Introduction
Pituitary adenomas account for ~10% of intracranial
tumors, and 5% are locally invasive (1). Although the etiology of pituitary
adenoma is not fully understood, numerous experimental and clinical
observations indicate that DNA repair enzyme
O6-methylguanine-DNA methyltransferase (MGMT) is
implicated in the pathogenesis of various tumor types, including
pituitary adenoma (2,3). MGMT is a ubiquitous highly expressed
enzyme that removes alkylating lesions at the O6
position of guanine to repair DNA damage (4).
MGMT is regulated by multiple molecular mechanisms,
including epigenetic silencing of the MGMT gene by promoter
methylation, histone modifications and aberrant expression of
transcriptional activators and repressors, microRNAs and activation
of the canonical Wnt/β-catenin signaling cascade (5). In the absence of Wnt ligands,
cytoplasmic β-catenin is phosphorylated and degraded by the
proteasome. However, a large pool of soluble non-phosphorylated
β-catenin exists in the presence of Wnt ligands. β-catenin is then
translocated to the nucleus where it interacts with T-cell factor
(TCF)/lymphoid-enhancing factor (LEF) family transcription factors,
such as TCF-4, to activate Wnt pathway target genes, including
cyclin D1 and MGMT (6). In fact,
growing evidence indicates that inhibition of Wnt/β-catenin
signaling may have antitumor effects (7).
Tanshinone IIA (TSA) is the main quinone compounds
isolated from Salvia miltiorrhiza, which have been used for
various medicinal purposes in traditional Chinese medicine for many
years. Experimental and clinical studies suggest that TSA has
antitumor effects (8,9). Whether TSA has antitumor effects
against pituitary adenomas, and whether the mechanisms are
associated with Wnt/β-catenin/MGMT signaling remains to be
clarified.
In the present study, AtT-20 mouse pituitary cells
were used to evaluate the potential antitumor effect of TSA against
pituitary adenoma and the underlying mechanism. Wnt/β-catenin/MGMT
pathway inactivation was involved in antitumor effect of TSA
against pituitary adenoma.
Materials and methods
Reagents
TSA (purity >98%), dimethyl sulfoxide (DMSO) and
4′,6-diamidino-2-phenylindole (DAPI) were purchased from
Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). Dulbecco's modified
Eagle's medium (DMEM), fetal bovine serum (FBS) and
phosphate-buffered saline (PBS) were purchased from Gibco (Thermo
Fisher Scientific, Inc., Waltham, MA, USA).
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT)
and 5,5′,6,6′-tetrachloro-1,1′,
3,3′-tetraethylbenzimidazolylcarbocyanine iodide (JC-1) were
obtained from Enzo Life Sciences, Inc. (Farmingdale, NY, USA). Cell
nuclear protein extraction kits, MGMT lentiviral activation
particles and and control lentiviral activation particles,
β-catenin short hairpin RNA (shRNA) lentiviral particle and control
shRNA plasmid, Polybrene, and puromycin dihydrochloride were
obtained from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA).
Cell culture and drug preparation
AtT-20 cells obtained from the Typical Culture
Preservation Commission Cell Bank, Chinese Academy of Sciences
(Shanghai, China) were cultured at 37°C in 5% CO2 and
95% atmosphere in DMEM medium supplemented with 5% FBS and
glutamine (2 mM). To overexpress MGMT in β-catenin knock down
cells, AtT-20 cells were cultured in 6-well plate (5×104
cells/ml) and cultured to 60% confluency. Cells were transfected
with MGMT lentiviral activation particles (1×105/ml) and
β-catenin shRNA lentiviral particle (1×105/ml) in
complete medium with Polybrene (5 µg/ml) and incubated overnight.
Stable clones were selected using puromycin dihydrochloride (5
µg/ml). The mRNA and protein expression of β-catenin or MGMT were
determined by reverse transcription-polymerase chain reaction
(RT-PCR) and western blotting, respectively. TSA stock solution (1
M) was prepared in DMSO and diluted with fresh complete medium
immediately prior to use. The control cells were treated with DMSO
(final concentration <0.1%).
Analysis of cell viability
Cell viability was determined by MTT assay. AtT-20
cells (5×104 cells/ml) were seeded in 96-well plates and
incubated overnight. After TSA (2.5, 5, 10, 20 µM) treatment for 4,
8, 12, 16 or 24 h, cells were incubated at 37°C for 4 h with MTT
solution (1 mg/ml), and then added 100 µl DMSO to dissolve formazan
crystals. The absorbance was detected on a microplate reader (570
nm; Tecan Group Ltd., Männedorf, Austria). Cell viability was
expressed as a percentage of the control group.
