Introduction
Breast cancer is the most commonly diagnosed cancer
and the leading cause of cancer mortality among females, accounting
for 23% of total cancer cases and 14% of cancer mortalities
(1). In developing countries,
including China, breast cancer remains a significant public health
issue due to its prevalence (2).
In recent years, medical advances have increased the availablity
and efficacy of breast cancer treatments, including improved
surgical methods for lumpectomy and mastectomy and radiation,
hormone and chemotherapies. However, the majority of drugs
currently used as chemotherapeutic agents exhibit low efficacy and
are associated with the development of drug resistance (3,4).
Therefore, new drugs with a higher therapeutic index are urgently
required to effectively treat this malignancy.
Over the last two decades, an increasing number of
bioactive compounds have been identified in traditional Chinese
medical herbs (5). Specific
compounds have been reported to kill tumor cells by generation of
reactive oxygen species (ROS). ROS are known to affect
mitochondrial membrane potential (ΔΨm) and trigger a series of
mitochondria-associated events (6,7). The
generation of ROS may contribute to mitochondrial damage, reduction
of ΔΨm, release of cytochrome c and Smac and subsequent caspase
activation and apoptosis (8,9).
Apoptosis is a major control mechanism of cell death when DNA
damage is not repaired. Apoptosis is a gene-directed programmed
cell death characterized by cell shrinkage, blebbing of the plasma
membrane, chromosomal DNA fragmentation and a number of other
morphological alterations (10).
There are two major pathways of apoptosis, the cell death receptor-
(extrinsic) and mitochondria-mediated apoptotic (intrinsic)
pathways (11). Mitochondria are
central to the intrinsic apoptotic pathway. Disruption of ΔΨm, loss
of mitochondrial potential and induction of cytochrome c release
from the mitochondria into the cytosol are all associated with
regulation of apoptosis and are recognized as key steps in the
mitochondrion-dependent apoptotic pathway (12-14).
Deltonin, diosgenin-3-O-β-D-glucopyranosyl
(1→4)-[α-L-rhamnopyranosyl (1→2)]-β-D-glucopyranoside, is a
steroidal saponin. The antiproliferative and apoptosis-inducing
effects of steroidal saponins, including compounds isolated from
plants and synthesized analogs, have been demonstrated in
vitro using a number of cancer cell lines (15-18).
We have previously demonstrated that deltonin has cytotoxic effects
against colon cancer cell lines and oral administration of deltonin
was found to significantly inhibit tumor growth and prolonged
survival of tumor bearing mice (19,20).
However, the effects of deltonin in breast cancer remain unknown.
To elucidate the anticancer activity and mechanisms by which
deltonin functions, the effects of deltonin on cell proliferation
and apoptosis in the MDA-MB-231 human breast carcinoma cell line
were evaluated. Deltonin induced apoptosis through loss of ΔΨm,
accumulation of intracellular ROS, caspase activation, regulation
of Bcl-2 family members and modulation of extracellular
signal-regulated kinase (ERK)/AKT signaling pathways.
Materials and methods
Reagents
Deltonin was obtained as previously described and
its purity determined by high performance liquid chromatography
(>98%) (19). The compound was
prepared as a stock solution in DMSO [final concentration <0.05%
(v/v)] and diluted in the relevant culture medium. ROS assay and
JC-1 ΔΨm detection kits were obtained from Beyotime Institute of
Biotechnology (Jiangsu, China).
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium (MTT), DMSO,
N-acetylcysteine (NAC) and primary antibody against GAPDH were
purchased from Sigma-Aldrich (St. Louis, MO, USA). Primary
antibodies against caspase-3 and -8, ERK1/2, phospho-ERK1/2
(Thr202/204), AKT and phospho-AKT (Ser473) were purchased from Cell
Signaling Technology (Beverly, MA, USA). Primary antibodies against
Bcl-2 (C-2), Bax (P19) and secondary antibodies were purchased from
Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA). All other
chemicals and reagents were of the highest purity grade
commercially available.
Cell lines and cell culture
Human breast carcinoma cell line, MDA-MB-231, was
obtained from the American Type Culture Collection (Manassas, VA,
USA) and maintained in RPMI-1640 medium containing 10% fetal bovine
serum (Gibco-BRL, Auckland, New Zealand), 100 U/ml penicillin and
100 μg/ml streptomycin in a humid chamber at 37°C under 5%
CO2(21).
Cytotoxicity assay
The inhibitory effect of deltonin on cell viability
was measured by MTT assay (22).
Cells were seeded in 96-well plates (Costar Corning, Rochester, NY,
USA) at a density of 3×103 cells/well. Deltonin
underwent serial dilution (0.5, 1.0, 2.0, 4.0, 8.0 μM) and DMSO
(<0.05%) was used as a control. Absorbance at 570 nm was
measured with Spectra Max M5 (Molecular Devices, LLC, Sunnyvale,
CA, USA) following drug treatment (12, 24, 36, 48 h). All
experiments were performed in triplicate. Dose- and time-dependent
curves of deltonin-treated MDA-MB-231 cells were generated as the
percentage cell growth inhibition, using the following formula: %
inhibition = 1 - A570treated cells/A570control
cells × 100. The 50% inhibiting concentration was calculated
using SPSS software v13.0.
Flow cytometry assay for apoptosis
The use of Annexin V-fluorescein isothiocyanate
(FITC) and propidium iodide (PI) for flow cytometry provides a
rapid and convenient assay for apoptosis as described previously
(23). Cells were exposed to
desired concentrations of deltonin for 24 h and the apoptotic index
was assessed using an Annexin V-FITC apoptosis kit (KeyGEN,
Nanjing, China) according to the manufacturer's instructions. For
each analysis, 30,000 events were acquired on a forward and side
scatter gate. Annexin V-positive PI-negative cells represented
early apoptotic populations and Annexin V-positive PI-positive
cells represented late apoptotic or secondary necrotic populations.
Data acquisition and analysis were performed by
fluorescence-activated cell sorting using list mode software
(Becton-Dickinson, Franklin Lakes, NJ, USA).
