Introduction
Gastric cancer (GC) ranks as the fifth most
prevalent malignancy worldwide and is also the fifth leading cause
of cancer-related mortality (1). For
patients with resectable disease, total gastrectomy with D2
lymphadenectomy remains the standard curative surgical approach,
typically followed by gastrointestinal reconstruction (2,3).
Depending on pre-operative staging and risk assessment, certain
patients may receive neoadjuvant therapy, while adjuvant
chemotherapy or chemoradiotherapy may be indicated based on
pathological findings, in accordance with widely accepted
international guidelines (2-4).
According to the 8th edition of the Tumour Node Metastasis (TNM)
classification, the 5-year survival rates following R0 resection
have improved significantly compared to previous decades, ranging
from 89.9% in stage IB cases to 20.2% in stage IIIC cases (5).
As survival outcomes have improved, health-related
quality of life (QoL) has gained increasing attention in GC care, a
domain historically underemphasized in surgical and oncological
literature (6-8).
Total gastrectomy, despite its curative intent, often leads to
persistent post-gastrectomy syndromes, such as nutritional
deficiencies, digestive dysfunction and psychosocial challenges,
all of which can significantly impair long-term QoL (8-11).
Recognising this, there is a growing emphasis on integrating QoL as
a core outcome measure in both clinical trials and routine care
(9,11,12). QoL
may be significantly impaired even in patients with otherwise
favourable clinical outcomes. Notably, QoL has been identified as
an independent prognostic factor for survival, underscoring its
importance in the post-operative management of patients undergoing
total gastrectomy (6,12).
Given its growing clinical relevance, it is
important to understand what QoL truly encompasses. QoL is a
multidimensional concept that includes physical, psychological,
social, and functional well-being, shaped by individual goals,
cultural context, and expectations. This holistic perspective
reinforces the need to balance oncological success with the overall
lived experience and recovery experienced by the patient (11,12).
To measure QoL in a meaningful manner,
patient-reported outcome (PRO) assessments are most commonly used,
which capture the experiences of patients directly, without
clinician interpretation. PRO assessments provide insight beyond
clinical metrics, assisting in the evaluation of the real-world
impact of surgery, as well as in both treatment decisions and
long-term care strategies (6,13).
Questionnaires are essential tools for documenting
the experience of a patient and assessing QoL. The most widely used
instrument for patients with cancer is the European Organization
for Research and Treatment of Cancer Quality of Life
Questionnaire-Core 30 (EORTC QLQ-C30; https://qol.eortc.org/questionnaires/) (14), a 30-item tool that evaluates multiple
dimensions of QoL. It includes five functional scales (physical,
role, cognitive, emotional and social functioning), a global QoL
scale, three symptom scales (fatigue, nausea/vomiting and pain),
and six single-item measures addressing appetite loss, diarrhoea,
dyspnoea, constipation, insomnia and financial difficulties
(6,9).
To complement the generic QLQ-C30, the EORTC
QLQ-STO22 (https://qol.eortc.org/questionnaire/gastric-cancer-update-of-qlq-sto22/)
module was developed specifically for patients with GC (15). It consists of 22 items assessing
disease-specific issues such as dysphagia, early satiety, reflux,
taste disturbances, eating-related anxiety, pain and body image
concerns. Collectively, the QLQ-C30 and QLQ-STO22 provide both a
general and a disease-specific evaluation of the QoL of patients
with GC.
The Postgastrectomy Syndrome Assessment Scale
(PGSAS) is a disease-specific tool developed to evaluate QoL after
gastrectomy. The PGSAS-37, derived from the original 45-item
version, is organised into three domains: Symptoms (oesophageal
reflux, abdominal pain, meal-related distress, indigestion,
diarrhoea, constipation, dumping) plus a total symptom score;
living status measures (food intake, need for additional meals,
meal quality and ability to work); and QoL subscales on
dissatisfaction with symptoms, meals, work and daily life (16,17).
The EuroQol 5 Dimensions (EQ-5D) is a generic,
standardised instrument developed by the EuroQol Group to assess
QoL across five dimensions: mobility, self-care, usual activities,
pain/discomfort and anxiety/depression. It also includes a visual
analogue scale for rating overall health (18).
Several other instruments have been created to
assess the QoL of patients with GC, including the Short Form-36
Health Survey (SF-36) (19), the
Functional Assessment of Cancer Therapy-Gastric (FACT-Ga) (11), the MD Anderson Symptom
Inventory-Gastrointestinal Module (MDASI-GI) (13) and the Korean Quality of Life in
Stomach Cancer Surgery (KOQUSS) (20) questionnaire. While some of these
tools assess overlapping domains, others, such as the KOQUSS, were
specifically designed to capture post-gastrectomy experiences.
Despite validation in selected languages, the broader international
use of these instruments remains limited.
It is evident that certain domains, such as physical
functioning, pain and social functioning, are consistently
represented across different questionnaires, allowing comparison,
whereas others are unique to individual tools, capturing aspects
that may not be addressed elsewhere; recognising these overlaps and
distinctions is essential for meaningful interpretation and for
structuring the results in a manner that facilitates cross-study
and cross-instrument analysis.
Moreover, the interpretation of these
questionnaires, although described in detail within the manual of
each instrument, varies between tools. Each questionnaire uses a
different scoring approach; for example, the PGSAS is based on raw
scores, whereas the EORTC can be calculated from either raw scores
or linear transformations, with a distinct transformation algorithm
for each domain (9). Consequently,
this necessitates separate analyses for each domain. It should be
noted that higher scores do not always indicate a better QoL; for
example, in symptom scales, a higher score reflects greater symptom
burden and therefore poorer QoL (10,16,17,21).
Certain studies have explored the use of an overall ‘summative
score’ (6,22); however, the majority of researchers
analyse outcomes on a domain-by-domain basis (8,9,12,15).
The aim of the present scoping review was to examine
recent evidence on post-operative QoL following total gastrectomy,
with a focus on both its chronological course and variations
related to surgical technique. Studies published within the
previous 5 years were used to reflect research conducted after
major updates to GC treatment guidelines, introduced in the West in
2013(23) and in the East in
2016(24). These updates redefined
the standard surgical approach by excluding routine removal of
spleen and pancreas from curative total gastrectomy. Given the
significance of this change, it is reasonable to expect that it has
had a substantial impact on the QoL of patients with GC, making it
a relevant factor to consider in recent studies. To the best of our
knowledge, this is the first scoping review to specifically examine
QoL following total gastrectomy in the context of the most recent
major guideline updates, providing a timely perspective on outcomes
in the modern surgical era.
Data and methods
A comprehensive search of the PubMed, Scopus and
EMBASE databases was performed using combinations of the
terms\total gastrectomy\,\gastric cancer\,\quality of
life\,\patient-reported outcomes', as well as the names of commonly
used QoL instruments (e.g., EORTC QLQ-C30, QLQ-STO22, PGSAS-37 and
EQ-5D). These terms were combined using Boolean operators (AND/OR)
as appropriate. To capture the most recent data, the search was
limited to studies published between 2020 and 2025, with the aim of
including research conducted after the most recent major updates in
GC treatment guidelines. The search and initial screening were
performed by the first author, and the selection of eligible
studies was discussed with the co-authors. Titles and abstracts
were initially screened for eligibility, followed by full-text
assessments of potentially relevant studies. Any discrepancies were
resolved through discussion and consensus among the authors.
The present scoping review was conducted and
reported in accordance with the Preferred Reporting Items for
Systematic Reviews and Meta-Analyses extension for Scoping Reviews
(PRISMA-ScR) guidelines (25).
Studies were eligible if they investigated total
gastrectomy, reported QoL outcomes using validated questionnaires
and explicitly referred to curative total gastrectomy without any
additional major organ resection. Non-English-language studies,
those conducted for non-oncological indications, those lacking
explicit QoL outcomes, or those not exclusively focused on total
gastrectomy were excluded. The search was limited to studies
published from 2020 onwards to reflect contemporary clinical
practice following the widespread adoption of recent guideline
updates. Studies involving multivisceral resections or comparing
total to subtotal gastrectomy without extractable data specific to
total gastrectomy were also excluded.
