Introduction
L-theanine (γ-glutamylethylamide;
C7H14N2O3) is a
water-soluble, edible, non-proteinogenic amino acid found
principally in Camellia sinensis, the source of green tea
(1). Previous studies in rats have
indicated that orally administered theanine is absorbed through the
intestinal tract, and hydrolyzed to glutamic acid and ethylamine in
the rat kidney (2,3). Theanine has been comprehensively
examined for its effects on mood-modulation and neuroprotection,
and for its lipid-lowering, antihypertensive, chemosensitizing and
hepatoprotective properties (4–8).
The pathogenesis of several diseases, including
cancer, is closely connected with abnormal apoptotic cell death
(9). The regulation of apoptosis
and associated mediators, which trigger or inhibit cell death, have
become the foundation for establishing therapeutic strategies in
cancer. Numerous studies have demonstrated that active components
from green tea, including catechins, theaflavins and polyphenols,
can induce apoptosis in several tumor cell lines (10–12).
Theanine, considered a novel biochemical modulator for
chemotherapeutics, can also suppress tumor cell invasion (13). It has been reported that theanine
suppressed the growth of A549 human non-small cell lung cancer and
K562 leukemic cell lines in a dose- and time-dependent manner
(14). Sugiyama and Sadzuka
(15) reported that theanine
significantly enhanced the inhibitory effect of doxorubicin (DOX)
on tumor growth in M5076 ovarian sarcoma-bearing mice, and
increased the concentration of DOX in tumor cells. Further
investigation has demonstrated that theanine increases the
antitumor efficacy of DOX via the inhibition of glutamate
transporters (16).
The aim of the present study was to investigate the
molecular mechanisms underlying the antitumor actions of theanine.
In the present study, theanine exhibited an enhanced apoptotic
efficacy on HepG2 cells when glutamine, an analogue of theanine,
was restricted in the cell culture medium. This suggested that
glutamine interfered with the action of theanine in the tumor
cells. It was also demonstrated that theanine exerted an apoptotic
effect on the HepG2 cells via mitochondrial pathways and the
activation of caspase-9. These findings indicated theanine is a
valuable antitumor component in food and in clinical treatment.
Materials and methods
Antibodies and reagents
Polyclonal rabbit antibodies against poly
(ADP-ribose) polymerase (PARP; cat no. 5625), caspase-3 (cat no.
9662), caspase-8 (cat no. 4790), caspase-9 (cat no. 9508),
cytochrome c (cat no. 4272) and β-actin (cat no. 4970) were
obtained from Cell Signaling Technology, Inc. (Beverly, MA, USA).
Monoclonal mouse antibody against cyclooxygenase (COX) IV (cat no.
ab16056), polyclonal rabbit antibodies against apoptosis-inducing
factor (AIF; cat no. ab32516) and endonuclease G (EndoG; cat no.
ab9647) were from Abcam (Cambridge, UK). L-theanine, L-glutamine
and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
(MTT) were purchased from Sigma-Aldrich; Merck KGaA (Darmstadt,
Germany). 4,6-diamidino-2-phenylindole was from Invitrogen; Thermo
Fisher Scientific, Inc. (Waltham, MA, USA). Z-LEHD-FMK, Z-IETD-FMK
and Z-VAD-FMK were provided by Merck KGaA.
Cells and culture conditions
Human hepatoblastoma HepG2 cells, human cervical
carcinoma HeLa cells, human hepatic L02 cells, human embryonic
kidney 293 (HEK293) cells and rat cardiac myoblast H9c2 cells were
purchased from the Cell Bank of Type Culture Collection of Chinese
Academy of Sciences (Shanghai, China). Cells were cultured in
Dulbecco's modified Eagle's medium (DMEM; Hyclone; GE Healthcare
Life Sciences, Logan, UT, USA) supplemented with 10% fetal bovine
serum (FBS; Hyclone; GE Healthcare Life Sciences), 100 U/ml
penicillin and 100 µg/ml streptomycin at 37°C with 5%
CO2. Glutamine restriction or deprivation were performed
by culturing cells with glutamine-containing (2.0 mM) or
glutamine-free DMEM (Hyclone; GE Healthcare Life Sciences)
supplemented with 10% FBS for 24 h.
Cell viability/toxicity assay
HepG2, HeLa, A549, L02 and HEK293 cells were seeded
into 96-well plates at a density of 5×103 cells/well 24
h prior to treatment. Following treatment with various
concentrations of L-theanine (0–600 µg/ml) at 37°C for 24 h, cell
viability was determined using an MTT assay. Briefly, 15 µl (5
mg/ml) MTT working solution were added into each well and,
following incubation at 37°C for 4 h, the MTT solution was removed
and 200 µl DMSO were added to dissolve the crystals. The absorbance
of each well at 570 nm was measured using a Synergy2 Multi-Mode
microplate reader (BioTek Instruments, Inc., Winooski, VT, USA).
The precise determination of cytotoxicity was achieved using a
CytoTox-Glo™ cytotoxicity assay (Promega Corporation, Madison, WI,
USA) to measure cell viability, according to the manufacturer's
protocol.
Terminal deoxynucleotidyl transferase
dUTP nick end labeling (TUNEL) assay
HepG2 cells were seeded at a density of
5×105 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 24 h, a TUNEL assay was performed using the Guava TUNEL
kit according to the manufacturer's protocol. The cells were added
to the Guava EasyCyte™ system, and the data were analyzed using
Guava TUNEL software version 2.0 (Guava Technologies, Inc.,
Hayward, CA, USA).
MultiCaspase apoptosis detection
HepG2 cells were seeded at a density of
5×105 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 24 h, the number of cells in the various stages of the
apoptotic pathway were determined via MultiCaspase apoptosis
detection (Guava Technologies, Inc.). The assay was performed
according to the manufacturer's protocol and the stained cells were
analyzed using the Guava EasyCyte™ flow cytometer. A
fluorochrome-conjugated inhibitor of caspases, termed
sulforhodamine-valyl-alanyl-aspartyl-fluoromethylketone, and the
cell impermeant DNA dye 7-aminoactinomycin (7-AAD) were used to
distinguish between non-apoptotic living cells, early/mid apoptotic
stage cells, late apoptotic/dying cells and dead cells. Data were
analyzed using the Guava TUNEL Software version 2.0 (Guava
Technologies, Inc.).
