Introduction
Lipopolysaccharide (LPS) is an endotoxin derived
from Gram-negative bacteria and it is a potent stimulus activating
glial cells in the brain. LPS-induced microglial activation affects
the survival of newly formed hippocampal neurons (1). The survival of hippocampal
progenitors has been shown to decrease following co-culture with
microglial cells activated by LPS (2), substantially preventing neuronal
differentiation and significantly increasing glial differentiation
(3). An LPS injection into the
lateral ventricle has been shown to cause deficits in the ability
of spatial learning as regards the Morris water maze test (4). Chronic neuroinflammation is a
hallmark of several neurological disorders associated with
cognitive loss and LPS-induced brain inflammation deteriorates
hippocampus-dependent cognitive deficits (5).
Physical exercise has been demonstrated to exert a
protective effect against various brain disorders (6–8). In
particular, physical exercise has been shown to increase
neurogenesis in the hippocampal dentate gyrus and newly generated
neurons in the hippocampal dentate gyrus improve learning ability
and memory function (7,9,10).
Neurogenesis in the hippocampal dentate gyrus occurs
throughout post-natal life, including adult life and is influenced
by the environment and behavior (10–12).
Adult hippocampal neurogenesis is considered to be composed of
several developmental stages: proliferation, differentiation,
migration, targeting and synaptic integration and various
characteristic neuronal markers are expressed during the specific
stages (13–15).
Among these neuronal markers, doublecortin (DCX) is
a brain-specific microtubule-associated protein and is considered
as the immature neuronal marker. DCX promotes microtubule
polymerization and is present in migrating neuroblasts and young
neurons (16). DCX is also present
in the tips of neurites of non-migratory immature neurons,
indicating that it plays a role in the growth of neuronal
processes, downstream of directional or guidance signals. Thus, DCX
is expressed during neurogenesis by mitotic and early post-mitotic
neurons (15,17).
On the other hand, the neuronal nuclear antigen
(NeuN) can be used as a marker of newly generated neuronal cells.
NeuN is considered as a specific marker of neurons, as it is not
expressed in non-neuronal cells (18). In addition, it does not stain the
nuclei of immature nerve cells until they achieve a stage of
development that approaches mature function. Therefore, NeuN is
regarded as a useful marker of neuronal maturation (19).
As mentioned above, physical exercise increases
neurogenesis in the hippocampal dentate gyrus, resulting in the
improvement or maintenance of learning ability and memory function
(7,9,10).
However, the memory-enhancing effects of exercise in association
with neuronal maturation have not yet been evaluated. In the
present study, we investigated the effects of forced treadmill
exercise and voluntary wheel exercise on the neuronal maturation in
rats with LPS-induced brain inflammation. Brain inflammation in
rats was induced by an injection of LPS into the cerebral
ventricle. Short-term memory was evaluated using a step-down
avoidance task. Cell proliferation was determined by
bromo-2′-deoxyuridine (BrdU) immunohistochemistry. Western blot
analysis for the determination of DCX and NeuN expression in the
hippocampus was performed in regards to neuronal maturation.
Materials and methods
Animals
Six-week-old male Sprague-Dawley rats weighing
200±10 g (n=60) were used for this experiment. The experimental
procedures were performed in accordance with the animal care
guidelines of the National Institutes of Health (NIH) and the
Korean Academy of Medical Sciences. The rats were housed under
controlled temperature (20±2°C) and lighting (07:00 to 19:00 h)
conditions with food and water available ad libitum. The
rats were randomly divided into 4 groups (n=15 each): the control
group, the LPS injection group, the LPS injection and treadmill
exercise group and the LPS injection and wheel exercise group. All
rats received 50 mg/kg (BrdU; Sigma Chemical Co., St. Louis, MO,
USA) intraperitoneally (i.p.) once a day 30 min prior to the onset
of treadmill and wheel exercise for the first 4 days.
Induction of brain inflammation
Brain inflammation was induced by an injection of
LPS (055:B5; Sigma Chemical Co.) into the cerebral ventricle, as
previously described (20).
Briefly, the rats were anesthetized with Zoletil 50® (10
mg/kg i.p.; Virbac Laboratories, Carros, France) and placed in a
stereotaxic frame. Through a hole drilled into the skull, a
26-gauge needle was implanted into the cerebral ventricle at the
following coordinates: 1.2 mm lateral to the midline, 0.9 mm
anterior to the coronal suture and at a depth of 3.3 mm from the
surface of the brain. LPS was dissolved in artificial cerebrospinal
fluid and 50 μg of LPS in 7 μl was infused for over 3 min. The
needle remained in place for an additional 3 min after the infusion
and was then gradually withdrawn.
Exercise protocols
Prior to the onset of exercise, the daily distance
for each rat on the wheel was calculated for 1 week, using an extra
number of rats (n=10). The exercise load of treadmill running was
adjusted to the exercise load of wheel running. Treadmill and wheel
exercise began 1 day following LPS injection.
The rats in the treadmill exercise group were forced
to run on a motorized treadmill for 30 min once a day for 6 weeks.
