Introduction
Cancer of the pancreas is often detected at a late
stage. At that juncture, it has commonly disseminated and is
therefore afflicted by a poor prognosis. A better understanding of
the molecular mechanisms underlying the disease, in particular its
progression and dissemination, should point the way to improved
clinical care. In tumor stage progression and metastasis formation
of this malignancy, osteopontin (also known as OPN or SPP1) has
been known to be a major contributor (1-3).
Hence, it is of importance to elucidate the knowledge base about
this secreted phosphoglycoprotein, pertaining to the induction of
its aberrant expression and its splice variants (4,5) in
pancreatic cancers. Such insights may improve early
detection/diagnosis and can point the way toward novel therapeutic
targets associated with tumor spread. For this purpose, the present
systematic review was conducted, analyzing studies found on
PubMed-listed database on this subject (Fig. S1).
Data and methods
Source literature
Publications in the PubMed database with the key
phrase ‘pancreas cancer OR pancreatic cancer AND osteopontin’
yielded 105 references (cut-off date, December 31, 2025). A total
of 26 articles were found not to be pertinent, predominantly as
they studied cancers of different organ origin (mostly liver
cancers, but also colorectal and gastric cancers); 1 article had
been retracted. Of note, ‘osteopontin AND pancreas NOT cancer’
produced 39 results (cut-off date, December 31, 2025). This search
captured papers on the roles of osteopontin in development and
organ physiology, as well as in various predisposing conditions. In
total, six hits were found not to be pertinent to pancreatic
function or to osteopontin (Fig.
S2).
Analysis
The mined articles were downloaded, summarized and
organized according to the aspect covered in the original studies.
They include the history and molecular characteristics of
osteopontin, pancreas development and function (development,
expression, function), tumor tissue (expression, marker
combination, tumor progression, survival), tumor blood (protein
biomarker, RNA biomarker, marker combination, progression,
post-treatment recovery), tumor immunology, tumor models
(metastasis, tropism, stemness, angiogenesis, chemoresistance),
predisposition and early lesions (prediagnosis and premalignancy,
smoking, pancreatitis, kidney disease), and diabetes and obesity
(prediabetes, type 1 diabetes, type 2 diabetes, cancer connection).
Multiple references are relevant to several subsections of the
present systematic review and are therefore cited accordingly.
Data extraction
All reviewed publications were tabulated in the
chronological order of their publication. In a separate table,
patient-derived results were organized with more detail. They are
arranged according to the cancer feature (or other disease feature)
addressed. Signal transduction connections are depicted in the
figures.
Results
History and molecular characteristics
of osteopontin
Studying various mammalian tumor cell lines in 1979,
Senger et al (6,7) described transformation-specific
secreted phosphoproteins, including one at a size of 58 kDa. The
cloning of osteopontin (also known as OPN, secreted phosphoprotein
1, SPP1 or bone sialoprotein) was reported independently in
1986(8). With the protein sequence
at hand, the description in 1988/89, by the groups of Denhardt
[Craig et al (9)] and Senger
et al (10,11), that the phosphoglycoprotein
osteopontin is prominently secreted by cancer cells, has prompted
much ensuing research into the roles played by this molecule in
tumor progression (to date, ~3,040 publications on ‘osteopontin AND
cancer’ in PubMed). Mechanistic insight was gained by the
identification of the receptor integrin αVβ3
(12) and the discovery that a
metastasis-associated splice variant of CD44 also serves as a
receptor (13). Additional integrins
have been identified as osteopontin binding partners. A new angle
arose in 2006, with the report that osteopontin splice variants are
selectively produced by malignant cells, but not by untransformed
cells (14). It has impacted the
study of osteopontin in pancreatic and other types of cancer.
The expression and distribution of osteopontin in
the luminal epithelial surfaces of tissues, including the pancreas,
was published in 1992(15). Initial
cursory mention of this protein in pancreatic cancer dates back to
a 1997 review on neoplastic cell adhesion to CD44(16). Since then, a literature has evolved
that analyzes osteopontin and its roles in cancers and other
conditions afflicting the pancreas (Table SI).
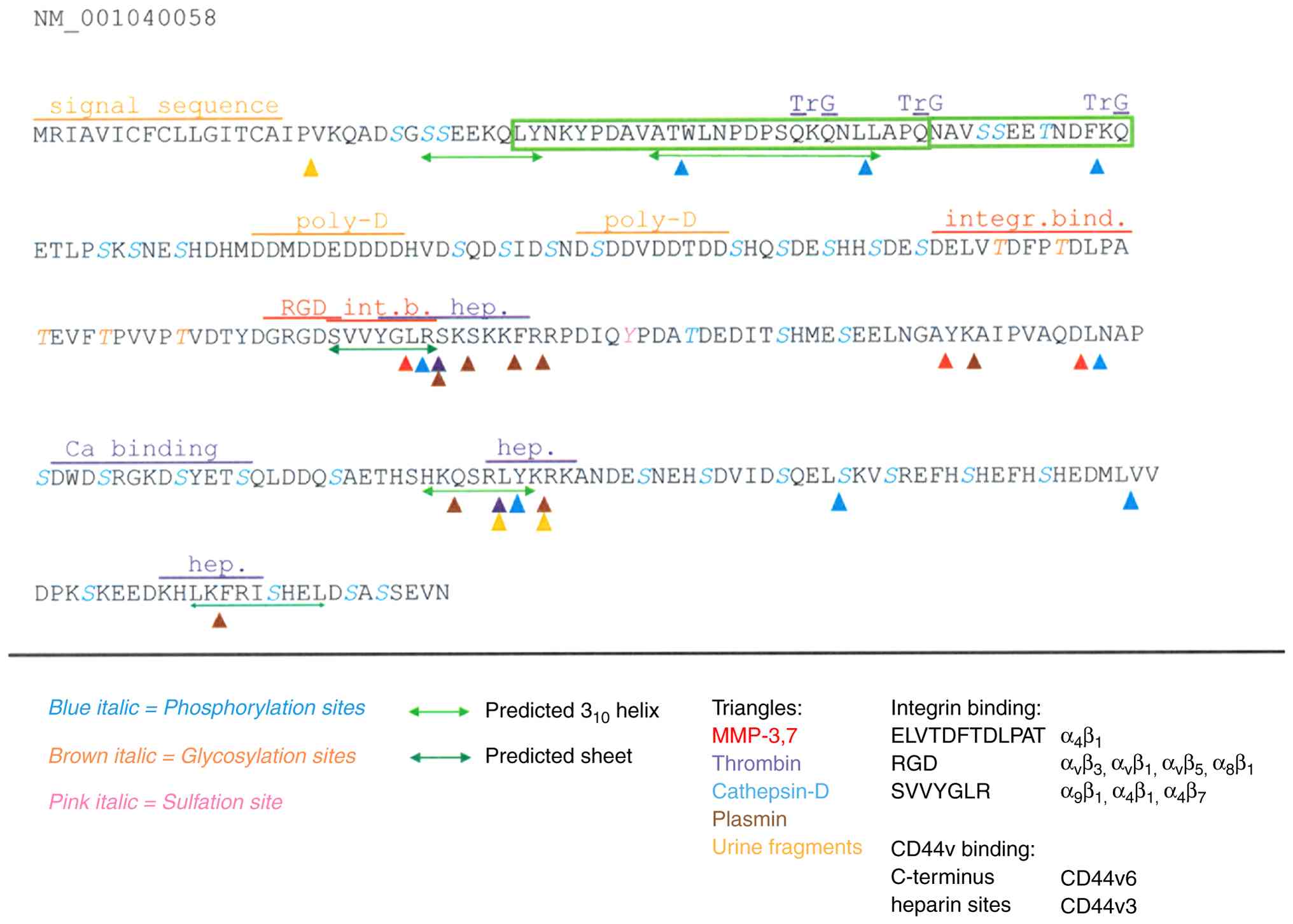
The osteopontin protein (Fig. 1) is characterized as very acidic,
largely unstructured, highly phosphorylated and glycosylated, as
well as possessing abundant calcium-binding capacity. Osteopontin
can engage several cell surface receptors, prominently integrins
and CD44v (a splice variant of CD44, entailing variant exons 3-6).
Thrombin cleavage (10) separates
the integrin-engaging N-terminus from the heparin- and
CD44v-ligating C-terminus (17).
There are two heparin binding sites (18) likely involved in the interaction with
CD44v, but also modulating the unfolding of a core element in the
protein (19,20). In the bone, osteopontin attaches to
hydroxyapatite (21). Furthermore,
the protein has been reported to harbor an interaction domain with
factor H (22), a site for
engagement of neuropilin-1(23), and
a domain to bind to inducible t-cell costimulator ligand (ICOSL)
(24). Whereas the physiologic role
of osteopontin lies in tissue remodeling and immune activation
(systemically), as well as the regulation of calcification (in
bone, breast and kidneys) (25-29),
in cancer, it mediates the survival of deadherent cells (30), as well as migration and invasion
(17) during the process of tumor
dissemination.
Humans generate multiple gene products of
osteopontin through alternative splicing (14,31) or
through an alternative transcription start site (32). With the exception of the alternative
transcriptional start site (which produces an intracellular form),
all osteopontin variants are secreted. Tumor progression genes are
aberrantly expressed or spliced in malignancies (4,5), and
osteopontin overexpression and/or splicing has been associated with
a number of aggressive tumors (33-35).
While alternative splicing of the osteopontin mRNA is formally
possible in several species and cell types, it has only been
reliably shown to arise in human transformed cells (36). Specifically, the role for the
metastasis-promoting variant OPN-c in cancer progression has been
elucidated as a gain-of-function at the splice junction that
ligates SLC7A11, activates peroxides, and facilitates mitochondrial
biogenesis (30).
Osteopontin is encoded by the spp1 gene, located on
chromosome 4 in locus 4q13.22, which has 7 exons, the first of
which is silent. The gene belongs to the SIBLING family (37), which stands for ‘small integrin
binding ligand N-linked glycoproteins’ (38).
Pancreas development and function.
Development
Matricellular proteins mediate tissue morphogenesis
and homeostasis by modulating cell-matrix and cell-cell
interactions. Osteopontin is a marker of undifferentiated
pancreatic precursors and ductal tissues. The specific, dynamic
profile of its expression in embryonic pancreatic tissues reflects
its participation in processes involving cell migration or
cell-cell interactions (39). In the
maintenance of duct cell identity, multiple cellular subpopulations
retain progenitor capacity. An epithelial-mesenchymal transitory
axis may arise in three duct subpopulations. Osteopontin marks a
cell type enriched in progenitor capacity. It serves a regulator of
the epithelial-mesenchymal transitory fate decision, and it is
required for differentiation past progenitor function and for
maintenance of duct function as reflected in carbonic anhydrase
activity. In its presence, the levels of markers of mature duct
cells, including HNF1B, SOX9 and KRT19, are increased. In its
absence, there is an increase in markers associated with
epithelial-mesenchymal transition, including VIM, ZEB1, TWIST1 and
MMP2(40). However,
osteopontin-deficient pancreata have not exhibited obvious
alterations in the morphology or differentiation of these tissues
(39).
Selenium forms part of glutathione peroxidase, which
assumes complex roles in metabolic syndrome. Offspring with
metabolic syndrome had a lower body weight, with males having a
lower body mass index and growth indicators in serum. Females, in
particular, had lower levels of serum insulin. All offspring
presented a repletion of selenium in pancreas, kidneys, and thyroid
as well as depletion in heart and muscle. Serum osteopontin was not
altered by either sex or high-fructose diet (41).
Expression. In healthy or tumor-surrounding
untransformed pancreatic tissue, osteopontin immunoreactivity has
been variably described. An in situ hybridization analysis
did not find detectable expression in healthy tissue (42); this was also the case by histology in
normal pancreatic glandular epithelia (43). Healthy pancreatic tissue samples
displayed a range from no staining (20%), via moderate staining, to
high staining (~10%) (44). In
another study, however, almost 60% of the healthy controls were
reported to express osteopontin (45).
Immunohistochemical staining for osteopontin was
positive for the epithelium lining in large and small pancreatic
ducts (15,46) or localized in ductal connective
tissue (47). A diffuse cytoplasmic
staining pattern according to immunohistochemistry was present in
5% of the ductal cells, with either occasional expression in islets
(48) or expression in all islet
cells (46). Osteopontin is
localized, to a varying degree, in connective tissue around acini
(47). A diffuse cytoplasmic
staining pattern according to immunohistochemistry was present in
30% of the acinar cells (48). By
immunofluorescence, the central areas of acini were exclusively
stained (49). No cells demonstrated
nuclear staining (44).
The discrepancies in the reported
immunohistochemistry staining may be a consequence of the rich
variations in posttranslational modifications of osteopontin
(including phosphorylation, glycosylation, multiple sites for
proteolytic cleavage, binding to calcium, heparin, or hyaluronate).
Due to these structural adaptations, the choice of the antibody to
be utilized for the staining can have a major impact on the results
[as was previously published for ELISA applications (50)]. Since the osteopontin field has grown
(currently almost 13,800 publications on this molecule in PubMed),
so has the number of commercially available antibodies (~1,120
antibodies to the human protein in Biocompare). Thorough
standardizations would be a requirement for diagnostic pathology
usage, but have not been conducted yet.
Function. In β-cells, osteopontin is
protective against both cytotoxicity and hyperglycemia (51-53).
Insulin secretion is impaired with increasing age, suggesting that
aging induces specific transcriptional changes in human islets. A
total of 346 genes that co-vary with age included increased
transcription of genes linked to senescence and downregulation in
several aspects of the cell cycle machinery. Also correlating with
age is an upregulation in osteopontin expression (51).
The maintenance of osteopontin expression in the
pancreatic tissues of adults argues for a function of this protein
in injury and pathologic responses (39). Pancreatic α-cells produce
osteopontin, which facilitates insulin release from stressed
β-cells. The protective stress response extends to metabolism
(52).
i) The incretin hormone glucose-dependent
insulinotropic polypeptide (GIP) promotes pancreatic β-cell
function by potentiating insulin secretion and β-cell
proliferation. GIP stimulated osteopontin mRNA and protein
expression, thus exerting effects on β-cell survival in islets
(proliferation by insulin-secreting cells, preservation of
functional β-cell mass), as well as the regulation of adipocyte
metabolism in fat tissue. Osteopontin expression is lower in
carriers of the A allele in the receptor GIPR (site rs10423928).
The effect is specific for GIP, as GLP-1 has no impact on
osteopontin expression, regardless of the glucose concentration
(53).
ii) The transmembrane receptor sortilin-related
VPS10-domain containing receptor 2 (SORCS2) in the pancreas is
predominantly expressed in islet α-cells. Its activity safeguards
insulin granule formation and release from glucose-stressed
β-cells. The loss of SORCS2 expression coincided with inability of
the α-cells to produce osteopontin. Consistently, β-cells in
SORCS2-deficient islets exhibited gene expression patterns
indicative of aggravated cell stress, exhibited defects in insulin
granule maturation, and had a blunted glucose response (52).
Tumor tissue. Expression
Rather consistently, immunohistochemistry
evaluations have found ~70% osteopontin expression in pancreatic
cancer (Table SII). In a comparison
between undifferentiated carcinoma and ductal adenocarcinoma, both
had close to 70% positivity for osteopontin (54). Osteopontin staining was present in
60% (48) or 74% (45) of primary adenocarcinomas. Osteopontin
was strongly expressed in 70% of adenocarcinoma tissues, but only
in ~30% of surrounding healthy tissue (55). The staining intensity in the tumors
displayed a range, being absent in ~35%, weak to moderate in 50%
and high in 15% (44).
Immunofluorescence analysis of cancer tissue from patients with
invasive ductal adenocarcinoma, the majority of whom were smokers,
revealed elevated amounts of osteopontin in the malignant ducts and
the surrounding pancreatic acini (56).
Stromal osteopontin was present in >90% of
cancers, but with large variations in intensity and staining
distribution (47). A substantial
portion of the surrounding fibroblasts displayed staining (44). Immunofluorescence revealed weak
intracellular staining in all cancer cells, while stroma stained
positive but even weaker than the cancer cells (49).
Consistent with the secretion of osteopontin via the
Golgi organ, its subcellular localization in histochemistry is
predominantly cytoplasmic. Where present, benign and malignant
pancreatic ductal cells displayed cytoplasmic and luminal staining.
The expression in adenocarcinoma cells was located mostly in the
cytoplasm (45). A substantial
portion of the surrounding fibroblasts in the stroma presented a
cytoplasmic staining pattern as well (44).
