Introduction
Infertility is a common health problem that affects
15% of couples worldwide (1). Male
partners may be involved in the etiology of half of the cases,
either as the primary or secondary cause (2). There are numerous known causes for
male infertility, however, important factors have been identified
recently, including oxidative stress (OS), DNA fragmentation and
aneuploidy of the sperm. OS is a product of the inequity between
reactive oxygen species (ROS) and antioxidants in the body. It is a
potent mechanism that leads to sperm damage and male infertility
(3). Normally, the seminal plasma
contains a specialized antioxidant system that provides effective
protection against OS (4). A
number of clinical studies have demonstrated a correlation between
male infertility and OS (5–8).
However, a fertility parameter that is not routinely evaluated is
DNA integrity. Sperm chromatin is an extremely organized structure
that consists of DNA and heterogeneous nucleoproteins. The
predominate nucleoproteins in the mature sperm are the protamines,
which replace the histones during spermiogenesis (9). Normal chromatin is essential for the
transmission of paternal genomic information and it is well
documented that fertility is negatively correlated with defective
sperm chromatin or DNA fragmentation (10,11).
Apart from DNA fragmentation, aneuploidy is another
form of abnormality in sperm DNA. Human spermatozoa are haploid
cells (n=23) that contain 22 autosomes and one sex chromosome,
either X or Y. Aneuploidy is the condition in which the sperm has
an extra chromosome (disomy) or a missing chromosome (nullisomy)
(12). The present study aimed to
achieve proper characterization of human semen, with particular
interest in the sperm DNA, together with an evaluation of the
oxidative status as a possible etiological factor for impaired
fertility.
Materials and methods
Study population and sample
collection
The present study was approved by the ethical review
board of the Arabian Gulf University. Verbal and written consents
were obtained from the volunteers. All patients received an
explanation of the study prior to obtaining informed consent. Semen
samples were collected from infertile male patients attending the
Salmania Medical Complex. All samples were collected by
masturbation into sterile containers following 3–4 days of sexual
abstinence. Subsequent to liquification, a conventional analysis
was performed according to the World Health Organization guidelines
(WHO Laboratory Manual for the Examination of Human Semen and
Sperm-Cervical Mucus Interaction, 1999). The variables taken into
account were the volume of the ejaculate (ml), the concentration of
round cells (x106/ml), the sperm concentration
(x106/ml), the forward motility (%) and the morphology
(% of normal forms). A leukocyte count (x106/ml) was
carried out using a standard peroxidase test. All the semen samples
were analyzed with flow cytometry (FCM) and the colorimetric assay
for total antioxidant capacity (TAC).
FCM analysis
Sample preparation
Native sampled preparations of a single cell
suspension were performed according to the method used by Ehemann
et al(13). The semen
samples were treated with two types of solutions; ethanol fixation
and citric acid preparation.
Sample staining for sperm DNA analysis by
FCM
The fixed samples were brought to room temperature
prior to staining. Each type of fixed cell had a specific staining
technique: The ethanol fixed cells were centrifuged at 337 × g for
10 min and resuspended in 3 ml 4′,6-diamidino-2-phenylindole
sulforhodamine (DAPI-SR) 101 staining solution. This was kept in
the dark for 20 min at room temperature. The citric acid prepared
cells were diluted with 3.5 ml of DAPI-phosphate staining solution
and directly analyzed.
Cytometry analysis of the fluorescence-stained
fixed samples
FCM analyses were performed using a CyFLOW space
flow cytometer (Partec, Münster, Germany) equipped with a UV laser
diode and a 488 nm argon laser and filter combination for
DAPI-stained single cells. The multicycle program was used for the
histogram analysis; each histogram represented 3×104
cells for measuring the DNA index. Human lymphocyte nuclei from
healthy donors were used as the internal standard for the
calibration of the diploid DNA-index.
