Introduction
An electrical storm (ES) is a life-threatening
cardiac emergency characterized by multiple episodes of sustained
ventricular arrhythmias occurring within a 24-h time period. ES is
commonly defined as ≥3 episodes of sustained ventricular
tachycardia (VT)/ventricular fibrillation (VF) [or appropriate
implantable cardioverter-defibrillator (ICD) therapies] in 24 h
(1,2). In Western populations, ES occurs in
~4-28% of patients with ICDs, with higher rates observed in certain
groups. Severe cases, particularly with hemodynamic instability or
recurrent arrhythmias, carry an early risk of mortality >10%
(2). VT storms are most frequently
encountered in patients with structural heart disease and reduced
ejection fraction (EF). However, they can also arise in individuals
with structurally normal hearts due to diverse triggers, such as
electrolyte disturbances, thyrotoxicosis, heightened sympathetic
activity, or inherited cardiac conditions (3). ES management is complex, requiring a
multidisciplinary approach that includes antiarrhythmic drugs,
electrolyte correction and ICD therapy for optimal outcomes
(4). Although refractory ES have
been reported, few have demonstrated the extreme arrhythmic burden
or intricate device-arrhythmia interactions seen here.
The present case report describes a rare,
catastrophic case of VT storm in a patient with ischemic
cardiomyopathy, marked by 42 ICD shocks in a period of 24 h,
despite dual anti-arrhythmics and intensive support. The present
case report also highlights the diagnostic and educational value of
real-time telemetry and electrocardiogram (ECG) capture.
Case report
The present case report describes a rare and
life-threatening case of ES in a 71-year-old male patient with
ischemic cardiomyopathy (EF, 35-40%), prior MitraClip placement
with persistent moderate mitral regurgitation, cardiac
resynchronization therapy defibrillator, multiple coronary stents
and a history of pericardiocentesis to the Wellstar Spalding
Medical Center (Griffin, GA, USA) on February 26, 2025. He
