Introduction
Irritable bowel syndrome (IBS) is a common chronic
gastrointestinal disorder with a prevalence of 5–20% and an
incidence of ~200 per 100,000 individuals (1). There are currently no radiological,
biochemical or endoscopic markers for the diagnosis of IBS
(2), which is therefore mainly
diagnosed by assessing the clinical symptoms of (3,4):
Abdominal discomfort/pain, altered bowel habits and
bloating/abdominal distension (5,6).
While IBS does not develop into serious diseases
(for example, colon cancer) and is not associated with increased
mortality rates (7,8), it considerably reduces quality of
life, and is an economic burden to the patient and society due to
direct and indirect costs (1). The
direct costs are attributable to the evaluation and treatment of
the condition, and the indirect costs may be due to absenteeism
from school or work, or presenteeism resulting in reduced
productivity while at school or work (1).
Chromogranin A (CgA) was first isolated from
secretory granules from the bovine adrenal medulla (9,10).
CgA is co-stored and co-released with monoamines and peptide
hormones from various sites, including the endocrine cells of the
gastrointestinal tract (9,10). CgA is commonly used as a marker for
gut endocrine cells and endocrine tumours (9–11).
Changes in the population of CgA-immunoreactive
endocrine cells, as a marker for the entire population of the
endocrine cells, have been investigated in the duodenum, ileum,
colon and rectum of IBS patients (12–14).
Whereas the density of CgA-immunoreactive cells is reduced in the
duodenum, ileum and colon, it does not differ from that of healthy
subjects in the rectum. To the best of our knowledge, alterations
in the population of CgA-immunoreactive cells in the stomach of
patients with IBS have not been investigated. Therefore, the
present study was undertaken to identify any changes in the total
population of endocrine cells in the stomach, as identified by CgA
immunoreactivity.
Materials and methods
Patients and controls
A total of 76 patients who fulfilled Rome III
Criteria (3,4) for IBS were included in the study.
These patients comprised 62 females and 14 males with a mean age of
32 years (range, 18–55 years). The predominant IBS symptoms were
diarrhoea (IBS-D), present in 26 patients, diarrhoea and
constipation (IBS-M) in 21 patients, and constipation (IBS-C) in 29
patients. All patients had suffered from IBS symptoms for a number
of years and none were able to associate the onset of these
symptoms with any particular event, such as a gastrointestinal
infection. All patients underwent a complete physical examination
and were submitted to blood analysis of the following parameters:
Full blood count, electrolytes, inflammatory markers, and liver and
thyroid function.
Healthy volunteers without any gastrointestinal
complaints were recruited as controls via a local announcement at
Stord Hospital (Stord, Norway), Haukelands University Hospital
(Bergen, Norway) and the University of Bergen (Bergen, Norway), as
well as in local newspapers. A total of 59 healthy subjects were
included: 15 were from the population of Stord city, and 44 were
students or hospital employees. The controls comprised 43 females
and 16 males with a mean age of 38 years (range, 20–67 years).
The study was performed in accordance with the
Declaration of Helsinki and was approved by the Committee for
Medical Research Ethics at the University of Bergen (Bergen,
Norway). All subjects gave oral and written consent to participate
in the study.
Gastroscopy, histopathology and
immunohistochemistry
Following an overnight fast, the patients and
controls underwent standard gastroscopy, during which three biopsy
samples were obtained from the antrum and three from the corpus
(major curvature) of the stomach. Two additional biopsy samples
were removed from the antrum and used for a rapid urease test to
identify the presence of Helicobacter pylori.
The biopsy samples were fixed overnight in 4%
buffered paraformaldehyde, embedded in paraffin and cut into 5-μm
sections. The sections were stained with haematoxylin and eosin and
immunostained using the avidin-biotin complex (ABC) method with the
Vectastain ABC kit (Vector Laboratories, Burlingame, CA, USA). The
primary antibody used was a monoclonal mouse antibody raised
against the N-terminal of purified CgA (M869; Dako, Carpinteria,
CA, USA). The sections were incubated with primary antibody diluted
to 1:1,000 at room temperature for 2 h. The sections were then
washed in phosphate-buffered saline (PBS; pH 7.4) and incubated
with biotinylated swine anti-mouse IgG (DakoCytomation, Glostrup,
Denmark) diluted to 1:200 for 30 min at room temperature.