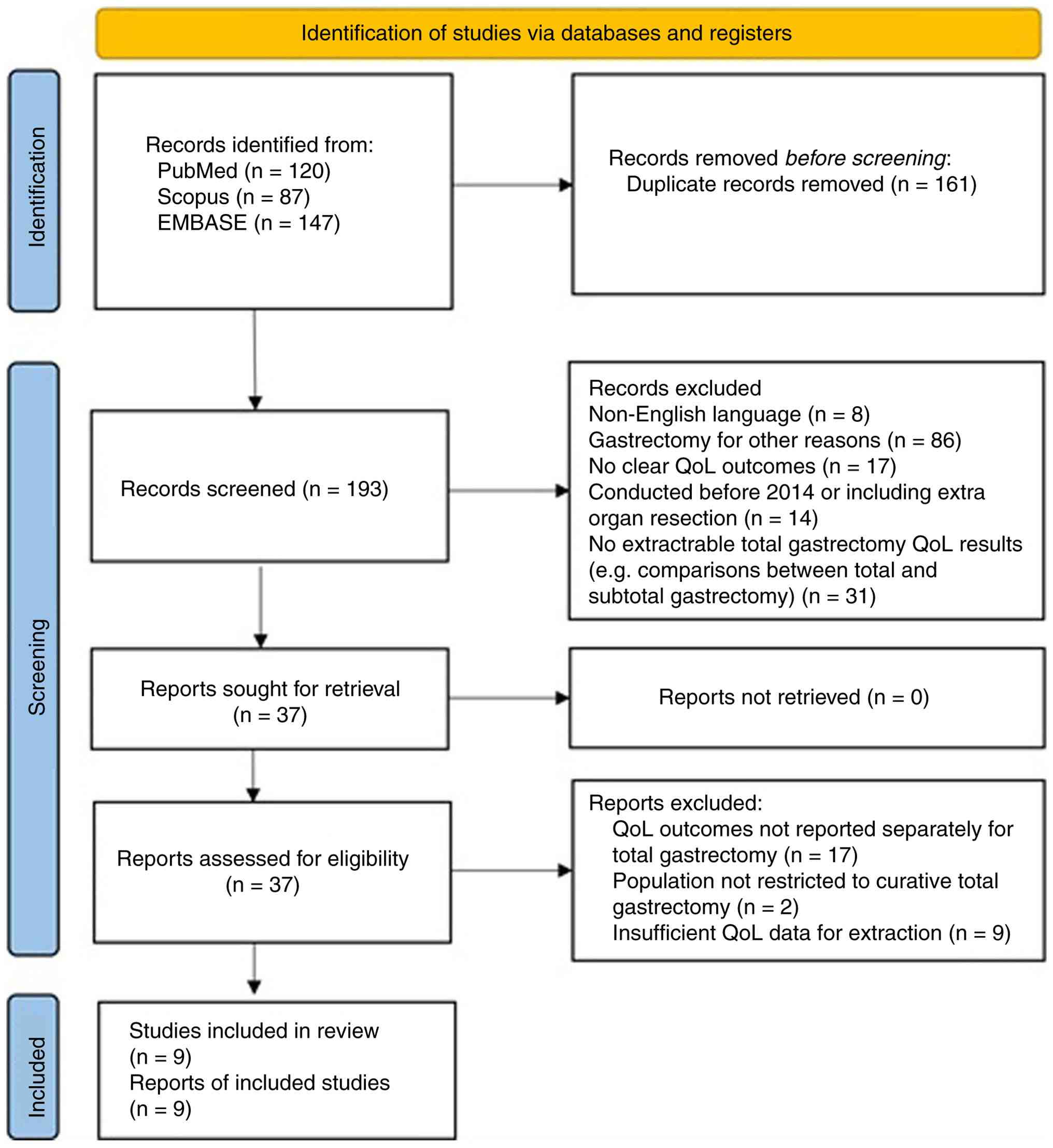
Following the removal of duplicates and applying the
predefined inclusion and exclusion criteria, a total of nine
studies were selected for analysis, as outlined in the PRISMA-ScR
flowchart (Fig. 1).
Results
A total of nine studies met the predefined
eligibility criteria and were included in the final analysis
(Fig. 1). Among these, Wei et
al (26) and Yan et al
(27) conducted retrospective
analyses comparing, among other outcomes, QoL following linear
stapling (LS) vs. circular stapling (CS) for the esophagojejunal
anastomosis. Wei et al (26)
supplemented their assessment with the Gastroesophageal Reflux
Disease Questionnaire (GERDQ) in order to more accurately capture
reflux symptoms. In addition, Yan et al (27) compared intracorporeal vs.
extracorporeal anastomosis, along with various anastomotic
configurations, such as overlap and π-shaped, as well as the
OrVil-assisted technique. Lin et al (28) compared QoL outcomes between totally
laparoscopic total gastrectomy (TLTG) and laparoscopic-assisted
total gastrectomy (LATG). Van der Wielen et al (29) assessed QoL outcomes between open
total gastrectomy (OTG) and minimally invasive total gastrectomy
(MITG) in patients with advanced GC treated with neoadjuvant
chemotherapy. A similar prospective comparison between open and
laparoscopic total gastrectomy was conducted by Tanaka et al
(17). Saeki et al (21) correlated high-resolution manometry
(HRM) findings with QoL, while Lu et al (30) retrospectively compared QoL outcomes
between patients undergoing standard Roux-en-Y anastomosis (RY) and
those receiving proximal jejunal pouch Roux-en-Y anastomosis
(PP-RY). Kubota et al (16)
compared QoL between elderly and non-elderly patients undergoing
aboral pouch reconstruction. Finally, Park et al (9) conducted a prospective longitudinal
study over a period of 3 years comparing QoL after total and distal
gastrectomy. Although the latter study would normally be
ineligible, it was the only study under consideration to present
clear, separate data on chronological changes in QoL domains for
the total gastrectomy subgroup. These data, which were explicitly
reported and independent from the study's other outcomes, were used
as a foundation for the assessment of how QoL evolved after total
gastrectomy. An outline of the characteristics of the included
studies is presented in Table I.
 | Table ICharacteristics of the studies
included in the scoping review of QoL following total
gastrectomy. |
Table I
Characteristics of the studies
included in the scoping review of QoL following total
gastrectomy.
| Authors, year of
publication | Study type | Study period | Study geographical
region | No. of cases | Total gastrectomy
cases only | Questionnaire
administration timeline | Comparison
parameters | Questionnaires
used | (Refs.) |
|---|
| Wei et al,
2021 | Retrospective | 2014-2018 | China | 120 | Yes | Post-operatively
after 6 months and 1 year | Linear stapler vs.
circular Stapler | EORTC QLQ C-30
EORTC STO-22 GERDQ | (26) |
| Yan et al,
2023 | Retrospective | 2015-2020 | China and USA | 105 | Yes | Pre-operatively and
post-operatively after 1 year | i) Linear stapler
vs. circular stapler; ii) Intracorporeal vs. extracorporeal
anastomosis; iii) Overlap vs. π-shaped vs. OrVil anastomosis | EORTC QLQ C-30
EORTC STO-22 | (27) |
| Lin et al,
2023 | Retrospective | 2014-2018 | China | 312 | Yes | Post-operatively
after 3 months, 6 months, and 1 year | TLTG vs. LATG | EORTC QLQ C-30
EORTC STO-22 | (28) |
| Van der Wielen
et al, 2022 |
Prospective-RCT | 2015-2018 | Europe | 96 | Yes | Pre-operatively and
post-operatively after 5 days, 3 months, 6 months, and 1 year | Open vs. MIS
gastrectomy in neoadjuvant patients | EORTC QLQ C-30
EORTC STO-22 EQ5D | (29) |
| Tanaka et
al, 2024 | Prospective | 2015-2020 | Japan | 59 | Yes | Pre-operatively and
post-operatively after 1 month, 3 months, 1 year, and 3 years | Laparoscopic vs.
open gastrectomy | EORTCC30 PGSAS
37 | (17) |
| Saeki et al,
2024 | Retrospective | 2014-2020 | Japan | 12 | Yes | Post-operatively
after 1 year | i) HRM findings;
ii) Linear stapler vs. circular stapler | PGSAS-37 | (21) |
| Lu et al,
2022 | Retrospective | 2019-2020 | China | 136 | Yes | Post-operatively
after 1 year | RY vs. PP-RY | EORTC QLQ C-30
EORTC STO-22 | (30) |
| Kubota et
al, 2024 | Retrospective | 2016-2022 | Japan | 36 | Yes | Post-operatively
after 1 year | Aboral pouch in the
elderly vs.non-elderly population | PGSAS-37 | (16) |
| Park et al,
2020 | Prospective | 2011-2014 | Korea | 114 | No | Pre-operatively,
and post-operatively after 1, 2 and 3 years | TG vs. DG (TG data
used only) | EORTC QLQ C-30
EORTC STO-22 | (9) |
The majority of the included studies were conducted
in East Asia, with the majority designed retrospectively. The
timepoints for questionnaire distribution ranged from the
pre-operative period up to 3 years post-operatively. In total, 990
cases were analysed in the studies. Given the exploratory nature of
the present scoping review, the aim was to synthesise recent
evidence and identify patterns in QoL outcomes rather than to
perform a formal methodological appraisal of individual
studies.
The eligible studies included in the present scoping
review used various QoL instruments: EORTC QLQ-C30, QLQ-STO22,
EQ-5D and PGSAS-37. Building on the observed overlap between
certain domains, outcomes were organised into conceptually grouped
categories to facilitate consistent comparison across instruments,
with the EORTC framework used as the primary reference given its
widespread use and established structure. The domains and
corresponding questions from other instruments were then mapped and
adapted accordingly, allowing for alignment under common thematic
categories (Table II). To the best
of our knowledge, this approach has not been previously described
and aims to improve clarity and comparability across studies. The
findings are therefore presented according to the unified domains.
