Introduction
Hepatocellular carcinoma (HCC) is a highly prevalent
type of cancer, with high incidence and mortality rates worldwide
(1). Although the current clinical
management strategies, involving surgical resection, local ablation
or liver transplantation, are able to cure a minority of cases in
the early stages of HCC, the majority of patients are clinically
diagnosed at advanced stages (2).
HCC is not sensitive to conventional cytotoxic agents. Previously,
the concept of targeted systemic therapies has been applied to the
treatment of HCC. Sorafenib, a tyrosine protein kinase inhibitor,
which selectively inhibits the mitogen-activated protein
kinase/extracellular signal-related kinase signaling pathway, has
been demonstrated to exhibit improved survival rates in patients
with advanced HCC (3,4). This finding indicates that therapy,
which targets metabolic pathways may enhance therapeutic efficacy
and improve the prognosis for patients with HCC.
The arachidonic acid (AA) metabolic pathway has been
recognized to be correlated with the occurrence and development of
various types of cancer. Phospholipase A2 (PLA2) and cyclooxygenase
(COX) are rate-limiting enzymes in the AA metabolic pathway, which
are important in the development and progression of a number of
types of cancer (5,6). PLA2 includes three subfamilies:
Cytosolic (c)PLA2, secreted PLA2 and calcium-independent PLA2.
Several studies have demonstrated that the overexpression of cPLA2α
is correlated with angiogenesis and the expression of vascular
endothelial growth factor in human colorectal cancer (7,8). By
contrast, cancer cell proliferation and xenograft tumor growth are
retarded following the suppression of cPLA2α (9,10).
Similarly, COX-2 is regarded as a prognostic predictor for cancer
in the liver, colon, pancreas, breast, prostate and lungs (11,12).
A selective inhibitor of COX-2, celecoxib, is able to induce
apoptosis by reducing the production of prostaglandin E2 (PGE2),
which has been accepted as a potent promoter of cell proliferation,
motility, invasion and angiogenesis (5). Accordingly, the development of drugs,
which act on the AA pathway may provide a novel avenue in
identifying chemotherapeutic agents for the treatment of HCC.
Berberine
(2,3-methylenedioxy-9,10-dimenthoxyprotoberberine chloride; BBR) is
an isoquinoline alkaloid, purified from Berberis species,
which has long been used as an anti-diarrhea drug in
gastrointestinal disorders in traditional Chinese medicine
(13,14). Previous studies have demonstrated
that BBR has favorable anticancer actions against several types of
tumor, including colon cancer, breast cancer, prostate cancer,
melanoma and HCC (15–17). However, the molecular mechanisms
underlying BBR-induced apoptosis mediated by metabolic pathways
remain to be fully elucidated. In the present study, the inhibitory
effect of BBR was investigated in HCC cell lines and in a
transplanted tumor model in BALB/c mice, in order to elucidate
whether BBR-induced apoptosis is correlated with the AA
pathway.
Materials and methods
Chemicals
A
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide
(MTT), penicillin and streptomycin were purchased from
Sigma-Aldrich (St. Louis, MO, USA). High-glucose Dulbecco's
modified Eagle's medium (DMEM) was obtained from Gibco Life
Technologies (Carlsbad, CA, USA). Fetal bovine serum (FBS), SDS,
dimethyl sulfoxide (DMSO), Tris and Tween-20 were purchased from
Beijing Dingguo Changsheng Biological Technology Co., Ltd.
(Beijing, China). The Bradford Protein Assay kit,
radioimmunoprecipitation assay (RIPA) lysis buffer, Annexin
V-fluorescein isothiocyanate (FITC) Apoptotic Detection kit and
Caspase Activity Assay kit were purchased from Beyotime Institute
of Biotechnology (Haimen, China). The primary antibodies against
apoptosis-inducing factor (AIF; mouse monoclonal; cat. no.
sc-55519), COX-2 (rabbit polyclonal; cat. no. sc-7951), cPLA2
(rabbit polyclonal; cat. no. sc-438) and secondary antibodies
(rabbit anti-mouse IgG-HRP, cat. no. sc-358914; goat anti-rabbit
IgG-HRP, cat. no. sc-2004) were from Santa Cruz Biotechnology, Inc.
(Santa Cruz, CA, USA). Anti-caspase-3 (rabbit polyclonal; cat. no.
9662) and anti-caspase-9 (mouse monoclonal; cat. no. 9508)
antibodies were from Cell Signaling Technology, Inc. (Shanghai,
China). β-actin primary antibody (rabbit monoclonal, cat. no.
1854-1) and GAPDH primary antibody (rabbit monoclonal, cat. no.
2251-1) were obtained from Epitomics, Inc. (Burlingame, CA, USA).
BBR (Purity >99%) was donated by the Northeast Pharmaceutical
Group Co., Ltd. (Shenyang, China) and was dissolved with 0.2% DMSO
culture medium to a final concentration of 200 µM as a stock
solution. All other chemicals and reagents were of analytical
grade.
