Introduction
As an effective mode of treatment for acute
respiratory distress syndrome (ARDS), mechanical ventilation (MV)
is widely performed in patients under general anesthesia (1). However, certain MV strategies can
result in ventilator-associated complications, such as
ventilator-induced lung injury (VILI) (2–4). In
addition to mechanical damage, there is growing evidence that
upregulation of inflammatory molecules, chemokines, leukocyte
sequestration and excessive inflammatory responses has a vital role
in the generation and progression of VILI (5–7).
However, the definite mechanism through which this pro-inflammatory
response is initiated and sustained, and how to relieve it, remain
unclear.
Recently, it was reported that dexmedetomidine (Dex)
exhibits analgesic and sedative properties for systemic anesthesia
by intravenous (i.v.) infusion through the activation of α2
adrenergic receptors (8). In
patients subjected to MV, Dex has been widely used in intensive
care units. Ample evidence has been reported on the protective
effects of MV on organs (9). It
has been demonstrated that Dex could ameliorate VILI-induced
pulmonary inflammation in animal models (10,11);
Chen et al (12) observed
that dex reduces VILI induced inflammation by inhibiting TLR4 and
NF-κB signaling through α2-adrenoreceptors. However, the exact
mechanism has yet to be established. It was hypothesized that the
protective signaling pathways in the lung cells are initiated
through Dex for the prevention of cellular apoptosis or necrosis
when subjected to MV.
ERK1/2 is a member of the MAPK family, and serves a
key role in the regulation of cell death and survival (13–15).
The inhibition of ERK1/2 signaling induces the activation of
apoptosis-associated proteins, such as BH3-interacting domain death
agonist, Bcl-2-like protein 11 and p53 upregulated modulator of
apoptosis. In addition, the MEK1/2-ERK1/2 signaling pathway may be
involved in modulating the inflammatory response to lung injury and
infection (16). However, little
attention has been dedicated to the effects of Dex administration
with respect to ERK1/2 and the expression of caspase-3, Bcl-2 and
Bcl-2 homologous antagonist/killer (Bak) in VILI. Therefore, the
present study aimed to provide evidence that supports Dex as a
treatment against VILI in a rat model of high-tidal volume MV
(HVT-MV) and to investigate the roles of phosphorylated
(p)-ERK1/2.
Materials and methods
Animals
A total of 168 adult male Sprague-Dawley rats (6–8
weeks old, 200–250 g) were purchased from Liaoning Changsheng
Biotechnology and housed one per cage at a stable temperature of
25±1°C with alternating 12-h light/dark cycle. Rats were allowed
access to food and water ad libitum until the start of the
experiments. All study procedures and animal handling were approved
by the Animal Review Board of Cangzhou Central Hospital (Cangzhou,
China) and were in accordance with the National Institutes of
Health guidelines.
Group assignment and experimental
protocol
After successful sevoflurane anesthesia (5% for
induction; 1.5–2.5% for maintenance), an indwelling needle (22G)
was placed into the catheterized caudal (tail) vein. The left
femoral artery was cannulated with a heparinized tube to monitor
arterial blood pressure. The level of sevoflurane was adjusted to
maintain stable hemodynamics. According to computer-based
randomization, the rats were assigned to one of the following seven
groups: i) Sham; ii) VILI; iii) VILI+low dose (L)-Dex; iv)
VILI+high dose (H)-Dex; v) VILI+L-Dex+PD98059 (PD; Calbiochem;
Merck KGaA); vi) VILI+H-Dex+PD; and vii) PD only group. In the Sham
group, rats were anesthetized with sevoflurane and kept on
spontaneous breathing without MV for 4 h, and IV catheterization
was performed. In the VILI group, the endotracheal intubation tube
was connected to a ventilator (model 683 Ventilator; Harvard
Apparatus) for 4 h. MV with a high tidal volume of 20 ml/kg,
respiratory frequency of 50 bpm, and compressed air was used. Each
rat could achieve a peak airway pressure of ~40 cm H2O,
as measured by an Exactus II pressure gauge (Omron Healthcare
Inc.). VILI+L-Dex group rats received the same MV strategy as those
in the VILI group and received i.v. administration of a loading
dose of Dex (Jiangsu Hengrui Medicine Co., Ltd.) of 1 µg/kg over 15
min before MV, followed by a maintenance dose of 1 µg/kg/h (i.v.)
during MV. Similarly, rats in the VILI+H-Dex group received the
same MV strategy and received i.v. administration of 10 µg/kg for
loading dose and 10 µg/kg/h for maintenance dose (17,18).
Rats in the VILI+L-Dex+PD group and the VILI+H-Dex+PD received
subcutaneous injections of 1 mg/kg PD, an ERK1/2 inhibitor
(19,20), 30 min prior to MV and Dex infusion.
The dose of 1 mg/kg was based on a preliminary experiment and a
previous study (21).
Subsequently, the ventilation strategy of the rats and the infusion
of Dex were consistent with the VILI+L-Dex group and the VILI+H-Dex
groups. Finally, rats in the PD group received 1 mg/kg PD
subcutaneously.
At the end of each experiment, the lungs and
bronchoalveolar lavage fluid (BALF) were collected under anesthesia
with sevoflurane. The left lung lobe was used for determining the
lung wet/dry (W/D) weight ratio after removing the blood and water
from the lung surface.
Histopathological analysis
After fixing in 4% paraformaldehyde at room
temperature overnight, the lung tissues were dehydrated and
embedded in paraffin. The tissue sections (thickness, 4 µm) were
stained with hematoxylin and eosin (H&E). The images were
captured under a light microscope and then assessed by a
pathologist who was blinded to the groups. Each sample was
evaluated according to the following three criteria: i) Alveolar
edema; 0, no edema in the interstitium; 1, slight edema in the
interstitium wit hyaline membrane formation; 2, interstitial edema
is evident; 3, interstitial edema is further aggravated and hyaline
membrane was formed. ii) Diffuse alveolar hemorrhage and
congestion; 0, none; 1, prominent congestion with extravasated
blood; 2, <25% extravasated blood; and 3, >25% extravasated
blood. iii) Intra-alveolar infiltration of inflammatory cells; 0,
none; 1, <10% aggregates; 2, 10–30% aggregates; and 3, >30%
aggregates. The indexes of the final score are accumulated.
ELISA
Interleukin-6 assay kit (cat. no. H007),
interleukin-1β assay kit (cat. no. H002) and tumor necrosis
factor-α (TNF-α) assay kit (cat. no. H052) were purchased from
Nanjing Jiancheng Bioengineering Institute and were used to measure
IL-6, IL-1β and TNF-α levels, respectively, in the BALF according
to the manufacturer's protocol.
