1. Introduction
Cancer remains a leading cause of morbidity and
mortality worldwide, accounting for almost ten million deaths
annually and imposing a growing burden on healthcare systems.
Modern oncology requires the integration of complex diagnostic,
prognostic and therapeutic information derived from medical
imaging, histopathology, molecular profiling and longitudinal
clinical data. Conventional workflows rely heavily on human
interpretation, which is inherently subjective and prone to inter-
and intra-observer variability, cognitive bias and information
overload. These limitations contribute to variability in diagnosis,
risk stratification and treatment selection.
Artificial intelligence (AI), defined as
computational systems capable of learning, reasoning and pattern
recognition, provides a paradigm shift by enabling automated,
objective and data-driven clinical decision support (1). Advances in machine learning (ML) and
deep learning (DL), particularly convolutional neural networks
(CNNs), have driven major breakthroughs in medical image analysis
and predictive modelling (2,3). Unlike early rule-based systems,
contemporary AI models can extract high-dimensional features from
complex data and model nonlinear relationships that are difficult
to capture using traditional statistical approaches.
Multiple systematic reviews and meta-analyses have
demonstrated that AI systems can achieve diagnostic performance
comparable to, and in some contexts exceeding, that of healthcare
professionals in selected oncologic tasks (4-7).
Despite this promise, widespread clinical adoption remains limited.
Barriers include dataset bias, limited external generalizability,
the lack of standardized data acquisition and annotation protocols,
insufficient interpretability of model outputs and complex
regulatory pathways (8). In
addition, concerns regarding data privacy, accountability and
equity have become increasingly prominent (9).
The present review aimed to provide a critical,
clinically oriented synthesis of AI applications across the
oncology continuum. Rather than providing a purely descriptive
overview, the present review aimed to discuss the methodological
strengths and limitations, unmet clinical needs, and emerging
technological paradigms, with the aim of providing a practical
roadmap for responsible and equitable integration of AI into
oncology practice.
2. AI in cancer imaging
Medical imaging represents the most mature and
clinically advanced domain of AI applications in oncology. AI has
been applied across the entire imaging pipeline, including
acquisition, reconstruction, segmentation, detection,
classification and prognostication, spanning modalities such as
computed tomography (CT), magnetic resonance imaging (MRI),
positron emission tomography, mammography and ultrasound.
DL-based reconstruction algorithms have enabled
substantial improvements in image quality by reducing noise and
artefacts, facilitating lower radiation doses and shorter
acquisition times without compromising diagnostic accuracy. These
advances are particularly relevant in CT and MRI, where dose
reduction and accelerated imaging have direct implications for
patient safety and workflow efficiency. AI-driven image
reconstruction has also enabled improved visualization of subtle
lesions, potentially enhancing early cancer detection (10).
CNN-based computer-aided diagnosis (CADx) systems
analyse lesion morphology, texture, and spatial context to support
malignancy detection, staging and risk stratification (11). Robust performance has been
demonstrated in melanoma classification (12), real-time colorectal polyp detection
during colonoscopy (13), the
identification of nodal metastases in head and neck cancer
(14) and breast cancer risk
prediction using mammography (15).
Several of these systems have progressed to regulatory approval,
reflecting the increasing acceptance of AI-enabled imaging tools by
agencies, such as the US Food and Drug Administration (16).
Beyond detection, AI-based imaging facilitates
tumour phenotyping and prognostication by integrating information
across multiple modalities (Table
I). Radiomic and DL features extracted from imaging data have
been associated with molecular subtypes, treatment response, and
survival outcomes (17). However, a
number of published studies remain retrospective and single-centre,
with limited demographic and technical diversity, increasing the
risk of overfitting and dataset bias (18-20).
Variability in scanner hardware, acquisition protocols and
reconstruction parameters further constrains generalizability
(Table II) (21).
 | Table IAI in clinical fields: Applications
and limitations. |
Table I
AI in clinical fields: Applications
and limitations.
| Clinical field | Key AI
applications | Representative
outcomes | Major
limitations |
|---|
| Cancer imaging (CT,
MRI, PET, mammography, USG) | Lesion detection,
segmentation, staging, radiomics-based prognostication, image
reconstruction | Improved detection
of melanoma, breast cancer risk prediction, colorectal polyp
identification, nodal metastasis classification | Retrospective
single-centre datasets, scanner/protocol variability, limited
interpretability, dataset bias |
| Digital
pathology | Tumour detection
and grading, lymph node metastasis identification, genomic mutation
inference from H&E slides, survival prediction | Gleason grading,
gastric/colonic tumour classification, prediction of actionable
mutations | Staining and
scanner heterogeneity, lack of standardization, insufficient
prospective validation |
| Clinical outcome
prediction | Survival
prediction, toxicity risk estimation, response assessment | Enhanced risk
stratification beyond conventional staging | Overfitting, poor
external generalizability, limited longitudinal modelling |
| Chemotherapy | Drug response
prediction, pharmacogenomic modelling, resistance detection | Improved in
silico prediction of sensitivity using multi-omics data | Scarcity of
high-quality clinical datasets, limited explainability, lack of
prospective trials |
| Radiotherapy | Automated
contouring, dose calculation, adaptive planning, toxicity
prediction | More rapid
treatment planning and adaptive workflows | Regulatory hurdles,
integration challenges, black-box models |
 | Table IIAI methodologies in oncology:
Applications and challenges. |
Table II
AI methodologies in oncology:
Applications and challenges.