Western blot analysis
HepG2 cells were seeded at a density of
5×105 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 12 h, cells were lysed for 30 min on ice in a lysis
buffer containing 135 mM NaCl, 20 mM Tris (pH 7.5), 25 mM
β-glycerophoshate, 2 mM EDTA, 2 mM DTT, 2 mM sodium pyrophosphate,
10% glycerol, 1% Triton X-100, 10 mM NaF, 1 mM sodium orthovanadate
and 1 mM PMSF, supplemented with complete protease inhibitor
cocktail (Roche Applied Science, Indianapolis, IN, USA). The
lysates were centrifuged (15,000 × g) at 4°C for 15 min. Equal
amounts of extracted protein samples (20 µg) were denatured in SDS,
separated by 12% SDS-PAGE, and transferred onto nitrocellulose
membranes (GE Healthcare Life Sciences). Membranes were blocked
with 5 % non-fat dry milk in Tris-buffered saline with 0.1%
Tween-20 for 2 h at room temperature. Subsequently, membranes were
incubated with the following primary antibodies overnight at 4°C:
Anti-caspase-3 (1:1,000), anti-PARP (1:1,000), anti-caspase-9
(1:1,000), anti-caspase-8 (1:1,000), anti-COX IV (1:1,000),
anti-cytochrome c (1:1,000), anti-AIF (1:1,000), anti-EndoG
(1:1,000) and anti-β-actin (1:1,000). The protein bands were
visualized using the LI-COR Odyssey infrared imaging system,
according to the manufacturer's protocol, using an IRDye800
fluorophore-conjugated goat anti-rabbit IgG (cat no. 926-32211;
1:5,000; LI-COR Biosciences, Lincoln, NE, USA) or anti-mouse IgG
(cat no. 926-32210; 1:5,000; LI-COR Biosciences) secondary antibody
in the dark at room temperature for 1 h. Blots were semi-quantified
by densitometric analysis using the LI-COR Odyssey analytical
software version 2.1 (LI-COR Biosciences).
Caspase activity measurement
HepG2 cells were seeded at a density of
5×105 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 12 h, caspase-8 and caspase-9 activity was measured
using the Caspase-Glo® 8 assay and
Caspase-Glo® 9 assay, respectively (Promega
Corporation). The assays were performed according to the
manufacturer's protocol. The luminescence was measured on a
Synergy2 Multi-Mode microplate reader (BioTek Instruments,
Inc.).
Measurement of mitochondrial membrane
potential (MMP)
HepG2 cells were seeded at a density of
5×105 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 12 h, the MMP of the HepG2 cells was assessed using the
Guava EasyCyte™ MitoPotential™ kit. In this assay, JC-1 cationic
dye is used to evaluate alterations in MMP. The cell impermeant
dye, 7-AAD, was used to simultaneously monitor cell membrane
permeability alterations in apoptosis. The stained cells were
analyzed using the Guava EasyCyte™ flow cytometer (Guava
Technologies, Inc.).
Mitochondrial isolation
HepG2 cells were seeded at a density of
2×107 cells/well 24 h prior to treatment. Following
treatment with various concentrations of L-theanine (0–600 µg/ml)
at 37°C for 12 h, intact mitochondria were separated from cytosolic
components of the HepG2 cells for further protein analysis using a
mitochondria isolation kit (cat no. 89874; Thermo Fisher
Scientific, Inc.). Dounce homogenization and differential
centrifugation were performed according to the manufacturer's
protocol.
Statistical analysis
All experimental data are expressed as the mean ±
standard deviation of three independent experiments in each group.
The statistical significance of the differences between the
experimental groups was assessed by Student's t-test for pair-wise
comparisons and one-way analysis of variance followed by Tukey's
post-hoc test for multiple comparisons. Statistical analysis was
performed using the SPPS software version 17.0 (SPSS, Inc.,
Chicago, IL, USA). P<0.05 was considered to indicate a
statistically significant difference.
Results
Theanine-induced cancer cell death is
potentiated by acute glutamine deprivation
To determine an anticancer activity of theanine, the
present study detected the effect of theanine on tumor and normal
cell viability. As shown in Fig.
1A, theanine significantly decreased the viability of the HepG2
and HeLa cells in a dose-dependent manner between from 75 and 600
µg/ml. Theanine treatment at the concentration of 600 µg/ml,
resulted in decreases in viability of ~45 and 25% in the HepG2 and
HeLa cells, respectively, compared with the untreated control
cells. By contrast, reductions in viability of <10% were
observed in the H9c2, L02 and HEK293 cells. These data, together
with previous findings (17)
indicated that the anticancer activity of theanine was cell-type
specific, and suggested that theanine possessed anticancer activity
with almost no harmful effect on normal cells.