MTT assay and determination of the half
maximal inhibitory concentration (IC50)
H22, HepG2 and Bel-7404 hepatoma cell lines and
normal hepatic embryo HL-7702 cells, were maintained in DMEM high
glucose medium, supplemented with 10% FBS, 100 U/ml penicillin and
100 µg/ml streptomycin, in an atmosphere of 95% air and 5%
CO2 at 37°C. An MTT assay was performed to assess cell
viability, which was performed in 96-well plates in octuplicate.
The cells were seeded at a density of 5×103 cells/well
overnight, and treated with BBR at final concentrations of 0, 12.5,
25, 50 and 100 µM for 24, 48 or 72 h. Subsequently 20
µl MTT (5 mg/ml) was added to each well for the final 3 h of
the 24, 48 or 72 h BBR treatment periods. Following this, the cell
supernatants were discarded, the MTT crystals were dissolved with
DMSO and the optical density (OD) was measured at 490 nm wavelength
(3360063; Tecan Austria GmbH, Grödig, Austria). The ratio of cell
proliferation to control group was calculated from the MTT data.
IC50 values of BBR were calculated from the percentages
of cell viability obtained from the MTT assay.
Apoptosis detection using flow
cytometry
Cell death and apoptosis were detected using an
Annexin V-FITC Apoptotic Detection kit by flow cytometry (BD
Biosciences, Franklin Lakes, NJ, USA). In brief, following
treatment with 0, 50 and 100 µM BBR at 37°C for 24 h, the
HepG2 cells plated in six-well plates at a density of
5×106 cells/well were harvested and washed twice with
cold phosphate-buffered saline. Following this, the cell pellets
were suspended with binding buffer (Beyotime Institute of
Biotechnology) at a density of 1×l06 cells/ml. Following
the addition of 5 µl FITC-conjugated annexin V to the
suspension, the suspension was incubated for 15 min at 4°C in the
dark, followed by the addition of 5 µl prop-idium iodide
(PI; Beyotime Institute of Biotechnology) for 5 min. The samples
were subsequently analyzed using flow cytometry (BD FACSCalibur; BD
Biosciences).
Western blot analysis
The cells were collected by centrifugation at 12,000
× g for 15 min at 4°C and were then washed twice with cold PBS. The
cell pellets were suspended in 200 µl RIPA lysis buffer for
30 min at 4°C, vortexed every 10 min, then centrifuged at 12,000 ×
g for 15 min at 4°C. The supernatant was collected as total protein
extract. In order to investigate AIF translocalization into the
nucleus, the nuclear protein extract was collected using a Nuclear
and Cytoplasmic Protein Extraction kit (Beyotime Institute of
Biotechnology), according to the manufacturer's instructions. The
protein concentration was measured using a Bradford Protein Assay
kit. The protein extract (50 µg) was aliquoted by 12%
SDS-PAGE and electrophoretically transferred onto polyvinylidene
difluoride membranes (EMD Millipore, Billerica, MA, USA). The blots
were blocked with Tris-buffered saline containing 0.05% Tween-20
and 5% non-fat dried milk for 1 h at room temperature, then they
were incubated with rabbit anti-mouse primary anti-COX-2,
anti-cPLA2, anti-caspase-3, anti-caspase-9 and anti-AIF antibodies
(all 1:1,000). The reaction was incubated overnight at 4°C with
gentle agitation, following which the membranes were incubated with
HRP-conjugated goat anti-rabbit secondary antibody (1:2,000) for 2
h at room temperature with gentle agitation. The membranes were
then washed and the bands were visualized using an Enhanced
Chemiluminescence Western Blotting Detection system (GE Healthcare
Life Sciences, Chalfont, UK).
Determination of the levels of AA and
PGE2
The cells were plated in 96-well plates in RPMI-1640
medium containing 10% (v/v) FBS. When ~70% confluence was reached,
the cells were treated with BBR at concentrations of 0, 12.5, 25,
50 or 100 µM for 24 h. At the end of treatment, the culture
supernatant was collected and centrifuged (10,000 × g, 3 min at
25°C) to remove cells or cell debris. The concentrations of PGE2
and AA in the supernatant were measured using an enzyme immunoassay
(PGE2 EIA and AA monoclonal EIA kits; Cayman Chemical Company, Ann
Arbor, MI, USA), according to the manufacturer's instructions. The
production of PGE2 (pg/ml) was normalized to the cell
counts/well.
H22 transplanted tumor model in mice
A total of 50 male BALB/c mice (6–8 weeks old,
weighing 18–22 g) were purchased from the Experimental Animal
Center of Norman Bethune College of Medicine, Jilin University
(Changchun, China). The mice were housed 5/cage under a 12/12 h
light/dark cycle with ad libitum access to food and water.
