Introduction
Depression is speculated to be a stress-related
mental disorder caused by mental and social burdens (1). While the precise mechanisms
underlying depression remain unknown, these will most likely
involve molecular and cellular abnormalities that interact with
genetic and environmental factors, such as abnormal metabolism of
monoamine neurotransmitters, neurogenesis and
hypothalamic-pituitary-adrenal axis dysfunction (2,3).
Although depression is characterized by multiple systemic
manifestations, inflammation is the key event associated with these
disturbances in the brain (4,5). It
has been shown that sustained stress leads to the activation of
inflammatory signaling and increases neuronal susceptibility to
damage (6,7). Moreover, inflammatory cytokines
impair neurogenesis, activity of neurotrophic factors and neuronal
plasticity (8,9). Inflammatory cytokines have been
proposed to regulate monoamine neurotransmitter metabolism by
inhibiting neurotransmitter synthesis and promoting monoamine
neurotransmitter hydrolysis (10,11).
Therefore, inhibiting inflammation may be a potential therapeutic
strategy for depression (12,13).
Nuclear factor (NF)-κB and nod-like receptor protein
3 (NLRP3) inflammasome are two archetypal molecular drivers of the
inflammatory response (14). NF-κB
is a critical transcription factor that induces the expression of
inflammatory mediators, such as interleukin (IL)-1β, IL-6 and tumor
necrosis factor α (TNF-α), and upregulates NLRP3 synthesis
(14). Dysregulated NF-κB
signaling has been reported to be associated with numerous
inflammatory, autoimmune and nervous system diseases (14). The NLRP3 inflammasome consists of a
NLRP3 pattern recognition receptor, an apoptosis-associated
speck-like protein (ASC) adaptor and the effector protein
caspase-1. Once activated, NLRP3 interacts with the ASC adaptor
protein and cleaves pro-caspase-1 to produce mature caspase-1,
which activates the processing of IL-1β to engage the immune
defense mechanism (15).
Furthermore, NF-κB- and NLRP3-induced inflammation may play an
important role in depression, Alzheimers disease, Parkinsons
disease and diabetes (16,17).
Although NF-κB and NLRP3 signaling have been
reported to contribute to inflammation, pathways such as the
cAMP/protein kinase A (PKA) signaling pathway may also play
important roles in regulating NF-κB and NLRP3-related inflammatory
responses (18–22). Previous studies have revealed the
anti-inflammatory role of cAMP, a prototypical second messenger,
which interferes with the immune response at multiple levels
(18–22). Furthermore, binding of cAMP to the
NLRP3 NOD-binding domain has been previously identified (22). In addition, the prototypical cAMP
effector kinase PKA, a serine/threonine kinase, regulates several
pathways involved in controlling inflammation by modulating gene
transcription and post-translational modification of effector
proteins, including NF-κB and NLRP3 (22). Moreover, PKA upregulation may also
increase the phosphorylation of cAMP-responsive element-binding
protein (CREB), which is an important transcription factor involved
in several critical functions in depression (23). Suppressing CREB signaling in the
hippocampus has been reported to induce depressive-like behavior by
interfering with the expression of CRE-regulated genes, such as
brain-derived neurotrophic factor (BDNF), which plays an important
role in depression (24).
Asiaticoside is one of the triterpenoid components
found in Centella asiatica (L.) (25–28).
It has been reported that asiaticoside exhibits anti-inflammatory,
antioxidative, wound-healing, hepatoprotective and antitumor
properties (25). Asiaticoside has
also been shown to exert a strong anti-inflammatory effect in
animal models of osteoarthritis, spinal cord injury and peritonitis
(26). Moreover, asiaticoside may
exert beneficial effects in central nervous system (CNS) disorders,
including Parkinsons disease and dementia (27). It has been reported that the
antidepressant properties of asiaticoside are mediated via
activation of the BDNF/tropomyosin receptor kinase B (TrkB)
signaling pathway in the chronic unpredictable mild stress (CMS)
mouse model (28). However, the
mechanism underlying asiaticoside-mediated alterations in BDNF
signaling is yet to be elucidated. A previous study showed the
neuroprotective effect of asiaticoside on Neuro-2a cells, which was
related to elevation of the cAMP/CREB signal (29). In line with these previous results,
it was hypothesized that the antidepressant-like properties and
anti-inflammatory effects of asiaticoside may be, at least partly,
via activation of the cAMP/PKA signaling pathway. The present study
aimed to investigate whether asiaticoside inhibits inflammation in
a mouse model of depression, and to identify the underlying
mechanisms regulating the cAMP/PKA pathway.
Materials and methods
Drugs and reagents
Asiaticoside (purity >98%) was purchased from
Nanjing Zelang Medical Technology Co., Ltd. Fluoxetine (FLX)
hydrochloride was purchased from Changzhou Siyao Pharmaceuticals
Co., Ltd. All other chemicals and reagents used were of analytical
grade.
Animals
In total, 60 male ICR mice (age, 2 months old;
weight, 18–22 g) were purchased from The Experimental Animal Center
in Jiangsu Province. Animals were randomly divided into five
treatment groups (n=12 mice/group) and housed in cages at room
temperature (22±2°C) under a 12-h light/dark cycle (lights on at
8:00 a.m.), with ad libitum access to food and water. The
mice were allowed to acclimate for 1 week before the experiment
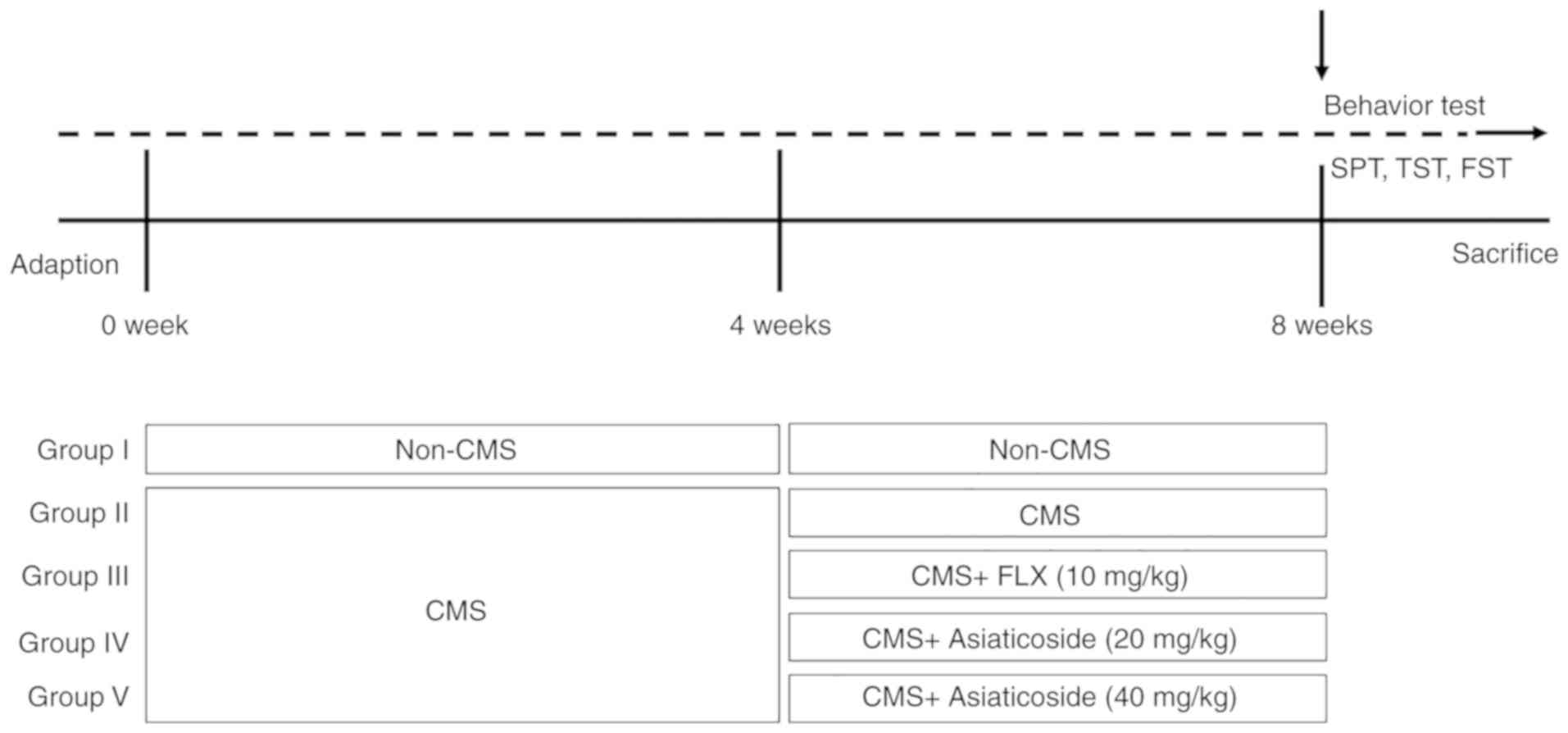
commenced. The groups were as follows: i) Normal control group; ii)
