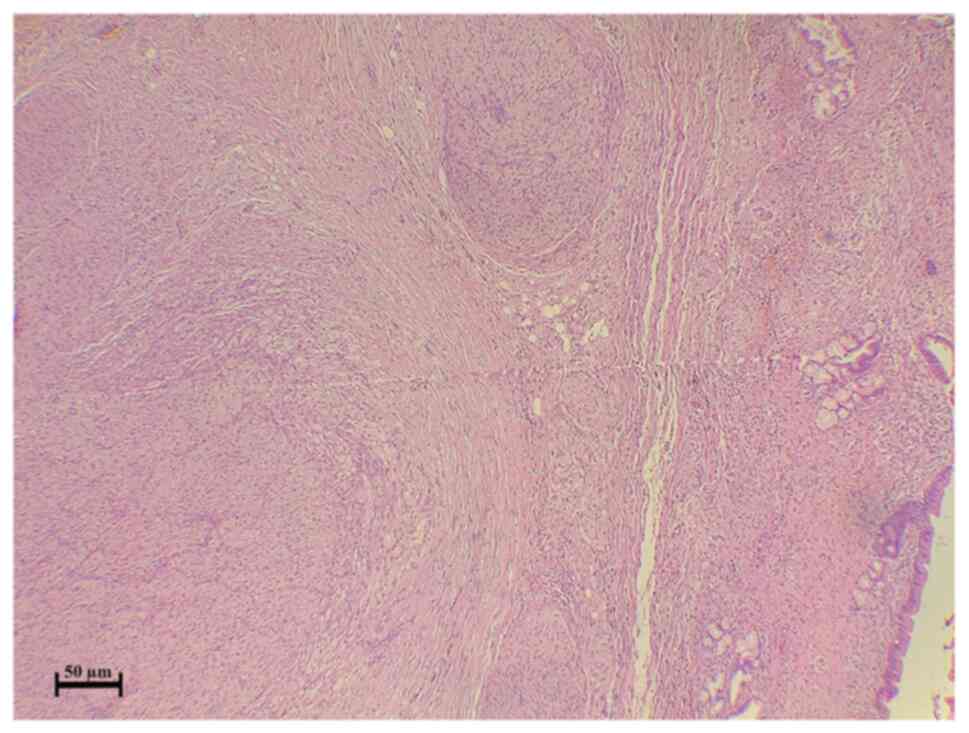
resected specimens were sent for histopathological analysis, which
revealed bile duct traumatic neuroma (Fig. 2). Immunostaining revealed diffuse and
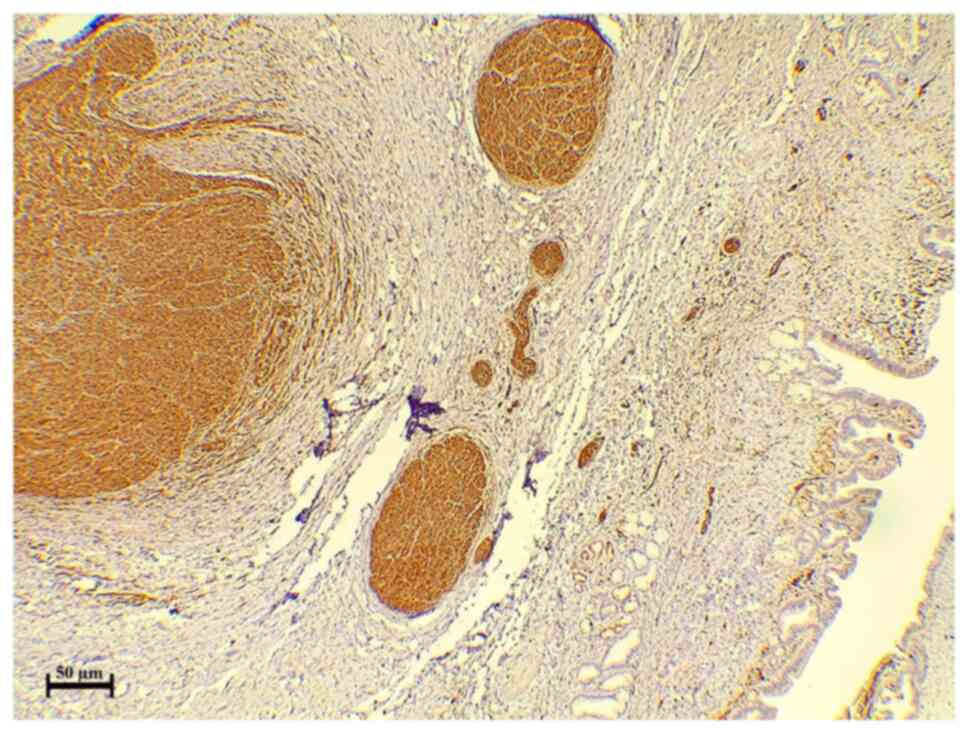
strong positivity in the disorganized nerve bundle (Fig. 3). SOX10 was used for the confirmation
of the neural nature of the lesion because SOX10 shows an increased
specificity for soft tissue tumors of neural crest origin (6).