Analysis of the frequency histograms
The DNA frequency histograms were evaluated using
cumulative frequency distributions ascribed to Hacker-Klom et
al(14). The term CC was used
to describe the condensed chromatin and indicate the haploid
spermatozoa, which had a normal DNA content. Five categories were
recognized in each histogram: Cells with a sub-haploid DNA content
<1CC (debris that may be of apoptotic origin); mature haploid
spermatozoa in the 1CC peak, haploid round spermatids in the 1C
peak; diploid spermatozoa in the 2CC peak, cells registered to the
right of the 2CC level, including 2C cells (leukocytes,
G1-spermatogonia and primary spermatocytes at
preleptotene), and to the right of 1CC, which are cells in the DNA
synthesis phase (S); and 4C cells (primary spermatocytes). The
presence of aneuploidy cells was marked as another bivarient
histogram with blue coloration. The software analyzed the two types
of population cells in the same diagram.
Semen TAC assay
Sample preparation
The samples were prepared according to the method
used by Said et al(15).
Following liquefaction, aliquots (300 μl) of each sample were
centrifuged at 300 × g for 7 min. The supernatant was aspirated and
recentrifuged at 300 × g for another 10 min. The seminal plasma was
frozen at −70°C until further use.
Colorimetric assay for TAC
The colorimetric assay for TAC was performed as per
the method of Mahfouz et al(4). Frozen seminal plasma was thawed by
placing the vials in an incubator at 37°C for 20 min and then
immediately assessed for the TAC. The TAC of the seminal plasma was
measured with the colorimetric method using the Cayman’s
Antioxidant Assay kit (Cayman’s Chemicals Company, Ann Arbor, MI,
USA). The seminal plasma samples were diluted at 1:10 with the
assay buffer prior to assaying. All the reagents and samples were
equilibrated to room temperature prior to beginning the assay. The
samples and Trolox standards were assayed twice by two observers.
The Trolox standards and reagent were prepared as per the
manufacturer’s instructions at the time of the assay. Trolox
standard (10 μl) and the samples were loaded into the corresponding
wells of a 96-well plate. Then, 10 μl metmyoglobin and 150 μl
chromogen were added to all the standard/sample wells. The reaction
was initiated by adding 40 μl hydrogen peroxide immediately. The
plate was covered and incubated for 5 min on a shaker at room
temperature. Absorbance was monitored at 750 nm using an ELx800
Absorbance Microplate Reader (Thomas Scientific, Swedesboro, NJ,
USA). The accuracy and sensitivity of the TAC assay kit were
calculated according to the manufacturer’s instructions.
Statistical analysis
Data analysis was performed using the SPSS 16 for
Windows Software Package (SPSS Inc., Chicago, IL, USA). Data were
expressed as percentages and mean ± SD. To identify the independent
association between the various seminal parameters and selected
variables, a logistic regression analysis was carried out.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Study population and sample
collection
The age range of the patients was 21–52 years, with
a mean age of 30.8±6.7 years. In total, 86.5% of the patients
presented with primary infertility, while 13.5% complained of
secondary infertility. Based on the WHO criteria, patient samples
were either normal or abnormal; 51.9% of the samples were abnormal
and the rest were normozoospermic. As shown in Table I, the normozoospermic group had
significantly higher sperm concentrations, motility and normal
forms than the abnormal group. Contrary to expectation, not all of
the normozoospermic samples had normal cytometry DNA analyses. Of
52 samples obtained from the infertile male patients, 36 semen
samples demonstrated abnormal chromatin at an overall percentage of
67.3, whereas 16 had normal chromatin at an overall percentage of
32.7.
 | Table IAnalysis of the standard seminal
parameters in the normozoospermic and abnormal semen samples (mean
± SD). |
Table I
Analysis of the standard seminal
parameters in the normozoospermic and abnormal semen samples (mean
± SD).
| Parameter | Normal | Abnormal | P-value |
|---|
| Concentration
(x106/ml) | 54.28±1.47 | 15.41±1.66 | <0.005 |
| Motility (% grade
A) | 64.12±8.60 | 16.81±20.47 | <0.005 |
| Vitality (%
alive) | 76.84±8.61 | 25.00±29.30 | <0.005 |
| Morphology (% normal
form) | 63.08±9.3 | 15.65±20.07 | <0.005 |
| Leukocyte count | 0.36±0.49 | 3.22±3.46 | <0.005 |
The samples were further divided into eight classes
based on the classification described by Haker-Klom et
al(14). A total of 8 samples
(15.4%) were classified as class 1 and all these samples where
normozoospermic. Class 2 consisted of 9 samples (17.3%), from which
only one ejaculate had an abnormal spermiogram (leukocytospermia).