CMS model group; iii) CMS + FLX (20 mg/kg, i.g) group; iv) CMS +
asiaticoside (20 mg/kg, i.g) group; and v) CMS + asiaticoside (40
mg/kg, i.g) group. Asiaticoside and FLX were prepared in
double-distilled water. Mice were exposed to the CMS stimuli for 4
weeks (weeks 1–4), followed by 4 weeks (weeks 5–8) of asiaticoside
or FLX treatment during which CMS stimulation continued. For the
control and CMS model groups, mice were given an equal volume of
double-distilled water. Asiaticoside doses were selected based on a
previous study (28). All
experiments were conducted as per the Guidelines of the
Institutional Animal Care and Use Committee of China and the
present study was approved by Experimental Animal Ethics Committee
of Xuzhou Medical University (Lianyungang, China).
Behavioral tests were performed after the last
administration of asiaticoside or FLX (Fig. 1). CMS was introduced as previously
described (17,30) with minor modifications (the order
of stressors was random). The normal control group animals were
left undisturbed in their cages in a separate room throughout the 8
weeks of treatment. The other four groups were individually housed
and subjected to a variety of stresses for 8 weeks: i) Soiled cage
(200 ml water in 100 g sawdust bedding; ii) foreign object
exposure; iii) light/dark perversion; iv) physical restraint for 2
h; v) food deprivation for 24 h; vi) water deprivation for 24 h;
vii) overnight illumination; viii) cage tilt (45°) for 10 h; ix)
white noise; x) 3-min oscillation; and xi) 1-min tail pinch (1 cm
from the root of the tail). All the stressors were applied randomly
to ensure the unpredictability of the experiment.
Sucrose preference test
Sucrose preference test was conducted every week
during the experimental period (17). Mice were first deprived of water
and food for 10 h before the test, and were then allowed to freely
choose between two bottles (one with tap water, and another with 1%
sucrose solution) for 10 h. To prevent possible effects of side
preference on drinking behavior, the positions of the bottles were
switched after 5 h. The consumption of the sucrose solution and tap
water was estimated by weighing the bottles. Sucrose preference was
calculated as sucrose preference (%) = sucrose intake (g)/[sucrose
intake (g) + water intake (g)] ×100.
Tail suspension test (TST)
TST was performed as described previously (17). Individual mice were acoustically
and visually isolated, and suspended ~50 cm above the floor by
placing adhesive tape ~1 cm from the tail tip. Mice were considered
immobile when they were passively suspended and remained completely
motionless. Each animal was suspended for 6 min, and total
immobility was recorded during the last 4 min of the test.
Forced swimming test (FST)
FST was carried out as described previously on the
day after TST (17). Mice were
forced to swim in a cylinder (20 cm height × 14 cm diameter)
containing fresh water (25±1°C) to a height of 10 cm. The
immobility time was recorded as the time the mice spent floating in
the water without struggling or only making movements necessary to
keep their heads above the water. Each animal was forced to swim
for 6 min, and total immobility time was recorded during the last 4
min of the test.
Collection of tissue samples
Mice were sacrificed after the final behavioral
tests were concluded. Ketamine and xylazine (100 and 10 mg/kg,
respectively) were intraperitoneally injected to anesthetize the
mice and the mice were then sacrificed by decapitation. The whole
hippocampus was rapidly dissected on an iced-plate and weighed. The
hippocampus was identified as described in Paxinos and Watsons
Atlas (30).
Determination of 5-hydroxytryptamine
(5-HT) and norepinephrine (NE) levels
The levels of 5-HT and NE in the mouse hippocampus
were determined by high-performance liquid chromatography (HPLC).
The hippocampus was homogenized in extract solution, which
consisted of 0.1 mM EDTA and 0.1 M HClO4 buffer, and the
mixture was centrifuged at a speed of 20,000 × g for 30 min at 4°C.
Then, 50 µl of the resulting supernatant was injected into the
liquid chromatography system equipped with a reversed phase C18
column (2.2 µm, 120 Å, 2.1×100 mm; Dionex; Thermo Fisher
Scientific, Inc.). The mobile phase was composed of solutions A
(0.1% formic acid, v/v) and B (acetonitrile), with a gradient
elution as follows: 0–6 min, 90–65% A; 6–8 min, 65–0% A; 8–10 min,
0% A. The flow rate was maintained at 0.4 ml/min, and the column
temperatures were maintained at 30°C, and was detected by ESA
Coulochem® III Electrochemical Detector (Dionex; Thermo
Fisher Scientific, Inc.). The detector was set at 350 mV. The
identification and purity were evaluated by the chromatographic
peaks as well as their quantitative evaluation, which was measured
by comparing their retention times and peak areas with those of
standard solutions. 5-HT (cat. no. H9523) and NE (cat. no.
N-069-1ML) standards were purchased from Sigma-Aldrich (Merck
KGaA).
Determination of IL-1β, IL-6, TNF-α
and cAMP levels in the hippocampus
Cytokine and cAMP concentrations in the mouse
hippocampus were measured by ELISA, according to the manufacturers
instructions. The following kits were used: Mouse IL-1β ELISA kit
(cat. no. 1210122); mouse IL-6 ELISA kit (cat. no. 1210602); mouse
TNF-α ELISA kit (cat. no. 1217202; all purchased from Dakewei
Biotechnology Co., Ltd.); and mouse cyclic adenosine monophosphate,
cAMP GENLISA™ ELISA kit (cat. no. KLM0071; Krishgen BioSystems).
The concentrations were measured using a microplate reader (450 nm
absorbance). The results were reported in picograms per milliliter
(pg/ml).
