Introduction
Retinoic acid-inducible gene-I (RIG-I)-like
receptors (RLRs) are pattern-recognition receptors that recognize
pathogen-associated molecular patterns. RLRs, which include RIG-I
and melanoma differentiation-associated gene 5, function as
cytosolic virus sensors and serve an important role in
mitochondrial-mediated antiviral immune systems (1). Once these receptors sense an RNA
virus invasion, they interact with mitochondrial
antiviral-signaling (MAVS) protein, an adaptor protein located on
the mitochondrial membrane, to induce antiviral immune responses,
including the production of type I interferons (2,3).
Furthermore, previous studies demonstrated that RLR activation
induces apoptosis in various cancer cells, including in lung cancer
(4–6).
Certain previous studies attempted to enhance
RLR-mediated antitumor and cytotoxic effects against cancer cells
(6–8). Yuan et al (6) examined the 5′end of ppp-RNA, which is
an agonist of RIG-I, and vascular endothelial growth factor (VEGF),
which promotes angiogenesis in cancer, and produced a
5′-triphosphate-small interfering (si)RNA targeting VEGF.
Consequently, the VEGF-targeting 5′-triphosphate-siRNA produced
multiple antitumor effects against human non-small cell lung cancer
(NSCLC) cells through not only the induction of RIG-I-mediated
apoptosis and antitumor immunity; however, additionally through
inhibition of tumor angiogenesis via VEGF knockdown. In another
approach, the cytotoxic effect of cotreatment with an RLR agonist
and ionizing radiation (IR) in human NSCLC was recently
investigated. RLR agonist-induced cell death was demonstrated to be
enhanced by IR, and the RLR agonist exhibited a radiosensitizing
effect (9). However, although it
was demonstrated that this cell death, which included apoptosis,
occurred in a caspase-dependent manner, the molecular mechanisms of
the effect remain unclear.
Apoptotic mechanisms may be broadly divided into two
pathways: The extrinsic pathway via death receptors and the
intrinsic pathway via mitochondria. In the extrinsic pathway,
activation of death receptors, including Fas cell surface death
receptor (Fas), induces apoptosis via activation of
caspase-8/caspase-3 (10). In
contrast, in the intrinsic pathway, cytochrome c is released from
mitochondria by external stressors, including DNA injury and
oxidative stress, resulting in the activation of
caspase-9/caspase-3 and the subsequent induction of apoptosis
(11). Furthermore, it is known
that apoptosis inhibitor proteins, including X-linked inhibitor of
apoptosis protein (XIAP), are involved in these pathways and
negatively regulate apoptosis by inhibiting caspase activation
(12).
It is additionally known that IR may activate the
extrinsic and intrinsic apoptosis pathways. Takahashi et al
(13) demonstrated that X-ray
irradiation induces caspase-8-mediated apoptosis accompanied with
Fas upregulation in a human leukemia cell line. Furthermore, Kim
et al (14) observed that
exposure of the HeLa cervical cancer cell line to radiation induced
the loss of the mitochondrial membrane potential, and the release
of cytochrome c and apoptosis-inducing factor from mitochondria,
which resulted in apoptotic cell death. Therefore, it is possible
that cotreatment with the RLR agonist and IR may effectively
activate the extrinsic and/or intrinsic apoptotic pathways.
To clarify the mechanism by which cotreatment with
an RLR agonist and IR induces apoptosis, the pathways involved in
the enhancement of apoptosis by cotreatment with RLR agonist and IR
in the A549 human NSCLC cell line were investigated.
Materials and methods
Reagents
Ca2+- and Mg2+-free PBS(−), propidium iodide (PI),
RNaseA and dimethyl sulfoxide (DMSO) were all purchased from
Sigma-Aldrich (Merck KGaA, Darmstadt, Germany).
Poly(I:C)-HMW/LyoVec™ [Poly(I:C)-HMW], which is a
complex of a synthetic double-stranded RNA analogue poly(I:C) and a
transfection reagent (LyoVec™), was purchased from
InvivoGen (San Diego, CA, USA). Fluorescein isothiocyanate
(FITC)-conjugated anti-human cluster of differentiation (CD)95
(Fas) antibody (cat. no. 305606) was purchased from BioLegend, Inc.
(San Diego, CA, USA). FITC-conjugated anti-mouse immunoglobulin
(Ig)G1 antibody (cat. no. A07795) was purchased from Beckman
Coulter, Inc. (Brea, CA, USA). Anti-rabbit horseradish peroxidase
(HRP)-conjugated IgG and anti-mouse HRP-conjugated IgG secondary
antibodies, anti-XIAP monoclonal (cat. no. 2045), anti-caspase-3
monoclonal (cat. no. 9661), anti-caspase-8 monoclonal (cat. no.
9746), anti-caspase-9 monoclonal (cat. no. 9502) and anti-β-actin
monoclonal (cat. no. 4967) primary antibodies were purchased from
Cell Signaling Technology Inc., (Danvers, MA, USA). Z-Val-Ala-Asp
(OMe)-CH2F (Z-VAD-fmk), Ac-Ile-Glu-Thr-Asp-H (aldehyde;
AC-IETD-CHO) and Ac-Leu-Glu-His-Asp-H (aldehyde; AC-LEHD-CHO)
peptides were purchased from Peptide Institute, Inc. (Osaka,
Japan). Ambion's Silencer® Select Pre-designed siRNA
against the gene encoding Fas (cat. no. s1506) was purchased from
Thermo Fisher Scientific, Inc. (Waltham, MA, USA).
Cell culture and treatment
A549 cells were purchased from RIKEN BioResource
Center (Tsukuba, Japan). Cells were maintained in Dulbecco's
modified Eagle's medium (Sigma-Aldrich; Merck KGaA) supplemented
with 1% penicillin/streptomycin (Gibco; Thermo Fisher Scientific,
Inc.) and 10% heat-inactivated fetal bovine serum (Japan Bioserum
Co., Ltd., Fukuyuma, Japan) at 37°C in a humidified atmosphere of
5% CO2.
Cells were seeded in 35-mm (6×104 cells)
or 60-mm culture dishes (1.2×105 cells; IWAKI Glass Co.
Ltd., Chiba, Japan) and cultured overnight to allow them to adhere
to the dish. The subsequent day, cells were treated with 250 ng/ml
RLR agonist Poly(I:C)-HMW for 72 h at 37°C in a humidified
atmosphere of 5% CO2. The cultured cells were harvested
using 0.1% trypsin-ethylenediaminetetraacetic acid (Gibco; Thermo
Fisher Scientific, Inc.) for subsequent analyses.
In experiments investigating the involvement of each
caspase in apoptosis induction, cells were pre-incubated with 50 µM
Z-VAD-fmk (a pan-caspase inhibitor), 100 µM AC-IETD-CHO (a
caspase-8 inhibitor) and 100 µM AC-LEHD-CHO (a caspase-9 inhibitor)
for 1 h at 37°C in a humidified atmosphere of 5% CO2,
prior to treatment with 250 ng/ml Poly(I:C)-HMW. As a vehicle
control, cells treated with the same amount of DMSO (0.2%) were
prepared.
siRNA transfection
A549 cells were transfected with siRNA targeting Fas
(cat. no. s1506; Ambion; Thermo Fisher Scientific, Inc.) using
Lipofectamine® RNAiMAX (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol. The
sense sequence for Fas was 5′-GGAAGACUGUUACUACAGUTT-3′.
