Introduction
House dust mites (HDMs) and storage mites (SMs) are
the major source of indoor or occupational allergens that cause
allergic diseases worldwide (1,2). To
date, 24 groups of allergens have been identified in HDMs and SMs
(3,4). A large number of these mite allergens
are cross-reactive. For example, tropomyosin, a group-10 allergen,
is a well-known pan-allergen that causes cross-reactivity among
mites, a variety of invertebrates (5,6)
including shrimps and crabs, and parasites, including
Anisakis nematodes and roundworms (7–9).
Group-8 mite allergens (glutathione S-transferase) have also
been shown to be cross-reactive with other arthropod allergens
(10,11). The group-2 allergens are
cross-reactive among mite species (12,13)
and are the primary allergens responsible for the cross-reactivity
between HDM and SM in Korea (14).
It has been suggested that a majority of the immunoglobulin (Ig)E
reactivity to SM allergens is due to cross-reactivity between
group-2, -8 and -10 allergens from HDMs. However, Son et al
(15) reported that IgE
cross-reactivity was observed between Dermatophagoides
farinae and Acarus siro extracts, even though strong IgE
reactive components were not detected by IgE immunoblot analysis
under reducing conditions. These results indicated the importance
of conformational epitopes for cross-reactivity (15). Tyrophagus putrescentiae is
one of the predominant SM species in Korea. Five Tyrophagus
putrescentiae allergens have been described to date: Tyr p 2,
Tyr p 3, Tyr p 8, Tyr p 10 and Tyr p 24 (16). Of these, Tyr p 2, Tyr p 8 and Tyr p
10 have been shown to cause cross-reactivity (6,11,15,17).
The allergenicity of group-5 allergens is moderate,
as only ~50% of HDM allergic patients are sensitized (18). In tropical regions, however, the
Blomia tropicalis allergen Blo t 5 causes reactions in
70–92% of patients with mite allergies, making it the predominant
reaction-associated group-5 allergen in that region (19,20).
Group-21 allergens, which are homologous with the group-5
allergens, have only been described in Blomia tropicalis and
Dermatophagoides pteronyssinus (21,22).
The primary as well as secondary structures of group-5 and -21
allergens are highly conserved. Blo t 21 and Der p 21 exhibit
moderate IgE reactivity and were found to co-localize in the
mid-gut epithelium, lumen and feces of mites (21–23).
Lin et al (18) reported
that Der p 5 was not abundant in house dust, with a concentration
of <100 ng/g in dust. Der p 5 and -21 are heat stable and can
sensitize allergic individuals in domestic environments over a long
period.
Arruda et al (19) failed to observe a correlation
between Blo t 5 and Der p 5 in eliciting IgE responses in Blomia
tropicalis-exposed and -unexposed individuals, despite 43%
identity at the amino acid level. Similarly, in a study on
Malaysian and Taiwanese patients, Kuo et al (20) reported a low correlation of IgE
reactivity between Blo t 5 and Der p 5 (Taiwanese, r=0.452;
Malaysian, r=0.346) as well as limited cross-reactivity. Gao et
al (21) demonstrated that Blo
t 21 was not highly cross-reactive to Blo t 5 despite a certain
amount of sequence and structural identity, while Weghofer et
al (22) showed that Der p 21
exhibited low IgE reactivity to Der p 5, Lep d 5 and Blo t 5
according to tests using sera from Der p 21-sensitized patients. In
addition, Blo t 21 was reported to have low to moderate
cross-reactivity with Blo t 5, Der p 5 and Der f 21 (24). While multiple studies have focused
on Der p 5 and Blo t 5, group-5 and -21 allergens from
Dermatophagoides farinae and Tyrophagus
putrescentiae, which are the predominant mite species in Korea,
have yet to be examined.
In the present study, six recombinant (r) allergens,
rDer f 5, rDer f 21, rTyr p 5, rTyr p 21, rBlo t 5 and rBlo t 21,
were purified using an Escherichia (E.) coli
expression system, and the IgE reactivity to each allergen was
determined using sera from Korean HDM-sensitized patients. The
cross-reactivities of these six recombinant allergens were then
examined using a competitive ELISA approach. Furthermore, the
potential of using rTyr p 5 and rTyr p 21 for mite species-specific
diagnoses was examined.
Materials and methods
Subjects and serum samples
Serum samples were obtained from patients attending
the Allergy-Asthma Clinic at Severance Hospital, Yonsei University
College of Medicine in Seoul, Korea. Allergy diagnoses were based
on any history of allergic reactions and skin prick testing. Sera
from patients were tested for IgE antibodies specific for HDM
(Dermatophagoides farinae) allergens using the Uni-CAP
system (Phadia, Uppsala, Sweden) and ELISA analysis. Sera from 194
HDM-sensitized subjects (males/females, 103:91; average age, 25.4
years; age range, 4–67 years) and 20 healthy controls were used to
assess the IgE reactivity of recombinant proteins. Serum samples
were collected after consent of the patients, and experiments using
the collected sera were approved by the Institutional Review Board
(no. 4-2009-0180). The study was approved by the ethics committee
of Sevrance Hospital (Yonsei University College of Medicine).