Protein extraction and western
blotting
Protein extracts were obtained by homogenizing
hippocampal tissue in lysis buffer [50 mM Tris HCl (pH 7.2)
containing 1% sodium deoxycholate, 1% NP-40, 0.15 mM NaCl and 0.1%
SDS; Roche Applied Science). A bicinchoninic acid assay was used to
determine protein concentrations (Sigma-Aldrich; Merck KGaA). Equal
amounts of protein (30 µg/µl) were separated by SDS-PAGE on 10%
gels and were then transferred to PVDF membranes (0.2 µm; EMD
Millipore). The membranes were blocked for 2 h with 5% non-fat dry
milk at room temperature, then incubated at 4°C overnight with the
following antibodies: Rabbit anti-phosphorylated
(p)NF-κBp65Ser536 (1:1,000; cat. no. orb501609; Biorbyt
Ltd.), rabbit anti-NF-κBp65 (1:1,000; cat. no. orb453023; Biorbyt
Ltd.), rabbit anti-NLRP3 (1:1,000; cat. no. orb101128; Biorbyt
Ltd.), rabbit anti-caspase-1 (1:1,000; cat. no. ab179515; Abcam),
rabbit anti-PKA (1:1,000; cat. no. ab75991; Abcam), rabbit
anti-p-VASPser157 (1:1,000; cat. no. ab47268; Abcam) and
rabbit anti-VASP (1:1,000; cat. no. ab205952; Abcam), rabbit
anti-pCREBSer133 (1:1,000; cat. no. ab32096; Abcam),
rabbit anti-CREB (1:1,000; cat. no. ab32515; Abcam), rabbit
anti-BDNF (1:1,000; cat. no. ab108319; Abcam) and rabbit
anti-β-actin (1:1,000; cat. no. ab8227; Abcam). After three washes
in TBS-Triton X-100 (1%) buffer, the membranes were incubated for 1
h at 24°C, with horseradish peroxidase-labeled anti-rabbit IgG
(1:5,000; cat. no. BS13271; Bioworld Technology, Inc.).
Densitometric measurements were performed using the ECL Western
detection system (EMD Millipore) and the Quantity One imaging
program (v7.1; Bio-Rad Laboratories, Inc.).
Statistical analyses
Experiments were repeated three times. Data are
presented as the mean ± SEM. Statistical significance was assessed
by one-way ANOVA followed by Tukeys test. All statistical analyses
were performed using GraphPad Prism 8 software (GraphPad Software,
Inc.). P<0.05 was considered to indicate a statistically
significant difference.
Results
Effects of asiaticoside on sucrose
preference, TST and FST
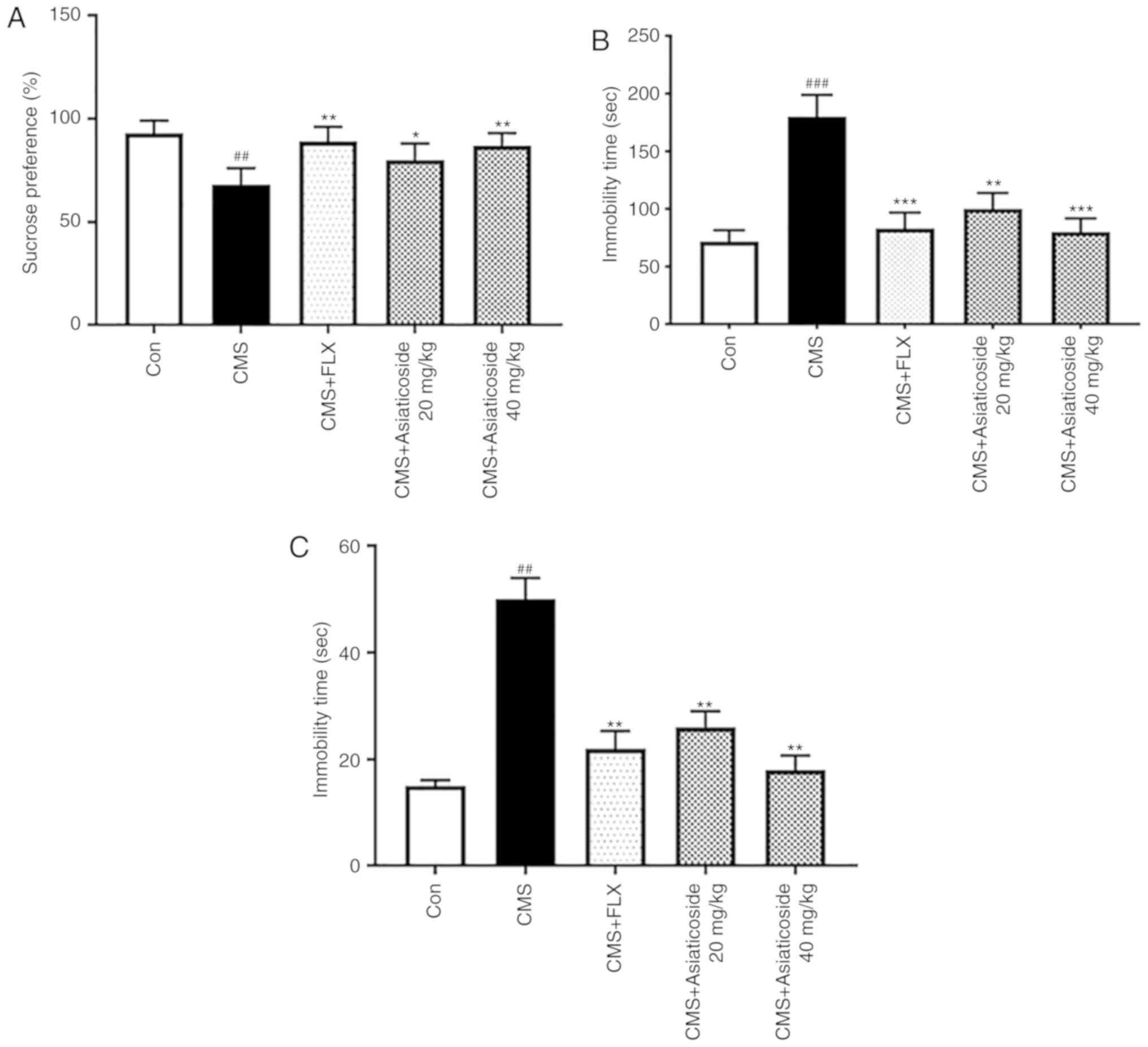
Sucrose preference in mice is considered a measure
of anhedonia (17). It was
revealed that the CMS model mice exhibited significantly reduced
sucrose consumption compared with the control group (P<0.01;
Fig. 2A). Moreover, asiaticoside
(20 and 40 mg/kg) or FLX (10 mg/kg) administration significantly
reversed the decreased sucrose consumption in the CMS model group
(P<0.05, P<0.01, respectively). It was also demonstrated that
CMS significantly increased the immobility time during TST
(P<0.001; Fig. 2B) and FST
(P<0.01; Fig. 2C). Asiaticoside
(20 and 40 mg/kg) or FLX (10 mg/kg) treatment significantly
reversed the increase in the immobility time in both TST
(P<0.01, P<0.001, respectively; Fig. 2B) and FST (P<0.01; Fig. 2C).
