Introduction
Chronic ultraviolet (UV) exposure is harmful and
hazardous to the human skin. Excessive UV light irradiation can
penetrate the skin and impair cellular antioxidant defense in
dermal human fibroblasts (HDFs), which play an important role in
maintaining normal structure and function of the skin (1,2).
Consequently, high production of reactive oxygen species (ROS)
leads to cellular oxidative stress, cell damage and apoptosis
(3,4). Since the generation of ROS has been
associated with the pathogenesis of UV-mediated damage in skin
cells, many antioxidants have been used to protect HDFs against UV
through induction of NF-E2-related factor 2(Nrf2)-dependent
antioxidant gene expression (5,6).
The transcription factor Nrf2 could bind antioxidant
response elements (AREs) and regulate the expression of antioxidant
genes such as NQO1, γ-GCS, HO-1 and others (7). It has been reported that Nrf2 acts as
a master regulator of the cellular antioxidant defense against
cutaneous photodamage mediated by UV radiation (8,9).
Notably, Nrf2 knock-out mice displayed UV-induced sunburn reaction
and oxidative DNA damage compared to wild-type mice (10). As a result, pharmacological
activation of Nrf2 has been a novel approach to skin
photoprotection. All-trans retinoic acid (ATRA) has been
clinically used to treat several skin diseases including UV-induced
skin oxidative damage (11,12).
Tan et al reported that ATRA could activate Nrf2 and induce
Nrf2 target genes expression (13). However, whether ATRA could increase
Nrf2 expression to protect skin fibroblasts against UV-induced
oxidative damage remains unclear.
3-Hydroxy-3-methylglutaryl reductase degradation
(Hrd1), also known as synoviolin, is a endoplasmic
reticulum-associated degradation (ERAD)-associated E3 ubiquitin
ligase (14). It contains a RING
finger domain at the C-terminus required for ubiquitin ligase
activity. HRD1 is implicated in the pathogenesis of rheumatoid
arthritis, Alzheimer's disease, liver fibrosis, renal fibrosis,
Parkinson's disease, cancer and Wolfram syndrome (15–19).
Wu et al (16) demonstrated
that Hrd1 is the specific E3 ubiquitin ligase of Nrf2 and induces
Nrf2 ubiquitylation and degradation in embryonic fibroblast cells.
Although Hrd1 could be upregulated in response to oxidative stress,
little is known about the role of Hrd1 in UV-induced skin oxidative
damage. Using skin tissues, upregulation of Hrd1 expression and
downregulation of Nrf2 expression were detected in UV-exposed
fibroblasts from human skin. It was also revealed that ATRA could
reverse the increase of Hrd1 expression induced by UV radiation
in vivo and in vitro. The aim of the present study
was to investigate the role of Hrd1 in the protective effect of
ATRA on human skin fibroblasts exposed to UV.
Materials and methods
Collection of clinical skin
samples
Samples of human skin (n=10) were obtained from
surgeries at the First Affiliated Hospital of Nanjing Medical
University. Samples were divided into 2 groups with the same sex
(all female) and age (average age of sun-exposed group:
40.20±11.39; average age of sun-protected group: 40.00±10.32). The
sun-protected group consisted of breast or back skin areas and the
sun-exposed group consisted of face or neck skin areas. For
immunohistochemical analysis, the skin biopsies were placed in 10%
phosphate-buffered formalin. The present study was approved by the
Local Ethics Committees of the First Affiliated Hospital of Nanjing
Medical University (Nanjing, China) (Permit no. 2016-SRFA-033).
Written informed consent was obtained from all participants in this
study.
Animal model
BALB/ C mice (age, 6–8 weeks; weight, 18–22 g; 20
male and 20 female) were obtained from the Chinese Academy of
Sciences, Shanghai SLAC Laboratory Animal Co., Ltd., and maintained
in a pathogen-free barrier facility at Nanjing Medical University.
The mice were maintained at 22–23°C, 55–60% humidity and a 12-h
light/dark cycle, and provided with certified standard chow and tap
water ad libitum. Mice were randomly divided into 4 groups:
the control group, the UV group, the ATRA group and the UV+ATRA
group. The dorsal area of all mice was shaved with electric
clippers twice a week. The UV source was supplied by Bio-spectra
system (Vilber Lourmat) with a UVB fluorescent bulb with a peak
wavelength at 312 nm and a UVA fluorescent bulb with a peak
wavelength at 365 nm. Mice in the ATRA group received 0.1% ATRA
cream (15 mg ATRA:15 g cream; Chongqing Huapont Pharmaceutical Co.,
Ltd.) on the back, coating 0.1 ml every time. Mice in the UV+ATRA
group received ATRA cream, and then were treated with UV. Mice were
exposed to UVA 10 J/cm2, UVB 30 mJ/cm2 every
day for 14 weeks and the duration of the exposure was 1 h/day.
During the entire experimental process, all efforts were made to
minimize the suffering of the animals, in accordance with the
Institutional Animal Care and Use Committee (IACUC) of Nanjing
Medical University.
Immunohistochemistry (IHC) and
immunofluorescence analyses
Human and mice skin tissues were prepared for
immunohistochemical analysis. Sections (6-µm thick) were
immunolabeled with primary antibodies (Hrd1 1:150; cat. no.
ab118483; Nrf2 1:200; cat. no. ab62352; Abcam). For
immunofluorescent staining, fibroblasts were incubated with primary
antibodies (Hrd1, 1:50; Nrf2, 1:100) for 1 h at room temperature.
After washing with PBST, cells were incubated with secondary
antibodies [Alexa546- (1:200; cat. no. A10040; Thermo Fisher
Scientific, Inc,) or Alexa488- (1:200; cat. no. A21206; Thermo
Fisher Scientific, Inc.) conjugated donkey anti-rabbit IgG,
Alexa350- (1:200; cat. no. A21081; Thermo Fisher Scientific, Inc.)
or Alexa546- (1:200; cat. no. A11056; Thermo Fisher Scientific,
Inc.) conjugated donkey anti-goat Ig) for 45 min. Images were
captured by LSM 700 confocal laser scanning microscope (Zeiss AG),
equipped with a ZEN 2009 software for image acquisition and
analyses.