Cloning and expression of recombinant
proteins in E. coli
Frozen Dermatophagoides farinae,
Tyrophagus putrescentiae and Blomia tropicalis mites
were obtained from the Arthropods of Medical Importance Resource
Bank (AMIB) at the Department of Environmental Medical Biology,
Yonsei University College of Medicine (Seoul, Korea). Total RNA was
isolated from frozen mite bodies using TRIzol reagent (Invitrogen
Life Technologies, Carlsbad, CA, USA), according to the
manufacturer's instructions. First-strand cDNA was synthesized from
5 µg total RNA using avian myeloblastosis reverse
transcriptase (Promega, Madison, WI, USA) and an oligo
(dT18) primer. Primers were designed based on the
published full-length nucleotide sequence of Der f 5 (GenBank
accession no. AY283283), Der f 21 (EF027122), Tyr p 5 (AY800358),
Tyr p 21 (AY800360), Blo t 5 (U59102) and Blo t 21 (AY800348), and
sequences were flanked by restriction enzyme sites for either
BamH I/Xho I or BamH I/Sal I. The
primer sequences were as follows: Der f 5 forward, 5′-GGATCCATG
AAATTCATCATTGCTATTGCTG-3′ and reverse,
5′-CTCGAGTTAAACTTCAATCTTCTTCACACGTTGCTC-3′; Der f 21 forward,
5′-GGATCCATGAAATTCATTATTTTCTGTGCCA-3′ and reverse,
5′-CTCGAGTTAATCATCCGATTTTACAGCTTTCACCTT-3′; Tyr p 5 forward,
5′-GGATCCATGAAGTTCGCCATTCTCGC-3′ and reverse,
5′-CTCGAGTTAGCGAGTCTTGACAGCCT-3′; Blo t 5 forward,
5′-GGATCCATGAAGTTCGCCATCGTT CTTATTG-3′ and reverse,
5′-CTCGAGTTATTGGGTTTG AATATCCTTCACTTTTTG-3′; Tyr p 21 forward,
5′-GGATCCATGAAGTTCGTCATCGCCCT-3′ and reverse,
5′-GTCGACTTAGACCTTGATGGCGTTCACT-3′; Blo t 21 forward,
5′-GGATCCATGAAATTTATCATCGCATTGGCTG-3′ and reverse,
5′-GTCGACTTATTCGGAATCTTGGACTCGCTTT-3′. The primers were synthesized
by GeneTech Corporation (Daejeon, Korea). The conditions for PCR
amplification were as follows: Pre-denaturation at 94°C for 5 min,
30 cycles of denaturation at 94°C for 30 sec, annealing at 55°C for
30 sec, extension at 72°C for 1 min and, following the final cycle,
an additional extension at 72°C for 5 min. The PCR-amplified cDNA
sequences encoding Der f 5, Der f 21, Tyr p 5, 21, Blo t 5 and 21
were cloned into the pCR4-TOPO vector (Invitrogen Life
Technologies), sub-cloned into the pET-28a expression vector
(Novagen, San Diego, CA, USA), and transformed into E. coli
Rosetta™ 2 (DE3) cells (Novagen). The expression of recombinant
proteins was induced by addition of 1 mM
isopropyl-1-thio-β-galactopyranoside (BioShop Canada Inc.,
Burlington, ON, Canada), and protein was purified from the
insoluble fraction of cell lysates using Ni sepharose excel resin
(GE Healthcare, Uppsala, Sweden) under denaturing conditions (20 mM
sodium phosphate, 500 mM NaCl, 500 mM imidazole, 6 M urea, pH 7.4).
The concentration of each of the recombinant allergens was
determined using a Bradford Assay (Bio-Rad Laboratories, Hercules,
CA, USA). The purified recombinant allergens were separated by 15%
SDS-PAGE, followed by staining with Coomassie Blue R250 (Amresco,
Solon, OH, USA).
Analysis of specific IgE binding to
recombinant proteins
Recombinant Der f 2 was obtained from the AMIB and
used as a control. Microtiter plates were coated with 100 µl
recombinant protein (2 µg/ml in 50 mM sodium carbonate, pH
9.6), and washed with phosphate-buffered saline (PBS) containing
0.05% Tween 20 (PBST). The plates were blocked with 1% bovine serum
albumin (BSA; EMD Millipore, Kankakee, IL, USA) in PBST for 1 h at
room temperature and then incubated for 1 h with 100 µl
serum per well diluted at 1:9 in PBST containing 1% BSA. IgE
antibodies were detected using biotinylated goat anti-human IgE
(Vector Laboratories, Burlingame, CA, USA) and
streptavidin-peroxidase (Sigma-Aldrich, St. Louis, MO, USA). The
assay was developed with 3,3′,5,5′-tetramethylbenzidine (Kirkegaard
and Perry Laboratories, Gaithersburg, MD, USA), which undergoes a
color change in the presence of the antibody/allergen complexes.
The absorbance (A) at 450 nm was measured using a Tecan sunrise
microplate reader (Tecan, Salzburg, Austria) and the Magellan CE
software after addition of 0.5 M H2SO4 to
stop color development. The cut-off value was determined as the
mean absorbance plus twice the value of the standard deviation of
20 negative controls.
Inhibition assay
The inhibitory effects of each allergen were
examined using a competitive ELISA approach. For these experiments,
recombinant protein was suspended in coating buffer (2
µg/ml, 50 mM carbonate buffer, pH 9.6) and added to the
wells of ELISA plates. After blocking with 1% BSA in PBST for 1 h
at room temperature, wells were incubated with selected serum
samples (1:9; a pooled serum of 10 subjects) that had been
pre-incubated overnight at 4°C with solutions containing various
concentrations (0.001, 0.01, 0.1, 1.0 or 10.0 µg/ml) of
recombinant proteins. IgE antibodies were detected as described
above. The percentage of inhibition was calculated as
(1−Ai/A0)×100, where Ai stands for
absorbance at 450 nm with an inhibitor and A0 for the
absorbance at 450 nm without an inhibitor. These assays were
conducted in duplicate.
