Introduction
Cardiovascular disease (CVD) is the leading cause of
mortality among patients undergoing hemodialysis (HD), with
cardiovascular-related mortality being 10-20-fold higher patients
undergoing HD than in the general population (1). Among the non-traditional factors that
increase the risk of CVD in patients undergoing HD, are uremic
toxins. Kynurenine is one such toxin. It is the initial molecule in
the tryptophan breakdown through the kynurenine pathway. There are
three reasons for kynurenine accumulation in patients undergoing
HD. First, persistent inflammation, a characteristic of patients
undergoing HD, increases indoleamine 2,3-dioxygenase (IDO)-1
expression in various tissues, mainly in monocytes, which then
degrades tryptophan into kynurenine (2-4).
The second enzyme that catalyzes tryptophan degradation to
kynurenine is liver L-tryptophan 2,3-dioxygenase (TDO), and
experimental data indicate that TDO activity increases in chronic
kidney disease (CKD) (5). Finally,
circulating kynurenine levels increase in patients undergoing HD
due to reduced renal excretion (3,6).
Notably, since kynurenine is a protein-bound toxin, HD or
hemodiafiltration does not effectively lower its blood levels
(3,7). Although an HD session may reduce the
plasma free fraction of the kynurenine concentration by ~60%, 67%
protein binding precludes the elimination of total kynurenine,
which rapidly replenishes free kynurenine levels (7,8). As
multiple factors affect circulating kynurenine levels, the use of
the serum kynurenine-to-tryptophan ratio (KTR) as an indicator of
IDO-1 activity has been challenged (9), particularly in patients undergoing HD
(10). More precisely, in a previous
study, in a cohort of 228 patients undergoing HD, plasma KTR and
peripheral blood mononuclear cell IDO-1 mean fluorescence intensity
(MFI) were higher than those in the healthy controls, but they did
not correlate with each other. Only the IDO-1 MFI was associated
with immune suppression, as evidenced by its negative correlation
with skin induration diameter in tuberculin skin testing and its
positive correlation with clinical infection (10). Once produced, kynurenine exerts its
effects by activating the aryl hydrocarbon receptor (AhR), thereby
altering multiple cellular functions (11-15).
In the general population, kynurenine pathway
metabolites have been associated with the incidence of heart
failure (HF) and atrial fibrillation (AF), as well as with worse
outcomes in patients with existing HF (16-18).
In patients undergoing HD, kynurenine pathway metabolites have been
linked to inflammation and prevalent atherosclerotic CVD (19), carotid intima-media thickness
(20,21), time to a new CV event (21), and a 1-year risk of mortality
(22). However, other researchers
have failed to find a connection between kynurenine pathway
metabolites and new CV events or overall mortality (7). Of note, a recent study on patients with
CKD not undergoing HD found that kynurenine levels were associated
with both the prevalence and incidence of HF (23).
Further mechanistic studies are required to confirm
the role of kynurenine in CKD-related complications. Nonetheless,
some data already exist. In a previous study on 116 patients with
CKD stage 3-5D, the sera of the patients exhibited potent
AhR-activating potential compared with the healthy controls
(24). Additionally, the expression
of the AhR target gene, CYP1A1, was upregulated in whole blood from
patients with CKD. Survival analysis revealed that cardiovascular
events were more common in patients with CKD with AhR-activating
potential above the median (24).
Another study analyzed samples from two large multicenter studies;
one on patients with end-stage kidney disease and another on
patients with CKD stage 2-3(25). In
the first study, patients with arteriovenous fistula thrombosis had
elevated kynurenine levels and AhR activity. In the second study,
patients with postangioplasty thrombosis had higher kynurenine
levels and AhR-activating potential. Mechanistic analyses in
vivo revealed that kynurenine promotes thrombosis following
vascular injury in an animal model and exerts its effects via AhR
pathways. In primary human vascular smooth muscle cell cultures,
kynurenine elevated AhR activity and tissue factor expression
(25).
Considering the critical role of CVD in the
mortality and morbidity of patients undergoing HD, it is crucial to
investigate the underlying pathophysiological pathways responsible
for CVD and, if possible, identify biomarkers for the early
detection of CVD in this population. Studies conducted in patients
undergoing HD have assessed kynurenine levels in relation to
prevalent atherosclerotic disease, defined collectively as coronary
heart disease (CHD), stroke, or peripheral arterial disease
(19,20); incident atherosclerotic events and
heart failure (21); incident
atherosclerotic disease and all-cause mortality (7); or overall mortality (22). However, although CVDs have
overlapping pathogenic pathways, these mechanisms are not
completely identical. Therefore, the present study examined whether
serum kynurenine levels are independently associated with prevalent
CHD, HF or AF among patients undergoing HD. To the best of our
knowledge, this is the first study to assess the association of
kynurenine with prevalent HF and AF among patients undergoing
HD.
Patients and methods
Patients
A total of 119 patients undergoing HD participated
in the present study. The mean age of the patients was 65.98±11.87
years, including 87 males and 32 females. The causes of end-stage
kidney disease were diabetic nephropathy (n=29), hypertension
(n=24), primary glomerulonephritis (n=22), cardiorenal syndrome
(n=7), autosomal dominant polycystic kidney disease (n=6),
secondary focal segmental glomerulosclerosis (n=6), vasculitis
(n=4), obstructive nephropathy (n=2), analgesic nephropathy (n=1)
and unknown (n=18).
In total, 41 patients had diabetes mellitus, 103
patients had hypertension, 41 patients had a history of CHD, 33
patients had AF, and, according to a recent, within-3-months,
transthoracic echocardiogram, 34 patients were diagnosed with HF
with preserved ejection fraction (HFpEF), while 27 patients were
diagnosed with HF with reduced ejection fraction (HFrEF). CHD was
ascertained by coronary artery angiography performed for angina
symptoms or after a myocardial infarction. The majority of patients
received anti-hypertensive medications, and statins were prescribed
to 79 of these patients. All patients with CHD were on clopidogrel
or aspirin, along with a β-blocker. Patients with HFrEF received a
β-blocker and a low-dose angiotensin receptor blocker. Those with
AF were prescribed a β-blocker and anticoagulation. The policy of
the HD unit of the University of Thessaly is to avoid
anti-inflammatory medications. The attending nephrologists
determined the need for phosphate binders, vitamin D analogs,
calcimimetics, erythropoietin and intravenous iron. All patients
had been undergoing HD for at least 6 months prior to inclusion.
Each patient underwent 4-h HD sessions, three times a week, using
polysulfone dialyzers and bicarbonate-based dialysate with calcium
concentrations of either 1.25 or 1.5 mmol/l.
The exclusion criteria included active infection,
autoimmune disease, malignancy, liver pathology, or treatment with
cytotoxic, immunosuppressive, or corticosteroid medications within
the prior 6 months. The clinical, laboratory and demographic
features of the patients undergoing HD are presented in Table I.
 | Table ICharacteristics of the patients
undergoing hemodialysis. |
Table I
Characteristics of the patients
undergoing hemodialysis.