An analysis of gene expression profiles in Oncomine
identified the upregulation of osteopontin in pancreatic cancer as
not reaching significance. However, in a separate evaluation of
ductal adenocarcinoma and intraepithelial neoplasia over healthy
tissue, each neoplasia was associated with significant
overexpression (57). Using RT-qPCR,
there was a 2.2-fold increase in osteopontin mRNA in adenocarcinoma
and a 1.6-fold increase in chronic pancreatitis samples,
respectively, compared to healthy pancreatic tissues (48). In patients with adenocarcinoma,
osteopontin was expressed in 43% with variable contributions by the
splice variants OPN-a, OPN-b and OPN-c (58). The variant form OPN-c was present in
87% of invasive ductal adenocarcinoma lesions, of whom 73% were
smokers. The levels of OPN-c correlated well with higher expression
levels of total osteopontin in the tissue and serum from these
patients (59,60). The splice variants have great marker
potential, as osteopontin splice forms were present only in ~15% of
tumor-surrounding healthy specimens (58). However, in situ hybridization
did not find detectable expression in the adenocarcinoma cells
(42), which could indicate that the
technique may lack sensitivity compared to PCR-based
approaches.
Osteopontin is also associated with less common
types of pancreatic cancers.
i) Ampullary adenocarcinoma is an aggressive cancer
with poor prognosis, which can be difficult to distinguish from
ampullary adenoma prior to resection. Osteopontin mRNA is
substantially elevated in this cancer. Its measurement may aid in
the early detection and differential diagnosis of patients with
periampullary lesions (61). By
in situ hybridization, tissue osteopontin was detectable
more strongly in ampullary cancer than in pancreatitis or in
healthy tissue (62). Osteopontin
expression in the cancer cells was not associated with prognosis;
however, the expression of osteopontin and location of
tumor-associated macrophages in bulky ampullary cancer predicted
recurrence (63).
ii) Osteoclast-like giant cell tumors are rare
neoplasms of the pancreas and mostly associated with ductal
adenocarcinomas. In the rare case of an osteoclast-like giant cell
tumor associated with mucinous cystadenocarcinoma, osteopontin was
expressed in the osteoclast-like giant cells but not in the
mononuclear tumor cells (64).
Marker combination. Since osteopontin had
been corroborated as a pancreatic cancer marker, the utility of its
association with other markers has been studied (Table SIII).
i) Osteopontin and the transcription factor FOXM1
were significantly upregulated in pancreatic cancer tissues and
were linked to poor clinical outcome (65). In tumor tissues, there was no
correlation of the RNA level between KRAS and TP53 mutational
status and osteopontin expression (58). In undifferentiated cancers and
adenocarcinomas, there was no correlation between osteopontin
expression and E-cadherin or β-catenin expression (54).
ii) Multiple reports have focused on ductal
adenocarcinoma lesions: The osteopontin staining levels correlated
with MMP-9 and vascular endothelial growth factor (VEGF) (66), correlated and colocalized with the
chemokine CCL-2 (MCP-1) in transformed cells and in the malignant
ducts (67), and, together with
immunostaining for LIM homeobox transcription factor 1α (LMX1A),
were associated with advanced nuclear grades and advanced stages
(while both markers were undetectable in healthy pancreatic
glandular epithelia) (43). Whereas
osteopontin and RAN mRNA levels highly correlated with each other
in tumor cells, adjacent non-malignant and benign pancreatic
tissues, the levels of either did not correlate with venous
lymphatic invasion, diabetes, obesity, T stage, body mass index, or
survival (68,69).
iii) Pancreatic cystic lesions may be benign,
requiring observation, or cancerous, requiring surgery. Lower
osteopontin, sTIE-2 (soluble form of the receptor for
angiopoietins) and leptin levels in cystic fluid were associated
with cancer (70).
A personalized adenocarcinoma-derived organoid chip
with functional endothelial barrier (to simulate the vascular
permeation and tumor interactions) was developed for biomarker
detection and functional drug sensitivity testing. Tumor-specific
biomarkers, including osteopontin, CA-19.9, TIMP-1, macrophage
inhibitory cytokine-1 (MIC-1), the adhesion receptor ICAM-1 and
soluble AXL (a receptor tyrosine kinase) were consistently present
in the chip outflows (71).
Tumor progression. Osteopontin staining was
present in 60% of primary adenocarcinomas and in 70% of lymph node
and liver metastases (48). The
small GTPase RAN drives pancreatic cancer metastasis by modulating
androgen receptor (AR) expression. In metastatic lymph node
tissues, there were elevated levels of RAN, osteopontin and AR
(69).
The association of total osteopontin with cancer
progression has been very variably reported as being absent, being
reflective of good prognosis, or being an indicator of poor
outcome. It is conceivable that the inconsistencies were due to the
lack of distinction between tumor-derived osteopontin splice
variants, which facilitate progression, and osteopontin produced by
the host in response to tissue damage, which supports remodeling
and healing. The measurement of cancer-associated osteopontin
splice forms may be a more accurate tool for prognostication of
progression than pan-osteopontin.
i) In premalignant intraductal papillary mucinous
neoplasms, osteopontin was a biomarker for the surveillance of
carcinogenic progression (72).
ii) Osteopontin expression and the location of
tumor-associated macrophages in bulky ampullary cancer predicted
recurrence (63). While being
undetectable in normal pancreatic glandular epithelia, osteopontin
immunostaining was associated with advanced nuclear grades and
advanced stages of ductal adenocarcinomas (43). In fine needle aspirations, abundance
of the splice variants OPN-b and OPN-c indicated a poor prognosis.
While OPN-b was a predictor for survival, OPN-c was associated with
metastatic disease (73).
iii) A low stromal deposition of osteopontin was
associated with a poor survival, independently of established
prognostic factors (47).
iv) In ductal adenocarcinoma, osteopontin staining
was not associated with grade and stage (44). In stage-oriented pancreas cancer
tissue arrays, high and preferentially cytoplasmic osteopontin
staining in 80% of carcinomas did not correlate with tumor stage
(74). Osteopontin mRNA levels in
ductal adenocarcinoma, adjacent non-malignant and benign pancreatic
tissues highly correlated with each other, but did not correlate
with venous lymphatic invasion, diabetes, obesity, T stage, body
mass index or survival (68).
v) The presence of osteopontin in adenocarcinoma may
have a protective effect, independently of tumor stage (45).
The infrequent presence of psammoma bodies in
pancreatic cancer may be associated with a less aggressive tumor
phenotype, potentially leading to a better prognosis. In very rare
cases of ductal adenocarcinomas, focal dystrophic calcification may
arise. In one case, numerous psammoma bodies, scattered throughout
the tumor, were positive for osteopontin. Due to its high
calcium-binding capacity, osteopontin can play a role in the
development of such psammoma bodies. Calcifications in imaging may
be early indicators of cancer (75).
In a case of intraductal tubulopapillary neoplasm with severe
calcification, psammoma body-type and non-psammoma body-type
calcifications stained positively for osteopontin, the macrophages
were weakly positive, and the tumor cells were also stained
strongly (76).
Survival. While (as with progression)
published studies on survival are not consistent, there is a
preponderance of findings that osteopontin, particularly its splice
variants, prognosticates a poor outcome.
i) In bulky ampullary cancer, osteopontin-positive
infiltrating tumor-associated macrophages and the expression of
macrophage migration inhibitory factor (MIF) have been found to be
associated with the worst disease-specific survival (63). In fine needle aspirations, the
abundance of the splice variants OPN-b and OPN-c indicated a poor
prognosis, with OPN-b being a predictor for survival. In
comparisons between long and short postsurgical survival of
adenocarcinoma, RNA for total osteopontin, as well as the splice
variants OPN-b and OPN-c, were more frequently expressed in
short-term survivors (58,73).
ii) Although osteopontin and RAN mRNA levels in
ductal adenocarcinoma highly correlated with each other, the levels
of either did not correlate with invasion or survival (68). In an exploration of seromarker levels
for outcomes in locally advanced cancer, patients who received
chemotherapy and stereotactic body radiation therapy showed no
association of osteopontin with improved survival (77).
iii) The median and 2-year overall survival was
longer when osteopontin was expressed in pancreatic cancer.
Osteopontin expression and T stage were independent predictors of
overall survival, while other histopathologic factors (tumor grade,
tumor size, nodal status) were not (45). A low stromal deposition of
osteopontin correlated with a poor survival, independently of
established prognostic factors for pancreatic cancer (47).
Tumor blood. Protein biomarker
The diagnostic and prognostic value of blood
osteopontin (Table SIV) has been
corroborated by various studies (78). Overall, diverse reports have been in
good agreement.
i) In profiling plasma biomarkers in pancreatic
cancer, osteopontin was detected at twice the level compared to
healthy controls (79) and higher in
patients with ductal adenocarcinoma compared with chronic
pancreatitis patients, type 2 diabetes and healthy controls
(80). Osteopontin was substantially
upregulated in late-stage ductal adenocarcinoma (81); however, alone, it was not a marker
for obesity or progenitor cell trafficking (82).
ii) Gastro-entero-pancreatic neuroendocrine tumors
are highly vascularized neoplasms. While the plasma concentrations
of TIE-2 and CgA were higher in patients (2 of 16 pancreatic) as
compared to healthy controls, the osteopontin values were not
significantly elevated (possibly due to a lack of power) (83).
iii) In a previous meta-analysis of serum levels for
the diagnosis of pancreatic cancer, osteopontin was higher in
patients than in healthy controls. Ethnicity-stratified analysis
indicated that this elevation occurred among both Caucasians and
Asians (84). Serum osteopontin
levels were elevated in patients with resectable adenocarcinoma
compared to healthy individuals (42). In an expanded comparison of
preoperative patients with resectable adenocarcinoma, compared to
patients with chronic pancreatitis and healthy controls,
osteopontin distinguished pancreatic cancer vs. chronic
pancreatitis or healthy controls (85). Osteopontin performed very highly in
distinguishing pancreatic cancer cases from healthy people. The
sensitivity dropped precipitously when tested on an expanded set of
controls, including patients with pancreatitis (86). ELISA of serum from patients with
adenocarcinoma, patients with chronic pancreatitis and healthy
donors revealed a 1.6-fold increase in osteopontin serum levels in
patients with tumors and a 1.9-fold increase in patients with
chronic pancreatitis (48).
Depending on the cut-off value, the sensitivity in these studies
varied over some range, but the specificity was always >90%.
iv) Mean pre-operative serum osteopontin levels in
patients with ampullary neoplasms (vs. patients with other
periampullary diseases and healthy controls) were elevated
(61).
RNA biomarker. By reverse transcription-PCR
from whole blood, OPN-b and OPN-c were elevated in patients with
pancreatic cancer as compared to healthy controls (87). This was consistent with protein
measurements in sera. No OPN-b or OPN-c was detected in healthy
sera. In patients with pancreatic lesions (comprising ductal
adenocarcinoma and IPMN), OPN-b was expressed in almost 50%, OPN-c
in 35%, and both in 5% (88).
Gene expression alterations indicative of pancreatic
cancer can be detected by profiling the RNA of pancreatic juice. In
cancer patients, it contained increased levels of osteopontin, as
well as IL-8, interferon-induced transmembrane protein 1 (IFITM1),
fibrinogen, the chemokine CXCR4, decay-accelerating factor (DAF or
CD55) and nicotinamide N-methyltransferase (NNMT) (89).
Marker combination. Information on markers is
presented in Table SIII.
Osteopontin shares characteristics of adipokines and holds the
promise of being complementary to the glycoprotein tumor marker
CA19-9 as an early clinical diagnostic marker (38). Its combination with CA19-9 improved
differentiation over either marker alone (80). In ductal adenocarcinoma, there was a
potential benefit of using osteopontin, CA19-9, and the
metalloproteinase inhibitor TIMP-1 in a panel (90). A 6-plex immunoassay, including
osteopontin, analyzed early- and late-stage adenocarcinoma vs.
intraductal papillary mucinous neoplasms (IPMN), pancreatitis and
healthy controls, osteopontin outperformed CA19-9 in separating
IPMN from chronic pancreatitis (91). In pre- and post-surgical samples, a
postoperative increase in plasma osteopontin raised the hazard for
poor survival. Carcinoembryonic antigen (CEA) levels correlated
with those of osteopontin (49).
Strong correlations with osteopontin existed for the cancer markers
CEA, C-reactive protein (CRP) and CA72-4. Osteopontin levels also
positively correlated with the liver function readouts bilirubin,
AST, GGT and ALP. A negative correlation was present between
osteopontin levels and albumin and HDL-cholesterol. Levels of
cholesterol, LDL, triglycerides, HOMA-IR and glucose did not
correlate with osteopontin concentrations (80). MIF and osteopontin performed very
highly in distinguishing pancreatic cancer cases from healthy
controls. The sensitivity dropped when the set of controls was
expanded to include patients with pancreatitis (86). In a conflicting report, an ELISA
comparison of preoperative serum from patients with resectable
adenocarcinoma, as well as sera from patients with chronic
pancreatitis and healthy controls, osteopontin did not provide
additional diagnostic power to the independent predictors of
diagnosis, MIC-1 and CA19-9(85).
Progression. Previously, using ELISA, a
post-operative increase in 10 ng/ml plasma osteopontin elevated the
hazard for reduced survival (49).
Although patients with ductal adenocarcinoma in stage IV had higher
osteopontin plasma levels than patients in stage II, there was no
difference in the levels of stage III compared to stage II
(80). In a 6-plex immunoassay,
osteopontin aided the delineation of cancer from benign lesions and
healthy controls, but it did not change significantly with stage
(91).
Post-treatment recovery. Post-operative
stress was assessed in serum from patients following hepatobiliary
pancreatic cancer surgery without post-operative complications.
While the low-stress group did not exhibit significant increases in
the levels of osteopontin on post-operative periods, the medium-
and high-stress groups did, which peaked on post-operative day
3(92). Plasma osteopontin levels
were not different between pre- and post-surgical specimens, but
both were significantly elevated over controls (49). Osteopontin levels in serum samples,
taken from the same patients before and 6 days after pancreatic
resection, displayed a decrease in ~30%, an increase in ~40%, and a
smaller than 20% change in ~25% (48).
Circulating biomarkers have been correlated with
efficacy and tolerability to antiangiogenic agents, such as
sunitinib. Osteopontin was associated with shorter progression-free
survival, independently of Ki-67. Its levels remained higher after
6 months of treatment in non-responders than in responders
(93). The combination of
galunisertib, the first small-molecule TGFβ Receptor inhibitor,
with gemcitabine has resulted in the improvement of survival in
patients with unresectable pancreatic cancer. In multi-marker
analysis from patient plasma, baseline proteins that were changed
during treatment included osteopontin, amphiregulin, the cancer
marker CA15-3, cathepsin D, P-Selectin, RAGE, sortilin, cartilage
oligomeric matrix protein (COMP), eotaxin-2, N-BNP and
thrombospondin-4(94). In another
study which explored seromarker levels for associations with
outcomes in locally advanced cancer, patients who received
chemotherapy and stereotactic body radiation therapy showed no
association of osteopontin with improved survival (77).
Tumor immunology
Osteopontin is expressed in tumor-infiltrating
immune cells (Table SV).
Macrophages are prominently involved.
i) Adenocarcinoma contained strong osteopontin mRNA
signals in tumor-infiltrating macrophages in close to 60% of
specimens, while its expression was not detectable in healthy
pancreatic tissue or in the macrophages distant from the
infiltrating cancer (42).
ii) In a comparison between ductal adenocarcinoma
and undifferentiated carcinoma, osteopontin was expressed, apart
from the tumor cells, in macrophages and osteoclast-like giant
cells. There was no correlation between the number of
osteopontin-positive macrophages and tumor cells (54). Occasional peritumoral inflammatory
cells (macrophages) exhibited osteopontin positive stain (74). M2 macrophages were significantly
accumulated. In cellular communication, the osteopontin-CD44
pathway between macrophages and epithelial cells was particularly
strengthened in ductal adenocarcinoma (compared to IPMN) (72).
iii) A 5-gene immune-related signature, including
osteopontin, SNHG10, CASC19, LINC00683 and LINC00237 enabled the
development of a risk score formula to predict the overall survival
of ductal adenocarcinoma patients, as well as a nomogram, combining
risk score, N stage, and margin status. The expression level of
osteopontin, mainly in ductal cells and macrophages, was related to
prognosis and immune regulators (95).
iv) Tumor-associated macrophages promote cancer cell
proliferation and distant metastases. They often overexpress
osteopontin. Osteopontin levels and location of tumor-associated
macrophages in bulky ampullary cancer predicted recurrence.
Patients with bulky tumor, osteopontin-positive infiltrating
tumor-associated macrophages and MIF-expression had the worst
disease-specific survival (63).
The abundance of osteopontin in tumor-associated
macrophages may serve as a poor prognostic indicator.
Fibroblasts and macrophages are heterogeneous cell
populations able to enhance metastasis by ductal adenocarcinoma.