presented with worsening exertional dyspnea, orthopnea, peripheral
edema and non-radiating chest pressure, symptoms emerging after a
recent reduction in diuretic therapy.
Upon admission, the patient was hypotensive (blood
pressure, 90/70 mmHg) and tachycardic (hear rate, 130 beats per
minute), with clinical features suggestive of evolving cardiogenic
shock, including cool extremities, +3 bilateral lower extremity
edema and a reduced urine output. Oxygen saturation remained 99% in
room air. A laboratory evaluation demonstrated acute kidney injury,
indicating early end-organ involvement. However, serum lactate
levels were within normal range (1 mmol/l), suggesting compensated
or early-stage shock physiology without overt systemic
hypoperfusion at the time of initial evaluation. An initial 12-lead
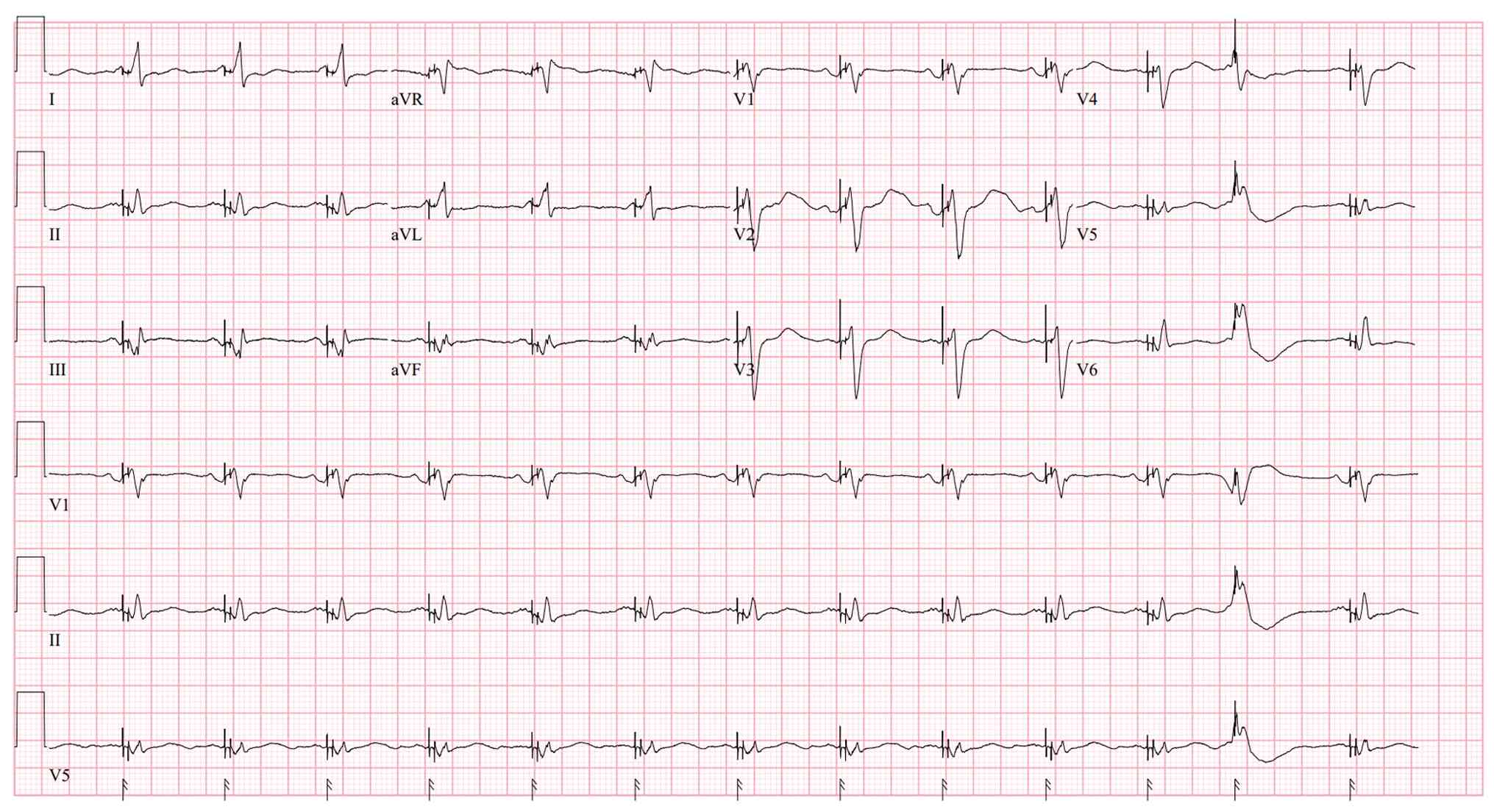
ECG (Fig. 1) demonstrated a
ventricular paced rhythm with an isolated premature ventricular
complex occurring at the end of the tracing, superimposed on a
pacemaker spike, suggesting potential ectopic trigger. Laboratory
analyses revealed a level of terminal pro-B-type natriuretic
peptide of 16,800 pg/ml, an elevated creatinine level of 2.41 mg/dl
(baseline, 1.80), and normal potassium (4.0 mmol/l), calcium (8.9
mg/dl), magnesium (2.0 mg/dl) and thyroid-stimulating hormone (2.2
µU/ml) levels, excluding common metabolic triggers.
Serum lactate levels were within normal range (1
mmol/l), suggesting adequate perfusion despite hypotension. The
patient demonstrated renal dysfunction consistent with early
end-organ involvement, while serum lactate levels remained normal,
suggesting preserved systemic perfusion at the time of evaluation.
The troponin level was mildly elevated at 102 ng/l, trending down
to 90 ng/l, consistent with demand-mediated ischemia amid an
ongoing ES. In the absence of dynamic ST changes and with prior
coronary evaluation, acute coronary syndrome was considered
unlikely, and no urgent angiography or stress testing was pursued.
