Introduction
Reactive oxygen species (ROS) are chemically
reactive molecules containing oxygen. ROS include free radicals,
such as superoxide radical (O2●−), hydroxyl
radical (OH●) and peroxyl radical (ROO●), as
well as non-radical species, such as hydrogen peroxide
(H2O2), which are products of normal
metabolism, inflammation, phagocytosis, intracellular signaling,
cell proliferation, apoptosis or other physiological processes
(1,2). Oxidative stress is defined as an
imbalance between the production of free radicals and the ability
of the organism to counteract or detoxify their harmful effects
through neutralization by antioxidants (1). As free radicals are very reactive
species, their overproduction during oxidative stress can cause
damage to all biological macromolecules, such as DNA, proteins and
lipids, thus leading to cell damage and subsequently, to the
manifestation of pathological conditions (1).
Oxidative stress occurs frequently in muscle tissue.
In particular, during intense exercise, there is a high rate of
O2 consumption in skeletal muscle that can cause
incomplete O2 reduction and electron leakage from the
electron transfer chain, leading to the generation of ROS and
oxidative stress, that in turn result in muscle fatigue, cell
damage and apoptosis (2–4).
Furthermore, oxidative stress-induced damage of the
vascular endothelium is one of the most important factors of
cardiovascular diseases (5–7). For
example, oxidative stress in endothelial cells induces acute and
chronic phases of leukocyte adhesion to the endothelium (8,9). In
addition, the interaction between ROS and nitric oxide sets off a
vicious circle which results in further endothelial activation and
inflammation (5). Moreover, ROS,
such as H2O2 can diffuse throughout
endothelial cells and react with cysteine groups in proteins to
modify their function (10). Thus,
under conditions of oxidative stress, endothelial cells can lose
integrity, progress to senescence and detach into the circulation
(11).
However, every living organism has antioxidant
mechanisms to counteract oxidative stress (1). Apart from the endogenous antioxidant
mechanisms, the supplementation of antioxidants through diet may be
used to reduce the detrimental effects of oxidative stress on human
health as suggested by our previous studies (12,13).
Some of the most well known food sources of antioxidants are grapes
and wine (14). Our research group
has conducted several studies on the antioxidant properties of
grapes which are attributed mainly to their polyphenolic content
(15–21). We have also conducted a series of
studies on the antioxidant activity of extracts from grape stems, a
by-product of the winemaking process which has been little
investigated (15,22–25).
For example, in one of our previous studies, we demonstrated that
grape stem extracts are rich in bioactive polyphenols (e.g.,
flavonoids, stilbenes and phenolic acids) (22) and exhibit strong free radical
scavenging activity in vitro (15).
Thus, the aim of the present study was firstly to
examine the antioxidant effects of stem extracts from three Greek
grape (Vitis vinifera) varieties (i.e., Mandilaria,
Mavrotragano and Moshomavro) at a cellular level, particularly in
human umbilical vein endothelial EA.hy926 and C2C12 muscle cells.
Moreover, since the polyphenolic composition has been shown to
affect the antioxidant activity of wine and grape seed extracts
(26,27), we also investigated whether the
polyphenolic composition of grape stem extracts affects their
antioxidant capacity. Grape stem extracts may be used as
antioxidant food supplements for reducing the oxidative
stress-induced detrimental effects on human health, and thus the
understanding of the mechanisms through which they exert their
bioactivities is of particular importance.
Materials and methods
Chemicals, reagents and culture
medium
Dulbecco's modified Eagle's medium (DMEM), fetal
bovine serum (FBS), phosphate-buffered saline (PBS) and trypsin
were purchased from Gibco (Grand Island, NY, USA).
Ethylenediaminetetraacetic acid (EDTA), 2,2′-azino-bis
(3-ethylbenzothiazoline-6-sulfonic acid), diammonium salt (ABTS),
horseradish peroxidase (HRP), 2,2-diphenyl-1-picrylhydrazyl (DPPH),
ferric chloride, 2-deoxyribose, nicotinamide adenine dinucleotide
(NADH), nitroblue tetrazolium (NBT), phenazine methosulfate (PMS),
mercury orange and 2,7-dichlorofluorescein diacetate (DCF-DA) were
obtained from Sigma-Aldrich (St. Louis, MO, USA). Trichloroacetic
acid (TCA) and 2-thiobarbituric acid (TBA) were purchased from
Merck (Darmstadt, Germany) and potassium ferricyanide was obtained
from AppliChem (Dresden, Germany). The cell proliferation kit II
(XTT) was purchased from Roche Diagnostics (Mannheim, Germany).
Preparation of the extracts
The stem extracts were prepared from the Greek grape
varieties, Mandilaria, Mavrotragano and Moshomavro. The isolation
of the extracts was performed as described in a previous study of
ours (15). In brief, 50 g of
dried sample (powdered grape stems) were poured into a 200 ml
mixture of methanol (MeOH)/H2O/1.0 N HCl (90:9.5:0.5
v/v) and sonicated in an ultrasonic bath for 10 min. The solvent
was separated by filtration, and the remaining solid was
re-extracted three additional times, using the same solvent system
and procedure. The combined extracts were evaporated under vacuum,
resulting in a slurry, which was dissolved in 30 ml of
MeOH/H2O (1:1) and centrifuged for 10 min (7,000 rpm).
The supernatant liquid was extracted with petroleum ether (3×30 ml)
to remove the lipids and concentrated under a vacuum. The remaining
residue was poured into 30 ml of brine and extracted repetitively
with ethyl acetate (EtOAc, 4×30 ml). Thus, all sugars remained in
the aqueous layer. The combined organic layers were then dried over
anhydrous magnesium sulfate and evaporated under a vacuum. The
remaining solid was weighed and dissolved in MeOH to 1 mg/ml, and
membrane filtered (0.45 µm). To avoid polyphenol
degradation, all of the aforementioned activities were performed in
the absence of direct sunlight and at temperatures below 35°C.
Assessment of the polyphenolic
composition of the extracts
The total polyphenolic content (TPC) of the extracts
was identified by the Folin-Ciocalteu method as previously
described (15). Briefly, a 100
µl sample of extract was added to a 10 ml flask containing 6
ml of deionized water. One milliliter of Folin–Ciocalteu reagent
was added to the mixture, and the flask was stoppered and allowed
to stand at room temperature for 3 min. A 1.5 ml portion of 20%
Na2CO3 was added and the solution was diluted
to the desire volume (10 ml) with deionized water. The absorbance
was measured at 725 nm versus a blank after 2 h at room
temperature. The results are expressed as gallic acid equivalents
using the standard curve (absorbance versus concentration) prepared
from authentic gallic acid.
The polyphenolic composition of the extracts was
assessed by high pressure liquid chromatography (HPLC) as
previously described (15).
Specifically, HPLC was carried out on a Hewlett Packard HP1100
system equipped with a quaternary pump and degasser. The column
used was a Kromasil C18 column (250 mm × 4.6 mm, particle size 5
µm) with a guard column of the same material (8×4 mm).
Injection was by means of a Rheodyne injection valve (model 7725I)
with a 20 µl fixed loop. For the chromatographic analyses,
HPLC grade water was prepared using a Milli-Q system, whereas all
HPLC solvents were filtered prior to use through cellulose acetate
membranes of 0.45 µm pore size. Chromatographic data were
acquired and processed using Chemstation software. More
specifically, the analysis was carried out at 30°C (maintained by a
column thermostat) using samples of 20 µl, which were
directly injected by means of a Rheodyne injection valve (model
7725I). The gradient eluted consisted of solvent A (obtained by the
addition of 3% acetic acid in 2 mM sodium acetate aqueous solution)
and solvent B (acetonitrile, CH3CN). Run time was set at
70 min with a constant flow rate at 1.0 ml/min in accordance with
the following gradient time table: at zero time, 95% A and 5% B;
after 45 min, the pumps were adjusted to 85% A and 15% B; at 60
min, 65% A and 35% B; at 65 min, 50% A and 50% B; and finally at 70
min, 100% B. This routine was followed by a 30-min equilibration
period with the zero time mixture prior to injection of the next
sample. The analysis was monitored at 280, 320, and 360 nm
simultaneously. Three replicate experiments were carried out for
each sample examined. Peaks were identified by comparing their
retention time and UV-vis spectra with the reference compounds, and
data were quantified using the corresponding curves of the
reference compounds as standards. All standards were dissolved in
methanol.
