Introduction
Pre-eclampsia (PE) is a common but complex condition
that can occur in pregnant women, characterized by hypertension and
proteinuria (1). It has been
reported to occur in 3–8% of pregnancies worldwide (2). According to the level of hypertension
and proteinuria, PE is classified from mild to severe, using the
American Congress of Obstetricians and Gynecologists Committee on
Practice Bulletin-Obstetrics (3).
Pre-existing conditions including diabetes mellitus, obesity, renal
disease and hypertension are considered to be risk factors for PE
development (4). PE is a
multi-system disease that threatens both maternal and newborn
health (5). PE development is
thought to be multifactorial, involving the disruption of vascular
homeostasis in the placenta (6),
altered placental transcriptome (7), reduced 2-methoxyestradiol level
(8) and hypoxia (9). It is well established that delivery
of the fetus and placenta is the most effective way to treat PE.
However, the exact mechanisms of PE development still remain
unclear and it is important to further elucidate the exact
pathophysiology of this disease.
microRNAs (miRNAs/miRs) are small, single-stranded
non-coding RNA molecules ~22 nucleotides in length. miRNAs suppress
the expression of their target mRNAs through RNA silencing and
post-transcriptional regulation (10–13).
It has been reported that the chromosome 19 miRNA cluster (C19MC),
C14MC, miRNA (miR-37-3) and miR-17-92 clusters are involved
placenta development (14,15). Accumulating evidence indicates that
the abnormal miRNA expression contributes to PE development
(16,17). For example, the upregulation of
miR-520g can promote PE via the inhibition of trophoblast migration
and invasion (18). The human
placenta expresses numerous angiogenic factors that may be involved
in maternal vascular adaptation to pregnancy, including vascular
endothelial growth factor (VEGF). Placental expression of VEGF has
been reported in villous trophoblasts, but immunostaining indicated
that VEGF is not expressed in the villous endothelium (19,20).
VEGFA in particular is a highly specific mitogen for vascular
endothelial cell growth. Previous studies have reported that
miR-203 is involved in the pathogenesis of tumor growth by
targeting VEGFA (21,22). Thus, the potential involvement of
miR-203 in trophoblast VEGFA expression and PE development were
investigated. In combination with previous research, the present
study may aid in the development of miRNAs or miRNA mimics as
effective biomarkers or therapeutic agents for the treatment of
placental diseases.
Materials and methods
Ethics statement
Human placental tissue used in this research was
collected from 38 patients that had provided written informed
consent. The present study was approved by the Ethics Committee of
Zhongnan Hospital of Wuhan University (Wuhan, China; permit no.
2016016).
Human placenta collection
Human placental tissues were collected from
artificial labor and caesarean sections at the Renmin Hospital of
Wuhan University (Wuhan, Hubei, China) between June 2016 and
February 2017. All samples were frozen in liquid nitrogen for 5 min
and stored at −80°C for subsequent analyses. Placental tissue was
collected from pregnant women who met the following inclusion
criteria: Monocyesis, normal pregnancy at the time of sample
collection and no pre-existing clinical conditions, including
diabetes, chronic nephritis, cardiovascular, hepatic or autoimmune
disease. Samples were subsequently categorized into the PE or
normal group according to the presence or absence of PE. Detailed
clinical characteristics of the sample groups are presented in
Table I.
 | Table I.Clinical characteristics of normal
and pre-eclampsia pregnancies. |
Table I.
Clinical characteristics of normal
and pre-eclampsia pregnancies.
| Characteristic | Pre-eclampsia
(n=18) | Normal (n=20) | P-value |
|---|
| Maternal age
(years) |
30.3±4.6 |
29.5±4.3 |
0.58 |
| Gestational age
(weeks) |
36.5±1.2 |
38.3±1.9 | <0.05 |
| Birth weight
(g) | 2,984.9±684.2 | 3,624.6±363.3 | <0.05 |
| Placenta weight
(g) |
559.2±79.4 |
642.9±108.6 | <0.05 |
| Mean arterial
pressure (mmHg) |
122.5±10.6 |
94.3±6.4 | <0.05 |
| Body mass index
(kg/m2) |
23.3±4.3 |
24.3±3.2 |
0.42 |
Hematoxylin and eosin (H&E)
staining
Placental tissue from different treatment groups was
sequentially fixed in 4% paraformaldehyde at room temperature for
12 h, dehydrated in 60, 70, 80, 90, 95% and absolute ethanol
respectively for 30 min at room temperature, embedded in paraffin
and cut into 5 µm sections. Following deparaffinization in xylene
for 5 min twice and rehydration sequentially in absolute, 95, 90,
80, 70 and 60% ethanol respectively for 5 min at room temperature,
placental tissue sections were stained with H&E. Images were
captured under ×100 magnification by a Nikon Eclipse Ci microscope
(Nikon Corporation, Tokyo, Japan).
Immunohistochemistry (IHC) detection
of VEGFA in the placenta
Immunohistochemical detection of VEGFA was conducted
on 5-µm-thick paraffin-embedded placental tissue sections using the
avidin-streptavidin-biotin method. IHC operation kits was purchased
from Beyotime Institute of Biotechnology (Haimen, Jiangsu, China).
The standard IHC protocol was performed according to the
manufacturer's instructions. Following blocking in immunnol
staining blocking buffer (P0102; Beyotime Institute of
Biotechnology) at room temperature for 1 h and incubation with a
human VEGFA primary antibody (1:50; ab1316; Abcam, Shanghai, China)
at 4°C overnight, sections were washed with PBS and subsequently
incubated with the biotin-conjugated goat anti-mouse IgG (TA130008;
1:200; OriGene Technologies, Inc., Beijing, China) at room
temperature for 45 min. Visualization was achieved with
diaminobenzidine staining (OriGene Technologies, Inc., Beijing,
China) for 5 min and counterstaining with hematoxylin solution for
2 min at room temperature. Images were captured with the Nikon
Eclipse Ci microscope (Nikon Corporation, Tokyo, Japan) at ×100
magnification.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Each human placental tissue sample for RT-qPCR was
obtained from the center between placental edge and umbilical cord.