Class 3 contained the histograms of 7 samples (13.5%) and among
them, only one ejaculate exhibited asthenoteratospermic features.
Class 4 included two ejaculatory samples (3.8%) that had abnormal
spermiograms (oligoasthenoteratozoospermia and teratozoospermia).
Class 5 contained the histograms of 4 samples (7.7%) and only one
ejaculate was normozoospermic. Class 6 contained 9 histograms
(17.3%) and barely two samples had normal spermiograms. Classes 7
and 8 had samples with abnormal spermiograms. Class 7 contained 6
samples (11.5%), whose spermiograms showed severe oligozoospermia,
while class 8 contained 7 azoospermic samples (13.5%). The
association between the parameters from the FCM analysis and the
conventional semen parameters is described in Table II.
 | Table IICorrelation between DNA integrity and
conventional semen parameters. |
Table II
Correlation between DNA integrity and
conventional semen parameters.
| Correlation with
chromatin condensation | Correlation with
presence of aneuploidy cells |
|---|
|
|
|
|---|
| Parameter | r | P-value | r | P-value |
|---|
| Concentration | 0.587 | <0.001 | −0.339 | <0.005 |
| Normal morphology
% | 0.658 | <0.001 | 0.245 | NS |
| Motility | 0.625 | <0.001 | 0.260 | NS |
| WBC count | −0.299 | <0.005 | −0.039 | NS |
Aneuploidy
There were significant correlations between the
