Introduction
Chemotherapeutic agents used for the treatment of
patients with cancer have been associated with serious adverse
effects, including tiredness, nephrotoxicity, anemia, leukopenia,
nausea, vomiting and loss of appetite (1–3).
Vomiting and appetite loss in patients with cancer have been linked
to the development of anorexia (4–6).
Numerous patients with cancer who suffer from chemotherapy-induced
anorexia have been reported to struggle with the continuation of
treatment, thus resulting in poor disease prognosis and impaired
quality of life (6,7). Megestrol acetate (MGA) is a steroidal
drug that is currently used as an appetite stimulant (8–11),
as it can increase appetite and promote weight gain in patients
with chemotherapy-induced anorexia (12,13).
However, the use of MGA has been associated with numerous adverse
effects, including excessive weight gain, fluctuations in appetite
and gastrointestinal complications (14,15).
Our previous study demonstrated that the traditional Korean
medicine sip-jeon-dae-bo-tang (SJDBT) was more effective in
improving food intake in cisplatin-induced and tumor-induced
anorexic mouse models compared with MGA (16,17).
Furthermore, the traditional Kampō medicine rikkunshito has been
demonstrated to ameliorate cisplatin-induced anorexia in rats and
humans (18,19). These findings suggested that herbal
formulas used in traditional Eastern medicine may have potential as
alternative therapeutic strategies for the prevention and treatment
of chemotherapy-associated anorexia in patients with cancer.
A herbal formula termed LCBP-Anocure-16001 (LA16001)
has been designed based on traditional Korean medicine and SJDBT,
and its effects have been investigated on chemotherapy-induced
anorexia (20). The formula
consists of a combination of herbal extracts from Atractylodes
japonica (AJ), Angelica gigas (AG), Astragalus
membranaceus (AM), which are included in SJDBT, with extracts
from Lonicera japonica Thunb. (LJ), Taraxacum
platycarpum H. Dahlstedt (TP), and Prunella vulgaris
var. asiatica (Nakai) Hara (PV), at a 1:1:1:1:1:1 ratio. AJ,
AG and AM have been used in traditional Korean medicine for the
treatment of anorexia, anemia, fatigue, general weakness, and for
immunopotentiation (21–26). In Korean tradition, LJ and TP have
been reported to alleviate drug-induced toxicity and fever, and
exert anti-inflammatory and diuretic effects (27,28),
whereas PV has traditionally been used for its antiviral and
antibacterial properties, and for the treatment of indigestion
(29,30).
In the present study, a cisplatin-induced anorexic
mouse model was employed to investigate the effects of LA16001 on
appetite. LA16001 was demonstrated to increase food intake and body
weight, and its effects were more potent compared with SJDBT and
MGA. The molecular mechanisms underlying its effects appeared to
involve altering the levels of ghrelin, leptin and interleukin
(IL)-6, potentially via regulation of the Janus kinase (JAK)
1/signal transducer and activator of transcription (STAT) 3
pathway. In addition, LA16001 was demonstrated to prevent
chemotherapy-associated leukopenia and neutropenia. These results
suggested that LA16001 may have potential as an alternative herbal
therapeutic strategy for the treatment of patients with
chemotherapy-induced anorexia.
Materials and methods
Herbal extracts and reagents
The herbal formulas were composed of extracts from
AJ, AG, AM, LJ, TP and PV, at w/w ratios of 1:1:1:1:1:1,
1:1:1:2:2:2 or 2:2:2:1:1:1. The herbal formulas were termed
LCBP-Anocure-16001, 16002 and 16003 (LA16001, LA16002, LA16003),
respectively. Herbal medicines were obtained from Kyung Hee Herb
Pharm (Wonju,) and Omniherb (Seoul, Korea). LA16001 was composed of
40 g each of AJ, AG, AM, LJ, TP and PV; LA16002 was composed of 25
g each of AJ, AG and AM and 50 g each of LJ, TP and PV; LA16003 was
composed of 50 g each of AJ, AG and AM and 25 g each of LJ, TP and
PV. The ingredients were placed in 15-fold volume of distilled
water and boiled at ~100°C for 3 h. The extracts were filtered
using 75 µm microfilters. The filtered residues were placed in a
15-fold volume of distilled water, boiled at ~100°C for 3 h and
refiltered through 75-µm microfilters. The extracted liquid was
concentrated under low pressure at 60°C. Finally, 81.8 g (LA16001),
84 g (LA16002) and 93.3 g (LA16003) dried extract was obtained; the
yields were 34.08, 37.33 and 41.47%, respectively. The extracts
were prepared according to the Good Manufacturing Practice
guidelines (31,32). SJDBT was obtained from Hanpoong
Pharm & Foods Co., Ltd. (Jeonju, Korea) and was prepared as
previously described (16).
Cisplatin (8 mg/kg) (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany)
was dissolved in distilled water.
Animal study
A total of 98 male BALB/c mice (age, 6 weeks) were
purchased from Orient Bio Inc. (Seongnam, Korea). Mice (n=4/group)
were randomly assigned to the following groups: Normal, control,
cisplatin, LA16001, LA16002, LA16003, SJDBT, MGA (Sigma-Aldrich;
Merck KGaA), AJ, AG, AP, LJ, TP and PV, and were maintained at
23±3°C (humidity, 55±15%), under a 12-h light/dark cycle. Access to
food and water was ad libitum, apart from during the
experimental period. All groups (except the normal group) were
fasted for 24 h prior to the start of the experiment (the normal
group was used as a control for starvation). The control group did
not receive any drug treatment, only starvation; this group was
used as a control for the cisplatin group. All groups (except the
normal and control groups) were induced to become anorexic through
administration of cisplatin. Therefore, the cisplatin group was
used as a control for the drug treatments. The drug was
administered at the concentration of 1,000 mg/kg on days 1–3. After
24 h, 8 mg/kg cisplatin was intraperitoneally injected into the
mice. During fasting, water was continuously provided and food was
provided following the oral administration of drugs. Food intake
and body weight were measured daily throughout the duration of the
study (Fig. 1A). An equal amount
of food was given at the same time each day and mice were weighed
the following day. All mice were sacrificed at the time labeled
‘Sac’ in the animal study design presented in Fig. 1A. Blood and adipose tissue,
stomach, and hypothalamus were obtained from the sacrificed
mice.
