Introduction
Lymphoblastic lymphoma (LBL) is an aggressive
neoplasm of precursor T and B cells, also known as lymphoblasts.
Precursor T-cell lymphoblastic lymphoma (T-LBL) accounts for ~90%
of LBL cases (1). It is predicted
to derive from malignant thymocytes that may appear at defined
stages of intrathymic T-cell differentiation (1). Patients are typically young males
with a median age of ~20 years (2). The disease is associated with
progressive infiltration of lymphoblasts into nodal structures or
extranodal structures, particularly the bone marrow, spleen and
central nervous system (CNS), leading to life-threatening
compression of adjacent structures (3). T-LBL is a chemotherapy-sensitive
disease with a 75–85% event-free survival rate using current
treatment regimens (2,4); however, 5-year overall survival
remains poor for T-LBL, mainly due to relapse, often as a result of
spread to the CNS (2,4). Thus, suppressing the viability and
invasive ability of T-LBL tumor cells may ameliorate T-LBL and
improve the prognosis of patients.
There is limited information regarding cytogenetic
and molecular abnormalities in T-LBL due to the rarity of the
disease; it has been reported that the disease is associated with
chromosomal rearrangements and submicroscopic DNA copy number
alterations (5). The majority of
chromosomal abnormalities involve juxtaposition of strong promoters
and enhancers from T-cell receptor genes with transcriptional
factor genes, including T-cell acute lymphocytic leukemia 1,
lymphoblastic leukemia-derived sequence 1 and homeobox 11 (HOX11)
(6). Additionally, a number of
T-LBL cases have been reported to exhibit mutations in the key
factors of Notch1 signaling, including those involved in
ubiquitin-mediated protein degradation to regulate Notch1 protein
levels, and various downstream targets that can induce cell
transformation via the regulation of cell survival, proliferation
and metabolism (7–11). Aberrantly upregulated Notch1
signaling has been observed in the majority of T-LBL cases, and
thus Notch1 has been considered to be one of the key regulators in
early T-cell development and T-LBL oncogenesis (12).
MicroRNAs (miRNAs/miRs) are endogenous small
noncoding RNAs composed of 22–24 nucleotides, acting as
post-transcriptional gene regulators (13,14).
Aberrant expression of miRNAs can be oncogenic or
tumor-suppressive, due to altered regulation of cell migration,
invasion, proliferation, apoptosis and chemoresistance (13,14).
Recent studies have indicated the importance of miRNAs in malignant
T-cell transformation (13,14).
miR-338-3p is encoded in the seventh intron of the
apoptosis-associated tyrosine kinase gene. Its function was first
associated with prion-induced neurodegeneration, as its expression
is reduced in the brains of mice infected with mouse-adapted
scrapie (15). Its expression was
also observed to be dysregulated in a variety of cancer types, such
as lung cancer (16); however, the
role of miR-338-3p in lymphoblastic lymphoma is yet to be
reported.
In the present study, it was reported that the
expression levels of miR-338-3p were reduced in the tissues of
patients with T-LBL and in a T-LBL cell line. Its normal expression
appeared to be required to inhibit the proliferation and migration
of cancer cells via direct regulation of HOXA3 mRNA. The present
study provided a novel insight into the molecular mechanisms
underlying dysregulated signaling networks that may be involved in
the pathogenesis of T-LBL.
Materials and methods
Tissue samples
T-cell lymphoblastic lymphoma (T-LBL) tissues and
adjacent normal tissues were surgically isolated from 38 patients
at The Affiliated Yantai Yuhuangding Hospital of Qingdao University
between February 2013 and March 2015. Inclusion criteria were: i)
Newly diagnosed cases; ii) no therapies performed. Exclusion
criteria were: i) Recurrent cases; ii) other clinical disorders
observed. Patients were staged according to Ann Arbor staging
system (17). The tissues were
flash-frozen in liquid nitrogen and stored at −80°C until
subsequent use. The application of human samples was approved by
the Human Ethics Committee/Institutional Review Board of the
Affiliated Yantai Yuhuangding Hospital of Qingdao University, and
all patients provided informed consent.
Cell culture and transfection
The T-LBL cell line SUP-T1, two T-cell lymphoblastic
leukemia cell lines (CCRF-CEM and Jurkat) and one human T-cell line
(H9) were purchased from the American Type Culture Collection. The
cells were cultured in RPMI-1640 medium (Gibco; Thermo Fisher
Scientific, Inc.) supplemented with 10% heat-inactivated fetal
bovine serum (FBS; Thermo Fisher Scientific, Inc.). The cultured
cells were incubated at 37°C with 5% CO2.