| Methodology | Typical
applications | Strengths | Challenges |
|---|
| Machine learning
(ML) | Survival
prediction, drug response modelling, radiomics | Handles structured
clinical and molecular data | Feature engineering
dependence, limited scalability |
| Deep learning
(CNNs, transformers) | Imaging analysis,
digital pathology, outcome prediction | Automatic feature
extraction, high diagnostic accuracy | Poor
interpretability, large data requirements |
| Radiomics | Tumour phenotyping,
prognostication | Quantifies imaging
heterogeneity | Sensitive to
acquisition variability |
| Multimodal AI | Integration of
imaging, pathology, genomics, EHRs | Captures tumour
biology and patient context comprehensively | Complex model
design, data harmonization |
| Foundation
models | Transfer learning
across cancers and institutions | Reduced labelling
needs, improved robustness | Computationally
intensive, transparency concerns |
| Federated
learning | Multi-centre model
training without data sharing | Preserves privacy,
improves generalizability | Infrastructure
demands, communication overhead |
| Explainable AI
(XAI) | Clinical decision
support transparency | Builds clinician
trust | Often adds
complexity, limited standardization |
Another persistent challenge is interpretability.
The black-box nature of DL models complicates the understanding of
decision pathways and limits clinician trust. Explainable AI
techniques, including saliency mapping and concept-based models,
are increasingly explored to address this limitation (22). Federated learning offers a promising
strategy for multi-institutional model development while preserving
data privacy (Table II) (23). Future research is required to
prioritize prospective, multi-centre validation studies,
standardized benchmarking datasets and transparent reporting to
enable meaningful clinical translation.
3. AI in digital pathology
The digitization of histopathology through
whole-slide imaging has catalysed rapid growth in AI-driven
computational pathology (Table I).
Digital slides enable large-scale analysis using DL architectures
such as CNNs, fully convolutional networks, and, more recently,
vision transformers. These models have demonstrated high accuracy
in tumour detection, grading, and prognostication across multiple
cancer types (Table II) (24).
Weakly supervised learning approaches have been
particularly impactful, enabling model training using slide-level
labels rather than exhaustive pixel-level annotations.
Clinical-grade performance has been reported for tasks, such as
Gleason grading in prostate cancer and lymph node metastasis
detection (25). Beyond
morphological assessment, AI models can infer genomic alterations
and clinically actionable mutations directly from routine
haematoxylin and eosin-stained slides, including in lung and
gastrointestinal cancers (26,27).
Several studies have demonstrated that AI-derived
histopathological features are associated with survival outcomes
and therapeutic response, particularly when integrated with
molecular and clinical data. These findings suggest that AI-enabled
pathology may serve not only as a diagnostic adjunct, but also as a
prognostic and predictive tool. In resource-limited settings, the
ability to infer molecular information from routine histology has
potential implications for cost-effective precision oncology
(26).
Despite these advances, digital pathology faces
substantial challenges. Variability in staining protocols, scanner
technologies and laboratory workflows limits reproducibility across
institutions. Dataset bias and relatively small sample sizes
further restrict external validity (28). Regulatory approval is complicated by
limited interpretability and the paucity of prospective validation
studies.
Future efforts should focus on standardized
pre-processing pipelines, harmonized data annotation practices and
pathology-specific reporting guidelines, including extensions of
frameworks, such as TRIPOD (29).