| Characteristic | N | Median | 25-75%
percentile |
|---|
| Age (years) | 119 | 67 | 59-76 |
| Duration of HD
treatment (months) | 119 | 42 | 22-42 |
| Males/females | 87/32 | | |
| Diabetes mellitus
(yes/no) | 44/75 | | |
| Hypertension
(yes/no) | 103/14 | | |
| Coronary heart
disease (yes/no) | 41/78 | | |
| HFrEF/HFpEF/no
HF | 27/34/58 | | |
| Atrial fibrillation
(yes/no) | 33/86 | | |
| White blood cell
count (c/µl) | 119 | 7,800 | 5,360-8,500 |
| Neutrophils
(c/µl) | 119 | 4,310 | 3,277-6,120 |
| Lymphocytes
(c/µl) | 119 | 1,725 | 1,236-2,064 |
|
Neutrophil-to-lymphocyte ratio | 119 | 3.039 | 1.839-3.778 |
| Hemoglobin
(g/dl) | 119 | 11.7 | 11.2-12.4 |
| Platelets
(c/µl) | 119 | 199,000 |
162,000-242,000 |
| Creatinine
(mg/dl) | 119 | 6.0 | 4.7-7.5 |
| Urea (mg/dl) | 119 | 122 | 107-144 |
| Urea reduction rate
(%) | 119 | 67.797 | 63.115-72.727 |
| Residual diuresis
(ml) | 119 | 0 | 0-600 |
| Body mass index
(kg/m²) | 119 | 25.333 | 22.773-29.201 |
| Protein catabolic
rate (g/kg/day) | 119 | 0.880 | 0.715-0.990 |
| Albumin (g/dl) | 119 | 3.6 | 3.5-3.8 |
| Cholesterol
(mg/dl) | 119 | 136.0 | 100.0-153.0 |
| Triglyceride
(mg/dl) | 119 | 109.0 | 82.0-167.0 |
| Calcium
(mg/dl) | 119 | 9.1 | 8.9-9.5 |
| Phosphorous
(mg/dl) | 119 | 5.1 | 4.5-5.9 |
| Parathyroid hormone
(pg/ml) | 119 | 240.81 | 155.61-471.80 |
| Alkaline
phosphatase (U/l) | 119 | 186.0 | 147.0-224.0 |
| SGOT (U/l) | 119 | 12.0 | 9.0-15.0 |
| SGPT (U/l) | 119 | 9.0 | 6.0-15.0 |
| Ferritin
(ng/ml) | 119 | 120 | 58.9-228.4 |
| TSAT (%) | 119 | 16.748 | 12.136-22.434 |
| CRP (mg/dl) | 119 | 0.88 | 0.69-1.4 |
| CRP >1 mg/dl
(yes/no) | 56/63 | | |
| Kynurenine
(µM) | 119 | 7.562 | 6.317-9.572 |
| Tryptophan
(µM) | 119 | 31.17 | 26.27-44.56 |
|
Kynurenine-to-tryptophan ratio | 119 | 0.235 | 0.187-0.326 |
A control group of 25 healthy individuals (mean age,
64.60±6.69 years; 16 males and 9 females) was included following a
review of their medical records and a physical examination.
Written informed consent was obtained from each
participant, and the study protocol was approved by the Ethics
Committee of the Faculty of Medicine, University of Thessaly,
Larissa, Greece (approval no. 558/10-2-2017).
Sample collection
Blood samples were collected at the beginning of the
second HD session of the week, and serum was stored at -80˚C. Serum
total kynurenine levels were measured using the Kynurenine ELISA
kit (cat. no. KA6140, Abnova Corporation), which has a detection
limit of 0.22 µM and a calibration range of 0.30-48 µM. Serum total
tryptophan levels were measured using the Tryptophan ELISA kit
(cat. no. BA E-2700, Labor Diagnostika Nord GmbH & CoKG), which
has a detection limit of 3.18 µM and a calibration range of 0-1223
µM. All other parameters were part of routine laboratory
assessments conducted alongside serum collection for kynurenine and
tryptophan measurement.
Statistical analysis
Statistical analysis was performed using IBM SPSS
Statistics version 29 (IBM Corp.) and JASP version 0.95.4
(University of Amsterdam, Amsterdam, The Netherlands). As the
one-sample Kolmogorov-Smirnov test revealed that the analyzed
variables were not normally distributed, non-parametric methods
were employed. Specifically, for group comparisons, the
Mann-Whitney U test was used, with data presented as the median
(interquartile range). A post hoc power analysis for the
kynurenine-related Mann-Whitney U test was performed using the
observed rank-biserial correlation (r), group sample sizes, and a
two-tailed significance level of α=0.05 to estimate achieved
statistical power under the nonparametric framework. Additionally,
Spearman's Rho was calculated to assess correlations between
continuous variables, while the Chi-squared test, with Fisher's
exact test when needed, were applied to evaluate the associations
between categorical variables. Receiver operating characteristic
(ROC) curve analysis was performed to assess the diagnostic utility
of kynurenine. However, as the pathogenesis of the evaluated
diseases is multifactorial, a single factor is unlikely to achieve
good discrimination (AUC >0.8). Therefore, the possible
pathogenetic role of kynurenine was evaluated using binary logistic
regression, including all factors associated with the evaluated
diseases. Multicollinearity among predictors was evaluated using
the variance inflation factor (VIF). Given the small sample size,
VIF values <2.5 were considered acceptable. To assess whether
kynurenine enhances discrimination of the evaluated diseases, along
with other confounding factors, the model fit was re-evaluated
using the change of Χ2 (ΔΧ2) and its
statistical significance, as well as AUC and Nagelkerke
R2, for each logistic regression analysis after removing
kynurenine from the model. A value of P<0.05 was considered to
indicate a statistically significant difference.
Results
Kynurenine levels in healthy
individuals and in patients undergoing HD
Serum kynurenine levels were significantly higher in
patients undergoing HD than in the healthy individuals [7.56 (3.26)
µM vs. 2.33 (1.32) µM, respectively; P<0.001] (Table II). A post hoc power analysis was
conducted using the observed effect size derived from the
Mann-Whitney U test (r=0.989; approximated Cohen's d=5.34), α=0.05,
and the achieved sample sizes (n1=25,
n2=119). The calculated power was 1.00 (Table III).
 | Table IIKynurenine, tryptophan and
kynurenine-to-tryptophan ratio in healthy controls and patients
undergoing hemodialysis, and subgroups of patients undergoing
hemodialysis. |
Table II
Kynurenine, tryptophan and
kynurenine-to-tryptophan ratio in healthy controls and patients
undergoing hemodialysis, and subgroups of patients undergoing
hemodialysis.
| Comparison | Kynurenine
(µM) | P-value | Tryptophan
(µM) | P-value |
Kynurenine-to-tryptophan ratio | P-value |
|---|
| HD patients vs.
healthy controls | 7.56 (3.26) vs.
2.33 (1.32) |
<0.001 | 31.17 (18.67) vs.
53.66 (18.87) |
<0.001 | (0.235 (0.143) vs.
0.05 (0.027) |
<0.001 |
| Patients undergoing
HD | | | | | | |
|
Males vs.
females | 7.53 (2.84) vs.
8.34 (4.27) | 0.801 | 31.49 (15.83) vs.
30.39 (28.81) | 0.584 | 0.239 (0.127) vs.
0.218 (0.189) | 0.540 |
|
Diabetes
mellitus, yes vs. no | 6.91 (3.86) vs.
7.60 (3.43) | 0.062 | 31.79 (19.74) vs.
30.83 (17.97) | 0.916 | 0.206 (0.167) vs.
0.242 (0.139) | 0.079 |
|
Hypertension,
yes vs. no | 7.60 (3.15) vs.
7.23 (3.10) | 0.271 | 30.83 (19.39) vs.
34.85 (8.07) | 0.312 | 0.236 (0.154) vs.
0.207 (0.064) | 0.210 |
|
Coronary
heart disease, yes vs. no | 6.87 (3.60) vs.
8.05 (3.14) | 0.345 | 36.86(19.99) vs.
28.62 (15.83) | 0.001 | 0.194 (0.095) vs.
0.249 (0.126) |
<0.001 |
|
Heart
failure, yes vs. no | 7.37 (3.53) vs.
7.62 (3.53) | 0.312 | 32.35 (18.50) vs.
29.82 (18.71) | 0.780 | 0.232 (0.147) vs.
0.242 (0.143) | 0.951 |
|
HFrEF, yes
vs. no | 8.78 (3.74) vs.
7.56 (3.32) | 0.030 | 28.48 (20.14) vs.
31.33 (18.6) | 0.741 | 0.246 (0.194) vs.
0.231 (0.121) | 0.201 |
|
Atrial
fibrillation, yes vs. no | 9.15 (3.57) vs.
7.30 (3.34) | 0.011 | 28.83 (18.68) vs.
31.90 (18.58) | 0.621 | 0.254 (0.158) vs.
0.226 (0.119) | 0.024 |
|
CRP >1
mg/dl, yes vs. no | 6.68 (4.13) vs.
8.08 (2.70) | 0.024 | 29.55 (18.98) vs.
32.08 (18.15) | 0.560 | 0.228 (0.060) vs.
0.249 (0.182) | 0.163 |
 | Table IIIPost hoc power analyses for
kynurenine-related Mann-Whitney U tests. |
Table III
Post hoc power analyses for
kynurenine-related Mann-Whitney U tests.
| Comparison | Observed effect
size (r) | Cohen's d
(approx.) | Sample size
(n) | Significance level
(α) | Post hoc power
(1-β) |
|---|
| Kynurenine:
Controls vs. patients undergoing HD | 0.989 | 5.34 | Control: 25 | 0.05 | 1.00 |
| | | | HD patients:
119 | | |
| Kynurenine: HFrEF
vs. no HFrEF | 0.275 | 0.57 | HFrEF: 27 | 0.05 | 0.77 |
| | | | No HFrEF: 92 | | |
| Kynurenine: AF vs.
no AF | 0.303 | 0.64 | AF: 33 | 0.05 | 0.88 |
| | | | No AF: 86 | | |
Notably, serum tryptophan levels were significantly
lower in patients undergoing HD than in the healthy individuals
[31.17 (18.67) µM vs. 53.66 (18.87) µM, respectively; P<0.001].
