Introduction
Immunoglobulin G4-related disease (IgG4-RD) is a
chronic disorder of an unknown etiology with multiple organ
involvement. It was first observed in the pancreas and is now known
as type 1 autoimmune pancreatitis (AIP)/IgG4-related pancreatitis
(1–3). This condition has been observed to
affect virtually every organ: Orbital contents; bile ducts;
salivary glands; retroperitoneum; pachymeninges; kidneys; lungs;
lymph nodes; aorta; arteries; breast; prostate; thyroid;
pericardium; and skin (3,4). Clinicopathological features
demonstrate striking similarities among the involved organs,
including a tendency to form tumefactive lesions, a dense
lymphoplasmacytic infiltrate rich in IgG4-positive plasma cells,
storiform fibrosis, frequent elevations of serum IgG4 levels and
good response to glucocorticoids (5,6).
However, as a member of a broad spectrum of IgG4-RD diseases,
IgG4-R ophthalmic (O)D (affects lacrimal glands, extraocular
muscles, orbital nerve and eyelid) differs from the other members
of the IgG4-RD group by two aspects: Fibrosis is not necessarily
marked histopathologically, and germinal centers (GCs) are
frequently observed (3,7). GCs are distinct structures within the
B cell follicles in the secondary lymphoid tissues and are major
sites of somatic hypermutation, class switching-associated
recombination and affinity maturation of activated B cells, and the
production of plasma cells and memory B cells (8,9). GCs
depend on cluster of differentiation 4-positive (CD4+) T
cells and it is now recognized that a subset of CD4+ T
cells, termed follicular B helper T (Tfh) cells, are the true
helper cells for B cells in antibody (Ab) responses (10,11).
Tfh cells were first described in the year 2000 when
several groups reported that a significant proportion of C-X-C
chemokine receptor type 5 (CXCR5)+CD4+ T
cells involved in B cell help for Ab responses in tonsils was
situated in GCs or follicles. These cells were subsequently termed
Tfh cells (12,13). Following 17 years of research,
there is now an improved understanding of the cellular and
molecular mechanisms of helper functions of Tfh cells. The CXCR5
serves as a surface marker for Tfh cells, CD4+ T cells expressing
CXCR5 can migrate in response to the follicular chemokine B
lymphocyte chemoattractant (also known as CXCL13), and relocate to
the border of T and B cell zone (T-B junction) and to the follicles
in secondary lymphoid organs (14,15).
B cell lymphoma 6 (Bcl-6) was strongly expressed in Tfh cells but
not in T helper (Th)1 or 2 cells (16). Bcl-6 has been identified as a
master regulator of Tfh cell differentiation in 2009 (17–19),
in the absence of Bcl-6, Tfh cell differentiation ceases in
vivo (17–19) while other CD4+ Th cell subsets are
relatively unaffected (18,19).
Tfh cells also express high levels of inducible T-cell costimulator
(ICOS) and interleukin (IL)-21 (13,16,20).
IL-21 is a Tfh cell-expressed helper cytokine (10) and the upregulation of ICOS is
essential for the initiation and maintenance of Tfh differentiation
(21,22).
The pathogenesis of IgG4-RD remains to be
elucidated. Researchers in the field of immunology identified
certain abnormal immunological mechanisms involved in the
pathogenesis of IgG4-RD. The results suggested that Th2 cells and
regulatory T cells (Tregs) as well as associated cytokines,
including IL-4, IL-10, IL-13 and transforming growth factor β1, may
serve key roles in the development of Mikulicz disease,
IgG4-related pancreatitis and cholangitis (23,24).
More recently, studies on the role of Th2 cytokines in IgG4-RD
indicated that IL-18 and interferon-γ are implicated in the
pathogenesis of IgG4-related dacryoadenitis and sialoadenitis
(25,26). Currently, increasing attention is
paid to Tfh cells; a higher frequency of GCs in patients with
IgG4-ROD indicates an increased number of activated B cells, which
may reflect the increase in Tfh cell number.
An association between Tfh cells and autoimmunity
has been suggested in many autoimmune diseases, including systemic
lupus erythematosus (27,28), autoimmune thyroid disease (29) and myasthenia gravis (30,31).
Since the major function of Tfh cells is to aid B cell activation
and to stimulate Ab responses, patients with IgG4-RD display
elevated IgG4 concentrations in sera and infiltrated IgG4-positive
cells in associated tissues, suggesting the involvement of Tfh
cells in the pathogenesis. In the studies performed with peripheral
blood samples, Tfh cells were related to the pathogenic process of
IgG4-RD (32,33). The profile and distribution of Tfh
cells in involved tissue, which demonstrates the association
between Tfh cells and IgG4-RD directly, has not been examined among
patients with IgG4-RD and IgG4-ROD patients demonstrating GC
formation.
In the following study, the expression of ICOS and
Bcl-6 was increased in tissues of patients with IgG4-RD compared
with IgG4-negative controls. It was also demonstrated that
CD4+CXCR5+ Tfh cells were more abundant in
patients with IgG4-RD compared with the IgG4-negative controls.
Among patients with IgG4-RD, CD4+CXCR5+ Tfh
cells were present in and outside GCs in patients with IgG4-ROD and
IgG4-related lymphadenopathy (IgG4-RL) while staining for CD4 and
CXCR5 in pancreatic lesions appeared as a randomly distributed
pattern. Fewer CD4+CXCR5+ Tfh cells were
observed in patients with type 1 AIP compared with patients with
IgG4-ROD and IgG4-RL. The expression of IL-21 was coincidental with
dual immunofluorescence of CD4 and CXCR5, and increased expression
of IL-21 was observed in patients with IgG4-ROD and IgG4-RL. IL-21
expression in patients with Ig-G4-ROD was positively correlated
with the IgG4/IgG ratio in immunohistochemically (IHC)-positive
cells.
Materials and methods
Patients and samples
A total of 7 patients with IgG4-ROD (4 men and 3
women; mean age ± standard deviation age 60.9±7.5 years) referred
to the Department of Ophthalmology at the Shanghai Changzheng
Hospital (Shanghai, China) between January 2013 and June 2016 were
included in the present study. A total of 7 patients with type 1
AIP or IgG4-RL (6 men and 1 women; mean age ± standard deviation
age 56.0±7.0 Years) who were referred to Renji Hospital (Shanghai,
China) between July 2012 and June 2016 were studied as disease
positive controls. Cases with normal serum IgG4 and no
IgG4-positive cells detected in tissue specimens (n=7) were
designated as IgG4-negative controls. IgG4-RD was diagnosed using
the following criteria (5,34): i) Clinical examination reveals
diffuse/localized swelling or masses in single or multiple organs;
ii) tissue biopsy meets at least two out of three major
histopathologic features of the disease: A lymphoplasmacytic
infiltrate, fibrosis (typically storiform); and obliterative
phlebitis; and iii) infiltration of IgG4-positive plasma cells:
Ratio of IgG4-positive cells to IgG-postive cells of >40% and
>10 IgG4-positive cells/high power field (magnification,
×400).
The lesion specimens were collected from all
patients with IgG4-RD from whom written informed consent was
obtained, and tissues were used only for research purposes. The
study was approved by the Ethics Committee of Renji Hospital.
IHC staining
The primary antibodies used for the IHC were as
follows: Anti IgG4 (cat. no. ab109493; Abcam, Shanghai, China; at
1/500 dilution); IgG (cat. no. ab109489; Abcam, Shanghai, China; at
1/300 dilution); Bcl-6 (cat. no. ab41845; Abcam, Shanghai, China;
at 1/50 dilution); ICOS (cat. no. ab105227; Abcam, Shanghai, China;
at 1/50 dilution); IL-21 (cat. no. 118510; and Abcam, Shanghai,
China; at 1/200 dilution).
IHC analyses
Paraffin sections (4 µm-thick) were deparaffinized
by immersion in xylene, followed by a hydration in ethanol. An
antigen retrieval was performed with EDTA pH 8.0 microwave repair
method. Endogenous peroxide activity was quenched by 3% hydrogen
peroxide in water. Then the sections were incubated for 30 min at
room temperature with 3% bovine serum albumin (BSA) (cat. no.
A8020; Beijing Solarbio Science and Technology Co., Ltd., Beijing,
China) and incubated overnight at 4°C with primary antibody.