Histopathological analysis and immunostaining were
performed as follows: For immunohistochemistry, the paraffin blocks
were cut into 4-6-µm-thick sections and transferred onto charged
glass slides. Subsequently, they were placed in an oven at 60˚C
overnight. Antigen retrieval was performed using the Dako PT Link
(Agilent Technologies, Inc.) by boiling the sections at 100˚C for 5
to 10 min. A solution of pH 6.0 or pH 9.0 was used regarding the
target antibody. The slides were then subjected to a 15-min wash
with a 20 ml buffer solution (0.05 mol/l Tris/HCl, 0.15 mol/L NaCl,
0.05% Tween-20, pH 7.6) at room temperature. To facilitate the
process, the slides were welled using the Dako Pen (Agilent
Technologies, Inc.). Furthermore, endogenous peroxidase was blocked
using 3% hydrogen peroxide. Subsequently, the primary antibodies
(SOX-10: EP268, ready-to-use, LOT no. 06560006, Master Diagnostica)
were applied at room temperature and left for 80 min. The secondary
antibody, which was horseradish peroxidase (PolyDetector Plus Link,
ready-to-use, LOT no. 0087XKD15, Bio SB PolyDetector Plus HRP,
ready-to-use, LOT no. 0088PKD15, Bio SB, Inc.), was then applied,
along with the chromogen (diaminobenzidine), both at room
temperature for 15 min. To achieve counterstaining, hematoxylin
Gill II (Leica Biosystems) was applied at room temperature for a
duration of 30 sec. The slides were dried and coverslips were
applied. A light microscope was used for the examination of the
slides (Leica Microsystems GmbH).
Follow-up
The post-operative recovery was smooth and
uneventful. The patient was discharged on the 4th post-operative
day in good health. Following 3 months of routine follow-up, the
patient's condition was stable.
Discussion
Adults can develop extra-hepatic bile duct
strictures as a result of malignant or benign biliary tumors
(4). Invasive adenocarcinoma is the
most frequent cause of primary biliary tumors, while benign tumors,
such as adenomas and papillomas are significantly less common,
accounting for only 6% of all biliary tumors (4,7). Biliary
tree traumatic neuroma is a rare benign, poorly defined,
non-encapsulated lesion with an unregulated proliferation of all
normal components of the nerve bundle, such as enlarged masses of
axons, endoneurial cells, Schwann cells, and perineural cells in a
rich collagenous matrix (8,9). The lesion is most typically observed
following a radical neck dissection, orthopedic surgery or limb
amputation (10,11). It is rarely reported following
parotidectomy, tooth extractions, cholecystectomy and liver
transplantation (12). The vast
majority of these lesions in the biliary tree arise in the cystic
duct remnants following cholecystectomy (13). The majority of the cases documented
in the literature occurred following open cholecystectomy. Only a
few cases have been recorded as a result of laparoscopic
cholecystectomy (3,4). The case described herein had a history
of laparoscopic cholecystectomy, which is in line with a previous
report by Nechi et al (3).
According to several case reports and reviews, the
interval between surgery and the diagnosis of biliary traumatic
neuroma ranged from 2 months to 46 years, with a mean age of 5
years (4). The interval in the
current case was only 4 weeks. Traumatic neuroma, often known as
amputation neuroma, is exceedingly rare in the gallbladder without
preceding surgery or cholelithiasis (7). Although the pathophysiology of biliary
traumatic neuroma has been shown to be associated with the process
of nerve regeneration, potential mechanisms encouraging its
production remain unclear. Calcineurin inhibitors may be one of the
elements involved in the development of biliary traumatic neuroma.