Measurement of ΔΨm
Alterations in ΔΨm were analyzed by flow cytometry
using a ΔΨm-sensitive molecular probe dye, JC-1, as described
previously (24). The JC-1 dye
bearing a delocalized positive charge enters the mitochondrial
matrix due to the negative charge established by the intact ΔΨm. In
healthy cells, JC-1 dye stains the mitochondria red due to
formation of JC-1 aggregates. In apoptotic cells, JC-1 dye
accumulates in the cytoplasm in monomeric form (green fluorescence)
due to collapse of ΔΨm. Cells were treated with 3.0 μM deltonin for
0, 4, 8, 12, 16 and 24 h. Following this, cells were harvested,
washed once in cold PBS and incubated with JC-1 dye at 37°C for 20
min. Stained cells were washed and re-suspended in 0.5 ml assay
buffer and the fluorescence was measured using flow cytometry. The
emission wavelengths of JC-1 monomers and JC-1 aggregates were 530
and 590 nm, respectively.
Measurement of intracellular ROS
Intracellular ROS generation was monitored using an
ROS assay kit and flow cytometry using fluorescence produced by
2′,7′-dichlorofluorescein following oxidation from
2′,7′-dichlorfluorescein-diacetate (DCFH-DA; Molecular Probes,
Grand Island, NY, USA) (25). In
brief, following treatment with 3.0 μM deltonin for 0, 4, 8, 12, 16
and 24 h, cells were harvested and then incubated with 10 μM
DCFH-DA in a culture medium at 37°C for 30 min. Cells were washed
and re-suspended in PBS. ROS generation was measured by flow
cytometry.
Inhibitor treatment
To confirm the role of intracellular ROS in
deltonin-induced apoptosis, a common quencher of ROS, NAC, was used
to inhibit intracellular alteration of redox states (26). NAC was dissolved in PBS and
adjusted to pH 7.4 to produce a 0.1 M stock solution. Cells were
pre-incubated with 6 mM NAC for 2 h and then treated with 3 μM
deltonin. After 8 h, alterations in ROS production were determined
by flow cytometry. After 24 h, percentage of apoptotic cells and
alterations in ΔΨm were analyzed by flow cytometry.
Protein extraction and western blot
analysis
Western blot analysis was performed as described
previously (27). Cells
(1×106/well) were grown in 6-well microplates overnight
and treated with various concentrations (0, 1, 3, 5 μM) of deltonin
for 24 h. Following this, cells were washed with ice-cold PBS and
total cell lysates were prepared by RIPA buffer. Lysates was
centrifuged at 12,000 g for 15 min at 4°C. The supernatant was
collected and total protein concentrations were determined using
the BCA assay, dissolved in 5X SDS sample buffer and denatured.
Proteins were separated on 10-15% SDS-PAGE and transferred to
polyvinylidene fluoride membrane (Bio-Rad, Hercules, CA, USA).
Membranes were incubated overnight at 4°C with the respective
primary antibodies [caspase-3 and -8, poly (ADP ribose) polymerase
(PARP), Bcl-2, Bax, ERK1/2, phospo-ERK1/2, AKT and phospo-AKT] and
horseradish peroxidase-conjugated secondary antibodies at 37°C for
1 h. Reactive bands were identified using an enhanced
chemiluminescent substrate to horseradish peroxidase (Amersham
Pharmacia Biotech, Amersham, UK). Expression of GAPDH was used as a
control.
Statistical analysis
Data are presented as mean ± SE and were analyzed
for statistical significance using analysis of variance, followed
by Scheffe's test for multiple comparisons. P<0.05 was
considered to indicate a statistically significant difference.
Results
Deltonin decreases cell viability and
induces apoptosis
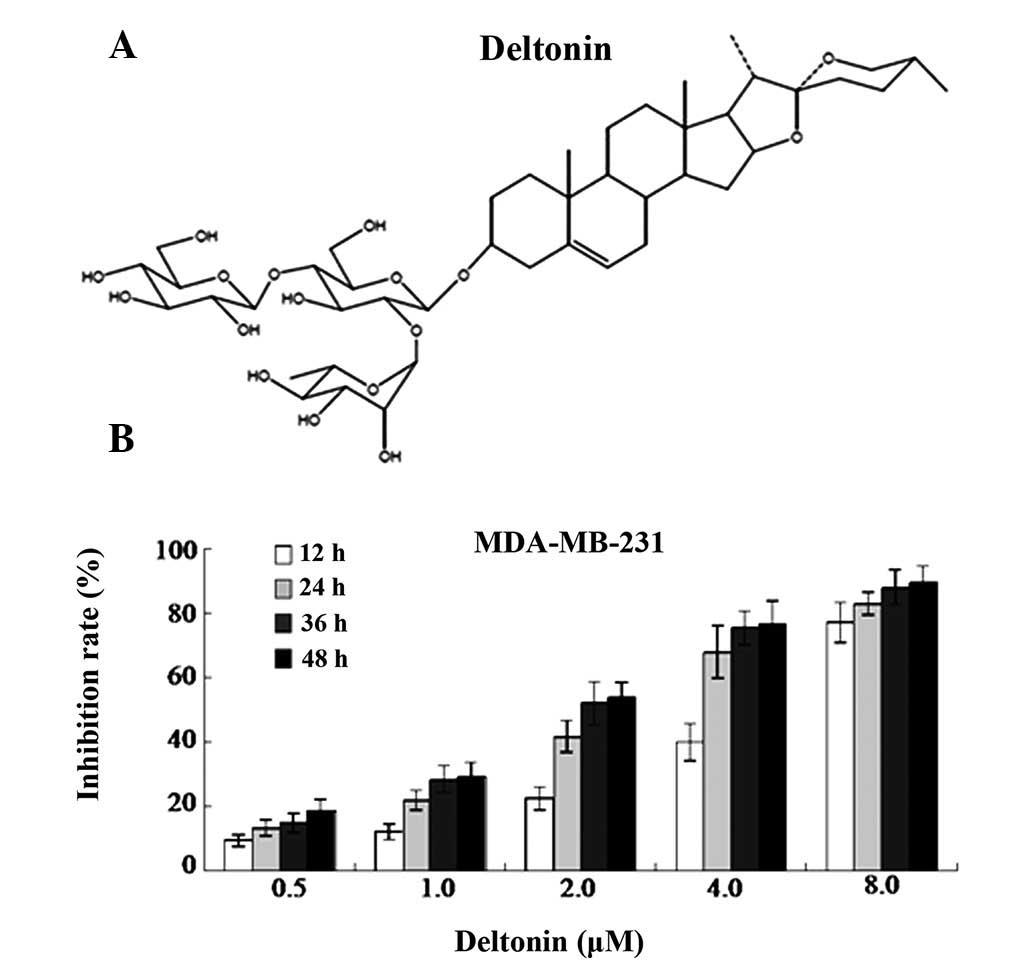
The chemical structure of deltonin is presented in
Fig. 1A. The cytotoxic effects of
deltonin were investigated by MTT assay. As demonstrated in
Fig. 1B, the maximum inhibition
ratio obtained with 8 μM deltonin treatment was 89.40%. Deltonin
inhibited the proliferation of MDA-MB-231 cells in a time- and
concentration-dependent manner.
