Introduction
Severe psychiatric disorders, including
schizophrenia, bipolar disorder and related psychotic illnesses,
are among the leading causes of long-term disability worldwide.
Although antipsychotic medications remain indispensable for symptom
control, relapse prevention and functional recovery, their
long-term use is frequently complicated by adverse physical health
outcomes. Among these, antipsychotic-associated weight gain (AAWG)
represents one of the most common, distressing and clinically
consequential adverse effects, contributing substantially to excess
cardiometabolic morbidity and premature mortality in this
population (1-3).
Individuals with severe mental illness experience a
markedly reduced life expectancy compared with the general
population, with cardiovascular disease accounting for a large
proportion of excess deaths (1,2). Weight
gain following the initiation of antipsychotics is a major
modifiable contributor to this disparity. Clinically significant
weight gain, often defined as a ≥7% increase from baseline body
weight, has been reported with almost all antipsychotic agents,
although the magnitude and trajectory of weight change vary across
drugs and individuals (3-5).
Notably, weight gain frequently emerges early in the course of
treatment, progresses rapidly during the first months, and may
persist long term, even when psychiatric symptoms are
well-controlled (6,7).
Second-generation antipsychotics (SGAs),
particularly olanzapine and clozapine, are consistently associated
with the highest risk of AAWG and metabolic disturbances, including
insulin resistance, dyslipidemia and impaired glucose tolerance
(5,6). These adverse effects are not merely
cosmetic; they are strongly associated with non-adherence to
treatment, a reduced quality of life, stigma, and the early
discontinuation of otherwise effective antipsychotic therapy
(8,9). Consequently, AAWG represents a critical
challenge at the intersection of psychiatric stability and physical
health, underscoring the need for effective and acceptable
management strategies.
The mechanisms underlying AAWG are multifactorial
and involve the dysregulation of appetite control, satiety
signaling, energy expenditure and peripheral metabolism.
Antipsychotic medications interact with central neurotransmitter
systems involved in energy homeostasis, including serotonergic,
histaminergic, muscarinic and dopaminergic pathways, as well as
peripheral metabolic processes that influence insulin sensitivity
and lipid handling (10-12).
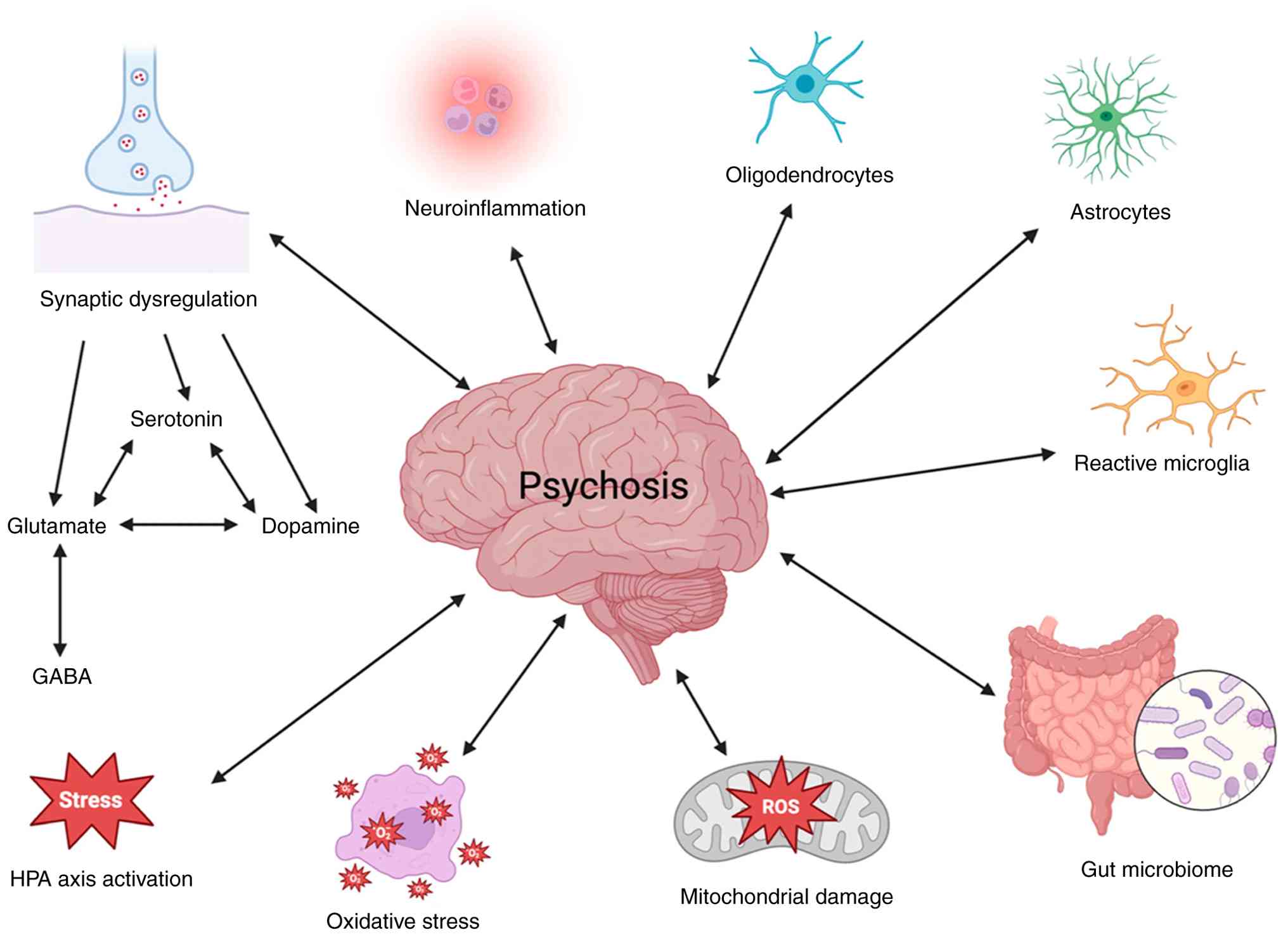
The broader neurobiological framework of psychosis, including
neurotransmission dysregulation, neuroinflammation, oxidative
stress and mitochondrial dysfunction, is illustrated in Fig. 1. Although these mechanisms are
central to psychotic disorders, only selected pathways are directly
implicated in antipsychotic-associated weight gain (13).