Effects of asiaticoside on
neurotransmitters in the hippocampus
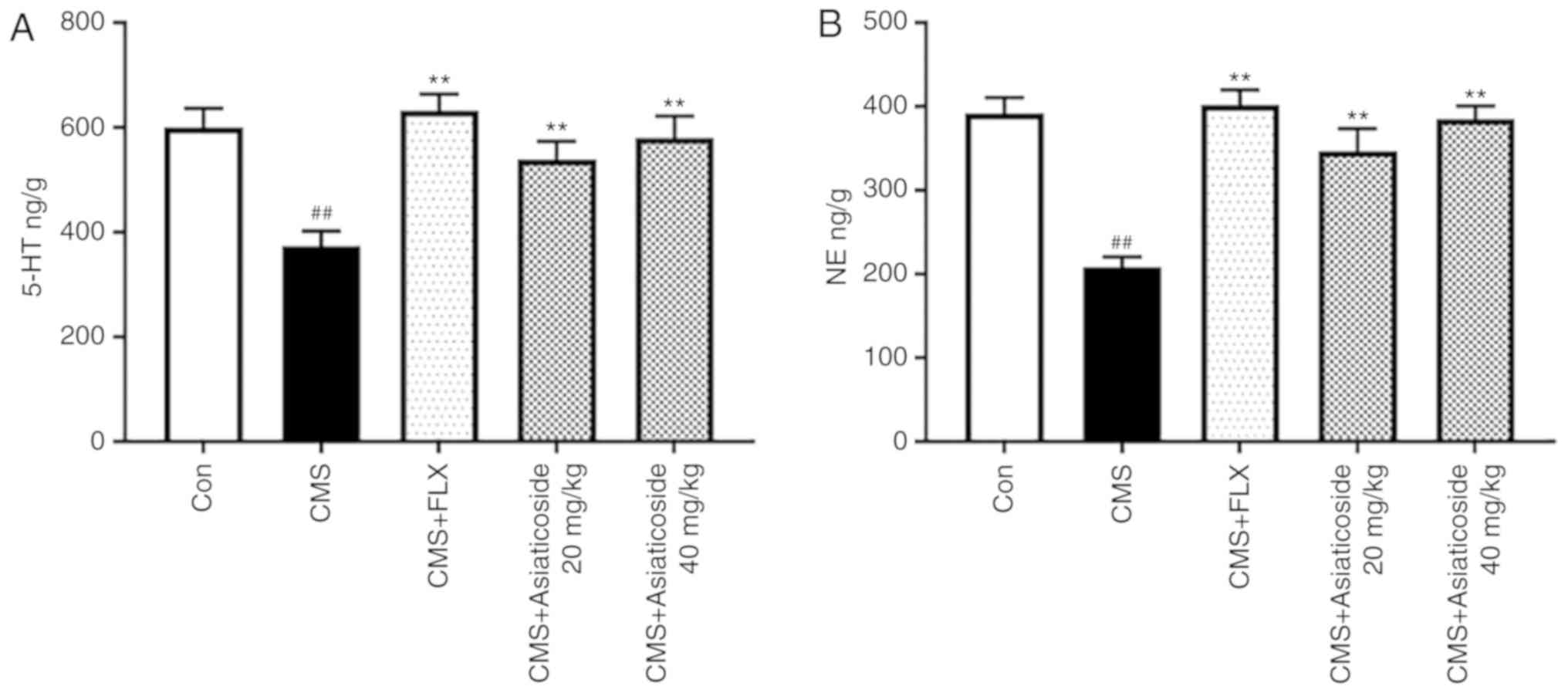
The present study also examined the levels of
neurotransmitters in the hippocampus. Fig. 3 shows the effect of asiaticoside on
the hippocampal levels of 5-HT and NE. The present results
suggested that CMS significantly decreased the levels of 5-HT and
NE in the hippocampus (P<0.01), whereas asiaticoside or FLX
administration significantly alleviated the levels of both 5-HT
(P<0.01; Fig. 3A) and NE
(P<0.01; Fig. 3B) in the
hippocampus of CMS model mice.
Effects of asiaticoside on IL-1β, IL-6
and TNF-α levels
The anti-inflammatory effect of asiaticoside in CMS
mice was identified in the present study (Fig. 4). It was found that treatment with
asiaticoside (20 and 40 mg/kg) or FLX (10 mg/kg) significantly
decreased the levels of IL-1β in CMS mice (P<0.01, P<0.001,
respectively; Fig. 4A). In the
hippocampus of the CMS mice, IL-6 and TNF-α levels were
significantly higher compared with the control group mice
(P<0.01, P<0.001, respectively). Asiaticoside (20 and 40
mg/kg) or FLX (10 mg/kg) administration significantly attenuated
the increase in IL-6 (P<0.05, P<0.01, respectively; Fig. 4B) and TNF-α (P<0.01, P<0.001;
respectively; Fig. 4C) levels in
these mice.
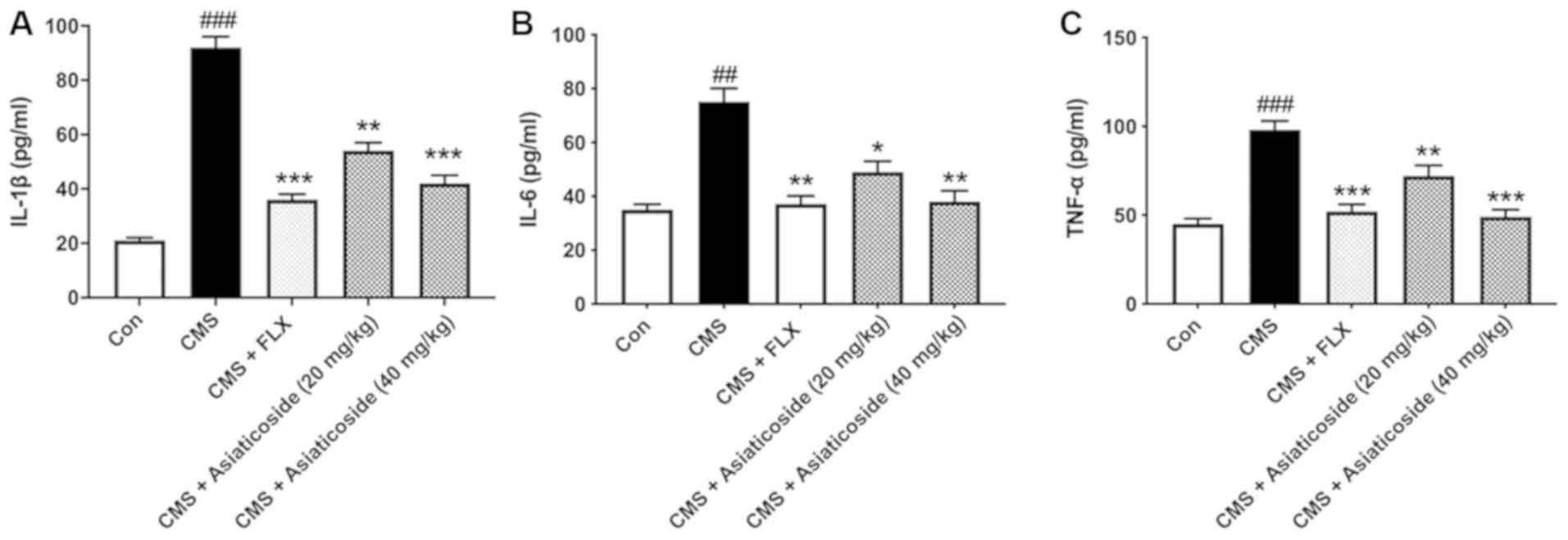 | Figure 4.Effects of asiaticoside treatment on
IL-1β, IL-6 and TNF-α levels. (A) IL-1β, (B) IL-6 and (C) TNF-α
were detected using ELISA and measured using a microplate reader at
450 nm absorbance. Data are presented as the mean ± SEM.