The exercise load consisted of running at a speed of 5 m/min for
the first 5 min, 8 m/min for the next 5 min and 10 m/min for the
last 20 min, with 0° inclination.
The rats in the wheel exercise group were
individually placed in cages equipped with a running wheel
(diameter, 20 cm; width, 9 cm). A magnet attached to a running
wheel triggered a magnetic reed switch that provided input to an
electrical counter. The number of revolutions was recorded and
wheel revolutions were counted irrespective of the direction of the
wheel. The running distance was calculated using the number of
wheel revolutions. The rats in the non-exercise groups were left in
the treadmill without running for the same period of time as the
rats in the treadmill exercise group.
Step-down avoidance task
The latency time of the step-down avoidance task was
determined in order to evaluate short-term memory, as previously
described (7). Briefly, the rats
were trained in a step-down avoidance task 38 days after beginning
the treadmill and wheel exercise. Twenty four hours after training,
the latency time (sec) in each group was measured.
The rats were placed on a 7×25-cm platform, with a
height of 2.5 cm. The platform faced a 42×25-cm grid of parallel
stainless steel bars, 0.1 cm in caliber, spaced 1 cm apart. In the
training sessions, the animals received a 0.5 mA scrambled foot
shock for 2 sec immediately upon stepping down. The time interval
that elapsed between the rats stepping down and placing all four
paws on the grid was defined as the latency time. A latency time
>300 sec was counted as 300 sec.
Tissue preparation
The rats were sacrificed immediately after
determining the latency time of the step-down avoidance task. All
rats were anesthetized using Zoletil 50 (10 mg/kg, i.p.; Virbac
Laboratories), transcardially perfused with 50 mM
phosphate-buffered saline (PBS) and fixed with a freshly-prepared
solution consisting of 4% paraformaldehyde in 100 mM phosphate
buffer (PB, pH 7.4). The brains were dissected and post-fixed in
the same fixative method overnight and transferred to a 30% sucrose
solution for cryoprotection. Coronal sections (49 μm-thick) were
made using a freezing microtome (Leica, Nussloch, Germany). On
average, 5 slice sections in the hippocampus were collected from
each rat. The sections obtained 2.5–2.7 mm posterior to the bregma
were used for BrdU immunohistochemistry.
Immunohistochemistry for BrdU
In order to detect newly generated cells in the
hippocampal dentate gyrus, BrdU-specific immunohistochemistry was
performed, as previously described (7). Briefly, the sections were first
permeabilized by incubation in 0.5% Triton X-100 in PBS for 20 min,
then pretreated in 50% formamide-2X standard saline citrate (SSC)
at 65°C for 2 h, denatured in 2N HCl at 37°C for 30 min and rinsed
twice in 100 mM sodium borate (pH 8.5). Afterwards, the sections
were incubated overnight at 4°C with a BrdU-specific mouse
monoclonal antibody (1:600; Roche Diagnostics GmbH, Mannheim,
Germany). The sections were then washed 3 times with PBS and
incubated with a biotinylated mouse secondary antibody (1:200;
Vector Laboratories, Burlingame, CA, USA) for 1 h. The sections
were then incubated for a further 1 h with an avidin-peroxidase
complex (1:100; Vector Laboratories). For visualization, the
sections were incubated in 50 mM Tris-HCl (pH 7.6) containing 0.03%
3,3′-diaminobenzidine tetrahydrochloride (DAB; Sigma Chemical Co.),
40 mg/ml nickel chloride and 0.03% hydrogen peroxide for 5 min. The
sections were then washed 3 times with PBS and mounted onto
gelatin-coated slides. The slides were air-dried overnight at room
temperature and the coverslips were mounted using
Permount®. The numbers of BrdU-positive cells in the
dentate gyrus were counted hemilaterally under a light microscope
(Olympus) and they were expressed as the numbers of cells per
mm2 in the dentate gyrus. The area of the dentate gyrus
was measured by the Image-Pro® Plus image analysis
system (Media Cybernetics Inc., Silver Spring, MD, USA).
Western blot analysis
Western blot analysis was performed as previously
described (7). The hippocampal
tissues were collected and were then immediately frozen at -70°C.
The hippocampal tissues were homogenized on ice and lysed in a
lysis buffer containing 50 mM HEPES (pH 7.5), 150 mM NaCl, 10%
glycerol, 1% Triton X-100, 1 mM PMSF, 1 mM EGTA, 1.5 mM
MgCl2·6H2O, 1 mM sodium orthovanadate and 100
mM sodium fluoride. Protein content was measured using a Bio-Rad
colorimetric protein assay kit (Bio-Rad, Hercules, CA, USA).
Protein (30 μg) was separated on SDS-polyacrylamide
gels and transferred onto a nitrocellulose membrane. The mouse
β-actin antibody (1:1,000; Santa Cruz Biotechnology Inc., Santa
Cruz, CA, USA), mouse anti-NeuN antibody (1:500; Chemicon
International) and goat anti-DCX antibody (1:500; Santa Cruz
Biotechnology Inc.) were used as the primary antibodies.
