Introduction
Osteoporosis is a widespread skeletal disorder
resulting from imbalances in bone homeostasis (1,2).
Osteoporosis is a critical contributor of mortality in the elderly
and is a socioeconomic challenge in terms of public health
(3). The incidence of osteoporosis
was 27.96% between 2012 and 2015 in China (4). During the process of bone remodeling,
osteoblasts generate new bone tissues, while osteoclasts contribute
to the destruction and reabsorption of bone tissues (5). Bone marrow-derived mesenchymal stem
cells (BMSCs) belong to a class of pluripotent stem cells, which
are characterized by their ability to differentiate into numerous
cell types, including chondrocytes, osteoblasts and adipocytes
(6,7). Calcium and vitamin D are the basic
treatment for osteoporosis (8).
Considering that osteoblasts serve a pivotal role in the dynamic
balance between bone formation and bone resorption (9), determining the latent mechanism
regulating the osteogenic differentiation of BMSCs is required to
improve the therapeutic treatment of osteoporosis.
Long non-coding RNAs (lncRNAs) are a type of
non-coding RNAs >200 nucleotides in length that lack
protein-coding capacity (10,11).
lncRNAs have attracted extensive attention and were reported to be
involved in the initiation and evolution of numerous types of
disease, such as atherosclerosis, inflammatory bowel disease and
cancer (12–14). Accumulating evidence has identified
that lncRNAs serve as vital modulators in a wide spectrum of
biological activities, including cell differentiation (15). Studies have also demonstrated that
lncRNAs are vital in the development of several orthopedic
disorders, such as osteoporosis and ankylosis spondylitis (16,17),
through their regulation in osteogenesis. For instance, lncRNA
metastasis associated lung adenocarcinoma transcript 1 impeded the
osteogenic differentiation of mesenchymal stem cells in
osteoporosis model rats via the MAPK signaling pathway (18); overexpression of lncRNA maternally
expressed gene 3 promoted the osteogenic differentiation of
mesenchymal stem cells of patients with multiple myeloma via
activating the transcription of bone morphogenetic protein (BMP) 4
(19) and the lncRNA HOX
transcript antisense RNA suppressed microRNA (miRNA/miR)-17-5p
expression, which affected mesenchymal stem cell osteogenic
differentiation and proliferation in non-traumatic osteonecrosis of
the femoral head (20). However,
although LINC01535 has been reported to exert oncogenic activities
in cervical cancer (21), to the
best of our knowledge, the biological function of LINC01535 in
osteogenesis remains elusive.
miRNAs are a category of small non-coding RNAs that
are 18–22 nucleotides in length (22). miRNAs regulate the expression
levels of target genes through binding with their 3′-untranslated
regions (3′-UTRs) to either inhibit their degradation or
translation (23,24). miRNAs may be implicated in the
pathogenesis of osteoporosis via regulating genes closely
associated with bone homeostatic processes at the
post-transcriptional level (25).
However, despite the reported tumor suppressive properties of
miR-3619-5p in human cancers (26), its role and regulatory mechanism in
osteogenic differentiation remains poorly understood. BMP2 belongs
to the TGF-β superfamily, where it serves a crucial role in bone
regeneration through regulating the differentiation of osteoblast
progenitors or stem cells (27,28).
Previous studies have confirmed that numerous miRNAs affect the
progression of osteogenesis via targeting BMP2 (29–31).
Therefore, the present study aimed to investigate the relationship
between miR-3619-5p and BMP2.
The current study hypothesized that LINC01535 may be
involved in the process of BMSC osteogenic differentiation. The
findings revealed that LINC01535 contributed to the differentiation
of BMSCs by targeting the miR-3619-5p/BMP2 axis.
Materials and methods
Cell culture
Human BMSCs (hBMSCs) were obtained from the American
Type Culture Collection. hBMSCs were cultured in DMEM (HyClone;
Cytiva), supplemented with 10% FBS (Gibco; Thermo Fisher
Scientific, Inc.), 100 mg/l streptomycin and 100 U/l penicillin,
and maintained at 37°C in the presence of 5% CO2. To
induce osteogenic differentiation, hBMSCs were maintained in
osteogenic supplement (OS) medium (Sigma-Aldrich; Merck KGaA) with
10 mM β-glycerophosphate (Sigma-Aldrich; Merck KGaA), 100 nM
dexamethasone (Sigma-Aldrich; Merck KGaA) and 100 nM ascorbic acid
(Sigma-Aldrich; Merck KGaA) for 14 days. The OS medium was replaced
every 2 days.
Cell transfection
A short hairpin RNA (shRNA) vector containing cDNA
oligonucleotides targeting LINC01535 (sh-LINC01535; Shanghai
GenePharma Co., Ltd.) was used to knock down LINC01535 expression
levels. BMP2-specific shRNA (sh-BMP2; Invitrogen; Thermo Fisher
Scientific, Inc.) was used to knock down BMP2 expression levels.
Scramble shRNAs (sh-NC; Shanghai GenePharma Co., Ltd.) were used as
the negative controls (NCs). miR-3619-5p mimic
(5′-AAGCGAGACAGTAGCATGCGGA-3′), miR-3619-5p inhibitor
(5′-TCAGCTTGCTACTGTSTCCGTT-3′) and their respective NCs (NC mimic,
5′-AATGAGGCGTGAGATGGAGGCT-3′ and NC inhibitor,
5′-GACCTGGATCTCAGCCTAGCTT-3′) were designed and generated by
Guangzhou RiboBio Co., Ltd. To explore whether LINC01535
facilitated the osteogenesis of hBMSCs via the miR-3619-5p/BMP2
axis, hBMSCs were transfected with sh-NC + NC inhibitor,
sh-LINC01535 + NC inhibitor, sh-LINC01535 + miR-3619 inhibitor or
sh-LINC01535 + miR-3619 inhibitor + sh-BMP2. hBMSCs were
transfected with the indicated plasmids (10 nM) using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.), according to the manufacturer's protocol.
