Introduction
Stem cell transplantation has been extensively
investigated as a therapy for heart failure (1,2).
Mesenchymal stem cells (MSCs), which have the ability to escape
detection by the host immune system upon transplantation and to
expand easily, become a promising source of cells for
transplantation (3).
Numerous studies have demonstrated that the
transplantation of MSCs can promote cardiac repair in acute
myocardial injury and cardiomyopathy (3–6). The
mechanisms mediating the beneficial effect of MSC transplantation
have been widely investigated. In a previous study of the authors,
the results demonstrated that the antifibrotic effect of MSCs
contributes to the improvement of heart function (6,7).
Despite progress, the transplantation of MSCs is insufficient to
fully restore heart function. Therefore, an important issue to be
solved is how to maximize the therapeutic effect of MSCs
transplantation. It has been proven that gene modification can
increase the efficacy of cell transplantation (8,9).
Adrenomedullin (ADM) is a cytokine that may modulate cardiac
fibroblast growth (10–12). ADM infusion during
ischemia/reperfusion has been proven to attenuate cardiac fibrosis
in rats (13).
Therefore, in the present study, the aim was to
examine whether ADM gene transfer can increase the therapeutic
effects of MSC transplantation in isoproterenol-induced global
heart failure in rats by attenuating cardiac fibrosis and to
investigate the mechanisms that mediate this effect.
Materials and methods
Culture and adenoviral transduction of
MSCs
MSCs were obtained and passaged according to
previously described methods (6,7). At
the third passage, the MSCs were transduced with an adenoviral
vector encoding the gene green fluorescent protein (Ad-CMV-GFP). To
select the best multiplicity of infection (MOI) for the
adenovirus-mediated gene transfer, MSCs were exposed to Ad-CMV-GFP
at MOIs of 1:50, 1:100, 1:200 and 1:400 for 8 h. The medium was
then replaced by normal Dulbecco's modified Eagle's medium
(DMEM)/F12 containing 10% fetal bovine serum and antibiotics. At 48
h following transfection, cell viability was determined through
observation of cellular morphology by fluorescence microscopy.
Based on these observations, 1:200 was chosen as the optimal MOI
because of its high efficiency and low toxicity. Transduction
efficacy was further assessed by fluorescence-activated cell
sorting (FACS) analysis. For FACS analysis, the transfected MSCs
were digested and centrifuged at 1,000 × g for 5 min. Following
removal of the supernatant, 500 µl PBS was added and the MSCs were
assayed by flow cytometry and analyzed using FACSDiva Pro software
(BD Biosciences, Franklin Lakes, NJ, USA). In addition, ADM
expression in the GFP-MSCs and ADM-MSCs was detected by western
blotting.
Experimental model and group
A total of 55 male Wistar rats (180–200 g) were
obtained from Harbin Medical University Laboratorial Animal Center
(Harbin, China). All experimental procedures were approved by the
Care of Experimental Animals Committee of Harbin Medical University
(Harbin, China). Isoproterenol (ISO)-induced heart failure was
induced as described previously (7). Briefly, ISO (170 mg/kg in 0.5 ml
saline; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was
subcutaneously injected into the rats every day for 4 consecutive
days. Sham-injected rats were injected subcutaneously with 0.5 ml
saline alone. At 4 weeks following the final injection, these rats
received transplants. Before transplantation, the animals were
randomized into four groups: The sham group and three ISO-induced
heart failure groups. The three heart failure groups each received
a different treatment: DMEM/F12 (medium-treated group), GFP-MSCs
(GFP-MSCs-treated group) or ADM-MSCs transplantation
(ADM-MSCs-treated group).
Cell transplantation
At 4 weeks following the final ISO or saline
injection, all of the rats received transplantation. In the
GFP-MSCs or ADM-MSCs-treated groups, a total of 5×106
GFP-MSCs or ADM-MSCs were suspended in 100 µl medium and injected
into the left anterior myocardium through a 30-G needle at 4
different sites. Medium (100 µl) was also injected into animals of
the sham group and the medium-treated group. At 4 weeks following
transplantation, the rats were sacrificed for further studies.
Echocardiography
Cardiac function was measured 4 weeks following
transplantation with an echocardiographic system equipped with a
12-MHz transducer (HP 5500; Hewlett-Packard, Palo Alto, CA, USA).
LV end-diastolic diameter (LVEDD), LV end-systolic diameter, LV
ejection fraction (LVEF) and fractional shortening (FS) were
measured.
Histological analysis
To detect the cell survival rates following MSC
implantation, prior to transplantation, transfected MSCs were also
fluorescently labeled with DAPI (Beyotime Institute of
Biotechnology, Haimen, China), according to the manufacturer's
protocol. At 4 weeks following transplantation, the hearts (n=3
each from the GFP-MSCs-treated group and ADM-MSCs-treated group)
were dissected and embedded in optimum cutting temperature
compound, snap-frozen in liquid nitrogen (−196°C), and cut into
6-µm sections. Transplanted cells that expressed green and blue
fluorescence were observed by fluorescence microscopy.
To detect fibrosis in cardiac muscle, the hearts
(n=6 from each group) were fixed in 10% formalin, cut transversely,
embedded in paraffin, and stained with Masson's trichrome staining.
The size of fibrotic areas was quantified as described (6). Transverse sections were randomly
obtained from two levels, and 10 randomly selected fields per
section were analyzed. After each field was scanned with a digital
image analyzer, the area of the collagenous fraction was calculated
as the sum of all areas containing connective tissue divided by the
total area of the image.
Reverse transcription
quantitative-polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the cardiac muscle
tissue using TRIzol reagent (Invitrogen; Thermo Fisher Scientific,
Inc., Waltham, MA, USA), following the manufacturer's protocol.
cDNA was generated using avian myeloblastosis virus reverse
transcriptase (Promega Corporation, Madison, WI, USA) and oligo
(dT) primers according to the manufacturer's instructions. Primer
sequences and reaction conditions are listed in Table I. Real-time PCR was performed in an
Applied Biosystems 7300 Fast Real-Time PCR System (Applied
Biosystems; Thermo Fisher Scientific, Inc.) with SYBR Green PCR
Master Mix (Bioneer Corporation, Daejeon, Korea). Primer sequences
used and thermocycling conditions are presented in Table I. β-actin mRNA amplified from the
same samples served as an internal control. The relative β-actin
expression of each targeted gene was normalized by subtracting the
corresponding threshold cycle (Ct) values using the
2−ΔΔCq comparative method (14).
 | Table I.Primer sequences used. |
Table I.