Statistical analysis
Correlations between the IgE reactivities to
recombinant proteins were analyzed by Pearson's correlation using
GraphPad Prism 6.0 (GrahPad, Inc., La Jolla, CA, USA). P<0.05
was considered to indicate a statistically significant
difference.
Results
Sequence analysis of Der f 5, Der f 21,
Tyr p 5, Tyr p 21, Blo t 5 and Blo t 21 suggests
cross-reactivity
The amino acid sequences of the six group-5 and
group-21 allergens, Der f 5, Der f 21, Tyr p 5, Tyr p 21, Blo t 5
and Blo t 21, were compared. As shown in Table I, the sequence of each allergen was
36.6–55.8% identical to that of the other group-5 and -21
allergens. While Der f 5 exhibited the highest level of identity to
Blo t 5 (44.4%), Der f 21 was most similar to Tyr p 5 (41.6%), Tyr
p 5 was most similar to Blo t 5 (52.9%) and Tyr p 21 was most
similar to Blo t 21 (55.8%). The lowest level of similarity was
observed between Der f 5 and Blo t 21 (36.6%), while the highest
sequence identity was observed between Tyr p 21 and Blo t 21
(55.8%) (Table I). This high level
of sequence identity between allergens suggested cross-reactivity
between group-5 and group-21 allergens. As depicted in Fig. 1, four amino acids that were
previously found to be crucial for IgE binding, were shown to be
conserved in five of the allergens: Der f 21 (E-77, D-82, E-87 and
E-92), Tyr p 5 (E-78, D-83, E-88 and E-93), Tyr p 21 (E-82, D-87,
E-92 and E-97) Blo t 5 (E-76, D-81, E-86 and E-91) and Blo t 21
(E-74, D-79, E-84 and E-89). Der f 5 (L-74, D-79, E-84 and E-89)
was the only allergen in which the binding site was not
conserved.
 | Table ISequence identity (%) of group-5 and
group-21 allergens from Dermatophagoides farinae, Tyrophagus
putrescentiae and Blomia tropicalis. |
Table I
Sequence identity (%) of group-5 and
group-21 allergens from Dermatophagoides farinae, Tyrophagus
putrescentiae and Blomia tropicalis.
| Amino acid
sequence | Der f 5 | Tyr p 5 | Blo t 5 | Der f 21 | Tyr p 21 | Blo t 21 |
|---|
| Der f 5 | 100 | 38.0 | 44.4 | 36.7 | 40.3 | 36.6 |
| Tyr p 5 | – | 100 | 52.9 | 41.6 | 45.3 | 43.4 |