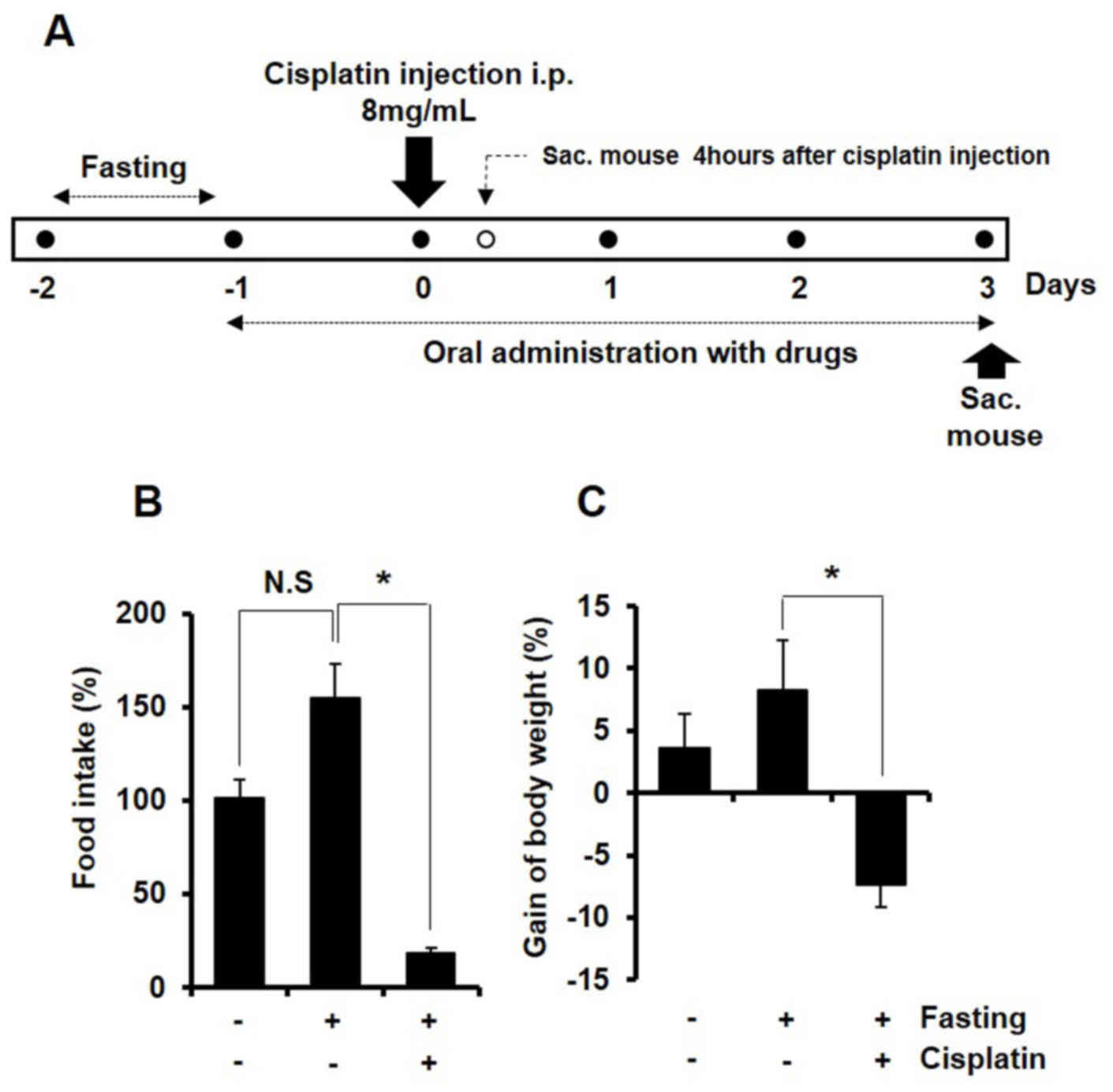 | Figure 1.Treatment with cisplatin decreases
food intake in BALB/c mice. (A) Time frame of the in vivo
study. All mice (except the normal group) fasted for 24 h and all
mice (except the normal and control groups) were intraperitoneally
administered with 8 mg/kg cisplatin. The groups were administered
orally LA16001, LA16002, LA16003, SJDBT, MAG, AP, AG, AJ, TP, LJ
and PV for three days, respectively. Mice were sacrificed 4 h or 3
days post-injection and tissue samples were collected for further
analysis. (B) Food intake and (C) body weight gain were monitored
daily throughout the duration of the study. Data are presented as
the mean ± standard deviation. *P<0.05. NS, not significant;
Sac., sacrifice. |
The present study was approved by the Animal Ethics
Committee of Kyung Hee University [approval no. KHUASP
(SE)-016-100].
ELISA and blood analysis
Samples from stomach, fat and hypothalamic tissue
were isolated from mice sacrificed 4 h subsequent to the
administration of cisplatin (n=4 mice/group), and lysed with
radioimmunoprecipitation assay lysis buffer (Biosesang, Inc.,
Seongnam, Korea). Lysates were centrifuged at 196 × g for 20 min at
room temperature, and supernatants were collected and used for the
assessment of target proteins using ELISA. The levels of ghrelin
(cat. no. EZRGRA-90K; EMD Millipore, Billerica, MA, USA), leptin
(cat. no. DY498; R&D Systems, Inc., Minneapolis, MN, USA) and
IL-6 (cat. no. 555240; BD Biosciences, San Jose, CA, USA) were
measured using ELISA kits according to the manufacturer's protocol.
The blood of the mice sacrificed 3 days subsequent to the
administration of cisplatin was used. Control group and cisplatin
group, LA16001 group mice were used, 4 mice/group. Exsanguination
was performed, and whole blood was analyzed using the
HEMAVET® 950 hematology system (Drew Scientific Inc.,
Miami Lakes, FL, USA). SigmaPlot, version 2001 (Systat Software,
Inc., San Jose, CA, USA) was used for analysis of the blood data.
To anesthetize, 400 µl/mouse 1.2% avertin solution was
intraperitoneally injected [1.2% avertin solution, 0.5 g
2,2,2-tribromoethanol powder (Sigma-Aldrich; Merck KGaA) dissolved
in 1 ml 2-methyl-2-butanol (Sigma-Aldrich; Merck KGaA) at 55°C, and
39 ml PBS]. The avertin solution was filtered through a Nalgene
0.22-µm filter (Thermo Fisher Scientific, Inc., Waltham, MA,
USA).
Western blot analysis
Adipose tissue was isolated from mice. To obtain
protein, the adipose tissue was lysed using
radioimmunoprecipitation assay buffer (Biosesang, Inc.) and was
centrifuged at 24,562 × g at 4°C for 20 min. Protein concentration
was measured using a Bradford assay. Equal amounts of extracted
protein samples (20 µg) were separated by 8% SDS-PAGE and
transferred onto polyvinylidene difluoride membranes (GE Healthcare
Life Sciences, Chalfont, UK). Membranes were blocked in 1% bovine
serum albumin (Sigma-Aldrich; Merck KGaA) and 2% skimmed milk for 1
h at room temperature. Membranes were then incubated at 4°C
overnight with the following primary antibodies:
Anti-phosphorylated (p)-JAK1 (1:1,000; cat no. 3331; Cell Signaling
Technology, Inc., Danvers, MA, USA), anti-p-STAT3 (1:1,000; cat no.