LDH leakage assay
AtT-20 cells (5×104 cells/ml) were seeded
in 6-well plates and incubated overnight. Following TSA treatment
(5, 10, 20 µM) for 4, 8, 12, 16 or 24 h, the culture medium was
used to measure the amounts of lactate dehydrogenase (LDH) released
using an LDH assay kit (Nanjing Jiancheng Bioengineering Institute,
Nanjing, China), according to the manufacturer's protocol.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL)
TUNEL kits (EMD Millipore, Billerica, MA, USA) were
used to detect DNA fragmentation of apoptotic AtT-20 cells. AtT-20
cells (1×105 cells/ml) were seeded in 6-well plates and
treated with increasing concentrations of TSA (5, 10, and 20 µM)
for 12 h. Cells were incubated at 37°C for 1 h with terminal
deoxynucleotidyl transferase enzyme, and then incubated for 30 min
with the anti-digoxigenin conjugate. The cell nuclei were
counterstained with DAPI. The fluorescence images were captured by
a fluorescence microscope (Leica Microsystems GmbH, Wetzlar,
Germany).
Quantification of the apoptosis
rate
The apoptosis rate was detected by Annexin
V/propidium iodide (PI) assay kits (Invitrogen; Thermo Fisher
Scientific, Inc.). AtT-20 cells (1×105 cells/ml) were
cultured in 6-well plates and treated with increasing
concentrations of TSA (5, 10 and 20 µM) for 12 h. Cells were
incubated for 15 min with Annexin V/PI (1 µg/ml) solution, and then
analyzed using a FACSCalibur flow cytometer. Data were analyzed
using CellQuest Pro software v1.0 (BD Biosciences, San Jose, CA,
USA).
Detection of mitochondrial membrane
potential
The mitochondrial membrane potential was detected
using JC-1. AtT-20 cells (1×105 cells/ml) were cultured
in 6-well plates and treated with increasing concentrations of TSA
(5, 10 and 20 µM) for 12 h. Cells were then incubated at 37°C for
30 min with JC-1 (2 µM) and analyzed using a FACSCalibur flow
cytometer.
Detection of caspase-3 activity
The caspase-3 activity was determined using a
fluorescein-based active caspase-3 staining kit (BioVision, Inc.,
Milpitas, CA, USA). AtT-20 cells (1×105 cells/ml) were
cultured in 6-well plates and treated with increasing
concentrations of TSA (5, 10 and 20 µM) for 12 h. Cells were
incubated for 10 min in chilled lysis buffer and then incubated at
37°C for additional 2 h in reaction buffer (containing 10 mM
dithiothreitol and DEVD-7-amino-4-trifluoromethylcoumarin
substrate). The fluorescence was detected using a microplate reader
(excitation, 400 nm; emission, 505 nm). The caspase-3 activity was
expressed as the percentage of the control.
Dual luciferase reporter assay
TCF-LEF reporter activity was determined using a
dual luciferase reporter assay. A TCF-LEF reporter, obtained from
SABiosciences (Qiagen, Inc., Valencia, CA, USA) is a mixture of
inducible TCF-LEF-responsive firefly luciferase construct and
constitutively expressed Renilla luciferase construct
(40:1). AtT-20 cells (1×105 cells/well) were seeded in
6-well plates. When 60% confluence was observed, the cells were
transiently transfected with TCF-LEF reporter using Lipofectamine
2000 (Thermo Fisher Scientific, Inc.). At 24 h after transfection,
cells were treated with increasing concentrations of TSA (5, 10 and
20 µM) for 12 h. TCF-LEF reporter activity was measured using the
dual luciferase reporter assay system (Promega Corporation,
Madison, WI, USA). Luciferase activity was detected busing a
GloMaxTM 96 microplate luminometer (Promega Corporation) The
firefly luciferase values were normalized to the Renilla
luciferase values. TCF-LEF activity was expressed as a percentage
of the control.
Co-immunoprecipitation (co-IP)
The interaction between β-catenin and TCF-4 was
detected using a Pierce co-immunoprecipitation kit (Thermo Fisher
Scientific, Inc.). Briefly, AtT-20 cells (1×105
cells/well) were treated with increasing concentrations of TSA (5,
10 and 20 µM) for 12 h. The nuclear protein was obtained using cell
nuclear protein extraction kits (Thermo Fisher Scientific, Inc.)
and pre-cleared using the control agarose resin, and then incubated
at 4°C with antibody against TCF-4 (1:200; ab185736, Abcam,
Cambridge, MA, USA) immobilized to AminoLink Plus (Thermo Fisher
Scientific, Inc.) coupling resin in reaction buffer.
Immunoprecipitates were washed with lysis buffer and eluted with
elution buffer. The level of β-catenin was analyzed by western
blotting.
Western blot
The expression level of protein was analyzed by
western blot. The total cytosol and nuclear protein extract were
prepared using cell cytosol and nuclear protein extraction kits
(Thermo Fisher Scientific, Inc.), respectively. The total protein
concentration was determined using a bicinchonic acid assay.
Proteins (20 µg) were resolved by 10% SDS-PAGE and transferred on
PVDF membranes. The membranes were blocked with 2% BSA
(Sigma-Aldrich; Merck KGaA) in TBS-Tween-20 (0.1%) at 4°C for 1 h.