Cell culture conditions
The EA.hy926 cells were a gift from Professor
Koukoulis (University of Thessaly, Larissa, Greece) and the C2C12
muscle cells were a gift from Professor Koutsilieris (National and
Kapodistrian University of Athens, Athens, Greece). All cells were
cultured in normal DMEM, containing 10% (v/v) FBS, 2 mM
L-glutamine, 100 U/ml of penicillin and 100 U/ml of streptomycin
(all from Gibco, Paisley, UK) in plastic disposable tissue culture
flasks at 37°C in 5% carbon dioxide.
Cell viability assay
The effects of the grape stem extracts on cell
viability were assessed using the XTT assay kit (Roche Diagnostics)
as previously described (21).
Briefly, the cells were subcultured in a 96-well plate with
1×104 cells per well in DMEM. After 24 h of incubation,
the cells were treated with increasing concentrations of the
extracts in serum-free DMEM for 24 h. A total of 50 µl of
XTT test solution, which was prepared by mixing 50 µl of
XTT-labeling reagent with 1 µl of electron coupling reagent,
was then added to each well. After 4 h of incubation, the
absorbance was measured at 450 nm and also at 690 nm as a reference
wavelength in a Bio-Tek EL×800 microplate reader (Winooski, VT,
USA). DMEM serum-free medium was used as a negative control. In
addition, the absorbance of each grape extract concentration alone
in DMEM serum-free medium and XTT test solution was tested at 450
nm. The absorbance values shown by the grape extracts alone were
subtracted from those derived from cells treated with the grape
extracts. Data were calculated as a percentage of inhibition using
the following formula: inhibition (%) = [(OD control − OD
sample)/OD control] ×100, where OD control and OD sample indicate
the optical density of the negative control and the tested
substances, respectively. All experiments were carried out in
triplicate and at least on 2 separate occasions.
DPPH radical scavenging assay
The DPPH radical scavenging activity of the grape
stem extracts was evaluated as previously described (15). Briefly, a 1.0 ml freshly prepared
methanolic solution of DPPH radical (100 µM) was mixed with
the test extract solution at different concentrations. The contents
were vigorously mixed, incubated at room temperature in the dark
for 20 min and the absorbance was read at 517 nm. In each
experiment, the test extract alone in methanol was used as a blank
and DPPH radical alone in methanol was used as a control. The
percentage of inhibition of DPPH radical by the tested extracts was
calculated according to the following equation: inhibition (%) =
[(Α control − Α sample)/Α control] ×100, where A control and A
sample are the absorbance values of the control and the test
sample, respectively. Moreover, in order to compare the radical
scavenging efficiency of the extracts, the IC50 value
showing the concentration that caused 50% scavenging of DPPH
radicals was calculated from the graph plotted inhibition
percentage against extract concentration. All experiments were
carried out in triplicate and at least on 2 separate occasions.
ABTS●+ radical scavenging
assay
ABTS●+ radical scavenging activity was
measured as described previously (15). In brief, ABTS●+ radicals
were produced by mixing 2 mM ABTS with 30 µM
H2O2 and 6 µM HRP enzyme in 50 mM
phosphate-buffered saline (PBS, pH 7.5). Immediately following the
addition of the HRP enzyme, the contents were vigorously mixed,
incubated at room temperature in the dark and the reaction was
monitored at 730 nm until stable absorbance was obtained.
Subsequenlty, 10 µL of different extract concentrations were
added to the reaction mixture and the decrease in absorbance at 730
nm was determined. In each experiment, the test extract alone
containing 1 mM ABTS and 30 µM H2O2 in
50 mM (PBS, pH 7.5) was used as a blank, while the formed
ABTS●+ radical solution alone with 10 µL
H2O was used as a control. The inhibition percentage and
the IC50 values were determined as described above for
the DPPH method. All experiments were carried out in triplicate and
at least on 2 separate occasions.
Reducing power assay
Reducing power was determined as previously
described (28). Briefly, the
extracts were dissolved in phosphate buffer (0.2 M, pH 6.6) at
different concentrations. An aliquot (2.5 ml) of the sample
solution was added to 2.5 ml of potassium ferricyanide (1%) and
incubated at 50°C for 20 min. The samples were cooled on ice for 5
min. Subsequenlty, 2.5 ml TCA (10%) were added and the samples were
centrifuged at 3,000 rpm for 10 min at 25°C. To the supernatant
(2.5 ml), 2.5 ml deionized water and 500 µl ferric chloride
(0.1%) were added and the samples were incubated at room
temperature for 10 min. The absorbance was measured at 700 nm. All
experiments were carried out in triplicate and at least on 2
separate occasions.
Treatment of EA.hy926 and C2C12 cells
with the grape stem extracts
The cells were seeded in culture flasks and
incubated for 24 h. The medium was then removed and replaced with
serum-free medium containing the grape extracts at non-cytotoxic
concentrations. Since one of the main purposes of the study was to
examine the effects of the polyphenolic composition on the
antioxidant activity of the extracts, the selected concentrations
for all three tested extracts had the same total polyphenolic
content and were expressed as 'µg of polyphenols/ml'. Thus,
the selected concentrations used were 0.20 µg of
polyphenols/ml for the EA.hy926 cells and 0.95 µg of
polyphenols/ml for the C2C12 cells. The cells were treated with the
extracts for 24 h and were then trypsinized, collected and
centrifuged twice at 300 × g for 10 min at 5°C. At the end of the
first centrifugation, the supernatant fluid was discarded and the
cellular pellet was resuspended in PBS. After the second
centrifugation, the cell pellet was collected and used for
measuring the levels of the oxidative stress markers, glutathione
(GSH), ROS, thiobarbituric acid reactive substances (TBARS) and
protein carbonyl (CARB).
Flow cytometric analysis of GSH and ROS
levels
The intracellular GSH and ROS levels were assessed
by flow cytometry using mercury orange and DCF-DA, respectively as
described previously (21). In
particular, the fluorescent mercury orange binds directly to GSH,
whilst DCF-DA within cells is deacetylated by esterases, and is
further converted to fluorescent DCF by the oxidative action of
ROS. A 400 µM stock solution of mercury orange was prepared
in acetone and stored at 4°C, while a fresh 400 µM stock
solution of DCF-DA was prepared in methanol. To assess the GSH and
ROS levels, the cells were resuspended in PBS at 1×106
cells per ml and incubated in the presence of mercury orange (40
µM) or DCF-DA (10 µM) in the dark at 37°C for 30 min.
The cells were then washed, resuspended in PBS and submitted to
flow cytometric analysis using a FACScan flow cytometer
(Becton-Dickinson, Franklin Lakes NJ, USA) with excitation and
emission wavelengths at 488 and 530 nm for ROS, and at 488 and 580
nm for GSH. In addition, forward angle and right angle light
scattering indicating the cell size and cell internal complexity,
respectively were measured. The cells were analysed at a flow rate
of 1,000 events per second. Analyses were performed on 10,000 cells
per sample and fluorescence intensities were measured on a
logarithmic scale of 4 decades of the log of fluorescence. Data was
analysed using BD Cell Quest software (Becton-Dickinson). Each
experiment was repeated at least 3 times.
Assessment of TBARS levels by
spectrophotometry
For the determination of TBARS levels, the cells
were resuspended in PBS at 1×106 cells/ml. Subsequently,
a slightly modified assay as desribed in the study by Keles et
al (29) was used as
previously described (21). A
total of 400 µl of cell suspension or 400 µl of PBS
for the blank was mixed with 500 µl of 35% TCA and 500
µl of tris(hydroxymethyl)aminomethane hydrochloride
(Tris-HCl) (200 mM, pH 7.4) and incubated for 10 min at room
temperature. Afterwards, 1 ml solution consisting of 2 M
Na2SO4 and 55 mM thiobarbituric acid was
added and the samples were incubated at 95°C for 45 min. The
samples were cooled on ice for 5 min and vortexed following the
addition of 1 ml of 70% TCA. The samples were then centrifuged at
15,000 × g for 3 min and the absorbance of the supernatant was read
at 530 nm. The assay requires >30 µg absolute amount of
protein in the test sample. The total protein concentration in the
cell suspension was assayed using a Bradford reagent from
Sigma-Aldrich. The calculation of the TBARS concentration was based
on the molar extinction co-efficient of malondialdehyde. Each
experiment was repeated at least 3 times.