Silencer® Select Negative No. 1 Control siRNA (cat. no.
AM4611, Ambion; Thermo Fisher Scientific, Inc.) was used as a
control (sequence not available). The final concentration of the
siRNAs was 5 nM. Following incubation for 24 h, transfected cells
were harvested and used for subsequent analyses.
In vitro irradiation
Cells were irradiated (150 kVp; 20 mA; 0.5-mm Al
filter and 0.3-mm Cu filter) using an X-ray generator (MBR-1520R-3;
Hitachi, Ltd., Tokyo, Japan) at a distance of 45 cm from focus and
at a dose rate of 0.99–1.02 Gy/min, and the duration of exposure
was ~4 min.
SDS-PAGE and western blotting
Harvested cells were lysed in 1XLaemmli sample
buffer (Bio-Rad Laboratories, Inc., Hercules, CA, USA) containing
2.5% 2-mercaptoethanol and the resulting cell lysates were boiled
for 10 min. Protein concentrations of the lysates were determined
using the XL-Bradford assay kit (APRO Science Corporation,
Tokushima, Japan) and a SmartSpec™ plus
spectrophotometer (Bio-Rad Laboratories, Inc.). SDS-PAGE and
western blotting were performed as previously described (15). Membranes were blocked in
Tris-buffered saline containing 0.1% Tween-20 and 5% non-fat
skimmed milk for 1 h at room temperature. The following primary
antibodies were used: Anti-XIAP (1:3,000), anti-caspase-3
(1:3,000), anti-caspase-8 (1:3,000), anti-caspase-9 (1:3,000) and
anti-β-actin (1:4,000). The following secondary antibodies were
used: HRP-conjugated anti-rabbit IgG (1:10,000) and HRP-conjugated
anti-rabbit IgG (1:10,000). Antigens were visualized using the
Enhanced Chemiluminescent (ECL) Prime Western Blotting Detection
System (GE Healthcare Life Sciences, Little Chalfont, UK) for the
detection of caspase-3, caspase-8 and caspase-9, or
Clarity™ Western ECL Substrate (Bio-Rad Laboratories,
Inc.) for the detection of XIAP and β-actin. Blots were stripped
using a Stripping Solution (Wako Pure Chemical Industries, Ltd.,
Osaka, Japan). Quantification of the bands was performed using
ImageJ software ver.1.51K (National Institutes of Health, Bethesda,
MD, USA) and the relative XIAP/β-actin ratio computed and
presented.
Cell cycle analysis
Cell cycle analysis was performed, as previously
described (16). Harvested cells
were fixed overnight in ice-cold 70% ethanol at −20°C. Fixed cells
were washed with and subsequently suspended in PBS(−) and treated
with 20 µg/ml RNase A for 30 min at 37°C. Following treatment,
cells were resuspended in PBS(−) containing 20 µg/ml PI and
incubated in the dark for 30 min. Finally, cells were passed
through a cell strainer (BD Falcon; BD Biosciences, Franklin Lakes,
NJ, USA) and analyzed using a flow cytometer (Cytomics FC500 with
CXP software ver.2; Beckman-Coulter, Inc.).
Analysis of cell surface Fas
expression
The analysis of cell surface Fas expression was
performed as previously described (17). Harvested cells were washed once
with PBS(−) and subsequently stained with FITC-conjugated
anti-human CD95 (Fas) antibodies (1:10) or FITC-conjugated mouse
IgG1 isotype control for 30 min at 4°C in the dark. Following
staining, cells were washed and analyzed using flow cytometry.
Prior to analysis, 20 µg/ml PI was added to cell suspensions to
discriminate dead cells from viable cells. Following gating PI
negative cells, the fluorescence intensity of CD95 staining was
analyzed.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from A549 cells using the
RNeasy® Plus Mini kit (Qiagen, Inc., Valencia, CA, USA)
and quantified using a NanoDrop spectrophotometer (Thermo Fisher
Scientific, Inc.). cDNA templates were synthesized from 1 µg RNA
using the iScript cDNA synthesis kit (Bio-Rad Laboratories, Inc.),
according to the manufacturer's protocol. The reaction conditions
were 5 min at 25°C, followed by 30 min at 42°C and 5 min at 85°C.
RT-qPCR was performed, as previously described (18). Power SYBR®-Green Master
Mix (Applied Biosystems; Thermo Fisher Scientific, Inc.) and a Step
One Plus™ system (Applied Biosystems; Thermo Fisher
Scientific, Inc.) were used with typical amplification parameters
(95°C for 10 min, followed by 40 cycles of 95°C for 15 sec and 60°C
for 1 min). Differences in gene expression relative to
non-irradiated controls were determined using a standard curve
based method (19). β-actin was
used as a housekeeping gene. β-actin primer sequences were as
previously described (20).
Primers for XIAP were sense, 5′-TTTTGGGACATGGATATACTCAGGT-3′ and
antisense, 5′-TGAAAGCACTTTACTTTATCACCTTC-3′.
Statistical analysis
Data are presented as the mean ± standard error of
the mean of at least three independent experiments. Comparisons
between the control and experimental groups were performed using a
two-sided Student's t-test or a two-sided Mann-Whitney's U-test
depending on the data distribution. Multiple data were analyzed
using one-way analysis of variance followed by the Tukey-Kramer
test. P<0.05 was considered to indicate a statistically
significant difference. All statistical analyses were performed
using Excel 2016 software (Microsoft Corporation, Redmond, WA, USA)
and with Statcel 4 (OMS Publishing, Inc., Tokyo, Japan).
Results
Poly(I:C)-HMW induces apoptosis
through caspase-8 and caspase-9 activation
The authors previously demonstrated that
Poly(I:C)-HMW activates caspase-3 and that a pan-caspase inhibitor
(Z-VAD-fmk) suppresses Poly(I:C)-HMW-induced cell death, including
apoptosis in A549 cells (9).
Although these results suggested that Poly(I:C)-HMW induces
apoptosis in a caspase-dependent manner, the pathways involved in
Poly(I:C)-HMW-induced apoptosis remained unclear and were
investigated in the present study.
As demonstrated in Fig.
1A, Poly(I:C)-HMW activated caspase-8 and caspase-9 in addition
to caspase-3. Whether caspase-8 and caspase-9 are involved in
apoptosis induction by Poly(I:C)-HMW was examined using caspase
inhibitors. The sub-G1 cell cycle peak, which contains cells with
fragmented DNA and is a hallmark of apoptosis (21), was analyzed. As demonstrated in
Fig. 1B, Poly(I:C)-HMW
significantly increased the sub-G1 population compared with the
DMSO control and treatment with Z-VAD-fmk significantly repressed
this effect (P<0.01). Furthermore, caspase-8 inhibitor (IETD)
and caspase-9 inhibitor (LEHD) significantly decreased
Poly(I:C)-HMW-induced apoptosis (P<0.05; Fig. 1B). These results suggested that
Poly(I:C)-HMW induces apoptosis in A549 cells through caspase-8 and
caspase-9 activation. However, the inhibitory effect of the
combination of caspase-8 inhibitor and caspase-9 on the
Poly(I:C)-HMW-induced sub-G1 population was similar to that of each
treatment alone (Fig. 1B).