From a clinical perspective, the trajectory of AAWG
is particularly relevant. Longitudinal studies have demonstrated
that the greatest proportion of weight gain occurs within the first
6-12 months of antipsychotic exposure, after which weight gain
often plateaus at a higher set point (6,14). This
pattern suggests that early intervention is crucial to prevent
excessive long-term weight accumulation. Once a new weight set
point is established, reversal becomes considerably more difficult,
even with intensive lifestyle or pharmacological interventions
(15). This understanding has
critical implications for the timing and selection of preventive
strategies.
Lifestyle modifications, including dietary
counseling, increased physical activity and behavioral
interventions, are is widely recommended as first-line management
for AAWG in clinical guidelines (16-18).
However, the real-world effectiveness of lifestyle interventions in
patients with severe mental illness is often limited. Cognitive
impairment, negative symptoms, sedation, social disadvantage and
illness-related functional limitations can significantly reduce
engagement and adherence (19,20).
Moreover, structured lifestyle programs are resource-intensive and
not consistently available, particularly in low- and middle-income
countries. As a result, reliance on lifestyle modifications alone
frequently fails to prevent clinically meaningful weight gain in
routine psychiatric practice.
These limitations have prompted growing interest in
adjunctive pharmacological strategies to mitigate AAWG. Among
available options, metformin has emerged as the most extensively
studied and consistently supported agent. Metformin is a
well-established insulin-sensitizing drug with decades of use in
the management of type 2 diabetes mellitus. Its mechanisms of
action extend beyond glucose lowering and include effects on
appetite regulation, gut hormone secretion, insulin sensitivity,
and energy balance (12,21). Of note, metformin does not cause
hypoglycemia when used as monotherapy and has a favorable safety
profile when appropriately prescribed and monitored.
Evidence from randomized controlled trials and
meta-analyses over the past two decades has demonstrated that
metformin can attenuate AAWG and improve metabolic parameters in
both diabetic and non-diabetic populations (22-25).
More recent analyses have refined this understanding by
highlighting the importance of timing. Meta-analyses and systematic
reviews published within the past 5 years have indicated that
metformin is particularly effective when initiated early in the
course of antipsychotic treatment or in antipsychotic-naïve
patients, where it can prevent further weight gain rather than
attempting to reverse established obesity (24-27).
This preventive effect is clinically meaningful, as even modest
reductions in weight trajectory can translate into substantial
long-term cardiometabolic benefit.
Perspectives in high-impact psychiatric literature,
including Schizophrenia Bulletin, have emphasized the need to
re-evaluate traditional stepwise algorithms that reserve metformin
as a last-line option only after lifestyle modifications and
antipsychotic switching have failed (18). Such approaches may inadvertently
delay an effective intervention until the window for maximal
benefit has passed. Instead, emerging evidence supports a more
proactive and individualized approach, integrating metformin
earlier in patients at a high risk of AAWG, particularly those
prescribed high-risk antipsychotics or those experiencing rapid
early weight gain.
Despite this growing evidence base, the real-world
implementation of metformin for AAWG remains inconsistent.
Clinician concerns regarding off-label use, perceived safety risks,
pill burden, and uncertainty about long-term benefits continue to
limit its uptake in psychiatric settings (28). In addition, the majority of available
data are derived from controlled trials conducted in high-income
countries, and there remains a relative paucity of prospective
observational data reflecting routine clinical practice in diverse
healthcare systems (13,19-21).
This gap is particularly relevant in low- and middle-income
countries, where resource constraints, limited access to structured
lifestyle programs, and high reliance on metabolically adverse
antipsychotics may amplify the burden of AAWG.
Furthermore, patient-centered considerations are
increasingly recognized as essential in the management of AAWG.
Qualitative research indicates that weight gain is among the most
distressing side effects of antipsychotic treatment from the
patient perspective, often perceived as more troubling than some
psychiatric symptoms themselves (29). Patients frequently report a desire
for early, effective interventions that acknowledge both physical
and mental health priorities. Pharmacological strategies, such as
metformin, which may reduce hunger and stabilize weight, are often
viewed as acceptable and empowering when appropriately explained
and monitored.