Horseradish peroxidase-conjugated anti-mouse antibodies for β-actin
and NeuN (1:2,000; Vector Laboratories) and horseradish
peroxidase-conjugated anti-goat antibody for DCX (1:5,000; Santa
Cruz Biotechnology Inc.) were used as the secondary antibodies.
The experiments were performed under normal
laboratory conditions and at room temperature, except for the
transferred membranes, which were performed at 4°C with a cold pack
and a pre-chilled buffer. Band detection was performed using the
enhanced chemiluminescence (ECL) detection kit (Santa Cruz
Biotechnology Inc.). In order to compare the relative protein
expression levels, the detected bands were calculated
densitometrically using Molecular Analyst™, version 1.4.1
(Bio-Rad).
Data analysis
Statistical analysis was performed using IBM PSS
software (version 20.0; IBM Corp., Armonk, NY, USA). For the
comparison among the groups, one-way ANOVA and Duncan’s post hoc
test were performed. All values were expressed as the means ±
standard error of the mean (SEM). A P-value <0.05 was considered
to indicate a statistically significant difference.
Results
Daily running distance
The daily running distance was 265.00±0.00 m in the
treadmill exercise group and 253.35±9.39 m in the wheel exercise
group. There was little difference in the daily running distance
between treadmill and wheel exercise.
Short-term memory
In order to evaluate the effects of forced treadmill
exercise and voluntary wheel exercise on short-term memory, a
step-down avoidance task was performed. The results of the
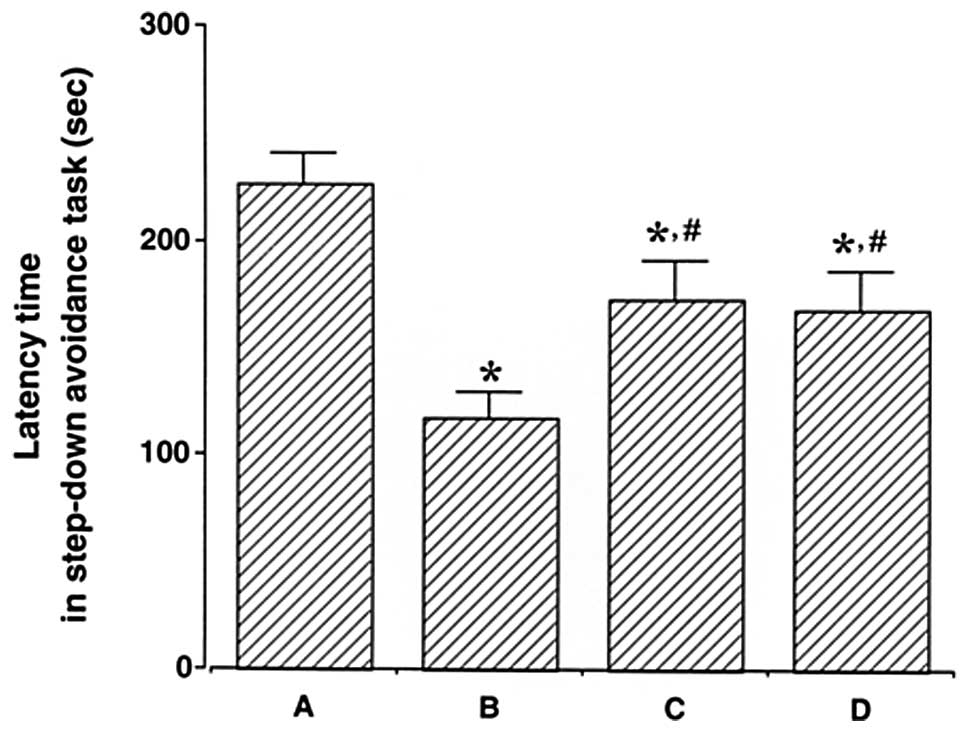
step-down avoidance task are presented in Fig. 1. The latency time was 227.25±13.70
sec in the control group, 117.75±12.15 sec in the LPS injection
group, 172.30±18.05 sec in the LPS injection and treadmill exercise
group and 167.33±18.53 sec in the LPS injection and wheel exercise
group.
The latency time in the LPS injection group was
conspicuously shorter than that in the control group (P<0.05).
On the other hand, treadmill and wheel exercise significantly
increased the latency time in the LPS-injected rats (P<0.05).
The effects of treadmill and wheel exercise on short-term memory
were similar. These results suggest that forced treadmill exercise
and voluntary wheel exercise significantly alleviated LPS-induced
short-term memory impairment.
