Introduction
The dental follicle, originating from the cranial
neural crest, is a loose ectomesenchyme-derived tissue, which
surrounds the dental papilla and enamel organ during the
development of teeth. The dental follicle develops into periodontal
supporting tissues, which serve important functions, including in
support, buffering, rebuilding and regeneration (1). hDFCs exhibit a high capacity for
proliferation, self-renewal and multi-directional differentiation,
and form structures, including the cementum and periodontal
ligament (PDL), when subcutaneously transplanted into
immunodeficient mice (2,3). hDFCs share a similar phenotype with
human PDLCs (hPDLCs), including a mineralization ability, and they
also exhibit apparent embryonic characteristics, including
pluripotency, heterogeneity and a higher potential for cementum
formation in vivo (4–6). It
has been suggested that hDFCs may provide a new source of seed
cells for stem cell therapy and periodontal tissue engineering.
Therefore, understanding the key target genes and underlying
molecular mechanisms of hDFC differentiation is required for
promoting periodontal development and regeneration.
Long non-coding RNAs (lncRNAs) are defined as
non-protein coding RNA molecules that are >200 nucleotides long
(7). lncRNAs perform their
biological effects through a number of mechanisms, including
genetic imprinting, chromatin remodeling, cell cycle regulation,
splicing and mRNA inactivation. lncRNAs control the pluripotency
and stemness of embryonic stem cells and induced pluripotent stem
cells, or promote the differentiation of pluripotent cells in the
opposite manner. Additionally, lncRNAs may transcriptionally or
post-transcriptionally regulate gene expression via different
molecular mechanisms (8,9).
An increasing number of studies indicate that
lncRNAs serve critical roles in the development of organs,
including the brain (10), heart
(11), liver (12), lungs (13) and bone (14). lncRNAs also serve significant roles
in tooth development. For example, the lncRNA differentiation
antagonizing non-protein coding RNA (DANCR) serves a role in
reparative dentin formation and regenerative endodontics (15). DANCR is an essential mediator in
the proliferation and differentiation of dental tissue-derived stem
cells, including dental pulp stem cells, stem cells from the apical
papilla and periodontal ligament stem cells (PDLSCs) (16). hDFCs and hPDLCs are essential cells
in different stages of periodontal development. However, it remains
unclear what potential roles lncRNAs serve in periodontal
development and whether lncRNAs are involved in specific activities
in different cells. Therefore the current study used microarrays to
obtain the different expression profiles of lncRNAs and mRNAs
between hDFCs and hPDLCs. Furthermore, the microarray data were
validated by reverse transcription-quantitative polymerase chain
reaction (RT-qPCR). Bioinformatics analyses were performed to
predict the possible roles of the differentially expressed lncRNAs
and mRNAs in periodontal development. The results demonstrated that
lncRNAs may serve critical roles in periodontal development, and
provided a solid foundation for further research.
Materials and methods
Cell culture
Human dental follicle and periodontal ligament
samples were obtained from four adolescents (2 males and 2 females)
between 12 and 16 years old following premolar and immature
impacted third molar (roots developed to <2/3 their full size)
extraction for orthodontic reasons. No significant differences were
identified in age or sex. Participants included in the study had no
history of systemic disease, smoking or specific medication. Tooth
extraction was performed at the Hospital of Stomatology, Sun
Yat-Sen University (Guangzhou, China) between June 2017 and July
2017. All experimental protocols were conducted under the
guidelines set by the Sun Yat-Sen University Ethics Committee and
written informed consent was obtained from all patients and their
parents. hDFCs and hPDLCs were isolated as previously described
(17,18). Briefly, dental follicle tissues
were gently removed with a scalpel from where they attached to the
root dentin and were digested in a solution containing 1 U/ml
collagenase type I (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany),
hyaluronidase (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) and 1
U/ml DNase I (Roche Applied Science, Mannheim, Germany).
Periodontal ligament tissues were isolated from the middle
one-third of the root surface and digested in a solution of 3 mg/ml
collagenase type I (Sigma-Aldrich; Merck KGaA) and 4 mg/ml dispase
(Sigma-Aldrich; Merck KGaA). hDFCs and hPDLCs were cultured in
Dulbecco's modified Eagle's medium supplemented with 20% fetal
bovine serum (both Gibco; Thermo Fisher Scientific, Inc., Waltham,
MA, USA), 100 U/ml penicillin and 100 µg/ml streptomycin
(Sigma-Aldrich; Merck KGaA) at 37°C in 5% CO2. Every 2–3
days, the medium was replaced. When cells reached 80% confluence,
they were passaged at a 1:3 ratio using 0.25% trypsin/EDTA (Gibco;
Thermo Fisher Scientific, Inc.). Third-generation cells were used
for the subsequent experiments.
RNA extraction
Total RNA was extracted using TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.).
Quantification and quality checks were conducted using a NanoDrop
ND-1000 spectrophotometer (Thermo Fisher Scientific, Inc.). RNA
integrity and genomic DNA contamination were determined by
denaturing agarose gel electrophoresis.
RNA microarray
The total RNA was purified using an RNeasy Mini kit
(Qiagen GmbH, Hilden, Germany) and labeled with a Quick Amp
Labeling kit (Agilent Technologies, Inc., Santa Clara, CA, USA).
Labeled RNA was purified again using the RNeasy Mini kit. The RNA
was subsequently hybridized onto an Arraystar Human lncRNA
Expression Microarray (version 4.0; Arraystar, Inc., Rockville, MA,
USA), which was designed for 30,586 lncRNAs and 26,109 coding genes
based on the RefSeq (https://ncbi.nlm.nih.gov/refseq/), UCSC Known Genes
and Gencode (https://genome.ucsc.edu/) and Ensembl
databases (http://ensemblgenomes.org/). Agilent
Feature Extraction software (version 11.0.1.1; Agilent
Technologies, Santa Clara, CA, US) was used to analyze the acquired
array images. The microarray data were deposited in the Gene
Expression Omnibus (GEO; http://www.ncbi.nlm.nih.gov/geo/) and are accessible
through GEO Series accession no. GSE124352. Differentially
expressed lncRNAs and mRNAs were identified to be statistically
significant (fold change >2.0 or <-2.0; P<0.05) using a
paired t-test. The microarray was performed by Kangchen BioTech
Co., Ltd. (Shanghai, China). Subgroup analysis was conducted to
classify the differentially expressed lncRNAs according to their
expression levels.
RT-qPCR analysis
Total RNA of hDFCs and hPDLCs was reverse
transcribed into complementary DNA using an RT kit (Takara Bio,
Inc., Otsu, Japan), conducted at 37°C for 15 min and 85°C for 5
sec. In total, six lncRNAs and six mRNAs were randomly selected for
RT-qPCR analysis using the RANDBETWEEN function in Microsoft Excel
(version 2010, Microsoft Corporation, Redmond, WA, USA). RT-qPCR
was performed using SYBR Green Real-Time PCR Master mix
(Invitrogen; Thermo Fisher Scientific, Inc.), and was run at 95°C
for 5 min, followed by 40 cycles at 95°C for 10 sec, 65°C for 20
sec and 72°C for 30 sec. Primers were synthesized by Invitrogen;
Thermo Fisher Scientific, Inc. (Table
I). The characteristics of the lncRNAs are presented in
Table II. Data were normalized to
GAPDH and the relative level of gene expression was calculated
using the 2−ΔΔCq method (19).
 | Table I.Primers used for the reverse
transcription-quantitative polymerase chain reaction. |
Table I.
Primers used for the reverse
transcription-quantitative polymerase chain reaction.
| Gene name | Forward
(5′-3′) | Reverse
(5′-3′) |
|---|
| NR_033917 |
TCACTGTCATGCACTAGCGG |
CAGCCCTTGAGGTTGTCCTT |
| NR_038367 |
CCCACCGTTCAATGAAAG |
GTTTCAAACACCCACATTTC |
| NR_026861 |
ACTCGCCTTTTGCGAGAAGA |
TCGCAGTTTCCTAGTGGGAC |
| NR_102703 |
AGCTGTGAACGGTAGCAGTG |
AGGCAGTTTTGTGGGTCAGT |
| NR_110162 |
GGCCCATTCCTGTGAATCGT |
GTGACTCAAAGCAGCAGAAGC |
|
ENST00000430859 |
CCCCTTAGCCTGCTTGTGAT |
CCAGTGAAAACTGCACAGAGC |
| KCNK12 |
CATGTACACCAGCGTGGAGG |
AGCGAGTAAATGCAGCACAC |
| CCL11 |
TCCCTGGAATCTCCCACACT |
CACTCAGGCTCTGGTTTGGT |
| MDK |
TCAGACCGGTTCTGGAGACA |
TTTGCTTTGGTCTTGGGGGT |
| SAA2 |
GAGTGGCAGAGACCCCAATC |
TCAGCTTCTCTGGACATAGACC |
| MGST1 |
CGGCCTCACCATTCCAGAC |
CAAGGTCATTCAGGTGGGCT |
| HIST1H2BG |
GCAGTGTCCGAAGGTACCAA |
TGTGAGACTTGAGTGGCTCTG |
| GAPDH |
CTGGGCTACACTGAGCACC |
AAGTGGTCGTTGAGGGCAATG |
 | Table II.Characteristics of lncRNAs for
validation. |
Table II.