For each domain, a definition is provided, followed by an overview
of its chronological evolution and a comparison across surgical
techniques.
 | Table IIUnified domains of QoL questionnaires
following total gastrectomy. |
Table II
Unified domains of QoL questionnaires
following total gastrectomy.
| Domain | EORTC
QLQ-C30a | EORTC STO22 | EQD5 |
PGSAS-37b |
|---|
| Functioning
scales | Physical
function | - | Mobility,
self-care | - |
| | Role function | - | Usual
activities | - |
| | Emotional
function | Anxiety/body
image |
Anxiety/depression | - |
| | Cognitive
function | - | - | - |
| | Social
function | Trouble eating with
others (item 46) | - | Meals (living
status) |
| Symptom scales | Fatigue | - | - | - |
| | Nausea and
vomiting | - | - | - |
| | Pain | Abdominal pain |
Pain/discomfort | Abdominal pain |
| | Dyspnoea | - | - | - |
| | Insomnia | - | - | - |
| |
Constipation/diarrhoea | - | - |
Constipation/diarrhoea |
| | - | Reflux | - | Oesophageal
reflux/indigestion |
| | - | Dysphagia | - | - |
| | - | Eating
restrictions | - | Meal-related
distress/Meals (ingestion) |
| | - | Taste | - | - |
| | - | Dry mouth | - | - |
| | - | Hair loss | - | - |
| | - | - | - | Dumping
subscale |
| Single items | Global
health/QoL | - | - |
Dissatisfaction |
| | Financial
impact | - | - | - |
Functioning scales
The functioning scales are demonstrated in Tables III and IV.
 | Table IIIChronological patterns of functional
and single-item quality of life following gastrectomy. |
Table III
Chronological patterns of functional
and single-item quality of life following gastrectomy.
| Authors, year of
publication | Domain | 0-3 months | 6-12 months | >12 months | Group
characteristicsa,b,c | (Refs.) |
|---|
| Park et al,
2020; Wei et al, 2021; Lin et al, 2023 | Physical
performance and functional capacity | Decline | Improvement | Improvement, but no
baseline levels | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Wei
et al: Mixed stage (I-III); MIS. Lin et al: Mixed
stage (I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy
in 58% (62.5% TLTG, 56.3% LATG) | (9,26,28) |
| Tanaka et
al, 2024; van der Wielen et al, 2022 | Physical
performance and functional capacity | Decline | Improvement | Baseline | Tanaka et
al: Early stage (IA-IIB); MIS and open. Van der Wielen et
al: Mixed stage (I-III); MIS and open; neoadjuvant
therapy. | (17,29) |
| Tanaka et
al, 2024; van der Wielen et al, 2022; Lin et al,
2023 | Role engagement and
daily living | Decline | Improvement, but no
baseline | Stabilisation by
year 3 | Tanaka et
al: Early stage (IA-IIB); MIS and open. Van der Wielen et
al: Mixed stage (I-III); MIS and open; neoadjuvant therapy. Lin
et al: Mixed stage (I-III); MIS; no neoadjuvant therapy;
adjuvant chemotherapy in 58% (62.5% TLTG, 56.3% LATG). | (17,28,29) |
| Park et al,
2020 | Role engagement and
daily living | Decline | Decline | Decline | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Park et al,
2020 | Social integration
and interaction | Improvement | Peak at 12
months | Decline | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Van der Wielen
et al, 2022 | Social integration
and interaction | Decline during
first 6 months | OTG returned to
baseline by 12 months; MITG remained below baseline | OTG exceeded
baseline; MITG did not | Mixed stage
(I-III); MIS and open; neoadjuvant therapy. | (29) |
| Tanaka et
al, 2024 | Emotional and
psychological well-being | Decline | Improvement | N/A | Early stage
(IA-IIB); MIS and open. | (17) |
| Yan et al,
2023; Lu et al, 2022; Lin et al, 2023 | Emotional and
psychological well-being | No significant
change | No significant
change | No significant
change | Yan et al:
Mixed stage (I-III); MIS. Lu et al: Mixed stage (I-III);
MIS. Lin et al: Mixed stage (I-III); MIS; no neoadjuvant
therapy; adjuvant chemotherapy in 58% (62.5% TLTG, 56.3%
LATG). | (27,29,30) |
| Park et al,
2020 | Emotional and
psychological well-being | Improvement | Improvement | Slow decline after
1-2 years | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Park et al,
2020 | Emotional and
psychological well-being (body image) | Improvement | Peak at 1 year | Decline | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Van der Wielen
et al, 2022 | Emotional and
psychological well-being (body image) | No significant
change (OTG); transient improvement (MIS) | Gradual return to
baseline (MIS) | Baseline | Mixed stage
(I-III); MIS and open; neoadjuvant therapy. | (29) |
| Wei et al,
2021; Yan et al, 2023 | Emotional and
psychological well-being (body image) | No significant
change | No significant
change | No significant
change | Mixed stage
(I-III); MIS. | (26,27) |
| Park et al,
2020 | Cognitive
resilience | Decline during
first year | Minimal
improvement | Minimal improvement
up to 3 years | Advanced stage
(II-III); MIS; 65% received adjuvant therapy | (9) |
| Tanaka et
al, 2024; Wei et al, 2021; Yan et al, 2023; Lu
et al, 2022; van der Wielen et al, 2022 | Cognitive
resilience | No significant
change | No significant
change | No significant
change | Tanaka et
al: Early stage (IA-IIB); MIS and open. Wei et al: Mixed
stage (I-III); MIS. Yan et al: Mixed stage (I-III); MIS. Lu
et al: Mixed stage (I-III); MIS. Van der Wielen et
al: Mixed stage (I-III); MIS and open; neoadjuvant
therapy. | (17,26,27,29,30) |
| Van der Wielen
et al, 2022 | Global health
perception and life satisfactiond | Decline | Improvement | Return to baseline
only in OTG | Mixed stage
(I-III); MIS and open; neoadjuvant therapy. | (29) |
| Park et al,
2020 | Global health
perception and life satisfactiond | Improvement | Improvement | Improvement | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Park et al,
2020 | Economic
consequences | No significant
change | No significant
change | Increase in year 2,
recovery by year 3 | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Wei et al,
2021; Yan et al, 2023; Lin et al, 2023; Tanaka et
al, 2024; van der Wielen et al, 2022; Lu et al,
2022 | Economic
consequences | No significant
change | No significant
change | No significant
change | Wei et al:
Mixed stage (I-III); MIS. Yan et al: Mixed stage (I-III);
MIS. Lin et al: Mixed stage (I-III); MIS; no neoadjuvant
therapy; adjuvant chemotherapy in 58% (62.5% TLTG, 56.3% LATG).
Tanaka et al: Early stage (IA-IIB); MIS and open. Van der
Wielen et al: Mixed stage (I-III); MIS and open; neoadjuvant
therapy. Lu et al: Mixed stage (I-III); MIS. | (17,26-30) |
 | Table IVComparative findings in functional
and single-item quality of life following gastrectomy. |
Table IV
Comparative findings in functional
and single-item quality of life following gastrectomy.
