Introduction
Ginseng, the root of Araliaceae plants, is a Chinese
herbal medicine that can be divided into different categories,
including wild ginseng, red ginseng and Korean ginseng among
others. Panax ginseng C.A. Meyer, a native herbal remedy
commonly used in China and Korea (1,2), has
been recognized as a life prolonging herb in Asia for thousands of
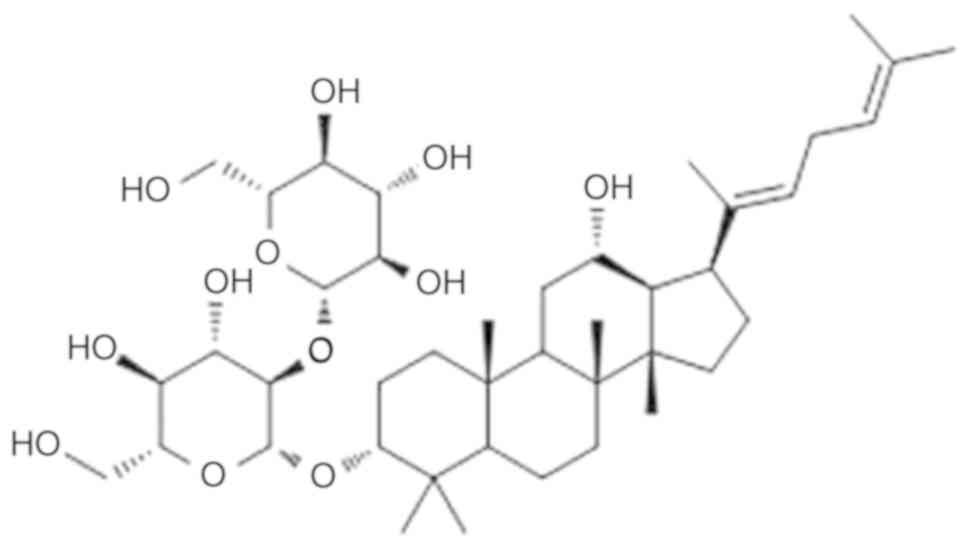
years (3–5). Ginsenoside Rg5 (Fig. 1) belongs to a family of
protopanaxadiol ginsenosides (1,6) and
has been demonstrated to exhibit marked anticancer activity
(7,8), antidermatitic activity (9), neuroprotective effects (10) and microglial activation (11).
Esophageal cancer is one of the most common
malignancies worldwide, with a high mortality rate of >400,000
cases per year (12). At present,
the main treatments include surgery, radiotherapy, chemotherapy and
targeted therapy. Squamous cell carcinoma accounts for >90% of
the esophageal pathological classifications worldwide, closely
followed by adenocarcinoma. Surgical resection remains the primary
treatment; however, the 5-year overall survival rate is only ~30%,
and the squamous cell carcinoma survival rate is 20–50%. Surgical
treatment is mainly limited by local recurrence and metastasis, as
well as the reduction of the patients' immune function
postoperatively (13). The
majority of patients are diagnosed at a relatively late stage due
to the lack of evident clinical symptoms during the early stages.
It has been reported that 40–60% of patients are unable to undergo
surgery as a result of suffering from late stage disease or due to
the high risks associated with surgery (14). Local radiotherapy is one of the
best treatment options for patients with local advancement. Since
the development of RTOG85-01, radiotherapy combined with
chemotherapy has been accepted as the optimal treatment option for
non-surgical esophageal cancer patients (15). However, the search for novel drugs
for the treatment of esophageal cancer remains of great
significance.
Apoptosis, also known as programmed cell death, is
important for controlling cell numbers and proliferation, and is
essential for the elimination of cancer cells. Apoptotic cells
usually avoid chromatin condensation, forming apoptotic bodies
(16). There are two major
apoptotic pathways, including extrinsic or death receptor pathways,
and intrinsic or mitochondrial pathways (17). These two pathways are associated
with the activation of caspases, which are responsible for inducing
apoptosis through nuclear DNA cleavage and regulatory cell
proteins. The mitochondrial pathway is mainly regulated by members
of the B-cell lymphoma 2 (Bcl-2) family, which includes
proapoptotic proteins, such as Bcl-2-associated X protein (Bax),
and antiapoptotic proteins, such as Bcl-2 and Bcl-extra large
protein (18). The
phosphoinositide-3 kinase (PI3K)/protein kinase B (Akt) signaling
pathway has a positive role in regulating cell growth,
proliferation, differentiation and cell cycle progression, and in
reversing drug resistance. Activated Akt in turn activates
downstream signaling pathways via phosphorylation in order to
participate in the regulation of cellular physiological and
biochemical changes that protect the surviving cells from
undergoing apoptosis. In addition, the inactivation of Akt may
contribute to the apoptosis of esophageal cancer cells.
The application of ginsenoside Rg5 in the treatment
of esophageal cancer is considered to have potentially promising
anti-tumor effects. Therefore, the aim of the present study was to
investigate the anti-tumor effect of ginsenoside Rg5 on esophageal
cancer cells and examine the possible molecular mechanisms in order
to provide an objective basis for its function.
Materials and methods
Chemicals and reagents
Ginsenoside Rg5 (purity, >99%), dissolved in 75%
ethanol at a density of 10 mM and stored at −20°C, was obtained
from Professor Yinghua Jin (College of Life Science, Jilin
University, Changchun, China). The Cell Counting Kit-8 (CCK-8)
assay kit and Fluo-3/acetoxymethyl (AM) were purchased from Dojindo
Molecular Technologies, Inc. (Kumamoto, Japan). The Annexin
V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis
assay kit and bicinchoninic acid (BCA) protein assay kit were
obtained from GenStar Biosolutions Co., Ltd. (Beijing, China).
Rhodamine 123 and LY294002 were purchased from Sigma-Aldrich (Merck
KGaA, Darmstadt, Germany). Insulin-like growth factor (IGF)-1 was
obtained from R&D Systems Europe, Ltd. (Abingdon, UK). Rabbit
polyclonal antibodies specific for cleaved caspase-3 (9661T;
1:800), Bax (5023T; 1:1,000), Bcl-2 (2872S; 1:1,000), Akt (4691S;
1:1,000), phosphorylated (p)-Akt (9611S; 1:1,000), cleaved
poly(adenosine diphosphate-ribose) polymerase (PARP) (5625S;
1:1,000) were obtained from Cell Signaling Technology, Inc.
