Introduction
Gastric cancer is the second most frequent cause of
cancer-related mortality worldwide, and the prognosis for gastric
cancer remains poor, with a five-year overall survival of just
5–15% (1). The chemotherapeutic
agents commonly administered in gastric cancer treatment include
5-fluoro-uracil (5-FU), docetaxel, cisplatin, and Adriamycin
(2,3), and the response rates to combination
regimens using these therapeutic agents are 20–50%, with a median
survival of 6–12 months (4).
However, chemotherapeutic options have become restricted due to
drug resistance and severe cellular toxicity (5). Thus, there is a continuing
requirement for novel therapeutic strategies to address drug
resistance and toxicity, including molecularly targeted agents.
5-FU is one of the most important agents for
gastrointestinal types of cancer. There are two signaling pathways
by which 5-FU exerts antitumor effects; abnormal RNA processing and
inhibition of DNA synthesis. 5-FU is catabolized to
dihydroflu-orouracil by dihydropyrimidine dehydrogenase (DPD), the
first and rate-limiting enzyme of its metabolic pathway. Up to 80%
of administered 5-FU is broken down by DPD in the liver (6). In tumor cells, 5-FU is functionally
converted to 5-fluorodeoxyuridine monophosphate, which forms a
tight covalent complex with thymidylate synthase (TS), the DNA
de novo synthesizing enzyme, in the presence of the folate
cofactor 5–10-methylene tetrahydrofolate (7). This complex blocks the conversion of
deoxyuridine monophosphate (dUMP) to thymidine monophosphate (dTMP)
and thus inhibits DNA synthesis (8). As an essential step in the
biosynthesis, TS catalyses the methylation of dUMP to dTMP, and a
decrease in TS levels within tumor cells blocks DNA synthesis in
the dividing cells (9). The
expression levels of TS in tumors have been reported as an
important indicator of chemosensitivity to 5-FU (10). Thus, pharmacogenetic variability in
5-FU-associated enzymes, such as TS, may be a major determinant for
treating gastrointestinal cancer patients with 5-FU (11).
Over the past 50 years, despite there being
advantages of 5-FU treatment, its clinical application has become
limited by drug resistance. The overall single-agent response rate
for agents, such as 5-FU, doxorubicin and methotrexate alone, in
gastric carcinoma patients remains at just 10–30% (12), and the combination of 5-FU with
other antitumor therapeutic agents has marginally improved the
response rates to 20–50%, with a median survival time of 6–12
months (13,14). The development of resistance is a
major problem in the administration of therapeutic agents, such as
5-FU, and limits their clinical utility (15). Thus, there is an urgent requirement
to establish novel treatments for gastric cancer therapy, and
resistance reversal may be achieved using combination treatments
comprising existing and/or novel agents to provide an important
strategy for treating gastric carcinoma.
Corosolic acid (CRA), a triterpenoid also known as
2α-hydroxyursolic acid, is found in numerous medicinal plants, such
as Lagerstroemia speciosa (banaba), Vaccinium
macrocarpon (cranberry), and Weigela subsessilis
(16–18). These plants are native to Asia,
although CRA has also been isolated from European and South
American plants (19). CRA
exhibited antidiabetic activities in animal studies and clinical
trials, including improvement of glucose metabolism by reducing
insulin resistance in KK-Ay diabetic mice, and by lowering
postchallenge plasma glucose levels in vivo, in humans
(20,21). CRA has demonstrated therapeutic
value due its biological activities, including anti-inflammatory
activity (22), as well as
anti-obesity (23) and
anti-atherosclerosis (24)
effects.
Additionally, CRA displays anticancer activities
against various human cancer cell lines. CRA induces apoptosis via
a mitochondrial signaling pathway in HeLa (cervix adenocarcinoma)
and MG-63 cells (osteosarcoma), and causes adenosine
monophosphate-activated protein kinase activation in SNU-601 cells
(stomach carcinoma) (18,25,26)
and HER2-downregulated cell cycle arrest in NCI-N87 cells (stomach
carcinoma) (27). Furthermore, CRA
causes suppression of the M2 polarization of macrophages and cell
proliferation, by inhibiting signal transducer and activator of
transcription 3 and nuclear factor κ-light-chain-enhancer of
activated B cells activation in glioblastoma cell lines (28), as well as exerting
immunosuppressive activity in myeloid-derived suppressor cells in a
murine sarcoma model (29).
However, the anticancer mechanism of CRA remains poorly
understood.
In the present study, the additive anticancer
activities of CRA are demonstrated in 5-FU-induced SNU-620 SNU-620
human gastric carcinoma cells, and the underlying mechanism is
described.
Materials and methods
Materials
RPMI-1640, fetal bovine serum (FBS) and
penicillin/streptomycin were obtained from Gibco Life Technologies
(Carlsbad, CA, USA). Trypsin/EDTA was purchased from GE Healthcare
Life Sciences (Logan, UT, USA). The following primary antibodies
were used: Rabbit polyclonal anti-human caspase-3 (1:1,000; no.
9662), rabbit polyclonal anti-human poly-(ADP-ribose) polymerase
(PARP) (1:1,000; no. 9542), rabbit polyclonal anti-human Bcl-2
(1:1,000; no. 2876), rabbit polyclonal anti-human mTOR (1:1,000;
no. 2972), rabbit polyclonal anti-human phospho-mTOR (1:1,000; no.
2971), rabbit polyclonal anti-human 4E-binding protein 1 (4-EBP1)
(1:1,000; no. 9452), rabbit polyclonal anti-human phospho-4-EBP1
(1:1,000; no. 9455) and rabbit polyclonal anti-human TS (1:1,000;
no. 3766) were purchased from Cell Signaling Technology, Inc.