miR-338-3p mimics, miR-338-3p inhibitor, negative
control (NC) and inhibitor NC were designed and synthesized by
Shanghai GenePharma Co., Ltd. Sequences were: miR-338-3p mimics,
5′-UUUGAGCAGCACUCAUUUUUGC-3′; negative control (NC),
5′-CAGUACUUUUAGUGUGUACAA-3′; miR-338-3p inhibitor,
5′-CAACAAAAUCACUGAUGCUGGA-3′; inhibitor NC,
5′-UUGUAAGUUGCGACAGCCACUCA-3′.A homeobox A3 (HOXA3) plasmid for
HOXA3 overexpression vector (pcDNA3.1 plasmid), and an empty vector
(pcDNA3.1 plasmid) used as the NC, were purchased from Guangzhou
RiboBio Co., Ltd. Nucleic acids (50 nM miRNA and 150 nM inhibitor)
were mixed with Lipofectamine® 2000 (Invitrogen; Thermo
Fisher Scientific, Inc.) and transfected into cells according to
the manufacturer's protocol. Briefly, approximately
5–6×105 SUP-T1 cells were seeded and cultured overnight
to reach 90–95% confluency at the time of transfection. Nucleic
acids and Lipofectamine® 2000 were diluted in
transfection medium. The reagents were then mixed and incubated at
room temperature for 5 min prior to the addition of cells. The
cells were further incubated at 37°C in a CO2 incubator
until subsequent assays were performed. Cells were harvested at 48
h post-transfection for subsequent experimentation.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cells or tissues using
TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.),
according to the manufacturer's protocol. Total RNA (1 µg) was
reverse transcribed into cDNA using specific stem-loop RT primers
and Mir-XTM miRNA First Strand Synthesis kit (Takara Biotechnology
Co., Ltd.) or the Affinity-Script QPCR cDNA Synthesis kit (Agilent
Technologies, Inc.); RT reaction was carried out at 42°C for 60 min
and was terminated by heating at 70°C for 5 min. qPCR reactions
were performed according to the standard SYBR Green assay protocol,
and the signals were detected using an ABI PRISM 7500 Sequence
Detection system (Applied Biosystems; Thermo Fisher Scientific,
Inc.). qPCR was performed using a SYBR Premix Ex Taq kit (Takara
Biotechnology Co., Ltd.) to determine HOXA3 expression. U6 small
nuclear RNA and GAPDH were used as normalization controls for the
quantification of miR-338-3p and HOXA3 expression, respectively.
PCR settings were: Preheating at 95°C for 10 min, followed by 40
cycles of 95°C for 10 sec, 60°C for 20 sec, and 72°C for 10 sec.
The relative expression of each gene was determined using the
2−∆∆Cq method (18).
The primers used for qPCR were as follows: miR-338-3p, forward
5′-TGCGGTCCAGCATCAGTGAT-3′, reverse 5′-CCAGTGCAGGGTCCGAGGT-3′;
HOXA3, forward 5′-TGGCAGTTTTACTTTATTGTCATAGC-3′, reverse
5′CATCAGAAATGTCCTGCTTGC-3′. U6, forward, 5′-GCTCGCTTCGGCAGCACA-3′;
U6, reverse, 5′-GAGGTATTCGCACCAGAGGA-3′; GAPDH, forward,
5′-ACACCCACTCCTCCACCTTT-3′; GAPDH, reverse,
5′-TTACTCCTTGGAGGCCATGT-3′.
Cell proliferation assay
Cells were transfected and seeded in 96-well plates
at 2×103 cells/well. The cells were further cultured in
a 37°C, 5% CO2 incubator for 0, 24, 48 and 72 h. Then,
10 µl of Cell Counting Kit-8 (CCK-8; Beyotime Institute of
Biotechnology) reagent was added to each well. The 96-well plates
were further incubated for 2 h under the aforementioned conditions.
The absorbance was detected at 450 nm using a microplate
reader.
Cell migration assay
Following transfection and incubation for 24 h,
cells were further cultured in serum-free medium for 12 h. The
cells were suspended, and the concentrations were adjusted to
4–5×105 cells/ml. Transwell chambers with 8-µm pores
(Corning, Inc.) were placed in 24-well plates. RPMI-1640 medium
(500 µl) containing 10% FBS was added to the lower chambers, and
suspended cells (200 µl) were plated in the upper chambers. The
plates were further incubated for 10 h at 37°C. Cells on the lower
surface of the chamber were fixed with 20% glacial acetic acid for
15–30 min and stained with 0.1% crystal violet for 30 min at 37°C.
The total number of cells was estimated from 10 randomly selected
fields. An inverted microscope (Nikon Corporation) was used;
magnification, ×400.
Luciferase reporter assay
According to predictions made using the TargetScan
database (release 7.2; http://www.targetscan.org), a putative
miR-338-3p-binding site was identified in the 3′-untranslated
region (3′-UTR) of the HOXA3 gene. The HOXA3 wild-type (WT) 3′-UTR
sequence and a mutant version (MT) were amplified via PCR and
cloned into the pGL3 dual-luciferase reporter vector (Promega
Corporation) to create pGL3-HOXA3-3′-UTR-WT (WT vector) and
pGL3-HOXA3-3′-UTR-MT (MT vector). Source of the DNA was human HOXA3
cDNA; polymerase: Pyrobest DNA polymerase (Fermentas; Thermo Fisher
Scientific, Inc.). The primers used were: WT, forward,
5′-CCGCTCGAGGAGCCAGGAGTCACTAGG-3′, and reverse,
5′-TAAGCGGCCGCTTGTTTTCAGAACGTGAG-3′. The PCR settings were 94°C for
3 min; 35 cycles at 94°C for 30 sec, 55°C for 30 sec and 72°C for
30 sec; terminated at 72°C for 5 min. SUP-T1 cells in the
logarithmic growth phase were seeded into 96-well plates at
1.5×104 cells/well, and co-transfected with the 100 ng
WT or 100 ng MT vector and one of the 50 nM miRNA or 150 nM
inhibitor (miR-338-3p mimics, NC, miR-338-3p inhibitor or inhibitor
NC) using Attractene Transfection Reagent (Qiagen, Inc.). Following
incubation for 48 h, the ratio of firefly to Renilla
luciferase activity was determined using a dual-luciferase reporter
system, according to the manufacturer's protocols (Promega
Corporation).
Western blotting
Cells were collected and lysed in ice-cold RIPA
buffer (Beyotime Institute of Biotechnology) with 10 nM PMSF.