Mesenchymal stem cells can be recruited by osteopontin from either
peripheral blood or bone marrow. Tumor-localized fibroblasts may be
reprogrammed by osteopontin to become cancer-associated
fibroblasts, as may be M1 anti-tumor macrophages, which are
reprogrammed to become tumor-associated macrophages (38). Metastasis-associated fibroblast
heterogeneity in the liver is regulated by macrophages via 3
functionally distinct subpopulations, among which the generation of
pro-metastatic and immunoregulatory myofibroblastic
metastasis-associated fibroblasts critically depends on the
macrophages. This subset was induced through a STAT3-dependent
mechanism, driven by macrophage-derived progranulin and cancer
cell-secreted leukemia inhibitory factor (LIF). In a reciprocal
manner, osteopontin secreted from myofibroblastic
metastasis-associated fibroblasts promoted an immunosuppressive
macrophage phenotype, resulting in the inhibition of cytotoxic
T-cell functions (Fig. S3). The
blockade or depletion of STAT3 restored an antitumor immune
response and reduced metastases (96).
PD-L1 is expressed in pancreatic cancer cells,
myeloid-derived suppressor cells, polymorphonuclear myeloid-derived
suppressor cells and tumor-associated macrophages. Despite
cytotoxic T-lymphocyte infiltration in the tumor microenvironment,
pancreatic cancer stands out as one of the malignancies responding
poorly to immune checkpoint blocker therapy. In non-response to
treatment, epigenomic dysregulation has emerged as a mechanism of
T-cell exhaustion. Mouse pancreatic tumors have a genome-wide
increase in H3K4me3 deposition (an epigenetic modification to the
DNA packaging protein Histone H3 that indicates tri-methylation at
the 4th lysine residue of the histone H3 protein) as compared with
healthy pancreas. Upstream, WD-repeat domain 5 (WDR5) is essential
for H3K4me3-specific histone methyltransferase activity.
Downstream, osteopontin and its receptor CD44v are upregulated by
their promoter H3K4me3 deposition and are primarily expressed in
tumor cells and monocytic myeloid-derived suppressor cells.
Osteopontin may compensate PD-L1 function to promote pancreatic
cancer immune escape. Pharmacological inhibition of the epigenetic
WDR5-H3K4me3 axis is effective in suppressing pancreatic tumor
immune escape and in improving efficacy of anti-PD-1 immunotherapy
in pancreatic cancer (97).
Leukocytes can be recruited by osteopontin from
either peripheral blood or bone marrow (38). Notably, a proinflammatory immune
component was distinctly present in intraductal papillary mucinous
neoplasms, comprising CD4+ T-cells, CD8+
T-cells and B-cells. Osteopontin is a biomarker for the
surveillance of carcinogenic progression (72).
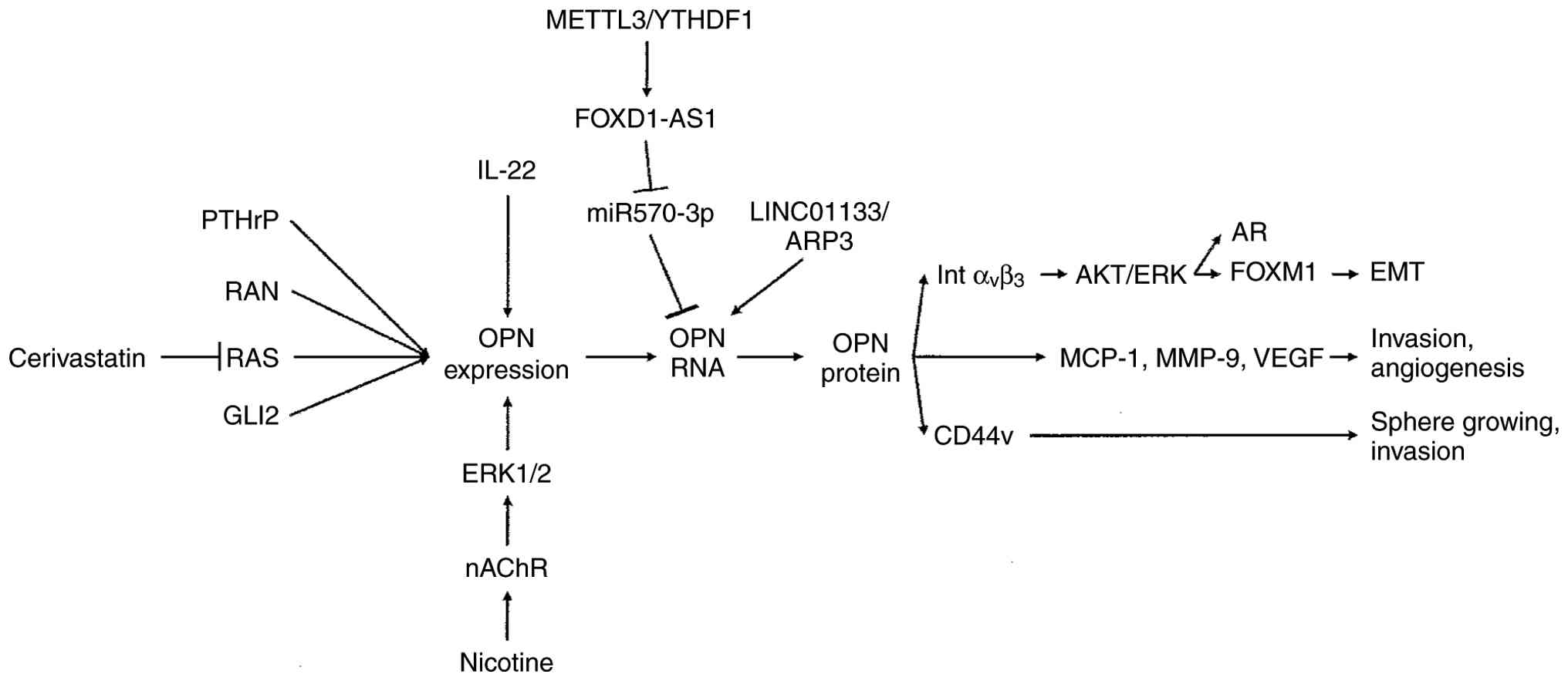
Tumor models
Osteopontin is integrated into a network of signal
transduction, which results in the activation of functional changes
that support cancer progression, including epithelial-mesenchymal
transition (mainly through integrin αVβ3),
sphere growing and invasion (mainly through CD44v), and the
secretion of soluble mediators (MCP-1, MMP-9 and VEGF) (Fig. 2).
Metastasis. Osteopontin promotes
epithelial-mesenchymal transition and cancer stem cell-like
properties.
i) Constant suppression of BMP activity by its
antagonist gremlin 1 (GREM1) is essential for maintaining the fate
of epithelial ductal adenocarcinoma cells.
ii) Osteopontin is an essential regulator of
mesenchymal cell fate.
Osteopontin, secreted from epithelial cells bound to
integrin β3 on mesenchymal ductal adenocarcinoma cells
to induce BMP2 and GREM1 expression. GREM1 inhibition of BMP
signaling was required for osteopontin expression in epithelial
cells, thereby forming an intercellular regulatory loop.
Mesenchymal and epithelial ductal adenocarcinoma cell fates are
determined by the reciprocal paracrine regulation of the soluble
factors GREM1 and osteopontin (81).
Long non-coding RNAs (lncRNAs) are involved in the tumorigenesis
and progression of ductal adenocarcinoma. Osteopontin is regulated
by some of them. LINC01133 is one of 16 hub genes that can predict
prognosis. Its overexpression compared to adjacent tissue may
promote proliferation and metastasis as well as inhibit apoptosis.
Expression of LINC01133 and osteopontin were positively correlated,
leading to enhanced epithelial-mesenchymal transition. LINC01133
bound to actin-related protein 3 (ARP3), and this complex reduced
osteopontin mRNA degradation, allowing for increased osteopontin
mRNA levels (98). Pancreatic
stellate cells (myofibroblast-like cells that are located in
exocrine regions of the pancreas) in the tumor microenvironment
contribute to invasion and metastasis. Osteopontin was highly
expressed and secreted, driven by hypoxia in a reactive oxygen
species-dependent manner. Signaling through integrin
αVβ3 was involved in the
epithelial-mesenchymal transition (65). Upon stimulation with certain
cytokines and accompanied by the increased production of
osteopontin, pancreatic stellate cells can be activated into
cancer-associated fibroblast (CAF) isoforms, such as inflammatory
CAF and myofibroblastic CAF, which participate in the desmoplastic
reaction to remodel the mesenchyme of pancreatic cancer (38).
Osteopontin increased the invasiveness of pancreatic
cancer cells, without having any impact on cell proliferation. The
effect was dependent on the receptor CD44(48). Cancer stem-like cells have increased
capacity to invade and grow as spheres in a manner that requires
CD44v6. They displayed elevated expression of markers for
metastases, including osteopontin, CD44v6, and the chemokine CXCR4,
when compared with their adherent counterparts (99). In cancer stem cells, FOXD1-AS1 was
upregulated. There, it promoted tumorigenesis and self-renewal. It
did so by upregulating osteopontin and acting as a ceRNA to sponge
miR-570-3p (88). The overexpression
of the splice variants OPN-b and OPN-c in ductal adenocarcinoma
cells increased their activity in soft-agar colony formation and
wound healing assays, induced the transcription of interleukin-6,
and reduced tumor necrosis factor-α (TNF-α), interferon-γ and
IL-10(88). One conflicting study
was unable to detect any osteopontin protein in the in the
supernatants or by western blotting in the lysates of three
commonly used ductal adenocarcinoma cell lines studied (44).
Osteopontin has long been known to be a downstream
target of RAS signaling (9,100). PTHrP is frequently amplified as
part of the KRAS amplicon in patients with pancreatic cancer. It is
highly enriched in ductal adenocarcinoma metastases, and its
upregulation drives the growth of both primary and metastatic
tumors. The osteopontin gene was a downstream effector of PTHrP,
overexpression, which enhanced migratory capacity and metastatic
ability (101). The small GTPase
RAN drives pancreatic cancer metastasis by modulating AR
expression. While RAN plays physiological roles in the regulation
of nuclear transport and microtubule spindle assembly, it also
mediates the invasive and liver-metastatic functions of
osteopontin. In RAN-silenced cells, osteopontin was necessary and
sufficient to restore AR levels via the PI3K/AKT signaling pathway.
AR reversed the inhibitory effects of RAN silencing or osteopontin
silencing on the mobility and invasion of cancer cells. However,
osteopontin did not have any significant effect on RAN
transcription (68,69). The anti-proliferative effects of
statins and hemin on pancreatic cancer cell lines do not appear to
be related to the heme oxygenase pathway. While the iron-containing
porphyrin hemin triggered reactive oxygen species-induced cell
death, the HMG-CoA reductase inhibitor cerivastatin targeted RAS
protein trafficking and affected markers of invasiveness.
Osteopontin mRNA expression was significantly suppressed at 12 h of
treatment with persisting effect of up to 48 h (102).
The zinc-finger DNA-binding transcription factors of
the GLI family are effectors of hedgehog signaling involved in cell
fate determination, proliferation and patterning in most organs
during embryo development. Sonic hedgehog (SHH)-GLI1 signaling and
osteopontin play vital roles in ductal adenocarcinoma.
Proliferation, migration and invasion were decreased, whereas
apoptosis was increased, when GLI1 or osteopontin was knocked down.
Exogenous osteopontin protein could partially reverse the effect of
both osteopontin and GLI1 knockdown. GLI1, but not SHH, was
associated with osteopontin expression, and GLI1 regulated
osteopontin production through a non-canonical pathway that did not
utilize smoothened (SMO). Hedgehog signaling promotes
proliferation, migration and invasion, but inhibits apoptosis of
pancreatic cancer cells through the upregulation of osteopontin
(55). Ductal adenocarcinoma is a
heterogeneous disease, comprised of a classical and a basal-like
subtype. These subtypes are not permanently encoded, but the
transcription factor GLI2 is a master regulator of their
inter-conversion. Its activation was sufficient to convert
classical to basal-like phenotypes. GLI2 upregulated the expression
of osteopontin, which was critical for metastatic growth and
adaptation to oncogenic KRAS ablation. Accordingly, elevated GLI2
and osteopontin levels predicted shortened overall survival. Thus,
the GLI2-osteopontin circuit is a driver of ductal adenocarcinoma
cell plasticity that establishes and maintains an aggressive
variant of this disease (103).
Tropism. From the parental pancreatic cancer
cell line HPC-4, two sublines were selected, resulting in a liver
metastatic cell line and a peritoneal disseminated cell line. The
liver metastatic cells expressed elevated levels of IL-8 and
integrin αVβ5. Among upregulated genes in
liver-tropic cells compared with peritoneum-tropic cells were
οsteopontin, VEGF and hepatocyte growth factor (HGF) (104). Osteopontin was upregulated in the
liver-metastatic cell line in comparison to the parental cells.
Inhibition by micro-RNA or antibody significantly reduced the
metastatic rate (105).
Osteopontin mRNA in a panel of human pancreatic
cancer cell lines was significantly related to their growth in the
liver of nude rats. In co-culture of cancer cells with hepatocytes,
οsteopontin mRNA was increased in the tumor cells, and its
downregulation was associated with reduced cell proliferation
(106,107). From a panel of pancreatic cancer
cell lines, some had the properties to grow in the liver of rats
and mimic liver metastasis of ductal adenocarcinoma. Among 33
associated genes and 5 signaling pathways, οsteopontin, MMP-1 and
IGF-1 stood out (108).
In the liver, macrophages regulate
metastasis-associated fibroblast heterogeneity via distinct
subpopulations. The generation of pro-metastatic and
immunoregulatory myofibroblastic metastasis-associated fibroblasts
critically depended on macrophages. Reciprocally, οsteopontin
secreted from myofibroblastic metastasis-associated fibroblasts
promoted an immunosuppressive macrophage phenotype (96).
Stemness. Cancer stem cells play a pivotal
role in the pathogenesis of human malignancies. Pancreatic cancer
stem-like cells have the capacity to grow as spheres and display
increased invasion capability. The sphere-growing population is not
only composed of cells expressing classical stem membrane markers,
but also needs CD44v6+ cells. The stem-like cells are
distinguished by upregulated expression of markers for metastasis,
including οsteopontin and CXCR4, when compared with their adherent
counterparts (99).
Cancer stem cells are considered responsible for the
recurrence of cancer. Dysregulated autophagy is highly prevalent in
many malignancies and has been implicated in cytoprotection and
tumor promotion. Induction of autophagy, mediated by
οsteopontin/NF-κB signaling, is required for the maintenance of
pancreatic cancer stem cell activity (109).
lncRNAs play a role in modulating cancer stemness
features. Specifically, in this subpopulation, the lncRNA FOXD1-AS1
is upregulated. It promotes tumorigenesis and self-renewal by
acting as a competitive endogenous RNA to sponge (bind to)
miR-570-3p, a microRNA that would otherwise suppress the expression
of osteopontin. Thus, the production of osteopontin is upregulated.
The elevated levels of FOXD1-AS1 in cancer are facilitated through
METTL3 and YTHDF1-dependent m6A methylation (110).
The interplay between tumor-microenvironment factors
and cancer stem cells plays critical roles in the aggressiveness of
pancreatic cancer.
i) CAFs promote cancer stem cell features. The
long-term treatment of pancreatic cancer cells with CAF-conditioned
medium enriched stemness, as reflected in increased tumor-sphere
formation and elevated self-renewal, as well as drug-resistance
markers. CAFs in 3-dimensional co-culture with pancreatic cancer
cells induced a substantial increase in stemness features. The
expression of CD44 and α-SMA progressively increased from the early
to late stages. Osteopontin was the top differentially
overexpressed gene, and its knockdown reduced the stemness
characteristics. There is an interplay between cancer-associated
fibroblasts and enrichment of stemness population through the
osteopontin/CD44 axis (111).
ii) Osteopontin from activated pancreatic stellate
cells interacts with integrin αVβ3 on
pancreatic cancer cells to upregulate FOXM1 expression. It promotes
epithelial-mesenchymal transition and cancer stem cell-like
properties of the cancer cells by activating the integrin
αVβ3-AKT/ERK-FOXM1 cascade in a paracrine
manner (65).
Angiogenesis. Obese visceral adipocytes
trigger aggressive transformation in ductal adenocarcinoma cells to
induce progression and accelerate angiogenesis via osteopontin
secretion. Conditioned media from these cells increased ductal
adenocarcinoma angiogenic capacity. The adipocytes directly
increased the migratory, cell growth and tube-forming capabilities
of endothelial cells in an osteopontin-dependent manner via
increased AKT phosphorylation and VEGF-A expression in both ductal
adenocarcinoma and endothelial cells. Tumor volume was increased in
obese mice compared with lean mice, whereas blocking osteopontin
inhibited obesity-accelerated tumor growth (112).