The KTR was higher in patients undergoing HD [0.235 (0.143) vs.
0.05 (0.027); P<0.001] (Table
II).
In patients undergoing HD, sex, diabetes mellitus,
hypertension, CHD and HF as a whole did not affect kynurenine
levels. However, kynurenine levels were higher in patients
undergoing HD with HFrEF [8.78 (3.74) Μm vs. 7.56 (3.32) µM;
P=0.030] (Table II). A post hoc
power analysis was conducted using the observed effect size derived
from the Mann-Whitney U test (r=0.275; approximated Cohen's
d=0.57), α=0.05, and the achieved sample sizes (n1=27,
n2=92) (Table III). The
calculated power was 0.77. Kynurenine levels were also higher in
patients with AF [9.15 (3.57) µM vs. 7.30 (3.34) µM; P=0.011]
(Table II). A post hoc power
analysis was conducted using the observed effect size derived from
the Mann-Whitney U test (r=0.303; approximated Cohen's d=0.64),
α=0.05, and the achieved sample sizes (n1=33,
n2=86). The calculated power was 0.88 (Table III).
In patients with C-reactive protein (CRP) levels
>1 mg/dl, serum kynurenine levels were lower [6.68 (4.13) µM vs.
8.08 (2.70) µM; P=0.024]. Notably, serum tryptophan levels did not
differ between patients with or without HFrEF [28.48 (20.14) µM vs.
31.33 (18.6) µM, respectively; P=0.741] or between patients with or
without AF [28.83 (18.68) µM vs. 31.90 (18.58) µM, respectively;
P=0.621]. KTR did not differ between patients undergoing HD with or
without HFrEF [0.246 (0.194) vs. 0.231 (0.121), respectively;
P=0.201]. However, KTR was higher in patients undergoing HD with AF
[0.254 (0.158) vs. 0.226 (0.119); P=0.024] (Table II).
Among the factors analyzed in the present study and
shown in Table I, serum kynurenine
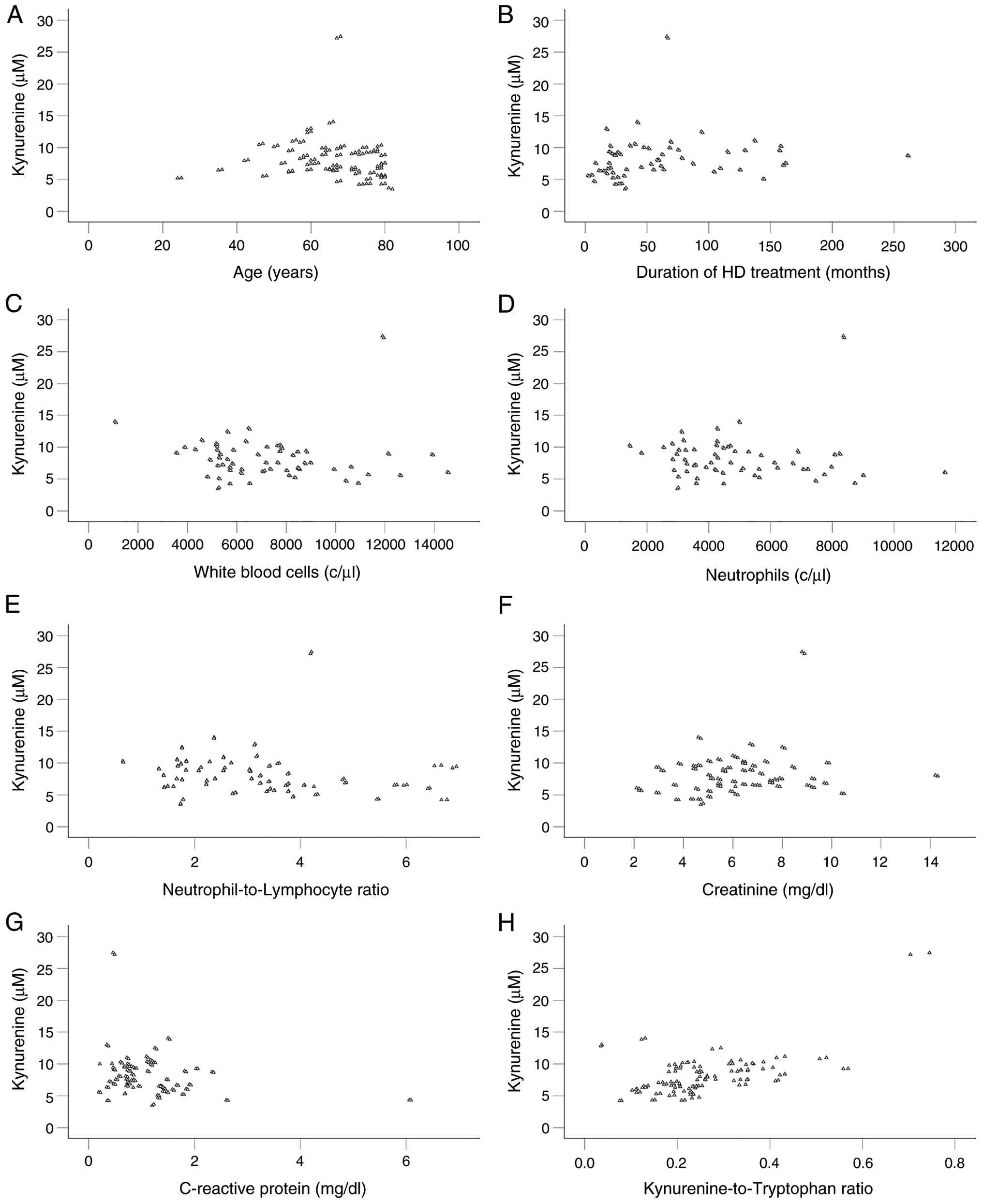
levels correlated with age (Rho=-0.245, P=0.007), the duration of
HD (Rho=0.330, P<0.001), white blood cell count (Rho=-0.255,
P=0.005), neutrophil count (Rho=-0.228, P=0.013),
neutrophil-to-lymphocyte ratio (Rho=-0.265, P=0.004), creatinine
(Rho=0.183, P=0.046) and CRP (Rho=-0.268, P=0.003). Notably,
kynurenine did not correlate with tryptophan (Rho=0.143, P=0.126),
whereas a positive correlation was observed with KRT (Rho=0.486,
P<0.001) (Table IV).
Scatterplots illustrating kynurenine and its statistically
significant correlated factors are presented in Fig. 1.
 | Table IVCorrelations of kynurenine with other
variables in patients undergoing hemodialysis. |
Table IV
Correlations of kynurenine with other
variables in patients undergoing hemodialysis.
| | Rho | P-value |
|---|
| Age | -0.245 | 0.007 |
| Duration of HD
treatment | 0.330 |
<0.001 |
| White blood
cells | -0.255 | 0.005 |
| Neutrophils | -0.228 | 0.013 |
| Lymphocytes | 0.070 | 0.451 |
|
Neutrophil-to-Lymphocyte ratio | -0.265 | 0.004 |
| Hemoglobin | -0.128 | 0.165 |
| Platelets | -0.110 | 0.235 |
| Creatinine | 0.183 | 0.046 |
| Urea | 0.034 | 0.714 |
| Urea reduction
ratio | -0.021 | 0.863 |
| Residual
diuresis | -0.008 | 0.934 |
| Body mass
index | 0.100 | 0.277 |
| Protein catabolic
ratio | 0.176 | 0.055 |
| Albumin | 0.100 | 0.281 |
| Cholesterol | -0.172 | 0.061 |
| Triglycerides | 0.071 | 0.442 |
| Calcium | 0.045 | 0.625 |
| Phosphoros | -0.152 | 0.099 |
| Parathyroid
hormone | 0.142 | 0.122 |
| Alkaline
phosphatase | 0.125 | 0.176 |
| SGOT | 0.059 | 0.521 |
| SGPT | 0.114 | 0.216 |
| Ferritin | 0.086 | 0.353 |
| Transferrin
saturation | -0.109 | 0.239 |
| C-reactive
protein | -0.268 | 0.003 |
| Tryptophan | 0.143 | 0.126 |
|
Kynurenine-to-tryptophan ratio | 0.486 |
<0.001 |
Kynurenine as a determinant of HFrEF
in patients undergoing HD
In addition to elevated kynurenine levels, patients
undergoing HD with HFrEF were on HD treatment for a longer period
of time [66 (54.0) months vs. 32 (49.5) months; P=0.034], had an
elevated neutrophil-to-lymphocyte ratio [3.600 (1.713) vs. 2.706
(1.725); P=0.026] and had lower triglyceride levels [87.0 (48.0)
vs. 114.5 (78.5) mg/dl; P=0.012]. HFrEF was associated with the
male sex (Pearson χ2=6.743, P=0.009) and CHD (Pearson
χ2=29.027, P<0.001) (Table
V).