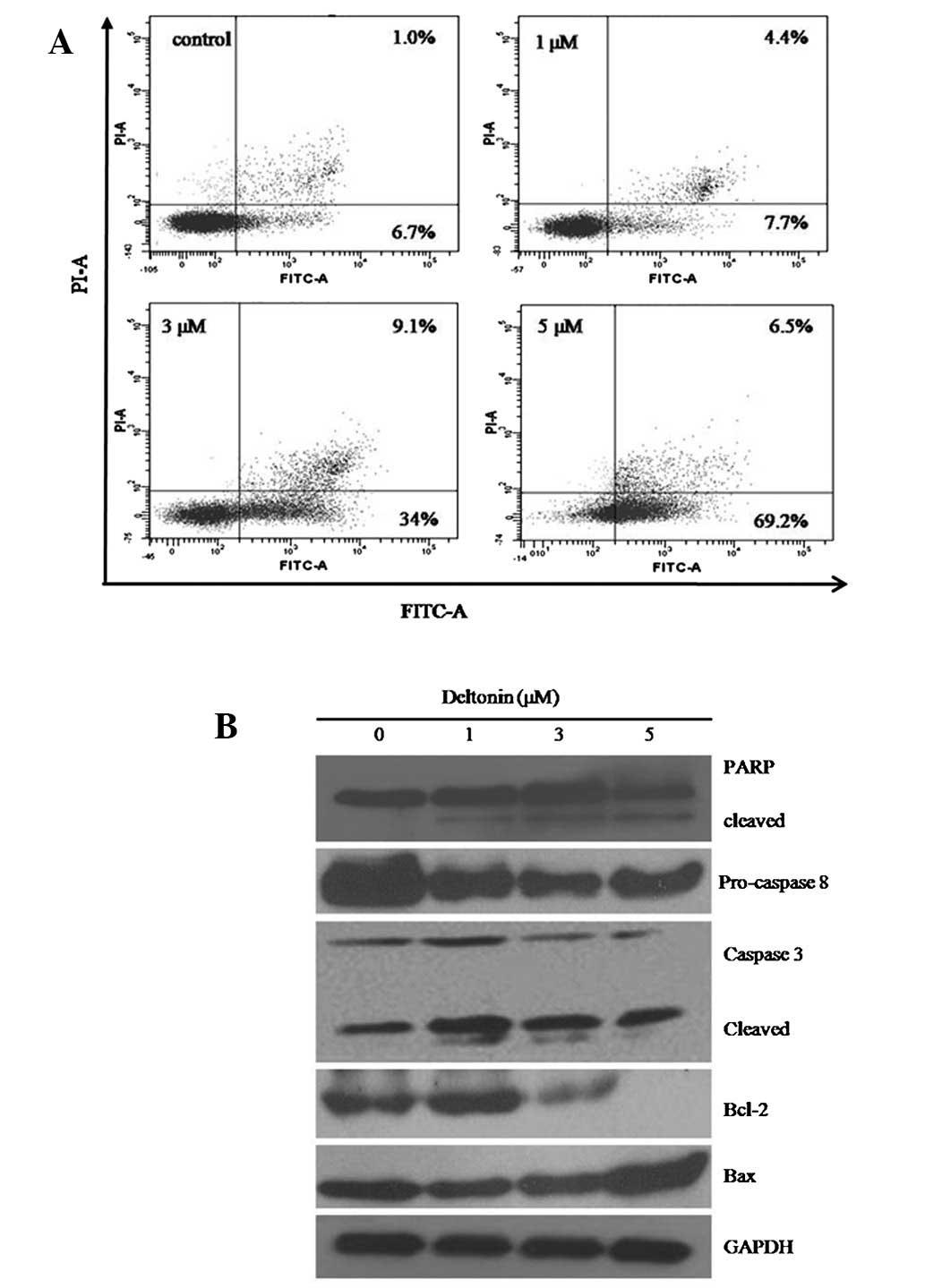
Flow cytometry analysis with an Annexin V-FITC
apoptosis kit was also performed to identify the effects of
deltonin. As shown in Fig. 2A,
deltonin treatment induced apoptosis in MDA-MB-231 cells in a
concentration-dependent manner. Compared with untreated cells,
cells treated with 5 μM deltonin resulted in up to 75.7% apoptosis
incidence, indicating that deltonin causes death in MDA-MB-231
cells by the induction of apoptosis.
Effect of deltonin on the expression of
apoptosis-related proteins
Apoptosis is executed by the coordinated actions of
caspase family members and is tightly regulated by Bcl-2 protein
family members. Therefore, protein expression levels of these
molecules were analyzed by western blot analysis. As demonstrated
in Fig. 2B, treatment of
MDA-MB-231 cells with deltonin led to activation of caspase-3 and
-8. It is well known that activation of caspase-3 during apoptosis
causes the cleavage of PARP, a major apoptotic enzyme. Deltonin
increased levels of cleaved PARP in a dose-dependent manner,
consistent with deltonin-induced apoptosis in MDA-MB-231 cells.
Following this, the protein expression levels of Bcl-2 family
members, Bcl-2 and Bax were determined. Fig. 2B shows a decrease in Bcl-2 and
increase in Bax levels.