(Danvers, MA, USA). Rabbit polyclonal antibodies specific for
cleaved caspase-8 (YT5688; 1:1,000) and caspase-9 (YC0013; 1:1,000)
were obtained from ImmunoWay Biotechnology Company (Plano, TX,
USA). Antibodies specific for β-actin (E-AB-20058; 1:1,000) and
horseradish peroxidase-conjugated goat anti-rabbit secondary
antibodies (E-AB-1003; 1:5,000) were purchased from Elabscience
Biotechnology Co., Ltd. (Wuhan, China).
Cell lines and cell culture
The human esophageal cancer cell line Eca-109 was
provided by Dr Xingyi Zhang (Second Hospital of Jilin University,
Changchun, China). Cells were incubated in RPMI-1640 medium with
10% fetal bovine serum, 100 U/ml penicillin and 100 µg/ml
streptomycin under 5% CO2 in humidified air at 37°C.
Cell viability assay
The effect of ginsenoside Rg5 on the viability of
cells was measured using the CCK-8 assay. Briefly, Eca-109 cells
were plated at a density of 2,000 cells/well with 200 µl complete
culture medium in 96-well plates, and treated with 2, 4, 8, 16 and
32 µM of ginsenoside Rg5 for 24 h. A total of 10 µl CCK-8 solution
was then added to the aforementioned cell cultures according to the
manufacturer's protocol. Subsequently, the plates were incubated
for 2 h at 37°C, and the optical density was read at a wavelength
of 450 nm on a microplate reader (Thermo Fisher Scientific, Inc.,
Waltham, MA, USA). The half maximal inhibitory concentration
(IC50) value was calculated using a fitted function
according to the concentration of each group and the corresponding
inhibition rate. This calculation was conducted automatically using
IBM SPSS software (version 22.0; IBM Corp., Armonk, NY, USA).
Analysis of apoptosis using flow
cytometry
Cell apoptosis was measured with a flow cytometer
using the Annexin V-FITC/PI apoptosis assay kits following the
manufacturer's protocol. Following exposure to the determined
concentrations of ginsenoside Rg5 for 24 h, cells were collected,
washed with cold PBS, and resuspended with binding buffer
containing 10 mM HEPES, 2.5 mM CaCl2 and 140 mM NaCl.
Next, cells were incubated with Annexin V in the dark at room
temperature for 10 min and then resuspended in binding buffer. PI
was added to the samples at 37°C immediately prior to analysis with
a FACScan flow cytometer (Beckman Coulter, Inc., Brea, CA,
USA).
Detection of mitochondrial membrane
potential
Mitochondrial membrane potential was detected using
rhodamine 123. Initially, Eca-109 cells were briefly treated with
Rg5 at a concentration of 5, 10 and 20 µM for 24 h. Next, the cell
suspension was routinely prepared, rinsed twice with PBS buffer,
cultured with rhodamine 123 (5 µg/ml) at 37°C for 30 min and washed
with PBS. The cells were collected again by centrifugation (800 × g
at 37°C for 5 min) and resuspended with 300 µl PBS. The
fluorescence intensity of cells incubated with rhodamine 123 was
finally determined by flow cytometric analysis.
Analysis of calcium levels
Calcium levels were measured using the calcium
indicator Fluo-3/AM and laser scanning confocal microscopic
imaging. Briefly, treated Eca-109 cells were incubated with
Fluo-3/AM for ~30 min in the dark at 37°C, and then washed with
D-Hanks medium. The fluorescence intensity was detected by confocal
microscopy at 488 nm, measuring the intensity of ~5–10 cells in
each microscopic field separately. Finally, the fluorescence
intensity was digitized, and all data were analyzed using the
affiliated software of the confocal microscope system.
Western blot analysis
Treated cells were washed with cold PBS and
extracted with lysis buffer, containing 50 mmol/l Tris (pH 8.0),
150 mmol/l NaCl, 5% glycerol, 5 mmol/l EDTA, 1% Triton X-100, 25
mmol/l NaF, 2 mmol/l Na2VO4, 10 µg/ml
aprotinin, 10 µg/ml pepstatin and 10 µg/ml leupeptin. The equal
amounts of cell lysates were frozen, thawed three times and then
centrifuged at 15,000 × g for 10 min at 4°C in order to remove the
insoluble pellet material. Protein determination was performed
using the BCA protein assay kit. SDS-PAGE was performed on 12 or
15% gels according to the protein molecular weight and a 5%
stacking gel. The proteins on the gels were then transferred to
prepared polyvinylidene difluoride membranes. Subsequently, the
membranes were blocked for ~2 h at 37°C in Tris-buffered saline
containing 0.5% Tween-20 (TBST) and 5% fat-free dried milk, and
then incubated with the corresponding antibodies overnight at 4°C.
The membranes were washed three times with TBST and incubated with
secondary antibodies at room temperature for 1 h. The
Hypersensitive Fluorescence and Chemiluminescence Imaging System
(Clinx Science Instruments Co., Ltd., Shanghai, China) was used to
visualize the signals. Band density was measured using Cell Quest
Research software (version 3.3; BD Biosciences, Franklin Lakes, NJ,
USA) and normalized to β-actin.
Inhibitor and activator of PI3K
treatments
Eca-109 cells were co-treated with Rg5 (10 µM) and
the PI3K-specific inhibitor LY294002 (20 µM) or the PI3K-specific
activator IGF-1 (100 ng/ml) for 24 h. Cell apoptosis was then
analyzed using a flow cytometer and the Annexin V-FITC/PI apoptosis
assay kit. Protein levels were assessed via western blotting, as
described earlier.
Statistical analysis
Data were analyzed by one-way analysis of variance,
followed by Student-Newman-Keuls test, and each value was expressed
as the mean ± standard deviation. Statistical analysis was
performed on IBM SPSS software (version 22.0). P<0.05 was
considered to indicate a statistically significant difference.
