Introduction
Breast cancer, the second most common type of
malignancy worldwide, is the most frequent type of cancer among
women and accounts for 29% of all new cancer diagnoses in women
(1,2). Every year, ~1,300,000 women are newly
diagnosed with breast cancer, with a mortality rate of almost
40,000 (3). Breast cancer is a
heterogeneous and complex disease. The carcinogenesis of breast
cancer involves genetic alterations, epigenetic alterations and
environmental factors (4,5). Currently, the primary therapeutic
strategies for patients with early breast cancer are surgery,
followed by hormonal therapy, chemotherapeutic approaches,
radiotherapy and biological therapies. However, for patients with
advanced disease, it remains a prevalent and life threatening
malignancy (6,7). Although several oncogenic and
tumor-suppressive genes have been demonstrated to have important
functions in the carcinogenesis and progression of breast cancer,
the molecular mechanisms underlying this process remain to be fully
elucidated. Therefore, it is important to understand these
molecular mechanisms and to develop novel targeted therapies for
breast cancer.
In breast cancer, changes in the expression of
microRNAs (miRNAs) have been demonstrated (8–10).
miRNAs are a group of non protein-coding, signal-stranded, small
RNAs, which are ~19–25 nucleotides in length (11). miRNAs function as negative
regulators of the expression of target mRNAs, and are involved in
cell proliferation, differentiation, cell cycle, survival and all
fundamental cellular processes implicated in carcinogenesis
(12). Accumulating evidence
suggests that over one third of all human genes may be targeted by
miRNAs (13). miRNAs inhibit the
translation or degradation of target mRNAs through binding to the
complementary 3′ untranslated region of target mRNAs by
base-pairing (14). There is also
increasing evidence to suggest that specific miRNAs can be
downregulated or upregulated in the different types of tumor
(15–17). The upregulation of miRNAs can
result in the degradation of tumor suppressor genes, whereas the
downregulation of miRNAs can lead to an increase in the expression
of oncogens (18). Therefore,
identification of the targets of miRNAs is important to understand
the functions of miRNAs in tumorigenesis and cancer development. It
also suggests that miRNAs may be a target for breast cancer
therapy.
The expression and functions of miR-411 have been
investigated in several types of cancer. However, until now,
miR-411 has not been investigated in human breast cancer. In the
present study, the expression, biological functions and molecular
mechanisms of miR-411 in human breast cancer were examined. The
results showed that the expression of miR-411 was significantly
decreased in human breast cancer, and was associated with lymph
node metastasis and histological grade. In addition, miR-411
suppressed cell proliferation, migration and invasion by directly
targeting specificity protein 1 (SP1). These findings have
therapeutic implications, which may be exploited for the treatment
of human breast cancer.
Materials and methods
Clinical specimens
The present study was approved by the Protection of
Human Subjects Committee of The Second People's Hospital of
Shenzhen (Shenzhen, China). Written informed consent was obtained
from all patients (age range, 26–75 years; stage range, 50 cases of
stage I–II, 64 cases of stage III) involved in the study. In total,
114 pairs of human breast cancer tissues and their corresponding
adjacent non-neoplastic tissues were obtained with patients'
approval. The tissue samples were snap frozen in liquid nitrogen at
the time of surgery and stored at −80°C in a refrigerator.
Cell culture and transfection
The human breast cancer cell lines, MCF-7,
MDA-MB-231 and SKBR3, and normal mammary epithelial cell line,
MCF-10A, were purchased from American Type Culture Collection
(Manassas, VA, USA). The cells were maintained in RPMI-1640 medium
(Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
containing 10% fetal bovine serum (FBS; Gibco; Thermo Fisher
Scientific, Inc.), 100 IU/ml penicillin (Gibco; Thermo; Fisher
Scientific, Inc.) and 100 ug/ml streptomycin (Gibco; Thermo Fisher
Scientific, Inc.) in a humidified 5% CO2 cell incubator
at 37°C.
The mature miR-411 mimics, negative control (NC) and
the luciferase reporter plasmid were synthesized and verified by
GenePharma (Shanghai, China). The cells were seeded into a 6-well
plate and cultured in RPMI-1640 medium without antibiotics. When
the cell density reached 30–40%, tansfections were performed using
Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocol.
RNA isolation and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
analysis
Total RNA was isolated from homogenized tissues and
cells using TRIzol reagent (Invitrogen; Thermo Fisher Scientific,
Inc.). The RT process was performed using an M-MLV Reverse
Transcription system (Promega Corporation, Madison, WI, USA) in a
25 µl volume. The temperature protocol for RT was as
follows: 95°C for 2 min; 20 cycles at 94°C for 1 min, 55°C for 1
min and 72°C for 2 min; and 72°C for 5 min. Following RT, qPCR was
performed using the reagents of the SYBR green I mix (Takara Bio,
Dalian, China) in a 20 µl reaction volume, according to the
manufacturer's protocol. The reaction system contained 10 µl
SYBR Green I mix, 2 µl cDNA, 2 µl forward primer, 2
µl reverse primer (Guangzhou RiboBio Co., Ltd., Guangzhou,
China) and 4 µl double-distilled water. The temperature
protocol for the reaction was as follows: 95°C for 10 min; 40
cycles at 95°C for 15 sec and 60°C for 1 min. Each sample was
analyzed in triplicate. U6 small RNA was used as an internal
control. Gene expression was quantified using the 2−ΔΔCq
method (19).
MTT assay
The assessment of cell proliferation was performed
using an MTT assay, according to the manufacturer's protocol.
Following transfection for 24 h, the transfected cells were seeded
into 96-well plates at a density of 3,000 cells/well. The MTT assay
was performed every 24 h until 96 h. A 20 µl volume of MTT
solution (5 mg/ml; Sigma-Aldrich, St. Louis, MO, USA) was added
into each well. Following incubation for 4 h at 37°C, the MTT
solution was removed and the formazan precipitates were dissolved
in 200 µl DMSO. The A490 of each well was measured using a
plate reader. The suppression rate was calculated using the
following formula: Suppression rate = (1 − A490miR-411 /
A490NC) × 100%. All experiments were performed in
triplicate.