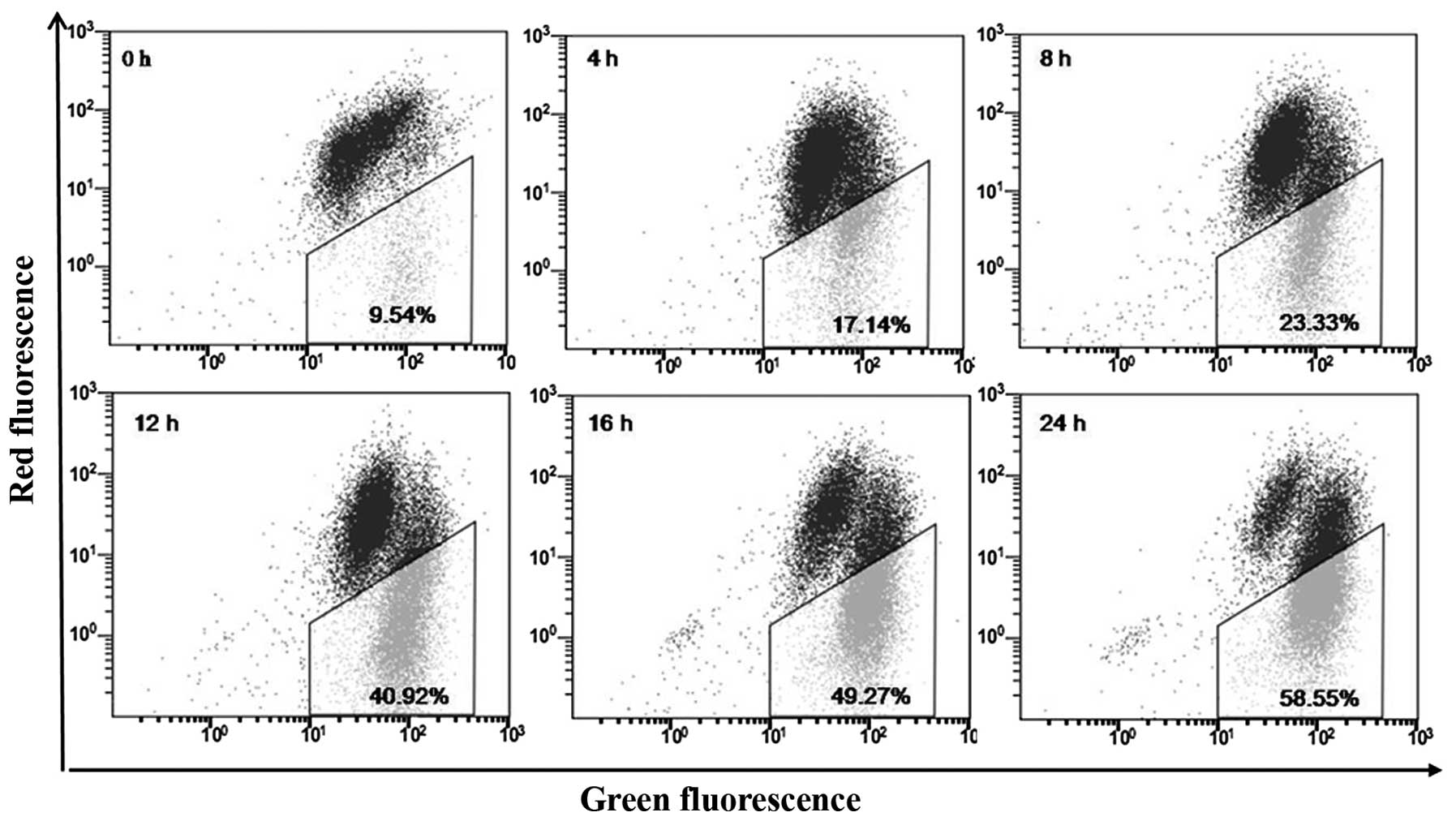
Effect of deltonin on ΔΨm change
Change of ΔΨm using JC-1 was performed to
investigate whether mitochondria are involved in deltonin-induced
apoptosis. Representative flow cytometric results are presented in
Fig. 3. Deltonin treatment led to
a time-dependent increase in the number of green
fluorescence-positive cells from 9.5 in untreated cells to 17.1,
23.3, 40.9, 49.3 and 58.6%.
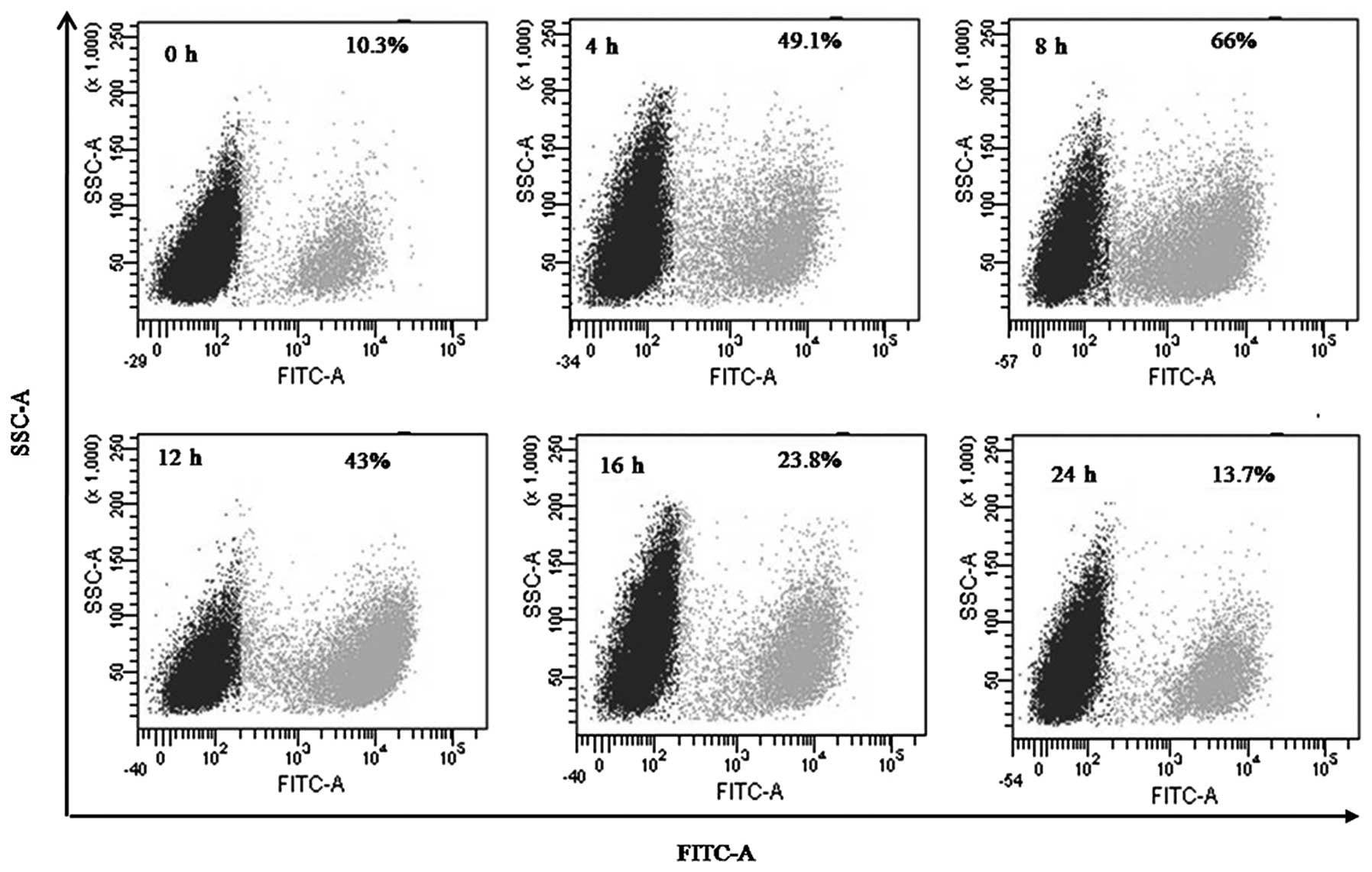
Effect of deltonin on intracellular
ROS
ΔΨm loss is an early event in apoptosis induction,
which is often accompanied by ROS production. Intracellular ROS was
examined using DCFH-DA to analyze the role of ROS in
deltonin-induced apoptosis. As demonstrated in Fig. 4, treatment of MDA-MB-231 cells with
3 μM deltonin for 0, 4, 8, 12, 16 and 24 h resulted in ROS increase
from 10.3 to 66% from 0 to 8 h, however, ROS levels decreased to
13.7% at 24 h. These results revealed that the ROS burst is
generated in a time-dependent manner.
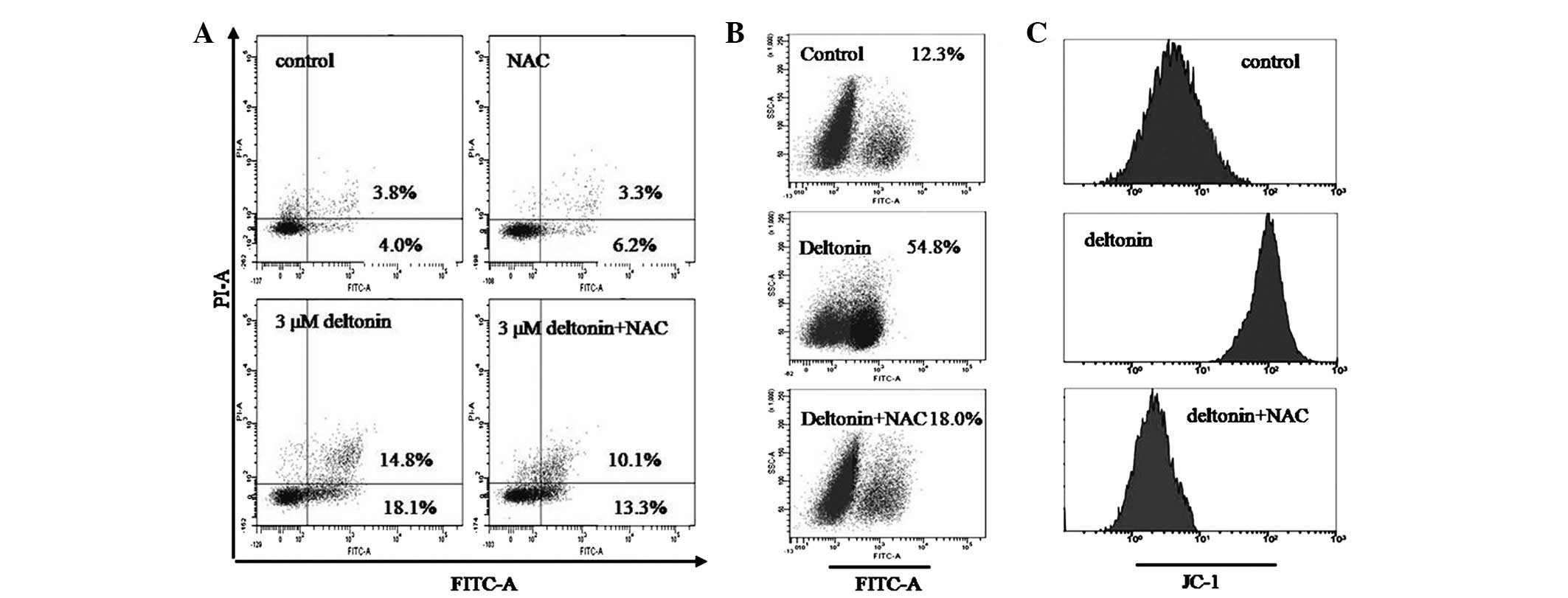
NAC inhibits deltonin-induced
apoptosis
To determine the association between intracellular
ROS and deltonin-induced apoptosis, MDA-MB-231 cells were
pretreated with 6 mM NAC for 2 h and then incubated with 3 μM
deltonin. As revealed in Fig. 5A,
pretreatment with NAC reduced the number of apoptotic cells,
whereby the percentage of apoptotic cells decreased from 32.9 to
23.4%.
NAC inhibits deltonin-induced
intracellular ROS accumulation
NAC markedly inhibited deltonin-induced accumulation
of ROS, the intensity of DCFH-DA fluorescence decreased from 54.8
to 18.0% (Fig. 5B).
NAC inhibits deltonin-induced
depolarization of ΔΨm
NAC is known to exert protective effects on cells,
therefore, the ability of NAC to prevent depolarization of ΔΨm in
deltonin-treated cells was invetigated. Fig. 5C reveals flow cytometry analysis of
ΔΨm in the presence of absence of NAC (6 mM) pretreament. The
results demonstrate that deltonin-induced apoptosis in MDA-MB-231
cells may be mediated by the mitochondrial pathway and, at least in
part, by accumulation of ROS.