Protein concentration was determined using the bicinchoninic acid
(BCA) protein assay kit (Beyotime Institute of Biotechnology), and
equal amounts of the samples (50 µg per lane) were separated via
10% SDS-PAGE. Proteins were then transferred to polyvinylidene
fluoride membranes at 100 V for 2.5 h. The membranes were blocked
for 2 h at room temperature with 5% fat-free milk in TBS with
Tween-20 and incubated with primary antibodies specific for HOXA3
(Abcam, ab28771) and GAPDH (Abcam, ab9485) at 4°C overnight. Then,
HRP Goat Anti-Rabbit (IgG) secondary antibody (Abcam, ab6721;
1:5,000) were added for incubation at room temperature for 2 h.
Chemiluminescence reagents were used to visualize the signals (EMD
Millipore). ImageJ v1.38 software (National Institutes of Health)
was used to quantify protein expression levels. GAPDH was used as
the loading control.
Statistical analysis
Statistical analyses were performed using SPSS 17.0
software (SPSS, Inc.). All the experiments were performed at least
three independent times. Data are presented as the mean ± standard
deviation. According to the miR-338-3p mean expression level, 38
cases of T-LBL tissues samples were divided into two groups,
including a higher miR-338-3p expression group (n=20) and a lower
miR-338-3p expression group (n=18). Correlations between
clinicopathological characteristics and miR-338-3p expression were
analyzed by χ2 test Independent-samples t-tests were
performed to compare two independent groups. One-way ANOVA followed
by Bonferroni's post-hoc test was performed to analyze differences
between three or more independent groups. P<0.05 was considered
to indicate a statistically significant difference.
Results
Reduced expression of miR-338-3p in
T-LBL
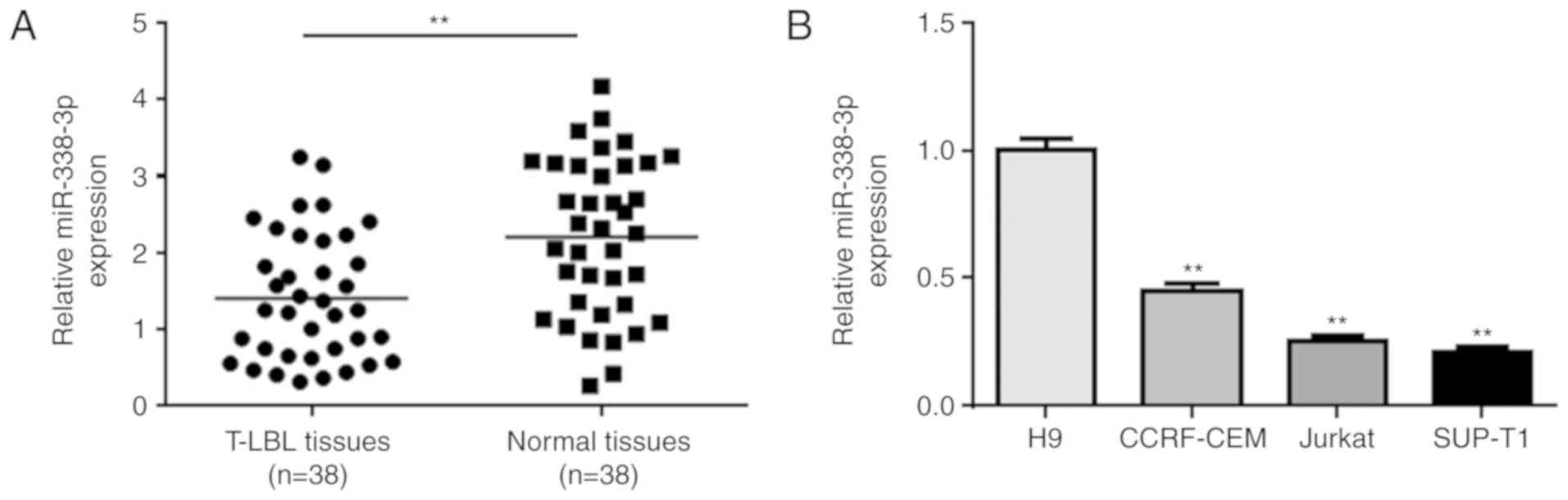
To investigate the role of miR-338-3p in T-LBL, the
expression levels of miR-338-3p were measured in T-LBL and adjacent
normal tissues collected from 38 patients with T-LBL. The mean
expression levels of miR-338-3p were significantly decreased in
T-LBL tumor tissues compared with those in the normal tissues
(Fig. 1A). The expression levels
were associated with certain clinicopathological characteristics
(Table I). The levels of
miR-338-3p were also determined in a number of cell lines, which
were predicted to be more homogeneous than those in patient
samples. miR-338-3p was significantly downregulated in the T-LBL
cell line SUP-T1, and the lymphoblastic leukemia cell lines
CCRF-CEM and Jurkat, compared with in the human H9 T-cell line
(Fig. 1B).
 | Table I.Clinicopathological characteristics of
patients with T-cell lymphoblastic lymphoma with high or low
miR-338-3p expression. |
Table I.