(Danvers, MA, USA), and rabbit polyclonal anti-human Bim (1:200;
sc-11425) and rabbit polyclonal anti-human GAPDH (1:1000; sc-25778)
were obtained from Santa Cruz Biotechnology, Inc. (Dallas, TX,
USA). Horseradish peroxidase-conjugated anti-mouse and anti-rabbit
antibodies were obtained from Transduction Lab (Lexington, KY,
USA). SuperSignal® West Pico Chemiluminescent Substrate
was purchased from Pierce Biotechnology, Inc. (Rockford, IL, USA)
and 5-FU was provided by Choongwae Pharmaceutical Co., Ltd. (Seoul,
Korea). Cell Counting Kit-8 (CCK-8) was purchased from Dojindo
Laboratories (Kumamoto, Japan) and the Annexin-V-FLUOS Staining kit
was purchased from Roche Diagnostics GmbH (Mannheim, Germany). A
Mitochondrial Apoptosis Staining kit was purchased from
PromoKine® (Heidelberg, Germany). CRA, rapamycin,
resveratrol, Tris base, EDTA and all other reagents were obtained
from Sigma-Aldrich (St. Louis, MO, USA).
Cells
SNU-620 human gastric carcinoma cells were purchased
from the Korean Cell Line Bank (Seoul, Korea). The cells were grown
in RPMI-1640 media supplemented with 10% (v/v) FBS, penicillin (100
U/ml)/streptomycin (100 µg/ml) at 37°C in a 5%
CO2 humidified incubator.
Cell growth inhibition assay
Cells were seeded at 5×103 cells/ml in
96-well microplates and allowed to attach for 24 h. 5-FU (1–80
µg/ml) or CRA (1–100 µM) was added to the medium at
various concentrations. Following treatment, the cell cytotoxicity
and/or proliferation was assessed using the CCK-8 assay. Briefly,
highly water-soluble tetrazolium salt
[2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)
-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt], produced
an orange-colored water-soluble product, formazan. The quantity of
formazan dye generated by dehydrogenases in the cells was directly
proportional to the number of living cells. CCK-8 (10 µl)
was added to each well and incubated for 3 h at 37°C; cell
proliferation and cytotoxicity were assessed by measuring the
absorbance at 450 nm using a microplate reader (Corning, Corning,
NY, USA). Three replicated wells were used per experimental
condition.
Annexin V/propidium iodide (PI)
staining
Cells were cultured in 6-well plates at
106 cells/well in RPMI-1640 medium, which were
pretreated with 5-FU (20 µg/ml) or CRA (50 µM) for 24
h. Cells were centrifuged at 400 ×g for 5 min and washed three
times with phosphate buffered saline (PBS), then the cell pellet
was resuspended in 100 µl Annexin-V-FLUOS labeling solution.
Following a 30-min incubation at room temperature, the samples were
analyzed using a flow cytometer (BD FACSCanto™ II; BD Biosciences,
Franklin Lakes, NJ, USA).
Western blot analysis
Cells were incubated with 5-FU (20 µg/ml) or
CRA (50 µM) for 24 h and washed twice in cold PBS. The cells
were lysed with lysis buffer [10 mM Tris (pH 7.4), 150 mM NaCl, 1
mM EDTA, 1% Triton X-100, 0.5% NP-40, 1 mM PI, 1 mM dithiothreitol,
1 mM phenylmethylsulfonyl fluoride] and placed on ice for 1 h with
occasional vortexing. Centrifugation was then conducted at 13,000
×g for 10 min and each of the cell lysates (50 µg) were
subjected to sodium dodecyl sulfate-polyacrylamide gel
electrophoresis and transferred to polyvinylidene difluoride
membranes (Invitrogen Life Technologies, Carlsbad, CA, USA). The
blots were blocked with 5% skimmed milk in PBS containing 0.05%
Tween-20 for 1 h at 25°C, then incubated with primary antibodies.
This was followed by incubation with anti-rabbit or anti-mouse
horseradish peroxidase-conjugated IgG and the blots were visualized
by enhanced chemiluminescence (Pierce Biotechnology, Inc.).
Results
Effects of 5-FU, CRA and the combination
treatment on the inhibition of cell proliferation in SNU-620 human
gastric carcinoma cells
The present study was initially performed to examine
whether 5-FU or CRA inhibited the growth of SNU-620 human gastric
carcinoma cells. The effects of these compounds on cell
proliferation were measured using the CCK-8 assay. Cells were
treated with 5-FU (1, 5, 20 and 80 µg/ml) or CRA (1, 10, 25,
50 and 100 µM), and following a 24-h exposure, growth
inhibition was measured and compared with the untreated control
cells. A concentration-dependent inhibition effect on cell growth
was observed (Fig. 1A and B). To
assess whether there was an additive effect, proliferation was
observed in the SNU-620 cells that had been exposed to 5-FU (20
µg/ml) and CRA (10, 25, 50, 75 and 100 µM) in
combination (Fig. 1C). A combined
treatment with 20 µg/ml 5-FU and 50 µM CRA resulted
in reduced growth of SNU-620 human gastric carcinoma cells compared
with that of 5-FU treatment alone; a 55.8% reduction in
proliferation was observed. No significant difference was
identified in the antiproliferative effect of the combined
treatments at the highest CRA concentrations (75 and 100
µM).