Characteristics of lncRNAs for
validation.
| lncRNA | Expression | Chromosome | Strand | Start | End | Class |
|---|
| NR_033917 | Down | Chr20 | + | 4173736 | 4176600 | Intergenic |
| NR_038367 | Down | Chr7 | + | 27135712 | 27139877 | Antisense |
| NR_026861 | Down | Chr6 | − | 166337535 | 166401527 | Intergenic |
| NR_102703 | Up | Chrx | − | 149007562 | 149025779 | Undefined |
| NR_110162 | Up | Chr7 | − | 112594689 | 112635698 | Undefined |
|
ENST00000430859 | Up | Chr7 | + | 20257208 | 20261315 | Bidirectional |
Bioinformatics analyses
Gene ontology (GO; http://geneontology.org/) was conducted to analyze the
functions of the differentially expressed genes. GO analysis
examines gene regulatory networks based on biological processes,
cellular components and molecular functions (20). A χ2 test and a two-sided
Fisher's exact test were used to classify the GO category.
Enrichment of the GO category was determined by the significance of
the functions. Kyoto Encyclopedia of Genes and Genomes (https://www.genome.jp/kegg/) pathway analysis of the
differentially expressed mRNAs was performed to identify the
associated pathways. Connections between the genes were revealed
based on their associations in the identified pathways. In
addition, a coding-non-coding gene co-expression (CNC) network was
constructed to investigate the interactions between the
differentially expressed lncRNAs and mRNAs. A number of
differentially expressed mRNAs were identified to be associated
with development. Pearson's correlation coefficient was calculated
for each pair of lncRNAs and mRNAs, and the most significantly
correlated genes (Pearson's correlation coefficient ≥0.90 or
≤-0.90; P<0.05) were selected to construct the network. The
network was generated using Cytoscape software (v2.8.2; http://cytoscape.org/). The default annotation
categories were selected in the software and an enrichment score
>2.0 was considered significant.
Transient transfection
hDFCs were transfected with 50 nM MEG3-small
interfering (si)RNA and NC-siRNA using Lipofectamine®
3000 (Invitrogen; Thermo Fisher Scientific, Inc.) at a density of
1.5×105 cells, according to the manufacturer's protocol.
The MEG3-siRNA and negative control (NC)-siRNA were synthesized by
Guangzhou RiboBio Co., Ltd. (Guangzhou, China). The MEG3-siRNA
sequences were as follows: GACTTAAACCAATGCCCTA,
CCTCTTACCTAAAGACTTA, CCCTCTTGCTTGTCTTACT. The expression of
pluripotency-associated genes was detected by RT-qPCR 72 h
post-transfection.
Statistical analysis
Data are presented as the mean ± standard deviation
(n=3) and were analyzed using a Student's t-test. All statistical
analysis was performed with SPSS 20.0 (IBM Corp., Armonk, NY, USA).
A fold-change >2.0 or <-2.0 and P<0.05 was considered to
indicate a statistically significant difference.
Results
lncRNA and mRNA expression profiles of
hDFCs and hPDLCs
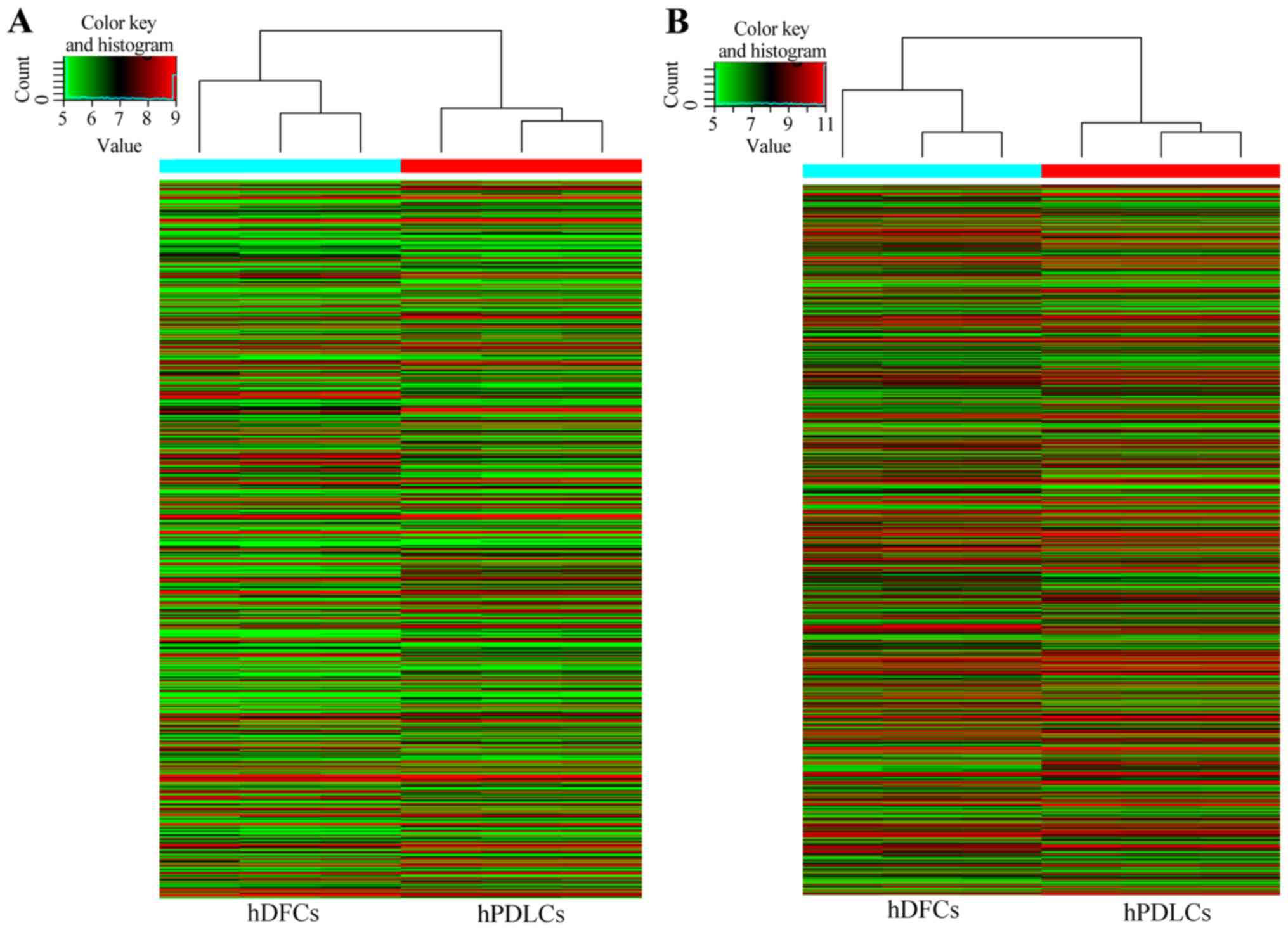
Gene expression patterns of hDFCs and hPDLCs were
screened with the high-throughput microarray method to reveal
potential molecular mechanisms underlying hDFC development into
periodontal tissues. Microarray probes detected 36,430 transcripts
in hDFCs and hPDLCs (Fig. 1). A
total of 845 lncRNAs were identified to be differentially expressed
in the hDFCs and hPDLCs, of which 460 were upregulated and 385 were
downregulated in the hDFCs compared with the hPDLCs. In addition, a
total of 1,012 mRNAs (6.12%) were differentially expressed, 553
mRNAs were upregulated and 459 mRNAs were downregulated in the
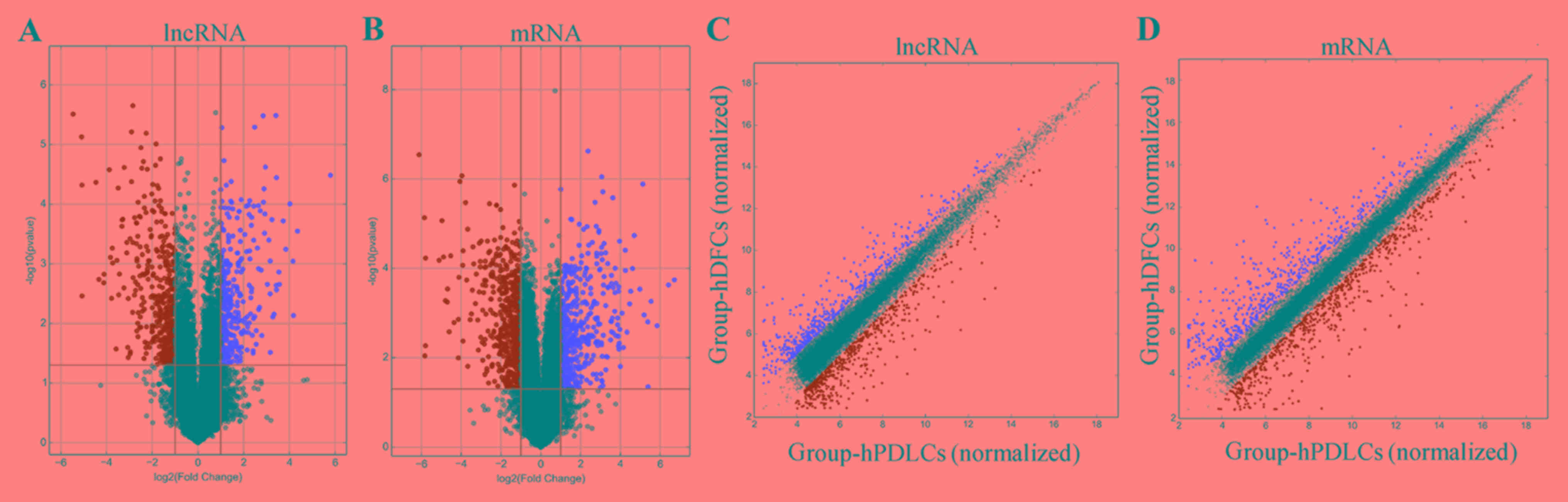
hDFCs compared with the hPDLCs. A scatter plot was generated to
visualize the variation in expression of lncRNAs and mRNAs in the
hDFCs and hPDLCs (Fig. 2A and B).