Clinicopathological characteristics of
patients with T-cell lymphoblastic lymphoma with high or low
miR-338-3p expression.
|
|
| miR-338-3p
expression |
|
|---|
|
|
|
|
|
|---|
| Characteristic | No. of patients
(n=38) | Low (n=18) | High (n=20) | P-value |
|---|
| Sex |
|
|
| 0.351 |
| Male | 28 | 12 | 16 |
|
|
Female | 10 | 6 | 4 |
|
| Age, years |
|
|
| 0.573 |
|
<40 | 29 | 13 | 16 |
|
|
>40 | 9 | 5 | 4 |
|
| B-symptoms |
|
|
| 0.564 |
| Yes | 13 | 7 | 6 |
|
| No | 25 | 11 | 14 |
|
| BM involvement |
|
|
| 0.184 |
|
Yes | 9 | 6 | 3 |
|
| No | 29 | 12 | 17 |
|
| Bulky disease
(>10 cm) |
|
|
| 0.018 |
|
Yes | 22 | 14 | 8 |
|
| No | 16 | 4 | 12 |
|
| Ann Arbor
stage |
|
|
| 0.021 |
| I/IE or
II/IIE | 11 | 2 | 9 |
|
|
III/IV | 27 | 16 | 11 |
|
| Albumin |
|
|
| 0.351 |
| ≤35
g/dl | 10 | 6 | 4 |
|
| >35
g/dl | 28 | 12 | 16 |
|
| Therapy
response |
|
|
| 0.386 |
| CR | 27 | 14 | 13 |
|
| Not
CR | 11 | 4 | 7 |
|
| Relapse in 2
years |
|
|
| 0.010 |
|
Yes | 23 | 7 | 16 |
|
| No | 15 | 11 | 4 |
|
Overexpression of miR-338-3p
suppresses cell proliferation and migration
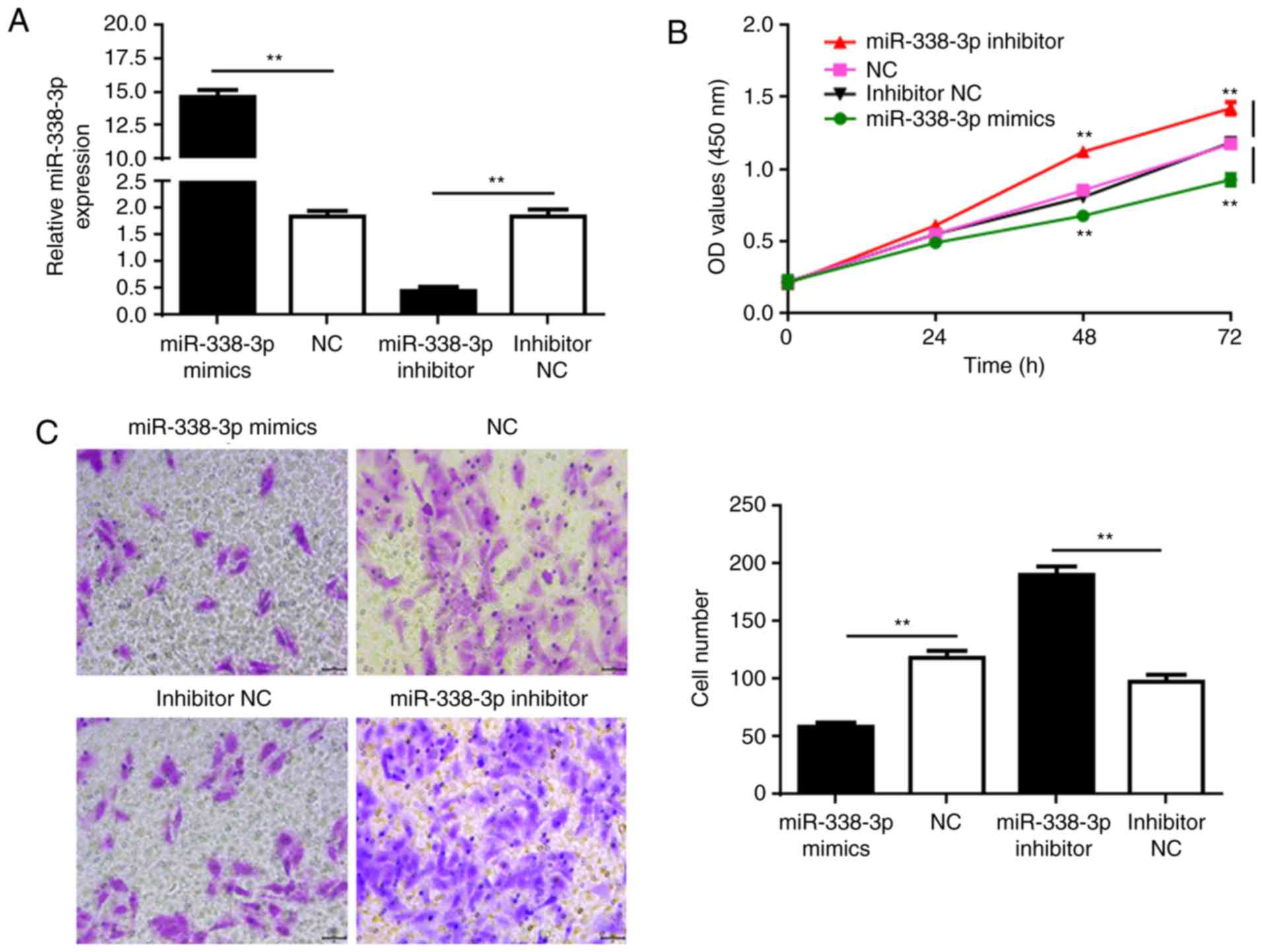
SUP-T1 cells were used for further functional
analyses. Synthetic miR-338-3p and miR-338-3p inhibitor molecules
were transfected into the SUP-T1 cells to rescue and further reduce
miR-336-3p expression, respectively (Fig. 2A). CCK-8 assays were performed to
determine the proliferation of cells at 24, 48, and 72 h
post-transfection. Transfection with miR-338-3p mimics
significantly decreased the proliferation of the cancer cells
compared with the NC, whereas further reducing miR-338-3p
expression with the inhibitor significantly promoted SUP-T1 cell
proliferation (Fig. 2B).