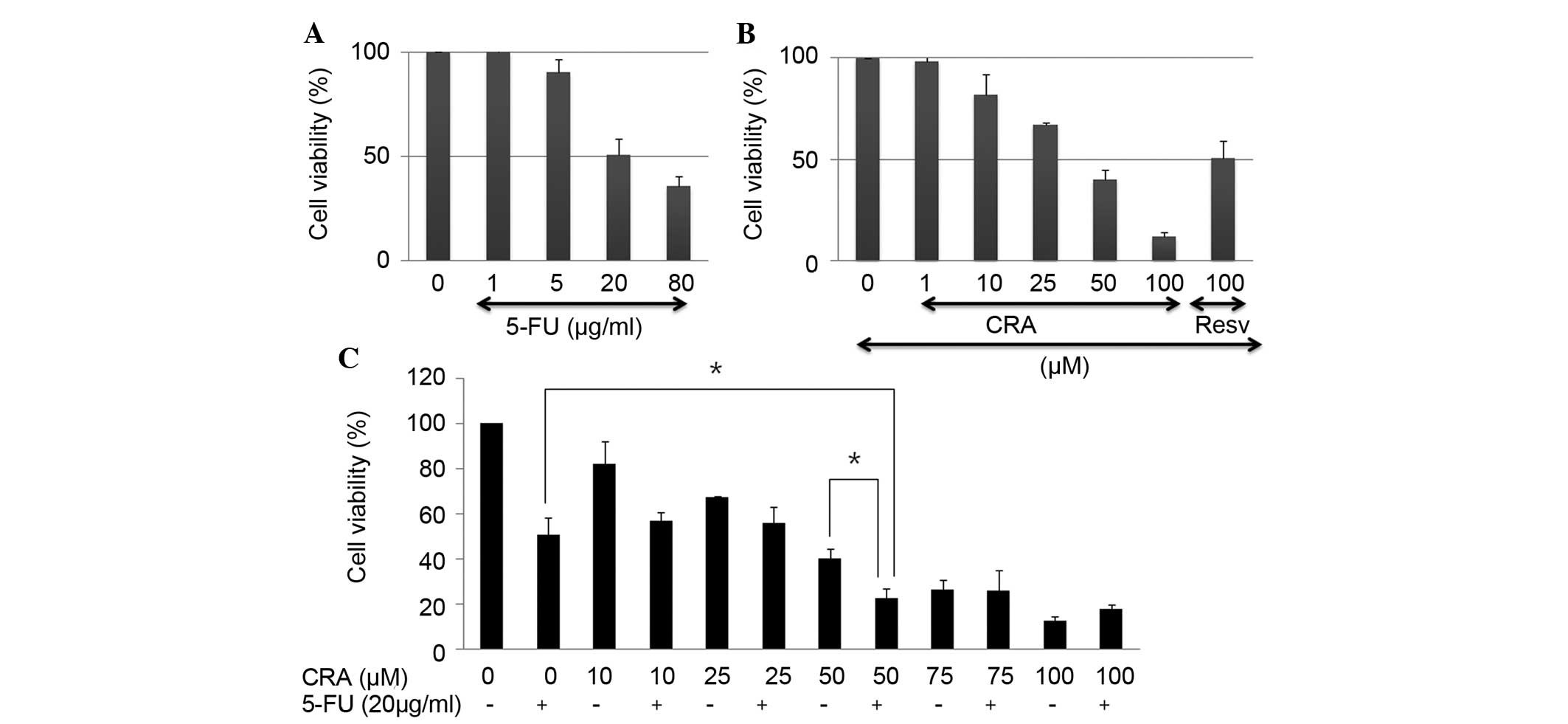 | Figure 1Antiproliferative activity of 5-FU,
CRA, or a combination treatment in SNU-620 human gastric carcinoma
cells. Cells were treated with increasing concentrations of either
(A) 5-FU (1, 5, 20 and 80 µg/ml) or (B) CRA (1, 10, 25, 50
and 100 µM) for 24 h. Cell viability was determined using a
Cell Counting Kit-8 assay measuring absorbance at 450 nm using a
96-well plate reader. The data are presented as the mean ± standard
deviation of three independent experiments. Resv (100 µM)
served as a control. (C) Cells were exposed to 20 µg/ml 5-FU
with CRA (10–100 µM) for 24 h. All data are reported as the
percentage change in comparison with the untreated control group,
which was arbitrarily assigned 100% viability.
*P<0.05 compared with either of the single
therapeutic agent-treatment groups. 5-Fu, 5-fluorouracil; CRA,
corosolic acid; Resv, resveratrol. |
Apoptosis induced by 5-FU, CRA or the
combination treatment in SNU-620 human gastric carcinoma cells
To investigate whether the combination of 5-FU and
CRA induced apoptotic cell death more effectively than either of
the treatments alone, the DNA contents were analyzed following
Annexin V/PI staining by flow cytometry. Compared with each of the
individual therapeutic agent-treated groups, the apoptotic
proportion was increased markedly in those cells that were treated
with 5-FU and CRA together (Fig.
2A). To determine the molecular basis of apoptotic cell death
induced by 5-FU, CRA, or the combination treatment, the cells were
treated with 20 µg/ml 5-FU and 50 µM CRA for 24 h,
and western blot analysis was performed to access the protein
levels of the following markers of apoptosis: Bcl-2, Bim, caspase-3
and cleaved PARP. The protein level of Bcl-2 (a protein of the
antiapoptotic family) was decreased significantly by the
combination, whereas the protein level of Bim (a protein of the
proapoptotic family) increased. The release of mitochondrial
cytochrome c increased, when compared with that of the
individual treatment groups (Fig.
2B). 5-FU and CRA activated caspase-3 and PARP, a nuclear
protein cleaved by activated caspase-3, and a combination of the
two therapeutic agents further potentiated these activities
(Fig. 2B). These results
demonstrate that CRA enhances 5-FU-induced apoptotic cell death in
SNU-620 human gastric carcinoma cells.