sperm chromatin condensation and the sperm count, morphology and
motility; yet a significant but low negative correlation was
observed between the chromatin condensation and the white blood
cell (WBC) count. Another notable abnormality also observed during
FCM analysis was the presence of aneuploidy cells. Of a total of 52
samples, 19 semen samples (36.54%) showed aneuploidy features. The
mean percentage of aneuploidy cells was higher in the samples with
severe chromatin condensation abnormalities, for example, in
classes 7 and 8, compared with other samples (Table III). These abnormal cells were
present even in the normozoospermic samples (Table III). A noteworthy finding was the
presence of aneuploidy cells in the semen samples with normal
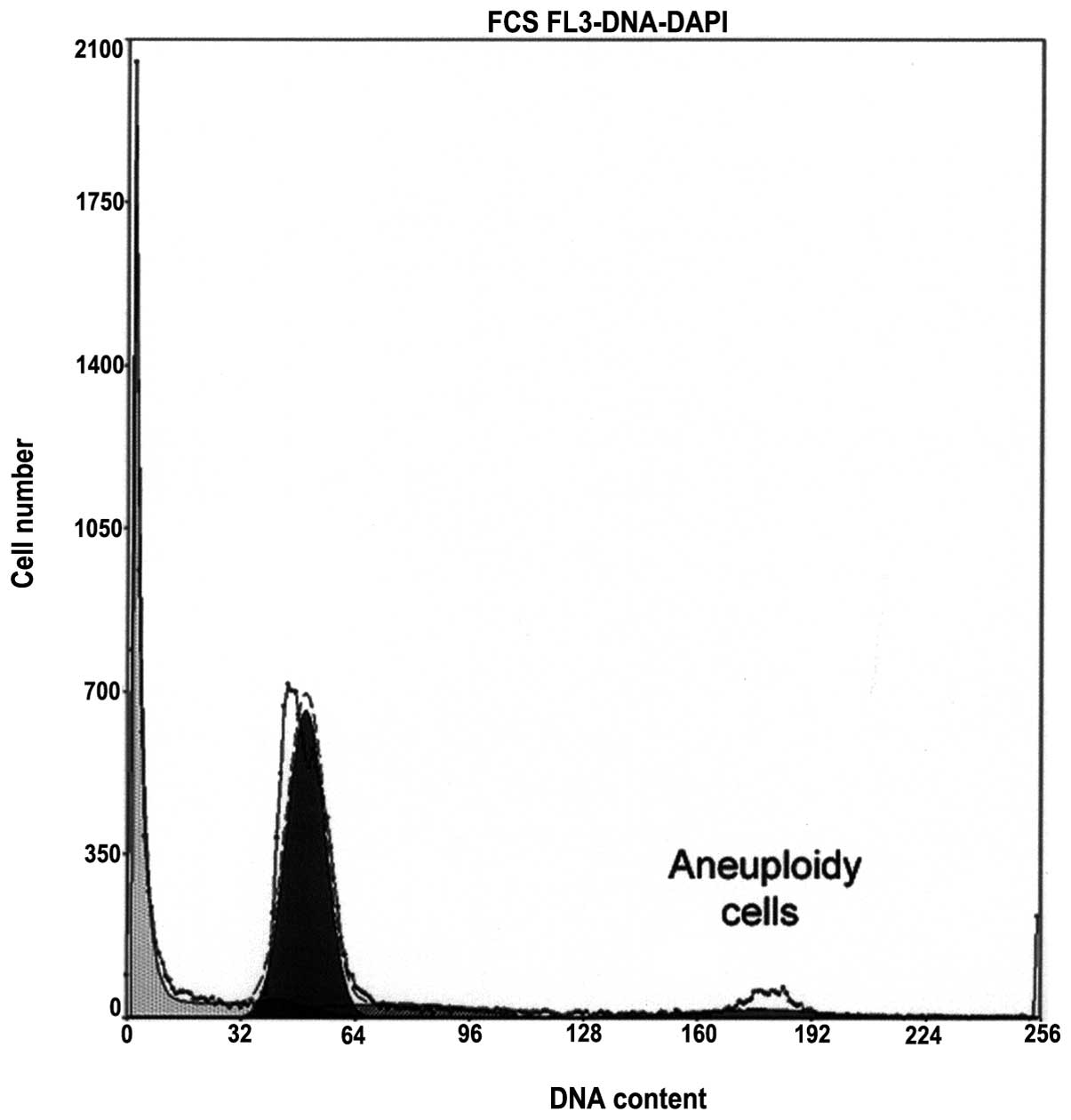
chromatin condensation (Fig. 1)
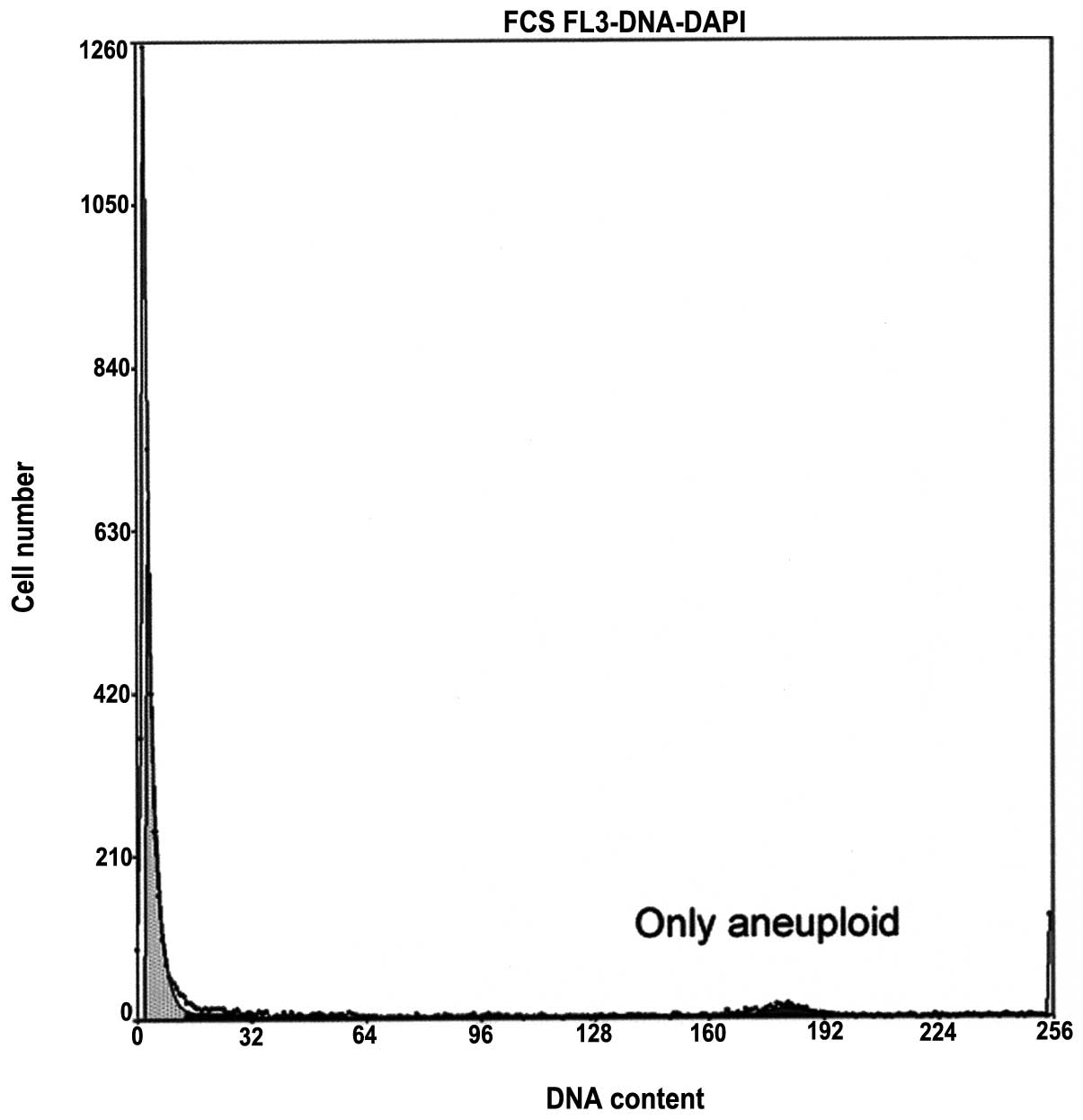
and even in the samples with complete azoospermia (class 8;
Fig. 2). There was a significant
negative correlation between the sperm concentration and the
presence of the aneuploidy cells. The samples with oligozoospermia
were more likely to have an abnormal aneuploidy rate than the other
samples (Table III). However, no
significant correlation existed between the occurrences of these
abnormal cells and the other semen parameters (Table II).