Subsequent experiments were performed 48 h post-transfection.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from hBMSCs using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.), according to the manufacturer's protocol. Total RNA was
reverse transcribed into cDNA using a SuperScript III kit
(Invitrogen; Thermo Fisher Scientific, Inc.), according to the
manufacturer's instructions. qPCR was subsequently performed on an
ABI 7500 Fast Real-Time PCR system (Applied Biosystems; Thermo
Fisher Scientific, Inc.) using SYBR Green Master mix (Thermo Fisher
Scientific, Inc.). The following thermocycling conditions were used
for qPCR: Initial denaturation at 95°C for 3 min, 40 cycles of 95°C
for 5 sec and 60°C for 30 sec. The following primer pairs were used
for the qPCR: LINC01535 forward, 5′-GGGATGGAAGTGTGATTGC-3′ and
reverse, 5′-TGATGCTAGGGGTGCTAAG-3′; miR-3619-5p forward,
5′-TCAGCAGGCAGGCTGGTGC-3′ and reverse, 5′-GAACATGTCTGCGTATCTC-3′;
BMP2 forward, 5′-AAGCGTCAAGCCAAACACAAAC-3′ and reverse,
5′-GCCACGATCCAGTCATTCCAC−3′; osteocalcin (OCN) forward,
5′-GCCGAGAAATGTTGGAGAAA-3′ and reverse, 5′-CTCCTTAATCTGGCCAACCA-3′;
osterix (OSX) forward, 5′-CAGGCTATGCTAATGATTACC−3′ and reverse,
5′-GGCAGACAGTCAGAAGAG−3′; runt-related transcription factor 2
(RUNX2) forward, 5′-CGAATAACAGCACGCTATTAA−3′ and reverse,
5′-GTCGCCAAACAGATTCATCCA−3′; GAP DH forward,
5′-AGGTCGGTGTGAACGGATTTG−3′ and reverse,
5′-TGTAGACCATGTAGTTGAGGTCA-3′ and U6 forward,
5′-CGCTTCGGCAGCACATATACTA-3′ and reverse,
5′-CGCTTCACGAATTTGCGTGTCA-3′. GAPDH and U6 were used as the
endogenous loading controls. The 2−ΔΔCq method (32) was used to quantify gene expression
levels.
Cell counting Kit-8 (CCK-8) assay
Cell proliferation was analyzed using a CCK-8 assay
(Dojindo Molecular Technologies, Inc.) according to the
manufacturer's protocol. Following transfection, hBMSCs were plated
into a 96-well plate at a density of 1×104 cells/well. A
total of 10 µl CCK-8 solution was added per well and incubated for
4 h at 37°C. Subsequently, the optical density value was determined
at a wavelength of 450 nm using a microplate reader (Bio-Rad
Laboratories, Inc.).
Alkaline phosphatase (ALP) activity
measurement
An ALP activity assay was performed using an ALP
detection kit (cat. no. A-059-2; Nanjing Jiancheng Bioengineering
Institute) according to the manufacturer's protocol. Briefly,
following osteogenic induction, hBMSCs were seeded into 96-well
plates at 6,000 cells per well, and subsequently treated with 50 µl
buffer solution and 50 µl substrate solution. Following incubation
for 15 min at 37°C, each well was supplemented with 150 µl
color-developing reagent and the absorbance was determined at a
wavelength of 520 nm using a microplate reader (Thermo Fisher
Scientific, Inc.). ALP activity was presented as King units/100 ml.
All experiments were repeated three times.
Western blotting
Total protein was extracted from hBMSCs using RIPA
lysis buffer (Beyotime Institute of Biotechnology) containing
protease inhibitors (Roche Diagnostics). Total protein was
quantified using a BCA Protein assay kit (Thermo Fisher Scientific,
Inc.), according to the manufacturer's instructions. A total of 10
µg protein/lane was separated via 10% SDS-PAGE. The separated
proteins were subsequently transferred onto PVDF membranes and
blocked with 5% non-fat milk for 2 h at room temperature. The
membranes were then incubated overnight with primary antibodies at
4°C. Following primary antibody incubation, the PVDF membranes were
incubated with horseradish peroxidase-conjugated goat anti-mouse
IgG (cat. no. ab205719) and goat anti-rabbit IgG (cat. no.
ab205718) secondary antibodies (1:1,000; both purchased form Abcam)
for 2 h at room temperature. Total protein was visualized using an
enhanced chemiluminescence kit (EMD Millipore) and semi-quantified
using Quantity One software (version 4.6.9; Bio-Rad Laboratories,
Inc.). The following primary antibodies were used: Anti-OCN
(1:1,000; cat. no. ab93876; Abcam), anti-OSX (1:1,000; cat. no.
ab209484; Abcam), anti-RUNX2 (1:1,000; cat. no. 12556; Cell
Signaling Technology, Inc.), anti-BMP2 (1:1,000; cat. no. ab214821;
Abcam) and anti-GAPDH (1:1,000; cat. no. ab8245; Abcam). GAPDH was
used as the loading control.
RNA immunoprecipitation (RIP)
assay
The EZMagna RIP kit (EMD Millipore) was used to
perform RIP experiments according to the manufacturer's protocol.
Briefly, hBMSCs were dissolved in lysis buffer and centrifuged at
10,000 × g for 10 min at 4°C. The cell lysates were added to the
RIP buffer, which was supplemented with magnetic beads coated with
anti-protein argonaute-2 (Ago2; cat. no. MABE253; EMD Millipore)
antibody and negative control anti-IgG antibody (cat. no. AB22-K;
EMD Millipore) and incubated overnight at 4°C. Samples were then
treated with proteinase K. Following elution from the beads, the
immunoprecipitated RNA was purified and subjected to RT-qPCR
analysis.
Luciferase reporter assay
StarBase 2.0 (http://starbase.sysu.edu.cn) was used to predict the
downstream target of LINC01535 and miR-3619-5p. The 3′-UTR
sequences of LINC01535 harboring miR-3619-5p binding sites were
cloned into the pmirGLO luciferase plasmid (Promega Corporation) to
construct LINC01535-wild-type (WT). To construct LINC01535-mutant
(Mut) vectors, miR-3619-5p binding sites were mutated. Following
the same protocol, the WT and Mut plasmids of BMP2 were generated.
hBMSCs were co-transfected with the corresponding luciferase
vectors and miR-3619-5p mimic or NC mimic using Lipofectamine 3000
(Invitrogen; Thermo Fisher Scientific, Inc.). Following 48 h of
incubation, the hBMSCs were collected and the relative luciferase
activity was analyzed using a Dual-Luciferase Reporter Assay system
(Promega Corporation) according to the manufacturer's protocol and
normalized to Renilla luciferase activity.
Statistical analysis
Experimental data are presented as the mean ± SD and
all assays were performed in triplicate. Statistical analysis was
performed using SPSS 21.0 software (IBM Corp.). Statistical
differences between two groups were determined using Student's
t-test. Statistical differences among ≥2 groups was determined
using one-way ANOVA followed by Tukey's post hoc test. P<0.05
was considered to indicate a statistically significant
difference.
Results
LINC01535 expression levels are
markedly upregulated during osteogenic induction
To stimulate the osteogenic differentiation of
hBMSCs, hBMSCs were treated with OS medium for 14 days. The results
revealed that the expression levels of osteogenesis-related genes,
OCN, OSX and RUNX2, were gradually upregulated as the osteogenic
induction time increased (Fig.
1A). Similarly, western blotting demonstrated that the protein
expression levels of OCN, OSX and RUNX2 were also markedly
upregulated following treatment with OS medium (Fig. 1B). The ALP activity assay also
showed that the administration of OS medium significantly enhanced
the ALP activity of hBMSCs (Fig.
1C). The CCK-8 assay revealed that the cell viability of hBMSCs
gradually increased during the process of osteogenic
differentiation (Fig. 1D). RT-qPCR
analysis illustrated that LINC01535 expression levels were
upregulated during the process of osteoblast differentiation
(Fig. 1E). Altogether, these
findings indicated that LINC01535 may be upregulated during
osteogenic induction.