 | Table VCharacteristics of patients
undergoing hemodialysis with and without heart failure with a
reduced ejection fraction. |
Table V
Characteristics of patients
undergoing hemodialysis with and without heart failure with a
reduced ejection fraction.
| | No HFrEF
(n=92) | HFrEF (n=27) | P-value |
|---|
| Age (years) | 67 (16.75) | 67 (15.0) | 0.706 |
| Duration of HD
treatment (months) | 32 (49.5) | 66 (54.0) | 0.034 |
| Males/females | 62/30 | 25/2 | 0.009 |
| Diabetes mellitus
(yes/no) | 36/56 | 8/19 | 0.369 |
| Hypertension
(yes/no) | 78/12 | 25/2 | 0.517 |
| Coronary heart
disease (yes/no) | 20/72 | 21/6 |
<0.001 |
| Atrial fibrillation
(yes/no) | 22/70 | 11/16 | 0.086 |
| White blood cell
count (c/µl) | 7,205
(2,817.5) | 6,890
(3,980.0) | 0.146 |
| Neutrophils
(c/µl) | 4,379.5
(2,369.3) | 4,226
(3,472.0) | 0.814 |
| Lymphocytes
(c/µl) | 1,790(703) | 1,645(974) | 0.075 |
|
Neutrophil-to-lymphocyte ratio | 2.706 (1.725) | 3.600 (1.713) | 0.026 |
| Hemoglobin
(g/dl) | 11.80 (1.20) | 11.70 (1.40) | 0.228 |
| Platelets
(c/µl) | 199,000
(74,500) | 202,000
(117,000) | 0.914 |
| Creatinine
(mg/dl) | 5.90 (2.97) | 6.10 (1.70) | 0.671 |
| Urea (mg/dl) | 122.5 (34.5) | 122.0 (37.0) | 0.542 |
| Urea reduction rate
(%) | 67.93 (9.75) | 67.68 (9.43) | 0.785 |
| Residual diuresis
(ml) | 0(600) | 300(600) | 0.464 |
| Body mass index
(kg/m2) | 25.183 (6.29) | 25.432 (6.07) | 0.310 |
| Protein catabolic
rate (g/kg/day) | 0.852 (0.263) | 0.926 (0.229) | 0.207 |
| Albumin (g/dl) | 3.65 (0.3) | 3.60 (0.2) | 0.336 |
| Cholesterol
(mg/dl) | 139 (53.5) | 124 (55.0) | 0.050 |
| Triglyceride
(mg/dl) | 114.5 (78.5) | 87.0 (48.0) | 0.012 |
| Calcium
(mg/dl) | 9.1 (0.4) | 9.2 (1.2) | 0.506 |
| Phosphorous
(mg/dl) | 5.1 (1.47) | 5.2 (1.30) | 0.295 |
| Parathyroid hormone
(pg/ml) | 285.66 (300.4) | 208.31 (238.5) | 0.243 |
| Alkaline
phosphatase (U/l) | 192 (97.5) | 179 (57.0) | 0.650 |
| SGOT (U/l) | 12 (6.75) | 12 (4.00) | 0.944 |
| SGPT (U/l) | 9(8) | 9(11) | 0.325 |
| Ferritin
(ng/ml) | 114.3 (145.82) | 141.1 (254.60) | 0.542 |
| Transferrin
saturation (%) | 17.17 (11.82) | 14.52 (4.82) | 0.129 |
| CRP (mg/dl) | 1.0 (0.72) | 0.85 (0.71) | 0.854 |
| CRP >1 mg/dl
(yes/no) | 46/46 | 10/17 | 0.235 |
| Kynurenine
(µM) | 7.52 (3.41) | 8.78 (3.74) | 0.030 |
| Tryptophan
(µM) | 31.33 (18.6) | 28.48 (20.14) | 0.741 |
|
Kynurenine-to-tryptophan ratio | 0.231 (0.121) | 0.246 (0.194) | 0.201 |
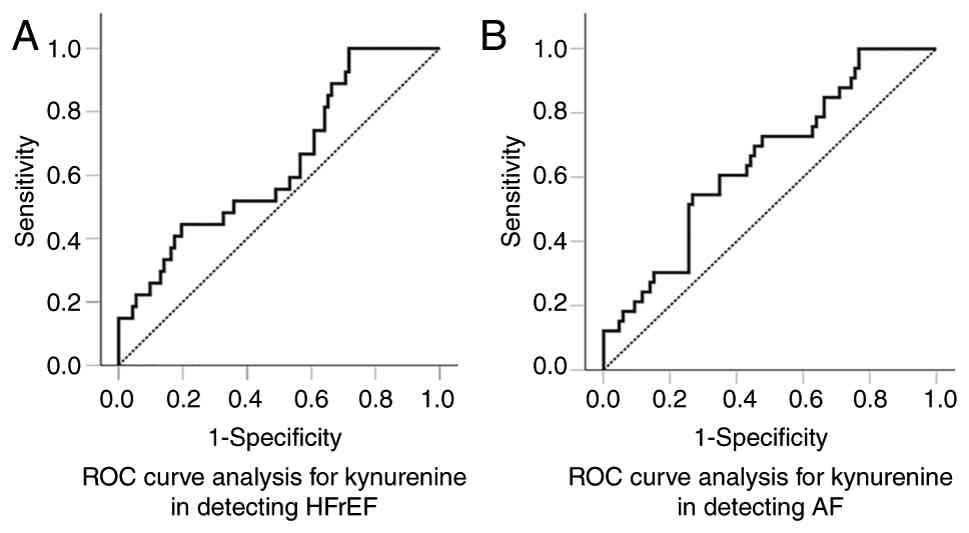
ROC curve analysis revealed a modest discriminative
ability of kynurenine for HFrEF, with an AUC of 0.637 [95%
confidence interval (CI), 0.520-0.754; P=0.022] (Fig. 2A). At the optimal cut-off of 6.11 µM,
the test had 100% sensitivity and 28.3% specificity, indicating
excellent rule-out capability but poor rule-in performance. Binary
logistic regression, including kynurenine and other factors linked
to HFrEF, as demonstrated in in Table
VI, indicated that kynurenine was a significant predictor of
HFrEF in patients undergoing HD. For every 1 µM increase in the
serum kynurenine level, the odds of HFrEF increased by 65% [odds
ratio (OR), 1.649; 95% CI, 1.141-2.381; P=0.008] (Table VI). Removing kynurenine from the
model decreased model fit (Χ2 decreases,
ΔΧ2=20.722, P<0.001), the AUC from 0.904 to 0.859,
and Nagelkerke R2 from 0.581 to 0.449. Thus, kynurenine
is unlikely to serve as a standalone diagnostic marker for HFrEF,
reflecting the multifactorial nature of the disease; however, it
remains a risk factor that may contribute to its pathogenesis.
 | Table VIBinary logistic regression analysis
of factors associated with HFrEF. |
Table VI
Binary logistic regression analysis
of factors associated with HFrEF.
| | | 95% CI for OR | |
|---|
| Comparison | B | S.E. | Sig. | OR | Lower | Upper | VIF |
|---|
| Duration of HD
treatment | 0.003 | 0.006 | 0.642 | 1.003 | 0.991 | 1.014 | 1.263 |
| Female sex | -0.105 | 0.954 | 0.913 | 0.901 | 0.139 | 5.838 | 1.295 |
| Coronary heart
disease | 3.535 | 0.839 |
<0.001 | 34.312 | 6.628 | 177.628 | 1.864 |
|
Neutrophile-to-lymphocyte ratio | 0.388 | 0.251 | 0.122 | 1.474 | 0.901 | 2.412 | 1.526 |
| Triglycerides (per
mg/dl) | -0.022 | 0.007 | 0.003 | 0.978 | 0.965 | 0.992 | 1.380 |
| Kynurenine (per
µM) | 0.500 | 0.188 | 0.008 | 1.649 | 1.141 | 2.381 | 1.809 |
Kynurenine as a determinant of AF in
patients undergoing HD
In addition to elevated kynurenine levels, patients
with AF undergoing HD were older [72 (13.5) years vs. 66 (18.0)
years; P=0.033], had an elevated white blood cell count [8,300
(2,245.0) vs. 6,220 (2,802.5) c/µl; P=0.006], neutrophil count
[5647.0 (2244.0) vs. 4194.5 (1868.5) c/µl; P<0.001] and
neutrophil-to-lymphocyte ratio [3.17 (2.06) vs. 2.72 (1.96);
P=0.024]. In addition, they had lower hemoglobin [11.40 (2.30) vs.
