Introduction
Cisplatin is a radiation sensitizer and cytotoxic
agent commonly used in cancer therapy. Cisplatin-induced
sensorineural hearing loss (SHL) is especially prevalent among
patients with brain tumors, head and neck cancer, and
nasopharyngeal carcinoma (1–3). The
incidence of low- and high-frequency SHL ranges from 10–97% in
patients who receive cisplatin-based chemoradiation (2–4).
Despite recent advances in treatments for cisplatin-induced SHL,
the prevalence of cisplatin-induced cytotoxicity and damage in
cochlear hair cells remains high (3). The primary cisplatin accumulation
site is the cochlea, where cisplatin can be retained for months, or
even years (1). As a result, the
time from cisplatin chemotherapy to SHL onset also ranges from
months to years (5).
Cisplatin-induced SHL is mechanistically related to
enhanced apoptosis and DNA damage (6,7). It
has also been reported that reactive oxygen species (ROS), as well
as Wnt and p53 signaling pathways, are activated in the cochlea
following cisplatin therapy (8–10).
Wnt activation protects against cytotoxicity in cochlear hair cells
(9). Moreover, abnormal expression
of genes and non-coding RNAs, such as microRNAs, is also associated
with SHL (11).
Several drugs with antagonistic effects against
cisplatin--induced cytotoxicity, DNA damage, and apoptosis in
cochlear hair cells might represent potential treatment strategies
for cisplatin-induced ototoxicity (10,12–14).
For instance, tetramethylpyrazine (Tet) and tanshinone IIA (Tan
IIA) can protect against hearing impairment and ototoxicity induced
by aminoglycoside antibiotics, cisplatin, and radiation (15–17).
Tet can also decrease caspase-3 expression in spiral ganglion and
apoptosis in guinea pig cochlea (15,17).
Tan IIA has been reported to protect House Ear Institute-Organ of
Corti 1 (HEI-OC1) auditory cells from cisplatin-induced ototoxicity
and may synergize with cisplatin, enhancing cytotoxicity against
cancer cells by promoting apoptosis and cell cycle arrest at the S
phase (18).
The molecular mechanisms underlying the protective
effects of Tet and Tan IIA against cisplatin-induced ototoxicity
are poorly understood. Therefore, the aim of the present study was
to determine the mechanistic basis of this otoprotective effect
in vitro. HEI-OC1 auditory cells were exposed to cisplatin
and treated with Tet or Tan IIA. Tet, but not Tan IIA, reversed the
inhibitory effect of cisplatin on HEU-OC1 viability. The underlying
molecular mechanisms were investigated using high-throughput,
next-generation sequencing, and bioinformatics analysis. The
findings of the present study provided insight into
cisplatin-induced ototoxicity in vitro and the Tet-mediated
protective effects against it.
Materials and methods
Cell culture
HEI-OC1 auditory cell line was a gift from Professor
Federico Kalinec (David Geffen School of Medicine at UCLA, CA, USA)
and maintained at 32°C in 10% CO2 in high-glucose
Dulbecco's modified Eagle's medium (DMEM; HyClone; GE Healthcare
Life Sciences) supplemented with 2 mM L-glutamine, 1 mM sodium
pyruvate and 10% fetal bovine serum (FBS; HyClone).
Cell treatments
HEI-OC1 auditory cells were separately treated for
48 h at 32°C in 10% CO2 with Tan IIA (3, 9, 27, 81, 243
or 729 mg/l; Shanghai Yuanye Biotechnology Co., Ltd.), Tet (125,
250, 500, 1×103, 2×103, 4 103,
8×103 or 1.60×104 mg/l; Shanghai Aladdin
Bio-Chem Technology Co., Ltd.) or cisplatin (5, 10, 20, 40, 80 and
160 µM; Sigma-Aldrich; Merck KGaA). In a separate experiment, the
cells were treated for 30 h with 30 µM cisplatin combined either
with Tan IIA (0.2, 0.5, 1.0, or 1.5 mg/l) or with Tet (37.5, 75,
125 or 250 mg/l) at 32°C in 10% CO2. The assays were
carried out in triplicate wells.
Cell viability assay
Cells were seeded into 96-well plates (Corning,
Inc.) at a density of 5×104 cells/ml and maintained in
high-glucose DMEM for 24 h. The culture medium was then replaced
with fresh medium supplemented with Tan IIA, Tet, cisplatin or
combinations as aforementioned. Cell viability was then assessed
using Cell Counting Kit-8 (MedChemExpress) according to the
manufacturer's instructions. The optical density was read at 450 nm
using a microplate reader (Bio-Rad Laboratories, Inc.).
Cell apoptosis assay
Cell apoptosis was measured using Annexin
V-fluorescein isothiocyanate (FITC) and propidium iodide (PI;
Beyotime Institute of Biotechnology) staining. Cells
(5×104 cells/well) were plated in 6-well plates, then
treated in triplicate with cisplatin for 48 h or with cisplatin and
Tet for 30 h at 32°C in 10% CO2. Following incubation,
cells were collected by centrifugation at 4°C and 1,500 × g for 5
min, then resuspended in 1X binding buffer. The samples were then
stained with 5 µl of Annexin-V-FITC solution and 5 µl of PI at 4°C
for 10–15 min in dark. The data were acquired using a BD
FACSCanto-II flow cytometer (BD Biosciences) and analyzed using an
Olympus BX51 fluorescence microscope (Olympus Optical, Tokyo,
Japan).
Western blot analysis
Proteins were extracted using RIPA lysis buffer
(Beyotime Institute of Biotechnology) according to the
manufacturer's instructions. Protein concentration was determined
using a Bradford protein assay kit (Thermo Fisher Scientific Inc.).
The samples (20 µg) were resolved by SDS-PAGE on 10% gels (Shanghai
Sangong Pharmaceutical Co., Ltd.), then transferred to PVDF
membranes (EMD Millipore). The membranes were then blocked with 5%
nonfat milk (Beyotime Institute of Biotechnology) at room
temperature for 1 h. The membranes were first incubated with
target-specific primary antibodies, including anti-P21 (1:1,000;
cat. no. ab188224; Abcam), anti-p16-INK4A (1:1,000; cat. no.
ab211542; Abcam), anti-FOXO3 (1:1,500; cat. no. 2497S; Cell
Signaling Technology, Inc.), anti-caspase 3 (1:1,000; cat. no.
ab13847; Abcam), anti-Fas (1:2,000; cat. no. ab216991; Abcam),
anti-Wnt receptor frizzled 6 (FZD6; 1:2,000; cat. no. DF4930;
Affinity Biosciences), anti-transcriptional repressor transcription
factor 7-like 1 (TCF7L1; 1:1,500; cat. no. ab86175; Abcam),
anti-wingless-type MMTV integration site family, member 2 (WNT2;
1:800; cat. no. ab109222; Abcam), anti-insulin-like growth factor 1
(IGF1; 1:1,500; cat. no. ab182408; Abcam), anti-SERPINE1 (1:1500;
cat. no. DF13553; Affinity Biosciences) and anti-GAPDH (1:10,000;
cat. no. KC-5G5; Shanghai Kangcheng Biotechnology Co. Ltd.) at 4°C
overnight. The membranes were then incubated with horseradish
peroxidase-conjugated secondary antibody (anti-rabbit IgG;
1:20,000; cat. no. ba1054; Boster Biological Technology, Ltd.) at
room temperature for 40 min. Lastly, protein bands were visualized
using an ECL kit (EMD Millipore).
RNA extraction and whole transcriptome
sequencing
Total RNA was extracted using the TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) at 48 and 30 h
post-treatment. RNA concentration and purity were determined using
a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, Inc.)
and an Agilent 2100 Bioanalyzer (Agilent Technologies Inc.). RNA
samples with an RNA integrity number (RIN) value of > eight were
used for library preparation. RNA was fragmented, reverse
transcribed to the first-strand cDNA using reverse transcriptase
(Invitrogen; Thermo Fisher Scientific, Inc.) and random hexamers
(Sangon Biotech Co., Ltd.), then synthesized to double-stranded DNA
using dNTPs (Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C
for 15 min and at 98°C for 5 min. The DNA sequencing libraries were
prepared using a TruSeq™ RNA Sample Preparation Kit (version 2;
Illumina, cat. nos. RS-122-2001 RS-122-2002) following the
protocols including DNA fragment end repair, adenylating, adapter
ligation, and fragment amplification. After verifying the library
quality using the Qubit2.0 (Thermo Fisher Scientific, Inc.),
Agilent 2100 (Agilent Technologies Inc.), and qPCR, sequencing
(loading concentration 3 nM/µl) was carried out using a V1
sequencing kit (Illumina, Inc.) on an Illumina HiSeq 4000
sequencing platform (paired-end; 150 bp; Illumina, Inc.).
Data processing
Base calling of the original sequencing image data
and fastq file extraction were carried out using Illumina CASAVA
software (version 1.8.2; Illumina, Inc.). The quality of raw data
was checked using the FastQC program (version 0.11.5; http://www.bioinformatics.babraham.ac.uk/projects/fastqc/).
After adaptor trimming and removal of duplicated and low-quality
reads (unknown base sequence >10%, and >50% of the read
consisted of reads with Phred scores of ≤3) using Trimmomatic
(version 0.30; http://www.usadellab.org/cms/?page=trimmomatic), the
clean reads were mapped to the ENSEMBL Mus musculus
GRCm38.p6 reference genome (http://www.ensembl.org/) with annotation version
GRCm38.97 using the TopHat 2 software (http://ccb.jhu.edu/software/tophat/index.shtml).
Transcript assembly and quantification was performed
using the Cufflinks software (version 2.2.1; http://cufflinks.cbcb.umd.edu/) by calculating the
expected number of fragments per kilobase of exon model per million
reads mapped (FPKM) values in each sample. Differentially expressed
genes (DEGs) between groups were identified using the edgeR
statistical software package (Bioconductor; http://www.bioconductor.org/) according to the
criteria of FDR <0.05 and |log2(FC)|≥1, where FDR is
the false discovery rate and FC is the fold change.
Enrichment analysis
Gene Ontology (GO; http://www.geneontology.org/) biological processes and
Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways associated
with the DEGs were identified from the Database for Annotation,
Visualization, and Integrated Discovery (DAVID; version 6.7;
http://david.ncifcrf.gov/). P<0.05 was used to
identify statistically significant terms.
Protein-protein interaction (PPI)
network analysis
The interaction between DEGs was predicted and
extracted from STRING (version 10; www.string-db.org). PPI networks were constructed and
visualized using Cytoscape (version 2.8; http://www.cytoscape.org/).
Statistical analysis
Quantitative data are presented as the mean ± SD of
triplicates. Statistical analysis was carried out using GraphPad
Prism software (version 6; GraphPad Software, Inc.). Multigroup
comparisons were analyzed using one-way ANOVA followed by Tukey's
post hoc test. The IC50 values of Tan IIA, Tet, and
cisplatin were calculated using nonlinear regression. P<0.05 was
considered to indicate a statistically significant difference.