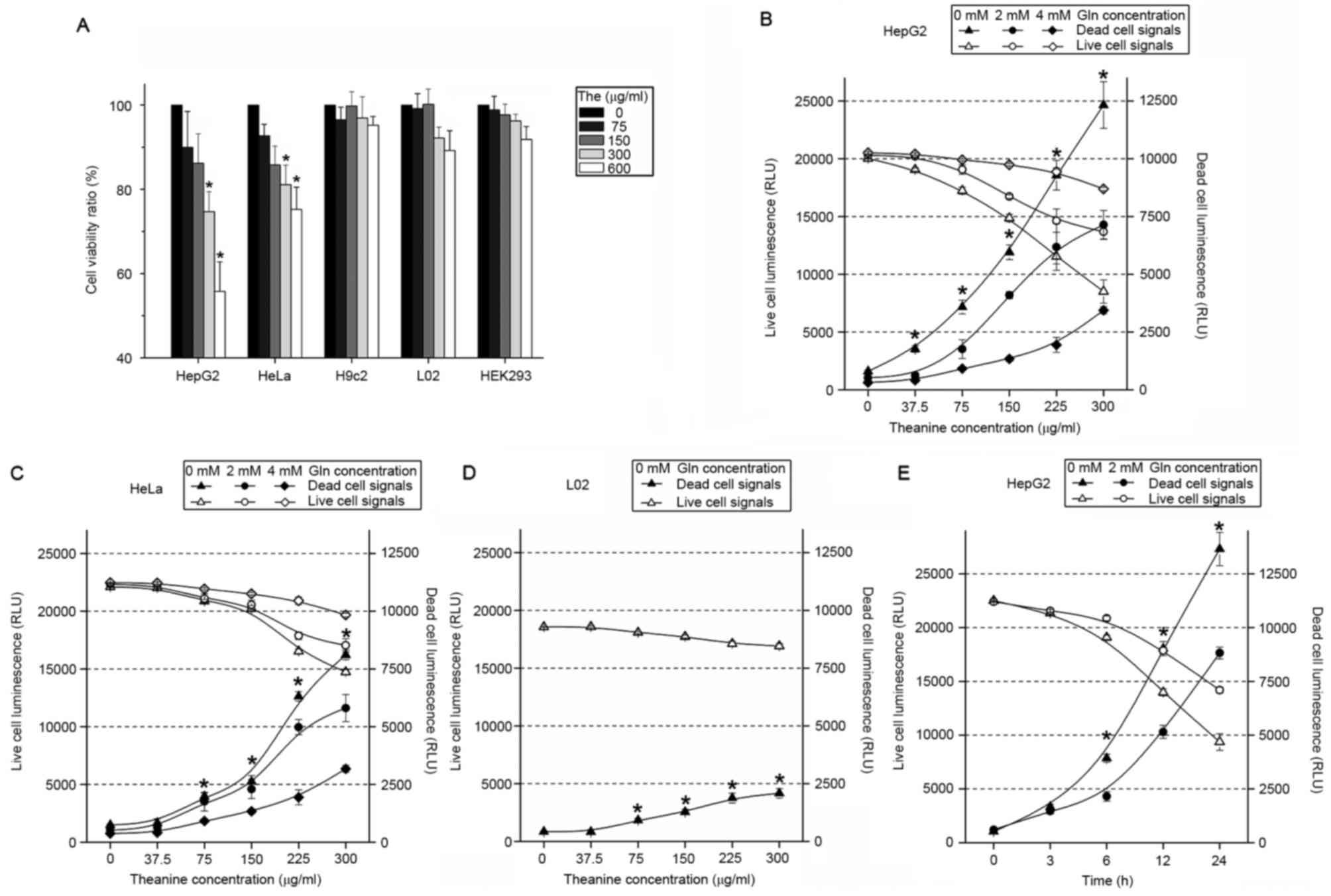 | Figure 1.Effects of glutamine concentration on
theanine-induced cytotoxicity. (A) HepG2, HeLa, H9c2, L02 and
HEK293 cells were incubated with various concentrations of
theanine, respectively. Cell viability was measured using a
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
assay. Data shown are representative of three independent
experiments. *P<0.05, compared with the untreated control. (B-E)
HepG2, HeLa or L02 cells in glutamine-free or glutamine-restricted
(2 or 4 mM) medium were treated with various concentrations of
theanine for 24 h respectively. HepG2 cells in glutamine-free or
glutamine-restricted (2 mM) media were treated with 300 µg/ml
theanine for the indicated time. In all cases, theanine
cytotoxicity was determined by a luminescence-based method as
described. Each value is a mean ± SD of three replicates. Gln,
glutamine; The, theanine. |
As theanine and glutamine are analogues, and are
mutually exchangeable due to their similar structure, the present
subsequently investigated whether glutamine present in the cell
culture medium interfered with the cytotoxic effect of theanine on
cancer cells. As it was noted that complete DMEM commonly contains
4 mM glutamine, the present study used 2 mM glutamine-containing
DMEM or glutamine-free DMEM for incubation of the HepG2, HeLa and
L02 cells in order to achieve acute glutamine restriction or
deprivation. The cells incubated under the acute glutamine
restriction or deprivation conditions were treated with 0–300 µg/ml
theanine for 24 h. As shown in Fig.
1B, theanine led to apparent dose-dependent cell death of HepG2
cells, dependent on glutamine concentration in the culture medium.
In addition to the decreased glutamine concentration,
theanine-induced cell death increased. A similar effect of
glutamine on theanine-induced cytotoxicity was also detected in the
HeLa cells (Fig. 1C). However,
neither glutamine restriction nor deprivation altered the effect of
theanine on the L02 cells (Fig.
1D). Furthermore, under acute glutamine deprivation, theanine
induced HepG2 cell death in a time-dependent manner (Fig. 1E). These data indicated that
glutamine prevented theanine-induced cancer cell death.
Theanine induces HepG2 cell apoptosis
under acute glutamine deprivation
To determine whether theanine-induced cytotoxicity
was due to apoptosis, a series of classical apoptosis detection
experiments were performed. The results of the TUNEL assay, using
flow cytometric analysis, indicated DNA damage in the
theanine-treated HepG2 cells (Fig.
2A). As the caspase family is pivotal in apoptosis (18), the present study performed
MultiCaspase apoptosis detection for evaluating theanine-induced
HepG2 cell death. It was found that theanine induced the activation
of caspases when the HepG2 cells were cultured in the glutamine
restricted and deprived media (Fig.
2B). During apoptosis, typical molecular events occur,
including the activation of initiators and effectors of caspases,
and cleavage of PARP (18). The
present study examined the protein levels of PARP, procaspase-8 and
procaspase-3 using western blot analysis (Fig. 2C). The data demonstrated that the
cleavage of PARP and procaspase-3 was increased in the
theanine-treated HepG2 cells exposed to acute glutamine
deprivation, whereas procaspase-8 remained unchanged. Taken
together, these results indicated that, under acute glutamine
deprivation, theanine induced typical apoptotic cell death in the
HepG2 cells.