Lung W/D weight ratio
Pulmonary edema development was assessed by the
ratio of lung W/D weight. Briefly, the lung tissues were separated
from the upper lobes of the left lung. After washing in saline, the
tissues were weighed immediately and reweighed followed dehydration
at 80°C for 24 h.
Immunofluorescence
To quantitatively measure alveolar epithelial cell
death, the TUNEL/propidium iodide (PI)/Hoechst 33342 assay was
used. After dewaxing with xylene and a serial ethanol gradient
concentration (100% ethanol for 3 min, 95% ethanol for 2 min, 80%
ethanol for 2 min, 75% ethanol for 2 min, H2O for 1
min), the tissue sections (4 µm) were incubated with 20 µg/ml
proteinase K (cat. no. ST533; Beyotime Institute of Biotechnology)
for 10 min at 37°C. Then, the slices were treated with
TUNEL-mixture (cat. no. C1088; Beyotime Institute of Biotechnology)
in the dark for 1 h at 37°C. After washing with PBS, 5 µg/ml of
Antifade Mounting Medium with Hoechst 33342 and propidium iodide
(PI; cat. no. P0137; Beyotime Institute of Biotechnology) were
added for 2 min at room temperature to stain the cell nuclei and
necrotic cell. Four nonoverlapping fields of view were evaluated in
each tissue section. The percentage of necrosis and apoptosis was
indicated by the ratio of PI-positive nuclei or TUNEL-positive
nuclei to the total cell nuclei counterstaining by Hoechst 33342,
respectively.
Western blot analysis
Western blotting was used to assess the expression
in ERK1/2, caspase-3, Bcl-2 and Bak. Total protein was extracted
from lung tissues and lysed in tissue lysis buffer (cat. no. P0013;
Beyotime Institute of Biotechnology), and the concentration was
measured using a bicinchoninic acid Protein Assay Kit (cat. no.
P0012S; Beyotime Institute of Biotechnology). Samples containing 30
µg of total protein were separated on a 12% SDS-PAGE and
transferred to a polyvinylidene fluoride membrane. The membranes
were blocked with QuickBlock™ blocking buffer for western blot
(Beyotime Institute of Biotechnology) at 25°C for 10 min, and then
incubated with primary antibodies of rabbit monoclonal anti-ERK1/2
(1:1,000; cat. no. AF1051; Beyotime Institute of Biotechnology),
anti-phospho-ERK1/2 (1:1,000; cat. no. AF1891; Beyotime Institute
of Biotechnology) polyclonal anti-caspase-3 (1:1,000; cat. no.
ab13847; Abcam), anti-Bak (1:500; cat. no. AB016; Beyotime
Institute of Biotechnology) and anti-Bcl-2 (1:500; cat. no.
K003505P; Beijing Solarbio Science & Technology Co., Ltd.)
overnight at 4°C. After rinsing with TBS+Tween-20 (0.05%) buffer
(Beyotime Institute of Biotechnology), the membranes were probed
with HRP-conjugated goat anti-rabbit secondary antibodies (1:2,000;
cat. no. A0208; Beyotime Institute of Biotechnology) for 1 h at
25°C. After incubation with an ECL plus kit (Beyotime Institute of
Biotechnology) for 5 min, the blot was visualized and
semi-quantified using Image Lab 5.1 software (Bio-Rad Laboratories,
Inc.). GAPDH (1:1,000; cat. no. K106389P; Beijing Solarbio Science
& Technology Co., Ltd.) was used as an internal reference.
Statistical analysis
All statistical analyses were performed using SPSS
Statistics version 20.0 (IBM Corp). Data are expressed as the mean
± SD. When only two groups were compared, an unpaired t-test was
used. Differences between more than two groups were assessed by
one-way analyses of variance followed by a Bonferroni's post hoc
test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Dex attenuates histopathological
damage in rat lungs
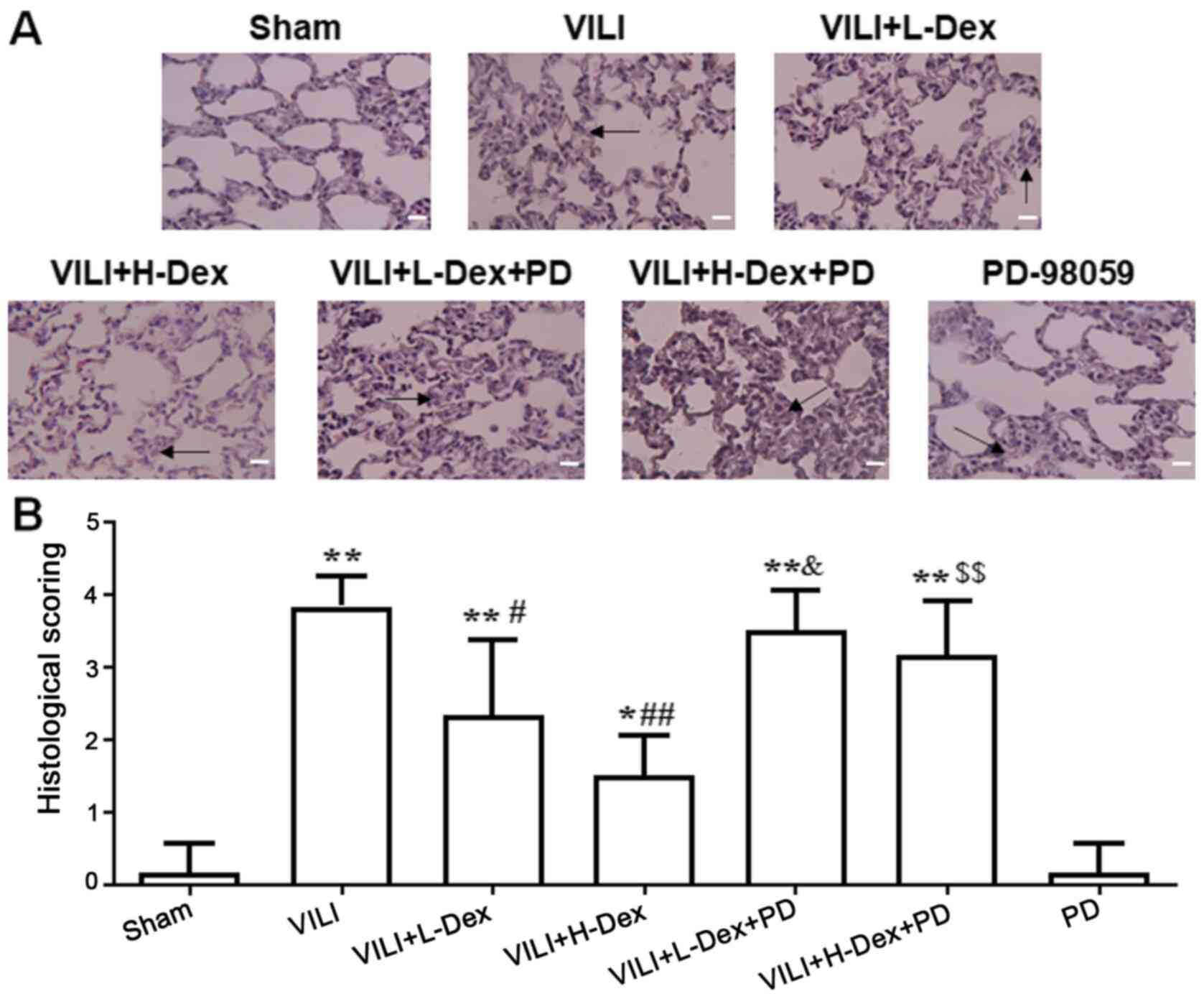
Lungs were stained with H&E to assess
histopathology based on morphology and scoring. The structural
changes and pulmonary edema in the VILI group were more serious
compared with those in the Sham group (Fig. 1). Dex treatment alleviated the lung
morphologic injury, especially in the VILI+H-Dex group compared
with the VILI group. However, after inhibition with PD98059, the
protective effect of Dex on morphology was weakened. PD98059
injection alone did not damage the lung tissue.