 | Table IIIAneuploidy rate in relation to
cytometry class in the 19 samples which had abnormal aneuploidy
rates.a |
Table III
Aneuploidy rate in relation to
cytometry class in the 19 samples which had abnormal aneuploidy
rates.a
| Sample No. | Aneuploidy,% | Class | Comment |
|---|
| 1 | 12.8 | 3 | Abnormal by WHO |
| 2 | 60 | 8 | Aspermia |
| 3 | 20.5 | 7 | Abnormal by WHO |
| 4 | 14.17 | 6 | Abnormal by WHO |
| 5 | 6.8 | 3 | Normal by WHO |
| 6 | 31.93 | 6 | Abnormal by WHO |
| 7 | 10.84 | 7 | Abnormal by WHO |
| 8 | 14.59 | 3 | Normal by WHO |
| 9 | 19.78 | 7 | Abnormal by WHO |
| 10 | 28.4 | 7 | Abnormal by WHO |
| 11 | 39.65 | 8 | Azoospermia |
| 12 | 51.31 | 8 | Azoospermia |
| 13 | 11.44 | 4 | Abnormal by
WHO |
| 14 | 11.2 | 3 | Normal by WHO |
| 15 | 51.85 | 6 | Abnormal by
WHO |
| 16 | 38.37 | 6 | Abnormal by
WHO |
| 17 | 10 | 2 | Normal CC by
WHO |
| 18 | 48.9 | 8 | Azoospermia |
| 19 | 8.9 | 5 | Abnormal by
WHO |
Concerning the TAC measurement, only 40.38% of the
patients had a normal level. The normozoospermic samples had a
higher mean level of TAC (1,500.92±280.29 μM) than the abnormal
samples (899.59±375.67 μM) (P<0.005). Moreover, the TAC levels
were much lower in the samples with leukocytospermia than the other
samples (1050.40±495.33 μM vs. 1,282.29±394.89 μM; P<0.005).