11.95 (1.20) g/dl, P=0.003], lower phosphorus [4.8 (1.28) vs. 5.2
(1.53) mg/dl; P=0.030] and higher ferritin [164.4 (258.15) vs.
106.9 (129.7) ng/ml, P=0.043] levels, and a lower transferrin
saturation [14.95 (9.11) vs. 17.13 (10.99)%; P=0.042]. As noted,
KTR was higher in patients with AF undergoing HD (Table VII).
 | Table VIICharacteristics of hemodialysis
patients with and without atrial fibrillation. |
Table VII
Characteristics of hemodialysis
patients with and without atrial fibrillation.
| Characteristic | No Atrial
fibrillation (n=86) | Atrial fibrillation
(n=33) | P-value |
|---|
| Age (years) | 66 (18.0) | 72 (13.5) | 0.033 |
| Duration of HD
treatment (months) | 33.5 (54.25) | 48.0 (79.00) | 0.276 |
| Males/females | 26/60 | 6/27 | 0.184 |
| Diabetes mellitus
(yes/no) | 30/56 | 14/19 | 0.446 |
| Hypertension
(yes/no) | 74/10 | 29/4 | 1.000 |
| Coronary heart
disease (yes/no) | 28/58 | 13/20 | 0.482 |
| HFrEF (yes/no) | 16/70 | 11/22 | 0.086 |
| White blood cell
count (c/µl) | 6,220
(2,802.5) | 8,300
(2,245.0) | 0.006 |
| Neutrophils
(c/µl) | 4,194.5
(1,868.5) | 5,647.0
(2,244.0) |
<0.001 |
| Lymphocytes
(c/µl) | 1,678.5
(841.5) | 1,821.0
(709.0) | 0.549 |
|
Neutrophil-to-lymphocyte ratio | 2.72 (1.96) | 3.17 (2.06) | 0.024 |
| Hemoglobin
(g/dl) | 11.95 (1.20) | 11.40 (2.30) | 0.003 |
| Platelets
(c/µl) | 200500 (77250) | 196000 (92000) | 0.523 |
| Creatinine
(mg/dl) | 6.10 (2.72) | 5.80 (3.20) | 0.553 |
| Urea (mg/dl) | 122.5 (39.5) | 122.0 (35.5) | 0.722 |
| Urea reduction rate
(%) | 68.19 (10.50) | 67.33 (7.14) | 0.854 |
| Residual diuresis
(ml) | 0(700) | 0(500) | 0.449 |
| Body mass index
(kg/m2) | 25.24 (6.35) | 26.81 (6.53) | 0.254 |
| Protein catabolic
rate (g/kg/day) | 0.848 (0.268) | 0.898 (0.252) | 0.219 |
| Albumin (g/dl) | 3.7 (0.3) | 3.6 (0.2) | 0.073 |
| Cholesterol
(mg/dl) | 137.5 (46.5) | 124.0 (66.0) | 0.157 |
| Triglyceride
(mg/dl) | 109.5 (85.5) | 109.0 (98.5) | 0.856 |
| Calcium
(mg/dl) | 9.1 (0.58) | 9.1 (0.8) | 0.260 |
| Phosphorus
(mg/dl) | 5.2 (1.53) | 4.8 (1.28) | 0.030 |
| Parathyroid hormone
(pg/ml) | 240.31 (196.9) | 300.10 (411.2) | 0.652 |
| Alkaline
phosphatase (U/l) | 175.0 (82.5) | 198.0 (57.5) | 0.854 |
| SGOT (U/l) | 12.0 (6.0) | 13.0 (4.5) | 0.128 |
| SGPT (U/l) | 9.0 (9.25) | 10.0 (8.5) | 0.793 |
| Ferritin
(ng/ml) | 106.9 (129.7) | 164.4 (258.15) | 0.043 |
| Transferrin
saturation (%) | 17.13 (10.99) | 14.95 (9.11) | 0.042 |
| CRP (mg/dl) | 0.81 (0.66) | 1.10 (0.83) | 0.181 |
| CRP >1 mg/dl
(yes/no) | 38/48 | 18/15 | 0.311 |
| Kynurenine
(µM) | 7.23 (3.51) | 9.15 (3.57) | 0.011 |
| Tryptophan
(µM) | 31.90 (18.58) | 28.83 (18.68) | 0.621 |
|
Kynurenine-to-tryptophan ratio | 0.226 (0.119) | 0.254 (0.158) | 0.024 |
ROC curve analysis demonstrated a modest
discriminative ability of kynurenine for AF, with an AUC of 0.652
(95% CI, 0.545-0.758; P=0.005) (Fig.
2B). At the optimal cut-off value of 9.0 µM, sensitivity was
54.5%, and specificity was 73.3%, indicating moderate diagnostic
performance without strong rule-in or rule-out. Binary logistic
regression, including kynurenine and other factors linked to AF,
demonstrated in Table VIII,
revealed that kynurenine was a significant predictor of AF in
patients undergoing HD. For each 1 µM increase in serum kynurenine
level, the odds of AF increase by 37% (OR, 1.372; 95% CI,
1.016-1.853; P=0.039) (Table
VIII). Removing kynurenine and KTR from the model decreased
model fit (Χ2 decreases, ΔΧ2=13.817,
P<0.001), the AUC from 0.871 to 0.814, and Nagelkerke
R2 from 0.498 to 0.371. Thus, kynurenine is unlikely to
serve as a standalone diagnostic marker for AF, which is consistent
with the multifactorial nature of the disease; however, it appears
to be a risk factor that may contribute to the pathogenesis of
AF.
 | Table VIIIBinary logistic regression analysis
of factors associated with atrial fibrillation. |
Table VIII
Binary logistic regression analysis
of factors associated with atrial fibrillation.
| | | 95% CI for OR | |
|---|
| | B | S.E. | Sig. | OR | Lower | Upper | VIF |
|---|
| Age (per year) | 0.092 | 0.035 | 0.009 | 1.096 | 1.023 | 1.175 | 1.363 |
|
Neutrophil-to-lymphocyte ratio | 0.374 | 0.211 | 0.077 | 1.453 | 0.960 | 2.198 | 1.229 |
| Hemoglobin (per
g/dl) | -1.168 | 0.423 | 0.006 | 0.311 | 0.136 | 0.712 | 1.173 |
| Phosphorus (per
mg/dl) | -0.537 | 0.352 | 0.127 | 0.584 | 0.293 | 1.165 | 1.185 |
| Ferritin (per
ng/ml) | 0.003 | 0.001 | 0.070 | 1.003 | 1.000 | 1.006 | 1.461 |
| Transferrin
saturation (%) | -0.072 | 0.041 | 0.076 | 0.930 | 0.859 | 1.007 | 1.521 |
| Kynurenine (per
µM) | 0.316 | 0.153 | 0.039 | 1.372 | 1.016 | 1.853 | 1.465 |
|
Kynurenine-to-tryptophan ratio | 4.225 | 2.783 | 0.129 | 68.401 | 0.292 | 16008 | 1.400 |
Discussion
Considering the high CV morbidity and mortality
rates in patients undergoing HD (1),
the present study investigated the impact of the uremic toxin
kynurenine on CVD by measuring its serum levels in patients
undergoing HD with or without CHD, HF or AF.