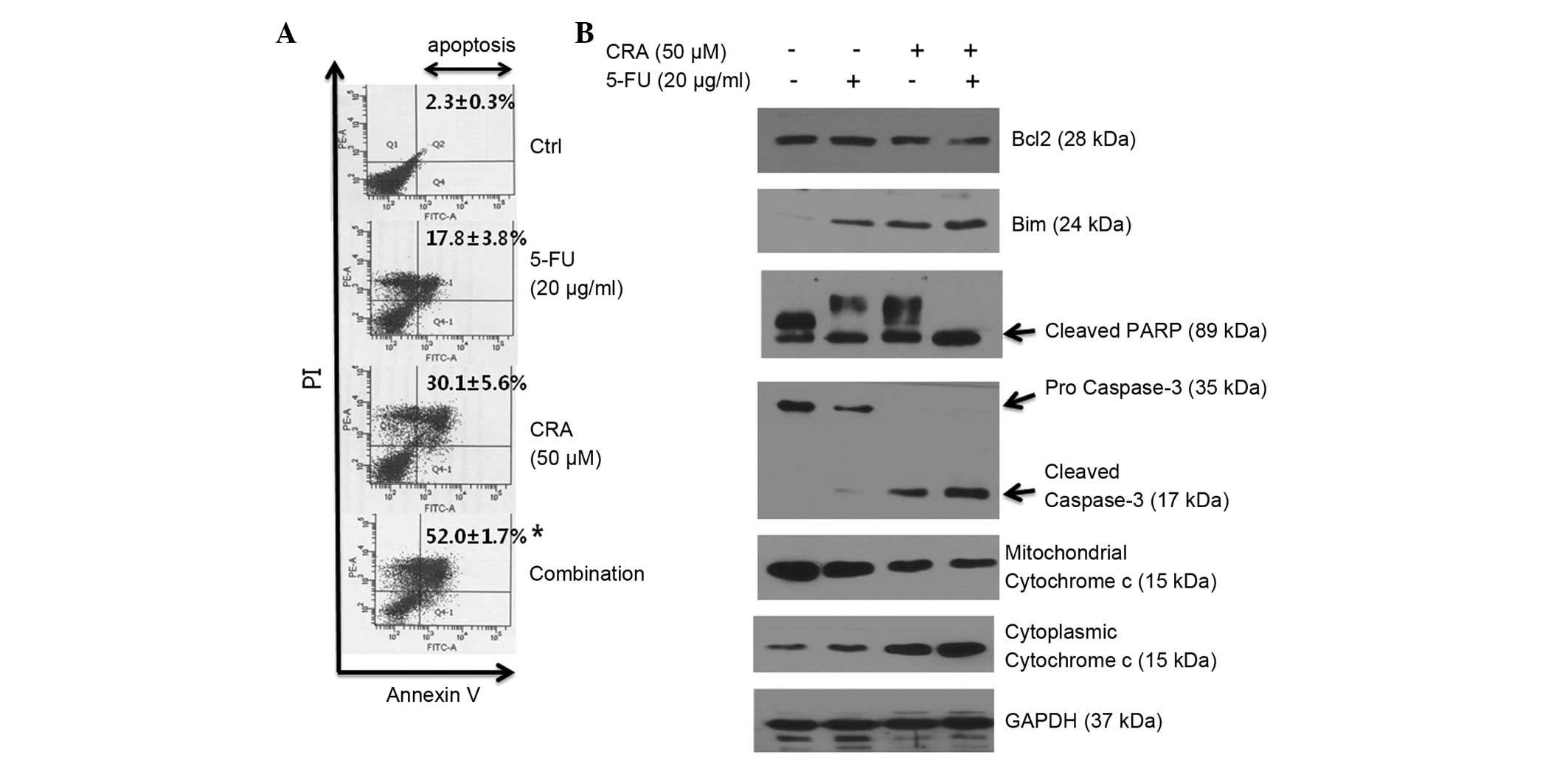 | Figure 2Apoptotic activity in SNU-620 human
gastric carcinoma cells induced by 5-FU, CRA or a combination
treatment. (A) Cells were treated with either 5-FU (20
µg/ml) or CRA (50 µM) or a combination of the two for
24 h, and flow cytometric analysis was performed using Annexin V/PI
staining. Values are presented as the mean of three independent
experiments (mean ± standard deviation). *P<0.05
compared with either of the single therapeutic agent-treatment
groups. (B) The expression of Bcl-2, Bim, PARP, caspase-3 and
cytochrome c proteins was assessed by western blot analysis.
GAPDH expression served as an internal control. PI, propidium
iodide; Ctrl, control; 5-FU, 5-fluorouracil; CRA, corosolic acid;
PARP, poly-(ADP-ribose) polymerase. |
Decreased mTOR/4-EBP1 signaling pathway
activity induced by 5-FU, CRA or the combination treatment in
SNU-620 human gastric carcinoma cells
The mTOR signaling pathway is highly activated in
gastric cancer, and is involved in gastric cancer cell growth and
apoptosis (30,31). In the present study, mTOR
phosphorylation was down-regulated by treatment with 5-FU or CRA
alone (Fig. 3A). The combination
treatment of 5-FU with CRA decreased mTOR/4-EBP1 signaling to a
greater extent, when compared with the individual treatment groups
(Fig. 3B).