Additionally, a volcano plot was used to visualize significantly
differentially expressed lncRNAs and mRNAs (Fig. 2C and D). Furthermore, the
microarray results revealed the top ten differentially expressed
lncRNAs and mRNAs (Tables III
and IV).
 | Table III.Top 10 downregulated and upregulated
lncRNAs of human dental follicle cells compared with human
periodontal ligament cells. |
Table III.
Top 10 downregulated and upregulated
lncRNAs of human dental follicle cells compared with human
periodontal ligament cells.
| A,
Downregulated |
|---|
|
|---|
| lncRNA | Log2 fold
change |
|---|
| G005378 | −55.6292005 |
| BC017988 | −20.5510994 |
| FGF13-AS1 | −18.1455729 |
| RP11-368I23.4 | −17.8927488 |
| LINC00473 | −16.2682486 |
| RP11-313F23.4 | −14.3828901 |
| RN7SKP240 | −13.2776605 |
| AP000619.5 | −11.8151652 |
| G075702 | −11.6414154 |
| RP11-256I23.3 | −10.831092 |
|
| B,
Upregulated |
|
| lncRNA | Log2 fold
change |
|
| LINC00944 | 43.9000497 |
| AK055386 | 33.9312365 |
| G048345 | 33.5797092 |
| LOC101929504 | 33.5392967 |
| LINC01021 | 21.9723651 |
| APCDD1L-AS1 | 20.0893837 |
| RP11-13N12.1 | 17.314853 |
| LOC100506457 | 17.1318902 |
| uc.176 | 14.6428966 |
| G060456 | 13.6926091 |
 | Table IV.Top 10 downregulated and upregulated
mRNAs of hDFCs compared with hPDLCs. |
Table IV.
Top 10 downregulated and upregulated
mRNAs of hDFCs compared with hPDLCs.
| A,
Downregulated |
|---|
|
|---|
| mRNA | Log2 fold
change |
|---|
| KRT5 | −105.4128548 |
| SOX11 | −85.9063061 |
| LRP1B | −57.6366695 |
| SLC7A3 | −46.7530193 |
| CTAG2 | −42.1623478 |
| SLITRK6 | −41.7426552 |
| SPATA22 | −36.689978 |
| SHISA2 | −35.1885461 |
| LAMP3 | −31.6925565 |
| MMP13 | −25.9685676 |
|
| B,
Upregulated |
|
| mRNA | Log2 fold
change |
|
| RPTN | 69.3550374 |
| PSG2 | 57.7064349 |
| ADRA2A | 56.7419608 |
| SOST | 55.5946956 |
| FLG | 55.4287499 |
| KCNB1 | 34.5671271 |
| SLC14A1 | 31.308326 |
| MFAP5 | 29.633329 |
| PSG5 | 27.7407034 |
| PSG7 | 25.9549841 |
Subgroup analysis classified the differentially
expressed lncRNAs as intergenic lncRNAs (lincRNAs) and antisense
lncRNAs (ASlncRNAs). The nearby protein-coding genes were
identified to predict potential functions of lncRNAs. A total of
113 lincRNAs and 35 ASlncRNAs, including NR_033932, T152410,
ENST00000512129 and ENST00000540293, were revealed to be located
near known protein-coding genes.
To verify the reliability of the microarray data,
six differentially expressed lncRNAs and six differentially
expressed mRNAs were randomly selected for analysis of their
expression levels by RT-qPCR. Each differentially expressed gene
was numbered and the RANDBETWEEN function in Microsoft Excel was
used. Compared with hPDLCs, hDFCs exhibited decreased expression
levels of the lncRNAs NR_033917, NR_038367 and NR_026861, and the
mRNAs potassium two pore domain channel subfamily K member 12, C-C
motif chemokine ligand 11 and midkine. By contrast, hDFCs
demonstrated increased expression levels of the lncRNAs NR_102703,
NR_110162 and ENST00000430859, and the mRNAs serum amyloid A2,
microsomal glutathione S-transferase 1 and histone cluster 1 H2B
family member g (Fig. 3). These
results were consistent with the microarray data.