An echocardiography revealed a large inferobasal left
ventricle/left ventricular (LV) aneurysm, severe left atrial
enlargement, moderate mitral regurgitation, and no evidence of LV
thrombus. Beta-blockers were avoided due to sustained hypotension
and concern for exacerbating cardiogenic shock.
Shortly following his arrival, the patient developed
sustained monomorphic VT, requiring ICD shocks. Despite intravenous
amiodarone and lidocaine, he progressed to fulminant ES with 42
appropriate ICD shocks over a period of 24 h, along with two
external cardioversions. Magnesium sulfate was administered
empirically without benefit. Persistent VT prompted admission to
the intensive care unit (ICU), intubation and initiation of deep
sedation with neuromuscular blockade, though arrhythmias continued.
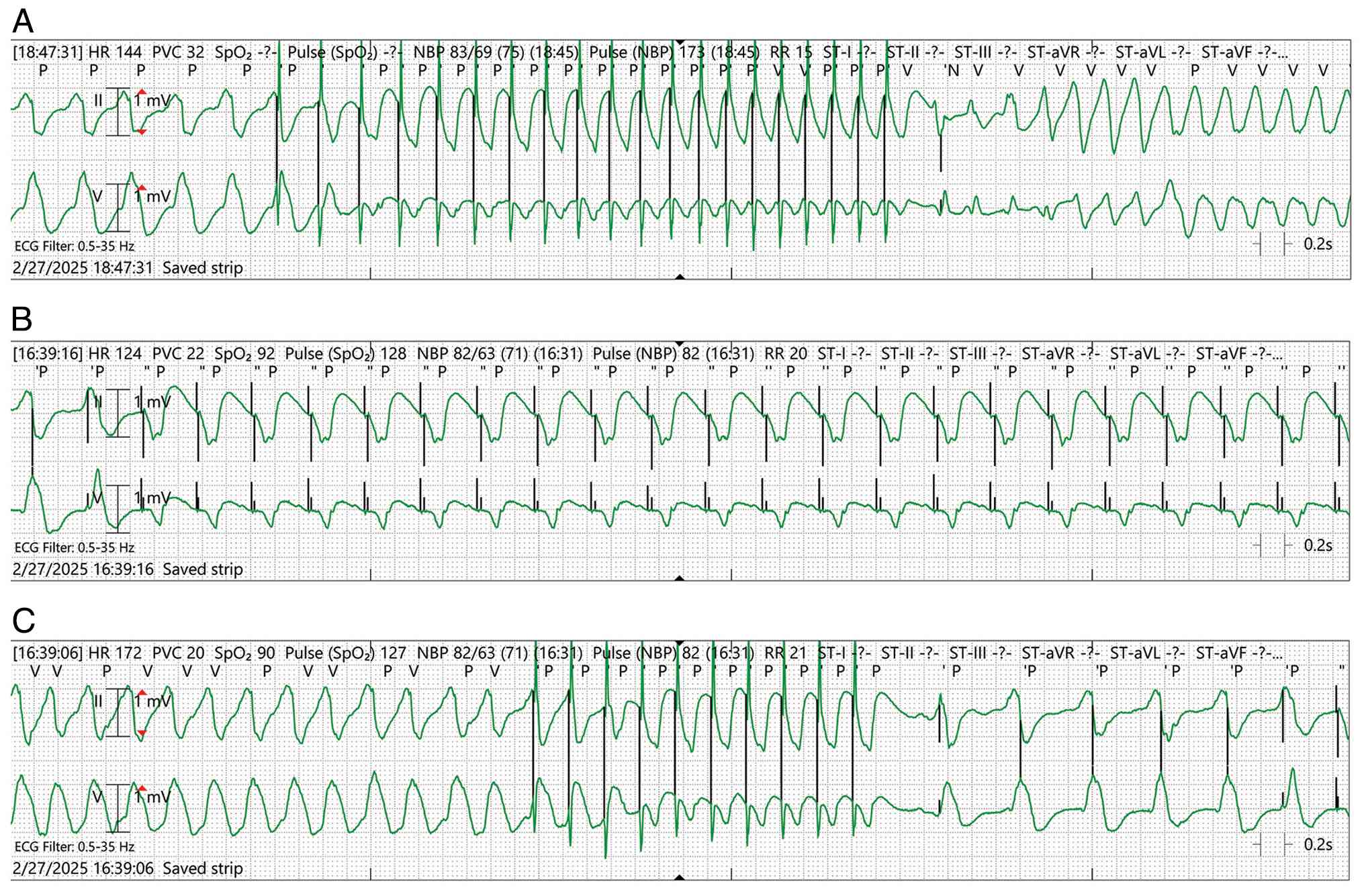
Telemetry proved diagnostically valuable: As demonstrated in
Fig. 2A, VT was transiently
suppressed by overdrive pacing, followed in some episodes by
degeneration into VF, suggesting a complex interaction between
pacing therapy and an unstable arrhythmogenic substrate rather than
inappropriate device function; As illustrated in Fig. 2B, sustained VT refractory to pacing
was observed; and Fig. 2C
illustrates VT interrupted by burst pacing, followed by slower
recurrence. These recordings highlight complex device-arrhythmia
interactions and the refractory nature of the storm.
ICD interrogation revealed optimal anti-tachycardia
pacing and shock settings with no malfunctions; thus, reprogramming
was not needed. The device documented a high VT burden with
appropriate therapies delivered. The patient received a 150 mg
intravenous amiodarone bolus followed by continuous infusion at 0.5
mg/min, and lidocaine was commenced at 1 mg/min after a 100 mg
bolus. Despite dual antiarrhythmic therapy, inotropes and
aggressive diuresis, VT persisted. Hemodynamics were tenuously
maintained with norepinephrine. Mechanical circulatory support
(e.g., intra-aortic balloon pump or Impella) was considered;
however, these were unavailable at the treating center.
Urgent electrophysiology consultation recommended
transfer for VT ablation and cardiac sympathetic denervation.
However, due to regional ICU bed shortages, the patient remained at
the referring facility. The prognosis remained poor. Despite
transient stabilization with aggressive medical and supportive