Cell proliferation in the hippocampal
dentate gyrus
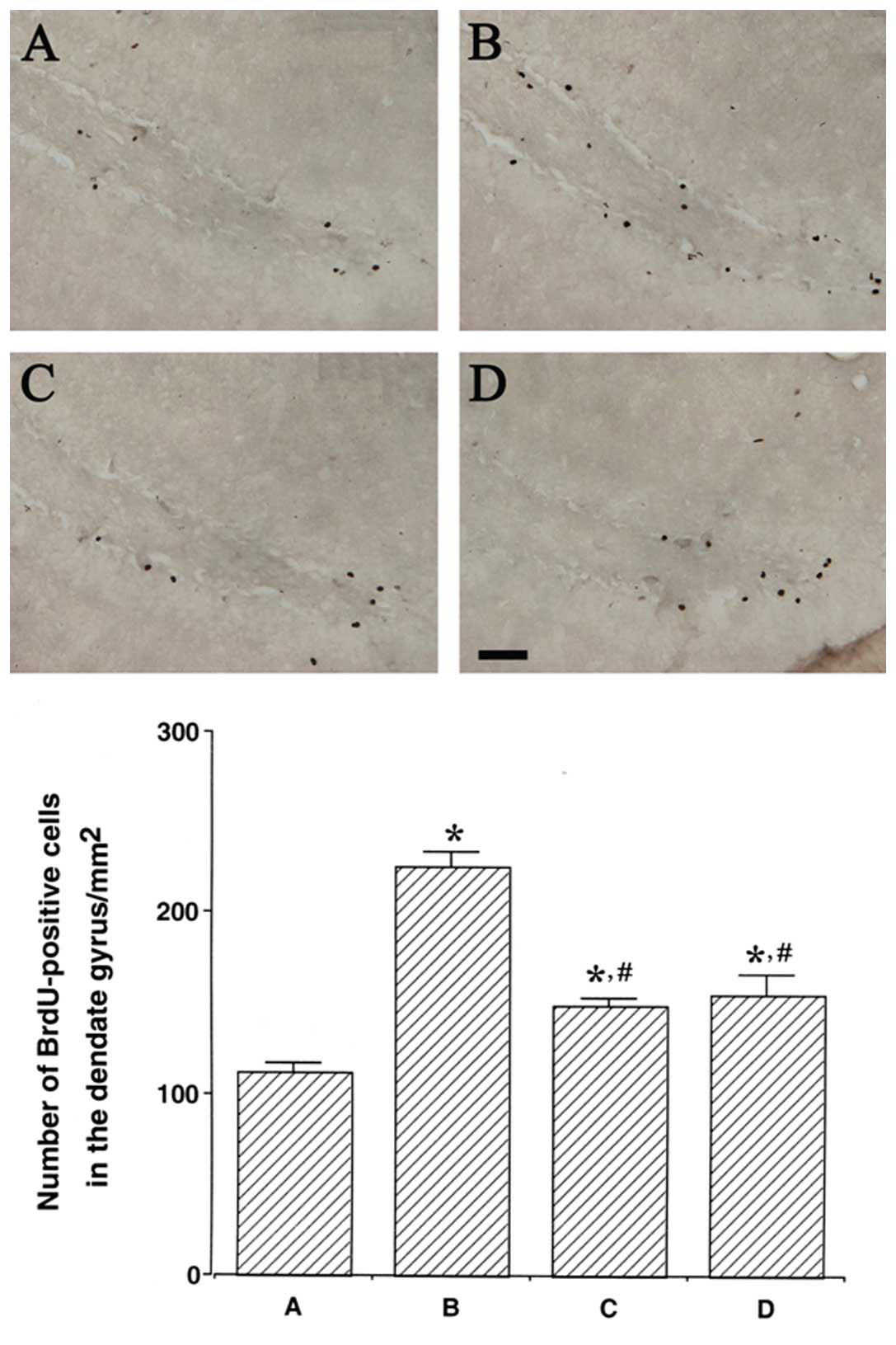
Photomicrographs of BrdU-positive cells in the
hippocampal dentate gyrus are presented in Fig. 2. The number of BrdU-positive cells
was 112.19±4.92/mm2 in the control group,
225.05±8.57/mm2 in the LPS injection group,
149.08±4.19/mm2 in the LPS injection and treadmill
exercise group and 155.43±10.26/mm2 in the LPS injection
and wheel exercise group.
The number of BrdU-positive cells in the hippocampal
dentate gyrus was remarkably increased following LPS injection
(P<0.05); however, treadmill and wheel exercise significantly
reduced the number of BrdU-positive cells in the LPS-injected rats
(P<0.05). These results demonstrated that the cell proliferation
in the hippocampal dentate gyrus was enhanced by LPS-induced brain
inflammation, while forced treadmill exercise and voluntary wheel
exercise suppressed cell proliferation in the hippocampal dentate
gyrus. The effects of treadmill and wheel exercise on cell
proliferation were similar.