| Authors, year of
publication | Domain | Comparison | Findings | Group
characteristicsa | (Refs.) |
|---|
| Wei et al,
2021; Yan et al, 2023 | Physical
performance and functional capacity | Stapling technique
(LS vs. CS) | No significant
differences | Wei et al:
Mixed stage (I-III); MIS. Yan et al: Mixed stage (I-III);
MIS | (26,27) |
| Tanaka et
al, 2024; Van der Wielen et al, 2022; Lin et al,
2023 | Physical
performance and functional capacity | MIS vs. open | No significant
differences | Tanaka et
al: Early stage (IA-IIB); MIS and open. Van der Wielen et
al: Mixed stage (I-III); MIS and open; neoadjuvant therapy. Lin
et al: Mixed stage (I-III); MIS; no neoadjuvant therapy;
adjuvant chemotherapy in 58% (62.5% TLTG, 56.3% LATG). | (17,28,29) |
| Kubota et
al, 2024; Lu et al, 2022 | Physical
performance and functional capacity | Pouch vs. no
pouch | No significant
differences | Kubota et
al: Mixed stage (I-III); MIS. Lu et al: Mixed stage
(I-III); MIS. | (16,30) |
| Lin et al,
2023 | Role engagement and
daily living | TLTG vs. LATG | TLTG patients had
better return-to-work ability at 6-12 months; no difference at 3
months | Mixed stage
(I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy in 58%
(62.5% TLTG, 56.3% LATG). | (28) |
| Van der Wielen
et al, 2022 | Role engagement and
daily living | MIS vs. open | Open group showed
persistently lower scores compared with MIS, but not statistically
significant | Mixed stage
(I-III); open and MIS; neoadjuvant therapy | (29) |
| Tanaka et
al, 2024; Van der Wielen et al, 2022 | Social integration
and interaction | Surgical approach;
MIS vs. open | Open group had
higher scores thanlaparoscopic group; OTG back to baseline at 6
months, MITG declined from 3 months onwards | Early stage
(IA-IIB); open and MIS, mixed stage (I-III); open and MIS;
neoadjuvant therapy. | (17,29) |
| Lin et al,
2023 | Social integration
and interaction | Surgical approach;
TLTG vs. LATG | TLTG associated
with better scores throughout first 12 months, but no significant
differences at assessed time points | Mixed stage
(I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy in 58%
(62.5% TLTG, 56.3% LATG). | (28) |
| Lu et al,
2022; Saeki et al, 2024; Kubota et al, 2024 | Social integration
and interaction | Body image; PP-RY
vs. RY; CS vs. LS; aboral pouch (elderly vs. non-elderly) | No significant
differences across comparisons | Lu et al:
Mixed stage (I-III); MIS. Saeki et al: Early stage (IA-IIB);
MIS and open. Kubota et al: Mixed stage (I-III); MIS. | (16,21,30) |
| Saeki et al,
2024 | Social integration
and interaction | Meal frequency | Increased need for
meals may have affected daily living patterns | Early stage
(IA-IIB); MIS and open. | (21) |
| Lu et al,
2022 | Emotional and
psychological well-being | PP-RY vs. RY | PP-RY associated
with significantly improved anxiety scores compared with standard
RY | Mixed stage
(I-III); MIS. | (30) |
| Wei et al,
2021 | Emotional and
psychological well-being | Stapling technique
(CS vs. LS) | CS patients more
likely to experience anxiety at 12 months compared to LS, although
not statistically significant; no significant differences in body
image | Mixed stage
(I-III); MIS. | (26) |
| Yan et al,
2023; van der Wielen et al, 2022; Lin et al,
2023 | Emotional and
psychological well-being | MIS vs. open | Some MIS cohorts
reported slightly improved emotional scores at 6 months, but no
significant differences overall; open group reported worse body
image scores than laparoscopic, although not statistically
significant | Yan et al:
Mixed stage (I-III); MIS. Van der Wielen et al: Mixed stage
(I-III); open and MIS; neoadjuvant therapy. Lin et al: Mixed
stage (I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy
in 58% (62.5% TLTG, 56.3% LATG). | (27-29) |
| Kubota et
al, 2024; Lu et al, 2022 | Emotional and
psychological well-being | Pouch
reconstructions | No significant
differences in emotional or body image outcomes | Kubota et
al: Mixed stage (I-III); MIS. Lu et al: Mixed stage
(I-III); MIS. | (16,30) |
| Yan et al,
2023; Tanaka et al, 2024; Lin et al, 2023; Lu et
al, 2022 | Cognitive
resilience | Stapling technique
(CS vs. LS); MIS vs. open; Pouch vs. no pouch reconstruction | No significant
differences | Yan et al:
Mixed stage (I-III); MIS. Tanaka et al: Early stage
(IA-IIB); open and MIS. Lin et al: Mixed stage (I-III); MIS;
no neoadjuvant therapy; adjuvant chemotherapy in 58% (62.5% TLTG,
56.3% LATG). Lu et al: Mixed stage (I-III); MIS. | (17,27,28,30) |
| Lin et al,
2023 | Global health
perception and life satisfaction | TLTG vs. LATG | TLTG patients
showed earlier improvements compared with LATG | Mixed stage
(I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy in 58%
(62.5% TLTG, 56.3% LATG). | (28) |
| Van der Wielen
et al, 2022 | Global health
perception and life satisfaction | MIS vs. open | MIS associated with
higher global health scores at 1 year; consistently better than
open gastrectomy | Mixed stage
(I-III); open and MIS; neoadjuvant therapy. | (29) |
| Lu et al,
2022 | Global health
perception and life satisfaction | PP-RY vs. RY | PP-RY
reconstructions associated with better overall QoL compared with
standard RY | Mixed stage
(I-III); MIS. | (30) |
| Kubota et
al, 2024; Tanaka et al, 2024; Saeki et al,
2024 | Global health
(dissatisfactionb) | Stapling technique
(CS vs. LS); elderly vs. non-elderly; MIS vs. open | No significant
differences in dissatisfaction scores across comparisons | Kubota et
al: Mixed stage (I-III); MIS. Tanaka et al: Early stage
(IA-IIB); open and MIS. Saeki et al: Early stage (IA-IIB);
MIS and open. | (16,17,21) |
| Wei et al,
2021; an et al, 2023 | Economic
consequences | Stapling technique
(LS vs. CS) | LS associated with
better financial scores at 12 months compared with CS | Wei et al:
Mixed stage (I-III); MIS. Yan et al: Mixed stage (I-III);
MIS. | (26,27) |
| Tanaka et
al, 2024 | Economic
consequences | MIS vs. open | Open group reported
higher burden scores, laparoscopic lower scores; differences not
statistically significant | Early stage
(IA-IIB); MIS and open. | (17) |
Physical performance and functional capacity.
This domain encompasses the recovery of mobility, self-care and
general physical strength following total gastrectomy.
In terms of chronological patterns, an early decline
with recovery complete by 6-12 months was found to be consistent
across the cohorts. At >12 months, recovery remained below the
baseline in adjuvant-exposed minimally invasive surgery (MIS)
cohorts (9,28) whereas early-stage (17), neoadjuvant-managed mixed-stage
(29) and mixed-stage MIS cohorts
without reported chemotherapy (26)
returned to baseline. Comparing the various techniques, physical
performance outcomes were shown to be comparable, with no
significant differences reported between stapling methods, surgical
approaches or reconstruction types (16,17,26-30).
Role engagement and daily living. ‘Role
functioning’ reflects the ability of the patient to resume work,
leisure activities and daily responsibilities following total
gastrectomy.
In early-stage (17)
and neoadjuvant-managed mixed-stage (29) cohorts, role functioning was found to
decline initially, but returned to the baseline within the first
year. By contrast, in mixed-stage MIS cohorts with adjuvant therapy
(28), improvements were observed,
although baseline levels were not restored, whereas advanced-stage
patients exposed to adjuvant therapy showed a sustained decline
(9).
Comparative analyses subsequently revealed limited
technique-associated differences in role functioning. In
mixed-stage MIS cohorts without neoadjuvant therapy, return to work
improved following TLTG compared with LATG, although only from 6-12
months onward (28). Open cohorts
reported worse role functioning compared with MIS cohorts, although
these differences were found not to be significant (29).
Social integration and interaction. ‘Social
functioning’ concerns how effectively a patient can engage in
social activities, maintain interpersonal relationships and adapt
to post-gastrectomy lifestyle changes.
Chronological trajectories were found to vary among
the cohorts. In advanced-stage MIS patients with adjuvant therapy,
the scores improved initially, peaked at 1 year, and subsequently
declined (9). In mixed-stage groups,
the OTG cohort returned to baseline by 12 months, and later
exceeded it, whereas the MITG cohort stayed below the baseline
throughout follow-up (29).
When the techniques were compared, in some studies,
open surgery cohorts scored higher than the MIS cohorts; the OTG
group returned to baseline by 6 months, whereas the MITG group
declined from 3 months onward (17,29). The
patients who underwent TLTG exhibited improved social interaction
compared with patients who underwent LATG during the first
post-operative year, although this improvement was found not to be
statistically significant (28).
Increased meal frequency was also reported in early-stage cohorts,
potentially affecting daily living patterns (21).
Emotional and psychological well-being. This
domain captures the patients' emotional functioning and
psychological distress, primarily assessed using the EORTC QLQ-C30
questionnaire and STO22 module (anxiety domain), with additional
contribution from the EQ-5D instrument (anxiety/depression
dimension).
Emotional outcomes varied according to the cohort
and over time. Advanced-stage patients with MIS who received
adjuvant therapy showed early improvement that declined after 1-2
years (9), whereas early-stage
patients treated without systemic therapy experienced only a
transient early decline, followed by recovery (17). Mixed-stage MIS cohorts generally
reported stable scores, with occasional transient improvement that
returned to baseline (26-30).
Technique-associated effects on emotional
functioning were found to be limited. PP-RY was associated with
improved anxiety scores compared with standard RY (30). Stapling methods exhibited no
consistent impact, although patients who underwent CS tended to
report more anxiety at 12 months compared with those who underwent
LS (26). MIS cohorts occasionally
demonstrated slightly improved emotional scores at 6 months,
although the overall differences compared with open surgery
patients were found not to be not significant; open cohorts
occasionally reported higher body image scores, although again,
these were not statistically significant (27-29).
Furthermore, pouch reconstructions exhibited no significant
differences in either emotional or body image outcomes (16,30).
Body image was found to be comparable across
reconstructions and technical variations, including pouch type,
stapling method, age-related pouch use and the surgical approach
(16,21,30).
Cognitive resilience. ‘Cognitive resilience’
relates to the ability of a patient to maintain concentration,
memory and mental clarity during recovery. It is specifically
assessed in the EORTC QLQ-C30 questionnaire, although it is not
routinely captured by the majority of other QoL instruments used in
post-gastrectomy studies.
Over time, a decline was observed during the first
year with only minimal recovery in advanced-stage MIS cohorts who
were receiving adjuvant therapy (9),
whereas early-stage and mixed-stage cohorts managed largely without
systemic therapy maintained stable scores throughout follow-up
(17,26,27,29,30).
Finally, no significant differences in cognitive
outcomes were reported across stapling methods, surgical approaches
or pouch reconstructions (17,27,28,30).