Results
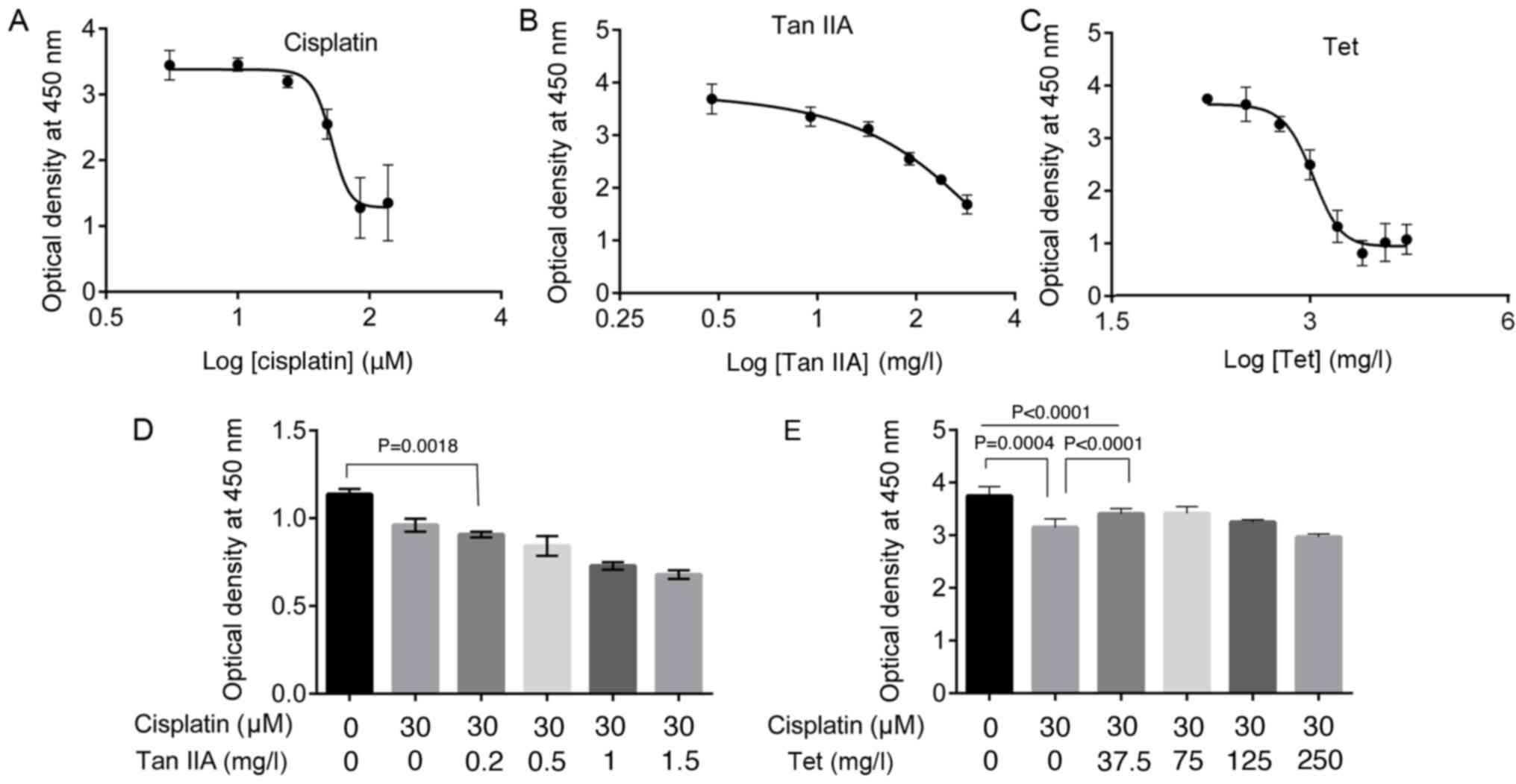
Cisplatin, Tan IIA, and Tet inhibit
HEI-OC1 auditory cell viability
Treatment with cisplatin, Tan IIA, and Tet for 48 h
inhibited the viability of HEI-OC1 auditory cells in a
dose-dependent manner (Fig. 1A-C),
with IC50 values of 42.89 µM, 151.80 and
1.04×103 mg/l, respectively.
Low Tet concentration prevents
cisplatin-induced cytotoxicity in HEI-OC1 auditory cells
To investigate whether Tan IIA and Tet
administration could suppress the inhibitory effect of cisplatin on
HEI-OC1 auditory cell viability, cells were treated with 30 µM
cisplatin combined with Tan IIA (0.2, 0.5, 1.0 and 1.5 mg/l) or Tet
(37.5, 75, 125 and 250 mg/l) for 30 h. To minimize drug-related
cytotoxicity, the selected concentrations were <IC50
values. The addition of Tan IIA at concentrations <1.5 mg/l
augmented the effect of cisplatin and further inhibited viability
significantly compared with cisplatin alone (P=0.0018, Fig. 1D). However, at concentrations of
37.5 and 75 mg/l Tet significantly increased viability, compared
with cisplatin alone (P<0.0001, Fig. 1E).
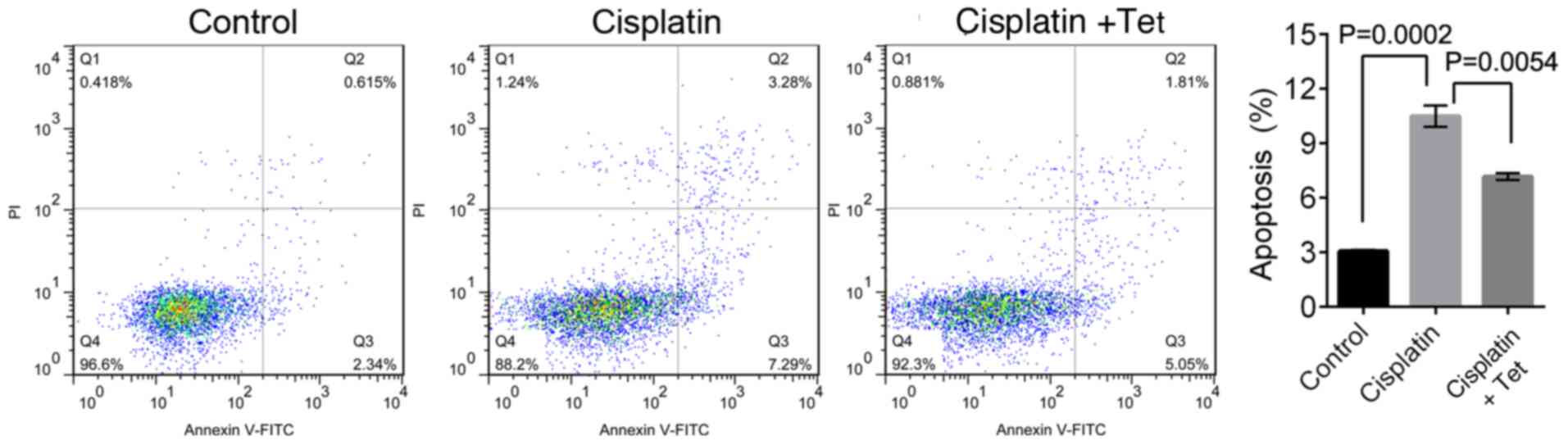
The percentage of apoptotic HEI-OC1 auditory cells
was significantly increased following cisplatin treatment, compared
with control cells (10.27±0.64% and 3.11±0.17% in the second (upper
right) and third (lower right) quadrants, respectively; P=0.0002;
Fig. 2). However, co-treatment
with cisplatin and Tet reduced the apoptosis rate to 7.18±0.33%
(P=0.0054). Collectively, these results indicated that low
concentrations of Tet could protect HEI-OC1 cells against
cisplatin-induced cytotoxicity.
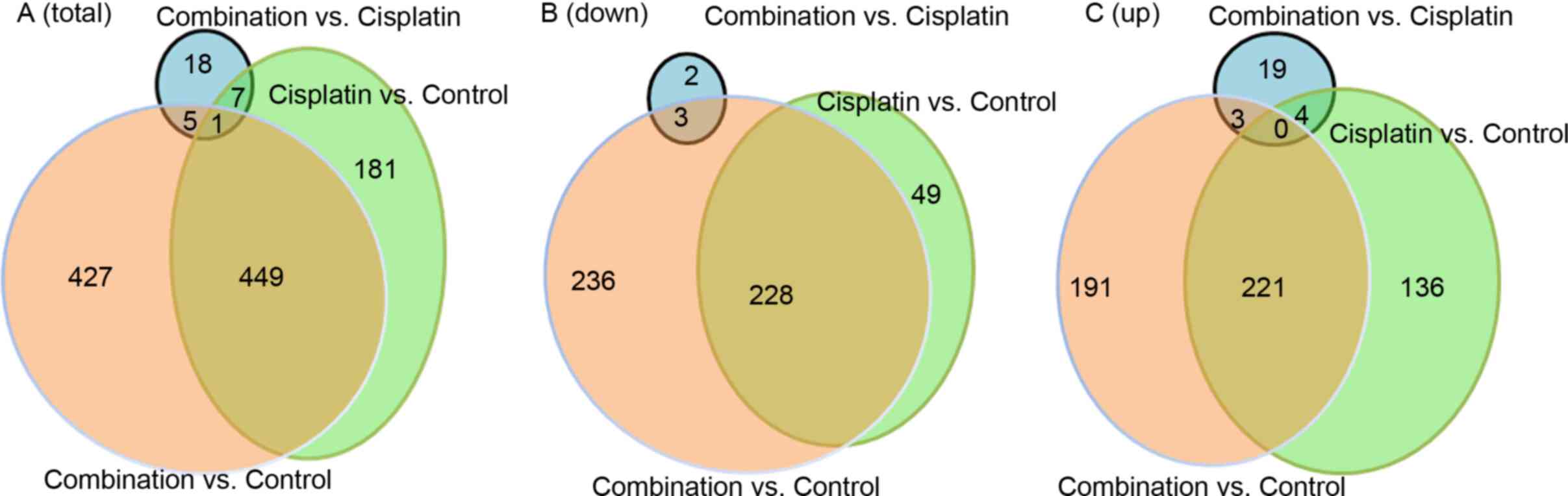
Summary of Illumina sequencing
To examine the molecular mechanisms associated with
the otoprotective properties of Tet, RNA sequencing (RNA-seq)
analysis was carried out on HEI-OC1 auditory cells treated with 30
µM cisplatin alone or combined with 37.5 mg/l Tet. Illumina
sequencing produced a total of 518.83 million raw reads,
corresponding to 510.65 million clean reads and 74.31 Gbp (Table SI). The average GC content was
50.26%, while the average frequency of reads >Q30 was 95.91%.
Transcriptome assembly generated 1,455,306 transcripts. In
comparison with control cells, 638 DEGs were identified following
cisplatin treatment, including 361 upregulated genes and 277
downregulated genes (Fig. 3A-C).