The mouse production permit number was SCXK (JI) 2010-0001 and the
usage license was SYXK (JI) 2010-0001. The animal experiments in
the present study were performed in accordance with the Good
Laboratory Practice Guidelines (18) and the experimental protocol was
approved by the Ethics Committee of the Norman Bethune Health
Science Center of Jilin University (Changchun, China). In brief,
H22 cells (2×106) suspended in 0.1 ml PBS were injected
subcutaneously into the right side of the dorsal flank of each
mouse. The mice were randomly divided into the following five
groups, each containing 10 mice: Control group, positive control
cyclophosphamide (CTX) group, 12.5 mg/kg BBR group, 25 mg/kg BBR
group and 50 mg/kg BBR group. The mice in the control group were
treated with vehicle (water; 2 ml/kg body weight). Mice in the BBR
groups were administrated with a daily gavage of BBR (dissolved in
water), beginning the day following transplantation of H22 mice
hepatoma tumor cells. The tumor volumes were measured every day,
calculated according to the following formula: Length ×
width2 × 0.52. Body weights were also measured every
day. The mice were sacrificed ~2 weeks subsequent to treatment.
Statistical analysis
All data are expressed as the mean ± standard error
of the mean. The statistical significance of the data was compared
using Student's t-test with SPSS software, version 14.0 (SPSS,
Inc., Chicago, IL, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
BBR inhibits viability of HCC cell lines
in a time- and dose-dependent manner
In order to evaluate the anticancer characteristics
of BBR, the viability of the cells was measured using an MTT assay
following exposure to BBR for different time-periods (24, 48 or 72
h). It was found that BBR significantly reduced cell viability in
mouse model and human hepatoma cell lines in a time- and
dose-dependent manner. BBR also affected the viability of normal
cells folloing longer durations of exposure (48 and 72 h), however,
the IC50 values in the HL-7702 hepatic embryonic cell
line (122.4 µM) at 72 h were markedly higher than the
Bel-7404, H22 and HepG2 HCC cell lines (9.21, 43.2 and 82.8
µM, respectively). The observed IC50 values
indicated that hepatoma cells are more susceptible to BBR, compared
with normal cells (Fig. 1).
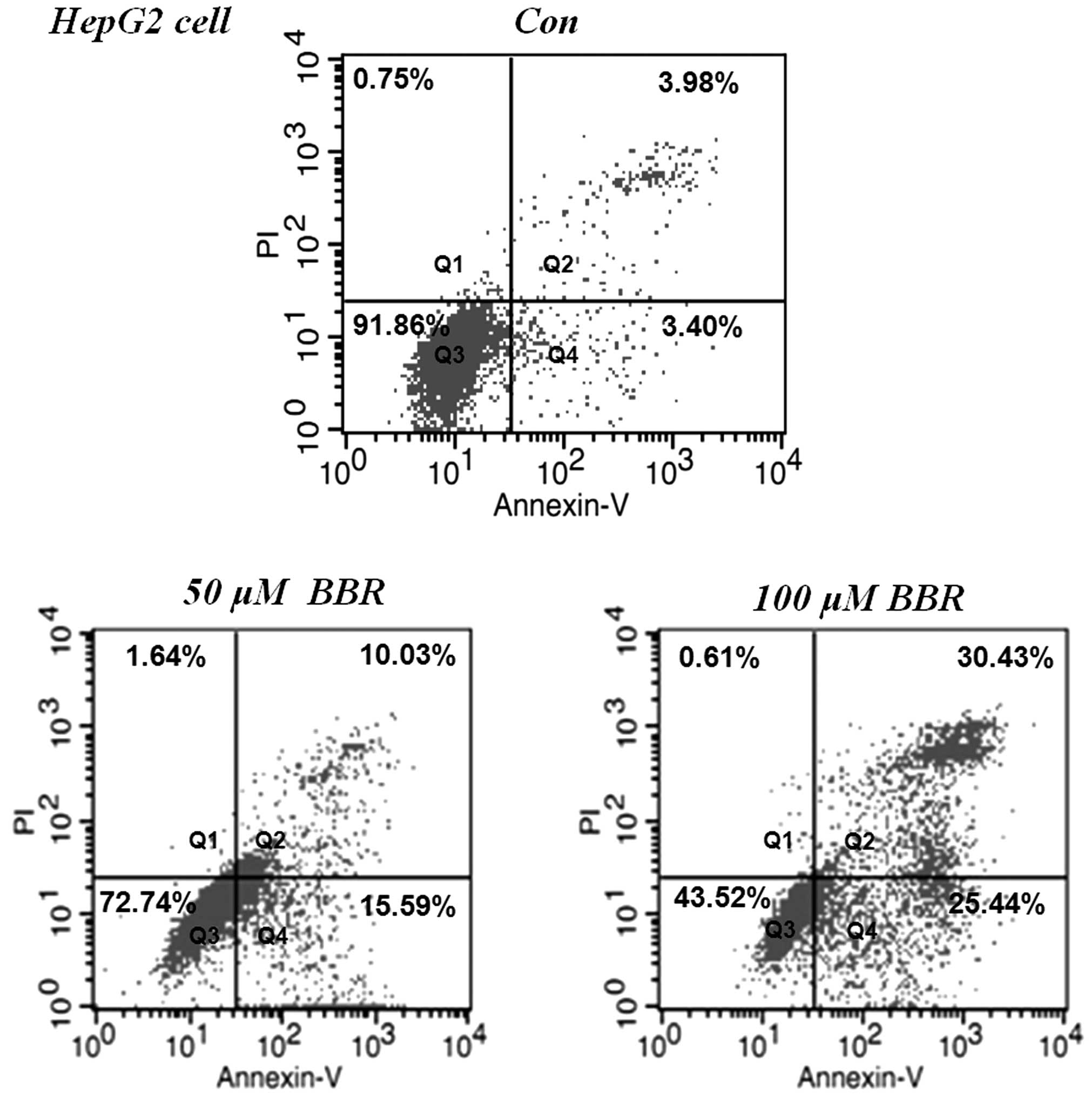
BBR increases apoptosis in HepG2
cells
The HepG2 cells were treated with 50 and 100
µM of BBR for 24 h. Apoptosis was detected using flow
cytometric analysis with annexin V-PI staining. As shown in
Fig. 2, HepG2 cells without BBR
treatment were observed in the quadrant (Q)3, however, BBR
treatment significantly induced early apoptosis, indicated by the
distribution of the cells in Q4, and late apoptotic cells,
indicated by distribution of the cells in Q2, in a dose-dependent
manner. The total apoptotic rates were 24.13±2.14 and 55.55±2.86%
in the 50 and 100 µM BBR treatment groups, respectively.