Explainable AI methods that highlight diagnostically relevant
histological features may improve transparency and pathologist
confidence. Federated learning frameworks can facilitate
multi-centre collaboration while preserving patient privacy, and
multimodal integration with radiologic and molecular data may
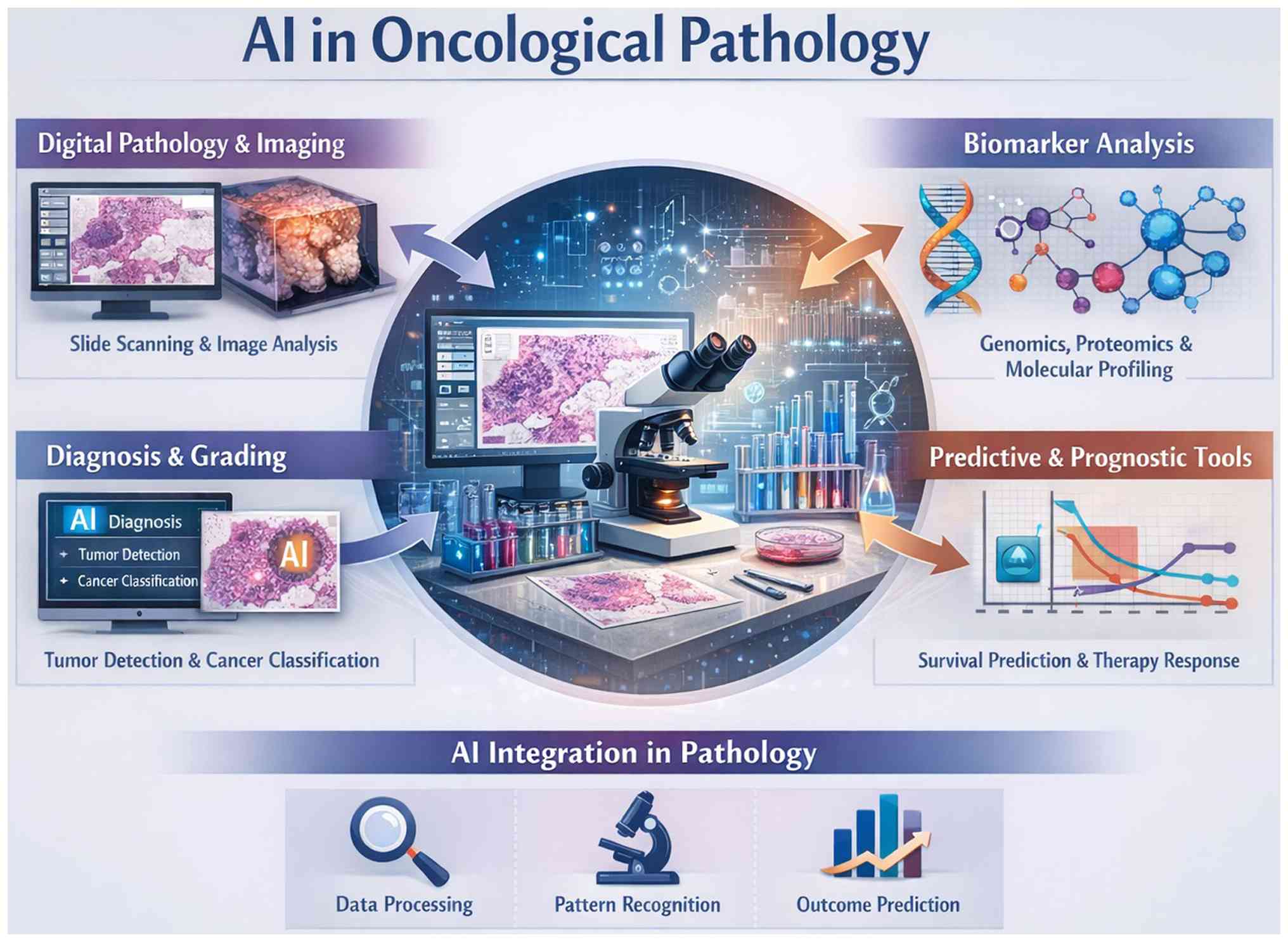
further enhance predictive performance (Table II). The integration of AI across
oncological pathology, highlighting its role in digital slide
analysis, biomarker profiling, tumour detection and predictive
prognostication is illustrated in Fig.
1. The schematic illustration in Fig. 1 emphasizes multimodal data processing
from histopathology and molecular features to outcome prediction,
demonstrating how AI supports diagnosis, grading and personalized
therapy planning within a unified pathology workflow.
4. AI in predicting clinical outcomes
Predicting clinical outcomes, such as survival,
treatment toxicity and therapeutic response is central to
personalized oncology (Table I). AI
models integrating radiologic, histopathologic, genomic and
clinical data have shown promise in improving risk stratification
beyond traditional staging systems (30-33).
Ensemble ML methods and deep neural networks have been applied to
predict progression-free and overall survival in lung, colorectal,
breast and head and neck cancers (34-36).
These models provide several potential advantages,
including the early identification of high-risk patients, the
proactive modification of treatment strategies and the improved
selection of therapeutic modalities. However, a number of outcome
prediction models are developed using limited or single-institution
datasets, increasing susceptibility to overfitting and reducing
external validity (37-40).
Interpretability remains a major challenge, particularly when
models generate probabilistic outputs without clear clinical
actionability.
Another limitation is the limited incorporation of
longitudinal data. Cancer progression and treatment response are
dynamic processes, and static baseline models may fail to capture
temporal changes. Recent research has emphasized the importance of
longitudinal modelling using real-world data and post-deployment
auditing to ensure safety and performance stability over time
(4,41,42)
(Table III). Prospective,
multi-centre validation and close collaboration between clinicians
and data scientists are essential to align AI tools with real
clinical needs.
 | Table IIIAI-driven challenges in oncology:
Computational, ethical/regulatory and data quality domains. |
Table III
AI-driven challenges in oncology:
Computational, ethical/regulatory and data quality domains.