Results
Ginsenoside Rg5 inhibits the
proliferation of Eca-109 cells
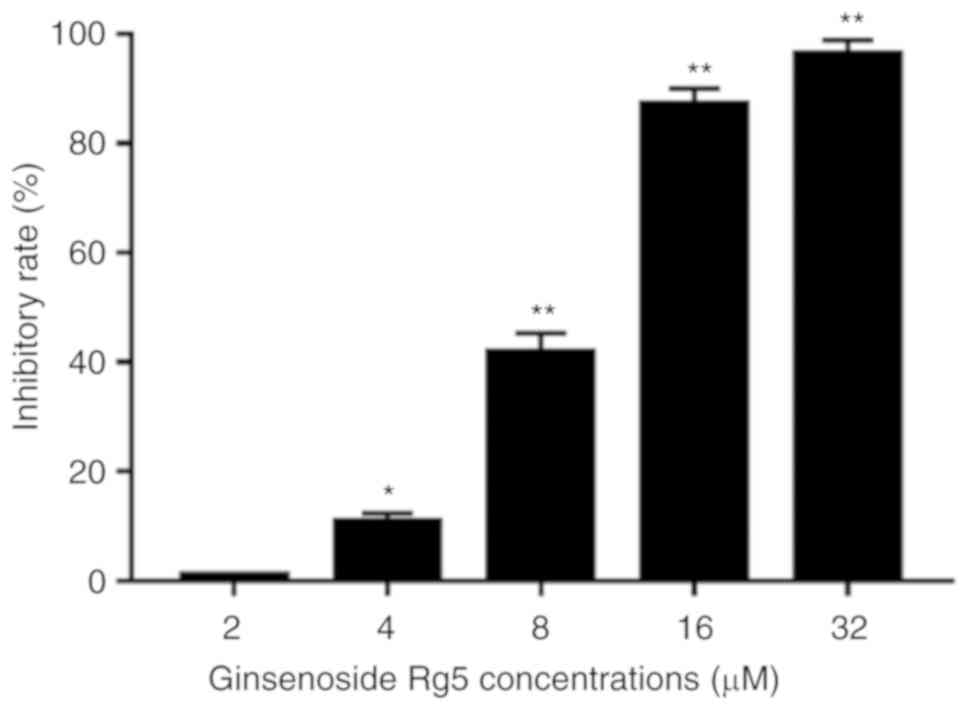
The anti-proliferative effect of ginsenoside Rg5 on
Eca-109 cells was measured by treating the cells with different
concentrations of ginsenoside Rg5 (2, 4, 8, 16 and 32 µM) for 24 h.
The results revealed that ginsenoside Rg5 significantly inhibited
the proliferation of Eca-109 cells in a concentration-dependent
manner. The IC50 value of ginsenoside Rg5 for inhibiting
Eca-109 cell proliferation was 8.245 µmol/l (Fig. 2).
Ginsenoside Rg5 induces apoptosis in
Eca-109 cells
In order to determine whether the anti-proliferative
effects of Rg5 were associated with the induction of apoptosis,
Eca-109 cells were treated with 0–20 µM ginsenoside Rg5 for 24 h,
and the Annexin V-FITC/PI apoptosis assay kit was used to determine
the rates of Rg5-induced apoptosis. As shown in Fig. 3A, treatment with ginsenoside Rg5 at
5, 10 and 20 µM induced a significant increase in apoptosis
compared with the control group.
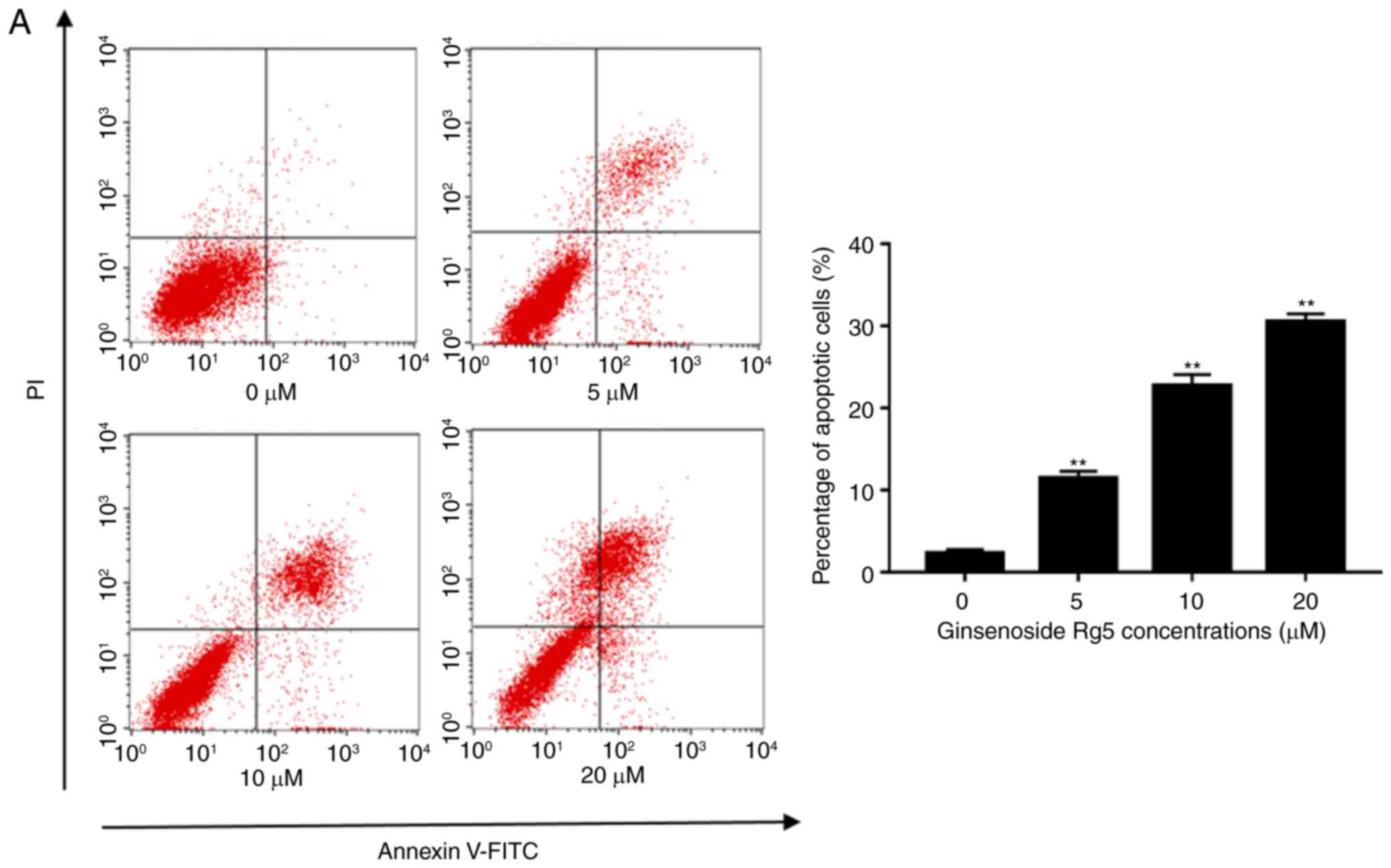 | Figure 3.Ginsenoside Rg5 induces apoptosis in
Eca-109 cells. (A) Annexin V-FITC/PI apoptosis assay kit was used
to determine the rate of Rg5-induced apoptosis following treatment
with ginsenoside Rg5 for 24 h. A concentration-dependent increase
in Eca-109 cell apoptosis was observed. (B) Cleaved caspase-3, −8
and −9, Bcl-2, Bax, Akt and p-Akt protein expression levels were
analyzed by western blot analysis. The quantified results are
presented in terms of the ratio of cleaved caspase-3/8/9 to
β-actin, Bcl-2 to Bax, and p-Akt to Akt. Data are presented as the
mean ± standard deviation. *P<0.05 and **P<0.01, vs. the
untreated group. FITC, fluorescein isothiocyanate; PI, propidium
iodide; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X protein;
p-, phosphorylated; Akt, protein kinase B. |