The size of the samples was ~1.5×1.5×1.5 cm3. Total RNA
was isolated with TRIzol reagent (Life Technologies; Thermo Fisher
Scientific, Inc., Waltham, MA, USA) according to the manufacturer's
protocol. Quantitative real-time PCR kits were purchased from
Takara Biotechnology Co., Ltd. (Dalian, China). Details of the RNA
extraction, cDNA conversion and RT-qPCR assays have been described
previously (23). To detect the
mRNA expression levels of miR-203 and VEGFA, RT-qPCR was performed
in 96-well reaction plates with a total volume of 10 µl, containing
the following: 1 µl cDNA template, 0.2 µl each primer, 3.6 µl
diethyl pyrocarbonate-H2O and 5 µl SYBR-Green dye using
the ABI Step One Plus™ Real-Time PCR System (Applied Biosystems;
Thermo Fisher Scientific, Inc.). The following primers were
designed using Primer Premier version 6.0 (Premier Biosoft
International, Palo Alto, CA, USA): miR-203 forward,
5′-GAATTCGGGGATCTGGGCGCAGGGGCC-3′ and reverse,
5′-CTCGAGCCGACCTGGAGCGCGGAGC-3′; VEGFA forward,
5′-ACCATGAACTTTCTGCTGTCTTGGGTGCAT-3′ and reverse,
5′-TCACCGCCTCGGCTTGTCACATCTGCAAGT-3′; GAPDH forward,
5′-TTGATGGCAACAATCTCCAC-3′ and reverse, 5′-CGTCCCGTAGACAAAATGGT-3′;
and 6 forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′. The PCR cycling parameters used were as
follows: Pre-denaturation at 95°C for 30 sec; 40 cycles of
denaturation at 95°C for 5 sec, annealing at 60°C for 10 sec and
extension at 72°C for 30 sec. GAPDH and U6 mRNA expression levels
were measured and applied as quantitative controls to normalize the
relative expression of VEGFA and miR-203, respectively. The
relative expression of each target amplicon was calculated using
the 2−ΔΔCq method relative to the endogenous control
(24).
Cell culture and transfection
The HTR-8/SVneo cell line was purchased from the
Cell Bank of the Chinese Academy of Sciences (Shanghai, China).
This cell line was established by the transfection of first
trimester villous explants of human primary trophoblasts. Cells
were cultured in RPMI 1640 (11875-093; Thermo Fisher Scientific,
Inc.) medium supplemented with 10% fetal bovine serum (FBS;
HyClone; GE Healthcare Life Sciences, Logan, UT, USA) without
antibiotics for 24 h. miR-203 mimic (Thermo Fisher Scientific,
Inc.), miR-203 inhibitor (anti-miR-203; Thermo Fisher Scientific,
Inc.) and a scramble control miRNA (Shanghai GenePharma Co., Ltd.,
Shanghai, China) were subsequently transfected into cells seeded on
a 6-well plate at 10,000 cells/well, as described previously
(21). Sequences were transfected
at a final concentration of 10 nM using Lipofectamine 3000
(Invitrogen; Thermo Fisher Scientific, Inc., Shanghai, China)
according to the manufacturer's protocol. This concentration was
determined based on dose-response experiments. Cells were further
cultured for the following cell proliferation and transwell assays
and subsequently harvested for RT-qPCR or western blot analysis 48
h after transfection. All experiments were conducted in
triplicate.
Cell proliferation
For the cell proliferation assay, transfected
HTR-8/SVneo cells were seeded into 96-well plates (5×104
cells/well). Cells were cultivated in RPMI 1640 medium for 3 days.
CCK-8 (10 µl; American Type Culture Collection; Manassas, VA, USA)
was subsequently added to each well and HTR-8/SVneo cells were
incubated for 2 h. Cell viability was measured by the absorbance at
a wavelength of 450 nm using an ELISA reader (Tecan Group, Ltd.,
Mannedorf, Switzerland).
Transwell assays
Cell migration and invasion was detected using
Transwell permeable supports (Corning Incorporated, Corning, NY,
USA). HTR-8/SVneo cells were transfected with the miR-203 mimic,
anti-miR-203 or the negative control (miRNC) for 24 h and
subsequently cultured in 200 µl of serum-free RPMI 1640 prior to
transfer onto the upper chambers of 24-well plates
(5×105 cells/well) with or without a Matrigel (BD
Biosciences, Franklin Lakes, NJ, USA) coating. The lower chamber
was supplemented with 800 µl of RPMI 1640 with 10% FBS. After 24 h
without Matrigel or 48 h with Matrigel, cells in the lower chamber
were fixed with absolute methanol for 20 min and stained with 0.1%
crystal violet solution (Sigma Aldrich; Merck KgaA, Darmstadt,
Germany) for 10 min at room temperature prior to cell counting
under a microscope.
Western blot analysis
Western blot analysis was performed as described
previously (21). Total protein
was extracted using radioimmunoprecipitation assay lysis buffer
(Beyotime Institute of Biotechnology) and Protease Inhibitor
Cocktail (Sigma Aldrich; Merck KgaA). A BCA Assay Kit (Thermo
Fisher Scientific, Inc.) was applied to detect the concentration of
extracted protein. A total of 30 µg was loaded onto each lane,
separated using 10% SDS-PAGE and blotted onto polyvinylidene
difluoride membranes (Merck KgaA). The membrane was subsequently
blocked in 5% non-fat milk for 1 h at room temperature and
incubated overnight at 4°C with the primary antibodies VEGFA
(1:1,000; ab1316, Abcam) and GAPDH (sc-47724; 1:3,000; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA). Following incubation with
horseradish peroxidase-conjugated anti-rabbit (A0208; 1:5,000;
Beyotime Institute of Biotechnology) and anti-mouse (A0216;
1:5,000; Beyotime Institute of Biotechnology) secondary antibodies
at room temperature for 45 min, antibody binding signals were
detected using the enhanced chemiluminescence luminol reagent
(PerkinElmer Inc., Waltham, MA, USA) and a Chemi-doc image analyzer
(Bio-Rad Laboratories, Inc., Hercules, CA, USA). The protein bands
were subsequently visualized with ImageJ software version 1.49
(National Institutes of Health, Bethesda, MD, USA). GAPDH was used
to normalize the band intensities.