Moreover, 882 DEGs were observed in cells treated with a
combination of cisplatin and Tet, including 415 upregulated and 467
downregulated genes (Fig. 3B and
C). Thus, relative to untreated cells, the combination of
cisplatin and Tet induced more DEGs than cisplatin alone. A total
of 449 DEGs were shared between the two datasets, including 221
upregulated and 228 downregulated DEGs (Fig. 3B and C). In comparison with
cisplatin alone, the combination with Tet (37.5 mg/l) only induced
31 DEGs, including 26 upregulated and 5 downregulated DEGs). In
total, 1,088 DEGs were identified, including 626 non-overlapping
DEGs (Fig. 3A-C).
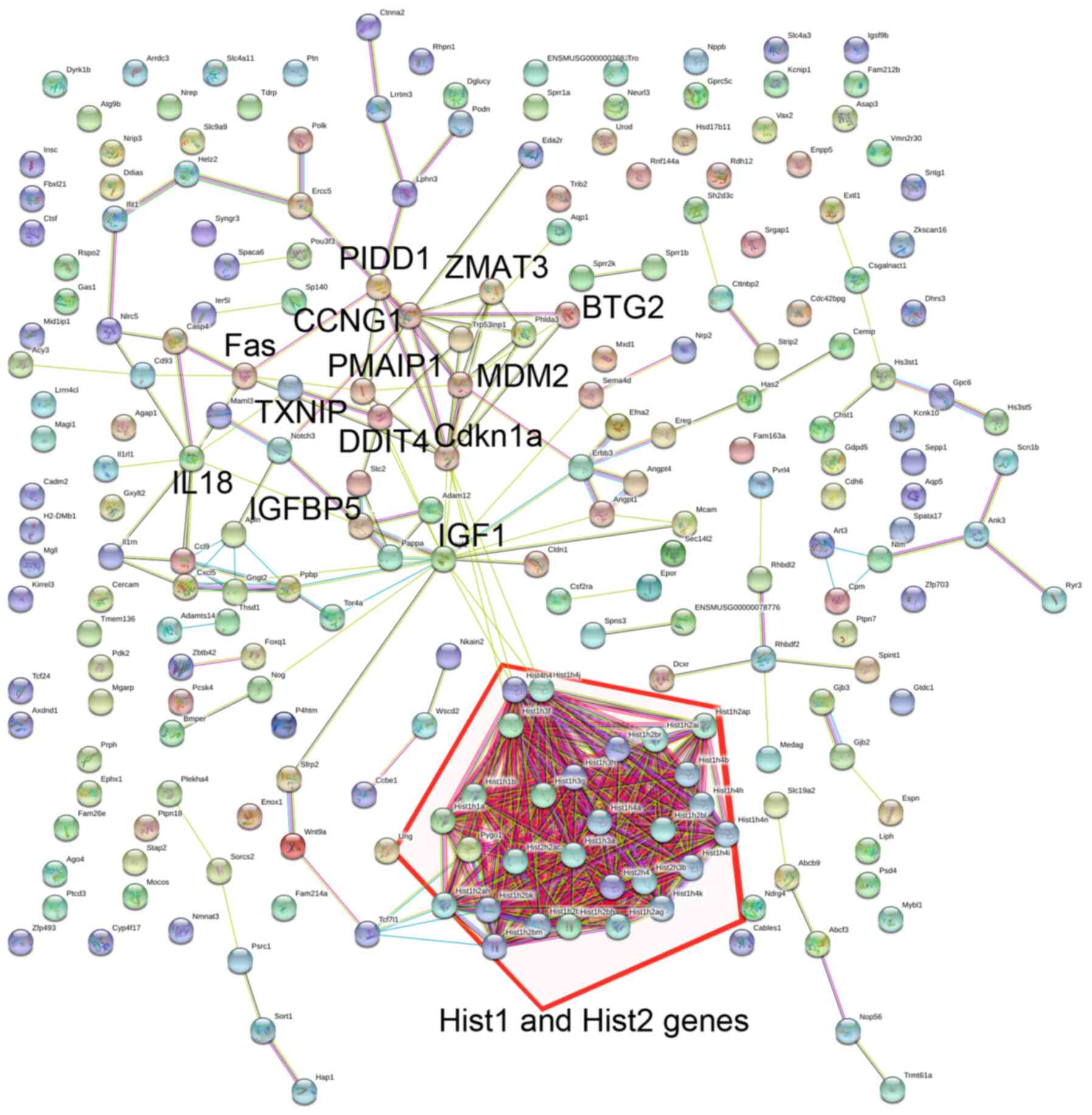
Summary and annotation of
cisplatin-induced DEGs
To examine the molecular mechanisms underlying
cisplatin-induced cytotoxicity, the biological processes and
pathways associated with the identified DEGs were examined using GO
and KEGG analysis. GO functional enrichment analysis indicated that
downregulated DEGs, such as histone (Hist)1 and Hist2 gene clusters
(Fig. 4), were associated with
biological processes related to ‘DNA replication-dependent
nucleosome assembly’, ‘nucleosome assembly’, and ‘DNA-templated
transcription, initiation’ (Table
I). Other downregulated DEGs were also associated with
biological processes, including ‘negative regulation of apoptotic
process’ (thrombospondin1, THBS1; TWIST2; insulin-1 receptor
antagonist gene, IL1RN; IGF1) and regulation of cell migration
(IL1RN; SERPINE1; IGF binding protein 5, IGFBP5).
 | Table I.Top 15 GO biological processes
associated with the differentially expressed genes induced by
cisplatin vs. control. |
Table I.
Top 15 GO biological processes
associated with the differentially expressed genes induced by
cisplatin vs. control.
| A, Downregulated
genes |
|---|
|
|---|
| Term | Count | P-value | Gene name |
|---|
| GO:0006334,
nucleosome assembly | 24 |
3.3010−22 | HIST2H3B,
HIST1H2BB, HIST1H1C, HIST1H1B, H1F0, ANP32B, H3F3B, HIST1H3F,
HIST1H4I, HIST1H3G, etc. |
| GO:0000183,
chromatin silencing at rDNA | 16 |
2.22×10−16 | HIST2H3B,
HIST1H2BB, HIST1H1C, HIST1H1B, BEND3, POLR1B, HIST2H4, SAP30, UBTF,
HIST1H4A, etc. |
| GO:0032776, DNA
methylation on cytosine | 12 |
1.37×10−13 | HIST2H3B, HIST4H4,
HIST1H4A, HIST1H4B, HIST1H3A, H3F3B, HIST1H3F, HIST1H4I, HIST1H3G,
HIST1H4J, etc. |
| GO:0045815,
positive regulation of gene expression, epigenetic | 12 |
2.03×10−13 | HIST2H3B, HIST4H4,
HIST1H4A, HIST1H4B, HIST1H3A, H3F3B, HIST1H3F, HIST1H4I, HIST1H3G,
HIST1H4J, etc. |
| GO:0006335, DNA
replication-dependent nucleosome assembly | 11 |
7.75×10−12 | HIST2H3B, HIST4H4,
HIST1H4A, HIST1H4B, HIST1H3A, HIST1H3F, HIST1H4I, HIST1H3G,
HIST1H4J, HIST1H4H, etc. |
| GO:0051290, protein
heterotetramerization | 11 |
3.70×10−10 | HIST2H3B, HIST4H4,
HIST1H4A, HIST1H4B, HIST1H3A, HIST1H3F, HIST1H4I, HIST1H3G,
HIST1H4J, HIST1H4H, etc. |
| GO:0006336, DNA
replication-inde pendent nucleosome assembly | 8 |
2.19×10−8 | HIST4H4, HIST1H4A,
HIST1H4B, H3F3B, HIST1H4I, HIST1H4J, HIST1H4H, HIST2H4 |
| GO:0045653,
negative regulation of megakaryocyte differentiation | 7 |
4.84×10−8 | HIST4H4, HIST1H4A,
HIST1H4B, HIST1H4I, HIST1H4J, HIST1H4H, HIST2H4 |
| GO:0001649,
osteoblast differentiation | 12 |
2.46×10−7 | NOG, IGF1, COL6A1,
TWIST2, IGFBP5, SPP1, FHL2, GJA1, H3F3B, MYBBP1A, etc. |
| GO:0043066,
negative regulation of apoptotic process | 25 |
3.61×10−7 | TWIST2, THBS1,
IL1RN, IGF1, GAS1, SERPINB9, PDGFRB, YBX3, FHL2, AQP1, etc. |
| GO:0001701, in
utero embryonic development | 17 |
1.89×10−6 | NOG, EDN1, YBX3,
TPM1, HES1, PDGFRB, ANGPT1, PTCH1, GJB3, GJA1, etc. |
| GO:0030324, lung
development | 11 |
4.89×10−6 | HES1, MIR92-1,
MIR17, MIR18, CCBE1, MIR20A, PTN, IGF1, LOX, MIR19B-1, etc. |
| GO:0006352,
DNA-templated transcription, initiation | 7 |
7.32×10−6 | HIST4H4, HIST1H4A,
HIST1H4B, HIST1H4I, HIST1H4J, HIST1H4H, HIST2H4 |
| GO:0030336,
negative regulation of cell migration | 10 |
9.64×10−6 | NOG, PDGFB, SFRP2,
IL1RN, SERPINE1, PTN, CNN2, TPM1, SRGAP1, IGFBP5 |
| GO:0030335,
positive regulation of cell migration | 13 |
1.38×10−5 | PDGFB, EDN1, IGF1,
AQP1, CXCL12, THBS1, IRS1, CEMIP, SEMA3C, PDGFRB, etc. |
|
| B, Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| GO:0006914,
autophagy | 9 |
1.97×10−4 | TRP53INP1, ATG9B,
ATG12, ATG2A, SESN2, HAP1, ABARAPL1, OPTN, ARSA |
| GO:0006915,
apoptotic process | 18 |
2.35×10−4 | ZMAT3, FOXO3,
PMAIP1, TRP53INP1, BBC3, PIDD1, FAS, DDIAS, DDIT4, PHLDA3,
etc. |
| GO:0072332,
intrinsic apoptotic signaling pathway by p53 class mediator | 5 |
3.76×10−4 | PDK2, BBC3, ZMAT3,
EDA2R, PMAIP1 |
| GO:0042771,
intrinsic apoptotic signaling pathway in response to DNA damage by
p53 class mediator | 5 |
3.76×10−4 | CDKN1A, BBC3,
PMAIP1, PHLDA3, DDIT4 |
| GO:0006919,
activation of cysteine-type endopeptidase activity involved in
apoptotic process | 6 |
9.12×10−4 | TNFRSF10B, NOD1,
BBC3, PMAIP1, APAF1, PIDD1 |
| GO:0006974,
cellular response to DNA damage stimulus | 14 |
9.13×10−4 | CDKN1A, BBC3,
TRP53INP1, PIDD1, PMAIP1, DDIAS, BTG2, ZMAT3, ERCC5, POLK,
etc. |
| GO:0000045,
autophagosome assembly | 5 |
1.45×10−3 | ATG9B, GABARAPL1,
ATG12, ATG2A, TRP53INP1 |
| GO:0048536, spleen
development | 5 |
1.7×10−3 | RIPK3, NFKB2, FAS,
CTC1, RC3H1 |
| GO:0007050, cell
cycle arrest | 6 |
2.13×10−3 | CDKN1A, AK1,
RASSF1, WDR6, TRP53INP1, DDIAS |
| GO:0016236,
macroautophagy | 4 |
2.34×10−3 | ATG12, NBR1, OPTN,
ZFYVE1 |
| GO:0051607, defense
response to virus | 8 |
2.75×10−3 | DDX58, NLRC5,
IFIT1, TMEM173, PMAIP1, EIF2AK2, CXCL10, DDIT4 |
| GO:0006977, DNA
damage response, signal transduction by p53 class mediator
resulting in cell cycle arrest | 3 |
4.25×10−3 | CDKN1A, MDM2,
PIDD1 |
| GO:0043029, T cell
homeostasis | 4 |
4.90×10−3 | RIPK3, PMAIP1, FAS,
RC3H1 |
| GO:0000422,
mitophagy | 4 |
6.37×10−3 | ATG9B, GABARAPL1,
ATG12, ATG2A |
| GO:0070059,
intrinsic apoptotic signaling pathway in response to endoplasmic
reticulum stress | 4 |
7.48×10−3 | TNFRSF10B, BBC3,
PMAIP1, APAF1 |
Moreover, the downregulated DEGs identified
following cisplatin treatment were also associated with several
KEGG pathways. Genes in the Hist1 and Hist2 clusters were
associated with ‘Systemic lupus erythematosus’, ‘Alcoholism’ and
‘Viral carcinogenesis’ (Table
II). IGF1 and THBS1 were involved in ‘PI3K-Akt signaling
pathway’, ‘Rap1 signaling pathway’, and ‘HIF-1 signaling
pathway’.