Against this background, there is a clear need for
prospective, real-world studies evaluating the effectiveness of
adjunctive metformin therapy for AAWG in routine psychiatric care.
Such studies can complement randomized controlled trials by
capturing broader patient populations, reflecting typical
prescribing practices, and addressing pragmatic outcomes such as
adherence and tolerability. Notably, observational data can help
inform context-specific clinical decision-making, particularly in
healthcare systems where guideline recommendations may not fully
account for local constraints and patient preferences.
Therefore, the present prospective observational
study was undertaken to evaluate the effectiveness of adjunctive
metformin therapy, in combination with lifestyle modifications, in
mitigating antipsychotic-associated weight gain in a real-world
psychiatric population. By focusing on clinically meaningful
outcomes such as changes in body weight and body mass index, and by
situating the findings within contemporary evidence and evolving
clinical perspectives, this study aims to contribute to the growing
body of literature supporting earlier and more individualized
management of AAWG.
Patients and methods
Study population
The present study was a 6-month, prospective
observational, non-randomized study conducted at the Department of
Psychiatry at Owaisi Hospital and Research Centre, Hyderabad,
India, which provides both inpatient and outpatient psychiatric
services. The primary objective was to assess the effectiveness of
adjunctive metformin therapy in the management and prevention of
AAWG. Clinical data were collected from the medical records of
patients, including case sheets and treatment charts. Patient
recruitment and data collection were conducted between June, 2024
and November, 2024. Eligible participants were adults aged 18-65
years, of either sex, who were receiving antipsychotic medications.
The median age of the study population was 27 years. Metformin was
prescribed at doses of 500, 850 mg, or 1 g according to routine
clinical judgment. Participants were allocated into two groups
based on usual prescribing practices: A test group receiving
metformin in addition to antipsychotic therapy and lifestyle
modifications, and a control group receiving antipsychotic therapy
with lifestyle modifications alone. The baseline demographic and
clinicopathological characteristics of the study population are
summarized in Table I.
 | Table IBaseline demographic and
clinicopathological characteristics of the study population. |
Table I
Baseline demographic and
clinicopathological characteristics of the study population.
| Variable | No. of
participants |
|---|
| Age (years), median
(range) | 27 (18-65) |
| Age group 18-29 | 39 |
| Age group 30-39 | - |
| Age group 40-49 | - |
| Age group 50-59 | - |
| ≥60 years | 2 |
| Sex (female) | 64 |
| Sex (male) | 36 |
| Primary psychiatric
diagnosis | |
|
Schizophrenia | 43 |
|
Bipolar
disorder | 17 |
|
Anxiety
disorders | 14 |
|
Major
depressive disorder | 13 |
|
Others | 13 |
| Antipsychotic
prescribed | |
|
Olanzapine | 34 |
|
Risperidone | 24 |
|
Amisulpride | 16 |
|
Others | 26 |
Lifestyle modifications
Lifestyle modifications were provided to the
participants in both study groups as part of routine clinical care.
These consisted of standardized verbal counseling delivered by the
treating psychiatrist or clinical pharmacist during outpatient
visits or inpatient rounds. Dietary advice included recommendations
for a balanced diet, reduction of calorie-dense and high-fat foods,
limitation of sugar-sweetened beverages, and the encouragement of
regular meal timing, in accordance with established guidelines for
the management of antipsychotic-associated weight gain and
cardiometabolic risk (6-8).
Physical activity advice focused on
moderate-intensity activities, such as brisk walking for ~30 min
per day on at least 3-5 days per week, tailored to the physical
capacity and psychiatric status of the patient, consistent with
international recommendations for individuals with severe mental
illness (9,11). Educational counseling emphasized the
association between antipsychotic therapy, weight gain and an
increased cardiometabolic risk, as recommended by contemporary
clinical guidelines (7,11).
No structured, supervised, or protocol-driven
dietary or exercise program was implemented. The intensity and
frequency of lifestyle interventions were not standardized, and
formal adherence assessment was not performed. Compliance with
lifestyle recommendations was assessed based on patient self-report
during routine follow-up visits, a commonly used approach in
real-world observational psychiatric studies (12,13).
Statistical analysis
Statistical analyses were performed using SPSS
version 25 software (IBM Corp.). Continuous variables are presented
as the mean ± standard deviation, and categorical variables are
summarized as frequencies and percentages. Within-group comparisons
of body weight and body mass index (BMI) before and after the
intervention were conducted using paired t-tests. Between-group
comparisons of mean changes in weight and BMI were performed using
independent samples t-tests. The comparison of BMI was analyzed
using a two-way mixed ANOVA with time (baseline vs. follow-up) with
the Bonferroni correction as the within-subject factor and study
group as the between-subject factor. For between-group analyses,
mean differences in change scores (follow-up minus baseline) were
calculated, and corresponding 95% confidence intervals (CIs) were
derived based on the standard error of the mean difference using
the independent samples t-test model. The CIs were computed as:
mean difference ± (t x standard error of the difference), where the
t-value was obtained from the appropriate degrees of freedom for
each comparison. All tests were two-tailed, and a P-value of
<0.05 was considered to indicate a statistically significant
difference.