Assessment of CARB levels by
spectrophotometry
For the determination of CARB levels, the cells were
resuspended in PBS at 1×106 cells/ml and then lysed by
sonication. Subsquently, a modified method as described in the
study by Patsoukis et al (30) was used as previously described
(21). In brief, 200 µL of
20% TCA were added to 200 µl of cell lysate, and this
mixture was incubated in an ice bath for 15 min and centrifuged at
15,000 × g for 5 min at 4°C. The supernatant fraction was
discarded. In addition, 500 µl of 10 mM
2,4-dini-trophenylhydrazine (DNPH; in 2.5 N HCl) for the sample or
500 µl of 2.5 N HCl for the blank were added to the pellet.
The samples were incubated in the dark at room temperature for 1 h,
with intermittent mixing every 15 min and were centrifuged at
15,000 × g for 5 min at 4°C. The supernatant fraction was
discarded, and 1 ml of 10% TCA was added to the pellet which was
mixed and centrifuged at 15,000 × g for 5 min at 4°C. The
supernatant fraction was discarded and 1 ml of ethanol-ethyl
acetate (1:1 v/v) was added, vortexed and centrifuged at 15,000 × g
for 5 min at 4°C. This washing step was repeated twice. The
supernatant fraction was discarded, and 1 ml of 5 M urea (pH 2.3)
was added, vortexed and incubated at 37°C for 15 min. The samples
were centrifuged at 15,000 × g for 3 min at 4°C, and the absorbance
was read at 375 nm. The calculation of the protein carbonyl
concentration was based on the molar extinction co-efficient of
DNPH. The assay requires >30 µg absolute amount of
protein in the test sample. The total plasma protein concentration
was evaluated using Bradford assay. Each experiment was repeated at
least 3 times.
Statistical analysis
Data were analyzed by one-way ANOVA followed by
Tukey's test for multiple pairwise comparisons. The level of
statistical significance was set at p<0.05. For all statistical
analyses, SPSS software, version 13.0 (SPSS Inc., Chicago, IL, USA)
was used. Data are presented as the means ± SEM.
Results
Polyphenolic composition of the grape
stem extracts
The TPC and the polyphenolic composition of the
extracts were identified by HPLC and are presented in Table I. The TPC values were similar
between the three extracts and ranged from 357 to 378 mg gallic
acid/g dried extract. These TPC values were similar to those
observed in one of our previous studies (15). Moreover, as we have previously
found in other grape stem extracts (15,22),
the extracts contained different classes of polyphenols, such as
phenolic acids (i.e., gallic acid, caffeic acid, coumaric acid,
ferulic acid and syringic acid), flavanols [(+)-catechin and
(−)-epicatechin], flavonols (quercetin, rutin and kaempferol) and
stilbenes (trans-resveratrol) (Table I). The amounts of some polyphenols
did not differ significantly between the three extracts. These
polyphenols were syringic acid (13.60–17.44 mg/g dried extract),
(+)-catechin (8.49–12.49 mg/g dried extract) and rutin (12.06–15.93
mg/g dried extract) (Table I).
However, the amounts of the other polyphenols presented marked
differences between the three tested extracts. Thus, their ranges
were 3.6-fold for gallic acid (7.39–26.96 mg/g dried extract),
4.2-fold for caffeic acid (0.54–2.32 mg/g dried extract), 1.9-fold
for (−)-epicatechin (9.83–19.13 mg/g dried extract), 2.1-fold for
coumaric acid (0.60–1.31 mg/g dried extract), 3.7-fold for ferulic
acid (0.97–3.59 mg/g dried extract), 3.5-fold for
trans-resveratrol (5.97–21.43 mg/g dried extract), 4.3-fold
for quercetin (3.94–17.33 mg/g dried extract) and 4.9-fold for
kaempferol (0.74–3.63 mg/g dried extract) (Table I).
 | Table IPolyphenolic composition of the grape
stem extracts. |
Table I
Polyphenolic composition of the grape
stem extracts.
| Grape variety | Mavrotragano | Mandilaria | Moshomavro |
|---|
| Gallic acid | 11.48a | 26.96a | 7.39a |
| Syringic acid | 17.44 | 13.60 | 15.98 |
| Caffeic acid | 0.54 | 2.32 | 0.64 |
| (+)-Catechin | 12.18 | 12.49 | 8.49 |
|
(−)-Epicatechin | 19.13 | 9.83 | 12.63 |
| Coumaric acid | 0.75 | 1.31 | 0.60 |
| Ferulic acid | 3.59 | 4.10 | 0.97 |
| Rutin | 15.93 | 12.06 | 14.81 |
|
trans-resveratrol | 9.09 | 21.43 | 5.97 |
| Quercetin | 3.94 | 17.33 | 8.01 |
| Kaempferol | 0.74 | 3.63 | 1.83 |
| TPCb | 357 | 378 | 373 |
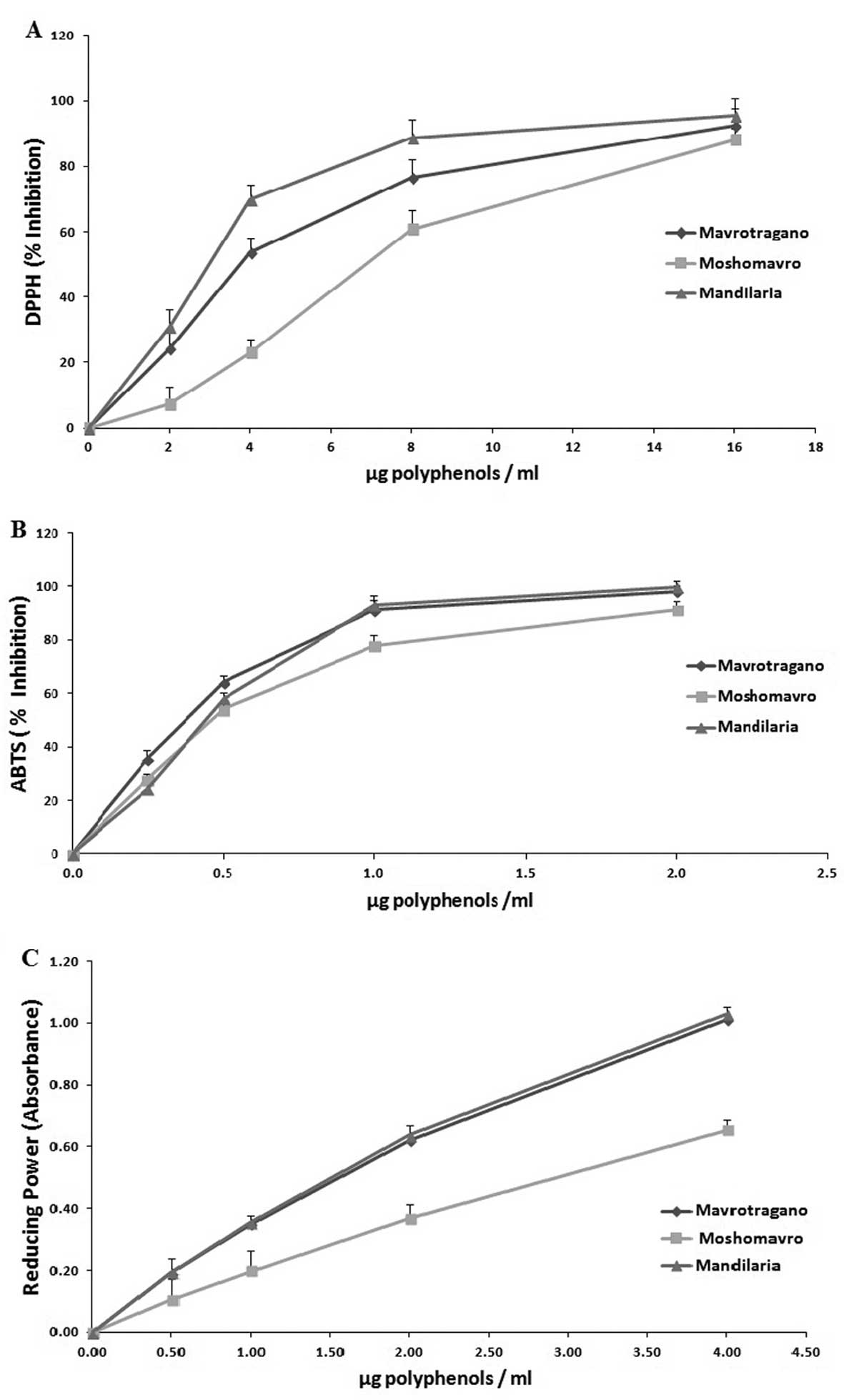
Scavenging of free radicals
All the extracts were examined for their free
radical scavenging activity. The results revealed that all the
extracts scavenged DPPH● and ABTS●+ radicals
in a dose-dependent manner at low concentrations (Fig. 1A and B). In order to compare the
radical scavenging efficiency of the extracts, the IC50
value indicating the concentration that caused the scavenging of
50% of DPPH● and ABTS●+ radicals was
calculated; graphs were created to plot the percentage of radical
scavenging capacity against the extract concentration. The
IC50 values calcuated from the DPPH radical scavenging
assay were in the following order: Mandilaria extract (2.8±0.2
µg polyphenols/ml) > Mavrotragano extract (3.6±0.17
µg polyphenols/ml) > Moshomavro extract (6.5±0.23
µg polyphenols/ml) (Table
II). In the ABTS●+ radical scavenging assay, the
order of potency of the extracts according to their IC50
values was: Mandilaria extract (0.42±0.08 µg polyphenols/ml)
> Mavrotragano extract (0.71±0.10 µg polyphenols/ml) >
Moshomavro extract (0.90±0.11 µg polyphenols/ml) (Table II).