Thereafter, the sections were incubated with a horseradish
peroxidase-conjugated secondary antibody (cat. no. K5007; Dako;
Agilent Technologies, Inc., Santa Clara, CA, USA; at 1/1 dilution)
for 50 min at room temperature. The stain was prepared using
diaminobenzidine substrate (cat. no. K5007; Dako; Agilent
Technologies, Inc.). Sections were counterstained 3 min at room
temperature with Harris hematoxylin and mounted prior to analysis.
Between each step of the analysis, sections were rinsed with PBS
three times, 5 min each. IHC images were captured using a light
microscope equipped with a digital camera (Olympus Corporation,
Tokyo, Japan). Stained IgG4- and IgG-positive cells were counted in
three high-power fields with the highest density of positive cells
and an average was calculated (5).
Semiquantification of IL-21 was performed as previously described
with a slight modification in the grade classification of staining
intensity and percentage of positive cells (35). Depending on the proportion of
positive stained cells (PP): <5% was scored 0, 6–25% was scored
1, 26–50% was scored 2, 51–75% was scored 3 and >75% was scored
4. According to the staining intensity (SI): No color was scored 0,
light yellow was scored 1, light brown was scored 2, brown was
scored 3. PP was multiplied by SI to obtain a total IHC score for
any given sample.
Double-labeled immunofluorescence for
CD4, CXCR5 and IL-21
Sections were de-waxed by xylene, then hydrated
through a graded ethanol series, and washed with distilled water.
Antigen retrieval and serum block were performed using a similar
method to the IHC staining. The primary antibodies used for the
analysis of co-expression of CD4 and CXCR5 were anti-CD4 (cat. no.
GB13064; Goodbio technology, Wuhan, Hubei, China; at 1/50
dilution), anti-CXCR5 (cat. no. GTX100351; GeneTex, Inc., Irvine,
CA, USA; at 1/100 dilution). The anti-IL-21 goat polyclonal
antibody was used to analyze the co-expression of IL-21 and CXCR5
(cat. no. GTX82914; GeneTex, Inc.; at 1/200 dilution while
anti-CXCR5 at 1/200 dilution). Cy3-conjugated goat anti-mouse IgG
(cat no. GB21301; Goodbio technology; at 1/300 dilution), Alexa
488-conjugated goat anti-rabbit IgG (cat no. GB25303; Goodbio
technology; at 1/400 dilution), Cy3-conjugated donkey anti-goat IgG
(cat no. GB21404; Goodbio technology; at 1/300 dilution) and Alexa
488-conjugated donkey anti-rabbit IgG (cat no. GB2540311; Goodbio
technology; at 1/400 dilution) were used as fluorochrome-conjugated
second antibodies. Then the sections were sequentially incubated
with primary antibodies overnight at 4°C, corresponding second
antibodies for 50 min at room temperature and DAPI for 10 min at
room temperature. Between each step, sections were rinsed with PBS
three times, 5 min each. Fluorescence images were captured using a
fluorescence microscope (Nikon Corporation).
Statistical analysis
Differences in the IHC staining scores between more
than two groups were determined using the Kruskal-Wallis test and
differences between two groups were determined using the
Mann-Whitney U test (two-tailed). Spearman's rank correlation
coefficient was used to assess the correlation between IL-21
expression and the ratio of IgG4- to IgG-positive cells in IgG4-ROD
samples. All statistical analyses were performed using SPSS
software (version 20; IBM Corp., Armonk, USA). P<0.05 was
considered to indicate a statistically significant difference.
Results
Patient profiles
The clinical and serological characteristics of
patients with IgG4-ROD are presented in Table I. Patients with IgG4-ROD were
predominantly middle-aged to elderly males. IgG4-ROD affected the
lacrimal gland in 6 patients the orbital soft tissue in 2 patients,
including the orbital apex. Computed tomography or magnetic
resonance imaging identified space-occupying lesions of lacrimal
gland or orbital cavity. Serum IgG and IgG4 were elevated in all
patients with a median level of 22.6 g/l (P25-P75: 16.2–31.3 g/l)
and 17.5 g/l (P25-P75: 8.5–38.5 g/l), respectively. With the
exception of 1 patient, the serum IgE value for all patients was
elevated above the normal limit (<165 IU/ml). Case 2 exhibited
paranasal sinusitis and case 5 exhibited bronchial asthma. Clinical
and serological characteristics of patients with type 1 AIP or
IgG4-RL and IgG4-negative controls are summarized in Table II. Immunohistochemistry results
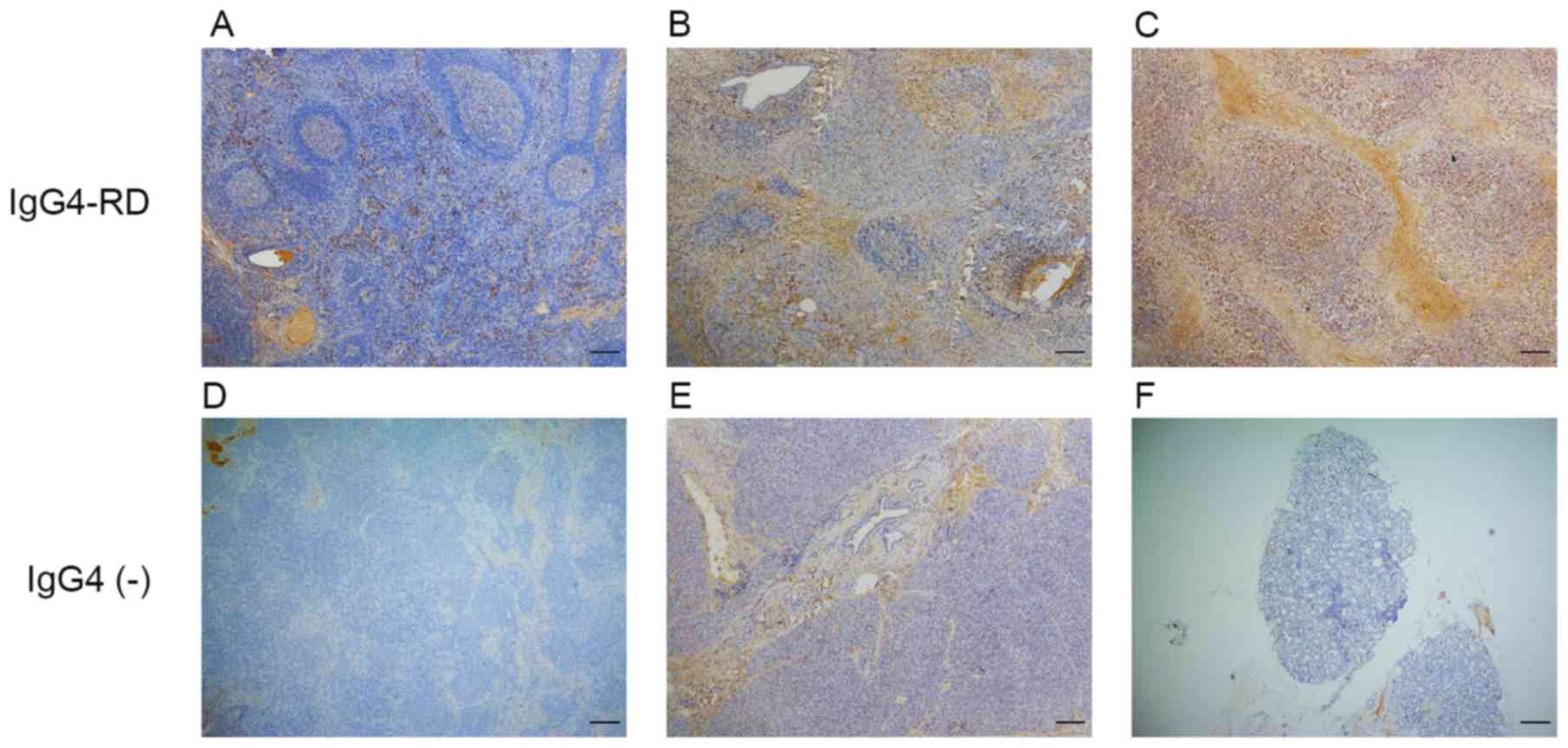
demonstrated that IgG4-positive cells mainly infiltrated the
interfollicular areas in lymph nodes (Fig. 1A), areas around the acinar and
ductal cells in pancreas (Fig. 1B)
and diffusely in lacrimal glands (Fig.