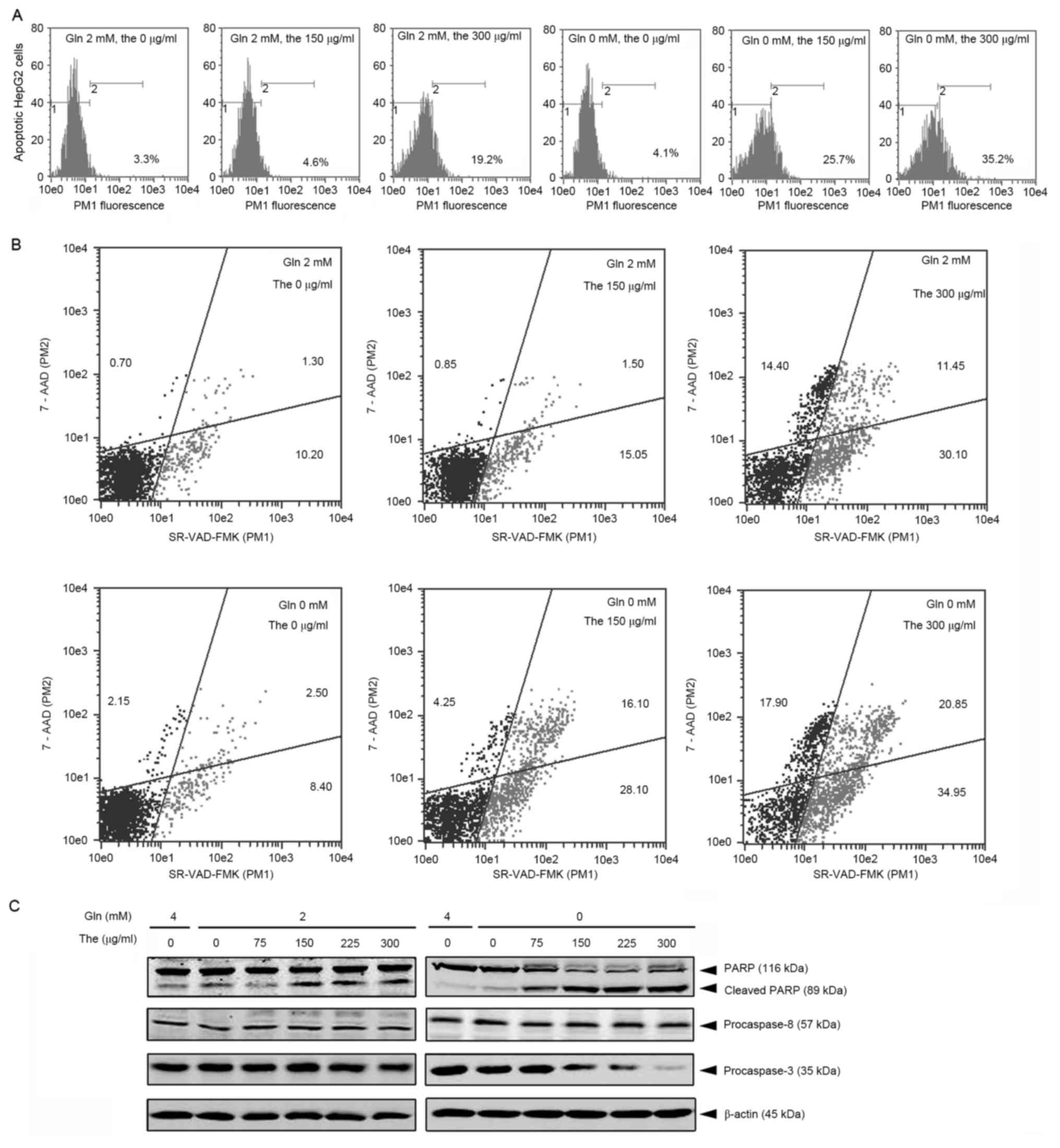 | Figure 2.Apoptosis of HepG2 cells is induced by
theanine. HepG2 cells in glutamine-free or glutamine-restriction (2
mM) media were treated with 0, 150 or 300 µg/ml theanine for 24 h.
Percentages of apoptotic cells were assessed using (A) TUNEL
analysis and (B) multicaspase detection, respectively. For TUNEL
analysis, the average apoptotic ratio of each group is shown. For
multicaspase detection, four quadrants denote non-apoptotic live
cells (bottom-left), early/mid apoptotic stage cells
(bottom-right), late apoptotic/dying cells (upper-right), and dead
cells (upper-left). Data shown are representative of three
independent experiments. (C) HepG2 cells in glutamine-free or
glutamine-containing (2 or 4 mM) medium were treated with 300 µg/ml
theanine for 24 h, and cell lysates were prepared. The cleavage of
PARP, caspase-8 and caspase-3 were determined using western blot
analysis. Expression of β-actin was determined to confirm equal
protein loading. Data shown are representative of three independent
experiments. Gln, glutamine; The, theanine; TUNEL, terminal
deoxynucleotidyl transferase dUTP nick end labeling; PARP, poly
(ADP-ribose) polymerase. |
Theanine treatment activates caspase-9
and caspase-3
The results described above showed that caspase
cascades were involved in theanine-induced apoptosis. Two pathways
have been considered to lead to caspase activation during
apoptosis, caspase-8 and caspase-9, being major extrinsic and
intrinsic initiators of caspase cascades, respectively. As shown in
Fig. 2C, theanine stimulated the
cleavage of caspase-3, but did not activate caspase-8. Therefore,
the present study determined changes in the protein levels of
procaspase-8, procaspase-9 and procaspase-3 in the HepG2 cells
following theanine treatment under glutamine deprivation
conditions. The results from the western blot analyses indicated
that procaspase-9 and procaspase-3 were cleaved following theanine
treatment, whereas caspase-8 was intact (Fig. 3A). The activity of caspase-8 was
then compared with caspase-9 using a luminescence-based assay. The
data showed that, following 12 h incubation with theanine, the
HepG2 cells exhibited 2.5 times higher caspase-9 activity, but no
change in caspase-8 activity (Fig.
3B). Caspase inhibitors were also used to investigate the role
of caspase-9 and caspase-8 in theanine-induced apoptosis. As shown
in Fig. 3C, the caspase-8
inhibitor Z-IETD-FMK failed to prevent theanine-induced apoptosis,
whereas the caspase-9 inhibitor Z-LEHD-FMK and pan caspase
inhibitor Z-VAD-FMK attenuated HepG2 cell death. Further
experiments showed that Z-LEHD-FMK and Z-VAD-FMK inhibited
theanine-induced PARP cleavage and caspase-3 activation, whereas
Z-IETD-FMK exerted minimal effect on the activation of PARP and
caspase-3 (Fig. 3D). Taken
together, the data demonstrated that caspase-9, but not caspase-8,
was essential in the theanine-induced apoptosis. It was also noted
that the pan caspase inhibitor Z-VAD-FMK did not completely prevent
theanine-induced apoptosis, suggesting that caspase-independent
cell death existed in this process.