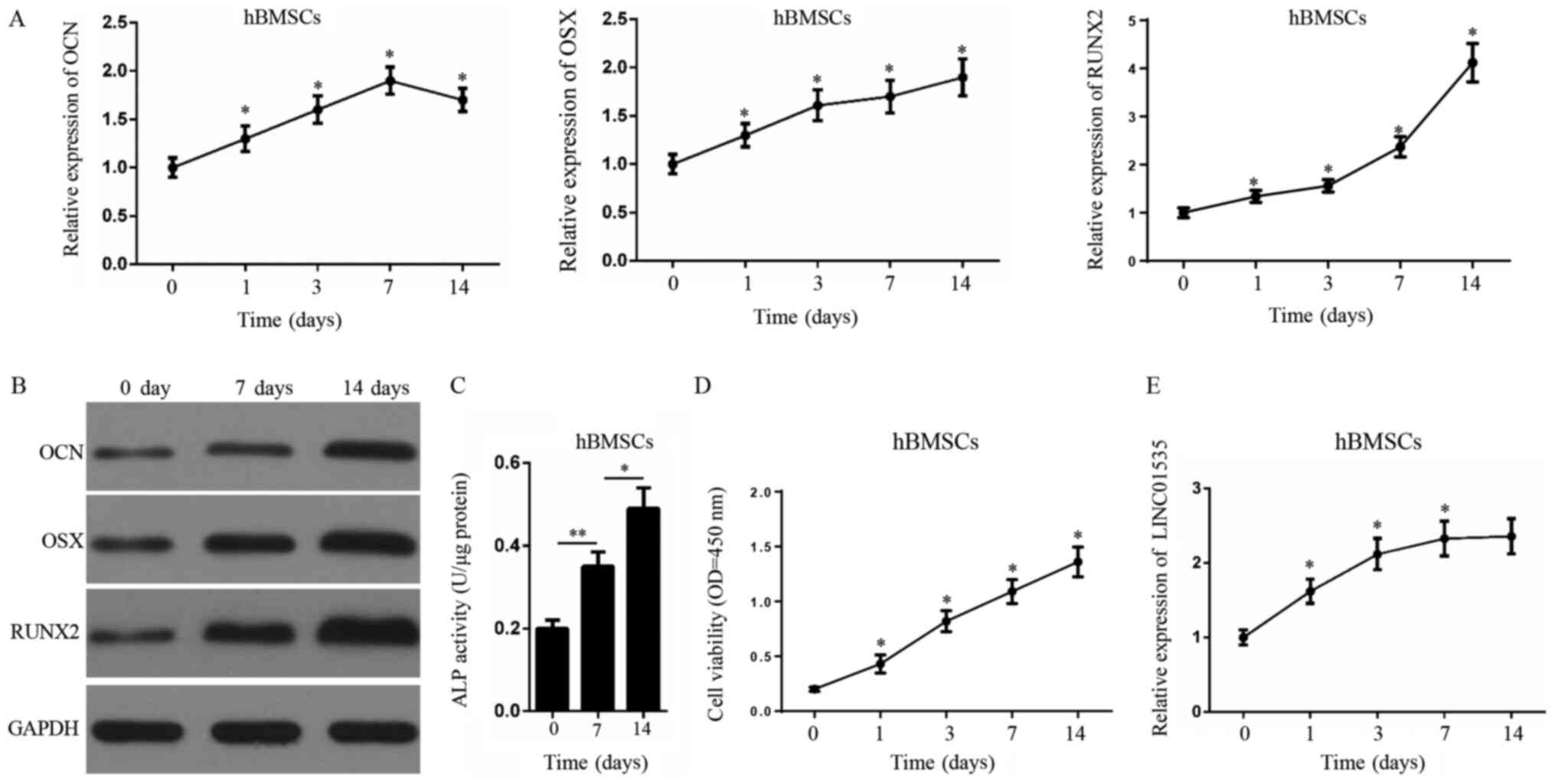 | Figure 1.LINC01535 expression is increased
during osteogenic induction. (A) RT-qPCR assay was performed to
assess the mRNA expression of OCN, OSX and RUNX2 in hBMSCs at 0, 1,
3, 7 and 14 days-post osteogenic induction. (B) Western blotting
was performed to assess the protein levels of OCN, OSX and RUNX2 in
hBMSCs during osteogenic induction. (C) ALP activity assay was
performed to estimate osteogenic differentiation of hBMSCs. (D)
Cell Counting Kit-8 assay showed the cell viability of hBMSCs
during the process of osteogenic differentiation. (E) LINC01535
expression in hBMSCs after treatment with OS medium as measured by
RT-qPCR. *P<0.05 and **P<0.01 vs. 0 day group or as
indicated. OCN, osteocalcin; OSX, osterix; RUNX2, runt-related
transcription factor 2; hBMSCs, human bone marrow-derived
mesenchymal stem cells; ALP, alkaline phosphatase; RT-qPCR, reverse
transcription-quantitative PCR; OD, optical density. |
Silencing of LINC01535 inhibits the
osteogenic differentiation and proliferation of hBMSCs
The present study subsequently aimed to investigate
the role of LINC01535 in osteogenesis. The knockdown transfection
efficiency of LINC01535 was verified using a RT-qPCR assay
(Fig. 2A) and loss-of-function
experiments were then performed. The mRNA and protein expression
levels of OCN, OSX and RUNX2 were markedly downregulated following
LINC01535 knockdown (Fig. 2B and
C). In addition, the effects of LINC01535 knockdown on
osteogenic differentiation were assessed using an ALP activity
assay and the results indicated that LINC01535 knockdown decreased
ALP activity (Fig. 2D). Moreover,
the CCK-8 assay demonstrated that the silencing of LINC01535
significantly inhibited hBMSC proliferation at 72 h (Fig. 2E). Altogether, these results
indicated that LINC01535 knockdown may inhibit hBMSC osteogenic
differentiation and proliferation.