| Domain | Key challenges |
|---|
| Computational | High hardware
requirements, model instability over time, lack of standardized
benchmarking, workflow integration difficulties |
|
Ethical/regulatory | Limited
transparency, accountability concerns, unclear liability, bias
amplification, delayed regulatory approval, failures such as IBM
Watson for Oncology due to inadequate validation |
| Data quality | Dataset bias, small
sample sizes, inconsistent acquisition and annotation, missing
longitudinal data, underrepresentation of LMIC populations |
| Equity (LMICs) | Infrastructure
gaps, workforce shortages, limited digitization, algorithmic bias,
restricted access to validated AI tools |
5. AI in chemotherapy
Chemotherapy selection and dosing require careful
consideration of tumour biology, patient characteristics and the
risk of toxicity (Table I). ML and
DL approaches have improved the prediction of the drug response by
modelling complex pharmacogenomic and molecular interactions that
are difficult to capture using conventional statistical methods
(43-47).
Models trained on large cancer cell-line datasets and multi-omics
data have demonstrated superior performance in predicting drug
sensitivity and resistance (48).
Despite encouraging preclinical and retrospective
results, translation to routine clinical care remains limited.
High-quality, well-annotated clinical pharmacogenomic datasets are
limited, and the majority of models lack prospective validation
(1,49). Limited interpretability further
constrains clinician confidence and shared decision-making
(Table II) (50,51).
Reporting guidelines for clinical trials evaluating AI
interventions underscore the need for rigorous study design and
transparency.
Future directions include the integration of
real-world clinical data, the development of explainable AI
frameworks and prospective clinical trials evaluating AI-guided
chemotherapy strategies. Reinforcement learning approaches may
enable adaptive treatment optimization based on individual patient
response trajectories.
6. AI in radiotherapy
Radiotherapy is a highly data-intensive discipline,
making it particularly amenable to AI-driven optimization.
Applications include automated contouring, dose calculation,
treatment planning, toxicity prediction, and adaptive radiotherapy
(Table I). While Monte Carlo
simulations remain the gold standard for dose calculation, they are
computationally intensive (52-54).
DL models can generate accurate dose distributions rapidly,
improving workflow efficiency (55).
Reinforcement learning has been explored for
adaptive radiotherapy, enabling treatment plans to evolve in
response to anatomical and biological changes during the course of
therapy (56). Generative
adversarial networks have also been investigated for synthetic data
generation to address limited sample sizes and class imbalance.
However, challenges related to interpretability, regulatory
approval and integration into existing planning systems persist
(Table I) (57).
Future research is warranted to prioritize
transparent and interpretable models that provide clinically
meaningful rationales for dose modification and toxicity
prediction. Standardized validation protocols, prospective clinical
evaluation, and integration with clinical decision-support systems
will be critical for safe and effective deployment.
7. Challenges and ethical
considerations
The clinical translation of AI in oncology is
hampered by regulatory requirements demanding robust evidence of
safety, efficacy and generalizability. Data-related challenges
(Table III) include inconsistent
acquisition, annotation and pre-processing protocols, as well as
sampling and observation bias. Models trained on unrepresentative
datasets may perform poorly in underrepresented populations,
exacerbating health disparities, particularly in low- and
middle-income countries (58-60).
Several AI systems in oncology have indeed been
withdrawn or scaled back following deployment due to inadequate
validation and poor real-world performance. Perhaps the most
notable example is IBM Watson for Oncology, which was marketed as
an AI-driven treatment recommendation tool, but lacked robust
clinical validation; it produced recommendations that often did not
align with real-world practice and was ultimately discontinued when
hospitals withdrew and IBM sold its Watson Health division after
failing to demonstrate clinical utility and safety (61,62).
Other older oncology decision support prototypes, including
rule-based systems such as OncoDoc and its successors, were never
widely adopted outside research settings due to high rates of
guideline-discordant recommendations and insufficient validation on
diverse clinical data (63). These
cases underscore the risks of implementing AI in oncology without
rigorous external validation and prospective outcome evaluation
before broad clinical use.
Ethical governance frameworks need to prioritize
fairness, transparency, accountability and inclusivity to prevent
AI from reinforcing existing inequities (Table III). Robust data governance
policies are required to balance innovation with privacy, informed
consent and security. The engagement of all stakeholders, including
patients, clinicians, developers, regulators and ethicists, is
essential, as is continuous performance monitoring and
post-deployment auditing.
8. Emerging frontiers
Several emerging paradigms have the potential to
overcome persistent scientific, clinical and implementation
barriers in AI-enabled oncology. Among these, multimodal AI
(Table II) represents a critical
advancement by integrating heterogeneous data sources, namely
radiological imaging, digital pathology, genomics, proteomics,
laboratory parameters and longitudinal electronic health records
into unified predictive frameworks. Such models better capture
tumour heterogeneity, temporal disease evolution and
patient-specific context, thereby enabling more accurate diagnosis,
risk stratification, treatment selection and outcome prediction
compared with unimodal approaches (64).
Foundation models (Table
II) pretrained on large, diverse and multi-institutional
datasets are increasingly influential in oncology. These models
leverage self-supervised or weakly supervised learning to acquire
generalizable representations that can be efficiently fine-tuned
for specific cancer types, modalities, or clinical tasks.