 | Table IIFree radical scavenging activity
against DPPH● and ABTS●+ radicals as well as
reducing power of grape stem extracts. |
Table II
Free radical scavenging activity
against DPPH● and ABTS●+ radicals as well as
reducing power of grape stem extracts.
| Grape variety |
DPPH●a
IC50b |
ABTS●+b
IC50b | Reducing
powera
RP0.5AUb |
|---|
| Mandilaria | 2.8±0.2 | 0.42±0.08 | 1.46±0.12 |
| Mavrotragano | 3.6±0.17 | 0.71±0.10 | 1.47±0.21 |
| Moshomavro | 6.5±0.23 | 0.90±0.11 | 2.92±0.23 |
Reducing power
For the reducing power assay, the
RP0.5AU, the concentration that produces an absorbance
of 0.5 at 700 nm, was determined (Fig.
1C). The RP0.5AU values of the tested extracts were
in the following order: Mandilaria extract (1.46±0.12 µg
polyphenols/ml) > Mavrotragano extract (1.47±0.21 µg
polyphenols/ml) > Moshomavro extract (2.92±0.23 µg
polyphe-nols/ml) (Table II).
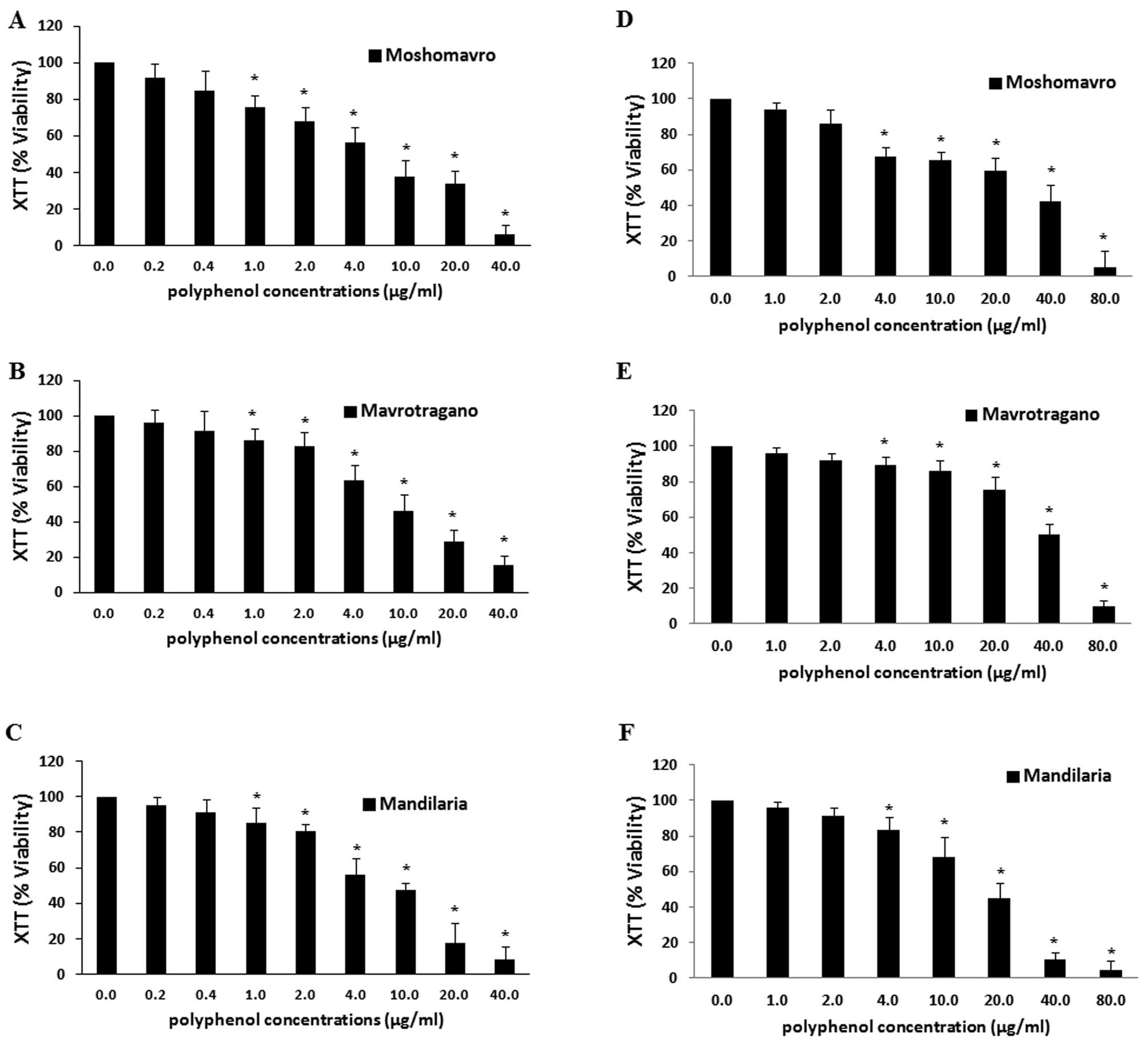
Effects of grape extracts on the
viability of C2C12 and EA.hy926 cells
XTT assay was used to assess the effects of the
grape extracts on the viability of the C2C12 and EA.hy926 cells in
order to select the non-cytotoxic concentrations for use in our
experiments. The results revealed that all three extracts exhibited
cytotoxicity at concentrations >1 µg polyphenols/ml in
the EA.hy926 cells and >4 µg polyphenols/ml in the C2C12
cells (Fig. 2). Thus, the
concentrations that were selected were 0.20 and 0.95 µg
polyphenols/ml for the EA.hy926 and C2C12 cells, respectively.