1C).
 | Table I.Clinical and serological
characteristics of patients with immunoglobulin G4-related
ophthalmic disease. |
Table I.
Clinical and serological
characteristics of patients with immunoglobulin G4-related
ophthalmic disease.
| Patient no. | Sex | Age at onset,
years | Onset time | Onset signs and
symptoms | Exophthalmos (mm)
Right eye (orbital distance) left eye) | Side | IgG (g/l) | IgG4 (g/l) | IgE IU/ml |
|---|
| 1 | M | 74 | 1 year | Diplopia,
proptosis, swelling | 15 (105) 14 | Bil | 33.7 | 38.5 | 368 |
| 2 | M | 57 | 8 years | Proptosis,
swelling | 24.5 (113) 31 | Bil | 31.3 | 43.2 | 415 |
| 3 | F | 68 | 3 months | Proptosis | 20 (103) 24 | Bil | 9.77 | 3.26 | 415 |
| 4 | F | 59 | 6 months | Swelling | 13 (110) 13 | Bil | 22.6 | 20.2 | 1140 |
| 5 | M | 59 | 5 years | Swelling | 14 (110) 20 | Bil | 18.9 | 12.3 | 5 |
| 6 | F | 52 | 5 months | Swelling,
ptosis | 14 (107)15 | Left | 16.2 | 8.48 | 495 |
| 7 | M | 57 | 7 years | Proptosis,
swelling | 26 (99) 28 | Bil | 25.8 | 17.5 | 319 |
 | Table II.Clinical and serological
characteristics of patients with type 1 AIP or IgG4-RL and
IgG4-negative controls. |
Table II.
Clinical and serological
characteristics of patients with type 1 AIP or IgG4-RL and
IgG4-negative controls.
| A, Patients with
type 1 AIP or IgG4-RL |
|---|
|
|---|
| Patient no. | Sex | Age at onset,
years | Affected organ | IgG4 (g/l) | Onset time | Onset signs and
symptoms |
|---|
| 8 | M | 57 | Pancreas, lymph
node | N | 0.5 months | Asyptomatic,
occupying |
| 9 | M | 67 | Pancreas | N | 10 days | Abdominal pain,
swelling |
| 10 | M | 62 | Pancreas | N | 2 months | Abdominal pain,
swelling |
| 11 | M | 49 | Pancreas, lymph
node | 10.1 | 2 weeks | Asyptomatic,
occupying |
| 12 | M | 54 | Lymph node | 28.1 | 1 years | Swelling |
| 13 | M | 56 | Lymph node | N | 0.5 months | Swelling |
| 14 | F | 47 | Lymph node | N | 4 years | Swelling |
|
| B, IgG4-negative
controls |
|
| Patient no. | Sex | Age at onset,
years | Affected organ | IgG4 (g/l) | Onset time | Onset signs and
symptoms |
|
| 15 | F | 25 | Labial gland | 0.85 | 1.5 years | Dry mouth, dry
skin |
| 16 | F | 52 | Orbital tissue | 0.35 | 10 years | Swelling |
| 17 | F | 44 | Peripancreatic
lymph node | 0.90 | 1 years | Abdominal pain,
swelling |
| 18 | M | 59 | Submandibular
gland | 1.27 | 1 month | Swelling |
| 19 | M | 43 | Pancreas | 0.71 | 1 year | Abdominal pain |
| 20 | F | 55 | Orbital tissue | 1.25 | 7 months | Orbital pain |
| 21 | F | 63 | Bile duct | 0.74 | 2 months | Yellowing and
pruritus of the skin |
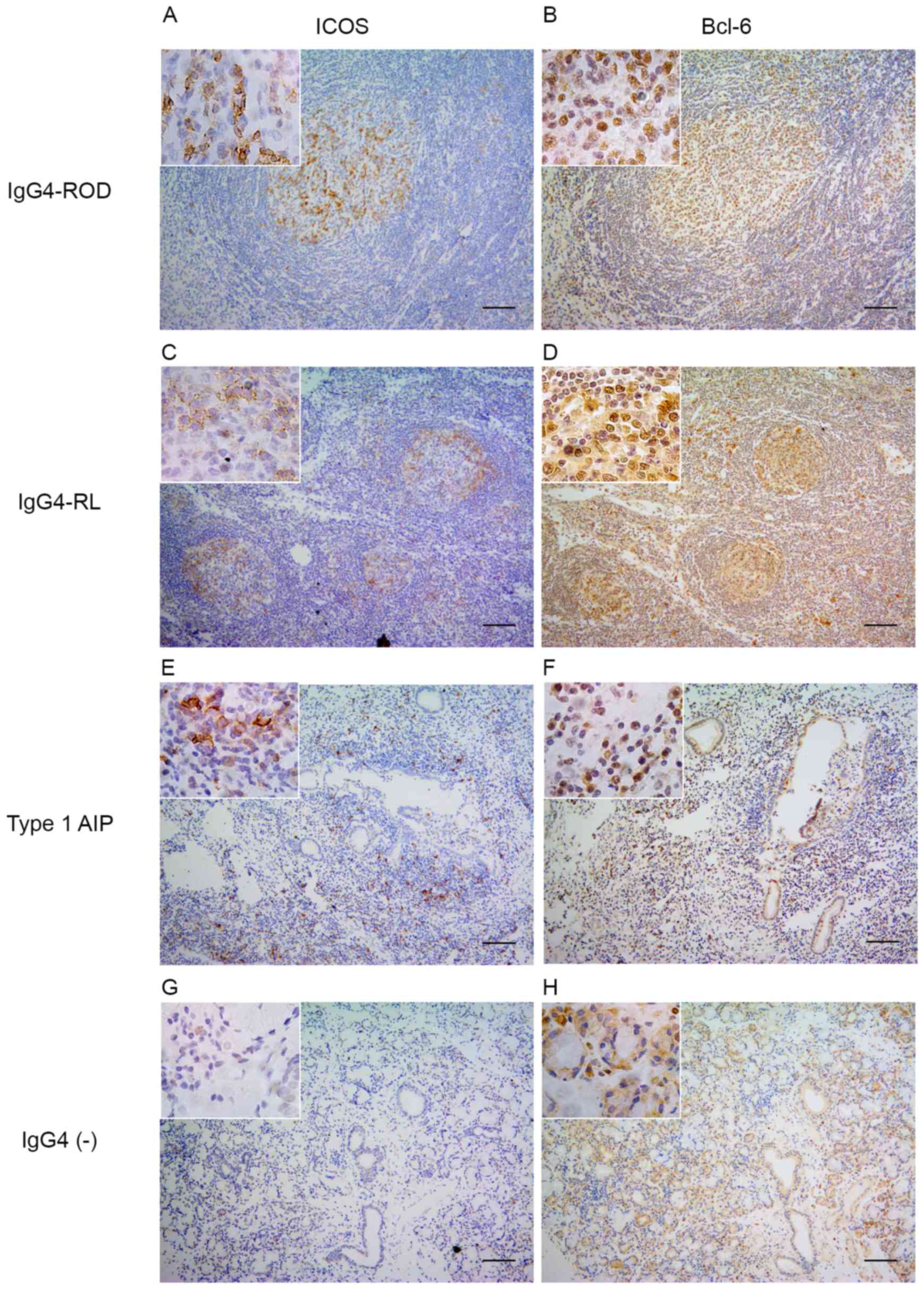
Expression of Bcl-6 and ICOS
The expression of Bcl-6, a master regulator
transcription factor in Tfh cells and ICOS, a surface marker of Tfh
cells, was examined (Fig. 2). In
patients with IgG4-ROD, the nuclear expression of Bcl-6 and
expression of ICOS were predominantly detected in GCs, and to a
lesser extent in infiltrating lymphocytes outside the GCs (Fig. 2A and B). A small proportion of
Bcl-6- and ICOS-positive cells were randomly distributed within the
pancreatic lesions (Fig. 2E and
F). In lymph nodes from patients with IgG4-RL positive staining
for Bcl-6 and ICOS was only observed in GCs (Fig. 2C and D). By contrast, the nuclear
expression of Bcl-6 was detected in relatively few lymphocytes in
all IgG4-negative controls and ICOS-positive cells were rarely
observed (Fig. 2G and H).