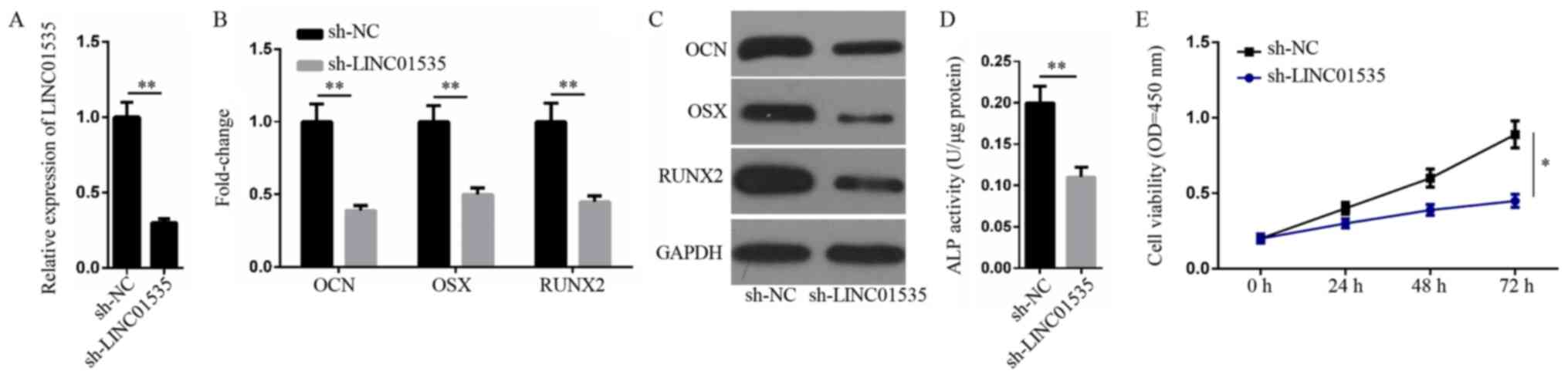 | Figure 2.Silencing of LINC01535 inhibits the
osteogenic differentiation and proliferation of hBMSCs. (A)
Following transfection with sh-NC or sh-LINC01535, RT-qPCR analysis
was performed to assess the transfection efficiency of
sh-LINC01535. The expression of OCN, OSX and RUNX2 at (B) mRNA and
(C) protein levels in hBMSCs was examined by RT-qPCR and western
blotting, respectively, in LINC01535 knockdown hBMSCs. (D) The
function of LINC01535 in hBMSC osteogenic differentiation was
evaluated by detecting ALP activity. (E) The cell viability in
LINC01535-knockdown hBMSCs was assessed using a Cell Counting Kit-8
assay. *P<0.05 and **P<0.01. OCN, osteocalcin; OSX, osterix;
RUNX2, runt-related transcription factor 2; hBMSCs, human bone
marrow-derived mesenchymal stem cells; sh, short hairpin RNA; NC,
negative control; OD, optical density; RT-qPCR, reverse
transcription-quantitative PCR; ALP, alkaline phosphatase. |
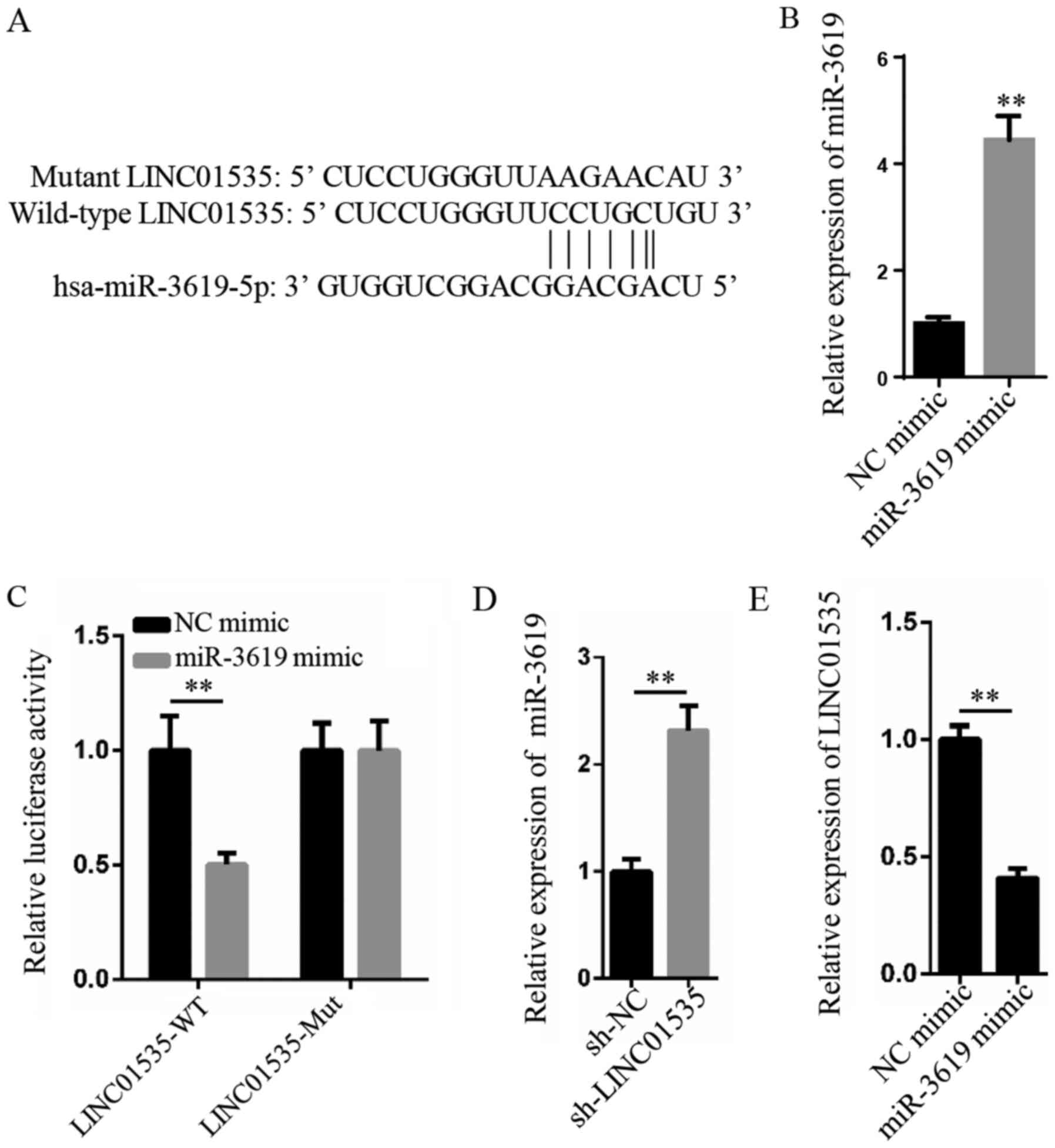
LINC01535 functions as a sponge for
miR-3619-5p
Using bioinformatics analysis, miR-3619-5p was
predicted to have complementary binding sites for LINC01535
(Fig. 3A). To further verify the
relationship between miR-3619-5p and LINC01535, a dual-luciferase
reporter assay was performed. Firstly, RT-qPCR demonstrated that
miR-3619 expression was significantly upregulated in hBMSCs
transfected with miR-3619 mimic compared with NC mimic-transfected
cells (Fig. 3B). Furthermore, the
dual-luciferase reporter assay revealed that miR-3619-5p
overexpression significantly decreased the relative luciferase
activity of the LINC01535-WT vector. Meanwhile, no significant
differences were observed in the relative luciferase activity of
the LINC01535-Mut vector, thus validating the interaction between
LINC01535 and miR-3619-5p (Fig.
3C). In addition, RT-qPCR analysis demonstrated that the
silencing of LINC01535 significantly upregulated miR-3619-5p
expression levels compared with the sh-NC group (Fig. 3D). Simultaneously, the upregulated
expression levels of miR-3619-5p resulted in the downregulated
expression levels of LINC01535 (Fig.
3E). Altogether, these findings suggested that LINC01535 may
interact with miR-3619-5p by direct binding.
