Introduction
Inflammatory pseudotumor (IPT) represents a
heterogenous group of mass-forming lesions that involve various
organs and is characterized by prominent inflammatory infiltrates
(1). Several lesions previously
considered to be IPT have been presently identified as different
entities. The neoplastic variants of IPT include inflammatory
myofibroblastic tumor that is associated with anaplastic lymphoma
kinase (ALK) translocation and inflammatory pseudotumor-like
follicular dendritic cell tumors of the liver and spleen that are
related to clonal Epstein-Barr virus infection (1). These lesions turned out to be true
neoplasms. In addition, there exist infectious and
autoimmune-induced IPTs, such as mycobacterial spindle-cell IPT of
lymph nodes and immunoglobulin (Ig) G4-related tumefactive lesions,
respectively (1). The etiology and
pathogenesis remain unclear for a subset of IPTs that lack the
entities described above.
In the liver, IPT is occasionally encountered when a
biopsy or resection of mass lesions is performed due to clinical
concerns of primary or metastatic liver tumors. In a previous study
of resected focal lesions in 403 patients, the incidence of hepatic
IPT was reported to be 0.7% (2). The
majority of IPTs are presumed to be an exuberant response to
cholangitis or infection, although in most cases, the infectious
agent is unknown (3). Hepatic IPTs
are classified into two types in terms of tumor location, that is,
parenchymal IPT and biliary IPT (4).
The diagnosis of hepatic parenchymal IPT is often possible
following imaging-directed percutaneous core needle biopsy, while
this is not the case with hepatic biliary IPT. Obstructive
jaundice, biliary stricture in endoscopic retrograde
cholangiopancreatography (ERCP), and cellular atypia that is
frequently documented at preoperative biliary cytology may suggest
cholangiocarcinoma (CC), leading to unnecessary surgery.
The present study descries a rare case of hepatic
biliary IPT in a patient who had been misdiagnosed with CC
pre-operatively based on the repeated biliary cytology through
endoscopic nasobiliary drainage (ENBD) and then underwent left
lobectomy. In addition, issues encountered with the cytological
diagnosis of this case are discussed by comparing the present case
with 2 cases of well-differentiated CC. To the best of our
knowledge, this is the first report to validate the limitations of
biliary cytology of hepatic biliary IPT by a multi-reviewer
re-evaluation.
Case report