Cell culture and treatment
HDFs were aseptically isolated from healthy adult
male circumcised foreskins with the approval of the Local Ethics
Committees of the First Affiliated Hospital of Nanjing Medical
University (Permit no. 2016-SRFA-033). Written informed consent was
obtained from all subjects in this study. Normal skin samples were
sterilized in 70% ethanol, minced, put in collagenase for 1 hour
and incubated in Dulbecco's modified Eagle's medium (DMEM, Gibco;
Thermo Fisher Scientific, Inc.) supplemented with 10% fetal bovine
serum (FBS; Sigma-Aldrich; Merck KGaA) and 1%
penicillin-streptomycin (Beijing Solarbio Science & Technology
Co., Ltd.) in an atmosphere of 5% CO2 at 37°C. HDFs were
washed once with PBS and then exposed to UVA irradiation (10
J/cm2) or UVB irradiation (30 mJ/cm2). ATRA
(product no. R2625; Sigma-Aldrich; Merck KGaA) was used to treat
fibroblasts according to a previous study (20). The dosage of ATRA was 1 µmol/l.
Real-time RT-PCR
Total RNA was extracted from cell samples by using
TRIzol (Invitrogen; Thermo Fisher Scientific, Inc.). RNA samples
were used to synthesize cDNA through RT using SuperScript III
Reverse Transcriptase kit (Thermo Fisher Scientific, Inc.)
according to manufacturer's instructions. PCR reaction systems were
prepared using SYBR®-Green Quantitative RTqPCR kit
(Sigma-Aldrich; Merck KGaA). Primers used to identify Hrd1 were:
Forward, 5′-AACCCCTGGGACAACAAGG-3′ and reverse,
5′-TCTGAGCTAGGGATGCTGGT-3′; and the primers for NRF2 were: Forward,
5′-ACACGGTCCACAGCTCATC-3′ and reverse,
5′-TGTCAATCAAATCCATGTCCTG-3′. GAPDH was used as an internal
control: Forward, 5′-TGTTGCCATCAATGACCCCTT-3′ and reverse,
5′-CTCCACGACGTACTCAGCG-3′. Real-time RT-PCR was carried out using
the following thermocycling conditions: 95°C for 50 sec, followed
by 40 cycles at 95°C for 12 sec and 60°C for 45 sec. All data were
processed using the 2−ΔΔCq method (21).
Western blot analysis
Cells were washed twice in ice-cold PBS, and then
solubilized in RIPA lysis buffer (Vazyme). Proteins extracted were
quantified using a Bicinchoninic Acid Protein Assay kit (cat. no.
P0012; Beyotime Institute of Biotechnology). Equal amounts of each
protein sample (30 µg/lane) were subjected to 10% SDS-PAGE and were
transferred to polyvinylidene fluoride membranes, which were
blocked with 5% skimmed milk at room temperature for 1 h. Members
were incubated overnight at 4°C with antibodies targeting Hrd1
(cat. no. ab170901; 1:2,000; Abcam), Nrf2 (cat. no. sc-13032;
1:2,000; Santa Cruz Biotechnology, Inc.) and β-actin (cat. no.
58169, 1:1,000; Cell Signaling Technology, Inc.), followed by
incubation with a horseradish peroxidase-labeled goat anti-rabbit
secondary antibody (cat. no. ab6721; 1:3,000; Abcam) for 1.5 h at
room temperature. β-Actin was used as a loading control. Protein
bands were visualized by enhanced chemiluminescence (cat. no.
WBKLS0500; EMD Millipore). ImageJ 1.45 software (National
Institutes of Health) was used to perform densitometric analysis of
each band.
Co-immunoprecipitation (Co-IP)
The lysates of fibroblasts were incubated with
anti-Hrd1 antibody, anti-Nrf2 antibody, or control IgG for 1 h,
followed by incubation overnight with protein A/G agarose beads
(Thermo Fisher Scientific, Inc.). The beads were collected by
centrifugation (14,000 × g at 4°C for 1 min), washed three times
with the lysis buffer and resuspended in 1X SDS loading buffer. The
IP were eluted from the beads by incubation at 95°C for 5 min. The
eluted proteins were separated by 10% SDS-PAGE and western blotting
was subsequently performed with the indicated antibodies.
Small interfering RNA and recombinant
adenoviruses infection
Small interfering RNA specific for Hrd1 (si-Hrd1-1
and si-Hrd1-2) and control siRNA (si-control) were synthesized
(Guangzhou RiboBio Co., Ltd.) and transfected using Lipofectamine
2000. The sequences of si-Hrd1-1 and si-Hrd1-2 were:
5′-CCAUGAGGCAGUUCAAGAAdTdT-3′ and 5′-UGUCUGGCCUUCACCGUUU-3,
respectively. Human fibroblasts (2×105/well) were
infected with 1×108 pfu of Hrd1 recombinant adenoviruses
according to the manufacturer's instructions (GeneChem, Shanghai,
China). The expression level of Hrd1 was assessed by western
blotting.
Intracellular reactive oxygen species
(ROS) detection
Intracellular ROS content were assessed by
determination of 2′, 7′-dichlorofluorescin diacetate (DCFH-DA,
Beyotime Institute of Biotechnology, Inc.), which is converted into
fluorescent 2′, 7′-dichlorofluorescin (DCF) in the presence of
peroxides (22). Human fibroblasts
were incubated with a 10-µM DCFH-DA probe for 30 min and then
harvested and washed with PBS. Images were captured using a
fluorescence microscope (DP70; Olympus Corporation) and the
fluorescence (emission, 525 nm; excitation, 488 nm) was assessed
using a fluorescence plate reader (BD Falcon; BD Biosciences).
WST-1 assay
Cell viability was determined using the WST-1 assay.
Human fibroblasts were seeded in 48-well dishes (4×104
cells/well) in 200 µl culture medium and transfected and treated as
aforementioned for 48 h. Then, each well was supplemented with 20
µl WST-1 (Roche Diagnostics) and incubated for 3 h. The absorbance
of the samples was measured with a spectrophotometer reader.
Statistical analysis
The data in the present study were analyzed using
SPSS 19.0 (IBM Corpοration) statistical software and presented as
the means ± SD. The analysis to determine the statistical
differences among the groups was performed using the Student's
t-test or one-way analysis of variance (ANOVA) followed by
Student-Newman-Keuls post hoc test. A P-value <0.05 was
considered to indicate a statistically significant difference.
