Introduction
Most follicles undergo degeneration during ovarian
follicular development in a process called follicular atresia
(1). It has been reported that
women ovulate ~400 primary oocytes from the 2 million cells present
at birth (2). Commonly, ~20
follicles mature each month, but only one follicle is ovulated, and
the remaining cells undergo follicular atresia (1). Furthermore, diseases such as premature
ovarian insufficiency (POI) are observed with increased follicular
atresia (3). Granulosa cell
apoptosis has been revealed to cause follicular atresia (4,5). It
has been demonstrated that granulosa cells are essential in
determining follicular growth or atresia, and are considered
initial cell populations that undergo apoptosis in atretic
follicles earlier than oocytes and theca cells, indicating their
role as the trigger of follicular atresia (1). It has been proposed that granulosa
cells are in continuous communication with the oocyte (6). Additionally, granulosa cells provide
the oocyte with biological products, including sugars, amino acids
and nucleotides, that sustain its metabolic activity, as well as
signals that regulate its differentiation or maturation (7). Therefore, the regulation of granulosa
cell apoptosis is important for the maintenance of ovarian health.
It has also been reported that oxidative stress induced by
3-nitropropionic acid decreases the number of large follicles,
increases follicular atresia in mouse ovaries and downregulates
SIRT1 expression, leading to FoxO1 activation and granulosa cell
apoptosis (8,9). Furthermore, knockout of mouse double
minute 2 (Mdm2), a major p53-negative regulator, results in a
decline in the number of growing follicles in the ovaries (10,11).
These findings suggest that Mdm2 regulating p53 serves a critical
role in ovary health, ultimately affecting follicular atresia or
POI.
Sirtuin1 (SIRT1) is a NAD(+)-dependent histone
deacetylase protein and is involved in various cellular processes,
including proliferation, inflammation, aging and apoptosis, via its
deacetylase activity (12–17). It has been suggested that SIRT1 acts
as an anti-apoptotic effector in granulosa cell apoptosis (18). p53 is a known tumor suppressor
protein and is a transcription factor regulated by SIRT1 (19). The activity of p53 as a
transcription factor controls the expression of target genes, such
as p21, PUMA and Bax, and induces cell cycle arrest, senescence and
cell death (20,21). Activated p53 can also induce
apoptosis via the Bcl-2-regulated pathway initiated by upregulation
of BH3-only members of the Bcl-2 protein family, including BCL2
like 11 (BIM), p53-upregulated modulator of apoptosis (PUMA) and
phorbol-12-myristate-13-acetate-induced protein 1 (PMAIP1), which
are direct transcriptional targets of p53 (22). PUMA or PMAIP1 directly bind to Bax
or Bcl2 antagonist/killer 1 to increase their proapoptotic
activity. Additionally, p53 has been implicated in granulosa cell
apoptosis (23). However, to the
best of our knowledge, there is no mechanistic information linking
SIRT1 with p53 activity in granulosa cell apoptosis.
The present study aimed to elucidate the regulatory
mechanism of granulosa cell apoptosis. Hydrogen peroxide was used
to generate reactive oxygen species. Flow cytometry and TUNEL
assays were employed to measure cell apoptosis, while small
interfering (si)RNA and short hairpin (sh)RNA against PUMA, PMAIP1
or p53 were used to suppress target gene expression. Furthermore,
immunoblotting and reverse transcription-quantitative (RT-qPCR)
were performed to examine the expression of PARP, caspase-3, p53,
PUMA and PMAIP1.
Materials and methods
Cell lines and reagents
Human granulosa COV434 cells were purchased from
Sigma-Aldrich (Merck KGaA), and were maintained in high glucose
DMEM (Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
(Atlas Biologicals, Inc.), 2 mM L-glutamine (Sigma-Aldrich; Merck
KGaA), 1% penicillin/streptomycin (Thermo Fisher Scientific, Inc.)
at 37°C in a CO2 incubator. 293T cells were purchased
from the American Type Culture Collection, and were maintained in
high glucose DMEM supplemented with 10% FBS, 1%
penicillin/streptomycin at 37°C in a CO2 incubator.