Primer sequences used.
| Primer | Direction | Sequence
(5′-3′) | Tm
(°C) |
|---|
| MMP-2 | Forward |
aaggatcggtttatttggcg | 55 |
|
| Reverse |
cattccctgcgaagaacaca |
|
| MMP-9 | Forward |
aagttctcgaatcacggaggaa | 56 |
|
| Reverse |
cattttcgcccagagaaga |
|
| HGF | Forward |
ttgtgagggagattatggtggc | 57 |
|
| Reverse |
caggacgatttgggatggcatag |
|
| TGF-β1 | Forward |
caacaattcctggcgtta | 56 |
|
| Reverse |
aagccctgtattccgtctcc |
|
| Collagen-1 | Forward |
cagattgggatggagggagttta | 56 |
|
| Reverse |
ctacagcacgcttgtggatggct |
|
| Collagen-III | Forward |
atagctgaactgaaagccaccat | 55 |
|
| Reverse |
cctgaactcaagagcggaata |
|
| β-actin | Forward |
cccatctatgagggttacgc | 56 |
|
| Reverse |
tttaatgtcacgcacgatttc |
|
Western blotting
To identify the protein expression of ADM, matrix
metalloproteinase (MMP)-2 and MMP-9, western blotting was
performed. Cardiac tissues were lysed using
radioimmunoprecipitation assay lysis buffer (Beyotime Institute of
Biotechnology). A bicinchoninic acid protein assay kit (Beyotime
Institute of Biotechnology) was used to determine protein
concentrations. Protein extracts (20 µg) were subjected to
electrophoresis through an 8–15% SDS-PAGE gel and were transferred
to a polyvinylidene difluoride membrane. Following blocking with 5%
skimmed milk at room temperature for 1 h, the membrane was
incubated with primary antibody at 37°C for 2 h: Anti-ADM (cat. no.
BA0629; 1:200; Wuhan Boster Biological Technology, Ltd., Wuhan,
China); anti-MMP-2 (cat. no. sc-13594; 1:200; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA); anti-MMP-9 (cat. no. BA2202;
1:200; Wuhan Boster Biological Technology, Ltd.); and anti-β-actin
(cat. no. BM3873; 1:200; Wuhan Boster Biological Technology, Ltd.).
The membrane was subsequently incubated at 37°C for an additional
hour in an alkaline phosphatase-conjugated secondary antibody (cat.
no. BM2007; 1:1,000; Wuhan Boster Biological Technology, Ltd.).
Immunoreactive bands were visualized by enhanced chemiluminescence
(Pierce; Thermo Fisher Scientific, Inc.) and exposure to x-ray
film. The x-ray films were photographed using an Alpha Innotech gel
analysis system (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Relative protein expression was quantified using Quantity One
software (version 4.62; Bio-Rad Laboratories, Inc.).
Statistical analysis
All values are expressed as mean values ± standard
error of the mean. Comparisons of parameters among groups were
analyzed using the one-way analysis of variance followed by Tukey's
post hoc test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Transduction of MSCs
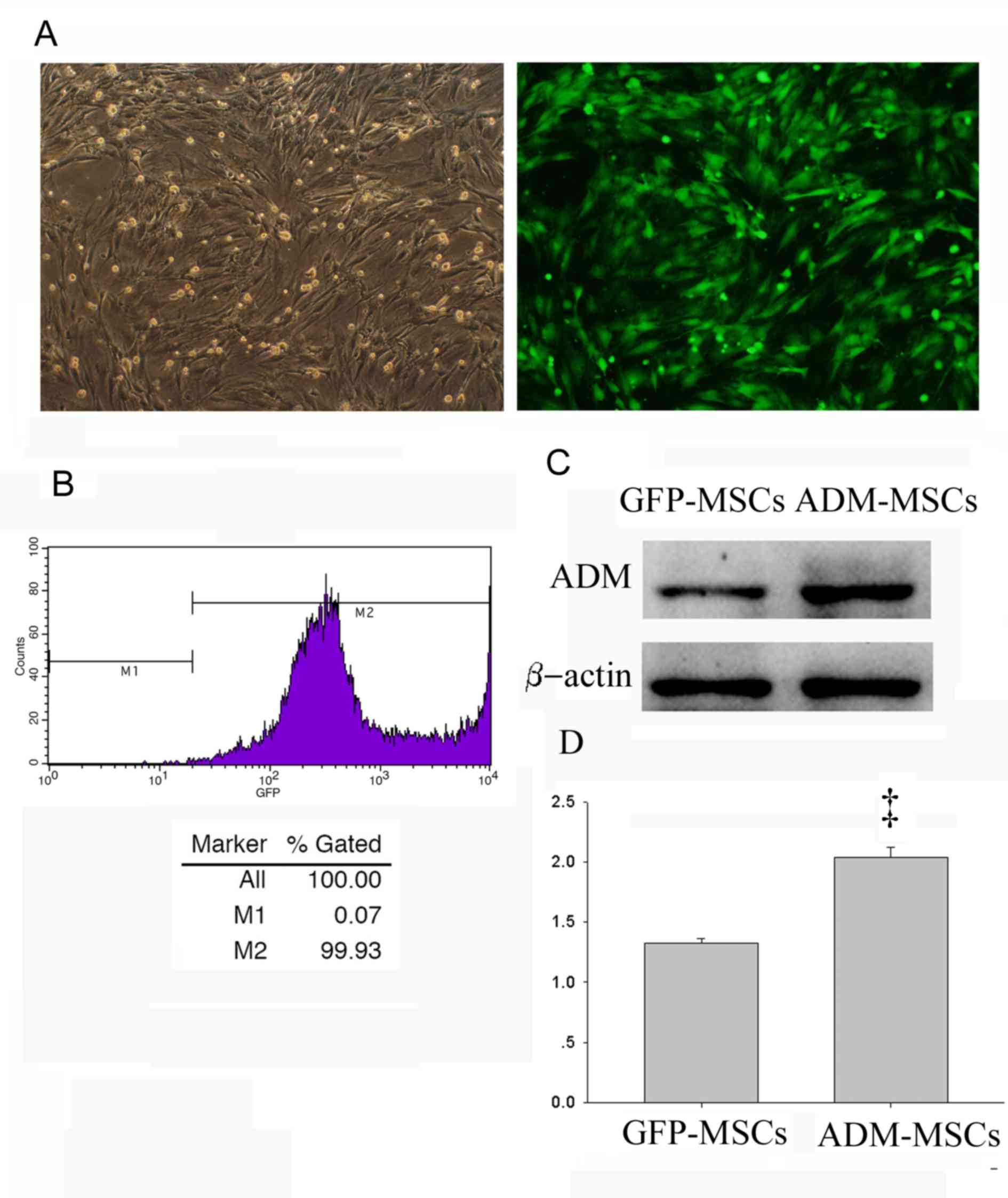
To choose the optimal MOI, MSCs were transduced with
the adenoviral vector at various MOIs. The results of observation
by fluorescence microscopy indicated that most of the MSCs became
round in shape and detached from the wall in 2 days at an MOI of
50, and that the percentage of GFP-positive cells was lower at an
MOI of 400. Therefore, adenoviral vectors were used to infect MSCs
at an MOI of 200 in the subsequent experiments. As presented in
Fig. 1A, most of these transfected
cells were alive observed by light microscopy, and these cells
presented green fluorescence observed by fluorescence microscopy.