Dex was demonstrated to have little
effect on hemodynamics
During the treatment process, the hemodynamics of
rats were found to be relatively stable (Tables SI and SII). This indicated that hemodynamics
have no effect on the pathology and inflammatory response.
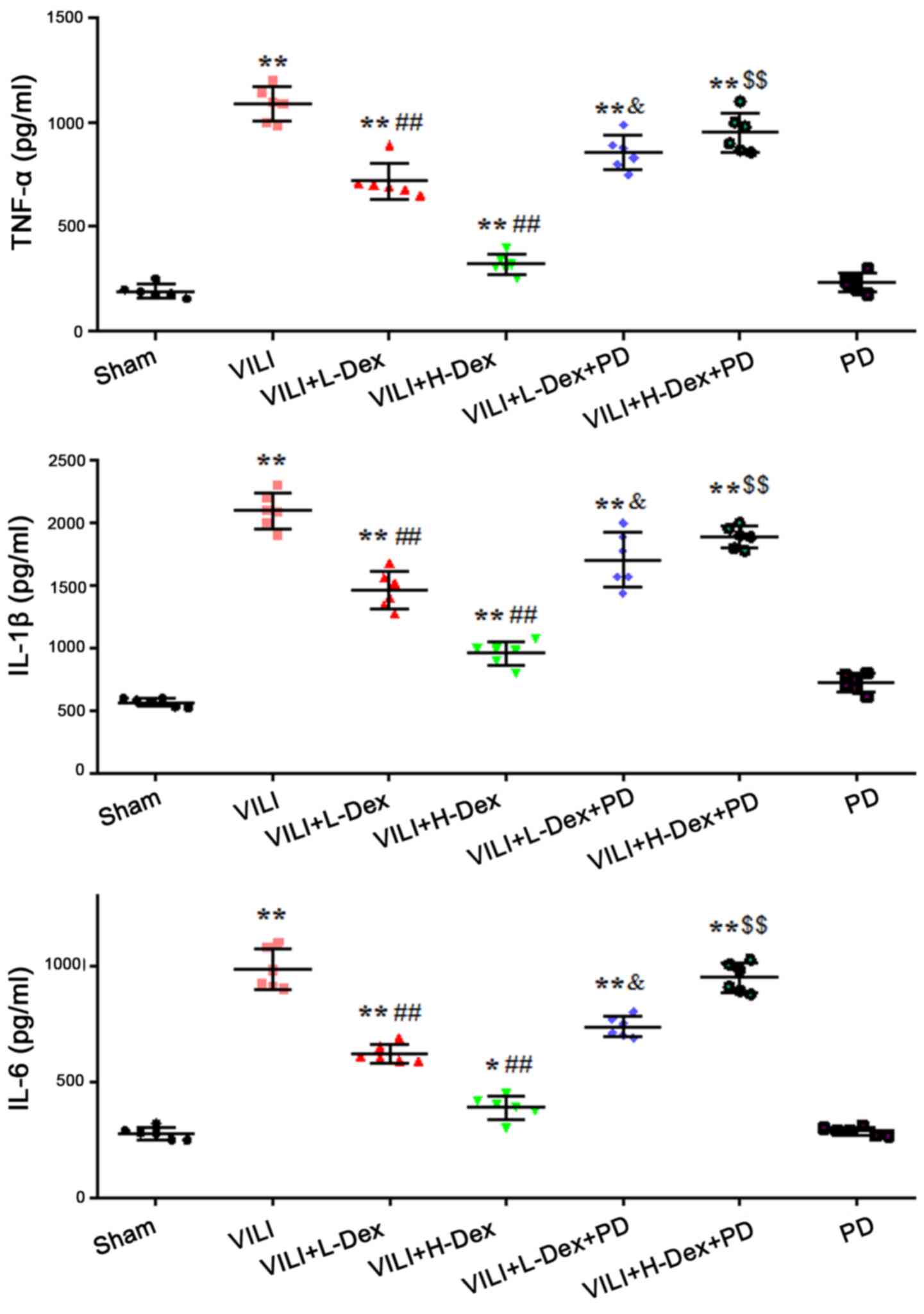
Dex reduces the level of inflammatory
cytokines in BALF
The effects of Dex on inflammatory cytokine levels
in BALF were measured using ELISAs. The results demonstrated that
the production of TNF-α, IL-1β and IL-6 in the VILI group were
significantly increased compared with levels in the Sham group
(P<0.05; Fig. 2). L-Dex
(P<0.05) and H-Dex (P<0.01) significantly downregulated the
expression levels of inflammatory cytokines compared with the VILI
group. PD98059 pretreatment significantly offset the effects of Dex
in the VILI+L-Dex and VILI+H-Dex groups, compared with the
VILI+L-Dex+PD and VILI+H-Dex+PD groups (both P<0.05; Fig. 2).