In 2025, a 76-year-old woman, who had been under
observation for diabetes and liver dysfunction, was referred to the
Department of Gastroenterology, Ota Memorial Hospital, Ota, Japan,
due to dilatation of the bile duct and portal vein (P3) obstruction
in the left lobe of the liver by contrast-enhanced computed
tomography (CECT). Laboratory tests at the first visit were
performed at the Department of Clinical Laboratory, Ota Memorial
Hospital, as routine testing, and the results were the following:
White blood cell count, 7,230/µl (reference range, 3,500-8,500/µl);
red blood cell count, 514x104/µl (reference range,
380-520x104/µl); hemoglobin, 15.0 g/dl (reference range,
11.5-15.5 g/dl); platelets, 23.6x104/µl (reference
range, 12.0-33.0x104/µl); total bilirubin, 0.74 mg/dl
(reference range, 0.4-1.5 mg/dl); aspartate aminotransferase, 32
U/l (reference range, 13-30 U/l); alanine aminotransferase, 27 U/l
(reference range, 7-23 U/l); alkaline phosphatase, 200 U/l
(reference range, 38-113 U/l); γ-glutamyl transferase, 215 U/l
(reference range, 9-32 U/l); HbA1c (NGSP), 6.3% (reference range,
4.6-6.2%); IgG, 1,228 mg/dl (reference range, 861-1,747 mg/dl);
carcinoembryonic antigen (CEA), 2.8 ng/ml (reference range, 0.0-5.0
ng/ml); CA19-9, <2.0 U/ml (reference range, 0.0-37.0 U/ml);
alpha-fetoprotein, 2.3 ng/ml (reference range, 0.0-10.0 ng/ml);
PIVKA-2, 20.47 mAU/ml (reference range, 0.0-39.0 mAU/ml); HBs-Ag
(-), HCV-Ab (-), anti-nuclear antibody (-) and anti-mitochondrial
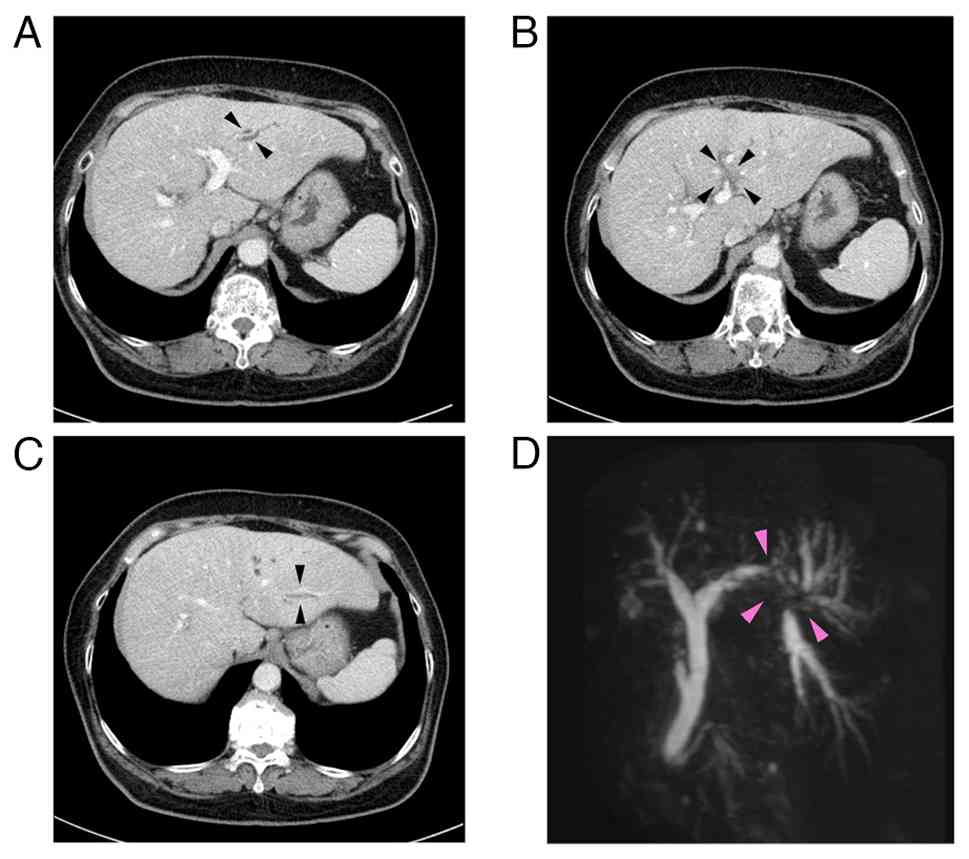
antibody (-). A CECT scan revealed an ~1 cm-sized low-density
lesion adjacent to the umbilical portion of the left portal vein
with peripheral bile duct dilatation in the lateral segment of the
liver (Fig. 1A-C). Magnetic
resonance cholangiopancreatography revealed the disruption of the
bile duct at the bifurcation of B2 and B3 bile ducts (arrows)
(Fig. 1D). The ERCP findings
demonstrated severe stenosis at the origin of the B3 bile duct,
which prevented fine needle aspiration. ENBD tube was placed in the
B2 bile duct and serial bile cytology was performed. Samples were
prepared with BD SurePathTM liquid-based cytology
(SP-LBC) (Becton, Dickinson and Company). In brief, bile juice was
centrifuged at 1,400 x g for 5 min at room temperature. The cell
pellet was fixed with CytorichTM Red (Becton Dickinson)
overnight and then washed. The cells were deposited onto BD
SurePathTM Precoated slides (Becton, Dickinson and
Company) according to the manufacturer's instructions. Following