Furthermore, flow cytometric analysis also indicated that the
transduction efficiency was >90% (Fig. 1B). These results were confirmed by
western blotting, which indicated that the expression of ADM
protein was significantly higher in the ADM-MSCs than the GFP-MSCs
(Fig. 1C and D).
Effect of cell transplantation on
heart function
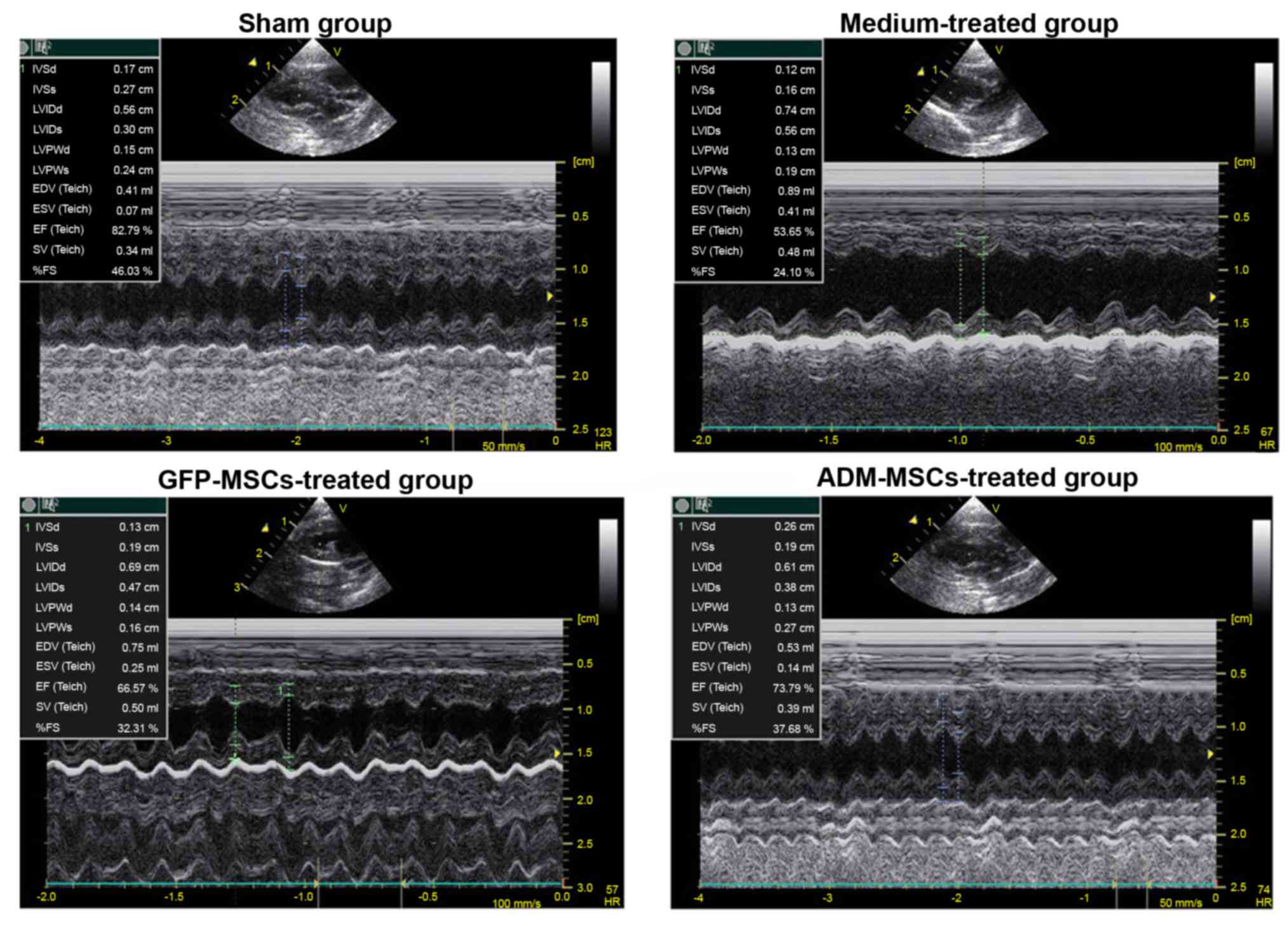
At 4 weeks following MSC transplantation, cardiac
function of the living rats was measured by echocardiography.
Results indicated that, compared with the sham group, the
medium-treated group exhibited a significant decrease in LVEF and
FS and an increase in LVEDD. These results demonstrated that the
ISO injection could impair heart function in rats. When compared
with the medium-treated group, cardiac function was significantly
improved in both of the cell-treated groups. More importantly, LVEF
and FS were noticeably higher in the ADM-MSCs-treated group than in
the GFP-MSCs-treated group. Therefore, the authors can conclude
that, compared with GFP-MSCs, ADM-MSC transplantation can further
improve heart function (Fig. 2 and
Table II).
 | Table II.Echocardiographic assessment of
cardiac function. |
Table II.