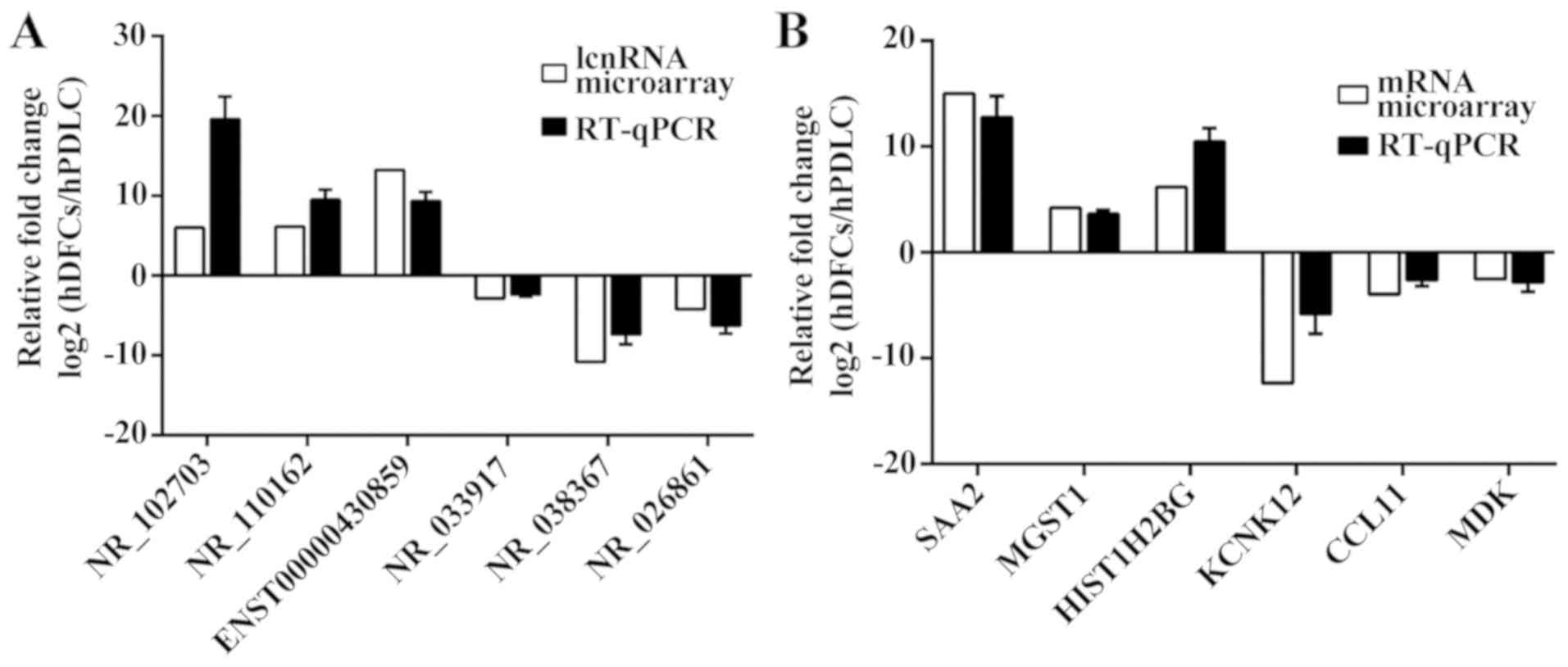 | Figure 3.Validation of the microarray results
using RT-qPCR. (A) Comparative expression levels of lncRNAs between
hDFCs and hPDLCs obtained by microarray and RT-qPCR. (B)
Comparative expression levels of mRNA between hDFCs and hPDLCs
obtained by microarray and RT-qPCR. The microarray analyses were
normalized and the RT-qPCR results were calculated using the
2−ΔΔCq method. SAA2, serum amyloid A2; MGST1, microsomal
glutathione S-transferase 1; HIST1H2BG, histone cluster 1 H2B
family member g; KCNK12, potassium two pore domain channel
subfamily K member 12; CCL11, C-C motif chemokine ligand 11; MDK,
midkine; RT-qPCR, reverse transcription-quantitative polymerase
chain reaction; lncRNA, long non-coding RNA; hDFC, human dental
follicle cell; hPDLC, human periodontal ligament cell. |
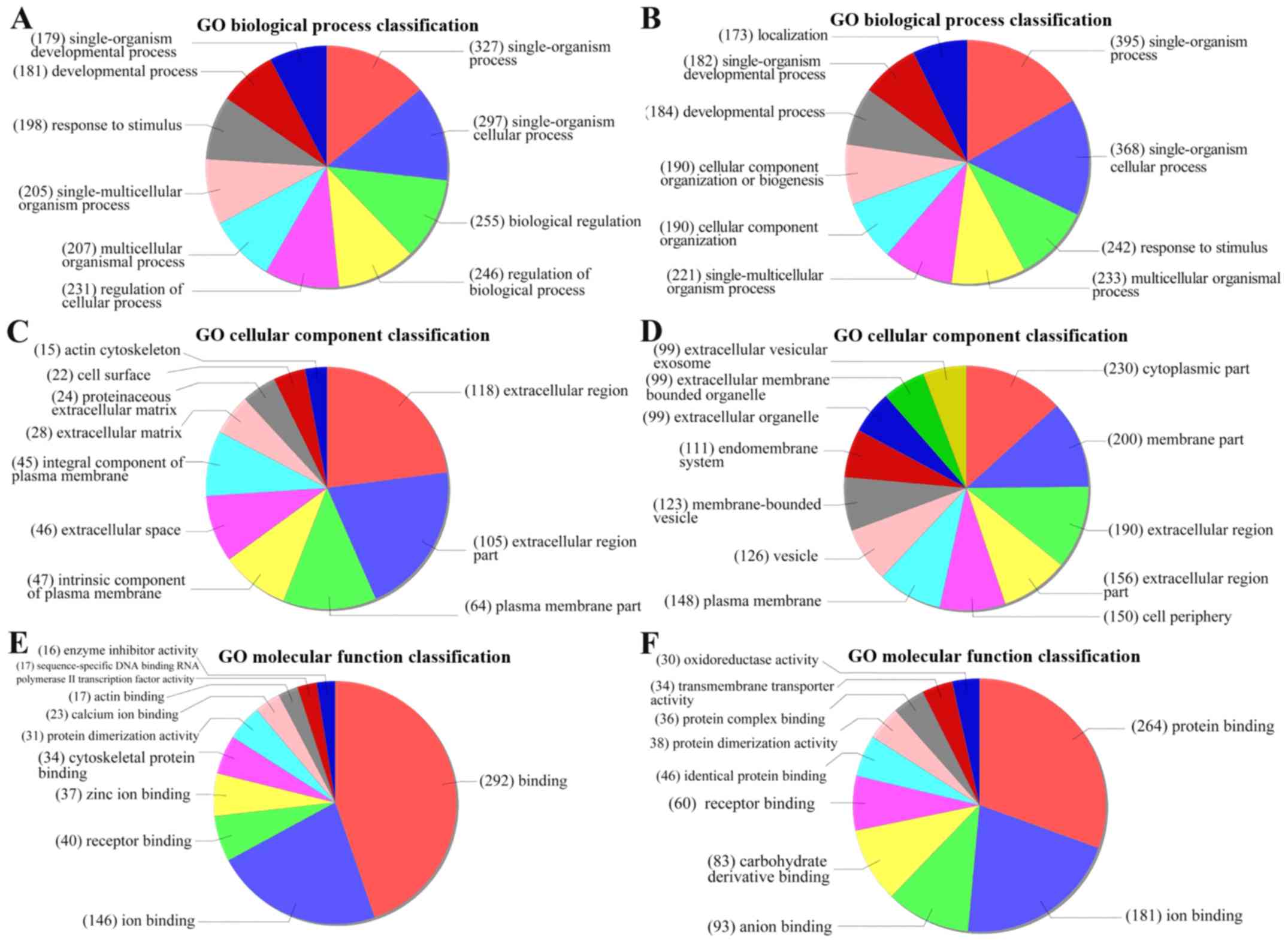
GO analysis and pathway analysis
GO analysis revealed functions associated with the
differentially expressed genes and provided annotations to describe
the genes and gene products (Fig.
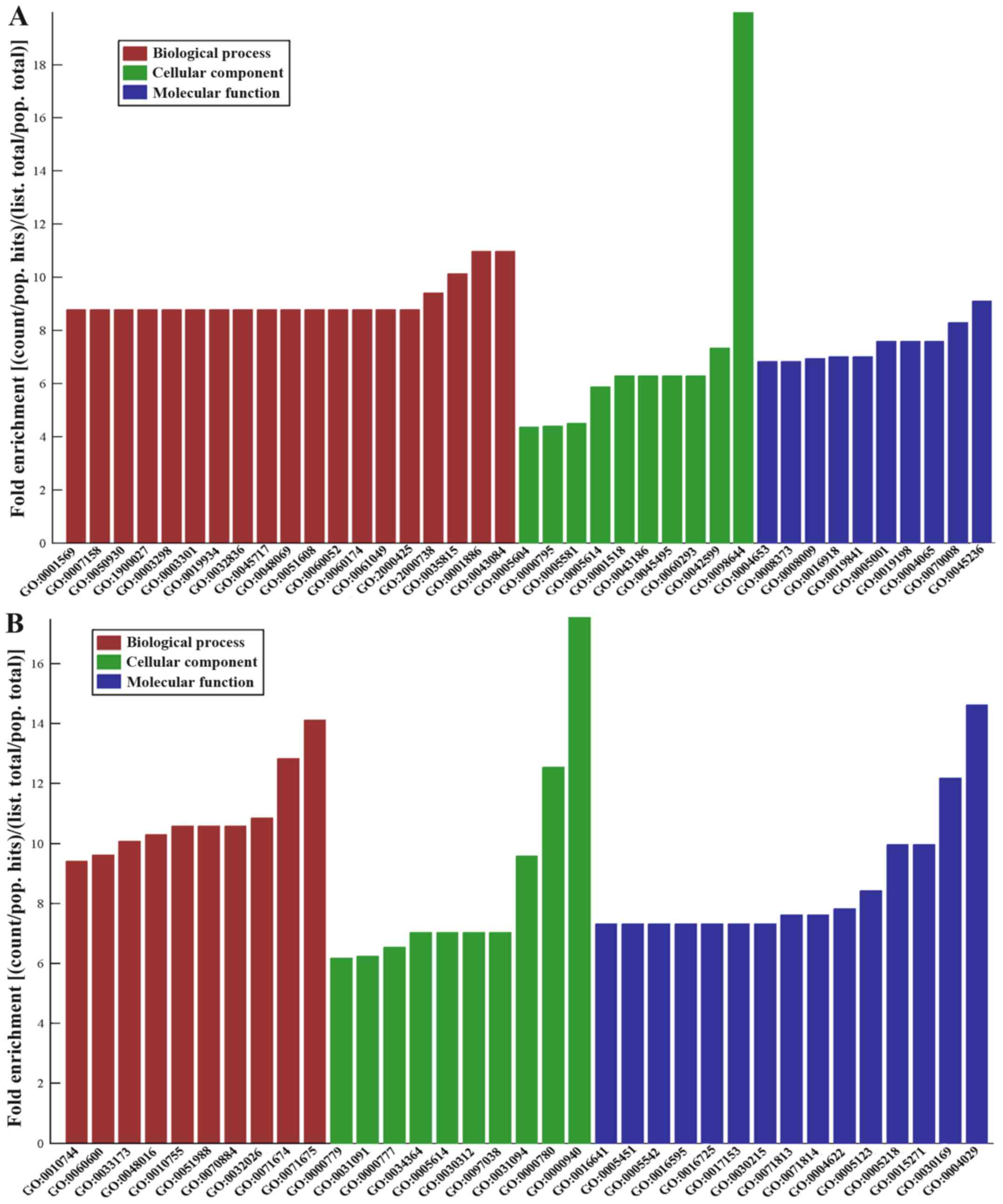
4). The top five downregulated GO functions were the following:
‘Complex of collagen trimers’; ‘endothelial cell morphogenesis’;
‘penile erection’; ‘positive regulation of renal sodium excretion’;
and ‘positive regulation of stem cell differentiation’ (Fig. 5A). The top five upregulated GO
functions were as follows: ‘Condensed chromosome outer
kinetochore’; ‘aldehyde dehydrogenase activity’; ‘regulation of
mononuclear cell migration’; ‘mononuclear cell migration’; and
‘condensed nuclear chromosome and centromeric region’ (Fig. 5B).