| Blo t 5 | – | – | 100 | 41.2 | 37.4 | 40.3 |
| Der f 21 | – | – | – | 100 | 39.3 | 37.5 |
| Tyr p 21 | – | – | – | – | 100 | 55.8 |
| Blo t 21 | – | – | – | – | – | 100 |
Production of recombinant group-5 and
group-21 allergens
Recombinant Der f 5, Der f 21, Tyr p 5, Tyr p 21,
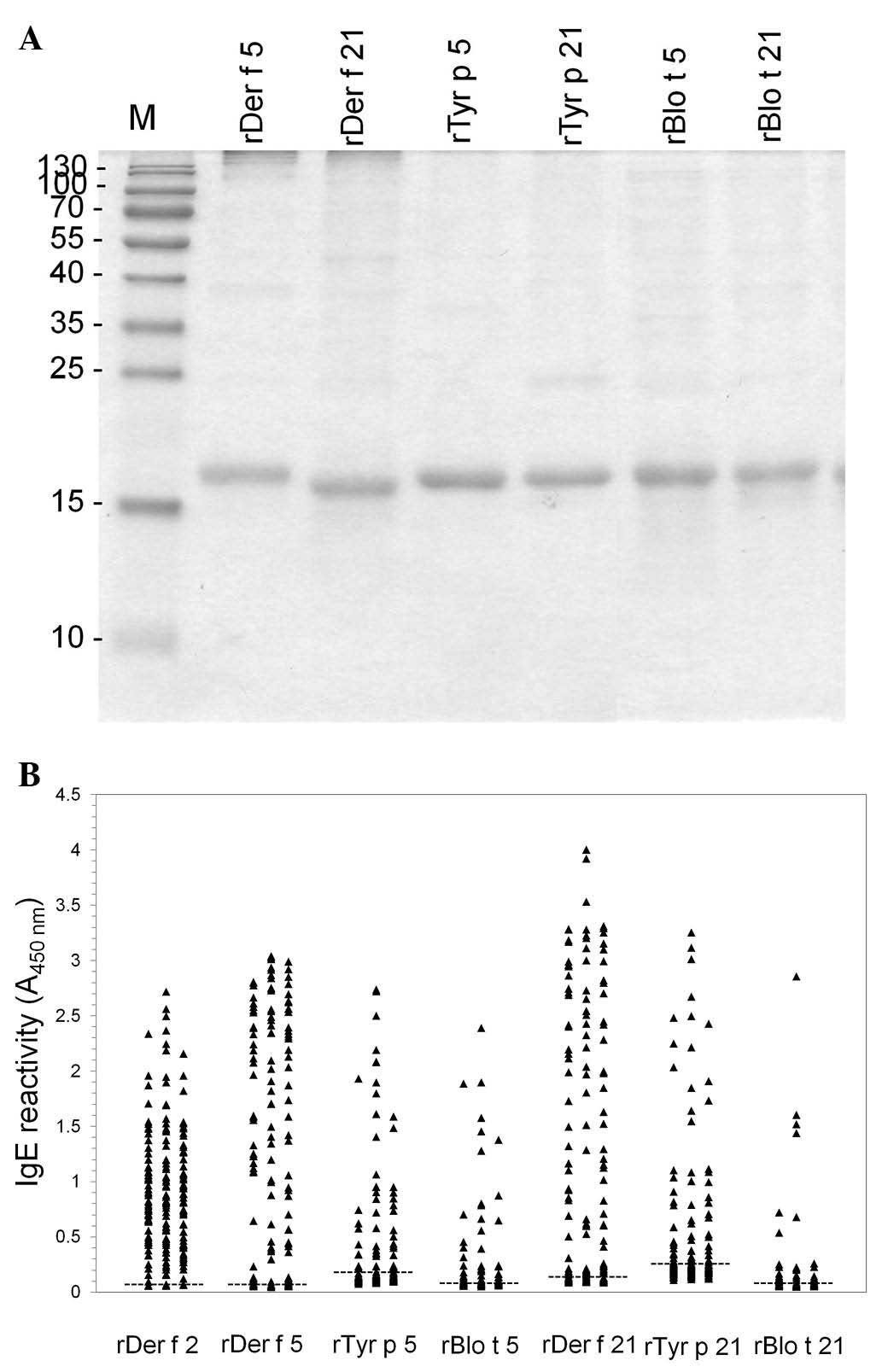
Blo t 5 and Blo t 21 were inducibly expressed in E. coli.
The recombinant proteins were purified from E. coli
inclusion bodies by affinity chromatography using a Ni-sepharose
resin. Purified proteins were then separated by 15% SDS-PAGE and
visualized by Coomassie Brilliant Blue staining. As expected, the
purified proteins migrated slightly slower than the 15-kDa marker,
which was consistent with the predicted 17-kDa size of the
allergens (Fig. 2A).
IgE reactivity is correlated between
HDM-associated recombinant proteins
The IgE reactivity of each purified recombinant
allergen was examined by ELISA using sera from 194 Korean patients
who were allergic to HDM. Der f 2 was used as a positive control.
While 187 of the 194 sera (96.39%) tested showed reactivity to rDer
f 2, 126 (64.95%) to rDer f 5, 128 (65.98%) to rDer f 21, 59
(30.41%) to rTyr p 5, 80 (41.24%) to rTyr p 21, 60 (30.93%) to rBlo
t 5 and 42 (21.65%) to rBlo t 21 (Fig.
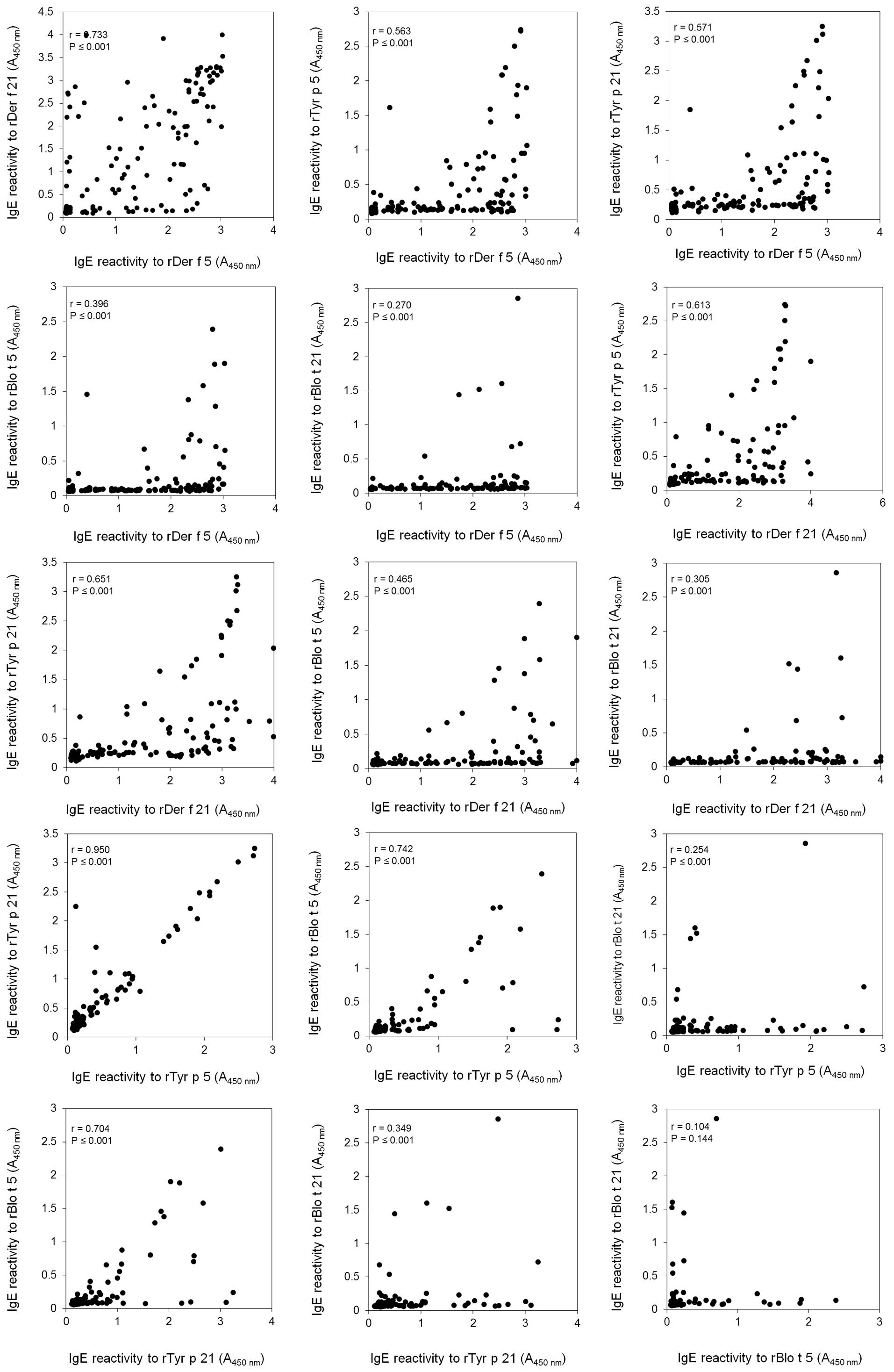
2B). The IgE reactivity of rDer f 5 was also strongly
correlated with that of rDer f 21 (r=0.733). The IgE reactivity to
rTyr p 5 exhibited the highest correlation with that to rTyr p 21
(r=0.950). It also moderately correlated with those of rDer f 5
(r=0.563), rDer f 21 (r=0.613) and Blo t 5 (r=0.742). The IgE
reactivity to rTyr p 21 correlated the highest with the
reactivities to rDer f 5 (r=0.571), rDer f 21 (r=0.651) and Blo t 5
(r=0.704), while the reactivity to rBlo t 5 was highly correlated
with those to rTyr p 5 (r=0.742) and rTyr p 21 (r=0.704). However,
the IgE reactivity to rBlo t 21 exhibited a poor correlation with
those to rDer f 5, rDerf 21, rTyr p 5 and rTyr p 21 (r=0.270,
0.305, 0.254, 0.349, respectively) (Fig. 3). These results demonstrated that