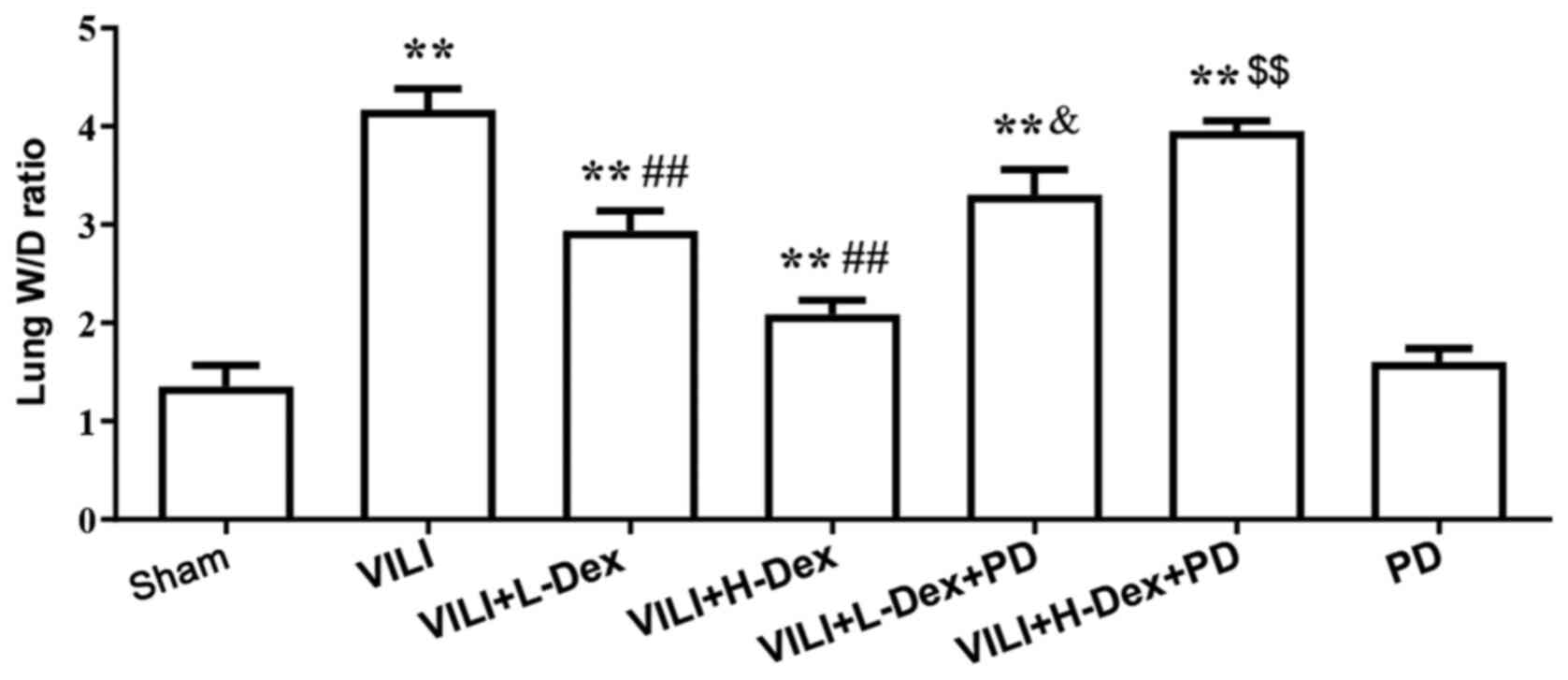
Dex reduces the lung W/D weight
ratio
Pulmonary edema was evaluated by the W/D weight
ratio of the lungs (Fig. 3). The
W/D ratio in the VILI group was found to be significantly higher
compared with that in the Sham group (P<0.05). In the VILI+L-Dex
(P<0.05) and the VILI+H-Dex treatment groups (P<0.01), Dex
significantly reduced the lung W/D ratios compared with the VILI
group. PD98059 pretreatment was found to reverse the protective
effects of Dex (P<0.05).
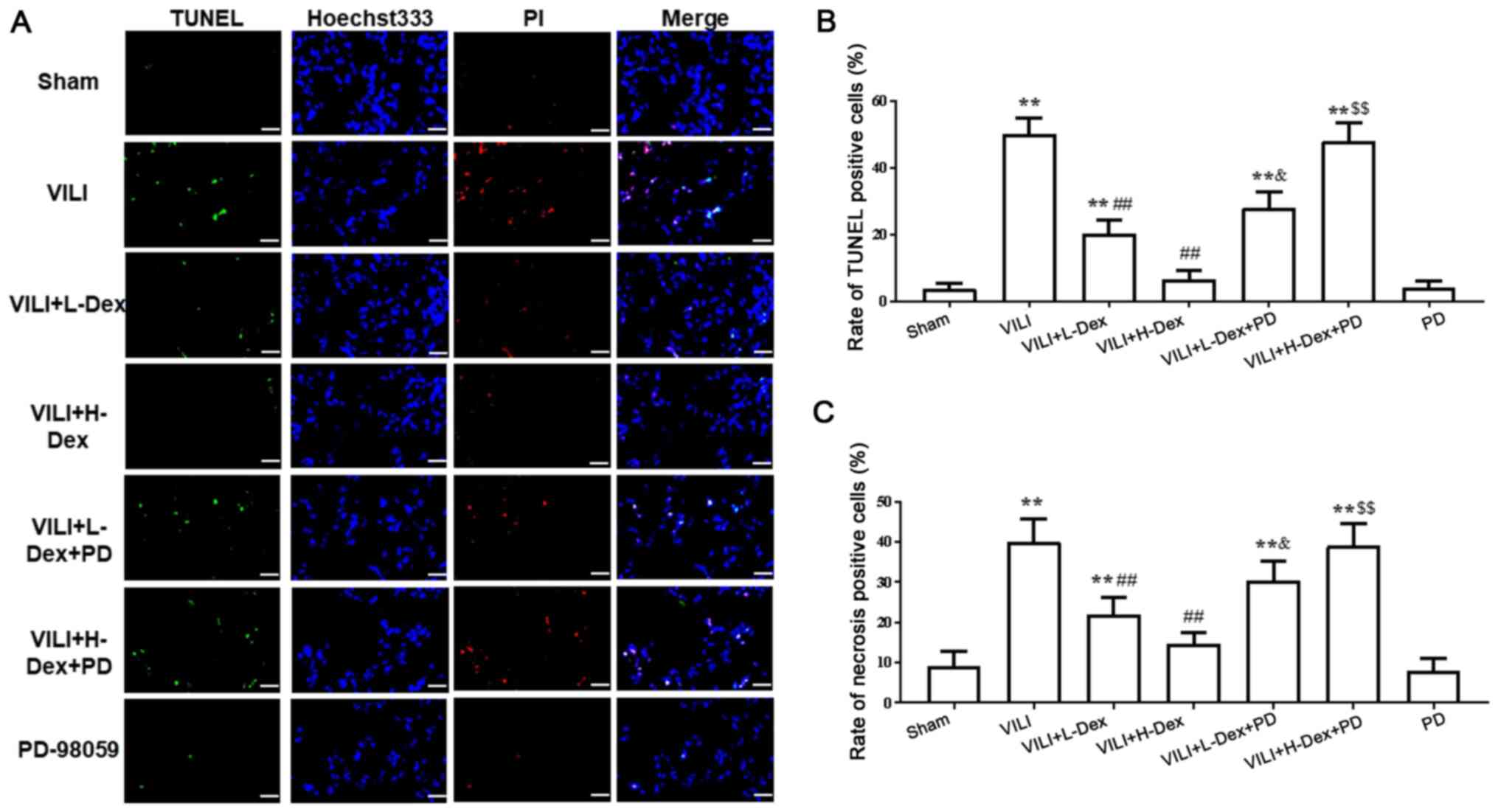
Dex reduces alveolar epithelial cell
death
Immunofluorescence assays demonstrated that,
compared with the Sham group, the necrosis and apoptosis of
alveolar epithelial cells significantly increased in the VILI
treated group (P<0.05; Fig. 4).