Additionally, Transwell assays were performed to investigate cell
migration. Similarly, upregulated miR-338-3p expression
significantly reduced cell migration, whereas transfection with
inhibitor enhanced migration compared with the respective controls
(Fig. 2C). The results suggested
that rescuing the expression of miR-338-3p may suppress the
development of T-LBL.
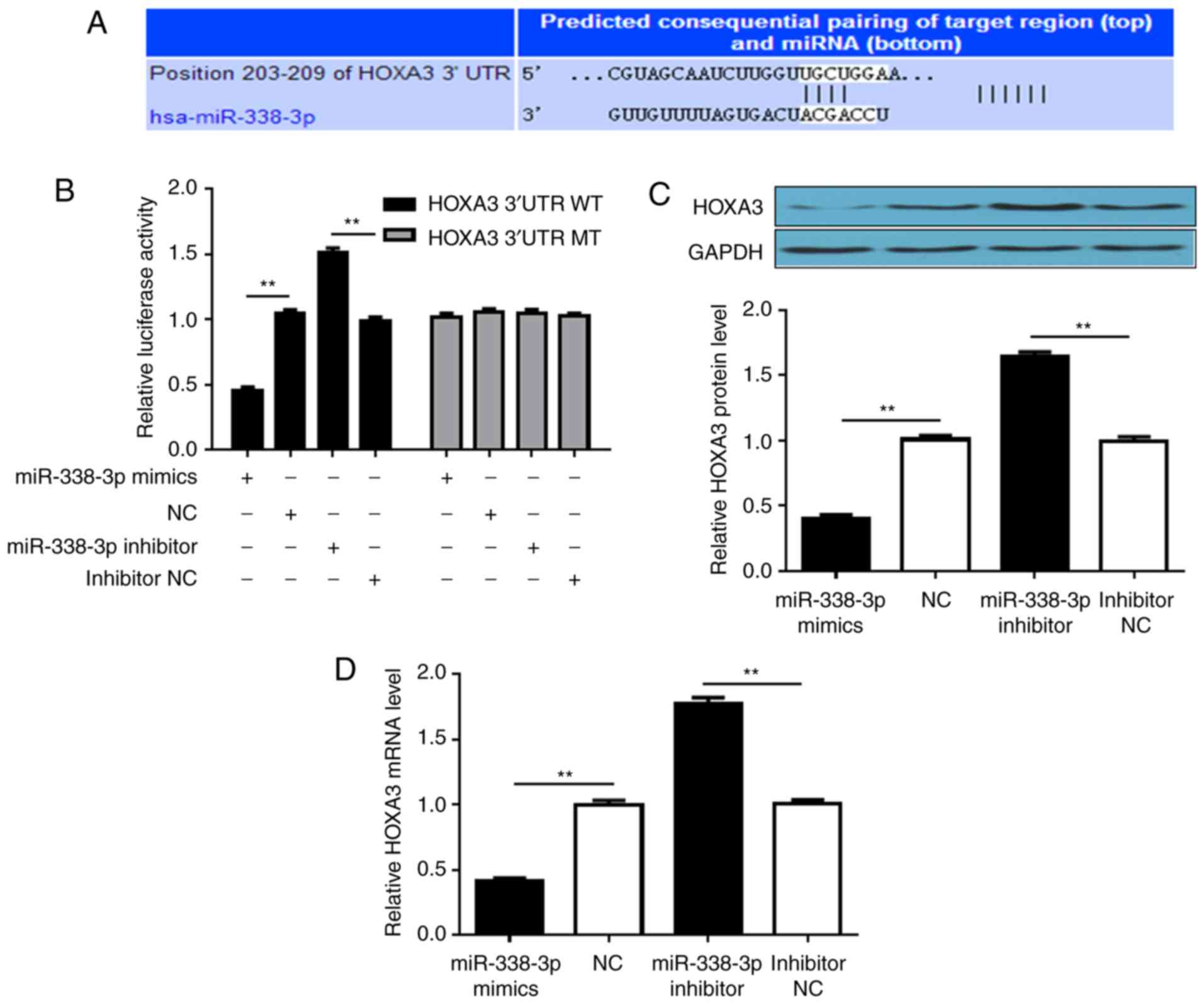
miR-338-3p directly regulates HOXA3
expression by targeting its 3′-UTR
As miRNAs mainly associate with the 3′-UTR of mRNAs
to regulate gene expression, searches were performed to identify
putative targets of miR-338-3p to investigate the molecular
mechanisms underlying its activity. TargetScan was employed to
search for miR-338-3p target genes, particularly those with
potential roles in promoting tumor cell proliferation and
migration. HOXA3 was considered a good candidate for further
analysis (Fig. 3A). The 3′-UTR of
HOXA3 was introduced into a dual-luciferase reporter system, and
the effects of altering miR-338-3p expression on luciferase
production were investigated. Introducing miR-338-3p mimics into
cells significantly suppressed luciferase signals, whereas
transfection with miR-338-3p inhibitor further increased luciferase
activity, compared with the respective controls. Conversely, cells
transfected with a dual-luciferase vector containing a mutant
3′-UTR that did not form a duplex with the seed-pairing region of
miR-338-3p did not exhibit alterations in luciferase activity
following transfection with mimics or inhibitor (Fig. 3B). The levels of HOXA3 protein and
mRNA in SUP-T1 cells were then further assessed. Transfection with
miR-338-3p mimics significantly downregulated HOXA3 at the protein
and mRNA levels compared with the NC, whereas decreasing the levels
of miR-338-3p upregulated HOX3A (Fig.