The membranes were incubated overnight at 4°C with the following
primary antibodies: β-catenin (C-18; sc-1496; 1:200) p-β-catenin
(Ser 33; sc-101650; 1:200), TCF-4 (F-7; sc-271288; 1:200), β-actin
(C-2; sc-8432; 1:1,000) and lamin B (C-20; sc-6216; 1:200) from
Santa Cruz Biotechnology, Inc., and cyclin D1 (ab134175; 1:1,000),
MGMT (ab39253; 1:1,000) from Abcam (Cambridge, MA, USA). The
membranes were incubated at 4°C for 1 h with respective horseradish
peroxidase-conjugated goat anti-mouse (ab6789, Abcam, Cambridge,
MA, USA) or goat anti-rabbit second antibodies (ab6721, Abcam,
Cambridge, MA, USA). The immunoblots were visualized using enhanced
chemiluminescence western blot detection kits (GE Healthcare Life
Sciences, Chalfont, UK) and then visualized using a molecular
imager with Image Lab 3.0 (Bio-Rad Laboratories, Inc., Hercules,
CA, USA). Equal loading of proteins was determined by β-actin or
lamin B. The expression levels of the proteins were quantified
using a densitometer (Molecular Devices, LLC, Sunnyvale, CA,
USA).
Statistical analysis
The data represent the results from three
independent experiments. The results are presented as the mean ±
standard error of the mean. Student's two-tailed t-test and one-way
ANOVA followed by Bonferroni's multiple comparison test were used
for comparison between two groups and multiple groups,
respectively. P<0.05 was considered statistically
significant.
Results
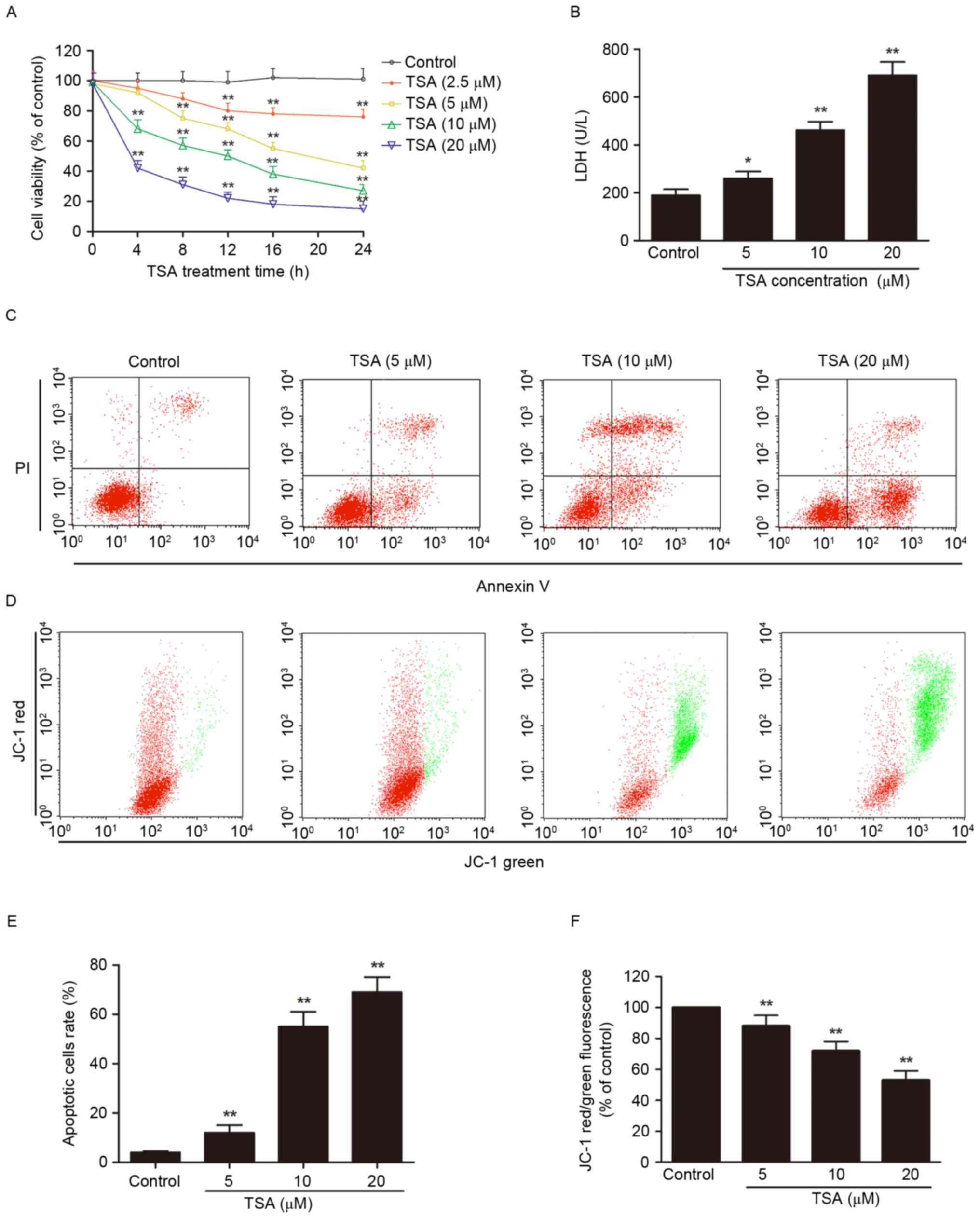
TSA induces apoptosis in AtT-20
cells
The potential antitumor effect of TSA against AtT-20
cells was investigated. Treatment of AtT-20 cells with increasing
concentrations of TSA for increasing periods of time reduced the
cell viability (P<0.01, Fig.