Expression of DCX protein in the
hippocampus
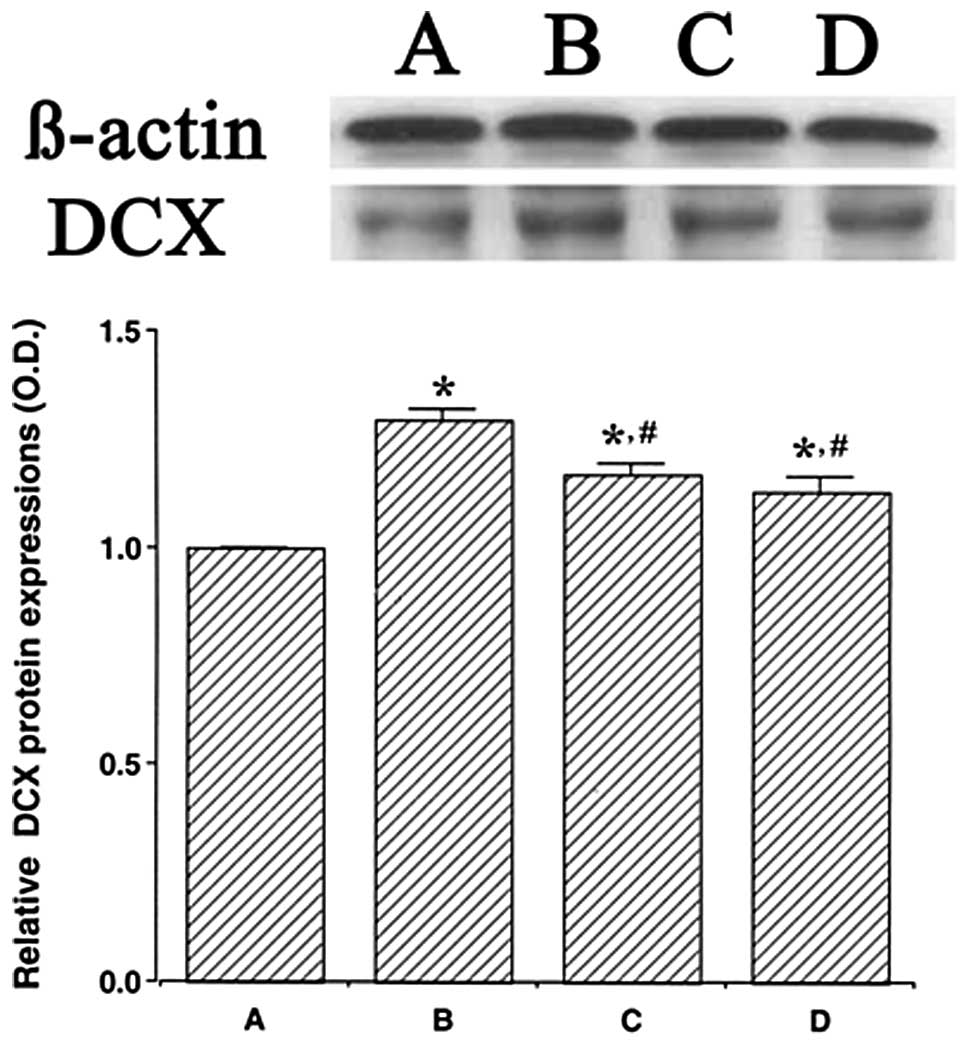
We determined the relative expression levels of DCX
in the hippocampus (Fig. 3). When
the level of DCX (45 kDa) in the control group was set at 1.00, the
level of DCX was 1.30±0.02 in the LPS injection group, 1.17±0.03 in
the LPS injection and treadmill exercise group and 1.13±0.03 in the
LPS injection and wheel exercise group.
The level of DCX protein in the LPS injection group
was significantly increased in comparison with that in the control
group (P<0.05). However, treadmill and wheel exercise
significantly reduced the expression of DCX protein in the
LPS-injected rats (P<0.05). These results suggest that
LPS-induced brain inflammation increases the generation of immature
neurons. However, forced treadmill exercise and voluntary wheel
exercise suppressed the generation of immature neurons. The effects
of treadmill exercise and wheel exercise on the DCX expression were
similar.