Echocardiographic assessment of
cardiac function.
| Group | n | LVDd (mm) | LVDs (mm) | EF (%) | FS (%) |
|---|
| Sham | 9 |
6.39±0.44 |
3.16±0.22 |
86.503±1.54 |
50.66±1.87 |
| Medium-treated | 8 |
7.25±1.69 |
5.35±1.57a |
56.67±6.86b |
26.27±4.01b |
|
GFP-MSCs-treated | 9 |
6.671±0.70 |
4.24±0.92c |
69.235±7.30b,c |
34.59±5.13b,c |
|
ADM-MSCs-treated | 9 |
6.696±0.81 |
3.95±0.55c |
79.403±1.70b–d |
43.48±2.15b–d |
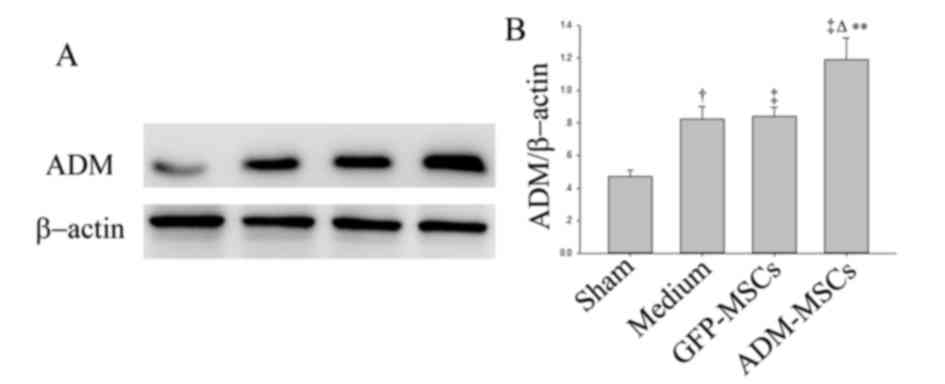
Expression of ADM
At 4 weeks following MSC transplantation, the
expression of ADM was examined by western blot analysis. Consistent
with a previous study of the authors (7), the current study demonstrated that
MSC transplantation could increase the expression of ADM in the
myocardium. Furthermore, the present study also showed that
ADM-MSCs transplantation could further increase the expression of
ADM compared with GFP-MSCs transplantation (Fig. 3A and B).
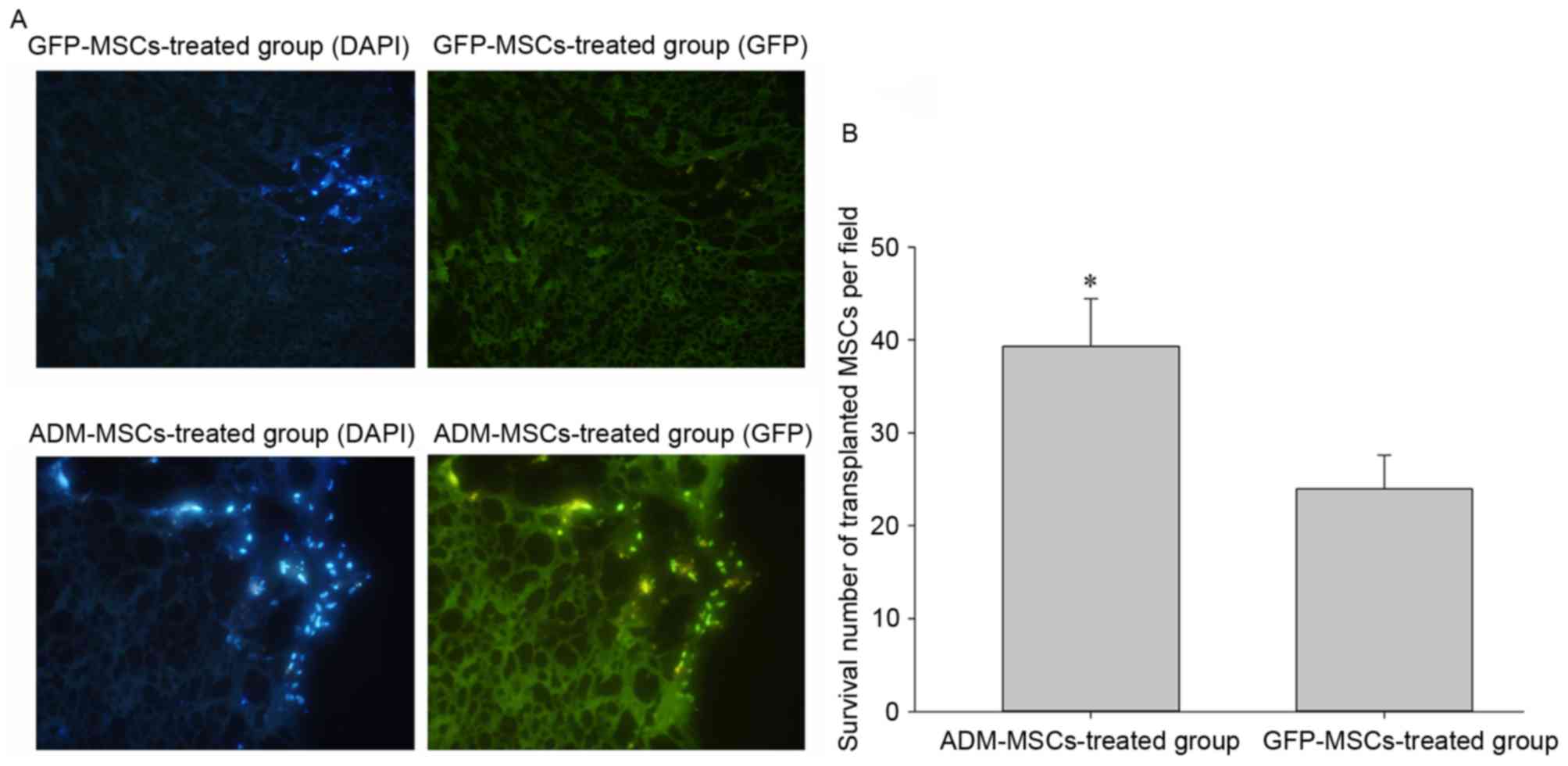
Survival of transplanted cells
To observe the survival of MSCs in vivo, the
nuclei of the transfected MSCs were also labeled with DAPI. As
demonstrated in Fig. 4A and B, the
survival of MSCs is higher in the ADM-MSCs-treated group than that
in the GFP-MSCs-treated group.