##P<0.01, ###P<0.001 vs. Con group;
*P<0.05, **P<0.01, ***P<0.001 vs. CMS-induced mice. CMS,
chronic unpredictable mild stress; FLX, fluoxetine; Con, control;
IL, interleukin; TNF-α, tumor necrosis factor α. |
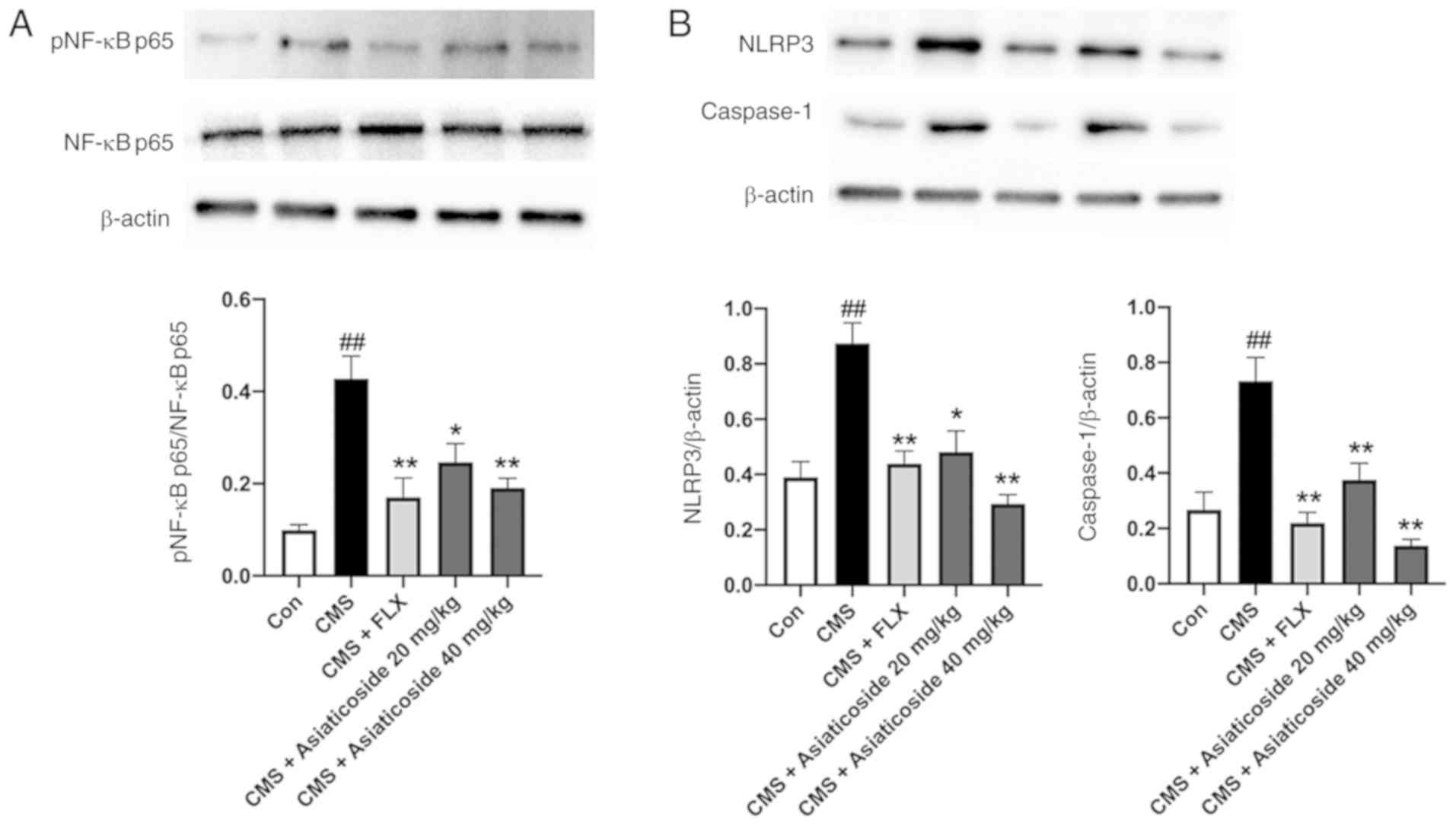
Effects of asiaticoside on pNF-κBp65,
NLRP3 and caspase-1 protein expression levels
The present study measured the protein expression
levels of pNF-κBp65 and NLRP3 inflammasome complex in the mice
hippocampus. It was identified that the protein expression of
pNF-κBp65 was significantly elevated (P<0.01) in the CMS mice
(Fig. 5A). Furthermore,
asiaticoside (20 and 40 mg/kg) or FLX administration significantly
reduced the expression of pNF-κBp65 (P<0.05, P<0.01,
respectively) in these mice. In addition, the protein expression
levels of NLRP3 and mature caspase-1 (Fig. 5B) were significantly increased in
the hippocampus of CMS mice (P<0.01). The present results also
indicated that asiaticoside (20 and 40 mg/kg) or FLX (10 mg/kg)
treatment significantly reduced the expression levels of NLRP3 and
mature caspase-1 in mice (P<0.05, P<0.01, respectively).
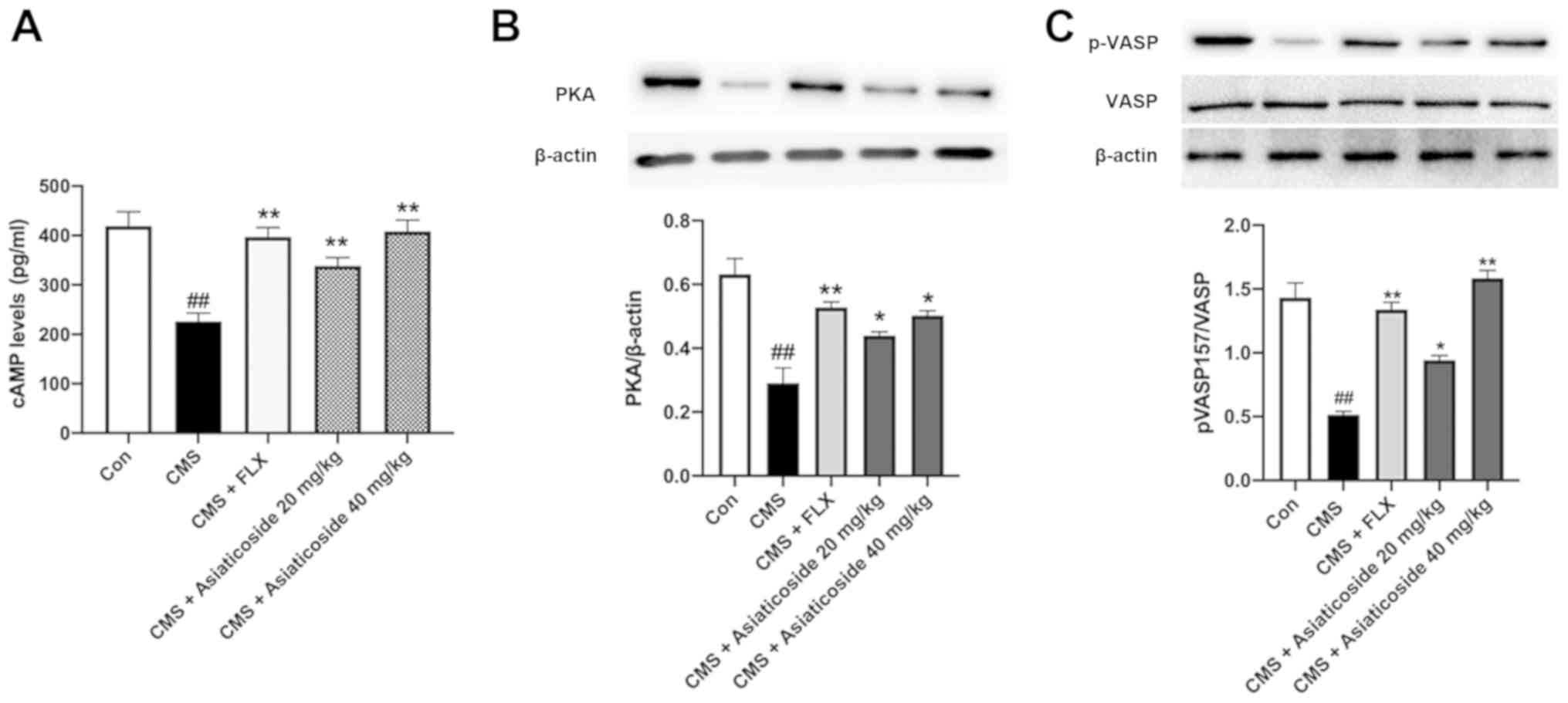
Effects of asiaticoside on cAMP, PKA
and p-vasodilator-stimulated phosphoprotein
(VASP)ser157
The protein expression levels of cAMP, PKA and
pVASPser157 were significantly decreased in the CMS mice
(Fig. 6) compared with the control
group (P<0.01). Moreover, treatment with asiaticoside (20 and 40
mg/kg) or FLX (10 mg/kg) reversed the CMS-induced decrease in the
expression levels of cAMP (P<0.01; Fig. 6A), PKA (P<0.05, P<0.01,
respectively; Fig. 6B) and
pVASPser157 (P<0.05, P<0.01, respectively;
Fig. 6C). Therefore, the present
results suggested that cAMP/PKA signaling may be involved in
asiaticoside-mediated inhibition of neuroinflammation.
