Introduction
Cervical cancer remains the second most common
cancer in women worldwide. In 2008, there were >529,800 new
cases and ~275,000 deaths caused by this malignancy (1,2).
Unfortunately, up to date, the molecular mechanisms involved in the
initiation and progression of cervical cancer have not been
elucidated. Understanding of the epigenetic modifications involved
in cervical cancer, such as those mediated by microRNAs, might shed
light on this issue (3,4).
microRNAs are a family of small, single-stranded
RNAs, which can degrade or block the transcription of mRNA via
directly binding to the 3′-untranslated region of their target
genes (5,6). microRNAs have been demonstrated to
regulate cell proliferation, division, differentiation, invasion,
and migration (7,8). A number of microRNAs, such as
microRNA-218 (miR-218) have been shown to be downregulated and play
a role as tumor suppressors in human malignancies (9–13).
miR-218 has been shown to suppress cellular proliferation and
invasion and to induce apoptosis both in vitro and in
vivo (13–15). In a previous study, we demonstrated
that the circulating miR-218 level is significantly reduced in
cervical cancer patients, and associates with the late tumor stage,
lymphatic node metastasis and poor prognosis (16). Here, using a mouse model and
primary cultured tumor tissues, we aimed to explore the effects of
miR-218 restoration in human cervical cancer.
Materials and methods
Clinical specimens and primary
cultures
A total of 25 fresh cervical cancer tissues were
collected from patients who underwent surgery at the Affiliated
Hospital of Jiangnan University (the Fourth People’s Hospital of
Wuxi, Jiangsu Province, China). For RNA extraction, the tissues
were instantly stored in RNAlater solution (Qiagen, Valencia, CA,
USA) at −80°C.
For primary cultures, fresh tumor tissues were
washed with Hank’s buffered salt solution (HBSS) and gently minced
with a sterilized scalpel. Then, the tissues were digested with
0.1% collagenase IA (product no., C5894; Sigma-Aldrich,
Minneapolis, MN, USA) for 2 h at 37°C. Following a wash with
phosphate buffer saline, the mixture was resuspended to a density
of ~1×105 cells/ml. Next, 2×104 cells/well
were seeded into 96-well plates and incubated for 24 h prior to
carboplatin treatment. Permissions for this study were obtained
from the Ethical Committee of the Fourth People’s Hospital of Wuxi,
and informed written consent was obtained from all the patients.
Patient information was collected; this information is summarized
in Table I.
 | Table IClinical features of cervical cancer
patients. |
Table I
Clinical features of cervical cancer
patients.
| Variable | Value |
|---|
| Total number | 25 |
| Median age
(range) | 53.5 (39–68) |
| Low miR-218
level | 49.2±7.8 |
| High miR-218
level | 55.1±9.1 |
| Stagea |
| I | 8 |
| II | 7 |
| III | 6 |
| IV | 4 |
| Lymph node
metastasis |
| Negative | 20 |
| Positive | 5 |
| Tumor size (cm) |
| ≤4.0 | 13 |
| >4.0 | 12 |
Cervical cancer cell lines, culture and
reagents
Four human cervical cancer cell lines were used in
this study, HeLa, SiHa, C33A and CaSki, obtained from the American
Type Culture Collection (ATCC; Manassas, VI, USA). The cells were
cultured in the following media: RPMI-1640 supplemented with 10%
fetal bovine serum (FBS) for CaSki, Dulbecco’s Modified Eagle’s
medium/Nutrient Mixture F-12 (DMEM/F12) with 10% FBS for HeLa, SiHa
and C33A. All cell lines were cultured at 37°C, with 5%
CO2. Carboplatin was purchased from Sigma-Aldrich
(product no., C2538) and was freshly prepared before each
experiment at a 5 mM stock concentration. The miR-218 mimic and its
negative control were purchased form GenePharma Co., Ltd.
(Shanghai, China).
Transient and stable transfection with
miR218
The pGenesil-1-miR-218 expression plasmid was
obtained as previously described (15). Transient transfection was performed
with the miR-218 mimic and negative control in all four cell lines.
A total of 1×105 cells/well were plated into the 6-well
plates and incubated overnight. Transfection with miR-218 mimic (or
the negative control) was performed using Lipofectamine®
2000 (Thermo Fisher Scientific, Waltham, MA, USA), and G418 (0.4
mg/ml, Sigma-Aldrich) was used to select the stably transfected
colonies.