Results
UV induces upregulation of Hrd1
expression and downregulation of Nrf2 expression in human skin
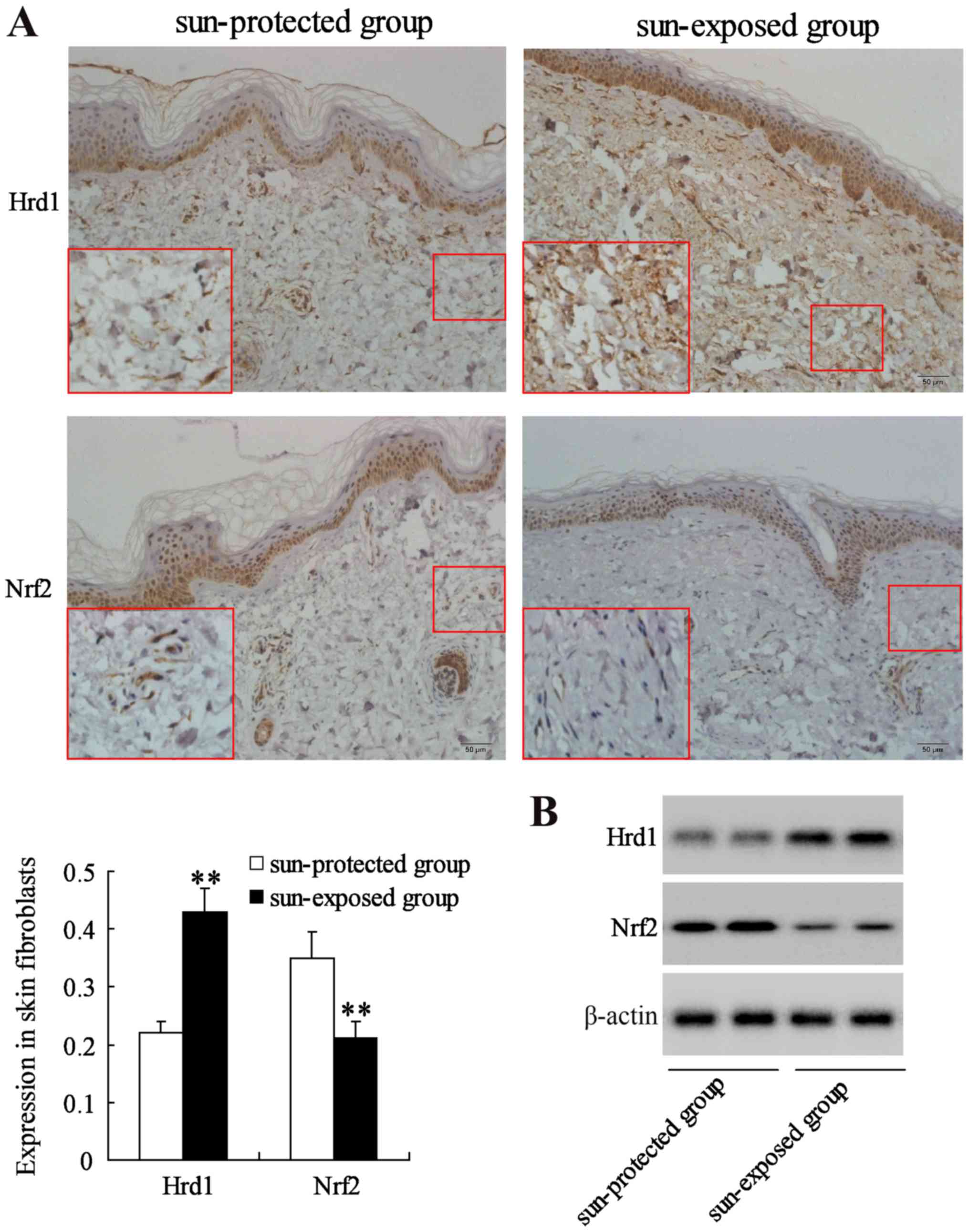
The expression of Hrd1 and Nrf2 was first evaluated
in human skin fibroblasts from a sun-protected group (10 samples)
and a sun-exposed group (10 samples) with same sex and age. IHC and
western blot analysis revealed that the expression level of Hrd1
was significantly increased but the expression level of Nrf2 was
significantly decreased in fibroblasts from sun-exposed skin
compared to that in fibroblasts from sun-protected skin (P<0.01)
(Fig. 1).
ATRA reverses Hrd1 expression
increased by UV radiation in an animal model
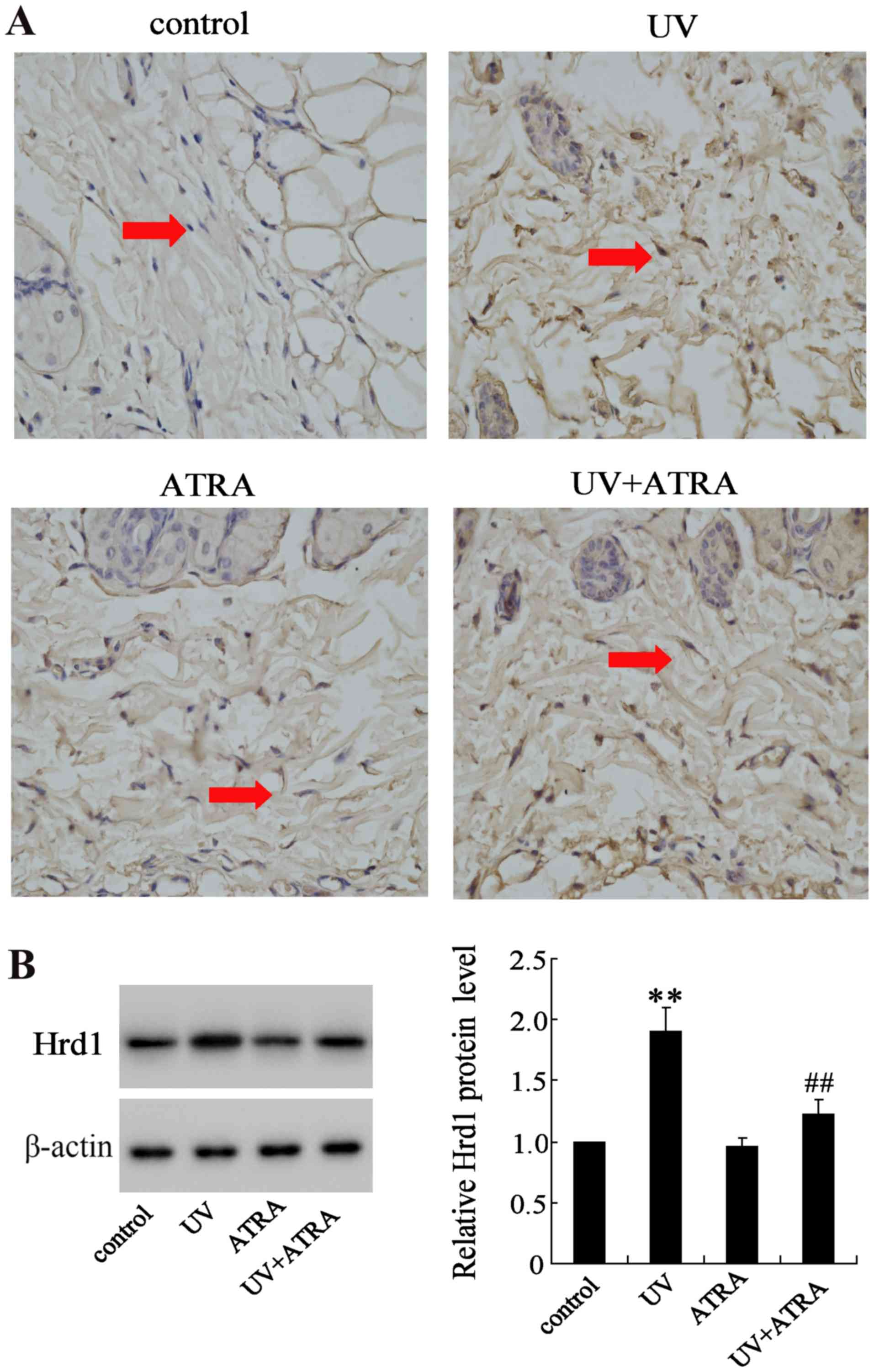
The effect of ATRA on Hrd1 expression was also
evaluated in fibroblasts from mice exposed to UV radiation. IHC and
western blot analysis revealed that expression level of Hrd1 was
significantly increased in fibroblasts from mice exposed to UV
compared with the control group, which could be partially reversed
by ATRA (Fig. 2).