Cell migration and invasion assays
The migration and invasion potentials of the cells
were evaluated using Transwell chambers with an 8-µm pore
polycarbonate membrane (Corning Costar, Cambridge, MA, USA). For
the invasion assay, the Transwell chambers were coated with
Matrigel (BD Biosciences, San Jose, CA, USA). Following
transfection for 24 h, 5×104 of the transfected cells in
300 µl serum-free medium were added into the upper chamber.
A 500 µl volume of medium containing 20% FBS was then added
to the lower chamber as a chemoattractant. Following 12 h
incubation for the migration assay 24 h incubation for the invasion
assay, cells, which had not migrated or invaded through the pores
were carefully removed using a cotton swab. The chambers were then
fixed with 100% methanol and stained with 0.5% crystal violet
(Beyotime Institute of Biotechnology, Haimen, China). The migrated
or invaded cells were counted under a light microscope (CKX41;
Olympus Corporation, Tokyo, Japan).
Western blot analysis
The rabbit anti-human monoclonal SP1 (1:1,000; cat.
no. 9389) and rabbit anti-human monoclonal GADPH (1:1,000; cat. no.
2118) primary antibodies used in the present study were purchased
from Cell Signaling Technology, Inc. (Danvers, MA, USA). The cells
were washed and lysed using RIPA lysis buffer (Beyotime Institute
of Biotechnology) 72 h post-transfection. Protein concentration was
determined using the bicinchoninc acid protein assay kit (Beyotime
Institute of Biotechnology). Equal quantities of protein (20
µg) were separated by 10% SDS-PAGE and electroblotted onto a
PVDF membrane (EMD Millipore, Billerica, MA, USA). The membrane was
blocked in phosphate-buffered saline (PBS) containing 0.1% Tween-20
(Beyotime Institute of Biotechnology) and 5% non-fat dry milk. The
membrane was then probed with a primary antibodies for overnight
incubation at 4°C. Horseradish peroxidase-conjugated goat
anti-rabbit secondary antibody (1:2,000; cat. no. BS13278; Bioworld
Technology, Inc., St. Louis Park, MN, USA) was then added for 1 h
at room temperature, followed by incubation at 1:1,000 dilution in
Tris-buffered saline with Tween-20. Finally, the protein bands were
detected using ECL solution (Pierce Biotechnology, Inc., Rockford,
IL, USA) and images were captured using a FluorChem imaging system
(Alpha Innotech, San Leandro, CA, USA). GADPH was used as a loading
control.
Luciferase assay
A luciferase assay was performed to determine
whether SP1 was a direct target of miR-411. The cells were seeded
into 12-well plates at 50% confluence. After 24 h at 37°C in an
atmosphere containing 5% CO2, the cells were transfected
with the luciferase reporter plasmid, miR-411 mimics or NC using
Lipofectamine 2000 reagent, performed in three independent
experiments. At 48 h post-transfection, the activities of firefly
and Renilla luciferase were examined using a Dual-Luciferase
Reporter Assay system (Promega Corporation). The activity of
firefly luciferase was normalized to the activity of Renilla
luciferase, respectively.
Statistical analysis
Data are presented as the mean ± standard deviation,
and were compared using SPSS 17 software (SPSS, Inc., Chicago, IL,
USA) using the Student's t-test or one-way analysis of variance
followed by least significant difference test. The associations
between miR-411 expression and clinicopathological factors were
analyzed using the χ2 test. P<0.05 was considered to
indicate a statistically significant difference.
Results
miR-411 is downregulated in breast cancer
tissues and cell lines
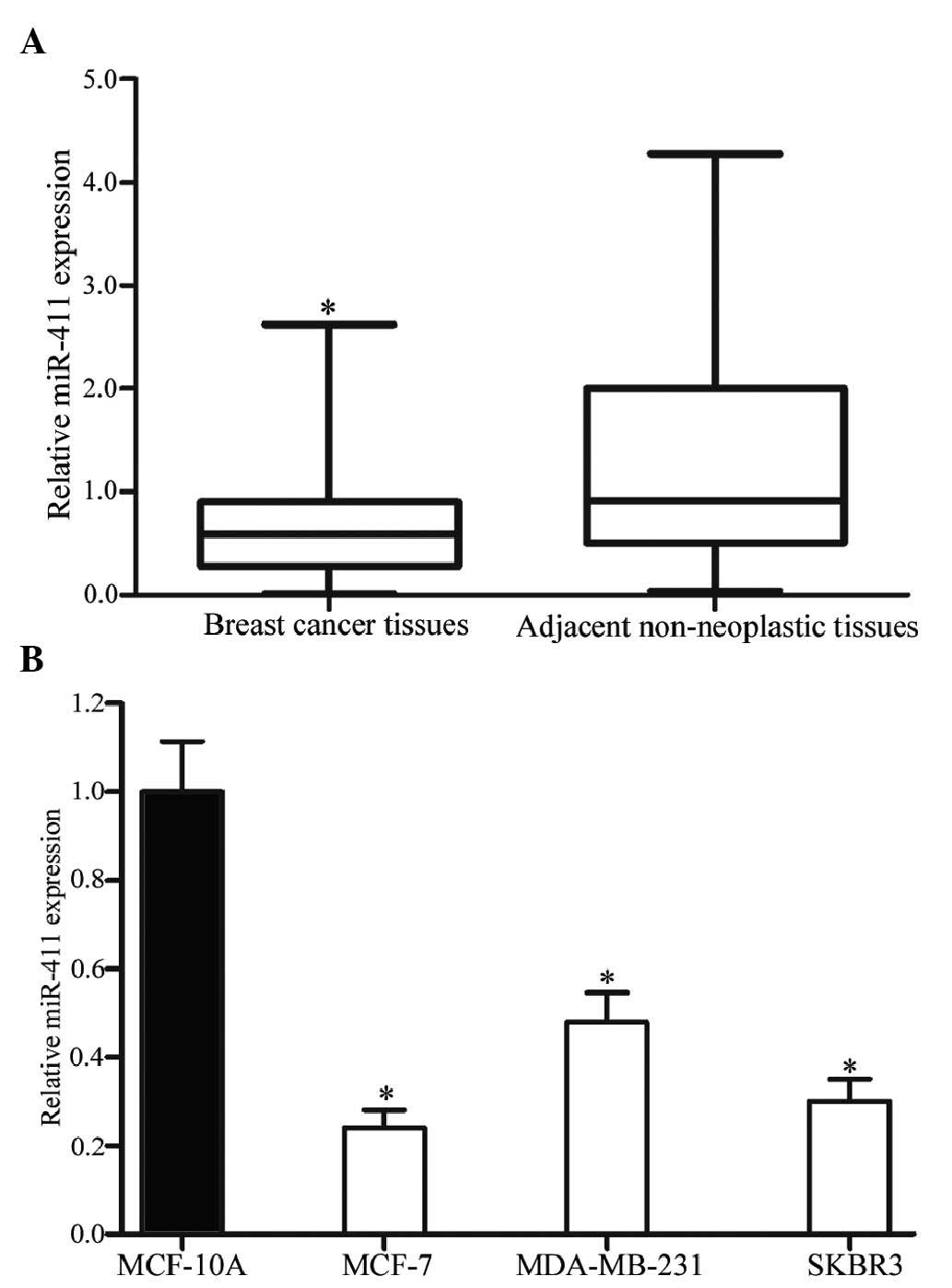
The expression levels of miR-411 in breast cancer
tissues and their corresponding adjacent non-neoplastic tissues
were measured using RT-qPCR. As shown in Fig. 1A, miR-411 was significantly
downregulated in the breast cancer tissues, compared with the
corresponding adjacent non-neoplastic tissues (P<0.05).