MTT assay
Cells (2×103 or 2×104
cells/well for primary cultured cells) were seeded into 96-well
plates and incubated overnight, then treated with different doses
of carboplatin for 72 h. Next, 5 μl MTT (5 mg/ml; Sigma-Aldrich)
and 100 μl dimethyl sulfoxide were sequentially added into each
well, and the absorbance was measured at 570 nm. All procedures
were performed in triplicate.
Cell-cycle analysis
Cells (3×106) were plated into a 60-mm
dish and incubated overnight before treatment with the miR-218
mimic or carboplatin. Twenty-four hours later, the cells were
harvested and suspended in 1 ml of staining solution, consisting of
50 μg/ml propidium iodide and 20 μg/ml RNase A (Thermo Fisher
Scientific). Then, the cells were analyzed on a FACScalibur flow
cytometer (BD Biosciences, San Diego, CA, USA). Flow cytometry data
were analyzed using CellQuest™ software (BD Biosciences). The
procedures were repeated in triplicate.
microRNA enrichment and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted using the Invitrogen TRIzol
reagent (Thermo Fisher Scientific). Then, the total RNA sample was
enriched in microRNAs using the Applied Biosystems®
mirVana miRNA Isolation kit (Thermo Fisher Scientific) according to
the manufacturer’s instructions. The expression of miR-218 was
measured by the Applied Biosystems Taqman miRNA assay kit that is
specific to miRNA-218, on an Applied Biosystems StepOnePlus™
Real-Time PCR system (all from Thermo Fisher Scientific). U6 RNA
was used as the endogenous control. The relative level of miR-218
was calculated with the 2−ΔΔCt method (17). All the procedures were repeated in
triplicate.
Western blotting
Total protein was extracted using RIPA buffer
(Beyotime Institute of Biotechnology, Shanghai, China) and the
protein concentration was measured using the BCA Protein Assay kit
(Bio-Rad Laboratories, Hercules, CA, USA). Equal amounts of protein
were separated by 12% SDS-PAGE and transferred onto polyvinylidene
fluoride membranes (Bio-Rad Laboratories). The membranes were
incubated overnight at 4°C with the primary antibodies rabbit
monoclonal anti-cyclin D1 and rabbit monoclonal anti-CDK4 (1:1,000
for anti-cyclin D1 and 1:500 for anti-CDK4; Cell Signaling
Technology, Beverly, MA, USA), and then incubated for 2 h at room
temperature with the secondary antibody horseradish
peroxidase-conjugated anti-rabbit IgG (monoclonal; Cell Signaling
Technology). The specific protein bands were detected using an
Enhanced Chemiluminescence system (Beyotime Institute of
Biotechnology).
Immunohistochemical staining (IHC)
IHC was performed as previously described (18). The primary antibodies used were:
anti-Ki 67 (1:200; Boster Biological Technology, Ltd., Wuhan,
China) and anti-cyclin D1 (1:300; Cell Signaling Technology).
Staining was performed with the Histostain-Plus IHC kit (Mingrui
Biotech Co., Ltd., Shanghai, China). The stained tissues were
visualized on an optical microscope (Leica Microsystems GmbH,
Wetzlar, Germany) and were separately evaluated by two
pathologists.
The tumor xenograft growth assay
The experimental procedures using animals in this
study were approved by the Animal Ethics Committee of the Jiangnan
University. Six- to eight-week-old female BALB/c nude mice were
purchased from SLAC Laboratory Animal Co., Ltd. (Shanghai, China).
SiHa cells (1×106) stably expressing miR-218 were
subcutaneously injected into the left posterior flanks of the nude
mice. As a negative control, an equal amount of SiHa cells
transfected with the empty vector was injected into the right
flanks. To test the effects of miR-218 on cellular sensitivity to
carboplatin, another group of nude mice bearing SiHa cells was
established following the same procedure, and carboplatin
(intraperitoneal injection, 30 mg/kg) was administrated at days 10,
17, 24 and 31. From 10 days after implantation, the tumor size was
measured every five days. Tumor volume = (length ×
width2)/2. Four weeks later, all mice were sacrificed,
and the tumor tissues were collected for further analysis.
Statistical analysis
The software SPSS 16.0 (SPSS Inc., Chicago, IL, USA)
was used for statistical analysis. The χ2 and the t-test
were used for categorical and quantitative data comparisons,
respectively. P<0.05 was considered to indicate a statistically
significant difference.