These results suggested that the BBR-induced apoptosis in the tumor
cells contributed to its anti-proliferative and cytotoxic
efficacies.
BBR induces AIF-dependent apoptosis in
HepG2 cells
In order to elucidate how BBR exerts its effects on
apoptosis in HepG2 cells, the effects of BBR on the intrinsic
apoptotic pathways of caspase-3 and caspase-9 were investigated.
The results demonstrated that the protein levels of total caspase-3
and total caspase-9 were not significantly altered following BBR
treatment. In addition, the no protein expression of cleaved
caspase-3 or cleaved caspase-9 proteins were detected using western
blotting (Fig. 3). Similar results
were observed in the caspase-3 activity assay, which demonstrated
that BBR had no significant effect on the activity of caspase-3
(data not shown). To further examine the apoptotic mechanisms of
BBR, total AIF and nucleic AIF were assessed using western blot
analysis. The results demonstrated that a wide range of BBR
concentrations between 12.5 and 100 µM, induced significant
increases in the protein expression of nucleic AIF, however, the
protein levels of total AIF were not affected. These results
indicated that there is a potential translocation of AIF between
the mitochondria and the nucleus, which is likely to be a pivotal
factor of apoptosis in the BBR-treated HCCs.
BBR affects the AA pathway and increases
the content ratio of AA to PGE2
To investigate how BBR affects the AA metabolic
pathway, two key enzymes (cPLA2 and COX-2) in the pathway were
assessed using western blot analysis. As shown in Fig. 4A, the protein expression levels of
cPLA2 and COX-2 were suppressed significantly in a dose-dependent
manner between concentrations of 12.5–100 µM BBR in the H22
and HepG2 cells. The contents of AA and PGE2 in the culture medium
of the H22 and HepG2 cells treated with BBR for 24 h were then
measured. As shown in Fig. 4B, the
content of AA was significantly increased, and increased as the
dose of BBR increased. The level of PGE2 was significantly reduced,
even with treated with a low dose of BBR (12.5 µM). The
concentration ratio of AA to PGE2 was increased following BBR
treatment in the H22 and HepG2 cells, suggesting that the ratio may
be important in BBR-induced apoptosis in the tumor cells.
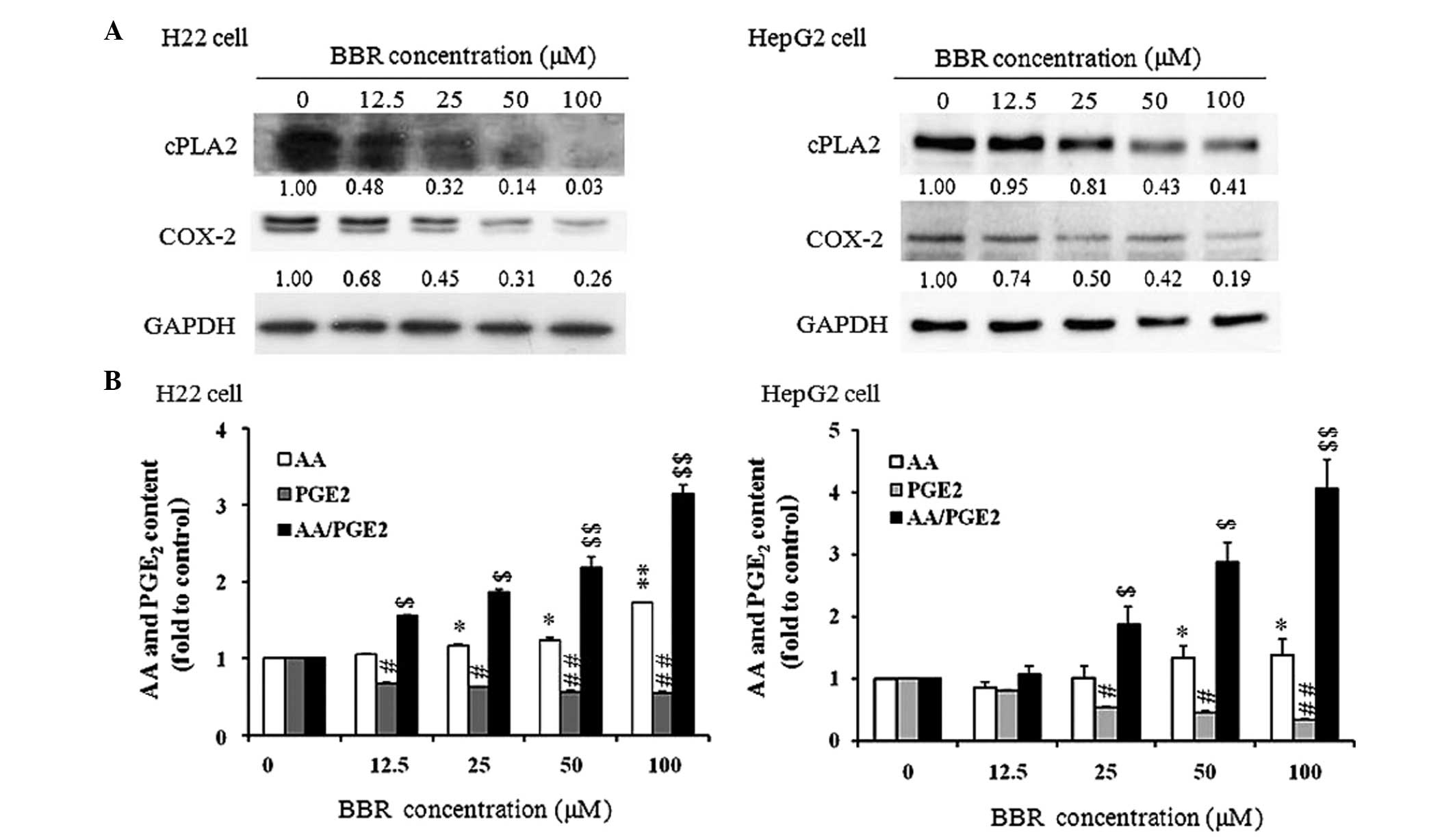 | Figure 4Effect of BBR on the AA metabolic