The tumor position, head and uncinate process, or
body and tail of the pancreas, are critical for surgical
strategies. Osteopontin is secreted by vessel endothelial cells in
body and tail pancreatic ductal adenocarcinoma and is associated
with increased tumor burden. Conversely, the number of tumor cells
marked by the retinoic acid carrier protein CRABP2 was lower in
body and tail pancreatic ductal adenocarcinoma and was a prognostic
marker of overall survival (113).
Chemoresistance. Osteopontin-induced
autophagy via activation of the NF-κB pathway contributes to
chemoresistance against gemcitabine in cancer cell lines. By
silencing osteopontin expression, gemcitabine conferred enhanced
cytotoxic effects. This implies that the combination of gemcitabine
with pharmacological autophagy inhibitors is a promising
therapeutic strategy (109).
A low level of FOXD1-AS1 may serve as a predictor
of 5-fluorouracil benefits in cancer patients. Pancreatic cancer
cells depleted of lncRNA FOXD1-AS1 exhibit heightened sensitivity
to 5-fluorouracil-indued cell growth inhibition and apoptosis. The
introduction of osteopontin could reverse the sensitivity of long
non-coding RNA FOXD1-AS1-knockdown cancer cells to
5-fluorouracil-induced cell apoptosis (110).
Bis-indole derivatives and substituted quinolines
exhibit anti-inflammatory activities. While both classes of drugs
are nuclear receptor 4A2 (NR4A2, NURR1) ligands, they interact
differentially with their drug target. The activation of gene
expression, including osteopontin, by bis-indole and
quinoline-derived activators of nuclear receptor 4A2 was
structure-dependent. These compounds induced osteopontin expression
at variable potency in distinct pancreatic cancer cell lines
(114).
Pancreatic stellate cells in the tumor
microenvironment contribute to chemoresistance. Osteopontin is
highly expressed and secreted by activated pancreatic stellate
cells (65).
Predisposition and early lesions.
Prediagnosis and premalignancy
Biomarkers were evaluated in prediagnostic sera
(collected before these patients had been clinically diagnosed with
pancreatic cancer) obtained from cases of pancreatic cancer
enrolled in a large prospective study. The panel of osteopontin,
CA19-9 and OPG, identified in a prior retrospective study, was not
effective (115). However, a 6-plex
immunoassay (including osteopontin) analyzed adenocarcinoma vs.
intraductal papillary mucinous neoplasm, pancreatitis and healthy
controls. It distinguished adenocarcinoma from non-cancer
conditions and outperformed CA19-9 in separating intraductal
papillary mucinous neoplasms from chronic pancreatitis (91). Intraductal papillary mucinous
neoplasms of the pancreas are bona fide precursor lesions for
ductal adenocarcinoma. According to a comparison between high-grade
IPMNs and IPMN-derived ductal adenocarcinomas, there were
heterogeneous alterations within the epithelium and the tumor
microenvironment during the progression of non-invasive dysplasia
to invasive cancer. For the epithelial cell component, there
existed acinar-ductal cells and isthmus-pit cells enriched in
precursor lesions. Osteopontin was a biomarker for the surveillance
of carcinogenesis by intraductal papillary mucinous neoplasms
(72).
Smoking. Smoking substantially increases the
risk of developing pancreatic cancer and accounts for up to 25% of
cases. In cancer tissue from patients with invasive ductal
adenocarcinoma, the majority of whom were smokers, there were
significant amounts of osteopontin in the malignant ducts and the
surrounding pancreatic acini (56).
The splice variant OPN-c was present in almost 90% of lesions, of
whom three fourth were smokers. The levels of OPN-c correlated well
with higher expression levels of total osteopontin in tissue and
serum from patients (59,60). The nicotine receptor was expressed in
the invasive and premalignant lesions without clear correlation
with smoking history (60).
Cigarette smoke and nicotine are among the leading
environmental risk factors for developing ductal adenocarcinoma. In
a previous study, exposure to cigarette smoke caused an increase in
pancreatic osteopontin that paralleled the rise of pancreatic and
plasma nicotine levels (56).
Nicotine activated the osteopontin promoter through the α7-nicotine
acetylcholine receptor (α7-nAChR) via the activation of ERK1/2 (but
not p38 or c-Jun NH2-terminal MAP kinases) in ductal adenocarcinoma
cells (56,60). While these cells expressed varying
levels of OPN-a, OPN-b, and α7-nAChR (67), their nicotine treatment selectively
induced the de novo expression of the splice variant OPN-c
(59) and increased α7-nAChR levels
(60).
Smoking and nicotine may also contribute to ductal
adenocarcinoma metastasis by inducing MMP-9 and VEGF expression.
Osteopontin plays a central role in mediating these effects via
promoter activation (66). OPN-c
induced MCP-1 promoter activity and increased its mRNA and protein.
Through this chemokine, nicotine may contribute to ductal
adenocarcinoma inflammation. MCP-1 was present in 60% of invasive
lesions, of whom two thirds were smokers (67).
Pancreatitis. During repeated or long-term
inflammation, cytokines and reactive oxygen species can cause DNA
damage, predisposing to cancer (40). Persistent pancreatitis is linked with
a substantially increased risk for pancreas cancer. Osteopontin was
elevated in pancreatitis, increasing progressively from normal to
recurrent acute pancreatitis and chronic pancreatitis (116).
Plasma osteopontin levels could serve as a highly
sensitive and specific early marker of mortality in patients with
acute pancreatitis. Its abundance was elevated, and at admission it
could uniquely predict mortality (117). Serum osteopontin levels increased
and osteocalcin levels decreased in the course of acute
pancreatitis in critically ill patients with systemic inflammation.
Osteopontin prognosticated the development of organ failure, even
after adjustment for high-sensitivity C-reactive protein and
demographic parameters, with its accuracy being similar to that of
the more complex APACHE II score (118). None of the bone biomarkers were
significantly parted between patients with alcohol-induced acute
pancreatitis and other etiologies, thus likely not reflecting a
potentially inferior nutritional status and impaired bone turnover
in subjects with alcohol-induced acute pancreatitis. Early in the
course of acute pancreatitis, osteopontin may help decide, who will
benefit from closer monitoring and aggressive therapy (118).
In fine needle aspirates, OPN-a was expressed in
50% of patients with chronic pancreatitis, OPN-b in <20%,
whereas OPN-c was totally absent (73). There was a 1.6-fold increase of
osteopontin mRNA in chronic pancreatitis samples compared to
healthy pancreatic tissues (48). In
a separate study, osteopontin levels were increased in patients
with chronic pancreatitis in comparison with those with type 2
diabetes and healthy controls. Osteopontin was lower in chronic
pancreatitis patients, type 2 diabetes and healthy controls
compared with patients with ductal adenocarcinoma. At a defined
cut-off, osteopontin differentiated chronic pancreatitis from
ductal adenocarcinoma with acceptable sensitivity and high
specificity. The combination of osteopontin with CA19-9 brought
about a better differentiation. There were no significant
differences in osteopontin concentrations among chronic
pancreatitis patients categorized according to the CP stage
(80). A 6-plex immunoassay,
including osteopontin, analyzed pancreatitis vs. adenocarcinoma,
IPMN and healthy controls. Osteopontin outperformed CA19-9 in
separating chronic pancreatitis from IPMN (91).
Cytokines are key mediators of inflammation. They
contribute to pancreatitis pathophysiology via several
mechanisms:
i) IL-22 acts primarily on epithelial and stromal
cells to protect against apoptosis, stimulate proliferation of
epithelial cells to repair injured tissues, and induce the
expression of antimicrobial peptides (including the REG family, see
type 2 diabetes below). Acinar cells respond to IL-22 with
activation of STAT3 and changes in gene transcription. IL-22
signals through a receptor comprising IL-10Rβ (CRF2-4) and IL-22R.
Whereas IL-10Rβ exhibits a broad distribution of expression, IL-22R
follows a restricted expression pattern, with the highest levels in
pancreas and detectable expression in multiple other tissues. IL-22
mediated robust inductions of expression for osteopontin and
pancreatitis-associated protein 1 (PAP1) through IL-10Rβ. It may
play a role in the pancreatic immune response (119).
ii) The transplantation of islets of Langerhans is
a potentially curative treatment for diabetes. Ensuing
immunosuppressive regimens have had some success in achieving
insulin independence. Nevertheless, transplanted islets are
exquisitely susceptible to the injurious effect of mediators
elicited by a very early host inflammatory response, which results
in islet cell dysfunction and possibly death of the transplanted
tissue. Pro-inflammatory cytokines and macrophage-derived IL-1β,
TNF-α, and nitric oxide perturb insulin secretion from β-cells and
whole islets. Osteopontin administration dose-dependently improved
islet cell-derived glucose-stimulated insulin secretion and
inhibited IL-1β-induced nitric oxide production in an
arginine-glycine-aspartate-dependent manner (the RGD motif is
important for integrin binding). The protective effect was
accompanied by inhibited transcription of iNOS and reduced
activation of NF-κB, resulting in decreased formation of the toxic
nitric oxide. Islets exposed to IL-1β revealed a naturally
occurring early upregulated osteopontin transcription, suggesting
the presence of a cross-talk between the IL-1β and osteopontin
pathways (120).
iii) While serum osteopontin was only marginally
increased in pancreatitis, gastrointestinal or muscle damage, it
was increased in liver and (to a lesser extent) kidney damage.
Biomarkers of tissue injury include osteopontin, GLDH, K18 and
ccK18 (intact and caspase-cleaved cytokeratin-18), MCSF, MCSFR, ALT
and miR-122(121).
Obesity increases the severity of acute
pancreatitis. The PPAR-γ agonist rosiglitazone serves as a
treatment choice for the disease.
i) Rosiglitazone, in mice without acute
pancreatitis fed both a low-fat diet and high-fat diet, increased
body weight and percent fat mass, with the upregulation of
adiponectin and the suppression of erythropoiesis.
ii) In mice with acute pancreatitis fed a high-fat
diet, rosiglitazone increased survival and hastened recovery from
pancreatic inflammation. This was associated with lower circulating
and pancreas-associated levels of osteopontin, IL-6, galectin-3 and
TIMP-1, particularly within one week post-acute pancreatitis.
iii) In mice with acute pancreatitis fed a low-fat
diet, rosiglitazone worsened the degree of intrapancreatic acinar
and fat necrosis as well as visceral fat saponification, without
affecting other parameters of disease severity or inflammation
(122).
Due to its high calcium-binding capability,
osteopontin maintains the homeostasis of this electrolyte. The
deregulation of osteopontin levels in disease may be associated
with calcifications.
i) Autoimmune pancreatitis has the potential for
calcification over a long-term clinical course. In surgical
specimens from autoimmune pancreatitis, chronic pancreatitis, and
healthy pancreas, as well as autoimmune pancreatic tissues from
rats, the incidence of osteopontin expression in centroacinar cells
in chronic pancreatitis with calcification and in autoimmune
pancreatitis was greater than that in healthy controls. Some cases
of autoimmune pancreatitis and chronic pancreatitis expressed the
receptor CD44 in centroacinar cells and ductal cells. The
inflammatory area and percentage of osteopontin/CD44-positive cells
increased with advancing age (123).
ii) Osteopontin is absent from the acinar or ductal
cells of the healthy pancreas. However, its RNA is detectable in
all cases of chronic calcifying pancreatitis and in over half of
cases of chronic pancreatitis without stones. The molecule may play
an important role in stone formation, and its targeting could lead
to an effective therapeutic approach to the inhibition of
calcification associated with chronic pancreatitis (124).
Kidney disease. An increased osteopontin
mRNA and protein expression correlated with proteinuria, reduced
creatinine clearance, and kidney fibrosis. In patients with chronic
kidney disease, there was a positive colocalization of the
osteopontin association signal at SPP1 with pancreas tissue.
Downstream analyses revealed colocalization of the osteopontin
association signal at KLKB1 (prekallikrein) with various plasma
proteins in trans, and with phenotypes (bone disorder, deep venous
thrombosis) (125).
The microangiopathy of diabetes may lead to renal
damage, for which the cysteine protease inhibitor cystatin C is a
biomarker. Urinary levels of cystatin C were increased in diabetic
fatty rats before the histopathological appearance of kidney
injury, and then further increased with the progression of disease.
In addition, urinary osteopontin, β2-microglobulin,
clusterin, the glutathione transferase GSTµ and kidney injury
molecule-1 (KIM-1) had the potency to detect renal damage (126).
Post-transplant diabetes mellitus is a complication
occurring following kidney transplantation, caused by increased
insulin resistance from glucocorticoids and decreased insulin
secretion from calcineurin inhibitors. Adverse outcomes include
reduced graft survival, heightened cardiovascular mortality and an
elevated risk of postoperative infections.
i) The long-term administration of the
immunosuppressant cyclosporine A causes hypoxic injury to the
kidneys with apoptotic cell death in renal tubular cells. It is
associated with renal tubular atrophy and the loss of tubular mass,
leading to progressive renal failure and irreversible renal striped
interstitial fibrosis. Mediators, including osteopontin in
conjunction with angiotensin II and TGF-β1, are involved in the
pathogenesis of chronic cyclosporine A nephropathy. Rosiglitazone
had a protective effect against pancreatic and renal injury caused
by cyclosporine A. It decreased blood glucose concentration,
increased plasma insulin concentration and preserved pancreatic
β-islet mass. The pro-inflammatory and pro-fibrotic molecules also
decreased (127).
ii) Cyclosporine a exerts a diabetogenic effect by
damaging pancreatic islet cell integrity. Continuous erythropoietin
receptor activator (CERA) mediates tissue-protective and
anti-inflammatory effects in various settings of organ injury. Its
low-dose administration did not lead to improved kidney function,
but it showed a trend toward upregulation of osteopontin,
accompanied by increased renal macrophage-infiltration, and
enhanced parenchymal TGF-β1 and IL-10. Moreover, CERA-treated
animals showed amelioration of pancreatic islet cell injury
(128).
In an evaluation of markers for renal graft
dysfunction in patients with type 1 diabetes mellitus following
kidney transplantation and simultaneous pancreas-kidney
transplantation, osteopontin was significantly elevated compared to
a control group of type 1 diabetes without diabetic nephropathy
(129).
Diabetes and obesity
Osteopontin is an islet protein and a pro-survival
factor that is protective against both cytotoxicity and
hyperglycemia, and may serve as an intrinsic feedback regulator of
nitric oxide signaling in β-cells. Its expression increases with
age. This phenomenon in aging islets could also affect the disease
course in diabetes (51,130). Pancreatic duct epithelial cells,
exposed to several combinations of glucose and insulin, displayed
accelerated proliferation under high glucose culture concentrations
and was more prominent in the presence of high insulin. The
expression of osteopontin mRNA was also increased. Furthermore, the
cells show increased oxidative stress (according to the expression
of 8-OHdG DNA) in the presence of high glucose (131).
Prediabetes. High levels of pancreatic
osteopontin mRNA and protein arose in prediabetic non-obese
pancreata, entailing co-localization of osteopontin with most of
the islet hormones. Transcripts for osteopontin were upregulated in
the pancreatic lymph nodes at the pre-diabetic stages, commencing
after the exposure of native normoglycemic non-obese predisposed
islets and β-cells to a high-dose combination of IL-1β, TNF-α and
IFN-γ. While cytokines induced the upregulation of osteopontin
promoter activity within one hour, glucose induced a dose-dependent
upregulation of promoter activity after 24 h. Long-term exposure to
cytokines or glucose reduced osteopontin promoter activity and
expression. There is a positive intrinsic mechanism that regulates
pancreatic osteopontin expression. Its temporal pattern is
inversely correlated with progression of insulitis and β-cell
destruction. Exhaustion of the local protective osteopontin system
in the islets is implicated in the associated loss of endogenous
islet protection and progression of the destructive insulitis with
resultant non-obese diabetes severity (130,132).
Hormones involved in bone remodeling and glucose
metabolism may be skewed in prediabetes. Specifically, the bone is
emerging as a key organ in the regulation of glucose metabolism.
Vitamin D has been implicated in the pathogenesis of
sub-inflammation, insulin-resistance and obesity. Its active form,
1,25-dihydroxyvitamin D, is the result of two hydroxylations that
take place in pancreas, liver, kidney, and immune cells. It is
conceivable that Vitamin D action and status in the regulation of
calcium-phosphorous balance and bone metabolism may also mirror the
interplay between other bone remodeling hormones such as
osteopontin, osteoprotegerin, CX3CL1, sclerosin, and insulin in
insulin-resistant states (133). In
prediabetes, hormones that are involved in bone remodeling may
affect glucose metabolism before overt type 2 diabetes mellitus
occurs with tissue-specific mechanisms.
i) Osteopontin expression was higher in impaired
glucose regulation compared to normal glucose tolerance and in
isolated impaired glucose tolerance compared to impaired fasting
glucose and impaired fasting glucose tolerance. Osteopontin was
positively correlated with HbA1c, PTH and fasting and 2h-plasma
glucose.
ii) Osteocalcin expression was similar in impaired
fasting glucose and normal glucose tolerance, but lower in impaired
glucose tolerance and impaired fasting glucose tolerance compared
to normal glucose tolerance.
iii) Osteoprotegerin correlated with TGD/SSPI,
endogenous glucose production and hepatic Insulin resistance index
in impaired glucose regulation.