Results
miR-218 inhibits the growth of cervical
cancer cells
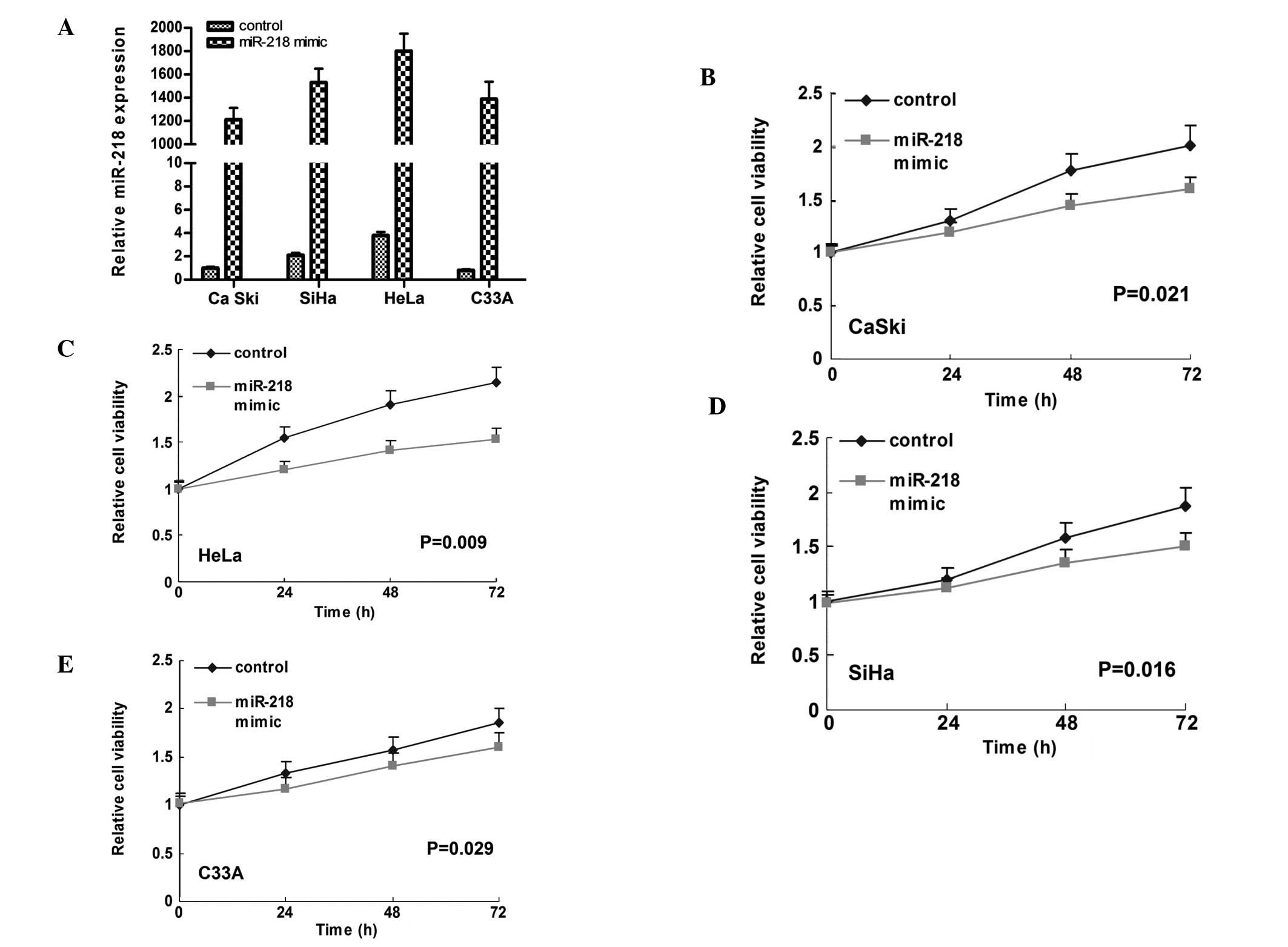
Compared to the negative control, 24-h treatment
with the miR-218 mimic increased the expression of miR-218 by
>1,000-fold in all four cell lines (Fig. 1A), and significantly inhibited
cellular growth (Fig. 1B–E,
P=0.021 for CaSki, P=0.009 for HeLa, P=0.016 for SiHa, and P=0.029
for C33A, respectively).