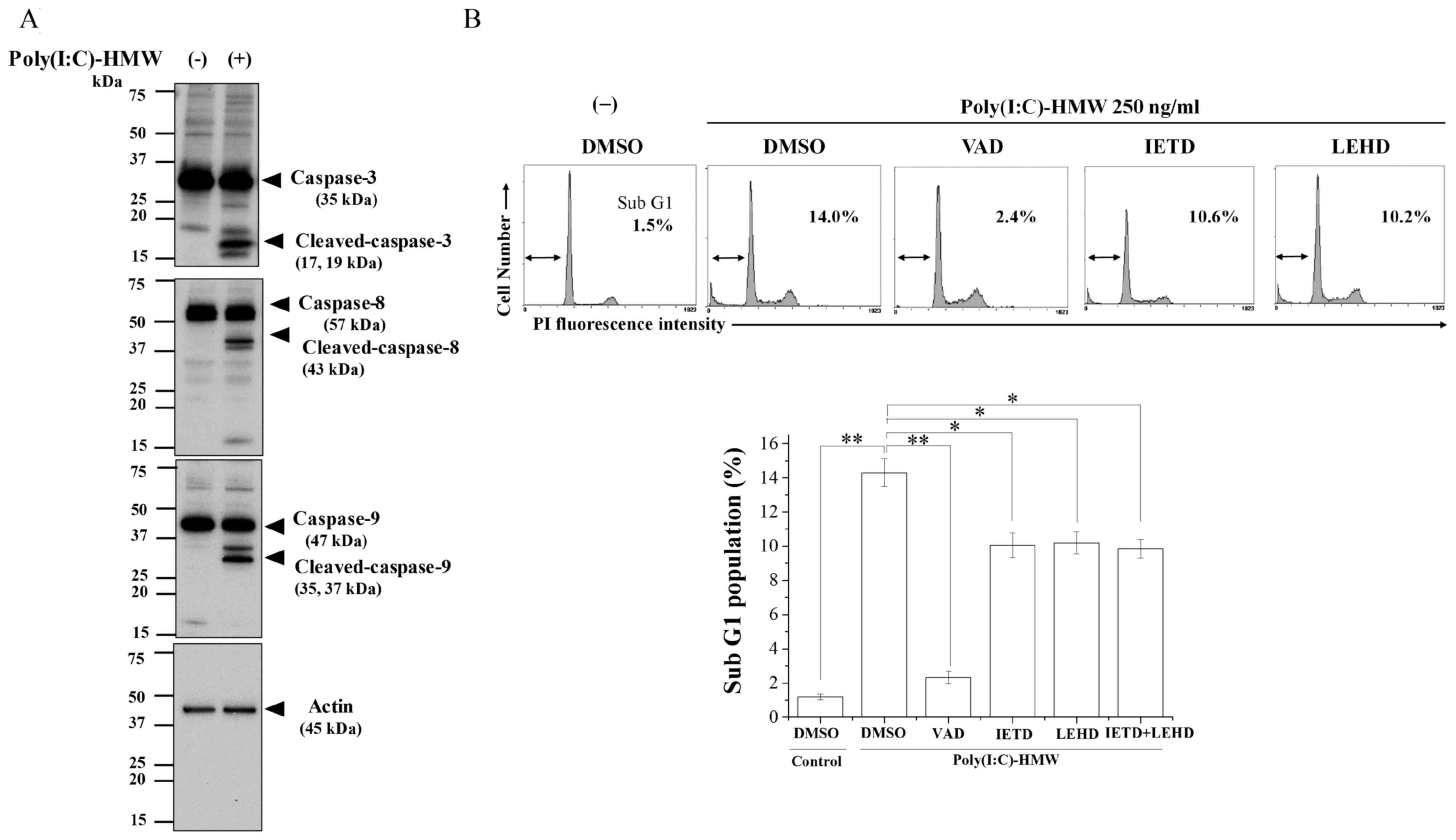 | Figure 1.Involvement of caspase-8 and
caspase-9 in Poly(I:C)-HMW-induced apoptosis. (A) A549 cells
treated with Poly(I:C)-HMW for 72 h were harvested for western blot
analyses of caspase-3, 8 and 9; β-actin was used as a loading
control. Representative blots are presented. (B) A549 cells were
pre-incubated with each caspase inhibitor for 1 h, followed by
treatment with 250 ng/ml Poly(I:C)-HMW. Following culturing for 72
h, cells were harvested for cell cycle analysis. Upper panel,
representative histograms and the data are presented. The
double-headed arrows indicate the sub-G1 population and the inset
numbers in the figure indicates the proportion of total cells in
the sub-G1 population. Lower panel, sub-G1 populations are
presented as the mean ± standard error of at least three
independent experiments. *P<0.05 and **P<0.01. DMSO, dimethyl
sulfoxide; VAD, Z-VAD-fmk; IETD, AC-IETD-CHO; LEHD, AC-LEHD-CHO;
Poly(I:C)-HMW, Poly(I:C)-HMW/LyoVec™; PI, propidium iodide. |
Caspase-8 involvement in apoptosis
enhancement by Poly(I:C)-HMW and IR cotreatment
The apoptotic pathway involved in the enhancement of
apoptosis by cotreatment with Poly(I:C)-HMW and IR was
investigated. As demonstrated in Fig.
2A, cotreatment with Poly(I:C)-HMW and 4 Gy X-ray irradiation
markedly increased the sub-G1 population compared with treatment
with Poly(I:C)-HMW or X-ray irradiation alone (P<0.01).
Furthermore, the active caspase-8 and caspase-9 expression in cells
treated with Poly(I:C)-HMW and IR increased compared with cells
treated with Poly(I:C)-HMW alone (Fig.
2B). The net increases in the sub-G1 population following IR in
the absence or presence of Poly(I:C)-HMW were ~2 and 20%,
respectively (Fig. 2C). This
elevated sub-G1 population in cotreated cells significantly
decreased to ~4 and 12% in the presence of Z-VAD-fmk or the
caspase-8 inhibitor IETD, respectively (P<0.05); however, there
was no significant difference when treated with the caspase-9
inhibitor LEHD. Furthermore, the inhibitory effect of the
combination of IETD and LEHD was similar to that of IETD alone
(Fig. 2C). Taken together, these
results suggested that caspase-8; however, not caspase-9, is
involved in the enhancement of apoptosis by cotreatment with
Poly(I:C)-HMW and IR.