However, a significant decrease was revealed in VILI model rats
treated with L-Dex or H-Dex compared with the VILI group (P<0.05
and P<0.01, respectively). Moreover, compared with the rats
treated with VILI+H-Dex and VILI+L-Dex, there was a significant
increase in cell death in the VILI+H-Dex+PD and VILI+L-Dex+PD
groups, respectively (P<0.05 for both; Fig. 4). Additionally, there was no
significant difference in cell death between the Sham group and the
PD group.
Dex may inhibit alveolar epithelial
cell apoptosis and decrease the Bak/Bcl-2 ratio via ERK1/2
activation
To further understand the mechanism of Dex on cell
death, the protein expression levels of Bak, Bcl-2, pro-caspase-3
and cleaved caspase-3 in the lung tissues were detected by western
blotting (Fig. 5). The expression
levels of Bak/Bcl-2 and cleaved caspase-3 significantly increased
in the VILI, VILI+L-Dex, VILI+H-Dex, VILI+L-Dex+PD and
VILI+H-Dex+PD groups compared with the Sham group (P<0.05;
Fig. 5), whereas compared with the
VILI group, L-Dex and H-Dex significantly reduced the expression
levels of apoptotic proteins Bak/Bcl-2 and cleaved caspase-3.
PD98059 co-treatment largely reversed the Bak/Bcl-2 and cleaved
caspase-3 in the VILI+L-Dex+PD and VILI+H-Dex+PD groups compared
with VILI+L-Dex and VILI+H-Dex. Moreover, there was no difference
in pro-caspase-3 expression in each group.
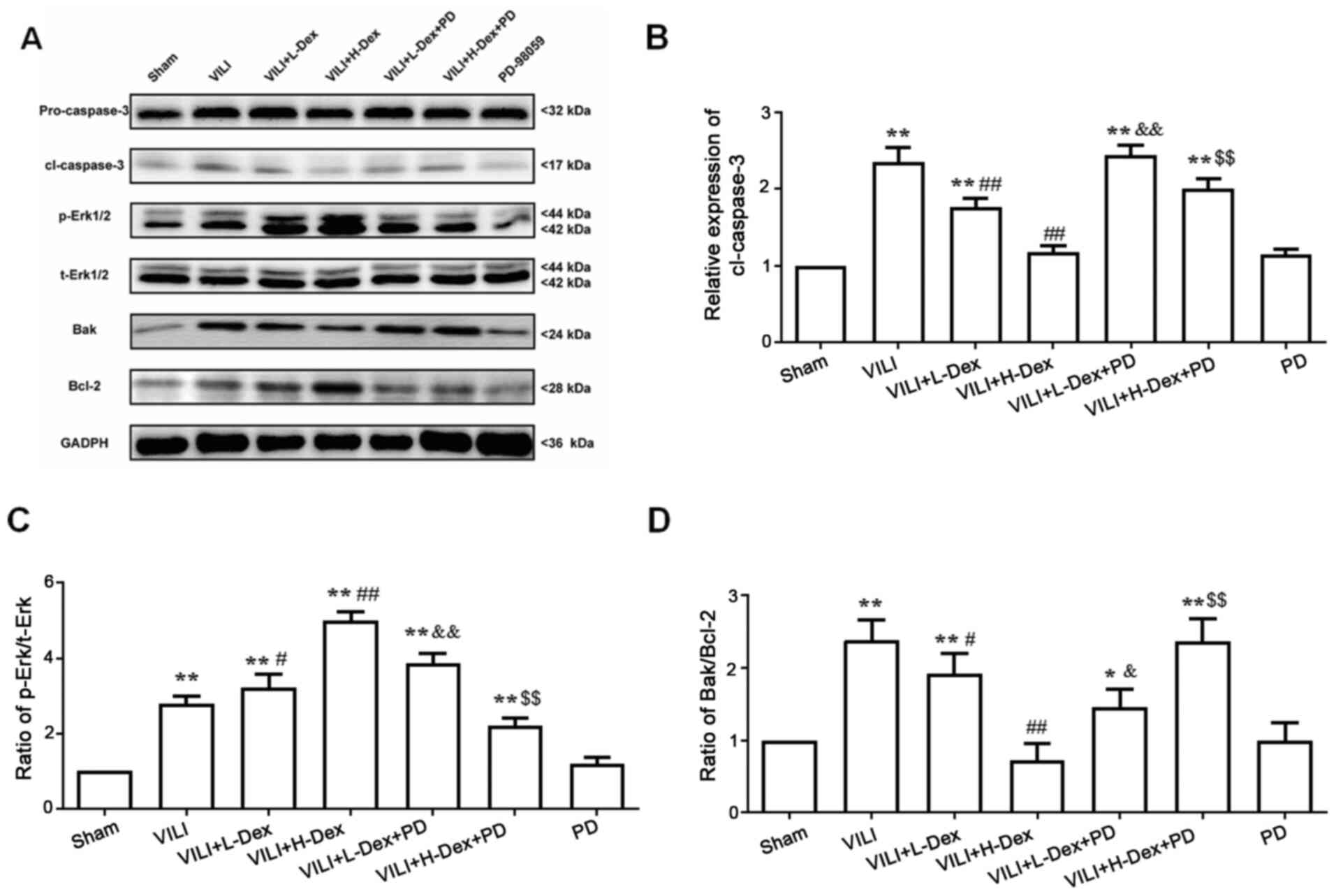 | Figure 5.Dex may inhibit alveolar epithelial
cell apoptosis and decrease the Bak/Bcl-2 ratio through ERK1/2
activation. (A) Representative western blotting images of pro- and
cl-caspase-3, p-ERK1/2, t-ERK1/2, Bak and Bcl-2 in the lung. The
semi-quantified protein expression level of (B) cl-caspase-3, (C)
p-ERK/t-ERK and (D) Bak/Bcl-2. Data are presented as the mean ± SD
(n=6). *P<0.05, **P<0.01 vs. Sham; #P<0.05,
##P<0.01 vs. VILI; &P<0.05,
&&P<0.01 VILI+L-Dex+PD vs. VILI+L-Dex group;
$$P<0.01 VILI+H-Dex+PD vs VILI+H-Dex. Cl, cleaved;
Dex, dexmedetomidine; H-Dex, high dose Dex at 10 µg/kg; L-Dex, low
dose Dex at 1 µg/kg; p-, phosphorylated; PD, PD98059; t-, total;
VILI, ventilator-induced lung injury. |
To investigate whether the antiapoptotic and
anti-inflammatory effects of Dex were associated with the ERK1/2
pathway, PD98059 was administered prior to Dex treatment. Western
blot analysis showed that the ratio of p-ERK/total (t)-ERK (which
included phosphorylated and dephosphorylated ERK) significantly
increased in VILI model rats compared with those in the Sham group
(P<0.05; Fig. 5C). However,
further analysis of the results indicated that the p-ERK/t-ERK
ratio was significantly higher in the VILI+L-Dex and VILI+H-Dex
group compared with the VILI group (P<0.05 and P<0.01,
respectively; Fig. 5C). PD98059
co-treatment largely reversed the p-ERK/t-ERK ratio in the
VILI+L-Dex+PD and VILI+H-Dex+PD group compared with VILI+L-Dex and
VILI+H-Dex, respectively (both P<0.05; Fig. 5C).
Discussion
MV is a life-saving therapy for ARDS and patients
with acute respiratory failure, such as acute lung injury, and is
critical in general anesthesia (12). However, excessive ventilation can
exacerbate pre-existing lung damage and damage healthy lungs, this
process is known as VILI (22,23).
The mechanisms of VILI involve atelectrauma, barotrauma, biotrauma
and volutrauma (24), although
there are numerous factors that have been considered as possible
triggers for VILI. Based on a previous study (20), a rat VILI model using the HVT-MV
method was successfully established in the present study.
Dex has a high affinity for α2 adrenoceptor and it
is widely used in critically ill patients (at 1–1.5 µg/kg/h) owing
to its sedative effects without respiratory inhibition (25,26).
Numerous studies have also reported the anti-inflammatory effects
of Dex by reducing the expression of inflammatory factors in