There was no correlation between PTH and insulin
sensitivity or β-cell function parameters (134).
Type 1 diabetes. Random peptide libraries
screening with sera from patients afflicted by insulin-dependent
diabetes mellitus found five disease-specific mimotopes displayed
on phages. The screen identified οsteopontin as an autoantigen of
the somatostatin cells in islets. An antibody raised against the
peptide corroborated the results in immunohistochemistry, western
blotting and cDNA libraries screening (135). The οsteopontin protein structure in
non-obese diabetes corresponds to the a-type allele of the
οsteopontin gene. According to comparative analysis of the single
nucleotide polymorphisms between the a-type and b-type alleles, the
majority of variations are within the non-coding regions of the
gene. The implication of οsteopontin in type 1 diabetes may render
this molecule an early marker of the disease (132).
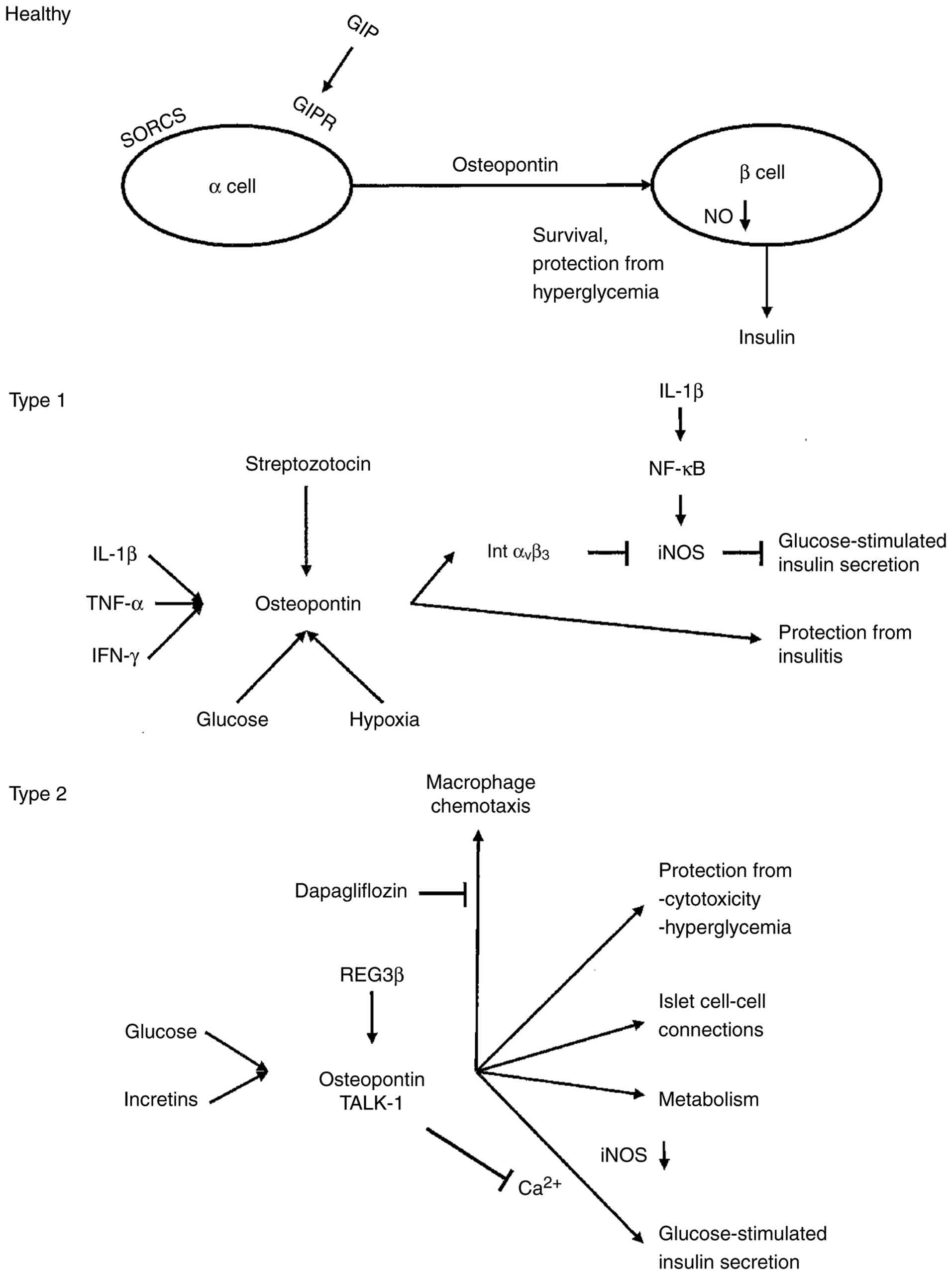
Osteopontin causes the chemotaxis of macrophages
and the downregulation of nitric oxide synthesis. It acts as a
regulator of the early islet autoimmune damage (Fig. 3), possibly by a shift in the steady
state of type 1 diabetes pathogenesis (132,135).
In the absence of osteopontin, type 1 diabetes was accelerated,
suggesting a protective role of this cytokine on the
insulin-producing cells of the pancreatic islets. However, there
were no significant differences in osteopontin levels between
patients with a duration of diabetes >3 years in comparison with
those with duration <3 years (80).
The onset of type 1 diabetes is preceded by a
pre-inflammatory stage, leading to insulitis. This manifestation
results from the selective targeted destruction of the
insulin-producing β-cells by an autoimmune phenomenon in
genetically predisposed individuals. Complete β-cell destruction
follows a massive invasion of the islets with a mixed population of
lymphocytes and macrophages (132,136).
The gene expression profiles of islets and pancreatic lymph nodes
displayed consistent changes in the islets before the appearance of
CD3+ T-cells in the islet periphery, associated with
dendritic cell attraction, lymphocyte homing, and apoptosis.
Whereas the level of the immunoregulatory cytokine IL-11 was low,
osteopontin, IL-1 and TNFSF13B, as well as genes involved in
angiogenesis were activated in the autoimmune diabetic islets
(136).
The islet protein osteopontin is differentially
regulated by streptozotocin and glucose (130,132).
In response to streptozotocin, both wild-type and osteopontin
knockout mice developed diabetes. In wild-type mice, osteopontin
serum levels were upregulated within one day, mild lymphocytic
infiltrate and apoptosis appeared in the diabetic islets, and
upregulation of both Th1 and Th2 cytokines occurred. Pancreatic
islets appeared larger in the osteopontin knockout group, no signs
of inflammation arose, and Th1 cytokines were mildly upregulated
with significant downregulation of IL-4. While the pancreatic
immune response to multiple low-dose streptozotocin diabetes was
balanced between Th1 and Th2 in wild-type animals, osteopontin
knockout mice displayed mild polarization towards Th1 responses
(137). Rats treated with a single
dose of streptozotocin showed acute and temporary upregulation of
serum osteopontin levels and pancreatic osteopontin mRNA and
protein, which within a day was localized towards the periphery of
the islets and surrounded the remaining insulin-positive cells.
Streptozotocin induced an upregulation of osteopontin promoter
activity within hours, while high glucose induced upregulation of
osteopontin promoter activity after two days. Initial osteopontin
upregulation after diabetes induction is likely due to
streptozotocin-induced toxicity, whereas maintenance of the high
osteopontin levels may be due to hyperglycemia (46).
Xenogeneic donors could potentially provide an
unlimited source of islets for the treatment of type 1 diabetes.
With donor xenoislet microencapsulation and host immunosuppression,
adult porcine islets were able to correct hyperglycemia after
intra-peritoneal transplantation in the short term. The islet
xenografts lost efficacy gradually, but at graft failure, some
viable islets remained, substantial porcine C-peptide was present
in the peritoneal graft site, and there was very little evidence of
a host immune response. Central necrosis arose in many of the
encapsulated islets after graft failure, and explanted islets
expressed endogenous markers of hypoxia (osteopontin, HIF-1α,
GLUT-1), suggesting a role for non-immunologic factors, likely
hypoxia, in graft failure. Chronic effects of non-immunologic
factors, such as hypoxia and hyperglycemia, damaged the
encapsulated islet xenografts (138).
Type 2 diabetes. Osteopontin secretion can,
in a variety of situations, help cells survive an otherwise lethal
insult (46). High glucose and
incretins simultaneously stimulated islet osteopontin secretion,
and osteopontin promoted cell metabolic activity when challenged by
high glucose (139). Patients with
type 2 diabetes suffer from insulin resistance and reduced insulin
secretion. There is a protective role of osteopontin in pancreatic
islets under diabetic conditions (Table
SVI), which may point to therapeutic targets for islet
protection in type 2 diabetes (139).
The osteopontin protein level, secretion and gene
expression were elevated in diabetic islets. External osteopontin
increased glucose-stimulated insulin secretion from diabetic, but
not from healthy islets in a Ca2+ dependent manner
(125,139). Ultra-structurally, islets reflected
weaker cell-cell connections between the islet cells in the
osteopontin knockout mice compared to wild-type mice. Although
osteopontin knockout and wild-type β-cells have the same number of
insulin granules, the former have significantly fewer docked
granules. The deletion of osteopontin results in several minor
β-cell defects that can be compensated for in a healthy system
(140).
Epigenetics is involved in the altered expression
of gene networks that underlie insulin resistance and
insufficiency. Major genes controlling β-cell differentiation and
function, such as PAX4, PDX1 and GLP1 receptor, are epigenetically
controlled. Epigenetics can cause insulin resistance through
immunomediated pro-inflammatory actions related to several factors,
such as osteopontin, NF-κB and Toll-like receptors (141).
Various mechanisms of intracellular signaling have
been elucidated, which contribute to the disease process in type 2
diabetes (Fig. 3).
i) In a previous study, osteopontin reduced the
streptozotocin-induced nitric oxide levels in the islets through an
arginine-glycine-aspartate (RGD)-dependent reduction of inducible
nitric oxide synthase (iNOS) mRNA levels, resulting in improved
glucose-stimulated insulin secretion (46).
ii) REG family proteins (small secretory proteins
that promote proliferation and differentiation) are implicated in
islet β-cell proliferation, survival and regeneration. The
expression of pancreatitis-associated protein (PAP; REG3β) is
highly induced in experimental diabetes and acute pancreatitis. It
is anti-inflammatory in the liver and pancreatic acinars. Upon
streptozotocin treatment, in contrast to wild-type littermates that
became hyperglycemic in 3 days and lost 15% of their weight,
RIP-I/REG3β mice were protected from hyperglycemia and weight loss.
Islet-protective osteopontin and the acute responsive nuclear
protein NUPR1 were induced by REG3β (142).
iii) Glucose-stimulated insulin secretion relies on
Ca2+ influx into β-cells, which is modulated by the
two-pore-domain K+ (K2P) channel, TALK-1, which
modulates glucose-stimulated insulin secretion. A gain-of-function
polymorphism in its encoding gene KCNK16 increases the risk for
developing type 2 diabetes. Intracellular osteopontin (iOPN) is
highly expressed in β-cells and is upregulated under pre-diabetic
conditions to help maintain normal β-cell function. iOPN
colocalized with and bound to TALK-1 channels in the pancreas. At
physiological voltages, iOPN activated TALK-1 transcript variant 3
channels and K+ currents, which also hyperpolarized the
resting membrane potential. Reducing β-cell iOPN decreased TALK-1
K+ currents and increased glucose-stimulated
Ca2+ influx (143). Both
osteopontin knockout and wild-type islets displayed synchronized
Ca2+ oscillations, indicative of intact β-cell
communication. Osteopontin knockout islets displayed higher
intracellular Ca2+ concentrations when stimulated with
glucose than wild-type islets and the initial dip upon elevated
glucose concentrations (which is associated with Ca2+
uptake into the endoplasmic reticulum) was significantly lower in
these islets (140).
iv) The pleiotropic effects of GIP on islet
function involve osteopontin. mRNA expression of its receptor GIPR
was decreased in human islets from carriers of the A allele of GIPR
rs10423928 (associated with impaired glucose- and GIP-stimulated
insulin secretion and a decrease in body-mass-index, lean body
mass, and waist circumference) or in patients with type 2 diabetes
(53).
The inhibition of sodium glucose cotransporter 2
(SGLT2) is a potential therapeutic strategy for treating diabetes.
The SGLT2 inhibitor dapagliflozin ameliorated hyperglycemia, β-cell
damage and albuminuria. Glomerular mesangial expansion and
interstitial fibrosis were suppressed in a dose-dependent manner.
Dapagliflozin treatment markedly decreased macrophage infiltration
and the gene expression of inflammation and oxidative stress in the
kidney. Osteopontin, TGF-α, MCP-1 and ICAM-1, were significantly
increased in diabetes, but suppressed by dapagliflozin.
Dapagliflozin ameliorates diabetic nephropathy by improving
hyperglycemia along with inhibiting inflammation and oxidative
stress (144).
Cancer connection. Diabetes and pancreatic
cancer reciprocally increase the risk of developing each other.
i) Among diseases that are complications of
pancreatic cancer, diabetes is the most common. Many cancer cases
become evident, when patients are investigated for a worsening in
glycemic control. Patients suffering from diabetes and obesity
exhibit an increased expression of osteopontin, which may act as a
critical regulator of insulin resistance and diabetes mellitus
(131).
ii) Patients with diabetes have an elevated risk of
developing pancreatic cancer (131). The levels of osteopontin were found
to be higher in patients with ductal adenocarcinoma compared with
patients with chronic type 2 diabetes (there were increased
osteopontin levels in patients with chronic pancreatitis in
comparison to those with type 2 diabetes and healthy controls)
(80). In sera from patients with
pancreatic lesions (comprising ductal adenocarcinoma and IPMN), the
presence of diabetes and/or obesity was associated with complete
disappearance of OPN-b and expression of only OPN-c. The splice
variant OPN-c was significantly associated with diabetes and
obesity (88).
Obesity is a risk and poor prognosis factor for
ductal adenocarcinoma. In those patients, osteopontin expression in
adipose tissues adjacent to adenocarcinoma tumor was higher than in
non-obese patients. Obese adipocytes triggered aggressive
transformation in ductal adenocarcinoma cells to induce progression
and accelerate angiogenesis via osteopontin secretion (112). There is potential biochemical
crosstalk between the metabolism of the adipose tissue (body mass)
and bone marrow (peripheral trafficking of bone marrow-derived stem
cells) in both healthy individuals and patients with
obesity-associated cancer. Plasma osteopontin was increased in
adenocarcinoma patients over healthy controls, but osteopontin
alone was not a marker for obesity or progenitor cell trafficking.
In adenocarcinoma patients, the osteonectin/osteopontin ratio
significantly associated with the body mass index, as well as with
the intensified systemic trafficking of mesenchymal and endothelial
progenitor cells (82).
Pancreatic stellate cells contribute to tumor
invasion, metastasis and chemoresistance. Osteopontin is highly
expressed and secreted upon their activation (65). These cells support increased
desmoplasia hyperplasia, a key mechanism by which obesity
contributes to the progression of pancreatic cancer. Upon
stimulation with TNF, IL-1/IL-2/IL-10, pancreatic stellate cells
can be activated into cancer-associated fibroblast isoforms, such
as iCAF and myCAF, which participate in the desmoplastic reaction
to remodel the mesenchyme of pancreatic cancer (38).
Discussion
Summary. Osteopontin has been understood as
a key factor in the progression of about 30 types of malignancies
(33,34). Specifically, its isoforms, entailing
the splice variants -a, -b, and -c, are critical players in
pancreatic cancer aggressiveness (35). Reported data have converged over the
years on an understanding that osteopontin overexpression and
aberrant splicing support the progression of cancers in the
pancreatis. Furthermore, due to the cytokine functions of the
full-length form OPN-a, the measurement of pan-osteopontin (total
osteopontin) lacks specificity compared to measurement of the
cancer-associated splice variants OPN-b and OPN-c. In support of
the biomarker studies, functional insight has been gained, over
several years of investigations, into the mechanisms of
osteopontin-mediated cancer progression. Osteopontin facilitates
the migration and invasion of cells (17). The full-length form and its
cancer-specific splice variants OPN-b or OPN-c synergize in
supporting the survival of deadherent cancer cells (14,30).
These functions support metastasis, tropism, stemness and
angiogenesis as outlined above.
Various precancerous conditions are capable of
producing molecules of aggressiveness early in transformation,
which reflects their cancer risk (34,145).
