|
1
|
Jia J, Wei C, Chen S, Li F, Tang Y, Qin W,
Zhao L, Jin H, Xu H, Wang F, et al: The cost of Alzheimer's disease
in China and re-estimation of costs worldwide. Alzheimers Dement.
14:483–491. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yiannopoulou KG and Papageorgiou SG:
Current and future treatments for Alzheimer's disease. Ther Adv
Neurol Disord. 6:19–33. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chu J, Li JG, Hoffman NE, Madesh M and
Praticò D: Degradation of gamma secretase activating protein by the
ubiquitin-proteasome pathway. J Neurochem. 133:432–439. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Esler WP and Wolfe MS: A portrait of
Alzheimer secretases-new features and familiar faces. Science.
293:1449–1454. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Glenner GG and Wong CW: Alzheimer's
disease: Initial report of the purification and characterization of
a novel cerebrovascular amyloid protein. Biochem Biophys Res
Commun. 120:885–890. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Agostinho P, Pliássova A, Oliveira CR and
Cunha RA: Localization and trafficking of amyloid-β protein
precursor and secretases: Impact on Alzheimer's disease. J
Alzheimers Dis. 45:329–347. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ashe KH and Zahs KR: Probing the biology
of Alzheimer's disease in mice. Neuron. 66:631–645. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sakono M and Zako T: Amyloid oligomers:
Formation and toxicity of Abeta oligomers. FEBS J. 277:1348–1358.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu LJ, Leung KH, Chan DS, Wang YT, Ma DL
and Leung CH: Identification of a natural product-like STAT3
dimerization inhibitor by structure-based virtual screening. Cell
Death Dis. 5:e12932014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ito M, Tanaka T, Toita A, Uchiyama N,
Kokubo H, Morishita N, Klein MG, Zou H, Murakami M, Kondo M, et al:
Discovery of
3-Benzyl-1-(trans-4-((5-cyanopyridin-2-yl)amino)cyclohexyl)-1-arylurea
derivatives as novel and selective cyclin-dependent kinase 12
(CDK12) inhibitors. J Med Chem. 61:7710–7728. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ma DL, Lin S, Wang W, Yang C and Leung CH:
Luminescent chemosensors by using cyclometalated iridium(iii)
complexes and their applications. Chem Sci. 8:878–889. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dhar Malhotra J, Chen C, Rivolta I, Abriel
H, Malhotra R, Mattei LN, Brosius FC, Kass RS and Isom LL:
Characterization of sodium channel alpha- and beta-subunits in rat
and mouse cardiac myocytes. Circulation. 103:1303–1310. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Vijayaragavan K, Powell AJ, Kinghorn IJ
and Chahine M: Role of auxiliary beta1-, beta2-, and beta3-subunits
and their interaction with Na (v) 1.8 voltage-gated sodium channel.
Biochem Biophys Res Commun. 319:531–540. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bechtold DA and Smith KJ: Sodium-mediated
axonal degeneration in inflammatory demyelinating disease. J Neurol
Sci. 233:27–35. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Waxman SG: Axonal conduction and injury in
multiple sclerosis: The role of sodium channels. Nat Rev Neurosci.
7:932–941. 2006. View
Article : Google Scholar : PubMed/NCBI
|
|
16
|
O'Malley HA, Shreiner AB, Chen GH,
Huffnagle GB and Isom LL: Loss of Na+ channel beta2
subunits is neuroprotective in amouse model of multiple sclerosis.
Mol Cell Neurosci. 40:143–155. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen C, Bharucha V, Chen Y, Westenbroek
RE, Brown A, Malhotra JD, Jones D, Avery C, Gillespie PJ III,
Kazen-Gillespie KA, et al: Reduced sodium channel density, altered
voltage dependence of inactivation, and increased susceptibility to
seizures in mice lacking sodium channel beta 2-subunits. Proc Natl
Acad Sci USA. 99:17072–17077. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lopez-Santiago LF, Pertin M, Morisod X,
Chen C, Hong S, Wiley J, Decosterd I and Isom LL: Sodium channel
beta2 subunits regulate tetrodotoxin-sensitive sodium channels in
small dorsal root ganglion neurons and modulate the response to
pain. J Neurosci. 26:7984–7994. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kim DY, Carey BW, Wang H, Ingano LA,
Binshtok AM, Wertz MH, Pettingell WH, He P, Lee VM, Woolf CJ and
Kovacs DM: BACE1 regulates voltage-gated sodium channels and
neuronal activity. Nat Cell Biol. 9:755–764. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wong HK, Sakurai T, Oyama F, Kaneko K,
Wada K, Miyazaki H, Kurosawa M, De Strooper B, Saftig P and Nukina
N: beta subunits of voltage-gated sodium channels are novel
substrates of beta-site amyloid precursor protein-cleaving enzyme
(BACE1) and gamma-secretase. Biol Chem. 280:23009–23017. 2005.