1A) and increase LDH leakage (P<0.01; Fig. 1B) in concentration- and
time-dependent manners.
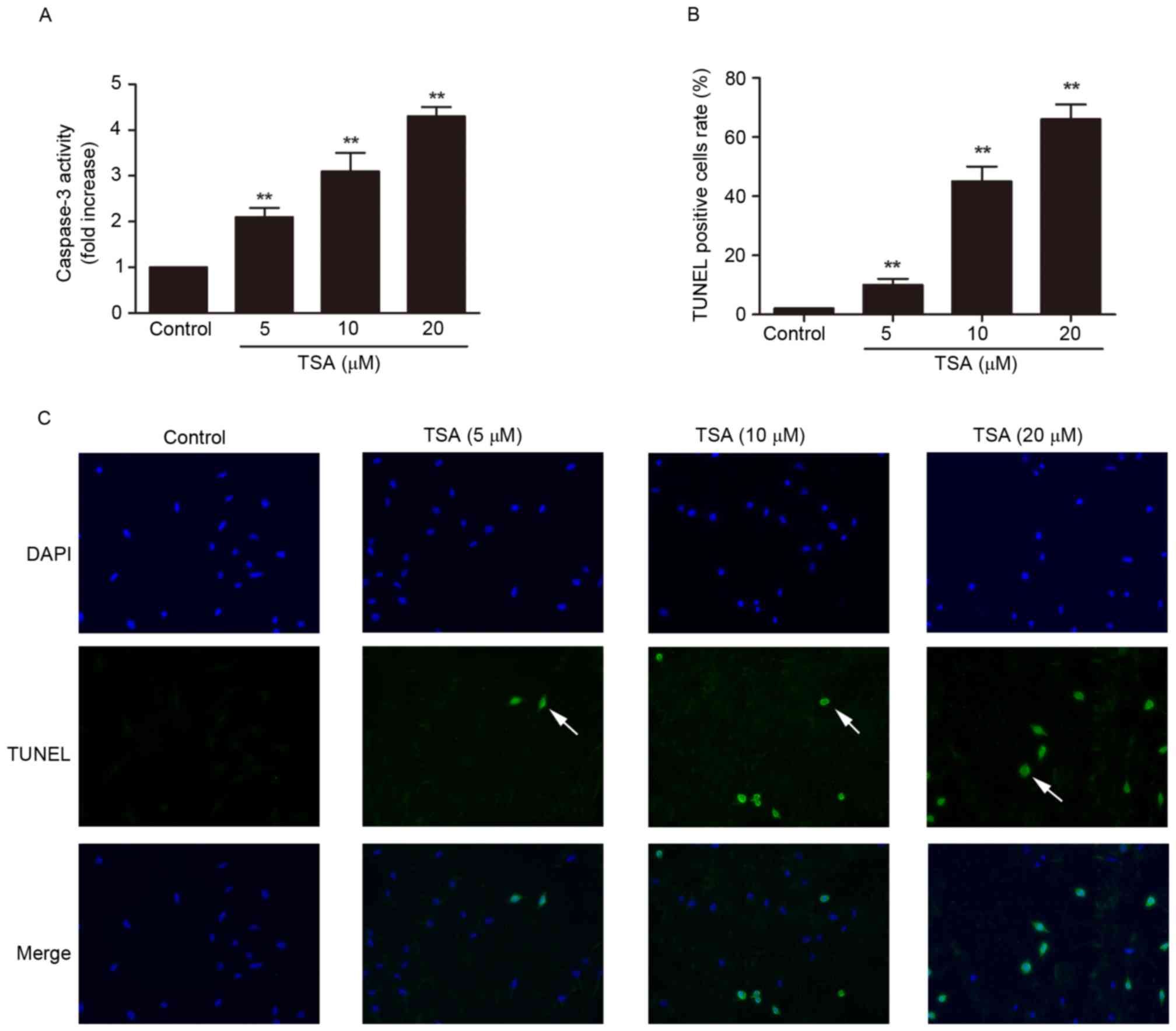
Phophatidylserine externalization, depolarization of
mitochondrial membrane, caspase-3 activation and DNA fragmentation
are the main features of apoptotic cells (10), and were detected by annexin V/PI
staining and JC-1 staining (Fig.
1C-F), and caspase-3 activity assay (Fig. 2A) and TUNEL staining (Fig. 2B and C), respectively. TSA
treatment significantly increased the percentage of apoptotic cells
(P<0.01; Fig. 1C and E) and
decreased mitochondrial membrane potential (P<0.01; Fig. 1D and F) compared with the control.