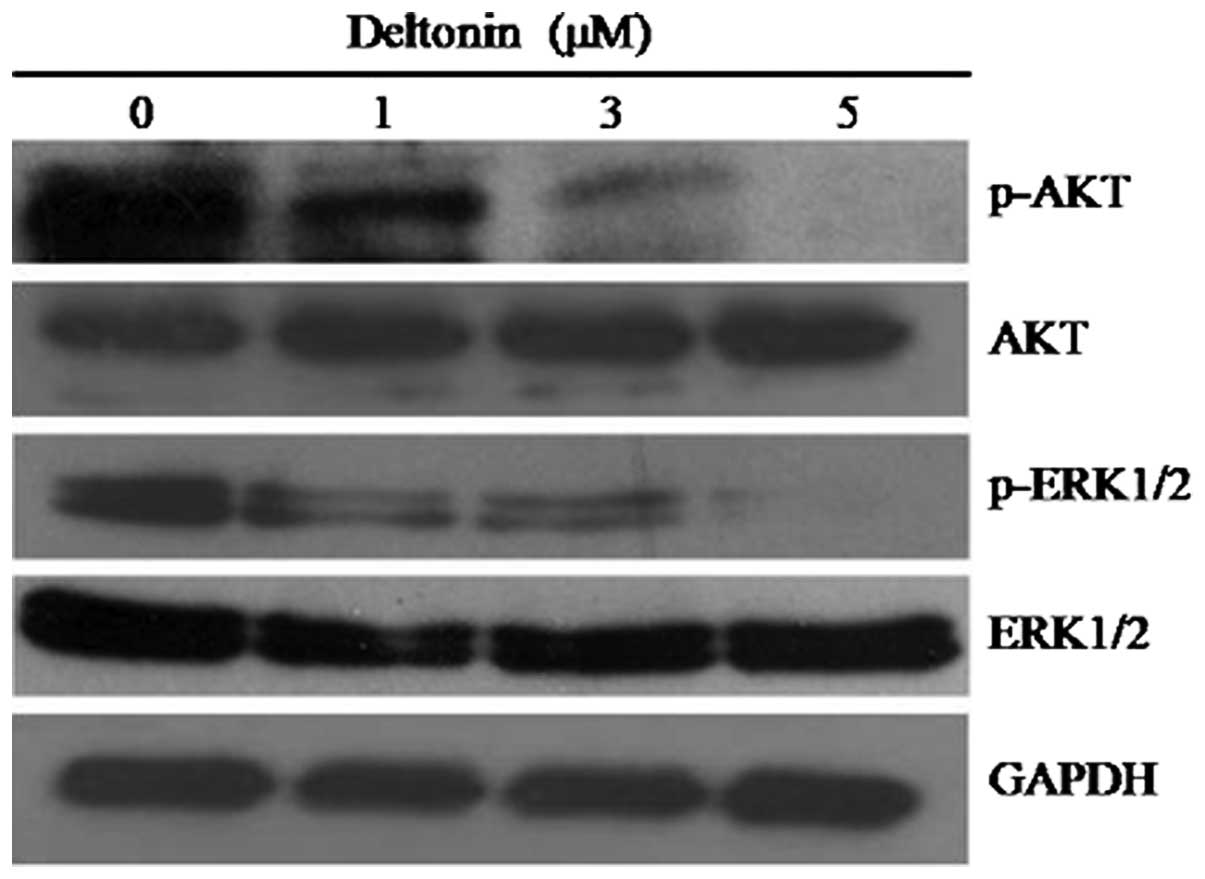
Effect of deltonin on the activation of
ERK1/2 and AKT
ERK and AKT are two notable pathways associated with
tumor development. Inhibition of the ERK pathway or AKT activation
has been identified to induce apoptosis in tumor cells (28-30).
To investigate the involvement of ERK/AKT signal pathways in
deltonin-induced apoptosis of MDA-MB-231 cells, levels of
phosphorylated and unphosphorylated AKT and ERK1/2 were determined
by western blot analysis. As shown in Fig. 6, treatment of deltonin decreased
levels of phospo-ERK1/2 and -AKT in a dose-dependent manner
compared with GAPDH (loading control). Results indicate that
interference with ERK and AKT signal pathways may contribute to the
anticancer activity of deltonin.
Discussion
Dioscorea zingiberensis Wright, a widely
distributed medicinal plant, has been used extensively for a number
of years in China. Deltonin is a major component of steroidal
saponins from Dioscorea zingiberensis Wright and it has been
reported to exhibit anticancer properties which may prove suitable
for cancer therapy. In the present study, the effect of deltonin on
breast cancer cells was evaluated and deltonin was found to
significantly inhibit the growth of human breast cancer MDA-MB-231
cells in a time- and dose-dependent manner.
Apoptosis is a form of programmed cell death which
occurs in response to a variety of stimuli and is characterized by
a series of morphological and biochemical changes. Apoptosis is
divided into two signaling pathways, death receptor-mediated
(extrinsic) and mitochondrial (intrinsic) (31). Caspases are central to the majority
of apoptotic responses. Activation of caspase-3 is considered to
initiate apoptosis and requires activation of initiator caspases,
including caspase-8 or -9, in response to proapoptotic signals
(32,33). In the apoptotic assay, results of
flow cytometry analysis confirmed that the percentage of apoptotic
cells increased with deltonin concentration. In addition, the
induction of apoptosis was confirmed by western blot analysis.
Treatment of MDA-MB-231 cells with deltonin led to the degradation
of caspase-3 and -8 and cleavage of PARP in a dose-responsive
manner. Bcl-2 family members are recognized as important regulators
of apoptosis. In the current study, the expression levels of key
Bcl-2 family proteins were determined. Western blot analysis
demonstrated that deltonin downregulated Bcl-2 levels and increased
levels of Bax. Bcl-2 is an important regulator of apoptosis by
control of mitochondrial function. Bcl-2 affects the release of
cytochrome c from the mitochondria and activates a number of
caspases (34,35). Therefore, additional analysis
focused on the mitochondrial pathway was performed.
Mitochondrial changes, including permeability
transition pore opening and the collapse of ΔΨm, induces apoptosis
by activation of caspases (36).
In the present study, the treatment of MDA-MB-231 cells with
deltonin resulted in the loss of ΔΨm in a time-dependent manner.
The results indicate that deltonin induced apoptosis of MDA-MB-231
cells, at least in part, through the mitochondrial damage-mediated
caspase pathway.