Expression of NeuN protein in the
hippocampus
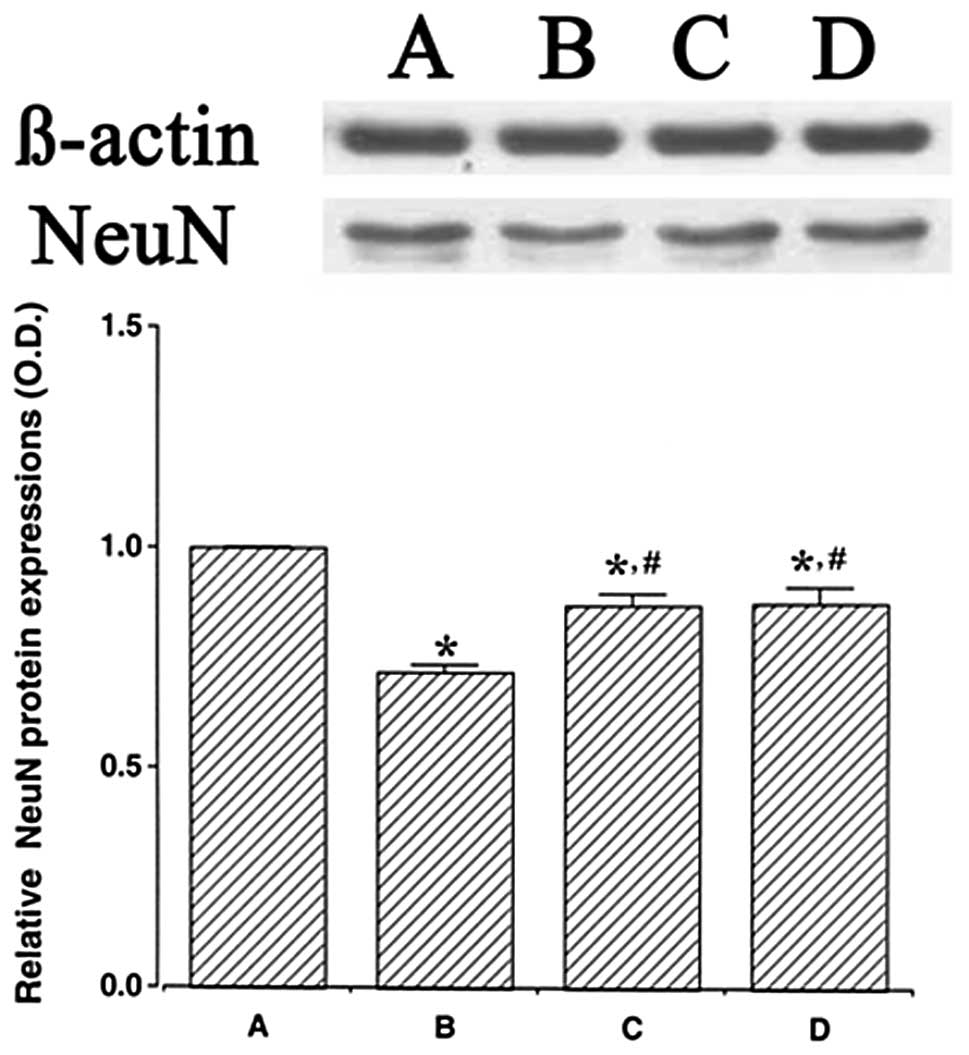
We determined the relative expression levels of NeuN
in the hippocampus (Fig. 4). When
the level of NeuN (46–48 kDa) in the control group was set at 1.00,
the level of NeuN was 0.72±0.02 in the LPS injection group,
0.87±0.02 in the LPS injection and treadmill exercise group and
0.87±0.03 in the LPS injection and wheel exercise group.
The expression of NeuN protein in the LPS injection
group was significantly decreased in comparison to the control
group (P<0.05). However, treadmill exercise and wheel exercise
significantly enhanced the expression of NeuN protein (P<0.05).
These results suggest that LPS-induced brain inflammation disturbed
the maturation of neuronal cells. However, forced treadmill
exercise and voluntary wheel exercise enhanced the generation of
mature neuronal cells. The effects of treadmill exercise and wheel
exercise on NeuN expression were similar.
Discussion
Belarbi et al(5) reported that brain inflammation
induced by LPS impaired learning ability and memory functions.
Similarly, our results revealed that LPS-induced brain inflammation
deteriorated short-term memory in rats. However, forced treadmill
exercise and voluntary wheel exercise improved short-term memory
incapacitated by brain inflammation, regardless of the type of
exercise (Fig. 1).
Exercise improves learning ability and memory
function by increasing neurogenesis in the hippocampus (21). Neurogenesis induced by exercise is
reported to be necessary in hippocampal-dependent learning, such as
spatial navigation learning or exploration in the elevated plus
maze (22,23). Neurogenesis is also influenced by
pathological conditions affecting the brain. Ekdahl et
al(1) reported that brain
inflammation impaired basal hippocampal neurogenesis by microglial
activation. As a result, they suggested that this suppression of
hippocampal neurogenesis might contribute to cognitive dysfunction
in aging, dementia, epilepsy and other brain inflammatory
conditions (1).
Of note, our results showed that brain inflammation
significantly increased cell proliferation, the first step of adult
neurogenesis (Fig. 2). Namely, our
data indicated that LPS-induced brain inflammation impaired
short-term memory in spite of the fact that brain inflammation
increased cell proliferation in the hippocampal dentate gyrus
(Figs. 1 and 2). Similar results were also found in
other studies. In brain ischemia, enhanced cell proliferation in
the hippocampal dentate gyrus was observed (8) and this enhanced cell proliferation in
the hippocampal dentate gyrus has been suggested as the
compensatory response to excessive apoptotic cell death (8,24).
The role of inflammation for adult neurogenesis, however, is much
more complex. Microglia activation, as an indicator of
inflammation, is not pro- or anti-neurogenic per se but the
net outcome is dependent on the balance between secreted molecules
with pro- and anti-inflammatory action (25). In the murine experimental
autoimmune encephalomyelitis model for multiple sclerosis, the most
common chronic inflammatory disease of the central nervous system,
proliferation regarding the hippocampal neuronal cells was
increased, however hippocampal-dependent cognitive functions were
massively impaired (26).