Foundation models reduce the dependence on extensive labelled
datasets, enhance robustness across populations and scanners, and
facilitate rapid deployment in resource-variable settings,
including smaller centres (65-67).
Federated learning (Table II) provides a pragmatic solution to
data-sharing constraints by enabling collaborative model training
across institutions without centralized transfer of sensitive
patient data. This paradigm is particularly valuable in oncology,
where data scarcity, privacy regulations, and institutional silos
limit model generalizability. Federated approaches can improve
performance across diverse demographic and clinical settings while
maintaining compliance with data protection frameworks (Table II) (68,69).
Despite these technical advances, explainable AI
remains central to clinical adoption, regulatory approval and
medicolegal accountability. Transparent models that provide
interpretable features, uncertainty estimates, and clinically
meaningful visualizations foster clinician trust and support safe
integration into decision-making workflows (Table II) (70,71).
Finally, ethical and equity considerations are
especially salient in low- and middle-income countries. Challenges
related to infrastructure, data quality, workforce training and
algorithmic bias (Table III) need
to be addressed through targeted capacity building, inclusive
dataset development and international collaboration. Without
deliberate governance and context-aware implementation, AI risks
exacerbating existing disparities (72,73).
Ensuring fairness, transparency and accessibility will be essential
for realizing the global promise of AI-driven oncology.
9. Conclusion
AI holds transformative potential to advance
precision oncology by improving diagnostic accuracy,
prognostication and treatment optimization. Realizing this
potential requires rigorous prospective validation, transparent
model design, multidisciplinary collaboration and robust ethical
safeguards. With responsible development and implementation, AI can
become an integral component of oncology practice and contribute
meaningfully to improved cancer outcomes worldwide.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
AR and AB jointly conceptualized the review. AR, AB,
AA and NC generated the outline of the review. AR, AB, AA and YP
drafted the manuscript. AA, YP and NC reviewed the manuscript. Data
authentication is not applicable. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Topol EJ: High-performance medicine: The
convergence of human and artificial intelligence. Nat Med.
25:44–56. 2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Esteva A, Chou K, Yeung S, Naik N, Madani
A, Mottaghi A, Liu Y, Topol E, Dean J and Socher R: Deep
learning-enabled medical computer vision. NPJ Digit Med.
4(5)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Biswas M, Kuppili V, Saba L, Edla DR, Suri
HS, Cuadrado-Godia E, Laird JR, Marinhoe RT, Sanches JM, Nicolaides
A and Suri JS: State-of-the-art review on deep learning in medical
imaging. Front Biosci (Landmark Ed). 24:392–426. 2019.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Xu Y, Li Y, Wang F, Zhang Y and Huang D:
Addressing the current challenges in the clinical application of
AI-based radiomics for cancer imaging. Front Med (Lausanne).
12(1674397)2025.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Abbas Q, Jeong W and Lee SW: Explainable
AI in clinical decision support systems: A meta-analysis of
methods, applications, and usability challenges. Healthcare
(Basel). 13(2154)2025.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Chua IS, Gaziel-Yablowitz M, Korach ZT,
Kehl KL, Levitan NA, Arriaga YE, Jackson GP, Bates DW and Hassett
M: Artificial intelligence in oncology: Path to implementation.
Cancer Med. 10:4138–4149. 2021.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Sultan AS, Elgharib MA, Tavares T, Jessri
M and Basile JR: The use of artificial intelligence, machine
learning and deep learning in oncologic histopathology. J Oral
Pathol Med. 49:849–856. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hong GS, Jang M, Kyung S, Cho K, Jeong J,
Lee GY, Shin K, Kim KD, Ryu SM, Seo JB, et al: Overcoming the
challenges in the development and implementation of artificial
intelligence in radiology: A comprehensive review of solutions
beyond supervised learning. Korean J Radiol. 24:1061–1080.
2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ueda D, Kakinuma T, Fujita S, Kamagata K,
Fushimi Y, Ito R, Matsui Y, Nozaki T, Nakaura T, Fujima N, et al:
Fairness of artificial intelligence in healthcare: Review and
recommendations. Jpn J Radiol. 42:3–15. 2024.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Melazzini L, Bortolotto C, Brizzi L,
Achilli M, Basla N, D'Onorio De Meo A, Gerbasi A, Bottinelli OM,
Bellazzi R and Preda L: AI for image quality and patient safety in
CT and MRI. Eur Radiol Exp. 9(28)2025.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Nie Y, Sommella P, Carratù M, O'Nils M and
Lundgren J: A deep CNN transformer hybrid model for skin lesion
classification of dermoscopic images using focal loss. Diagnostics
(Basel). 13(72)2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tschandl P, Rinner C, Apalla Z, Argenziano
G, Codella N, Halpern A, Janda M, Lallas A, Longo C, Malvehy J, et
al: Human-computer collaboration for skin cancer recognition. Nat
Med. 26:1229–1234. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Goyal H, Mann R, Gandhi Z, Perisetti A,
Ali A, Aman Ali K, Sharma N, Saligram S, Tharian B and Inamdar S:
Scope of artificial intelligence in screening and diagnosis of
colorectal cancer. J Clin Med. 9(3313)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ho TY, Chao CH, Chin SC, Ng SH, Kang CJ
and Tsang NM: Classifying neck lymph nodes of head and neck
squamous cell carcinoma in MRI images with radiomic features. J
Digit Imaging. 33:613–618. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yala A, Lehman C, Schuster T, Portnoi T
and Barzilay R: A deep learning mammography-based model for
improved breast cancer risk prediction. Radiology. 292:60–66.