Subsequent to washing the slides in PBS buffer, the sections were
incubated for 30 min with avidin-biotin-peroxidase complex
(DakoCytomation) diluted to 1:100 and then immersed in
3,3′-diaminobenzidine peroxidase substrate (Vector Laboratories),
followed by counterstaining with haematoxylin.
Computerised image analysis
The number of immunoreactive cells and the area of
epithelial cells were measured using Olympus cellSens imaging
software (version 1.7; Olympus, Tokyo, Japan) on a computer linked
to an Olympus microscope (type BX 43) with built-in Koehler
illumination for transmitted light, a light-intensity manager, a
switchable high-colour-reproductivity LED light source, a 6 V/30 W
halogen bulb and an Olympus camera (DP 26). The number of
CgA-immunoreactive cells in each field was counted manually and the
area of epithelium containing these cells was drawn manually using
a computer. The intensity of CgA immunoreactivity in each field was
measured using an automatic threshold setting. A ×40 objective was
used, which resulted in each frame (field) on the monitor
representing a tissue area of 0.035 mm2. For each
subject, measurements were performed in ten randomly selected
fields. Immunostained sections from the IBS patients and controls
were coded and mixed, and measurements were conducted by the same
individual who was blinded to the identity of the sections. The
data from the fields were tabulated, and the cell density of the
epithelium (in cells/mm2) and the immunoreactivity
intensity were computed and analysed statistically.
Statistical analysis
The differences in gender and the occurrence of
H. pylori infection between patients and controls were
examined using Fisher’s exact test, and the age difference was
analysed by the Mann-Whitney nonparametric test. The differences
among controls, all IBS patients (IBS-total), IBS-D, IBS-M and
IBS-C patients were investigated using the Kruskal-Wallis
nonparametric test with Dunn’s post test. The data are presented as
the mean ± the standard error of the mean and P<0.05 was
considered to indicate a statistically significant difference.
Results
Patients and controls
The gender and age distributions did not
significantly differ between the patients and the controls (P=0.297
and P=0.824, respectively). H. pylori infection was
identified in three of the patients and two of the control subjects
(as evidenced by the urease test and by histopathological
examination), and its incidence did not significantly differ
between the two groups (P=0.64).
Gastroscopy, histopathology and
immunohistochemistry
The oesophagus, stomach and duodenum of the patients
and the controls were macroscopically normal. Histopathological
examination of the stomach revealed normal histology.