Single items. Global health perception
and life satisfaction
This domain represents the subjective well-being,
satisfaction or dissatisfaction and adaptation to life of a
patients following total gastrectomy.
The course of global health recovery differed among
the cohorts. In mixed-stage groups with neoadjuvant therapy, the
scores declined early on, improved by 1 year, and returned to the
baseline only with the OTG group, whereas the MITG group remained
below baseline throughout follow-up (29). By contrast, advanced-stage MIS
cohorts with adjuvant therapy exhibited gradual improvement over
time (9).
In terms of comparing techniques, TLTG was
associated with earlier improvements compared with LATG in a
mixed-stage MIS cohort who did not receive neoadjuvant therapy
(28). MIS was associated with
higher global health scores in some cohorts compared with open
surgery in mixed-stage groups who were treated with neoadjuvant
therapy (29). Finally, PP-RY
reconstructions were found to be associated with an improved
overall quality of life compared with standard RY (30).
With regard to dissatisfaction, no significant
differences were observed across the stapling techniques, between
elderly and non-elderly patients, or between MIS and open surgery
approaches (16,17,21).
Economic consequences. Financial difficulties
were assessed in a subset of studies as a measure of post-operative
socioeconomic burden. This domain was not evaluated in studies
utilizing the PGSAS instrument.
Chronological patterns were found to be largely
stable across the studies. The majority of the cohorts, including
early- and mixed-stage groups managed with MIS or open approaches,
reported no significant changes over time (17,26-30).
By contrast, patients with advanced-stage MIS exposed to adjuvant
therapy experienced an increase in financial strain during the
second year, with recovery by the 3rd year (9).
Upon comparing the techniques, the stapling method
appeared to influence financial outcomes, with LS associated with
improved scores at 12 months compared with CS in mixed-stage MIS
cohorts (26,27). Open surgery groups reported higher
burden scores than the MIS groups in early-stage cohorts, although
these differences were found not to be statistically significant
(17).
Symptom experience and disease
burden
A detailed summary of the chronological evolution of
symptom-related QoL outcomes following gastrectomy is provided in
Table V. The most concerning
post-gastrectomy symptoms, indicatively the eating restrictions,
pain, diarrhoea and reflux, worsened in the early post-operative
period, and gradually improved within the first year, although
reflux often persisted, whereas eating restrictions were slower to
improve and sometimes remained in the long term.
 | Table VChronological patterns of
symptom-related quality of life following gastrectomy. |
Table V
Chronological patterns of
symptom-related quality of life following gastrectomy.
| Authors, year of
publication | Symptom | 0-6 months | 6-12 months | >12 months | Group
characteristicsa,b,c | (Refs). |
|---|
| Park et al,
2020 | Fatigue | Peak | Plateau | Return to baseline
by year 3 | Advanced stage
(II-III); MIS; 65% received adjuvant therapy | (9) |
| Van der Wielen
et al, 2022 | Fatigue | Peak | Gradual return to
baseline | Baseline | Mixed stage
(I-III); MIS and open; neoadjuvant therapy | (29) |
| Van der Wielen
et al, 2022 |
Nausea/vomiting | Peak | Return near
baseline by 6 months | Stable by year
3 | Mixed stage
(I-III); MIS and open; neoadjuvant therapy | (29) |
| Park et al,
2020 |
Nausea/vomiting | No significant
change | No significant
change | No significant
change | Advanced stage
(II-III); MIS; 65% received adjuvant therapy | (9) |
| Van der Wielen
et al, 2022 | Dyspnoea | Peak | Return near
baseline by 6 months | Stable by year
3 | Mixed stage
(I-III); MIS and open; neoadjuvant therapy | (29) |
| Park et al,
2020 | Dyspnoea | No significant
change | No significant
change | No significant
change | Advanced stage
(II-III); MIS; 65% received adjuvant therapy | (9) |
| Park et al,
2020 | Pain | Peak | Improvement | Secondary peak in
year 2, slight improvement after secondary peak | Advanced stage
(II-III); MIS; 65% received adjuvant therapy | (9) |
| Tanaka et
al, 2024 | Pain | Peak | Return to baseline
by 12 months | Baseline | Early stage
(IA-IIB); MIS and open | (17) |
| Van der Wielen
et al, 2022 | Pain | Peak | Return to baseline
by 12 months for the open group, MIS remained elevated | Baseline for open
surgery, elevated for MIS | Mixed stage
(I-III); MIS and open; neoadjuvant therapy | (29) |
| Wei et al,
2021; Yan et al, 2023 | Pain | Not reported | Improved scores at
6-12m | Not reported | Mixed stage
(I-III); MIS; proportion receiving neoadjuvant therapy not
reported. | (26,27) |
| Park et al,
2020; van der Wielen et al, 2022 | Constipation | No change | No change | No change | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Mixed stage (I-III); MIS and open;
neoadjuvant therapy. | (9,29) |
| Wei et al,
2021; Yan et al, 2023; van der Wielen et al,
2022 | Diarrhoea | Decline; worse in
neoadjuvant patients at 3 months | Return to base line
by 6–12 months | Baseline | Wei et al:
Mixed stage (I-III); MIS. Yan et al: Mixed stage (I-III);
MIS; proportion receiving neoadjuvant therapy not reported. Van der
Wielen et al: Mixed stage (I-III); MIS and open; neoadjuvant
therapy. | (26,27,29) |
| Park et al,
2020 | Diarrhoea | Decline; worse in
neoadjuvant patients at 3 months | Elevated | Elevated up to 2
years | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Park et al,
2020; van der Wielen et al, 2022 | Dysphagia | Peak immediately
postoperatively and at 3 months | Return to baseline
by 6 months | No change | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Mixed stage (I-III); MIS and open;
neoadjuvant therapy. | (9,29) |
| Park et al,
2020; van der Wielen et al, 2022; Lin et al,
2023 | Reflux | Increased | Remained elevated;
improvement noted TLTG vs. LATG | Remained
elevated | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Mixed stage (I-III); MIS and open;
neoadjuvant therapy. Lin et al: Mixed stage (I-III); MIS; no
neoadjuvant therapy; adjuvant chemotherapy in 58% (62.5% TLTG,
56.3% LATG). | (9,28,29) |
| Park et al,
2020; van der Wielen et al, 2022 | Eating
restrictions | Decline | No improvement | Persisted in MITG;
improved in OTG | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Stages I-III; MIS and open; neoadjuvant
therapy. | (9,29) |
| Park et al,
2020; van der Wielen et al, 2022; Wei et al, 2021;
Yan et al, 2023; Lin et al, 2023 | Taste
disturbances | Peak; worse in
MIS | Peak; more evident
in neoadjuvant patients at 6 months | No significant
further changes | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Mixed stage (I-III); MIS and open;
neoadjuvant therapy. Wei et al: Mixed stage (I-III); MIS.
Yan et al: Mixed stage (I-III); MIS; proportion receiving
neoadjuvant therapy not\ reported. Lin et al: Mixed stage
(I-III); MIS; no neoadjuvant therapy; adjuvant chemotherapy in 58%
(62.5% TLTG, 56.3% LATG). | (9,26-29) |
| Park et al,
2020; van der Wielen et al, 2022; Lin et al,
2023 | Appetite loss | No significant
change; worse scores in neoadjuvant patients | Recovery to
baseline by 6 months; better scores after PP-RY | No change | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Stages I-III; MIS and open; neoadjuvant
therapy. Lin et al: Mixed stage (I-III); MIS; no neoadjuvant
therapy; adjuvant chemotherapy in 58% (62.5% TLTG, 56.3%
LATG). | (9,28,29) |
| Park et al,
2020; van der Wielen et al, 2022 | Sleep | Stable; worse in
MIS | Recovery to
baseline by 6 months | Baseline | Park et al:
Advanced stage (II-III); MIS; 65% received adjuvant therapy. Van
der Wielen et al: Mixed stage (I-III); MIS and open;
neoadjuvant therapy. | (9,29) |
| Park et al,
2020 | Mouth dryness | Increased; worse in
MIS | Increased | Slight improvement
after 2 years | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Van der Wielen
et al, 2022 | Mouth dryness | Increased;
worsening scores in MIS for the first 3 months | Recovery to
baseline by 6 months | Baseline | Mixed stage
(I-III); MIS and open; neoadjuvant therapy. | (29) |
| Park et al,
2020 | Hair loss | Worsening | Worsening | Improved after year
2 | Advanced stage
(II-III); MIS; 65% received adjuvant therapy. | (9) |
| Tanaka et
al, 2024 | Dumping | No significant
findings | No significant
findings | No significant
findings | Early stage
(IA-IIB); MIS and open. | (17) |
Variations across studies were observed in the
context of differing patient characteristics and treatment
exposures. Prolonged fatigue, pain, diarrhoea and taste disruptions
were found to be more pronounced in advanced-stage patients, the
majority of whom received adjuvant therapy (9), whereas earlier recovery was described
in mixed-stage cohorts treated with neoadjuvant therapy (29). Stable or improving outcomes were
observed both in early-stage cohorts (17) and in mixed-stage MIS groups where
chemotherapy exposure was absent or not reported (26-28).