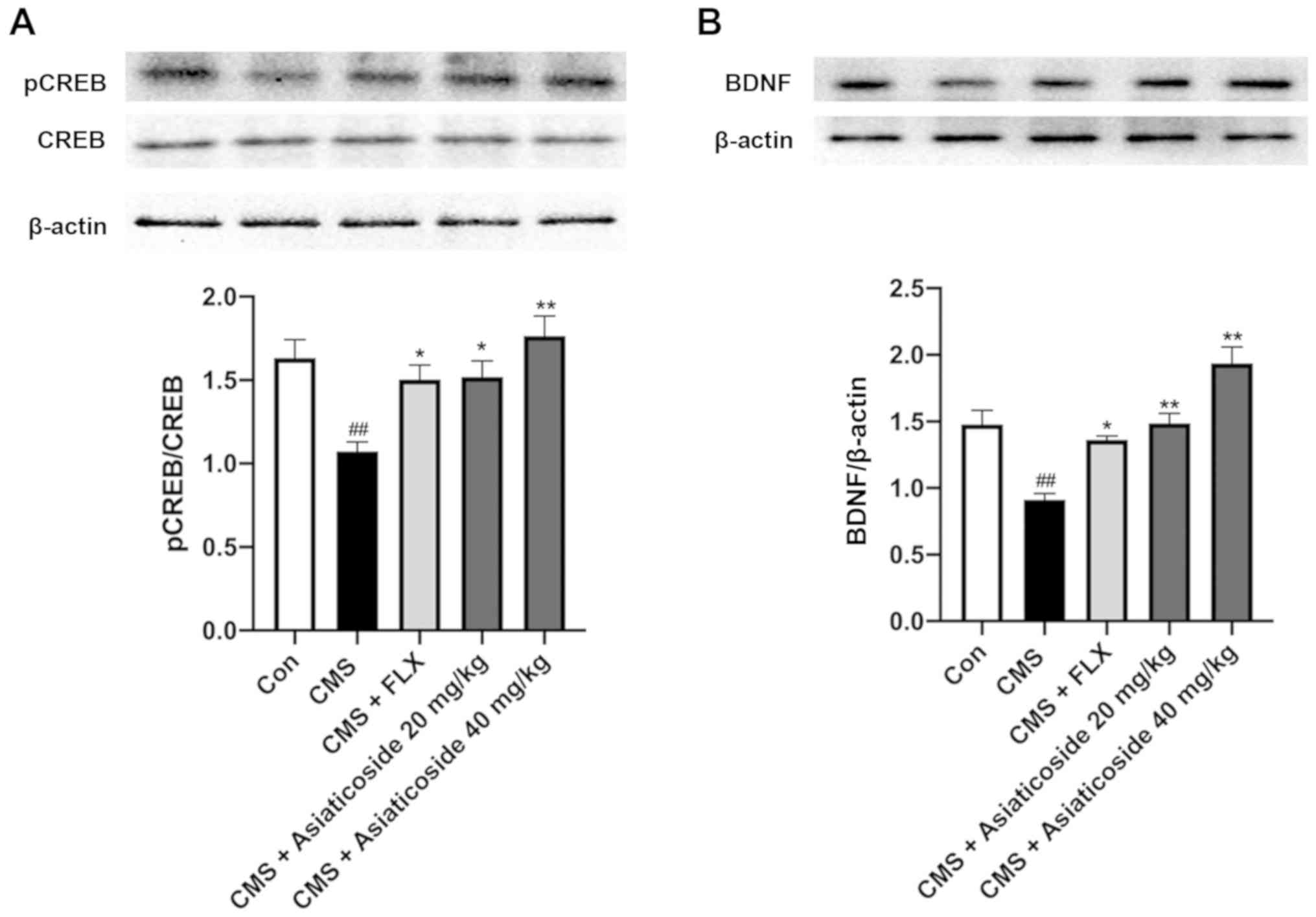
Effects of asiaticoside on
pCREBSer133 and BDNF expression levels
It has been reported that the antidepressant-like
effect of asiaticoside is mediated via the activation of BDNF
signaling (29). However, the
mechanism underlying asiaticoside-mediated alterations in BDNF
levels is yet to be elucidated. cAMP/PKA activation has been
reported to increase CREB phosphorylation and induce the expression
of CRE-regulated genes, such as BDNF (24). Therefore, the present study
examined the protein expression levels of pCREBSer133
and BDNF in CMS mice (Fig. 7). It
was found that the ratio of pCREBSer133 to CREB was
significantly decreased in the hippocampus of CMS mice (P<0.01),
which was reversed by asiaticoside or FLX treatments (P<0.05,
P<0.01, respectively; Fig. 7A).
Furthermore, BDNF expression was significantly lower in the
hippocampus of the CMS mice compared with the control mice
(P<0.01). However, this effect was completely reversed upon
asiaticoside or FLX treatment (P<0.01, P<0.05, respectively;
Fig. 7B), thus supporting the role
of asiaticoside in activating the cAMP/PKA signaling pathway.
Discussion
The present results suggested that asiaticoside
administration can reverse depressive-like behavior, increase the
levels of monoamine neurotransmitters, inhibit hippocampal
inflammation, and ameliorate pNF-κBp65 and NLRP3 inflammasome
levels in the CMS mouse model. Moreover, it was speculated that the
antidepressant-like effect of asiaticoside may occur via modulation
of the cAMP/PKA/NF-κB/NLRP3/CREB/BDNF signaling pathway.
The CMS mouse model is a validated animal model of
depression. CMS results in complex behavioral alterations, as well
as biochemical and cellular cascades, such as neurotransmitter,
endocrine, neurotrophic factors and inflammation, which are similar
to those observed in patients with major depression (13). The present mouse model demonstrated
decreased sucrose preference index in the sucrose preference test,
and increased immobility time during TST and FST. Moreover, the
present results are in line with those of previous studies using
CMS mice (17,31,32).
It has been shown that abnormal levels of monoamine
neurotransmitters are a typical feature of depression and stress in
animals, and have been proposed as the principal pathogenic factor
involved in this condition (10).
The present results indicated that the levels of neurotransmitters,
5-HT and NE, decreased significantly after CMS exposure, which is
consistent with previous studies (3,32).