Rabbit anti-SIRT1 (cat. no. ab32441), mouse anti-p53
(cat. no. ab26) and mouse anti-PMAIP1 (cat. no. ab13654) antibodies
(1:2,000) were purchased from Abcam. Rabbit anti-cleaved caspase-3
(cat. no. sc7148; 1:500) antibody was purchased from Santa Cruz
Biotechnology, Inc. Rabbit anti-cleaved poly(ADP-ribose) polymerase
(PARP; cat. no. 9541), rabbit anti-PUMA (cat. no. 4976) and rabbit
anti-acetyl p53 (Lys382; cat. no. 2525) antibodies (1:2,000) were
purchased from Cell Signaling Technology, Inc. HRP-conjugated goat
anti-rabbit IgG polyclonal (cat. no. ADI-SAB-300-J) and
HRP-conjugated goat anti-mouse IgG F(ab′)2, polyclonal (cat. no.
ADI-SAB-100-J) antibodies (1:2,000) were purchased from Enzo Life
Sciences, Inc. Z-VAD-FMK was purchased from Selleck Chemicals.
Mouse anti-β-actin antibody (cat. no. A5441; 1:4,000),
hexadimethrine bromide, H2O2, EX527 and
N-acetyl-L-cysteine (NAC) were purchased from Sigma-Aldrich (Merck
KGaA). SRT1720 was purchased from MedChemExpress.
2′,7′-Dichlorofluorescein diacetate (H2DCFDA), Annexin V-FITC
antibody and PI were purchased from Thermo Fisher Scientific, Inc.
The TUNEL assay kit was purchased from Promega Corporation. The
Transcriptor First Strand cDNA Synthesis kit and FastStart
Essential DNA Green Master were purchased from Roche
Diagnostics.
Plasmid and production of lentiviral
vectors
ON-TARGET plus non-targeting control pool (cat. no.
D-001810-10-05), ON-TARGET plus Human TP53 siRNA-SMART pool (cat.
no. L-003329-00-0005), ON-TARGET plus Human PUMA siRNA-SMART pool
(cat. no. L-004380-00-0005) and ON-TARGET plus Human PMAIP1
siRNA-SMART pool (cat. no. L-005275-00-0005) were purchased from GE
Healthcare Dharmacon, Inc. pMDLg/pRRe (Addgene plasmid no. 12251),
pRSV-Rev (Addgene plasmid no. 12253) and pMD2.G (Addgene plasmid
no. 12259) were gifted by Didier Trono. pLKO.1-luc and SIRT1
MISSION short hairpin (sh)RNA were purchased from Sigma-Aldrich
(Merck KGaA).
A total of 1×106 293T cells were seeded
in 100-mm cell culture dishes. According to Trono Lab protocols,
packaging plasmids 2 µg pMDLg/pRRe and 1 µg pRSV-Rev plus envelope
plasmid 1.2 µg pMD2.G were transfected with each 4 µg SIRT1
knockdown vector or 4 µg pLKO.1-luc control vector into 293T cells
using Effectene transfection reagents (Qiagen GmbH). Following 48 h
of transfection, media containing shRNA-generated lentivirus were
collected, purified via 0.45-µm syringe filters, concentrated using
a Lenti-X™ concentrator (Takara Bio, Inc.) and then titered using a
Lenti-X™ p24 Rapid titer kit (Takara Bio, Inc.). The absorbance was
determined at 450 nm using SPECTROstar® Nano.
Gene silencing using siRNA and
shRNA
COV434 cells (1×106) growing on 60-mm
cell culture dishes were incubated with fresh media with a final
concentration of hexadimethrine bromide at 8 mg/ml to enhance
transduction of most cell types. Lentivirus-mediated SIRT1 shRNA
transduction was performed at a multiplicity of infection of 10.