2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sivakumar R, Lue B and Kundu S: FDA
approval of artificial intelligence and machine learning devices in
radiology: A systematic review. JAMA Netw Open.
8(e2542338)2025.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Evangelou K, Kotsantis I, Kalyvas A,
Kyriazoglou A, Economopoulou P, Velonakis G, Gavra M, Psyrri A,
Boviatsis EJ and Stavrinou LC: Artificial intelligence in the
diagnosis and treatment of brain gliomas. Biomedicines.
13(2285)2025.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kelly CJ, Karthikesalingam A, Suleyman M,
Corrado G and King D: Key challenges for delivering clinical impact
with artificial intelligence. BMC Med. 17(195)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Roberts M, Driggs D, Thorpe M, Gilbey J,
Yeung M, Ursprung S, Aviles-Rivero AI, Etmann C, McCague C, Beer L,
et al: Common pitfalls and recommendations for using machine
learning to detect and prognosticate for COVID-19 using chest
radiographs and CT scans. Nat Mach Intell. 3:199–217. 2021.
|
|
20
|
Zech JR, Badgeley MA, Liu M, Costa AB,
Titano JJ and Oermann EK: Variable generalization performance of a
deep learning model to detect pneumonia in chest radiographs: A
cross-sectional study. PLoS Med. 15(e1002683)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Tran AT, Zeevi T and Payabvash S:
Strategies to Improve the Robustness and Generalizability of Deep
Learning Segmentation and Classification in Neuroimaging.
BioMedInformatics. 5(20)2025.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chaddad A, Peng J, Xu J and Bouridane A:
Survey of explainable AI techniques in healthcare. Sensors (Basel).
23(634)2023.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yang M, Huang D, Wan W and Jin M:
Federated learning for privacy-preserving medical data sharing in
drug development. Appl Comput Eng. 134:80–84. 2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Iizuka O, Kanavati F, Kato K, Rambeau M,
Arihiro K and Tsuneki M: Deep learning models for histopathological
classification of gastric and colonic epithelial tumours. Sci Rep.
10(1504)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Butt MA, Kaleem MF, Bilal M and Hanif MS:
Using multi-label ensemble CNN classifiers to mitigate labelling
inconsistencies in patch-level Gleason grading. PLoS One.
19(e0304847)2024.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wang X, Jiang Y, Yang S, Wang F, Zhang X,
Wang W, Chen Y, Wu X, Xiang J, Li Y, et al: Foundation model for
predicting prognosis and adjuvant therapy benefit from digital
pathology in GI cancers. J Clin Oncol. 43:3468–3481.
2025.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Qaiser T, Lee CY, Vandenberghe M, Yeh J,
Gavrielides MA, Hipp J, Scott M and Reischl J: Usability of deep
learning and H&E images predict disease outcome-emerging tool
to optimize clinical trials. NPJ Precis Oncol. 6(37)2022.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Hsu CY, Askar S, Alshkarchy SS, Nayak PP,
Attabi KAL, Khan MA, Mayan JA, Sharma MK, Islomov S and Soleimani
Samarkhazan H: AI-driven multi-omics integration in precision
oncology: Bridging the data deluge to clinical decisions. Clin Exp
Med. 26(29)2025.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Lekadir K, Feragen A, Fofanah AJ, Frangi
AF, Buyx A, Emelie A, Lara A, Porras AR, Chan AW, Navarro A, et al:
FUTURE-AI: International consensus guideline for trustworthy and
deployable artificial intelligence in healthcare. arXiv:
2309.12325, 2023.
|
|
30
|
Elemento O, Khozin S and Sternberg CN: The
use of artificial intelligence for cancer therapeutic
decision-making. NEJM AI. 2(10.1056/aira2401164)2025.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Shao J, Ma J, Zhang Q, Li W and Wang C:
Predicting gene mutation status via artificial intelligence
technologies based on multimodal integration (MMI) to advance
precision oncology. Semin Cancer Biol. 91:1–15. 2023.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Awuah WA, Ben-Jaafar A, Roy S,
Nkrumah-Boateng PA, Tan JK, Abdul-Rahman T and Atallah O:
Predicting survival in malignant glioma using artificial
intelligence. Eur J Med Res. 30(61)2025.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Bera K, Braman N, Gupta A, Velcheti V and
Madabhushi A: Predicting cancer outcomes with radiomics and
artificial intelligence in radiology. Nat Rev Clin Oncol.