Effect of cell transplantation on
cardiac fibrosis
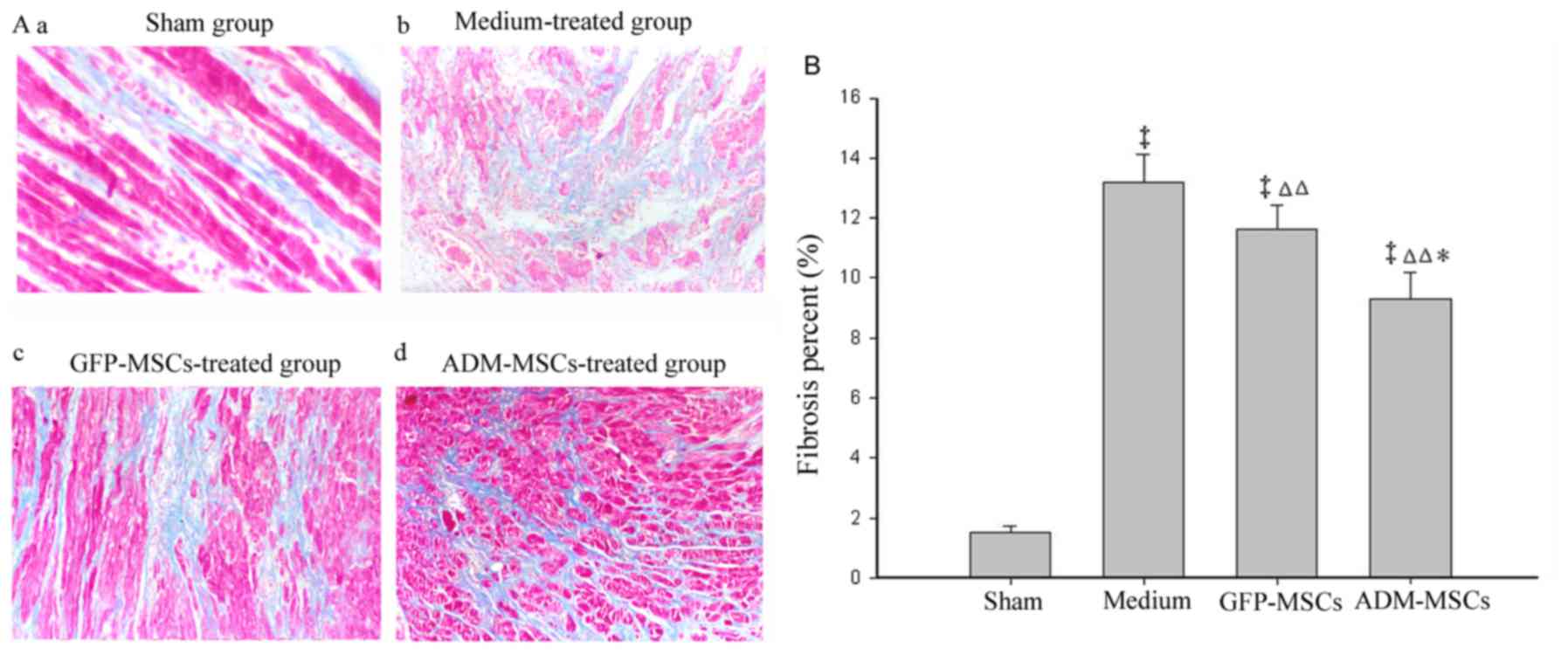
The extent of myocardial fibrosis was assessed by
Masson's trichrome staining. Fig.
5A presents representative photographs of Masson's trichrome
staining of heart sections 4 weeks following transplantation.
Quantitative analysis of the fibrotic area was also performed. As
identified in Fig. 5B, the
fibrotic area was significantly greater in the medium-treated group
than the GFP-MSCs-treated group and the ADM-MSCs-treated group.
Furthermore, the fibrotic area of the ADM-MSCs-treated group was
significantly smaller than that of the GFP-MSCs-treated group.
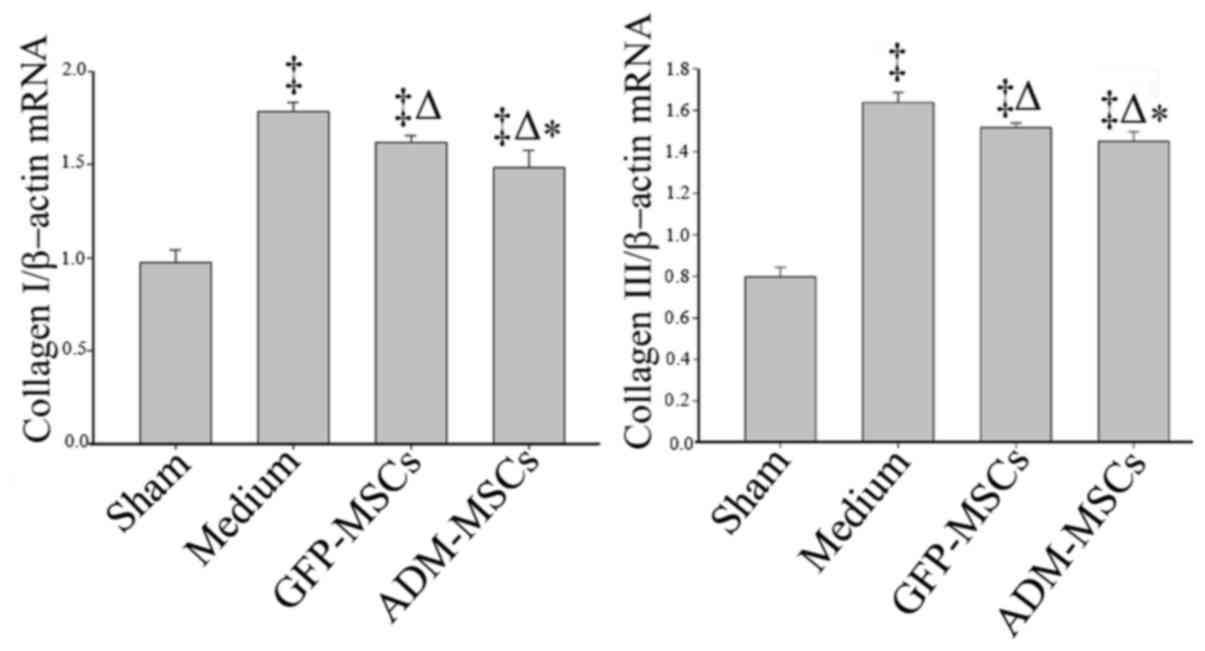
Next, the expression of collagen I and III were
investigated by RT-qPCR. Similar to the authors' previous study
(6), the present study indicated
that, compared with the sham group, the expressions of collagen I
and III were significantly higher in the medium-treated group. In
addition, cell transplantation significantly decreased the
expression of collagen I and III. Importantly, the expressions of
collagen I and III were further decreased in the ADM-MSCs-treated
group compared with the GFP-MSCs-treated group (Fig. 6).