View Article : Google Scholar
|
|
21
|
Kim DY, Ingano LA, Carey BW, Pettingell WH
and Kovacs DM: Presenilin/gamma-secretase-mediated cleavage of the
voltage-gated sodium channel beta2-subunit regulates cell adhesion
and migration. J Biol Chem. 280:23251–23261. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huth T, Schmidt-Neuenfeldt K, Rittger A,
Saftig P, Reiss K and Alzheimer C: Non-proteolytic effect of
beta-site APP cleaving enzyme 1 (BACE1) on sodium channel function.
Neurobiol Dis. 33:282–289. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Corbett BF, Leiser SC, Ling HP, Nagy R,
Breysse N, Zhang X, Hazra A, Brown JT, Randall AD, Wood A, et al:
Sodium channel cleavage is associated with aberrant neuronal
activity and cognitive deficits in a mouse model of Alzheimer's
disease. J Neurosci. 33:7020–7026. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
XiYang YB, Wang YC, Zhao Y, Ru J, Lu BT,
Zhang YN, Wang NC, Hu WY, Liu J, Yang JW, et al: Sodium
channel-voltage-gated-beta 2 plays a vital role in brain aging
associated with synaptic plasticity and expression of COX5A and
FGF-2. Mol Neurobiol. 53:955–967. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
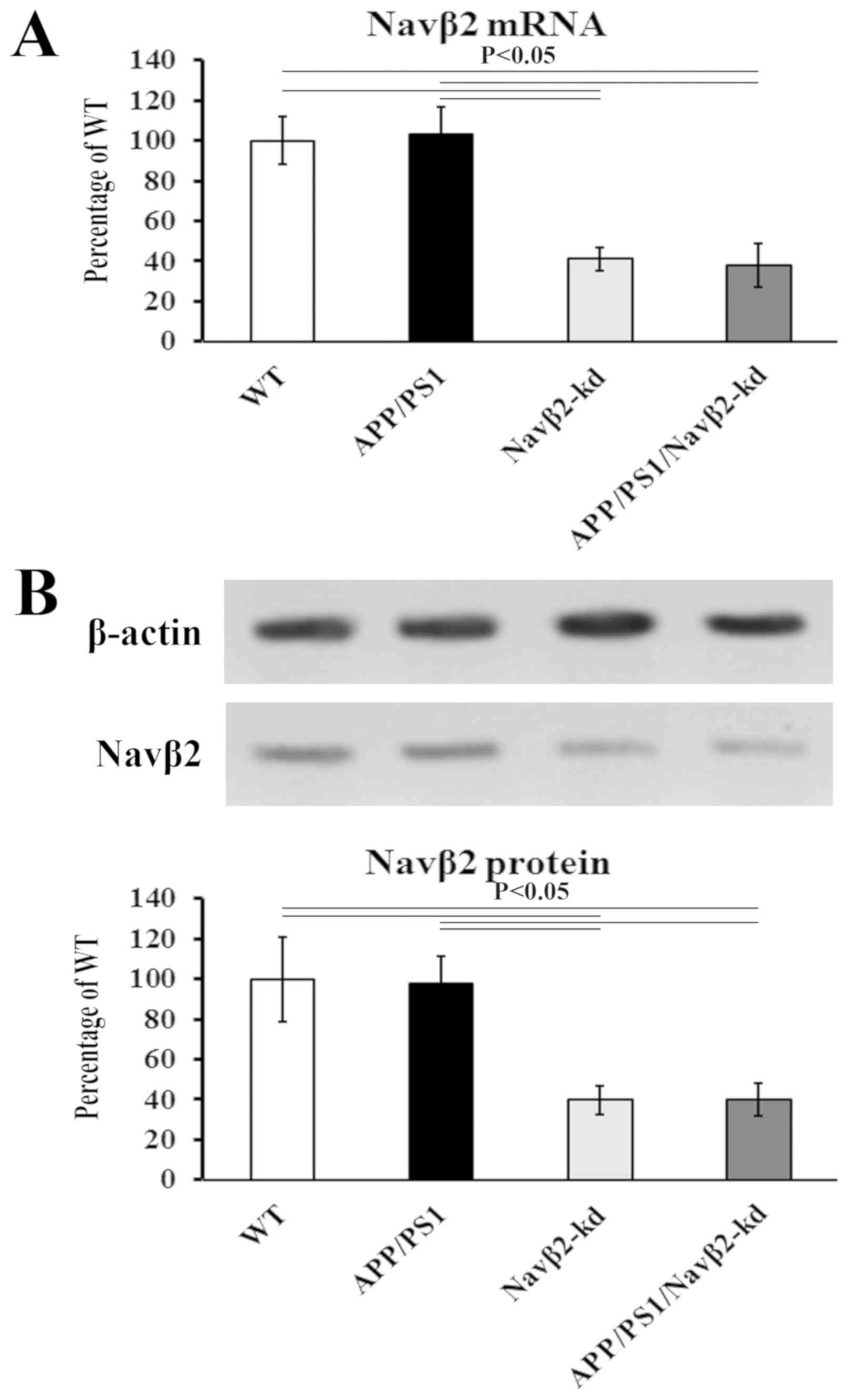
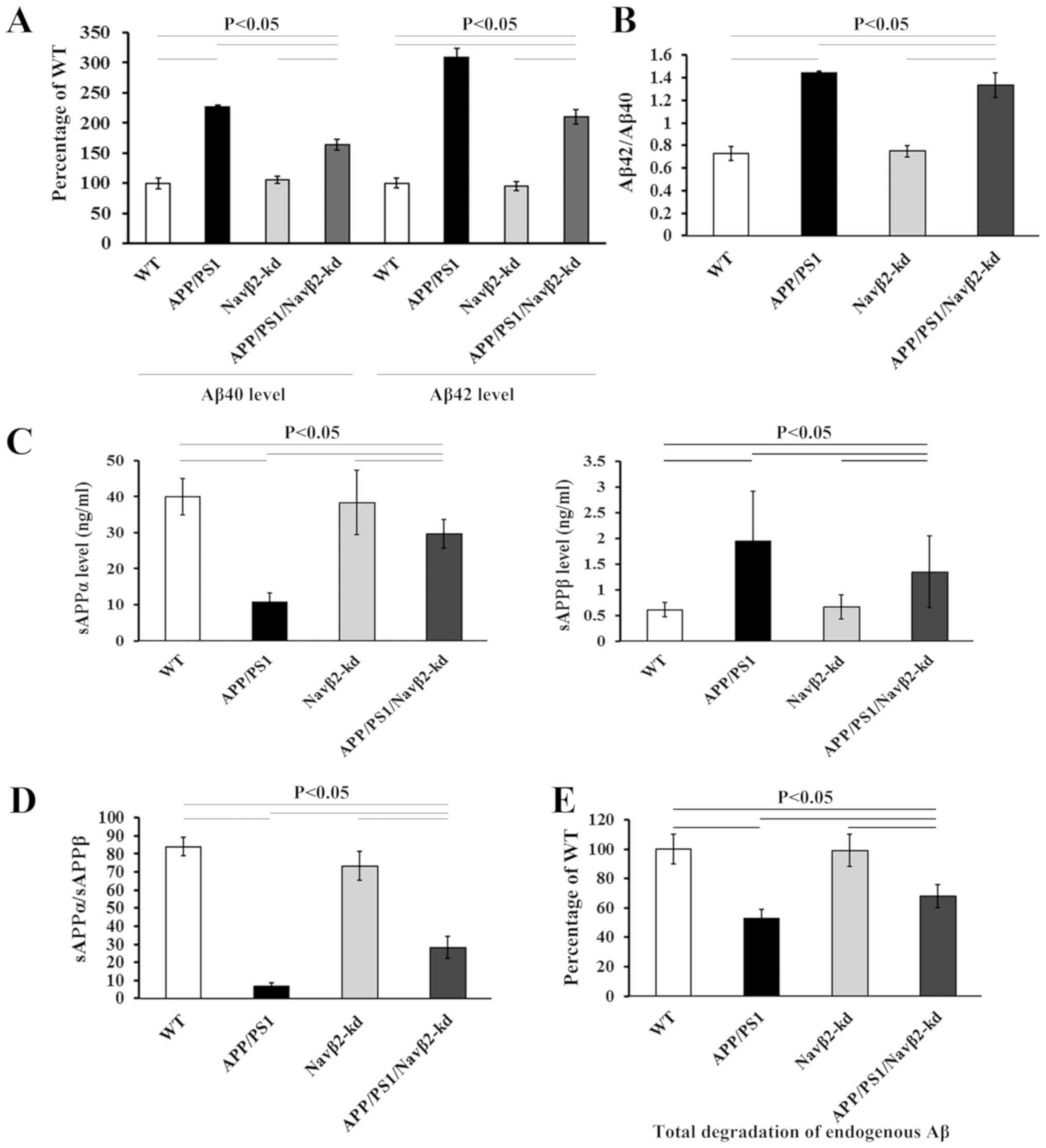