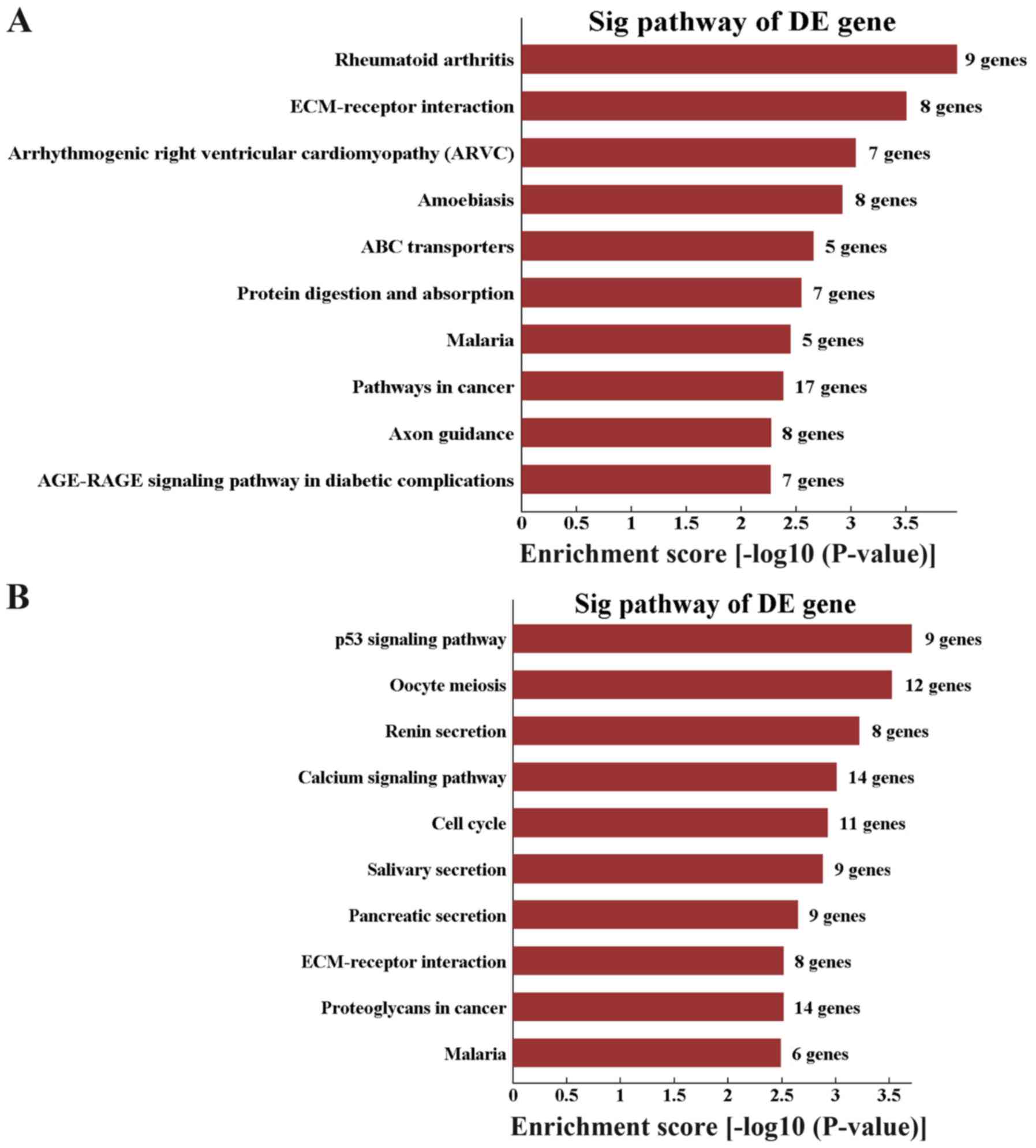
Pathway analysis revealed 20 downregulated pathways.
The top three enriched networks were ‘rheumatoid arthritis’,
‘ECM-receptor interaction’ and ‘arrhythmogenic right ventricular
cardiomyopathy (ARVC)’ (Fig. 6A).
In addition, 36 upregulated pathways were identified and the top
three enriched networks were ‘p53 signaling pathway’, ‘oocyte
meiosis’ and ‘renin secretion’ (Fig.
6B).
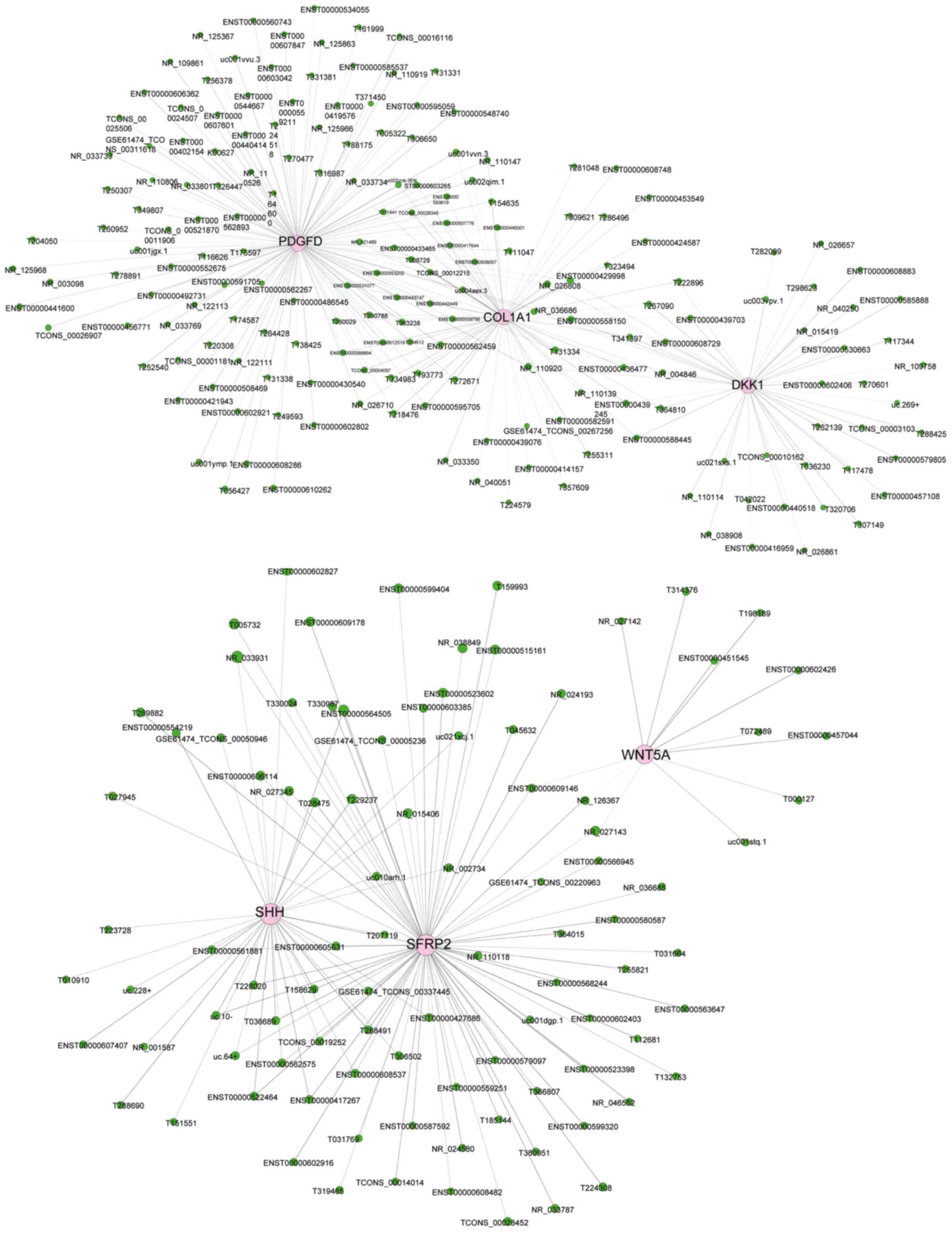
CNC network
A total of 615 lncRNAs and 16 mRNAs were selected to
construct the CNC network (Fig.