9131; Cell Signaling Technology, Inc.) and anti-α-tubulin
(1:10,000; cat no. T-5168; Sigma-Aldrich; Merck KGaA).
Subsequently, membranes were incubated with horseradish
peroxidase-conjugated secondary antibody (1:2,000 in 1× PBS with
0.05% Tween-20; cat. no. 5450-0010; KPL, Inc., Gaithersburg, MD,
USA) for 1 h at room temperature. Protein bands were visualized
using Enhanced Chemiluminescence kit solution (DoGen, Seoul, Korea)
and quantified using ImageJ software version 1.49v (National
Institutes of Health, Bethesda, MD, USA) and normalized to
α-tubulin.
Statistical analysis
The data are expressed as the mean ± standard
deviation and experiments were performed on 4 mice/group. The
statistical significance of the differences between groups was
assessed using Student's t-test. Statistical analysis was performed
using Microsoft Excel 2013 (Microsoft Corporation, Redmond, WA,
USA). P<0.05 was considered to indicate a statistically
significant difference. One-way analysis of variance was
additionally performed with post hoc test (Tukey), using SPSS
software (version 23; IBM Corp., Armonk, NY, USA).
Results
LA16001-3 increases food intake in
cisplatin-induced anorexic mice
Mice were fasted for 24 h and intraperitoneally
injected with 8 mg/kg cisplatin. Experimental drugs were orally
administrated 24 h before cisplatin injection. To investigate the
effects of the experimental drugs on food intake and body weight,
mice were maintained for 3 days following the injection of
cisplatin (Fig. 1A). The results
of the present study demonstrated that following cisplatin
administration, food intake and body weight decreased (Figs. 1B and C, respectively). Notably,
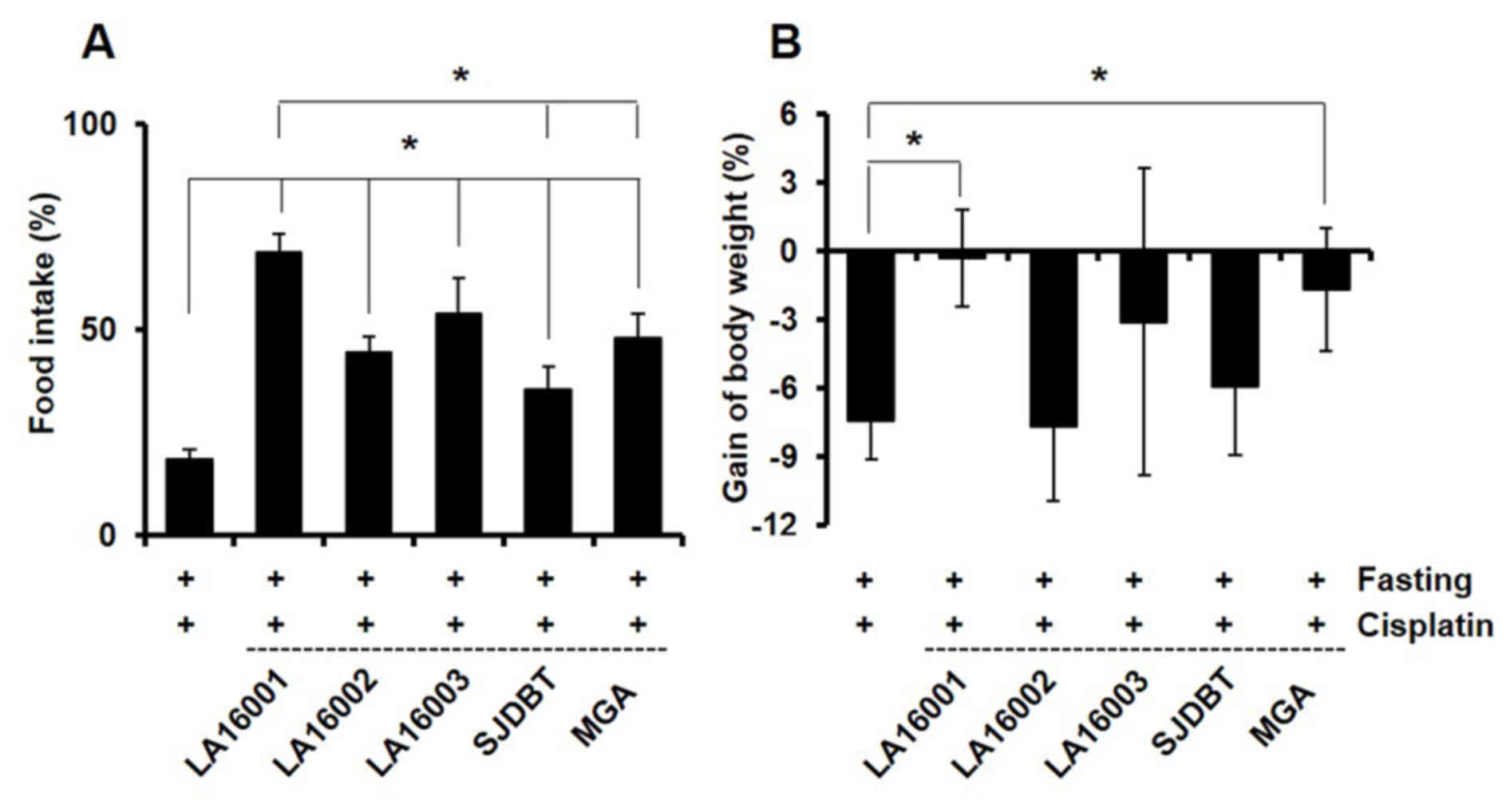
LA16001, LA16002, LA16003, SJDBT and MGA alleviated
cisplatin-induced loss of appetite (Fig. 2A), and LA16001 and MGA
significantly reversed decreases in body weight (Fig. 2B). LA16001 appeared to have more
potent orexigenic effects on food intake, compared with all other
treatment groups (Fig. 2A). In
addition, all treatment groups demonstrated significantly reversed
cisplatin-induced loss of appetite, with LA16001 exerting the most
pronounced effect (Fig. 3A).
Furthermore, LA16001, AP, AG and TP treatment significantly
reversed loss of body weight in experimental mice, again with
LA16001 being the most effective (Fig.