Calcineurin inhibitors, in general, aid in nerve repair by
promoting axon development. In addition, tacrolimus acts as a
neuroprotectant and neurotrophic drug, enhancing neurological
recovery following peripheral nerve and spinal cord injuries
(14).
These tumors are frequently identified by accident.
The tumor site may determine the patient's symptoms, which may
include upper abdominal pain with features of obstructive jaundice
in the common bile duct, hilar neuromas, or post-cholecystectomy
pain in cystic duct stump tumors (13,15). The
lesion is commonly misdiagnosed as cholangiocarcinoma, and a
comprehensive treatment strategy is undertaken (15). The case described herein presented
with upper abdominal pain, anorexia and yellow discoloration of the
sclera. This is in contrast to the study by Nechi et al
(3) that reported only jaundice as
the presentation of their case.
The most difficult aspect of the management of
biliary traumatic neuroma is the pre-operative diagnosis (13). Although various imaging techniques,
such as ultrasound (US), computed tomography (CT) and MRI, are
useful to some extent, the diagnosis of biliary traumatic neuroma
pre-operatively remains challenging (16,17).
Typically, these tumors are found following the compression of the
surrounding structures, leading to the misdiagnosis as hilar
cholangiocarcinoma (13). Previous
studies have observed that CT and MRI clearly demonstrate biliary
duct dilatation without proof of a tumor-like lesion (18,19).
MRCP can reveal bile duct stenosis (18). However, none of these modalities has
the sensitivity and specificity required for proper pre-surgical
identification of biliary traumatic neuroma, and none can
definitively rule out the existence of malignancy (13). In the case described in the present
study, MRCP revealed a dilated proximal bile duct and mild
dilatation of the intrahepatic biliary tree due to a stricture
distal to the confluence of the right and left hepatic ducts. A
dynamic liver MRI was performed to exclude tumors and revealed the
same finding of bile duct stricture due to a clip on the bile
duct.
Shimura et al (20) demonstrated that intraductal
ultrasonography can differentiate traumatic neuroma from other
malignancies by providing accurate details. Biopsies utilizing
cholangioscopy and endoscopic US were previously used to diagnose
two cases of traumatic neuroma pre-operatively (21,22).
Surgery is recommended to confirm the diagnosis and
treat biliary obstruction. This is performed by resecting the
extrahepatic biliary system that is closely attached to the
neuroma, followed by a Roux-en-Y hepaticojejunostomy (9). The clinical manifestations in the
patient described herein and the intraoperative results all
suggested malignancy. Therefore, an extensive surgical approach was
performed.
In conclusion, biliary tree traumatic neuroma is an
uncommon occurrence following laparoscopic cholecystectomy. The
rarity of this condition, combined with the absence of a
standardized diagnostic modality, renders its diagnosis difficult
and can even lead to its misdiagnosis as biliary cancer.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author upon reasonable
request.
Authors' contributions
HHKA was a major contributor to the conception of
the study, as well as to the literature search for related studies.
MNH, DSH, and FHK were involved in the literature review, in the
writing of the manuscript, as well as in the analysis and
interpretation of the patient's data. AMA was the pathologist
examining the specimen. DTG, DAI, DHA, BAA and OHGH, were involved
in the literature review, in the design of the study, in the
revision of the manuscript, and in the processing of the figures.
HHKA and DTG confirm the authenticity of all the raw data. All
authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient described in the present study for the inclusion of her
data.
Patient consent for publication
The patient provided verbal consent for publication
and for her data and any related images in the present study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lee JY: Traumatic neuroma at the
mastectomy site, unusual benign lesion, mimicking tumor recurrence:
A report of two cases. Radiol Case Rep. 17:662–666. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kang J, Yang P, Zang Q and He X: Traumatic
neuroma of the superficial peroneal nerve in a patient: A case
report and review of the literature. World J Surg Oncol.
14(242)2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Nechi S, Nakhli A, Ben Hamida W, Bani A,
Khsiba A, Ben Mohamed A, Chelbi E, Hamzaoui L and Touinsi H:
Traumatic neuroma of the bile duct: A case report. Clin Case Rep.