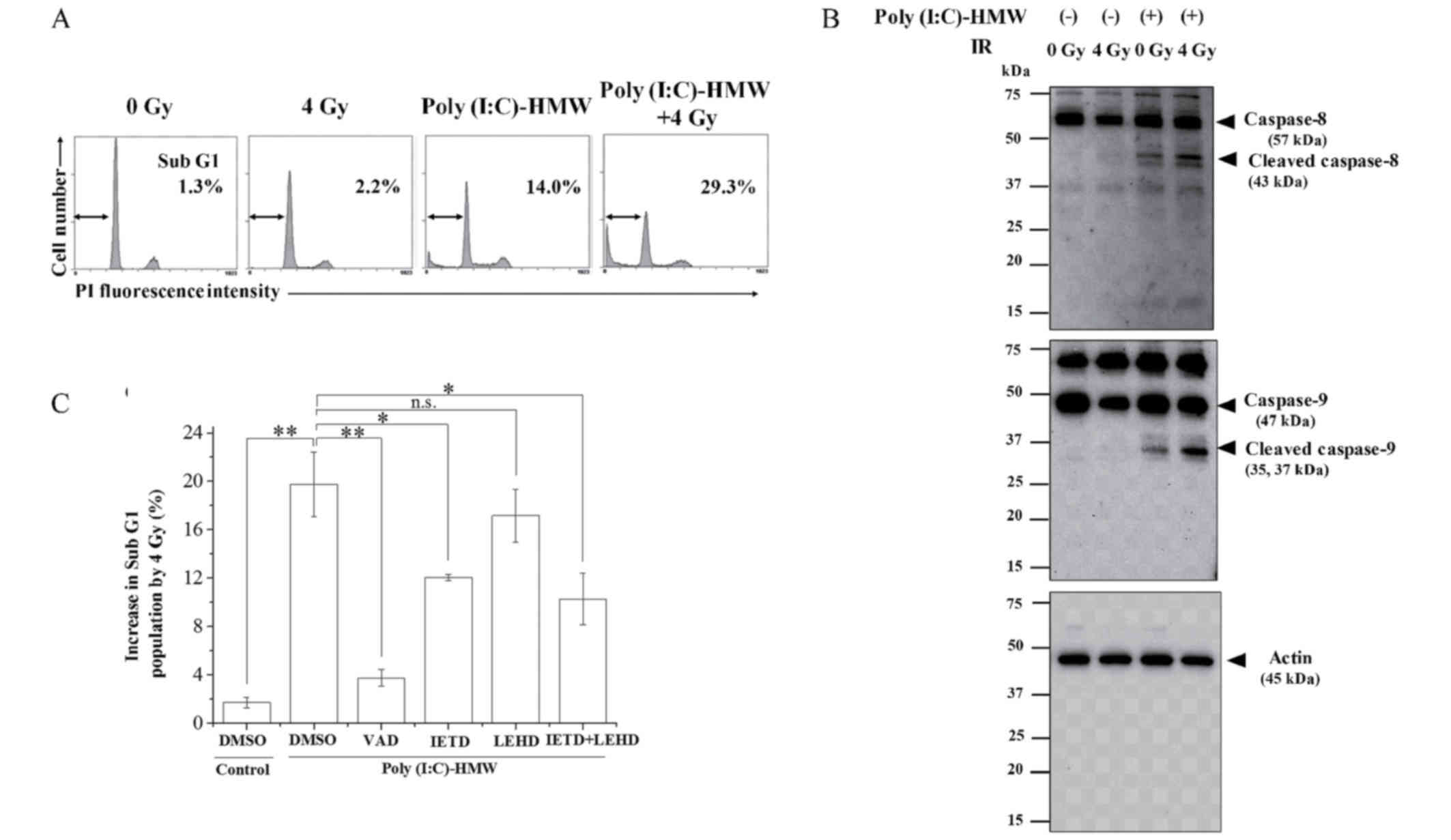 | Figure 2.Involvement of caspase-8 and 9 in the
enhancement of apoptosis by cotreatment with Poly(I:C)-HMW and IR.
A549 cells were pre-incubated with 250 ng/ml Poly(I:C)-HMW.
Following incubation for 1 h, the cells were irradiated with 4 Gy
X-rays. Following culturing for 72 h, the cells were harvested for
(A) cell cycle and (B) western blot analyses. Representative
histograms of cell cycle analyses are presented. The double-headed
arrows indicate the sub-G1 population and the inset numbers
indicate the proportion of cells in the sub-G1 population.
Representative blots of caspase-8 and caspase-9 are presented.
β-actin was used as a loading control. (C) Cells were pre-incubated
with each caspase inhibitor for 1 h, followed by treatment with 250
ng/ml Poly(I:C)-HMW. Following incubation for 1 h, the cells were
irradiated with 4 Gy X-rays. Following culturing for 72 h, the
cells were harvested for cell cycle analysis. Data of sub-G1
population are presented as the mean ± standard error of at least
three independent experiments. *P<0.05 and **P<0.01. DMSO,
dimethyl sulfoxide; VAD, Z-VAD-fmk; IETD, AC-IETD-CHO; LEHD,
AC-LEHD-CHO; n.s., not significant; Poly(I:C)-HMW,
Poly(I:C)-HMW/LyoVec™; PI, propidium iodide; IR, ionizing
radiation. |
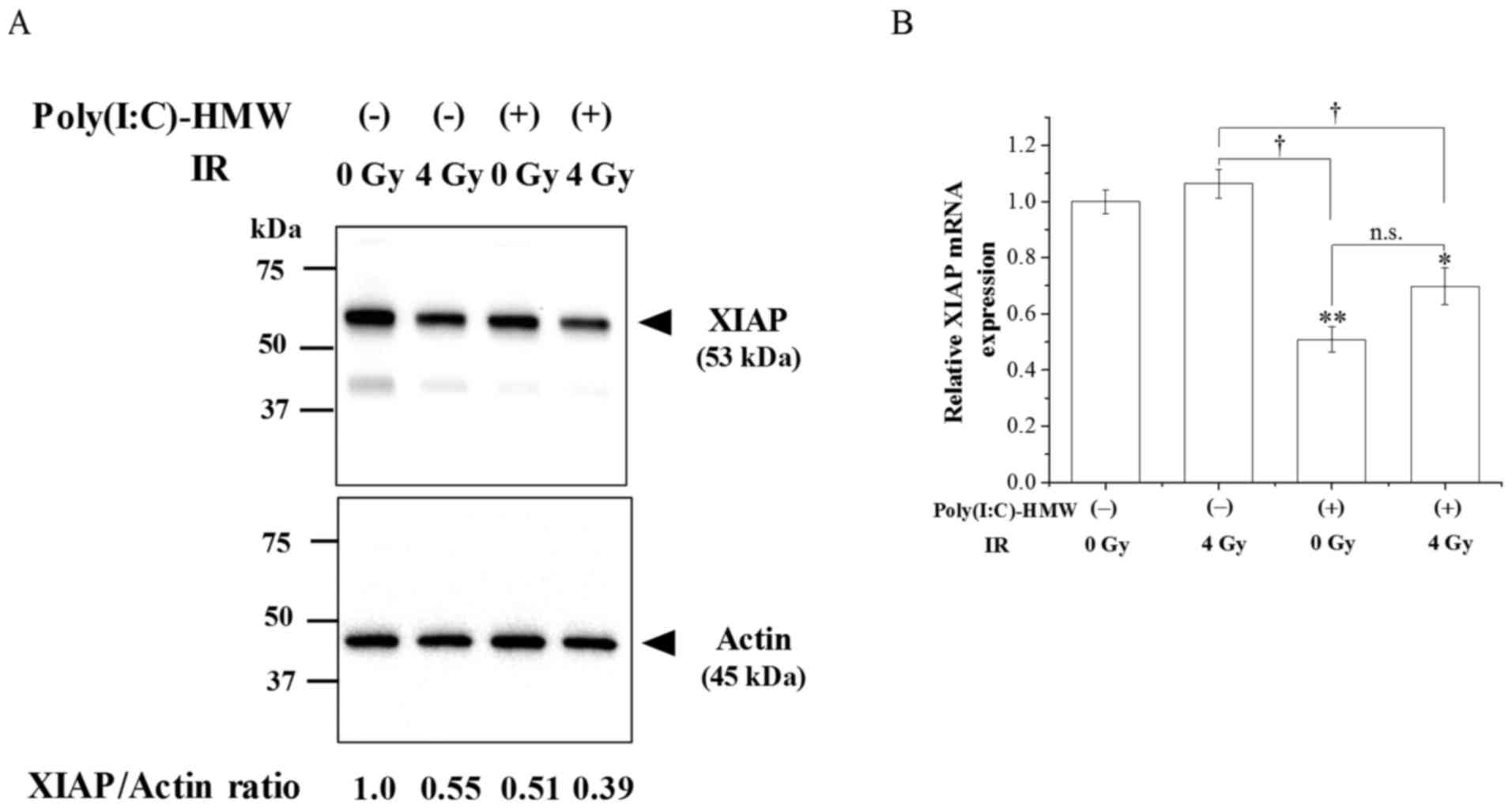
Effects of Poly(I:C)-HMW and IR
cotreatment on XIAP expression
The expression of the apoptosis inhibitor protein
XIAP was examined. As demonstrated in Fig. 3A, XIAP protein expression in cells
treated with Poly(I:C)-HMW and/or IR 72 h following treatment was
decreased compared with the control cells. Notably, XIAP protein
expression levels were lowest in the cotreatment group. When the
expression of XIAP mRNA was investigated, there was a significant
difference in expression between the control group and the
Poly(I:C)-HMW treatment and Poly(I:C)-HMW and IR cotreatment groups
(Fig. 3B; P<0.05).