Luciferase reporter assay
The luciferase reporter assay was conducted as
described previously (21). The
3′-untranslated region (UTR) sequence of VEGFA was amplified and
inserted into the luciferase reporter vector pGL3-enhancer (Promega
Corporation, Madison, WI, USA). The primers for VEGFA 3′-UTR were
5′-CAGCTCGAGTGTGTGAGTGGTTGACCTTCCT-3′ (forward) and
5′-CCGAAGCTTTCAGGGAGAGAGAGATTGGAAA-3′ (reverse). HTR-8/SVneo cells
were seeded into 24-well plates (5×105 cells/well) and
incubated for 24 h. Wild-type or mutant VEGFA 3′-UTR vectors were
subsequently co-transfected with the miR-203 mimic into HTR-8/SVneo
cells using Lipofectamine 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.). After incubation for 48 h, cells were lysed and
assayed for luciferase activity with the Luciferase Assay System
(Promega Corporation) according to the manufacturer's instructions.
Renilla-luciferase was used for normalization.
Statistical analysis
Data analysis was performed with the Stata 13.0
software (StataCorp LP, College Station, TX, USA), using the
Student's t-test, one way analysis of variance and the Scheffe post
hoc test with Spearman's correlation analysis. Results are
presented as the mean ± standard error. P<0.05 was considered to
indicate a statistically significant difference.
Results
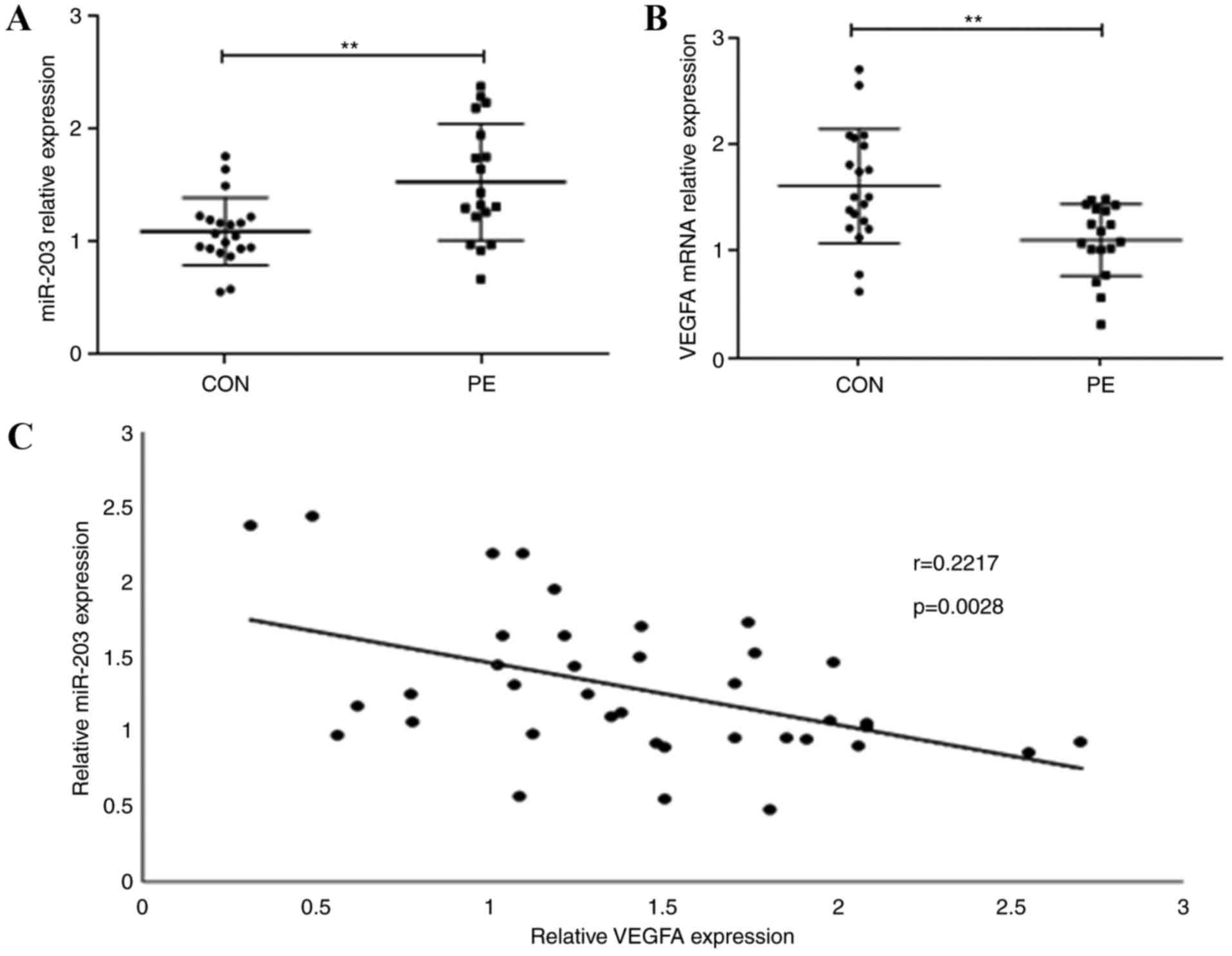
Expression levels of miR-203 and VEGFA
in human placenta
To evaluate the role of miR-203 and VEGFA in the
placenta development, miR-203 and VEGFA expression levels in all 38
placental tissue samples were measured using RT-qPCR. miR-203
expression levels were significantly increased in PE placentas
compared with normal placentas (Fig.