3C and D). The results indicated that miR-338-3p directly
targeted the 3′-UTR of HOXA3.
miR-338-3p suppresses SUP-T1 cell
proliferation and migration via the inhibition of HOXA3 protein
expression
To determine whether the regulation of HOXA3
expression was involved in the tumor-suppressive role of
miR-338-3p, the effects of miR-338-3p upregulation in the presence
or absence of HOXA3 overexpression were determined. Transfection of
SUP-T1 cells with HOXA3 vector significantly upregulated the
expression of HOXA3 mRNA and protein compared with HOXA3 NC,
whereas transfection with miR-338-3p mimics significantly reduced
the expression of HOXA3 protein compared with the NC (Fig. 4A and B). Co-transfection of HOXA3
vector and miR-338-3p mimics did not significantly alter the
protein level compared with co-transfection with the respective
controls (Fig. 4B). Upregulation
of HOXA3 significantly increased the proliferation and migration of
the SUP-T1 cells compared with HOXA3 NC, and rescued proliferation
and migration in miR-338-3p mimics-transfected cells (Fig. 4C and D). The results suggested that
miR-338-3p suppressed T-LBL by inhibiting HOXA3 protein
expression.
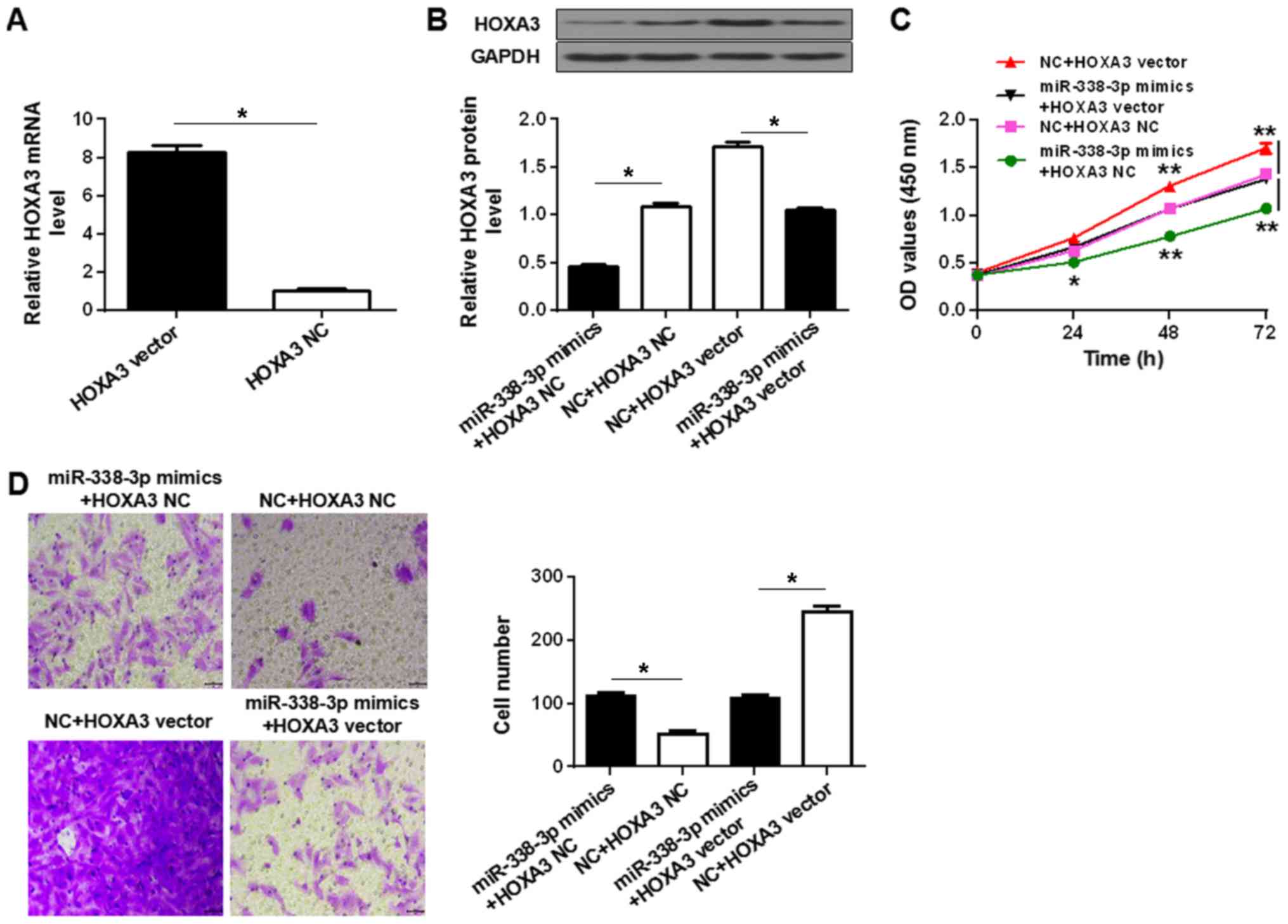 | Figure 4.miR-338-3p-dependent downregulation of
HOXA3 is required for the suppression of T-LBL malignancy. (A)
Reverse transcription-quantitative polymerase chain reaction
analysis of HOXA3 expression in SUP-T1 cells transfected with HOXA3
vector or NC. (B) Western blot analysis of HOXA3 protein levels in
SUP-T1 cells co-transfected with HOXA3 vector or HOXA3 NC, and
miR-338-3p mimics or NC. (C) Proliferation of SUP-T1 cells
co-transfected with the aforementioned vectors and RNAs. (D)
Migration of SUP-T1 cells transfected with the aforementioned
vectors and miRNAs. Left, crystal violet-stained cells that
migrated during the Transwell assay. Right, quantification of cell
migration. Scale bar, 40 µm. *P<0.05, **P<0.01. miR-338-3p,
microRNA-338-3p; HOXA3, homeobox A3; NC, negative control; HOXA3
NC, empty vector; OD, optical density. |
Discussion
T-LBL is an aggressive disease caused by the
malignant transformation of immature progenitors primed towards
T-cell development. At present, chemotherapy remains the main
treatment option (1). This often
includes intensive remission induction chemotherapy, CNS
prophylaxis, consolidation chemotherapy and subsequent maintenance
therapy for 12–18 months (19);
however, relapse is common and overall survival is poor (1). The present study observed that the
expression profile of miR-338-3p may be important for the
suppression of T-LBL. This miRNA was shown to directly target the
3′-UTR of HOXA3 mRNA to inhibit T-LBL cell proliferation and
migration. The results provide insight for further investigation
into signaling networks associated with miR-338-3p, and may aid the
development of novel therapeutic options to reduce the incidence of
relapse.