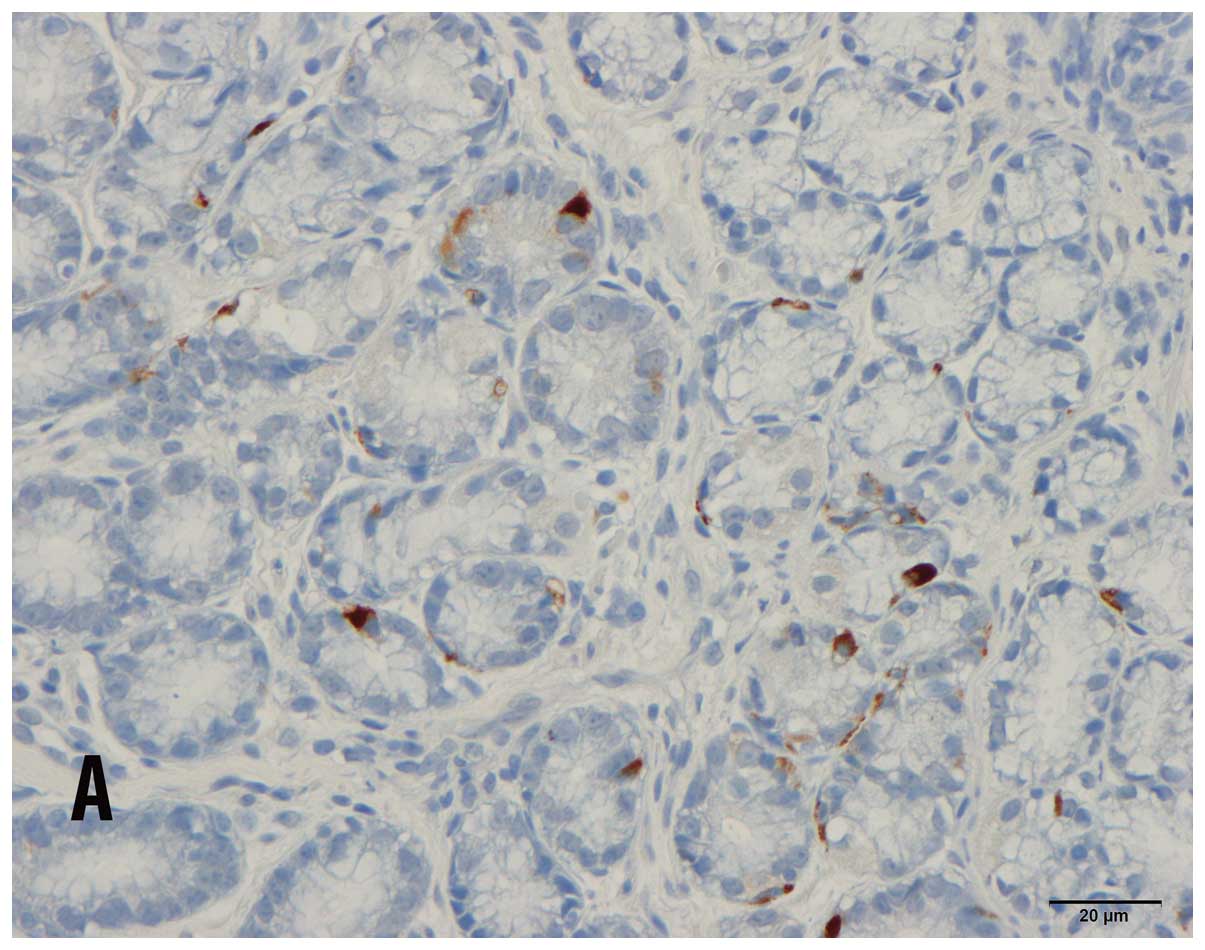
CgA-immunoreactive cells were observed in the antra and corpora of
patients and controls, and were basket- or flask-shaped,
occasionally with a long basal cytoplasmic process.
Computerised image analysis
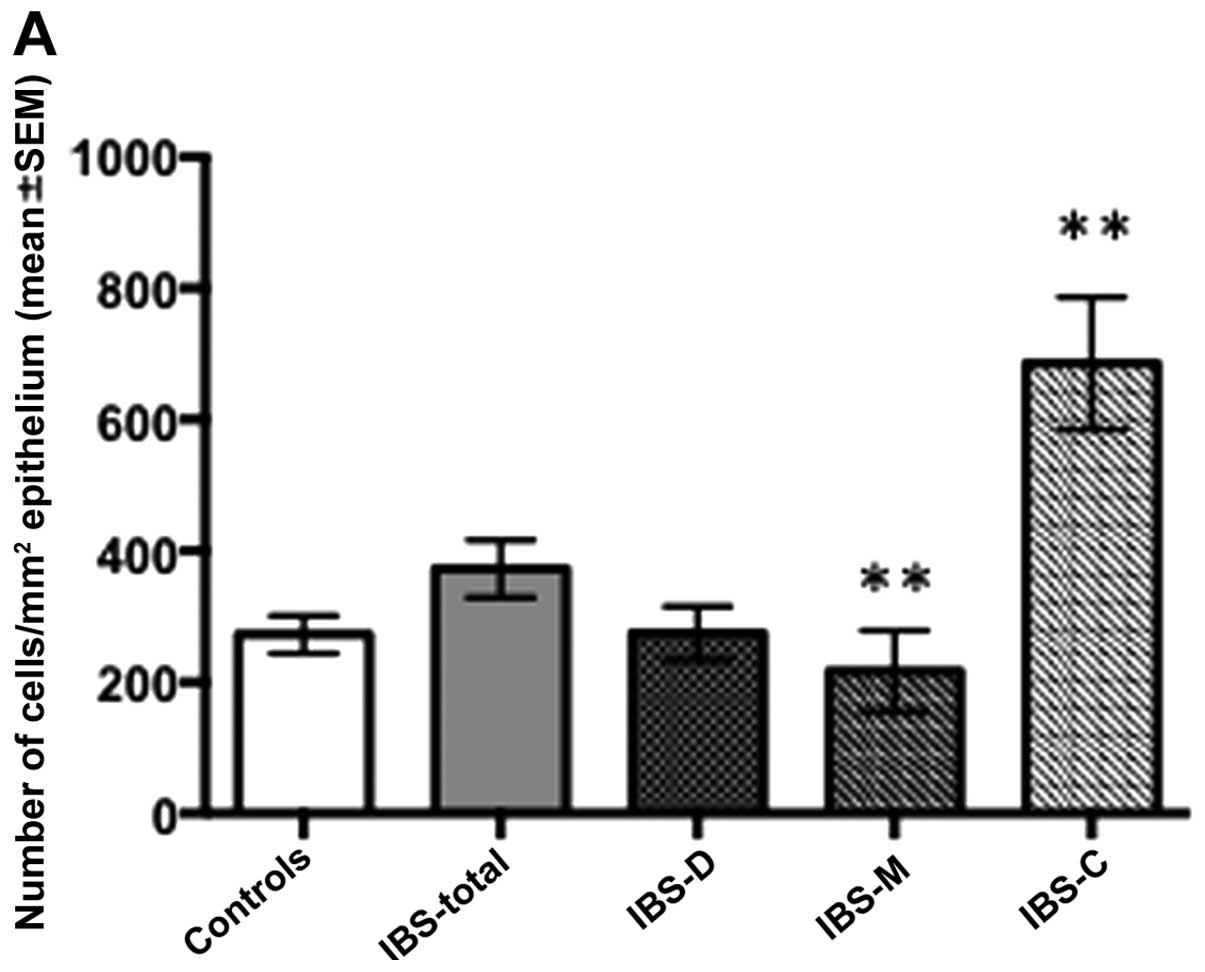
In the antra, the densities of CgA cells were
measured as 272.5±28.6, 372.7±43.6, 274.6±41.2, 217.0±61.9 and
685.3±100.5 cells/mm2 in the controls and the IBS-total,
IBS-D, IBS-M and IBS-C patients, respectively. The Kruskal-Wallis
test demonstrated that the CgA cell density differed significantly
between the controls and the IBS-total and IBS subgroups (P=0.01).
The Cga cell density was significantly lower in the IBS-M group and
significantly higher in the IBS-C group relative to the controls
(P=0.003 and P<0.01, respectively; Figs. 1 and 2). The intensities of the anti-CgA
immunoreactivity were 122.2±1.4, 126.0±0.8, 125.7±0.9, 124.7±3.7
and 127.0±1.4, in the controls and the IBS-total, IBS-D, IBS-M and
IBS-C patients, respectively. No significant differences in this
parameter were detected among any of the groups (P=0.14; Figs. 1 and 2).