the IgE reactivity of all six recombinant allergens exhibited at
least a certain level of correlation to one another.
IgE cross-reactivity is present among
recombinant group-5 and group-21 allergens
In order to investigate the cross-reactivity between
the allergens, the present study performed an IgE inhibition on
each allergen, using a competitive ELISA approach with serum pooled
from 10 allergy patients (Table
II). The IgE reactivities to rDer f 2, rDer f 5 and rDer f 21
were only inhibited by the selfsame allergens. rDer f 21 was found
to be the strongest inhibitor of IgE reactivity to the other
allergens, inhibiting 93.01% of IgE reactivity to rTyr p 5, 92.12%
of that to rTyr p 21, 86.99% of that to rBlo t 5 and 70.30% of IgE
reactivity to rBlo t 21, at an inhibitor concentration of 10
µg/ml (Fig. 4). The IgE
reactivity to rBlo t 21 was inhibited by up to 35.27% by rDer f 5,
and by up to 43.57% by rTyr p 21. rTyr p 5 inhibited 76.77% of IgE
reactivity to rTyr p 21, while rTyr p 21 inhibited 86.0% of the IgE
reactivity to rTyr p 5. The IgE reactivity of Blo t 5 was inhibited
by up to 77.35% by rTyr p 5 and by 73.44% by rTyr p 21. In
addition, rBlo t 21 inhibited the IgE reactivity to rTyr p 21 by up
to 43.57%. These results suggested that each of the allergens
tested exhibited a certain degree of cross-reactivity to one
another; however, there was an extremely low degree of
cross-reactivity between Der f 5 and the other allergens tested,
which may be due to the lack of a conserved IgE binding site of Der
f 5.
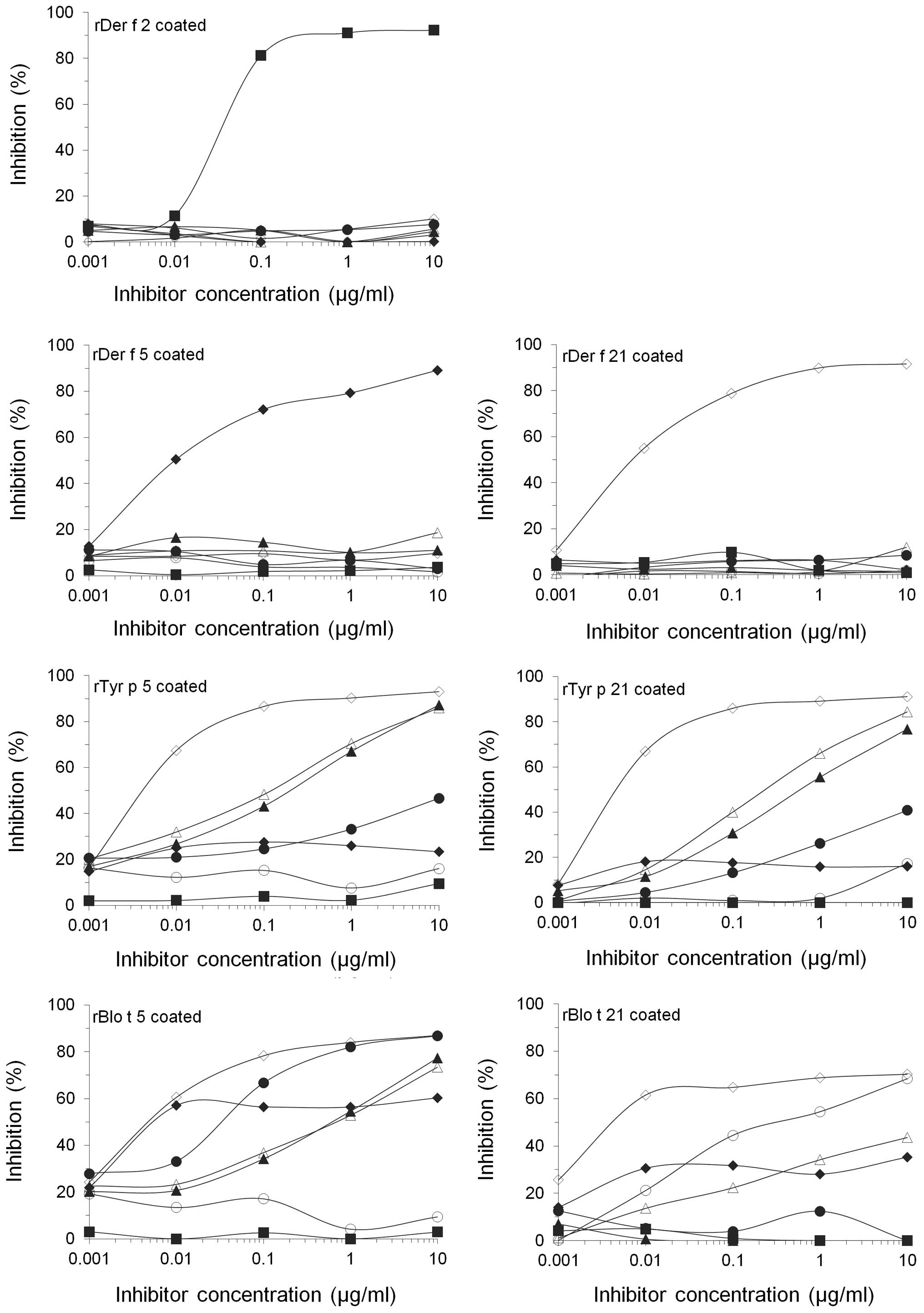 | Figure 4Inhibition analyses of six recombinant
allergens. Competitive IgE inhibition ELISA was conducted using
pooled sera from 10 patients, pre-absorbed with 0.001, 0.01, 0.1, 1
or 100 µg/ml of each allergen (■, rDer f 2; ♦, rDer f
5; ⋄, rDer f 21; ▲, rTyr p 5; Δ, rTyr p 21; •, rBlo t 5; ○, rBlo t
21). IgE, immunoglobulin E; r, recombinant; Der,
Dermatophagoides farinae; Tyr, Tyrophagus
putrescentiae; Blo, Blomia tropicalis. |
 | Table IICharacteristics of patients used for
the competitive inhibition ELISA. |
Table II