Studies on a variety of cancer types have reported
the tumor-suppressive effects of miR3383p; therefore, there has
been increasing interest in utilizing miR-338-3p to treat different
solid tumors, including gastric cancer, hepatocellular carcinoma
and malignant melanoma (20–22).
The present study has associated miR-338-3p expression with the
suppression of T-LBL for the first time, to the best of our
knowledge. Notably, its target mRNA is encoded by the homeobox gene
HOXA3. Numerous members of this gene family encode transcriptional
factors that are mainly involved in embryonic development and
hematopoietic stem cell function (23,24).
Normal expression of HOXA3 in the developing embryo is required for
the proper formation of the thymus, thyroid and parathyroid organs
(23,24). HOXA3 overexpression, which can
activate oncogenic signaling pathways, has been reported to be
involved in cancer biology (25).
Further investigation into the mechanism of action of HOXA3 in
T-LBL may aid the identification of molecular mechanisms involved
in the development of the disease.
The molecular mechanisms of action of miRNAs on
mRNAs have been extensively studied; however, the functional
consequences remain unclear. It was initially hypothesized that
miRNAs may inhibit the translation of target mRNAs without
significantly affecting their levels in animal cells (26,27);
however, it was subsequently proposed that the immediate outcome of
miRNA-target mRNA interactions is translation inhibition, but that
mRNA degradation can follow (28,29).
The present study observed that HOXA3 protein and mRNA were
downregulated by miR-338-3p in SUP-T1 cells, potentially supporting
the idea that miRNAs induce target mRNA degradation; however, the
possibility that miR-338-3p regulates other cellular factors that
ultimately affect the transcription of the HOXA3 mRNA cannot be
ruled out. A time-course analysis of the effects of miR-338-3p may
help to further demonstrate the mechanisms underlying the actions
of miR-338-3p.
Small animal models of human diseases, particularly
rare diseases such as T-cell lymphoma/leukemia, are useful tools
for investigating the molecular mechanisms underlying pathogenesis
and evaluating novel therapeutic approaches. T-cell lymphoma has
been modeled in various types of genetically modified mice, and
although important insight has been obtained from these models, the
limitations cannot be ignored (30), as no single animal model can
capture all aspects of a human disease. Studies revealing novel
molecular details regarding the development of T-LBL may improve
rodent models, and thus aid research and development for the next
generation of therapies.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XW designed the experiments. LW and MS performed the
experiments and collected data. XW analyzed the data and drafted
this manuscript. All authors approved this manuscript.
Ethics approval and consent to
participate
Informed consent was obtained from all participants.
The use of human samples was approved by the Human Ethics
Committee/Institutional Review Board of the Affiliated Yantai
Yuhuangding Hospital of Qingdao University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Uckun FM, Gaynon PS, Sensel MG, Nachman J,
Trigg ME, Steinherz PG, Hutchinson R, Bostrom BC, Sather HN and
Reaman GH: Clinical features and treatment outcome of childhood
T-lineage acute lymphoblastic leukemia according to the apparent
maturational stage of T-lineage leukemic blasts: A children's
cancer group study. J Clin Oncol. 15:2214–2221. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lepretre S, Graux C, Touzart A, Macintyre
E and Boissel N: Adult T-type lymphoblastic lymphoma: Treatment
advances and prognostic indicators. Exp Hematol. 51:7–16. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Portell CA and Sweetenham JW: Adult
lymphoblastic lymphoma. Cancer J. 18:432–438. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee WJ, Moon HR, Won C, Chang SE, Choi JH,
Moon KC and Lee MW: Precursor B- or T-lymphoblastic lymphoma
presenting with cutaneous involvement: A series of 13 cases
including 7 cases of cutaneous T-lymphoblastic lymphoma. J Am Acad
Dermatol. 70:318–325. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Aifantis I, Raetz E and Buonamici S:
Molecular pathogenesis of T-cell leukaemia and lymphoma. Nat Rev
Immunol. 8:380–390. 2008. View
Article : Google Scholar : PubMed/NCBI
|
|
6
|
Graux C, Cools J, Michaux L, Vandenberghe
P and Hagemeijer A: Cytogenetics and molecular genetics of T-cell
acute lymphoblastic leukemia: From thymocyte to lymphoblast.