therapy, the clinical condition of the patient progressively
deteriorated, and he unfortunately passed away on hospital day 5
from admission before advanced electrophysiologic therapies could
be initiated.
Discussion
ES may manifest as polymorphic VT, monomorphic VT,
or VF, and is more commonly observed in patients with structural or
infiltrative heart diseases, such as amyloidosis or sarcoidosis.
Potentially reversible triggers include acute myocardial ischemia,
electrolyte abnormalities, thyrotoxicosis, QT prolongation and drug
toxicity. It may also be precipitated by reperfusion injury or
myocardial scarring that serves as an arrhythmogenic substrate.
Polymorphic VT is often associated with ischemia or metabolic
disturbances, whereas monomorphic VT generally arises from
scar-mediated reentry (2,5). The diverse pathophysiology of ES
underscores the need to identify underlying triggers. In the case
described herein, a large LV aneurysm, ischemic cardiomyopathy and
prior pericardial intervention likely formed the substrate for the
refractory monomorphic VT storm.
ES from VT commonly occurs in patients with
structural heart disease, as in the case described in the present
study. It can present with symptoms, such as palpitations, chest
pain, dyspnea, or even cardiac arrest. Early resuscitation and
ICU-level care are critical for stabilization and monitoring
(1). The rapid progression from
stable presentation to ICU intubation and multi-drug therapy over
24-48 h underscores the fulminant nature of electrical storm. The
primary approach to treating patients with recurrent VT or VF
relies on pharmacologic therapy, with antiarrhythmic agents such as
amiodarone serving as a foundational component (6). Current guidelines support early
beta-blocker use (e.g., IV esmolol or metoprolol) in ES. However,
in the patient described herein, beta-blockade was deferred due to
hypotension and suspected cardiogenic shock. Management followed
guideline-based strategies, including dual antiarrhythmics,
sedation to reduce sympathetic drive and vasopressors for
hemodynamic support, aligning with the American College of
Cardiology recommendations for aggressive, team-based ES care
(2). Despite a continuous amiodarone
infusion, the arrhythmia of the patient remained refractory,
prompting the addition of lidocaine for enhanced rhythm
control.