Compared with the control group, caspase-3 activity (P<0.01;
Fig. 2A) and TUNEL-positive cell
rate (P<0.01; Fig. 2B and C)
were increased by TSA in a dose-dependent manner.
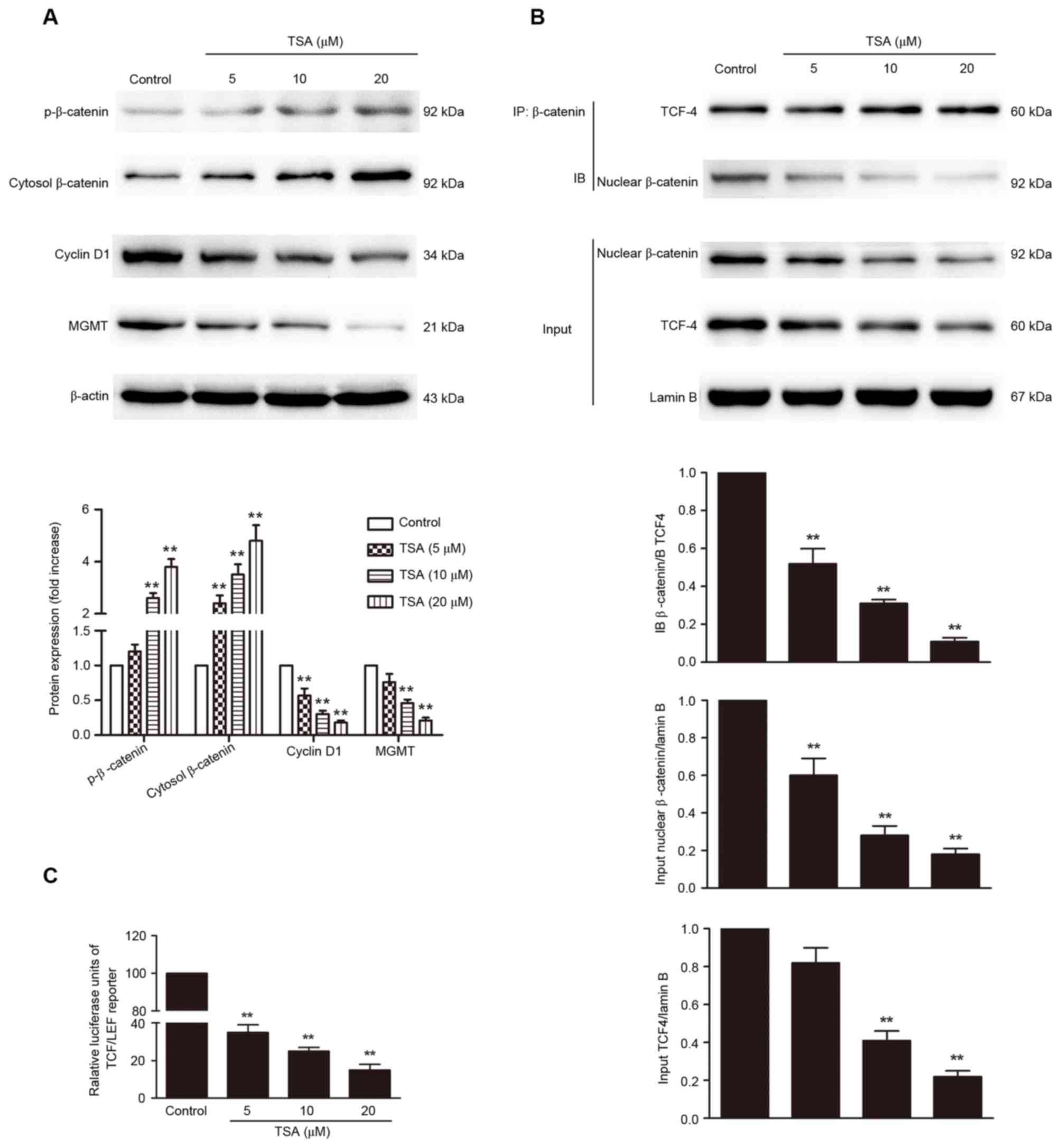
TSA inhibited Wnt/β-catenin/MGMT
pathway in AtT-20 cells
Dysregulation of the Wnt/β-catenin/MGMT pathway has
a pivotal role in the pathogenesis of pituitary adenoma. A strategy
to suppress the expression of MGMT by inhibition of Wnt/β-catenin
signaling may be useful as a novel treatment for pituitary adenoma.
Thus, whether TSA affects the Wnt/β-catenin/MGMT pathway was
investigated. TSA treatment increased β-catenin phosphorylation and
inhibited nuclear translocation of β-catenin in a dose-dependent
manner (P<0.01; Fig. 3A and B).
The levels of cyclin D1 and MGMT were detected to determine whether
the inactivation of β-catenin mediated by TSA decreases the
transcription of target genes. The protein levels of cyclin D1 and
MGMT were significantly decreased by TSA treatment compared with
the control (P<0.01; Fig. 3A).