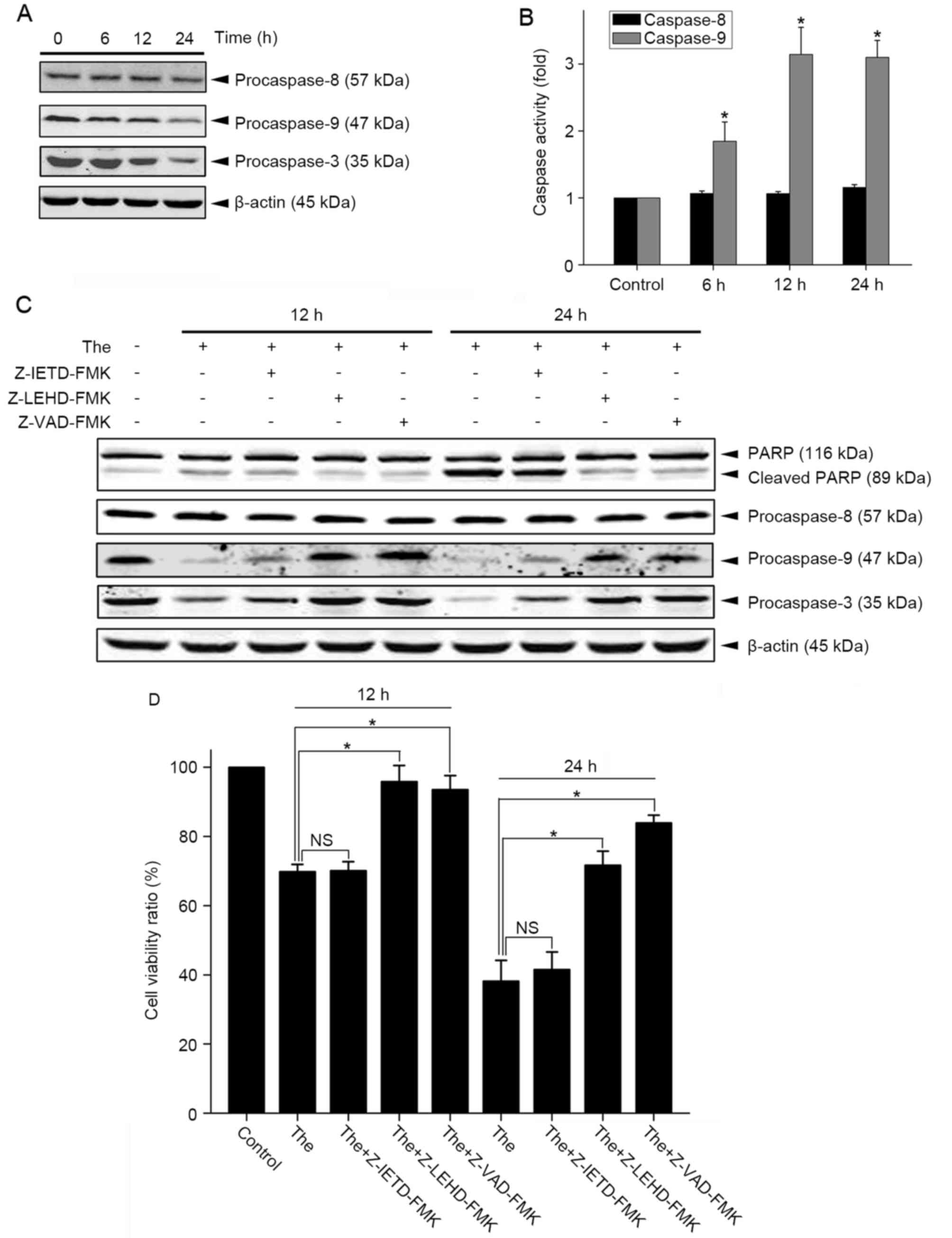 | Figure 3.Caspase pathway activation in
theanine-induced apoptosis. (A) HepG2 cells in glutamine-free media
were treated with 300 µg/ml theanine for the indicated durations,
respectively. Cell lysates were prepared, and the levels of
caspase-8, caspase-9 and caspase-3 were assessed using western blot
analysis. Expression of β-actin was determined to confirm equal
protein loading. Data shown are representative of three independent
experiments. (B) Following treatment with 300 µg/ml theanine for
the indicated time, activities of caspase-8 and caspase-9 in the
HepG2 cells were detected using a luminescence-based method. Data
shown are representative of three independent experiments.
*P<0.05, compared with the untreated control. (C) HepG2 cells
were exposed to 300 µg/ml theanine for 12 or 24 h, accompanied with
caspase-8 inhibitor Z-IETD-FMK, caspase-9 inhibitor Z-LEHD-FMK or
pan-caspase inhibitor Z-VAD-FMK treatment, respectively, as
indicated. Levels of caspase-8, caspase-9 and caspase-3 were
determined using western blot analysis. Expression of β-actin was
assessed to confirm equal protein loading. Data shown are
representative of two independent experiments. (D) Cell viability
was measured using a 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl
tetrazolium bromide assay. Cell viability without treatment was
taken as 100%. Data are presented as the mean ± standard deviation
of three independent experiments. *P<0.05, compared with
untreated cells. PARP, poly (ADP-ribose) polymerase; The, theanine;
NS, not significant. |
Involvement of the mitochondrial
pathway in theanine-induced apoptosis
In the intrinsic pathway of apoptosis, MMP is the
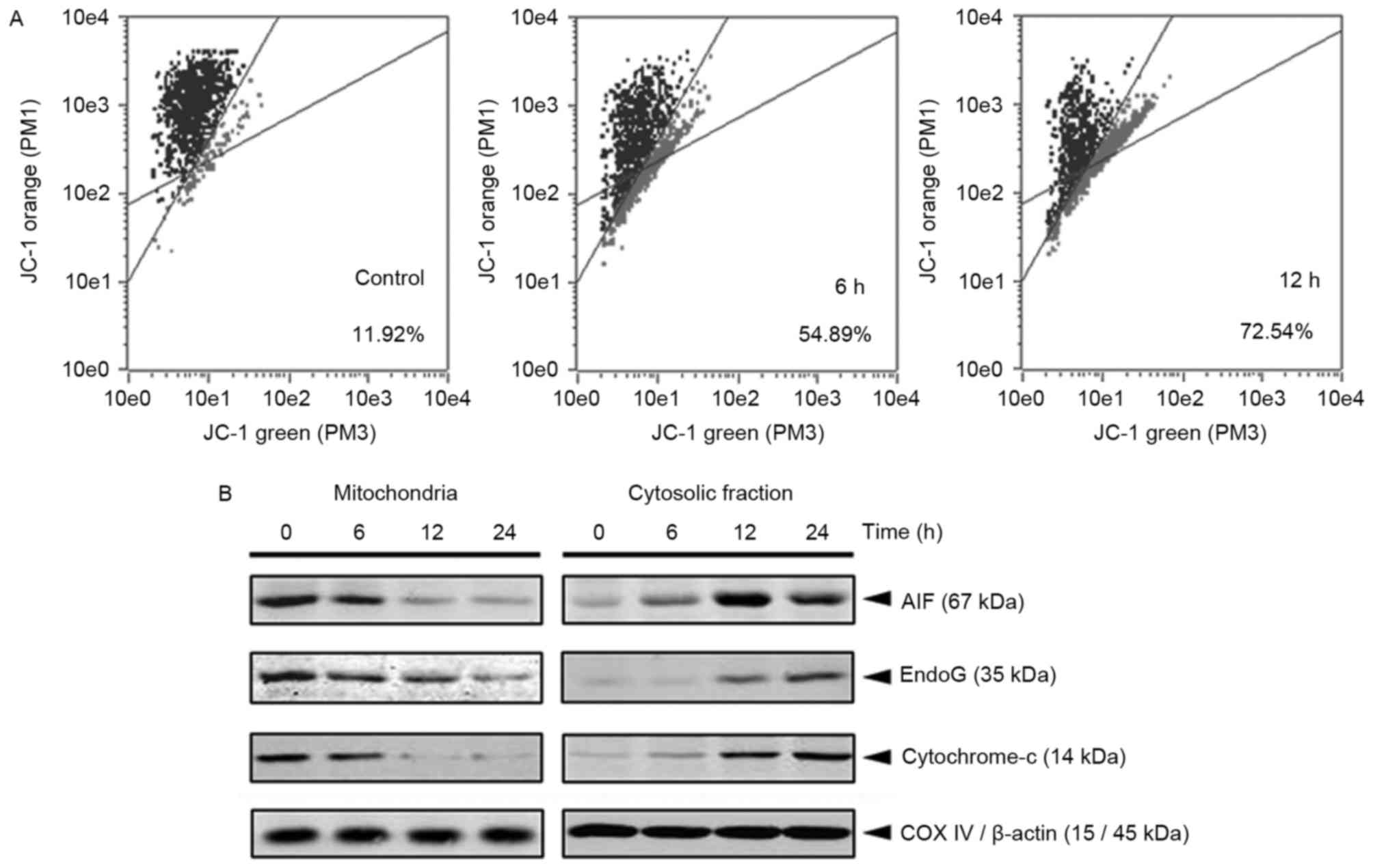
critical event responsible for caspase activation (19). The cationic dye, JC-1, was used to
evaluate alterations in MMP during theanine-induced HepG2 cell
apoptosis. The results revealed ~73% depolarized MMP was detected
in the cells treated with 300 µg/ml theanine for 12 h (Fig. 4A). Caspase-9 activation has been
reported to be associated with the mitochondrial pathway in
apoptosis. Once cytochrome c is released from mitochondria, it
binds to cytosolic protein adapter molecule apoptosis protease
activating factor 1, which recruits and cleaves the inactive
procaspase-9 thereby promoting it into the active form. As the
above data indicated that caspase-9 was important in
theanine-induced apoptosis, the present study detected the release
of cytochrome c from mitochondria of the HepG2 cells following
theanine treatment. As expected, a time-dependent increase in
cytosolic cytochrome c induced by theanine was detected (Fig. 4B). It has been recognized that the
release of the caspase-independent death effectors AIF and EndoG
from the mitochondria is a hallmark of caspase-independent cell
death. The results of the present study further demonstrated that