As expected, an increased kynurenine production
(2-5),
reduced renal elimination (3,6) and the
inability of HD to achieve sufficient kynurenine clearance
(3,7)
led to significantly higher kynurenine levels in patients
undergoing HD compared to healthy volunteers. Notably, although
previous studies have reported a positive correlation between
kynurenine pathway metabolites and inflammation (3,19,20),
others have failed to detect such an association (6,7). The
present study observed a negative correlation between serum
kynurenine and CRP or the neutrophil-to-lymphocyte ratio. IDO-1 is
present in various cell types and, upon upregulation by
inflammatory stimuli, exerts immunosuppressive and
anti-inflammatory effects (26,27). In
the immune microenvironment, the upregulation of IDO-1 in monocytes
leads to local tryptophan depletion and kynurenine production,
which activate general control nonderepressible-2 kinase and AhR,
respectively. As a result, cell metabolism shifts to suppress the
adaptive immune response by reducing T-cell proliferation and
favoring CD4+ T-cell differentiation toward regulatory
rather than effector phenotypes (14,28-30).
As regards natural killer cells, IDO-1 impairs their function by
downregulating NKG2D ligand via A disintegrin and metalloproteinase
domain-containing protein 10(31).
As regards neutrophils, IDO-1 activity limits neutrophil abundance
and pro-inflammatory function (32,33). In
epithelial cells, IDO-1 is upregulated following viral infection
and subsequently downregulates NF-κB signaling and the production
of the pro-inflammatory cytokines, IFN-α, IFN-β and IL-6(34). Of note, in antigen-presenting cells,
AhR activation is required for IDO-1 expression, suggesting a
positive feedback loop that may enhance IDO-1 immunosuppressive
properties (35). It is likely that
the negative correlation between serum kynurenine levels and the
inflammatory markers CRP and neutrophil-to-lymphocyte ratio
detected in the present study reflects IDO-1 upregulation due to
chronic inflammation that characterizes patients undergoing HD,
which subsequently counteracts inflammation. Notably, compared with
healthy controls, IDO-1 expression is higher in monocytes from
patients undergoing HD (4).
Remarkably, in a previous study, in a group of stable patients
undergoing HD, plasma IDO-1 levels were higher than those in
healthy subjects and negatively correlated with CRP, IL-6 and TNF-α
(36).
In the present study, serum kynurenine levels did
not differ between patients undergoing HD with or without CHD.
Previous studies have identified a link between kynurenine pathway
metabolites and subclinical atherosclerosis, prevalent
atherosclerotic CVD, new CV events, and mortality in patients
undergoing HD (19-22).
However, in a cohort of patients undergoing HD from the CONTRAST
trial with a median follow-up of 4.3 years, baseline kynurenine
pathway metabolite levels were not associated with all-cause
mortality or new CV events (7).
Differences in the characteristics of the patients across these
studies may account for this discrepancy.
In the general population, kynurenine pathway
metabolites have been linked to the occurrence of HF and AF, as
well as to worse outcomes in patients with existing HF (16-18).
In patients with CKD, the prevalence of HF is high and is
associated with increased mortality (37). In a previous study, in a cohort of
673 non-dialysis patients with CKD stages 1-5, kynurenine levels
were connected to both prevalent and incident HF (23). The present study assessed whether
kynurenine levels varied between patients with and without HF
undergoing HD. It was found that serum kynurenine was elevated only
in patients with HFrEF. ROC curve analysis demonstrated a modest
discriminatory ability of kynurenine as a biomarker for HFrEF in
patients undergoing HD, with an AUC of 0.637 (P=0.022). However,
given that HFrEF is a multifactorial disorder, a single variable is
unlikely to provide strong diagnostic accuracy. Therefore, a binary
logistic regression analysis was performed to determine whether
kynurenine is an independent risk factor and a potential
pathogenetic contributor to HFrEF in this population. After
adjusting for other factors associated with HFrEF, kynurenine
remained an independent predictor. For each 1 µM increase in serum
kynurenine level, the odds of HFrEF increased by 65%. Notably, in
the patient cohort in the present study, 21 of the 27 patients with
HFrEF also had CHD. Experimental data support the role of
kynurenine pathway products in the development of HF. Following
myocardial infarction, IDO-1 is upregulated in endothelial cells,
leading to increased kynurenine production. This, through the AhR
pathway, causes cardiomyocyte apoptosis (38). An elevated expression of IDO-1 has
been observed in the hypertrophic myocardium, and increased
kynurenine activates the AhR, leading to cardiac hypertrophy
(39). Finally, monocyte
infiltration plays a significant role in cardiac fibrosis (40), and in patients undergoing HD,
monocytes express higher levels of IDO-1(4).
The present study then examined whether kynurenine
levels vary between patients with and without AF undergoing HD. In
patients undergoing HD, the prevalence of AF is higher than that in
the general population and is linked to increased mortality
(41). Even following effective
treatment of AF with pulmonary vein isolation (42,43), the
recurrence rate is significantly higher in patients undergoing HD,
indicating the vulnerability of this population to AF (44,45). The
present study found that serum kynurenine levels were higher in
patients with AF. ROC curve analysis revealed the modest
performance of kynurenine as a biomarker for AF in patients
undergoing HD, with an AUC of 0.652 (P=0.005). However, for a
multifactorial condition such as AF, a single factor is unlikely to
achieve good discrimination. Thus, binary logistic regression
analysis was used to evaluate whether kynurenine is an independent
risk factor and possibly a pathogenetic factor for AF in this
population. After adjusting for other factors associated with AF,
kynurenine remained an independent predictor. For each 1 µM
increase in serum kynurenine, the odds of AF increased by 37%.
Whether this is caused by cardiac fibrosis or a direct
arrhythmogenic effect of kynurenine pathway products remains to be
clarified. Experimentally, kynurenine exerts pro-arrhythmogenic
effects by modulating cardiac repolarization, possibly by reducing
Kv11.1 channel levels (46). In
other cell types, an increased expression of IDO-1 has been shown
to upregulate P2X7 purinoceptor expression (47). Experimentally, P2X7 predisposes to AF
by altering ion channel protein expression and inducing myocardial
hypertrophy and fibrosis (48,49).
To the best of our knowledge, this is the first
study to assess the role of kynurenine in HF and AF among patients
undergoing HD. However, there are some limitations. Although our
study detected an association between serum kynurenine levels and
the prevalence of HFrEF and AF, its cross-sectional design cannot
establish causality. Future studies that measure kynurenine
longitudinally could clarify causal associations and may help
establish serum kynurenine as a risk factor for HFrEF and AF among
patients undergoing HD. For this reason, the authors plan to
follow-up with the patients and enroll additional patients. In
addition, the enrollment of patients from other centers will verify
the stability of the association between kynurenine and
cardiovascular complications. Another limitation of the present
study is the inclusion of patients with a known history of CHD,
which may have led to an underestimation of the true prevalence of
CHD, as no specific diagnostic imaging was performed to detect
clinically silent cases. Nevertheless, patients undergoing HD
attend the HD unit three times per week, facilitating the early
detection of CHD symptoms. Furthermore, these patients receive
regular cardiological assessments and transthoracic
echocardiography. As regards HF, all patients underwent a
transthoracic echocardiogram within 3 months of the study. AF was
unlikely to remain undiagnosed, given the frequency of patient
visits to the HD unit.
In the event that the contribution of kynurenine to
the development of HF or AF in patients undergoing HD is confirmed,
strategies to reduce its levels may warrant consideration. As IDO-1
significantly contributes to kynurenine production, reducing its
expression by alleviating the chronic inflammation typical of HD is
a viable approach. Notably, clinical trials, such as the large
phase III clinical trials NCT05021835 and the NCT05485961, are
already underway using monoclonal antibodies targeting
pro-inflammatory cytokines to reduce chronic inflammation (50). Alternatively, the direct inhibition
of IDO-1 is possible. Several specific IDO-1 inhibitors, as well as
inhibitors of both IDO-1 and TDO, are available and have been shown
to be safe, having been tested as immunomodulatory agents in cancer
treatment (51).