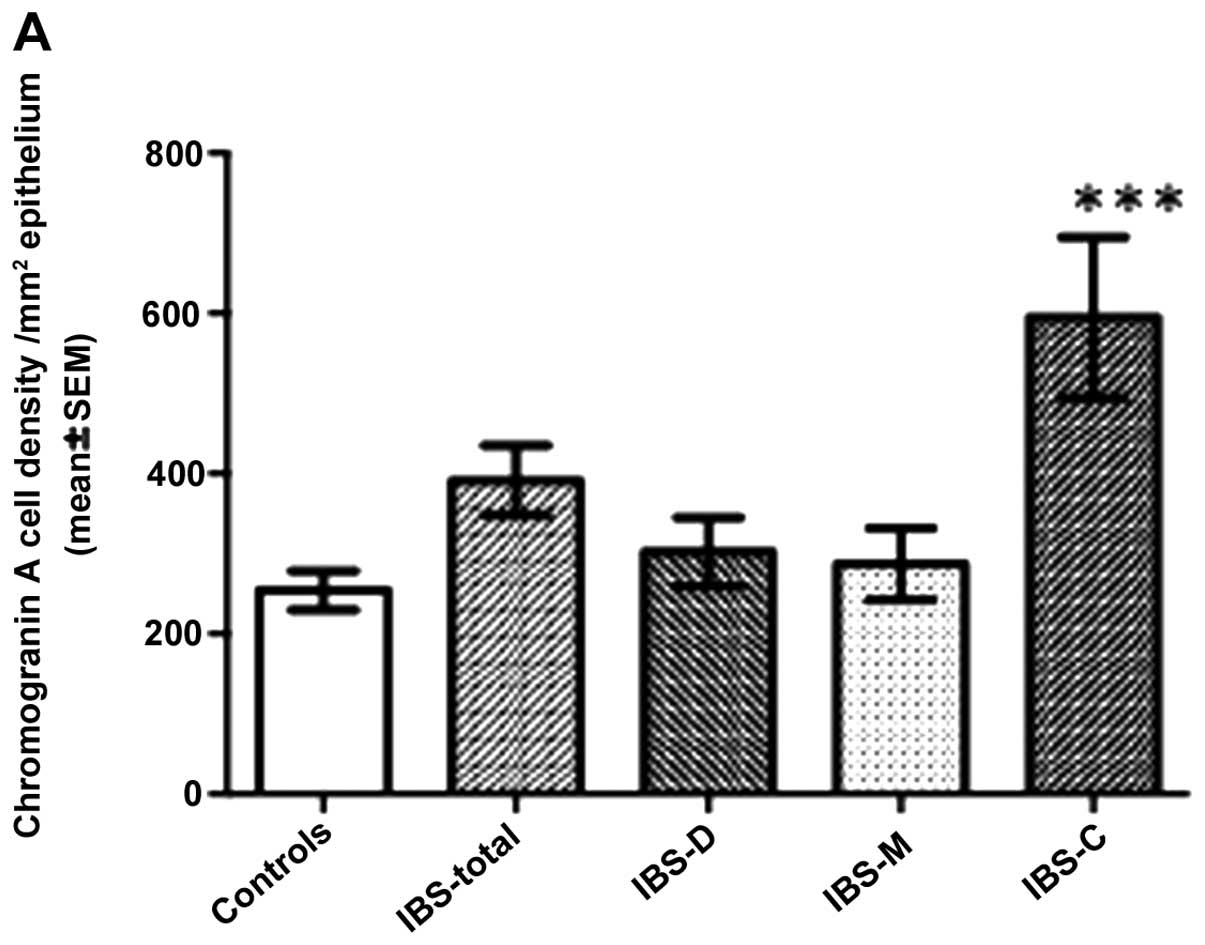
In the corpora, the densities of CgA cells were
254.1±24.6, 391.3±43.4, 302.4±42.6, 286.9±44.9 and 594.7±100.1
cells/mm2 in the controls and the IBS-total, IBS-D,
IBS-M and IBS-C patients, respectively. Multi-comparison with the
Kruskal-Wallis test demonstrated that the CgA cell density differed
significantly between the controls and the IBS-total and all IBS
subgroups (P=0.004), and particularly between patients with IBS-C
and the controls (P=0.0002; Figs.