3B). These results suggested that LA16001 may be able to
counteract cisplatin-induced anorexia in vivo.
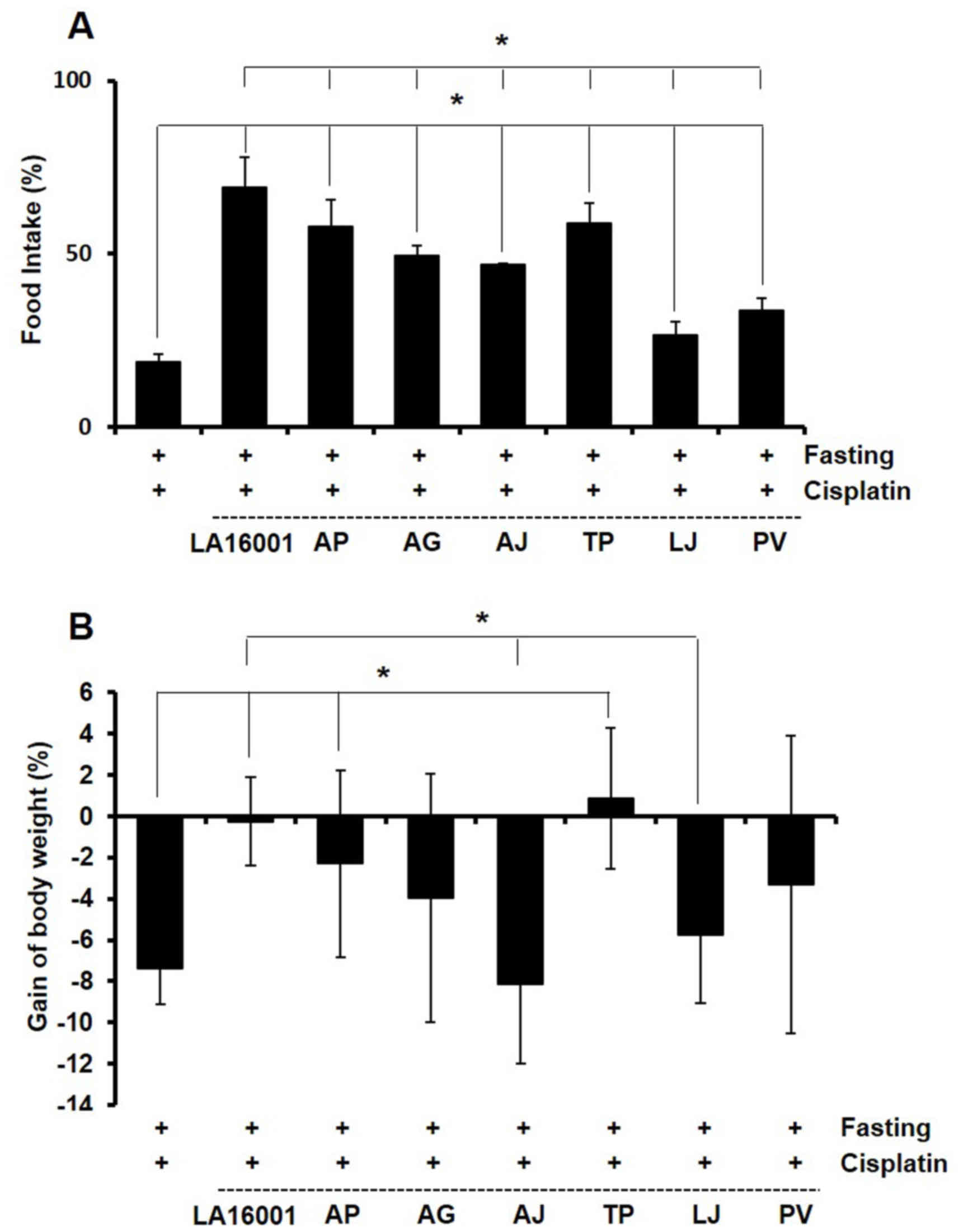 | Figure 3.Effects of LA16001 on food intake and
body weight in cisplatin-induced anorexic mice. Mice were fasted
for 24 h and orally administered with LA16001, AJ, AG, AP, LJ, TP
and PV (1,000 mg/kg). After 24 h, mice received an intraperitoneal
cisplatin (8 mg/kg) injection. (A) Food intake and (B) body weight
gain were monitored daily throughout the duration of the study.
Data are presented as the mean ± standard deviation. *P<0.05.
LA16001, LCBP-Anocure 16001; AJ, Atractylodes japonica; AG,
Angelica gigas; AM, Astragalus membranaceus; LJ,
Lonicera japonica Thunb.; TP, Taraxacum platycarpum
H. Dahlstedt; PV, Prunella vulgaris var. asiatica
(Nakai) Hara. |
LA16001 increases the levels of
appetite-associated hormones in cisplatin-treated mice
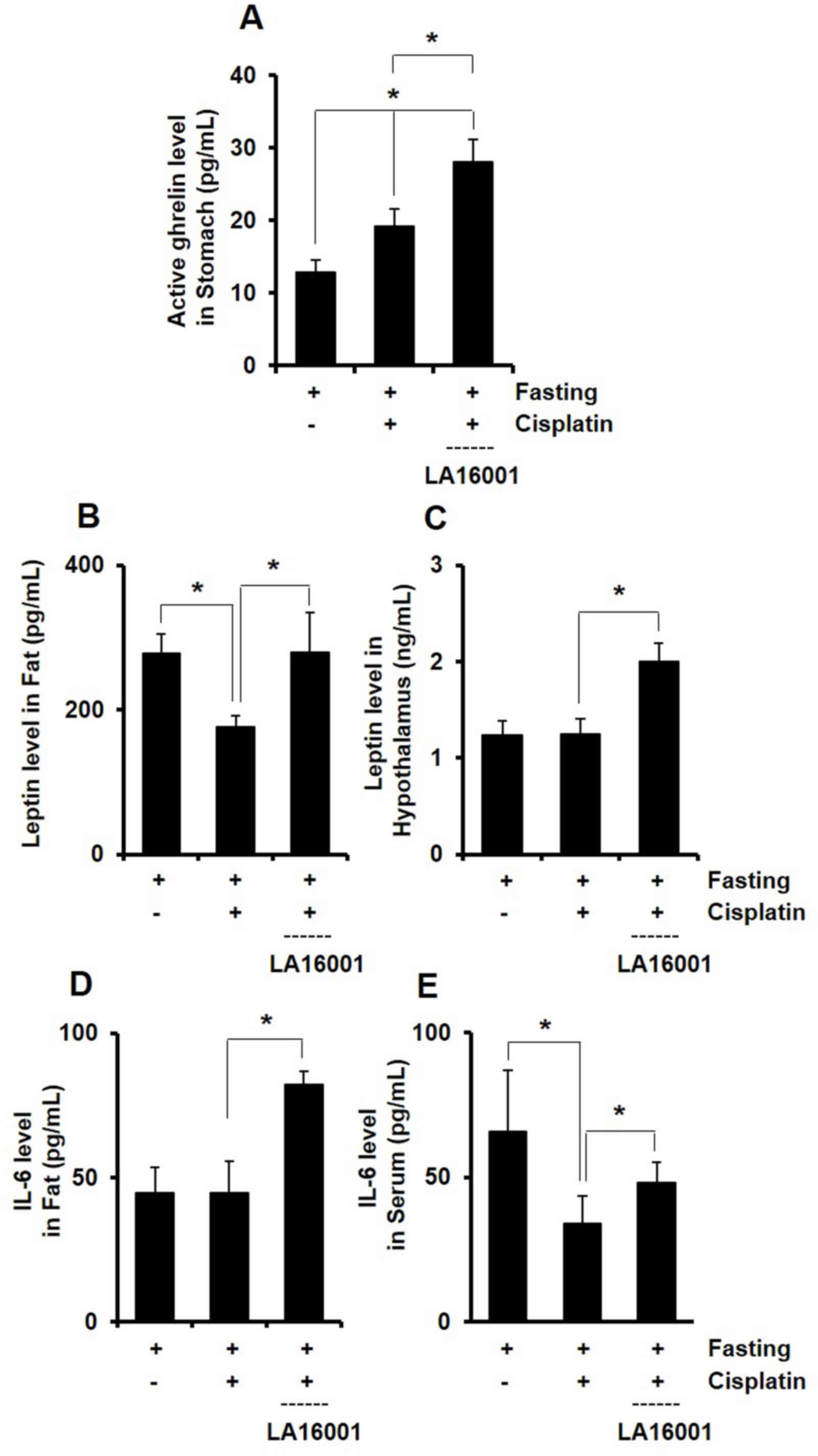
A total of 4 h post-cisplatin injection, the hormone
levels of ghrelin, leptin and IL-6 were assessed in mouse tissue
and serum samples. Mice in the cisplatin and LA16001 groups
exhibited increased ghrelin levels in stomach tissue samples