certain specific cases (27–29).
Considering previous literature reports and clinical dose analysis
(11,12), two doses of Dex were examined in
the present study. The data showed that high doses of Dex at 10
µg/kg/h significantly decreased expression of the
inflammation-related cytokines IL-1β, IL-6 and TNF-α, W/D ratios
and pathological changes in the VILI model rats. Several reports
have demonstrated that Dex reduces the apoptosis of various
parenchymal and epithelial cells following inflammation or ischemia
(30–33). Similarly, H-Dex was found to
significantly reduce alveolar epithelial cell apoptosis. The
present results suggested that Dex may reduce lung injury via
suppressing inflammatory responses and cell death. These protective
effects may, in part, be due to α2 adrenoceptors.
In the present study, p-ERK1/2 expression was
significantly increased in the lung tissue exposed to VILI or
VILI+Dex. Moreover, Dex pretreatment appeared to induce more
phosphorylation of ERK1/2. In addition, VILI treatment was
demonstrated to induce a significant increase in cell death,
whereas Dex pretreatment helped to attenuate these effects.
However, when the phosphorylation of ERK1/2 was inhibited by
PD98059, the protective effects of Dex appeared to be largely
weakened. The neuroprotective role of Dex against cerebral ischemic
injury may function via phosphorylation of ERK1/2 or the MAPK/ERK
pathway (34,35). Similarly, the present study
indicated that Dex pretreatment may protect alveolar epithelial
cells against VILI-mediated effects through ERK1/2 activation.
A previous study has demonstrated that the
activation of ERK1/2 may inhibit the NF-κB signaling pathway, which
results in a reduction in the expression levels of inflammatory
factors (for example, IL-1β, IL-6 and TNF-α) and apoptotic
proteins, such as caspase-3 and Bak, thereby reducing
hypoxia-induced damage (36). The
present study results indicated that Dex reduced the expression
levels of Bak, Bcl-2 and cleaved caspase-3, and decreases the
secretion of inflammatory cytokines. However, these protective
effects also partially following with the administration of
PD98059. It has also been reported that activation of the ERK1/2
signal pathway may regulate the expression of aquaporins (37), in the present study, Dex reduced
the ratio of W/D lung tissue weight, possibly by regulating the
expression of aquaporins and activating ERK1/2. A study that
compared the gene expression characteristics of lung injury in
rodents and humans have found that conserved pathways, including
MEK1/2-ERK1/2, are potential targets against the injury pathway
(38). The MEK1/2-ERK1/2 pathway
has an important role in inflammatory responses. It can be
stimulated by signals downstream of Ras and Raf, and extracellular
stimuli, such as growth factors and cytokines (39). PD98059 binds inactive enzymes of
MEK1 and MEK2, which prevents their activation by Raf kinase,
thereby inhibiting ERK1/2 activation (40). U0126 is known to directly inhibit
the catalytic activity of active MEK1 and block MEK1/2 activity;
U0126 primarily inhibits the phosphorylation of ERK (41). This study focused on ERK1/2 in the
MEK/ERK pathway and demonstrated that PD98059 had a greater effect
compared with U0126 in preliminary experiments, thus, PD98059 was
chosen as an upstream inhibitor in this study.
In summary, the present study emphasized the
protective effects of Dex against VILI and the role of Dex in the
treatment of VILI. However, this study was still in its preliminary
stage and further in vitro and in vivo studies of
molecular mechanisms and signaling pathways are necessary to
confirm these results.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
CHZ and SQS conceptualized the study. CHZ, JY, BQW,
YN, LW and SQS performed the experiments and collected the data.