pathway. (A) Protein expression levels of cPLA2 and COX-2 in the
H22 and HepG2 cells. Cells were treated with 0, 12.5, 25, 50 and
100 µM BBR for 24 h. The numbers below the bands indicate
the relative density ratio of each protein, normalized by the
internal control (GAPDH). The experiments were performed three
times and produced similar results. (B) Ratio of AA to PGE2 in the
culture medium of H22 and HepG2 cells. The cells were treated with
BBR at concentrations of 0, 12.5, 25, 50 or 100 µM for 24 h.
At the end of treatment, the culture supernatant was collected and
used to measure the content of PGE2 and AA using an enzyme
immunoassay, according to the manufacturer's instructions. The
values of PGE2, AA and the ratio of AA/PGE2 in the control group
were set as 1 as normalization. Data are expressed as the mean ±
standard error of the mean. *P<0.05 and
**P<0.01, vs. control group for PGE2;
$P<0.05 and $$P<0.01, vs. control group
for AA; #P<0.05 and ##P<0.01, vs.
control group for AA/PGE2. BBR, berberine; AA, arachidonic acid;
cPLA2, cytosolic phospholipase A2; COX-2, cyclooxygenase 2; PGE2,
prostaglandin E2. |
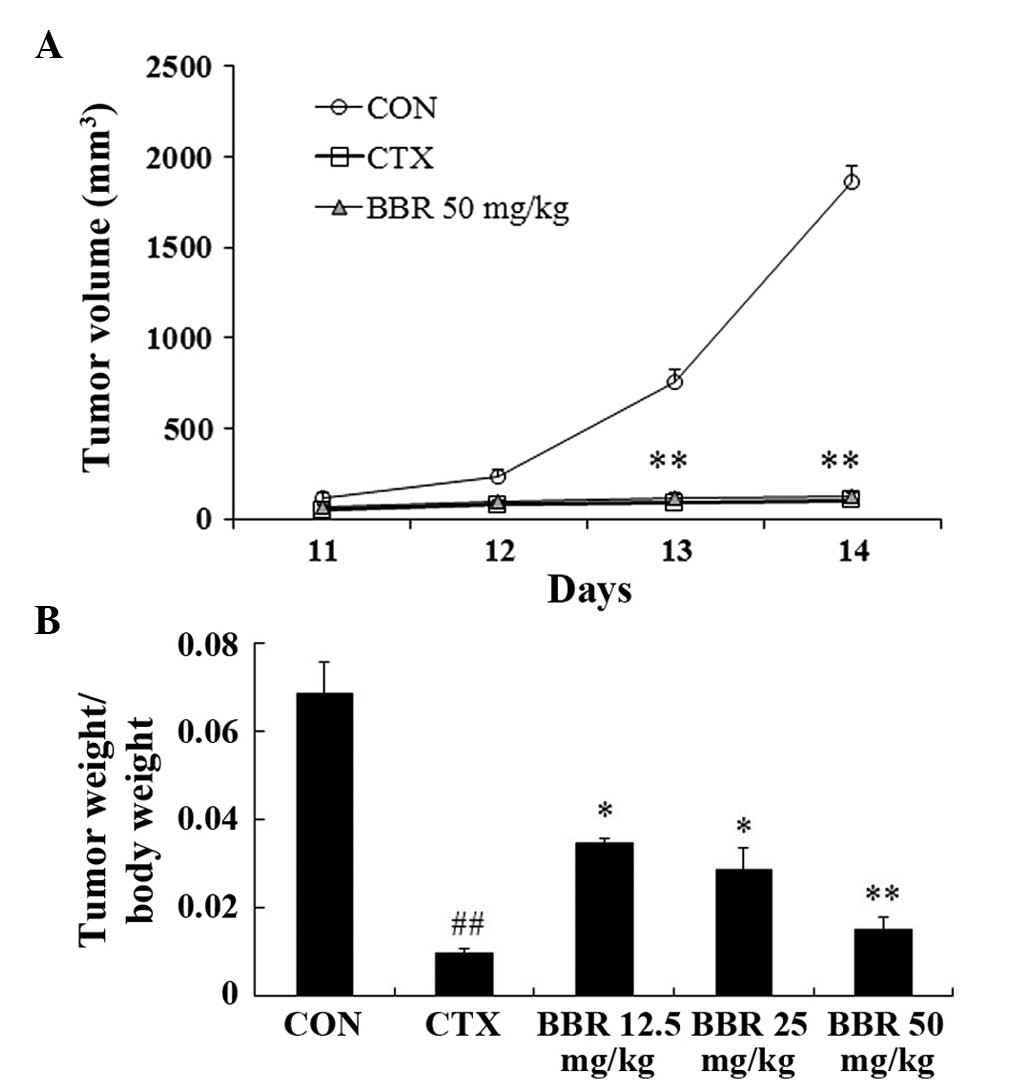
BBR inhibits H22 transplanted tumor
growth
To evaluate the efficacy of BBR on tumor growth
in vivo, a H22 transplanted tumor model was established in
BALB/c mice. As shown in Fig. 5A,
the tumor volume in the control group reached the logarithmic
growth phase 12 days following inoculation, and the same inhibitory
effects were observed in the 50 mg/kg BBR and the CTX positive
control group. Treatment with BBR reduced tumor volume in a
dose-dependent matter (Fig. 5A),
and this was confirmed by the ratios of tumor weight relative to
mouse body weight (Fig. 5B).
Discussion
HCC has higher prevalence and mortality rates,
compared with other types of cancer, including breast, prostate and
stomach cancer (1). Therefore,
prolonging life expectancy and improving quality of life have
become key objectives in the treatment and management of patients
with advanced HCC. The anticancer properties of BBR have been
previously investigated (15,16,19–21).
A key factor for the use of natural BBR as an alternative to the
chemotherapeutic approach is its low cytotoxicity (22). In the present study the effects of
BBR on cell viability and apoptosis were investigated in normal and