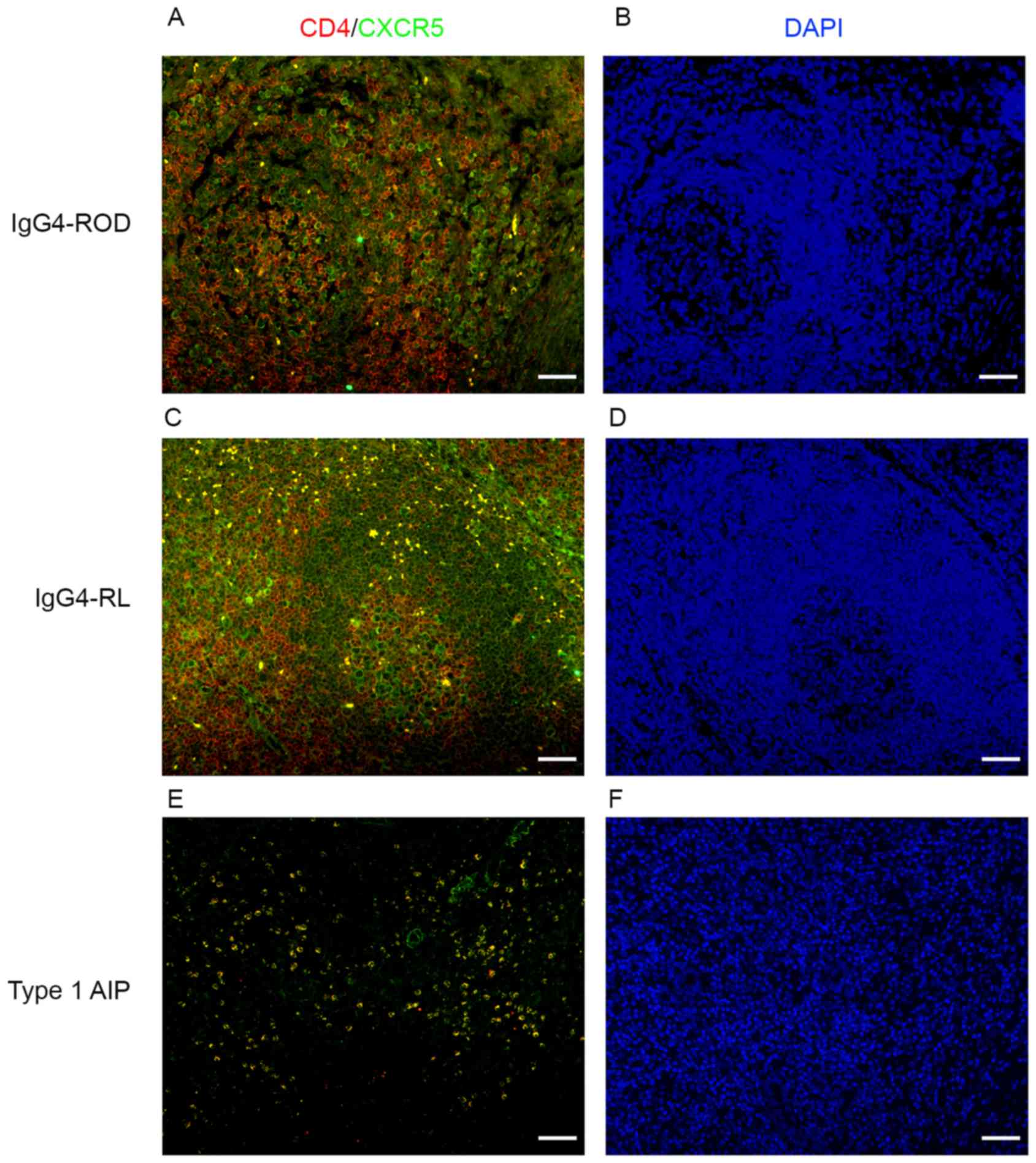
Increased number of
CD4+CXCR5+ cells in involved tissues of
patients with IgG4-RD
The above results indicated that expression of Bcl-6
and ICOS, which are the markers for Tfh cells, were increased in
patients with IgG4-RD compared with patients with IgG4-negative.
CD4+CXCR5+ cells were examined using dual
immunofluorescence staining for CD4 and CXCR5 using
paraffin-embedded specimens from patients with IgG4-RD and
IgG4-negative patients (Figs. 3
and 4). In the IgG4-RD group,
CD4+CXCR5+ cells were randomly distributed in
areas of type 1 AIP (Figs. 3 and
4E) and were predominantly
detected in GCs and T-B junctions in lymph nodes from IgG4-RL
(Fig. 4C).
CD4+CXCR5+ cells were scattered and
distributed in GCs within the orbital lesions in patients with
IgG4-ROD (Figs. 3 and 4A). In IgG4-negative controls, numerous
CD4-positive cells were observed; however, few
CD4+CXCR5+ cells could be identified
(Fig. 3). The number of
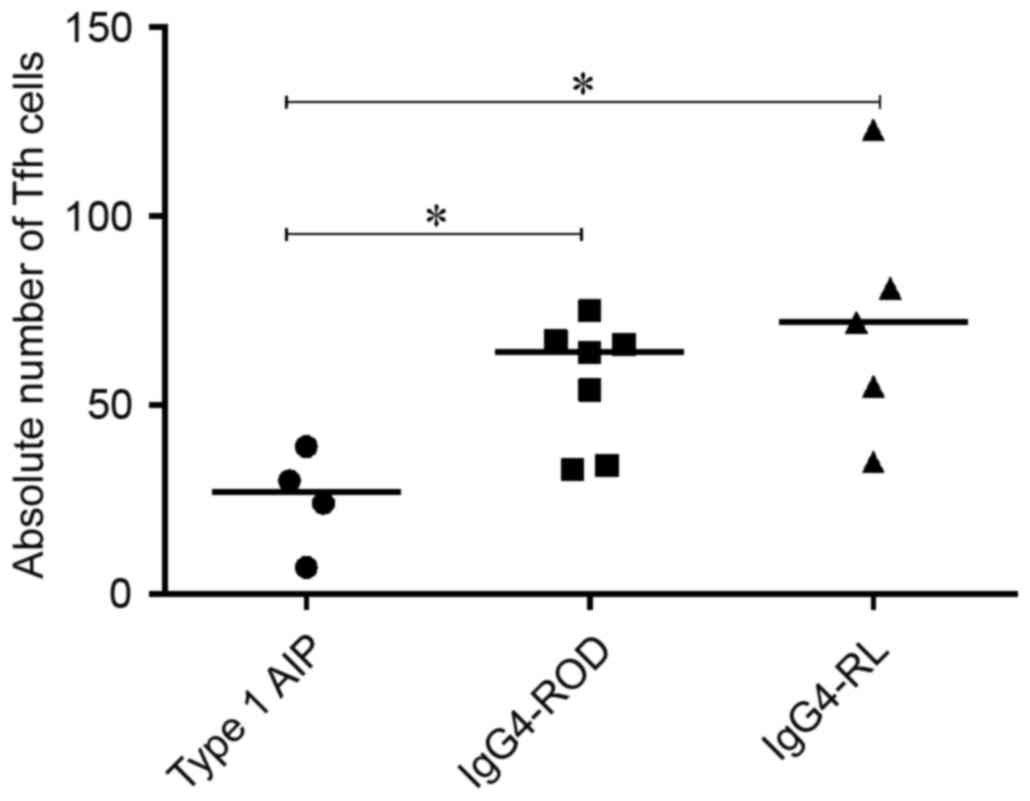
CD4+CXCR5+ cells among involved tissues from
patients with IgG4-RD demonstrated significantly decreased
CD4+CXCR5+ cells in patients with type 1 AIP
compared with patients with IgG4-ROD and IgG4-RL (P=0.024 and
0.032, respectively), there was no significant difference between
IgG4-ROD and IgG4-RL (Fig. 5).