Differences in recovery trajectories were also
observed between surgical techniques. Notably, minimally invasive
approaches were reported in some studies to be associated with
slower resolution of pain compared with open surgery (9,29). By
contrast, in a single study, totally laparoscopic procedures were
found to be associated with an earlier improvement in reflux
compared with laparoscopic-assisted techniques (29), although this observation was made
from a single study, and therefore should be interpreted with
caution.
Table VI summarises
comparative findings in symptom-associated QoL. Symptom outcomes
were similar across studies regardless of stage distribution or
perioperative therapy. Most of the technique-associated comparisons
revealed no significant differences; when present, the differences
were small and inconsistent. Within MIS, the stapler type and
anastomotic method were associated with isolated differences,
including less constipation and dysphagia, but more cases of early
diarrhoea and worse reflux in certain subgroups (26,27). A
short-term advantage was observed for TLTG over LATG at 6 months,
although without long-term differences (28). The reconstruction method also
appeared relevant, with PP-RY associated with reduced pain and
improved appetite outcomes compared with standard RY (30).
 | Table VIComparative findings in
symptom-associated quality of life following gastrectomy. |
Table VI
Comparative findings in
symptom-associated quality of life following gastrectomy.
| | |
Statistically significant findings of
symptom burden on surgical approach comparison | |
|---|
| Authors, year of
publication | Group
characteristicsa | Comparison
parameters | Fatigue | Nausea/vomiting,
dyspnoea | Pain | Constipation | Diarrhoea | Dysphagia | Reflux | Eating
restrictions, meal quantity | Taste
disturbances | Appetite loss | Sleep
disturbances | Mouth dryness | Hair loss | Dumping
symptoms | (Refs.) |
|---|
| Wei et al,
2021 | Stages I-III,
MIS | Linear stapler vs.
circular stapler | No | No | No | Yese | No | Yesg | No | No | No | No | No | No | No | No | (26) |
| Yan et al,
2023 | Stages I-III,
MIS | Linear stapler i)
vs. circular stapler ii) Intracorporeal vs. extracorporeal
anastomosis iii) Overlap vs. π-shaped vs. OrVil anastomosis | No | No | No | No | Yesf | Yesg | Yesi | Yesj | No | No | No | No | No | No | (27) |
| Lin et al,
2023 | Stages I-III, MIS;
no neoadjuvant therapy; adjuvant chemotherapy in 58% (62.5% TLTG,
56.3% LATG). | TLTG vs. LATG | No | No | No | No | No | Yesh | No | Yesk | No | No | No | No | No | No | (28) |
| Tanaka et
al, 2024 | Stages IA-IIB, open
and MIS | Laparoscopic vs.
open gastrectomy | Yesb | No | No | No | No | No | No | No | No | No | No | No | No | No | (17) |
| Van der Wielen
et al, 2022 | Stages I-III,
neoadjuvant treatment | Open vs. MIS
gastrectomy in neoadjuvant patients | No | No | No | No | No | No | No | No | No | No | Non | No | No | No | (29) |
| Saeki et al,
2024 | Stages IA-IIB, open
and MIS | i) HRM findings ii)
Linear stapler vs. Circular Stapler | No | No | Yesc | No | No | No | No | No | No | No | No | No | No | No | (21) |
| Lu et al,
2022 | Stages I-III,
MIS | R-Y vs. PP-RY | No | No | Yesd | No | No | No | No | Yesl | No | Yesm | No | No | No | No | (30) |
| Kubota et
al, 2024 | Stages I-III,
MIS | Aboral pouch in the
elderly vs. non-elderly population | No | No | No | No | No | No | No | No | No | No | No | No | No | No | (16) |
| Park et al,
2020 | Stages II-III, MIS,
65% adjuvant | TG vs. DG (TG data
used only) | No | No | No | No | No | No | No | No | No | No | No | No | No | No | (9) |
Influence of other clinical
variables
In addition to questionnaire domains, the present
scoping review recorded whether the studies reported on general
health status, peri-operative therapy or post-operative
complications, as these may influence QoL outcomes. Adjuvant
therapy was described in two studies (9,28),
whereas three reported on neoadjuvant therapy (27-29).
Post-operative complications were mentioned in five studies
(16,17,27,28,30).
However, none of the studies analysed these variables in
conjunction with QoL.
Discussion
QoL is increasingly recognised as a key outcome in
cancer care, reflecting not only treatment success but also the
ability of a patient to adapt and recover following major
interventions, such as total gastrectomy. While oncological results
remain essential, understanding functional recovery is equally
important and may guide surgical decision-making (26).
The present review found that physical, role and
emotional functioning consistently declined at an early stage
post-operatively, with recovery typically beginning ~6 months. The
pattern observed in the present scoping review is in agreement with
other mixed gastrectomy studies (12,15).
While this early decline is partly expected as the body heals from
a major surgery, it is also influenced by post-gastrectomy symptoms
that can significantly affect daily functioning (12). Moreover, this period often coincides
with the initiation of adjuvant chemotherapy or chemoradiotherapy
for several patients, which can further impact QoL. While recovery
to baseline values may occur by 6 months, it should be noted that
baseline does not necessarily reflect an optimal state,
particularly in patients with advanced disease (9,29). As
highlighted in the wider literature, recognising this trajectory
can support patient expectation management and inform tailored
rehabilitation planning (8,9).
The present scoping review identified mixed outcomes
on social functioning following total gastrectomy. The studies
demonstrating an early decline are in agreement with existing
literature; Hu et al (15)
reported a marked decline in social functioning after surgery,
particularly during the first 45 post-operative days, with
subsequent improvement to near-baseline levels. Vaccaro et
al (31) observed a similar
pattern, suggesting that changes in body image, disruption of
established routines and pleasures, and the need to adapt to new
eating patterns may hinder social engagement, particularly during
meals. While a decline in social functioning over time is well
documented in the literature, the improvement reported in some
studies, particularly at ~12-months, may reflect the gradual
adaptation of patients to post-operative changes and the resumption
of social activities (17,29). Surgical approach did not appear to
have a consistent or statistically significant impact on social
functioning. Given the multifactorial nature of this domain,
including physical recovery, nutritional adaptation, emotional
well-being, and social support, isolating the effect of surgery
alone is challenging. Longitudinal, standardised assessment may be
needed to clarify the true trajectory of social functioning after
total gastrectomy.
In the present scoping review, only the studies that
used the EORTC QLQ-C30 questionnaire assessed cognitive status. Of
note, one study reported a measurable decline (9), while the others found no change over
time or variation based on surgical technique (17,26,27,29,30).
Overall, the available evidence is insufficient to draw conclusive
interpretations in this domain, and several factors may account for
this inconsistency. A likely contributing factor is the early
timing of assessment in certain studies. This may have captured
short-term post-operative cognitive dysfunction, which typically
resolves within 3 months (32,33).
Another contributing factor may be the limited structure of the
cognitive domain within these QoL instruments, which typically
includes only two questions and does not constitute a formal
cognitive assessment, such as the Mini-Mental State Examination
(34). As such, their sensitivity to
subtle or transient cognitive changes may be limited. The
aforementioned considerations highlight the need for more
thoughtful application and interpretation of existing QoL tools
when assessing post-operative cognitive function.
The present scoping review found no evidence of
significant financial strain over time between subgroups; a
non-significant trend favouring linear over circular stapler
techniques may relate to the higher post-operative complication
rates reported with circular stapling, including bleeding,
anastomotic stenosis and dysphagia (26,27). The
only study demonstrating a decrease in financial burden involved
early-stage GC, suggesting the improvement may be linked to a
reduced need for post-operative therapy rather than surgical
approach (17). Literature on
oncologic surgery for upper gastrointestinal cancers, highlights
that chemotherapy, financial demands of treatment-including
personal expenses, travel and accommodation for care- and other
related costs, can place a considerable strain on patients'
economic well-being (35,36). Given the distinct patient groups and
the varied socioeconomic contexts across study sites, firm
conclusions regarding the financial domain remain challenging.
Symptom trajectories appeared to vary according to
patient and treatment characteristics rather than surgical approach
alone. Patients with advanced-stage disease receiving adjuvant
therapy experienced more prolonged fatigue, pain, diarrhoea and
taste disruptions (9), whereas
mixed-stage cohorts treated with neoadjuvant therapy reported
earlier recovery in several domains (29). Stable or improving symptom profiles
were more often observed in early-stage cohorts (17) and in mixed-stage MIS groups without
chemotherapy exposure (26-28).
These findings suggest that disease stage and peri-operative
therapy may exert greater influence on symptom burden than surgical
technique itself. Variability in follow-up schedules and symptom
reporting tools further complicates interpretation, underscoring
the need for longitudinal, standardised assessment to clarify how
patient and treatment factors interact in shaping post-operative
symptom profiles.
The observation that global QoL improved even when
symptoms persisted suggests that overall well-being is not simply
the cumulative effect of individual complaints, but also reflects
the capacity of patients to adapt and reframe their
post-gastrectomy experience. Similar findings have been described
in other oncologic populations, where coping mechanisms,
resilience, and social support play a decisive role in maintaining
global QoL despite ongoing treatment-related burdens (35,36).