cytosolic AIF and EndoG were increased in the theanine-treated
HepG2 cells (Fig. 4B). Taken
together, these data suggested that caspase-9 and the
caspase-independent mitochondrial pathway were involved in
theanine-induced apoptosis.
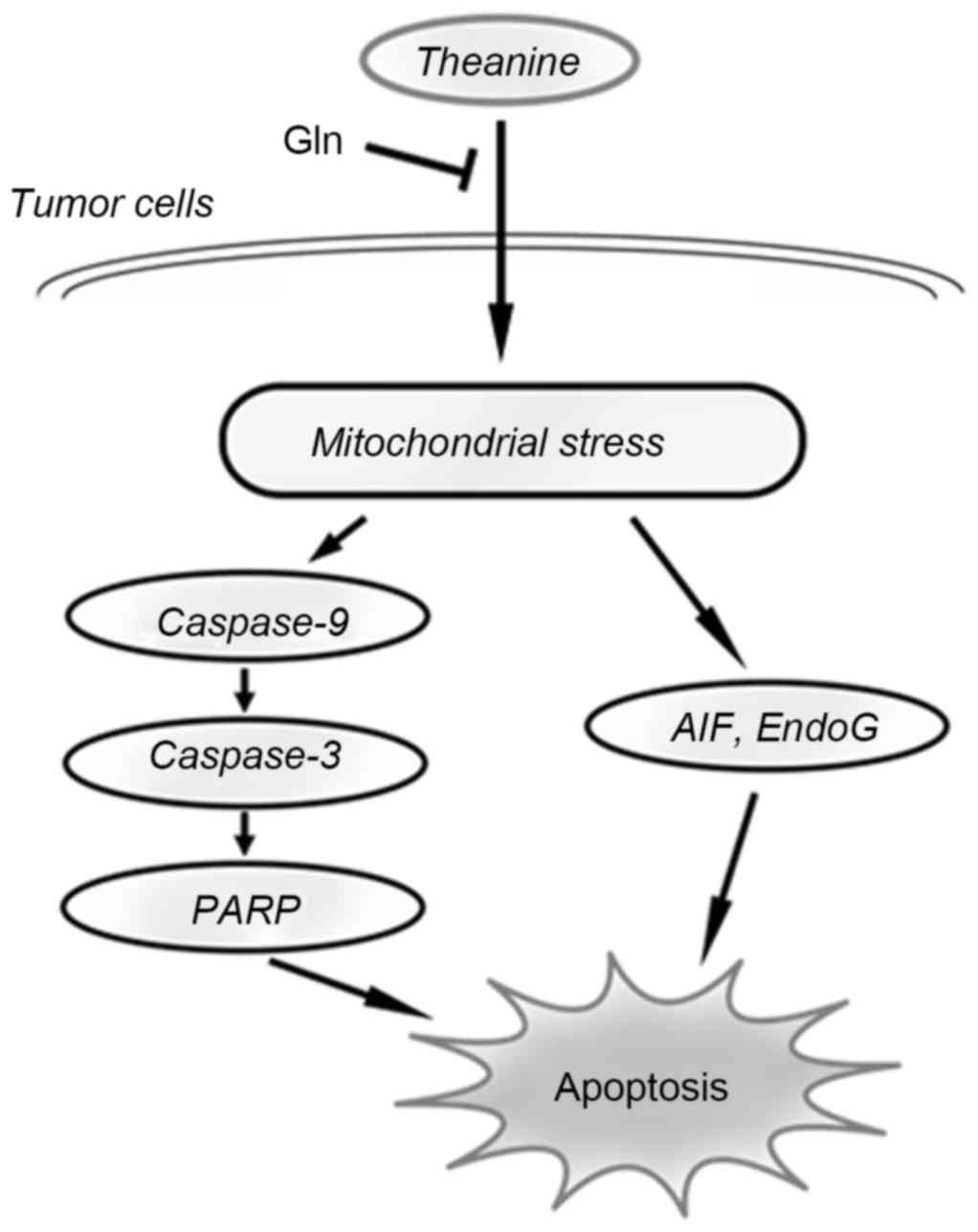
Discussion
Liver cancer has long been considered a therapeutic
challenge due to its cytotoxic drug resistant nature and associated
liver function disorder, which reduces the safety of a number of
conventional chemotherapeutic agents (20). In addition to cytotoxic drugs,
there are certain chemicals from traditional Chinese medicine or
dietary foods, which target apoptosis and ate being investigated
for cancer treatment (9). In the
present study, it was demonstrated that theanine, a free amino acid
from green tea, induced human hepatocellular carcinoma HepG2 cell
death, and this effect was significantly enhanced by acute
glutamine restriction or deprivation. It was subsequently found
that theanine induced HepG2 cell death through apoptosis.
In the present study, it was found that a
concentration >300 µg/ml of theanine was required for inducing
significant cell apoptosis when the cells were cultured in DMEM
supplemented with 4 mM glutamine (Fig.
1A). It is reasonable to suggest that glutamine antagonized the
apoptotic effect of theanine due to their similar structures. The
present study then imposed acute restriction/deprivation of
glutamine, and marked enhancement in the efficacy of theanine in
apoptosis was induced. This finding is consistent with those of a
previous report, which demonstrated that the accumulation of
theanine in the brain synaptosomal fraction was markedly inhibited
by glutamine, whereas glutamine accumulation was inhibited by
theanine and vice versa (16).
Therefore, it was hypothesized that glutamine interfered with the
action of theanine in tumor cells. In addition, the results also
showed that the acute restriction/deprivation of glutamine alone
within 24 h did not lead to nutrient withdrawal stress towards the
HepG2 and HeLa cells (Fig. 1B and
C).
In various clinical situations, appropriate
exogenous glutamine supply is considered to contribute beneficially
to reduce risks of high-dose chemotherapy (21). Similarly, theanine has been shown
to protect normal cells from damage by chemotherapeutic drugs
(22). As theanine is a glutamate
derivative, it may be enzymatically metabolized to glutamate in the
liver, and consequently be utilized as a constituent of glutathione
(GSH). Therefore, theanine can attenuate the DOX-induced adverse
reactions involved in oxidative damage, due to increase in
glutamate and the recovery of GSH levels in normal tissues
(15,22). However, tumor cells rapidly consume
glutamine and use it to supply metabolic pathways, which support
cell growth and proliferation. Glutamine is also a multipurpose
nutrient, feeding several additional pathways, which enhance the