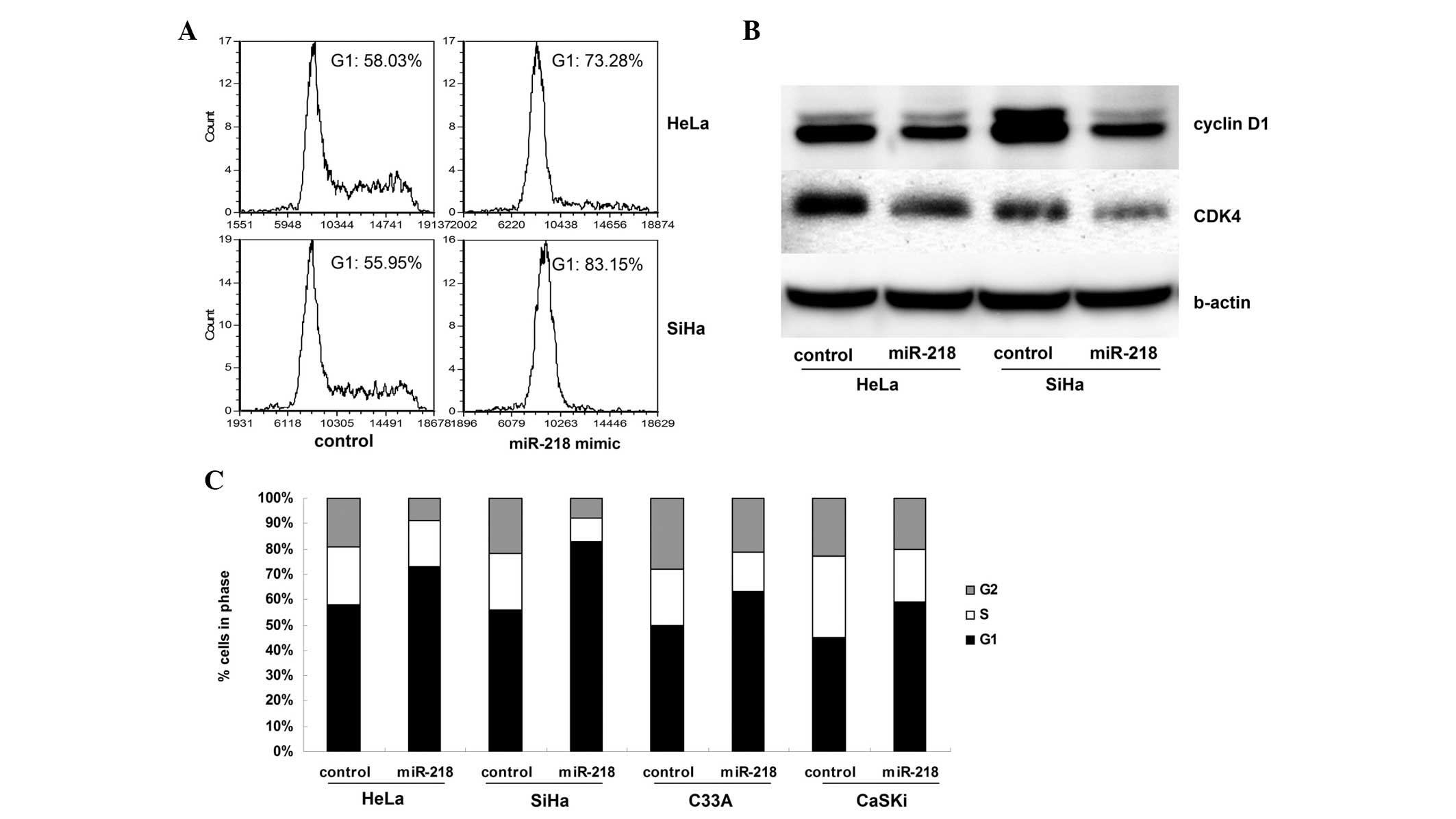
miR-218 induces G1 phase cell-cycle
arrest and inhibits cyclin D1 and CDK4
In HeLa and SiHa cells, overexpression of miR-218
arrested the majority of the cells at the G1 phase (Fig. 2A), but this arrest was not
prominent in the C33A and CaSKi cell lines (Fig. 2C). In addition, the expression of
cyclin D1 and CDK4 was reduced in miR-218-overexpressing HeLa and
SiHa cells (Fig. 2B).