Effect of cell transplantation on
expression of MMPs
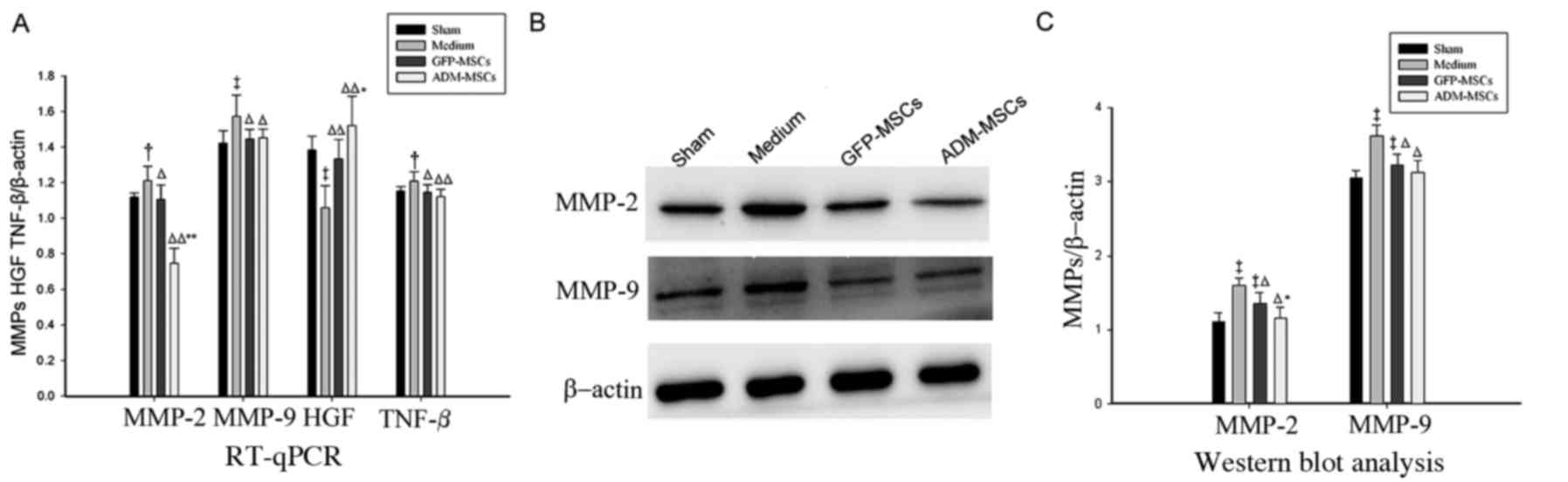
The authors investigated the expression of MMP-2 and
MMP-9 by RT-qPCR and western blotting. The results demonstrated
that, compared with the medium-treated group, the gene expression
of MMP-2 and MMP-9 was significantly lower in both of the cell
treated groups. Importantly, compared with the GFP-MSCs-treated
group, the ADM-MSCs-treated group exhibited even lower expression
of MMP-2 (Fig. 7A). In addition,
the groups treated with both cell types had significantly lower
MMP-2 and MMP-9 protein levels compared with the medium-treated
group. The protein level of MMP-2 was also lower in the
ADM-MSCs-treated group than the GFP-MSCs-treated group (Fig. 7B and C). Although the gene and
protein expression of MMP-9 were lower in the ADM-MSCs-treated
group than that in the GFP-MSCs-treated group, the difference
between these two groups was not statistically significant.
Paracrine signaling by cell
transplantation
The expression of the hepatocyte growth factor (HGF)
and transforming growth factor (TGF)-β genes, which mediate cardiac
remodeling, were then investigated. As presented in Fig. 7A, compared with the expression in
the medium-treated group, the gene expression of HGF was
significantly lower in both the GFP-MSCs-treated group and the
ADM-MSCs-treated group. Importantly, compared with the
GFP-MSCs-treated group, the ADM-MSCs-treated group exhibited lower
levels of HGF gene expression. In addition, it was also indicated
that, compared with the GFP-MSCs-treated group, the
ADM-MSCs-treated group had not lower levels of TGF-β gene
expression (Fig. 7A).
Discussion
In the present article, the authors demonstrated
that, compared to the GFP-MSCs transplantation, ADM-MSC
transplantation greatly improved cardiac function and decreased the
cardiac fibrosis associated with ISO-induced heart failure. In
addition, the beneficial effects of ADM-MSC transplantation may be
partially mediated by significantly influencing the expression of
fibrosis-related cytokines, such as ADM and HGF.
Studies have reported that subcutaneous
administration of ISO in rats could produce myocardial cell death
and cardiac fibrosis and lead to heart failure, while maintaining
an intact coronary vasculature (15,16).
The effectiveness of reperfusion therapy and the potential
confounding effects of an abnormal blood supply underscore the
importance of studying cardiac fibrosis arising in the presence of
an intact vascular system (16).
For these reasons, the ISO-treated rat heart is an excellent model
system for investigations into cardiac fibrosis and heart failure.
Previous studies, including the authors' published study, have
proven that ISO may induce heart failure and cardiac fibrosis 4
weeks subsequent to injection (6,16,17).
Additionally, it is well-known that cardiac fibrosis may have
already formed in patients with previous myocardial infarction and
dilated cardiomyopathy (18).
Therefore, in order to investigate whether cell transplantation can
inhibit the progress of cardiac fibrosis of these patients, the
authors chose the interval of 4 weeks following ISO injection to
initiate MSC transplantation.