Involvement of Fas in apoptosis
induction by Poly(I:C)-HMW and IR cotreatment
As caspase-8 is involved in the sub-G1 population
increase following cotreatment with Poly(I:C)-HMW and IR, the
involvement of death receptor Fas in cotreatment apoptosis
induction was investigated. As demonstrated in Fig. 4A and B, Poly(I:C)-HMW and IR
increased cell surface Fas expression. Furthermore, cell surface
Fas expression in cells cotreated with Poly(I:C)-HMW and IR was
significantly increased compared with Poly(I:C)-HMW or IR alone
(P<0.05; Fig. 4B). Therefore,
A549 cells were transfected with a Fas-targeting siRNA and
apoptosis induction by Poly(I:C)-HMW and/or IR was analyzed in
these cells. The knockdown of Fas decreased its cell surface
expression (Fig. 4C); however, did
not decrease apoptosis induction by Poly(I:C)-HMW and/or IR
(Fig. 4D).
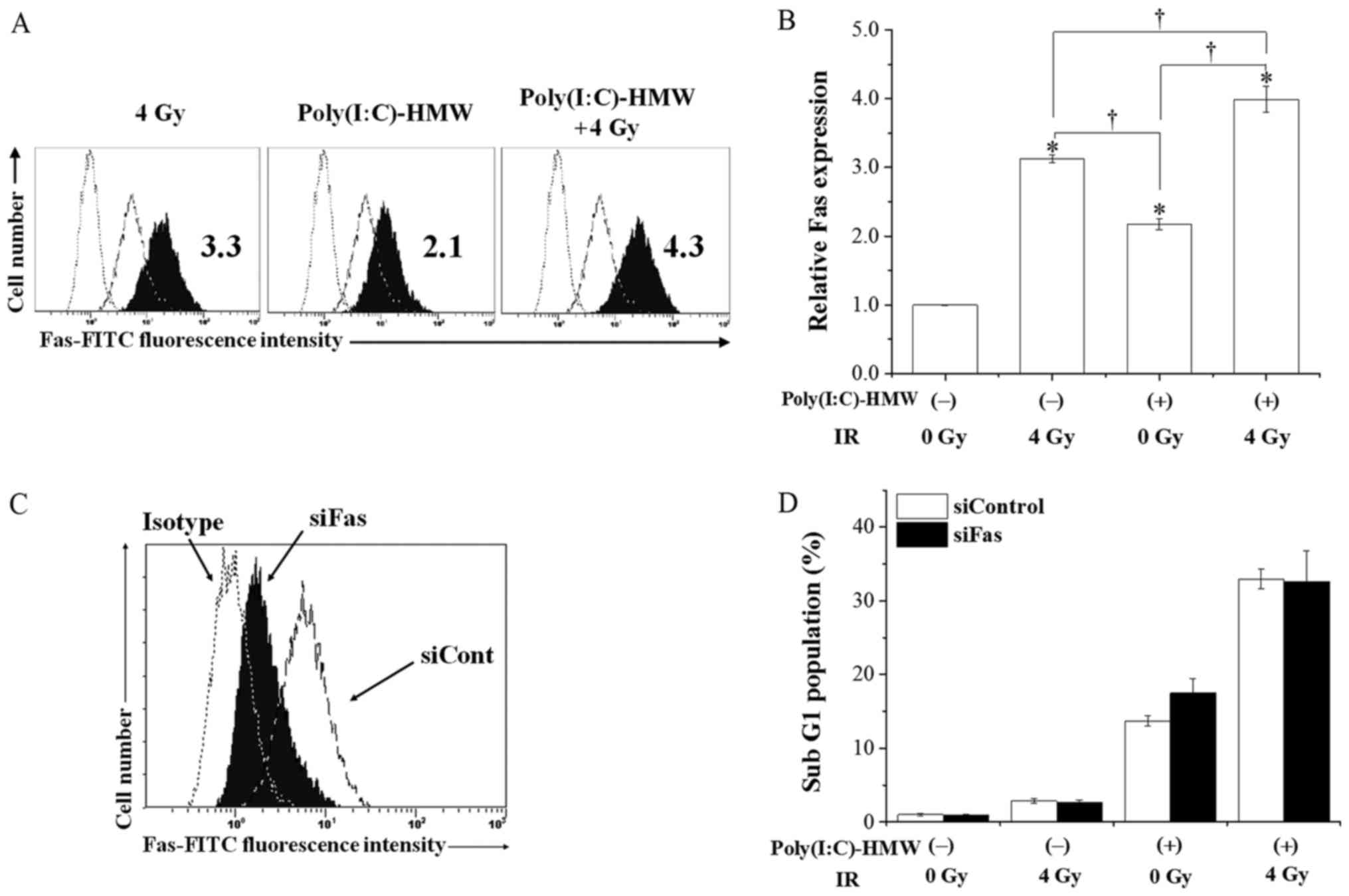 | Figure 4.Involvement of Fas in apoptosis
induction by Poly(I:C)-HMW and/or IR. A549 cells were pre-incubated
with 250 ng/ml Poly(I:C)-HMW. Following incubation for 1 h, the
cells were irradiated with 4 Gy X-rays. After culturing for 72 h,
the cells were harvested for analysis of cell surface Fas
expression. (A) Representative histograms are presented. The dotted
line indicates the isotype control. The broken line and filled
black histograms indicate the results from untreated cells and
treated cells, respectively. The inset numbers indicate the
relative values of median fluorescence intensity of Fas compared
with non-treated control group. (B) Relative Fas expression in the
control, Poly(I:C)-HMW, IR and cotreatment groups. Data are
presented as the mean ± standard error of three independent
experiments; *P<0.05 vs. the control group.
†P<0.05. (C) A549 cells treated with an siRNA against
Fas were harvested for the analysis of cell surface Fas expression.