ATRA decreases Hrd1 expression and
increases Nrf2 expression in human fibroblasts exposed to UV
radiation
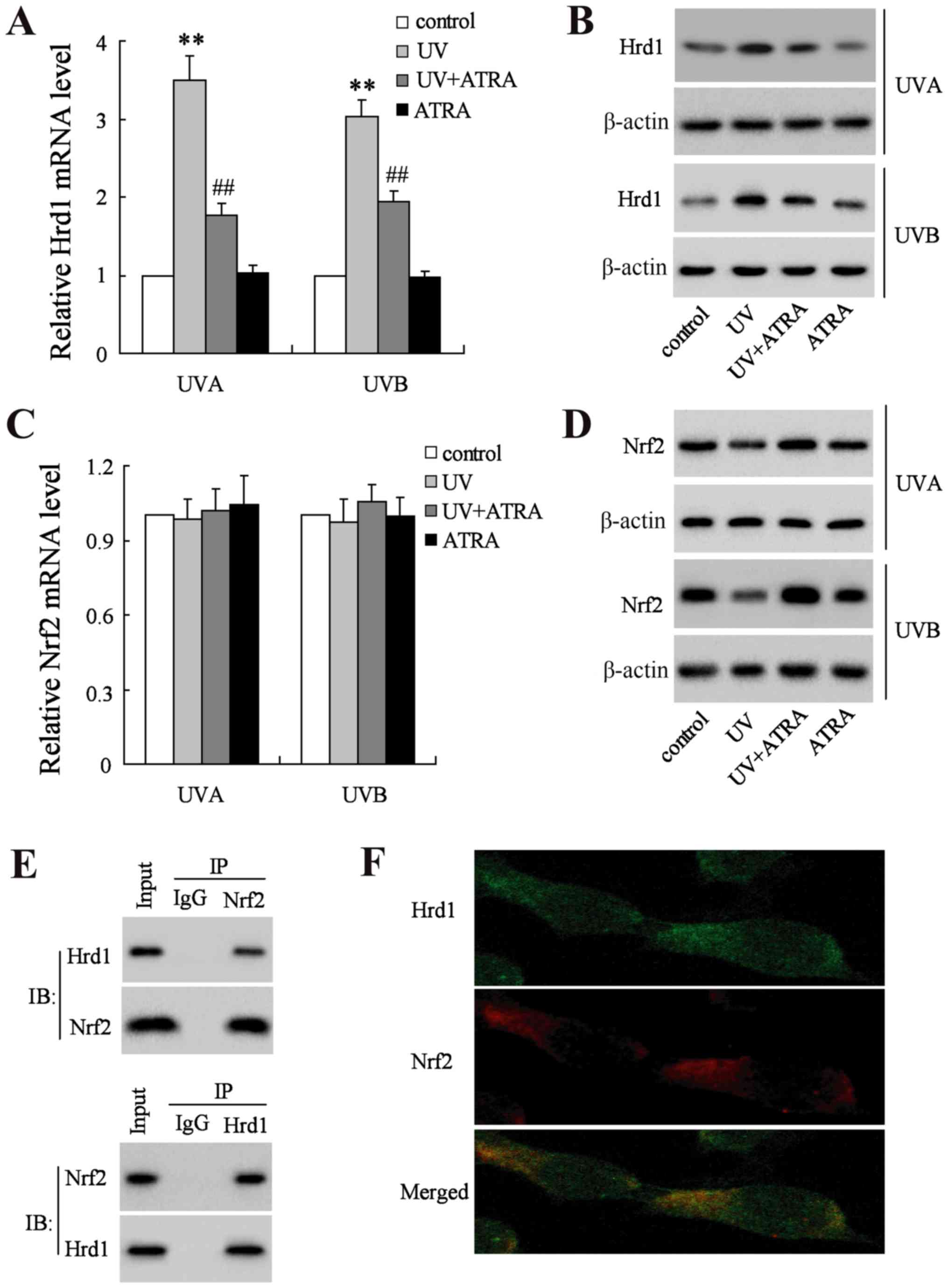
UVA and UVB increased the mRNA and protein levels of
Hrd1, which could be reversed by ATRA in human fibroblasts
(Fig. 3A and B). Next, the mRNA
and protein level of Nrf2 was assessed. As revealed in Fig. 3C, neither UV nor ATRA had an effect
on the mRNA level of Nrf2. Notably, the level of Nrf2 protein was
significantly decreased in human fibroblasts exposed UV radiation,
which could be restored by ATRA (Fig.
3D). To explore whether Hrd1 could interact with Nrf2 in
fibroblasts, Co-IP and immunofluorescence analyses were used to
assess the co-localization between Hrd1 and Nrf2. Co-IP analysis
demonstrated that endogenously expressed Hrd1 and Nrf2 co-existed
in precipitated complexes in fibroblasts (Fig. 3E). In addition, Hrd1 co-localized
with the Nrf2 in fibroblasts as revealed in Fig. 3F.