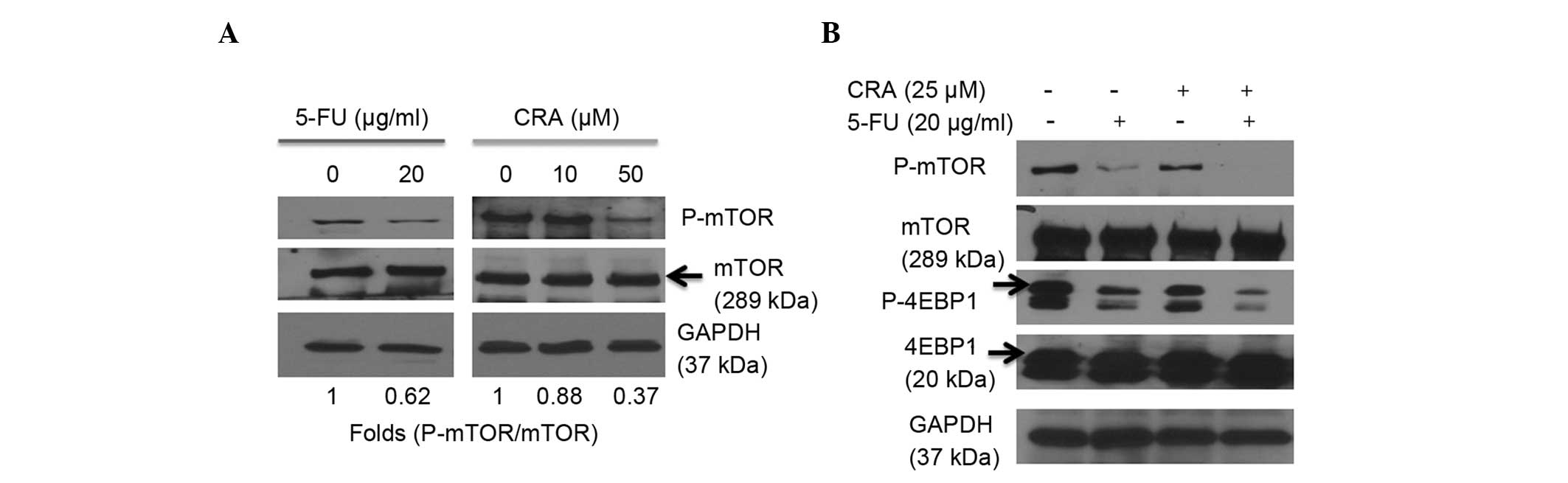 | Figure 3Decreased mTOR/4-EBP1 signaling
induced by 5-FU, CRA or a combination treatment in SNU-620 human
gastric carcinoma cells. Western blot analysis of the cells
following treatment with (A) either 5-FU (20 µg/ml) or CRA
(10 or 50 µM) and (B) a combination treatment (20
µg/ml 5-FU + 25 µM CRA) for 24 h, equal amounts of
protein samples were resolved by sodium dodecyl
sulfate-polyacrylamide gel electrophoresis, followed by
immunoblotting with antibodies against mTOR, P-mTOR, 4EBP1 and
P-4EBP1. GAPDH expression served as an internal control. 5-FU,
5-fluorouracil; CRA, corosolic acid; P, phosphorylated; mTOR,
mammalian target of rapamycin; 4EBP1, 4E-binding protein 1. |
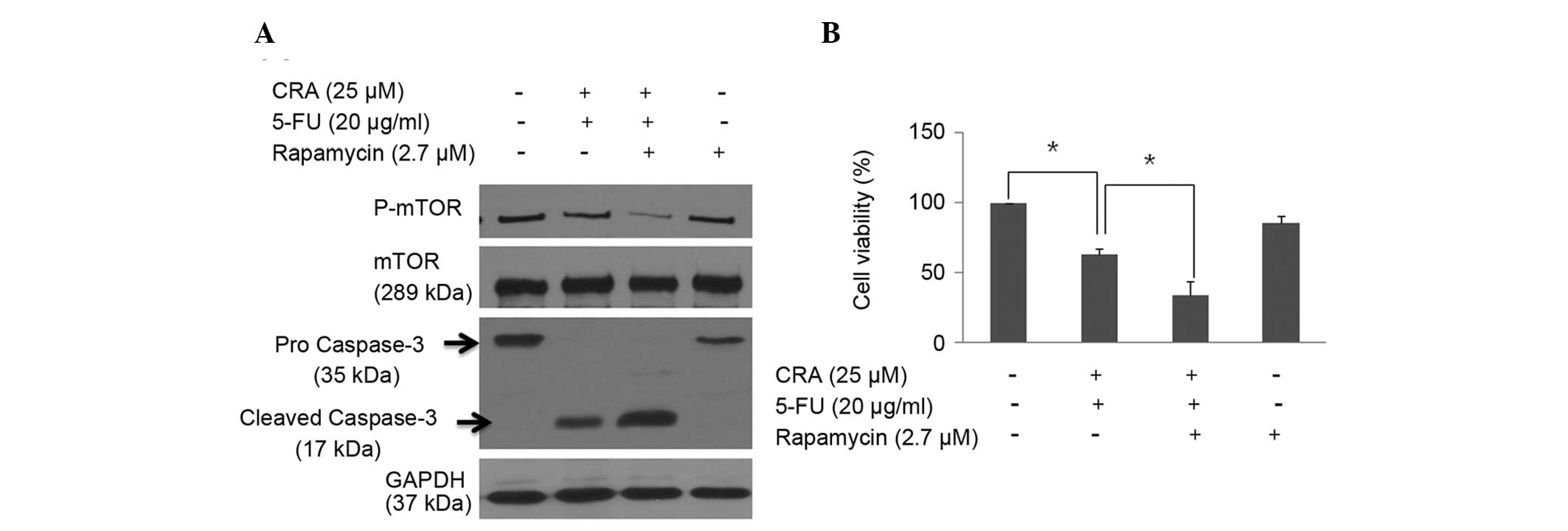
Enhanced antiproliferative and apoptotic
effects of 5-FU in combination with CRA via the mTOR signaling
pathway in SNU-620 human gastric carcinoma cells
To examine whether the apoptosis and
antiproliferative effect of the combination treatment of 5-FU and
CRA involved inhibition of the mTOR signaling pathway, the
phosphorylation levels of mTOR and 4-EBP1 were investigated by
western blot analysis using the mTOR inhibitor, rapamycin (Fig. 4A). The caspase-3 cleavage of the
combination treatment was increased further with rapamycin
(Fig. 4A). In addition, the
antiproliferative effect of the combination treatment of 5-FU and
CRA was enhanced with rapamycin (Fig.