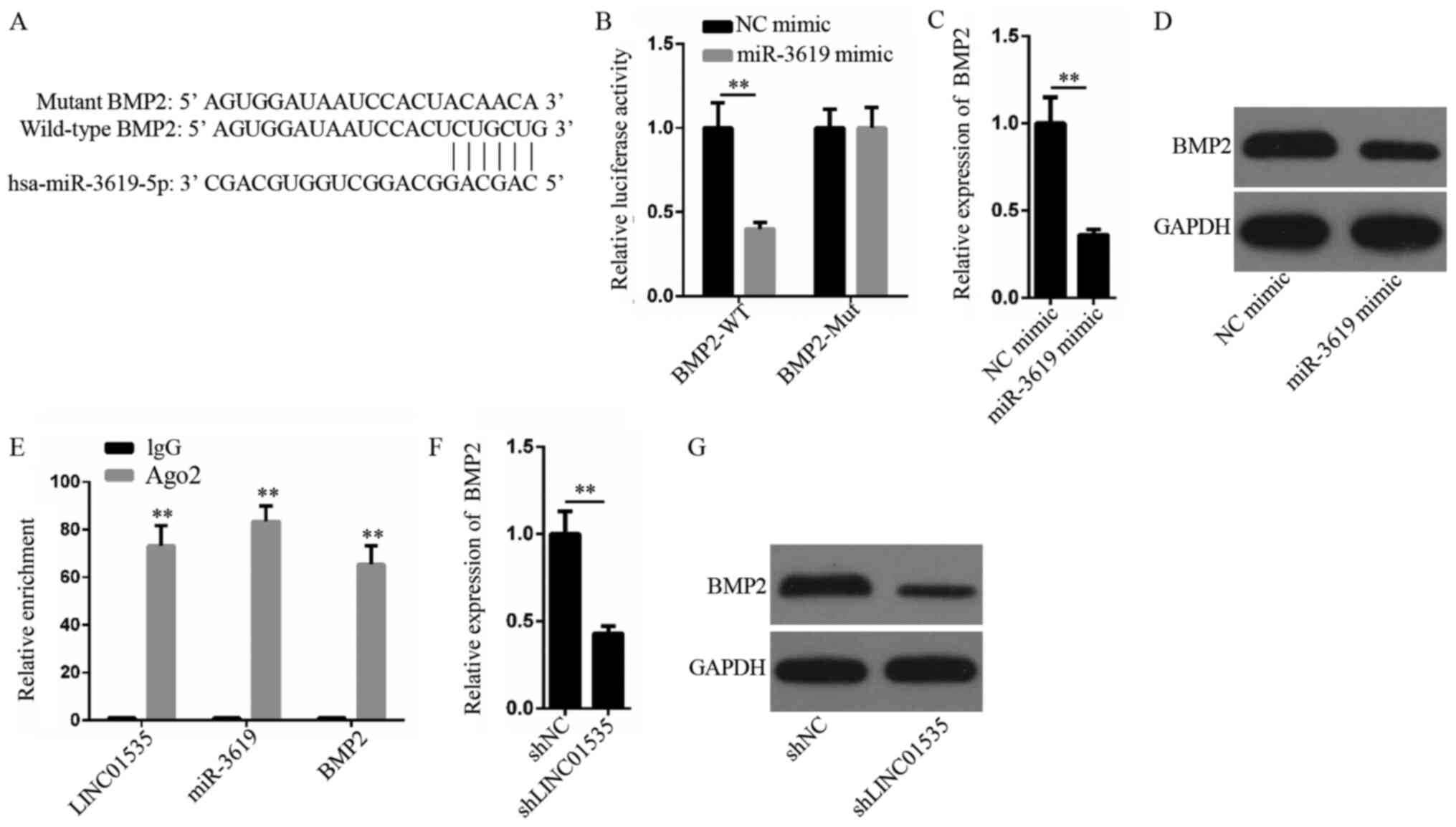
LINC01535 promotes BMP2 expression
levels by competing for miR-3619-5p
The starBase website was used to identify target
genes of miR-3619-5p. The analysis identified a complementary
binding site between BMP2 and miR-3619-5p (Fig. 4A). A luciferase reporter gene assay
further revealed that the relative luciferase activity of the
BMP2-WT vector significantly decreased when co-transfected with the
miR-3619-5p mimic compared with the NC mimic, whereas the
co-transfection with the NC mimic did not affect relative
luciferase activity (Fig. 4B). In
addition, the results demonstrated that the upregulation of
miR-3619-5p expression levels markedly downregulated the BMP2
expression at both mRNA and protein levels (Fig. 4C and D). RIP assay results
illustrated that LINC01535, miR-3619-5p and BMP2 were all
abundantly expressed in the complexes precipitated by the anti-Ago2
antibody (Fig. 4E). In addition,
LINC01535 decreased the mRNA and protein expression levels of BMP2
compared with the sh-NC group (Fig. 4F
and G). Therefore, based on the aforementioned findings, it was
hypothesized that LINC01535 may sponge miR-3619-5p to upregulate
the BMP2 expression levels.
LINC01535 facilitates the osteogenesis
of hBMSCs via the miR-3619-5p/BMP2 axis
Due to the competing nature of LINC01535 over
miR-3619-5p, rescue assays were performed to investigate whether
LINC01535 exerted its role in osteogenic differentiation via the
miR-3619-5p/BMP2 axis. Following transfection with miR-3619
inhibitor and sh-BMP2, the expression levels of miR-3619-5p and
BMP2 were significantly downregulated compared with their
respective NCs (Fig. 5A).
Furthermore, RT-qPCR and western blotting revealed that the
downregulated mRNA and protein expression levels of OCN, OSX and
RUNX2 caused by LINC01535 silencing increased following miR-3619-5p
suppression, while the expression was partially recovered following
BMP2 depletion (Fig. 5B and C).
The ALP activity assay further validated that miR-3619-5p inhibitor
reversed the regulatory impact of LINC01535 knockdown on ALP
activity, while the restoration of osteogenic differentiation
occurred following BMP2 downregulation (Fig. 5D). Moreover, miR-3619-5p inhibition
significantly increased the cell viability repressed by LINC01535
inhibition, while the silencing of BMP2 restored the viability of
hBMSCs at 72 h (Fig. 5E). These
observations suggested that LINC01535 may function as a facilitator
in osteogenesis via modulating the miR-3619-5p/BMP2 axis.