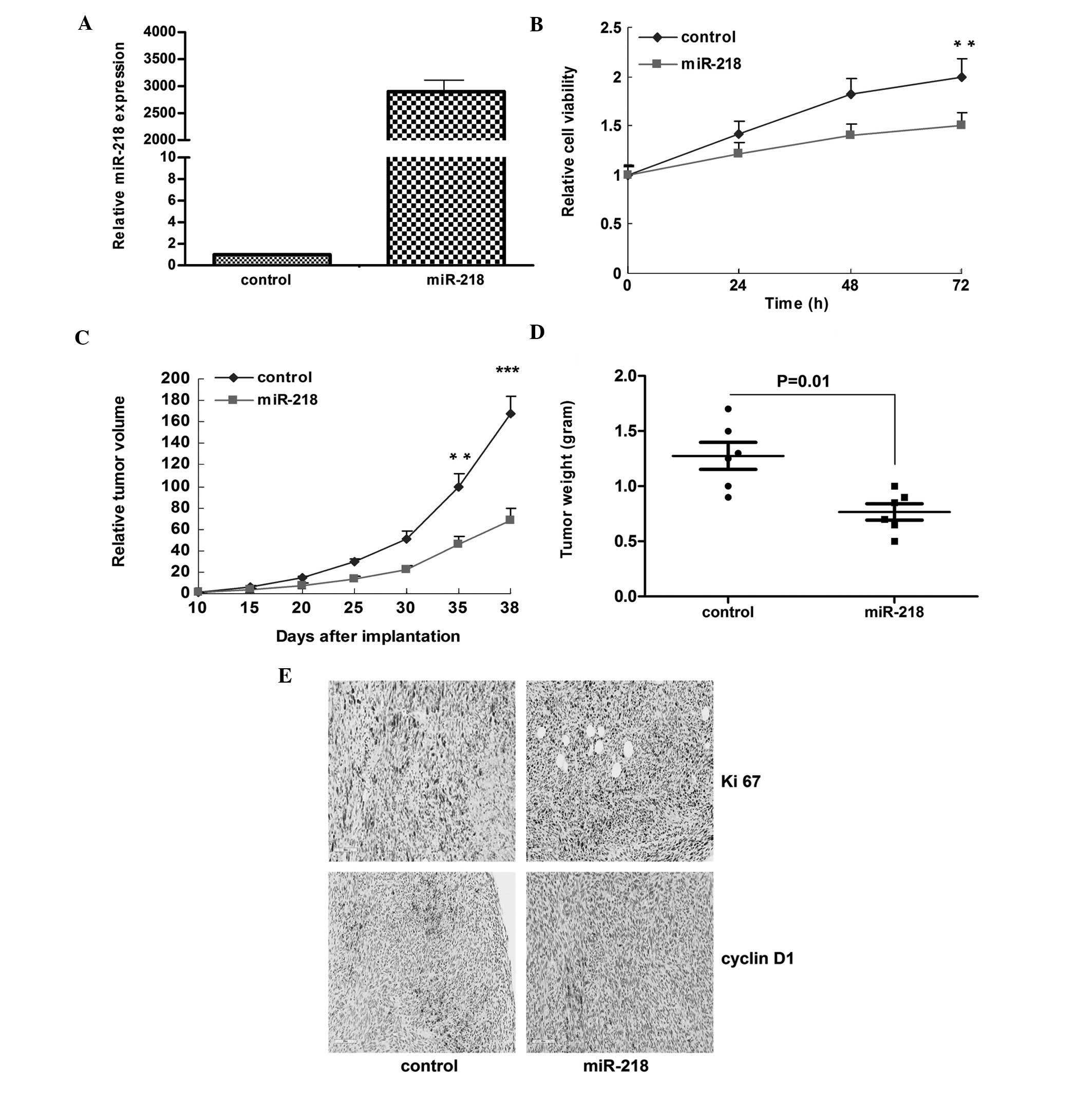
Stable overexpression of miR-218 inhibits
tumor growth in vivo
By transfection of SiHa cells with the
miR-218-expressing plasmid, we stably overexpressed miR-218, as
much as 3,000-fold (Fig. 3A). The
growth of the transfected SiHa cells was significantly reduced
(Fig. 3B, P<0.01), similarly to
the transient transfection experiments. In the mouse model
established by injection of the miR-218-transfected SiHa cells,
growth of the xenograft was also impaired (Fig. 3C, P<0.001); moreover, the tumor
weight in these mice was reduced compared to the control group
(Fig. 3D, P=0.01). Overexpression
of miR-218 also inhibited the expression of Ki 67 (a marker of
proliferation) and cyclin D1 (Fig.
3E).