Paracrine mechanisms have been proven to serve an
important role in the transplantation of MSCs. The authors'
previous study had also indicated that MSCs could secrete ADM and
that the level of ADM in the myocardium significantly increased
after the transplantation of MSCs, coinciding with the decrease in
cardiac fibrosis (6). Therefore,
it was concluded that ADM secreted by the MSCs may play an
important role in MSC transplantation. ADM has been reported to
inhibit myocardial remodeling by attenuating cardiomyocyte
hypertrophy, the proliferation of myocardial fibroblasts and the
production of extracellular matrix (10–13).
Thus, in the current study, the authors sought to investigate
whether, compared with MSCs alone, ADM gene-modified MSCs can
further decrease cardiac fibrosis and increase heart function.
In the present study, GFP-MSCs, ADM-MSCs or medium
were grafted into the myocardium in a rat model of ISO-induced
heart failure. At 4 weeks following transplantation, heart function
was examined, and the results showed that, compared with GFP-MSCs
and medium, ADM-MSCs could significantly improve heart function, as
indicated by an increase in LVEF. Therefore, it may be concluded
that modifying MSCs with ADM can increase the beneficial effects of
MSC transplantation.
Cardiac fibrosis serves an important role in the
pathogenesis of heart failure. Many previous studies have shown
that inhibiting the development of cardiac fibrosis could lead to
the improvement of heart function (19,20).
A previous study reported that MSCs transplantation could improve
heart function partly by inhibiting myocardial fibrosis (6). Furthermore, ADM have been proven to
play a role as an autocrine/paracrine modulator in the process of
cardiac remodeling by suppressing mitogenesis and collagen
synthesis in fibroblasts (11).
Therefore, the effect of ADM modified MSCs on cardiac fibrosis were
investigated in the study. Histological and molecular analysis
demonstrated that transplantation of ADM-MSCs could result in the
greatest decrease in cardiac fibrosis, compared with medium
transplantation and GFP-MSC transplantation.
The MMPs are a family of >25 species of
zinc-dependent proteases, which have been shown to be important in
ventricular remodeling by degrading the extracellular matrix. In
the failing heart, the normal collagens are degraded by increased
levels of MMPs, and fibrous interstitial deposits of poorly
cross-linked collagens are synthesized. This, in turn, may lead to
myocardial fibrosis, dilation of the ventricles and cardiac
dysfunction (21,22). Inhibition of the activities of the
MMPs prevents progressive left ventricular remodeling and improves
heart function in an animal model of heart failure (23,24).
Regardless of the inciting cause, there appears to be increased
expression of MMPs in the initial phase and the final phase of
heart failure, leading to marked ventricular dilation and fibrosis
(25,26). The authors' previous results have
indicated that the expression of MMP-2 and MMP-9 was significantly
increased in ISO-induced heart failure (6). In addition, ADM has been proven to
have an ability to inhibit the expression of MMP-2 and MMP-1 in
hepatic stellate cells and rheumatoid synovial fibroblasts
(27,28). Therefore, the effect of ADM-MSC
transplantation on the expression of MMP-2 and MMP-9 was
investigated in the current study. Importantly, the results also
showed that, compared with the GFP-MSCs-treated group, the
ADM-MSCs-treated groups exhibited a significant decrease in MMP-2
levels. Therefore, the authors propose that compared with MSCs, ADM
gene-modified MSCs can decrease the expression of MMP-2
significantly, resulting in a greater decrease in the degradation
of normal collagen and greater improvement in heart function.
Next, the effect of ADM on the paracrine ability of
MSCs was explored. The expression of ADM in the myocardium was
investigated. Results demonstrated that the level of ADM was
significantly increased in the ADM-MSCs-treated group than that of
the GFP-MSCs-treated group, which is coinciding with the decrease
of myocardial fibrosis. Therefore, it may be concluded that
ADM-MSCs can inhibit myocardial fibrosis by secreting ADM.
Furthermore, HGF (29) and TGF-β
(30), which is related to
myocardial fibrosis, were investigated. The authors' previous study
had indicated that the transplantation of MSCs increased the
expression of HGF in the myocardium (6). In the present study, results
demonstrated that the ADM-MSCs-treated group exhibited a
significant increase in HGF, when compared with the
GFP-MSCs-treated and medium-treated groups. The expression of TGF-β
was also observed. However, these results presented no significant
difference between the ADM-MSCs-treated group and the
GFP-MSCs-treated group. These observations suggested that ADM-MSCs
may decrease myocardial fibrosis not only by secreting ADM, but by
changing the expression of fibrosis-related cytokines, such as HGF.
Further studies will be required to detect the mechanism by which
the ADM-MSCs influence the expression of these fibrosis-related
cytokines. In addition, the current study demonstrated that ADM
modification could increase the survival of transplanted cells.
In conclusion, the transplantation of ADM-MSCs can
greatly improve cardiac function by attenuating myocardial
fibrosis. The beneficial effect of ADM-MSC transplantation may be
partially mediated through changes in the expression of
fibrosis-related genes, such as ADM and HGF.
Acknowledgements
The authors would like to thank Dr Wei Liu for her
expert assistance with cell culture and western blot analysis. Dr
Wei Liu is a member of the Key Laboratory of Myocardial Ischemia
Mechanism and Treatment, Harbin Medical University (Harbin, China).
The present study was supported by grants from the Natural Science
Foundation of Heilongjiang Province (grant no. QC2013C108).
References
|
1
|
Sun R, Li X, Liu M, Zeng Y, Chen S and
Zhang P: Advances in stem cell therapy for cardiovascular disease
(Review). Int J Mol Med. 38:23–29. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhang C, Nong Y, Tong S, Yao Q, Wen L,
Zhang Z, Wei L, Cheng J, Feng Y and Song Z: Triptolide improves
early survival of mesenchymal stem cells transplantation into rat
myocardium. Cardiology. 128:73–85. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Choi SH, Jung SY, Kwon SM and Baek SH:
Perspectives on stem cell therapy for cardiac regeneration:
Advances and challenges. Circ J. 76:1307–1312. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chin SP, Poey AC, Wong CY, Chang SK, Tan
CS, Ng MT, Chew KH, Lam KH and Cheong SK: Intramyocardial and
intracoronary autologous bone marrow-derived mesenchymal stromal
cell treatment in chronic severe dilated cardiomyopathy.