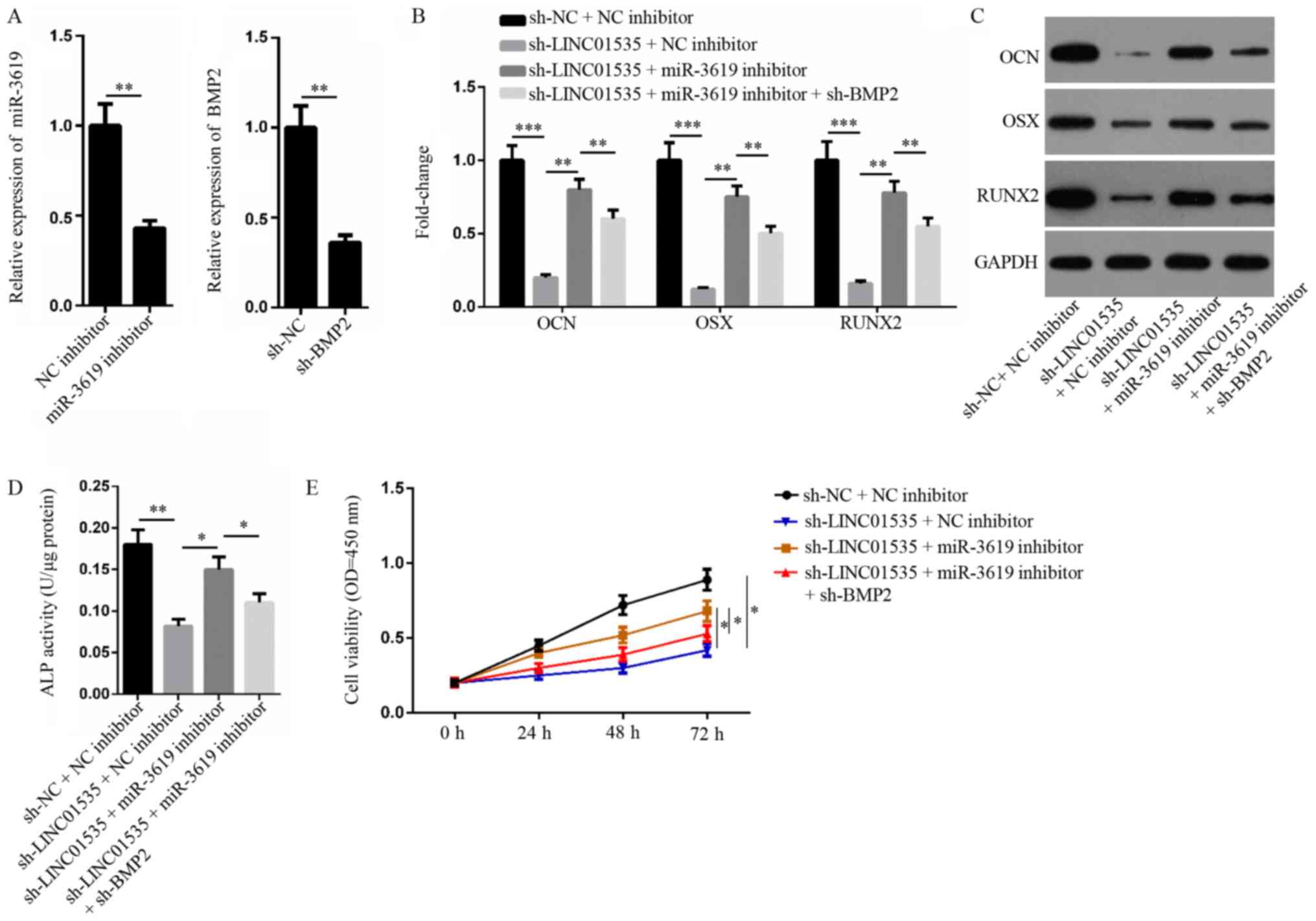 | Figure 5.LINC01535 facilitates the
osteogenesis of hBMSCs via the miR-3619-5p/BMP2 axis. To assess the
role of LINC01535/miR-3619-5p/BMP2 in hBMSC osteogenesis, hBMSCs
were transfected with different vectors to generate four groups:
sh-NC + NC inhibitor, sh-LINC01535 + NC inhibitor, sh-LINC01535 +
miR-3619-5p inhibitor and sh-LINC01535 + miR-3619-5p inhibitor +
sh-BMP2. (A) The inhibition efficiency of miR-3619-5p and sh-BMP2
was examined by RT-qPCR. The (B) mRNA and (C) protein levels of
OCN, OSX and RUNX2 in the four groups were measured by RT-qPCR and
western blotting, respectively. (D) The osteogenic differentiation
of hBMSCs in described groups was evaluated by ALP activity
detection. (E) Cell Counting Kit-8 assay was performed to assess
hBMSC proliferation following transfection with indicated vectors
and reagents. *P<0.05, **P<0.01 and ***P<0.005. miR,
microRNA; RT-qPCR, reverse transcription-quantitative PCR; NC,
negative control; BMP2, bone morphogenetic protein 2; OCN,
osteocalcin; OSX, osterix; RUNX2, runt-related transcription factor
2; hBMSCs, human bone marrow-derived mesenchymal stem cells; sh,
short hairpin RNA; ALP, alkaline phosphatase. |
Discussion
Osteoporosis is a common type of bone metabolic
disorder disease that is attributed to the excess of bone
absorption over bone formation (33,34).
The characteristics of osteoporosis include diminished bone mineral
density, bone loss, microarchitecture destruction and increased
risks of fractures, ultimately leading to bone collapse and
fracture (35,36). Despite significant progress being
made in the treatment of osteoporosis, the underlying mechanisms of
osteoporosis remain poorly understood (37). BMSCs can differentiate into
osteoblasts, which neutralize bone absorption mediated by
osteoclasts (38). Accordingly,
identifying regulators of osteogenic differentiation in BMSCs is of
significance to help shed light on the pathogenesis of
osteoporosis.
An increasing number of investigations have reported
that lncRNAs regulate a multitude of cellular processes that
participate in a diverse range of diseases, including osteoporosis
(29,39,40).
In fact, emerging evidence has identified that lncRNAs participated
in osteoporosis progression (41–43).
For example, the suppression of lncRNA anti-differentiation
noncoding RNA facilitated the osteogenesis of osteoblasts in
postmenopausal osteoporosis via modulating histone-lysine
N-methyltransferase EZH2 and RUNX2 (44); the lncRNA X inactive specific
transcript enhanced osteoporosis via repressing bone marrow
mesenchymal stem cell differentiation (45) and the lncRNA KCNQ1OT1 upregulated
BMP2 expression levels to promote osteogenic differentiation by
competitively binding to miR-214 (31). It was also shown that LINC01535
functioned as an oncogene in the development of cervical cancer
(21). However, to the best of our
knowledge, there are very few investigations into the role and
potential mechanism of LINC01535 in the osteogenic differentiation
of BMSCs. The findings of the present study revealed that LINC01535
was highly expressed during osteogenic induction. Further
functional assays indicated that the silencing of LINC01535
attenuated the growth and osteogenic differentiation of hBMSCs.
lncRNAs serve as natural sponges of miRNAs, which
subsequently interferes with downstream target genes (46–48).
Therefore, the present study hypothesized that LINC01535 may exert
its function in osteogenic differentiation by acting as a sponge
for a specific miRNA. Using the starBase website, a potential
miR-3619-5p binding site was identified in the 3′-UTR of LINC01535.