Ginsenoside Rg5 induces the cleavage
of caspase-3, −8 and −9, and the phosphorylation of Akt in Eca-109
cells
The activation of specific caspases, including
caspase-8 and −9, and the downstream effector caspase-3 were
analyzed by western blotting. The results revealed that the levels
of cleaved caspase-3, −8 and −9 increased following treatment with
ginsenoside Rg5 in a dose-dependent manner (Fig. 3B). In addition, the proteolytic
cleavage of PARP was increased. The levels of the pro-apoptotic
protein Bax and the anti-apoptotic protein Bcl-2 were also
measured. The results indicated that the ratio of Bcl-2/Bax
expression decreased following ginsenoside Rg5 treatment. The
PI3K/Akt signaling pathway is considered to be an important
upstream apoptotic pathway; thus, this was also examined in the
present study. Western blot analysis revealed that Eca-109 cells
treated with ginsenoside Rg5 (5, 10 and 20 µM) presented
significantly inhibited expression of p-Akt (Fig. 3B). It was speculated that the
promotion of apoptosis by Rg5 may be associated with the
downregulation of the PI3K/Akt pathway.
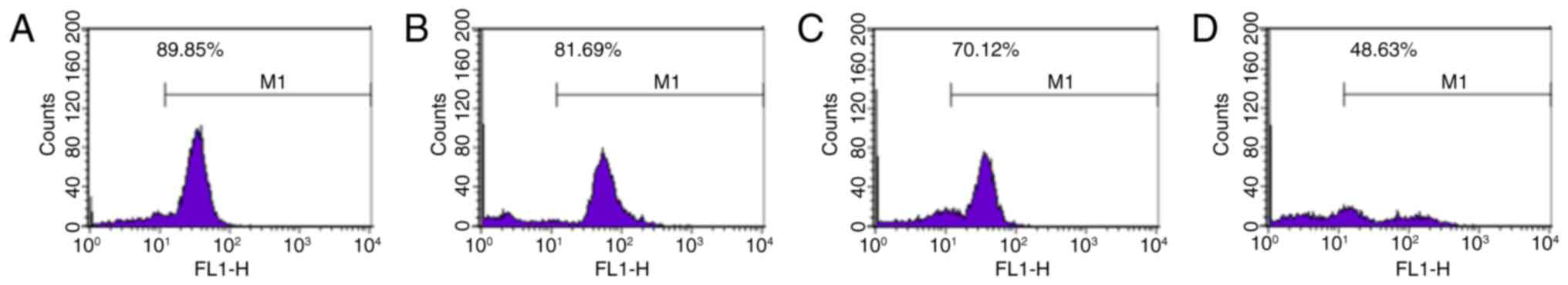
Ginsenoside Rg5 decreases the
mitochondrial transmembrane potential of Eca-109 cells
The decrease in mitochondrial transmembrane
potential serves an important role in the progression of apoptosis.
Rhodamine 123 is a lipophilic cationic fluorescent probe that can
pass through a living cell membrane. Rhodamine 123 has a positive
charge due to electrical adsorption and can be used to specifically
label the mitochondria (19). In
the present study, the results revealed that treatment with
ginsenoside Rg5 at 5, 10 and 20 µM significantly reduced the
mitochondrial transmembrane potential in Eca-109 cells
(81.69±1.414, 70.12±1.957 and 48.63±1.224%, respectively) when
compared with the control group (89.85±1.829%; P<0.05; Fig. 4).