3 and 4). The intensities of
the CgA immunoreactivity were 118.3±1.6, 121.4±1.0, 120.7±1.7,
124.0±2.2 and 120.7±1.7 in the controls and the IBS-total, IBS-D,
IBS-M and IBS-C patients, respectively. As in the antrum, this
parameter did not differ significantly among any of the groups
(P=0.29; Figs. 3 and 4).
Discussion
The findings of the present study revealed that the
density of all endocrine cells in the antrum and corpus of the
stomach, as identified by CgA immunoreactivity, is abnormal in
certain patients with IBS. This abnormality was not present for all
IBS subtypes, the density of CgA-immunoreactive cells was increased
in the antra and corpora of patients with IBS-C, and reduced in the
antra of patients with IBS-M relative to the controls. Whereas
CgA-immunoreactive cells in the antrum represent the population of
gastrin-, serotonin- and somatostatin-secreting cells, in the
corpus CgA-immunoreactive cells represent ghrelin-, serotonin- and
somatostatin-secreting cells (1,15,16).
Thus, although CgA cell density in the antrum and the corpus was
unchanged in IBS-D patients, changes in particular endocrine cells
cannot be excluded.
Computerised image analysis has been used for the
past 15 years for determining the cellular concentrations of
certain substances in a section of tissue by measuring the
intensity of immunoreactivity produced by the colour of the
immunohistochemical staining (17). When this practise was initially
implemented, measuring these parameters was tedious, with
instability in the light source also rendering the measurements
unreliable. Subsequent advances in computer software and
microscopic illumination have greatly improved the measurement
reliability. However, the intensity of immunoreactivity is
expressed in arbitrary units and thus is only useful for
comparisons among groups immunostained under the same conditions.
In the present study, no significant differences were detected in
the intensities of CgA immunoreactivity in the antra and the
corpora of the controls and IBS patients, including all IBS
subtypes. This indicates that the cellular content of CgA was
similar in the controls and all types of IBS patients. The cellular
content of CgA reflects the synthesis and release of CgA, and the
findings suggest that these functions are not affected in IBS
patients.
With the exception of ghrelin in the oxyntic mucosa
of the stomach of IBS patients (15,18),
to the best of our knowledge the endocrine cells in the stomach of
IBS patients have not been previously investigated. The density of
ghrelin-secreting cells in the stomach was reported to be lower in
IBS-C and higher in IBS-D than in healthy controls (19). Abnormalities in the endocrine cells
in other parts of the gastrointestinal tract, including the
duodenum, ileum, colon and rectum, have been reported in patients
with IBS (12,13,19–30).
In the duodenum, the cell densities of gastric inhibitory
polypeptid- and somatostatin-secreting cells were reduced in IBS-D
and IBS-C (21). The densities of
duodenal secretin- and cholecystokinin (CCK)-secreting cells were
reduced in IBS-D but unchanged in IBS-C, while the duodenal
serotonin cells were not affected in either IBS-D or IBS (21). Furthermore, in postinfectious IBS,
an increased number of duodenal CCK cells but a reduced number of
serotonin-secreting cells have been reported (20). Densities of colonic serotonin- and
peptide YY (PYY)-secreting cells were reportedly low in IBS-D and
IBS-C (22). In the rectum, the
densities of PYY and enteroglucagon-secreting cells were
significantly lower and the density of somatostatin-secreting cells
was significantly higher in IBS-D and IBS-C than in controls,
whereas the density of serotonin-secreting cells in these patients
did not differ from that in healthy controls (24,23).
Densities of rectal serotonin- and PYY-secreting cells in
post-infectious IBS were reported to be elevated (26,28,30,31,32).
In conclusion, the total numbers of endocrine cells
were abnormal in the stomachs of patients with IBS. This
abnormality was not associated with a change in the intensity of
the immunoreactivity, possibly indicating unchanged synthesis
and/or release of the hormones. In addition, the changes in
endocrine cells observed in the present study varied among the IBS
subtypes, which may indicate a role of the endocrine cells in
differential symptomologies. The findings of the present study add
to the evidence supporting the presence of endocrine cell
abnormalities throughout the gastrointestinal tract in IBS
patients.