Characteristics of patients used for
the competitive inhibition ELISA.
| No. | Age (years) | Gender (M/F) | Clinical
symptoms | ELISA results
(A450nm)a
|
|---|
| rDer f 5 | rDer f 21 | rTyr p 5 | rTyr p 21 | rBlo t 5 | rBlo t 21 |
|---|
| 1 | 23 | M | Rhinitis | 2.916 | 3.281 | 2.739 | 3.251 | 0.239 | 0.723 |
| 2 | 14 | M | Asthma | 3.029 | 3.991 | 1.898 | 2.035 | 1.899 | 0.146 |
| 3 | 31 | M | Rhinitis | 2.128 | 2.285 | 0.420 | 1.544 | 0.071 | 1.519 |
| 4 | 25 | F | Rhinitis | 2.560 | 3.252 | 0.402 | 1.114 | 0.078 | 1.602 |
| 5 | 55 | M | Asthma | 2.343 | 1.804 | 1.404 | 1.641 | 0.800 | 0.074 |
| 6 | 9 | M | Asthma | 2.866 | 3.168 | 1.932 | 2.483 | 0.703 | 2.855 |
| 7 | 16 | M | Asthma | 2.559 | 3.113 | 2.084 | 2.495 | 0.785 | 0.071 |
| 8 | 12 | M | Asthma | 2.336 | 2.994 | 1.588 | 1.909 | 1.377 | 0.104 |
| 9 | 24 | M | Rhinitis | 2.838 | 2.999 | 1.798 | 2.214 | 1.884 | 0.090 |
| 10 | 16 | M | Atopic
dermatitis | 2.623 | 3.289 | 2.190 | 2.672 | 1.578 | 0.087 |
Discussion
In the present study, six recombinant group-5 and
group-21 mite allergens were produced using an E. coli
expression system, and their IgE reactivities and
cross-reactivities were examined. The recombinant proteins
exhibited moderate IgE reactivities (21.65–65.98%) and
low-to-moderate cross-reactivities in Korean patients with HDM
allergies. Previous studies reported that group-5 and group-21 mite
allergens exhibited little to no cross-reactivity (20,21,24),
and Tan et al (24)
reported low to moderate cross-reactivity between Blo t 21 and Blo
t 5, Der p 5 and Der f 21. Blo t 21 and Blo t 5 encode a conserved
IgE epitope, and there is little difference in their secondary
structure. A linear IgE epitope of Blo t 5 was mapped in the loop
region (76-ELKRTDLNILERFNYE-91). However, the IgE epitope of Blo t
21 is not thought to be linear, as one (E-89) of the four critical
amino acids of the IgE binding site (E-74, D-79, E-84 and E-89) was
predicted to be part of another amino acid cluster (24). It is likely that the
cross-reactivity of group-5 and -21 allergens may be affected by
their tertiary structure.