7). The CNC network included 488 positive pairs and 438
negative pairs, and each mRNA could be associated with 1–20
lncRNAs, and vice versa. The CNC network indicated the different
molecular mechanisms of hDFCs compared with hPDLCs associated with
the inter-regulation of lncRNAs and mRNAs.
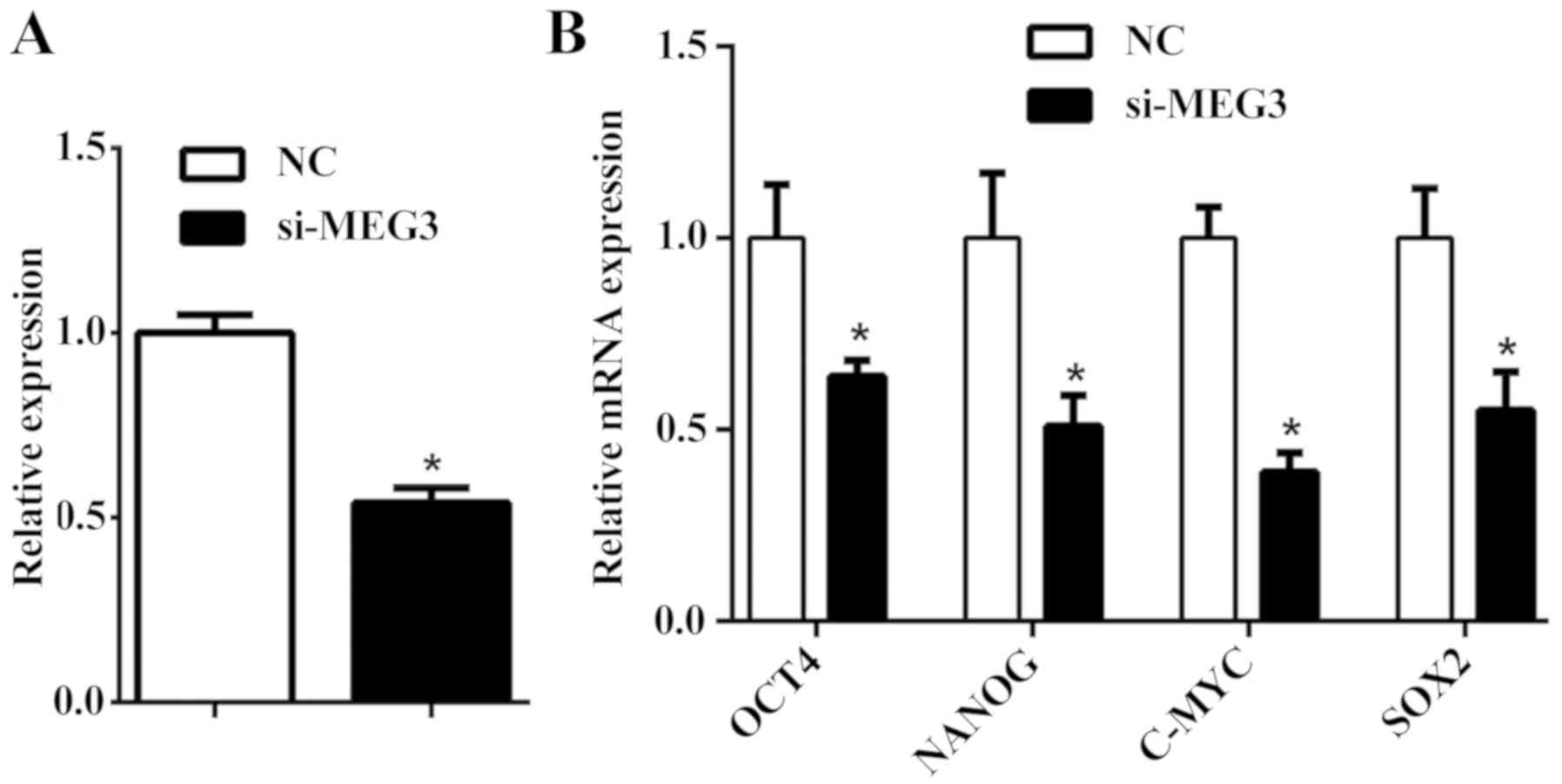
MEG3 regulates the pluripotency of
hDFCs
MEG3 was reported to serve a crucial role in
initiating embryogenesis and development (21). The current study depleted MEG3
expression in hDFCs and identified a reduction in the transcription
of pluripotency-associated genes (Fig.
8). In summary, these results may suggest a possible regulatory
role of MEG3 in the pluripotency of hDFCs.
Discussion
The dental follicle contains precursor cells that
may develop into periodontal ligament, cementum and alveolar bone
during periodontal development. hDFCs and hPDLCs are the major
cells of the dental follicle and periodontal ligaments,
respectively. These cells are key markers that represent different
stages of periodontal tissue development; however, hDFCs exhibit
more obvious embryonic characteristics, including pluripotency and
heterogeneity. Therefore, understanding the mechanisms associated
with the differentiation of hDFCs to hPDLCs is crucial for
promoting periodontal development and regeneration.
An increasing number of studies have indicated that
numerous lncRNAs regulate cell differentiation through epigenetics
or in-cis/in-trans gene transcription. Previous studies have
demonstrated that differential expression levels of lncRNAs are
associated with human diseases and biological processes. lncRNAs
serve vital roles in cell development and lineage commitment.
Therefore, lncRNAs may serve as key regulators of human tissue
development and regeneration (22). lncRNAs are differentially expressed
during the differentiation of human bone marrow mesenchymal stem
cells and PDLSCs (23,24). To investigate the changes and roles
of lncRNAs during the development of hDFCs, the current study used
high-throughput microarrays to detect the expression levels of
genes. Only the middle one-third of the root surface was taken
while obtaining the periodontal ligament from the extracted tooth;
the quantity was small, and collagen fibers and blood vessels in
the tissues may have affected the array results. The aim of the
present study was to compare the differentially expressed genes
between hDFCs and hPDLCs with differentiation capability. Cells
obtained at P3 have a stronger differentiation capability. A total
of 845 lncRNAs and 1,012 mRNAs were detected to be differentially
expressed in hDFCs compared with hPDLCs. By comparing the
expression of lncRNAs and mRNAs in hDFCs and hPDLCs, the present
results identified variation in the expression levels of lncRNAs
and mRNAs during the development of hDFCs. The results also
indicated that upregulated lncRNAs and mRNAs were predominant
during the development of hDFCs, and differentially expressed mRNAs
and lncRNAs were used as candidates to screen the key genes
associated with the differentiation of hDFCs by bioinformatics
analysis. The RT-qPCR results confirmed the reliability of the
microarray data. Markedly differential expression levels between
the two types of cells indicated that lncRNAs may serve crucial
roles in the differentiation and development of hDFCs.
lncRNAs may be categorized as sense, antisense,
intronic, intergenic and bidirectional (25). Among them, ASlncRNAs are a type of
endogenous lncRNA that complement other transcripts. Antisense
transcription is a common phenomenon in humans, and is based on
whether the antisense RNA acts in cis or in trans. ASlncRNAs
regulate gene expression at the transcriptional or
post-transcriptional level via a number of biological mechanisms,
including RNA-DNA interaction (chromatin remodeling), transcription
interference and RNA-RNA interaction in the nucleus/cytoplasm.