4B). These findings indicate that CRA additively regulates
5-FU-induced apoptosis and viability of SNU-620 cells through the
mTOR signaling pathway.
Discussion
Gastric cancer is the most common cause of
cancer-related mortality in eastern Asia, with a high incidence and
mortality rate. Furthermore, the survival rates of gastric cancer
patients are substantially worse than those of patients with the
majority of other types of cancer (32). Numerous gastric cancer patients
continue to be diagnosed at a late stage, and recurrent tumors are
often detected subsequent to curative surgery. For various clinical
reasons, chemotherapy is an important treatment option. Among the
chemotherapeutic agents, 5-FU is considered to be significant in
the treatment of gastric cancer. Single-agent responses are usually
partial and relatively transient, and toxicity to normal tissues
has been one of the major obstacles to successful cancer
chemotherapy (33). Thus, there
has been increasing focus on the application of combined treatments
using natural products for the treatment of gastric cancer. For
example, thymoquinone, isolated from Nigella sativa seeds
(34) and gambogic acid from
Garcinia hanburyi trees (8)
have been reported to induce anti-gastric cancer effects when
combined with 5-FU. Recent studies have shown that CRA exerts
anticancer activities in various cell types, including gastric
cancer (18,25–29).
However, to the best of our knowledge, the combined
chemotherapeutic effect of 5-FU with CRA and the underlying
biological mechanisms in gastric cancer have not been examined. In
the present study, the additive anticancer activity of 5-FU
combined with CRA in SNU-620 human gastric carcinoma cells was
investigated and CRA was observed to inhibit proliferation in
SNU-620 gastric cancer cells in a dose-dependent manner (Fig. 1B). It has been reported that CRA
exhibits antiproliferative effects in various human gastric cancer
cell lines, such as NCI-N87, SNU-484, and SNU-601, with half
maximal inhibitory concentration (IC50) values for CRA
between 16.9 and 43.7 µM (18,27).
In the current study, in SNU-620 human gastric carcinoma cells, the
IC50 value determined for CRA (40.6 µM) was
within this range. The results revealed that 5-FU-induced
proliferative reduction was synergistically enhanced as a result of
combination treatment with CRA (50 µM; Fig. 1C).
Apoptosis is a tightly regulated signaling process
that involves the coordination of antiapoptotic and proapoptotic
proteins (35). In the present
study, apoptosis in gastric cancer cells induced by 5-FU and CRA
was investigated using Annexin V/PI staining. The results
demonstrated that 5-FU combined with CRA induced apoptosis in
SNU-620 human gastric carcinoma cells more markedly than either
5-FU or CRA alone (Fig. 2A). 5-FU
and CRA combination treatment resulted in a decrease in Bcl-2
expression, an increase in Bim protein expression, and release of
cytochrome c from the mitochondria into the cytoplasm.
Additionally, caspase-3 and PARP activation were observed with 5-FU
and CRA induction in the SNU-620 human gastric carcinoma cells
(Fig. 2B). These results
demonstrate that CRA exposure may potentiate apoptosis induced by
5-FU with mitochondrial dysfunction, and indicates that CRA may be
an effective adjuvant treatment with 5-FU.
mTOR, a serine/threonine kinase protein (290 kDa),
has been considered as a potential target in cancer therapy. mTOR
is a member of the phosphatidylinositol-3-kinase (PI3K) family and
appears to operate downstream of PI3K/Akt (36). Major functions of mTOR include the
activation of p70 S6 kinase (S6K) and inhibition of 4-EBP1.
Activation of S6K leads to translation of ribosomal proteins and
ribosome biogenesis, and the inhibition of 4-EBP1 results in
inhibition of eukaryotic translation initiation factor 4E, as well
as activation of cap-dependent translation of critical mRNAs
(2). The activation of mTOR
results in the control of protein synthesis, metabolism,
proliferation, growth and apoptosis (37). The mTOR inhibitor, rapamycin is an
approved therapeutic agent for preventing allograft rejection in
organ transplantation due to its potent inhibition of T-cell
activation; furthermore, rapamycin exhibits anticancer activity
against various types of cancer (4,30,31).
The mTOR signaling pathway is highly activated in gastric cancers
and presents a promising novel molecular target for cancer therapy;
thus, mTOR inhibitors may act effectively against gastric cancer
cells (30). It has been reported
that mTOR inhibition is necessary to enhance 5-FU-induced apoptosis
in gastric cancer cells (38), and
CRA also regulates mTOR signaling in gastric cancer cells, leading
to cell viability reduction (18).
In the present study, 5-FU in combination with CRA was assessed to
establish whether this treatment combination inhibited cell
viability through the mTOR signaling pathway in gastric cancer
cells. The results indicate that 20 µg/ml 5-FU markedly
decreases mTOR phosphorylation and signaling (Fig. 3A), and the additive activity of
mTOR/4-EBP1 signaling was observed by administering 25 µM
CRA (Fig. 3B). Additional
rapamycin treatment showed increasingly potent inhibition of mTOR
phosphorylation, apoptosis and cell proliferation (Fig. 4A and B). In the present study,
combination chemotherapy using an anticancer drug (5-FU) and a
natural compound (CRA), which exhibited cell signal inhibitory
activity achieved an improved response rate by affecting cell
viability.