In the pancreas, IPMN are bona fide precursor lesions of ductal
adenocarcinoma. They often express and splice osteopontin, which
serves as a biomarker for surveillance of their carcinogenesis risk
(72). In pancreatic lesions
(comprising ductal adenocarcinoma and IPMN), OPN-b and OPN-c were
expressed in a substantial fraction of patients (88). Osteopontin aids the delineation of
cancer from benign lesions (IPMN, pancreatitis) and healthy
controls (91).
Clinical implications
While several meta-analyses have corroborated the
association of tumor-derived osteopontin and its variants with
pancreatic cancer progression (33-35)
and original reports on the abundance of osteopontin in cancerous
tissue have had good agreement, initial analyses (with still
maturing techniques) for the presence in surrounding tissue or for
prognostic value had conflicting outcomes. The incongruences in the
results by individual reports could reflect i) insufficient
sensitivity or specificity when measuring total osteopontin; ii)
structural variations in the protein combined with a lack of
standardization of antibodies to be utilized for detection; iii)
differences among pancreatic cancer subtypes; iv) tumor vs. stroma
sources of osteopontin may also generate heterogeneity. Much more
consistency has been achieved with osteopontin as a blood
biomarker. It has potential for clinical applications when combined
with organ-specific biomarkers (outlined in Table SIII).
Strong immune responses against pancreatic cancer
have been hard to achieve. Osteopontin derived from
tumor-infiltrating macrophages or in macrophage-fibroblast
interactions has been associated with overall poor prognosis.
Furthermore, despite the frequent presence of PD-L1, immune
checkpoint blockers have had little efficacy. Osteopontin may
compensate PD-L1 function to promote pancreatic cancer immune
escape. However, pancreatic cancer may not have the levels of
antigenicity required for prompting anti-tumor immunity. Whether
the suppression of osteopontin in this setting could suffice for
triggering an immune reaction to the pancreatic malignancy will
require further research.
In non-transformed tissues, only the full-length
form OPN-a is pertinent. In the healthy pancreas, osteopontin
fulfills safeguarding functions, including islet survival and
protection from hyperglycemia. However, its increased production is
associated with pancreatitis, and there it may contribute to the
inflammation.
The roles of osteopontin in diabetes are
multi-faceted. In type 1 diabetes, the protein could be an
autoantigen. However, it is also secreted during the disease and
may have an ameliorating effect on the destructive inflammation. In
type 2 diabetes, the β-cell protective functions seem to be in the
foreground. Osteopontin is an islet protein and a pro-survival
factor that is protective against both cytotoxicity and
hyperglycemia, and may serve as an intrinsic feedback regulator of
nitric oxide signaling in β-cells. Its temporal pattern of
expression is inversely correlated with progression of insulitis
and β-cell destruction.
Connections. The present systematic review
has brought to the forefront several connections of osteopontin to
other molecules and mechanisms.
Osteopontin avidly binds calcium and hydroxyapatite
(18,146,147).
This may be relevant for diseases of the pancreas.
i) Osteopontin is detectable in all cases of
chronic calcifying pancreatitis and in approximately half of cases
of chronic pancreatitis without stones. The molecule may play a
critical role in stone formation (124). Autoimmune pancreatitis has the
potential for calcification over a long-term clinical course. The
incidence of osteopontin expression in centroacinar cells in
chronic pancreatitis with calcification and in autoimmune
pancreatitis was significantly greater than that in healthy
controls (123).
ii) The ectopic mineralization of soft tissues is a
typical response to systemic imbalance of various metabolic factors
as well as tissue injury. Coxsackievirus B3 infection resulted in
significant tissue injury, particularly in the pancreas and heart,
involving substantial ectopic calcification. Infection induced
upregulation of osteopontin, SPARC and collagen type I in the
pancreas. α-lipoic acid diminished the tissue damage, subsequently
ameliorating ectopic calcification via the suppression of
osteogenic signals (148).
iii) Calcification is very rare in ductal
adenocarcinomas, but sometimes focal dystrophic calcification
arises. Psammoma bodies contain osteopontin, and their development
may be facilitated by the high calcium-binding capacity of
osteopontin (75,76).
The regulation of nitric oxide by osteopontin has
long been known (149-152):
i) Pro-inflammatory cytokines and
macrophage-derived IL-1β, TNF-α and nitric oxide perturb insulin
secretion from transformed β-cells and whole islets. Osteopontin
regulates nitric oxide production via inhibition of iNOS
transcription. It dose-dependently improved islet cell-derived,
glucose-stimulated insulin secretion and inhibited IL-1β-induced
nitric oxide production in an arginine-glycine-aspartate
(RGD)-dependent manner (120).
ii) The addition of osteopontin to diabetic islets
significantly improved their glucose-stimulated insulin secretion.
Osteopontin significantly reduced the streptozotocin-induced nitric
oxide levels in the islets through an RGD-dependent reduction of
iNOS mRNA levels (46).
The PPAR-γ agonist rosiglitazone is administered to
improve glycemic control in adults with type 2 diabetes. It is
typically prescribed as a monotherapy or in combination with
metformin or sulfonylureas. In addition, the agent may be a
treatment choice in acute pancreatitis and in post-transplant organ
damage by cyclosporine A. In mice with acute pancreatitis fed a
high-fat diet, the drug increased survival and hastened recovery
from inflammation. This was associated with significantly lower
circulating and pancreas-associated levels of osteopontin (122). The long-term administration of the
immunosuppressant cyclosporine A post-transplantation causes
hypoxic injury to the kidneys. Mediators include osteopontin.
Rosiglitazone had a protective effect against pancreatic and renal
injury caused by cyclosporine A. Pro-inflammatory and pro-fibrotic
molecules, including osteopontin, also decreased under treatment
(127).
Conclusion. The prognosis of pancreatic
cancer is poor. This is due to the fact that, to a large extent, it
is typically detected at advanced stages. Insights into its
mechanisms of progression may lead to improved early detection and
better late-stage treatments. Osteopontin variants, once
sufficiently elucidated and delineated from non-cancerous diseases
of the pancreas, are prominent candidate markers for this goal. The
cancer-specific variant forms also represent candidate drug
targets.
Supplementary Material
PubMed-listed publications by year.
The y-axis indicates the number of hits per calendar year. In the
stacked bars, the lower set (dotted) shows articles in PubMed with
the key phrase ‘pancreas cancer OR pancreatic cancer AND
osteopontin’, the upper panel (hatched) displays articles in PubMed
with the key phrase ‘osteopontin AND pancreas NOT cancer’.
PRISMA flow chart. The standard chart
for identification of studies via databases and registers is
displayed with the applicable numbers (153). Note that the reference citation
pertains to the reference list in the main manuscript.
Fibroblast-macrophage interactions in
ductal adenocarcinoma. Recruitment and reprogramming of fibroblasts
and macrophages may generate an immunosuppressive environment by
inhibiting cytotoxic T-cell functions.
Synopsis of the pertinent
literature.
Patient studies.
Cancer markers used in conjunction
with Osteopontin.
Blood biomarker osteopontin.
Osteopontin in pancreatic cancer
immune responses.
Osteopontin in diabetes.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
The author GFW conceived and designed the study,
collected all data for the systematic review, and wrote and edited
the manuscript. GFW confirms the authenticity of all the raw data.
The author has read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The author declares that he has no competing
interests.
References
|
1
|
Zhao H, Chen Q, Alam A, Cui J, Suen KC,
Soo AP, Eguchi S, Gu J and Ma D: The role of osteopontin in the
progression of solid organ tumour. Cell Death Dis.
9(356)2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chen L, Huan X, Xiao GH, Yu WH, Li TF, Gao
XD and Zhang YC: Osteopontin and its downstream carcinogenic
molecules: Regulatory mechanisms and prognostic value in cancer
progression. Neoplasma. 69:1253–1269. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Liu L, Niu K, Yang Z, Song J, Wei D, Zhang
R and Tao K: Osteopontin: An indispensable component in common
liver, pancreatic, and biliary related disease. J Cancer Res Clin
Oncol. 150(508)2024.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Weber GF and Ashkar S: Stress response
genes: The genes that make cancer metastasize. J Mol Med (Berl).
78:404–408. 2000.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Weber GF: Molecular mechanisms of
metastasis. Cancer Lett. 270:181–190. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Senger DR, Wirth DF and Hynes RO:
Transformed mammalian cells secrete specific proteins and
phosphoproteins. Cell. 16:885–893. 1979.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Senger DR, Wirth DF and Hynes RO:
Transformation-specific secreted phosophoproteins. Nature.
286:619–621. 1980.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Oldberg A, Franzen A and Heinegard D:
Cloning and sequence analysis of rat bone sialoprotein
(osteopontin) cDNA reveals an Arg-Gly-Asp cell-binding sequence.
Proc Natl Acad Sci USA. 83:8819–8823. 1986.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Craig AM, Nemir M, Mukherjee BB, Chambers
AF and Denhardt DT: Identification of the major phosphoprotein
secreted by many rodent cell lines as 2ar/osteopontin: Enhanced
expression in H-ras-transformed 3T3 cells. Biochem Biophys Res
Commun. 157:166–173. 1988.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Senger DR, Perruzzi CA, Papadopoulos A and
Tenen DG: Purification of a human milk protein closely similar to
tumor-secreted phosphoproteins and osteopontin. Biochim Biophys
Acta. 996:43–48. 1989.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Senger DR, Perruzzi CA and Papadopoulos A:
Elevated expression of secreted phosphoprotein I (osteopontin, 2ar)
as a consequence of neoplastic transformation. Anticancer Res.
9:1291–1299. 1989.PubMed/NCBI
|
|
12
|
Miyauchi A, Alvarez J, Greenfield EM, Teti
A, Grano M, Colucci S, Zambonin-Zallone A, Ross FP, Teitelbaum SL,
Cheresh D, et al: Binding of osteopontin to the osteoclast integrin
alpha v beta 3. Osteoporos Int. 3 (Suppl 1):S132–S135.
1993.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Weber GF, Ashkar S, Glimcher MJ and Cantor
H: Receptor-ligand interaction between CD44 and osteopontin
(Eta-1). Science. 271:509–512. 1996.PubMed/NCBI View Article : Google Scholar
|
|
14
|
He B, Mirza M and Weber GF: An osteopontin
splice variant induces anchorage independence in human breast
cancer cells. Oncogene. 25:2192–2202. 2006.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Brown LF, Berse B, Van de Water L,
Papadopoulos-Sergiou A, Perruzzi CA, Manseau EJ, Dvorak HF and
Senger DR: Expression and distribution of osteopontin in human
tissues: Widespread association with luminal epithelial surfaces.
Mol Biol Cell. 3:1169–1180. 1992.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rudzki Z and Jothy S: CD44 and the
adhesion of neoplastic cells. Mol Pathol. 50:57–71. 1997.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Weber GF, Zawaideh S, Hikita S, Kumar VA,
Cantor H and Ashkar S: Phosphorylation-dependent interaction of
osteopontin with its receptors regulates macrophage migration and
activation. J Leukoc Biol. 72:752–761. 2002.PubMed/NCBI
|
|
18
|
Prince CW: Secondary structure predictions
for rat osteopontin. Connect Tissue Res. 21:15–20. 1989.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kurzbach D, Schwarz TC, Platzer G, Hofler
S, Hinderberger D and Konrat R: Compensatory adaptations of
structural dynamics in an intrinsically disordered protein complex.
Angew Chem Int Ed Engl. 53:3840–3843. 2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Kurzbach D, Canet E, Flamm AG, Jhajharia
A, Weber EM, Konrat R and Bodenhausen G: Investigation of
intrinsically disordered proteins through exchange with
hyperpolarized water. Angew Chem Int Ed Engl. 56:389–392.
2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Reinholt FP, Hultenby K, Oldberg A and
Heinegard D: Osteopontin-a possible anchor of osteoclasts to bone.
Proc Natl Acad Sci USA. 87:4473–4475. 1990.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Fedarko NS, Fohr B, Robey PG, Young MF and
Fisher LW: Factor H binding to bone sialoprotein and osteopontin
enables tumor cell evasion of complement-mediated attack. J Biol
Chem. 275:16666–16672. 2000.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen Y, Gialeli C, Shen J, Duner P, Walse
B, Duelli A, Caing-Carlsson R, Blom AM, Zibert JR, Nilsson AH, et
al: Identification of an osteopontin-derived peptide that binds
neuropilin-1 and activates vascular repair responses and
angiogenesis. Pharmacol Res. 205(107259)2024.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Raineri D, Dianzani C, Cappellano G,
Maione F, Baldanzi G, Iacobucci I, Clemente N, Baldone G, Boggio E,
Gigliotti CL, et al: Osteopontin binds ICOSL promoting tumor
metastasis. Commun Biol. 3(615)2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mark MP, Butler WT, Prince CW, Finkelman
RD and Ruch JV: Developmental expression of 44-kDa bone
phosphoprotein (osteopontin) and bone gamma-carboxyglutamic acid
(Gla)-containing protein (osteocalcin) in calcifying tissues of
rat. Differentiation. 37:123–136. 1988.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Nagata T, Todescan R, Goldberg HA, Zhang Q
and Sodek J: Sulphation of secreted phosphoprotein I (SPPI,
osteopontin) is associated with mineralized tissue formation.
Biochem Biophys Res Commun. 165:234–240. 1989.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hirota S, Ito A, Nagoshi J, Takeda M,
Kurata A, Takatsuka Y, Kohri K, Nomura S and Kitamura Y: Expression
of bone matrix protein messenger ribonucleic acids in human breast
cancers. Possible involvement of osteopontin in development of
calcifying foci. Lab Invest. 72:64–69. 1995.PubMed/NCBI
|
|
28
|
Bellahcene A and Castronovo V: Increased
expression of osteonectin and osteopontin, two bone matrix
proteins, in human breast cancer. Am J Pathol. 146:95–100.
1995.PubMed/NCBI
|
|
29
|
Yasui T, Fujita K, Sasaki S, Sato M,
Sugimoto M, Hirota S, Kitamura Y, Nomura S and Kohri K: Expression
of bone matrix proteins in urolithiasis model rats. Urol Res.
27:255–261. 1999.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Fnu G and Weber GF: Osteopontin induces
mitochondrial biogenesis in deadherent cancer cells. Oncotarget.
14:957–969. 2023.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Briones-Orta MA, Avendano-Vazquez SE,
Aparicio-Bautista DI, Coombes JD, Weber GF and Syn WK: Osteopontin
splice variants and polymorphisms in cancer progression and
prognosis. Biochim Biophys Acta Rev Cancer. 1868:93–108.A.
2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Shinohara ML, Kim HJ, Kim JH, Garcia VA
and Cantor H: Alternative translation of osteopontin generates
intracellular and secreted isoforms that mediate distinct
biological activities in dendritic cells. Proc Natl Acad Sci USA.
105:7235–7239. 2008.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Weber GF, Lett GS and Haubein NC:
Osteopontin is a marker for cancer aggressiveness and patient
survival. Br J Cancer. 103:861–869. 2010.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Weber GF, Lett GS and Haubein NC:
Categorical meta-analysis of Osteopontin as a clinical cancer
marker. Oncol Rep. 25:433–441. 2011.PubMed/NCBI View Article : Google Scholar
|
|
35
|
An Y, Fnu G, Xie C and Weber GF:
Meta-analysis of osteopontin splice variants in cancer. BMC Cancer.
23(373)2023.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Weber GF: The phylogeny of
Osteopontin-analysis of the protein sequence. Int J Mol Sci.
19(2557)2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Fisher LW, Torchia DA, Fohr B, Young MF
and Fedarko NS: Flexible structures of SIBLING proteins, bone
sialoprotein, and osteopontin. Biochem Biophys Res Commun.
280:460–465. 2001.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wang Q, Wang H, Ding Y, Wan M and Xu M:
The role of adipokines in pancreatic cancer. Front Oncol.
12(926230)2022.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Kilic G, Wang J and Sosa-Pineda B:
Osteopontin is a novel marker of pancreatic ductal tissues and of
undifferentiated pancreatic precursors in mice. Dev Dyn.
235:1659–1667. 2006.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Hendley AM, Rao AA, Leonhardt L, Ashe S,
Smith JA, Giacometti S, Peng XL, Jiang H, Berrios DI, Pawlak M, et
al: Single-cell transcriptome analysis defines heterogeneity of the
murine pancreatic ductal tree. Elife. 10(e67776)2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ojeda ML, Nogales F, Muñoz Del Valle P,
Díaz-Castro J, Murillo ML and Carreras O: Metabolic syndrome and
selenium in fetal programming: Gender differences. Food Funct.