In addition, the expression levels of miR-411 were
detected in breast cancer cell lines and the normal mammary
epithelial cell line, MCF-10A. As shown in Fig. 1B, downregulation in the levels of
miR-411 were also observed in the MCF-7, MDA-MB-231 and SKBR3
cells, compared with the MCF-10A cells (P<0.05). In these three
cell lines, the expression levels in the MCF-7 and SKBR3 cells were
lowest, compared with that in the MDA-MB-231 cells. Therefore,
MCF-7 and SKBR3 were selected for use in the present study to
examine the functions of miR-411 in breast cancer.
Association between the expression of
miR-411 and clinicopathological features in patients with breast
cancer
To further assess the association between the
expression of miR-411 and clinicopathological features in patients
with breast cancer, statistical analysis was performed. As shown in
Table I, a low expression level of
miR-411 was closely associated with lymph node metastasis (P=0.034)
and histological grade (P=0.003). However, no correlation was found
between the expression of miR-411 and other clinicopathological
factors, including age, tumor diameter, stage, and the expression
of human epidermal growth factor receptor 2, Ki-67 or P53
(P>0.05).
 | Table IAssociation between the expression of
miR-411 and clinicopathological factors. |
Table I
Association between the expression of
miR-411 and clinicopathological factors.
| Clinical
feature | Cases (n) | miR-411 expression
| P-value |
|---|
| Low | High |
|---|
| Age | | | | 0.257 |
| <50 years | 59 | 37 | 22 | |
| ≥50 years | 55 | 28 | 27 | |
| Tumor diameter | | | | 0.230 |
| <2 cm | 76 | 40 | 36 | |
| ≥2 cm | 38 | 25 | 13 | |
| Lymph node
metastasis | | | | 0.034 |
| Negative | 46 | 32 | 14 | |
| Positive | 68 | 33 | 35 | |
| Stage | | | | 0.349 |
| I–II | 50 | 26 | 24 | |
| III | 64 | 39 | 25 | |
| Histological
grade | | | | 0.003 |
| I–II | 72 | 49 | 23 | |
| III | 42 | 16 | 26 | |
| HER2
expression | | | | 0.456 |
| Negative | 56 | 34 | 22 | |
| Positive | 58 | 31 | 27 | |
| Ki-67
expression | | | | 0.848 |
| Negative | 46 | 27 | 19 | |
| Positive | 68 | 38 | 30 | |
| P53 expression | | | | 0.835 |
| Negative | 33 | 18 | 15 | |
| Positive | 81 | 47 | 34 | |
miR-411 is upregulated in breast cancer
cells following transfection with miR-411 mimics
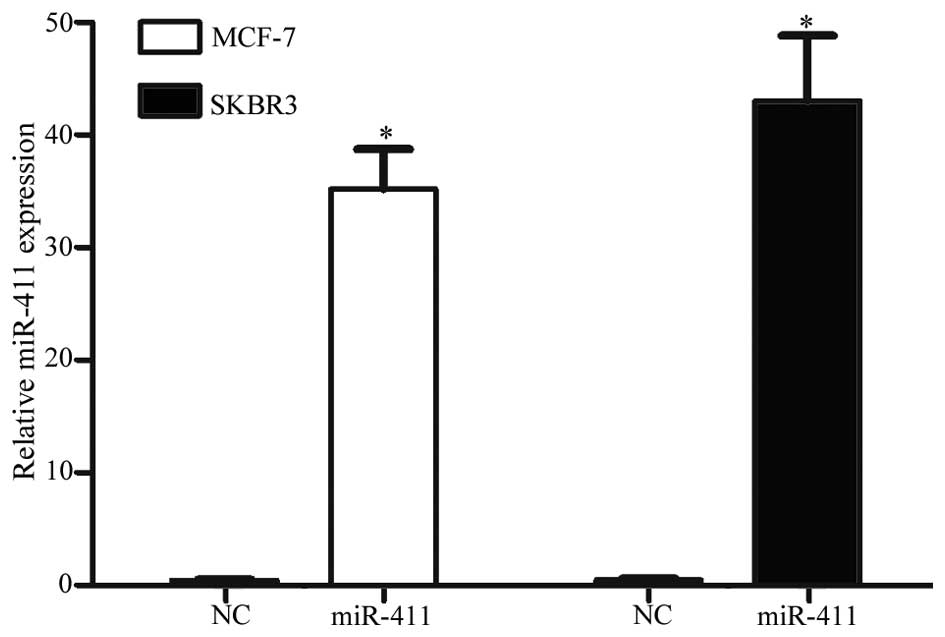
In order to investigate the effects of miR-411 on
breast cancer, the present study transfected the miR-411 mimic into
the MCF-7 and SKBR3 cells. RT-qPCR was performed to assess
transfection efficiency. At 72 h post-transfection, miR-411 was
significantly overexpressed in the cells transfected with miR-411
mimics, compared with the cells transfected with NC (Fig. 2; P<0.05).