compared with mice in the fasting control group (Fig. 4A). Cisplatin was demonstrated to
reduce leptin levels in fat and hypothalamic tissue, whereas
LA16001 appeared to counteract this effect (Figs. 4B and C). IL-6 levels were assessed
in serum and fat tissue samples. IL-6 levels in fat tissue were not
altered following cisplatin administration; however, they were
significantly decreased in serum samples. Notably, LA16001
administration was demonstrated to significantly increase fat
tissue and serum IL-6 levels (Figs. 4D
and E). These results indicated that LA16001 administration
increased the levels of ghrelin, leptin and IL-6 in
cisplatin-treated mice.
LA16001 activates the JAK1/STAT3
signaling pathway in fat tissue
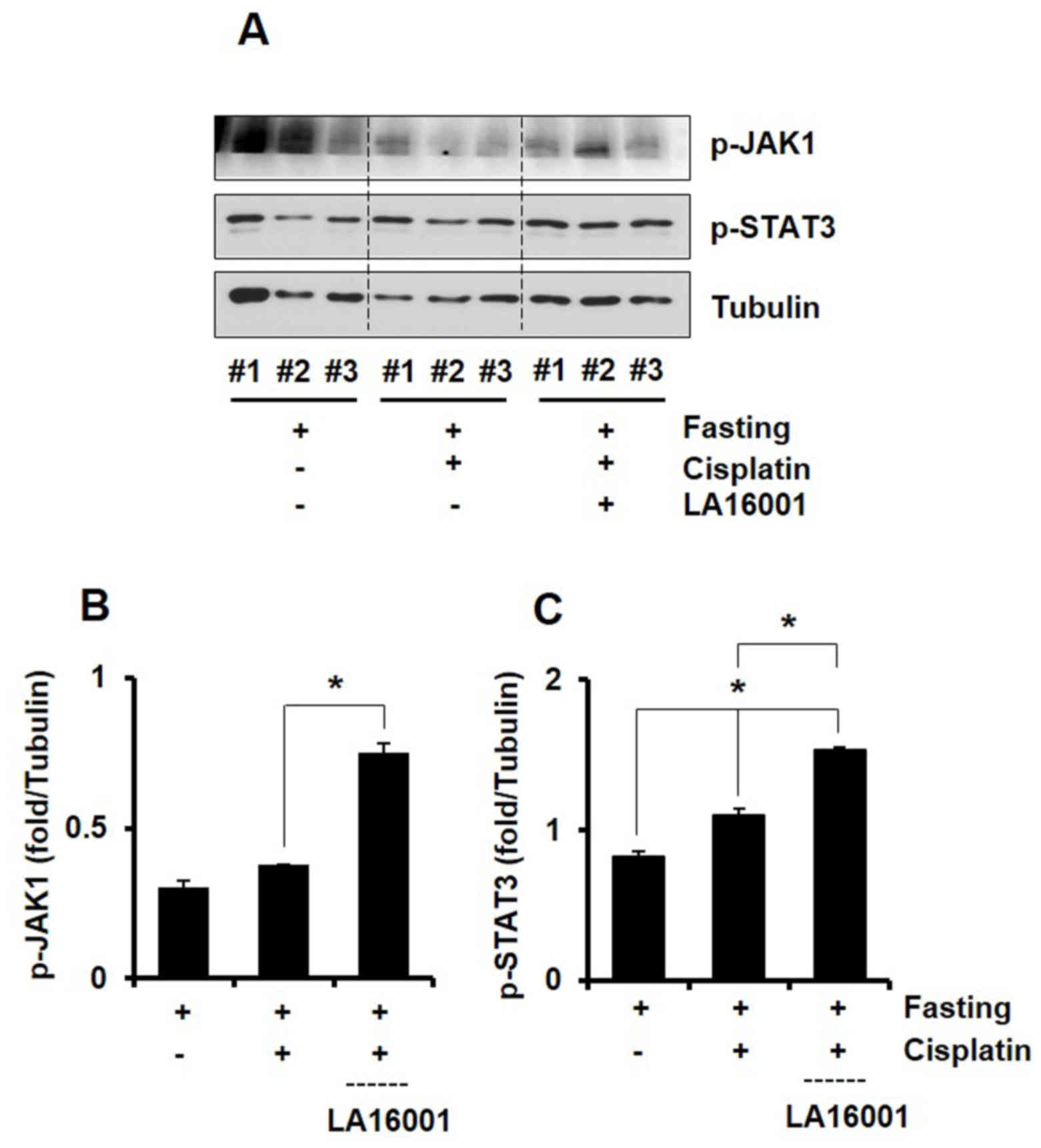
A total of 4 h post-cisplatin injection, the
phosphorylation status of JAK1 and STAT3 was investigated in fat
tissue samples. LA16001 administration resulted in significantly
increased protein expression levels of p-JAK1 and p-STAT3 compared
with the cisplatin group (Figs. 5A and
B). These results suggested that the molecular mechanisms
underlying the orexigenic effects of LA16001 in cisplatin-induced
anorexic mice may involve the activation of the JAK1/STAT3
intracellular signaling pathway.