cancer cell lines. BBR was found to suppress cell growth in dose-
and time-dependent manner in mouse H22 and in human HepG2 and
Bel-7404 HCC cell lines. This indicated that the inhibitory effect
of BBR on liver tumor was not species-specific, and was associated
with a specific type of HCC cells, as previously reported (22). In the control, the normal HL-7702
hepatic embryo cells exhibited depressed cell viability only in the
higher dose (50–100 µM) BBR and longer (48–72 h) duration
treatment groups, and exhibited higher IC50 values,
compared with all the HCC cell lines. Comparable results of a
previous study indicated that BBR does not significantly affect
cell viability in an immortalized non-tumor cell line, which is
consistent to Chang liver cells, following 48 h treatment (22). The higher IC50 observed
in the normal cells in the present study indicated that BBR
selectively reduces tumor cell viability with lower toxicity in
normal cells. The divergent actions of BBR in the normal and
hepatic cancer cells suggested that BBR may be a practical
alternative therapeutic agent for HCC.
Apoptosis may proceed predominantly through a death
receptor-dependent pathway (extrinsic pathway) or a
mitochondria-dependent pathway (intrinsic pathway) (23). The permeabilization of the outer
mitochondrial membrane and the subsequent release of pro-apoptotic
proteins from the intermembrane space of mitochondria are key
events in caspase-dependent and caspase-independent pathways. AIF
is one of the mitochondrial proteins, which is released into the
cytosol and translocated to the nucleus, where it binds to DNA and
provokes caspase-independent chromatin condensation resulting in
apoptosis (24–26). Therefore, AIF is a reliable
indicator of caspase-independent apoptosis in cells. The results of
the present study demonstrated that the intrinsic mitochondrial
pathway, triggered by the activation of caspase-9 and -3 activation
appear to be independent of BBR-induced apoptosis due to the fact
that the protein expression levels of cleaved caspase-9 and -3 were
undetectable, and that the activity of caspase-3 remained unchanged
following treatment with BBR. This was consistent with reports from
previous studies, which indicated that the caspase pathway was not
significantly activated in HepG2 cells and other cancer cells
following treatment with BBR (19,27).