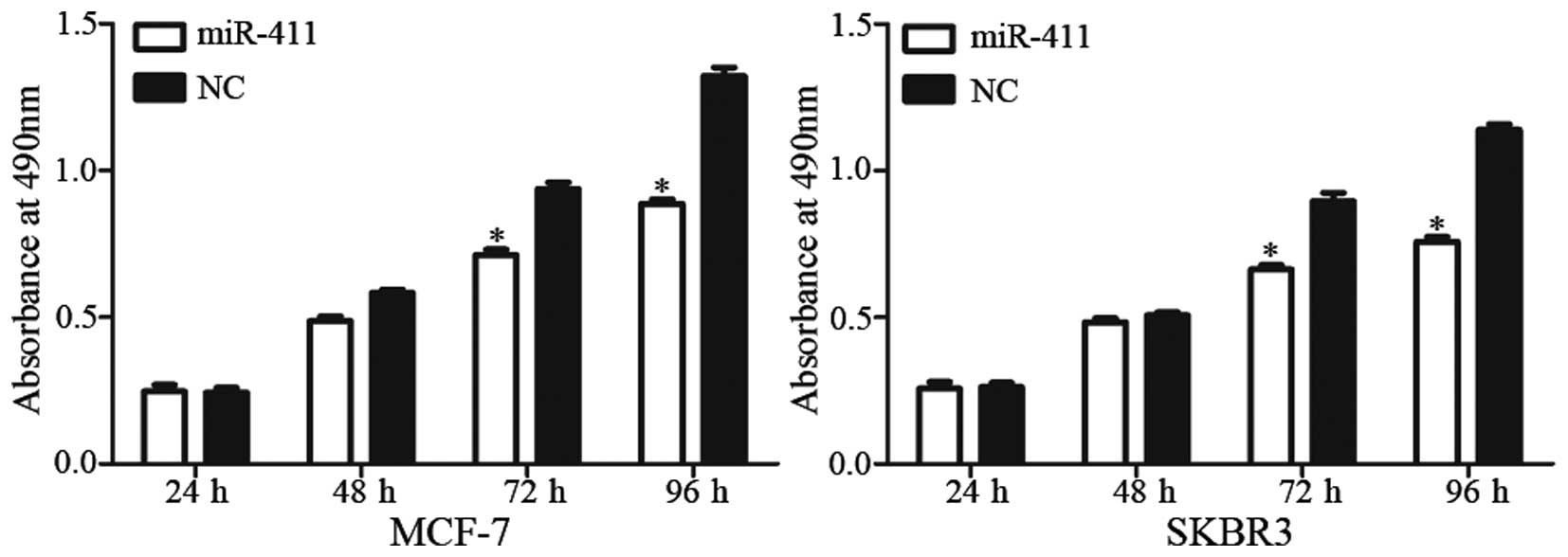
miR-411 suppresses the proliferation of
breast cancer cells
An MTT assay was performed to examine the effect of
miR-411 on cell proliferation. It was revealed that the
upregulation of miR-411 significantly inhibited cell proliferation
in the MCF-7 and SKBR3 cells (Fig.
3; P<0.05). At 96 h post-transfection, the inhibitory rates
were 28.55±4.34% in the MCF-7 cells and 33.09±5.17% in the SKBR3
cells. These results verified that miR-411 functioned as a tumor
suppressor in human breast cancer.
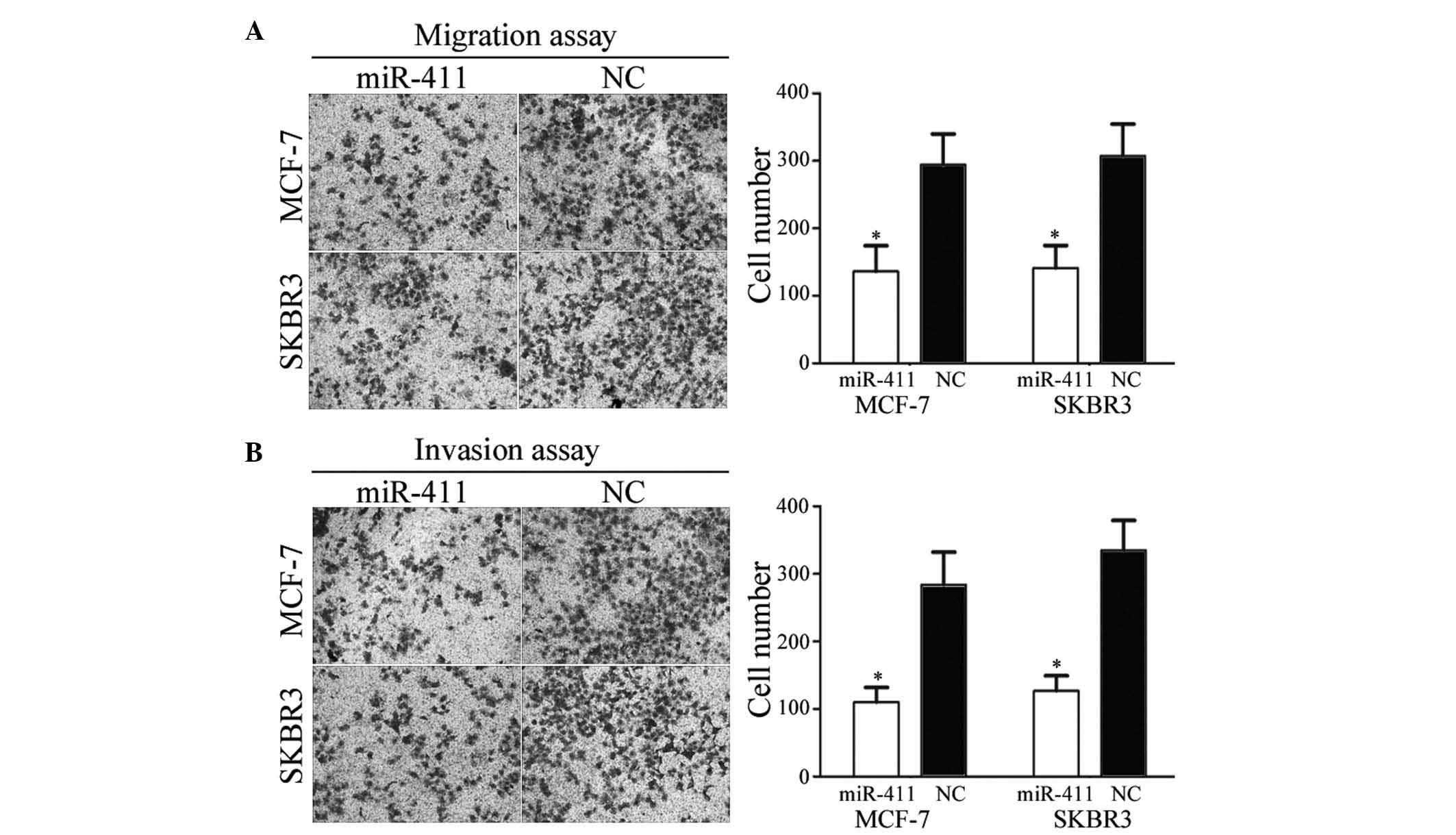
miR-411 decreases the migration and
invasion of breast cancer cells
To investigate the effect of miR-411 on cell
motility, cell migration and invasion assays were performed. As
shown in Fig. 4A and B, the
migration and invasive abilities of the MCF-7 and SKBR3 cells
transfected with miR-411 were significantly downregulated, compared
with those of the cells transfected with the NC (P<0.05). These
results indicated that miR-411 decreased the migration and invasive
abilities of the breast cancer cells.