LA16001 prevents immunosuppression in
cisplatin-induced anorexic mice
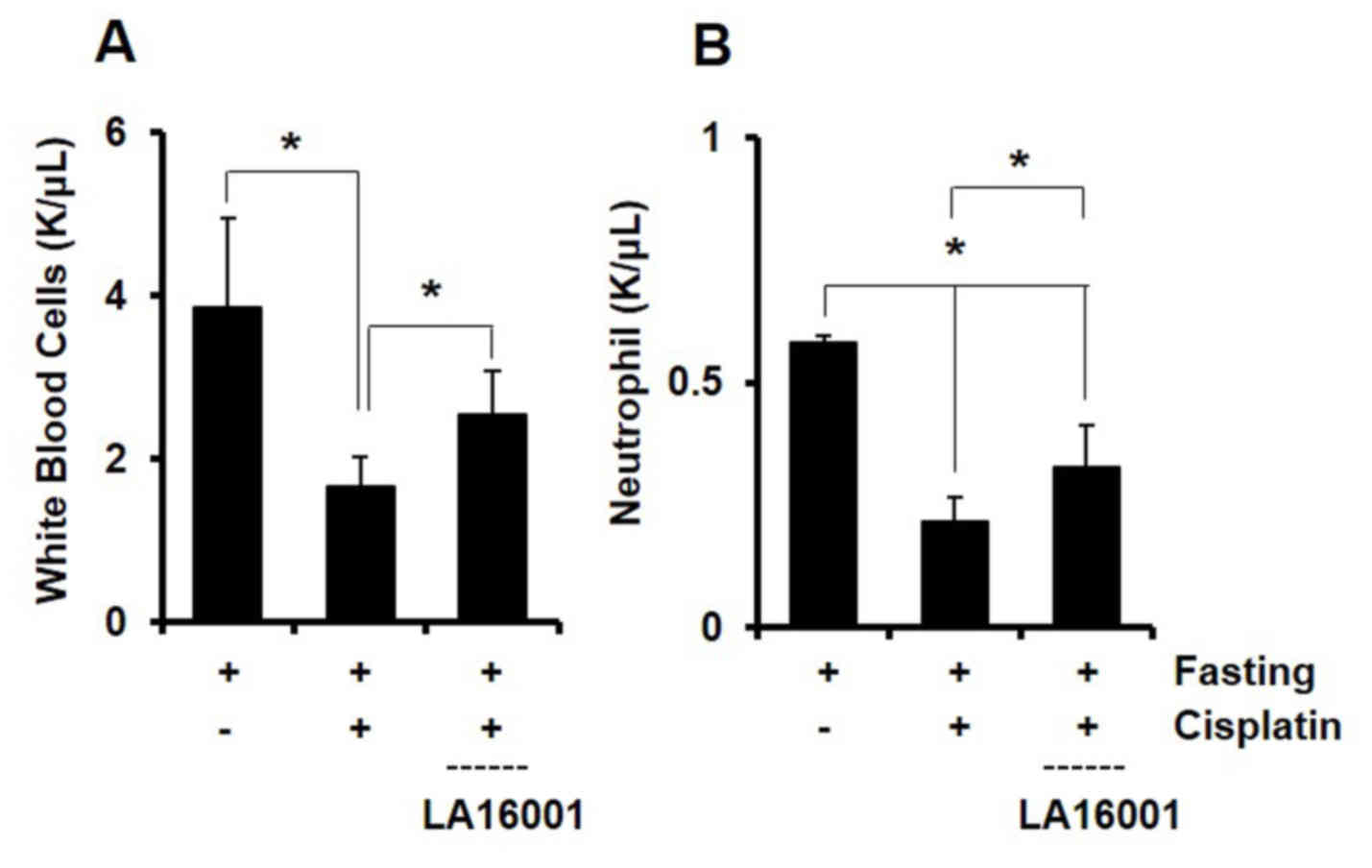
White blood cell and neutrophil counts were
performed 3 days after cisplatin administration to evaluate the
effects of LA16001 on chemotherapy-induced immunosuppression.
Treatment with cisplatin was demonstrated to reduce the numbers of
white blood cells (Fig. 6A) and
neutrophils (Fig. 6B) in mouse
whole blood samples. Notably, LA16001 administration appeared to
counteract the effects of cisplatin on immune cell numbers
(Fig. 6). These results suggested
that LA16001 may mitigate the immunosuppressive effects associated
with cisplatin treatment.
Discussion
Chemotherapy induces various side effects, including
anorexia. Anticancer drugs are toxic to healthy cells as they share
characteristics with rapidly dividing cancer cells. The healthy
cells that are primarily affected are hair follicle cells, and
cells of the mouth, stomach, large intestine and the mucous
membrane of the anus, which exhibit a rapid growth rate compared
with other cells (33,34). Anorexia is particularly induced by
gastrointestinal toxicity (35).
Anorexia is a frequent adverse event following cancer chemotherapy,
and has been associated with poor disease prognosis and treatment
discontinuation (1).
Previous studies have suggested that herbal formulas
used in traditional Eastern medicine may have potential as
alternative strategies for the treatment of chemotherapy-induced
anorexia (16,36,37).
In the present study, the herbal formula LA16001 was developed, and
was demonstrated to effectively counteract the development of
chemotherapy-induced anorexia in mice. LA16001 prevented the loss
of appetite and body weight following cisplatin administration.
Ghrelin is a hormone critical for appetite regulation, and its
levels have been reported to decrease following food intake
(38). Leptin is a hormone
functioning as an appetite suppressant, and its levels increase
following feeding (39). IL-6 is a
proinflammatory cytokine that has been revealed to function
together with leptin to increase appetite (40). Patients with cancer with anorexia
due to anticancer drugs exhibit increased ghrelin and decreased
leptin (41). Leptin and IL-6 have
been demonstrated to act through the regulation of the JAK1/STAT3
intracellular signaling pathway. STAT3 is involved in the food
intake and glucose metabolism of leptin. Leptin was observed to
function abnormally in STAT3-deficient mice (41,42).