Pathological conditions such as acute central nervous system
insults and experimental autoimmune encephalomyelitis were
accompanied by an increase in neurogenesis as well as by the
induction of neurogenesis in nonneurogenic areas. These conditions
might be induced by the activation of microglia and resulted in the
loss of cognitive activity (27).
DCX-positive cells in the hippocampal dentate gyrus
were increased in mice exhibiting autoimmune encephalomyelitis
compared with those in the control group (28) DCX, a marker of newly generated
neurons, is frequently regarded as an immature neuronal marker. DCX
is present in the tips of neurites of non-migratory immature
neurons and expressed only during three stages of neurogenesis
(differentiation, migration and targeting), not the synaptic
integration stage in which the newly formed cells become
functionally integrated into the hippocampal network (15). In our study, the expression of DCX
protein was increased through brain inflammation, indicating that
brain inflammation increased the generation of immature neurons.
However, both forced treadmill exercise and voluntary wheel
exercise significantly suppressed the expression of DCX protein,
suggesting that the generation of immature neurons by brain
inflammation was inhibited by forced treadmill exercise and
voluntary wheel exercise (Fig.
3).
NeuN, a specific marker of neuronal cells,
recognizes nuclear proteins rather than cytoplasmic antigens or
cytoplasmic organelles and does not stain the nuclei of immature
nerve cells until they achieve a stage of development that
approaches mature function. Thus, NeuN satisfies the primary
criterion of a tissue marker regarding neuronal maturation
(19). Huehnchen et
al(28) also reported that
despite the initial increase in the number of progenitor cells, the
succeeding differentiation of BrdU-positive cells into mature
NeuN-positive neurons was delayed in autoimmune encephalomyelitis
mice when compared with mice in the control group. In light of the
mechanism whereby cognitive deficits may be explained by altered
proliferation and the differentiation of newborn hippocampal
progenitors (29,30), in this study, we investigated the
expression of NeuN protein in the hippocampus. Brain inflammation
reduced the capacity of neuronal progenitors to differentiate into
mature neurons, whereas forced treadmill exercise and voluntary
wheel exercise significantly increased the generation of mature
neurons suppressed by brain inflammation (Fig. 4). Ekdahl et al(1) reported that the LPS-induced
inflammation remarkably reduced the number of new neurons, the
BrdU-immunoreactive cells double-labeled with NeuN in the
subgranular zone of the dentate gyrus. On the other hand, physical
exercise improved memory function and spatial learning ability
through the enhancement of neurogenesis, which was ascertained by
increasing the number of BrdU- and NeuN-double positive neurons
(31).
In conclusion, the results from our study
demonstrated that brain inflammation induced by LPS weakened
short-term memory by increasing the generation of immature neurons
and contrarily suppressed the generation of mature neurons. On the
other hand, both forced treadmill exercise and voluntary wheel
exercise significantly improved short-term memory by enhancing
neuronal maturation. Moreover, forced treadmill exercise and
voluntary wheel exercise showed similar efficacy. From these
results, it can be inferred that forced treadmill exercise and
voluntary wheel exercise may improve memory function deteriorated
by brain inflammation, such as Alzheimer’s disease and Parkinson’s
disease.
Acknowledgements
This study was supported by the National Research
Foundation of Korea Grant funded by the Korean Government
(NRF-2010-327-G00127).
References
|
1
|
Ekdahl CT, Claasen JH, Bonde S, Kokaia Z
and Lindvall O: Inflammation is detrimental for neurogenesis in
adult brain. Proc Natl Acad Sci USA. 100:13632–13637. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cacci E, Claasen JH and Kokaia Z:
Microglia-derived tumor necrosis factor-alpha exaggerates death of
newborn hippocampal progenitor cells in vitro. J Neurosci Res.
80:789–797. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cacci E, Ajmone-Cat MA, Anelli T, Biagioni
S and Minghetti L: In vitro neuronal and glial differentiation from
embryonic or adult neural precursor cells are differently affected
by chronic or acute activation of microglia. Glia. 56:412–425.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Guo J, Li F, Wu Q, Gong Q, Lu Y and Shi J:
Protective effects of icariin on brain dysfunction induced by
lipopolysaccharide in rats. Phytomedicine. 17:950–955. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Belarbi K, Jopson T, Tweedie D, Arellano
C, Luo W, Greig NH and Rosi S: TNF-α protein synthesis inhibitor
restores neuronal function and reverses cognitive deficits induced
by chronic neuroinflammation. J Neuroinflammation. 9:232012.
|
|
6
|
Kim DH, Ko IG, Kim BK, et al: Treadmill
exercise inhibits traumatic brain injury-induced hippocampal
apoptosis. Physiol Behav. 101:660–665. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kim SE, Ko IG, Kim BK, et al: Treadmill
exercise prevents aging-induced failure of memory through an
increase in neurogenesis and suppression of apoptosis in rat
hippocampus. Exp Gerontol. 45:357–365. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sim YJ, Kim SS, Kim JY, Shin MS and Kim
CJ: Treadmill exercise improves short-term memory by suppressing
ischemia-induced apoptosis of neuronal cells in gerbils. Neurosci
Lett. 372:256–261. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Cao L, Jiao X, Zuzga DS, Liu Y, Fong DM,
Young D and During MJ: VEGF links hippocampal activity with
neurogenesis, learning and memory. Nat Genet. 36:827–835. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
van Praag H, Christie BR, Sejnowski TJ and
Gage FH: Running enhances neurogenesis, learning and long-term
potentiation in mice. Proc Natl Acad Sci USA. 96:13427–13431.