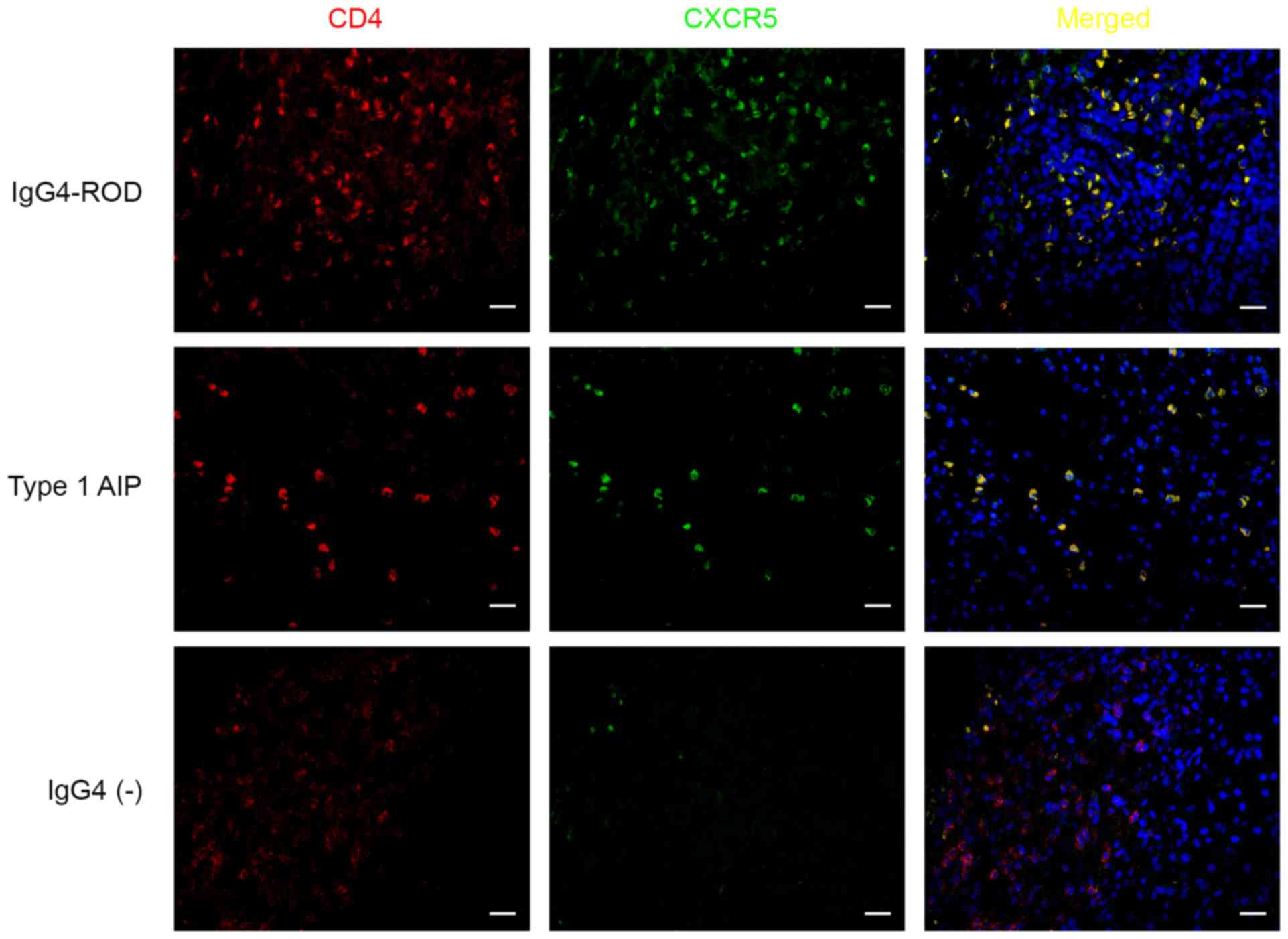 | Figure 3.CD4+CXCR5+
cells in involved tissues of patients with IgG4-RD. Sections were
stained for CD4 (red), CXCR5 (green), cells in yellow indicate the
coexpression of CD4 and CXCR5. Upper row, lacrimal gland; middle
row, pancreas; lower row, pancreas. Scale bars, 20 µm;
magnification, ×400. IgG4-RD, immunoglobulin G4-related disease;
CD4, cluster of differentiation 4; CXCR5, C-X-C chemokine receptor
type 5. |
Expression of IL-21 in involved
tissues of patients with IgG4-RD
IL-21 is the main effector cytokine produced by Tfh
cells (10) and the present study,
using immunohistochemical analyses, determined whether IL-21 is
expressed in samples from patients with IgG4-RD. All 16 IgG4-RD
samples expressed IL-21 (Fig. 6A).
IL-21 was prominent in GCs and less evident in infiltrating
lymphocytes outside the GCs in orbital lesions and lymph nodes.
Staining for IL-21 in pancreatic lesions appeared as a scattered
pattern, primarily in infiltrating lymphocytes around the acinar
and slightly detected in ductal cells. Fluorescence microscopy
revealed that the IL-21 expression in involved tissues from
patients with IgG4-RD coincided with their CXCR5 expression
(Fig. 6B). While the scores from
the IHC staining exhibit decreased expression of patients with
IL-21 in type 1 AIP compared with patients with IgG4-ROD and
IgG4-RL (P=0.042 and 0.016, respectively), the scores were
comparable between IgG4-ROD and IgG4-RL patients (Fig. 6C).
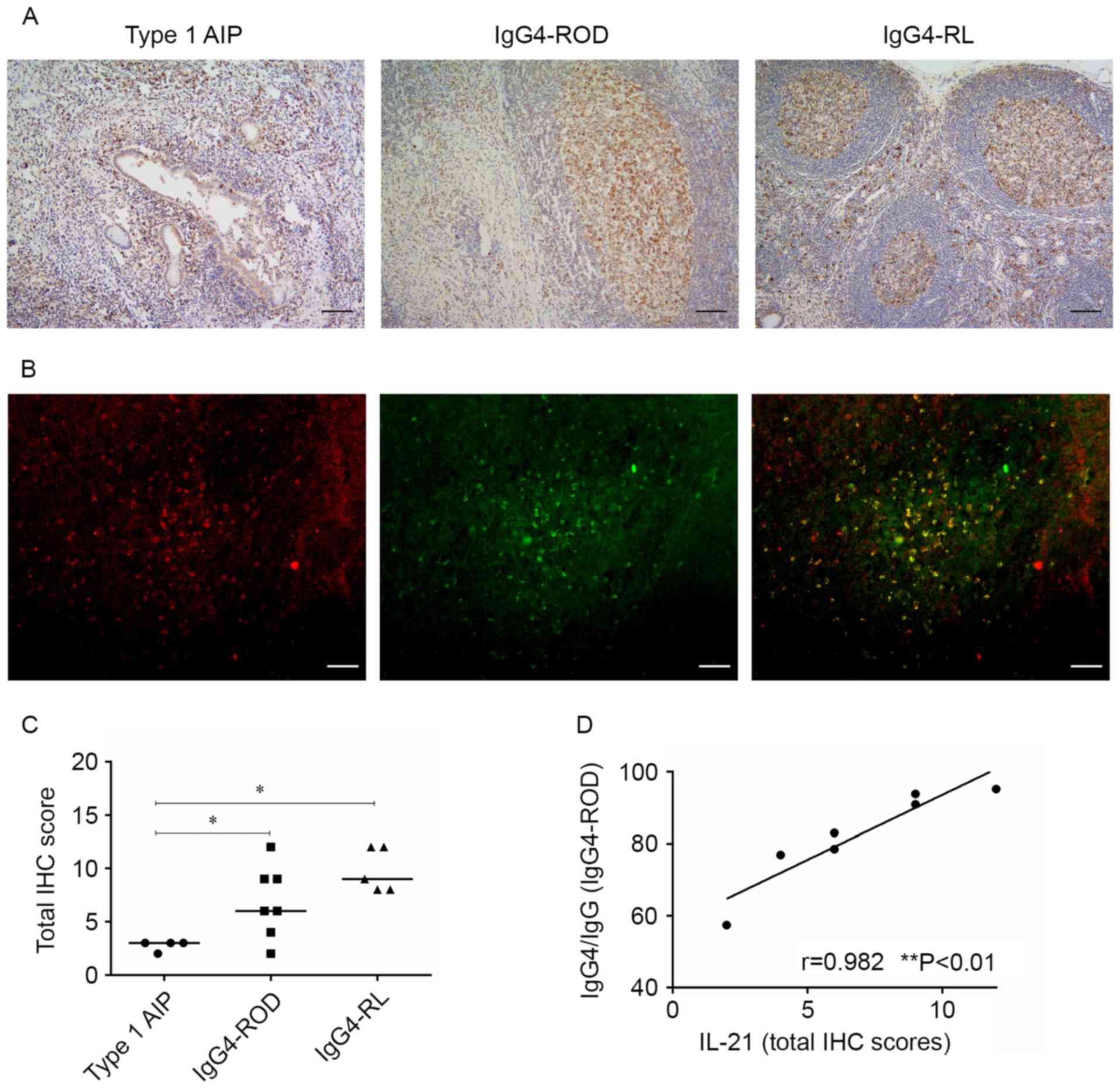 | Figure 6.Expression of IL-21 in involved
tissues of patients with IgG4-RD. (A) Representative micrographs
stained for IL-21 of pancreas (left), orbital soft tissue (middle),
and lymph node (right) with IgG4-RD (scale bar, 100 µm;
magnification, ×100). (B) Representative micrographs were from the
orbital soft tissue in IgG4-ROD patients. Immunofluorescent double
staining of IL-21-positive cells (red) and CXCR5+ cells
(green), cells in yellow indicate the coexpression of IL-21 and
CXCR5 (scale bars, 50 µm; magnification, ×200). (C) Total IHC
scores for IL-21 for patients with IgG4-RD. *P<0.05. (D)
Correlation between IgG4 production and IL-21 IHC scores in orbital
specimens from IgG4-ROD as determined by Spearman's rank test.