rDer f 5 and rDer f 21 were inhibited by the
selfsame proteins, but not by any other allergens. This reflects
the fact that the sera utilized in the present study are from
subjects who are sensitized to Dermatophagoides farinae, but
not to SMs. The IgE reactivity to Der f 21 was effectively
inhibited by Tyr p 5, Tyr p 21, Blo t 5 and Blo t 21. This may
suggest that the IgE reactivity to the group-5 and -21 allergens of
SMs are cross-reactive to Der f 21. It is likely that the IgE
epitopes of Der f 21 are similar to or partially overlap with those
of the group-5 and -21 allergens from Tyrophagus
putrescentiae and Blomia tropicalis. By contrast, rDer f
5 exhibited poor cross-reactivity to the other group-5 and -21
allergens tested. Therefore, Der f 5 may potentially be used for
species-specific diagnoses. By contrast, due to its high level of
cross-reactivity, Der f 21 would likely cause an allergic response
in patients that are not sensitized to SMs.
Interestingly, although Blomia tropicalis is
not native to Korea, rBlo t 5 and rBlo t 21 displayed IgE
reactivity to the sera of Korean patients with HDM allergies. This
may presumably be due to the cross-reactivity of Blomia
tropicalis allergens to those of Dermatophagoides
farinae, Tyrophagus putrescentiae or Blomia
kulagini. Of note, Blomia kulagini, though not being
common, has been found in Korea and was shown to be cross-reactive
with Blomia tropicalis (25). In the present study, the binding of
IgE to rBlo t 5 and rBlo t 21 was inhibited by rDer f 5, rDer f 21,
rTyr p 5 and rTyr p 21. However, rBlo t 5 and rBlo t 21 were unable
to inhibit IgE binding to the other group-5 and -21 allergens from
Dermatophagoides farinae or Tyrophagus putrescentiae.
In 2004, Chew and co-workers deposited the mRNA sequences of a Tyr
p 5 (accession name, Tyr p 5.01 allergen; accession number,
AY800358) and Tyr p 21 (Tyr p 5.03 allergen; AY800360; in the
present study, this allergen was named Tyr p 21 due to its high
similarity to Blo t 21) in GenBank. However, their IgE reactivity
had not yet been reported. The present study provided the first
analysis of the IgE reactivities to Tyr p 5 and Tyr p 21 as well as
their cross-reactivities with Der f 5, Der f 21, Blo t 5 and Blo t
21. Tyr p 5 exhibited 52.9% identity with Blo t 5 and 55.8% with
rTyr p 21 and Blo t 21 at the amino acid level. rTyr p 5 inhibited
77.35% of the IgE reactivity to rBlo t 5, indicating a high degree
of cross-reactivity. Furthermore, rTyr p 21 inhibited 43.57% of the
IgE reactivity to rBlo t 21. According to Casset et al
(26), Der p 5 and Der p 21 were
not thought to be stable in allergen extracts as they were not
detected in commercially available extracts. Indeed, the batch
variation of crude extracts can result in difficulties with allergy
diagnosis and immunotherapy. These issues, however, may be solved
by the use of recombinant allergens, which allow for easy
standardization and preparation of allergen mixtures (27). Recombinant allergens may also
enable component-resolved diagnoses and personalized
allergen-specific immunotherapies.
In conclusion, the present study examined the IgE
reactivity and cross-reactivity of recombinant group-5 and group-21
allergens from three mite species Dermatophagoides farinae,
Tyrophagus putrescentiae and Blomia tropicalis.
Cross-reactivity to the storage allergens (Tyr p 5, Tyr p 21, Blo t
5 and Blo t 21) was observed when high titers of IgE antibodies,
specific for HDM allergens (Der f 5 and Der f 21), were detected.
This was particularly the case for Der f 21. It is therefore
concluded that mite group-5 and -21 allergens are at least in part
responsible for these cross-reactions. However, further studies are
required to identify the IgE epitope that is the cause of
cross-reactivity. Understanding the cross-reactivity that occurs
between allergens from different mite species may be useful for the
development of improved component-resolved diagnoses and for
enhancing treatment of mite-associated allergies.
Acknowledgments
This study was supported by Yonsei University
College of Medicine (grant no. 6-2011-0160).
References
|
1
|
Thomas WR, Hales BJ and Smith WA: House
dust mite allergens in asthma and allergy. Trends Mol Med.
16:321–328. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jeong KY, Park JW and Hong CS: House dust
mite allergy in Korea: The most important inhalant allergen in
current and future. Allergy Asthma Immunol Res. 4:313–325. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Yong TS and Jeong KY: Household arthropod
allergens in Korea. Korean J Parasitol. 47(Suppl 47): S143–S153.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bessot JC and Pauli G: Mite allergens: An
overview. Eur Ann Allergy Clin Immunol. 43:141–156. 2011.PubMed/NCBI
|
|
5
|
Reese G, Ayuso R and Lehrer SB:
Tropomyosin: An invertebrate pan-allergen. Int Arch Allergy
Immunol. 119:247–258. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jeong KY, Hong CS and Yong TS: Allergenic
tropomyosins and their cross-reactivities. Protein Pept Lett.