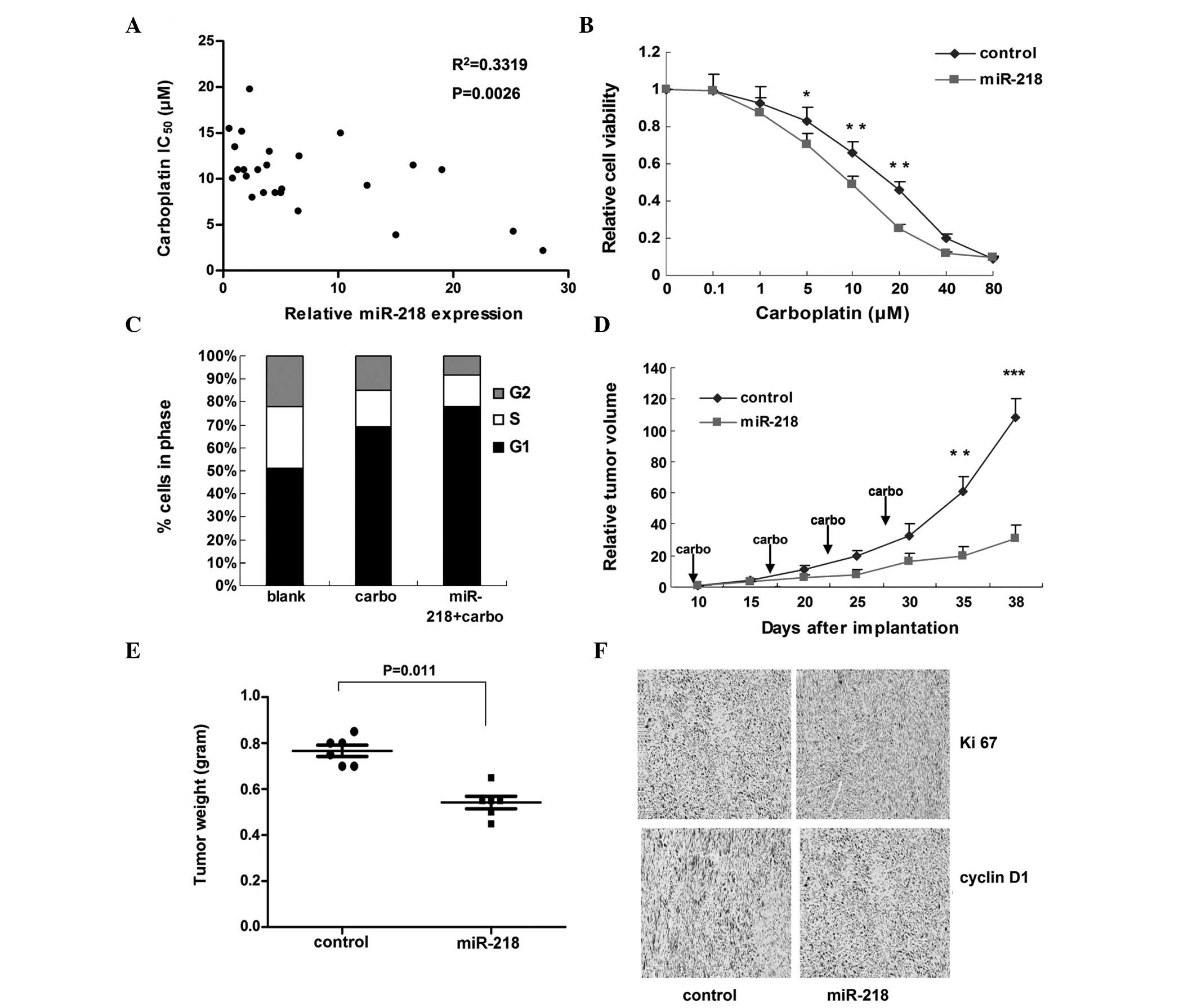
miR-218 expression correlates to tumor
sensitivity to carboplatin in primary cultured cervical cancer
samples
As shown in Fig.
4A, reduced expression of miR-218 was associated with cervical
cancer cell resistance to carboplatin, and as the level of miR-218
increased, the half maximal inhibitory concentration
(IC50) of carboplatin (72-h treatment) gradually
decreased. The correlation between miR-218 expression and
IC50 was significant (R2=0.3319,
P=0.0026).
Restoration of miR-218 sensitizes SiHa
cells to carboplatin
In vitro, miR-218 overexpression increased
cellular sensitivity to carboplatin and induced cell-cycle arrest
(Fig. 4B and C). In vivo,
tumor growth in the group overexpressing miR-218 was markedly lower
compared to the control group, where miR-218 expression was lower
(Fig. 4D, P<0.001), while tumor
weight was also reduced (Fig. 4E,
P=0.011). Carboplatin treatment further reduced the expression of
Ki 67 and cyclin D1 when combined with overexpression of miR-218
(Fig. 4F).
Discussion
Uncontrolled tumor growth and chemo-/radio-
resistance remain the leading causes of cancer-related death,
especially in cervical cancer, where patients often experience
tumor recurrence and poor outcome (19). Thus, valid approaches to suppress
tumor growth and improve cellular sensitivity to routinely used
therapeutic agents are urgently needed. The group of small,
single-stranded nucleotide chains, microRNAs, has been demonstrated
to be a potent tool to target the genes involved in tumor growth
and reactions to drugs (20). For
instance, SPC3649, a miR-122 antagonist, is currently being
evaluated in a phase II clinical trial (NCT01872936) and may
constitute the first commercial microRNA-based drug (21). Another example is MRX34, a drug
designed to mimic the activity of human miR-34, now under phase I
clinical trials (NCT01829971) (22).