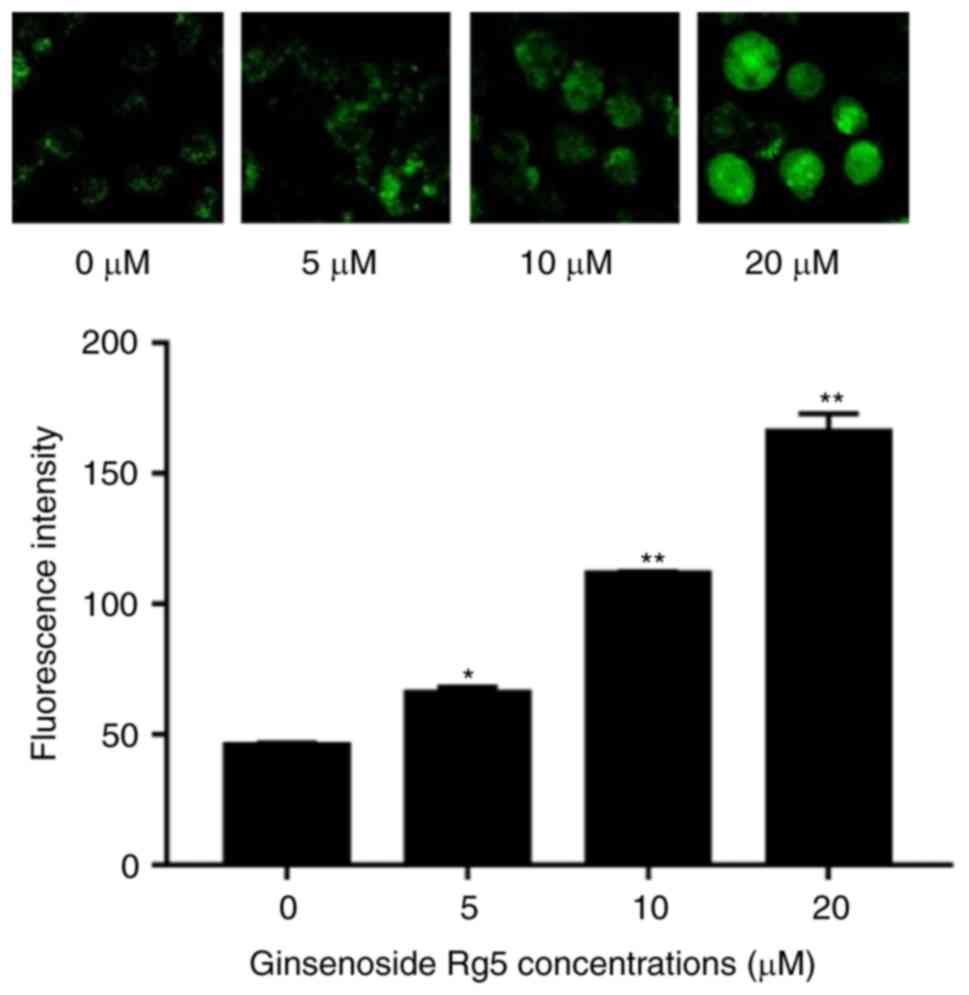
Ginsenoside Rg5 increases cytoplasmic
calcium levels in Eca-109 cells
As a major intracellular messenger, calcium is
involved in regulating the physiological activities of numerous
types of cells and tissues. The fluorescent probe Fluo-3/AM enters
the cell and generates fluorescence at the excitation wavelength of
488 nm, and its intensity is proportional to the concentration of
intracellular free calcium ions (20). The results of the current study
revealed that, following 24-h treatment with Rg5, the cytoplasmic
calcium levels of Eca-109 cells were significantly increased in a
dose-dependent manner, compared with the control group levels, as
observed by the enhanced fluorescence intensity (Fig. 5).
Inhibition of the PI3K/Akt signaling
pathway induces the anti-apoptotic effects of Rg5, while activation
of the pathway inhibits this effect
The present study then examined whether the PI3K/Akt
signaling pathway is important in the anti-apoptotic effect of Rg5.
Eca-109 cells were treated with the PI3K inhibitor LY294002 or with
the PI3K activator IGF-1. Co-treatment with ginsenoside Rg5 and
LY294002 induced apoptosis when compared with the group treated
with Rg5 alone (Fig. 6A), while it
also markedly increased the levels of cleaved caspase-9 and cleaved
PARP (Fig. 6B). In addition,
LY294002 significantly lowered the expression levels of p-Akt and
Bcl-2. Furthermore, co-treatment with IGF-1 markedly inhibited the
anti-apoptotic effect of Rg5 (Fig.
6A). The cleavage of caspase-9 and PARP caused by Rg5 was
weakened by IGF-1; however, the decline in p-Akt and Bcl-2 protein
expression was reversed (Fig. 6B).
These results indicated that ginsenoside Rg5 induced Eca-109 cell
apoptosis via the PI3K/Akt signaling pathway.
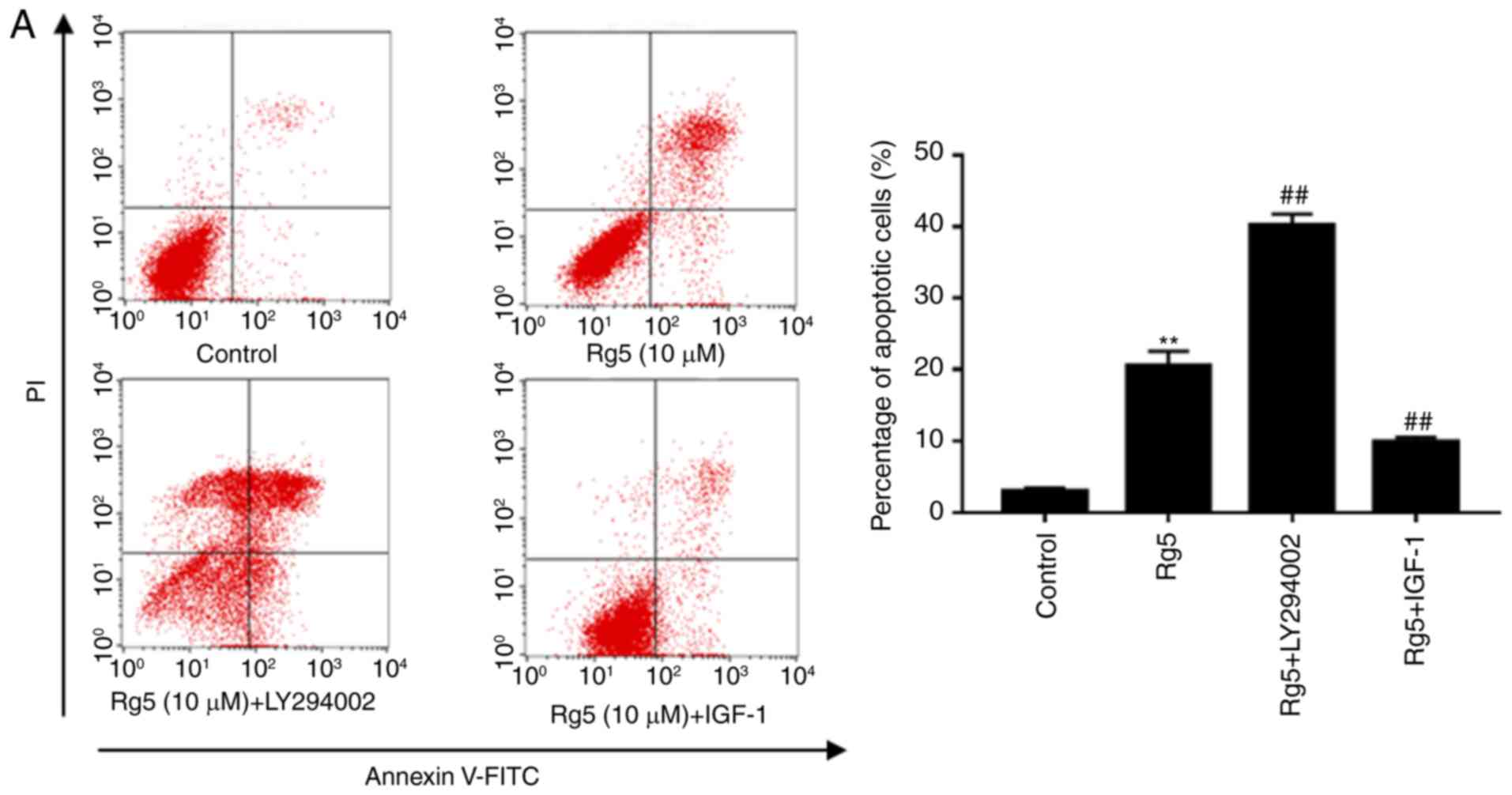 | Figure 6.LY294002 enhances the anti-apoptotic
effects of Rg5 and IGF-1 inhibits these effects. (A) Annexin
V-FITC/PI apoptosis assay kit was used to determine the apoptotic
rate of the untreated, Rg5-treated (10 µM), LY294002-treated (20
µM) and IGF-1-treated (100 ng/ml) groups following treatment for 24
h. (B) Cleaved caspase-9, cleaved PARP, Bcl-2, Bax, Akt and p-Akt
protein expression levels were analyzed by western blot analysis.