 | Table II.Kyoto Encyclopedia of Genes and
Genomes pathways associated with the differentially expressed genes
induced by cisplatin vs. control. |
Table II.
Kyoto Encyclopedia of Genes and
Genomes pathways associated with the differentially expressed genes
induced by cisplatin vs. control.
| A, Downregulated
genes |
|---|
|
|---|
| Term | Count | P-value | Gene name |
|---|
| mmu05322, Systemic
lupus erythematosus | 26 |
7.48×10−19 | HIST1H2AF,
HIST1H2AG, HIST1H2BM, HIST1H4A, HIST1H2BK, HIST1H4B, HIST1H2BL,
HIST2H2AC, HIST1H2BJ, HIST3H2A, etc. |
| mmu05034,
Alcoholism | 26 |
2.04×10−15 | HIST1H4A,
HIST1H2BK, HIST1H4B, HIST1H2BL, HIST2H2AC, HIST1H2BJ, HIST3H2A,
H2AFX, HIST1H4I, HIST1H4J, etc. |
| mmu05203, Viral
carcinogenesis | 15 |
2.98×10−5 | HIST1H2BB, HIST4H4,
HIST1H2BH, HIST2H4, HIST1H2BM, MRPS18B, HIST1H4A, HIST1H2BK,
HIST1H4B, HIST1H2BL, etc. |
| mmu04510, Focal
adhesion | 11 |
2.43×10−3 | PDGFB, TLN2,
COL1A2, PDGFRB, IGF1, THBS1, COL5A3, SPP1, PDGFC, COL3A1, SPP1,
etc. |
| mmu05202,
Transcriptional misregulation in cancer | 9 |
6.42×10−3 | MAF, HIST2H3B,
HIST1H3A, IGF1, PBX1, HIST1H3G, ID2, H3F3B, HIST1H3F, |
| mmu05206, MicroRNAs
in cancer | 12 |
6.50×10−3 | PDGFB, PDGFRB,
MARCKS, THBS1, IRS1, TPM1, MIR17, MIR18, MIR20A, etc. |
| mmu04151, PI3K-Akt
signaling pathway | 13 |
1.45×10−2 | PDGFB, PDGFRB,
IGF1, ANGPT1, THBS1, IRS1, ANGPT4, SPP1, COL5A3, COL3A1, etc. |
| mmu04512,
ECM-receptor interaction | 6 |
1.59×10−2 | COL3A1, COL1A2,
COL6A1, THBS1, COL5A3, SPP1 |
| mmu04015, Rap1
signaling pathway | 9 |
2.72×10−2 | PDGFB, TLN2, IGF1,
ANGPT1, THBS1, ANGPT4, MAGI1, PDGFRB, PDGFC |
| mmu04066, HIF-1
signaling pathway | 6 |
2.81×10−2 | EDN1, SERPINE1,
HK2, IGF1, ANGPT1, ANGPT4 |
| mmu04330, Notch
signaling pathway | 4 |
4.86×10−2 | DTX4, HES1, MAML3,
LFNG |
|
| B, Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| mmu04115, p53
signaling pathway | 12 |
1.77×10−10 | CDKN1A, BBC3,
ZMAT3, MDM2, PMAIP1, FAS, CCNG1, PIDD1, SESN2, CCNG2, etc. |
| mmu05169,
Epstein-Barr virus infection | 8 |
8.93×10−4 | DDX58, CDKN1A,
NFKBIE, RELB, NFKBIA, MDM2, NFKB2, EIF2AK2 |
| mmu05164, Influenza
A | 8 |
3.32×10−3 | DDX58, TNFRSF10B,
NFKBIA, H2-DMB1, FAS, EIF2AK2, NXT2, CXCL10 |
| mmu04068, FoxO
signaling pathway | 7 |
4.21×10−3 | CDKN1A, GABARAPL1,
ATG12, FBXO25, MDM2, FOXO3, CCNG2 |
| mmu04623, Cytosolic
DNA-sensing pathway | 5 |
6.03×10−3 | DDX58, TMEM173,
RIPK3, NFKBIA, CXCL10 |
| mmu04622,
RIG-I-like receptor signaling pathway | 5 |
7.47×10−3 | DDX58, TMEM173,
ATG12, NFKBIA, CXCL10 |
| mmu04142,
Lysosome | 6 |
1.27×10−2 | ABCB9, AP3M2,
SMPD1, ARSA, SORT1, CTSF |
| mmu05162,
Measles | 6 |
1.95×10−2 | DDX58, TNFRSF10B,
BBC3, NFKBIA, FAS, EIF2AK2 |
| mmu04064 NF-kappa B
signaling pathway | 5 |
2.48×10−2 | DDX58, RELB,
NFKBIA, NFKB2, PIDD1 |
| mmu04210,
Apoptosis | 4 |
3.10×10−2 | TNFRSF10B, NFKBIA,
FAS, APAF1 |
| mmu05206, MicroRNAs
in cancer | 8 |
3.82×10−2 | NOTCH3, CDKN1A,
RASSF1, MDM2, CCNG1, DDIT4, PDCD4, MIR34A |
| mmu05203, Viral
carcinogenesis | 7 |
4.85×10−2 | CDKN1A, KAT2B,
NFKBIA, MDM2, PMAIP1, NFKB2, EIF2AK2 |
Notably, the upregulated DEGs induced by cisplatin
in HEI-OC1 auditory cells were significantly associated with
autophagy (autophagy-related gene 12, ATG12; ATG9B; ATG2A),
apoptosis-associated processes, response to DNA damage and cell
cycle arrest. These genes included
phorbol-12-myristate-13-acetate-induced protein 1 (PMAIP1), Bcl-2
binding component 3 (BBC3), zinc finger matrin-type 3 (ZMAT3),
p53-induced death domain protein 1 (PIDD1), B-cell translocation
gene protein 2 (BTG2), thioredoxin-interacting protein (TNXIP), DNA
damage induced apoptosis suppressor (DDIAS), cycle-dependent kinase
inhibitor 1A (CDKN1A), transformation related protein 53-induced
nuclear protein 1 (TRP53INP1), FOXO3, and Fas (Table I). These DEGs interacted closely
(Fig. 4) and some (including
CDKN1A, BBC3, ZMAT3, PMAIP1, FAS, PIDD1, and FOXO3) were
significantly associated with ‘p53 signaling pathway’ and ‘FoxO
signaling pathway’ (Table
II).
Common effect of Tet and cisplatin on
transcriptome expression profiles
As indicated in Fig.
3, 450 DEGs were shared between the two treatments. The shared
DEGs included Fas, PMAIP1, ATG12, CDKN1A, and ZMAT3, which were
upregulated, and THBS1 and SERPINE1, which were downregulated
(Fig. 5A-D). Enrichment analysis
showed these shared genes enriched in similar functional categories
and KEGG pathways as DEGs induced by cisplatin alone (Tables III and IV). The upregulated genes including
THBS1, CDKN1A, Fas, PMAIP1, TXNIP, and ZMAT3 were associated with
biological processes, such as ‘programmed cell death’ and ‘cell
cycle’ (Table III). Genes
including CDKN1A, Fas, PMAIP1, TXNIP, and ZMAT3 and pathways
including ‘p53 signaling pathway’ and/or ‘Apoptosis’ (Table IV). The downregulated genes
including the histone genes, endothelin 1 (EDN1), suppressor of
cytokine signaling 3 (SOCS3), and insulin receptor substrate 1
(IRS1) were associated with biological processes, such as
‘nucleosome assembly’ and ‘regulation of phosphate metabolic
process’ (Table III). The
downregulated DEGs including THBS1, collagen type III alpha 1 chain
(COL3A1), and platelet derived growth factor subunit B (PDGFB) were
involved in ‘ECM-receptor interaction’ and ‘Focal adhesion’
(Table IV).
 | Table III.The top 20 biological processes
associated with the differentially expressed genes overlapping
between cisplatin vs. control and combination vs. control
groups. |
Table III.