Importantly, co-IP results indicated that the interaction between
β-catenin and TCF-4 was significantly reduced by TSA treatment in a
dose-dependent manner (Fig. 3B,
P<0.01). In accordance with these results, TSA treatment of
AtT-20 cells transfected with a TCF-LEF reporter resulted in a
dose-dependent reduction in TCF-LEF reporter activity (P<0.01;
Fig. 3C).
TSA induces apoptosis via inhibition
of Wnt/β-catenin/MGMT pathway in AtT-20 cells
To investigate whether Wnt/β-catenin/MGMT pathway
was involved in TSA-mediated apoptosis, β-catenin knock down AtT-20
cells were infected with MGMT lentiviral activation particles. The
inhibition of β-catenin nuclear translocation (Fig. 4A), decrease in TCF-LEF reporter
activity (Fig. 4B, P<0.01),
reduction in MGMT expression (Fig.
4A), phophatidylserine externalization (Fig. 4C), caspase-3 activation (Fig. 4D), DNA fragmentation (Fig. 4E), LDH leakage (Fig. 4F) and decrease in cell viability
(Fig. 4G) induced by TSA were all
significantly augmented in β-catenin knock down AtT-20 cells
(P<0.01). However, overexpression of MGMT in β-catenin knock
down AtT-20 cells reversed TSA-mediated phophatidylserine
externalization (P<0.01; Fig.
4C), caspase-3 activation (P<0.01; Fig. 4D), DNA fragmentation (P<0.01;
Fig. 4E), LDH leakage (P<0.01;
Fig. 4F) and decrease in cell
viability (P<0.01; Fig. 4G),
suggesting that Wnt/β-catenin/MGMT inactivation is responsible for
TSA-mediated antitumor effects.
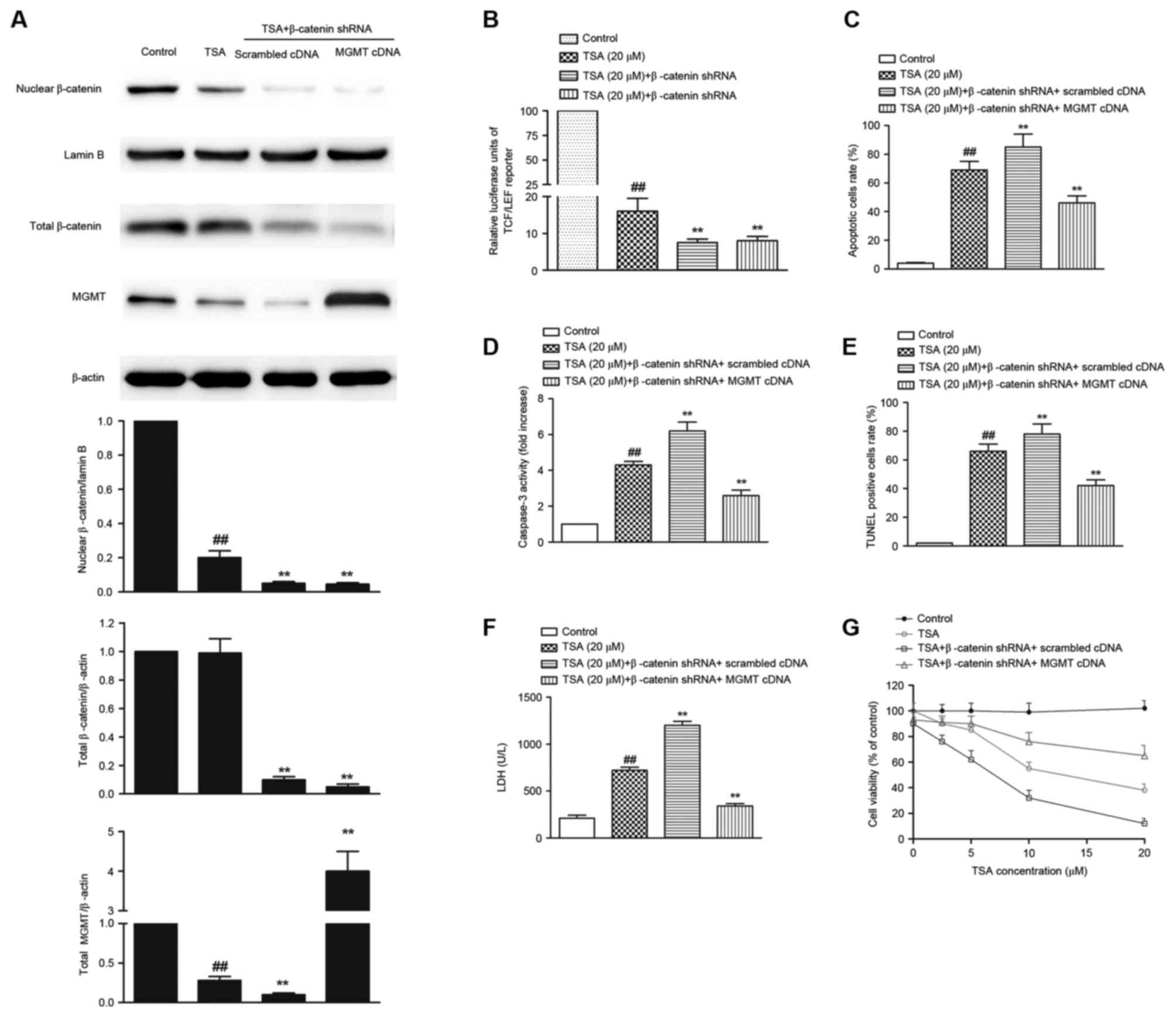 | Figure 4.MGMT overexpression abolishes
TSA-mediated apoptosis in β-catenin knock down AtT-20 cells. (A)
The protein expression of nuclear β-catenin, total β-catenin and
MGMT determined by western blot. (B) TCF-LEF reporter luciferase
activity in β-catenin knock down AtT-20 cells. MGMT overexpression
abolished TSA-mediated increase in (C) the apoptotic cells rate,
(D) caspase-3 activation, (E) DNA fragmentation, (F) LDH leakage
and (G) the decrease in cell viability. ##P<0.01 vs.