Downregulation of Hrd1 expression
increases the expression level of Nrf2 in UV-irradiated human
dermal fibroblasts
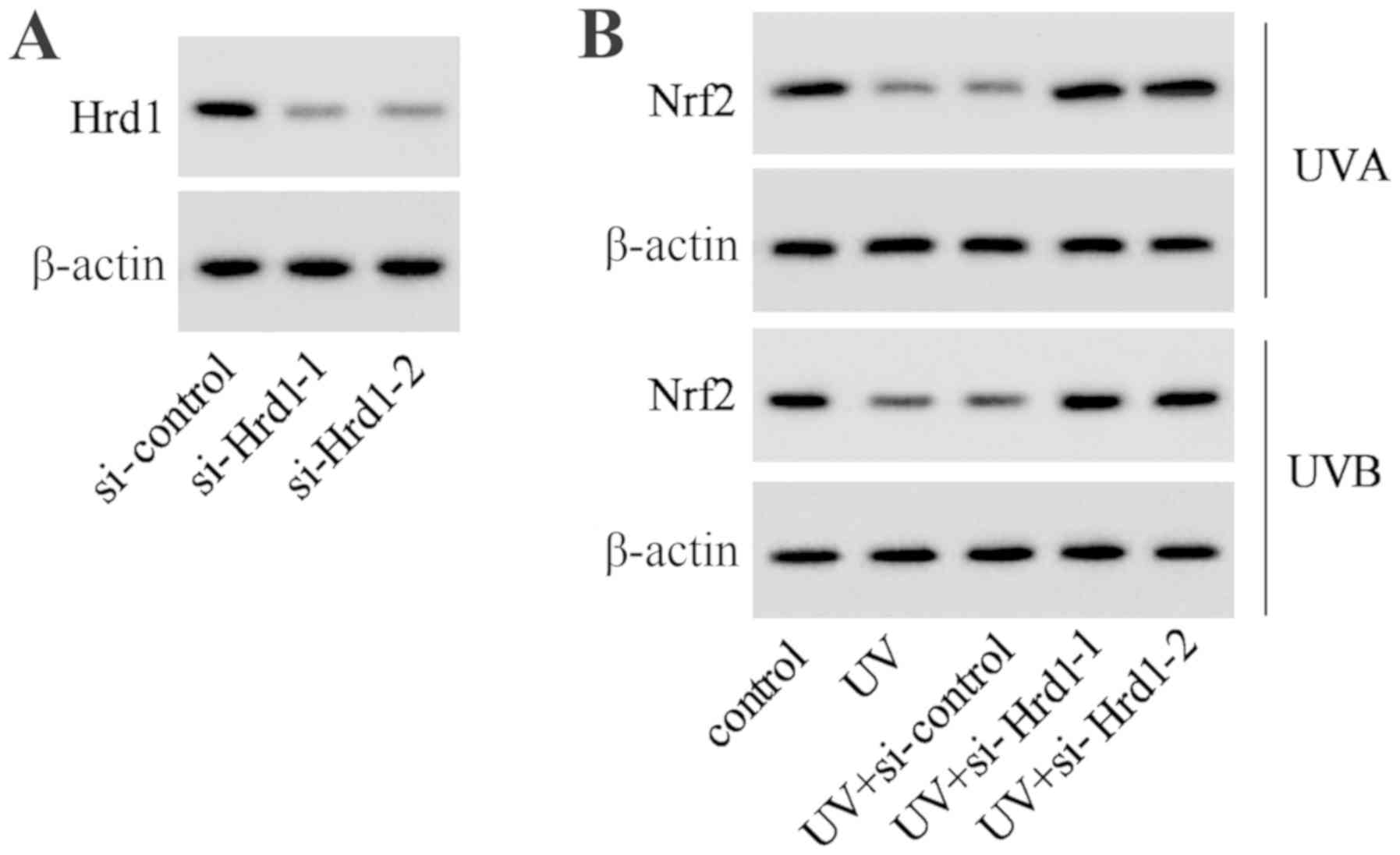
To investigate whether Hrd1 was involved in the
decrease of Nrf2 expression induced by UV, siRNA was used to
knockdown Hrd1 expression in human dermal fibroblasts. As revealed
in Fig. 4A, Hrd1 siRNA (si-Hrd1-1
and si-Hrd1-2) could decrease Hrd1 expression, indicating that
si-Hrd1-1 and si-Hrd1-2 were efficiently introduced into human
fibroblasts and acted to knock down Hrd1. After UV exposure, the
expression of Nrf2 was significantly decreased. Silencing of Hrd1
could increase Nrf2 expression in human fibroblasts exposed to UV
radiation (Fig. 4B).
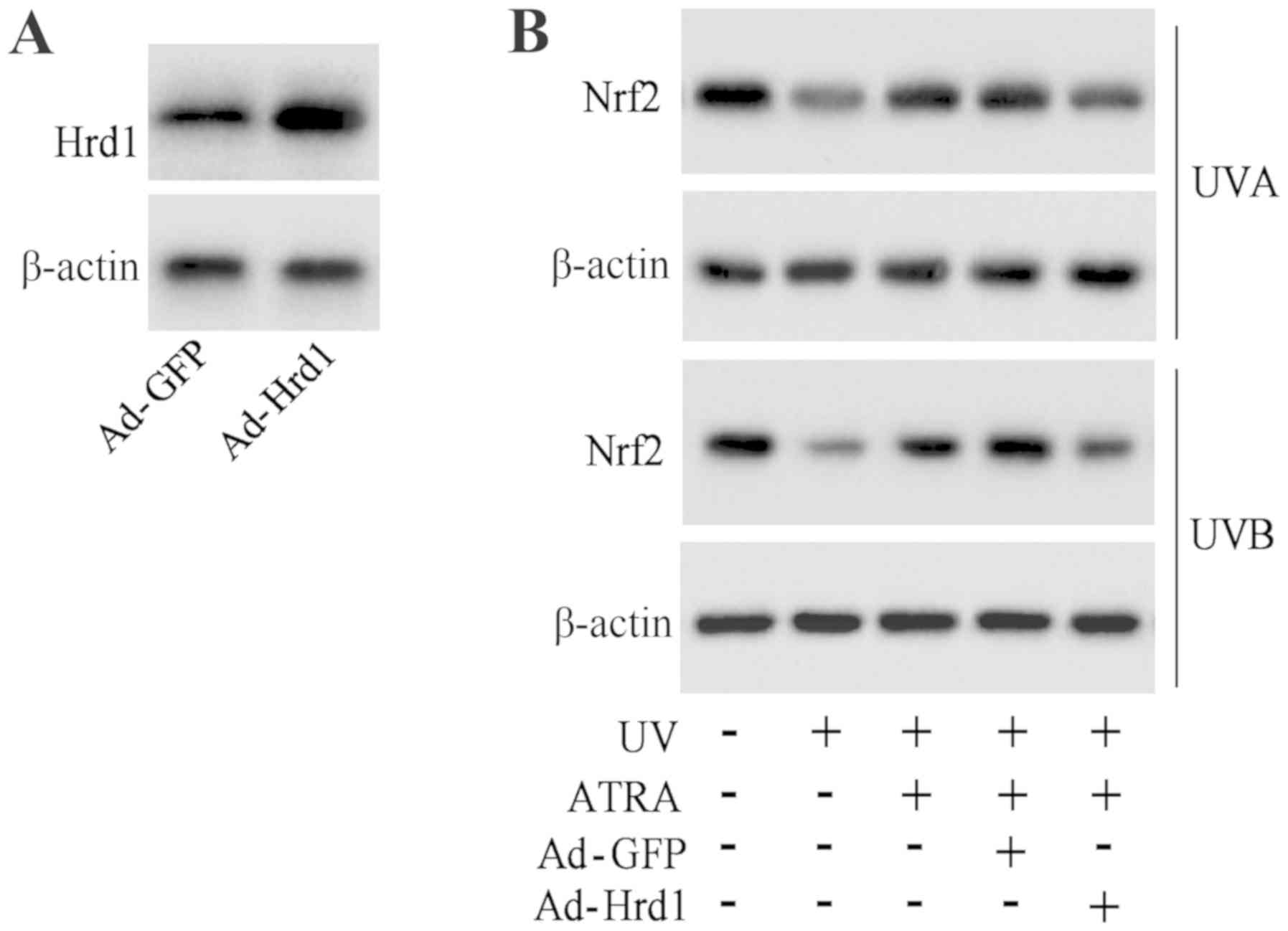
Overexpression of Hrd1 abolishes the
protective effect of ATRA against the UV-induced decrease of Nrf2
expression
To investigate that ATRA prevented the decrease of
Nrf2 protein expression against UV radiation through Hrd1, human
fibroblasts were infected with Hrd1 recombinant adenoviruses
(Ad-Hrd1). As revealed in Fig. 5A,
Ad-Hrd1 significantly increased Hrd1 expression. ATRA treatment
significantly increased Nrf2 expression in fibroblasts exposed to
UV, which was reversed by overexpression of Hrd1 (Fig. 5B).