Results
Baseline characteristics
The majority of the study participants were young
adults aged 18-29 years (39%), while only 2% were aged >60
years. Female patients constituted a higher proportion of the study
population (64%) compared with males. Olanzapine was the most
frequently prescribed antipsychotic (34%), followed by risperidone
(24%) and amisulpride (16%). Clonazepam (12%) was prescribed as an
adjunctive medication and was not classified as an antipsychotic.
Cariprazine (6%), quetiapine (4%), and aripiprazole (4%) were less
commonly used. Schizophrenia was the most prevalent psychiatric
diagnosis (43%), followed by bipolar disorder (17%) and anxiety
disorders (14%). Other diagnoses included major depressive disorder
(13%), psychosis not otherwise specified (4%), obsessive-compulsive
disorder (3%), post-traumatic disorder (3%), absence seizures (2%),
and alcohol dependence (1%) (Table
I).
Weight change analysis
An exploratory comparison indicated that patients
with ongoing treatment with antipsychotics exhibited numerically
lower weight gain than the treatment-naïve patients; however, this
difference was not statistically significant (mean difference, 6
kg; P=0.47; 95% CI, -2.3 to 1.09) (Table II).
 | Table IIComparison of mean change in body
weight across clinical subgroups. |
Table II
Comparison of mean change in body
weight across clinical subgroups.
| Variable | Mean weight change
(kg) | Standard
deviation | P-value | 95% confidence
interval |
|---|
| Treatment
status | | | | |
|
Ongoing
antipsychotic use | -0.62 | 2.31 | 0.47 | -2.34 to 1.09 |
|
Treatment-naïve | Reference | - | - | - |
| Study group | | | | |
|
Test group
(metformin + lifestyle modifications) | -4.99 | 1.29 | <0.001 | -7.53 to -2.47 |
|
Control
group (lifestyle modifications only) | Reference | - | - | - |
| Adverse drug
reactions (ADRs) | | | | |
|
Present | +0.26 | 1.25 | 0.83 | -2.18 to 2.71 |
|
Absent | Reference | - | - | - |
| Sex | | | | |
|
Female | +1.69 | 0.80 | 0.39 | -0.88 to 2.27 |
|
Male | Reference | - | - | - |
Between-group analysis demonstrated a statistically
significant difference in weight change. Participants in group 1
(metformin plus lifestyle modifications) experienced a mean weight
reduction of ~4 kg compared with group 2 (lifestyle modifications
alone) (P<0.001; 95% CI, -7.5 to -2.4) (Table II).
Adverse drug reactions exhibited no significant
association with weight change. Patients reporting adverse
reactions had a mean weight increase of 0.263 kg compared with
those without adverse reactions (P=0.833; 95% CI, -2.1 to 2.7).
Sex-based analysis revealed no statistically significant difference
in weight change between female and male patients (P=0.387; 95% CI,
-0.88 to 2.27) (Table II).
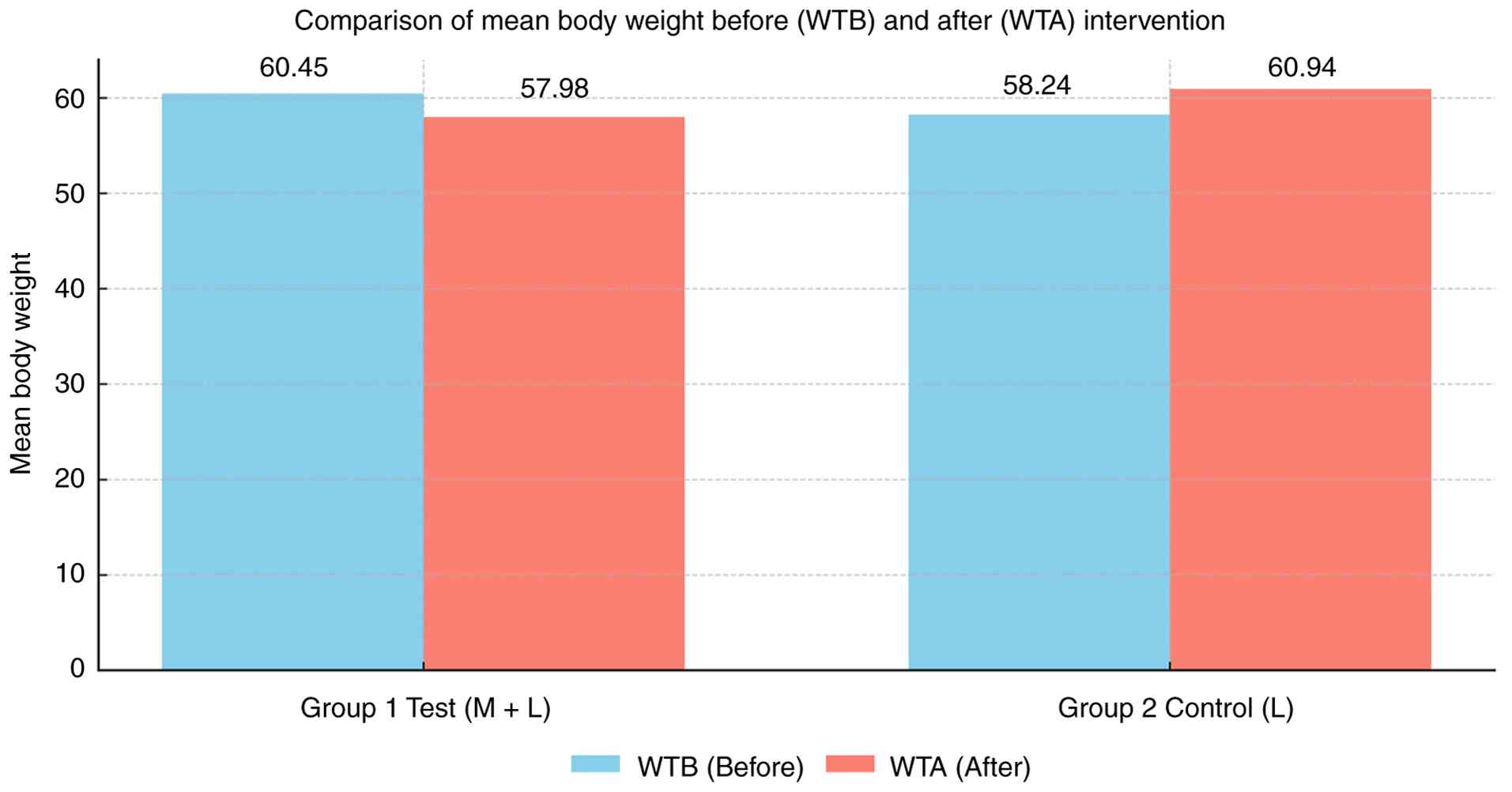
Comparison of body weight
Within-group comparisons revealed that in group 1
(metformin plus lifestyle modifications), the mean body weight
decreased from 60.45 kg at baseline to 57.98 kg following the
intervention, indicating a net weight reduction. By contrast, group