control; **P<0.01 vs. TSA treatment alone. TSA, Tanshinone IIA;
p-β-catenin; phosphorylated β-catenin; MGMT,
O6-methylguanine-DNA methyltransferase; TCF-4, T-cell
factor; LEF, lymphoid-enhancing factor; TUNEL, terminal
deoxynucleotidyl transferase dUTP nick-end labeling; LDH, lactate
dehydrogenase. |
Discussion
The current study confirmed that TSA induces
apoptosis in AtT-20 cells. Apoptosis is generally characterized by
phophatidylserine externalization, depolarization of mitochondrial
membrane, caspase-3 activation and DNA fragmentation (11,12).
Previous studies have indicated that TSA has anti-tumor effects on
glioma cells (13), osteosarcoma
cells (14) and gastric cancer
cells (15). In the present study,
TSA treatment resulted in cell viability reduction,
phophatidylserine externalization, mitochondrial membrane potential
disruption, caspases-3 activation and DNA fragmentation in AtT-20
cells. These results suggest that TSA induced apoptosis in AtT-20
cells, suggesting that TSA may be a candidate drug for treatment of
pituitary adenoma.
The principal finding of this study is that TSA
induces apoptosis in AtT-20 cells via downregulating
Wnt/β-catenin/MGMT signaling. The canonical Wnt/β-catenin cascade,
the non-canonical planar cell polarity pathway and the
Wnt/Ca2+ pathway are involved in Wnt receptor activation
(16). The Wnt/β-catenin pathway
can regulate MGMT gene expression in cancer, and inhibition of Wnt
signalling can prevent chemoresistance (17). Dysregulation of the
Wnt/β-catenin/MGMT pathway has a pivotal role in the pathogenesis
of pituitary adenoma (18,19). In the present study, TSA increased
the phosphorylation of β-catenin, and inhibited its nuclear
translocation in AtT-20 cells. Notably, co-IP results revealed that
β-catenin/TCF-4 binding was significantly reduced by TSA treatment,
meanwhile, TSA treatment of AtT-20 cells transiently transfected
with TCF-LEF reporter resulted in a marked decrease in TCF-LEF
activity. These results indicated that when activated β-catenin
translocated to the nucleus, combined with TCF-4 and interacted
with TCF-LEF, resulting in transcription of target genes, including
cyclin D1 and MGMT. The results of the present study are in
agreement with a previous study, which observed that TSA inhibited
the transdifferentiation of renal tubular epithelial cells induced
by high glucose via downregulation of the Wnt/beta-catenin
signaling pathway activity (20).
Notably, TSA-mediated suppression of
Wnt/β-catenin/MGMT pathway and apoptosis were exacerbated in
β-catenin KO cells, however, TSA-mediated apoptosis was abolished
by MGMT overexpression in β-catenin knock down cells. To the best
of our knowledge, this study is the first to demonstrate that
Wnt/β-catenin/MGMT inactivation is mediates for TSA-induced
apoptosis in AtT-20 cells.
In conclusion, TSA induced apoptosis by inactivating
the Wnt/β-catenin/MGMT pathways in AtT-20 cells, which provides a
novel insight into the mechanism underlying the antitumor effects
of Salvia miltiorrhiza extract. The results of the present
study must be further investigated, particularly through the use of
an in vivo model.
Acknowledgements
This project was supported by the Basic research
projects of Shenzhen Science and Technology Program (grant nos.
JCYJ20140414170821309, JCYJ20150306114616, JCYJ20150330102720152
and JCYJ2920831101643895), the Natural Science Foundation of
Guangdong (grant no. 2015A030313777), the Research Fund from
Shenzhen Key Laboratory of Neurosurgery (grant no.
ZDSYS20140509173142601).
References
|
1
|
Gurcan O, Gurcay AG, Kazanci A, Yildirim
AE, Turkoglu OF, Komurcu HF and Beskonakli E: Trigeminal neuralgia
as an unusual isolated symptom of pituitary Adenoma: Case report
and review of the literature. Turk Neurosurg. 26:180–183.