The observed transition from pacing-mediated VT
suppression to subsequent VF likely reflects dynamic
electrophysiologic substrate instability rather than pro-arrhythmic
device behavior. In scar-mediated ventricular arrhythmias, pacing
may transiently terminate re-entrant circuits, but can also alter
conduction pathways, occasionally facilitating degeneration into
more malignant rhythms in highly unstable myocardial substrates.
Importantly, device interrogation in our patient confirmed
appropriate sensing, detection and therapy delivery, supporting
appropriate device function despite arrhythmia recurrence.
In ES, the prompt management of pain and
psychological distress is critical, as recurrent external or
ICD-mediated shocks can increase sympathetic activation through
catecholamine surge, thereby exacerbating ventricular arrhythmias.
Sedation with appropriate analgesia and anxiolysis is recommended
to mitigate this adrenergic response and improve patient comfort.
In the case described in the present study, deep sedation with
neuromuscular blockade was implemented for sympathetic suppression
and ventilator synchrony; however, the arrhythmia remained
refractory. Current expert consensus supports sedation ranging from
mild to deep levels for both arrhythmia control and patient comfort
in ES management (2,7).
Percutaneous stellate ganglion block has emerged as
a potential temporizing therapy for refractory ES by reducing
sympathetic outflow and ventricular arrhythmia burden. Although
this strategy has shown promising results as a bridge to definitive
therapies (2), such as catheter
ablation or surgical denervation, bedside SGB was not available at
the authors' institution (Wellstar Saplding Medical Center) at the
time of patient management and therefore, could not be pursued.
ES in patients with structural heart disease is
associated with a high risk of early mortality, and catheter
ablation, particularly when achieving complete VT elimination,
provides a favorable outcome with ES-free survival rates >90%
(8). Catheter ablation is a key
therapy for refractory VT storm, particularly when complete VT
suppression is possible. In the case in the present study, VT
persisted despite dual antiarrhythmics, sedation and support,
leading to plans for urgent ablation and sympathectomy. Unlike
prior reports [e.g., Nakayama et al (8), Ahadzi et al (1) and Rahman and Sohail (4)], the patient in the present study
experienced an unusually high ICD shock burden with no response to
aggressive treatment.
In a previous study, a pooled meta-analysis of 471
patients with ES treated using catheter, ethanol, or surgical
ablation reported favorable outcomes: In total, 72% achieved
complete arrhythmia elimination, and 91% had successful suppression
of clinical VTs (9). Complications
were rare (2%), with <1% procedure-related mortality. At 1.2
years, 94% remained free of ES and 72% had no VT recurrence, though
overall mortality was 17%, primarily due to progressive heart
failure (9). In line with these
findings, the patient in the present study was being prepared for
transfer to a tertiary care center for consideration of VT ablation
and cardiac sympathetic denervation, highlighting the critical role
of advanced interventional strategies in refractory ES
management.
Trans-coronary ethanol ablation has emerged as an
option for refractory VT in structural heart disease, particularly
when catheter ablation fails. In a series of 46 patients, selective
coronary angiography guided ethanol delivery achieved partial
success in 66%. However, VT has been shown to recur in 74% of
patients at 6 months and 82% of patients at 12 months, with a 32%
complication rate, including one procedural death (10).
Transcoronary ethanol ablation may benefit select
patients; however, the patient described herein, with a large LV
aneurysm and complex arrhythmogenic substrate, had not undergone
ablation. Despite dual antiarrhythmics and supportive care, he
experienced 42 ICD shocks in 24 h, highlighting a rare, severe VT
storm and the urgent need for timely advanced electrophysiologic
intervention in high-risk cases.