We previously reported that loss of miR-218 relates
to late tumor stage and lymphatic node metastasis, indicating the
roles of miR-218 in the progression of cervical cancer. We now
investigated the relationship between the miR-218 level and tumor
sensitivity to carboplatin, as well as the effects of miR-218
restoration in cervical cancer progression.
miR-218 has been shown to participate in numerous
signaling pathways through which it can suppress tumor growth,
migration and invasion, such as IKK-β, NF-κB, Slits-Robo, survivin
and PI3K/AKT (9,23,24).
Using two transfection protocols, we upregulated miR-218 in four
human cervical cancer cell lines, and in both series of
experiments, cellular growth and cell-cycle progression were
significantly inhibited. In vivo, overexpression of miR-218
also significantly suppressed the growth of SiHa xenografts. Our
findings are supported by previous studies on nasopharyngeal cancer
(14), gastric carcinoma (15)and other malignancies (12,13).
For cervical cancer patients at the late stage,
platinum-based chemotherapy is commonly combined with surgery or
radiotherapy. Since severe dose-dependent toxicity is frequently
observed, oncologists nowadays opt for weekly chemotherapy, which
has been demonstrated to be more efficient and have limited
side-effects (25,26). In this study, we found that the low
miR-218 level correlates to resistance to carboplatin in the
primary cultured human cervical cancer samples, which was
consistent with the data observed in the mouse model. Tumors where
miR-218 expression was restored were more sensitive to carboplatin
treatment than those with relatively reduced expression of miR-218,
which indicates that upregulating miR-218 may be a promising
strategy for patients who both show low expression of miR-218 and
resistance to carboplatin. Apart for its suppressive effects on
tumor proliferation (as shown by the Ki 67 IHC detection), we also
found the miR-218 overexpression or miR-218 overexpression combined
with carboplatin treatment significantly inhibits cell cycle
progression, in agreement with a previous study (27).
In other malignancies, microRNAs such as miR-193b
and miR-21, were proved to predict tumor sensitivity to
chemotherapy (28,29). Here, we demonstrated that miR-218
expression correlates to carboplatin sensitivity. Moreover, we
showed that restoration of miR-218 allows to overcome carboplatin
resistance when miR-218 expression is low. In the near future, we
will focus on development of miR-218 as a predictive marker for
resistance to chemotherapy in cervical cancer.
In summary, based on our previous and the present
findings, we conclude that restoration of the reduced miR-218 level
in the cells can significantly suppress the growth of cervical
cancer, potentially via the induction of cell-cycle arrest;
furthermore, we found that both in vitro and in vivo,
the high level of miR-218 can sensitize cervical cancer cells to
carboplatin. Our findings provide strong arguments for the
development of a novel therapy for cervical cancer based on
miR-218, especially for patients with a reduced miR-218 level.
Acknowledgements
This study was supported by grants to Jinjin Yu from
the Wuxi Science and Technology Bureau (no. CSE01N1113), to Yuan
Wang from the Affiliated Hospital of Jiangnan University (no.
SY201305) and from the Health Bureau of Wuxi (MS201417), and to
Guiqiang Du from the Preliminary Plan for Excellent Young Medical
Talent training from Shanghai First People’s Hospital (no.
12RC21).
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2013. CA Cancer J Clin. 63:11–30. 2013. View Article : Google Scholar
|
|
2
|
American Cancer Society. Cancer Facts
& Figures 2011. American Cancer Society; Atlanta: 2011
|
|
3
|
Davis-Dusenbery BN and Hata A: microRNA in
cancer: the involvement of aberrant microRNA biogenesis regulatory
pathways. Genes Cancer. 1:1100–1114. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee JW, Choi CH, Choi JJ, et al: Altered
microRNA expression in cervical carcinomas. Clin Cancer Res.
14:2535–2542. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bartel DP: MicroRNAs: genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
He L and Hannon GJ: MicroRNAs: small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kloosterman WP and Plasterk RH: The
diverse functions of microRNAs in animal development and disease.
Dev Cell. 11:441–450. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lu J, Getz G, Miska EA, et al: MicroRNA
expression profiles classify human cancers. Nature. 435:834–838.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Uesugi A, Kozaki K, Tsuruta T, et al: The
tumor suppressive microRNA miR-218 targets the mTOR component
Rictor and inhibits AKT phosphorylation in oral cancer. Cancer Res.