7:3031–3038. 2016.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Koopmann J, Fedarko NS, Jain A, Maitra A,
Iacobuzio-Donahue C, Rahman A, Hruban RH, Yeo CJ and Goggins M:
Evaluation of osteopontin as biomarker for pancreatic
adenocarcinoma. Cancer Epidemiol Biomarkers Prev. 13:487–491.
2004.PubMed/NCBI
|
|
43
|
Tsai WC, Lin CK, Yang YS, Chan DC, Gao HW,
Chang FN and Jin JS: The correlations of LMX1A and osteopontin
expression to the clinicopathologic stages in pancreatic
adenocarcinoma. Appl Immunohistochem Mol Morphol. 21:395–400.
2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Weber CE, Erşahin ÇH, Kuo PC and Mi Z:
Pancreatic cancer and osteopontin: The relationship remains
unclear. Pancreas. 45:e35–e36. 2016.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Collins AL, Rock J, Malhotra L, Frankel WL
and Bloomston M: Osteopontin expression is associated with improved
survival in patients with pancreatic adenocarcinoma. Ann Surg
Oncol. 19:2673–2678. 2012.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Katakam AK, Chipitsyna G, Gong Q, Vancha
AR, Gabbeta J and Arafat HA: Streptozotocin (STZ) mediates acute
upregulation of serum and pancreatic osteopontin (OPN): A novel
islet-protective effect of OPN through inhibition of STZ-induced
nitric oxide production. J Endocrinol. 187:237–247. 2005.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Franklin O, Billing O, Öhlund D, Berglund
A, Herdenberg C, Wang W, Hellman U and Sund M: Novel prognostic
markers within the CD44-stromal ligand network in pancreatic
cancer. J Pathol Clin Res. 5:130–141. 2019.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Kolb A, Kleeff J, Guweidhi A, Esposito I,
Giese NA, Adwan H, Giese T, Büchler MW, Berger MR and Friess H:
Osteopontin influences the invasiveness of pancreatic cancer cells
and is increased in neoplastic and inflammatory conditions. Cancer
Biol Ther. 4:740–746. 2005.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Franklin O, Öhlund D, Lundin C, Öman M,
Naredi P, Wang W and Sund M: Combining conventional and
stroma-derived tumour markers in pancreatic ductal adenocarcinoma.
Cancer Biomark. 15:1–10. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Anborgh PH, Wilson SM, Tuck AB, Winquist
E, Schmidt N, Hart R, Kon S, Maeda M, Uede T, Stitt LW and Chambers
AF: New dual monoclonal ELISA for measuring plasma osteopontin as a
biomarker associated with survival in prostate cancer: Clinical
validation and comparison of multiple ELISAs. Clin Chem.
55:895–903. 2009.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Seiron P, Stenwall A, Hedin A, Granlund L,
Esguerra JLS, Volkov P, Renström E, Korsgren O, Lundberg M and Skog
O: Transcriptional analysis of islets of Langerhans from organ
donors of different ages. PLoS One. 16(e0247888)2021.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Kalnytska O, Qvist P, Kunz S, Conrad T,
Willnow TE and Schmidt V: SORCS2 activity in pancreatic α-cells
safeguards insulin granule formation and release from
glucose-stressed β-cells. iScience. 27(108725)2024.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Lyssenko V, Eliasson L, Kotova O, Pilgaard
K, Wierup N, Salehi A, Wendt A, Jonsson A, De Marinis YZ, Berglund
LM, et al: Pleiotropic effects of GIP on islet function involve
osteopontin. Diabetes. 60:2424–2433. 2011.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Sedivy R, Peters K and Klöppel G:
Osteopontin expression in ductal adenocarcinomas and
undifferentiated carcinomas of the pancreas. Virchows Arch.
446:41–45. 2005.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Wu W, Yang H, Wang Z, Zhang Z, Lu X, Yang
W, Xu X, Jiang Y, Li Y, Fan X, et al: A Noncanonical hedgehog
signaling exerts a Tumor-promoting effect on pancreatic cancer
cells via induction of osteopontin expression. Cancer Biother
Radiopharm: Dec 31, 2021 doi: 10.1089/cbr.2021.0317 (Epub ahead of
print).
|
|
56
|
Chipitsyna G, Gong Q, Anandanadesan R,
Alnajar A, Batra SK, Wittel UA, Cullen DM, Akhter MP, Denhardt DT,
Yeo CJ and Arafat HA: Induction of osteopontin expression by
nicotine and cigarette smoke in the pancreas and pancreatic ductal
adenocarcinoma cells. Int J Cancer. 125:276–285. 2009.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Tu Y, Chen C and Fan G: Association
between the expression of secreted phosphoprotein-related genes and
prognosis of human cancer. BMC Cancer. 19(1230)2019.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Fiorino S, Visani M, Masetti M, Acquaviva
G, Tallini G, De Leo A, Fornelli A, Ragazzi M, Vasuri F, Grifoni D,
et al: Periostin, tenascin, osteopontin isoforms in long- and
non-long survival patients with pancreatic cancer: A pilot study.
Mol Biol Rep. 47:8235–8241. 2020.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Sullivan J, Blair L, Alnajar A, Aziz T, Ng
CY, Chipitsyna G, Gong Q, Witkiewicz A, Weber GF, Denhardt DT, et
al: Expression of a prometastatic splice variant of osteopontin,
OPNC, in human pancreatic ductal adenocarcinoma. Surgery.
146:232–240. 2009.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Sullivan J, Blair L, Alnajar A, Aziz T,
Chipitsyna G, Gong Q, Yeo CJ and Arafat HA: Expression and
regulation of nicotine receptor and osteopontin isoforms in human
pancreatic ductal adenocarcinoma. Histol Histopathol. 26:893–904.
2011.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Van Heek NT, Maitra A, Koopmann J, Fedarko
N, Jain A, Rahman A, Iacobuzio-Donahue CA, Adsay V, Ashfaq R, Yeo
CJ, et al: Gene expression profiling identifies markers of
ampullary adenocarcinoma. Cancer Biol Ther. 3:651–656.
2004.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Bloomston M, Ellison EC, Muscarella P,
Al-Saif O, Martin EW, Melvin WS and Frankel WL: Stromal osteonectin
overexpression is associated with poor outcome in patients with
ampullary cancer. Ann Surg Oncol. 14:211–217. 2007.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Hsu HP, Shan YS, Lai MD and Lin PW:
Osteopontin-positive infiltrating tumor-associated macrophages in
bulky ampullary cancer predict survival. Cancer Biol Ther.
10:144–154. 2010.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Sedivy R, Kalipciyan M, Mazal PR, Wolf B,
Wrba F, Karner-Hanusch J, Mühlbacher F and Mader RM:
Osteoclast-like giant cell tumor in mucinous cystadenocarcinoma of
the pancreas: An immunohistochemical and molecular analysis. Cancer
Detect Prev. 29:8–14. 2005.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Cao J, Li J, Sun L, Qin T, Xiao Y, Chen K,
Qian W, Duan W, Lei J, Ma J, et al: Hypoxia-driven paracrine
osteopontin/integrin αvβ3 signaling promotes pancreatic cancer cell
epithelial-mesenchymal transition and cancer stem cell-like
properties by modulating forkhead box protein M1. Mol Oncol.
13:228–245. 2019.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Lazar M, Sullivan J, Chipitsyna G, Gong Q,
Ng CY, Salem AF, Aziz T, Witkiewicz A, Denhardt DT, Yeo CJ and
Arafat HA: Involvement of osteopontin in the matrix-degrading and
proangiogenic changes mediated by nicotine in pancreatic cancer
cells. J Gastrointest Surg. 14:1566–1577. 2010.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Lazar M, Sullivan J, Chipitsyna G, Aziz T,
Salem AF, Gong Q, Witkiewicz A, Denhardt DT, Yeo CJ and Arafat HA:
Induction of monocyte chemoattractant protein-1 by nicotine in
pancreatic ductal adenocarcinoma cells: Role of osteopontin.
Surgery. 148:298–309. 2010.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Saxena S, Gandhi A, Lim PW, Relles D,
Sarosiek K, Kang C, Chipitsyna G, Sendecki J, Yeo CJ and Arafat HA:
RAN GTPase and osteopontin in pancreatic cancer. Pancreat Disord
Ther. 3(113)2013.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Deng L, Ren J, Liu D, Li H, Yang G, Wang
K, Song Y and Su H: Ran drives pancreatic cancer metastasis by
activating the osteopontin-PI3K/AKT-androgen receptor signaling
cascade. Toxicol Appl Pharmacol. 499(117328)2025.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Grudzinska E, Szmigiel P, Majewska K,
Mrowiec S and Czuba ZP: Pancreatic cancer and benign pancreatic
cystic lesions: Differences in cytokines, growth factors, and
immunological markers concentrations in serum and cystic fluid.
Cancers (Basel). 17(2783)2025.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Goluba K, Parfejevs V, Rostoka E,
Jekabsons K, Blake I, Neimane A, Ule AA, Rimsa R, Vangravs R,
Pcolkins A and Riekstina U: Personalized PDAC chip with functional
endothelial barrier for tumour biomarker detection: A platform for
precision medicine applications. Mater Today Bio.
29(101262)2024.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Li J, Wei T, Ma K, Zhang J, Lu J, Zhao J,
Huang J, Zeng T, Xie Y, Liang Y, et al: Single-cell RNA sequencing
highlights epithelial and microenvironmental heterogeneity in
malignant progression of pancreatic ductal adenocarcinoma. Cancer
Lett. 584(216607)2024.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Siddiqui AA, Jones E, Andrade D, Shah A,
Kowalski TE, Loren DE, Chipitsyna G and Arafat HA: Osteopontin
splice variant as a potential marker for metastatic disease in
pancreatic adenocarcinoma. J Gastroenterol Hepatol. 29:1321–1327.
2014.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Coppola D, Szabo M, Boulware D, Muraca P,
Alsarraj M, Chambers AF and Yeatman TJ: Correlation of osteopontin
protein expression and pathological stage across a wide variety of
tumor histologies. Clin Cancer Res. 10:184–190. 2004.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Ohike N, Sato M, Kawahara M, Ohyama S and
Morohoshi T: Ductal adenocarcinoma of the pancreas with
psammomatous calcification. Report of a case. JOP. 9:335–338.
2008.PubMed/NCBI
|
|
76
|
Takayama S, Maeda T, Nishihara M, Kanazawa
A, Chong HS, Oka H, Hirota S and Ishikawa O: A case of intraductal
tubulopapillary neoplasm of pancreas with severe calcification, a
potential pitfall in diagnostic imaging. Pathol Int. 65:501–506.
2015.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Rao AD, Liu Y, von Eyben R, Hsu CC, Hu C,
Rosati LM, Parekh A, Ng K, Hacker-Prietz A, Zheng L, et al:
Multiplex proximity ligation assay to identify potential prognostic
biomarkers for improved survival in locally advanced pancreatic
cancer patients treated with stereotactic body radiation therapy.
Int J Radiat Oncol Biol Phys. 100:486–489. 2018.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Kaleağasıoğlu F and Berger MR: SIBLINGs
and SPARC families: Their emerging roles in pancreatic cancer.
World J Gastroenterol. 20:14747–14759. 2014.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Fredriksson S, Horecka J, Brustugun OT,
Schlingemann J, Koong AC, Tibshirani R and Davis RW: Multiplexed
proximity ligation assays to profile putative plasma biomarkers
relevant to pancreatic and ovarian cancer. Clin Chem. 54:582–589.
2008.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Rychlíková J, Vecka M, Jáchymová M,
Macášek J, Hrabák P, Zeman M, Vávrová L, Řoupal J, Krechler T and
Ák A: Osteopontin as a discriminating marker for pancreatic cancer
and chronic pancreatitis. Cancer Biomark. 17:55–65. 2016.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Li H, Lan L, Chen H, Zaw Thin M, Ps H,
Nelson JK, Evans IM, Ruiz EJ, Cheng R, Tran L, et al: SPP1 is
required for maintaining mesenchymal cell fate in pancreatic
cancer. Nature. 648:203–209. 2025.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Blogowski W, Dolegowska K, Deskur A,
Dolegowska B and Starzyńska T: An attempt to evaluate selected
aspects of ‘Bone-Fat Axis’ Function in healthy individuals and
patients with pancreatic cancer. Medicine (Baltimore).
94(e1303)2015.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Melen-Mucha G, Niedziela A, Mucha S,
Motylewska E, Lawnicka H, Komorowski J and Stepien H: Elevated
peripheral blood plasma concentrations of tie-2 and angiopoietin 2
in patients with neuroendocrine tumors. Int J Mol Sci.
13:1444–1460. 2012.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Li JJ, Li HY and Gu F: Diagnostic
significance of serum osteopontin level for pancreatic cancer: A
meta-analysis. Genet Test Mol Biomarkers. 18:580–586.
2014.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Koopmann J, Rosenzweig CN, Zhang Z, Canto
MI, Brown DA, Hunter M, Yeo C, Chan DW, Breit SN and Goggins M:
Serum markers in patients with resectable pancreatic
adenocarcinoma: Macrophage inhibitory cytokine 1 versus CA19-9.
Clin Cancer Res. 12:442–446. 2006.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Chen R, Crispin DA, Pan S, Hawley S,
McIntosh MW, May D, Anton-Culver H, Ziogas A, Bronner MP and
Brentnall TA: Pilot study of blood biomarker candidates for
detection of pancreatic cancer. Pancreas. 39:981–988.
2010.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Hartung F and Weber GF: RNA blood levels
of osteopontin splice variants are cancer markers. Springerplus.
2(110)2013.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Sarosiek K, Jones E, Chipitsyna G,
Al-Zoubi M, Kang C, Saxena S, Gandhi AV, Sendiky J, Yeo CJ and
Arafat HA: Osteopontin (OPN) isoforms, diabetes, obesity, and
cancer; what is one got to do with the other? A new role for OPN. J
Gastrointest Surg. 19:639–650. 2015.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Rogers CD, Fukushima N, Sato N, Shi C,
Prasad N, Hustinx SR, Matsubayashi H, Canto M, Eshleman JR, Hruban
RH and Goggins M: Differentiating pancreatic lesions by microarray
and QPCR analysis of pancreatic juice RNAs. Cancer Biol Ther.
5:1383–1389. 2006.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Poruk KE, Firpo MA, Scaife CL, Adler DG,
Emerson LL, Boucher KM and Mulvihill SJ: Serum osteopontin and
tissue inhibitor of metalloproteinase 1 as diagnostic and
prognostic biomarkers for pancreatic adenocarcinoma. Pancreas.
42:193–197. 2013.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Song J, Sokoll LJ, Pasay JJ, Rubin AL, Li
H, Bach DM, Chan DW and Zhang Z: Identification of serum biomarker
panels for the early detection of pancreatic cancer. Cancer
Epidemiol Biomarkers Prev. 28:174–182. 2019.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Sekiguchi K, Matsuda A, Yamada M,
Matsumoto S, Sakurazawa N, Kawano Y, Yamada T, Miyashita M and
Yoshida H: The utility of serum osteopontin levels for predicting
postoperative complications after colorectal cancer surgery. Int J
Clin Oncol. 27:1706–1716. 2022.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Jiménez-Fonseca P, Martín MN,
Carmona-Bayonas A, Calvo A, Fernández-Mateos J, Redrado M,
Capdevila J, Lago NM, Lacasta A, Muñarriz J, et al: Biomarkers and
polymorphisms in pancreatic neuroendocrine tumors treated with
sunitinib. Oncotarget. 9:36894–36905. 2018.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Melisi D, Garcia-Carbonero R, Macarulla T,
Pezet D, Deplanque G, Fuchs M, Trojan J, Kozloff M, Simionato F,
Cleverly A, et al: TGFβ receptor inhibitor galunisertib is linked
to inflammation- and remodeling-related proteins in patients with
pancreatic cancer. Cancer Chemother Pharmacol. 83:975–991.
2019.PubMed/NCBI View Article : Google Scholar
|
|
95
|
Chen K, Wang Q, Liu X, Wang F, Ma Y, Zhang
S, Shao Z, Yang Y and Tian X: Single cell RNA-Seq identifies
Immune-related prognostic model and key Signature-SPP1 in
pancreatic ductal adenocarcinoma. Genes (Basel).
13(1760)2020.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Raymant M, Astuti Y, Alvaro-Espinosa L,
Green D, Quaranta V, Bellomo G, Glenn M, Chandran-Gorner V, Palmer
DH, Halloran C, et al: Macrophage-fibroblast JAK/STAT dependent
crosstalk promotes liver metastatic outgrowth in pancreatic cancer.