1A; P<0.01), whereas VEGFA expression levels were
significantly decreased (Fig. 1B;
P<0.01). The correlation between miR-203 and VEGFA expression
was further evaluated and a significant negative correlation
(r=0.2217; P=0.0028; Fig. 1C) was
identified. These results indicate that miR-203 upregulation may be
associated with VEGFA downregulation.
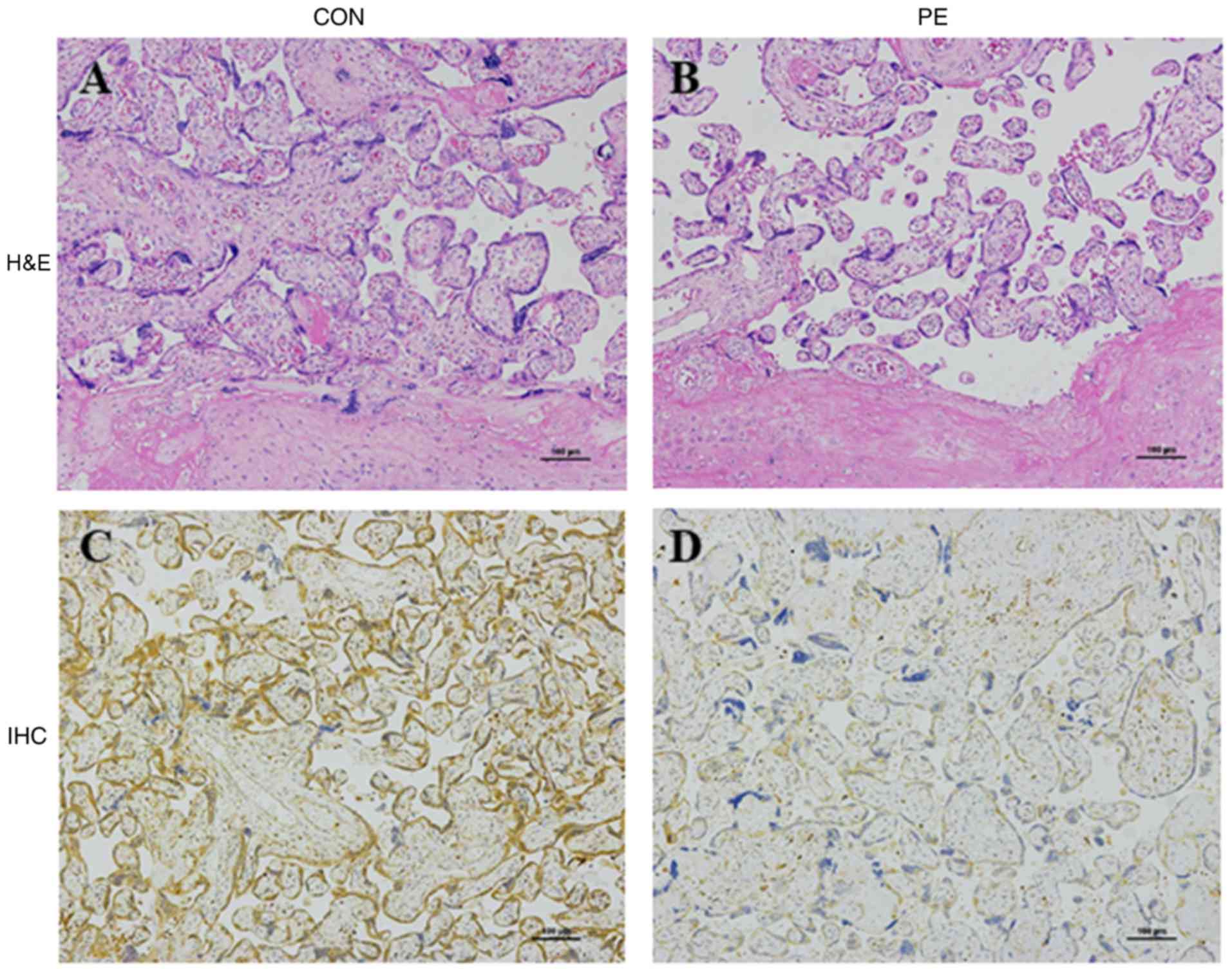
Placenta pathology and distribution of
VEGFA expression
To confirm morphological changes in the PE placenta,
H&E staining was applied to the normal and PE placenta tissue
samples. A disordered structure and a prominent decrease in
chorionic villi was observed in the PE placenta in comparison with
normal placenta tissue (Fig. 2A and
B). IHC revealed abundant VEGF expression in villous
trophoblasts and a lack of expression in the villous endothelium.
VEGFA expression levels in the PE placentas was clearly
downregulated in comparison with normal placenta samples (Fig. 2C and D). These results suggest that
VEGFA downregulation in the placenta may be associated with PE
occurrence.