A luciferase reporter assay subsequently validated the interaction
between LINC01535 and miR-3619-5p. Previous studies reported that
lncRNAs could regulate the progression of various cancers, such as
papillary thyroid carcinoma (49),
hemangioma (50) and
retinoblastoma (51) by targeting
miR-3619. Furthermore, BMP2, as a crucial modulator of osteogenic
differentiation, was confirmed to be a target of miR-3619-5p. BMP2
has been reported to be involved in osteogenic differentiation.
Jiang et al (52) indicated
that miR-204 suppressed the osteogenic differentiation of
mesenchymal stem cells by regulating BMP2. Furthermore, Zhang et
al (53) demonstrated that
overexpression of miR-93-5p inhibited osteogenic differentiation by
downregulating BMP2. In the present study, the results suggested
that LINC01535 may affect the expression levels of BMP2 via
sponging miR-3619-5p. Ultimately, these findings verified that
LINC01535 promoted BMP2 expression to exert its promoting role in
the osteogenic regulation of hBMSCs by competing for
miR-3619-5p.
To the best of our knowledge, the present study was
the first to characterize the expressional pattern and regulatory
function of LINC01535 during the osteogenic differentiation
process. The results suggested that LINC01535 may accelerate the
osteogenic differentiation of hBMSCs via targeting the
miR-3619-5p/BMP2 pathway, which provided an improved understanding
of the pathogenesis of osteoporosis and suggested that the
LINC01535/miR-3619-5p/BMP2 network may represent a latent
therapeutic strategy for the treatment of osteoporosis. However,
the present study has several limitations, which remain to be
addressed; for example, the findings were obtained from in
vitro assays, thus, the role of the LINC01535/miR-3619-5p/BMP2
axis should be further investigated in vivo.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YZ and FW designed the present study. YC and XH
performed all the experiments. NZ and XH analyzed the data and
prepared the figures. YZ drafted the initial manuscript. FW
reviewed and revised the manuscript. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Feng X and McDonald JM: Disorders of bone
remodeling. Annu Rev Pathol. 6:121–145. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mulari MT, Qu Q, Härkönen PL and Väänänen
HK: Osteoblast-like cells complete osteoclastic bone resorption and
form new mineralized bone matrix in vitro. Calcif Tissue Int.
75:253–261. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bennett CN, Longo KA, Wright WS, Suva LJ,
Lane TF, Hankenson KD and MacDougald OA: Regulation of
osteoblastogenesis and bone mass by Wnt10b. Proc Natl Acad Sci USA.
102:3324–3329. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen P, Li Z and Hu Y: Prevalence of
osteoporosis in China: A meta-analysis and systematic review. BMC
Public Health. 16:10392016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Phan TC, Xu J and Zheng MH: Interaction
between osteoblast and osteoclast: Impact in bone disease. Histol
Histopathol. 19:1325–1344. 2004.PubMed/NCBI
|
|
6
|
Pittenger MF, Mackay AM, Beck SC, Jaiswal
RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S and
Marshak DR: Multilineage potential of adult human mesenchymal stem
cells. Science. 284:143–147. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bruder SP, Kurth AA, Shea M, Hayes WC,
Jaiswal N and Kadiyala S: Bone regeneration by implantation of
purified, culture-expanded human mesenchymal stem cells. J Orthop
Res. 16:155–162. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang W, Yang GJ, Wu SX, Li DQ, Xu YB, Ma
CH, Wang JL and Chen WW: The guiding role of bone metabolism test
in osteoporosis treatment. Am J Clin Exp Immunol. 7:40–49.
2018.PubMed/NCBI
|
|
9
|
Xiong L, Jung JU, Wu H, Xia WF, Pan JX,
Shen C, Mei L and Xiong WC: Lrp4 in osteoblasts suppresses bone
formation and promotes osteoclastogenesis and bone resorption. Proc
Natl Acad Sci USA. 112:3487–3492. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Quinn JJ and Chang HY: Unique features of
long non-coding RNA biogenesis and function. Nat Rev Genet.
17:47–62. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Iyer MK, Niknafs YS, Malik R, Singhal U,
Sahu A, Hosono Y, Barrette TR, Prensner JR, Evans JR, Zhao S, et
al: The landscape of long noncoding RNAs in the human
transcriptome. Nat Genet. 47:199–208. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Josefs T and Boon RA: The long non-coding
road to atherosclerosis. Curr Atheroscler Rep. 22:552020.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ghafouri-Fard S, Eghtedarian R and Taheri
M: The crucial role of non-coding RNAs in the pathophysiology of
inflammatory bowel disease. Biomed Pharmacother. 129:1105072020.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chen W, Yang J, Fang H, Li L and Sun J:
Relevance function of linc-ROR in the pathogenesis of cancer. Front
Cell Dev Biol. 8:6962020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Guttman M, Donaghey J, Carey BW, Garber M,
Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L, et al:
lincRNAs act in the circuitry controlling pluripotency and
differentiation. Nature. 477:295–300. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Wang CG, Hu YH, Su SL and Zhong D: lncRNA
DANCR and miR-320a suppressed osteogenic differentiation in
osteoporosis by directly inhibiting the Wnt/β-catenin signaling
pathway. Exp Mol Med. 52:1310–1325. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ma J, Zhang X, Zhang H and Chen H: lncRNA
MEG3 suppresses the progression of ankylosis spondylitis by
regulating the Let-7i/SOST axis. Front Mol Biosci. 7:1732020.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zheng S, Wang YB, Yang YL, Chen BP, Wang
CX, Li RH and Huang D: lncRNA MALAT1 inhibits osteogenic
differentiation of mesenchymal stem cells in osteoporosis rats
through MAPK signaling pathway. Eur Rev Med Pharmacol Sci.
23:4609–4617. 2019.PubMed/NCBI
|
|
19
|
Zhuang W, Ge X, Yang S, Huang M, Zhuang W,
Chen P, Zhang X, Fu J, Qu J and Li B: Upregulation of lncRNA MEG3
promotes osteogenic differentiation of mesenchymal stem cells from
multiple myeloma patients by targeting BMP4 transcription. Stem
Cells. 33:1985–1997. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wei B, Wei W, Zhao B, Guo X and Liu S:
Long non-coding RNA HOTAIR inhibits miR-17-5p to regulate
osteogenic differentiation and proliferation in non-traumatic
osteonecrosis of femoral head. PLoS One. 12:e01690972017.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Song H, Liu Y, Jin X, Liu Y, Yang Y, Li L,
Wang X and Li G: Long non-coding RNA LINC01535 promotes cervical
cancer progression via targeting the miR-214/EZH2 feedback loop. J
Cell Mol Med. 23:6098–6111. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Carthew RW and Sontheimer EJ: Origins and
mechanisms of miRNAs and siRNAs. Cell. 136:642–655. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rana TM: Illuminating the silence:
Understanding the structure and function of small RNAs. Nat Rev Mol
Cell Biol. 8:23–36. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Garzon R, Calin GA and Croce CM: MicroRNAs
in cancer. Annu Rev Med. 60:167–179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
An JH, Ohn JH, Song JA, Yang JY, Park H,
Choi HJ, Kim SW, Kim SY, Park WY and Shin CS: Changes of microRNA
profile and microRNA-mRNA regulatory network in bones of
ovariectomized mice. J Bone Miner Res. 29:644–656. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhang H, Hua Y, Jiang Z, Yue J, Shi M,
Zhen X, Zhang X, Yang L, Zhou R and Wu S: Cancer-associated
fibroblast-promoted lncRNA DNM3OS confers radioresistance by
regulating DNA damage response in esophageal squamous cell
carcinoma. Clin Cancer Res. 25:1989–2000. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kim J, Kim IS, Cho TH, Lee KB, Hwang SJ,
Tae G, Noh I, Lee SH, Park Y and Sun K: Bone regeneration using
hyaluronic acid-based hydrogel with bone morphogenic protein-2 and
human mesenchymal stem cells. Biomaterials. 28:1830–1837. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shu B, Zhang M, Xie R, Wang M, Jin H, Hou
W, Tang D, Harris SE, Mishina Y, O'Keefe RJ, et al: BMP2, but not
BMP4, is crucial for chondrocyte proliferation and maturation
during endochondral bone development. J Cell Sci. 124:3428–3440.