Moreover, it was demonstrated that asiaticoside administration
significantly reversed the decrease in 5-HT and NE levels in a
mouse model of CMS-induced depression. In addition, it was found
that CMS-induced neurobehavioral and biochemical changes could be
restored by FLX, which was the positive control, and asiaticoside
administration, thus indicating that asiaticoside had
antidepressant effects in this model.
Previous studies revealed that inflammatory
cytokines are an important risk factor in the development of
depression in patients and animal models, and inhibiting
inflammatory processes could ameliorate depressive symptoms
(2,31,32).
In particular, the degradation pathways of the monoamine
neurotransmitter precursors have been reported to be activated by
inflammatory factors, which may lead to an increase in the levels
of these neurotransmitters (10).
A previous study also showed that asiaticoside exhibited a strong
anti-inflammatory effect in animal models of cognitive deficits,
spinal cord injury and cerebral ischemia-reperfusion injury
(27,33,34).
In the present study, it was demonstrated that CMS increased
hippocampal IL-1β, IL-6 and TNF-α levels, which were alleviated by
asiaticoside administration, thus indicating that the
antidepressant effect of asiaticoside may be due to its
anti-inflammatory properties.
CMS has previously been reported to induce
activation of NF-κB and the NLRP3 inflammasome, which in turn
increased the levels of IL-1β, IL-6 and TNF-α in the hippocampus of
animal models of depression (16).
Consistent with this previous study (16), the present results suggested that
the expression levels of pNF-κBp65 and the NLRP3 inflammasome,
which constitutes NLRP3 and caspase-1, were significantly increased
in the CMS group compared with the control group. Furthermore, it
was demonstrated that asiaticoside inhibited the activation of
pNF-κBp65, NLRP3 and caspase-1, leading to a reduction in the
levels of inflammatory cytokines and subsequent inflammatory
response. It has also been reported that asiaticoside may attenuate
inflammation by downregulating pNF-κBp65 and the levels of
inflammatory cytokines, such as IL-1β, IL-6 and TNF-α, in a rat
model of β-amyloid-induced Alzheimers disease (27).
The role of the cAMP/PKA signaling pathway in
depression and motor activity disturbances has been previously
identified (24). Moreover,
studies have shown a prominent role of the cAMP/PKA signaling
pathway in regulating NF-κB- and NLRP3-related inflammation
(18–22). NF-κB is a nuclear transcription
factor, which is regulated by cAMP via PKA. In the dimeric form,
NF-κB regulates the expression of genes involved in immune and
inflammatory responses (18–21).
Furthermore, the direct binding of cAMP to NLRP3 has also been
revealed (22). Therefore, it was
speculated that the cAMP/PKA signaling pathway may have an
important role in suppressing inflammatory responses. Moreover, it
has been shown that chronic stress inhibited cAMP/PKA signaling in
animal models of depression (35–37).
Consistent with previous studies, the present results indicated
that CMS downregulated the expression levels of cAMP and PKA in the
mouse hippocampus, whereas asiaticoside administration ameliorated
these changes (35–37). In addition, phosphorylation of
VASP157, a marker of PKA activation, was increased in the
asiaticoside-treated mice. Therefore, the present results suggested
that the antidepressant and anti-inflammatory activities of
asiaticoside in the CMS mouse model may be mediated via cAMP/PKA
signaling.
Exposure to chronic stress may induce
depressive-like behavior in animal models, inducing some of the
main features of depression, such as reduced levels of pCREB and
BDNF (24). Moreover, increased
PKA activation has been reported to enhance CREB phosphorylation,
which regulates the transcription of several genes, including BDNF,
which is involved in several critical functions of the CNS such as
learning and emotional responses (24). While previous studies using
senescence-accelerated mice or depression mouse models have
identified the involvement of CREB or BDNF/TrkB signaling in
regulating asiaticoside-mediated improvements in behavioral
response (28,38), relatively few studies have
evaluated the potential role of cAMP/PKA signaling in this process.
The present results suggested that CMS caused a significant
reduction in pCREB and BDNF levels in the hippocampus. Furthermore,
it was found that asiaticoside treatment increased pCREB and BDNF
levels, and cAMP/PKA signaling in a CMS model. Based on these
results, it was speculated that activation of the cAMP/PKA
signaling pathway by asiaticoside may play an important role in its
antidepressant effect.
In conclusion, the present results indicated that
asiaticoside may have an antidepressant-like effect in the CMS
mouse model. It was revealed that asiaticoside treatment increased
the levels of monoamine neurotransmitters, and reversed the
increase in inflammatory cytokines induced by CMS. Moreover, the
antidepressant and anti-inflammatory mechanism of action of
asiaticoside may be via regulation of the cAMP/PKA signaling
pathway. The present study may facilitate the development of
asiaticoside as a novel therapeutic for the treatment of
depression. However, further studies are required to further
identify the mechanism of asiaticoside in the activation of PKA
signalling in depression, and to assess its clinical effectiveness
in patients with depression or similar disorders. Future studies
will use neurons or glial cells, astrocytes or microglia, in
vivo and in vitro to further support the conclusion.
Collectively, the present results suggested that administration of
asiaticoside may be a useful natural adjuvant in antidepressant
therapy.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural
Science Foundation of China (grant no. 81870943) and the
Postdoctoral Research Fund of Xuzhou Medical University (grant no.
2015107026).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors contributions
LW and TG made substantial contributions to
acquisition of data, and analysis and interpretation of data. YG
interpreted data and revised the manuscript critically for
important intellectual content. YX made substantial contributions
to conception and design, and agreed to be accountable for all
aspects of the work in ensuring that questions related to the
accuracy or integrity of any part of the work are appropriately
investigated and resolved. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
All experiments were conducted as per the Guidelines
of the Institutional Animal Care and Use Committee of China and the
present study was approved by Experimental Animal Ethics Committee
of Xuzhou Medical University
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ota KT and Duman RS: Environmental and
pharmacological modulations of cellular plasticity: Role in the
pathophysiology and treatment of depression. Neurobiol Dis.
57:28–37. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yu HY, Yin ZJ, Yang SJ, Ma SP and Qu R:
Baicalin reverses depressive-like behaviours and regulates
apoptotic signalling induced by olfactory bulbectomy. Phytother
Res. 30:469–475. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lee WY, Jang SW, Lee JS, Kim YH, Kim HG,
Han JM, Kim DW, Yi MH, Choi MK and Son CG: Uwhangchungsimwon, a
traditional herbal medicine, protects brain against oxidative
injury via modulation of hypothalamus-pituitary-adrenal (HPA)
response in a chronic restraint mice model. J Ethnopharmacol.
151:461–469. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Miller AH and Raison CL: The role of
inflammation in depression: From evolutionary imperative to modern
treatment target. Nat Rev Immunol. 16:22–34. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Inserra A, Mastronardi CA, Rogers G,
Licinio J and Wong ML: Neuroimmunomodulation in major depressive
disorder: focus on caspase 1, inducible nitric oxide synthase, and
interferon-gamma. Mol Neurobiol. 56:4288–4305. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liberman AC, Trias E, da Silva Chagas L,
Trindade P, Dos Santos Pereira M, Refojo D, Hedin-Pereira C and
Serfaty CA: Neuroimmune and inflammatory signals in complex
disorders of the central nervous system. Neuroimmunomodulation.