In conclusion, as demonstrated in the present study,
in patients undergoing HD, serum kynurenine levels are elevated and
associated with HFrEF and AF, suggesting a potential role for this
uremic toxin in the development of these CV disorders. If proven,
efforts to decrease tryptophan metabolism via the kynurenine
pathway by reducing inflammation or inhibiting IDO-1 would be
beneficial, as removing already formed kynurenine pathway products
is challenging due to their binding to serum proteins.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
TE was involved in the experimental design of the
study and wrote the draft of the manuscript. TE, MD, AK, PM, CP,
EL, AB and IS were involved in data collection. AK performed the
ELISA. TE, MD, AK and MT performed the statistical analysis. TE,
MD, AK, CP, EL, AB, MT and IS were involved in the editing of the
manuscript. TE, AK and MD confirm the authenticity of all the raw
data. All authors have read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was conducted in accordance with
the Declaration of Helsinki and was approved by the Ethics
Committee of the University of Thessaly, Faculty of Medicine
(Approval Number: 558/10-2-2017). Informed consent was obtained
from all subjects involved in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Jankowski J, Floege J, Fliser D, Böhm M
and Marx N: Cardiovascular disease in chronic kidney disease.
Circulation. 143:1157–1172. 2021.
|
|
2
|
Alberati-Giani D, Ricciardi-Castagnoli P,
Kohler C and Cesura AM: Regulation of the kynurenine metabolic
pathway by interferon-gamma in murine cloned macrophages and
microglial cells. J Neurochem. 66:996–1004. 1996.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Schefold JC, Zeden JP, Fotopoulou C, von
Haehling S, Pschowski R, Hasper D, Volk HD, Schuett C and Reinke P:
Increased indoleamine 2,3-dioxygenase (IDO) activity and elevated
serum levels of tryptophan catabolites in patients with chronic
kidney disease: A possible link between chronic inflammation and
uraemic symptoms. Nephrol Dial Transplant. 24:1901–1908.
2009.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Eleftheriadis T, Pissas G, Antoniadi G,
Tsogka K, Makri P, Liakopoulos V and Stefanidis I: Increased
indoleamine 2,3-dioxygenase in monocytes of patients on
hemodialysis. Iran J Kidney Dis. 10:91–93. 2016.PubMed/NCBI
|
|
5
|
Saito K, Fujigaki S, Heyes MP, Shibata K,
Takemura M, Fujii H, Wada H, Noma A and Seishima M: Mechanism of
increases in L-kynurenine and quinolinic acid in renal
insufficiency. Am J Physiol Renal Physiol. 279:F565–F572.
2000.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Debnath S, Velagapudi C, Redus L, Thameem
F, Kasinath B, Hura CE, Lorenzo C, Abboud HE and O'Connor JC:
Tryptophan metabolism in patients with chronic kidney disease
secondary to type 2 diabetes: Relationship to inflammatory markers.
Int J Tryptophan Res. 10(1178646917694600)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
van Gelder MK, Middel IR, Vernooij RWM,
Bots ML, Verhaar MC, Masereeuw R, Grooteman MP, Nubé MJ, van den
Dorpel MA, Blankestijn PJ, et al: Protein-Bound uremic toxins in
hemodialysis patients relate to residual kidney function, are not
influenced by convective transport, and do not relate to outcome.
Toxins (Basel). 12(234)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zakrocka I, Wicha-Komsta K, Boczkowska S,
Załuska W, Papadopoulou E and Nasioulas G: The impact of clinical
variables and dialysis modality on kynurenine pathway enzymes
expression. Int J Mol Sci. 27(1313)2026.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Badawy AAB and Guillemin G: The plasma
[Kynurenine]/[Tryptophan] ratio and indoleamine 2,3-dioxygenase:
Time for appraisal. Int J Tryptophan Res.
12(1178646919868978)2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen Y, Xie Z, Xiao C, Zhang M, Li Z, Xie
J, Zhang Y, Zhao X, Zeng P, Mo L, et al: Peripheral
kynurenine/tryptophan ratio is not a reliable marker of systemic
indoleamine 2,3-dioxygenase: A lesson drawn from patients on
hemodialysis. Oncotarget. 8:25261–25269. 2017.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Opitz CA, Litzenburger UM, Sahm F, Ott M,
Tritschler I, Trump S, Schumacher T, Jestaedt L, Schrenk D, Weller
M, et al: An endogenous tumour-promoting ligand of the human aryl
hydrocarbon receptor. Nature. 478:197–203. 2011.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Beischlag TV, Morales JL, Hollingshead BD
and Perdew GH: The aryl hydrocarbon receptor complex and the
control of gene expression. Crit Rev Eukaryot Gene Expr.
18:207–250. 2008.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Eleftheriadis T, Pissas G, Antoniadi G,
Liakopoulos V and Stefanidis I: Kynurenine, by activating aryl
hydrocarbon receptor, decreases erythropoietin and increases
hepcidin production in HepG2 cells: A new mechanism for anemia of
inflammation. Exp Hematol. 44:60–67.e61. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Eleftheriadis T, Pissas G, Liakopoulos V
and Stefanidis I: IDO decreases glycolysis and glutaminolysis by
activating GCN2K, while it increases fatty acid oxidation by
activating AhR, thus preserving CD4+ T-cell survival and
proliferation. Int J Mol Med. 42:557–568. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Eleftheriadis T, Pissas G, Golfinopoulos
S, Liakopoulos V and Stefanidis I: Role of indoleamine
2,3-dioxygenase in ischemia-reperfusion injury of renal tubular
epithelial cells. Mol Med Rep. 23(472)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Razquin C, Ruiz-Canela M, Toledo E,
Hernández-Alonso P, Clish CB, Guasch-Ferré M, Li J, Wittenbecher C,
Dennis C, Alonso-Gómez A, et al: Metabolomics of the
tryptophan-kynurenine degradation pathway and risk of atrial
fibrillation and heart failure: Potential modification effect of
Mediterranean diet. Am J Clin Nutr. 114:1646–1654. 2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Dschietzig TB, Kellner KH, Sasse K,
Boschann F, Klüsener R, Ruppert J, Armbruster FP, Bankovic D,
Meinitzer A, Mitrovic V and Melzer C: Plasma kynurenine predicts
severity and complications of heart failure and associates with
established biochemical and clinical markers of disease. Kidney
Blood Press Res. 44:765–776. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Guillemin GJ, Lund A, Nordrehaug JE, Lund
A, Nordrehaug JE, Slettom G, Solvang SH, Pedersen EK, Midttun Ø,
Ulvik A, et al: Plasma kynurenines and prognosis in patients with
heart failure. PLoS One. 15(e0227365)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pawlak K, Domaniewski T, Mysliwiec M and
Pawlak D: The kynurenines are associated with oxidative stress,
inflammation and the prevalence of cardiovascular disease in
patients with end-stage renal disease. Atherosclerosis.
204:309–314. 2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Pawlak K, Brzosko S, Mysliwiec M and
Pawlak D: Kynurenine, quinolinic acid—The new factors linked to
carotid atherosclerosis in patients with end-stage renal disease.
Atherosclerosis. 204:561–566. 2009.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Benitez T, VanDerWoude E, Han Y, Byun J,
Konje VC, Gillespie BW, Saran R and Mathew AV: Kynurenine pathway
metabolites predict subclinical atherosclerotic disease and new
cardiovascular events in chronic kidney disease. Clin Kidney J.
15:1952–1965. 2022.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Al Awadhi S, Myint L, Guallar E, Clish CB,
Wulczyn KE, Kalim S, Thadhani R, Segev DL, DeMarco MM, Moe SM, et
al: A metabolomics approach to identify metabolites associated with
mortality in patients receiving maintenance hemodialysis. Kidney
Int Rep. 9:2718–2726. 2024.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Mohiti S, Christensen J, Landler NE,
Sørensen IM, Thomassen JQ, Bjergfelt SS, Hansen D, Feldt-Rasmussen
B, Bro S, Ebrahimi-Mameghani M, et al: Serum tryptophan and
kynurenine levels and risk of heart failure among patients with
chronic kidney disease. Clin Nutr. 47:14–20. 2025.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Dou L, Poitevin S, Sallée M, Addi T,
Gondouin B, McKay N, Denison MS, Jourde-Chiche N, Duval-Sabatier A,
Cerini C, et al: Aryl hydrocarbon receptor is activated in patients
and mice with chronic kidney disease. Kidney Int. 93:986–999.