In the present study, LA16001 was revealed to increase ghrelin,
leptin, IL-6, p-JAK1 and p-STAT3 levels in cisplatin-treated mice,
suggesting that it may mitigate cisplatin-associated appetite
suppression via a mechanism involving the JAK1/STA3 signaling
pathway.
Our previous study revealed that SJDBT counteracted
the appetite-suppressing effects associated with chemotherapy in
cisplatin-induced anorexic mice (16). Notably, SJDBT appeared to prevent
cisplatin-induced anorexia more effectively compared with MGA
(16). In the present study,
LA16001 appeared to exert more potent effects against
cisplatin-induced anorexia compared with SJDBT. The effects of
LA16001 on the expression of ghrelin, leptin and IL-6 suggested
that this novel herbal composition may effectively treat
cisplatin-induced anorexia.
Anticancer drugs target the bone marrow cells from
which blood cells are derived, thereby decreasing white blood cell
count and inducing immune degradation (43–45).
Immunosuppression is a frequent and serious adverse event observed
in patients with cancer receiving chemotherapy (43–45).
The present study assessed the effects of LA16001 on
cisplatin-induced immunosuppression, through the evaluation of
white blood cell and neutrophil counts in cisplatin-treated mice.
The present results suggested that LA16001 may prevent immune
disorders associated with chemotherapy in cisplatin-induced
anorexic mice.
In conclusion, the present study revealed that the
herbal mixture LA16001 ameliorated the appetite-suppressing effects
of cisplatin in a chemotherapy-induced anorexic mouse model.
LA16001 was demonstrated to modulate hormone and cytokine levels,
potentially via activation of the JAK1/STAT3 intracellular
signaling pathway. Furthermore, LA16001 counteracted the
immunosuppressive effects of cisplatin, as demonstrated by an
increase in immune cell numbers. The present results suggested that
LA16001 may have potential as an alternative therapeutic strategy
for the prevention and treatment of chemotherapy-associated
anorexia in patients with cancer. Further studies are required to
validate the orexigenic effects of LA16001, and to investigate the
cellular and molecular mechanisms underlying its actions.
Acknowledgements
The authors would like to thank Hanpoong Pharm &
Foods Co., Ltd. (Jeonju, Korea) for providing some of the herbal
extracts used in the present study. The present study was supported
by the Korean Medicine R&D project of the Ministry of Health
and Welfare (grant no. B120014) and by the National Research
Foundation of Korea, funded by the Korean Ministry of Science
(grant no. 2007-0054931).
References
|
1
|
Yao X, Panichpisal K, Kurtzman N and
Nugent K: Cisplatin nephrotoxicity: A review. Am J Med Sci.
334:115–124. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Coates A, Abraham S, Kaye SB, Sowerbutts
T, Frewin C, Fox RM and Tattersall MH: On the receiving end-patient
perception of the side-effects of cancer chemotherapy. Eur J Cancer
Clin Oncol. 19:203–208. 1983. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Decker-Baumann C, Buhl K, Frohmüller S,
von Herbay A, Dueck M and Schlag PM: Reduction of
chemotherapy-induced side-effects by parenteral glutamine
supplementation in patients with metastatic colorectal cancer. Eur
J Cancer. 35:202–207. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Mantovani G, Maccio A, Bianchi A, Curreli
L, Ghiani M, Santona MC and Del Giacco GS: Megestrol acetate in
neoplastic anorexia/cachexia: Clinical evaluation and comparison
with cytokine levels in patients with head and neck carcinoma
treated with neoadjuvant chemotherapy. Int J Clin Lab Res.
25:135–141. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bender CM, McDaniel RW, Murphy-Ende K,
Pickett M, Rittenberg CN, Rogers MP, Schneider SM and Schwartz RN:
Chemotherapy-induced nausea and vomiting. Clin J Oncol Nurs.
6:94–102. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bosnjak SM, Dimitrijevic J and Djordjevic
F: Cancer and chemotherapy-induced nausea and vomiting: A focus on
olanzapine. Curr Opin Support Palliat Care. 10:180–188. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Metri K, Bhargav H, Chowdhury P and Koka
PS: Ayurveda for chemo-radiotherapy induced side effects in cancer
patients. J Stem Cells. 8:115–129. 2013.PubMed/NCBI
|
|
8
|
Aoyagi T, Terracina KP, Raza A, Matsubara
H and Takabe K: Cancer cachexia, mechanism and treatment. World J
Gastrointest Oncol. 7:17–29. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Fernández-Lucas M, Díaz-Domínguez ME,
Ruiz-Roso G, Raoch V, Teruel-Briones JL and
Quereda-Rodríguez-Navarro C: Anorexia and megestrol acetate:
Treatment versus placebo controlled study. Nefrologia. 34:416–417.
2014.PubMed/NCBI
|
|
10
|
Cuvelier GD, Baker TJ, Peddie EF, Casey
LM, Lambert PJ, Distefano DS, Wardle MG, Mychajlunow BA, Romanick
MA, Dix DB and Wilson BA: A randomized, double-blind,
placebo-controlled clinical trial of megestrol acetate as an
appetite stimulant in children with weight loss due to cancer
and/or cancer therapy. Pediatr Blood Cancer. 61:672–679. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
De Conno F, Martini C, Zecca E, Balzarini
A, Venturino P, Groff L and Caraceni A: Megestrol acetate for
anorexia in patients with far-advanced cancer: A double-blind
controlled clinical trial. Eur J Cancer. 34:1705–1709. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ruiz Garcia V, López-Briz E, Carbonell
Sanchis R, Gonzalvez Perales JL and Bort-Marti S: Megestrol acetate
for treatment of anorexia-cachexia syndrome. Cochrane Database Syst
Rev. 28:CD0043102013.
|
|
13
|
Sung JH, An HS, Jeong JH, Shin S and Song
SY: Megestrol acetate increases the proliferation, migration, and
adipogenic differentiation of adipose-derived stem cells via
glucocorticoid receptor. Stem Cells Transl Med. 4:789–799. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Nelson KA, Walsh D and Hussein M: A phase
II study of low-dose megestrol acetate using twice-daily dosing for
anorexia in nonhormonally dependent cancer. Am J Hosp Palliat Care.
19:206–210. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Votaw ML: Excessive sweating: Unusual side
effect of megestrol acetate therapy. Tenn Med. 90:591997.PubMed/NCBI
|
|
16
|
Woo SM, Choi YK, Kim AJ, Yun YJ, Shin YC,
Cho SG and Ko SG: Sip-jeon-dea-bo-tang, a traditional herbal
medicine, ameliorates cisplatin-induced anorexia via the activation
of JAK1/STAT3-mediated leptin and IL-6 production in the fat tissue
of mice. Mol Med Rep. 13:2967–2972. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Choi YK, Jung KY, Woo SM, Yun YJ, Jun CY,
Park JH, Shin YC, Cho SG and Ko SG: Effect of Sipjeondaebotang on
cancer-induced anorexia and cachexia in CT-26 tumor-bearing mice.