71:5765–5778. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang B, Pan X, Cobb GP and Anderson TA:
microRNAs as oncogenes and tumor suppressors. Dev Biol. 302:1–12.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tatarano S, Chiyomaru T, Kawakami K, et
al: miR-218 on the genomic loss region of chromosome 4p15.31
functions as a tumor suppressor in bladder cancer. Int J Oncol.
39:13–21. 2011.PubMed/NCBI
|
|
12
|
Wu DW, Cheng YW, Wang J, Chen CY and Lee
H: Paxillin predicts survival and relapse in non-small cell lung
cancer by microRNA-218 targeting. Cancer Res. 70:10392–10401. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Song L, Huang Q, Chen K, et al: miR-218
inhibits the invasive ability of glioma cells by direct
downregulation of IKK-β. Biochem Biophys Res Commun. 402:135–140.
2010.PubMed/NCBI
|
|
14
|
Alajez NM, Lenarduzzi M, Ito E, et al:
MiR-218 suppresses nasopharyngeal cancer progression through
downregulation of survivin and the SLIT2-ROBO1 pathway. Cancer Res.
71:2381–2391. 2011. View Article : Google Scholar
|
|
15
|
Tie J, Pan Y, Zhao L, et al: MiR-218
inhibits invasion and metastasis of gastric cancer by targeting the
Robo1 receptor. PLoS Genet. 6:e10008792010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Yu J, Wang Y, Dong R, Huang X, Ding S and
Qiu H: Circulating microRNA-218 was reduced in cervical cancer and
correlated with tumor invasion. J Cancer Res Clin Oncol.
138:671–674. 2012. View Article : Google Scholar
|
|
17
|
Schmittgen TD and Livak KJ: Analyzing
real-time PCR data by the comparative C(T) method. Nat Protoc.
3:1101–1108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu D, Wu Y, Wang Y, et al: CREPT
accelerates tumorigenesis by regulating the transcription of
cell-cycle-related genes. Cancer Cell. 21:92–104. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Cancer Research UK. Cervical cancer
survival statistics. Let’s Beat Cancer Sooner. http://info.cancerresearchuk.org/cancerstats/types/cervix/survival/.
Accessed, April 2011
|
|
20
|
Nana-Sinkam SP and Croce CM: Clinical
applications for microRNAs in cancer. Clin Pharmacol Ther.
93:98–104. 2013. View Article : Google Scholar
|
|
21
|
Janssen HL, Reesink HW, Lawitz EJ, et al:
Treatment of HCV infection by targeting microRNA. N Engl J Med.
368:1685–1694. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang R, Ma J, Wu Q, et al: Functional role
of miR-34 family in human cancer. Curr Drug Targets. 14:1185–1191.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fish JE, Wythe JD, Xiao T, et al: A
Slit/miR-218/Robo regulatory loop is required during heart tube
formation in zebrafish. Development. 138:1409–1419. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gao C, Zhang Z, Liu W, Xiao S, Gu W and Lu
H: Reduced microRNA-218 expression is associated with high nuclear
factor kappa B activation in gastric cancer. Cancer. 116:41–49.
2009.PubMed/NCBI
|
|
25
|
Leath CA III and Straughn JM Jr:
Chemotherapy for advanced and recurrent cervical carcinoma: results
from cooperative group trials. Gynecol Oncol. 129:251–257. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Scatchard K, Forrest JL, Flubacher M,
Cornes P and Williams C: Chemotherapy for metastatic and recurrent
cervical cancer. Cochrane Database Syst Rev.
10:CD0064692012.PubMed/NCBI
|
|
27
|
He X, Dong Y, Wu CW, et al: MicroRNA-218
inhibits cell cycle progression and promotes apoptosis in colon
cancer by downregulating BMI1 polycomb ring finger oncogene. Mol
Med. 18:1491–1498. 2012.PubMed/NCBI
|
|
28
|
Ziliak D, Gamazon ER, Lacroix B, Kyung Im
H, Wen Y and Huang RS: Genetic variation that predicts platinum
sensitivity reveals the role of miR-193b* in
chemotherapeutic susceptibility. Mol Cancer Ther. 11:2054–2061.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wei J, Gao W, Zhu CJ, et al:
Identification of plasma microRNA-21 as a biomarker for early
detection and chemosensitivity of non-small cell lung cancer. Chin
J Cancer. 30:407–414. 2011. View Article : Google Scholar : PubMed/NCBI
|