In conclusion, the present case report underscores
the extreme and life-threatening nature of ES in patients with
structural heart disease, particularly when refractory to optimized
device settings and dual antiarrhythmic therapy. The documentation
of 42 appropriate ICD shocks within 24 h represents one of the
highest recorded burdens of therapy-resistant VT, highlighting the
limitations of standard pharmacological and device-based
management. The early recognition of ES physiology, combined with
real-time diagnostic tools such as telemetry, is critical for
timely intervention. The present case report reinforces the need
for rapid multidisciplinary escalation, including electrophysiology
consultation, catheter ablation and sympathetic denervation for
improving outcomes in refractory cases. It also provides valuable
electrophysiological insights into device-arrhythmia interactions
that can inform future management of complex arrhythmias.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
PA was involved in the conception and design of the
case report, drafting the article or revising it critically for
important intellectual content. NN was involved in the literature
review and obtained medical images. ARQ obtained medical images. HS
was involved in the drafting of the article and in the conception
of the study. NO analyzed the patient's data. AT advised on patient
treatment. DP analyzed the patient's data and gave the final
approval of the version to be submitted. PA and NO confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from the
patient's family member for his inclusion in the present case
report.
Patient consent for publication
Written informed consent for publication of this
case and any accompanying images was obtained from the patient's
family member.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ahadzi D, Agyekum F, Doku A, Yakubu AS,
Hoedofia G and Ayetey H: Electrical storm in a middle-aged man.
Ghana Med J. 57:156–160. 2023.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Jentzer JC, Noseworthy PA, Kashou AH, May
AM, Chrispin J, Kabra R, Arps K, Blumer V, Tisdale JE and Solomon
MAL: American College of Cardiology Critical Care Cardiology and
Electrophysiology Sections. Multidisciplinary Critical Care
Management of Electrical Storm: JACC State-of-the-Art Review. J Am
Coll Cardiol. 81:2189–2206. 2023.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Muser D, Santangeli P and Liang JJ:
Management of ventricular tachycardia storm in patients with
structural heart disease. World J Cardiol. 9:521–530.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rahman I and Sohail M: The mystery of
electrical storm: A case report. Cureus. 16(e57202)2024.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Rodriguez AP, Badiye A, Lambrakos LK,
Ghodsizad A, Myerburg RJ and Goldberger JJ: Refractory ventricular
tachycardia storm associated with severe hypokalemia in Fanconi
syndrome. HeartRhythm Case Rep. 5:374–378. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kowey PR: An overview of antiarrhythmic
drug management of electrical storm. Can J Cardiol. 12 (Suppl
B):3B–8B; discussion 27B-28B. 1996.PubMed/NCBI
|
|
7
|
Arya A, Bode K, Piorkowski C, Bollmann A,
Sommer P, Gaspar T, Wetzel U, Husser D, Kottkamp H and Hindricks G:
Catheter ablation of electrical storm due to monomorphic
ventricular tachycardia in patients with nonischemic
cardiomyopathy: acute results and its effect on long-term survival.
Pacing Clin Electrophysiol. 33:1504–1509. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Nakayama H, Komatsu J, Nishimura Y, Sugane
H, Hosoda H, Imai RI, Nakaoka Y, Nishida K, Mito S, Seki SI, et al:
Refractory electrical storm in a patient with hypertrophic
cardiomyopathy and an implantable cardioverter-defibrillator. J
Cardiol Cases. 27:233–236. 2023.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Nayyar S, Ganesan AN, Brooks AG, Sullivan
T, Roberts-Thomson KC and Sanders P: Venturing into ventricular
arrhythmia storm: A systematic review and meta-analysis. Eur Heart
J. 34:560–571. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kowlgi GN and Cha YM: Management of
ventricular electrical storm: a contemporary appraisal. Europace.
22:1768–1780. 2020.PubMed/NCBI View Article : Google Scholar
|