ability of cells to communicate with each other, and to cope with
stress by oncogenic signaling and the tumor microenvironment
(23). Therefore, oncologists
often avoid supplying glutamine to tumor-bearing host to avoid any
potential risk.
In the present study, the cytotoxicity of theanine
towards five cell lines, including two tumor cell lines and three
types normal cell lines, was examined. Consistent with the above,
the apparent cytotoxicity of theanine towards HepG2 and HeLa cells
was detected when glutamine was restricted in the medium, and the
higher cytotoxicity of theanine was exhibited in HepG2 cells.
Therefore, theanine may be a valuable therapeutic reagent when
supplementation of glutamine is refused to avoid interference
during treatment.
The understanding of apoptosis has provided the
basis for novel targeted therapies, which can induce cancer cell
death or sensitize cells to established cytotoxic agents and
radiation therapy. In the present study, it was found that
caspase-9, but not caspase-8, was involved in theanine-induced
HepG2 cell apoptosis. As is commonly known, apoptosis occurs
through two main pathways, which includes the extrinsic
(cytoplasmic) pathway triggered through death receptor and
intrinsic (mitochondrial) pathway, which leads to the release of
cytochrome c from the mitochondria and activation of the
death signal. Caspase-8 and caspase-9 are important in the
extrinsic and intrinsic apoptotic pathways, respectively (18). To clarify the underlying mechanism
of theanine-induced apoptosis, the present study measured the
activation of caspase-3, MMP, release of cytochrome c, and
protein levels of AIF and EndoG. These experiments demonstrated
that theanine induced HepG2 cell apoptosis by activating the
intrinsic apoptotic pathway. Sadzuka et al (22) demonstrated that glutaminase reacted
with theanine to generate glutamate in vitro, and that the
metabolism of theanine mediated by glutaminase, and the increase of
glutamate-mediated GSH is important for theanine anticancer action
in vivo. HepG2 cells accumulate higher levels of glutamine,
compared with normal cells, and are more sensitive to glutamine
withdrawal from the medium (24).
When extrinsic glutamine is restricted, it may be easier for
theanine to disturb glutamine/glutamate metabolism and trigger the
generation of inappropriate metabolites, followed by activation of
the intrinsic apoptosis pathway. In mammals, glutaminase exists in
the matrix of mitochondria, therefore, it was expected that
theanine may alter glutamine/glutamate metabolism at the site of
mitochondria. Taken together, the results of the present study
indicated the function of theanine on cancer cells (Fig. 5). Further investigations are
required to demonstrate the intracellular interference between
theanine and glutamine, and the signal transduction networks
underlying the theanine-induced apoptosis.