2 (lifestyle modifications alone) demonstrated an increase in mean
body weight from 58.24 to 60.94 kg over the study period. These
findings indicate a beneficial effect of adjunctive metformin
therapy in preventing antipsychotic-associated weight gain
(Table III and Fig. 2).
 | Table IIIComparison of mean body weight before
and after the intervention. |
Table III
Comparison of mean body weight before
and after the intervention.
| Parameter | Group 1: Test
(metformin + lifestyle modifications) | Group 2: Control
(lifestyle modifications only) |
|---|
| Weight before
intervention (kg) | 60.45±9.1 | 58.24±8.9 |
| Weight after
intervention (kg) | 57.98±8.8 | 60.94±9.3 |
| Mean change
(kg) | -2.47 | +2.70 |
| Within-group
P-value | <0.001 | <0.001 |
Comparison of BMI
BMI was assessed at baseline and after 6 months of
follow-up in both study groups (Table
IV). In group 1 (metformin plus lifestyle modifications), the
mean BMI decreased from 23.6±3.4 kg/m2 at baseline to
22.6±3.3 kg/m2 at follow-up, representing a mean
reduction of approximately 1.0 kg/m2, which was
statistically significant (P<0.001).
 | Table IVComparison of BMI before and after
the intervention. |
Table IV
Comparison of BMI before and after
the intervention.
| A, BMI in the
groups before and after the intervention |
|---|
| Parameter | Group 1: Test
(metformin + lifestyle modifications) | Group 2: Control
(lifestyle modifications only) |
|---|
| BMI before
intervention (kg/m2) | 23.6±3.4 | 22.9±3.2 |
| BMI after
intervention (kg/m2) | 22.6±3.3 | 24.0±3.5 |
| B, Results of
two-way mixed ANOVA |
| Effect | F value | P-value |
| Time effect | F (1,98)=21.4 | P<0.001 |
| Group effect | F (1,98)=9.6 | P=0.003 |
| Time x group
interaction | F (1,98)=28.7 | P<0.001 |
| C, Post-hoc
Bonferroni comparisons |
| Comparison | Mean
difference | P-value |
| Test group:
Baseline vs. follow-up | -1.0 kg/m² | P<0.001 |
| Control group:
Baseline vs. follow-up | +1.1 kg/m² | P<0.001 |
| Between groups at
follow-up | -1.4 kg/m² | P=0.002 |
The comparison of BMI was analyzed using a two-way
mixed ANOVA with time (baseline vs. follow-up) as the
within-subject factor and study group as the between-subject
factor. The analysis demonstrated a significant main effect of time
(P<0.001) and a significant group x time interaction
(P<0.001), indicating that BMI changes over time differed
between the two groups. Post-hoc comparisons with the Bonferroni
correction revealed that the metformin group experienced a
significant reduction in BMI from 23.6±3.4 to 22.6±3.3
kg/m2 (P<0.001). By contrast, the control group
exhibited a significant increase in BMI from 22.9±3.2 to 24.0±3.5
kg/m2 (P<0.001). Between-group comparisons at
follow-up also showed a significant difference favoring the
metformin group (P=0.002) (Table
IV).
Discussion
In the present prospective observational study
conducted in a real-world psychiatric setting, adjunctive metformin
therapy was associated with a significant reduction in body weight
and BMI among patients receiving antipsychotic treatment.