emersion in 95% ethanol, the slides were subject to Papanicoloau
staining with Tissue-Tek Prima® Plus (Sakura Finetek)
according to the manufacturer's instructions. The stained glass
slides were inspected under a light microscope (BX53; Olympus
Corporation).
Cytologically abnormal findings, such as irregular
nuclear overlapping, irregular cluster margins, and irregular
nuclear arrangement were observed against an inflammatory
background primarily composed of numerous neutrophils (Fig. 2A and B). These finding met the criteria of
Cytology Guidelines 5, Digestive System, edited by the Japanese
Society of Clinical Cytology for adenocarcinoma (Table I) (5,6), and a
diagnosis of intrahepatic CC was made. A left hepatic lobectomy was
performed, and the cut surface of the resected liver revealed
multiple yellowish-white polypoid tumors, up to 8x5 mm in size,
which obstructed the B2 and B3 bile ducts (Fig. 3A and B).
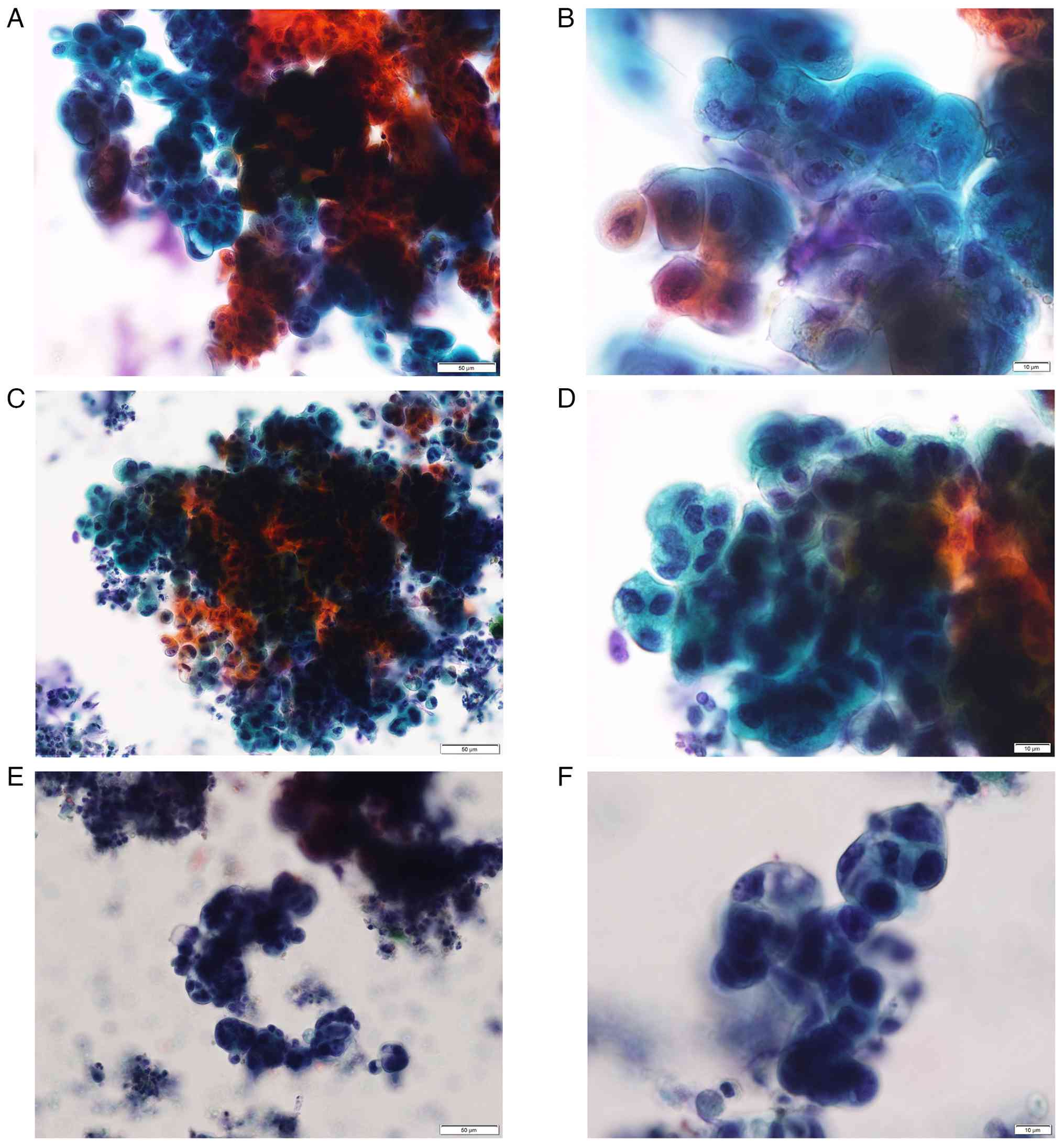 | Figure 2Cytomorphology of the present IPT case
and 2 cases of well-differentiated CC for comparison. (A) Bile
cytology from the present IPT case (case 1). Large clusters of
cells presenting irregularly overlapped nuclei, irregularly
arranged nuclei and irregular cluster margins, suggesting
malignancy based on the diagnostic criteria for bile cytology by
the Japanese Society of Clinical Cytology 2015 (Pap; magnification,
x20). (B) Bile cytology from the present IPT case (case 1).
Enlarged nuclei, irregular shaped nuclei and nucleoli swelling in
the small clusters were also compatible with malignancy (Pap;
magnification, x60). (C) Bile cytology from a 67-year-old male
patient with CC (case 2). Large clusters, demonstrating similar
findings with those of Case 1 (Pap; magnification, x20). (D) Bile
cytology from case 2. Small clusters, demontstrating similar
findings with those of case 1. Hyperchromasia may be more prominent
than case 1 (Pap; magnification, x60). (E) Bile cytology from a
70-year-old male patient with CC (case 3). Large clusters,
demonstrating similar findings with those of case 1 (Pap;
magnification, x20). (F) Bile cytology from case 3. Small clusters,
demonstrating similar findings with those of case 1. Hyperchromasia
may be more prominent than case 1 (Pap; magnification, x60). IPT,
inflammatory pseudotumor; CC, cholangiocarcinoma; Pap,
Papanicoloau. |
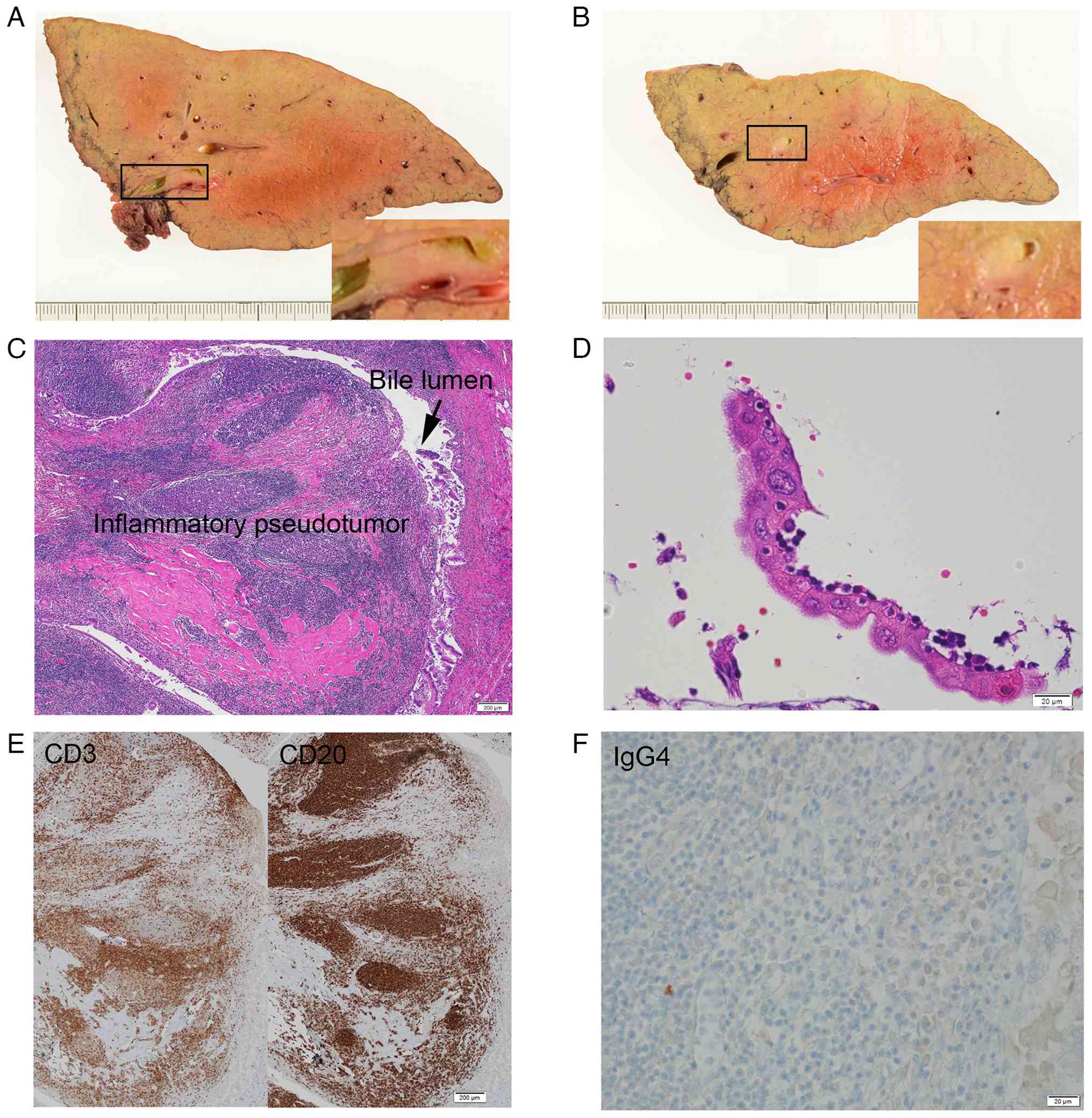 | Figure 3Pathological examination of the
resected liver. (A) Cut surface of the resected left lobe of the