In conclusion, the present study showed that
theanine induced HepG2 cell death under acute glutamine
restriction/deprivation conditions by triggering
caspase-9-dependent and caspase-independent mitochondrial pathways.
It was found that glutamine restriction and/or deprivation
amplified the apoptotic action of theanine in HepG2 cells. These
observations shed light on the potential use of theanine in the
therapeutic treatment of liver cancer and support the view that
glutamine supplementation requires cautious use during
chemotherapy.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Nature Science Foundation of China (grant no. 81471557), the
Jiangsu Perspective Study (grant no. BY2013001-03), the Natural
Science Research Project of Anhui Provincial Education Department
(grant no. KJ2017A908) and the Science Research Project of Chuzhou
City Vocation College (grant no. 2016zk02).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
YX was responsible for the experimental design and
implementation. PB was a major contributor in writing the
manuscript and conducted some of the experiments. QW was
responsible for image processing and data analysis. YZ was
responsible for cell culture and performed the experiments. ZY and
LL interpreted the data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Liang YR, Liu C, Xiang LP and Zheng XQ:
Health benefits of theanine in green tea: A review. Trop J
Pharmaceut Res. 14:1943–1949. 2015. View Article : Google Scholar
|
|
2
|
Terashima T, Takido J and Yokogoshi H:
Time-dependent changes of amino acids in the serum, liver, brain
and urine of rats administered with theanine. Biosci Biotechnol
Biochem. 63:615–618. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Juneja LR, Chu DC, Okubo T, Nagato Y and
Yokogoshi H: L-theanine-a unique amino acid of green tea and its
relaxation effect in humans. Trends Food Sci Technol. 10:199–204.
1999. View Article : Google Scholar
|
|
4
|
Gomez-Ramirez M, Kelly SP, Montesi JL and
Foxe JJ: The effects of L-theanine on alpha-band oscillatory brain
activity during a visuo-spatial attention task. Brain Topogr.
22:44–51. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cho HS, Kim S, Lee SY, Park JA, Kim SJ and
Chun HS: Protective effect of the green tea component, L-theanine
on environmental toxins-induced neuronal cell death.
Neurotoxicology. 29:656–662. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bozzetti F, Biganzoli L, Gavazzi C,
Cappuzzo F, Carnaghi C, Buzzoni R, Dibartolomeo M and Baietta E:
Glutamine supplementation in cancer patients receiving
chemotherapy: A double-blind randomized study. Nutrition.
13:748–751. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Yokogoshi H and Kobayashi M: Hypotensive
effect of gamma-glutamylmethylamide in spontaneously hypertensive
rats. Life Sci. 62:1065–1068. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang G, Ye X, Ji D, Zhang H, Sun F, Shang
C, Zhang Y, Wu E, Wang F, Wu F, et al: Inhibition of lung tumor
growth by targeting EGFR/VEGFR-Akt/NF-κB pathways with novel
theanine derivatives. Oncotarget. 5:8528–8543. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Martin KR: Targeting apoptosis with
dietary bioactive agents. Exp Biol Med (Maywood). 231:117–129.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nakazato T, Ito K, Miyakawa Y, Kinjo K,
Yamada T, Hozumi N, Ikeda Y and Kizaki M: Catechin, a green tea
component, rapidly induces apoptosis of myeloid leukemic cells via
modulation of reactive oxygen species production in vitro and
inhibits tumor growth in vivo. Haematologica. 90:317–325.
2005.PubMed/NCBI
|
|
11
|
Yang GY, Liao J, Kim K, Yurkow EJ and Yang
CS: Inhibition of growth and induction of apoptosis in human cancer
cell lines by tea polyphenols. Carcinogenesis. 19:611–616. 1998.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kundu T, Dey S, Roy M, Siddiqi M and
Bhattacharya RK: Induction of apoptosis in human leukemia cells by
black tea and its polyphenol theaflavin. Cancer Lett. 230:111–121.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhang G, Miura Y and Yagasaki K:
Inhibitory effects of theanine and sera from theanine-fed rats on
receptor-mediated cancer cell invasion beneath mesothelial-cell
monolayers. Cytotechnology. 36:195–200. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Q, Duan H, Luan J, Yagasaki K and
Zhang G: Effects of theanine on growth of human lung cancer and
leukemia cells as well as migration and invasion of human lung
cancer cells. Cytotechnology. 59:211–217. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sugiyama T and Sadzuka Y: Theanine, a
specific glutamate derivative in green tea, reduces the adverse
reactions of doxorubicin by changing the glutathione level. Cancer
Lett. 212:177–184. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kakuda T, Hinoi E, Abe A, Nozawa A, Ogura
M and Yoneda Y: Theanine, an ingredient of green tea, inhibits
[3H]glutamine transport in neurons and astroglia in rat brain. J
Neurosci Res. 86:1846–1856. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Friedman M, Mackey BE, Kim HJ, Lee IS, Lee
KR, Lee SU, Kozukue E and Kozukue N: Structure-activity
relationships of tea compounds against human cancer cells. J Agric
Food Chem. 55:243–253. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shi Y: Mechanisms of caspase activation
and inhibition during apoptosis. Mol Cell. 9:459–470. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kroemer G, Galluzzi L and Brenner C:
Mitochondrial membrane permeabilization in cell death. Physiol Rev.
87:99–163. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kerr SH and Kerr DJ: Novel treatments for
hepatocellular cancer. Cancer Lett. 286:114–120. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kuhn KS, Muscaritoli M, Wischmeyer P and
Stehle P: Glutamine as indispensable nutrient in oncology:
Experimental and clinical evidence. Eur J Nutr. 49:197–210. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sadzuka Y, Sugiyama T, Nagamine M, Umegaki
K and Sonobe T: Efficacy of theanine is connected with theanine
metabolism by any enzyme, not only drug metabolizing enzymes. Food
Chem Toxicol. 44:286–292. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shanware NP, Mullen AR, DeBerardinis RJ
and Abraham RT: Glutamine: Pleiotropic roles in tumor growth and
stress resistance. J Mol Med (Berl). 89:229–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Tardito S, Chiu M, Uggeri J, Zerbini A, Da
Ros F, Dall'Asta V, Missale G and Bussolati O: L-Asparaginase and
inhibitors of glutamine synthetase disclose glutamine addiction of
β-catenin-mutated human hepatocellular carcinoma cells. Curr Cancer
Drug Targets. 11:929–943. 2011. View Article : Google Scholar : PubMed/NCBI
|