Overexpression of Hrd1 abolishes the
protective effect of ATRA against UV-induced ROS production and
cytotoxicity
The role of Hrd1 on the protective effect of ATRA
against UV-induced oxidative stress was assessed in human dermal
fibroblasts. As revealed in Fig.
6A, ATRA treatment efficiently inhibited UV-induced ROS
production, which could be abolished by overexpression of Hrd1.
Next, the role of Hrd1 on the protective effect of ATRA against
UV-induced cytotoxicity was investigated. As observed in Fig. 6B, ATRA treatment significantly
increased viability of fibroblasts exposed to UV radiation.
Similarly, overexpression of Hrd1 abolished the protective effect
of ATRA against UV-induced cytotoxicity.
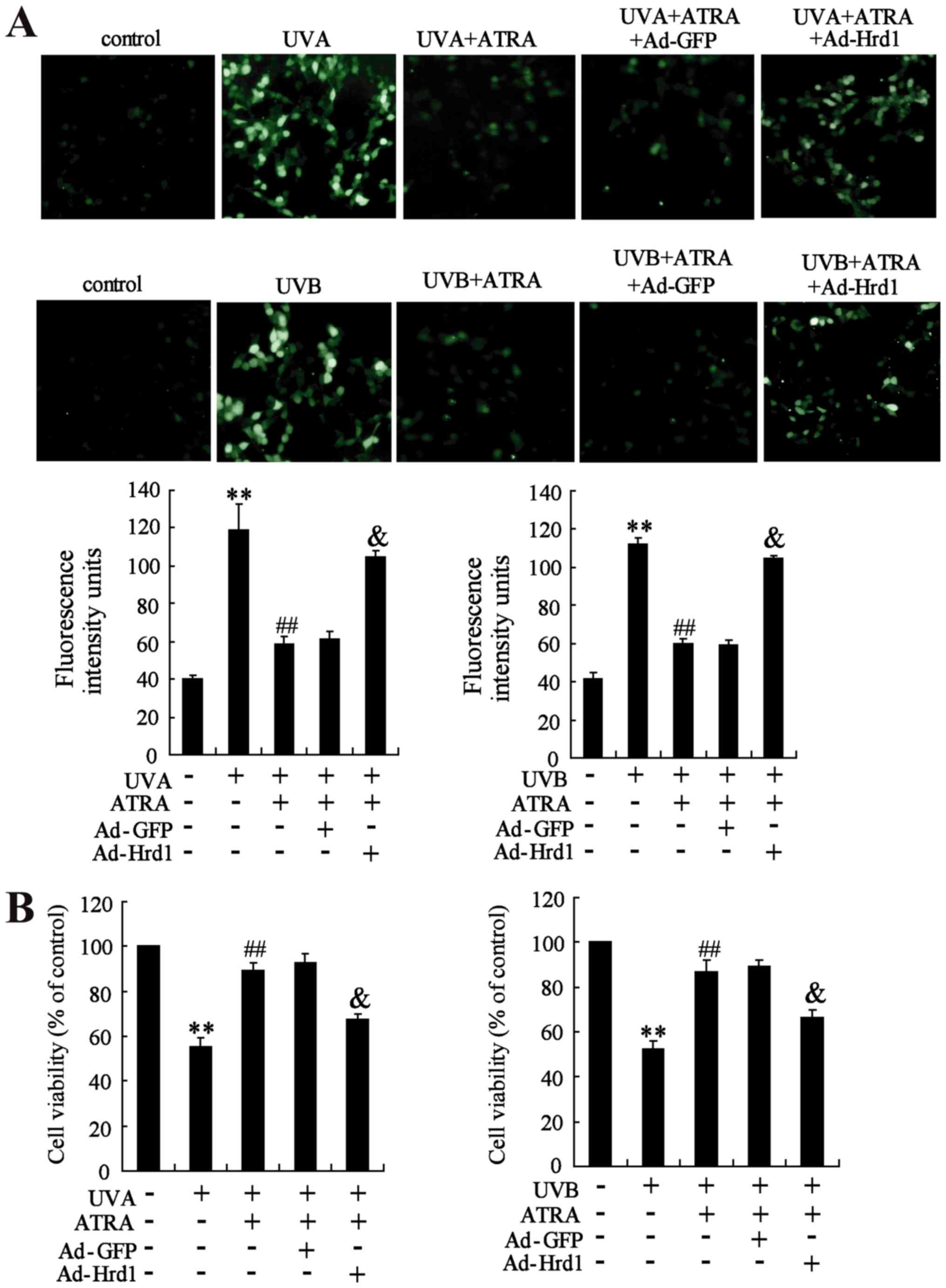 | Figure 6.Overexpression of Hrd1 abolishes the
protective effect of ATRA against UV-induced ROS production and
cytotoxicity. Human fibroblasts were infected with ad-Hrd1 for 24
h, and then, exposed to UVA and UVB, and treated with ATRA for 24
h. (A) Fluorescence analyses were performed using a fluorescence
microscope and quantified ROS production was detected. (B) Cell
viability was assessed by WST-1 assay. Compared to control,
**P<0.01; compared to UV, ##P<0.01; compared to
UV+ATRA+Ad-GFP, &P<0.01. Hrd1,
3-hydroxy-3-methylglutaryl reductase degradation; ATRA,
all-trans retinoic acid; UV, ultraviolet; ROS, reactive
oxygen species. |
Discussion
ATRA has been clinically used to treat several skin
diseases including UV-induced skin oxidative damage (11,12).