Participants in the metformin group experienced a mean weight
reduction of ~4 kg compared with those receiving lifestyle
modification alone, supporting the effectiveness of metformin as an
adjunctive strategy for managing AAWG. The study population
reflects routine clinical practice, with a predominance of
second-generation antipsychotics, particularly olanzapine and
risperidone, agents well recognized for their high metabolic
liability. Benzodiazepines, including clonazepam, were prescribed
as concomitant medications and were not classified as antipsychotic
agents for analytical purposes.
AAWG remains a major clinical challenge in long-term
psychiatric care, contributing to increased cardiometabolic risk,
reduced quality of life, and poor treatment adherence (14,15).
Recent large-scale observational studies and meta-analyses have
reaffirmed that SGAs such as olanzapine and clozapine are
associated with the greatest magnitude of weight gain, particularly
during the early phases of treatment (16,17).
Contemporary evidence emphasizes that early and proactive
intervention is critical, as weight gain trajectories established
within the first 6-12 months are often sustained over time and
difficult to reverse (18).
The findings of the present study are consistent
with those of previous randomized controlled trials and
meta-analyses published between 2021 and 2025, which demonstrate
that adjunctive metformin significantly attenuates AAWG and
improves metabolic parameters in patients receiving antipsychotic
therapy (12,19-21).
An updated meta-analysis by Zheng et al reported a
clinically meaningful reduction in body weight and BMI with
metformin compared with placebo, even among non-diabetic patients
(21). Similarly, the study by
Seifarth et al (20)
confirmed moderate-quality evidence supporting metformin for
reducing antipsychotic-related weight gain and insulin resistance.
Other analyses have further highlighted that metformin is
particularly effective when initiated early or in patients at high
metabolic risk, reinforcing its role as both a preventive and
therapeutic agent (21).
In the present study, longitudinal follow-up
demonstrated a clear divergence in weight trajectories between
groups. Patients receiving metformin exhibited a reduction in mean
body weight from 60.45 to 57.98 kg, whereas those managed with
lifestyle modifications alone experienced an increase from 58.24 to
60.94 kg. Corresponding changes in BMI mirrored these trends. These
findings are consistent with previous randomized controlled trials
and meta-analyses demonstrating significant reductions in body
weight and BMI with adjunctive metformin in patients receiving
antipsychotic therapy (3,21,24,27).
Notably, these real-world data complement evidence from controlled
trials by demonstrating effectiveness under routine prescribing
conditions.
Mechanistically, metformin exerts its beneficial
effects on weight regulation through multiple pathways, including
improved insulin sensitivity, the suppression of hepatic
gluconeogenesis, modulation of gut microbiota, and the enhancement
of anorexigenic gut hormones such as glucagon-like peptide-1 and
peptide YY (24,25). Recent translational studies suggest
that these mechanisms may counteract antipsychotic-induced
disruptions in appetite signaling and peripheral metabolism,
thereby stabilizing weight trajectories in vulnerable patients
(26). The observed benefits of
metformin in non-diabetic populations further support its broader
applicability in psychiatric practice (27).
An exploratory analysis in the present study
suggested that patients with ongoing exposure to antipsychotics
exhibited numerically lower weight gain compared with
treatment-naïve individuals; however, this difference did not reach
statistical significance and should be interpreted cautiously.
Similar findings have been reported in recent cohort studies, where
early metabolic changes were highly variable and influenced by
baseline risk factors, antipsychotic type, and duration of exposure
(28). These observations underscore
the complexity of weight dynamics in psychiatric populations and
highlight the need for individualized risk stratification.
Sex-based analysis revealed a higher adherence to
metformin therapy among female patients, although no statistically
significant difference in weight change between sexes was observed.
Recent literature suggests that sex may influence medication
adherence and health-seeking behaviors, with women often
demonstrating greater engagement in weight-related interventions
(29,30). However, adherence in the present
study was assessed using self-report and pill counts rather than
validated adherence instruments, limiting the strength of this
conclusion. Nonetheless, these findings highlight the importance of
considering psychosocial and behavioral factors when implementing
metabolic interventions in psychiatric care.
Overall, the results of the present study add to the
growing body of evidence supporting metformin as a safe and
effective adjunctive intervention for mitigating AAWG, particularly
when combined with lifestyle modification. Recent expert
commentaries and consensus statements have increasingly advocated
for earlier integration of metformin in high-risk patients, rather
than reserving its use as a last-line option after significant
weight gain has occurred (21,31). The
present findings provide further real-world support for this
evolving clinical approach.
Several limitations of the present study should be
acknowledged. The single-center design may limit generalizability,
and the 6-month follow-up period restricts assessment of long-term
metabolic outcomes. The observational, non-randomized and
non-blinded nature of the study introduces potential selection bias
and residual confounding. Metformin dosing was guided by routine
clinical practice, resulting in dose heterogeneity (500 mg to 1 g),
which may have influenced treatment response. Lifestyle
modifications were delivered as non-standardized counseling without
objective adherence monitoring, limiting the ability to isolate the
independent effects of metformin. Adherence assessment relied on
patient self-report and pill counts rather than validated
instruments, which may reduce precision, particularly in
gender-based comparisons.
Despite these limitations, the present study
provides valuable real-world evidence from a routine psychiatric
setting. The findings should be interpreted with appropriate
caution, and causal inferences cannot be definitively established.