The dotted line indicates the isotype control. The broken line and
filled black histogram indicate the results of cells treated with
control siRNA and Fas-targeting siRNA, respectively. (D) A549 cells
treated with siRNA against Fas were incubated with 250 ng/ml
Poly(I:C)-HMW, followed by 4 Gy X-ray irradiation. Following
culturing for 72 h, the cells were harvested for cell cycle
analysis. Data of sub-G1 population are presented as the mean ±
standard error of four independent experiments. IR, ionizing
radiation; Poly(I:C)-HMW, Poly(I:C)-HMW/LyoVec™; si, small
interfering; Cont, control; FITC, fluorescein isothiocyanate; Fas,
Fas cell surface death receptor. |
Discussion
The authors previously investigated the effect of IR
on RLRs in human monocytic cells and demonstrated that it
negligibly affected expression of RLRs and their response to their
agonists (22), suggesting a
potential for RLR agonists as effective immunostimulants during
radiation therapy. Furthermore, the authors recently identified
that RLR agonist Poly(I:C)-HMW exhibited cytotoxicity against human
NSCLC cells and that its cytotoxicity was enhanced by cotreatment
with IR (9). The authors
additionally demonstrated that Poly(I:C)-HMW, or cotreatment with
Poly(I:C)-HMW and IR, induced caspase-mediated apoptosis (9). In the present study, the involvement
of caspase-8 and caspase-9 in apoptosis induction by Poly(I:C)-HMW
or cotreatment with Poly(I:C)-HMW and IR was investigated. As a
result, Poly(I:C)-HMW was demonstrated to induce apoptosis through
caspase-8 and caspase-9. Furthermore, cotreatment with
Poly(I:C)-HMW and IR was demonstrated to effectively activate
caspase-8 and caspase-9 compared with each treatment alone, and
that caspase-8 inhibitor IETD decreased the apoptosis increase
induced by cotreatment. These results suggested that caspase-8;
however, not caspase-9, mediates the enhancement of apoptosis
induced by cotreatment with Poly(I:C)-HMW and IR.
It has been demonstrated that RLR activation may
induce apoptosis via the intrinsic (caspase-9) and/or extrinsic
(caspase-8) apoptotic pathways (4,5,23).
For example, Besch et al (5) demonstrated that the RIG-I agonist
5′-triphosphate RNA activates caspase-8 and caspase-9 in human
melanoma cells; however, caspase-9, not caspase-8, serves an
important role in 5′-triphosphate RNA-induced apoptosis. However,
El Maadidi et al (23)
observed that the activation of melanoma differentiation-associated
gene 5 by Semliki Forest Virus (SFV) induces caspase-9 and
caspase-8-mediated apoptosis. These previous studies suggested that
the apoptotic pathway induced by RLR activation depends on the type
of cell or on the RLR agonists.
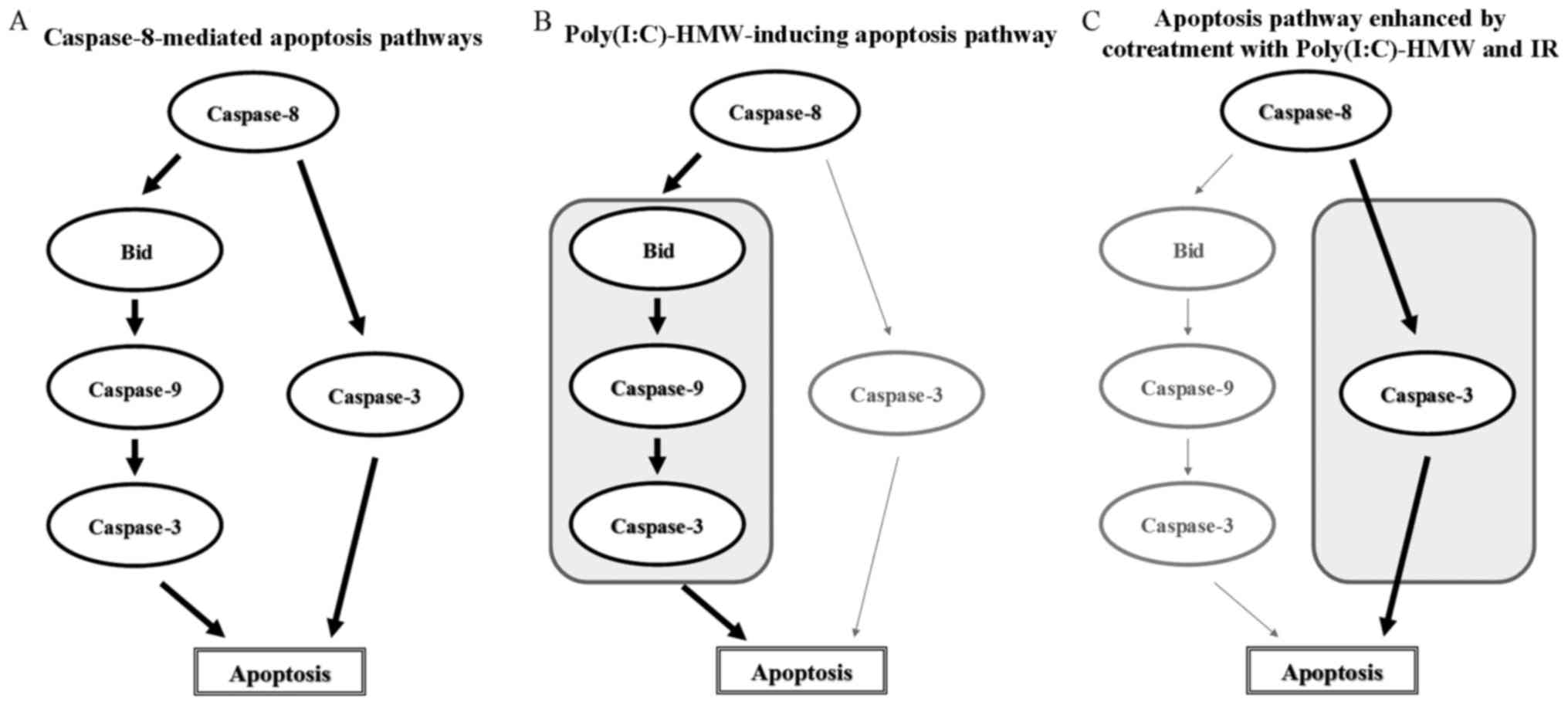
In the present study, it was demonstrated that
Poly(I:C)-HMW induced apoptosis in A549 cells through caspase-8 and
caspae-9 activation. It is known that caspase-8 may activate the
mitochondrial-mediated apoptotic pathway by regulating
mitochondrial outer membrane permeabilization (MOMP) (24). The activated caspase-8 cleaves a
pro-apoptotic Bcl-2 family member, BH3-interacting domain death
agonist (Bid), which subsequently induces MOMP and results in
caspase-9 activation followed by cytochrome c release (Fig. 5A) (24). When the combination of caspase-8
inhibitor and caspase-9 inhibitor was examined, the inhibitory
effect of it on the Poly(I:C)-HMW-induced sub-G1 population was
similar to that of caspase-8 or caspase-9 inhibitor alone.