The role of ROS in mediating apoptosis in various
cancer cells is well established (37). ROS generation causes functional
disorder of mitochondria and leads to cell apoptosis (38,39).
Results indicate that deltonin induced generation of ROS and
collapse of ΔΨm. Intracellular ROS levels were observed to
significantly increase in a time-dependent manner and peaked at 8 h
of deltonin treatment, indicating that deltonin may result in
accumulation of intracellular ROS. To determine whether increased
production of ROS was critical for deltonin-induced apoptosis,
cells were pretreated with the ROS scavenger, NAC. Results revealed
that NAC had a suppressive effect on deltonin-induced apoptosis,
intracellular ROS generation and depolarization of ΔΨm, indicating
that ROS is associated with activation of the mitochondrial
pathway. In general, results demonstrate that deltonin induces
apoptosis by a mechanism involving increased intracellular ROS
levels in MDA-MB-231 cells.
AKT signaling is crucial for initiation and
progression of breast cancer (40)
and also regulates several downstream targets responsible for cell
survival and proliferation (41-44).
In the current study, deltonin downregulated expression of
phospo-AKT, indicating that deltonin inhibits AKT-mediated survival
signaling in breast cancer cells. In addition, the ERK signaling
pathway is an important pathway associated with mediating cell
growth, survival and death (45,46).
In the present study, deltonin treatment altered the expression of
phospo-ERK1/2 in MDA-MB-231 cells, demonstrating that
deltonin-induced apoptosis of MDA-MB-231 cells is associated with
the inhibition of phosphorylation of AKT and ERK1/2.
In conclusion, our results demonstrate that the
cytotoxic effect of deltonin in MDA-MB-231 cells is mediated by the
induction of apoptosis. Deltonin-induced apoptosis involves the
regulation of Bcl-2 family members and caspase-dependent
mitochondrial dysfunction and also appears to be markedly
associated with ROS production and regulation of ERK and AKT
signaling pathways. Results indicate that deltonin may be an
effective therapeutic agent for the treatment of breast cancer.
Acknowledgements
The present study was supported by grants from the
the National Major Scientific and Technological Special Project
(no. 2009ZX09102-045), National 12th 5-Year Plan Project of China
(no. 2011BAJ07B04) and the National Natural Science Foundation of
China (no. 20972105).
References
|
1
|
Jemal A, Bray F, Center MM, et al: Global
cancer statistics. CA Cancer J Clin. 61:69–90. 2011. View Article : Google Scholar
|
|
2
|
Lambert R, Sauvaget C and Sankaranarayanan
R: Mass screening for colorectal cancer is not justified in most
developing countries. Int J Cancer. 125:253–256. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Anderson BO, Shyyan R, Eniu A, et al:
Breast cancer in limited-resource countries: An overview of the
Breast Health Global Initiative 2005 Guidelines. Breast J. 12(Suppl
1): S3–S15. 2006.PubMed/NCBI
|
|
4
|
Gottesman MM: Mechanisms of cancer drug
resistance. Annu Rev Med. 53:615–627. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Efferth T, Li PC, Konkimalla VS and Kaina
B: From traditional Chinese medicine to rational cancer therapy.
Trends Mol Med. 13:353–361. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Chatterjee S, Kundu S and Bhattacharyya A:
Mechanism of cadmium induced apoptosis in the immunocyte. Toxicol
Lett. 177:83–89. 2008. View Article : Google Scholar
|
|
7
|
Pathak N and Khandelwal S: Role of
oxidative stress and apoptosis in cadmium induced thymic atrophy
and splenomegaly in mice. Toxicol Lett. 169:95–108. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gupta S, Yel L, Kim D, et al: Arsenic
trioxide induces apoptosis in peripheral blood T lymphocyte subsets
by inducing oxidative stress: a role of Bcl-2. Mol Cancer Ther.
2:711–719. 2003.PubMed/NCBI
|
|
9
|
Fan TJ, Han LH, Cong RS, et al: Caspase
family proteases and apoptosis. Acta Biochim Biophys Sin
(Shanghai). 37:719–727. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Reed JC: Mechanisms of apoptosis. Am J
Pathol. 157:1415–1430. 2000. View Article : Google Scholar
|
|
11
|
Ghobrial IM, Witzig TE and Adjei AA:
Targeting apoptosis pathways in cancer therapy. CA Cancer J Clin.
55:178–194. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Desagher S and Martinou JC: Mitochondria
as the central control point of apoptosis. Trends Cell Biol.
10:369–377. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Li H, Zhu H, Xu CJ and Yuan J: Cleavage of
BID by caspase 8 mediates the mitochondrial damage in the Fas
pathway of apoptosis. Cell. 94:491–501. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Green D and Kroemer G: The central
executioners of apoptosis: caspases or mitochondria? Trends Cell
Biol. 8:267–271. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Moalic S, Liagre B, Corbière C, et al: A
plant steroid, diosgenin, induces apoptosis, cell cycle arrest and
COX activity in osteosarcoma cells. FEBS Lett. 506:225–230. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kaskiw MJ, Tassotto ML, Th'ng J and Jiang
ZH: Synthesis and cytotoxic activity of diosgenyl saponin
analogues. Bioorgan Med Chem. 16:3209–3217. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kaskiw MJ, Tassotto ML, Mok M, et al:
Structural analogues of diosgenyl saponins: synthesis and
anticancer activity. Bioorgan Med Chem. 17:7670–7679. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Liu MJ, Yue PY, Wang Z and Wong RN: Methyl
protodioscin induces G2/M arrest and apoptosis in K562 cells with
the hyperpolarization of mitochondria. Cancer Lett. 224:229–241.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tong QY, Qing Y, Shu D, et al: Deltonin, a
steroidal saponin, inhibits colon cancer cell growth in vitro and
tumor growth in vivo via induction of apoptosis and
antiangiogenesis. Cell Physiol Biochem. 27:233–242. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shu D, Qing Y, Tong QY, et al: Deltonin
isolated from Dioscorea zingiberensis inhibits cancer cell
growth through inducing mitochondrial apoptosis and suppressing AKT
and mitogen activated protein kinase signals. Biol Pharml Bull.