This perspective highlights the importance of integrating
psychosocial and rehabilitative support into survivorship care, in
parallel with symptom management. While surgical modifications such
as minimally invasive or pouch reconstructions may influence
short-term recovery, their impact on long-term global QoL appears
limited when compared with the broader determinants of adaptation
and support.
Overall, the patterns observed across the included
studies suggest that post-operative QoL trajectories following
total gastrectomy may be influenced more strongly by disease stage
and exposure to systemic therapy than by differences in surgical
technique alone. Nevertheless, interpretation of the patterns
observed across the reviewed studies should be undertaken with
caution, as the included studies differ substantially in stage
distribution, exposure to perioperative systemic therapy, and
timing of QoL assessment. It would indeed be of considerable
interest to better understand the independent contribution of
surgical technique, systemic therapy, and disease-related factors
to post-operative QoL, particularly the effect of each factor
considered in isolation. In the currently available literature,
however, these elements are closely intertwined within diverse
patient populations, and the combination of sample size and study
design limits the ability to isolate their individual effects.
Further studies specifically designed to address these questions
may help clarify the relative contribution of each factor.
Reviewing the current literature revealed several
key concerns regarding QoL assessment following total gastrectomy.
While QoL is increasingly recognised as an important component of
GC care, relatively few studies consider it as a primary outcome,
with the majority still focusing on traditional surgical endpoints
such as complications, resection margins, and survival. When QoL is
assessed, it is often used to compare surgical techniques or
patient subgroups rather than to chart the full course of recovery.
This tendency provides only a partial view of recovery, limiting
insight into the longer-term physical, emotional and social
challenges faced by patients after surgery.
Another dimension of heterogeneity relates to
geography. The present scoping review found a marked imbalance in
QoL research on curative total gastrectomy over the past 5 years,
with the majority of studies conducted in East Asia, where GC is
more common (1). While these data
provide valuable clinical insight, the predominance of Asian study
populations limits the generalisability of findings, particularly
in culturally sensitive domains such as role functioning, dietary
practices and social engagement. Notably, despite the development
of one of the most comprehensive and widely validated QoL
instruments for cancer patients, the EORTC QLQ-C30 and its
gastric-specific module, Western centres have contributed
relatively few QoL studies focused specifically on total
gastrectomy. Addressing the regional imbalance requires
consideration of cultural and geographical context when
interpreting QoL instruments and when formulating clinical
recommendations.
Building on the challenges outlined above, a major
difficulty in interpreting QoL outcomes is the marked heterogeneity
among available studies. This includes differences in study design,
such as retrospective and prospective approaches, differences in
setting between multicentre and single-centre studies and
variations in focus, with certain studies comparing surgical
techniques within specific subgroups. Such variability limits the
generalisability of findings and complicates efforts to compare and
synthesise results across the literature.
Marked variability in the timing of QoL distribution
further complicates interpretation. Only a small number of studies
applied questionnaires pre-operatively or in the early
post-operative period (9,17,27,29),
when physical and psychological effects are most acute. While
certain studies tracked changes over time (9,21,17,28),
others relied on a single time point (16,26,27,29,30). The
fact that QoL is primarily considered in relation to long-term
outcomes may explain why questionnaires are not commonly used to
capture the impact of short-term complications, which are often
described under the separate concept of ‘quality of recovery’
(37,38). Yet, nothing in the instrument manuals
precludes their early use, leaving an opportunity to better
understand the immediate post-operative experience; an approach
that could yield valuable insights into the trajectory from
short-term recovery to long-term well-being (12,27).
These methodological differences are further
influenced by the limitations of the instruments themselves. In the
present scoping review, no single questionnaire was found to be
capable of comprehensively assessing QoL following total
gastrectomy. The EORTC QLQ-C30, developed for patients with cancer,
covers a broad range of domains, but requires the STO22 module to
capture GC-specific symptoms, resulting in 52 questions, which can
be burdensome for patients and may reduce response rates.
Similarly, researchers using the PGSAS-37 often supplemented it
with the EORTC or omitted domains, such as functioning and
financial status entirely. Overlap between certain domains and
omission of others not only limits comparability between studies,
but also impedes the ability to track the full course of recovery.
The lack of uniformity in scoring and scale direction across
instruments requires familiarity with each tool's methodology,
increases the complexity of analysis, and makes cross-study
comparisons more difficult. The aforementioned challenges emphasise
the importance of applying existing instruments within a
standardised framework to minimise respondent burden while ensuring
all relevant domains are assessed.
Finally, although the majority of studies reported
clinical variables, such as pre-operative treatment or
post-operative complications, their potential influence on QoL
outcomes was rarely explored. Most importantly, none of the studies
considered the impact of post-operative chemotherapy on QoL,
despite its recognised effect on recovery and overall well-being.
These gaps are particularly critical, given that several patients
with GC may already be malnourished, anxious, or psychologically
burdened at the time of diagnosis. Without consideration of these
variables, QoL results may reflect not only the effects of surgery
but also broader clinical and psychosocial conditions. This lack of
adjustment can confound observed QoL patterns, making it difficult
to separate the effects of surgery from those of the underlying
disease, treatment-related side effects, or pre-existing patient
conditions. To improve interpretability and comparability, future
research would benefit from clearly defined assessment timelines
and a systematic evaluation of pre-operative status, post-operative
course, and adjuvant therapies.
The present scoping review has certain limitations.
As a scoping rather than a systematic review, it may not capture
all available literature despite a focused and thorough search.
Additionally, by limiting inclusion to patients undergoing total
gastrectomy without additional major organ resection, certain
relevant data may have been excluded, potentially affecting the
completeness of the findings. Finally, the inconsistency in how QoL
is assessed and reported across studies makes comparison
challenging and may weaken the overall interpretability of
results.
In conclusion, total gastrectomy remains a
life-altering procedure with long-term consequences for the daily
life of a patient. Beyond survival and surgical endpoints, QoL is a
critical outcome as it can influence treatment decisions and guide
both clinicians and patients through post-operative challenges. A
clear understanding of QoL in both the early and late phases of
recovery is essential for informing expectations and optimising
follow-up strategies. Equally, evaluating how different surgical
techniques shape these outcomes can provide valuable guidance for
shared decision-making. To achieve this, more QoL-focused studies
are needed, designed to capture both chronological changes and
procedure-specific outcomes across a range of cultural and clinical
settings.
Acknowledgements
Part of the material presented in this manuscript
was previously presented as a poster at the 34th Panhellenic
Congress of Surgery and International Surgical Forum, Athens,
Greece, November 2025.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
ME conceived and designed the scoping review,
performed the literature search, data extraction, synthesis and
drafted the manuscript. DA contributed to data extraction and
interpretation, and provided a critical review and revisions of the
manuscript. AP created the tables and figures, and contributed to
the interpretation of the findings. MD contributed to manuscript
drafting, data interpretation and provided critical revisions. DT
contributed to the conception of the study and offered surgical
expertise to ensure clinical accuracy. TT assisted with the
literature search and data cross-checking. GZ and KT contributed to
manuscript revisions, data interpretation and ensured intellectual
rigour. ME and DA confirm the authenticity of all the raw data. All
authors reviewed and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Use of artificial intelligence tools
During the preparation of this work, AI tools were
used to improve the readability and language of the manuscript or
to generate images, and subsequently, the authors revised and
edited the content produced by the AI tools as necessary, taking
full responsibility for the ultimate content of the present
manuscript.
References
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263.
2024.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lordick F, Carneiro F, Cascinu S, Fleitas
T, Haustermans K, Piessen G, Vogel A and Smyth EC: ESMO Guidelines
Committee. Electronic address: clinicalguidelines@esmo.org. Gastric
cancer: ESMO Clinical Practice Guideline for diagnosis, treatment
and follow-up. Ann Oncol. 33:1005–1020. 2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Ajani JA, D'Amico TA, Bentrem DJ, Chao J,
Cooke D, Corvera C, Das P, Enzinger PC, Enzler T and Fanta P:
Gastric cancer, version 2.2022, NCCN clinical practice guidelines
in oncology. J Natl Compr Canc Netw. 20:167–19. 2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Japanese Gastric Cancer A: Japanese
gastric cancer treatment guidelines 2018 (5th edition). Gastric
Cancer. 24:1–21. 2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kim SG, Seo HS, Lee HH, Song KY and Park
CH: Comparison of the Differences in Survival Rates between the 7th
and 8th Editions of the AJCC TNM Staging System for Gastric
Adenocarcinoma: A Single-Institution Study of 5,507 Patients in
Korea. J Gastric Cancer. 17:212–219. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Husson O, de Rooij BH, Kieffer J,
Oerlemans S, Mols F, Aaronson NK, van der Graaf WTA and van de
Poll-Franse LV: The EORTC QLQ-C30 summary score as prognostic
factor for survival of patients with cancer in the ‘Real-World’:
Results from the Population-Based PROFILES registry. Oncologist.
25:e722–e732. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Grosek J, Zavrtanik H and Tomažič A:
Health-related quality of life after curative resection for gastric
adenocarcinoma. World J Gastroenterol. 27:1816–1827.