Acknowledgements
This study was supported by a grant from
Helse-Fonna.
References
|
1
|
El-Salhy M, Gundersen D, Hatlebakk JG and
Hausken T: Irritable Bowel Syndrome: Diagnosis, Pathogenesis and
Treatment Options. Nova Science Publishers Inc.; New York, NY:
2012
|
|
2
|
Spiller RC: Potential biomarkers.
Gastroenterol Clin North Am. 40:121–139. 2011. View Article : Google Scholar
|
|
3
|
Spiller R, Aziz Q, Creed F, et al;
Clinical Services Committee of The British Society of
Gastroenterology. Guidelines on the irritable bowel syndrome:
mechanisms and practical management. Gut. 56:1770–1798. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Longstreth GF, Thompson WG, Chey WD,
Houghton LA, Mearin F and Spiller RC: Functional bowel disorders.
Gastroenterology. 130:1480–1491. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Drossman DA, Li Z, Andruzzi E, et al: U.S.
householder survey of functional gastrointestinal disorders.
Prevalence, sociodemography, and health impact. Dig Dis Sci.
38:1569–1580. 1993. View Article : Google Scholar
|
|
6
|
Thompson WG: A world view of IBS.
Irritable bowel syndrome: diagnosis and treatment. Camilleri M and
Spiller RC: Saunders; Philadelphia: pp. 17–26. 2002
|
|
7
|
Harvey RF, Mauad EC and Brown AM:
Prognosis in the irritable bowel syndrome: a 5-year prospective
study. Lancet. 1:963–965. 1987.PubMed/NCBI
|
|
8
|
Nørgaard M, Farkas DK, Pedersen L, et al:
Irritable bowel syndrome and risk of colorectal cancer: a Danish
nationwide cohort study. Br J Cancer. 104:1202–1206.
2011.PubMed/NCBI
|
|
9
|
Taupenot L, Harper KL and O’Connor DT: The
chromogranin-secretogranin family. N Engl J Med. 348:1134–1149.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wiedenmann B and Huttner WB: Synaptophysin
and chromogranins/secretogranins - widespread constituents of
distinct types of neuroendocrine vesicles and new tools in tumor
diagnosis. Virchows Arch B Cell Pathol Incl Mol Pathol. 58:95–121.
1989. View Article : Google Scholar
|
|
11
|
Deftos LJ: Chromogranin A: its role in
endocrine function and as an endocrine and neuroendocrine tumor
marker. Endocr Rev. 12:181–187. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
El-Salhy M, Lomholt-Beck B and Hausken T:
Chromogranin A as a possible tool in the diagnosis of irritable
bowel syndrome. Scand J Gastroenterol. 45:1435–1439. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
El-Salhy M, Mazzawi T, Gundersen D and
Hausken T: Chromogranin A cell density in the rectum of patients
with irritable bowel syndrome. Mol Med Report. 6:1223–1225.
2012.PubMed/NCBI
|
|
14
|
El-Salhy M, Wendelbo IH and Gundersen D:
Reduced chromogranin A cell density in the ileum of patients with
irritable bowel syndrome. Mol Med Rep. 7:1241–1244. 2013.PubMed/NCBI
|
|
15
|
El-Salhy M, Seim I, Chopin L, Gundersen D,
Hatlebakk JG and Hausken T: Irritable bowel syndrome: the role of
gut neuroendocrine peptides. Front Biosci (Elite Ed). 4:2783–2800.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
El-Salhy M, Ostgaard H, Gundersen D,
Hatlebakk JG and Hausken T: The role of diet in the pathogenesis
and management of irritable bowel syndrome (Review). Int J Mol Med.