The top 20 biological processes
associated with the differentially expressed genes overlapping
between cisplatin vs. control and combination vs. control
groups.
| A, Downregulated
genes |
|---|
|
|---|
| Term | Count | P-value | Gene name |
|---|
| GO:0006334,
nucleosome assembly | 18 |
4.24×10−18 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0031497,
chromatin assembly | 18 |
6.98×10−18 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0034728,
nucleosome organization | 18 |
8.89×10−18 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0065004,
protein-DNA complex assembly | 18 |
8.89×10−18 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0006323, DNA
packaging | 18 |
1.55×10−15 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0006333,
chromatin assembly or disassembly | 18 |
5.73×10−15 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0034622,
cellular macromolecular complex assembly | 19 |
5.45×10−11 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, CTTNBP2, etc. |
| GO:0034621,
cellular macromolecular complex subunit organization | 19 |
4.01×10−10 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, CTTNBP2, etc. |
| GO:0065003,
macromolecular complex assembly | 19 |
6.33×10−8 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, CTTNBP2, etc. |
| GO:0006325,
chromatin organization | 18 |
1.25×10−7 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0043933,
macromolecular complex subunit organization | 19 |
2.18×10−7 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, CTTNBP2, etc. |
| GO:0051276,
chromosome organization | 18 |
3.94×10−6 | H1F0, HIST1H2BB,
HIST4H4, HIST1H1C, HIST1H2AF, HIST1H1B, HIST1H2AG, HIST1H1A,
HIST1H2BH, HIST2H4, etc. |
| GO:0030855,
epithelial cell differentiation | 8 |
5.36×10−4 | SPRR1A, SPRR1B,
GJA1, SPRR2G, PTCH1, ID3, FZD2, SPRR2K |
| GO:0007507, heart
development | 10 |
1.09×10−3 | NRP2, ID2, EDN1,
GJA1, FHL2, PTCH1, ADAMTS1, ID3, MECOM, TPM1 |
| GO:0031424,
keratinization | 4 |
3.94×10−3 | SPRR1A, SPRR1B,
SPRR2G, SPRR2K |
| GO:0060429,
epithelium development | 10 |
4.08×10−3 | SPRR1A, LMO4,
SPRR1B, GJA1, SPRR2G, PTCH1, ID3, FZD2, MECOM, SPRR2K |
| GO:0042325,
regulation of phosphorylation | 10 |
6.31×10−3 | SPRY1, PDGFB,
SOCS3, EDN1, PDGFRB, TRIB3, CD24A, PPP1R14B, IRS1, TRIB1 |
| GO:0019220,
regulation of phosphate metabolic process | 10 |
7.97×10−3 | SPRY1, PDGFB,
SOCS3, EDN1, PDGFRB, TRIB3, CD24A, PPP1R14B, IRS1, TRIB1 |
| GO:0051174,
regulation of phosphorus metabolic process | 10 |
7.97×10−3 | SPRY1, PDGFB,
SOCS3, EDN1, PDGFRB, TRIB3, CD24A, PPP1R14B, IRS1, TRIB1 |
| GO:0043009,
chordate embryonic development | 12 |
9.30×10−3 | HES1, SFRP2, EDN1,
NLE1, PDGFRB, GJA1, PTCH1, GAS1, MECOM, TPM1, etc. |
|
| B, Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| GO:0012501,
programmed cell death | 16 |
1.95×10−5 | TRP53INP1, FAS,
DDIT4, CKAP2, ZMAT3, EDA2R, PMAIP1, PDCD4, TMEM173, NOD1, etc. |
| GO:0008219, cell
death | 16 |
4.35×10−5 | TRP53INP1, FAS,
DDIT4, CKAP2, ZMAT3, EDA2R, PMAIP1, PDCD4, TMEM173, NOD1, etc. |
| GO:0016265,
death | 16 |
5.68×10−5 | TRP53INP1, FAS,
DDIT4, CKAP2, ZMAT3, EDA2R, PMAIP1, PDCD4, TMEM173, NOD1, etc. |
| GO:0006915,
apoptosis | 15 |
6.61×10−5 | TRP53INP1, APAF1,
FAS, CKAP2, ZMAT3, PMAIP1, PDCD4, DDIT4, TMEM173, NOD1, etc. |
| GO:0007049, cell
cycle | 16 |
3.41×10−4 | CKAP2, TXNIP,
KAT2B, CCNG1, SESN2, CDKN1A, EREG, RASSF1, TRP53INP1, MDM2,
etc. |
| GO:0007050, cell
cycle arrest | 5 |
1.64×10−3 | CDKN1A, AK1,
RASSF1, TRP53INP1, SESN2 |
| GO:0022402, cell
cycle process | 11 |
2.67×10−3 | CDKN1A, EREG, AK1,
RASSF1, TRP53INP1, MDM2, SESN2, CCNG1, CCNG2, SMC4, etc. |
| GO:0033554,
cellular response to stress | 11 |
3.25×10−3 | POLK, CLSPN,
CDKN1A, ERCC5, ATG9B, ATG12, BTG2, ZMAT3, PMAIP1, EIF2AK2,
etc. |
| GO:0012502,
induction of programmed cell death | 7 |
3.78×10−3 | NOD1, RIPK3,
TRP53INP1, PMAIP1, FAS, APAF1, PHLDA3 |
| GO:0006917,
induction of apoptosis | 7 |
3.78×10−3 | NOD1, RIPK3,
TRP53INP1, PMAIP1, FAS, APAF1, PHLDA3 |
| GO:0043068,
positive regulation of programmed cell death | 8 |
7.02×10−3 | CDKN1A, NOD1,
RIPK3, TRP53INP1, PMAIP1, FAS, APAF1, PHLDA3 |
| GO:0010942,
positive regulation of cell death | 8 |
7.32×10−3 | CDKN1A, NOD1,
RIPK3, TRP53INP1, PMAIP1, FAS, APAF1, PHLDA3 |
| GO:0006974,
response to DNA damage stimulus | 8 |
1.43×10−2 | POLK, CLSPN,
CDKN1A, ERCC5, BTG2, ZMAT3, PMAIP1, PHLDA3 |
| GO:0045596,
negative regulation of cell differentiation | 6 |
2.33×10−2 | NOTCH3, EREG,
GDF11, NFKBIA, RC3H1, TOB1 |
| GO:0043065,
positive regulation of apoptosis | 7 |
2.35×10−2 | NOD1, RIPK3,
TRP53INP1, PMAIP1, FAS, APAF1, PHLDA3 |
| GO:0008104, protein
localization | 13 |
3.59×10−2 | ARL6IP1, TXNIP,
GDI1, ABCB9, CHMP5, ZMAT3, NFKBIA, PMAIP1, OPTN, CXCL10, etc. |
| GO:0007033, vacuole
organization | 3 |
3.67×10−2 | ATG9B, ATG12,
CHMP5 |
| GO:0009411,
response to UV | 3 |
3.87×10−2 | CDKN1A, ERCC5,
PMAIP1 |
 | Table IV.Kyoto Encyclopedia of Genes and
Genomes (KEGG) pathways associated with the differentially
expressed genes overlapping between cisplatin vs. control and
combination vs. control groups. |
Table IV.
Kyoto Encyclopedia of Genes and
Genomes (KEGG) pathways associated with the differentially
expressed genes overlapping between cisplatin vs. control and
combination vs. control groups.
| A, Downregulated
genes |
|---|
| Term | Count | P-value | Gene name |
|---|
| mmu05322, Systemic
lupus erythematosus | 14 |
3.29×10−10 | HIST1H2BB, HIST4H4,
HIST1H2AF, HIST1H2AG, HIST1H2BH, HIST2H4, HIST1H2BM, HIST1H4A,
H3F3B, H2AFX, etc. |
| mmu04510, Focal
adhesion | 9 |
3.42×10−3 | GM12715, PDGFB,
COL3A1, COL1A2, PDGFRB, COL6A1, THBS1, COL5A3, SPP1 |
| mmu04512,
ECM-receptor interaction | 6 |
3.90×10−3 | COL3A1, COL1A2,
COL6A1, THBS1, COL5A3, SPP1 |
| mmu04330, Notch
signaling pathway | 4 |
2.53×10−2 | DTX4, HES1, MAML3,
LFNG |
|
| B, Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| mmu04115, p53
signaling pathway | 10 |
6.27×10−9 | CDKN1A, ZMAT3,
MDM2, PMAIP1, FAS, APAF1, SESN2, CCNG1, CCNG2, GTSE1 |
| mmu04623, Cytosolic
DNA-sensing pathway | 5 |
1.35×10−3 | DDX58, TMEM173,
RIPK3, NFKBIA, CXCL10 |
| mmu04622,
RIG-I-like receptor signaling pathway | 5 |
2.97×10−3 | DDX58, TMEM173,
ATG12, NFKBIA, CXCL10 |
| mmu04210,
Apoptosis | 4 |
4.16×10−2 | TNFRSF10B, NFKBIA,
FAS, APAF1 |
Specific effect of cisplatin and Tet
combination on HEI-OC1 auditory cells
Of the 882 DEGs induced by the cisplatin and Tet
combination, the 467 downregulated histone genes were involved in
biological processes including ‘nucleosome assembly’ and
‘nucleosome organization’ (Table
V), and one KEGG pathway of ‘Systemic lupus erythematosus’
(Table VI). The downregulated
DEGs that were specifically induced by combination treatment,
including cyclin-dependent kinase inhibitor 2A (CDKN2A) were
related to biological processes like ‘rRNA processing’ and ‘ncRNA
metabolic process’ (Table V). The
downregulated CCND1 specifically responded to combination treatment
was also involved in ‘Focal adhesion’ (Table VI).
 | Table V.The top 20 biological processes
associated with the differentially expressed genes induced by
combination treatment compared with control. |
Table V.
The top 20 biological processes
associated with the differentially expressed genes induced by
combination treatment compared with control.