In the present study, it was demonstrated that Hrd1 was involved in
the protective effect of ATRA against UV-induced ROS production and
cytotoxicity in skin fibroblasts. Hrd1 expression was significantly
increased in UV-exposed human or mice skin fibroblasts. Moreover,
ATRA could reverse the increase of Hrd1 expression induced by UV
radiation in vivo and in vitro. In addition, ATRA or
knockdown of Hrd1 could increase Nrf2 expression in fibroblasts
exposed to UV radiation, and Hrd1 could directly interact with Nrf2
in skin fibroblasts. Notably, overexpression of Hrd1 abolished the
protective effect of ATRA on the UV-induced decrease of Nrf2
expression, the production of reactive oxygen species (ROS) and the
decrease of cell viability.
Hrd1 has been demonstrated to be upregulated in
response to oxidative stress (23). In the present study, it was
determined that the level of Hrd1 was higher in UV-exposed human or
mice skin fibroblasts. A previous study revealed that Hrd1 was the
specific E3 ubiquitin ligase of Nrf2 for ubiquitylation and
degradation (16,24). It was revealed that Hrd1 and Nrf2
were both in the cytoplasm, and interaction between Hrd1 and Nrf2
was strong in skin fibroblasts. It was also determined that there
was an inverse correlation of HRD1 expression and Nrf2 expression
in fibroblasts treated with UV radiation. Notably, knockdown of
Hrd1 could reverse the decrease of Nrf2 expression in fibroblasts
exposed to UV radiation. In the present study, a connection among
UV radiation, Hrd1 increase, and Nrf2 degradation was established.
Hrd1 was responsible for the decrease of Nrf2 under UV light
irradiation.
It has been reported that Nrf2 acts as a master
regulator of the cellular antioxidant defense against cutaneous
photodamage mediated by UV radiation (8,9).
However, the effect of UV irradiation on Nrf2 expression and
activity in skin fibroblasts has remained controversial. Certain
studies demonstrated that UV irradiation led to Nrf2 activation and
accumulation in the nucleus (25,26),
while others revealed that Nrf2 was downregulated and transported
from the nucleus in dermal fibroblasts exposed to UV irradiation
(27,28). The different results may be due to
the cell source, the time and the dose of UV exposure in different
laboratories. It was observed that UV could downregulate Nrf2
expression using human-derived fibroblasts. Given that Nrf2 could
combat oxidative damage, it was speculated that pharmacological
upregulation of Nrf2 could protect skin fibroblasts against
UV-induced injury.
ATRA has been reported to protect skin fibroblasts
against UV-induced injury during which oxidative stress plays an
important pathological role (29,30).
Τhe role of ROS and antioxidant signaling was therefore examined in
the ATRA-mediated protective effect. It was demonstrated that ATRA
could abolish the decrease of Nrf2 expression in fibroblasts
exposed to UV radiation. Although ATRA at high concentrations (≥30
µmol/l) can induce cytotoxicity and activate Nrf2 (13), low concentrations of ATRA alone (1
µmol/l) had no effect on Nrf2 expression (Fig. 3D) and cell viability (data not
shown) in the present study. Thus, ATRA treatment only resulted in
the decrease of ROS production induced by UV. These results led us
to ask the question of how ATRA eliminated UV-induced ROS
generation. The present study, for the first time, revealed that
the ubiquitin E3 ligate Hrd1 was involved in the protective effect
of ATRA on UV-induced oxidative damage in skin fibroblasts.
In conclusion, our observations revealed that ATRA
decreased Hrd1 expression in skin fibroblasts exposed to UV
irradiation, leading to upregulation of Nrf2 expression and
ultimately to the reduction of ROS production and cytotoxicity. The
present study shed light on the molecular mechanisms responsible
for the protective effect of ATRA on skin fibroblasts against
UV-induced oxidative damage.
Acknowledgements
Not applicable.
Funding
The present study was supported by the National
Natural Science Foundation of China Grant (no. 81673082) and the
Six Talent Peaks Project of Jiangsu Province (no. 2008101).
Availability of data and materials
All data generated and/or analyzed during the
present study are available from the corresponding author upon
reasonable request.
Authors' contributions
XC and WQ performed the experiments, analyzed the
data and drafted the manuscript. FC performed the supplementary
experiments, contributed to the interpretation of the data and
revised the manuscript. YJ and SRL performed the experiments of the
clinical skin samples and the animal model. FW and XL assisted with
the molecular biology experiments. DS and BC conceived and designed
the study and revised the manuscript. All authors read and approved
the final manuscript and agree to be accountable for all aspects of
the research in ensuring that the accuracy or integrity of any part
of the work are appropriately investigated and resolved.
Ethics approval and consent to
participate
This study was approved by the Local Ethics
Committees of the First Affiliated Hospital of Nanjing Medical
University (Nanjing, China) (Permit no. 2016-SRFA-033). Written
informed consent was obtained from all participants in this study.
All animal protocols were approved by the Institutional Animal Care
and Use Committee (IACUC) of Nanjing Medical University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Vedrenne N, Coulomb B, Danigo A, Bonté F
and Desmoulière A: The complex dialogue between (myo)fibroblasts
and the extracellular matrix during skin repair processes and
ageing. Pathol Biol (Paris). 60:20–27. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cavinato M and Jansen-Dürr P: Molecular
mechanisms of UVB-induced senescence of dermal fibroblasts and its
relevance for photoaging of the human skin. Exp Gerontol. 94:78–82.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wenk J, Brenneisen P, Meewes C, Wlaschek
M, Peters T, Blaudschun R, Ma W, Kuhr L, Schneider L and
Scharffetter-Kochanek K: UV-induced oxidative stress and
photoaging. Curr Probl Dermatol. 29:83–94. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Oliveira MM, Daré RG, Barizão ÉO,
Visentainer JV, Romagnolo MB, Nakamura CV and Truiti MDCT:
Photodamage attenuating potential of Nectandra hihua against
UVB-induced oxidative stress in L929 fibroblasts. J Photochem
Photobiol B. 181:127–133. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liang B, Peng L, Li R, Li H, Mo Z, Dai X,
Jiang N, Liu Q, Zhang E, Deng H, et al: Lycium barbarum
polysaccharide protects HSF cells against ultraviolet-induced
damage through the activation of Nrf2. Cell Mol Biol Lett.