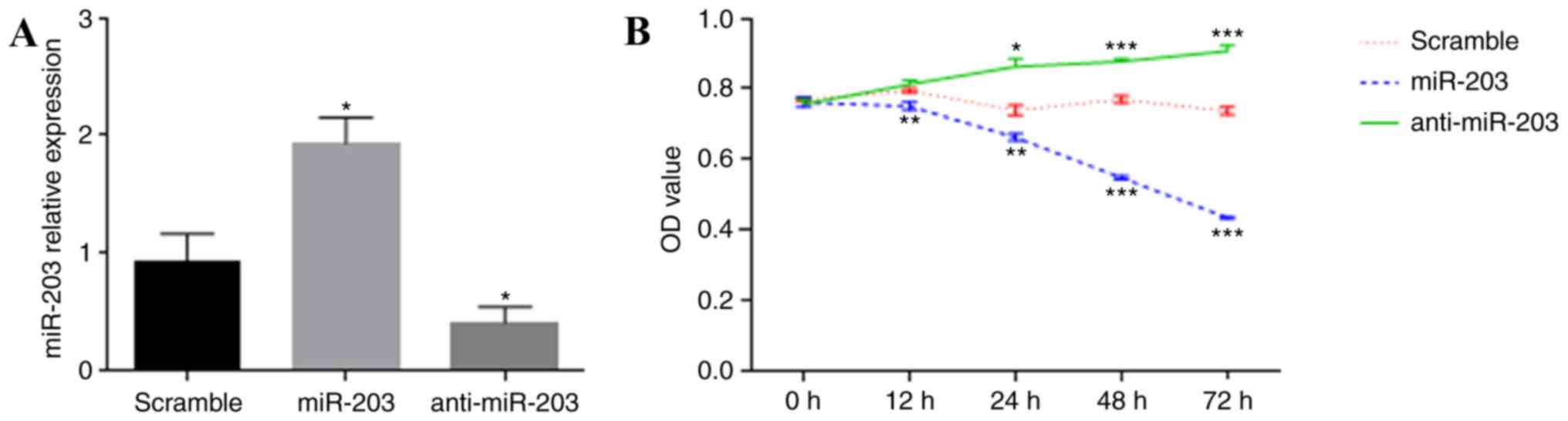
miR-203 affects the proliferation,
migration and invasion of HTR-8/SVneo cells
The HTR-8/SVneo cell line is established by the
transfection of human primary trophoblasts originating from first
trimester villous explants, with a gene encoding the simian virus
40 large T antigen, resulting in immortalization (25). To determine the effect of miR-203
on cell proliferation in the human placenta, HTR-8/SVneo cells were
transiently transfected with miR-203 mimics and anti-miR-203, and a
CCK-8 assay was performed to test the cell proliferation rate.
RT-qPCR was used to confirm the overexpression or inhibition of
miR-203 expression in HTR-8/SVneo cells (Fig. 3A). Overexpression of miR-203 was
demonstrated to reduce the proliferative capacity of HTR-8/SVneo
cells, whereas the inhibition of miR-203 expression resulted in a
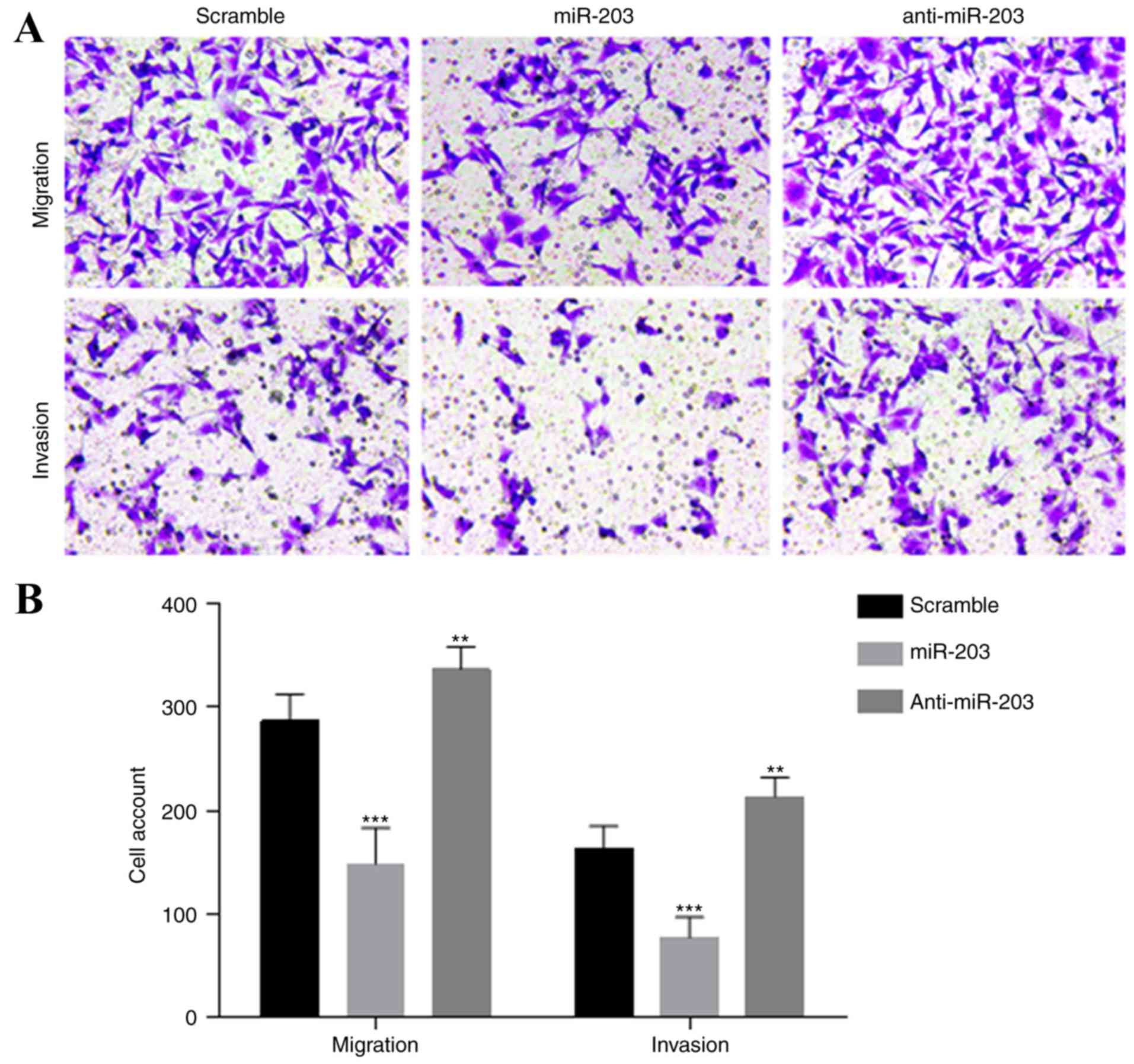
significant increase in cell proliferation (Fig. 3B). To assess the effects of miR-203
on cell migration and invasion, Transwell migration and invasion
assays were performed in the HTR-8/SVneo cells. Overexpression of
miR-203 significantly suppressed the migration and invasion
abilities of HTR-8/SVneo cells compared to the control cells
(Fig. 4A and B). The migration and
invasion capacity of HTR-8/SVneo cells in the anti-miR-203 group
was also significantly enhanced.