Future well-designed randomized controlled trials with longer
follow-up are warranted to confirm these results, explore
gender-specific adherence patterns, and evaluate the long-term
cardiometabolic benefits of adjunctive metformin therapy.
In conclusion, the present prospective observational
study demonstrated that adjunctive metformin therapy, when combined
with lifestyle modifications, was associated with a statistically
significant reduction in mean body weight and BMI among patients
receiving antipsychotic treatment. Patients in the metformin group
experienced a decrease in mean body weight from 60.45 to 57.98 kg,
whereas those managed with lifestyle modifications alone showed an
increase from 58.24 to 60.94 kg over the study period. These
findings support the potential role of metformin in mitigating
antipsychotic-associated weight gain in routine clinical practice.
Although higher self-reported adherence to metformin therapy was
observed among female patients, adherence was not assessed using
validated instruments, and no statistically significant sex-based
differences in weight outcomes were identified. Given the
observational design, the findings should be interpreted with
caution, and further well-designed randomized controlled trials are
warranted to confirm these results and to evaluate long-term
metabolic outcomes.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
SS and MA conceived and designed the study, and
supervised the overall conduct of the research. SLE, MB, MAN, WFAO
and MN were involved in patient recruitment, data collection and
data processing. SS and MA performed the statistical analysis and
interpreted the results. SAH provided methodological input
regarding metabolic risk assessment and contributed to data
interpretation. All authors participated in drafting or critically
revising the manuscript for important intellectual content and
approved the final version. All authors agree to be accountable for
the integrity of the work. SS and MA confirm the authenticity of
all the raw data.
Ethics approval and consent to
participate
The study was conducted in accordance with the
approval of the Institutional Review Board of Deccan College of
Medical Sciences, Hyderabad, Telangana, India (IEC no.
2024/59/080). Written informed consent for publication was obtained
from all participants.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ahuja N: A Short Textbook of Psychiatry
(7th edition). Jaypee Brothers Medical Publishers, Ltd., New Delhi,
2011.
|
|
2
|
Allison DB, Mentore JL, Heo M, Chandler
LP, Cappelleri JC, Infante MC and Weiden PJ: Antipsychotic-induced
weight gain: A comprehensive research synthesis. Am J Psychiatry.
156:1686–1696. 1999.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Baptista T, Martínez J, Lacruz A, Rangel
N, Beaulieu S, Serrano A, Arapé Y, Martinez M, de Mendoza S, Teneud
L and Hernández L: Metformin for prevention of weight gain and
insulin resistance with olanzapine: A double-blind
placebo-controlled trial. Can J Psychiatry. 51:192–196.
2006.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Corcoran C and Jacobs TF: Metformin. In:
StatPearls. StatPearls Publishing, Treasure Island, FL, 2026.
|
|
5
|
Correll CU, Manu P, Olshanskiy V,
Napolitano B, Kane JM and Malhotra AK: Cardiometabolic risk of
second-generation antipsychotic medications during first-time use
in children and adolescents. JAMA. 302:1765–1773. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
De Hert M, Detraux J, van Winkel R, Yu W
and Correll CU: Metabolic and cardiovascular adverse effects
associated with antipsychotic drugs. Nat Rev Endocrinol. 8:114–126.
2011.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Correll CU, Solmi M, Veronese N, Bortolato
B, Rosson S, Santonastaso P, Thapa-Chhetri N, Fornaro M, Gallicchio
D, Collantoni E, et al: Prevalence, incidence and mortality from
cardiovascular disease in patients with pooled and specific severe
mental illness: A large-scale meta-analysis of 3,211,768 patients
and 113,383,368 controls. World Psychiatry. 16:163–180.
2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Tek C, Kucukgoncu S, Guloksuz S, Woods SW,
Srihari VH and Annamalai A: Antipsychotic-induced weight gain in
first-episode psychosis patients: A meta-analysis of differential
effects of antipsychotic medications. Early Interv Psychiatry.
10:193–202. 2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Vancampfort D, Firth J, Schuch FB,
Rosenbaum S, Mugisha J, Hallgren M, Probst M, Ward PB, Gaughran F,
De Hert M, et al: Sedentary behavior and physical activity levels
in people with schizophrenia, bipolar disorder and major depressive
disorder: A global systematic review and meta-analysis. World
Psychiatry. 16:308–315. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Schuch FB, Vancampfort D, Richards J,
Rosenbaum S, Ward PB and Stubbs B: Exercise as a treatment for
depression: A meta-analysis adjusting for publication bias. J
Psychiatr Res. 77:42–51. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
lSayed NA, Aleppo G, Aroda VR, Bannuru RR,
Brown FM, Bruemmer D, Collins BS, Das SR, Hilliard ME, Isaacs D, et
al: 10. Cardiovascular disease and risk management: Standards of
care in diabetes-2023. Diabetes Care. 46 (Suppl 1):S158–S190.
2023.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Velligan DI, Weiden PJ, Sajatovic M, Scott
J, Carpenter D, Ross R and Docherty JP: Expert Consensus Panel on
Adherence Problems in Serious and Persistent Mental Illness. The
expert consensus guideline series: Adherence problems in patients
with serious and persistent mental illness. J Clin Psychiatry. 70
(Suppl 4):S1–S48. 2009.PubMed/NCBI
|
|
13
|
Fiedorowicz JG, Miller DD, Bishop JR,
Calarge CA, Ellingrod VL and Haynes WG: Systematic review and
meta-analysis of pharmacological interventions for weight gain from
antipsychotics and mood stabilizers. Curr Psychiatry Rev. 8:25–36.