SP1 is a direct target gene of miR-411 in
vitro
To identify the target of miR-411, TargetScan was
used. SP1 was predicted to be a target of miR-411 (Fig. 5A). To verify whether miR-411
directly targets SP1, western blot analysis was performed to
determine whether SP1 was downregulated at the protein level in
breast cancer cells following transfection with miR-411. As shown
in Fig. 5B, SP1 was significantly
downregulated in the MCF-7 and SKBR3 cells following transfection
with miR-411 (P<0.05).
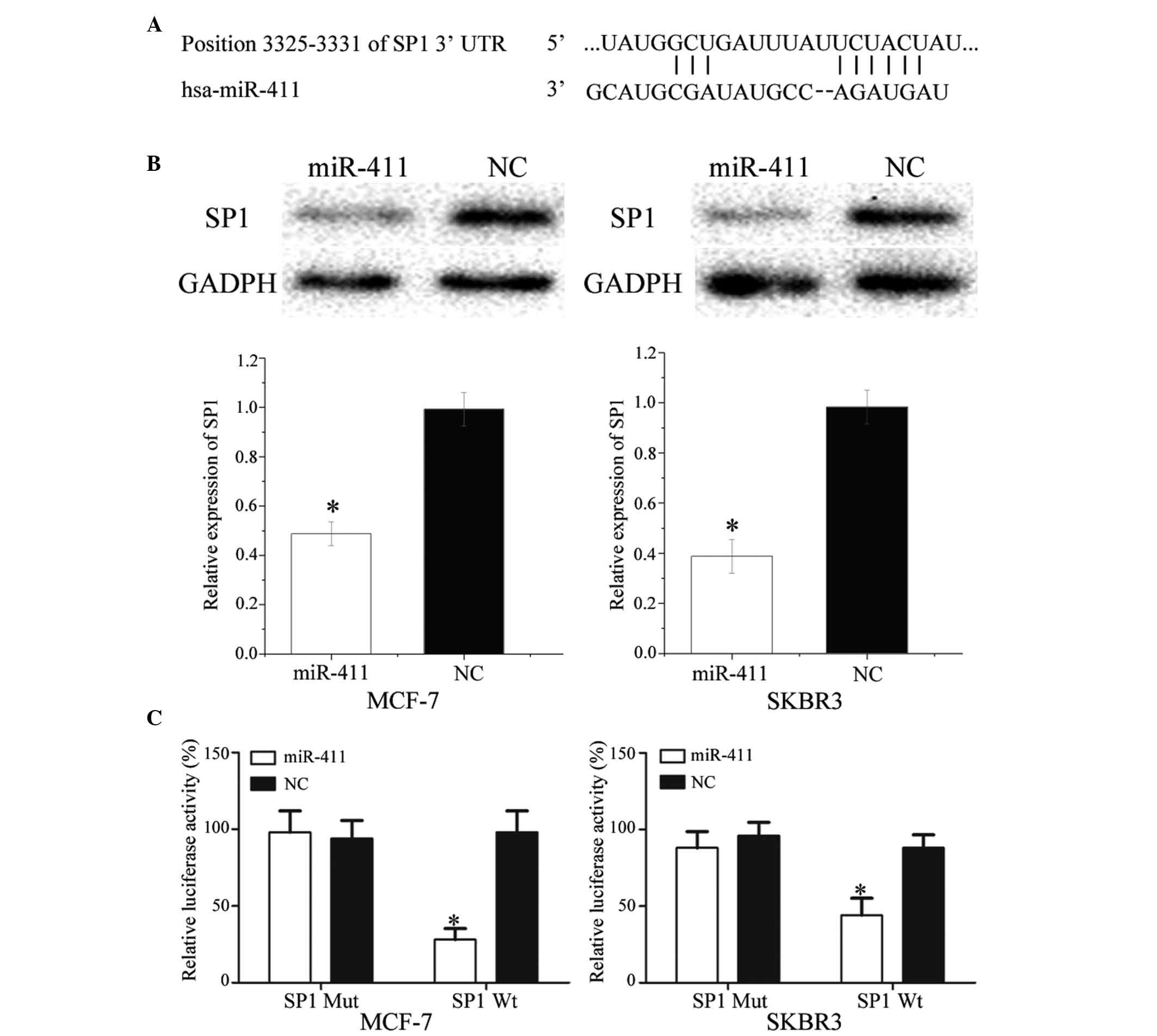 | Figure 5SP1 is a direct target gene of
miR-411 in vitro. (A) TargetScan revealed that SP1 mRNA
contained an miR-411 seed match at position 3,325–3,331 of the SP1
3′UTR. (B) Western blot analysis revealed that SP1 was
significantly downregulated in MCF-7 and SKBR3 cells following
transfection with miR-411. (C) Overexpression of miR-411
significantly inhibited the luciferase activity of SP1 Wt, but not
SP1 Mut in the MCF-7 and SKBR3 cells. Data are presented as the
mean ± standard deviation. *P<0.05, compared with
their respective NC. SP1, specificity protein 1; miR, microRNA; Wt,
wild-type; Mut, mutant; NC, negative control; 3′ UTR, 3′
untranslated region. |
Luciferase assays were also performed to determine
whether miR-411 directly targeted SP1. As shown in Fig. 5C, miR-411 significantly inhibited
the luciferase activity of SP1 WT, but not SP1 Mut, in the MCF-7
and SKBR3 cells (P<0.05). Taken together, these results
indicated SP1 was a direct target gene of miR-411 in
vitro.