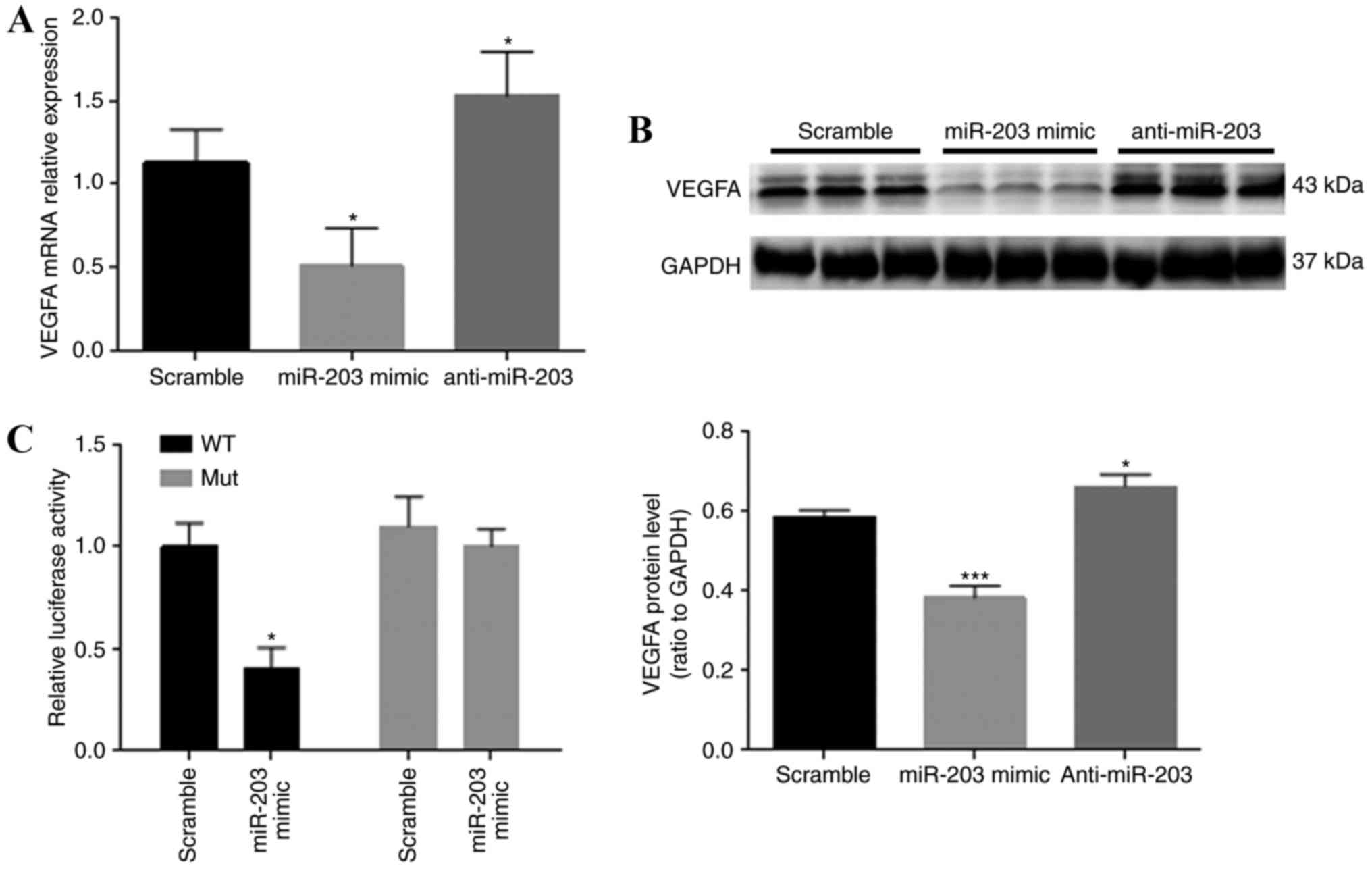
VEGFA is directly targeted by miR-203
in HTR-8/SVneo cells
To evaluate the possible effect of miR-203 on VEGFA
expression, VEGFA mRNA and protein expression levels in HTR-8/SVneo
cells were examined with RT-qPCR and western blot analysis,
respectively (Fig. 5A and B). Both
VEGFA mRNA and protein levels were significantly reduced in
HTR-8/SVneo cells transfected with the miR-203 mimics. By contrast,
the expression levels of VEGFA mRNA and protein in the anti-miR-203
group were significantly upregulated in comparison with the control
group. Results indicated that VEGFA mRNA expression levels of VEGFA
in the different groups was consistent with the protein expression
levels. The luciferase reporter assay was conducted to evaluate the
direct relationship between the VEGFA 3′-UTR and miR-203. miR-203
overexpression in HTR-8/SVneo cells significantly suppressed the
luciferase activity of the wild-type reporter. This inhibition was
eliminated when the target miR-203 was mutated (Fig. 5C).
Discussion
miRNAs are considered to be major contributors to
gene expression regulation and various biological processes. It is
well established that the proliferation, migration and invasion of
trophoblast cells into the pregnant uterus nourishes the developing
fetus. Abnormal trophoblast invasion or migration may lead to the
development of PE (18,26). The mechanisms of PE development are
unclear and are thought to be multifactorial. Following the
discovery of a potential association between the dysregulation of
placental miRNA expression and PE (27), other studies subsequently
demonstrated the expression of numerous miRNAs in the placenta and
a minimally overlapping pattern was identified (16). However, contradictory findings
concerning miRNA regulation have also been identified (28,29).