| Downregulated
genes |
|---|
|
|---|
| Term | Count | P-value | Gene name |
|---|
| GO:0006334,
nucleosome assembly | 19 |
3.03×10−14 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0031497,
chromatin assembly | 19 |
5.06×10−14 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0065004,
protein-DNA complex assembly | 19 |
6.50×10−14 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0034728,
nucleosome organization | 19 |
6.50×10−14 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0034622,
cellular macromo lecular complex assembly | 28 |
6.42×10−13 | EIF6, FKBP4, WNT2,
HIST1H2BM, HIST1H4A, PRMT5, TUBA1C, NIP7, HIST1H1A, HIST1H2BH,
FLNA, etc. |
| GO:0042254,
ribosome biogenesis | 20 |
7.62×10−12 | EIF6, NAF1, EXOSC4,
NIP7, BOP1, IMP3, CDKN2A, NPM1, RRS1, NHP2, etc. |
| GO:0034470, ncRNA
processing | 23 |
9.92×10−12 | NAF1, BOP1, RRP9,
QTRT1, EXOSC1, TRMT61A, FBL, IMP3, NOP2, CDKN2A, etc. |
| GO:0006323, DNA
packaging | 19 |
1.16×10−11 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0034621,
cellular macromolecular complex subunit organization | 28 |
1.17×10−11 | HIST1H2AB, EIF6,
FKBP4, WNT2, CTTNBP2, NIP7, FLNA, HIST2H4, HIST1H3A, CACNA1A,
etc. |
| GO:0022613,
ribonucleoprotein complex biogenesis | 21 |
3.69×10−11 | EIF6, NAF1, RRP9,
FBL, IMP3, NOP2, CDKN2A, PRMT5, NPM1, RRS1, etc. |
| GO:0006333,
chromatin assembly or disassembly | 19 |
4.39×10−11 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| GO:0034660, ncRNA
metabolic process | 24 |
2.17×10−10 | NAF1, PUS1, EXOSC4,
BOP1, QTRT1, EXOSC1, TRMT61A, IMP3, CDKN2A, METTL1, etc. |
| GO:0065003,
macromolecular complex assembly | 30 |
8.60×10−10 | HIST4H4, FKBP4,
WNT2, CTTNBP2, PRMT5, NPM1, MYC, TUBA1C, NIP7, FLNA, etc. |
| GO:0043933,
macromolecular complex subunit organization | 30 |
5.71×10−9 | HIST1H2AB, EIF6,
FKBP4, WNT2, CTTNBP2, PRMT5, NPM1, MYC, NIP7, etc. |
| GO:0006364, rRNA
processing | 13 |
9.96×10−8 | NAF1, EXOSC4,
EXOSC1, BOP1, FBL, RCL1, IMP3, NOP2, CDKN2A, NHP2, etc. |
| GO:0016072, rRNA
metabolic process | 13 |
1.16×10−7 | NAF1, EXOSC4,
EXOSC1, BOP1, FBL, RCL1, IMP3, NOP2, CDKN2A, NHP2, etc. |
| GO:0006396, RNA
processing | 29 |
8.47×10−7 | NAF1, BOP1, QTRT1,
IMP3, CDKN2A, METTL1, PRMT5, TSEN2, EXOSC4, RPP40, etc. |
| GO:0008033, tRNA
processing | 11 |
3.90×10−6 | PUS7L, ELAC2, PUS1,
METTL1, WDR4, QTRT1, TSEN2, TRMT61A, NSUN2, FBL, etc. |
| GO:0006399, tRNA
metabolic process | 12 |
5.36×10−5 | PUS7L, ELAC2, PUS1,
METTL1, WDR4, QTRT1, TSEN2, TRMT61A, RPP40, FBL, etc. |
| GO:0006325,
chromatin organization | 20 |
1.14×10−4 | HIST1H2AB, PRMT5,
H2AFZ, H1F0, HIST1H2BB, HIST1H1C, HIST1H1B, HIST1H1A, HIST1H2BH,
HIST2H4, etc. |
|
| Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| GO:0012501,
programmed cell death | 21 |
1.23×10−5 | CKAP2, ZMAT3,
PMAIP1, BCL2L11, DDIT4, TMEM173, CASP3, TRP53INP1, FAS, PHLDA3,
etc. |
| GO:0007049, cell
cycle | 24 |
1.75×10−5 | TXNIP, CKAP2,
NUSAP1, CCNG1, NCAPD3, BRCA1, CDKN1A, RASSF1, TRP53INP1, MDM2,
etc. |
| GO:0012502,
induction of programmed cell death | 12 |
2.50×10−5 | SERINC3, CASP3,
CASP4, NOD1, IL18, RIPK3, TRP53INP1, PMAIP1, FAS, APAF1, etc. |
| GO:0006917,
induction of apoptosis | 12 |
2.50×10−5 | SERINC3, CASP3,
CASP4, NOD1, IL18, RIPK3, TRP53INP1, PMAIP1, FAS, APAF1, etc. |
| GO:0006915,
apoptosis | 20 |
3.28×10−5 | CKAP2, ZMAT3,
PMAIP1, PDCD4, DDIT4, CASP3, TMEM173, TRP53INP1, JAK2, FAS,
etc. |
| GO:0008219, cell
death | 21 |
3.32×10−5 | JAK2, FAS, CKAP2,
ZMAT3, EDA2R, PMAIP1, DDIT4, TMEM173, CASP3, TRP53INP1, etc. |
| GO:0022402, cell
cycle process | 18 |
4.28×10−5 | NUSAP1, CCNG1,
SESN2, CCNG2, NCAPD3, BRCA1, CDKN1A, RASSF1, TRP53INP1, MDM2,
etc. |
| GO:0016265,
death | 21 |
4.61×10−5 | TRP53INP1, JAK2,
FAS, CKAP2, ZMAT3, PMAIP1, PDCD4, TAX1BP1, BCL2L11, DDIT4,
etc. |
| GO:0043068,
positive regulation of programmed cell death | 14 |
5.61×10−5 | PMAIP1, BCL2L11,
BRCA1, SERINC3, CDKN1A, CASP3, CASP4, NOD1, TRP53INP1, FAS,
etc. |
| GO:0010942,
positive regulation of cell death | 14 |
6.06×10−5 | PMAIP1, BCL2L11,
BRCA1, SERINC3, CDKN1A, CASP3, CASP4, NOD1, TRP53INP1, FAS,
etc. |
| GO:0043065,
positive regulation of apoptosis | 13 |
2.13×10−4 | PMAIP1, BCL2L11,
BRCA1, SERINC3, CASP3, RIPK3, TRP53INP1, APAF1, FAS, PHLDA3,
etc. |
| GO:0007050, cell
cycle arrest | 6 |
1.23×10−3 | GAS2L3, CDKN1A,
AK1, RASSF1, TRP53INP1, SESN2 |
| GO:0042981,
regulation of apoptosis | 18 |
2.19×10−3 | IL18, PMAIP1,
TAX1BP1, BRCA1, SERINC3, CASP3, CDKN1A, BTG2, TRP53INP1, FAS,
etc. |
| GO:0043067,
regulation of programmed cell death | 18 |
2.49×10−3 | IL18, PMAIP1,
TAX1BP1, BRCA1, SERINC3, CASP3, CDKN1A, BTG2, TRP53INP1, FAS,
etc. |
| GO:0010941,
regulation of cell death | 18 |
2.63×10−3 | IL18, PMAIP1,
TAX1BP1, BRCA1, SERINC3, CASP3, CDKN1A, BTG2, TRP53INP1, FAS,
etc. |
| GO:0007076, mitotic
chromosome condensation | 3 |
3.98×10−3 | NUSAP1, NCAPD3,
SMC4 |
| GO:0033554,
cellular response to stress | 14 |
4.91×10−3 | POLK, ATG9B, ATG12,
NEIL3, ZMAT3, PMAIP1, BRCA1, CASP3, CDKN1A, BTG2, etc. |
| GO:0007067,
mitosis | 9 |
5.62×10−3 | SPC25, KNTC1,
NUSAP1, NDC80, CEP55, CCNG1, CCNG2, NCAPD3, SMC4 |
| GO:0000280, nuclear
division | 9 |
5.62×10−3 | SPC25, KNTC1,
NUSAP1, NDC80, CEP55, CCNG1, CCNG2, NCAPD3, SMC4 |
| GO:0000087, M phase
of mitotic cell cycle | 9 |
6.38×10−3 | SPC25, KNTC1,
NUSAP1, NDC80, CEP55, CCNG1, CCNG2, NCAPD3, SMC4 |
 | Table VI.Kyoto Encyclopedia of Genes and
Genomes pathways associated with the differentially expressed genes
induced by combination treatment compared with control. |
Table VI.
Kyoto Encyclopedia of Genes and
Genomes pathways associated with the differentially expressed genes
induced by combination treatment compared with control.
| A, Downregulated
genes |
|---|
|
|---|
| Term | Count | P-value | Gene name |
|---|
| mmu05322, Systemic
lupus erythematosus | 15 |
1.94×10−7 | HIST1H2AB, HIST4H4,
HIST1H4K, HIST1H2AF, HIST1H2AG, HIST1H2BM, HIST1H2BK, HIST1H4A,
HIST1H4B, HIST2H2AC, etc. |
| mmu03010,
Ribosome | 10 |
3.36×10−4 | RPS19, RPL41,
RPLP1, GM10020, RPL26, RPL3, RPL10, RPL36, RPL37, RPS5 |
| mmu04510, Focal
adhesion | 15 |
3.76×10−4 | PDGFB, COL3A1,
COL2A1, COL5A3, FLNA, CCND1, COL1A2, PDGFRB, THBS1, SPP1, etc. |
| mmu00670, One
carbon pool by folate | 4 |
6.69×10−3 | SHMT2, ATIC,
MTHFD1L, GART |
| mmu00100, Steroid
biosynthesis | 4 |
7.98×10−3 | CYP51, SQLE, LSS,
DHCR24 |
| mmu04512,
ECM-receptor interaction | 7 |
1.69×10−2 | COL3A1, COL1A2,
COL6A1, COL2A1, THBS1, COL5A3, SPP1 |
|
| B, Upregulated
genes |
|
| Term | Count | P-value | Gene
name |
|
| mmu04115, p53
signaling pathway | 11 |
3.09×10−8 | CDKN1A, CASP3,
ZMAT3, MDM2, PMAIP1, FAS, APAF1, SESN2, CCNG1, CCNG2, etc. |
| mmu04623, Cytosolic
DNA-sensing pathway | 7 |
1.10×10−4 | DDX58, TMEM173,
IL18, RIPK3, NFKBIA, CHUK, CXCL10 |
| mmu05200, Pathways
in cancer | 13 |
1.74×10−3 | NFKBIA, TCF7L1,
FZD6, CTNNA2, CASP3, CDKN1A, RASSF1, MDM2, WNT9A, FAS, etc. |
| mmu04622,
RIG-I-like receptor signaling pathway | 6 |
2.57×10−3 | DDX58, TMEM173,
ATG12, NFKBIA, CHUK, CXCL10 |
| mmu04210,
Apoptosis | 6 |
7.41×10−3 | CASP3, TNFRSF10B,
NFKBIA, FAS, APAF1, CHUK |
| mmu04621, NOD-like
receptor signaling pathway | 5 |
1.11×10−2 | NOD1, IL18, NFKBIA,
TAB2, CHUK |
| mmu04920,
Adipocytokine signaling pathway | 5 |
1.44×10−2 | CPT1C, NFKBIE,
NFKBIA, JAK2, CHUK |
| mmu04142,
Lysosome | 6 |
2.59×10−2 | SGSH, ABCB9, AP3M2,
IGF2R, SORT1, CTSF |
| mmu05215, Prostate
cancer | 5 |
3.77×10−2 | CDKN1A, MDM2,
NFKBIA, TCF7L1, CHUK |
The 415 upregulated genes (such as ZMAT3, PMAIP1,
TRP53INP1, CDKN1A, BTG2, and FAS) were clustered in biological
processes including ‘programmed cell death’, ‘cell cycle’,
‘induction of apoptosis’ and ‘apoptosis’ (Table V), as were the DEGs that were
specifically upregulated by the combination treatment (including
Caspase-3 (CASP3) and IL18 (Table
V). Genes like CDKN1A, CASP3, ZMAT3, PMAIP1, and FAS were
involved in ‘p53 signaling pathway’, and genes including CASP3,
CDKN1A, and FAS were related to ‘pathways in cancer’, as were
TCF7L1 and FZD6 genes that were specifically responded to
combination treatment (Table
VI).
Most of the 432 DEGs that were specifically induced
by the combination treatment were associated with the biological
processes and KEGG pathways that were similar to the functional
categories associated with the 882 DEGs related to combination
treatment (Table SII). For
instance, downregulated genes including CDKN2A were related to the
biological processes that related to the metabolism and biogenesis
of ribosome and nucleosome, and the processing of RNAs (Table SII). upregulated CASP3 and IL18,
were associated with ‘positive regulation of apoptotic process’ and
‘positive regulation of cell death’. The ‘Pathways in cancer’
pathway enriched upregulated WNT9A, TCF7L1, and FZD6 (Table SII).