The quantified results are presented in terms of the ratio of
cleaved caspase-9 to β-actin, Bcl-2 to Bax, and p-Akt to Akt. Data
are presented as the mean ± standard deviation. *P<0.05 and
**P<0.01, vs. untreated group; #P<0.05 and
##P<0.01, vs. the Rg5-treated group. IGF-1,
insulin-like growth factor 1; FITC, fluorescein isothiocyanate; PI,
propidium iodide; PARP, poly(adenosine diphosphate-ribose)
polymerase; Bcl-2, B-cell lymphoma 2; Bax, Bcl-2-associated X
protein; p-, phosphorylated; Akt, protein kinase B. |
Discussion
Recently, a number of traditional Chinese medicine
agents have been utilized for combination and adjuvant tumor
treatment in order to reduce the side effects of therapy. With the
development of molecular biology and in-depth research, the
anti-tumor mechanism of ginseng is now better understood. In total,
46 potential target genes of (20S) G-Rh2 have been identified by
performing phage display screening with the T7 select human liver
tumor cDNA phage library (21).
This indicates that ginsenosides may be multi-targeting drugs,
triggering various anti-tumor pathways.
Several previous studies have confirmed that
ginsenosides exert an anti-tumor effect through apoptotic signaling
pathways. For instance, it has been reported that ginsenoside Rg3
induces apoptosis in human glioblastoma cells through the
mitogen-activated protein kinase kinase signaling pathway (22). In addition, apoptosis is induced in
cervical cancer cells via the mitochondrial pathway by the compound
JRS-15 (23). The anti-tumor
effect of Rg5 in cervical cancer (24), breast cancer (25) and hepatoma (9) cells has also been explored
previously; however, to the best of our knowledge, no previous
studies have investigated esophageal cancer cells. In addition to
research into its anti-tumor effects, the protective effect of
ginsenoside Rg5 has received increasing attention in recent years.
Previous studies have demonstrated that ginsenoside Rg5
significantly improved cisplatin-associated chemotherapy-induced
nephrotoxicity (26). Its roles in
improving immunity and protecting the heart have also been
confirmed (27,28). Combined with the current research
in progress, it is clear that ginsenoside Rg5 serves a wide range
of protective anti-tumor effects and has great potential for
clinical application as an anti-tumor drug. The aim of the present
study was to elucidate the apoptotic effect of Rg5 on esophageal
cancer cells and its possible mechanism. In the present study, it
was identified that Rg5 inhibited the proliferation of esophageal
cancer cells and promoted apoptosis. Significant increases in the
activity of caspase-3, −8, −9 and PARP were observed.
It has been reported that the decrease in the
mitochondrial transmembrane potential is associated with intrinsic
apoptotic pathways that increase the permeability due to the
release of cytochrome c in the cytoplasm (29). The mitochondrial permeability
transition pore (MPTP) is located in the mitochondrial inner and
outer membrane, and is composed of a variety of protein
non-selective complexes (30). Its
periodic opening serves an important role in maintaining
homeostasis and electrochemical balance in the mitochondria. When
the mitochondrial transmembrane potential decreases, apoptosis is
eventually induced in cells following opening of the MPTP. This
indicates that the decrease in transmembrane potential may be
associated with the opening of the MPTP (31). Prolonged opening of the MPTP leads
to the reduction of the H+ concentration gradient inside
and outside of the mitochondrial membrane, while the transmembrane
potential gradually decreases (32). The respiratory chain is uncoupled
and the synthesis of adenosine triphosphate (ATP) is interrupted,
further promoting the release of cytochrome c, apoptosis-inducing
factor and protein kinases, such as Akt, into the cytoplasm
(33). In the present study,
ginsenoside Rg5 treatment induced a significant decrease in the
mitochondrial transmembrane potential of Eca-109 cells, thereby
promoting apoptosis via intrinsic pathways, as measured by flow
cytometry.
Ca2+ serves a major role in triggering
mitosis in various types of cells, as well as in regulating cell
death (34). Low cytoplasmic
Ca2+ concentration is critical for maintaining normal
cellular function, whereas an overload of cellular Ca2+
is highly toxic, leading to extensive activation of proteases and
phospholipases (35). Mitochondria
and ATPase pumps serve an important role in the preservation of
Ca2+ levels and are closely associated with the release
of Ca2+. The release of intracellular Ca2+ or
increased Ca2+ influx from the mitochondria or
endoplasmic reticulum may serve as an apoptotic signal, thereby
leading to apoptosis, particularly via extrinsic pathways. While
the release of Ca2+ may lead to the breakdown of
organelles, activation of apoptosis-associated transcription
factors triggers apoptosis (36).
The present study detected changes in Ca2+ levels using
Fluo-3/AM, which can enter into the cell and generate fluorescence
at the excitation wavelength of 488 nm, following binding with
intracellular free calcium ions. The results of the present study
revealed that Rg5 increased the intracellular Ca2+
levels and caused cell apoptosis.