25:246–270. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Lanquillon S, Krieg JC, Bening-Abu-Shach U
and Vedder H: Cytokine production and treatment response in major
depressive disorder. Neuropsychopharmacology. 22:370–379. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Jeon SW and Kim YK: The role of
neuroinflammation and neurovascular dysfunction in major depressive
disorder. J Inflamm Res. 11:179–192. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Yu H, Zhang F and Guan X: Baicalin reverse
depressive-like behaviors through regulation SIRT1-NF-kB signaling
pathway in olfactory bulbectomized rats. Phytother Res.
33:1480–1489. 2019. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Barua CC, Haloi P, Saikia B, Sulakhiya K,
Pathak DC, Tamuli S, Rizavi H and Ren X: Zanthoxylum alatum
abrogates lipopolysaccharide-induced depression-like behaviours in
mice by modulating neuroinflammation and monoamine
neurotransmitters in the hippocampus. Pharm Biol. 56:245–252. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Raison CL, Capuron L and Miller AH:
Cytokines sing the blues: Inflammation and the pathogenesis of
depression. Trends Immunol. 27:24–31. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Duarte-Silva E, Macedo D, Maes M and
Peixoto CA: Novel insights into the mechanisms underlying
depression-associated experimental autoimmune encephalomyelitis.
Prog Neuropsychopharmacol Biol Psychiatry. 93:1–10. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yirmiya R, Rimmerman N and Reshef R:
Depression as a microglial disease. Trends Neurosci. 38:637–658.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Afonina IS, Zhong Z, Karin M and Beyaert
R: Limiting inflammation-the negative regulation of NF-κB and the
NLRP3 inflammasome. Nat Immunol. 18:861–869. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ratajczak MZ, Adamiak M, Thapa A, Bujko K,
Brzezniakiewicz-Janus K and Lenkiewicz AM: NLRP3 inflammasome
couples purinergic signaling with activation of the complement
cascade for the optimal release of cells from bone marrow.
Leukemia. 33:815–825. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Shao BZ, Cao Q and Liu C: Targeting NLRP3
inflammasome in the treatment of CNS diseases. Front Mol Neurosci.
11:3202018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yu HY, Yin ZJ, Yang SJ and Ma SP: Baicalin
reverse AMPA receptor expression and neuron apoptosis in chronic
unpredictable mild stress rats. Biochem Biophys Res Commun.
451:467–472. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Gerlo S, Kooijman R, Beck IM, Kolmus K,
Spooren A and Haegeman G: Cyclic AMP: A selective modulator of
NF-κB action. Cell Mol Life Sci. 68:3823–3841. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zuo L, Shi L and Yan F: The reciprocal
interaction of sympathetic nervous system and cAMP-PKA-NF-kB
pathway in immune suppression after experimental stroke. Neurosci
Lett. 627:205–210. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gilmore TD: Introduction to NF-kappaB:
Players, pathways, perspectives. Oncogene. 25:6680–6684. 2006a.
View Article : Google Scholar
|
|
21
|
Gilmore TD and Herscovitch M: Inhibitors
of NF-kappaB signaling: 785 and counting. Oncogene. 25:6887–6899.
2006b. View Article : Google Scholar
|
|
22
|
Mortimer L, Moreau F, MacDonald JA and
Chadee K: NLRP3 inflammasome inhibition is disrupted in a group of
auto-inflammatory disease CAPS mutations. Nat Immunol.
17:1176–1186. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Keil MF, Briassoulis G, Stratakis CA and
Wu TJ: Protein kinase A and anxiety-related behaviors: A
mini-review. Front Endocrinol (Lausanne). 7:832016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Ramezany Yasuj S, Nourhashemi M,
Keshavarzi S, Motaghinejad M and Motevalian M: Possible role of
cyclic AMP response element binding/brain-derived neurotrophic
factor signaling pathway in mediating the pharmacological effects
of duloxetine against methamphetamine use-induced cognitive
impairment and withdrawal-induced anxiety and depression in rats.
Adv Biomed Res. 8:112019. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Gao YZ, Zhao LF, Ma J, Xue WH and Zhao H:
Protective mechanisms of wogonoside against
Lipopolysaccharide/D-galactosamine-induced acute liver injury in
mice. Eur J Pharmacol. 780:8–15. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
He L, Hong G, Zhou L, Zhang J, Fang J, He
W, Tickner J, Han X, Zhao L and Xu J: Asiaticoside, a component of
Centella asiatica attenuates RANKL-induced
osteoclastogenesis via NFATc1 and NF-κB signaling pathways. J Cell
Physiol. 234:4267–4276. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang C, Chen S, Zhang Z, Xu H, Zhang W,
Xu D, Lin B and Mei Y: Asiaticoside alleviates cerebral
ischemia-reperfusion injury via NOD2/mitogen-activated protein
kinase (MAPK)/nuclear factor kappa B (NF-κB) signaling pathway. Med
Sci Monit. 26:e9203252020.PubMed/NCBI
|
|
28
|
Luo L, Liu XL, Mu RH, Wu YJ, Liu BB, Geng
D, Liu Q and Yi LT: Hippocampal BDNF signaling restored with
chronic asiaticoside treatment in depression-like mice. Brain Res
Bull. 114:62–69. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Nalinratana N, Meksuriyen D and
Ongpipattanakul B: Differences in neuritogenic activity and
signaling activation of madecassoside, asiaticoside, and their
aglycones in neuro-2a cells. Planta Med. 84:1165–1173. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Paxinos G and Watson C: The Rat
Hippocampus in Stereotaxic Coordinates. Academic Press. (San Diego,
CA). 1998.
|
|
31
|
Du RH, Tan J, Sun XY, Lu M, Ding JH and Hu
G: Fluoxetine inhibits NLRP3 inflammasome activation: Implication
in depression. Int J Neuropsychopharmacol. 19:192016. View Article : Google Scholar
|
|
32
|
Deng XY, Li HY, Chen JJ, Li RP, Qu R, Fu Q
and Ma SP: Thymol produces an antidepressant-like effect in a
chronic unpredictable mild stress model of depression in mice.
Behav Brain Res. 291:12–19. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen S, Yin ZJ, Jiang C, Ma ZQ, Fu Q, Qu R
and Ma SP: Asiaticoside attenuates memory impairment induced by
transient cerebral ischemia-reperfusion in mice through
anti-inflammatory mechanism. Pharmacol Biochem Behav. 122:7–15.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Luo Y, Fu C, Wang Z, Zhang Z, Wang H and
Liu Y: Asiaticoside attenuates the effects of spinal cord injury
through antioxidant and anti-inflammatory effects, and inhibition
of the p38-MAPK mechanism. Mol Med Rep. 12:8294–8300. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li XY, Qi WW, Zhang YX, Jiang SY, Yang B,
Xiong L and Tong JC: Helicid ameliorates learning and cognitive
ability and activities cAMP/PKA/CREB signaling in chronic
unpredictable mild stress rats. Biol Pharm Bull. 42:1146–1154.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Zhang R, Guo L, Ji Z, Li X, Zhang C, Ma Z,
Fu Q, Qu R and Ma S: Radix scutellariae attenuates CUMS-induced
depressive-like behavior by promoting neurogenesis via cAMP/PKA
pathway. Neurochem Res. 43:2111–2120. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Jiang H, Zhang X, Wang Y, Zhang H, Li J,
Yang X, Zhao B, Zhang C, Yu M, Xu M, et al: Mechanisms underlying
the antidepressant response of acupuncture via PKA/CREB signaling
pathway. Neural Plast. 2017:41351642017. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lin X, Huang R, Zhang S, Wei L, Zhuo L, Wu
X, Tang A and Huang Q: Beneficial effects of asiaticoside on
cognitive deficits in senescence-accelerated mice. Fitoterapia.
87:69–77. 2013. View Article : Google Scholar : PubMed/NCBI
|