CHZ and JY analyzed the data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
All study procedures and animal handling were
approved by the Animal Review Board of Cangzhou Central Hospital
(Cangzhou, China; approval no. 2015-009-01).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ball L, Dameri M and Pelosi P: Modes of
mechanical ventilation for the operating room. Best Practice and
Research Clinical Anaesthesiology. 29. Elsevier; Amsterdam: pp.
285–299. 2015, PubMed/NCBI
|
|
2
|
Hartmann EK, Boehme S, Bentley A, Duenges
B, Klein KU, Elsaesser A, Baumgardner JE, David M and Markstaller
K: Influence of respiratory rate and end-expiratory pressure
variation on cyclic alveolar recruitment in an experimental lung
injury model. Crit Care. 16:R82012.PubMed/NCBI
|
|
3
|
Rocco PR, Dos Santos C and Pelosi P:
Pathophysiology of ventilator-associated lung injury. Curr Opin
Anaesthesiol. 25:123–130. 2012.PubMed/NCBI
|
|
4
|
Usatyuk PV and Natarajan V:
Hydroxyalkenals and oxidized phospholipids modulation of
endothelial cytoskeleton, focal adhesion and adherens junction
proteins in regulating endothelial barrier function. Microvasc Res.
83:45–55. 2012.PubMed/NCBI
|
|
5
|
Frank JA, Parsons PE and Matthay MA:
Pathogenetic significance of biological markers of
ventilator-associated lung injury in experimental and clinical
studies. Chest. 130:1906–1914. 2006.PubMed/NCBI
|
|
6
|
Scheiermann J and Klinman DM: Suppressive
oligonucleotides inhibit inflammation in a murine model of
mechanical ventilator induced lung injury. J Thorac Dis.
8:2434–2443. 2016.PubMed/NCBI
|
|
7
|
Ko YA, Yang MC, Huang HT, Hsu CM and Chen
LW: NF-κB activation in myeloid cells mediates ventilator-induced
lung injury. Respir Res. 14:692013.PubMed/NCBI
|
|
8
|
Dent BT, Aarnes TK, Wavreille VA, Lakritz
J, Lerche P, KuKanich B, Riccó Pereira CH and Bednarski RM:
Pharmacokinetics and pharmacodynamic effects of oral transmucosal
and intravenous administration of dexmedetomidine in dogs. Am J Vet
Res. 80:969–975. 2019.PubMed/NCBI
|
|
9
|
Ma D, Hossain M, Rajakumaraswamy N, Arshad
M, Sanders RD, Franks NP and Maze M: Dexmedetomidine produces its
neuroprotective effect via the alpha 2A-adrenoceptor subtype. Eur J
Pharmacol. 502:87–97. 2004.PubMed/NCBI
|
|
10
|
Taniguchi T, Kurita A, Kobayashi K,
Yamamoto K and Inaba H: Dose- and time-related effects of
dexmedetomidine on mortality and inflammatory responses to
endotoxin-induced shock in rats. J Anesth. 22:221–228.
2008.PubMed/NCBI
|
|
11
|
Chen C, Zhang Z, Chen K, Zhang F, Peng M
and Wang Y: Dexmedetomidine regulates inflammatory molecules
contributing to ventilator-induced lung injury in dogs. J Surg Res.
187:211–218. 2014.PubMed/NCBI
|
|
12
|
Chen H, Sun X, Yang X, Hou Y, Yu X, Wang
Y, Wu J, Liu D, Wang H, Yu J, et al: Dexmedetomidine reduces
ventilator-induced lung injury (VILI) by inhibiting Toll-like
receptor 4 (TLR4)/nuclear factor (NF)-κB signaling pathway. Bosn J
Basic Med Sci. 18:162–169. 2018.PubMed/NCBI
|
|
13
|
Johnson GL and Lapadat R:
Mitogen-activated protein kinase pathways mediated by ERK, JNK, and
p38 protein kinases. Science. 298:1911–1912. 2002.PubMed/NCBI
|
|
14
|
Gong G, Yuan L, Cai L, Ran M, Zhang Y,
Gong H, Dai X, Wu W and Dong H: Tetramethylpyrazine suppresses
transient oxygen-glucose deprivation-induced connexin32 expression
and cell apoptosis via the ERK1/2 and p38 MAPK pathway in cultured
hippocampal neurons. PLoS One. 9:e1059442014.PubMed/NCBI
|
|
15
|
Zhang L, Jiang B, Zhu N, Tao M, Jun Y,
Chen X, Wang Q and Luo C: Mitotic checkpoint kinase Mps1/TTK
predicts prognosis of colon cancer patients and regulates tumor
proliferation and differentiation via PKCα/ERK1/2 and PI3K/Akt
pathway. Med Oncol. 37:52019.PubMed/NCBI
|
|
16
|
Long ME, Gong KQ, Eddy WE, Volk JS,
Morrell ED, Mikacenic C, West TE, Skerrett SJ, Charron J, Liles WC,
et al: MEK1 regulates pulmonary macrophage inflammatory responses
and resolution of acute lung injury. JCI Insight.
4:e1323772019.
|
|
17
|
Goyagi T and Tobe Y: Dexmedetomidine
improves the histological and neurological outcomes 48 h after
transient spinal ischemia in rats. Brain Res. 1566:24–30.
2014.PubMed/NCBI
|
|
18
|
Sanders RD, Giombini M, Ma D, Ohashi Y,
Hossain M, Fujinaga M and Maze M: Dexmedetomidine exerts
dose-dependent age-independent antinociception but age-dependent
hypnosis in Fischer rats. Anesth Analg. 100:1295–1302.
2005.PubMed/NCBI
|
|
19
|
Gao Y, Zhou S, Wang F, Zhou Y, Sheng S, Qi
D, Huang JH, Wu E, Lv Y and Huo X: Hepatoprotective effects of limb
ischemic post-conditioning in hepatic ischemic rat model and liver
cancer patients via PI3K/ERK pathways. Int J Biol Sci.