Cytotherapy. 13:814–821. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Suresh SC, Selvaraju V, Thirunavukkarasu
M, Goldman JW, Husain A, Palesty Alexander J, Sanchez JA, McFadden
DW and Maulik N: Thioredoxin-1 (Trx1) engineered mesenchymal stem
cell therapy increased pro-angiogenic factors, reduced fibrosis and
improved heart function in the infracted rat myocardium. Int J
Cardiol. 201:517–528. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Li L, Zhang Y, Li Y, Yu B, Xu Y, Zhao S
and Guan Z: Mesenchymal stem cell transplantation attenuates
cardiac fibrosis associated with isoproterenol-induced global heart
failure. Transpl Int. 21:1181–1189. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Li L, Zhang S, Zhang Y, Yu B, Xu Y and
Guan Z: Paracrine action mediate the antifibrotic effect of
transplanted mesenchymal stem cells in a rat model of global heart
failure. Mol Biol Rep. 36:725–731. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xu B, Luo Y, Liu Y, Li BY and Wang Y:
Platelet-derived growth factor-BB enhances MSC-mediated
cardioprotection via suppression of miR-320 expression. Am J
Physiol Heart Circ Physiol. 308:H980–H989. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wen Z, Huang W, Feng Y, Cai W, Wang Y,
Wang X, Liang J, Wani M, Chen J, Zhu P, et al: MicroRNA-377
regulates mesenchymal stem cell-induced angiogenesis in ischemic
hearts by targeting VEGF. PLoS One. 9:e1046662014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tsuruda T, Kato J, Kitamura K, Kawamoto M,
Kuwasako K, Imamura T, Koiwaya Y, Tsuji T, Kangawa K and Eto T: An
autocrine or a paracrine role of adrenomedullin in modulating
cardiac fibroblast growth. Cardiovasc Res. 43:958–967. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Horio T, Nishikimi T, Yoshihara F, Matsuo
H, Takishita S and Kangawa K: Effects of adrenomedullin on cultured
rat cardiac myocytes and fibroblasts. Eur J Pharmacol. 382:1–9.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nishikimi T, Yoshihara F, Mori Y, Kangawa
K and Matsuoka H: Cardioprotective effect of adrenomedullin in
heart failure. Hypertens Res. 26 Suppl:S121–S127. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Okumura H, Nagaya N and Kangawa K:
Adrenomedullin infusion during ischemia/reperfusion attenuates left
ventricular remodeling and myocardial fibrosis in rats. Hypertens
Res. 26 Suppl:S99–S104. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Okuda N, Hayashi T, Mori T, Inamoto S,
Okabe M, Mieno S, Horimoto H and Kitaura Y: Nifedipine enhances the
cardioprotective effect of an angiotensin-II receptor blocker in an
experimental animal model of heart failure. Hypertens Res.
28:431–438. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Teerlink JR, Pfeffer JM and Pfeffer MA:
Progressive ventricular remodeling in response to diffuse
isoproterenol-induced myocardial necrosis in rats. Circ Res.
75:105–113. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Brouri F, Hanoun N, Mediani O, Saurini F,
Hamon M, Vanhoutte PM and Lechat P: Blockade of beta1- and
desensitization of beta 2-adrenoceptors reduce isoprenaline-induced
cardiac fibrosis. Eur J Pharmacol. 485:227–234. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Maisch B: Ventricular remodeling.
Cardiology. 87 Suppl 1:S2–S10. 1996. View Article : Google Scholar
|
|
19
|
Eckhouse SR and Spinale FG: Changes in the
myocardial interstitium and contribution to the progression of
heart failure. Heart Fail Clin. 8:7–20. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Deschamps AM and Spinale FG: Matrix
modulation and heart failure: New concepts question old beliefs.
Curr Opin Cardiol. 20:211–216. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Spinale FG: Myocardial matrix remodeling
and the matrix metalloproteinases: Influence on cardiac form and
function. Physiol Rev. 87:1285–1342. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Vanhoutte D, Schellings M, Pinto Y and
Heymans S: Relevance of matrix metalloproteinases and their
inhibitors after myocardial infarction: A temporal and spatial
window. Cardiovasc Res. 69:604–613. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song YH, Cai H, Gu N, Qian CF, Cao SP and
Zhao ZM: Icariin attenuates cardiac remodelling through
down-regulating myocardial apoptosis and matrix metalloproteinase
activity in rats with congestive heart failure. J Pharm Pharmacol.
63:541–549. 2001. View Article : Google Scholar
|
|
24
|
Creemers EE, Cleutjens JP, Smits JF and
Daemen MJ: Matrix metalloproteinase inhibition after myocardial
infarction: A new approach to prevent heart failure? Circ Res.
89:201–210. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Li YY, McTiernan CF and Feldman AM:
Interplay of matrix metalloproteinases, tissue inhibitors of
metalloproteinases and their regulators in cardiac matrix
remodeling. Cardiovasc Res. 46:214–224. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Boixel C, Fontaine V, Rücker-Martin C,
Milliez P, Louedec L, Michel JB, Jacob MP and Hatem SN: Fibrosis of
the left atria during progression of heart failure is associated
with increased matrix metalloproteinases in the rat. J Am Coll
Cardiol. 42:336–344. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang Y, Zhang JS, Qian J, Huang GC and
Chen Q: Adrenomedullin regulates expressions of transforming growth
factor-beta1 and beta1-induced matrix metalloproteinase-2 in
hepatic stellate cells. Int J Exp Pathol. 87:177–184. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Lee EG, Lee S, Chae HJ, Park SJ, Lee YC
and Yoo WH: Adrenomedullin inhibits IL-1β-induced rheumatoid
synovial fibroblast proliferation and MMPs, COX-2 and PGE2
production. Inflammation. 34:335–343. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Li Y, Takemura G, Kosai K, Yuge K, Nagano
S, Esaki M, Goto K, Takahashi T, Hayakawa K, Koda M, et al:
Postinfarction treatment with an adenoviral vector expressing
hepatocyte growth factor relieves chronic left ventricular
remodeling and dysfunction in mice. Circulation. 107:2499–2506.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Edgley AJ, Krum H and Kelly DJ: Targeting
fibrosis for the treatment of heart failure: A role for
transforming growth factor-β. Cardiovasc Ther. 30:e30–e40. 2012.
View Article : Google Scholar : PubMed/NCBI
|