ASlncRNAs can serve as scaffolds between DNA and proteins, and
participate in disease processes by assisting with interactions
between ASlncRNAs, DNA and proteins (26). The current microarray results
indicated the ASlncRNAs and nearby mRNAs that may regulate
important biological processes. For example, the lncRNA NR_033932
was identified as one of the transcripts of lncRNA repulsive
guidance molecule b (RGMB)-AS1, which was revealed to be
upregulated in hDFCs compared with hPDLCs. lncRNA RGMB-AS1 is
located in the antisense region of RGMB, and RGMB carries the exon
of the lncRNA RGMB-AS1 gene in the reverse strand and orientation
of its intron region. lncRNA RGMB-AS1 silencing suppresses lung
adenocarcinoma and hepatocellular carcinoma cell proliferation,
migration and invasion, and leads to cell cycle arrest at the
G1/G0 phase (27,28).
lncRNA T152410 was identified to be upregulated in hDFCs compared
with hPDLCs. The nearby gene SMAD specific E3 ubiquitin protein
ligase 2 (SMURF2) is a member of the HECT family of E3 ubiquitin
ligases that regulate the polarity of cells during embryonic
development and other signaling pathways for osteoblast migration,
proliferation and differentiation. Notably, the anti-proliferative
effect of SMURF2 siRNA is mediated by arresting cells in the
G0/G1 phase, similar to the lncRNA RGMB-AS1
(29,30). This suggests that these two
ASlncRNAs may promote hDFC migration, proliferation and
differentiation by regulating the cell cycle.
lncRNAs may exert effects by controlling the
transcriptional regulation of nearby coding genes (31). Subgroup analysis of the microarray
demonstrated that a number of differentially expressed lincRNAs
located near mRNAs are associated with tissue development. For
example, lncRNA ENST00000512129 and nearby mRNA lymphoid enhancer
binding factor 1 (LEF1) were downregulated in hDFCs compared with
hPDLCs. As a key mediator of the Wnt/β-catenin signaling pathway
and epithelial-mesenchymal interaction, LEF1 has been revealed to
regulate incisor development. LEF1 also serves a key role in stem
cell maintenance along with SOX2 and paired like homeodomain 2
(32,33). lncRNA ENST00000540293 and some
nearby matrix metalloproteinase (MMP) mRNAs were revealed to be
downregulated in hDFCs. MMPs are effective proteolytic mediators
during ECM remodeling, and regulate the Notch signaling pathway,
which is involved in differentiation. MMPs are able to regulate the
necessary changes in the microenvironment, and overexpression of
MMPs increases cell differentiation during development by altering
the bioavailability of chemokines and cytokines that affect stem
cell function (34). These results
could be explained by evidence that lncRNAs regulate the expression
of neighboring genes to promote cellular survival and
differentiation (35).
Pathway analysis revealed that the top three
downregulated pathways were ‘rheumatoid arthritis’, ‘ECM-receptor
interaction’ and ‘arrhythmogenic right ventricular cardiomyopathy
(ARVC)’. In addition, the top three upregulated pathways were ‘p53
signaling pathway’, ‘oocyte meiosis’ and ‘renin secretion’. Among
these pathways, the p53 pathway is a vital signaling pathway in
tumor biology. The expression level of p53 is high during early
embryonic development. p53 serves an important role in self-renewal
and human embryonic stem cell differentiation by regulating
specific target genes or miRNAs and reactivating developmental
pathways for tissue regeneration (36). The calcium pathway is a ubiquitous
intracellular signaling pathway that participates in numerous
cellular processes, including cell proliferation, differentiation
and apoptosis. As an intracellular messenger, calcium serves an
important role in the cellular signaling pathways at different
stages of stem cell differentiation (37). Upregulation of calcium-mediated
signaling is essential for the maintenance of stem cells. The
current results indicated that these pathways may be involved in
the development of hDFCs.
In addition, CNC analysis revealed a potential
regulatory network between lncRNAs and mRNAs, and the mRNAs
selected for CNC analysis were closely associated with the pathways
in the development process of stem cells (38,39).
Among the lncRNA-mRNA pairs, lncRNA uc021sxs.1 was differentially
expressed in hDFCs compared with hPDLCs and was correlated with
Dickkopf-1 (DKK1). DKK1 serves as an antagonist of the canonical
Wnt signaling pathway by binding to the Wnt receptor Lrp5/6, which
is central to embryonic and adult bone development (40). The lncRNA ENST00000609146 was
upregulated in hDFCs compared with hPDLCs and it was negatively
correlated with Wnt5a. Wnt5a regulates a variety of biological
processes, including proliferation, differentiation, migration,
adhesion and polarity (41). Wnt5a
appears to serve important roles in the fate of DFSCs in the
development and regeneration of the periodontium. Wnt5a can
directly or indirectly activate the canonical Wnt signaling pathway
to promote mesendoderm differentiation (42). The correlations between these
lncRNAs and mRNAs indicate that lncRNA uc021sxs.1 and lncRNA
ENST00000609146 may be involved in associated signaling pathways by
regulating mRNA expression. In addition, the results suggest that
the activation of the Wnt signaling pathway may serve a critical
role in hDFC development.
Although numerous studies have been conducted to
identify cell factors that maintain the pluripotency of periodontal
cells (43), potential lncRNAs
with similar functions remain unknown. The current results
demonstrated that MEG3 may serve a role in the establishment or
maintenance of the stem cell state.
A study conducted by Lee et al (44) reported that 1.49% of mRNAs in hDFCs
were differentially expressed (fold change >2.0 or <-2.0;
P<0.05) compared with hPDLCs. The current microarray results
indicated that among a total of 16,531 mRNAs, the expression levels
of 1,012 mRNAs (6.12%) were significantly different in hDFCs
compared with hPDLCs. Differences in the ages of participants and
cell state may explain this discrepancy. Compared with the study by
Lee et al (44), the
current analysis predominantly focused on the potential lncRNAs
instead of mRNAs, which may serve critical roles in hDFC
differentiation.
The present results may promote further studies to
investigate the functions of genes and the regulatory mechanisms in
hDFCs and periodontal tissue, and may advance the use of stem
cell-based therapies in regenerative medicine. Furthermore, more
studies should be conducted to investigate the potential functions
that differentially expressed lncRNAs serve in multiple biological
processes of hDFCs and periodontal tissue.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China (grant no. 81170932) and the
Natural Science Foundation of Guangdong Province (grant no.
2015A030313083).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LW, LD and JL conceived and designed the
experiments. LD performed the experiments. LW, LD and HH analyzed
the data. LD drafted the manuscript. CP, XZ, and ZC performed the
experiments, analyzed the data and revised the manuscript. All
authors reviewed and approved the final manuscript.
Ethics approval and consent to
participate
The protocol to acquire human tissues was approved
by the Ethical Guidelines of the Ethics Committee of the Hospital
of Stomatology, Sun Yat-Sen University (Guangzhou, China). Written
informed consent was obtained from all patients and their
parents.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Cho MI and Garant PR: Development and
general structure of the periodontium. Periodontol 2000. 24:9–27.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Itaya S, Oka K, Ogata K, Tamura S,
Kira-Tatsuoka M, Fujiwara N, Otsu K, Tsuruga E, Ozaki M and Harada
H: Hertwig's epithelial root sheath cells contribute to formation
of periodontal ligament through epithelial-mesenchymal transition
by TGF-β. Biomed Res. 38:61–69. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sowmya S, Chennazhi KP, Arzate H,
Jayachandran P, Nair SV and Jayakumar R: Periodontal specific
differentiation of dental follicle stem cells into osteoblast,
fibroblast, and cementoblast. Tissue Eng Part C Methods.
21:1044–1058. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Seo BM, Miura M, Gronthos S, Bartold PM,
Batouli S, Brahim J, Young M, Robey PG, Wang CY and Shi S:
Investigation of multipotent postnatal stem cells from human
periodontal ligament. Lancet. 364:149–155. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Menicanin D, Mrozik KM, Wada N, Marino V,
Shi S, Bartold PM and Gronthos S: Periodontal-ligament-derived stem
cells exhibit the capacity for long-term survival, self-renewal,
and regeneration of multiple tissue types in vivo. Stem Cells Dev.
23:1001–1011. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo S, Guo W, Ding Y, Gong J, Zou Q, Xie
D, Chen Y, Wu Y and Tian W: Comparative study of human dental
follicle cell sheets and periodontal ligament cell sheets for
periodontal tissue regeneration. Cell Transplant. 22:1061–1073.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kashi K, Henderson L, Bonetti A and
Carninci P: Discovery and functional analysis of lncRNAs:
Methodologies to investigate an uncharacterized transcriptome.