2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Kolachalama VB, Shashar M, Alousi F,
Shivanna S, Rijal K, Belghasem ME, Walker J, Matsuura S, Chang GH,
Gibson CM, et al: Uremic solute-aryl hydrocarbon receptor-tissue
factor axis associates with thrombosis after vascular injury in
humans. J Am Soc Nephrol. 29:1063–1072. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Muller AJ, Sharma MD, Chandler PR,
Duhadaway JB, Everhart ME, Johnson BA III, Kahler DJ, Pihkala J,
Soler AP, Munn DH, et al: Chronic inflammation that facilitates
tumor progression creates local immune suppression by inducing
indoleamine 2,3 dioxygenase. Proc Natl Acad Sci USA.
105:17073–17078. 2008.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Mor A, Tankiewicz-Kwedlo A, Ciwun M,
Lewkowicz J and Pawlak D: Kynurenines as a novel target for the
treatment of inflammatory disorders. Cells. 13(1259)2024.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Eleftheriadis T, Pissas G, Antoniadi G,
Spanoulis A, Liakopoulos V and Stefanidis I: Indoleamine
2,3-dioxygenase increases p53 levels in alloreactive human T cells,
and both indoleamine 2,3-dioxygenase and p53 suppress glucose
uptake, glycolysis and proliferation. Int Immunol. 26:673–684.
2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Eleftheriadis T, Pissas G, Antoniadi G,
Liakopoulos V and Stefanidis I: Indoleamine 2,3-dioxygenase
depletes tryptophan, activates general control non-derepressible 2
kinase and down-regulates key enzymes involved in fatty acid
synthesis in primary human CD4+ T cells. Immunology. 146:292–300.
2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Eleftheriadis T, Pissas G, Sounidaki M,
Tsogka K, Antoniadis N, Antoniadi G, Liakopoulos V and Stefanidis
I: Indoleamine 2,3-dioxygenase, by degrading L-tryptophan, enhances
carnitine palmitoyltransferase I activity and fatty acid oxidation,
and exerts fatty acid-dependent effects in human alloreactive CD4+
T-cells. Int J Mol Med. 38:1605–1613. 2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Fang X, Guo L, Xing Z, Shi L, Liang H, Li
A, Kuang C, Tao B and Yang Q: IDO1 can impair NK cells function
against non-small cell lung cancer by downregulation of NKG2D
Ligand via ADAM10. Pharmacol Res. 177(106132)2022.PubMed/NCBI View Article : Google Scholar
|
|
32
|
El-Zaatari M, Chang YM, Zhang M, Franz M,
Shreiner A, McDermott AJ, van der Sluijs KF, Lutter R, Grasberger
H, Kamada N, et al: Tryptophan catabolism restricts
IFN-γ-expressing neutrophils and clostridium difficile
immunopathology. J Immunol. 193:807–816. 2014.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Guo SX, Zhang L, Jiang W and Ma JJ:
Indoleamine 2,3-dioxygenase adjusts neutrophils recruitment and
chemotaxis in Aspergillus fumigatus keratitis. Int J Ophthalmol.
15:380–387. 2022.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Zhao F, Huang Y, Ji J, Liu X, Li X, Zou L,
Wu K, Liu XD, Zeng S, Wang X, et al: IDO1 promotes CSFV replication
by mediating tryptophan metabolism to inhibit NF-κB signaling. J
Virol. 98(e0045824)2024.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Nguyen NT, Kimura A, Nakahama T, Chinen I,
Masuda K, Nohara K, Fujii-Kuriyama Y and Kishimoto T: Aryl
hydrocarbon receptor negatively regulates dendritic cell
immunogenicity via a kynurenine-dependent mechanism. Proc Natl Acad
Sci USA. 107:19961–19966. 2010.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Eleftheriadis T, Liakopoulos V, Antoniadi
G, Stefanidis I and Galaktidou G: Indoleamine 2,3-dioxygenase is
increased in hemodialysis patients and affects immune response to
hepatitis B vaccination. Vaccine. 29:2242–2247. 2011.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Damman K and Testani JM: The kidney in
heart failure: An update. Eur Heart J. 36:1437–1444.
2015.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Melhem NJ, Chajadine M, Gomez I, Howangyin
KY, Bouvet M, Knosp C, Sun Y, Rouanet M, Laurans L, Cazorla O, et
al: Endothelial cell indoleamine 2, 3-dioxygenase 1 alters cardiac
function after myocardial infarction through kynurenine.
Circulation. 143:566–580. 2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Wang Y, Song J, Yu K, Nie D, Zhao C, Jiao
L, Wang Z, Zhou L, Wang F, Yu Q, et al: Indoleamine 2,3-dioxygenase
1 deletion-mediated kynurenine insufficiency inhibits pathological
cardiac hypertrophy. Hypertension. 80:2099–2111. 2023.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Sager HB, Hulsmans M, Lavine KJ, Moreira
MB, Heidt T, Courties G, Sun Y, Iwamoto Y, Tricot B, Khan OF, et
al: Proliferation and recruitment contribute to myocardial
macrophage expansion in chronic heart failure. Circ Res.
119:853–864. 2016.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Winkelmayer WC, Patrick AR, Liu J,
Brookhart MA and Setoguchi S: The increasing prevalence of atrial
fibrillation among hemodialysis patients. J Am Soc Nephrol.
22:349–357. 2011.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Nesapiragasan V, Hayıroğlu Mİ, Sciacca V,
Sommer P, Sohns C and Fink T: Catheter Ablation approaches for the
treatment of arrhythmia recurrence in patients with a durable
pulmonary vein isolation. Balkan Med J. 40:386–394. 2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Yumurtaş AÇ, Pay L, Tezen O, Çetin T,
Yücedağ FF, Arter E, Kadıoğlu H, Akgün H, Özkan E, Uslu A, et al:
Evaluation of risk factors for long-term atrial fibrillation
development in patients undergoing typical atrial flutter ablation:
A multicenter pilot study. Herz. 50:51–58. 2024.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Takigawa M, Kuwahara T, Takahashi A,
Kobori A, Takahashi Y, Okubo K, Watari Y, Sugiyama T, Kimura S,
Takagi K, et al: The impact of haemodialysis on the outcomes of
catheter ablation in patients with paroxysmal atrial fibrillation.
Europace. 16:327–334. 2013.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Onishi N, Kaitani K, Nakagawa Y, Kobori A,
Inoue K, Kurotobi T, Morishima I, Matsui Y, Yamaji H, Nakazawa Y,
et al: Radiofrequency catheter ablation for atrial fibrillation
patients on hemodialysis (From the Kansai Plus Atrial Fibrillation
Registry)-clinical impact of early recurrence. Circ J.
88:1057–1064. 2024.PubMed/NCBI View Article : Google Scholar
|
|
46
|
van Ham WB, Cornelissen CM, Polyakova E,
van der Voorn SM, Ligtermoet ML, Monshouwer-Kloots J, Vos MA, Bossu
A, van Rooij E, van der Heyden MAG and van Veen TAB: Pro-arrhythmic
potential of accumulated uremic toxins is mediated via
vulnerability of action potential repolarization. Int J Mol Sci.
24(5373)2023.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Eleftheriadis T, Pissas G, Golfinopoulos
S, Liakopoulos V and Stefanidis I: Indoleamine 2,3-dioxygenase
controls purinergic receptor-mediated ischemia-reperfusion injury
in renal tubular epithelial cells. J Basic Clin Physiol Pharmacol.
34:745–754. 2023.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Ye T, Yang J, Liu Z, Yu Y, Zhang C, Guo Y,
Yu F, Zhou Y, Song Z, Shi J, et al: Inhibition of the P2X7 receptor
prevents atrial proarrhythmic remodeling in experimental
post-operative atrial fibrillation. Int Immunopharmacol.
129(111536)2024.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Zhou Y, Ye T, Yu F, Song Z, Wang L, Zhang
C, Yang B, Yang J and Wang X: Inhibition of P2X7 receptor mitigates
atrial fibrillation susceptibility in isoproterenol-induced rats.
Biochem Biophys Res Commun. 749(151340)2025.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Nowak KL and Chonchol M: Targeting
inflammation in CKD. Am J Kidney Dis. 86:803–813. 2025.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Al-Zoubi RM, Elaarag M, Al-Qudimat AR,
Al-Hurani EA, Fares ZE, Farhan A, Al-Zoubi SR, Khan A, Agouni A,
Shkoor M, et al: IDO and TDO inhibitors in cancer immunotherapy:
Mechanisms, clinical development, and future directions. Front
Pharmacol. 16(1632446)2025.PubMed/NCBI View Article : Google Scholar
|