Discussion
In the last decade, accumulating evidence has
suggested that the aberrant expression of miRNAs is a
characteristic of malignancies, including breast cancer (8,20,21).
miR-411 is located at the 14q32 region (22). In hepatocellular cell carcinoma
(HCC), miR-411 has been found to be overexpressed in HCC tissues,
compared with adjacent non-neoplastic tissues, and its expression
was also found to be significantly upregulated in eight HCC cell
lines, compared with the human hepatic cell line, LO2. Harafuji
et al (23) reported that
miR-411 was upregulated in primary and immortalized myoblasts of
facioscapulohumeral muscular dystrophy (FSHD), compared with
control myoblasts. However, the present study demonstrated that
miR-411 was downregulated in breast cancer tumor samples and cell
lines. These conflicting results suggest that the expression of
miR-411 in cancer is tissue-type dependent, and the present study
expanded current knowledge of the expression of miR-411 in
cancer.
Previous studies have demonstrated that miRNAs are
important in tumorigenesis and development (24). Studies have also indicated that the
expression of miRNAs is significantly associated with the clinical
recurrence and progression of metastasis (25,26).
In HCC, miR-411 enhances cell growth and has been confirmed as an
oncogene through the regulation of ITCH (27). In FSHD, miR-411 has been found to
inhibit myogenic factors and may have an important function in
myogenesis (23). However, there
are no previous reports on the functions of miR-411 in breast
cancer. In the present study, it was found that miR-411 suppressed
breast cancer cell proliferation, migration and invasion. These
results indicated that miR-411 has important functions in the
tumorigenesis and development of breast cancer, and may have
clinical implications in the treatment of breast caner.
The identification of miR-411 target genes is
essential for understanding its functions in the carcinogenesis and
progression of breast cancer. It is also important for
investigating novel targeted therapies for breast cancer. In the
present study, a molecular link between miR-411 and SP1 was
identified. SP1, a member of the Sp/Krüppel-like factor
transcription factor family, was the first transcription factor to
be cloned from mammalian cells in 1983 (28). SP1 regulates gene expression
through binding to GC-rich sequences (29). It has been found to be upregulated
in several types of cancer, including breast cancer (30), gastric cancer (31), hepatocellular carcinoma (32), thyroid cancer (33), colorectal cancer (34), pancreatic cancer (35) and lung cancer (36). Accumulated evidence has indicated
that SP1 is critical in a variety of physiological processes,
including cell proliferation, apoptosis, cell cycle,
differentiation, tumor metastasis and tumor development (37–40).
The elevated expression of SP1 has also been found to be associated
with poor prognosis in gastric cancer and breast cancer (41,42).
These findings suggest that selecting SP1 as a therapeutic approach
is practicable for breast cancer.
SP1 has been found to be regulated by multiple
miRNAs in several types of cancer. For example, in HCC, miR-324-5p
inhibits cell invasion by targeting SP1 (43). In addition, in gastric cancer,
miR-145, miR-133a, miR-133b, miR-22 and miR-335 act as tumor
suppressors via the direct inhibition of SP1 (31,44,45).
In non-small cell lung cancer, miR-27b decreases cell proliferation
and invasion through the regulation of SP1 (46). In addition, in human glioblastoma,
miR-377 suppresses cell growth and invasion by inhibiting the
activity of SP1 (47). In ovarian
cancer, miR-145 enhances cell chemosensitivity to paclitaxel
through targeting SP1. In esophageal carcinoma, miR-429 targets SP1
to inhibit cell invasion and enhance cell apoptosis (48). In prostate cancer, miR-330
decreases cell migration and invasion through the downregulation of
SP1 (49). Studies have also
revealed that miRNAs regulate the expression of SP1 (50–52).
For example, miR-200b functions as a tumor suppressor by directly
targeting SP1. In the present study, it was demonstrated that
miR-411 has a suppressive role in breast cancer through the
regulation of SP1. Therefore, miR-411/SP1-based targeted therapy
may offer potential as a novel treatment for breast cancer.
In conclusion, the present study showed that miR-411
was downregulated in breast cancer, and was associated with lymph
node metastasis and histological grade. It was also demonstrated
that miR-411 inhibited breast cancer cell proliferation, migration
and invasion by directly targeting SP1. Therefore, miR-411 may
offer potential as a targeted therapy for breast cancer. Future
investigations are required to address whether the potential of
miR-411 may be fully realized in breast cancer therapy.
Acknowledgments
This study was supported by the National Natural
Science Fund (grant no. 30500599), the Guangdong Natural Science
Fund Project (grant no.≈9151503102000019), the Guangdong province
Science and Technology Plan Project (grant no. 2013021800097), the
Shenzhen Science and Technology Plan (Medical Treatment and Public
Health) Key Project (grant no. 201201028) and the Shenzhen
Technical Research and Development Fund Project (grant nos.
CXZZ20140414170821163, GIHZ20130412153906740,
NJCYJ20120615085512920, JCYJ20120613171430264 and
ZYC201006180477A).
References
|
1
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar
|
|
2
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2015. CA Cancer J Clin. 65:5–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Mangolini A, Ferracin M, Zanzi MV,
Saccenti E, Ebnaof SO, Poma VV, Sanz JM, Passaro A, Pedriali M,
Frassoldati A, et al: Diagnostic and prognostic microRNAs in the
serum of breast cancer patients measured by droplet digital PCR.
Biomark Res. 3:122015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sharma S, Kelly TK and Jones PA:
Epigenetics in cancer. Carcinogenesis. 31:27–36. 2010. View Article : Google Scholar :
|
|
5
|
Beckmann MW, Niederacher D, Schnürch HG,
Gusterson BA and Bender HG: Multistep carcinogenesis of breast
cancer and tumour heterogeneity. J Mol Med (Berl). 75:429–439.