2001. View Article : Google Scholar
|
|
29
|
Zhang N, Hu X, He S, Ding W, Wang F, Zhao
Y and Huang Z: lncRNA MSC-AS1 promotes osteogenic differentiation
and alleviates osteoporosis through sponging microRNA-140-5p to
upregulate BMP2. Biochem Biophys Res Commun. 519:790–796. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Qiao L, Li CG and Liu D: CircRNA_0048211
protects postmenopausal osteoporosis through targeting miRNA-93-5p
to regulate BMP2. Eur Rev Med Pharmacol Sci. 24:3459–3466.
2020.PubMed/NCBI
|
|
31
|
Wang CG, Liao Z, Xiao H, Liu H, Hu YH,
Liao QD and Zhong D: lncRNA KCNQ1OT1 promoted BMP2 expression to
regulate osteogenic differentiation by sponging miRNA-214. Exp Mol
Pathol. 107:77–84. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ahmed SF and Elmantaser M: Secondary
osteoporosis. Endocr Dev. 16:170–190. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Teitelbaum SL: Bone resorption by
osteoclasts. Science. 289:1504–1508. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Li WF, Hou SX, Yu B, Li MM, Férec C and
Chen JM: Genetics of osteoporosis: Accelerating pace in gene
identification and validation. Hum Genet. 127:249–285. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Tu KN, Lie JD, Wan CKV, Cameron M, Austel
AG, Nguyen JK, Van K and Hyun D: Osteoporosis: A review of
treatment options. P T. 43:92–104. 2018.PubMed/NCBI
|
|
37
|
Adler RA: Osteoporosis treatment:
Complexities and challenges. J Endocrinol Invest. 39:719–720. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Clarke B: Normal bone anatomy and
physiology. Clin J Am Soc Nephrol. 3 (Suppl 3):S131–S139. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Liang C, Zhao T, Li H, He F, Zhao X, Zhang
Y, Chu X, Hua C, Qu Y, Duan Y, et al: Long non-coding RNA ITIH4-AS1
accelerates the proliferation and metastasis of colorectal cancer
by activating JAK/STAT3 signaling. Mol Ther Nucleic Acids.
18:183–193. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Li FP, Ling DQ and Gao LY: Long noncoding
RNA Chaer mediated Polycomb Repressor Complex 2 (PRC2) activity to
promote atherosclerosis through mTOR signaling. Eur Rev Med
Pharmacol Sci. 23:7639–7648. 2019.PubMed/NCBI
|
|
41
|
Mei B, Wang Y, Ye W, Huang H, Zhou Q, Chen
Y, Niu Y, Zhang M and Huang Q: lncRNA ZBTB40-IT1 modulated by
osteoporosis GWAS risk SNPs suppresses osteogenesis. Hum Genet.
138:151–166. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shen JJ, Zhang CH, Chen ZW, Wang ZX, Yang
DC, Zhang FL and Feng KH: lncRNA HOTAIR inhibited osteogenic
differentiation of BMSCs by regulating Wnt/β-catenin pathway. Eur
Rev Med Pharmacol Sci. 23:7232–7246. 2019.PubMed/NCBI
|
|
43
|
Zhang RF, Liu JW, Yu SP, Sun D, Wang XH,
Fu JS and Xie Z: lncRNA UCA1 affects osteoblast proliferation and
differentiation by regulating BMP-2 expression. Eur Rev Med
Pharmacol Sci. 23:6774–6782. 2019.PubMed/NCBI
|
|
44
|
Cai N, Li C and Wang F: Silencing of
lncRNA-ANCR promotes the osteogenesis of osteoblast cells in
postmenopausal osteoporosis via targeting EZH2 and RUNX2. Yonsei
Med J. 60:751–759. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Chen X, Yang L, Ge D, Wang W, Yin Z, Yan
J, Cao X, Jiang C, Zheng S and Liang B: Long non-coding RNA XIST
promotes osteoporosis through inhibiting bone marrow mesenchymal
stem cell differentiation. Exp Ther Med. 17:803–811.
2019.PubMed/NCBI
|
|
46
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: A ceRNA hypothesis: The rosetta stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Sun F, Liang W, Tang K, Hong M and Qian J:
Profiling the lncRNA-miRNA-mRNA ceRNA network to reveal potential
crosstalk between inflammatory bowel disease and colorectal cancer.
PeerJ. 7:e74512019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Zhou RS, Zhang EX, Sun QF, Ye ZJ, Liu JW,
Zhou DH and Tang Y: Integrated analysis of lncRNA-miRNA-mRNA ceRNA
network in squamous cell carcinoma of tongue. BMC Cancer.
19:7792019. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang G, Wang X and Jin Y:
LINC01410/miR-3619-5p/FOXM1 feedback loop regulates papillary
thyroid carcinoma cell proliferation and apoptosis. Cancer Biother
Radiopharm. 34:572–580. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu Z, Kang Z, Dai Y, Zheng H and Wang Y:
Long noncoding RNA LINC00342 promotes growth of infantile
hemangioma by sponging miR-3619-5p from HDGF. Am J Physiol Heart
Circ Physiol. 317:H830–H839. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yan G, Su Y, Ma Z, Yu L and Chen N: Long
noncoding RNA LINC00202 promotes tumor progression by sponging
miR-3619-5p in retinoblastoma. Cell Struct Funct. 44:51–60. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Jiang X, Zhang Z, Peng T, Wang G, Xu Q and
Li G: miR-204 inhibits the osteogenic differentiation of
mesenchymal stem cells by targeting bone morphogenetic protein 2.
Mol Med Rep. 21:43–50. 2020.PubMed/NCBI
|
|
53
|
Zhang Y, Wei QS, Ding WB, Zhang LL, Wang
HC, Zhu YJ, He W, Chai YN and Liu YW: Increased microRNA-93-5p
inhibits osteogenic differentiation by targeting bone morphogenetic
protein-2. PLoS One. 12:e01826782017. View Article : Google Scholar : PubMed/NCBI
|