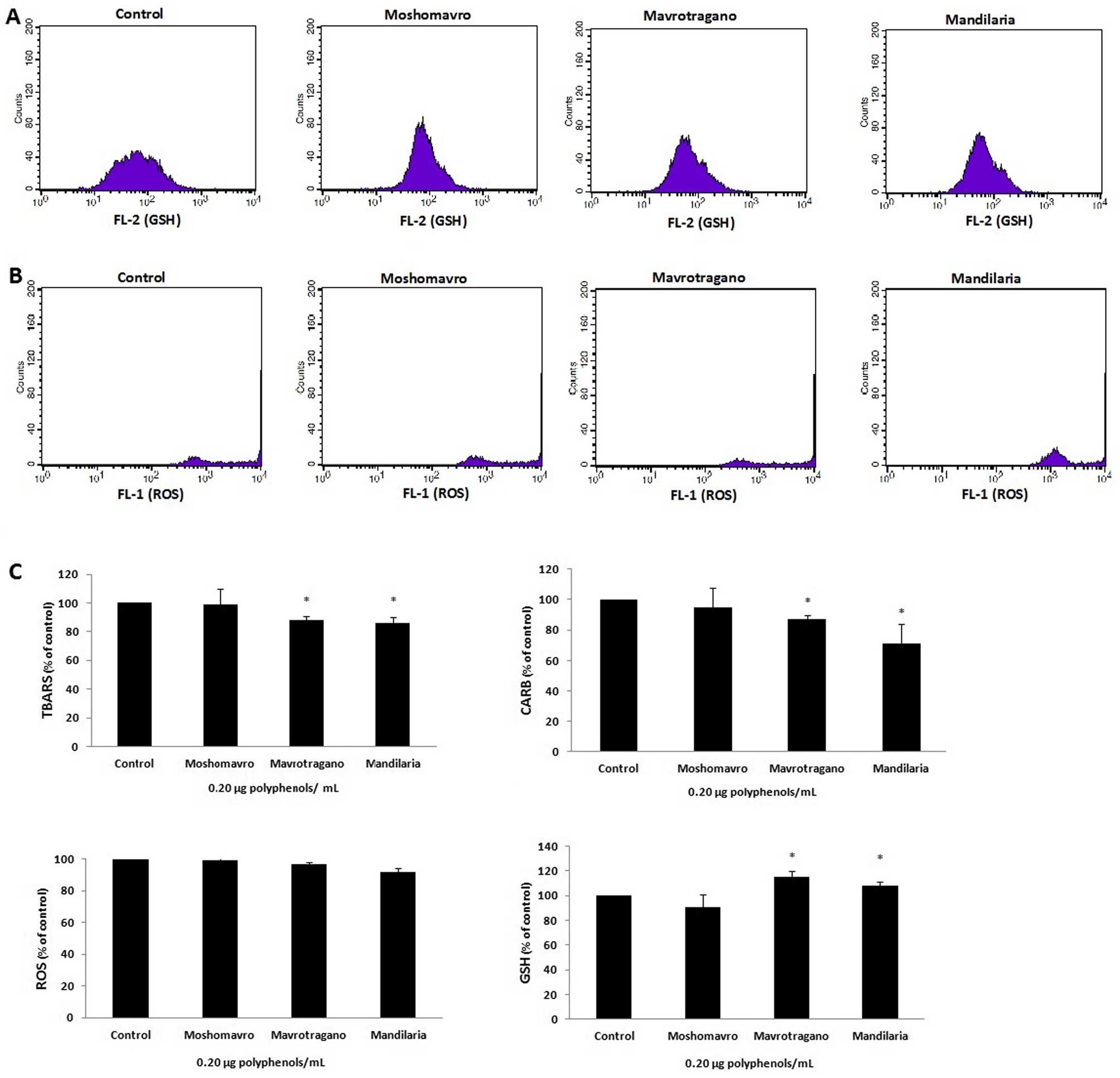
Effects of treatment with grape extracts
on the levels of oxidative stress markers in EA.hy926 cells
The levels of oxidative stress markers were measured
in order to assess effects of the extracts on the redox status of
EA.hy926cells. The ROS and GSH levels were measured by flow
cytometry using DCF-DA and mercury orange, respectively. Histograms
demonstrating the cell counts versus the fluorescence of the
mercury orange or DCF-DA are shown in Fig. 3A and B, respectively. The mean
fluorescence intensity values were evaluated using BD CellQuest
software and are expressed as a percentage of the controls
(untreated cells). None of the tested extracts had any effect on
the ROS levels (Fig. 3C).
Moreover, the Mavrotragano and Mandilaria extracts significantly
increased the GSH levels by 20.2±1.5 and 15.8±4.1%, respectively
compared to the controls (Fig.
3C). On the contrary, Moshomavro extract did not affect the GSH
levels.
Furthermore, lipid peroxidation and protein
oxidation were assessed by measuring spectrophotometrically the
levels of TBARS and CARB, respectively (Fig. 3C). Specifically, Mandilaria extract
significantly reduced lipid peroxidation by 14.8±2.3% compared to
the controls, Mavrotragano extract reduced lipid peroxidation by
12.5±1.9%, while Moshomavro extract had no effect. Similar to lipid
peroxidation, treatment with Mandilaria extract exerted the most
significant reducing effect on CARB levels by 25.9±6.3% (Fig. 3C). Mavrotragano extract reduced the
CARB levels by 16.6±1.3% and Moshomavro extract did not have any
effect on these levels.
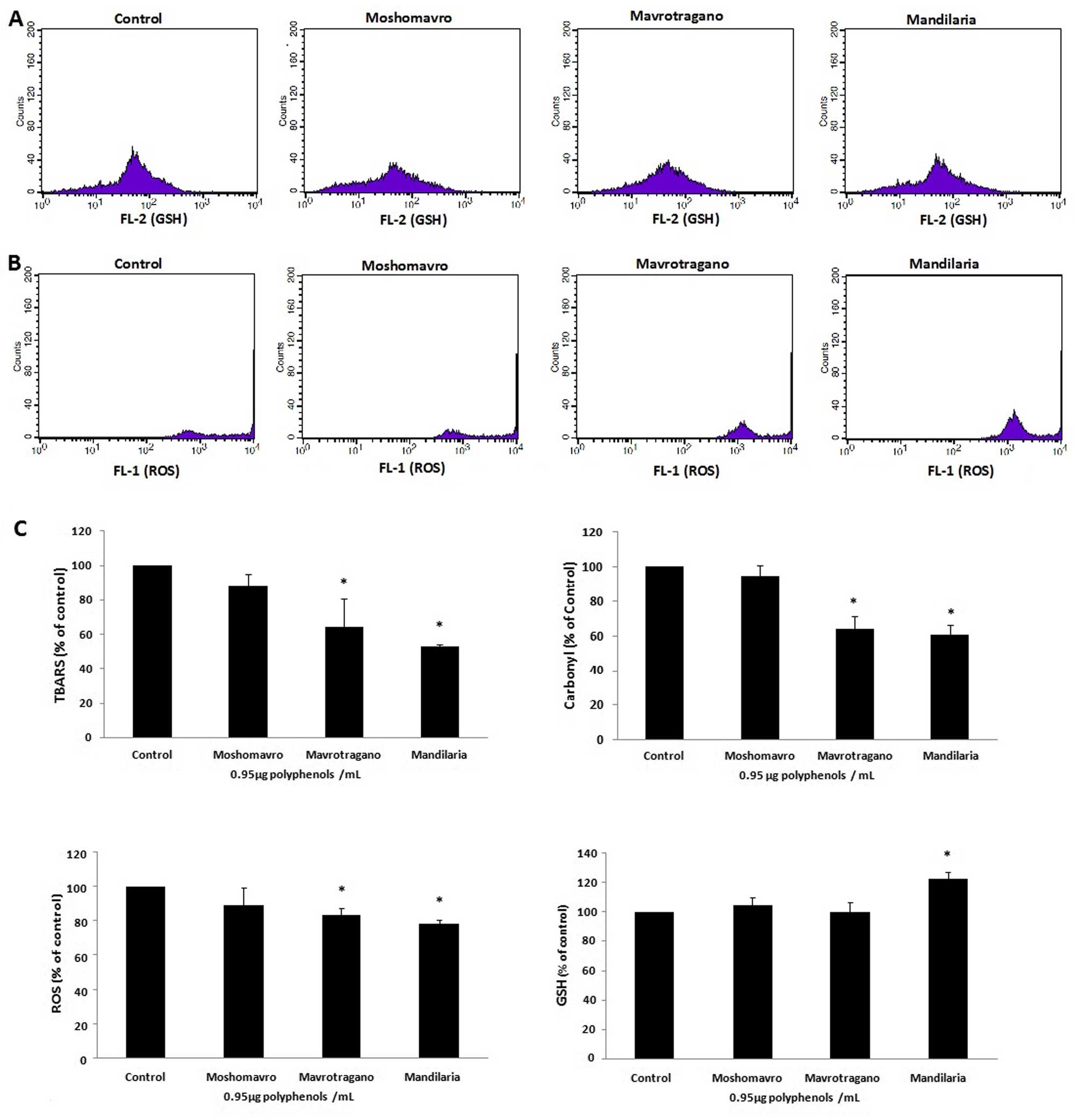
Effects of treatment with grape extracts
on the levels of oxidative stress markers in C2C12 cells
The histograms demonstrating the cell counts versus
the fluorescence of mercury orange or DCF-DA in the C2C12 cells
following treatment with the grape extracts are shown in Fig. 4A and B, respectively. The
Mandilaria and Mavrotragano extracts significantly decreased the
ROS levels by 21.8±2 and 16.5±3.7%, respectively, while Moshomavro
extract had no effect (Fig. 4C).