liver demonstrated yellowish-white tumors adjacent to the B3 bile
duct, which caused stenosis of the bile duct. (B) Cut surface of
the resected left lobe of the liver demonstrated yellowish-white
tumors adjacent to the B2 bile duct, which caused stenosis of the
bile duct. (C) The tumor consisted of mature lymphoid tissue with
hyalinized collagen deposit, which was diagnosed as biliary IPT
(H&E; magnification, x4). (D) The overlying bile eithelium
revealed strong reactive atypia, such as enlarged nuclei,
anisokaryosis and prominent nucleoli (H&E; magnification, x40).
(E) This IPT consisted of both CD3-positive (left panel) and
CD20-positive (right panel) cells, suggesting a reactive lesion
(magnification, x4). (F) There were few IgG4-positive cells in this
IPT (magnification, x40). H&E, hematoxylin and eosin; IPT,
inflammatory pseudotumor. |
 | Table IDiagnostic criteria for bile cytology
by the JSCC 2015. |
Table I
Diagnostic criteria for bile cytology
by the JSCC 2015.
| A, Large
clusters |
|---|
|
1.
Irregularly overlapped nuclei |
|
2.
Irregularly arranged nuclei |
|
3. Irregular
cluster margins |
| B, Small clusters and
isolated cells |
|
1. Enlarged
nuclei |
|
2.
Irregularly shaped nuclei |
|
3. Abnormal
chromatin |
| C, Other notable
findings |
|
1. Necrotic
background |
|
2. Varying
cell cluster size |
| D, Point to note |
|
1. Do not
make a judgment from limited abnormalities |
|
2. Even if
morphological changes occur when bile juice is left as it is,
cytological diagnosis may be possible by observing the nuclear
structure |
|
3. Benign
tumors and normal tissue display cytological features of equal
internuclear distance and regularly arranged cytoplasm at cluster
margins |
The resected specimen was routinely processed for
pathological diagnosis at the Department of Pathology, Ota Memorial
Hospital. In brief, the specimen was fixed with 10% neutral
buffered formalin for 48 h at room temperature and then embedded in
paraffin. The subsequent 3 µm-thick sections were stained with
hematoxylin and eosin (H&E) with Tissue-Tek Prima®
Plus (Sakura Finetek) for 55 min at room temperature according to
the manufacturer's instructions. The immunohistochemical staining
of CD3 (clone 2GV6; cat. no. 518110079; Roche Diagnostics), CD20
(clone L26; cat. no. 518110086; Roche Diagnostics) and IgG4 (clone
MRQ-44; cat. no. 06523854001; Roche Diagnostics) was outsourced to
SRL Central Laboratory. The stained glass slides were inspected
under a light microscope (BX53; Olympus Corporation).