Hu T, Xiao Z, Mao R, Chen B, Lu MN, Tong
J, Mei R, Li SS, Xiao ZC, Zhang LF and Xiyang YB: Navβ2 knockdown
improves cognition in APP/PS1 mice by partially inhibiting seizures
and APP amyloid processing. Oncotarget. 8:99284–99295. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
National Research Council (US) Committee
for the Update of the Guide for the Care and Use of Laboratory
Animals, . Guide for the Care and Use of Laboratory Animals. 8th.
National Academies Press; Washington, DC: 2011
|
|
27
|
Hu T, Li YS, Chen B, Chang YF, Liu GC,
Hong Y, Chen HL and Xiyang YB: Elevated glucose-6-phosphate
dehydrogenase expression in the cervical cancer cases is associated
with the cancerigenic event of high-risk human papillomaviruses.
Exp Bio Med (Maywood). 240:1287–1297. 2015. View Article : Google Scholar
|
|
28
|
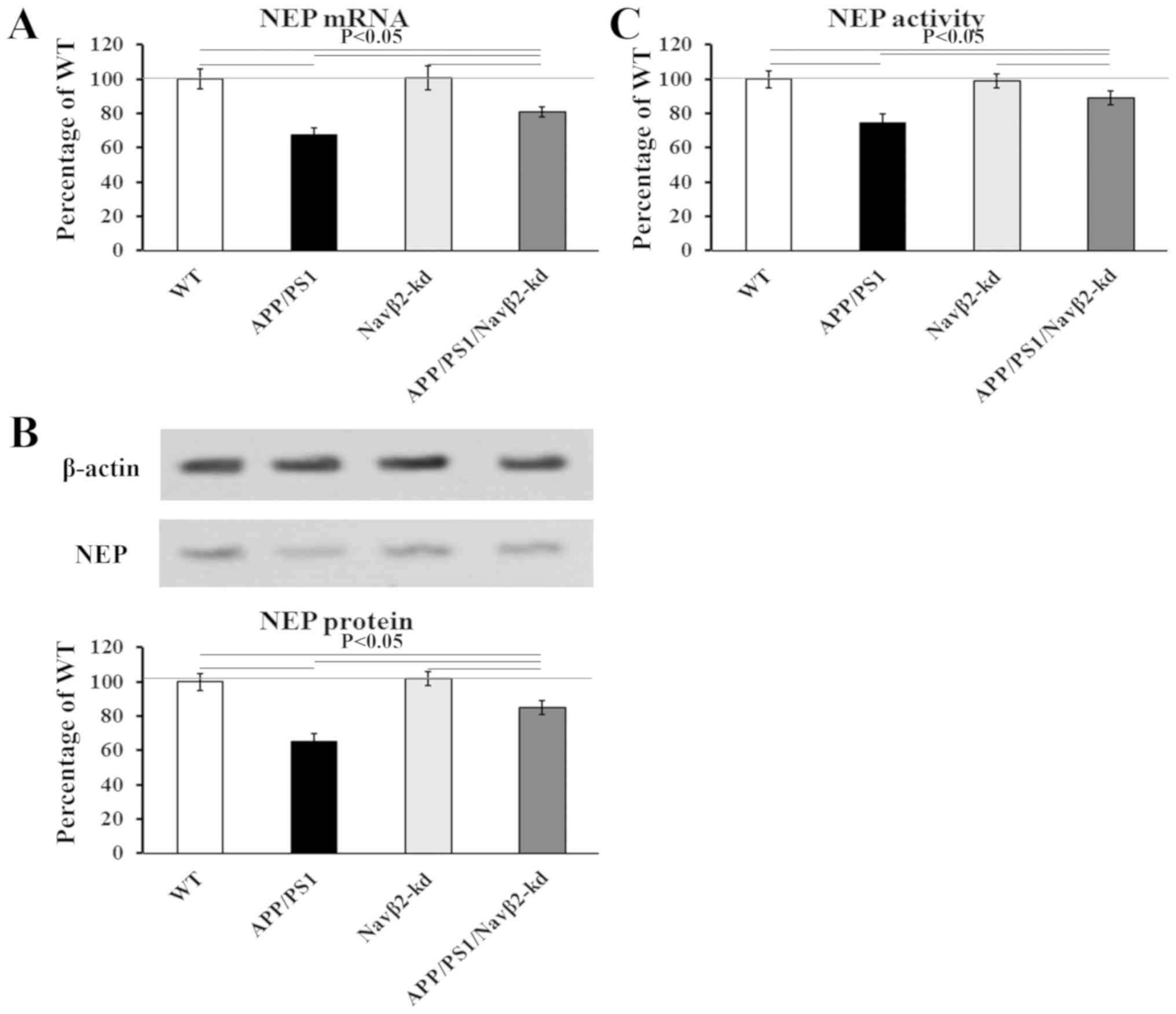
Grimm MO, Mett J, Stahlmann CP, Grösgen S,
Haupenthal VJ, Blümel T, Hundsdörfer B, Zimmer VC, Mylonas NT,
Tanila H, et al: APP intracellular domain derived from
amyloidogenic β- and γ-secretase cleavage regulates neprilysin
expression. Front Aging Neurosci. 7:772015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hu T, Chang YF, Xiao Z, Mao R, Tong J,
Chen B, Liu GC, Hong Y, Chen HL, Kong SY, et al: miR-1 inhibits