2016.PubMed/NCBI
|
|
2
|
Bengtsson D, Schrøder HD, Andersen M,
Maiter D, Berinder K, Rasmussen U Feldt, Rasmussen ÅK, Johannsson
G, Hoybye C, Van Der Lely AJ, et al: Long-term outcome and MGMT as
a predictive marker in 24 patients with atypical pituitary adenomas
and pituitary carcinomas given treatment with temozolomide. J Clin
Endocrinol Metab. 100:1689–1698. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
McCormack A, Kaplan W, Gill AJ, Little N,
Cook R, Robinson B and Clifton-Bligh R: MGMT expression and
pituitary tumours: Relationship to tumour biology. Pituitary.
16:208–219. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen YP, Hou XY, Yang CS, Jiang XX, Yang
M, Xu XF, Feng SX, Liu YQ and Jiang G: DNA methylation and histone
acetylation regulate the expression of MGMT and chemosensitivity to
temozolomide in malignant melanoma cell lines. Tumour Biol.
37:11209–11218. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wickström M, Dyberg C, Milosevic J, Einvik
C, Calero R, Sveinbjörnsson B, Sandén E, Darabi A, Siesjö P, Kool
M, et al: Wnt/β-catenin pathway regulates MGMT gene expression in
cancer and inhibition of Wnt signalling prevents. chemoresistance.
6:89042015.
|
|
6
|
Khattak MN, Buchfelder M, Kleindienst A,
Schöfl C and Kremenevskaja N: CRH and SRIF have opposite effects on
the Wnt/β-catenin signalling pathway through PKA/GSK-3β in
corticotroph pituitary cells. Cancer Invest. 28:797–805. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Das A, Miller R, Lee P, Holden CA,
Lindhorst SM, Jaboin J, Vandergrift WA III, Banik NL, Giglio P,
Varma AK, et al: A novel component from citrus, ginger, and
mushroom family exhibits antitumor activity on human meningioma
cells through suppressing the Wnt/β-catenin signaling pathway.
Tumour Biol. 36:7027–7034. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xing Y, Tu J, Zheng L, Guo L and Xi T:
Anti-angiogenic effect of tanshinone IIA involves inhibition of the
VEGF/VEGFR2 pathway in vascular endothelial cells. Oncol Rep.
33:163–170. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bai Y, Zhang L, Fang X and Yang Y:
Tanshinone IIA enhances chemosensitivity of colon cancer cells by
suppressing nuclear factor-κB. Exp Ther Med. 11:1085–1089. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Prokhorova EA, Zamaraev AV, Kopeina GS,
Zhivotovsky B and Lavrik IN: Role of the nucleus in apoptosis:
Signaling and execution. Cell Mol Life Sci. 72:4593–4612. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Jellinger KA: Challenges in neuronal
apoptosis. Curr Alzheimer Res. 3:377–391. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hunter AL, Choy JC and Granville DJ:
Detection of apoptosis in cardiovascular diseases. Methods Mol Med.
112:277–289. 2005.PubMed/NCBI
|
|
13
|
Ding L, Ding L, Wang S, Wang S, Wang W,
Wang W, Lv P, Lv P, Zhao D, Zhao D, et al: Tanshinone IIA affects
autophagy and apoptosis of glioma cells by inhibiting
phosphatidylinositol 3-kinase/Akt/mammalian target of rapamycin
signaling pathway. Pharmacology. 99:188–195. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Huang ST, Huang CC, Huang WL, Lin TK, Liao
PL, Wang PW, Liou CW and Chuang JH: Tanshinone IIA induces
intrinsic apoptosis in osteosarcoma cells both in vivo and in vitro
associated with mitochondrial dysfunction. Sci Rep. 7:403822017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu J, Wang X, Li Y and Tang B: Tanshinone
IIA suppresses gastric cancer cell proliferation and migration by
downregulation of FOXM1. Oncol Rep. 37:1394–1400. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Clevers H: Wnt/beta-catenin signaling in
development and disease. Cell. 127:469–480. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wickström M, Dyberg C, Milosevic J, Einvik
C, Calero R, Sveinbjörnsson B, Sandén E, Darabi A, Siesjö P, Kool
M, et al: Wnt/β-catenin pathway regulates MGMT gene expression in
cancer and inhibition of Wnt signalling prevents chemoresistance.
Nat Commun. 6:89042015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Formosa R, Gruppetta M, Falzon S, Santillo
G, DeGaetano J, Xuereb-Anastasi A and Vassallo J: Expression and
clinical significance of Wnt players and survivin in pituitary
tumours. Endocr Pathol. 23:123–131. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Maïza JC and Caron P: Pituitary carcinomas
and aggressive adenomas: An overview and new therapeutic options.
Ann Endocrinol (Paris). 70 Suppl 1:S12–S19. 2009.(In French).
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Huang BY, Cao LY and Fu XG: Effects of
tanshinone IIA on Wnt/beta-catenin signaling pathway of high
glucose induced renal tubular epithelial cell transdifferentiation.
Zhongguo Zhong Xi Yi Jie He Za Zhi. 32:965–969. 2012.(In Chinese).
PubMed/NCBI
|