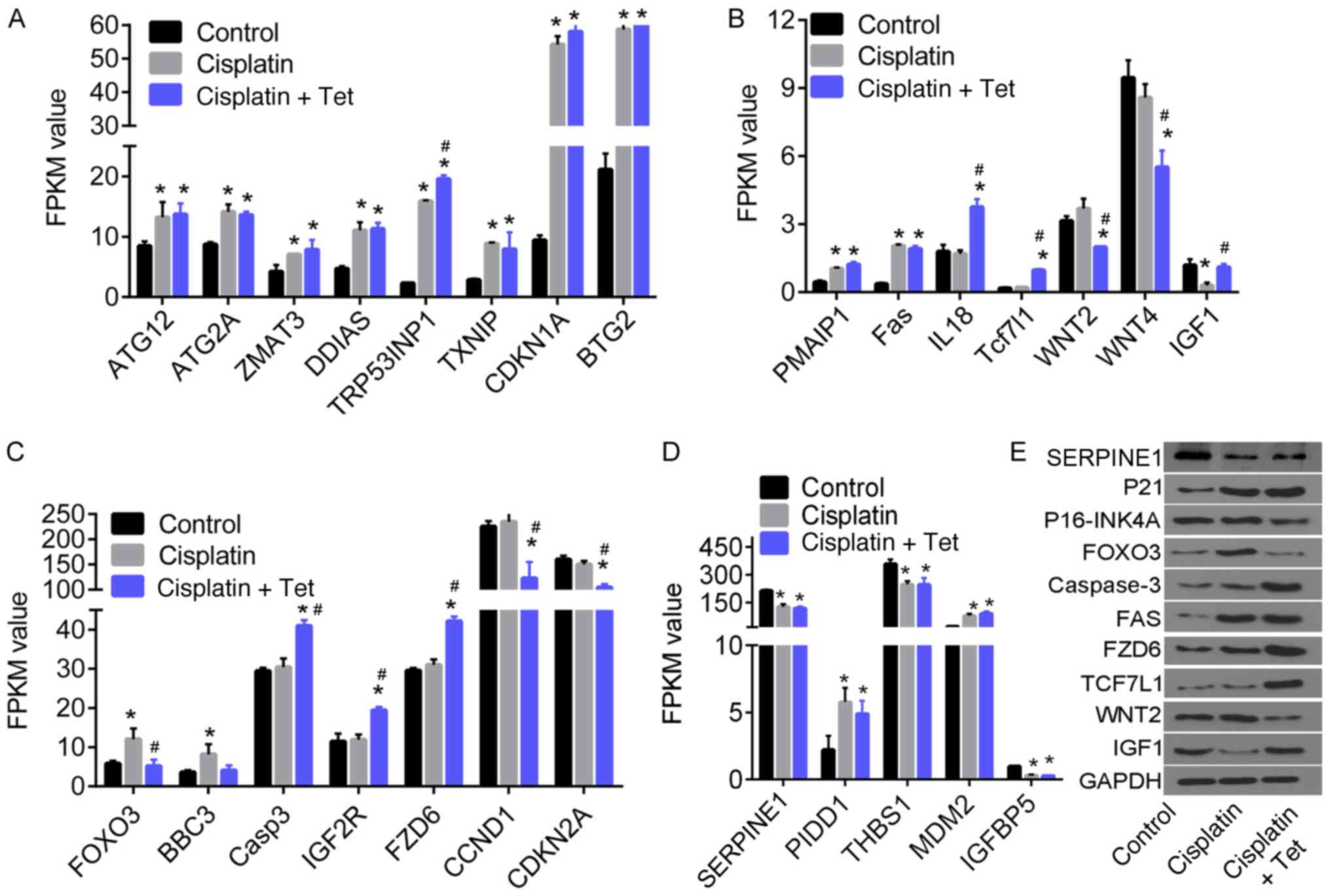
The expression patterns of several DEGs are shown in
Fig. 5. The FPKM of ATG12, ATG2A,
ZMAT3, DDIAS, TRP53INP1, TXNIP, CDKN1A, BTG2, PMAIP1, PIDD1 and Fas
increased following treatment with cisplatin alone or in
combination with Tet, compared with untreated control cells. By
contrast, serpine1, THBS1 and IGFBP5 were downregulated.
The expression of IGF1 (Fig. 5B) decreased following cisplatin
treatment but was rescued by the addition of Tet (P<0.05). FOXO3
expression followed the opposite pattern (Fig. 5C). In comparison with control and
cisplatin alone, Tet treatment increased the expression of caspase
3, IGF2R, FZD6, IL18, TCF7L1, and decreased the expression of
WNT2/4, CCND1 and CDKN2A (Fig. 5B and
C). The gene expression changes induced specifically by Tet,
and their associated biological processes, may serve an important
role in inhibiting cisplatin-induced cytotoxicity in HEI-OC1
auditory cells.
Validation of protein expression
Protein expression of several DEGs was determined
using western blot analysis. CDKN1A/p21 and Fas upregulation was
cisplatin-dependent and Tet-independent (Fig. 5E). However, expression of
CDKN2A/p16-INK4A and WNT2 were decreased, whereas TCF7L1, FZD6, and
CASP3 were specifically upregulated by the combination of cisplatin
and Tet.
Discussion
Cisplatin-associated ototoxicity is a major
complication of cisplatin-based chemotherapy (2,3,5).
Cisplatin-induced cytotoxicity, DNA damage and apoptosis in
cochlear hair cells contribute to cisplatin-associated ototoxicity
(10,12–14).
The present study demonstrated that cisplatin-induced ototoxicity
in HEI-OC1 auditory cells was associated with the inhibition of
cell viability and dysregulation of genes related to apoptosis,
cell cycle arrest and several pathways, including the p53, HIF-1,
Wnt and PI3K-Akt signaling pathways. Treatment with Tet protected
HEI-OC1 auditory cells against cisplatin-induced cytotoxicity in
vitro and regulated several signaling pathways.
The synergistic effect of Tan IIA on
cisplatin-induced cytotoxicity in cancer cells has been previously
reported (18). Indeed, Tan IIA
was previously observed to enhance cisplatin-induced apoptosis and
cell cycle arrest at the S phase in human prostate cancer cells
(18). In another study, Tan IIA
promoted apoptosis and cell cycle arrest at the sub-G1
phase in human hepatocellular carcinoma cells (19). Du et al (16) demonstrated that 24-h Tan IIA
treatments at concentrations <64 mg/l could alleviate
radiation-induced cytotoxicity and apoptosis of HEI-OC1 auditory
cells by inhibiting p65/nuclear factor κB p53 and p21 signaling
pathways. Du et al (16)
also indicated that Tan IIA at concentrations >16 mg/l resulted
in significant cytotoxicity, whereas Tan IIA <8 mg/l had no
cytotoxic effects on HEI-OC1 cells. However, the present study
determined that Tan IIA concentrations as low as 1.5 mg/l augmented
the effect of cisplatin on cell viability of HEI-OC1 auditory
cells. This difference might be due to the longer treatment period
in the present study, compared with Du et al (16) (30 and 24 h, respectively). Thus, it
may be hypothesized that Tan IIA-induced cytotoxicity to HEI-OC1
auditory cells is time-dependent, since the concentration used in
the present study was far below the IC50 value of 151.8
mg/l.
By contrast, low concentrations of Tet (37.5 and 70
mg/l) exerted a protective effect against cisplatin-induced
cytotoxicity. RNA-seq analysis was carried out to examine the
underlying molecular mechanism. Cisplatin inhibited the viability
of HEI-OC1 auditory cells by decreasing the expression of Hist1 and
Hist2 gene clusters that are associated with DNA replication and
actively transcribed in differentiating cells (20). Cisplatin also inhibited the
expression of genes related to cell migration and proliferation,
including IGF1 and IGFBP5. The expression of IGF1, IGF receptors
and IGFBPs has been reported to induce or promote the proliferation
of various cell types through several signaling pathways, such as
PI3K/Akt (21–23). It has been demonstrated that IGF1
could counteract cisplatin-induced DNA damage by inhibiting
cisplatin-induced phosphorylation of histone H2AX and
ataxia-telangiectasia mutated, and blocking DNA double-strand break
repair (24). IGFBP5
overexpression and IGF1R inhibition are associated with increased
cisplatin sensitivity in esophageal carcinoma cells, lung and
ovarian cancer cells (25–27). The present study demonstrated that,
compared with cisplatin alone, Tet treatment increased the
expression of IGF1, but not IGFBP5. These results suggested that
Tet protected HEI-OC1 auditory cells from cisplatin-induced
cytotoxicity by restoring IGF1 signaling.
Exposure to cisplatin upregulated genes that were
associated with apoptosis and autophagy, including ATG12, ZMAT3,
PMAIP1, TRP53INP1 and PIDD1. However, the expression of these genes
was not modulated by Tet treatment. By contrast, treatment with Tet
downregulated FOXO3 (significantly) and BBC3 (insignificantly),
compared with cisplatin alone. A previous study demonstrated that
the proapoptotic FoxO signaling pathway was activated by
amikacin-induced ototoxicity (28). FOXO3 mediates a chemo-protection
effect in advanced cancer by interacting with TP53 and mutations in
TP53 prevent FOXO3 binding, thereby enhancing FOXO3-induced cell
death in high-stage neuroblastoma (29). Additionally, FOXO3 links autophagy
and apoptosis by regulating the transactivation of the proapoptotic
gene BBC3 (30). Under the
condition of autophagy inhibition, BBC3 is transactivated by FOXO3
(29,30). In the present study, the expression
of FOXO3 and BBC3 following cisplatin exposure was increased,
consistent with increased apoptosis. It could be hypothesized that
increased FOXO3 and BB3-mediated apoptosis may be associated with
inhibition of autophagy following cisplatin treatment. Moreover,
the protective effect of Tet treatment might be mediated by
modulation of autophagy.
The proapoptotic p53 signaling pathway is activated
in the cochlea and in HEI-OC1 auditory cells following cisplatin
exposure (8,10,31,32).
Xiong et al (32) and Ma
et al (31) indicated that
the expression or acetylation of p53 in HEI-OC1 auditory cells
could be increased by cisplatin treatment, thereby promoting
apoptosis. In particular, Ma et al (31) demonstrated that ginkgolide B
prevented cisplatin-induced cytotoxicity and decreased p53
expression in cisplatin-treated cochlear cells. Moreover,
Benkafadar et al (33)
suggested that the knockdown of p53 could attenuate
cisplatin-induced ototoxicity and cochlear cell apoptosis.
Consistent with these previous reports, the present study
demonstrated that cisplatin induced significant upregulation of p53
signaling-related genes, including ZMAT3, PMAIP1, TRP53INP1 and
PIDD1. However, the expression of these genes was not affected by
Tet treatment, indicating that the protective effect of Tet on
HEI-OC1 auditory cells did not involve p53 signaling.
A previous study suggested that the Wnt signaling
pathway protects against neomycin-induced hair cell damage
(9). Liu et al (9) demonstrated that β-catenin activation
prevented apoptosis of hair cells. By contrast, inhibition of
β-catenin increased FOXO3 expression, ROS production, and apoptosis
in these cells (9). In addition,
the inhibition of Wnt signaling in spiral ganglion neurons may
increase the levels of ROS (8).
The present study suggested that Tet addition into HEI-OC1 auditory
cells increased the expression of two mediators of the Wnt
signaling pathway, including TCF7L1 and FZD6. Thus, activation of
Wnt signaling may be involved in the Tet-mediated protective effect
on HEI-OC1 auditory cells. However, this study demonstrated that
WNT2 and WNT4 genes, which are necessary for the activation of
FZD6, were downregulated specifically by combination with Tet.