19:132–146. 2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wang S, Zhang H, Liu Z and Liu Y: A novel
deep learning method to predict lung cancer long-term survival with
biological knowledge incorporated gene expression images and
clinical data. Front Genet. 13(800853)2022.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Akselrod-Ballin A, Chorev M, Shoshan Y,
Spiro A, Hazan A, Melamed R, Barkan E, Herzel E, Naor S, Karavani
E, et al: Predicting breast cancer by applying deep learning to
linked health records and mammograms. Radiology. 292:331–342.
2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Chiu YC, Chen HIH, Zhang T, Zhang S,
Gorthi A, Wang LJ, Huang Y and Chen Y: Predicting drug response of
tumors from integrated genomic profiles by deep neural networks.
BMC Med Genomics. 12 (Suppl 1)(S18)2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Huynh BN, Groendahl AR, Tomic O, Liland
KH, Knudtsen IS, Hoebers F, van Elmpt W, Malinen E, Dale E and
Futsaether CM: Head and neck cancer treatment outcome prediction: A
comparison between machine learning with conventional radiomics
features and deep learning radiomics. Front Med (Lausanne).
10(1217037)2023.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Luo Y, Tseng HH, Cui S, Wei L, Ten Haken
RK and El Naqa I: Balancing accuracy and interpretability of
machine learning approaches for radiation treatment outcomes
modeling. BJR Open. 1(20190021)2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Sinha T, Khan A, Awan M, Bokhari SFH, Ali
K, Amir M, Jadhav AN, Bakht D, Puli ST and Burhanuddin M:
Artificial intelligence and machine learning in predicting the
response to immunotherapy in non-small cell lung carcinoma: A
systematic review. Cureus. 16(e61220)2024.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Pesapane F, Nicosia L, D'Amelio L,
Quercioli G, Pannarale MR, Priolo F, Marinucci I, Farina MG, Penco
S, Dominelli V, et al: Artificial intelligence-driven
personalization in breast cancer screening: From population models
to individualized protocols. Cancers (Basel).
17(2901)2025.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ennab M and Mcheick H: Enhancing
interpretability and accuracy of AI models in healthcare: A
comprehensive review on challenges and future directions. Front
Robot AI. 11(1444763)2024.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Niu S, Ma J, Yin Q, Wang Z, Bai L and Yang
X: Modelling patient longitudinal data for clinical decision
support: A case study on emerging AI healthcare Technologies. Inf
Syst Front. 27:409–427. 2025.
|
|
43
|
Sartori F, Codicè F, Caranzano I, Rollo C,
Birolo G, Fariselli P and Pancotti C: A comprehensive review of
deep learning applications with multi-omics data in cancer
research. Genes (Basel). 16(648)2025.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Clayton EA, Pujol TA, McDonald JF and Qiu
P: Leveraging TCGA gene expression data to build predictive models
for cancer drug response. BMC Bioinformatics. 21 (Suppl
14)(S364)2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Liu X, Song C, Huang F, Fu H, Xiao W and
Zhang W: GraphCDR: A graph neural network method with contrastive
learning for cancer drug response prediction. Brief Bioinform.
23(bbab457)2021.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Ali M and Aittokallio T: Machine learning
and feature selection for drug response prediction in precision
oncology applications. Biophys Rev. 11:31–39. 2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Kalinin AA, Higgins GA, Reamaroon N,
Soroushmehr S, Allyn-Feuer A, Dinov ID, Najarian K and Athey BD:
Deep learning in pharmacogenomics: From gene regulation to patient
stratification. Pharmacogenomics. 19:629–650. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Sharifi-Noghabi H, Jahangiri-Tazehkand S,
Smirnov P, Hon C, Mammoliti A, Nair SK, Mer AS, Ester M and
Haibe-Kains B: Drug sensitivity prediction from cell line-based
pharmacogenomics data: Guidelines for developing machine learning
models. Brief Bioinform. 22(bbab294)2021.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Beam AL and Kohane IS: Big data and
machine learning in health care. JAMA. 319:1317–1318.
2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Vamathevan J, Clark D, Czodrowski P,
Dunham I, Ferran E, Lee G, Li B, Madabhushi A, Shah P, Spitzer M
and Zhao S: Applications of machine learning in drug discovery and
development. Nat Rev Drug Discov. 18:463–477. 2019.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Holzinger A, Langs G, Denk H, Zatloukal K
and Müller H: Causability and explainability of artificial
intelligence in medicine. Wiley Interdiscip Rev Data Min Knowl
Discov. 9(e1312)2019.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Damilakis J and Stratakis J: Descriptive
overview of AI applications in x-ray imaging and radiotherapy. J
Radiol Prot. 44(041001)2024.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Psoroulas S, Paunoiu A, Corradini S,
Hörner-Rieber J and Tanadini-Lang S: MR-linac: Role of artificial
intelligence and automation. Strahlenther Onkol. 201:298–305.