Histologically, the polypoid tumor protruded into
the bile lumen (Fig. 3C). With the
background of micronodular liver cirrhosis, dense lymphocyte and
plasma cell infiltration were observed under the bile duct
epithelium, accompanied by the formation of multiple lymphoid
follicles and the dense collagen deposit with hyalinization
(Fig. 3C). Eosinophilic infiltration
was not remarkable. Although the portal vein was compressed by the
lymphoid tissue, no obliterative phlebitis was observed. Portions
of the biliary epithelium exhibited severe atypia, such as nuclear
enlargement, anisokaryosis and swollen nucleoli (Fig. 3D), but no interstitial infiltration
was observed. The lymphoid tissue comprised similar amounts of
CD3-positive T cells and CD20-positive B cells, suggesting reactive
lesion (Fig. 3E). The histological
final diagnosis was lymphoplasmacytic pseudotumor. Immunostaining
revealed few IgG4-positive cells, and the lesion did not meet the
criteria for IgG4-related disease (Fig.
3F) (7).
Reflecting on the cytological diagnosis before
surgery, three board-certified cytotechnologists (YH, TK and TT)
and two board-certified cytopathologists (SS and YI) at Ota
Memorial Hospital and five board-certified cytotechnologists (TF,
MU, TY, TI and AS) from neighboring institutions blindly
re-evaluated the ENBD cytology specimens prepared with SP-LBC from
this case (case 1) (Fig. 2A and
B) and two cases of
well-differentiated CC at the hepatic hilus (cases 2 and 3)
(Fig. 2C, D, E and
F), which had been histologically
confirmed following surgical resection at Ota Memorial Hospital in
2024. Well-differentiated CC was selected for comparison, as poorly
differentiated CC is typically not a diagnostic challenge, but
well-differentiated CC is difficult to distinguish from benign
and/or reactive changes cytologically. Glass slides of the three
cases were re-evaluated simultaneously. Although the rate of
diagnosis as malignant was somewhat lower in case 1 than those in
cases 2 and 3, five investigators (50%) evaluated case 1 as
malignant (Table II). There were no
cases in which all observers agreed on the diagnosis.
 | Table IIA multi-reviewer re-evaluation of
cytomorphology of the present IPT case and 2 cases of
well-differentiated CC. |
Table II
A multi-reviewer re-evaluation of
cytomorphology of the present IPT case and 2 cases of
well-differentiated CC.
| | Tumor markers | Cytological
diagnosis |
|---|
| Case no. (age,
years/sex) | CEA (ng/ml;
reference range, 0.0-5.0) | CA19-9 (U/ml;
reference range, 0.0-37.0 | Benign | Atypia | Suspicious for
malignancy | Malignancy |
|---|
| Case 1 (76/F) | 2.8 | <2.0 | 0 | 5 | 0 | 5 |
| Case 2 (67/M) | 5.4 | 73.8 | 0 | 1 | 1 | 8 |
| Case 3 (70/M) | 1.9 | 144.1 | 1 | 2 | 1 | 6 |
The authors then presented this IPT case in a slide
conference at the 39th Annual Meeting of the Kanto Society of
Clinical Cytology held in Shizuoka prefecture, Japan in
2025(8). Virtual slides of this case
were posted on the society's website 1 month prior to the
conference, and board-certified cytotechnologists affiliated with
the society voted on several possible diagnoses. Of the 42
respondents, 61.9% selected adenocarcinoma and 23.8% selected other
types of cancer, such as adenosquamous carcinoma and hepatocellular
carcinoma, while only 14.3% selected regenerative atypia.
Discussion
IPT is a term used to describe a benign and rare
process found in almost every site of the body. Histologically, an
IPT contains cells associated with both acute and chronic
inflammation, including lymphocytes, plasma cells, myofibroblastic
cells and a fibrous reaction. The cause of IPT is unknown, although
it is hypothesized that the causes may be trauma, surgical
inflammation, autoimmune condition and low-grade mesenchymal tumor.
Some IPTs have been associated with IgG4-related sclerosing
disease, such as autoimmune pancreatitis, sclerosing cholangitis,
sialoadenitis, retroperitoneal fibrosis, tubulointerstitial
nephritis, interstitial pneumonia, prostatitis and lymphadenopathy
(9).
Hepatic IPT was first reported by Pack and Baker
(10) in 1953. Although there are
several methods for classifying hepatic IPT, such as etiology and
tumor location, some authors proposed histological classification.
In 1978, Someren (11) classified
hepatic IPT into three distinct types: Hyalinized sclerosing,
plasma cell granuloma and xanthogranuloma. Later, with the
recognition of IgG4-related disease, Zen et al (7) classified hepatic IPT (16 cases) into
two types, namely fibrohistiocytic (10 cases) and lymphoplasmacytic
(6 cases) variants. They reported that lymphoplasmacytic variants
could belong to the IgG4-related diseases, while all 16 cases
negatively stained with ALK by immunohistochemistry (7). In the present case report, abundant
lymphoplasmacytic inflammation illustrating periductal infiltration
was observed, but neither xanthogranulomatous inflammation nor
multinucleated giant cells was observed, favoring lymphoplasmacytic
type. On the other hand, eosinophilic infiltration or IgG4-positive
plasma cell infiltration was not remarkable throughout the lesion,
while remarkable deposit of hyalinized collagen was observed. These
findings are inconsistent with pure lymphoplasmacytic type. More
refined histological classification of the hepatic ITP awaits the
further accumulation of cases.
The authors could not make a correct diagnosis of
this hepatic biliary IPT prior to surgery, and the patient
underwent surgery. The imaging analyses strongly suggested
intrahepatic CC, and cytodiagnosis of the repeated ENBD bile
cytology was malignancy by 5/10 of the
cytopathologists/cytotechnologists in our and related institutions.