1997. View Article : Google Scholar
|
|
6
|
Nakamura S, Yagata H, Ohno S, Yamaguchi H,
Iwata H, Tsunoda N, Ito Y, Tokudome N, Toi M, Kuroi K and Suzuki E:
Multi-center study evaluating circulating tumor cells as a
surrogate for response to treatment and overall survival in
metastatic breast cancer. Breast Cancer. 17:199–204. 2010.
View Article : Google Scholar
|
|
7
|
Gong Y, He T, Yang L, Yang G, Chen Y and
Zhang X: The role of miR-100 in regulating apoptosis of breast
cancer cells. Sci Rep. 5:116502015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Mar-Aguilar F, Luna-Aguirre CM,
Moreno-Rocha JC, Araiza-Chávez J, Trevino V, Rodríguez-Padilla C
and Reséndez-Pérez D: Differential expression of miR-21, miR-125b
and miR-191 in breast cancer tissue. Asia Pac J Clin Oncol.
9:53–59. 2013. View Article : Google Scholar
|
|
9
|
Hu Y, Zhu Q and Tang L: MiR-99a antitumor
activity in human breast cancer cells through targeting of mTOR
expression. PLoS One. 9:e920992014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lin H, Dai T, Xiong H, Zhao X, Chen X, Yu
C, Li J, Wang X and Song L: Unregulated miR-96 induces cell
proliferation in human breast cancer by downregulating
transcriptional factor FOXO3a. PLoS One. 5:e157972010. View Article : Google Scholar
|
|
11
|
Lee JC, Gundara JS, Glover A, Serpell J
and Sidhu SB: MicroRNA expression profiles in the management of
papillary thyroid cancer. Oncologist. 19:1141–1147. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hwang HW and Mendell JT: MicroRNAs in cell
proliferation, cell death, and tumorigenesis. Br J Cancer.
96(Suppl): R40–R44. 2007.PubMed/NCBI
|
|
13
|
Lewis BP, Burge CB and Bartel DP:
Conserved seed pairing, often flanked by adenosines, indicates that
thousands of human genes are microRNA targets. Cell. 120:15–20.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li X, Abdel-Mageed AB, Mondal D and Kandil
E: MicroRNA expression profiles in differentiated thyroid cancer, a
review. Int J Clin Exp Med. 6:74–80. 2013.
|
|
15
|
Xia M, Li H, Wang JJ, Zeng HJ and Wang SH:
MiR-99a suppress proliferation, migration and invasion through
regulating insulin-like growth factor 1 receptor in breast cancer.
Eur Rev Med Pharmacol Sci. 20:1755–1763. 2016.PubMed/NCBI
|
|
16
|
Wu D, Niu X, Pan H, Zhou Y, Qu P and Zhou
J: MicroRNA-335 is downregulated in bladder cancer and inhibits
cell growth, migration and invasion via targeting ROCK1. Mol Med
Rep. 13:4379–4385. 2016.PubMed/NCBI
|
|
17
|
Chen X, Bo L, Lu W, Zhou G and Chen Q:
MicroRNA-148b targets Rho-associated protein kinase 1 to inhibit
cell proliferation, migration and invasion in hepatocellular
carcinoma. Mol Med Rep. 13:477–482. 2016.
|
|
18
|
Marini F, Luzi E and Brandi ML: MicroRNA
role in thyroid cancer development. J Thyroid Res. 2011:4071232011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) Method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
20
|
Abedi N, Mohammadi-Yeganeh S, Koochaki A,
Karami F and Paryan M: miR-141 as potential suppressor of β-catenin
in breast cancer. Tumour Biol. 36:9895–9901. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li D, Zhao Y, Liu C, Chen X, Qi Y, Jiang
Y, Zou C, Zhang X, Liu S, Wang X, et al: Analysis of MiR-195 and
MiR-497 expression, regulation and role in breast cancer. Clin
Cancer Res. 17:1722–1730. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Nadal E, Zhong J, Lin J, Reddy RM, Ramnath
N, Orringer MB, Chang AC, Beer DG and Chen G: A MicroRNA cluster at
14q32 drives aggressive lung adenocarcinoma. Clin Cancer Res.
20:3107–3117. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Harafuji N, Schneiderat P, Walter MC and
Chen YW: miR-411 is up-regulated in FSHD myoblasts and suppresses
myogenic factors. Orphanet J Rare Dis. 8:552013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yi B, Piazza GA, Su X and Xi Y: MicroRNA
and cancer chemoprevention. Cancer Prev Res (Phila). 6:401–409.
2013. View Article : Google Scholar
|
|
25
|
Zhang JX, Song W, Chen ZH, Wei JH, Liao
YJ, Lei J, Hu M, Chen GZ, Liao B, Lu J, et al: Prognostic and
predictive value of a microRNA signature in stage II colon cancer:
A microRNA expression analysis. Lancet Oncol. 14:1295–1306. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Heinzelmann J, Unrein A, Wickmann U,
Baumgart S, Stapf M, Szendroi A, Grimm MO, Gajda MR, Wunderlich H
and Junker K: MicroRNAs with prognostic potential for metastasis in
clear cell renal cell carcinoma: A comparison of primary tumors and
distant metastases. Ann Surg Oncol. 21:1046–1054. 2014. View Article : Google Scholar
|
|
27
|
Xia K, Zhang Y, Cao S, Wu Y, Guo W, Yuan W
and Zhang S: miR-411 regulated ITCH expression and promoted cell
proliferation in human hepatocellular carcinoma cells. Biomed
Pharmacother. 70:158–163. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Dynan WS and Tjian R: The
promoter-specific transcription factor Sp1 binds to upstream
sequences in the SV40 early promoter. Cell. 35:79–87. 1983.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kadonaga JT, Courey AJ, Ladika J and Tjian
R: Distinct regions of Sp1 modulate DNA binding and transcriptional
activation. Science. 242:1566–1570. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Yue L, Li L, Liu F, Hu N, Zhang W, Bai X,
Li Y, Zhang Y, Fu L, Zhang X and Ye L: The oncoprotein HBXIP
activates transcriptional coregulatory protein LMO4 via Sp1 to
promote proliferation of breast cancer cells. Carcinogenesis.