Thus, the stepwise mechanisms of miRNA promotion of PE development
remain incompletely understood. It is essential to elucidate the
precise contribution of miRNAs in PE development in order to
clarify the pathogenesis of this disease. To the best of our
knowledge, this is the first report to demonstrate the
participation of miR-203 in PE development through the placenta.
The data of the present study suggests that miR-203 has critical
involvement in PE development through the downregulation of VEGFA
expression and consequentially the proliferation, invasion and
migration ability of trophoblast cells in the human placenta.
A previous study confirmed the involvement of miRNA
clusters in the development of the placenta (16), particularly the miR-37-3 cluster.
It has been reported that expression of the miR-37-3 cluster
decreases during the development of the placenta (30). The results of the present study
also revealed a significant upregulation of miR-203 expression in
PE placentas in comparison with the normal placenta samples,
indicating that miR-203 upregulation may participate in the
development of PE. Studies have demonstrated the involvement of
miRNAs in the behaviors of endothelial and trophoblast cells via
the regulation of the expression of proteins, including
metalloproteinase 2, ephrin-B and VEGFA (30,31).
Thus, the present study investigated the expression of VEGFA in the
PE placenta and demonstrated a significant downregulation of VEGFA
expression in PE placental tissue in comparison with the normal
placental tissue with RT-qPCR and IHC, suggesting that VEGFA may
participate in PE, which is in line with the results of previous
research (32).
The negative effects of miR-203 on HTR-8/SVneo cells
was evaluated to further investigate the role of miR-203 in PE
development. The results revealed that the overexpression or
deletion of miR-203 markedly suppressed or promoted cell
proliferation, migration and invasion, respectively. Similar
results have been demonstrated in studies investigating miR-29b and
miR-519d, which also contribute to PE development by affecting the
proliferation, invasion and migration of trophoblast cells
(26,33,34).
Furthermore, several studies have also reported the ability of
miR-203 to function as a tumor suppressor (35–38).
Viticchiè et al (37)
reported that miR-203 can influence cell proliferation, migration
and invasion ability in prostate cancer. Notably, miR-203 has been
demonstrated to suppress tumor growth and angiogenesis by targeting
VEGFA (21,22). The present study demonstrated that
VEGFA mRNA and protein expression levels were significantly reduced
in HTR-8/SVneo cells transfected with miR-203 mimics and expression
was increased in the anti-miR-203 group. A luciferase reporter
assay confirmed the interaction between the 3′-UTR of VEGFA and
miR-203. Taken together, it can be speculated that miR-203
participates in PE through the downregulation of VEGFA expression.
Nevertheless, it is still necessary to delineate the numerous
pathways that miR-203 may affect and to investigate these pathways
to development possible therapeutic targets for the treatment of
PE.
In summary, the results of the present study suggest
that miR-203 suppressed trophoblast cell proliferation, migration
and invasion through the downregulation of VEGFA expression in the
placenta samples. A correlation between miR-203 and VEGFA was
identified, which may contribute to the onset and/or progression of
PE. The miR-203/VEGFA pathway may be an effective biomarker or a
viable drug target for therapeutic intervention in PE.
References
|
1
|
Redman CW and Sargent IL: Latest advances
in understanding preeclampsia. Science. 308:1592–1594. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Duley L: The global impact of
pre-eclampsia and eclampsia. Semin Perinatol. 33:130–137. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
ACOG Committee on Practice
Bulletins-Obstetrics, . ACOG practice bulletin. Diagnosis and
management of preeclampsia and eclampsia. Number 33, January 2002.
Obstet Gynecol. 99:159–167. 2002.PubMed/NCBI
|
|
4
|
McDonald SD, Best C and Lam K: The
recurrence risk of severe de novo pre-eclampsia in singleton
pregnancies: A population-based cohort. BJOG. 116:1578–1584. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Phipps E, Prasanna D, Brima W and Jim B:
Preeclampsia: Updates in pathogenesis, definitions, and guidelines.
Clin J Am Soc Nephrol. 11:1102–1113. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jia R, Li J, Rui C, Ji H, Ding H, Lu Y, De
W and Sun L: Comparative proteomic profile of the human umbilical
cord blood exosomes between normal and preeclampsia pregnancies
with high-resolution mass spectrometry. Cell Physiol Biochem.
36:2299–2306. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Steegers EA, von Dadelszen P, Duvekot JJ
and Pijnenborg R: Pre-eclampsia. Lancet. 376:631–644. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Shen Z, Wu Y, Chen X, Chang X, Zhou Q,
Zhou J, Ying H, Zheng J, Duan T and Wang K: Decreased maternal
serum 2-methoxyestradiol levels are associated with the development
of preeclampsia. Cell Physiol Biochem. 34:2189–2199. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Thompson LP, Pence L, Pinkas G, Song H and
Telugu BP: Placental hypoxia during early pregnancy causes maternal
hypertension and placental insufficiency in the hypoxic guinea pig
model. Biol Reprod. 95:1282016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li W, Huang H, Su J, Ji X, Zhang X, Zhang
Z and Wang H: miR-124 Acts as a tumor suppressor in glioblastoma
via the inhibition of signal transducer and activator of
transcription 3. Mol Neurobiol. 54:2555–2561. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
He Y, Zhao C, Liu Y, He Z, Zhang Z, Gao Y
and Jiang J: RETRACTED ARTICLE: miR-124 Functions as a Tumor
Suppressor via Targeting hCLOCK1 in Glioblastoma. Mol Neurobiol.