progression of high-risk papillomavirus-associated human cervical
cancer by targeting G6PD. Oncotarget. 7:86103–86116. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zuccato C, Belyaev N, Conforti P, Ooi L,
Tartari M, Papadimou E, MacDonald M, Fossale E, Zeitlin S, Buckley
N and Cattaneo E: Wide spread disruption of repressor element-1
silencing transcription factor/neuron-restrictive silencer factor
occupancy at its target genes in Huntington's disease. J Neurosci.
27:6972–6983. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Belyaev ND, Nalivaeva NN, Makova NZ and
Turner AJ: Neprilysin gene expression requires binding of the
amyloid precursor protein intracellular domain to its promoter:
Implications for Alzheimer disease. EMBO Rep. 10:94–100. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kerridge C, Kozlova DI, Nalivaeva NN and
Turner AJ: Hypoxia affects neprilysin expression through caspase
activation and an APP intracellular domain-dependent Mechanism.
Front Neurosci. 9:4262015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Mullan M, Crawford F, Axelman K, Houlden
H, Lilius L, Winblad B and Lannfelt L: A pathogenic mutation for
probable Alzheimer's disease in the APP gene at the N-terminus of
beta-amyloid. Nat Genet. 1:345–347. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Citron M, Oltersdorf T, Haass C,
McConlogue L, Hung AY, Seubert P, Vigo-Pelfrey C, Lieberburg I and
Selkoe DJ: Mutation of the beta-amyloid precursor protein in
familial Alzheimer's disease increases beta-protein production.
Nature. 360:672–674. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Felsenstein KM, Hunihan LW and Roberts SB:
Altered cleavage and secretion of a recombinant beta-APP bearing
the Swedish familial Alzheimer's disease mutation. Nat Genet.
6:251–255. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Esch FS, Keim PS, Beattie EC, Blacher RW,
Culwell AR, Oltersdorf T, McClure D and Ward PJ: Cleavage of
amyloid beta peptide during constitutive processing of its
precursor. Science. 248:1122–1124. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lichtenthaler SF: α-secretase in
Alzheimer's disease: Molecular identity, regulation and therapeutic
potential. J Neurochem. 116:10–21. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hardy J and Selkoe DJ: The amyloid
hypothesis of Alzheimer's disease: Progress and problems on the
road to therapeutics. Science. 297:353–356. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Xie Z and Xu Z: General anesthetics and
β-amyloid protein. Prog Neuropsychopharmacol Biol Psychiatry.