Finally, Mandilaria extract significantly increased the GSH levels
by 22.6±4.5% compared to the controls, while the other two extracts
did not have any affect on these levels (Fig. 4C).
Moreover, Mandilaria extract significantly reduced
the TBARS levels by 47.3±1.2%, and Mavrotragano extract by
36.2±9.6%, while Moshomavro extract had no effect compared to the
controls in the C2C12 cells (Fig.
4C). In addition, Mandilaria extract exerted the greatest
reducing effect on CARB levels, decreasing them by 39.0±5.1%
(Fig. 4C). Mavrotragano extract
reduced the CARB levels by 35.9±7.1% and Moshomavro extract did not
have any effect on these levels (Fig.
4C).
Discussion
Grape extracts and wine have been the center of
attention of researchers, demonstrating their beneficial effects on
human health (14). For example,
food supplements termed as 'grape seed extracts' are consumed
worldwide for their beneficial effects on human health. However,
the majority of studies on grapes refer to extracts from seeds,
skins and pomace (20,31,32).
Of note, in previous studies of ours, we demonstrated that grape
stems, a by-product produced in large amounts (approximately 5% of
the original grape material) during the vinification process, which
has been little investigated, contained bioactive phytochemicals
and presented important biological activities (15,24).
In particular, we demonstrated that grape stem extracts exhibited
strong antioxidant activity in vitro comparable even to that
of grape seed extracts (15). In
the present study, we investigated at a cellular level (i.e., in
endothelial and muscle cells) the antioxidant activity of stem
extracts from Greek grape varieties.
Before examining the effects of grape stem extracts
on the redox status of cells, their potency of scavenging free
radicals in vitro was assessed. As also demonstrated in a
previous study of ours (15), in
this study, the tested grape stem extracts were very potent
scavengers of DPPH● and ABTS●+ radicals. In
both DPPH and ABTS assays, Mandilaria extract was the most potent
and Moshomavro extract the weakest. Thus, although the three tested
extracts had similar TPC values, their ability to scavenge free
radicals varied greatly. Specifically, Mandilaria extract was
almost 2-fold more potent as scavenger against DPPH● and
ABTS●+ radicals than Moshomavro extract. This
discordance between the TPC values and free radical scavenging
activity is in agreement with the results of our previous study in
which the TPC values of 12 grape stem extracts had a weak
correlation with their free radical scavenging activity (15). This lack of correlation between the
TPC values and free radical scavenging activity suggests that the
qualitative polyphenolic composition of the stem extracts may be
more important than their quantitative polyphenolic content for
their antioxidant potency. For example, it has been suggested that
polyphenolic extracts exhibiting high antioxidant activity have a
high content of special polyphenols, such as
trans-resveratrol, gallic acid, quercetin, (+)-catechin,
(−)-epicatechin and rutin that are very labile to oxidation, and
thus can potentially scavenge free radicals before they are able to
attack and damage other macromolecules (33–35).
Indeed, Mandilaria extract contained at least 2-fold greater
amounts of trans-resveratrol, gallic acid and quercetin than
Moshomavro extract.
Moreover, the reducing power of the grape stem
extracts was examined. The reducing power of a substance is
associated with its antioxidant activity, since it indicates that
the substances are electron donors and can reduce the oxidized
intermediates of the lipid peroxidation processes, so that they can
act as primary and secondary antioxidants (36). All the three grape stem extracts at
increasing concentrations showed increased reducing power. As in
free radical scavenging assays, Mandilaria extract exhibited the
greatest reducing power, but in this case, along with Mavrotragano
extract. Again, it was observed that apart from the similar TPC
values of the grape stem extracts, the Mandilaria and Mavrotragano
extracts had 2-fold greater reducing power than the Moshomavro
extract. This difference may be attributed to the differences in
the polyphenolic composition of the tested extracts. For example, a
previous study reported that proanthocyanidins, a class of
oligomeric flavonoids, of grape stem extracts were mainly
responsible for reducing power possibly due to the relatively high
number of hydroxyl groups that their monomer units contain
(37).
As mentioned above, in this study, the antioxidant
activity of grape stem extracts was also examined at a cellular
level, specifically in endothelial and muscle cells. In a previous
study, we demonstrated that grape extracts from pomace improved the
redox status of EA.hy926 and C2C12 cells (21). Pomace is also a by-product of the
winemaking process, but it includes, apart from the stems, the
seeds and skins as well. To the best of our knowledge, this is the
first study on the effects of grape extracts from stems on the
redox status of endothelial and muscle cells. Two of the tested
extracts, the Mandilaria and Mavrotragano extracts, improved the
redox status of the endothelial and muscle cells, since they
reduced lipid peroxidation, as shown by the decrease in TBARS
levels. Moreover, the Mandilaria and Mavrotragano extracts
diminished protein oxidation, as shown by the decrease in CARB
levels in the EA.hy926 and C2C12 cells. However, the Moshomavro
extract was the only one of the tested extracts that did not
decrease the CARB and TBARS levels compared to the untreated cells
in either the EA.hy926 or C2C12 cells.
The reduction of protein oxidation and lipid
peroxidation by the Mandilaria and Mavrotragano extracts may be
attributed to their ability to reduce ROS levels, particularly in
muscle cells. The direct free radical scavenging activity and
reducing power of the Mandilaria and Mavrotragano extracts may
account at least in part for their capacity to decrease ROS levels
in muscle cells. On the contrary, these two extracts did not affect
the ROS levels in EA.hy926 endothelial cells, possibly due to the
fact that the concentrations of the extracts used in these cells
were lower than those used in C2C12 muscle cells. The Moshomavro
extract did not alter the ROS levels in either the endothelial or
muscle cells. Remarkably, Moshomavro extract was also the least
potent extract in free radical scavenging and reducing power
assays.
The decrease in ROS-induced damage of lipids and
proteins by the Mandilaria and Mavrotragano extracts in endothelial
cells may also be explained by their ability to enhance antioxidant
mechanisms, such as GSH. However, in the muscle cells, only the
Mandilaria extract increased GSH levels, indicating that this
extract was the most potent for GSH induction. The Moshomavro
extract, which did not decrease the TBARS, CARB and ROS levels was
also unable to increase the GSH levels in either the endothelial or
in muscle cells. The increase in GSH levels by grape stem extracts
is of particular importance, since GSH, the most abundant
non-protein thiol source in cells, is one of the major
intracellular antioxidant molecules protecting against oxidative
stress (38). In previous studies
of ours, we demonstrated that GSH was a key molecule for the
antioxidant activity of a grape pomace extract in both EA.hy926 and
C2C12 cells (20,21). Specifically, grape pomace extract
was shown to increase GSH levels and the expression or activity of
enzymes involved in synthesis (γ-glutamylcysteine synthetase) and
metabolism (e.g., glutathione S-transferase) of GSH (20,21).