29:723–731. 2012.PubMed/NCBI
|
|
17
|
El-Salhy M and Mahdavi J: Image analysis
of the duodenal endocrine cells in mice with particular regard to
optic densitomertry. Uppsala J Med Sci. 102:175–184. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
El-Salhy M: Ghrelin in gastrointestinal
diseases and disorders: a possible role in the pathophysiology and
clinical implications (review). Int J Mol Med. 24:727–732. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
El-Salhy M, Lillebø E, Reinemo A and
Salmelid L: Ghrelin in patients with irritable bowel syndrome. Int
J Mol Med. 23:703–707. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dizdar V, Spiller R, Singh G, et al:
Relative importance of abnormalities of CCK and 5-HT (serotonin) in
Giardia-induced post-infectious irritable bowel syndrome and
functional dyspepsia. Aliment Pharmacol Ther. 31:883–891. 2010.
|
|
21
|
El-Salhy M, Vaali K, Dizdar V and Hausken
T: Abnormal small-intestinal endocrine cells in patients with
irritable bowel syndrome. Dig Dis Sci. 55:3508–3513. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
El-Salhy M, Gundersen D, Ostgaard H,
Lomholt-Beck B, Hatlebakk JG and Hausken T: Low densities of
serotonin and peptide YY cells in the colon of patients with
irritable bowel syndrome. Dig Dis Sci. 57:873–878. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
El-Salhy M, Gundersen D, Hatlebakk JG,
Gilja OH and Hausken T: Abnormal rectal endocrine cells in patients
with irritable bowel syndrome. Regul Pept. 188:60–65. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Coates MD, Mahoney CR, Linden DR, et al:
Molecular defects in mucosal serotonin content and decreased
serotonin reuptake transporter in ulcerative colitis and irritable
bowel syndrome. Gastroenterology. 126:1657–1664. 2004. View Article : Google Scholar
|
|
25
|
Wang SH, Dong L, Luo JY, et al: Decreased
expression of serotonin in the jejunum and increased numbers of
mast cells in the terminal ileum in patients with irritable bowel
syndrome. World J Gastroenterol. 13:6041–6047. 2007. View Article : Google Scholar
|
|
26
|
Lee KJ, Kim YB, Kim JH, Kwon HC, Kim DK
and Cho SW: The alteration of enterochromaffin cell, mast cell, and
lamina propria T lymphocyte numbers in irritable bowel syndrome and
its relationship with psychological factors. J Gastroenterol
Hepatol. 23:1689–1694. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Park JH, Rhee PL, Kim G, et al:
Enteroendocrine cell counts correlate with visceral
hypersensitivity in patients with diarrhoea-predominant irritable
bowel syndrome. Neurogastroenterol Motil. 18:539–546. 2006.
View Article : Google Scholar
|
|
28
|
Kim HS, Lim JH, Park H and Lee SI:
Increased immunoendocrine cells in intestinal mucosa of
postinfectious irritable bowel syndrome patients 3 years after
acute Shigella infection - an observation in a small case control
study. Yonsei Med J. 51:45–51. 2010.
|
|
29
|
Dunlop SP, Coleman NS, Blackshaw E, et al:
Abnormalities of 5-hydroxytryptamine metabolism in irritable bowel
syndrome. Clin Gastroenterol Hepatol. 3:349–357. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Spiller RC, Jenkins D, Thornley JP, et al:
Increased rectal mucosal enteroendocrine cells, T lymphocytes, and
increased gut permeability following acute Campylobacter enteritis
and in post-dysenteric irritable bowel syndrome. Gut. 47:804–811.
2000. View Article : Google Scholar
|
|
31
|
Wang LH, Fang XC and Pan GZ: Bacillary
dysentery as a causative factor of irritable bowel syndrome and its
pathogenesis. Gut. 53:1096–1101. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Dunlop SP, Jenkins D, Neal KR and Spiller
RC: Relative importance of enterochromaffin cell hyperplasia,
anxiety, and depression in postinfectious IBS. Gastroenterology.
125:1651–1659. 2003. View Article : Google Scholar : PubMed/NCBI
|