However, a number of previous studies have presented contradictory
results. Hwang et al (22)
demonstrated increases in the expression levels of cleaved
caspase-3 and -8 in HepG2 cells, and proposed that the potential of
anti-hepatoma activity of BBR may be mediated through a
caspase-dependent pathway. Wang et al (19) reported that the increase of
activity observed in caspase-3, -8 and -9 is involved in HepG2 cell
apoptosis induced by BBR. There is no clear explanation for the
contradictory results at present, which require further
investigation. The results of the present study also demonstrated
that BBR induced the translocation of AIF from the mitochondria to
the nucleus in a dose-dependent manner in the HepG2 cells.
PLA2 and COX-2 in the AA metabolic pathway have been
demonstrated to be important in the development and progression of
various types of cancer (7,9,28,29).
Tumor tissues surgically obtained from patients with HCC have been
previously observed to exhibit significantly higher enzymatic
activities, and protein and mRNA levels of PLA2, compared with
those in surrounding liver tissues or control liver tissues
(30). The activation of cPLA2 may
lead to the accumulation of intracellular AA and/or
lysophospholipid, inducing the alteration of mitochondrial function
(31). However, the released AA is
degraded by COX-2, which is also overexpressed in the development
of cancer (12,32). Consequently, PLA2 and COX-2 affect
the production of AA and PGE2, with the final biological effects
depending on the coupling of the PLA2 and COX-2 pathways and the
combined actions of AA and PGE2. The present study demonstrated
that overexpression of cPLA2 and COX2 in the H22 and HepG2 cells
was significantly depressed by treatment with BBR, and the
production of PGE2 was also reduced in a dose-dependent manner. In
addition, the ratio of AA to PGE2 was consistently elevated in the
two cell lines, indicating that the ratio of pro-apoptotic AA and
anti-apoptotic PGE2 determined the survival of the cancer
cells.
In conclusion, the present study demonstrated that
BBR selectively exerted cytotoxicity towards the HCC cell lines and
suppressed H22 transplanted tumor growth in BALB/c mice. In
addition, BBR induced AIF-mediated apoptosis and increased the
ratio of AA to PGE2, through suppressing the protein expression of
cPLA2 and COX-2, which was involved in the inhibitory effect of BBR
on HCC. These results suggested that BBR may be used as a potent
and alternative chemotherapeutic agent for HCC treatment.
Acknowledgments
This study was supported by the Science and
Technology Support Program of Jilin Province (grant nos.
20130206051YY and 20140203011YY). The majority of the experiments
were performed at the Preclinical Pharmacology R&D Center of
Jilin Province (China).
Abbreviations:
|
BBR
|
berberine
|
|
HCC
|
hepatocellular carcinoma
|
|
AA
|
arachidonic acid
|
|
cPLA2
|
cytosolic phospholipase A2
|
|
PGE2
|
prostaglandin E2
|
|
COX
|
cyclooxygenase
|
|
AIF
|
apoptosis-inducing factor
|
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Gomaa AI and Waked I: Recent advances in
multidisciplinary management of hepatocellular carcinoma. World J
Hepatol. 7:673–687. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Xie B, Wang DH and Spechler SJ: Sorafenib
for treatment of hepatocellular carcinoma: A systematic review. Dig
Dis Sci. 57:1122–1129. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Woo HY and Heo J: Sorafenib in liver
cancer. Expert Opin Pharmacother. 13:1059–1067. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang M, Xu ZG, Shi Z, Shao D, Li O, Li W,
Li ZJ, Wang KZ and Chen L: Inhibitory effect of celecoxib in lung
carcinoma by regulation of cyclooxygenase-2/cytosolic phospholipase
A2 and peroxisome proliferator-activated receptor gamma.
Mol Cell Biochem. 355:233–240. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dan P, Rosenblat G and Yedgar S:
Phospholipase A2 activities in skin physiology and
pathology. Eur J Pharmacol. 691:1–8. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lim SC, Cho H, Lee TB, et al: Impacts of
cytosolic phospholipase A2, 15-prostaglandin dehydrogenase and
cyclooxygenase-2 expressions on tumor progression in colorectal
cancer. Yonsei Med J. 51:692–699. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Tosato G, Segarra M and Salvuccil O:
Cytosolic phospholipase A2{alpha} and cancer: A role in tumor
angiogenesis. J Natl Cancer Inst. 102:1377–1379. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zheng Z, He X, Xie C, Hua S, Li J, Wang T,
Yao M, Vignarajan S, Teng Y, Hejazi L, et al: Targeting cytosolic
phospholipase A2 α in colorectal cancer cells inhibits
constitutively activated protein kinase B (AKT) and cell
proliferation. Oncotarget. 15:12304–12316. 2014.