Therefore, it is thought that Poly(I:C)-HMW induces apoptosis
thorough caspase-8-mediated caspase-9 activation; however, the
present study did not investigate whether Bid is involved in
caspase-8-mediated apoptosis pathways (Fig. 5B). Notably, it was demonstrated
that caspase-8 mediates the apoptosis enhancement from cotreatment
with Poly(I:C)-HMW and IR, whereas caspase-9 does not, even though
cotreatment with Poly(I:C)-HMW and IR effectively activated
caspase-8 and caspase-9 compared with Poly(I:C)-HMW alone. Similar
to the results of the present study, Liu et al (25) identified that the combination of
measles virus virotherapy and radiation therapy effectively induced
cleavage of poly(ADP-ribose) polymerase (PARP), a substrate of
capsase-3, in glioma cells, and that the increase in PARP cleavage
by the combination of them was inhibited by a pan-caspase inhibitor
and a caspase-8 inhibitor; however, not a caspase-9 inhibitor. As
caspase-3 may process pro-caspase-9 (26), it is possible that caspase-9 was
cleaved following the caspase-8/caspase-3 apoptotic pathway. El
Maadidi et al (23)
demonstrated that the apoptotic pathway induced by SFV is
independent of the MOMP induced by the pro-apoptotic Bcl-2 family
member protein apoptosis regulator BAX/Bcl-2 homologous
antagonist/killer and involves an SFV-induced recruitment of
caspase-8 to MAVS on the mitochondrial membrane where caspase-8 is
activated and required to cleave and activate caspase-3.
Furthermore, considering that the inhibitory effects of a
combination of caspase-8 inhibitor and caspase-9 inhibitor on the
increase in sub-G1 population by IR in the presence of
Poly(I:C)-HMW was similar to that of caspase-8 alone, it is likely
that the caspase-9-independent (or MOMP-independent) and
caspase-8-mediated apoptotic pathway is involved in the enhancement
of apoptosis by cotreatment with Poly(I:C)-HMW and IR (Fig. 5C).
In the present study, treatment with a pan-caspase
inhibitor, Z-VAD-fmk, inhibited apoptosis induction by
Poly(I:C)-HMW or by cotreatment with Poly(I:C)-HMW and IR. In
contrast, a caspase-8 inhibitor only partially decreased their
effects. These results suggested that other factors, other than
caspase-8, are involved in apoptosis induction by Poly(I:C)-HMW or
by cotreatment with Poly(I:C)-HMW and IR. It was demonstrated that
the inhibitor of apoptosis protein, XIAP, was downregulated in
cells treated with Poly(I:C)-HMW and/or IR. Nakhaei et al
(27) observed that the
downregulation of XIAP protein expression sensitizes cells to
virus-induced apoptosis. Furthermore, it has been demonstrated that
the suppression of XIAP overexpression enhances the radiation
sensitivity of human lung cancer (28), suggesting an association between
the radiation sensitivity of malignant tumors and XIAP expression.
As XIAP is known to be a potent inhibitor of caspases 3, 7 and 9
(29), it is likely that the
downregulation of XIAP by cotreatment with Poly(I:C)-HMW and IR
enhanced caspase-mediated apoptosis. There is a possibility that
other apoptotic mechanisms, including the endoplasmic
reticulum-mediated apoptotic pathway, are involved in apoptosis
induction by Poly(I:C)-HMW or cotreatment with Poly(I:C)-HMW and
IR. Further studies are required to investigate other apoptotic
mechanisms.
Downregulation of XIAP protein expression was
observed in the cells treated with Poly(I:C)-HMW and/or IR.
Notably, Poly(I:C)-HMW alone and cotreatment decreased XIAP mRNA
expression, whereas IR alone did not. These results suggested that
Poly(I:C)-HMW downregulated XIAP protein expression by decreasing
the transcription of XIAP mRNA; whereas, X-ray irradiation affects
XIAP expression through a transcription-independent manner. It is
known that XIAP protein expression is tightly regulated not only at
the transcriptional level; however, additionally
post-transcriptionally (30,31).
Yang et al (32)
demonstrated that DNA damaging agents, including etoposide, may
degrade XIAP through the ubiquitin-proteasome pathway. Therefore,
it is possible that IR decreases XIAP protein expression through
proteasome-mediated degradation. The proteasomal degradation of
XIAP may additionally be involved in the downregulation of XIAP
protein by Poly(I:C)-HMW, as it was identified that Sendai virus
infection induces the proteasomal degradation of XIAP by
phosphorylating XIAP at Ser430 through Iκ B kinase ε- or
Tank-binding kinase 1, which function downstream of MAVS (27). Further studies are required to
elucidate the mechanisms by which Poly(I:C)-HMW and/or IR regulate
XIAP protein expression.
As IR increases Fas expression, which causes
caspase-8-mediated apoptosis (13,33),
Fas was predicted to be involved in apoptosis induction by
cotreatment with Poly(I:C)-HMW and IR. However, although the
upregulation of cell surface Fas expression in cotreated cells was
observed (Fig. 4A and B), Fas
knockdown did not decrease apoptosis induction (Fig. 4C). Therefore, it is unlikely that
Fas upregulation is associated with apoptosis enhancement by
cotreatment. El Maadidi et al (23) observed that the RLR adaptor protein
MAVS directly associates with caspase-8 and subsequently activates
caspase-3, leading to apoptosis. Therefore, it is possible that
MAVS-mediated caspase-8 activation is involved in the apoptosis
enhancement observed upon cotreatment with Poly(I:C)-HMW and IR. As
the expression of other death receptors, including death receptor
5, is known to be enhanced by X-ray irradiation (14,34),
there is a possibility that other death receptors are involved in
the cotreatment-induced apoptosis enhancement of Poly(I:C)-HMW and
IR. In a future study, the authors aim to investigate the
mechanisms by which cotreatment with Poly(I:C)-HMW and IR
effectively activate caspase-8.
In conclusion, in the present study it was
demonstrated that caspase-8 is involved in the enhancement of
apoptosis by cotreatment with Poly(I:C)-HMW and IR. Furthermore, it
was demonstrated that cotreatment effectively activates caspase-8
independently of the upregulation of cell surface Fas expression.
As it has been demonstrated that RLR agonists increase Fas
expression in pancreatic cells and that cells treated with RLR
agonists become sensitive to Fas-mediated cell killing effects
(35), it is hypothesized that
combinations of RLR agonists, X-ray irradiation and Fas agonists
may be effective in cancer treatments. This hypothesis requires
further verification in a future study.
Acknowledgements
Not applicable.
Funding
The present study was supported by JSPS KAKENHI
(grant no. JP25861053) and was partially supported by JSPS KAKENHI
(grant no. JP15K09985) and the Takeda Science Foundation (for HY;
Osaka, Japan).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
HY and IK contributed to the conception of the
study. HY, YS and YK performed experiments, collected data, and
analyzed data. YS, HY and IK wrote, reviewed and revised the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Yoneyama M and Fujita T: Structural
mechanism of RNA recognition by the RIG-I-like receptors. Immunity.
29:178–181. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kawai T, Takahashi K, Sato S, Coban C,
Kumar H, Kato H, Ishii KJ, Takeuchi O and Akira S: IPS-1, an
adaptor triggering RIG-I- and MDA5-mediated type I interferon
induction. Nat Immunol. 6:981–988. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Johnson CL and Gale M Jr: CARD games
between virus and host get a new player. Trends Immunol. 27:1–4.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Wu Y, Wu X, Wu L, Wang X and Liu Z: The
anticancer functions of RIG-I-like receptors, RIG-I and MDA5, and
their applications in cancer therapy. Transl Res. 190:51–60. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Besch R, Poeck H, Hohenauer T, Senft D,
Häcker G, Berking C, Hornung V, Endres S, Ruzicka T, Rothenfusser S
and Hartmann G: Proapoptotic signaling induced by RIG-I and MDA-5
results in type I interferon-independent apoptosis in human
melanoma cells. J Clin Invest. 119:2399–2411. 2009.PubMed/NCBI
|
|
6
|
Yuan D, Xia M, Meng G, Xu C, Song Y and
Wei J: Anti-angiogenic efficacy of 5′-triphosphate siRNA combining
VEGF silencing and RIG-I activation in NSCLCs. Oncotarget.