2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Wang CJ, Suh YS, Lee HJ, Park JH, Park SH,
Choi JH, Alzahrani F, Alzahrani K, Kong SH, Park DJ, et al:
Post-operative quality of life after gastrectomy in gastric cancer
patients: A prospective longitudinal observation study. Ann Surg
Treat Res. 103:19–31. 2022.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Park KB, Park JY, Lee SS, Chung HY and
Kwon OK: Chronological changes in quality of life and body
composition after gastrectomy for locally advanced gastric cancer.
Ann Surg Treat Res. 98:262–269. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ikeda M, Yoshida M, Mitsumori N, Etoh T,
Shibata C, Terashima M, Fujita J, Tanabe K, Takiguchi N, Oshio A
and Nakada K: Assessing optimal Roux-en-Y reconstruction technique
after total gastrectomy using the Postgastrectomy Syndrome
Assessment Scale-45. World J Clin Oncol. 13:376–387.
2022.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pinheiro RN, Mucci S, Zanatto RM, Picanço
Junior OM, Bottino AAG, Fontoura RP and Lopes Filho GJ: Quality of
life as a fundamental outcome after curative intent gastrectomy for
adenocarcinoma: Lessons learned from patients. J Gastrointest
Oncol. 10:989–998. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Hu Y and Zaydfudim VM: Quality of life
after curative resection for gastric cancer: Survey metrics and
implications of surgical technique. J Surg Res. 251:168–179.
2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Xu R, Gu Q, Xiao S, Zhao P and Ding Z:
Patient-reported gastrointestinal symptoms following surgery for
gastric cancer and the relative risk factors. Front Oncol.
12(951485)2022.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Aaronson NK, Ahmedzai S, Bergman B,
Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman
SB, de Haes JC, et al: The european organization for research and
treatment of cancer QLQ-C30: A quality-of-life instrument for use
in international clinical trials in oncology. J Natl Cancer Inst.
85:365–376. 1993.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hu Y, Vos EL, Baser RE, Schattner MA,
Nishimura M, Coit DG and Strong VE: Longitudinal analysis of
Quality-of-Life recovery after gastrectomy for cancer. Ann Surg
Oncol. 28:48–56. 2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Kubota A, Yamauchi S, Yoshimoto Y, Tsuda
K, Yube Y, Kaji S, Orita H, Brock MV and Fukunaga T: Impact of the
Aboral Pouch in Roux-en-Y reconstruction after laparoscopic total
gastrectomy for elderly patients. Juntendo Iji Zasshi. 70:204–213.
2024.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Tanaka C, Kanda M, Misawa K, Mochizuki Y,
Hattori M, Sueoka S, Watanabe T, Yamada T, Murotani K and Kodera Y:
Long-term quality of life after open and laparoscopic total
gastrectomy for stage I gastric cancer: A prospective
multi-institutional study (CCOG1504). Ann Gastroenterol Surg.
8:999–1007. 2024.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Brooks R, Boye KS and Slaap B: EQ-5D: A
plea for accurate nomenclature. J Patient Rep Outcomes.
4(52)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Pinheiro RN, Mucci S, Zanatto RM, Picanço
Junior OM, Bottino AAG, Fontoura RP and Lopes Filho GJ: Influence
of the centralizing gastric cancer surgery on the health-related
quality of life in Brazil. J Gastrointest Oncol. 14:1235–1249.
2023.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Eom BW, Lee J, Lee IS, Son YG, Ryu KW, Kim
SG, Kim HI, Kim YW, Kong SH, Kwon OK, et al: Development and
validation of a Symptom-Focused quality of life questionnaire
(KOQUSS-40) for gastric cancer patients after gastrectomy. Cancer
Res Treat. 53:763–772. 2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Saeki Y, Tanabe K, Ota H, Chikuie E,
Takemoto Y, Karakuchi N, Kohata A, Miura O, Toyama E, Kugimiya N
and Ohdan H: Exploratory study on the impact of intraesophageal
pressure on quality of life in patients following total
gastrectomy: A retrospective cohort study. BMC Surg.
24(217)2024.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Giesinger JM, Kieffer JM, Fayers PM,
Groenvold M, Petersen MA and Scott NW: EORTC Quality of Life Group.
Replication and validation of higher order models demonstrated that
a summary score for the EORTC QLQ-C30 is robust. J Clin Epidemiol.
69:79–88. 2016.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Waddell T, Verheij M, Allum W, Cunningham
D, Cervantes A and Arnold D: Gastric cancer: ESMO-ESSO-ESTRO
Clinical Practice Guidelines for diagnosis, treatment and
follow-up. Ann Oncol. 24 (Suppl 6):vi57–vi63. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sano T, Sasako M, Mizusawa J, Yamamoto S,
Katai H, Yoshikawa T, Nashimoto A, Ito S, Kaji M, Imamura H, et al:
Randomized controlled trial to evaluate splenectomy in total
gastrectomy for proximal gastric carcinoma. Ann Surg. 265:277–283.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tricco AC, Lillie E, Zarin W, O'Brien KK,
Colquhoun H, Levac D, Moher D, Peters MDJ, Horsley T, Weeks L, et
al: PRISMA extension for scoping reviews (PRISMA-ScR): Checklist
and explanation. Ann Intern Med. 169:467–473. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wei M, Wang N, Yin Z, Wu T, Zhou S, Dang
L, Zhang Z, Wu D, Gao P, Zhang B, et al: Short-term and quality of
life outcomes of patients using linear or circular stapling in
esophagojejunostomy after laparoscopic total gastrectomy. J
Gastrointest Surg. 25:1667–1676. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yan Y, Wang D, Mahuron K, Wang X, Lu L,
Zhao Z, Melstrom L, Li C, Paz IB, Liu J, et al: Different methods
of minimally invasive esophagojejunostomy after total gastrectomy
for gastric cancer: Outcomes from two experienced centers. Ann Surg
Oncol. 30:6718–6727. 2023.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lin GT, Chen JY, Chen QY, Que SJ, Liu ZY,
Zhong Q, Wang JB, Lin JX, Lu J, Lin M, et al: Patient-reported
outcomes of individuals with gastric cancer undergoing totally
laparoscopic versus Laparoscopic-assisted total gastrectomy: A
Real-World, propensity Score-matching analysis. Ann Surg Oncol.
30:1759–1769. 2023.PubMed/NCBI View Article : Google Scholar
|
|
29
|
van der Wielen N, Daams F, Rosati R,
Parise P, Weitz J, Reissfelder C, Diez Del Val I, Loureiro C,
Parada-González P, Pintos-Martínez E, et al: Health related quality
of life following open versus minimally invasive total gastrectomy
for cancer: Results from a randomized clinical trial. Eur J Surg
Oncol. 48:553–560. 2022.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Lu J, Wu Z, Liu G, Wang B and Shi L: The
clinical effectiveness of establishing a proximal jejunum pouch
after laparoscopic total gastrectomy: A propensity score-based
analysis. Asian J Surg. 45:425–430. 2022.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Vaccaro S, Díaz Crescitelli ME,
Mastrangelo S, Fornaciari N, Reverberi E, Di Leo S and Ghirotto L:
Patients' experiences in early satiety after total gastrectomy for
gastric cancer: A phenomenological study. Front Nutr.
11(1511113)2024.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Droc G, Isac S, Nita E, Martac C, Jipa M,
Mihai DI, Cobilinschi C, Badea AG, Ojog D, Pavel B, et al:
Postoperative cognitive impairment and pain perception after
Abdominal Surgery-could immersive virtual reality bring more? A
clinical approach. Medicina (Kaunas). 59(2034)2023.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ou-Young J, Royse C, Clarke-Errey S,
El-Ansary D, Riedel B, Griffiths J and Bowyer A: Recovery
trajectories after major abdominal surgery: A retrospective pooled
cohort study. Acta Anaesthesiol Scand. 69(e14576)2025.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Lloyd DG, Ma D and Vizcaychipi MP:
Cognitive decline after anaesthesia and critical care. Continuing
Education in Anaesthesia, Critical Care and Pain. 12:105–109.
2012.
|
|
35
|
Hirata Y, To C, Lyu H, Smith GL, Samuel
JP, Tran Cao HS, Badgwell BD, Katz MHG and Ikoma N: Prevalence of
and factors associated with financial toxicity after pancreatectomy
and gastrectomy. Ann Surg Oncol. 31:4361–4370. 2024.PubMed/NCBI View Article : Google Scholar
|
|
36
|
McCall MD, Graham PJ and Bathe OF: Quality
of life: A critical outcome for all surgical treatments of gastric
cancer. World J Gastroenterol. 22:1101–1113. 2016.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Kleif J, Waage J, Christensen KB and
Gögenur I: Systematic review of the QoR-15 score, a
patient-reported outcome measure measuring quality of recovery
after surgery and anaesthesia. Br J Anaesth. 120:28–36.
2018.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Myles PS, Shulman MA, Reilly J, Kasza J
and Romero L: Measurement of quality of recovery after surgery
using the 15-item quality of recovery scale: A systematic review
and meta-analysis. Br J Anaesth. 128:1029–1039. 2022.PubMed/NCBI View Article : Google Scholar
|