These results suggested that the activation of Wnt signaling may be
mediated by novel factors, rather than WNT2/4.
The present study has limitations. Firstly, the
concentrations of cisplatin used in this study were too low. To
minimize its cytotoxicity in vitro, cells were treated with
cisplatin and Tet at concentrations <IC50 values.
Obvious reverse changes in cell viability and genomics were
observed between combination treatment of 37.5 mg/l Tet and 30 µM
cisplatin, and 30 µM cisplatin alone. Moreover, the potential
mechanisms underlying the role of Tet were only identified using
RNA-seq and bioinformatics analysis. Experimental validation would
provide further insight into the molecular basis of Tet-mediated
inhibition of ototoxicity.
In conclusion, the present study confirmed that low
doses of Tet could attenuate cisplatin-induced cytotoxicity in
HEI-OC1 auditory cells. Gene expression analysis suggested that
cisplatin induced ototoxicity in vitro by activating the p53
and FoxO signaling pathways, and inhibiting IGF signaling. Tet
attenuated ototoxicity through activation of the Wnt/β-catenin and
IGF pathways, and inhibition of FOXO3/BBC3 signaling. Further
validation is required to directly demonstrate the roles of these
pathways in auditory cells.
Supplementary Material
Supporting Data
Acknowledgements
The authors would like to thank Professor Federico
Kalinec (David Geffen School of Medicine at UCLA, CA, USA) for the
generous donation of the HEI-OC1 auditory cells.
Funding
No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article. The fastq files of raw
sequencing data are available from the corresponding author upon
reasonable request.
Authors' contributions
GG and XT designed the study. GG, XH, JC and LB
acquired, analyzed and interpreted the data. XH, JC and LB carried
out the statistical analysis. GG drafted the manuscript. XH and XT
revised the manuscript for important intellectual content. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Breglio AM, Rusheen AE, Shide ED,
Fernandez KA, Spielbauer KK, McLachlin KM, Hall MD, Amable L and
Cunningham LL: Cisplatin is retained in the cochlea indefinitely
following chemotherapy. Nat Commun. 8:16542017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shorter P, Harden F, Owen R, Panizza B,
Burmeister B, Sommerville J, Mengersen K and Foote M: Risk profiles
for sensorineural hearing loss in patients with head and neck
cancer receiving cisplatin-based chemoradiation. J Med Imaging
Radiat Sci. 48:61–67. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wang J, Chen YY, Tai A, Chen XL, Huang SM,
Yang C, Bao Y, Li NW, Deng XW, Zhao C, et al: Sensorineural hearing
loss after combined intensity modulated radiation therapy and
cisplatin-based chemotherapy for nasopharyngeal carcinoma. Transl
Oncol. 8:456–462. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Robertson MS, Hayashi SS, Camet ML,
Trinkaus K, Henry J and Hayashi RJ: Asymmetric sensorineural
hearing loss is a risk factor for late-onset hearing loss in
pediatric cancer survivors following cisplatin treatment. Pediatr
Blood Cancer. 66:e274942019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bass JK, Hua CH, Huang J, Onar-Thomas A,
Ness KK, Jones S, White S, Bhagat SP, Chang KW and Merchant TE:
Hearing loss in patients who received cranial radiation therapy for
childhood cancer. J Clin Oncol. 34:1248–1255. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wu X, Li X, Song Y, Li H, Bai X, Liu W,
Han Y, Xu L, Li J, Zhang D, et al: Allicin protects auditory hair
cells and spiral ganglion neurons from cisplatin-Induced apoptosis.
Neuropharmacology. 116:429–440. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Sheth S, Mukherjea D, Rybak LP and
Ramkumar V: Mechanisms of cisplatin-induced ototoxicity and
otoprotection. Front Cell Neurosci. 11:3382017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liu W, Xu X, Fan Z, Sun G, Han Y, Zhang D,
Xu L, Wang M, Wang X, Zhang S, et al: Wnt signaling activates
TP53-induced glycolysis and apoptosis regulator and protects
against cisplatin-induced spiral ganglion neuron damage in the
mouse cochlea. Antioxid Redox Signal. 30:1389–1410. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Liu L, Chen Y, Qi J, Zhang Y, He Y, Ni W,
Li W, Zhang S, Sun S, Taketo MM, et al: Wnt activation protects
against neomycin-induced hair cell damage in the mouse cochlea.
Cell Death Dis. 7:e21362016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Guo X, Bai X, Li L, Li J and Wang H:
Forskolin protects against cisplatin-induced ototoxicity by
inhibiting apoptosis and ROS production. Biomed Pharmacother.
99:530–536. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang Q, Liu H, McGee J, Walsh EJ, Soukup
GA and He DZ: Identifying microRNAs involved in degeneration of the
organ of corti during age-related hearing loss. PLoS One.
8:e627862013. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang P, Zhang P, Huang J, Li M and Chen X:
Trichostatin a protects against cisplatin-induced ototoxicity by
regulating expression of genes related to apoptosis and synaptic
function. Neurotoxicology. 37:51–62. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bhatta P, Dhukhwa A, Sheehan K, Al Aameri
RFH, Borse V, Ghosh S, Sheth S, Mamillapalli C, Rybak L, Ramkumar V
and Mukherjea D: Capsaicin protects against cisplatin ototoxicity
by changing the STAT3/STAT1 ratio and activating Cannabinoid (CB2)
receptors in the Cochlea. Sci Rep. 9:41312019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kitcher SR, Kirkwood NK, Camci ED, Wu P,
Gibson RM, Redila VA, Simon JA, Rubel EW, Raible DW, Richardson GP
and Kros CJ: ORC-13661 protects sensory hair cells from
aminoglycoside and cisplatin ototoxicity. JCI Insight.
4:e1267642019. View Article : Google Scholar
|
|
15
|
Cui C, Liu D and Qin X: Attenuation of
streptomycin ototoxicity by tetramethylpyrazine in guinea pig
cochlea. Otolaryngol Head Neck Surg. 152:904–911. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Du S, Yao Q, Tan P, Xie G, Ren C, Sun Q,
Zhang X, Zheng R, Yang K, Yuan Y and Yuan Q: Protective effect of
tanshinone IIA against radiation-induced ototoxicity in HEI-OC1
cells. Oncol Lett. 6:901–906. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bayram A, Kaya A, Akay E, Hıra İ and Özcan
İ: The protective role of tetramethylpyrazine against
cisplatin-induced ototoxicity. Int J Pediatr Otorhinolaryngol.
94:1–7. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Hou LL, Xu QJ, Hu GQ and Xie SQ:
Synergistic antitumor effects of tanshinone II A in combination
with cisplatin via apoptosis in the prostate cancer cells. Yao Xue
Xue Bao. 48:675–679. 2013.(In Chinese). PubMed/NCBI
|
|
19
|
Chang TW, Lin CY, Tzeng YJ and Lur HS:
Synergistic combinations of tanshinone IIA and trans-resveratrol
toward cisplatin-comparable cytotoxicity in HepG2 human
hepatocellular carcinoma cells. Anticancer Res. 34:5473–5480.
2014.PubMed/NCBI
|
|
20
|
Banday AR, Baumgartner M, Al Seesi S,
Karunakaran DK, Venkatesh A, Congdon S, Lemoine C, Kilcollins AM,
Mandoiu I, Punzo C and Kanadia RN: Replication-dependent histone
genes are actively transcribed in differentiating and aging retinal
neurons. Cell Cycle. 13:2526–2541. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kadri Z, Lefevre C, Goupille O, Penglong
T, Granger-Locatelli M, Fucharoen S, Maouche-Chretien L, Leboulch P
and Chretien S: Erythropoietin and IGF-1 signaling synchronize cell
proliferation and maturation during erythropoiesis. Genes Dev.
29:2603–2616. 2015.PubMed/NCBI
|
|
22
|
Yu M, Wang H, Xu Y, Yu D, Li D, Liu X and
Du W: Insulin-like growth factor-1 (IGF-1) promotes myoblast
proliferation and skeletal muscle growth of embryonic chickens via
the PI3K/Akt signalling pathway. Cell Biol Int. 39:910–922. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bruchim I, Attias Z and Werner H:
Targeting the IGF1 axis in cancer proliferation. Expert Opin Ther
Targets. 3:1179–1192. 2009. View Article : Google Scholar
|
|
24
|
Jeon JH, Kim SK, Kim HJ, Chang J, Ahn CM
and Chang YS: Insulin-like growth factor-1 attenuates
cisplatin-induced gammaH2AX formation and DNA double-strand breaks
repair pathway in non-small cell lung cancer. Cancer Lett.
272:232–241. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Du J, Shi HR, Ren F, Wang JL, Wu QH, Li X
and Zhang RT: Inhibition of the IGF signaling pathway reverses
cisplatin resistance in ovarian cancer cells. BMC Cancer.
17:8512017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Sun Y, Zheng S, Torossian A, Speirs CK,
Schleicher S, Giacalone NJ, Carbone DP, Zhao Z and Lu B: Role of
insulin-like growth factor-1 signaling pathway in
cisplatin-resistant lung cancer cells. Int J Radiat Oncol Biol
Phys. 82:e563–e572. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chan D, Zhou Y, Chui CH, Lam KH, Law S,
Chan AS, Li X, Lam AK and Tang JCO: Expression of insulin-like
growth factor binding protein-5 (IGFBP5) reverses
cisplatin-resistance in esophageal carcinoma. Cells. 7:1432018.
View Article : Google Scholar
|
|
28
|
Liu S, Zhang X, Sun M, Xu T and Wang A:
FoxO3a plays a key role in the protective effects of pomegranate
peel extract against amikacin-induced ototoxicity. Int J Mol Med.
40:175–181. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Rupp M, Hagenbuchner J, Rass B, Fiegl H,
Kiechl-Kohlendorfer U, Obexer P and Ausserlechner MJ:
FOXO3-mediated chemo-protection in high-stage neuroblastoma depends
on wild-type TP53 and SESN3. Oncogene. 36:6190–6203. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Fitzwalter BE and Thorburn A: FOXO3 links
autophagy to apoptosis. Autophagy. 14:1467–1468. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Ma W, Li J, Hu J, Cheng Y, Wang J, Zhang X
and Xu M: MiR214-regulated p53-NOX4/p66shc pathway plays a crucial
role in the protective effect of Ginkgolide B against
cisplatin-induced cytotoxicity in HEI-OC1 cells. Chem Biol
Interact. 245:72–81. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xiong H, Pang J, Yang H, Dai M, Liu Y, Ou
Y, Huang Q, Chen S, Zhang Z, Xu Y, et al: Activation of
miR-34a/SIRT1/p53 signaling contributes to cochlear hair cell
apoptosis: Implications for age-related hearing loss. Neurobiol
Aging. 36:1692–1701. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Benkafadar N, Menardo J, Bourien J,
Nouvian R, François F, Decaudin D, Maiorano D, Puel JL and Wang J:
Reversible p53 inhibition prevents cisplatin ototoxicity without
blocking chemotherapeutic efficacy. EMBO Mol Med. 9:7–26. 2017.
View Article : Google Scholar : PubMed/NCBI
|