As the results from the assessment of the oxidative
stress markers showed, there were differences in the effects of the
tested stem extracts on the redox status of the cells. These
differences of the extracts were profoundly attributed to their
polyphenolic composition. This conclusion is supported by the fact
that the concentrations of all the three extracts had the same TPC
values in the cellular experiments. Thus, it was excluded that the
observed differences in the antioxidant activity between the
extracts may be due to their different amount of polyphenols. Among
the tested extracts, the Moshomavro extract exhibited much less
potency for improving the redox status of the cells compared to the
Mandilaria and Mavtotragano extracts. The Moshomavro extract also
had less amounts of gallic acid, (+)-catechin, ferulic acid and
trans-resveratrol than the Mandilaria and Mavtotragano
extracts, and thus these polyphenols may account, at least in part,
for the higher antioxidant activity of the latter two extracts. For
example, a previous study demonstrated that the
trans-resveratrol increased GSH levels and decreased
oxidative stress-induced intracellular ROS and malondialdehyde
(MDA) production in vascular smooth muscle cells (39). Gallic acid has also been shown to
reduce ROS levels in vascular smooth muscle cells and their
corresponding mediator p47phox, a molecule translocating to the
membrane and inducing O2●− production
(40). Moreover, in rat vascular
smooth muscle cells, ferulic acid has been shown to decrease lipid
peroxidation and increase the activity of antioxidant enzymes
(e.g., superoxide dismutase) (41). Between the Mandilaria and
Mavrotragano extracts, the former exhibited more potent antioxidant
activity. As regards the differences in the polyphenolic
composition between these two extracts, Mandilaria had higher
amounts of caffeic acid, coumaric acid, quercetin and kaempferol.
Among these polyphenols, caffeic acid has been reported to decrease
O2●− levels at very low concentrations
(100–1,000 nM) in human umbilical vein-derived endothelial cells
under oxidative stress conditions (42). Furthermore, quercetin and
kaempferol have been shown to reduce O2●−
levels in rat vascular smooth muscle cells through the inhibition
of nicotinamide adenine dinucleotide phosphate (NADPH)-oxidase
activity, the main source of O2●− in the
vessel wall (43).
In conclusion, to the best of our knowledge, the
findings of the present study indicate for the first time that
grape stem extracts at low concentrations improve the redox status
of endothelial and muscle cells. Therefore, grape stem extracts may
be used, for instance, in the development of antioxidant food
supplements used for counteracting oxidative stress-induced
pathological conditions in endothelial or skeletal muscles.
However, it was shown that the antioxidant activity of the grape
stem extracts depended on their qualitative polyphenolic
composition. For example, considerable amounts of polyphenols, such
as trans-resveratrol, gallic acid, (+)-catechin, ferulic
acid, caffeic acid, quercetin, coumaric acid and kaempferol may be
essential for the observed antioxidant activity of the stem
extracts. The knowledge of the polyphenolic composition that is
responsible for a strong antioxidant activity would help to assess
the quality of grape stem extracts intended for uses as antioxidant
supplements.
Acknowledgments
This study was funded in part by the MSc programmes
'Biotechnology-Nutrition and Environment' and 'Molecular Biology
and Genetics Applications' in the Department of Biochemistry and
Biotechnology at the University of Thessaly.
Abbreviations:
|
ABTS
|
2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid)
|
|
CARB
|
protein carbonyl
|
|
DMEM
|
Dulbecco's modified Eagle's medium
|
|
DCF-DA
|
2,7-dichlorofluorescein diacetate
|
|
DNPH
|
2,4-dinitrophenylhydrazine
|
|
DPPH
|
2,2-diphenyl-1-picryl-hydrazyl
|
|
EDTA
|
ethylenediaminetetraacetic acid
|
|
FBS
|
fetal bovine serum
|
|
GSH
|
glutathione
|
|
H2O2
|
hydrogen peroxide
|
|
HRP
|
horseradish peroxidase
|
|
NADH
|
2-deoxyribose, nicotinamide adenine
dinucleotide
|
|
NBT
|
nitroblue tetrazolium
|
|
O2●−
|
superoxide radical
|
|
OH●
|
hydroxyl radical
|
|
PBS
|
phosphate-buffered saline
|
|
PMS
|
phenazine methosulfate
|
|
ROO●
|
peroxyl radical
|
|
ROS
|
reactive oxygen species
|
|
TBA
|
thiobarbituric acid
|
|
TBARS
|
thiobarbituric acid reactive
substances
|
|
TCA
|
trichloroacetic acid
|
|
TPC
|
total polyphenolic content
|
References
|
1
|
Halliwell B: Free radicals and other
reactive species in disease. Nature Encyclopaedia of Life Sciences.
J. Wiley and Sons: Nature Publishing Group; New York: 2001
|
|
2
|
Reid MB: Free radicals and muscle fatigue:
Of ROS, canaries, and the IOC. Free Radic Biol Med. 44:169–179.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nunes-Silva A, Bernardes PT, Rezende BM,
Lopes F, Gomes EC, Marques PE, Lima PM, Coimbra CC, Menezes GB,
Teixeira MM, et al: Treadmill exercise induces neutrophil
recruitment into muscle tissue in a reactive oxygen
species-dependent manner. An intravital microscopy study. PLoS One.
9:e964642014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Di Meo S and Venditti P: Mitochondria in
exercise-induced oxidative stress. Biol Signals Recept. 10:125–140.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Deanfield JE, Halcox JP and Rabelink TJ:
Endothelial function and dysfunction: Testing and clinical
relevance. Circulation. 115:1285–1295. 2007.PubMed/NCBI
|
|
6
|
Victor VM, Rocha M, Solá E, Bañuls C,
Garcia-Malpartida K and Hernández-Mijares A: Oxidative stress,
endothelial dysfunction and atherosclerosis. Curr Pharm Des.
15:2988–3002. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Higashi Y, Noma K, Yoshizumi M and Kihara
Y: Endothelial function and oxidative stress in cardiovascular
diseases. Circ J. 73:411–418. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kokura S, Wolf RE, Yoshikawa T, Granger DN
and Aw TY: Molecular mechanisms of neutrophil-endothelial cell
adhesion induced by redox imbalance. Circ Res. 84:516–524. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zou Y, Yoon S, Jung KJ, Kim CH, Son TG,
Kim MS, Kim YJ, Lee J, Yu BP and Chung HY: Upregulation of aortic
adhesion molecules during aging. J Gerontol A Biol Sci Med Sci.
61:232–244. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rhee SG: Cell signaling.
H2O2, a necessary evil for cell signaling.
Science. 312:1882–1883. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Woywodt A, Bahlmann FH, De Groot K, Haller
H and Haubitz M: Circulating endothelial cells: Life, death,
detachment and repair of the endothelial cell layer. Nephrol Dial
Transplant. 17:1728–1730. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Matthaiou CM, Goutzourelas N, Stagos D,
Sarafoglou E, Jamurtas A, Koulocheri SD, Haroutounian SA, Tsatsakis
AM and Kouretas D: Pomegranate juice consumption increases GSH
levels and reduces lipid and protein oxidation in human blood. Food
Chem Toxicol. 73:1–6. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kerasioti E, Kiskini A, Veskoukis A,
Jamurtas A, Tsitsimpikou C, Tsatsakis AM, Koutedakis Y, Stagos D,
Kouretas D and Karathanos V: Effect of a special
carbohydrate-protein cake on oxidative stress markers after
exhaustive cycling in humans. Food Chem Toxicol. 50:2805–2810.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bagchi D, Swaroop A, Preuss HG and Bagchi
M: Free radical scavenging, antioxidant and cancer chemoprevention
by grape seed proanthocyanidin: An overview. Mutat Res. 768:69–73.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Apostolou A, Stagos D, Galitsiou E, Spyrou
A, Haroutounian S, Portesis N, Trizoglou I, Wallace Hayes A,
Tsatsakis AM and Kouretas D: Assessment of polyphenolic content,
antioxidant activity, protection against ROS-induced DNA damage and
anticancer activity of Vitis vinifera stem extracts. Food Chem
Toxicol. 61:60–68. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Spanou C, Veskoukis AS, Stagos D, Liadaki
K, Anastasiadi M, Haroutounian SA, Tsouka M, Tzanakouli E and
Kouretas D: Effects of grape extracts on the in vitro activity of
enzymes involved in oxidative stress regulation. In Vivo.