Nat Commun. 15(3593)2024.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Lu C, Liu Z, Klement JD, Yang D, Merting
AD, Poschel D, Albers T, Waller JL, Shi H and Liu K: WDR5-H3K4me3
epigenetic axis regulates OPN expression to compensate PD-L1
function to promote pancreatic cancer immune escape. J Immunother
Cancer. 9(e002624)2021.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Yang Y, Gong Y, Ding Y, Sun S, Bai R, Zhuo
S and Zhang Z: LINC01133 promotes pancreatic ductal adenocarcinoma
epithelial-mesenchymal transition mediated by SPP1 through binding
to Arp3. Cell Death Dis. 15(492)2024.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Gaviraghi M, Tunici P, Valensin S, Rossi
M, Giordano C, Magnoni L, Dandrea M, Montagna L, Ritelli R, Scarpa
A and Bakker A: Pancreatic cancer spheres are more than just
aggregates of stem marker-positive cells. Biosci Rep. 31:45–55.
2011.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Nose K, Saito H and Kuroki T: Isolation of
a gene sequence induced later by tumor-promoting
12-O-tetradecanoylphorbol-13-acetate in mouse osteoblastic cells
(MC3T3-E1) and expressed constitutively in ras-transformed cells.
Cell Growth Differ. 1:511–518. 1990.PubMed/NCBI
|
|
101
|
Pitarresi JR, Norgard RJ, Chiarella AM,
Suzuki K, Bakir B, Sahu V, Li J, Zhao J, Marchand B, Wengyn MD, et
al: PTHrP drives pancreatic cancer growth and metastasis and
reveals a new therapeutic vulnerability. Cancer Discov.
11:1774–1791. 2021.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Vanova K, Boukalova S, Gbelcova H, Muchova
L, Neuzil J, Gurlich R, Ruml T and Vitek L: Heme oxygenase is not
involved in the anti-proliferative effects of statins on pancreatic
cancer cells. BMC Cancer. 16(309)2016.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Adams CR, Htwe HH, Marsh T, Wang AL,
Montoya ML, Subbaraj L, Tward AD, Bardeesy N and Perera RM:
Transcriptional control of subtype switching ensures adaptation and
growth of pancreatic cancer. Elife. 8(e45313)2019.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Nishimori H, Yasoshima T, Hata F, Denno R,
Yanai Y, Nomura H, Tanaka H, Kamiguchi K, Sato N and Hirata K: A
novel nude mouse model of liver metastasis and peritoneal
dissemination from the same human pancreatic cancer line. Pancreas.
24:242–250. 2002.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Ohno K, Nishimori H, Yasoshima T,
Kamiguchi K, Hata F, Fukui R, Okuya K, Kimura Y, Denno R, Kon S, et
al: Inhibition of osteopontin reduces liver metastasis of human
pancreatic cancer xenografts injected into the spleen in a mouse
model. Surg Today. 40:347–356. 2010.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Zhivkova-Galunska M, Adwan H, Eyol E,
Kleeff J, Kolb A, Bergmann F and Berger MR: Osteopontin but not
osteonectin favors the metastatic growth of pancreatic cancer cell
lines. Cancer Biol Ther. 10:54–64. 2010.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Delany AM: Matricellular proteins
osteopontin and osteonectin/SPARC in pancreatic carcinoma. Cancer
Biol Ther. 10:65–67. 2010.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Eyol E, Murtaga A, Zhivkova-Galunska M,
Georges R, Zepp M, Djandji D, Kleeff J, Berger MR and Adwan H: Few
genes are associated with the capability of pancreatic ductal
adenocarcinoma cells to grow in the liver of nude rats. Oncol Rep.
28:2177–2187. 2012.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Yang MC, Wang HC, Hou YC, Tung HL, Chiu TJ
and Shan YS: Blockade of autophagy reduces pancreatic cancer stem
cell activity and potentiates the tumoricidal effect of
gemcitabine. Mol Cancer. 14(179)2015.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Ouyang L, Sun MM, Zhou PS, Ren YW, Liu XY,
Wei WY, Song ZS, Lu K and Yang LX: LncRNA FOXD1-AS1 regulates
pancreatic cancer stem cell properties and 5-FU resistance by
regulating the miR-570-3p/SPP1 axis as a ceRNA. Cancer Cell Int.
24(4)2024.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Nallasamy P, Nimmakayala RK, Karmakar S,
Leon F, Seshacharyulu P, Lakshmanan I, Rachagani S, Mallya K, Zhang
C, Ly QP, et al: Pancreatic tumor microenvironment factor promotes
cancer stemness via SPP1-CD44 Axis. Gastroenterology.
161:1998–2013.e7. 2021.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Fukusada S, Shimura T, Natsume M,
Nishigaki R, Okuda Y, Iwasaki H, Sugimura N, Kitagawa M, Katano T,
Tanaka M, et al: Osteopontin secreted from obese adipocytes
enhances angiogenesis and promotes progression of pancreatic ductal
adenocarcinoma in obesity. Cell Oncol (Dordr). 47:229–244.
2024.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Liu Y, Fu Y, Liu T, Wang J, Zhang Z, Shi
Y, Cao Z, Yang G, Chen H, Luo W, et al: SPP1+ venous endothelial
cells and CRABP2+ tumor cells contribute to favorable body and tail
pancreatic ductal adenocarcinoma tumor microenvironment. Mol
Carcinog. 64:1377–1391. 2025.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Li X, Tjalkens RB, Shrestha R and Safe S:
Structure-dependent activation of gene expression by bis-indole and
quinoline-derived activators of nuclear receptor 4A2. Chem Biol
Drug Des. 94:1711–1720. 2019.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Nolen BM, Brand RE, Prosser D,
Velikokhatnaya L, Allen PJ, Zeh HJ, Grizzle WE, Huang Y, Lomakin A
and Lokshin AE: Prediagnostic serum biomarkers as early detection
tools for pancreatic cancer in a large prospective cohort study.
PLoS One. 9(e94928)2014.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Hill RL, Yadav D, Hart PA, Whitcomb DC,
McQuerry KJ, Karnik KN, Stello KM, Conwell DL and Rao M:
Circulating molecular drivers of bone remodeling in pancreatitis.
Clin Transl Gastroenterol. 16(e00926)2025.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Rao C, Bush N, Rana SS, Sharma RK, Rana S,
Jeyashree K and Gupta R: Plasma osteopontin levels as an early
predictor of mortality in acute pancreatitis: A preliminary study.
Pancreas. 51:e77–e79. 2022.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Sward P, Bertilsson S, Struglics A and
Kalaitzakis E: Serum osteopontin is associated with organ failure
in patients with acute pancreatitis. Pancreas. 47:e7–e10.
2018.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Aggarwal S, Xie MH, Maruoka M, Foster J
and Gurney AL: Acinar cells of the pancreas are a target of
interleukin-22. J Interferon Cytokine Res. 21:1047–1053.
2001.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Arafat HA, Katakam AK, Chipitsyna G, Gong
Q, Vancha AR, Gabbeta J and Dafoe DC: Osteopontin protects the
islets and beta-cells from interleukin-1 beta-mediated cytotoxicity
through negative feedback regulation of nitric oxide.
Endocrinology. 148:575–584. 2007.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Llewellyn HP, Vaidya VS, Wang Z, Peng Q,
Hyde C, Potter D, Wang J, Zong Q, Arat S, Martin M, et al:
Evaluating the sensitivity and specificity of promising circulating
biomarkers to diagnose liver injury in humans. Toxicol Sci.
181:23–34. 2021.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Pini M, Rhodes DH, Castellanos KJ, Cabay
RJ, Grady EF and Fantuzzi G: Rosiglitazone improves survival and
hastens recovery from pancreatic inflammation in obese mice. PLoS
One. 7(e40944)2012.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Takada H, Nakazawa T, Ohara H, Ando T,
Hayashi K, Naito I, Okumura F, Tanaka H, Yamada T, Takahashi S and
Joh T: Role of osteopontin in calcification in autoimmune
pancreatitis. Dig Dis Sci. 54:793–801. 2009.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Nakamura M, Oka M, Iizuka N, Kawauchi S,
Gondo T, Ueno T and Tangoku A: Osteopontin expression in chronic
pancreatitis. Pancreas. 25:182–187. 2002.PubMed/NCBI View Article : Google Scholar
|
|
125
|
Cheng Y, Li Y, Scherer N,
Grundner-Culemann F, Lehtimäki T, Mishra BH, Raitakari OT, Nauck M,
Eckardt KU, Sekula P, et al: Genetics of osteopontin in patients
with chronic kidney disease: The German Chronic Kidney Disease
study. PLoS Genet. 18(e1010139)2022.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Togashi Y and Miyamoto Y: Urinary cystatin
C as a biomarker for diabetic nephropathy and its
immunohistochemical localization in kidney in Zucker diabetic fatty
(ZDF) rats. Exp Toxicol Pathol. 65:615–622. 2013.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Chung BH, Li C, Sun BK, Lim SW, Ahn KO,
Yang JH, Choi YH, Yoon KH, Sugawara A, Ito S, et al: Rosiglitazone
protects against cyclosporine-induced pancreatic and renal injury
in rats. Am J Transplant. 5:1856–1867. 2005.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Meerwein C, Korom S, Arni S, Inci I, Weder
W and Jungraithmayr W: The effect of low-dose Continuous
Erythropoietin receptor activator in an experimental model of acute
Cyclosporine A induced renal injury. Eur J Pharmacol. 671:113–119.
2011.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Glazunova AM, Arutyunova MS, Tarasov EV,
Nikankina LV, Ilyin AV, Shamkhalova MS, Shestakova MV and Moysyuk
YG: Evaluation of markers for renal graft dysfunction in patients
with type 1 diabetes mellitus after kidney transplantation and
simultaneous pancreas-kidney transplantation. Ter Arkh. 88:25–34.
2016.PubMed/NCBI View Article : Google Scholar : (In Russian).
|
|
130
|
Gong Q, Chipitsyna G, Gray CF,
Anandanadesan R and Arafat HA: Expression and regulation of
osteopontin in type 1 diabetes. Islets. 1:34–41. 2009.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Ito M, Makino N, Matsuda A, Ikeda Y,
Kakizaki Y, Saito Y, Ueno Y and Kawata S: High glucose accelerates
cell proliferation and increases the secretion and mRNA expression
of osteopontin in human pancreatic duct epithelial cells. Int J Mol
Sci. 18(807)2017.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Melanitou E: Investigation of type 1
diabetes in NOD mice knockout for the osteopontin gene. Gene.
753(144785)2020.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Garbossa SG and Folli F: Vitamin D,
sub-inflammation and insulin resistance. A window on a potential
role for the interaction between bone and glucose metabolism. Rev
Endocr Metab Disord. 18:243–258. 2017.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Daniele G, Winnier D, Mari A, Bruder J,
Fourcaudot M, Pengou Z, Hansis-Diarte A, Jenkinson C, Tripathy D
and Folli F: The potential role of the
osteopontin-osteocalcin-osteoprotegerin triad in the pathogenesis
of prediabetes in humans. Acta Diabetol. 55:139–148.
2018.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Fierabracci A, Biro PA, Yiangou Y, Mennuni
C, Luzzago A, Ludvigsson J, Cortese R and Bottazzo GF: Osteopontin
is an autoantigen of the somatostatin cells in human islets:
Identification by screening random peptide libraries with sera of
patients with insulin-dependent diabetes mellitus. Vaccine.
18:342–354. 1999.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Aspord C, Rome S and Thivolet C: Early
events in islets and pancreatic lymph nodes in autoimmune diabetes.
J Autoimmun. 23:27–35. 2004.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Arafat HA, Lada E, Katakam AK and Amin N:
Osteopontin deficiency impacts the pancreatic TH1/TH2 cytokine
profile following multiple low dose streptozotocin-induced
diabetes. Exp Clin Endocrinol Diabetes. 114:555–562.
2006.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Safley SA, Kenyon NS, Berman DM, Barber
GF, Willman M, Duncanson S, Iwakoshi N, Holdcraft R, Gazda L,
Thompson P, et al: Microencapsulated adult porcine islets
transplanted intraperitoneally in streptozotocin-diabetic non-human
primates. Xenotransplantation. 25(e12450)2018.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Cai M, Bompada P, Salehi A, Acosta JR,
Prasad RB, Atac D, Laakso M, Groop L and De Marinis Y: Role of
osteopontin and its regulation in pancreatic islet. Biochem Biophys
Res Commun. 495:1426–1431. 2018.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Wendt A, Mollet IG, Knutsson A, Bolmgren
VS, Hultgardh-Nilsson A, Gomez MF and Eliasson L: Osteopontin
affects insulin vesicle localization and Ca2+ homeostasis in
pancreatic beta cells from female mice. PLoS One.
12(e0170498)2017.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Sommese L, Zullo A, Mancini FP, Fabbricini
R, Soricelli A and Napoli C: Clinical relevance of epigenetics in
the onset and management of type 2 diabetes mellitus. Epigenetics.
12:401–415. 2017.PubMed/NCBI View Article : Google Scholar
|
|
142
|
Xiong X, Wang X, Li B, Chowdhury S, Lu Y,
Srikant CB, Ning G and Liu JL: Pancreatic islet-specific
overexpression of Reg3β protein induced the expression of pro-islet
genes and protected the mice against streptozotocin-induced
diabetes mellitus. Am J Physiol Endocrinol Metab. 300:E669–E680.
2011.PubMed/NCBI View Article : Google Scholar
|
|
143
|
Dickerson MT, Vierra NC, Milian SC, Dadi
PK and Jacobson DA: Osteopontin activates the diabetes-associated
potassium channel TALK-1 in pancreatic β-cells. PLoS One.
12(e0175069)2017.PubMed/NCBI View Article : Google Scholar
|
|
144
|
Terami N, Ogawa D, Tachibana H, Hatanaka
T, Wada J, Nakatsuka A, Eguchi J, Horiguchi CS, Nishii N, Yamada H,
et al: Long-term treatment with the sodium glucose cotransporter 2
inhibitor, dapagliflozin, ameliorates glucose homeostasis and
diabetic nephropathy in db/db mice. PLoS One.
9(e100777)2014.PubMed/NCBI View Article : Google Scholar
|
|
145
|
Walaszek K, Lower EE, Ziolkowski P and
Weber GF: Breast cancer risk in premalignant lesions: Osteopontin
splice variants indicate prognosis. Br J Cancer. 119:1259–1266.
2018.PubMed/NCBI View Article : Google Scholar
|
|
146
|
Shiraga H, Min W, VanDusen WJ, Clayman MD,
Miner D, Terrell CH, Sherbotie JR, Foreman JW, Przysiecki C,
Neilson EG, et al: Inhibition of calcium oxalate crystal growth in
vitro by uropontin: Another member of the aspartic acid-rich
protein superfamily. Proc Natl Acad Sci USA. 89:426–430.
1992.PubMed/NCBI View Article : Google Scholar
|
|
147
|
Gorski JP and Shimizu K: Isolation of new
phosphorylated glycoprotein from mineralized phase of bone that
exhibits limited homology to adhesive protein osteopontin. J Biol
Chem. 263:15938–15945. 1988.PubMed/NCBI
|
|
148
|
Kim HS, Shin HI, Lim HS, Lee TY, Lee K and
Jeong D: α-Lipoic acid attenuates coxsackievirus B3-induced ectopic
calcification in heart, pancreas, and lung. Biochem Biophys Res
Commun. 432:378–383. 2013.PubMed/NCBI View Article : Google Scholar
|
|
149
|
Hwang SM, Lopez CA, Heck DE, Gardner CR,
Laskin DL, Laskin JD and Denhardt DT: Osteopontin inhibits
induction of nitric oxide synthase gene expression by inflammatory
mediators in mouse kidney epithelial cells. J Biol Chem.
269:711–715. 1994.PubMed/NCBI
|
|
150
|
Miyazaki Y, Tashiro T, Higuchi Y,
Setoguchi M, Yamamoto S, Nagai H, Nasu M and Vassalli P: Expression
of osteopontin in a macrophage cell line and in transgenic mice
with pulmonary fibrosis resulting from the lung expression of a
tumor necrosis factor-alpha transgene. Ann N Y Acad Sci.
760:334–341. 1995.PubMed/NCBI View Article : Google Scholar
|
|
151
|
Singh K, Balligand JL, Fischer TA, Smith
TW and Kelly RA: Glucocorticoids increase osteopontin expression in
cardiac myocytes and microvascular endothelial cells. Role in
regulation of inducible nitric oxide synthase. J Biol Chem.
270:28471–2848. 1995.PubMed/NCBI View Article : Google Scholar
|
|
152
|
Rollo EE, Laskin DL and Denhardt DT:
Osteopontin inhibits nitric oxide production and cytotoxicity by
activated RAW264.7 macrophages. J Leukoc Biol. 60:397–404.
1996.PubMed/NCBI View Article : Google Scholar
|
|
153
|
Page MJ, McKenzie JE, Bossuyt PM, Boutron
I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan
SE, et al: The PRISMA 2020 statement: An updated guideline for
reporting systematic reviews. BMJ. 372(n71)2021.PubMed/NCBI View Article : Google Scholar
|