The difficulty in this cytological diagnosis was further verified
later by a large number of reviewers (n=42) at the regional slide
conference (8). Of note, a core
needle biopsy was not performed and the tumor markers for
adenocarcinoma were within normal ranges.
Since its first report in 1953(10), the pre-operative diagnosis of hepatic
IPT remains very difficult, and surgery is currently performed when
malignancy cannot be completely ruled out. A histological
confirmation of a biopsy is thus critical for avoiding unnecessary
surgery. Some reports have shown the efficacy of the percutaneous
core needle biopsy for the diagnosis of hepatic IPT (12); however, Okamoto et al
(13) reported that hepatic IPT was
accurately diagnosed in only 12/23 cases (52.1%) by percutaneous
needle biopsy. In addition, ‘seeding’ of the needle tract has been
a concern in patients who are surgical candidates (14). Accordingly, cytological techniques
have become the initial diagnostic modality when malignant biliary
stricture is suspected. Bile aspiration and brush cytology via ERCP
drainage tube and repeated ENBD bile cytology are preferably
performed. ENBD is simple, repeatable and suitable for
comprehensive screening, but diagnosis is often difficult due to
low cell recovery rates and the effects of bile acid-induced
cytopathies (15). False-positive
results for regenerative atypia and false-negative results for low
cell recovery rate in well-differentiated CC should also be noted
(16). The sensitivity of bile
cytology for malignant stricture has been reported to be 30%
(28/93) for ERCP aspiration bile cytology, 48% (62/130) for brush
cytology and 24% (19/79) for ENBD bile aspiration cytology
(17). Low sensitivity of ENBD bile
cytology may be overcome by repeated aspiration cytology (1 to 14
times), which increases the diagnostic yield up to 72.3% (34/47),
and 32 positive results have been obtained at the maximum of six
samplings (18). As far as is known,
there is no report of false-positive rates of ENBD bile cytology to
date. Moreover, false-negative and false-positive rates in common
bile duct brushing cytology during ERCP have been reported to be
13.3% (22/166) and 4.2% (7/166), respectively (19). False-positive results of ERCP brush
cytology occur most often in patients with primary sclerosing
cholangitis, IgG4-related cholangitis and autoimmune pancreatitis
(19,20). Marked periductal inflammation,
fibrosis and epithelial degenerative changes can be the cause of
atypical cells mimicking malignancy. Taken together, the definitive
diagnosis of hepatic biliary IPT may be intrinsically difficult
only by ENBD biliary cytology.
Several studies have attempted to identify definite
cytologic criteria that can predict malignancy in bile juice. In
1995, Cohen et al (21) first
reported the cytologic criteria for biliary malignancy. Their
analysis revealed that nuclear molding, chromatin clumping and an
increased nuclear/cytoplasmic (N/C) ratio, and less importantly,
anisonucleosis, nuclear irregularity and nuclear enlargement were
frequently associated with malignancy (primary Iowa criteria)
(21). In 1998, Renshaw et al
(22) similarly reported that
nuclear molding, chromatin clumping and loss of polarity were
associated with malignancy (Boston criteria). In 2002, Henke et
al (23) successfully applied
the Iowa criteria to liquid-based specimens. In 2017, Avadhani
et al (24) pointed to 11
characteristics significantly associated with malignancy, including
3-dimension clusters, pleomorphism, 2-cell population, hypo/hyper
chromasia, a high N/C ratio, cytoplasmic vacuoles, nuclear
irregularity, cellular discohesion, hypercellularity, nuclear
molding and prominent nucleoli. They concluded that the
identification of 3/11 characteristics markedly improves diagnostic
performance. In Japan, the criteria proposed by Hirooka et
al (5) have been used for
cytological diagnosis of pooled bile juice (5,6). The
rate of accurate diagnosis by these criteria was at most only 61%
(5), although it is assumed to be
superior to individual criteria of respective cytotechnologists by
~20% (25). These criteria are also
used at Ota Memorial Hospital, and the case presented herein was
diagnosed as malignant according to the three factors met for A
(large clusters) (Table I). It is
now deemed that further attention should have been paid to the
abnormalities of individual cells in small clusters, such as an
increased N/C ratio and nuclear hyperchromasia. However, it was
assumed that enlarged nuclei and irregular shaped nuclei (B.1 and 2
in Table I) and enlarged nucleoli
may not be useful, as these findings were observed in this IPT case
(Fig. 3D).
The final key for differentiation between IPT and CC
may be tumor markers. In this case, CA19-9 and CEA levels were both
within normal ranges. If these data had been seriously considered
and percutaneous core needle biopsy was performed, unnecessary
surgery may have been avoided. Of note, CA19-9 levels may be
elevated in benign diseases in pancreaticobiliary stricture
lesions. In a previous study, out of the 17 cases of hepatic IPT,
CA19-9 was elevated beyond its normal range (<37 U/ml), as high
as 518.8 U/ml in 4 cases (23.5%) (26). The optimal cut-off value for CA19-9
varies depending on the study. Kuzu et al (27) stated that CA19-9 at the cut-off value
(72.5 U/ml) was an effective predictive factor in diagnosing tumors
in the pancreaticobiliary region. On the other hand, CA19-9 is not
produced in patients belonging to the Lewis blood group
(α-β-), which occurs in 5-10% of the
population that lacks the enzyme 1,4-fucosyltransferase required
for antigen epitope production (28). In such cases, DU-PAN-2 may be an
alternative to CA19-9 testing (29).