34:927–935. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xu Y, Zhao F, Wang Z, Song Y, Luo Y, Zhang
X, Jiang L, Sun Z, Miao Z and Xu H: MicroRNA-335 acts as a
metastasis suppressor in gastric cancer by targeting Bcl-w and
specificity protein 1. Oncogene. 31:1398–1407. 2012. View Article : Google Scholar :
|
|
32
|
Yin P, Zhao C, Li Z, Mei C, Yao W, Liu Y,
Li N, Qi J, Wang L, Shi Y, et al: Sp1 is involved in regulation of
cystathionine gamma-lyase gene expression and biological function
by PI3K/Akt pathway in human hepatocellular carcinoma cell lines.
Cell Signal. 24:1229–1240. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Bonofiglio D, Qi H, Gabriele S, Catalano
S, Aquila S, Belmonte M and Ando S: Peroxisome
proliferator-activated receptor gamma inhibits follicular and
anaplastic thyroid carcinoma cells growth by upregulating
p21Cip1/WAF1 gene in a Sp1-dependent manner. Endocr Relat Cancer.
15:545–557. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Pathi S, Jutooru I, Chadalapaka G, Nair V,
Lee SO and Safe S: Aspirin inhibits colon cancer cell and tumor
growth and downregulates specificity protein (Sp) transcription
factors. PLoS One. 7:e482082012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huang C and Xie K: Crosstalk of Sp1 and
Stat3 signaling in pancreatic cancer pathogenesis. Cytokine Growth
Factor Rev. 23:25–35. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang YT, Chuang JY, Shen MR, Yang WB,
Chang WC and Hung JJ: Sumoylation of specificity protein 1 augments
its degradation by changing the localization and increasing the
specificity protein 1 proteolytic process. J Mol Biol. 380:869–885.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Black AR, Black JD and Azizkhan-Clifford
J: Sp1 and krüppel-like factor family of transcription factors in
cell growth regulation and cancer. J Cell Physiol. 188:143–160.
2001. View
Article : Google Scholar : PubMed/NCBI
|
|
38
|
Li L, He S, Sun JM and Davie JR: Gene
regulation by Sp1 and Sp3. Biochem Cell Biol. 82:460–471. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Safe S and Abdelrahim M: Sp transcription
factor family and its role in cancer. Eur J Cancer. 41:2438–2448.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang L, Guan X, Zhang J, Jia Z, Wei D, Li
Q, Yao J and Xie K: Targeted inhibition of Sp1-mediated
transcription for antiangiogenic therapy of metastatic human
gastric cancer in orthotopic nude mouse models. Int J Oncol.
33:161–167. 2008.PubMed/NCBI
|
|
41
|
Zhang J, Zhu ZG, Ji J, Yuan F, Yu YY, Liu
BY and Lin YZ: Transcription factor Sp1 expression in gastric
cancer and its relationship to long-term prognosis. World J
Gastroenterol. 11:2213–2217. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wright C, Angus B, Napier J, Wetherall M,
Udagawa Y, Sainsbury JR, Johnston S, Carpenter F and Horne CH:
Prognostic factors in breast cancer: Immunohistochemical staining
for SP1 and NCRC 11 related to survival, tumour epidermal growth
factor receptor and oestrogen receptor status. J Pathol.
153:325–331. 1987. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Cao L, Xie B, Yang X, Liang H, Jiang X,
Zhang D, Xue P, Chen D and Shao Z: MiR-324-5p suppresses
hepatocellular carcinoma cell invasion by counteracting ECM
degradation through post-transcriptionally downregulating ETS1 and
SP1. PLoS One. 10:e01330742015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Guo MM, Hu LH, Wang YQ, Chen P, Huang JG,
Lu N, He JH and Liao CG: miR-22 is down-regulated in gastric
cancer, and its overexpression inhibits cell migration and invasion
via targeting transcription factor Sp1. Med Oncol. 30:5422013.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Qiu T, Zhou X, Wang J, Du Y, Xu J, Huang
Z, Zhu W, Shu Y and Liu P: MiR-145, miR-133a and miR-133b inhibit
proliferation, migration, invasion and cell cycle progression via
targeting transcription factor Sp1 in gastric cancer. FEBS Lett.
588:1168–1177. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Jiang J, Lv X, Fan L, Huang G, Zhan Y,
Wang M and Lu H: MicroRNA-27b suppresses growth and invasion of
NSCLC cells by targeting Sp1. Tumour Biol. 35:10019–10023. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Zhang R, Luo H, Wang S, Chen W, Chen Z,
Wang HW, Chen Y, Yang J, Zhang X, Wu W, et al: MicroRNA-377
inhibited proliferation and invasion of human glioblastoma cells by
directly targeting specificity protein 1. Neuro Oncol.
16:1510–1522. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Wang Y, Li M, Zang W, Ma Y, Wang N, Li P,
Wang T and Zhao G: MiR-429 up-regulation induces apoptosis and
suppresses invasion by targeting Bcl-2 and SP-1 in esophageal
carcinoma. Cell Oncol (Dordr). 36:385–394. 2013. View Article : Google Scholar
|
|
49
|
Mao Y, Chen H, Lin Y, Xu X, Hu Z, Zhu Y,
Wu J, Xu X, Zheng X and Xie L: microRNA-330 inhibits cell motility
by downregulating Sp1 in prostate cancer cells. Oncol Rep.
30:327–333. 2013.PubMed/NCBI
|
|
50
|
Cui F, Wang S, Lao I, Zhou C, Kong H,
Bayaxi N, Li J, Chen Q, Zhu T and Zhu H: miR-375 inhibits the
invasion and metastasis of colorectal cancer via targeting SP1 and
regulating EMT-associated genes. Oncol Rep. 36:487–493.
2016.PubMed/NCBI
|
|
51
|
Kang M, Xiao J, Wang J, Zhou P, Wei T,
Zhao T and Wang R: MiR-24 enhances radiosensitivity in
nasopharyngeal carcinoma by targeting SP1. Cancer Med. 5:1163–1173.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Li X, Chen L, Wang W, Meng FB, Zhao RT and
Chen Y: MicroRNA-150 inhibits cell invasion and migration and is
downregulated in human osteosarcoma. Cytogenet Genome Res.
146:124–135. 2015. View Article : Google Scholar : PubMed/NCBI
|