13:835–845. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Gámez C, Sánchez-García S, Ibáñez MD,
López R, Aguado E, López E, Sastre B, Sastre J and del Pozo V:
Tropomyosin IgE-positive results are a good predictor of shrimp
allergy. Allergy. 66:1375–1383. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nieuwenhuizen NE and Lopata AL: Anisakis:
A food-borne parasite that triggers allergic host defences. Int J
Parasitol. 43:1047–1057. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Acevedo N and Caraballo L: IgE
cross-reactivity between Ascaris lumbricoides and mite allergens:
Possible influences on allergic sensitization and asthma. Parasite
Immunol. 33:309–321. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Huang CH, Liew LM, Mah KW, Kuo IC, Lee BW
and Chua KY: Characterization of glutathione S-transferase from
dust mite, Der p 8 and its immunoglobulin Ecross-reactivity with
cockroach glutathione S-transferase. Clin Exp Allergy. 36:369–376.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liao EC, Lin YH, Chiu CL, Lin TC and Tsai
JJ: Identification of allergeniccomponent Tyr p 8 from Tyrophagus
putrescentiae and cross-reactivity with Der p 8. Clin Vaccine
Immunol. 20:506–512. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Johannessen BR, Skov LK, Kastrup JS,
Kristensen O, Bolwig C, Larsen JN, Spangfort M, Lund K and Gajhede
M: Structure of the house dust mite allergen Der f 2: Implications
for function and molecular basis of IgE cross-reactivity. FEBS
Lett. 579:1208–1212. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Barber D, Arias J, Boquete M, Cardona V,
Carrillo T, Gala G, Gamboa P, García-Robaina JC, Hernández D, Sanz
ML, et al: Analysis of mite allergic patients in a diverse
territory by improved diagnostic tools. Clin Exp Allergy.
42:1129–1138. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Park JW, Ko SH, Yong TS, Ree HI, Jeoung BJ
and Hong CS: Cross-reactivity of Tyrophagus putrescentiae with
Dermatophagoides farinae and Dermatophagoides pteronyssinus in
urban areas. Ann Allergy Asthma Immunol. 83:533–539. 1999.
View Article : Google Scholar
|
|
15
|
Son M, Jeong KY, Kim BJ, Lim KJ, Lee JH
and Park JW: IgE reactivity to Acarus siro extract in Korean dust
mite allergic patient. Exp Appl Acarol. 63:57–64. 2014. View Article : Google Scholar
|
|
16
|
Jeong KY, Kim CR, Un S, Yi MH, Lee IY,
Park JW, Hong CS and Yong TS: Allergenicity of recombinant troponin
C from Tyrophagus putrescentiae. Int Arch Allergy Immunol.
151:207–213. 2010. View Article : Google Scholar
|
|
17
|
Fernández-Caldas E, Iraola V and Carnés J:
Molecular and biochemical properties of storage mites (except
Blomia species). Protein Pept Lett. 14:954–959. 2007. View Article : Google Scholar
|
|
18
|
Lin KL, Hsieh KH, Thomas WR, Chiang BL and
Chua KY: Characterization of Der p V allergen, cDNA analysis and
IgE-mediated reactivity to the recombinant protein. J Allergy Clin
Immunol. 94:989–996. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Arruda LK, Fernandez-Caldas E, Naspitz CK,
Montealegre F, Vailes LD and Chapman MD: Identification of Blomia
tropicalis allergen Blo t 5 by cDNA cloning. Int Arch Allergy
Immunol. 107:456–457. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kuo IC, Cheong N, Trakultivakorn M, Lee BW
and Chua KY: An extensive study of human IgE cross-reactivity of
Blo t 5 and Der p 5. J Allergy Clin Immunol. 111:603–609. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Gao YF, Wang de Y, Ong TC, Tay SL, Yap KH
and Chew FT: Identification and characterization of a novel
allergen from Blomia tropicalis: Blo t 21. J Allergy Clin Immunol.
120:105–112. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Weghofer M, Dall'Antonia Y, Grote M,
Stöcklinger A, Kneidinger M, Balic N, Krauth MT, Fernández-Caldas
E, Thomas WR, van Hage M, et al: Characterization of Der p 21, a
new important allergen derived from the gut of house dust mites.
Allergy. 63:758–767. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Weghofer M, Grote M, Dall'Antonia Y,
Fernández-Caldas E, Krauth MT, van Hage M, Horak F, Thomas WR,
Valent P, Keller W, et al: Characterization of folded recombinant
Der p 5, a potential diagnostic marker allergen for house dust mite
allergy. Int Arch Allergy Immunol. 147:101–109. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tan KW, Ong TC, Gao YF, Tiong YS, Wong KN,
Chew FT and Mok YK: NMR structure and IgE epitopes of Blo t 21, a
major dust mite allergen from Blomia tropicalis. J Biol Chem.
287:34776–3485. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Cardona G, Guisantes J, Postigo I, Eraso
E, Serna LA and Mar tínez J: Allergenic cross-reactivity between
Blomia tropicalis and Blomia kulagini (Acari: Echymiopodidae)
extracts from optimized mite cultures. J Investig Allergol Clin
Immunol. 15:259–265. 2005.
|
|
26
|
Casset A, Mari A, Purohit A, Resch Y,
Weghofer M, Ferrara R, Thomas WR, Alessandri C, Chen KW, de Blay F,
et al: Varying allergen composition and content affects the in vivo
allergenic activity of commercial Dermatophagoides pteronyssinus
extracts. Int Arch Allergy Immunol. 159:253–262. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Vrtala S, Huber H and Thomas WR:
Recombinant house dust mite allergens. Methods. 66:67–74. 2014.
View Article : Google Scholar
|