As regards CEA, Juntermanns et al (30) reported that only 55 (40%) out of the
136 patients with hilar CC exhibited elevated CEA levels prior to
surgery. Thus, normal tumor marker levels do not necessarily
exclude malignancy, but it may be a reason to consider needle
biopsy.
In conclusion, a case of biliary IPT of the liver
was presented herein. Although tumor markers had been within normal
ranges and core needle biopsy had not been performed, this case was
diagnosed as CC and underwent left hepatic lobectomy based on
imaging analyses and ENBD bile cytology. As ENBD bile cytology may
play a limited role in differentiation between benign and malignant
bile strictures, surgical indication should be carefully determined
in patients with normal tumor marker levels.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study are included
in the figures and/or tables of this article.
Authors' contributions
YI conceptualized this study. YH, TK and YI made a
cytological diagnosis before surgery. TT, SS, TF, MU, TY, TI and AS
contributed to the re-evaluation of the cytological diagnosis. YI
made a histological diagnosis of the surgically resected specimen.
EK and YO contributed to the endoscopic procedures. SM, KH and NT
performed the surgery. YH and YI wrote the manuscript. MO and FE
prepared the pathological specimens and critically revised the
manuscript. All authors have read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Ota Memorial Hospital, Ota, Japan (Approval no.
OR25014). A written informed consent was obtained from the patient
with biliary IPT for participation in the present case report,
while the patients with CC were not required to give informed
consent to the study as the analysis used anonymous cytological
images that were obtained after each patient agreed to treatment by
written consent.
Patient consent for publication
A written informed consent was obtained from the
patient with biliary IPT for the publication of the present case
report and any accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Balabaud C, Bioulac-Sage P, Goodman ZD and
Makhlouf HR: Inflammatory pseudotumor of the liver: A rare but
distinct tumor-like lesion. Gastroenterol Hepatol (NY). 8:633–634.
2012.PubMed/NCBI
|
|
2
|
Torzilli G, Inoue K, Midorikawa Y, Hui AM,
Takayama T and Makuuchi M: Inflammatory pseudotumors of the liver:
Prevalence and clinical impact in surgical patients.
Hepatogastroenterology. 48:1118–1123. 2001.PubMed/NCBI
|
|
3
|
Wang D and Misdraji J: Inflammatory
pseudotumor of the liver. Surg Pathol Clin. 16:565–580.
2023.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Tublin ME, Moser AJ, Marsh JW and Gamblin
TC: Biliary inflammatory pseudotumor: Imaging features in seven
patients. AJR Am J Roentgenol. 188:W44–W48. 2007.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hirooka Y, Nakaizumi A, Oka T, Naito Y,
Arisaka Y, Minamiguchi S, Haba R, Takenaka A, Furuhata A and Masuda
D: Report of the clinical study for methods to improve the
diagnostic accuracy of bile cytology (1) - Diagnostic bile cytology
criteria. J Jpn Soc Clin Cytol. 49:7–14. 2010.(In Japanese).
|
|
6
|
Hirooka Y, Oka T, Okada S, Katayama H,
Kato T, Kijima H, Sakatani T, Takenaka A, Naito Y, Naito Y, et al:
Hepatobiliary System. In: Cytology Guidelines 5, Digestive System.
1st edition. Japanese Society of Clinical Cytology (ed.) Kanehara,
Tokyo, p220, 2015.
|
|
7
|
Zen Y, Fujii T, Sato Y, Masuda S and
Nakanuma Y: Pathological classification of hepatic inflammatory
pseudotumor with respect to IgG4-related disease. Mod Pathol.
20:884–894. 2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Higuchi Y: Case 3 in Slide Conference. In:
Proceedings of the 39th Annual Meeting of the Kanto Society of
Clinical Cytology. p15, 2025 (In Japanese). https://plaza.umin.ac.jp/~jscck39/wp-content/uploads/20250920_jscck39_program.pdf.
|
|
9
|
Patnana M, Sevrukov AB, Elsayes KM,
Viswanathan C, Lubner M and Menias CO: Inflammatory pseudotumor:
The great mimicker. AJR Am J Roentgenol. 198:W217–W227.
2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Pack GT and Baker HW: Total right hepatic
lobectomy report of a case. Ann Surg. 138:253–258. 1953.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Someren A: ‘Inflammatory pseudotumor’ of
liver with occlusive phlebitis: Report of a case in a child and
review of the literature. Am J Clin Pathol. 69:176–181.
1978.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nakama T, Hayashi K, Komada N, Ochiai T,
Hori T, Shioiri S and Tsubouchi H: Inflammatory pseudotumor of the
liver diagnosed by needle liver biopsy under ultrasonographic
tomography guidance. J Gastroenterol. 35:641–645. 2000.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Okamoto K, Yoshimi F, Shida D, Oka D,
Hagihara T, Shioyama Y, Shimazaki J and Itabashi M: A case of
inflammatory pseudotumor of the liver which presented difficulty in
differential diagnosis cholangiocellular carcinoma. Jpn J
Gastroenterol Surg. 33:1900–1904. 2000.(In Japanese).
|
|
14
|
Mahmoudi N, Enns R, Amar J, AlAli J, Lam E
and Telford J: Biliary brush cytology: Factors associated with
positive yields on biliary brush cytology. World J Gastroenterol.