23:182018. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Parzonko A and Kiss AK: Caffeic acid
derivatives isolated from Galinsoga parviflora herb protected human
dermalfibroblasts from UVA-radiation. Phytomedicine. 57:215–222.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Silva-Palacios A, Ostolga-Chavarría M,
Zazueta C and Königsberg M: Nrf2: Molecular and epigenetic
regulation during aging. Ageing Res Rev. 47:31–40. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhong JL, Edwards GP, Raval C, Li H and
Tyrrell RM: The role of Nrf2 in ultraviolet a mediated heme
oxygenase 1 induction in human skin fibroblasts. Photochem
Photobiol Sci. 9:18–24. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Schäfer M, Dütsch S, auf dem Keller U and
Werner S: Nrf2: A central regulator of UV protection in the
epidermis. Cell Cycle. 9:2917–2918. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Saw CL, Huang MT, Liu Y, Khor TO, Conney
AH and Kong AN: Impact of Nrf2 on UVB-induced skin
inflammation/photoprotection and photoprotective effect of
sulforaphane. Mol Carcinog. 50:479–486. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Francz PI, Conrad J and Biesalski HK:
Modulation of UVA-induced lipid peroxidation and suppression of
UVB-induced ornithine decarboxylase response by all-trans retinoic
acid in human skin fibroblasts in vitro. Biol Chem. 379:1263–1269.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hiraishi Y, Hirobe S, Iioka H, Quan YS,
Kamiyama F, Asada H, Okada N and Nakagawa S: Development of a novel
therapeutic approach using a retinoic acid-loaded microneedle patch
for seborrheic keratosis treatment and safety study in humans. J
Control Release. 171:93–103. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tan KP, Kosuge K, Yang M and Ito S: NRF2
as a determinant of cellular resistance in retinoic acid
cytotoxicity. Free Radic Biol Med. 45:1663–1673. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kaneko M, Okuma Y and Nomura Y: Molecular
approaches to the treatment, prophylaxis, and diagnosis of
Alzheimer's disease: Possible involvement of HRD1, a novel molecule
related to endoplasmic reticulum stress, in Alzheimer's disease. J
Pharmacol Sci. 118:325–330. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Amano T, Yamasaki S, Yagishita N,
Tsuchimochi K, Shin H, Kawahara K, Aratani S, Fujita H, Zhang L,
Ikeda R, et al: Synoviolin/Hrd1, an E3 ubiquitin ligase, as a novel
pathogenic factor for arthropathy. Genes Dev. 17:2436–2449. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wu T, Zhao F, Gao B, Tan C, Yagishita N,
Nakajima T, Wong PK, Chapman E, Fang D and Zhang DD: Hrd1
suppresses Nrf2-mediated cellular protection during liver
cirrhosis. Genes Dev. 28:708–722. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim H, Bhattacharya A and Qi L:
Endoplasmic reticulum quality control in cancer: Friend or foe.
Semin Cancer Biol. 33:25–33. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Nomura J, Hosoi T, Kaneko M, Ozawa K,
Nishi A and Nomura Y: Neuroprotection by endoplasmic reticulum
stress-induced HRD1 and chaperones: Possible therapeutic targets
for Alzheimer's and Parkinson's disease. Med Sci (Basel). 4(pii):
E142016.PubMed/NCBI
|
|
19
|
Fonseca SG, Ishigaki S, Oslowski CM, Lu S,
Lipson KL, Ghosh R, Hayashi E, Ishihara H, Oka Y, Permutt MA and
Urano F: Wolfram syndrome 1 gene negatively regulates ER stress
signaling in rodent and human cells. J Clin Invest. 120:744–755.
2010. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Shim JH, Shin DW, Noh MS and Lee TR:
Reduced collagen internalization via down-regulation of MRC2
expression by UVA irradiation and its recovery by all-trans
retinoic acid. J Dermatol Sci. 73:163–166. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Afri M, Frimer AA and Cohen Y: Active
oxygen chemistry within the liposomal bilayer. Part IV: Locating
2′,7′-dichlorofluorescein (DCF), 2′,7′-dichlorodihydrofluorescein
(DCFH) and 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) in
the lipid bilayer. Chem Phys Lipids. 131:123–133. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Saito R, Kaneko M, Kitamura Y, Takata K,
Kawada K, Okuma Y and Nomura Y: Effects of oxidative stress on the
solubility of HRD1, a ubiquitin ligase implicated in Alzheimer's
disease. PLoS One. 9:e945762014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
O'Connell MA and Hayes JD: The Keap1/Nrf2
pathway in health and disease: From the bench to the clinic.
Biochem Soc Trans. 43:687–689. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Hirota A, Kawachi Y, Itoh K, Nakamura Y,
Xu X, Banno T, Takahashi T, Yamamoto M and Otsuka F: Ultraviolet A
irradiation induces NF-E2-related factor 2 activation in dermal
fibroblasts: Protectiverole in UVA-induced apoptosis. J Invest
Dermatol. 124:825–832. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Gęgotek A, Rybałtowska-Kawałko P and
Skrzydlewska E: Rutin as a mediator of lipid metabolism and
cellular signaling pathways interactions in fibroblasts altered by
UVA and UVB radiation. Oxid Med Cell Longev. 2017:47213522017.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Saito Y, Tsuruma K, Ichihara K, Shimazawa
M and Hara H: Brazilian green propolis water extract up-regulates
the early expression level of HO-1 and accelerates Nrf2 after UVA
irradiation. BMC Complement Altern Med. 15:4212015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kannan S and Jaiswal AK: Low and high dose
UVB regulation of transcription factor NF-E2-related factor 2.
Cancer Res. 66:8421–8429. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nilsson J, Gritli-Linde A and Heby O: Skin
fibroblasts from spermine synthase-deficient hemizygous gyro male
(Gy/Y) mice overproduce spermidine and exhibit increased resistance
to oxidative stress but decreased resistance to UV irradiation.
Biochem J. 352:381–387. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu PY, Huang CC, Chu Y, Huang YH, Lin P,
Liu YH, Wen KC, Lin CY, Hsu MC and Chiang HM: Alleviation of
ultraviolet B-induced photodamage by coffea arabica extract in
human skin fibroblasts and hairless mouse skin. Int J Mol Sci.
18:E7822017. View Article : Google Scholar : PubMed/NCBI
|