54:23752017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kagiya T: MicroRNAs and osteolytic bone
metastasis: The Roles of MicroRNAs in tumor-induced osteoclast
differentiation. J Clin Med. 4:1741–1752. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Park JH and Shin C: MicroRNA-directed
cleavage of targets: Mechanism and experimental approaches. BMB
Rep. 47:417–423. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang W, Feng L, Zhang H, Hachy S, Satohisa
S, Laurent LC, Parast M, Zheng J and Chen DB: Preeclampsia
up-regulates angiogenesis-associated microRNA (i.e., miR-17, −20a,
and −20b) that target ephrin-B2 and EPHB4 in human placenta. J Clin
Endocrinol Metab. 97:E1051–E1059. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Morales-Prieto DM, Ospina-Prieto S,
Chaiwangyen W, Schoenleben M and Markert UR: Pregnancy-associated
miRNA-clusters. J Reprod Immunol. 97:51–61. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jairajpuri DS and Almawi WY: MicroRNA
expression pattern in pre-eclampsia (Review). Mol Med Rep.
13:2351–2358. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Gunel T, Hosseini MK, Gumusoglu E,
Kisakesen HI, Benian A and Aydinli K: Expression profiling of
maternal plasma and placenta microRNAs in preeclamptic pregnancies
by microarray technology. Placenta. 52:77–85. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Jiang L, Long A, Tan L, Hong M, Wu J, Cai
L and Li Q: Elevated microRNA-520g in pre-eclampsia inhibits
migration and invasion of trophoblasts. Placenta. 51:70–75. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zygmunt M, Herr F, Münstedt K, Lang U and
Liang OD: Angiogenesis and vasculogenesis in pregnancy. Eur J
Obstet Gynecol Reprod Biol. 110 Suppl 1:S10–S18. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shiraishi S, Nakagawa K, Kinukawa N,
Nakano H and Sueishi K: Immunohistochemical localization of
vascular endothelial growth factor in the human placenta. Placenta.
17:111–121. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhu X, Er K, Mao C, Yan Q, Xu H, Zhang Y,
Zhu J, Cui F, Zhao W and Shi H: miR-203 suppresses tumor growth and
angiogenesis by targeting VEGFA in cervical cancer. Cell Physiol
Biochem. 32:64–73. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Xu L, Shen B, Chen T and Dong P: miR-203
is involved in the laryngeal carcinoma pathogenesis via targeting
VEGFA and Cox-2. Onco Targets Ther. 9:4629–4637. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Di Stefano V, Wang B, Parobchak N, Roche N
and Rosen T: RelB/p52-mediated NF-κB signaling alters histone
acetylation to increase the abundance of corticotropin-releasing
hormone in human placenta. Sci Signal. 8:ra852015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Graham CH, Hawley TS, Hawley RG,
MacDougall JR, Kerbel RS, Khoo N and Lala PK: Establishment and
characterization of first trimester human trophoblast cells with
extended lifespan. Exp Cell Res. 206:204–211. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Enquobahrie DA, Abetew DF, Sorensen TK,
Willoughby D, Chidambaram K and Williams MA: Placental microRNA
expression in pregnancies complicated by preeclampsia. Am J Obstet
Gynecol. 204:178.e12–21. 2011. View Article : Google Scholar
|
|
27
|
Pineles BL, Romero R, Montenegro D, Tarca
AL, Han YM, Kim YM, Draghici S, Espinoza J, Kusanovic JP, Mittal P,
et al: Distinct subsets of microRNAs are expressed differentially
in the human placentas of patients with preeclampsia. Am J Obstet
Gynecol. 196:261.e1–6. 2007. View Article : Google Scholar
|
|
28
|
Zhu XM, Han T, Sargent IL, Yin GW and Yao
YQ: Differential expression profile of microRNAs in human placentas
from preeclamptic pregnancies vs normal pregnancies. Am J Obstet
Gynecol. 200:661.e1–7. 2009. View Article : Google Scholar
|
|
29
|
Xu P, Zhao Y, Liu M, Wang Y, Wang H, Li
YX, Zhu X, Yao Y, Wang H, Qiao J, et al: Variations of microRNAs in
human placentas and plasma from preeclamptic pregnancy.
Hypertension. 63:1276–1284. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bentwich I, Avniel A, Karov Y, Aharonov R,
Gilad S, Barad O, Barzilai A, Einat P, Einav U, Meiri E, et al:
Identification of hundreds of conserved and nonconserved human
microRNAs. Nat Genet. 37:766–770. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hu TX, Wang G, Guo XJ, Sun QQ, He P, Gu H,
Huang Y, Gao L and Ni X: miR 20a,-20b and −200c are involved in
hydrogen sulfide stimulation of VEGF production in human placental
trophoblasts. Placenta. 39:101–110. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hong F, Li Y and Xu Y: Decreased placental
miR-126 expression and vascular endothelial growth factor levels in
patients with pre-eclampsia. J Int Med Res. 42:1243–1251. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li P, Guo W, Du L, Zhao J, Wang Y, Liu L,
Hu Y and Hou Y: microRNA-29b contributes to pre-eclampsia through
its effects on apoptosis, invasion and angiogenesis of trophoblast
cells. Clin Sci (Lond). 124:27–40. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Xie L and Sadovsky Y: The function of
miR-519d in cell migration, invasion, and proliferation suggests a
role in early placentation. Placenta. 48:34–37. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bo J, Yang G, Huo K, Jiang H, Zhang L, Liu
D and Huang Y: microRNA-203 suppresses bladder cancer development
by repressing bcl-w expression. FEBS J. 278:786–792. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang C, Zheng X, Shen C and Shi Y:
MicroRNA-203 suppresses cell proliferation and migration by
targeting BIRC5 and LASP1 in human triple-negative breast cancer
cells. J Exp Clin Cancer Res. 31:582012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Viticchiè G, Lena AM, Latina A, Formosa A,
Gregersen LH, Lund AH, Bernardini S, Mauriello A, Miano R, Spagnoli
LG, et al: miR-203 controls proliferation, migration and invasive
potential of prostate cancer cell lines. Cell Cycle. 10:1121–1131.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Furuta M, Kozaki KI, Tanaka S, Arii S,
Imoto I and Inazawa J: miR-124 and miR-203 are epigenetically
silenced tumor-suppressive microRNAs in hepatocellular carcinoma.
Carcinogenesis. 31:766–776. 2010. View Article : Google Scholar : PubMed/NCBI
|