The PI3K/Akt signaling pathway is associated with
numerous cellular functions, including cell viability,
proliferation and apoptosis, while it is also involved in cell
survival pathways (37,38). Inhibition of this pathway can
decrease the phosphorylation of Akt (39). The PI3K/Akt signaling pathway is
normally activated in human cancer. Previous studies have reported
that the inhibition of PI3K/Akt signaling pathway activity can
effectively inhibit the proliferation and invasion of breast cancer
cells (40). Furthermore,
inhibition of PI3K/Akt signaling in lung cancer induces apoptosis
and arrests the cell cycle (41).
The present study further revealed that Rg5 treatment resulted in a
concentration-dependent decrease in the level of p-Akt in Eca109
cells. This may indicate that the pro-apoptotic effect of Rg5 is
associated with the inhibition of the PI3K/Akt pathway. To further
determine whether the PI3K/Akt signaling pathway is important in
the apoptosis of esophageal cancer cells, Eca-109 cells were
co-treated with Rg5 and the PI3K inhibitor LY294002, and the
expression levels of apoptosis markers were detected. Compared with
Rg5 alone, co-treatment with LY294002 significantly inhibited the
level of p-Akt, and in addition, significantly decreased the
expression of Bcl2; however, it also induced the expression of
cleaved caspase-9. The PI3K promoter IGF-1 was also used to treat
cells to determine whether it was able to reverse Rg5-induced
apoptosis. The results revealed that IGF-1 treatment reversed the
decrease in p-Akt induced by Rg5, while it significantly reversed
the decrease in Bcl-2 expression and increase in cleaved caspase-9
expression when compared with Rg5 treatment alone.
In conclusion, the present study revealed that
ginsenoside Rg5 inhibited the proliferation and induced the
apoptosis of human esophageal cancer cells. In addition, its role
in promoting tumor apoptosis was associated with the downregulation
of the PI3K/Akt signaling pathway. However, the present study had
certain limitations. The specific signaling pathways involved in
ginsenoside Rg5-induced apoptosis in esophageal cancer cells
require further verification. Furthermore, in vivo
experiments are required to understand the anti-tumor effects of
Rg5 and to conduct pharmacokinetic studies.
Acknowledgements
The authors would like to thank Professor Yinghua
Jin (College of Life Science, Jilin University, Changchun, China)
for providing ginsenoside Rg5.
Funding
The present study was supported by the Scientific
and Technical Foundation of Jilin Province (grant no.
20160204022YY).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LL and HR, DZ conceived and designed the study. DZ,
AW and JF performed the experiments. QZ and DZ analyzed the data.
DZ and LL wrote the manuscript. All authors read and approved the
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Yun TK, Lee YS, Lee YH, Kim SI and Yun HY:
Anticarcinogenic effect of Panax ginseng C.A. Meyer and
identification of active compounds. J Korean Med Sci. 16
(Suppl):S6–S18. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
De Souza LR, Jenkins AL, Sievenpiper JL,
Jovanovski E, Rahelić D and Vuksan V: Korean red ginseng (Panax
ginseng C.A. Meyer) root fractions: Differential effects on
postprandial glycemia in healthy individuals. J Ethnopharmacol.
137:245–250. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Varjas T, Nowrasteh G, Budán F, Nadasi E,
Horváth G, Makai S, Gracza T, Cseh J and Ember I: Chemopreventive
effect of Panax ginseng. Phytother Res. 23:1399–1403. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Helms S: Cancer prevention and
therapeutics: Panax ginseng. Altern Med Rev. 9:259–274.
2004.PubMed/NCBI
|
|
5
|
Shin HR, Kim JY, Yun TK, Morgan G and
Vainio H: The cancer-preventive potential of Panax ginseng: A
review of human and experimental evidence. Cancer Causes Control.
11:565–576. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kim SN, Ha YW, Shin H, Son SH, Wu SJ and
Kim YS: Simultaneous quantification of 14 ginsenosides in Panax
ginseng C.A. Meyer (Korean red ginseng) by HPLC-ELSD and its
application to quality control. J Pharm Biomed Anal. 45:164–170.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Nag SA, Qin JJ, Wang W, Wang MH, Wang H
and Zhang R: Ginsenosides as anticancer agents: In vitro and in
vivo activities, structure-activity relationships, and molecular
mechanisms of action. Front Pharmacol. 3:252012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee KY, Lee YH, Kim SI, Park JH and Lee
SK: Ginsenoside-Rg5 suppresses cyclin E-dependent protein kinase
activity via up-regulating p21Cip/WAF1 and down-regulating cyclin E
in SK-HEP-1 cells. Anticancer Res. 17:1067–1072. 1997.PubMed/NCBI
|
|
9
|
Shin YW, Bae EA and Kim DH: Inhibitory
effect of ginsenoside Rg5 and its metabolite ginsenoside Rh3 in an
oxazolone-induced mouse chronic dermatitis model. Arch Pharm Res.
29:685–690. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kim EJ, Jung IH, Van Le TK, Jeong JJ, Kim
NJ and Kim DH: Ginsenosides Rg5 and Rh3 protect scopolamine-induced
memory deficits in mice. J Ethnopharmacol. 146:294–299. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lee YY, Park JS, Jung JS, Kim DH and Kim
HS: Anti-inflammatory effect of ginsenoside Rg5 in
lipopolysaccharide-stimulated BV2 microglial cells. Int J Mol Sci.
14:9820–9833. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ferlay J, Soerjomataram I, Dikshit R, Eser
S, Mathers C, Rebelo M, Parkin DM, Forman D and Bray F: Cancer
incidence and mortality worldwide: Sources, methods and major
patterns in GLOBOCAN 2012. Int J Cancer. 136:E359–E386. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Song Y, Li L, Ou Y, Gao Z, Li E, Li X,
Zhang W, Wang J, Xu L, Zhou Y, et al: Identification of genomic
alterations in oesophageal squamous cell cancer. Nature. 509:91–95.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Tachibana M, Kinugasa S, Hirahara N and
Yoshimura H: Lymph node classification of esophageal squamous cell
carcinoma and adenocarcinoma. Eur J Cardiothorac Surg. 34:427–431.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ouyang L, Shi Z, Zhao S, Wang FT, Zhou TT,
Liu B and Bao JK: Programmed cell death pathways in cancer: A
review of apoptosis, autophagy and programmed necrosis. Cell
Prolif. 45:487–498. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Streeter OE Jr, Martz KL, Gaspar LE,
Delrowe JD, Asbell SO, Salter MM and Roach M III: Does race
influence survival for esophageal cancer patients treated on the
radiation and chemotherapy arm of RTOG #85-01? Int J Radiat Oncol
Biol Phys. 44:1047–1052. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Danial NN and Korsmeyer SJ: Cell death:
Critical control points. Cell. 116:205–219. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Schönfelder U, Radestock A, Elsner P and
Hipler UC: Cyclodextrin-induced apoptosis in human keratinocytes is
caspase-8 dependent and accompanied by mitochondrial cytochrome c
release. Exp Dermatol. 15:883–890. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kale J, Liu Q, Leber B and Andrews DW:
Shedding light on apoptosis at subcellular membranes. Cell.