IgG4-RD, immunoglobulin G4-related disease; IgG4-ROD,
immunoglobulin G4-related ophthalmic disease; type 1 AIP, type 1
autoimmune pancreatitis; IgG4-RL, immunoglobulin G4-related
lymphadenopathy; CXCR5, C-X-C chemokine receptor type 5; IL-21,
interleukin 2; IHC, immunohistochemistry. |
Association between IgG4 production
and expression of IL-21 in patients with IgG4-ROD
The association between IgG4 production and the
levels of IL-21 IHC scores in orbital lesions was examined.
Spearman's rank correlation analysis demonstrated that the IL-21
IHC scores in orbital lesions were positively correlated with the
ratio of IgG4-positive cells to IgG-positive cells in patients with
IgG4-ROD (Fig. 6D).
Discussion
The present study analyzed the expression of the key
Tfh cell immunological proteins ICOS, Bcl-6, CXCR5 and IL-21 in the
affected tissues of patients with IgG4-RD, using IHC and dual
immunofluorescence. The results demonstrated increased expression
of ICOS and Bcl-6 in the involved tissues of patients with IgG4-RD
compared with the IgG4-negative control group. The
CD4+CXCR5+ Tfh cells were more prevalent in
IgG4-RD and scarce in the IgG4-negative control group. Among
patients with IgG4-RD, CD4+CXCR5+ Tfh cells
were detected inside and ouside GCs in patients with IgG4-ROD and
IgG4-RL, whereas staining for CD4 and CXCR5 in pancreatic lesions
appeared as a randomly distributed pattern. A decreased number of
CD4+CXCR5+ Tfh cells was observed in patients
with type 1 AIP compared with IgG4-ROD and IgG4-RL patients. The
expression of IL-21 was almost coincidental with the dual
immunofluorescence of CD4 and CXCR5 and elevated expression of
IL-21 was observed in patients with IgG4-ROD and IgG4-RL. IL-21
expression was positively correlated with the IgG4/IgG ratio in IHC
positive cells.
Naive CD4+ T cells may differentiate into one of
several lineages of Th cells and exert a variety of immunological
functions. These include clearing viruses, helminths and fungi by
Th1, Th2 and Th17 cells, respectively, and suppressing immune
responses by induced Treg cells (36). Tfh cells are a subset of CD4+ T
cells that provide help for B cells during the GC reaction
(10). A large GC is frequently
formed within involved organs in individuals with IgG4-ROD
(7,37) and is presumably the source of
IgG4-positive plasma cells. Based on this observation, the present
study targeted IgG4-ROD and investigated the Tfh cell abundance in
affected tissues.
Tfh cells depend on the expression of the master
regulator transcription factor Bcl-6 and distinguishing features of
Tfh cells are the expression of ICOS, CXCR5, IL-21, programmed cell
death protein 1 and SLAM-associated protein (10). The analysis of the expression of
Bcl-6 and ICOS in patients with IgG4-RD and IgG4-negative controls,
revealed elevated expression of ICOS and Bcl-6 in the involved
tissues of patients with IgG4-RD. Tfh cells require upregulation of
Bcl-6 to develop and perform their function in B cell maturation in
GCs (38). Bcl-6 binding is
associated with the control of Tfh cell migration and repression of
alternative cell fates (39). The
upregulation of ICOS is essential for the initiation and
maintenance of Tfh differentiation, and
CD4+CXCR5+ cells were scarce in mice with B
cell-specific deletion of ICOS ligand (11,21).
Increased expression of both Bcl-6 and ICOS in involved tissues of
patients with IgG4-RD indicated the presence of Tfh cells.
Dual immunofluorescence of paraffin-embedded
specimens revealed increased numbers of
CD4+CXCR5+ Tfh cells in all cases of IgG4-RD
irrespective of the organs involved, and an enhanced expression of
Bcl-6 and ICOS, which histopathologically indicated that this cell
population was involved in the pathogenesis of IgG4-RD. The
frequency and distribution of CD4+CXCR5+ Tfh
cells differed in patients with IgG4-ROD and type 1 AIP.
CD4+CXCR5+ Tfh cells were observed inside and
outside GCs in patients with IgG4-ROD, however they were randomly
distributed and fewer in areas of type 1 AIP compared with
IgG4-ROD. The distribution of CD4+CXCR5+ Tfh cells corresponded
with the expression of Bcl-6 and ICOS. CXCR5 is the canonical Tfh
marker and in the present study, CD4+CXCR5+
cells which localized in GCs were considered as GC Tfh cells and
predecessors of those cells with Tfh-like characteristics
(localized in extrafollicles) were termed Pre-Tfh cells (10,11,40).
Human autoimmune diseases predominantly affect nonlymphoid tissues,
which frequently contain large numbers of infiltrating, activated
lymphocytes, as well as lymphoid-like tissues with a GC. Tfh cells
have been identified in nonlymphoid tissues, particularly in
autoimmune diseases (41). High
frequency of GC formation and an increased number of Tfh cells in
IgG4-ROD may suggest that IgG4-ROD is an autoimmune disease and may
also indicate that Tfh cells serve a different role in IgG4-ROD and
type 1 AIP, since GCs are not commonly observed in type 1 AIP
(42). Previous studies considered
blood CD4+CXCR5+ cells as equivalents of Tfh cells. Akiyama et
al (32) demonstrated that the
proportion of circulating Tfh cells was significantly increased in
patients with IgG4-RD compared with patients with allergic rhinitis
or with healthy controls, and these results are consistent with
those obtained in the present study (11,15).
Tfh cells are a predominant source of IL-21
(43). In the present study, IL-21
was abundant in GCs in orbital lesions and lymph nodes, whereas
staining for IL-21 in pancreatic lesions appeared as a scattered
pattern, primarily in infiltrating lymphocytes around the acinar
and ductal cells. Expression of IL-21 was weaker in Type 1 AIP
patients compared with IgG4-ROD and IgG4-RL patients. The IL-21
expression in involved tissues from patients with IgG4-RD was
coincidental with their CXCR5 expression. Concurrent expression of
IL-21 and CXCR5, together with the distribution of IL-21-positive
cells indicates that the IL-21 in affected tissues of IgG4-ROD is
mainly produced by Tfh cells and the high expression of IL-21 in
IgG4-ROD is due to the increased number of Tfh cells. IgG4
production was positively correlated with the IL-21 IHC scores in
patients with IgG4-ROD. IL-21 can promote CD40L-mediated GC B cell
proliferation and drive human B cell differentiation into
Ig-secreting cells in vitro (44,45).