In conclusion, the present results demonstrate that
the anticancer effect of 5-FU combined with CRA was more marked
than treatment with 5-FU or CRA alone. It was found that apoptotic
and antiproliferative effects were induced, when the two
therapeutic agents were used in combination, via the mTOR/4-EBP1
signaling pathway. These findings indicate the potential combined
application of these therapeutic agents in adjuvant clinical
treatment of gastric cancer.
Acknowledgments
The present study was supported by the research fund
of Chungnam National University (Daejeon, Korea).
References
|
1
|
Shigematsu H, Yoshida K, Sanada Y, Osada
S, Takahashi T, Wada Y, Konishi K, Okada M and Fukushima M:
Rapamycin enhances chemotherapy-induced cytotoxicity by inhibiting
the expressions of TS and ERK in gastric cancer cells. Int J
Cancer. 126:2716–2725. 2010.
|
|
2
|
Ohtsu A: Chemotherapy for metastatic
gastric cancer: Past, present and future. J Gastroenterol.
43:256–264. 2008. View Article : Google Scholar
|
|
3
|
Ajani JA, Moiseyenko VM, Tjulandin S,
Majlis A, Constenla M, Boni C, Rodrigues A, Fodor M, Chao Y, Voznyi
E, et al V-325 Study Group: Clinical benefit with docetaxel plus
fluorouracil and cisplatin compared with cisplatin and fluorouracil
in a phase III trial of advanced gastric or gastroesophageal cancer
adenocarcinoma: The V-325 Study Group. J Clin Oncol. 25:3205–3209.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee KH, Hur HS, Im SA, Lee J, Kim HP, Yoon
YK, Han SW, Song SH, Oh DY, Kim TY, et al: RAD001 shows activity
against gastric cancer cells and overcomes 5-FU resistance by
downregulating thymidylate synthase. Cancer Lett. 299:22–28. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Benson AB III: New approaches to the
adjuvant therapy of colon cancer. Oncologist. 11:973–980. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Longley DB, Harkin DP and Johnston PG:
5-fluorouracil: Mechanisms of action and clinical strategies. Nat
Rev Cancer. 3:330–338. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sasaki E, Tominaga K, Kuwamura H, Watanabe
T, Fujiwara Y, Oshitani N, Higuchi K and Arakawa T: Synergistic
antitumor effect of combined 5-fluorouracil (5-FU) with
5-chloro-2,4-di-hydroxypyridine on 5-FU-resistant gastric cancer
cells: Possible role of a dihydropyrimidine
dehydrogenase-independent mechanism. J Gastroenterol. 42:816–822.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wang J, Liu W, Zhao Q, Qi Q, Lu N, Yang Y,
Nei FF, Rong JJ, You QD and Guo QL: Synergistic effect of
5-fluorouracil with gambogic acid on BGC-823 human gastric
carcinoma. Toxicology. 256:135–140. 2009. View Article : Google Scholar
|
|
9
|
Parker WB and Cheng YC: Metabolism and
mechanism of action of 5-fluorouracil. Pharmacol Ther. 48:381–395.
1990. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Peters GJ, van der Wilt CL, van Triest B,
Codacci-Pisanelli G, Johnston PG, van Groeningen CJ and Pinedo HM:
Thymidylate synthase and drug resistance. Eur J Cancer.
31:1299–1305. 1995. View Article : Google Scholar
|
|
11
|
Papamichael D: The use of thymidylate
synthase inhibitors in the treatment of advanced colorectal cancer:
Current status. Stem Cells. 18:166–175. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Murad AM, Santiago FF, Petroianu A, Rocha
PR, Rodrigues MA and Rausch M: Modified therapy with
5-fluo-rouracil, doxorubicin and methotrexate in advanced gastric
cancer. Cancer. 72:37–41. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Douillard JY, Cunningham D, Roth AD,
Navarro M, James RD, Karasek P, Jandik P, Iveson T, Carmichael J,
Alakl M, et al: Irinotecan combined with fluorouracil compared with
fluorouracil alone as first-line treatment for metastatic
colorectal cancer: A multicentre randomised trial. Lancet.
355:1041–1047. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chon HJ, Rha SY, Im CK, Kim C, Hong MH,
Kim HR, An JR, Noh SH, Chung HC and Jeung HC: Docetaxel versus
paclitaxel combined with 5-FU and leucovorin in advanced gastric
cancer: combined analysis of two phase II trials. Cancer Res Treat.
41:196–204. 2009. View Article : Google Scholar
|
|
15
|
Choi EJ and Kim GH: 5-Fluorouracil
combined with apigenin enhances anticancer activity through
induction of apoptosis in human breast cancer MDA-MB-453 cells.
Oncol Rep. 22:1533–1537. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Hou W, Li Y, Zhang Q, Wei X, Peng A, Chen
L and Wei Y: Triterpene acids isolated from Lagerstroemia speciosa
leaves as alpha-glucosidase inhibitors. Phytother Res. 23:614–618.
2009. View
Article : Google Scholar
|
|
17
|
Kim E, Sy-Cordero A, Graf TN, Brantley SJ,
Paine MF and Oberlies NH: Isolation and identification of
intestinal CYP3A inhibitors from cranberry (Vaccinium macrocarpon)
using human intestinal microsomes. Planta Med. 77:265–270. 2011.
View Article : Google Scholar :
|
|
18
|
Lee MS, Lee CM, Cha EY, Thuong PT, Bae K,
Song IS, Noh SM and Sul JY: Activation of AMP-activated protein
kinase on human gastric cancer cells by apoptosis induced by
corosolic acid isolated from Weigela subsessilis. Phytother Res.