2025.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Smolders A, Lomax A, Weber DC and
Albertini F: Deep learning based uncertainty prediction of
deformable image registration for contour propagation and dose
accumulation in online adaptive radiotherapy. Phys Med Biol.
68(245027)2023.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Chen X, Men K, Li Y, Yi J and Dai J: A
feasibility study on an automated method to generate
patient-specific dose distributions for radiotherapy using deep
learning. Med Phys. 46:56–64. 2019.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Li C, Guo Y, Lin X, Feng X, Xu D and Yang
R: Deep reinforcement learning in radiation therapy planning
optimization: A comprehensive review. Phys Med.
125(104498)2024.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Akpinar MH, Sengur A, Salvi M, Seoni S,
Faust O, Mir H, Molinari F and Acharya UR: Synthetic data
generation via generative adversarial networks in healthcare: A
systematic review of image- and signal-based studies. IEEE Open J
Eng Med Biol. 6:183–192. 2024.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Chen Y, Clayton EW, Novak LL, Anders S and
Malin B: Human-centered design to address biases in artificial
intelligence. J Med Internet Res. 25(e43251)2023.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Abràmoff MD, Tarver ME, Loyo-Berrios N,
Trujillo S, Char D, Obermeyer Z and Eydelman MB: Foundational
Principles of Ophthalmic Imaging and Algorithmic Interpretation
Working Group of the Collaborative Community for Ophthalmic Imaging
Foundation. Washington D.C..Maisel WH: Considerations for
addressing bias in artificial intelligence for health equity. NPJ
Digit Med. 6(170)2023.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Tejani AS, Ng YS, Xi Y and Rayan JC:
Understanding and mitigating bias in imaging artificial
intelligence. Radiographics. 44(e230067)2024.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Ross C and Swetlitz I: IBM's Watson
supercomputer recommended ‘unsafe and incorrect’ cancer treatments,
internal documents show. STAT, Boston, MA, 2018.
|
|
62
|
Strickland E: IBM Watson, heal thyself:
How IBM overpromised and underdelivered on AI health care. IEEE
Spectr. 56:24–31. 2019.
|
|
63
|
Séroussi B, Laouénan C, Gligorov J, Uzan
S, Mentré F and Bouaud J: Which breast cancer decisions remain
non-compliant with guidelines despite the use of computerised
decision support? Br J Cancer. 109:1147–1156. 2013.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Jandoubi B and Akhloufi MA: Multimodal
artificial intelligence in medical diagnostics. Information.
16(591)2025.
|
|
65
|
Tak D, Garomsa BA, Chaunzwa TL,
Zapaishchykova A, Climent Pardo JC, Ye Z, Zielke J, Ravipati Y,
Vajapeyam S, Mahootiha M, et al: A foundation model for generalized
brain MRI analysis. medRxiv [Preprint]: 2024.12.02.24317992,
2024.
|
|
66
|
Yan S, Yu Z, Primiero C, Vico-Alonso C,
Wang Z, Yang L, Tschandl P, Hu M, Ju L, Tan G, et al: A multimodal
vision foundation model for clinical dermatology. Nat Med.
31:2691–2702. 2025.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Ding T, Wagner SJ, Song AH, Chen RJ, Lu
MY, Zhang A, Vaidya AJ, Jaume G, Shaban M, Kim A, et al: A
multimodal whole-slide foundation model for pathology. Nat Med.
31:3749–3761. 2025.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Hao R, Chang WC, Hu J and Gao M: Federated
Learning-Driven Health Risk Prediction on Electronic Health Records
Under Privacy. Constraints. Preprints: https://doi.org/10.20944/preprints202510.1471.v1.
|
|
69
|
Mu J, Kadoch M, Yuan T, Lv W, Liu Q and Li
B: Explainable federated medical image analysis through causal
learning and blockchain. IEEE J Biomed Health Inform. 28:3206–3218.
2024.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Rezaeian O, Bayrak AE and Asan O:
Explainability and AI confidence in clinical decision support
systems: Effects on trust, diagnostic performance, and cognitive
load in breast cancer care. arXiv: https://doi.org/10.48550/arXiv.2501.16693.
|
|
71
|
Salimparsa M, Sedig K, Lizotte DJ,
Abdullah SS, Chalabianloo N and Muanda FT: Explainable AI for
clinical decision support systems: Literature review, key gaps, and
research synthesis. Informatics. 12(119)2025.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Marey A, Ambrozaite O, Afifi A, Agarwal R,
Chellappa R, Adeleke S and Umair M: A perspective on AI
implementation in medical imaging in LMICs: Challenges, priorities,
and strategies. Eur Radiol: October 23, 2025 (Epub ahead of
print).
|
|
73
|
Kaushik A, Barcellona C, Mandyam NK, Tan
SY and Tromp J: Challenges and opportunities for data sharing
related to artificial intelligence tools in health care in low- and
middle-income countries: Systematic review and case study from
Thailand. J Med Internet Res. 27(e58338)2025.PubMed/NCBI View
Article : Google Scholar
|