14:2037–2050. 2018.PubMed/NCBI
|
|
20
|
Li LF, Liao SK, Ko YS, Lee CH and Quinn
DA: Hyperoxia increases ventilator-induced lung injury via
mitogen-activated protein kinases: A prospective, controlled animal
experiment. Crit Care. 11:R252007.PubMed/NCBI
|
|
21
|
Sammons MJ, Raval P, Davey PT, Rogers D,
Parsons AA and Bingham S: Carrageenan-induced thermal hyperalgesia
in the mouse: Role of nerve growth factor and the mitogen-activated
protein kinase pathway. Brain Res. 876:48–54. 2000.PubMed/NCBI
|
|
22
|
Biehl M, Kashiouris MG and Gajic O:
Ventilator-induced lung injury: Minimizing its impact in patients
with or at risk for ARDS. Respir Care. 58:927–937. 2013.PubMed/NCBI
|
|
23
|
Cortjens B, Royakkers AA, Determann RM,
van Suijlen JD, Kamphuis SS, Foppen J, de Boer A, Wieland CW,
Spronk PE, Schultz MJ, et al: Lung-protective mechanical
ventilation does not protect against acute kidney injury in
patients without lung injury at onset of mechanical ventilation. J
Crit Care. 27:261–267. 2012.PubMed/NCBI
|
|
24
|
Ye L, Zeng Q, Dai H, Zhang W, Wang X, Ma
R, Hong X, Zhao C and Pan L: Endoplasmic reticulum stress is
involved in ventilator-induced lung injury in mice via the
IRE1α-TRAF2-NF-κB pathway. Int Immunopharmacol.
78:1060692020.PubMed/NCBI
|
|
25
|
Cruickshank M, Henderson L, MacLennan G,
Fraser C, Campbell M, Blackwood B, Gordon A and Brazzelli M:
Alpha-2 agonists for sedation of mechanically ventilated adults in
intensive care units: a systematic review. Health Technol Assess.
20:v–xx, 1–117. 2016.PubMed/NCBI
|
|
26
|
Shehabi Y, Howe BD, Bellomo R, Arabi YM,
Bailey M, Bass FE, Bin Kadiman S, McArthur CJ, Murray L, Reade MC,
et al ANZICS Clinical Trials Group and the SPICE III investigators,
: Early sedation with dexmedetomidine in critically ill patients. N
Engl J Med. 380:2506–2517. 2019.PubMed/NCBI
|
|
27
|
Gao J, Sun Z, Xiao Z, Du Q, Niu X, Wang G,
Chang YW, Sun Y, Sun W, Lin A, et al: Dexmedetomidine modulates
neuroinflammation and improves outcome via alpha2-adrenergic
receptor signaling after rat spinal cord injury. Br J Anaesth.
123:827–838. 2019.PubMed/NCBI
|
|
28
|
Cheng F, Yan FF, Liu YP, Cong Y, Sun KF
and He XM: Dexmedetomidine inhibits the NF-κB pathway and NLRP3
inflammasome to attenuate papain-induced osteoarthritis in rats.
Pharm Biol. 57:649–659. 2019.PubMed/NCBI
|
|
29
|
Zhou XY, Liu J, Xu ZP, Fu Q, Wang PQ and
Zhang HA: Dexmedetomidine inhibits the
lipopolysaccharide-stimulated inflammatory response in microglia
through the pathway involving TLR4 and NF-κB. Kaohsiung J Med Sci.
35:750–756. 2019.PubMed/NCBI
|
|
30
|
Xie Y, Guo C, Liu Y, Shi L and Yu J:
Dexmedetomidine activates the PI3K/Akt pathway to inhibit
hepatocyte apoptosis in rats with obstructive jaundice. Exp Ther
Med. 18:4461–4466. 2019.PubMed/NCBI
|
|
31
|
Zhai M, Liu C, Li Y, Zhang P, Yu Z, Zhu H,
Zhang L, Zhang Q and Wang J and Wang J: Dexmedetomidine inhibits
neuronal apoptosis by inducing Sigma-1 receptor signaling in
cerebral ischemia-reperfusion injury. Aging (Albany NY).
11:9556–9568. 2019.PubMed/NCBI
|
|
32
|
Sun J, Zheng S, Yang N, Chen B, He G and
Zhu T: Dexmedetomidine inhibits apoptosis and expression of COX-2
induced by lipopolysaccharide in primary human alveolar epithelial
type 2 cells. Biochem Biophys Res Commun. 517:89–95.
2019.PubMed/NCBI
|
|
33
|
Cui J, Zhao H, Wang C, Sun JJ, Lu K and Ma
D: Dexmedetomidine attenuates oxidative stress induced lung
alveolar epithelial cell apoptosis in vitro. Oxid Med Cell Longev.
2015:3583962015.PubMed/NCBI
|
|
34
|
Shi Y, Peng XH, Li X, Luo GP and Wu MF:
Neuroprotective role of dexmedetomidine pretreatment in cerebral
ischemia injury via ADRA2A-mediated phosphorylation of ERK1/2 in
adult rats. Exp Ther Med. 16:5201–5209. 2018.PubMed/NCBI
|
|
35
|
Qiu Z, Lu P, Wang K, Zhao X, Li Q, Wen J,
Zhang H, Li R, Wei H, Lv Y, et al: Dexmedetomidine inhibits
neuroinflammation by altering microglial M1/M2 polarization through
MAPK/ERK pathway. Neurochem Res. 45:345–353. 2020.PubMed/NCBI
|
|
36
|
Jin H and Yu J: Lidocaine protects H9c2
cells from hypoxia-induced injury through regulation of the
MAPK/ERK/NF-κB signaling pathway. Exp Ther Med. 18:4125–4131.
2019.PubMed/NCBI
|
|
37
|
Shen Q, Ma X, Hua Y, Chen M, Wang Y, Zhou
Q, Ye W and Zhu X: Aquaporin 3 expression induced by salvia
miltiorrhiza via ERK1/2 signal pathway in the primary human amnion
epithelium cells from isolated oligohydramnios. Curr Mol Med.
16:312–319. 2016.PubMed/NCBI
|
|
38
|
Sweeney TE, Lofgren S, Khatri P and Rogers
AJ: Gene expression analysis to assess the relevance of rodent
models to human lung injury. Am J Respir Cell Mol Biol. 57:184–192.
2017.PubMed/NCBI
|
|
39
|
Caunt CJ, Sale MJ, Smith PD and Cook SJ:
MEK1 and MEK2 inhibitors and cancer therapy: The long and winding
road. Nat Rev Cancer. 15:577–592. 2015.PubMed/NCBI
|
|
40
|
Dudley DT, Pang L, Decker SJ, Bridges AJ
and Saltiel AR: A synthetic inhibitor of the mitogen-activated
protein kinase cascade. Proc Natl Acad Sci USA. 92:7686–7689.
1995.PubMed/NCBI
|
|
41
|
Chen Y, Zhang H, Liu E, Xu CB and Zhang Y:
Homocysteine regulates endothelin type B receptors in vascular
smooth muscle cells. Vascul Pharmacol. 87:100–109. 2016.PubMed/NCBI
|