Another interesting feature was that the infertile subjects with an
abnormal chromatin condensation had significantly lower TAC levels
than the other subjects and that the level was markedly decreased
in the samples with severe abnormalities (class 7 and 8). For class
1 the mean level of TAC was 1,715.25±321.43 μM, while the mean
level of TAC in classes 7 and 8 was 769.83±241.81 and 842.29±
415.09 μM, respectively.
Discussion
In clinical practice, an unsatisfactory diagnostic
strategy remains for male infertility. This is due to the fact that
despite the variety of etiological factors, the traditional
technique for assessing human semen maintains its key role in the
evaluation of male fertility (16). Traditional analysis provides a
descriptive assessment of semen parameters, however, the normal
values of these factors do not truly reflect the fertilization
capacity of the sperm (17,18).
A previous study indicated the limited value of traditional
analysis, particularly in assisted reproductive technology (ART)
(8).
In the present study, traditional analysis revealed
that 48.1% of the study group were normozoospermic and that they
had higher parameter values than the abnormal samples, with the
exception of the leukocyte content, as shown in Table I. Fertility workers depend on these
results to differentiate between fertile and infertile males.
However, this appeared to be an incorrect method of judgment when
comparing the results of semen testing according to the WHO
criteria and analyzing the other recent fertility parameters of the
present study. Sperm DNA integrity has been recognized as an
important parameter for semen quality (19), although it is not assessed
routinely. A variety of assays have been developed to measure sperm
DNA damage. Certain techniques detect the actual breaks in the DNA
strands, whereas other approaches assess the vulnerability of the
DNA to denaturation (20). FCM is
a new method that has the ability to discriminate between the
variations in DNA damage in the sperm (21). The principles of FCM are based on
staining the sperm nuclei with a DNA stain to evaluate the ratio of
single- and double-stranded DNA (following acid exposure that
causes denaturation of the double-stranded DNA in the sperm with
abnormal chromatin) (13). This
technique assesses the sperm chromatin condensation and scores it
according to the Hacker-Klom scoring system (14). Briefly, the Hacker-Klom scoring
system divides the chromatin condensation state into eight classes,
considering only classes 1 and 2 as normal. The present study
revealed that 67.3% of the samples had abnormal chromatin. A number
of these samples had normal class 1 chromatin and they represented
15.4% of the samples. Other samples had severe abnormalities and
typical features of class 8 chromatin. The rest of the samples
exhibited the features of the other classes (Fig. 2). The DNA fragmentation was greater
in the males with abnormal semen parameters. The semen samples with
a low sperm concentration, low motility and poor morphology
exhibited more DNA fragmentation than the normal samples (Table II). These results were supported
by several previously published studies (8,22)
that revealed an increase in the number of DNA abnormalities in the
semen when there was an increase in the number of semen
abnormalities. Another fertility parameter assessed by FCM was the
aneuploidy rate of the spermatozoa. Aneuploidy gametes are a major
cause of pregnancy loss, aneuploid births and developmental defects
(23). In the present study,
measurements of the aneuploidy rate demonstrated that 36.53% of the
samples had abnormal aneuploidy cells. These abnormal cells mostly
appeared in the samples with abnormal chromatin condensation
(Table III). Nevertheless, the
present study has also shown that these cells may be detected in
samples with normal chromatin condensation (Fig. 1) and even in azoospermic samples
(Fig. 2). A previous study
detected abnormal aneuploidy cells in azoospermic samples (24), however, no study has recorded a
high percentage of these cells in samples with normal chromatin
condensation. The present study has demonstrated a negative
correlation between the sperm count and the aneuploidy rate
(r=−0.339, P<0.005), a result that is in agreement with that of
other studies that considered the patients with oligozoospermia at
high risk of carrying chromosomal abnormalities, particularly if
they chose to undergo the intracytoplasmic sperm injection (ICSI)
program (25,26).