Mediators Inflamm. 2014:7365632014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ohno T, Yanai M, Ando H, Toyomasu Y, Ogawa
A, Morita H, Ogata K, Mochiki E, Asao T and Kuwano H: Rikkunshito,
a traditional Japanese medicine, suppresses cisplatin-induced
anorexia in humans. Clin Exp Gastroenterol. 4:291–296. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Takeda H, Sadakane C, Hattori T, Katsurada
T, Ohkawara T, Nagai K and Asaka M: Rikkunshito, an herbal
medicine, suppresses cisplatin-induced anorexia in rats via 5-HT2
receptor antagonism. Gastroenterology. 134:2004–2013. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Song BK, Won JH and Kim S: Historical
Medical Value of Donguibogam. J Pharmacopuncture. 19:16–20. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zee-Cheng RK: Shi-quan-da-bu-tang (ten
significant tonic decoction), SQT. A potent Chinese biological
response modifier in cancer immunotherapy, potentiation and
detoxification of anticancer drugs. Methods Find Exp Clin
Pharmacol. 14:725–736. 1992.PubMed/NCBI
|
|
22
|
Piao JH, Zhang L, Zhang H, Gao TH and Li
XH: Experimental studies on antianimia effect of shengxuesu.
Zhongguo Zhong Yao Za Zh. 28:544–547. 2003.(In Chinese).
|
|
23
|
Sun W, Meng K, Qi C, Yang X, Wang Y, Fan
W, Yan Z, Zhao X and Liu J: Immune-enhancing activity of
polysaccharides isolated from Atractylodis macrocephalae Koidz.
Carbohydr Polym. 126:91–96. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lv Y, Feng X and Zhu B: Study on effect of
Astragalus membranaceus injection on hematopoiesis in anemic mice
with myelosuppression. Zhong Yao Cai. 28:791–793. 2005.(In
Chinese). PubMed/NCBI
|
|
25
|
Zhu X and Zhu B: Effect of Astragalus
membranaceus injection on megakaryocyte hematopoiesis in anemic
mice. Hua Xi Yi Ke Da Xue Xue Bao. 32:590–592. 2001.(In Chinese).
PubMed/NCBI
|
|
26
|
Auyeung KK, Han QB and Ko JK: Astragalus
membranaceus: A review of its protection against inflammation and
gastrointestinal cancers. Am J Chin Med. 44:1–22. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Shang X, Pan H, Li M, Miao X and Ding H:
Lonicera japonica Thunb.: Ethnopharmacology, phytochemistry and
pharmacology of an important traditional Chinese medicine. J
Ethnopharmacol. 138:1–21. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rasool S and Sharma B: Taraxacum
officinale: A high value less known medicinal plant. Ann Plant Sci.
3:908–915. 2014.
|
|
29
|
Kirbağ S, Zengin F and Kursat M:
Antimicrobial activities of extracts of some plants. Pak J Bot.
41:2067–2070. 2009.
|
|
30
|
Fang X, Yu MM, Yuen WH, Zee SY and Chang
RC: Immune modulatory effects of Prunella vulgaris L. On
monocytes/macrophages. Int J Mol Med. 16:1109–1116. 2005.PubMed/NCBI
|
|
31
|
FDA K: Safety MOFAD: Guidance on good
manufacturing practices (GMP) for active pharmaceutical
ingredients. 2015.
|
|
32
|
Organization WH: Annex 3 supplementary
guidelines on good manufacturing practices for the manufacture of
herbal medicines. 85–106. 2006.
|
|
33
|
Damia G and D'Incalci M: Mechanisms of
resistance to alkylating agents. Cytotechnology. 27:165–173. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Malhotra V and Perry MC: Classical
chemotherapy: Mechanisms, toxicities and the therapeutic window.
Cancer Biol Ther. 2 4 Suppl 1:S2–S4. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lee CS, Ryan EJ and Doherty GA:
Gastro-intestinal toxicity of chemotherapeutics in colorectal
cancer: The role of inflammation. World J Gastroenterol.
20:3751–3761. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Ohnishi S and Takeda H: Herbal medicines
for the treatment of cancer chemotherapy-induced side effects.
Front Pharmacol. 6:142015. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Takeda H, Muto S, Nakagawa K, Ohnishi S
and Asaka M: Rikkunshito and ghrelin secretion. Curr Pharm Des.
18:4827–4838. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dixit VD, Schaffer EM, Pyle RS, Collins
GD, Sakthivel SK, Palaniappan R, Lillard JW Jr and Taub DD: Ghrelin
inhibits leptin- and activation-induced proinflammatory cytokine
expression by human monocytes and T cells. J Clin Invest.
114:57–66. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Zhang F, Chen Y, Heiman M and Dimarchi R:
Leptin: Structure, function and biology. Vitam Horm. 71:345–372.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Shirazi R, Palsdottir V, Collander J,
Anesten F, Vogel H, Langlet F, Jaschke A, Schürmann A, Prévot V,
Shao R, et al: Glucagon-like peptide 1 receptor induced suppression
of food intake, and body weight is mediated by central IL-1 and
IL-6. Proc Natl Acad Sci USA. 110:pp. 16199–16204. 2013; View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Buettner C, Pocai A, Muse ED, Etgen AM,
Myers MG Jr and Rossetti L: Critical role of STAT3 in leptin's
metabolic actions. Cell Metab. 4:49–60. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Engineer DR and Garcia JM: Leptin in
anorexia and cachexia syndrome. Int J Pept. 2012:2874572012.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Suprasert P, Aue-Aungkul A and Pautad N:
Lack of Relationship of Egg white intake with occurrence of
leukopenia in gynecologic cancer patients during chemotherapy.
Asian Pac J Cancer Prev. 17:1265–1267. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Rodgers GM III, Becker PS, Bennett CL,
Cella D, Chanan-Khan A, Chesney C, Cleeland C, Coccia PF,
Djulbegovic B, Garst JL, et al: Cancer- and chemotherapy-induced
anemia. J Natl Compr Canc Netw. 6:536–564. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Munoz-Langa J, De Castro J, Gasco P, et
al: Chemotherapy-associated anemia in patients with lung cancer: An
epidemiological, retrospective and multicenter study. Future Oncol.
11:1665–1674. View Article : Google Scholar : PubMed/NCBI
|