14:569–573. 2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kinjo Y, Naito Y, Nagayama D, Tsukamoto T,
Umeda A, Takei M, Fukagawa Y, Araki Y, Kimura Y and Higaki K: Key
points of cytogram in bile cytology. J Kyushu-Okinawa Soc Clin
Cytol. 56:15–20. 2025.(In Japanese). https://koscc.jp/magazine/pdf/vol56.pdf.
|
|
16
|
Giovannini D, Bailly A, Seigneurin A,
Fior-Gozlan M, Eyraud PY, Roth G, Laverrière MH, McLeer A and Sturm
N: Biliary cytology: A diagnostic tree for adenocarcinoma based on
a cohort of 135 patients with endoscopic retrograde
cholangiopancreatography for stenosis of the extrahepatic bile
duct. Cancer Cytopathol. 130:433–442. 2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yagioka H, Hirano K, Isayama H, Tsujino T,
Sasahira N, Nagano R, Hamada T, Miyabayashi K, Ito Y, Mohri D, et
al: Clinical significance of bile cytology via an endoscopic
nasobiliary drainage tube for pathological diagnosis of malignant
biliary strictures. J Hepatobiliary Pancreat Sci. 18:211–215.
2011.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Abdelghani YA, Arisaka Y, Masuda D, Takii
M, Ashida R, Makhlouf MM, Fouad YM, Tsuji M, Kurisu Y and Higuchi
K: Bile aspiration cytology in diagnosis of bile duct carcinoma:
Factors associated with positive yields. J Hepatobiliary Pancreat
Sci. 19:370–378. 2012.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Geramizadeh B, Moughali M, Shahim-Aein A,
Memari S, Ghetmiri Z, Taghavi A and Bagheri Lankarani K: False
negative and false positive rates in common bile duct brushing
cytology, a single center experience. Gastroenterol Hepatol Bed
Bench. 11:296–300. 2018.PubMed/NCBI
|
|
20
|
Ding SM, Lu AL, Xu BQ, Shi SH, Edoo MIA,
Zheng SS and Li QY: Accuracy of brush cytology in biliopancreatic
strictures: A single-center cohort study. J Int Med Res.
49(300060520987771)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cohen MB, Wittchow RJ, Johlin FC, Bottles
K and Raab SS: Brush cytology of the extrahepatic biliary tract:
Comparison of cytologic features of adenocarcinoma and benign
biliary strictures. Mod Pathol. 8:498–502. 1995.PubMed/NCBI
|
|
22
|
Renshaw AA, Madge R, Jiroutek M and
Granter SR: Bile duct brushing cytology: Statistical analysis of
proposed diagnostic criteria. Am J Clin Pathol. 110:635–640.
1998.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Henke AC, Jensen CS and Cohen MB:
Cytologic diagnosis of adenocarcinoma in biliary and pancreatic
duct brushings. Adv Anat Pathol. 9:301–308. 2002.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Avadhani V, Hacihasanoglu E, Memis B,
Pehlivanoglu B, Hanley KZ, Krishnamurti U, Krasinskas AM, Osunkoya
AO, Daniels LM, Freedman AA, et al: Cytologic predictors of
malignancy in bile duct brushings: A multi-reviewer analysis of 60
cases. Mod Pathol. 30:1273–1286. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Furuhata A, Hirooka Y, Toui Y, Abe K, Abe
Y and Gonda H: Application of diagnostic bile cytology criteria for
cytotechnologists. J Jpn Soc Clin Cytol. 54:292–298. 2015.(In
Japanese).
|
|
26
|
Nigam N, Rajani SS, Rastogi A, Patil A,
Agrawal N, Sureka B, Arora A and Bihari C: Inflammatory
pseudotumors of the liver: Importance of a multimodal approach with
the insistance of needle biopsy. J Lab Physicians. 11:361–368.
2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kuzu UB, Ödemiş B, Turhan N, Parlak E,
Dişibeyaz S, Suna N, Öztaş E, Akpınar MY, Aksoy A, Torun S, et al:
The diagnostic value of brush cytology alone and in combination
with tumor markers in pancreaticobiliary strictures. Gastroenterol
Res Pract. 2015(580254)2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Ballehaninna UK and Chamberlain RS: The
clinical utility of serum CA 19-9 in the diagnosis, prognosis and
management of pancreatic adenocarcinoma: An evidence based
appraisal. J Gastrointest Oncol. 3:105–119. 2012.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Narimatsu H, Iwasaki H, Nakayama F,
Ikehara Y, Kudo T, Nishihara S, Sugano K, Okura H, Fujita S and
Hirohashi S: Lewis and secretor gene dosages affect CA19-9 and
DU-PAN-2 serum levels in normal individuals and colorectal cancer
patients. Cancer Res. 58:512–518. 1998.PubMed/NCBI
|
|
30
|
Juntermanns B, Radunz S, Heuer M, Hertel
S, Reis H, Neuhaus JP, Vernadakis S, Trarbach T, Paul A and Kaiser
GM: Tumor markers as a diagnostic key for hilar cholangiocarcinoma.
Eur J Med Res. 15:357–361. 2010.PubMed/NCBI View Article : Google Scholar
|