1999.PubMed/NCBI
|
|
11
|
Eriksson PS, Perfilieva E, Bjök-Eriksson
T, Alborn AM, Nordborg C, Peterson DA and Gage FH: Neurogenesis in
the adult human hippocampus. Nat Med. 4:1313–1317. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kempermann G, Kuhn HG and Gage FH: More
hippocampal neurons in adult mice living in an enriched
environment. Nature. 386:493–495. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kempermann G, Jessberger S, Steiner B and
Kronenberg G: Milestones of neuronal development in the adult
hippocampus. Trends Neurosci. 27:447–452. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ming GL and Song H: Adult neurogenesis in
the mammalian central nervous system. Annu Rev Neurosci.
28:223–250. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
von Bohlen and Halbach O:
Immunohistological markers for proliferative events, gliogenesis
and neurogenesis within the adult hippocampus. Cell Tissue Res.
345:1–19. 2011.PubMed/NCBI
|
|
16
|
Francis F, Koulakoff A, Boucher D, et al:
Doublecortin is a developmentally regulated, microtubule-associated
protein expressed in migrating and differentiating neurons. Neuron.
23:247–256. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Friocourt G, Koulakoff A, Chafey P,
Boucher D, Fauchereau F, Chelly J and Francis F: Doublecortin
functions at the extremities of growing neuronal processes. Cereb
Cortex. 13:620–626. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wolf HK, Buslei R, Schmidt-Kastner R,
Schmidt-Kastner PK, Pietsch T, Wiestler OD and Blümcke I: NeuN: a
useful neuronal marker for diagnostic histopathology. J Histochem
Cytochem. 44:1167–1171. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sarnat HB, Nochlin D and Born DE: Neuronal
nuclear antigen (NeuN): a marker of neuronal maturation in early
human fetal nervous system. Brain Dev. 20:88–94. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Tyagi E, Agrawal R, Nath C and Shukla R:
Influence of LPS-induced neuroinflammation on acetylcholinesterase
activity in rat brain. J Neuroimmunol. 205:51–56. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
van Praag H, Kempermann G and Gage FH:
Running increases cell proliferation and neurogenesis in the adult
mouse dentate gyrus. Nat Neurosci. 2:266–270. 1999.
|
|
22
|
O’Callaghan RM, Ohle R and Kelly AM: The
effects of forced exercise on hippocampal plasticity in the rat: A
comparison of LTP, spatial- and non-spatial learning. Behav Brain
Res. 176:362–366. 2007.PubMed/NCBI
|
|
23
|
Shors TJ, Townsend DA, Zhao M,
Kozorovitskiy Y and Gould E: Neurogenesis may relate to some but
not all types of hippocampal-dependent learning. Hippocampus.
12:578–584. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu J, Solway K, Messing RO and Sharp FR:
Increased neurogenesis in the dentate gyrus after transient global
ischemia in gerbils. J Neurosci. 18:7768–7778. 1998.PubMed/NCBI
|
|
25
|
Ekdahl CT, Kokaia Z and Lindvall O: Brain
inflammation and adult neurogenesis: the dual role of microglia.
Neuroscience. 158:1021–1029. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Aharoni R, Arnon R and Eilam R:
Neurogenesis and neuroprotection induced by peripheral
immunomodulatory treatment of experimental autoimmune
encephalomyelitis. J Neurosci. 25:8217–8228. 2005. View Article : Google Scholar
|
|
27
|
Ziv Y, Ron N, Butovsky O, et al: Immune
cells contribute to the maintenance of neurogenesis and spatial
learning abilities in adulthood. Nat Neurosci. 9:268–275. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Huehnchen P, Prozorovski T, Klaissle P, et
al: Modulation of adult hippocampal neurogenesis during
myelin-directed autoimmune neuroinflammation. Glia. 59:132–142.
2011. View Article : Google Scholar
|
|
29
|
Deng W, Aimone JB and Gage FH: New neurons
and new memories: how does adult hippocampal neurogenesis affect
learning and memory? Nat Rev Neurosci. 11:339–350. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kempermann G, Gast D and Gage FH:
Neuroplasticity in old age: sustained fivefold induction of
hippocampal neurogenesis by long-term environmental enrichment. Ann
Neurol. 52:135–143. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wong-Goodrich SJ, Pfau ML, Flores CT,
Fraser JA, Williams CL and Jones LW: Voluntary running prevents
progressive memory decline and increases adult hippocampal
neurogenesis and growth factor expression after whole-brain
irradiation. Cancer Res. 70:9329–9338. 2010. View Article : Google Scholar
|