The seminal TAC level was also assessed in the
present study. A low seminal TAC level is frequently associated
with male infertility (6). The
present study has shown that 59.62% of the samples had an abnormal
seminal TAC level; the seminal TAC was higher in the
normozoospermic samples. Additionally, the TAC level was negatively
correlated with leukocytospermia. This finding may be explained by
the fact that the excess WBCs generated more ROS. These results are
in concordance with the study by Sharma et al(27), which concluded that the presence of
WBCs in semen samples is associated with OS and impaired fertility.
Furthermore, there was a significant correlation between the sperm
chromatin fragmentation and oxidative stress of the semen. The
samples with a high degree of sperm DNA fragmentation had a low
seminal TAC level. This agrees with previous studies that observed
a negative correlation between the seminal TAC level and sperm
chromatin fragmentation (2,28).
In the present study, a significant difference in the results of
the various cellular and molecular tests compared with the
classical semen analysis was observed. The semen analysis revealed
that 48.1% of the study group had normal semen parameters, while by
contrast, the aneuploidy rate measurements demonstrated that 63.47%
of the samples were normal. The results of the chromatin
condensation analysis showed that 32.7% of the samples were normal.
Additionally, the measurement of the seminal TAC level revealed
that only 40.38% of the samples were normal. Another finding of the
present study was that only 25% of the patients had a normal
spermiogram combined with normal chromatin condensation, a normal
aneuploidy rate and a normal level of TAC. Only 23% of the patients
had abnormal ejaculates by all analytical measurements. These
variations in the results agreed with the study by Aitikan et
al which considered that the semen parameters measured by
conventional analysis are not able to assist the development of a
diagnosis but may only provide a description of the seminal
phenotype (17).
References
|
1
|
Sharma RS, Gaur KK, Pal PC, Manocha M,
Tomar D, Khan AA, et al: Semen characteristics: Advancement in
andrological assessment. Indian J Clin Biochem. 20:173–183. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tremellen K: Oxidative stress and male
infertility - a clinical perspective. Hum Reprod Update.
14:243–258. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Makker K, Agarwal A and Sharma R:
Oxidative stress and male infertility. Indian J Med Res.
129:357–367. 2009.
|
|
4
|
Mahfouz R, Sharma R, Lackner J, Aziz N and
Agarwal A: Evaluation of chemiluminescence and flow cytometry as
tools in assessing production of hydrogen peroxide and superoxide
anion in human spermatozoa. Fertil Steril. 92:819–827. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Eskenazi B, Kidd SA, Marks AR, Sloter E,
Block G and Wyrobek AJ: Antioxidant intake is associated with semen
quality in healthy men. Hum Reprod. 20:1006–1012. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Agarwal A, Sharma RK, Nallella KP, Thomas
AJ Jr, Alvarez Jg and Sikka SC: Reactive oxygen species as an
independent marker of male factor infertility. Fertil Steril.
86:878–885. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pasqualotto FF, Sundaram A, Sharma RK,
Borges E Jr, Pasqualotto EB and Agarwal A: Semen quality and
oxidative stress scores in fertile and infertile patients with
varicocele. Fertil Steril. 89:602–607. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Varghese AC, Bragais FM, Mukhopadahyay D,
Kundu S, Pal M, Bhattacharyya AK and Agarwal A: Human sperm DNA
integrity in normal and abnormal semen samples and its correlation
with sperm characterstics. Andrologia. 41:207–215. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Carrell DT, Emery BR and Hammoud S:
Altered protamine expression and diminished spermatogenesis: what
is the link? Hum Reprod Update. 13:313–327. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Evenson DP and Wixon R: Clincal aspects of
sperm DNA fragmentation detection and male infertility.