34:1231–1239. 2011.
|
|
21
|
Moon DO, Choi YH, Moon SK, et al: Gossypol
decreases tumor necrosis factor-α-induced intercellular adhesion
molecule-1 expression via suppression of NF-κB activity. Food Chem
Toxicol. 49:999–1005. 2011.PubMed/NCBI
|
|
22
|
Zhang Z, Li M, Wang H, et al: Antisense
therapy targeting MDM2 oncogene in prostate cancer: effects on
proliferation, apoptosis, multiple gene expression and
chemotherapy. Proc Natl Acad Sci USA. 100:11636–11641. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Edwards LA, Thiessen B, Dragowska WH, et
al: Inhibition of ILK in PTEN-mutant human glioblastomas inhibits
PKB/Akt activation, induces apoptosis and delays tumor growth.
Oncogene. 24:3596–3605. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cossarizza A, Baccarani-Contri M,
Kalashnikova G and Franceschi C: A new method for the
cytofluorimetric analysis of mitochondrial membrane potential using
the J-aggregate forming lipophilic cation
5,5′,6,6′-tetrachloro-1,1′,3,3′-tetraethylbenzimidazolcarbocyanine
iodide (JC-1). Biochem Biophys Res Commun. 197:40–45.
1993.PubMed/NCBI
|
|
25
|
Kroemer G, Zamzami N and Susin SA:
Mitochondrial control of apoptosis. Immunol Today. 18:44–51. 1997.
View Article : Google Scholar
|
|
26
|
Mitsopoulos P and Suntres ZE: Protective
effects of liposomal N-Acetylcysteine against paraquat-induced
cytotoxicity and gene expression. J Toxicol. 2011:1–14. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Konopleva M, Contractor R, Tsao T, et al:
Mechanisms of apoptosis sensitivity and resistance to the BH3
mimetic ABT-737 in acute myeloid leukemia. Cancer Cell. 10:375–388.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Nguyen TT, Tran E, Nguyen TH, et al: The
role of activated MEK-ERK pathway in quercetin-induced growth
inhibition and apoptosis in A549 lung cancer cells. Carcinogenesis.
25:647–659. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cusimano A, Foderà D, D'Alessandro N, et
al: Potentiation of the antitumor effects of both selective
cyclooxygenase-1 and cyclooxygenase-2 inhibitors in human hepatic
cancer cells by inhibition of the MEK/ERK pathway. Cancer Biother.
6:1461–1468. 2007.PubMed/NCBI
|
|
30
|
Kunnimalaiyaan M, Ndiaye M and Chen H:
Apoptosis-mediated medullary thyroid cancer growth suppression by
the PI3K inhibitor LY294002. Surgery. 140:1009–1014. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kong CS, Kimb JA, Yoon NY and Kim SK:
Induction of apoptosis by phloroglucinol derivative from
Ecklonia cava in MCF-7 human breast cancer cells. Food Chem
Toxicol. 47:1653–1658. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Haruna S, Kuroi R, Kajiwara K, Hashimoto
R, Matsugo S, Tokumaruc S and Kojoa S: Induction of apoptosis in
HL-60 cells by photochemically generated hydroxyl radicals.
Bioorgan Med Chem Lett. 12:675–676. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liu H, Xiao Y, Xiong C, Wei A and Ruan J:
Apoptosis induced by a new flavonoid in human hepatoma HepG2 cells
involves reactive oxygen species-mediated mitochondrial dysfunction
and MAPK activation. Eur J Pharmacol. 654:209–216. 2011. View Article : Google Scholar
|
|
34
|
Kim MJ, Liao J, Dowling ML, et al: TRAIL
inactivates the mitotic checkpoint and potentiates death induced by
microtubule-targeting agents in human cancer cells. Cancer Res.
68:3440–3449. 2008. View Article : Google Scholar
|
|
35
|
Danial NN and Korsmeyer SJ: Cell death:
critical control points. Cell. 116:205–219. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kluck RM, Bossy-Wetzel E, Green DR and
Newmeyer DD: The release of cytochrome c from mitochondria: a
primary site for Bcl-2 regulation of apoptosis. Science.
275:1132–1136. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Monteghirfo S, Tosetti F, Ambrosini C, et
al: Antileukemia effects of xanthohumol in Bcr/Abl-transformed
cells involve nuclear factor-kappaB and p53 modulation. Mol Cancer
Ther. 7:2692–2702. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Won SJ, Chung KS, Ki YS, Choi JH, Cho WJ
and Lee KT: CWJ-081, a novel 3-arylisoquinoline derivative, induces
apoptosis in human leukemia HL-60 cells partially involves reactive
oxygen species through c-Jun NH2-terminal kinase pathway. Bioorgan
Med Chem Lett. 20:6447–6451. 2010. View Article : Google Scholar
|
|
39
|
Han YM, Shin DS, Lee YJ, Ismail IA, Hong
SH, Han DC and Kwon BM: 2-Hydroxycurcuminoid induces apoptosis of
human tumor cells through the reactive oxygen species-mitochondria
pathway. Bioorgan Med Chem Lett. 21:747–751. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dillon RL, White DE and Muller WJ: The
phosphatidyl inositol 3-kinase signaling network: implications for
human breast cancer. Oncogene. 26:1338–1345. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Viglietto G, Motti ML, Bruni P, et al:
Cytoplasmic relocalization and inhibition of the cyclin-dependent
kinase inhibitor p27 (Kip1) by PKB/Akt-mediated phosphorylation in
breast cancer. Nat Med. 8:1136–1144. 2002. View Article : Google Scholar
|
|
42
|
Brunet A, Bonni A, Zigmond MJ, et al: AKT
promotes cell survival by phosphorylating and inhibiting a Forkhead
transcription factor. Cell. 96:857–868. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Bartkova J, Lukas J, Muller H, Lutzhoft D,
Strauss M and Bartek J: Cyclin D1 protein expression and function
in human breast cancer. Int J Cancer. 57:353–361. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li Y, Corradetti MN, Inoki K and Guan KL:
TSC2: filling the GAP in the mTOR signaling pathway. Trends Biochem
Sci. 29:32–38. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Vivanco I and Sawyers CL: The
phosphatidylinositol 3-kinase-AKT pathway in human cancer. Nat Rev
Cancer. 2:489–501. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
46
|
Fang JY and Richardson BC: The MAPK
signaling pathways and colorectal cancer. Lancet Oncol. 6:322–327.
2005. View Article : Google Scholar : PubMed/NCBI
|