Additionally, IL-21 controls the maintenance and optimal affinity
maturation of the GC reaction by maintaining the Bcl-6 expression
in GC B cells in vivo (46,47).
IL-21 can also contribute to IgG4 production in Mikulicz's disease
(48) and the data obtained in the
present study demonstrates that Tfh-derived IL-21 can promote B
cell differentiation into IgG4-secreting cells in patients with
IgG4-ROD.
Based on the available data (32,33,41),
it can be hypothesized that chronic stimulation of orbital tissues
by an unknown antigen induces production of CXCL13, which in turn
recruits circulating Tfh and B cells. Interaction between Tfh and B
cells triggers GC formation and B cell differentiation into
IgG4-producing cells through secretion of IL-21 in IgG4-ROD.
Steroids represent an effective short-term treatment
of IgG4-RD, their effect typically becomes evident within weeks;
however, £40% of patients relapse within the 1st year (49). Rituximab is a promising medication
but resistant cases have been reported (50). The aforementioned results of the
present study suggest that the use of T cell activation inhibitors
for Tfh cells, including abatacept, could represent an effective
treatment of IgG4-RD. Yamamoto et al (51) recently reported a
rituximab-resistant IgG4-RD patient who showed a good response to
treatment with abatacept. Since abatacept may affect Tfh cells in
the GCs (51), it may exert
greater effect on patients with IgG4-ROD. Understanding of the
pathgenetic relevance of IL-17 in rheumatoid arthritis and
collagen-induced arthritis led to the development of a
semi-specific immunotherapy based on cytokine antagonism for the
treatment of these disorders (52,53).
Identification of IL-21 as a potentially key cytokine in IgG4-ROD
may enable the use of specific IL-21 antagonists in the treatment
of IgG4-ROD.
However, the significance of the present study is
limited by the number of patients analyzed. As clinicians are
becoming increasingly aware of IgG4-RD and the administration of
glucocorticoid therapy is more prevalent, invasive medical
procedures are less prevalent, decreasing the availability of
samples from IgG4-RD patients, especially those with IgG4-ROD.
Larger cohort studies performed in the future may further elucidate
the underlying mechanism of Tfh cell action in affected
tissues.
In summary, in the present study
CD4+CXCR5+ Tfh cells were highly prevalent in
IgG4-RD, and the frequency and distribution of CD4+CXCR5+ Tfh cells
differed between patients with IgG4-ROD and type 1 AIP, which may
suggest different roles for Tfh cells in IgG4-ROD and type 1 AIP.
IL-21 was highly expressed in patients with IgG4-ROD and
Tfh-derived IL-21 can promote B cell to differentiate into
IgG4-secreting cells in IgG4-ROD, suggesting that Tfh cells may
have a direct role in the pathogenesis of IgG4-ROD. These results
make Tfh cells and IL-21 an important focus of potential
therapeutic methods to treat IgG4-ROD.
Acknowledgements
The present study was supported by the Science and
Technology Commission Foundation of Shanghai (grant no.
13ZR1424800).
References
|
1
|
Hamano H, Kawa S, Horiuchi A, Unno H,
Furuya N, Akamatsu T, Fukushima M, Nikaido T, Nakayama K, Usuda N
and Kiyosawa K: High serum IgG4 concentrations in patients with
sclerosing pancreatitis. N Engl J Med. 344:732–738. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Hamanou H, Kawa S, Ochi Y, Unno H, Shiba
N, Wajiki M, Nakazawa K, Shimojo H and Kiyosawa K: Hydronephrosis
associated with retroperitoneal fibrosis and sclerosing
pancreatitis. Lancet. 359:1403–1404. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Stone JH, Khosroshahi A, Deshpande V, Chan
JK, Heathcote JG, Aalberse R, Azumi A, Bloch DB, Brugge WR,
Carruthers MN, et al: Recommendations for the nomenclature of
IgG4-related disease and its individual organ system
manifestations. Arthritis Rheum. 64:3061–3067. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Umehara H, Okazaki K, Masaki Y, Kawano M,
Yamamoto M, Saeki T, Matsui S, Sumida T, Mimori T, Tanaka Y, et al:
A novel clinical entity, IgG4-related disease (IgG4RD): General
concept and details. Mod Rheumatol. 22:1–14. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Deshpande V, Zen Y, Chan JK, Yi EE, Sato
Y, Yoshino T, Klöppel G, Heathcote JG, Khosroshahi A, Ferry JA, et
al: Consensus statement on the pathology of IgG4-related disease.
Mod Pathol. 25:1181–1192. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stone JH, Zen Y and Deshpande V:
IgG4-Related Disease. N Engl J Med. 366:539–551. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Goto H, Takahira M and Azumi A; Japanese
Study Group for IgG4-Related Ophthalmic Disease, : Diagnostic
criteria for IgG4-related ophthalmic disease. Jpn J Ophthalmol.
59:1–7. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
MacLennan IC: Germinal centers. Annu Rev
Immunol. 12:117–139. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Klein U and Dalla-Favera R: Germinal
centres: Role in B-cell physiology and malignancy. Nat Rev Immunol.
8:22–33. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Crotty S: Follicular helper CD4 T cells
(TFH). Annu Rev Immunol. 29:621–663. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vinuesa CG, Linterman MA, Yu D and
MacLennan IC: Follicular Helper T Cells. Annu Rev Immunol.
34:335–368. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Schaerli P, Willimann K, Lang AB, Lipp M,
Loetscher P and Moser B: Cxc chemokine receptor 5 expression
defines follicular homing T cells with B cell helper function. J
Exp Med. 192:1553–1562. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Breitfeld D, Ohl L, Kremmer EK, Ellwart J,
Sallusto F, Lipp M and Förster R: Follicular B helper T Cells
express Cxc chemokine receptor 5, Localize to B cell follicles and
support immunoglobulin production. J Exp Med. 192:1545–1552. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ansel KM, McHeyzer-Williams LJ, Ngo VN,
McHeyzer-Williams MG and Cyster JG: In vivo activated CD4 T cells
upregulate CXC chemokine receptor 5 and reprogram their response to
lymphoid chemokines. J Exp Med. 190:1123–1134. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zlotnik A and Yoshie O: Chemokines: A new
classification system and their role in immunity. Immunity.