|
|
10
|
Patel MI, Singh J, Niknami M, et al:
Cytosolic phospholipase A2-alpha: A potential therapeutic target
for prostate cancer. Clin Cancer Res. 14:8070–8079. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Matsubayashi H, Infante JR, Winter J, et
al: Tumor COX-2 expression and prognosis of patients with
resectable pancreatic cancer. Cancer Biol Ther. 6:1569–1575. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Singh B, Berry JA, Shoher A, Ramakrishnan
V and Lucci A: COX-2 overexpression increases motility and invasion
of breast cancer cells. Int J Oncol. 26:1393–1399. 2005.PubMed/NCBI
|
|
13
|
Chen C, Yu Z, Li Y, Fichna J and Storr M:
Effects of berberine in the gastrointestinal tract - a review of
actions and therapeutic implications. Am J Chin Med. 42:1053–1070.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Birdsall TC and Kelly GS: Berberine:
Therapeutic potential of an alkaloid found in several medicinal
plants. Altern Med Rev. 2:94–103. 1997.
|
|
15
|
Li J, Cao B, Liu X, et al: Berberine
suppresses androgen receptor signaling in prostate cancer. Mol
Cancer Ther. 10:1346–1356. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chidambara Murthy KN, Jayaprakasha GK and
Patil BS: The natural alkaloid berberine targets multiple pathways
to induce cell death in cultured human colon cancer cells. Eur J
Pharmacol. 688:14–21. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhang M, Xu ZG, Shi Z, et al: Inhibitory
effect of celecoxib in lung carcinoma by regulation of
cyclooxygenase-2/cytosolic phospholipase A2 and peroxisome
proliferator-activated receptor gamma. Mol Cell Biochem.
355:233–240. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Good Laboratory Practice for Drugs. China
Food and Drug Administration; Beijing: 1999
|
|
19
|
Wang L, Liu L, Shi Y, et al: Berberine
induces caspase-independent cell death in colon tumor cells through
activation of apoptosis-inducing factor. PLoS One. 7:e364182012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kuo HP, Chuang TC, Yeh MH, et al: Growth
suppression of HER2-overexpressing breast cancer cells by berberine
via modulation of the HER2/PI3K/Akt signaling pathway. J Agric Food
Chem. 59:8216–8224. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Patil JB, Kim J and Jayaprakasha GK:
Berberine induces apoptosis in breast cancer cells (MCF-7) through
mitochondrial-dependent pathway. Eur J Pharmacol. 645:70–78. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Hwang JM, Kuo HC, Tseng TH, Liu JY and Chu
CY: Berberine induces apoptosis through a mitochondria/caspases
pathway in human hepatoma cells. Arch Toxicol. 80:62–73. 2006.
View Article : Google Scholar
|
|
23
|
Goldar S, Khaniani MS, Derakhshan SM and
Baradaran B: Molecular mechanisms of apoptosis and roles in cancer
development and treatment. Asian Pac J Cancer Prev. 16:2129–2144.
2015.PubMed/NCBI
|
|
24
|
Fabregat I, Roncero C and Fernández M:
Survival and apoptosis: A dysregulated balance in liver cancer.
Liver Int. 27:155–162. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Norberg E, Orrenius S and Zhivotovsky B:
Mitochondrial regulation of cell death: Processing of
apoptosis-inducing factor (AIF). Biochem Biophys Res Commun.
396:95–100. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Candé C, Vahsen N, Garrido C and Kroemer
G: Apoptosis-inducing factor (AIF): Caspase-independent after all.
Cell Death Differ. 11:591–595. 2004.PubMed/NCBI
|
|
27
|
Tan YL, Goh D and Ong ES: Investigation of
differentially expressed proteins due to the inhibitory effects of
berberine in human liver cancer cell line HepG2. Mol Biosyst.
2:250–258. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dan P, Rosenblat G and Yedgar S:
Phospholipase A2 activities in skin physiology and
pathology. Eur J Pharmacol. 691:1–8. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Singh T, Vaid M, Katiyar N, Sharma S and
Katiyar SK: Berberine, an isoquinoline alkaloid, inhibits melanoma
cancer cell migration by reducing the expressions of
cyclooxygenase-2, prostaglandin E2 and prostaglandin
E2 receptors. Carcinogenesis. 32:86–92. 2011. View Article : Google Scholar
|
|
30
|
Ying Z, Tojo H, Komatsubara T, Nakagawa M,
Inada M, Kawata S, Matsuzawa Y and Okamoto M: Enhanced expression
of group II phospholipase A2 in human hepatocellular carcinoma.
Biochim Biophys Acta. 1226:201–205. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Dymkowska D, Szczepanowska J, Wieckowski
MR and Wojtczak L: Short-term and long-term effects of fatty acids
in rat hepatoma AS-30D cells: The way to apoptosis. Biochim Biophys
Acta. 1763:152–163. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Greenhough A, Smartt HJ, Moore AE, et al:
The COX-2/PGE2 pathway: Key roles in the hallmarks of cancer and
adaptation to the tumour microenvironment. Carcinogenesis.
30:377–386. 2009. View Article : Google Scholar : PubMed/NCBI
|