Theriogenology. 65:979–991. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bungum M, Humaidan P, Axmon A, Spano M,
Bungum L, Erenpreiss J and Giwercman A: Sperm DNA integrity
assessment in prediction of assisted reproduction technology
outcome. Hum Reprod. 22:174–179. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Prisant N, Escalier D, Soufir JC, Morillon
M, Schoevaert D, Misrahi M and Tachdjian G: Ultrastructural nuclear
defects and increased chromosome aneuploidies in spermatozoa with
elongated heads. Hum Reprod. 22:1052–1059. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ehemann V, Sykora J, Vera-Delgado J, Lange
A and Otto HF: Flow cytometric detection of spontaneous apoptosis
in human breast cancer using the TUNEL-technique. Cancer Lett.
194:125–131. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hacker-Klom UB, Göhde W, Nieschlag E and
Behre HM: DNA flow cytometry of human semen. Hum Reprod.
14:2506–2512. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Said TM, Kattal N, Sharma RK, Sikka SC,
Thomas AJ Jr, Mascha E and Agarwal A: Enhanced chemiluminescence
assay vs colorimetric assay for measurement of the total
antioxidant capacity of human seminal plasma. J Androl. 24:676–680.
2003.
|
|
16
|
Aziz N and Agarwal A: Evaluation of sperm
damage: beyond the World Health Organization criteria. Fertil
Steril. 90:484–485. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Aitken RJ, Baker MA and O’Bryan M:
Shedding light on chemiluminescence:the application of
chemiluminescence in diagnostic andrology. J Androl. 25:455–465.
2004.PubMed/NCBI
|
|
18
|
Marcus-Braun N, Braun G, Potashnik G and
Har-Vardi I: Effect of cryopreservation on quality and
fertilization capacity of human sperm. Eur J Obstet Gynecol Reprod
Biol. 116:63–66. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Agarwala A and Allamaneni SS: The effect
of sperm DNA damage on assisted reproduction outcomes. A review
Minerva Ginecol. 56:235–245. 2004.PubMed/NCBI
|
|
20
|
Zini A and Sigman M: Are tests of sperm
DNA damage clinically useful? Pros and cons. J Androl. 30:219–229.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Elshal M, El-Sayed IH, Elsaied MA,
El-Masry SA and Kumosani TA: Sperm head defects and disturbances in
spermatozoal chromatin and DNA integrities in idiopathic infertile
subjects: association with cigarette smoking. Clin Biochem.
42:589–594. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Trisini AT, Singh NP, Duty SM and Hauser
R: Relationship between human semen parameters and deoxyribonucleic
acid damage assessed by the neutral comet assay. Fertil Steril.
82:1623–1632. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rubes J, Vozdova M, Robbins WA, Rezacova
O, Perreault SD and Wyrobek AJ: Stable variants of sperm aneuploidy
among healthy men show associations between germinal and somatic
aneuploidy. Am J Hum Genet. 70:1507–1519. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Matzuk MM and Lamb DJ: Genetic dissection
of mammalian fertility pathways. Nat Cell Biol. 4(Suppl): S41–S49.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Vegetti W, Van Assche E, Frias A, et al:
Correlation between semen parameters and sperm aneuploidy rates
investigated by fluorescence in-situ hybridization in infertile
men. Hum Reprod. 15:351–365. 2000. View Article : Google Scholar
|
|
26
|
Calogero AE, De Palma A, Grazioso C,
Barone N, Romeo R, Rapazzo G and D’Agata R: Aneuploidy rate in
spermatozoa of selected men with abnormal semen parameters. Hum
Reprod. 16:1172–1179. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sharma RK, Pasqualotto AE, Nelson DR,
Thomas AJ Jr and Agarwal A: Relationship between seminal white
blood cell counts and oxidative stress in men treated at an
infertility clinic. J Androl. 22:575–583. 2001.
|
|
28
|
Aitken S and Roman SD: Antioxidative
system and oxidative stress in the testis. Molecular mechanisms in
Spermatogenesis. Cheng CY: Landes Bioscience; New York: 2008
|