12:121–127. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chtanova T, Tangye SG, Newton R, Frank N,
Hodge MR, Rolph MS and Mackay CR: T Follicular helper cells express
a distinctive transcriptional profile, reflecting their role as
Non-Th1/Th2 effector cells that provide help for B Cells. J
Immunol. 173:68–78. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Johnston RJ, Poholek AC, DiToro D, Yusuf
I, Eto D, Barnett B, Dent AL, Craft J and Crotty S: Bcl6 and
Blimp-1 are reciprocal and antagonistic regulators of T follicular
helper cell differentiation. Science. 325:1006–1010. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nurieva RI, Chung Y, Martinez GJ, Yang XO,
Tanaka S, Matskevitch TD, Wang YH and Dong C: Bcl6 mediates the
development of T follicular helper cells. Science. 325:1001–1005.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu D, Rao S, Tsai LM, Lee SK, He Y,
Sutcliffe EL, Srivastava M, Linterman M, Zheng L, Simpson N, et al:
The transcriptional repressor Bcl-6 directs T follicular helper
cell lineage commitment. Immunity. 31:457–468. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
King C: New insights into the
differentiation and function of T follicular helper cells. Nat Rev
Immunol. 9:757–766. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Choi YS, Kageyama R, Eto D, Escobar TC,
Johnston RJ, Monticelli L, Lao C and Crotty S: ICOS receptor
instructs T follicular helper cell versus effector cell
differentiation via induction of the transcriptional repressor
Bcl6. Immunity. 34:932–946. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Weber JP, Fuhrmann F, Feist RK, Lahmann A,
Al Baz MS, Gentz LJ, Vu Van D, Mages HW, Haftmann C, Riedel R, et
al: ICOS maintains the T follicular helper cell phenotype by
down-regulating Kruppel-like factor 2. J Exp Med. 212:217–233.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zen Y, Fujii M, Harada K, Kawano M, Yamada
K, Takahira M and Nakanuma Y: Th2 and regulatory immune reactions
are increased in immunoglobin G4-related sclerosing pancreatitis
and cholangitis. Hepatology. 45:1538–1546. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tanaka A, Moriyama M, Nakashima H, Miyake
K, Hayashida JN, Maehara T, Shinozaki S, Kubo Y and Nakamura S: Th2
and regulatory immune reactions contribute to IgG4 production and
the initiation of Mikulicz disease. Arthritis Rheum. 64:254–263.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Komori T, Kondo S, Wakisaka N, Nakanishi
Y, Nakanishi-Yagi S, Tsuji A, Endo K, Murono S and Yoshizaki T:
IL-18 is highly expressed in inflammatory infiltrates of
submandibular glands in patients with immunoglobulin G4-related
disease. Hum Pathol. 46:1850–1858. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Maehara T, Mattoo H, Ohta M, Mahajan VS,
Moriyama M, Yamauchi M, Drijvers J, Nakamura S, Stone JH and Pillai
SS: Lesional CD4+ IFN-γ+ cytotoxic T lymphocytes in IgG4-related
dacryoadenitis and sialoadenitis. Ann Rheum Dis. 76:377–385. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Simpson N, Gatenby PA, Wilson A, Malik S,
Fulcher DA, Tangye SG, Manku H, Vyse TJ, Roncador G, Huttley GA, et
al: Expansion of circulating T cells resembling follicular helper T
cells is a fixed phenotype that identifies a subset of severe
systemic lupus erythematosus. Arthritis Rheum. 62:234–244. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Choi JY, Ho JH, Pasoto SG, Bunin V, Kim
ST, Carrasco S, Borba EF, Goncalves CR, Costa PR, Kallas EG, et al:
Circulating follicular helper-like T cells in systemic lupus
erythematosus: Association with disease activity. Arthritis
Rheumatol. 67:988–999. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhu C, Ma J, Liu Y, Tong J, Tian J, Chen
J, Tang X, Xu H, Lu L and Wang S: Increased frequency of follicular
helper T cells in patients with autoimmune thyroid disease. J Clin
Endocrinol Metab. 97:943–950. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang M, Zhou Y, Guo J, Li H, Tian F, Gong
L, Wang X, Lan M, Li Z and Zhang W: Thymic TFH cells involved in
the pathogenesis of myasthenia gravis with thymoma. Exp Neurol.
254:200–205. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang CJ, Gong Y, Zhu W, Qi Y, Yang CS, Fu
Y, Chang G, Li Y, Shi S, Wood K, et al: Augmentation of Circulating
Follicular Helper T Cells and Their Impact on Autoreactive B Cells
in Myasthenia Gravis. J Immunol. 197:2610–2617. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Akiyama M, Suzuki K, Yamaoka K, Yasuoka H,
Takeshita M, Kaneko Y, Kondo H, Kassai Y, Miyazaki T, Morita R, et
al: Number of circulating follicular helper 2 T cells correlates
with IgG4 and interleukin-4 levels and plasmablast numbers in
IgG4-related disease. Arthritis Rheumatol. 67:2476–2481. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Akiyama M, Yasuoka H, Yamaoka K, Suzuki K,
Kaneko Y, Kondo H, Kassai Y, Koga K, Miyazaki T, Morita R, et al:
Enhanced IgG4 production by follicular helper 2 T cells and the
involvement of follicular helper 1 T cells in the pathogenesis of
IgG4-related disease. Arthritis Res Ther. 18:1672016. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Umehara H, Okazaki K, Masaki Y, Kawano M,
Yamamoto M, Saeki T, Matsui S, Yoshino T, Nakamura S, Kawa S, et
al: Comprehensive diagnostic criteria for IgG4-related disease
(IgG4-RD), 2011. Mod Rheumatol. 22:21–30. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Friedrichs K, Gluba S, Eidtrnann H and
Jonat W: Overexpression of p53 and Prognosis in Breast Cancer.
Cancer. 72:3641–3647. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhu J, Yamane H and Paul WE:
Differentiation of effector CD4 T cell populations (*). Annu Rev
Immunol. 28:445–489. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Sato Y, Ohshima K, Ichimura K, Sato M,
Yamadori I, Tanaka T, Takata K, Morito T, Kondo E and Yoshino T:
Ocular adnexal IgG4-related disease has uniform clinicopathology.
Pathol Int. 58:465–470. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Poholek AC, Hansen K, Hernandez SG, Eto D,
Chandele A, Weinstein JS, Dong X, Odegard JM, Kaech SM, Dent AL, et
al: In vivo regulation of Bcl6 and T follicular helper cell
development. J Immunol. 185:313–326. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hatzi K, Nance JP, Kroenke MA, Bothwell M,
Haddad EK, Melnick A and Crotty S: BCL6 orchestrates Tfh cell
differentiation via multiple distinct mechanisms. J Exp Med.
212:539–553. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Fazilleau N, Mark L, McHeyzer-Williams LJ
and McHeyzer-Williams MG: Follicular helper T cells: Lineage and
location. Immunity. 30:324–335. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
King C, Tangye SG and Mackay CR: T
follicular helper (TFH) cells in normal and dysregulated immune
responses. Annu Rev Immunol. 26:741–766. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zen Y: The Pathology of IgG4-Related
Disease in the Bile Duct and Pancreas. Semin Liver Dis. 36:242–256.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Spolski R and Leonard WJ: IL-21 and T
follicular helper cells. Int Immunol. 22:7–12. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Good KL, Bryant VL and Tangye SG: Kinetics
of Human B cell behavior and amplification of proliferative
responses following stimulation with IL-21. J Immunol.
177:5236–5247. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bryant VL, Ma CS, Avery DT, Li Y, Good KL,
Corcoran LM, de Waal Malefyt R and Tangye SG: Cytokine-mediated
regulation of human B cell differentiation into ig-secreting cells:
Predominant Role of IL-21 produced by CXCR5+ T follicular helper
cells. J Immunol. 179:8180–8190. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Zotos D, Coquet JM, Zhang Y, Light A,
D'Costa K, Kallies A, Corcoran LM, Godfrey DI, Toellner KM, Smyth
MJ, et al: IL-21 regulates germinal center B cell differentiation
and proliferation through a B cell-intrinsic mechanism. J Exp Med.
207:365–378. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Linterman MA, Beaton L, Yu D, Ramiscal RR,
Srivastava M, Hogan JJ, Verma NK, Smyth MJ, Rigby RJ and Vinuesa
CG: IL-21 acts directly on B cells to regulate Bcl-6 expression and
germinal center responses. J Exp Med. 207:353–363. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Maehara T, Moriyama M, Nakashima H, Miyake
K, Hayashida JN, Tanaka A, Shinozaki S, Kubo Y and Nakamura S:
Interleukin-21 contributes to germinal centre formation and
immunoglobulin G4 production in IgG4-related dacryoadenitis and
sialoadenitis, so-called Mikulicz's disease. Ann Rheum Dis.
71:2011–2019. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Martinez-Valle F, Fernandez-Codina A,
Pinal-Fernandez I, Orozco-Galvez O and Vilardell-Tarres M:
IgG4-related disease: Evidence from six recent cohorts. Autoimmun
Rev. 16:168–172. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yamamoto M, Awakawa T and Takahashi H: Is
rituximab effective for IgG4-related disease in the long term?
Experience of cases treated with rituximab for 4 years. Ann Rheum
Dis. 74:e462015. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yamamoto M, Takahashi H, Takano K, Shimizu
Y, Sakurai N, Suzuki C, Naishiro Y, Yajima H, Awakawa T, Himi T and
Nakase H: Efficacy of abatacept for IgG4-related disease over 8
months. Ann Rheum Dis. 75:1576–1578. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Bai F, Tian H, Niu Z, Liu M, Ren G, Yu Y,
Sun T, Li S and Li D: Chimeric anti-IL-17 full-length monoclonal
antibody is a novel potential candidate for the treatment of
rheumatoid arthritis. Int J Mol Med. 33:711–721. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Yoshiga Y, Goto D, Segawa S, Ohnishi Y,
Matsumoto I, Ito S, Tsutsumi A, Taniguchi M and Sumida T: Invariant
NKT cells produce IL-17 through IL-23-dependent and -independent
pathways with potential modulation of Th17 response in
collagen-induced arthritis. Int J Mol Med. 22:369–374.
2008.PubMed/NCBI
|