24:1857–1861. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
19
|
Stohs SJ, Miller H and Kaats GR: A review
of the efficacy and safety of banaba (Lagerstroemia speciosa L.)
and corosolic acid. Phytother Res. 26:317–324. 2012.
|
|
20
|
Miura T, Ueda N, Yamada K, Fukushima M,
Ishida T, Kaneko T, Matsuyama F and Seino Y: Antidiabetic effects
of corosolic acid in KK-Ay diabetic mice. Biol Pharm Bull.
29:585–587. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Fukushima M, Matsuyama F, Ueda N, Egawa K,
Takemoto J, Kajimoto Y, Yonaha N, Miura T, Kaneko T, Nishi Y, et
al: Effect of corosolic acid on postchallenge plasma glucose
levels. Diabetes Res Clin Pract. 73:174–177. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Banno N, Akihisa T, Tokuda H, Yasukawa K,
Higashihara H, Ukiya M, Watanabe K, Kimura Y, Hasegawa J and
Nishino H: Triterpene acids from the leaves of Perilla frutescens
and their anti-inflammatory and antitumor-promoting effects. Biosci
Biotechnol Biochem. 68:85–90. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zong W and Zhao G: Corosolic acid
isolation from the leaves of Eriobotrta japonica showing the
effects on carbohydrate metabolism and differentiation of 3T3-L1
adipocytes. Asia Pac J Clin Nutr. 16:346–352. 2007.PubMed/NCBI
|
|
24
|
Chen H, Yang J, Zhang Q, Chen LH and Wang
Q: Corosolic acid ameliorates atherosclerosis in apolipoprotein
E-deficient mice by regulating the nuclear factor-κB signaling
pathway and inhibiting monocyte chemoattractant protein-1
expression. Circ J. 76:995–1003. 2012. View Article : Google Scholar
|
|
25
|
Xu Y, Ge R, Du J, Xin H, Yi T, Sheng J,
Wang Y and Ling C: Corosolic acid induces apoptosis through
mitochondrial pathway and caspase activation in human cervix
adenocarcinoma HeLa cells. Cancer Lett. 284:229–237. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cai X, Zhang H, Tong D, Tan Z, Han D, Ji F
and Hu W: Corosolic acid triggers mitochondria and
caspase-dependent apoptotic cell death in osteosarcoma MG-63 cells.
Phytother Res. 25:1354–1361. 2011.PubMed/NCBI
|
|
27
|
Lee MS, Cha EY, Thuong PT, Kim JY, Ahn MS
and Sul JY: Down-regulation of human epidermal growth factor
receptor 2/neu oncogene by corosolic acid induces cell cycle arrest
and apoptosis in NCI-N87 human gastric cancer cells. Biol Pharm
Bull. 33:931–937. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Fujiwara Y, Komohara Y, Ikeda T and Takeya
M: Corosolic acid inhibits glioblastoma cell proliferation by
suppressing the activation of signal transducer and activator of
transcription-3 and nuclear factor-kappa B in tumor cells and
tumor-associated macrophages. Cancer Sci. 102:206–211. 2011.
View Article : Google Scholar
|
|
29
|
Horlad H, Fujiwara Y, Takemura K, Ohnishi
K, Ikeda T, Tsukamoto H, Mizuta H, Nishimura Y, Takeya M and
Komohara Y: Corosolic acid impairs tumor development and lung
metastasis by inhibiting the immunosuppressive activity of
myeloid-derived suppressor cells. Mol Nutr Food Res. 57:1046–1054.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lang SA, Gaumann A, Koehl GE, Seidel U,
Bataille F, Klein D, Ellis LM, Bolder U, Hofstaedter F, Schlitt HJ,
et al: Mammalian target of rapamycin is activated in human gastric
cancer and serves as a target for therapy in an experimental model.
Int J Cancer. 120:1803–1810. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Matsuzaki T, Yashiro M, Kaizaki R, Yasuda
K, Doi Y, Sawada T, Ohira M and Hirakawa K: Synergistic
antipro-liferative effect of mTOR inhibitors in combination with
5-fluorouracil in scirrhous gastric cancer. Cancer Sci.
100:2402–2410. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jackson C, Cunningham D and Oliveira J;
ESMO Guidelines Working Group: Gastric cancer: ESMO clinical
recommendations for diagnosis, treatment and follow-up. Ann Oncol.
20(Suppl 4): 34–36. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Glimelius B, Ekström K, Hoffman K, Graf W,
Sjödén PO, Haglund U, Svensson C, Enander LK, Linné T, Sellström H,
et al: Randomized comparison between chemotherapy plus best
supportive care with best supportive care in advanced gastric
cancer. Ann Oncol. 8:163–168. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Lei X, Lv X, Liu M, Yang Z, Ji M, Guo X
and Dong W: Thymoquinone inhibits growth and augments
5-fluoro-uracil-induced apoptosis in gastric cancer cells both in
vitro and in vivo. Biochem Biophys Res Commun. 417:864–868. 2012.
View Article : Google Scholar
|
|
35
|
Tang D, Lotze MT, Kang R and Zeh HJ:
Apoptosis promotes early tumorigenesis. Oncogene. 30:1851–1854.
2011. View Article : Google Scholar
|
|
36
|
Bjornsti MA and Houghton PJ: The TOR
pathway: A target for cancer therapy. Nat Rev Cancer. 4:335–348.
2004. View
Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wullschleger S, Loewith R and Hall MN: TOR
signaling in growth and metabolism. Cell. 124:471–484. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Tomioka H, Mukohara T, Kataoka Y,
Ekyalongo RC, Funakoshi Y, Imai Y, Kiyota N, Fujiwara Y and Minami
H: Inhibition of the mTOR/S6 K signal is necessary to enhance
fluorouracil-induced apoptosis in gastric cancer cells with HER2
amplification. Int J Oncol. 41:551–558. 2012.PubMed/NCBI
|