9(e04619)2021.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Lalchandani P, Korn A, Lu JG, French SW,
Hou L and Chen KT: Traumatic bile duct neuroma presenting with
acute cholangitis: A case report and review of literature. Ann
Hepatobiliary Pancreat Surg. 23:282–285. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sleiman YA, Hassoun ZA, Nasser HA, Abs L
and Allouch M: A late unusual complication after an open
cholecystectomy: Amputation neuroma of the CBD causing obstructive
jaundice. Int J Surg Case Rep. 39:123–125. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Karamchandani JR, Nielsen TO, van de Rijn
M and West RB: Sox10 and S100 in the diagnosis of soft-tissue
neoplasms. Appl Immunohistochem Mol Morphol. 20:445–450.
2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lin T, Bissessur AS, Zhu Y, Fukuyama T,
Ding G and Cao L: Case report: Idiopathic traumatic neuroma of the
gallbladder without previous surgery. Front Surg.
9(851205)2022.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Skovronsky DM and Oberholtzer JC:
Pathologic classification of peripheral nerve tumors. Neurosurg
Clin N Am. 15:157–166. 2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Iannelli A, Fabiani P, Karimdjee BS,
Converset S, Saint-Paul MC and Gugenheim J: Traumatic neuroma of
the cystic duct with biliary obstruction. Report of a case. Acta
Gastroenterol Belg. 66:28–29. 2003.PubMed/NCBI
|
|
10
|
Foltán R, Klíma K, Špačková J and Šedý J:
Mechanism of traumatic neuroma development. Med Hypotheses.
71:572–576. 2008.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kim NR, Cho HY, Chung DH, Kim KK, Cho JH
and Choi SJ: Post-transplant amputation traumatic neuroma of the
hilum and extrahepatic duct in a liver donor. J Pathol Transl Med.
52:191–194. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nazlan MA, Chik I, Fahmy F, Azman A and
Zuhdi Z: Traumatic neuroma of bile duct: A case report. HPB.
23(S384)2021.
|
|
13
|
Rahnemai-Azar AA, Ganjei-Azar P, Levi D
and Sleeman D: Non-traumatic biliary duct neuroma masquerading as a
Klatskin tumour. ANZ J Surg. 87:165–166. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Navez J, Golse N, Bancel B, Rode A, Ducerf
C, Mezoughi S, Mohkam K and Mabrut JY: Traumatic biliary neuroma
after orthotopic liver transplantation: A possible cause of
‘unexplained’ anastomotic biliary stricture. Clin Transplant.
30:1366–1369. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Rastogi A, Bihari C, Arora A, Sinha PK and
Chattopadhyay TK: Hilar traumatic neuroma masquerading as Klatskin
tumor. Trop Gastroenterol. 36:54–56. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Cheng Y, Jia Q, Xiong X and Cheng N:
Traumatic bile duct neuroma developing in a remnant choledochal
cyst. Dig Liver Dis. 46(e3)2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kaymakcioglu N, Gorgulu S, Zeybek N, Yagci
G, Demiriz M, Akdeniz A and Tufan T: Amputation neuroma of
extrahepatic bile duct as a rare cause of postcholecystectomy pain.
Eur Surg. 37:166–168. 2005.
|
|
18
|
Tsitouridis J, Kouklakis G, Xiarhos P and
Patakiuta F: Non traumatic neuroma of the bile duct: Report of a
case. Dig Endosc. 10:323–326. 1998.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Ueno Y, Ikeda K, Maehara M, Sakaida N,
Omura N, Kurokawa H and Sawada S: Traumatic neuroma of the bile
duct. Abdom Imaging. 33:560–562. 2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shimura K, Tamada K, Asada M, Watabiki N,
Wada I, Tanaka N and Suzuki Y: Intraductal ultrasonography of
traumatic neuroma of the bile duct. Abdom Imaging. 26:632–634.
2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Toyonaga H, Taniguchi Y, Inokuma T and
Imai Y: Traumatic bile duct neuroma diagnosed by boring biopsy with
cholangioscopy. Gastrointest Endosc. 87:1361–1362. 2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yasuda I, Kobayashi S, Nagata K, Takahashi
K and Entani T: Endoscopic images of amputation neuroma at the
cystic duct stump. Gastrointest Endosc. 90:986–987. 2019.PubMed/NCBI View Article : Google Scholar
|