151:1179–1184. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Robinson JA, Jenkins NS, Holman NA,
Roberts-Thomson SJ and Monteith GR: Ratiometric and nonratiometric
Ca2+ indicators for the assessment of intracellular free Ca2+ in a
breast cancer cell line using a fluorescence microplate reader. J
Biochem Biophys Methods. 58:227–237. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang YS, Lin Y, Li H, Li Y, Song Z and Jin
YH: The identification of molecular target of (20S) ginsenoside Rh2
for its anti-cancer activity. Sci Rep. 7:124082017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Choi YJ, Lee HJ, Kang DW, Han IH, Choi BK
and Cho WH: Ginsenoside Rg3 induces apoptosis in the U87MG human
glioblastoma cell line through the MEK signaling pathway and
reactive oxygen species. Oncol Rep. 30:1362–1370. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sun C, Guo XX, Zhu D, Xiao C, Bai X, Li Y,
Zhan Z, Li XL, Song ZG and Jin YH: Apoptosis is induced in cancer
cells via the mitochondrial pathway by the novel xylocydine-derived
compound JRS-15. Int J Mol Sci. 14:850–870. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liang LD, He T, Du TW, Fan YG, Chen DS and
Wang Y: Ginsenoside-Rg5 induces apoptosis and DNA damage in human
cervical cancer cells. Mol Med Rep. 11:940–946. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kim SJ and Kim AK: Anti-breast cancer
activity of Fine Black ginseng (Panax ginseng Meyer) and
ginsenoside Rg5. J Ginseng Res. 39:125–134. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Li W, Yan MH, Liu Y, Liu Z, Wang Z, Chen
C, Zhang J and Sun YS: Ginsenoside Rg5 Ameliorates
Cisplatin-induced nephrotoxicity in mice through inhibition of
inflammation, oxidative Stress, and apoptosis. Nutrients. 8(pii):
E5662016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yang YL, Li J, Liu K, Zhang L, Liu Q, Liu
B and Qi LW: Ginsenoside rg5 increases cardiomyocyte resistance to
ischemic injury through regulation of mitochondrial hexokinase-II
and dynamin-related protein 1. Cell Death Dis. 8:e26252017.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shin MS, Song JH, Choi P, Lee JH, Kim SY,
Shin KS, Ham J and Kang KS: Stimulation of innate immune function
by Panax ginseng after heat processing. J Agric Food Chem.
66:4652–4659. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Heiskanen KM, Bhat MB, Wang HW, Ma J and
Nieminen A: Mitochondrial depolarization accompanies cytochrome c
release during apoptosis in PC6 cells. J Biol Chem. 26:5654–5658.
1999. View Article : Google Scholar
|
|
30
|
Chen H, Xu J, Lv Y, He P, Liu C, Jiao J,
Li S, Mao X and Xue X: Proanthocyanidins exert a neuroprotective
effect via ROS/JNK signaling in MPTP-induced Parkinson's disease
models in vitro and in vivo. Mol Med Rep. 18:4913–4921.
2018.PubMed/NCBI
|
|
31
|
Rasheed MZ, Tabassum H and Parvez S:
Mitochondrial permeability transition pore: A promising target for
the treatment of Parkinson's disease. Protoplasma. 254:33–42. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wu HY, Huang CH, Lin YH, Wang CC and Jian
TR: Cannabidiol induced apoptosis in human monocytes through
mitochondrial permeability transition pore-mediated ROS production.
Free Radic Biol Med. 124:311–318. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shibayama-Imazu T, Aiuchi T and Nakaya K:
Vitamin K2-mediated apoptosis in cancer cells: Role of
mitochondrial transmembrane potential. Vitam Horm. 78:211–226.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Giorgi C, Romagnoli A, Pinton P and
Rizzuto R: Ca2+ signaling, mitochondria and cell death. Curr Mol
Med. 8:119–130. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pinton P, Giorgi C, Siviero R, Zecchini E
and Rizzuto R: Calcium and apoptosis: ER-mitochondria Ca2+ transfer
in the control of apoptosis. Oncogene. 27:6407–6418. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Oppenheim R: Naturally occurring cell
death during neural development. Trends Neurosci. 8:487–493. 1985.
View Article : Google Scholar
|
|
37
|
Granado-Serrano AB, Martin MA, Bravo L,
Goya L and Ramos S: Quercetin induces apoptosis via caspase
activation, regulation of Bcl-2, and inhibition of PI-3-kinase/Akt
and ERK pathways in a human hepatoma cell line (HepG2). Nutr.
136:2715–2721. 2006. View Article : Google Scholar
|
|
38
|
Xiang T, Fang Y and Wang SX: Quercetin
suppresses HeLa cells by blocking PI3K/Akt pathway. J Huazhong Univ
Sci Technol Med Sci. 34:740–744. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Wong MH, Xue A, Baxter RC, Pavlakis N and
Smith RC: Upstream and downstream co-inhibition of
mitogen-activated protein kinase and PI3K/Akt/mTOR pathways in
pancreatic ductal adenocarcinoma. Neoplasia. 18:425–435. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Dillon LM, Bean JR, Yang W, Shee K,
Symonds LK, Balko JM, McDonald WH, Liu S, Gonzalez-Angulo AM, Mills
GB, et al: P-REX1 creates a positive feedback loop to activate
growth factor receptor, PI3K/AKT and MEK/ERK signaling in breast
cancer. Oncogene. 34:3968–3976. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Riquelme E, Behrens C, Lin HY, Simon G,
Papadimitrakopoulou V, Izzo J, Moran C, Kalhor N, Lee JJ, Minna JD
and Wistuba II: Modulation of EZH2 expression by MEK-ERK or
PI3K-AKT Signaling in lung cancer is Dictated by different KRAS
oncogene mutations. Cancer Res. 76:675–685. 2016. View Article : Google Scholar : PubMed/NCBI
|