6:29664–29674. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Poeck H, Besch R, Maihoefer C, Renn M,
Tormo D, Morskaya SS, Kirschnek S, Gaffal E, Landsberg J, Hellmuth
J, et al: 5′-Triphosphate-siRNA: Turning gene silencing and Rig-I
activation against melanoma. Nat Med. 14:1256–1263. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li D, Gale RP, Liu Y, Lei B, Wang Y, Diao
D and Zhang M: 5′-Triphosphate siRNA targeting MDR1 reverses
multi-drug resistance and activates RIG-I-induced
immune-stimulatory and apoptotic effects against human myeloid
leukaemia cells. Leuk Res. 58:23–30. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yoshino H, Iwabuchi M, Kazama Y, Furukawa
M and Kashiwakura I: Effects of retinoic acid-inducible gene-I-like
receptors activations and ionizing radiation cotreatment on
cytotoxicity against human non-small cell lung cancer in
vitro. Oncol Lett. 15:4697–4705. 2018.PubMed/NCBI
|
|
10
|
Green DR and Llambi F: Cell death
signaling. Cold Spring Harb Perspect Biol. 7:pii: a006080. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Rongvaux A: Innate immunity and tolerance
toward mitochondria. Mitochondrion. 41:14–20. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mohamed MS, Bishr MK, Almutairi FM and Ali
AG: Inhibitors of apoptosis: Clinical implications in cancer.
Apoptosis. 22:1487–1509. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Takahashi K, Inanami O, Hayashi M and
Kuwabara M: Protein synthesis-dependent apoptotic signalling
pathway in X-irradiated MOLT-4 human leukaemia cell line. Int J
Radiat Biol. 78:115–124. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kim MJ, Lee KH and Lee SJ: Ionizing
radiation utilizes c-Jun N-terminal kinase for amplification of
mitochondrial apoptotic cell death in human cervical cancer cells.
FEBS J. 275:2096–2108. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yoshino H, Kumai Y and Kashiwakura I:
Effects of endoplasmic reticulum stress on apoptosis induction in
radioresistant macrophages. Mol Med Rep. 15:2867–2872. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wakasaya T, Yoshino H, Fukushi Y,
Yoshizawa A and Kashiwakura I: A liquid crystal-related compound
induces cell cycle arrest at the G2/M phase and apoptosis in the
A549 human non-small cell lung cancer cell line. Int J Oncol.
42:1205–1211. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Fukushi S, Yoshino H, Yoshizawa A and
Kashiwakura I: p53-independent structure-activity relationships of
3-ring mesogenic compounds' activity as cytotoxic effects against
human non-small cell lung cancer lines. BMC Cancer. 16:5212016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yoshino H, Chiba K, Saitoh T and
Kashiwakura I: Ionizing radiation affects the expression of
Toll-like receptors 2 and 4 in human monocytic cells through c-Jun
N-terminal kinase activation. J Radiat Res. 55:876–884. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Larionov A, Krause A and Miller W: A
standard curve based method for relative real time PCR data
processing. BMC Bioinformatics. 6:622005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yoshino H and Kashiwakura I: Involvement
of reactive oxygen species in ionizing radiation-induced
upregulation of cell surface toll-like receptor 2 and 4 expression
in human monocytic cells. J Radiat Res. 58:626–635. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Darzynkiewicz Z, Buruno S, Del Bino G,
Gorczyca W, Hotz MA, Lassota P and Traganos F: Features of
apoptotic cells measured by flow cytometry. Cytometry. 13:795–808.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Yoshino H, Saitoh T, Kozakai M and
Kashiwakura I: Effects of ionizing radiation on retinoic
acid-inducible gene-I-like receptors. Biomed Rep. 3:59–62. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
El Maadidi S, Faletti L, Berg B, Wenzl C,
Wieland K, Chen ZJ, Maurer U and Borner C: A novel mitochondrial
MAVS/Caspase-8 platform links RNA virus-induced innate antiviral
signaling to Bax/Bak-independent apoptosis. J Immunol.
192:1171–1183. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Kalkavan H and Green DR: MOMP, cell
suicide as a BCL-2 family business. Cell Death Differ. 25:46–55.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Liu C, Sarkaria JN, Petell CA,
Paraskevakou G, Zollman PJ, Schroeder M, Carlson B, Decker PA, Wu
W, James CD, et al: Combination of measles virus virotherapy and
radiation therapy has synergistic activity in the treatment of
glioblastoma multiforme. Clin Cancer Res. 13:7155–7165. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Porter AG and Jänicke RU: Emerging roles
of caspase-3 in apoptosis. Cell Death Differ. 6:99–104. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Nakhaei P, Sun Q, Solis M, Mesplede T,
Bonneil E, Paz S, Lin R and Hiscott J: IκB kinase ε-dependent
phosphorylation and degradation of X-linked inhibitor of apoptosis
sensitizes cells to virus-induced apoptosis. J Virol. 86:726–737.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Cao C, Mu Y, Hallahan DE and Lu B: XIAP
and survivin as therapeutic targets for radiation sensitization in
preclinical models of lung cancer. Oncogene. 23:7047–7052. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Eckelman BP, Salvesen GS and Scott FL:
Human inhibitor of apoptosis proteins: Why XIAP is the black sheep
of the family. EMBO Rep. 7:988–994. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hiramatsu N, Messah C, Han J, LaVail MM,
Kaufman RJ and Lin JH: Translational and posttranslational
regulation of XIAP by eIF2α and ATF4 promotes ER stress-induced
cell death during the unfolded protein response. Mol Biol Cell.
25:1411–1420. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Galbán S and Duckett CS: XIAP as a
ubiquitin ligase in cellular signaling. Cell Death Differ. 7:54–60.
2010. View Article : Google Scholar
|
|
32
|
Yang Y, Fang S, Jensen JP, Weissman AM and
Ashwell JD: Ubiquitin protein ligase activity of IAPs and their
degradation in proteasomes in response to apoptotic stimuli.
Science. 288:874–877. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Afshar G, Jelluma N, Yang X, Basila D,
Arvold ND, Karlsson A, Yount GL, Dansen TB, Koller E and Haas-Kogan
DA: Radiation-induced caspase-8 mediates p53-independent apoptosis
in glioma cells. Cancer Res. 66:4223–4232. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hamasu T, Inanami O, Asanuma T and
Kuwabara M: Enhanced induction of apoptosis by combined treatment
of human carcinoma cells with X rays and death receptor agonists. J
Radiat Res. 46:103–110. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Duewell P, Steger A, Lohr H, Bourhis H,
Hoelz H, Kirchleitner SV, Stieg MR, Grassmann S, Kobold S, Siveke
JT, et al: RIG-I-like helicases induce immunogenic cell death of
pancreatic cancer cells and sensitize tumors toward killing by
CD8(+) T cells. Cell Death Differ. 21:1825–1837. 2014. View Article : Google Scholar : PubMed/NCBI
|