Biochim Biophys Acta. 1859:3–15. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ghosal S, Das S and Chakrabarti J: Long
noncoding RNAs: New players in the molecular mechanism for
maintenance and differentiation of pluripotent stem cells. Stem
Cells Dev. 22:2240–2253. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schmitz SU, Grote P and Herrmann BG:
Mechanisms of long noncoding RNA function in development and
disease. Cell Mol Life Sci. 73:2491–2509. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Aprea J and Calegari F: Long non-coding
RNAs in corticogenesis: Deciphering the non-coding code of the
brain. EMBO J. 34:2865–2884. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Korostowski L, Sedlak N and Engel N: The
Kcnq1ot1 long non-coding RNA affects chromatin conformation and
expression of Kcnq1, but does not regulate its imprinting in the
developing heart. PLoS Genet. 8:e10029562012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Lv J, Huang Z, Liu H, Liu H, Cui W, Li B,
He H, Guo J, Liu Q, Zhang Y and Wu Q: Identification and
characterization of long intergenic non-coding RNAs related to
mouse liver development. Mol Genet Genomics. 289:1225–1235. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Herriges MJ, Swarr DT, Morley MP, Rathi
KS, Peng T, Stewart KM and Morrisey EE: Long noncoding RNAs are
spatially correlated with transcription factors and regulate lung
development. Genes Dev. 28:1363–1379. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hassan MQ, Tye CE, Stein GS and Lian JB:
Non-coding RNAs: Epigenetic regulators of bone development and
homeostasis. Bone. 81:746–756. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Chen L, Song Z, Huang S, Wang R, Qin W,
Guo J and Lin Z: lncRNA DANCR suppresses odontoblast-like
differentiation of human dental pulp cells by inhibiting
wnt/β-catenin pathway. Cell Tissue Res. 364:309–318. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jia Q, Chen X, Jiang W, Wang W, Guo B and
Ni L: The regulatory effects of long noncoding RNA-ANCR on dental
tissue-derived stem cells. Stem Cells Int. 2016:31468052016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Viale-Bouroncle S, Klingelhöffer C, Ettl T
and Morsczeck C: The WNT inhibitor APCDD1 sustains the expression
of β-catenin during the osteogenic differentiation of human dental
follicle cells. Biochem Biophys Res Commun. 457:314–317. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wei M, Zhang M, Adams A and Duan Y: JNK
and AKT/GSK3β signaling pathways converge to regulate periodontal
ligament cell survival involving XIAP. Biochem Biophys Res Commun.
448:485–491. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ashburner M, Ball CA, Blake JA, Botstein
D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT,
et al: Gene ontology: Tool for the unification of biology. The gene
ontology consortium. Nat Genet. 25:25–29. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Sellers ZP, Schneider G, Maj M and
Ratajczak MZ: Analysis of the paternally-imprinted DLK1-MEG3 and
IGF2-H19 tandem gene loci in NT2 embryonal carcinoma cells
identifies DLK1 as a potential therapeutic target. Stem Cell Rev.
14:823–836. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Peng S, Cao L, He S, Zhong Y, Ma H, Zhang
Y and Shuai C: An overview of long noncoding RNAs involved in bone
regeneration from mesenchymal stem cells. Stem Cells Int.
2018:82736482018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Farzi-Molan A, Babashah S, Bakhshinejad B,
Atashi A and Fakhr TM: Down-regulation of the non-coding RNA H19
and its derived miR-675 is concomitant with up-regulation of
insulin-like growth factor receptor type 1 during neural-like
differentiation of human bone marrow mesenchymal stem cells. Cell
Biol Int. 42:940–948. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gu X, Li M, Jin Y, Liu D and Wei F:
Identification and integrated analysis of differentially expressed
lncRNAs and circRNAs reveal the potential ceRNA networks during
PDLSC osteogenic differentiation. BMC Genet. 18:1002017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kunej T, Obsteter J, Pogacar Z, Horvat S
and Calin GA: The decalog of long non-coding RNA involvement in
cancer diagnosis and monitoring. Crit Rev Clin Lab Sci. 51:344–357.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tsai MC, Manor O, Wan Y, Mosammaparast N,
Wang JK, Lan F, Shi Y, Segal E and Chang HY: Long noncoding RNA as
modular scaffold of histone modification complexes. Science.
329:689–693. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Li P, Zhang G, Li J, Yang R, Chen S, Wu S,
Zhang F, Bai Y, Zhao H, Wang Y, et al: Long noncoding RNA RGMB-AS1
indicates a poor prognosis and modulates cell proliferation,
migration and invasion in lung adenocarcinoma. PLoS One.
11:e1507902016.
|
|
28
|
Sheng N and Li Y, Qian R and Li Y: The
clinical significance and biological function of lncRNA RGMB-AS1 in
hepatocellular carcinoma. Biomed Pharmacother. 98:577–584. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Choi YH, Kim YJ, Jeong HM, Jin YH, Yeo CY
and Lee KY: Akt enhances Runx2 protein stability by regulating
Smurf2 function during osteoblast differentiation. FEBS J.
281:3656–3666. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
David D, Jagadeeshan S, Hariharan R, Nair
AS and Pillai RM: Smurf2 E3 ubiquitin ligase modulates
proliferation and invasiveness of breast cancer cells in a CNKSR2
dependent manner. Cell Div. 9:22014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Li B, Chen P, Qu J, Shi L and Zhuang W, Fu
J, Li J, Zhang X, Sun Y and Zhuang W: Activation of LTBP3 gene by a
long noncoding RNA (lncRNA) MALAT1 transcript in mesenchymal stem
cells from multiple myeloma. J Biol Chem. 289:29365–29375. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Sun Z, Yu W, Sanz Navarro M, Sweat M,
Eliason S, Sharp T, Liu H, Seidel K, Zhang L, Moreno M, et al: Sox2
and Lef-1 interact with Pitx2 to regulate incisor development and
stem cell renewal. Development. 143:4115–4126. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Santiago L, Daniels G, Wang D, Deng FM and
Lee P: Wnt signaling pathway protein LEF1 in cancer, as a biomarker
for prognosis and a target for treatment. Am J Cancer Res.
7:1389–1406. 2017.PubMed/NCBI
|
|
34
|
Kessenbrock K, Wang CY and Werb Z: Matrix
metalloproteinases in stem cell regulation and cancer. Matrix Biol.
44-46:184–190. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Bunch H: Gene regulation of mammalian long
non-coding RNA. Mol Genet Genomics. 293:1–15. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Levine AJ and Berger SL: The interplay
between epigenetic changes and the p53 protein in stem cells. Genes
Dev. 31:1195–1201. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Tonelli FM, Santos AK, Gomes DA, da Silva
SL, Gomes KN, Ladeira LO and Resende RR: Stem cells and calcium
signaling. Adv Exp Med Biol. 740:891–916. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu Q, Hu CH, Zhou CH, Cui XX, Yang K,
Deng C, Xia JJ, Wu Y, Liu LC and Jin Y: DKK1 rescues osteogenic
differentiation of mesenchymal stem cells isolated from periodontal
ligaments of patients with diabetes mellitus induced periodontitis.
Sci Rep. 5:131422015. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xiang L, Chen M, He L, Cai B, Du Y, Zhang
X, Zhou C, Wang C, Mao JJ and Ling J: Wnt5a regulates dental
follicle stem/progenitor cells of the periodontium. Stem Cell Res
Ther. 5:1352014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ou L, Fang L, Tang H, Qiao H, Zhang X and
Wang Z: Dickkopf Wnt signaling pathway inhibitor 1 regulates the
differentiation of mouse embryonic stem cells in vitro and in vivo.
Mol Med Rep. 13:720–730. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kikuchi A, Yamamoto H, Sato A and
Matsumoto S: Wnt5a: Its signalling, functions and implication in
diseases. Acta Physiol (Oxf). 204:17–33. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Yu CY and Kuo HC: The trans-spliced long
noncoding RNA tsRMST impedes human embryonic stem cell
differentiation through WNT5A-mediated inhibition of the
epithelial-to-mesenchymal transition. Stem Cells. 34:2052–2062.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu X, Tan GR, Yu M, Cai X, Zhou Y, Ding
H, Xie H, Qu F, Zhang R, Lam CU, et al: The effect of tumour
necrosis factor-alpha on periodontal ligament stem cell
differentiation and the related signaling pathways. Curr Stem Cell
Res Ther. 11:593–602. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee HS, Lee J, Kim SO, Song JS, Lee JH,
Lee SI, Jung HS and Choi BJ: Comparative gene-expression analysis
of the dental follicle and periodontal ligament in humans. PLoS
One. 8:e842012013. View Article : Google Scholar : PubMed/NCBI
|