25:657–662. 2011.PubMed/NCBI
|
|
17
|
Stagos D, Kazantzoglou G, Magiatis P,
Mitaku S, Anagnostopoulos K and Kouretas D: Effects of plant
phenolics and grape extracts from Greek varieties of Vitis vinifera
on mitomycin C and topoisomerase I-induced nicking of DNA. Int J
Mol Med. 15:1013–1022. 2005.PubMed/NCBI
|
|
18
|
Stagos D, Kazantzoglou G, Theofanidou D,
Kakalopoulou G, Magiatis P, Mitaku S and Kouretas D: Activity of
grape extracts from Greek varieties of Vitis vinifera against
mutagenicity induced by bleomycin and hydrogen peroxide in
Salmonella typhimurium strain TA102. Mutat Res. 609:165–175. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Stagos D, Spanou C, Margariti M,
Stathopoulos C, Mamuris Z, Kazantzoglou G, Magiatis P and Kouretas
D: Cytogenetic effects of grape extracts (Vitis vinifera) and
polyphenols on mitomycin C-induced sister chromatid exchanges
(SCEs) in human blood lymphocytes. J Agric Food Chem. 55:5246–5252.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Goutzourelas N, Stagos D, Housmekeridou A,
Karapouliou C, Kerasioti E, Aligiannis N, Skaltsounis AL, Spandidos
DA, Tsatsakis AM and Kouretas D: Grape pomace extract exerts
antioxidant effects through an increase in GCS levels and GST
activity in muscle and endothelial cells. Int J Mol Med.
36:433–441. 2015.PubMed/NCBI
|
|
21
|
Goutzourelas N, Stagos D, Demertzis N,
Mavridou P, Karterolioti H, Georgadakis S, Kerasioti E, Aligiannis
N, Skaltsounis L, Statiri A, et al: Effects of polyphenolic grape
extract on the oxidative status of muscle and endothelial cells.
Hum Exp Toxicol. 33:1099–1112. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Anastasiadi M, Chorianopoulos NG, Nychas
GJ and Haroutounian SA: Antilisterial activities of polyphenol-rich
extracts of grapes and vinification byproducts. J Agric Food Chem.
57:457–463. 2009. View Article : Google Scholar
|
|
23
|
Sahpazidou D, Geromichalos GD, Stagos D,
Apostolou A, Haroutounian SA, Tsatsakis AM, Tzanakakis GN, Hayes AW
and Kouretas D: Anticarcinogenic activity of polyphenolic extracts
from grape stems against breast, colon, renal and thyroid cancer
cells. Toxicol Lett. 230:218–224. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Stagos D, Apostolou A, Poulios E,
Kermeliotou E, Mpatzilioti A, Kreatsouli K, Koulocheri SD,
Haroutounian SA and Kouretas D: Antiangiogenic potential of grape
stem extract through inhibition of vascular endothelial growth
factor expression. J Physiol Pharmacol. 65:843–852. 2014.
|
|
25
|
Kantsadi AL, Apostolou A, Theofanous S,
Stravodimos GA, Kyriakis E, Gorgogietas VA, Chatzileontiadou DS,
Pegiou K, Skamnaki VT, Stagos D, et al: Biochemical and biological
assessment of the inhibitory potency of extracts from vinification
byproducts of Vitis vinifera extracts against glycogen
phosphorylase. Food Chem Toxicol. 67:35–43. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu L, Zhang Y, Deng J, Li H and Lu J:
Phenolic concentrations and antioxidant properties of wines made
from north american grapes grown in china. Molecules. 17:3304–3323.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Meng JF, Fang YL, Qin MY, Zhuang XF and
Zhang ZW: Varietal differences among the phenolic profiles and
antioxidant properties of four cultivars of spine grape (Vitis
davidii Foex) in Chongyi County (China). Food Chem. 134:2049–2056.
2012. View Article : Google Scholar
|
|
28
|
Kerasioti E, Stagos D, Priftis A,
Aivazidis S, Tsatsakis AM, Hayes AW and Kouretas D: Antioxidant
effects of whey protein on muscle C2C12 cells. Food Chem.
155:271–278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Keles MS, Taysi S, Sen N, Aksoy H and
Akçay F: Effect of corticosteroid therapy on serum and CSF
malondialdehyde and antioxidant proteins in multiple sclerosis. Can
J Neurol Sci. 28:141–143. 2001.PubMed/NCBI
|
|
30
|
Patsoukis N, Zervoudakis G, Panagopoulos
NT, Georgiou CD, Angelatou F and Matsokis NA: Thiol redox state
(TRS) and oxidative stress in the mouse hippocampus after
pentylenetetrazol-induced epileptic seizure. Neurosci Lett.
357:83–86. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Feringa HH, Laskey DA, Dickson JE and
Coleman CI: The effect of grape seed extract on cardiovascular risk
markers: A meta-analysis of randomized controlled trials. J Am Diet
Assoc. 111:1173–1181. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sri Harsha PS, Mesias M, Lavelli V and
Morales FJ: Grape skin extracts from winemaking by-products as a
source of trapping agents for reactive carbonyl species. J Sci Food
Agric. In press.
|
|
33
|
Alov P, Tsakovska I and Pajeva I:
Computational studies of free radical-scavenging properties of
phenolic compounds. Curr Top Med Chem. 15:85–104. 2015. View Article : Google Scholar :
|
|
34
|
Li AN, Li S, Zhang YJ, Xu XR, Chen YM and
Li HB: Resources and biological activities of natural polyphenols.
Nutrients. 6:6020–6047. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yoshimura M: Structure elucidation of
antioxidative polyphenols and their biological properties. Yakugaku
Zasshi. 134:957–964. 2014.In Japanese. View Article : Google Scholar
|
|
36
|
Chanda S and Dave R: In vitro models for
antioxidant activity evaluation and some medicinal plants
possessing antioxidant properties: An overview. Afr J Microbiol
Res. 3:981–996. 2009.
|
|
37
|
Karvela E, Makris DP, Kalogeropoulos N and
Karathanos VT: Deployment of response surface methodology to
optimise recovery of grape (Vitis vinifera) stem polyphenols.
Talanta. 79:1311–1321. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Livingstone C and Davis J: Targeting
therapeutics against glutathione depletion in diabetes and its
complications. Br J Diabetes Vasc Dis. 7:258–265. 2007. View Article : Google Scholar
|
|
39
|
Guo R, Li W, Liu B, Li S, Zhang B and Xu
Y: Resveratrol protects vascular smooth muscle cells against high
glucose-induced oxidative stress and cell proliferation in vitro.
Med Sci Monit Basic Res. 20:82–92. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yang HL, Huang PJ, Liu YR, Kumar KJ, Hsu
LS, Lu TL, Chia YC, Takajo T, Kazunori A and Hseu YC: Toona
sinensis inhibits LPS-induced inflammation and migration in
vascular smooth muscle cells via suppression of reactive oxygen
species and NF-κB signaling pathway. Oxid Med Cell Longev.
2014:9013152014. View Article : Google Scholar
|
|
41
|
Wu L, Li X, Li Y, Wang L, Tang Y and Xue
M: Proliferative inhibition of danxiongfang and its active
ingredients on rat vascular smooth muscle cell and protective
effect on the VSMC damage induced by hydrogen peroxide. J
Ethnopharmacol. 126:197–206. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Migliori M, Cantaluppi V, Mannari C,
Bertelli AA, Medica D, Quercia AD, Navarro V, Scatena A, Giovannini
L, Biancone L, et al: Caffeic acid, a phenol found in white wine,
modulates endothelial nitric oxide production and protects from
oxidative stress-associated endothelial cell injury. PLoS One.
10:e01175302015. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Jimenez R, Lopez-Sepulveda R, Romero M,
Toral M, Cogolludo A, Perez-Vizcaino F and Duarte J: Quercetin and
its metabolites inhibit the membrane NADPH oxidase activity in
vascular smooth muscle cells from normotensive and spontaneously
hypertensive rats. Food Funct. 6:409–414. 2015. View Article : Google Scholar : PubMed/NCBI
|