Leukemia. 20:1496–1510. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Palomero T, Lim WK, Odom DT, Sulis ML,
Real PJ, Margolin A, Barnes KC, O'Neil J, Neuberg D, Weng AP, et
al: NOTCH1 directly regulates c-MYC and activates a
feed-forward-loop transcriptional network promoting leukemic cell
growth. Proc Natl Acad Sci USA. 103:18261–18266. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Weng AP, Millholland JM, Yashiro-Ohtani Y,
Arcangeli ML, Lau A, Wai C, Del Bianco C, Rodriguez CG, Sai H,
Tobias J, et al: c-Myc is an important direct target of Notch1 in
T-cell acute lymphoblastic leukemia/lymphoma. Genes Dev.
20:2096–2109. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vilimas T, Mascarenhas J, Palomero T,
Mandal M, Buonamici S, Meng F, Thompson B, Spaulding C, Macaroun S,
Alegre ML, et al: Targeting the NF-kappaB signaling pathway in
Notch1-induced T-cell leukemia. Nat Med. 13:70–77. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Palomero T, Sulis ML, Cortina M, Real PJ,
Barnes K, Ciofani M, Caparros E, Buteau J, Brown K, Perkins SL, et
al: Mutational loss of PTEN induces resistance to NOTCH1 inhibition
in T-cell leukemia. Nat Med. 13:1203–1210. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
O'Neil J, Grim J, Strack P, Rao S,
Tibbitts D, Winter C, Hardwick J, Welcker M, Meijerink JP, Pieters
R, et al: FBW7 mutations in leukemic cells mediate NOTCH pathway
activation and resistance to gamma-secretase inhibitors. J Exp Med.
204:1813–1824. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Weng AP, Ferrando AA, Lee W, Morris JP IV,
Silverman LB, Sanchez-Irizarry C, Blacklow SC, Look AT and Aster
JC: Activating mutations of NOTCH1 in human T cell acute
lymphoblastic leukemia. Science. 306:269–271. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ye F: MicroRNA expression and activity in
T-cell acute lymphoblastic leukemia. Oncotarget. 9:5445–5458. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wallaert A, Van Loocke W, Hernandez L,
Taghon T, Speleman F and Van Vlierberghe P: Comprehensive miRNA
expression profiling in human T-cell acute lymphoblastic leukemia
by small RNA-sequencing. Sci Rep. 7:79012017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Saba R, Goodman CD, Huzarewich RL,
Robertson C and Booth SA: A miRNA signature of prion induced
neurodegeneration. PLoS One. 3:e36522008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li Y, Chen P, Zu L, Liu B, Wang M and Zhou
Q: MicroRNA-338-3p suppresses metastasis of lung cancer cells by
targeting the EMT regulator Sox4. Am J Cancer Res. 6:127–140.
2016.PubMed/NCBI
|
|
17
|
Lister TA, Crowther D, Sutcliffe SB,
Glatstein E, Canellos GP, Young RC, Rosenberg SA, Coltman CA and
Tubiana M: Report of a committee convened to discuss the evaluation
and staging of patients with Hodgkin's disease: Cotswolds meeting.
J Clin Oncol. 7:1630–1636. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2-ΔΔCT method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Sweetenham JW: Treatment of lymphoblastic
lymphoma in adults. Oncology (Williston Park). 23:1015–1020.
2009.PubMed/NCBI
|
|
20
|
Guo B, Liu L, Yao J, Ma R, Chang D, Li Z,
Song T and Huang C: miR-338-3p suppresses gastric cancer
progression through a PTEN-AKT axis by targeting P-REX2a. Mol
Cancer Res. 12:313–321. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Huang XH, Wang Q, Chen JS, Fu XH, Chen XL,
Chen LZ, Li W, Bi J, Zhang LJ, Fu Q, et al: Bead-based microarray
analysis of microRNA expression in hepatocellular carcinoma:
miR-338 is downregulated. Hepatol Res. 39:786–794. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Caramuta S, Egyhazi S, Rodolfo M, Witten
D, Hansson J, Larsson C and Lui WO: MicroRNA expression profiles
associated with mutational status and survival in malignant
melanoma. J Invest Dermatol. 130:2062–2070. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chojnowski JL, Masuda K, Trau HA, Thomas
K, Capecchi M and Manley NR: Multiple roles for HOXA3 in regulating
thymus and parathyroid differentiation and morphogenesis in mouse.
Development. 141:3697–3708. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Manley NR and Capecchi MR: The role of
Hoxa-3 in mouse thymus and thyroid development. Development.
121:1989–2003. 1995.PubMed/NCBI
|
|
25
|
Zhang X, Liu G, Ding L, Jiang T, Shao S,
Gao Y and Lu Y: HOXA3 promotes tumor growth of human colon cancer
through activating EGFR/Ras/Raf/MEK/ERK signaling pathway. J Cell
Biochem. 119:2864–2874. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lee RC, Feinbaum RL and Ambros V: The
C. elegans heterochronic gene lin-4 encodes small RNAs with
antisense complementarity to lin-14. Cell. 75:843–854. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wightman B, Ha I and Ruvkun G:
Posttranscriptional regulation of the heterochronic gene lin-14 by
lin-4 mediates temporal pattern formation in C. elegans.
Cell. 75:855–862. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guo H, Ingolia NT, Weissman JS and Bartel
DP: Mammalian microRNAs predominantly act to decrease target mRNA
levels. Nature. 466:835–840. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Bazzini AA, Lee MT and Giraldez AJ:
Ribosome profiling shows that miR-430 reduces translation before
causing mRNA decay in zebrafish. Science. 336:233–237. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kohnken R, Porcu P and Mishra A: Overview
of the use of murine models in leukemia and lymphoma research.
Front Oncol. 7:222017. View Article : Google Scholar : PubMed/NCBI
|