47:140–146. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Nalivaeva NN, Beckett C, Belyaev ND and
Turner AJ: Are amyloid-degrading enzymes viable therapeutic targets
in Alzheimer's disease? J Neurochem. 120 (Suppl 1):S167–S185. 2012.
View Article : Google Scholar
|
|
42
|
Nalivaeva NN, Belyaev ND, Kerridge C and
Turner AJ: Amyloid- clearing proteins and their epigenetic
regulation as a therapeutic target in Alzheimer's disease. Front
Aging Neurosci. 6:2352014. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Marr RA and Spencer BJ: NEP-like
endopeptidases and Alzheimer's disease. Curr Alzheimer Res.
7:223–229. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Pacheco-Quinto J and Eckman EA:
Endothelin-converting enzymes degrade intracellular β-amyloid
produced within the endosomal/lysosomal pathway and autophagosomes.
J Biol Chem. 288:5606–5615. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Miners JS, Baig S, Palmer J, Palmer LE,
Kehoe PG and Love S: Abeta-degrading enzymes in Alzheimer's
disease. Brain Pathol. 18:240–252. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Turner AJ, Brown CD, Carson JA and Barnes
K: The neprilysin family in health and disease. Adv Exp Med Biol.
477:229–240. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Turner AJ, Isaac RE and Coates D: The
neprilysin (NEP) family of zinc metalloendopeptidases: Genomics and
function. Bioessays. 23:261–269. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Selkoe DJ: Preventing Alzheimer's disease.
Science. 337:1488–1492. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Pluta R, Jabłoński M, Ułamek-Kozioł M,
Kocki J, Brzozowska J, Januszewski S, Furmaga-Jabłońska W,
Bogucka-Kocka A, Maciejewski R and Czuczwar SJ: Sporadic
Alzheimer's disease begins as episodes of brain ischemia and
ischemically dysregulated Alzheimer's disease genes. Mol Neurobiol.
48:500–515. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Li C, Booze RM and Hersh LB:
Tissue-specific expression of rat neutral endopeptidase
(neprilysin) mRNAs. J Biol Chem. 270:5723–5728. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Pardossi-Piquard R, Petit A, Kawarai T,
Sunyach C, Alves da Costa C, Vincent B, Ring S, D'Adamio L, Shen J,
Müller U, et al: Presenilin-dependent transcriptional control of
the Abeta-degrading enzyme neprilysin by intracellular domains of
betaAPP and APLP. Neuron. 46:541–554. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Belyaev ND, Kellett KA, Beckett C, Makova
NZ, Revett TJ, Nalivaeva NN, Hooper NM and Turner AJ: The
transcriptionally active amyloid precursor protein (APP)
intracellular domain is preferentially produced from the 695
isoform of APP in a {beta}-secretase-dependent pathway. J Biol
Chem. 285:41443–41454. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Kerridge C, Belyaev ND, Nalivaeva NN and
Turner AJ: The Aβ-clearance protein transthyretin, like neprilysin,
is epigenetically regulated by the amyloid precursor protein
intracellular domain. J Neurochem. 130:419–431. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Li S, Wang X, Ma QH, Yang WL, Zhang XG,
Dawe GS and Xiao ZC: Amyloid precursor protein modulates Nav1.6
sodium channel currents through a Go-coupled JNK pathway. Sci Rep.
6:393202016. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Hansen TB, Jensen TI, Clausen BH, Bramsen
JB, Finsen B, Damgaard CK and Kjems J: Natural RNA circles function
as efficient microRNA sponges. Nature. 495:384–388. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Hébert SS and De Strooper B: Alterations
of the microRNA network cause neurodegenerative disease. Trends
Neurosci. 32:199–206. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Lukiw WJ: Micro-RNA speciation in fetal,
adult and Alzheimer's disease hippocampus. Neuroreport. 18:297–300.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Liu W, Liu C, Yin B and Peng XZ: Functions
of miR-9 and miR-9* during aging in SAMP8 mice and their possible
mechanisms. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 37:253–258.
2015.PubMed/NCBI
|