2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Jones HM and Pilowsky LS: Dopamine and
antipsychotic drug action revisited. Br J Psychiatry. 181:271–275.
2002.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Malin SK and Kashyap SR: Effects of
metformin on weight loss: Potential mechanisms. Curr Opin
Endocrinol Diabetes Obes. 21:323–329. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Schwinghammer TL, DiPiro JT, Ellingrod VL
and DiPiro CV: McGraw Hill Medical: Pharmacotherapy Handbook (11th
edition). AccessPharmacy, 2021. Available: https://accesspharmacy.mhmedical.com/book.aspx?bookID=3012.
|
|
17
|
McGrath J, Saha S, Welham J, El Saadi O,
MacCauley C and Chant D: A systematic review of the incidence of
schizophrenia: The distribution of rates and the influence of sex,
urbanicity, migrant status and methodology. BMC Med.
2(13)2004.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mizuno Y, Suzuki T, Nakagawa A, Yoshida K,
Mimura M, Fleischhacker WW and Uchida H: Pharmacological strategies
to counteract antipsychotic-induced weight gain and metabolic
adverse effects in schizophrenia: A systematic review and
meta-analysis. Schizophr Bull. 40:1385–1403. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Newcomer JW: Antipsychotic medications:
Metabolic and cardiovascular risk. J Clin Psychiatry. 68 (Suppl
4):S8–S13. 2007.PubMed/NCBI
|
|
20
|
Seifarth C, Schehler B and Schneider HJ:
Effectiveness of metformin on weight loss in non-diabetic
individuals with obesity. Exp Clin Endocrinol Diabetes. 121:27–31.
2013.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Jiang WL, Cai DB, Yin F, Zhang L, Zhao XW,
He J, Ng CH, Ungvari GS, Sim K, Hu ML, et al: Adjunctive metformin
for antipsychotic-induced dyslipidemia: a meta-analysis of
randomized, double-blind, placebo-controlled trials. Transl
Psychiatry. 10(117)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Rawani NS, Chan AW, Dursun SM and Baker
GB: The underlying neurobiological mechanisms of psychosis: Focus
on neurotransmission dysregulation, neuroinflammation, oxidative
stress, and mitochondrial dysfunction. Antioxidants (Basel).
13(709)2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Penninx BWJH and Lange SMM: Metabolic
syndrome in psychiatric patients: Overview, mechanisms, and
implications. Dialogues Clin Neurosci. 20:63–73. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
de Silva VA, Suraweera C, Ratnatunga SS,
Dayabandara M, Wanniarachchi N and Hanwella R: Metformin in
prevention and treatment of antipsychotic induced weight gain: A
systematic review and meta-analysis. BMC Psychiatry.
16(341)2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Pillinger T, McCutcheon RA, Vano L, Mizuno
Y, Arumuham A, Hindley G, Beck K, Natesan S, Efthimiou O, Cipriani
A and Howes OD: Comparative effects of 18 antipsychotics on
metabolic function in patients with schizophrenia, predictors of
metabolic dysregulation, and association with psychopathology: A
systematic review and network meta-analysis. Lancet Psychiatry.
7:64–77. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
American Diabetes Association Professional
Practice Committee. 9. Pharmacologic approaches to glycemic
treatment: Standards of care in diabetes-2024. Diabetes Care. 47
(Suppl 1):S158–S178. 2024.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Siskind DJ, Russell AW, Gamble C, Winckel
K, Mayfield K, Hollingworth S, Hickman I, Siskind V and Kisely S:
Treatment of clozapine-associated obesity and diabetes with
exenatide in adults with schizophrenia: A randomized controlled
trial (CODEX). Diabetes Obes Metab. 20:1050–1055. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Firth J, Siddiqi N, Koyanagi A, Siskind D,
Rosenbaum S, Galletly C, Allan S, Caneo C, Carney R, Carvalho AF,
et al: The Lancet Psychiatry Commission: A blueprint for protecting
physical health in people with mental illness. Lancet Psychiatry.
6:675–712. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Koomen LEM, van der Horst MZ, Deenik J and
Cahn W: Lifestyle interventions for people with a severe mental
illness living in supported housing: A systematic review and
meta-analysis. Front Psychiatry. 13(966029)2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Vancampfort D, Stubbs B, Mitchell AJ, De
Hert M, Wampers M, Ward PB, Rosenbaum S and Correll CU: Risk of
metabolic syndrome and its components in people with schizophrenia
and related psychotic disorders, bipolar disorder and major
depressive disorder: A systematic review and meta-analysis. World
Psychiatry. 14:339–347. 2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Dayabandara M, Hanwella R, Ratnatunga S,
Seneviratne S, Suraweera C and de Silva VA:
Antipsychotic-associated weight gain: Management strategies and
impact on treatment adherence. Neuropsychiatr Dis Treat.
13:2231–2241. 2017.PubMed/NCBI View Article : Google Scholar
|