Following 48 h of transduction, cells were collected and lysed.
For siRNA knockdown, COV434 cells (1×106)
growing on 60-mm cell culture dishes were transfected with 40 nM
non-targeting siRNA or 40 nM p53 siRNA using
Lipofectamine® 3000 (Invitrogen; Thermo Fisher
Scientific, Inc.). Following 48 h of transfection, cells were
collected and lysed.
Flow cytometry analysis
COV434 cells growing on 60-mm dishes were pretreated
with the SIRT1 inhibitor 20 µM EX527 or the SIRT1 activator 1 µM
SRT1720 for 2 h, and then treated with 0.5 mM
H2O2 for 6 h at 37°C. The time gradient was
determined based on recent study (24). Additionally, COV434 cells were
transfected with non-targeting siRNA or p53 siRNA for 48 h, and
then incubated with H2O2 for 6 h at 37°C.
Apoptosis analysis was performed using Annexin V-FITC and PI
according to the manufacturer's instructions (Thermo Fisher
Scientific, Inc.). Data were acquired using FACSCalibur (BD
Biosciences) and were analyzed using BD Cell Quest™ Pro version
6.5.1 (BD Biosciences). The apoptotic rate was calculated using the
percentage of early and late apoptotic cells.
Cellular reactive oxygen species
detection assay
COV434 cells growing on confocal dishes at 80%
confluency were pretreated with 5 mM NAC for 2 h and incubated at
37°C with H2O2 for 2, 4 or 6 h. The
conditioned media were removed, and fresh media containing 10 mM
DCF-DA were added. The cells were incubated at 37°C for 20 min.
After incubation, the conditioned media were removed and washed
with prewarmed PBS. The media were replaced with prewarmed PBS
containing 1% FBS, and the cells were incubated at 37°C for 10 min.
Data analysis was performed using a Zeiss LSM 710 laser-scanning
confocal microscope (magnification, ×20; Zeiss GmbH).
TUNEL assay
COV434 cells (1×106) were seeded on cover
glasses in 6-well plates. After 24 h, cells were pretreated with 20
µM EX527 or 1 µM SRT1720 for 2 h, and then incubated at 37°C with
H2O2 for 6 h. Additionally, COV434 cells were
transfected with 40 nM non-targeting siRNA or 40 nM PMAIP1 siRNA
for 48 h, and then treated with 0.5 mM H2O2
for 6 h at 37°C. Cells were washed with ice-cold Dulbecco's PBS
(DPBS; Welgene, Inc.) and fixed with 4% formaldehyde in DPBS at 4°C
for 25 min. Cells were washed with DPBS for 5 min at room
temperature twice and permeabilized with 0.2% Triton X-100 solution
in DPBS for 5 min. After rinsing the cover glasses using DPBS,
equilibration buffer was added to pre-equilibrate for 10 min at
room temperature. The fragmented DNA of apoptotic cells was stained
with equilibration buffer, nucleotide mix and terminal
deoxynucleotidyl transferase (rTdT) for 1 h at 37°C. 2X SSC was
added to stop the reaction for 15 min at room temperature. After
washing twice, cells were stained with 1:10,000 DAPI in DPBS for 15
min at room temperature. After washing twice, cover glasses were
mounted using mounting medium (Thermo Fisher Scientific, Inc.).
Equilibration buffer, nucleotide mix, rTdT and SSC were contained
in the TUNEL assay kit (Promega Corporation) and their
concentration was determined according to manufacturer's
instructions. Five sections of the cover glass were imaged at ×20
magnification. Data analysis was performed using a laser-scanning
confocal microscope Zeiss LSM 710 (Zeiss GmbH).
Immunoblot analysis
COV434 cells (1×106) grown on 60 mm
dishes were pretreated with 5 mM NAC, 50 µM Z-VAD-FMK, 20 µM Ex527
or DMSO at 37°C for 2 h, and then treated with 0.5 mM
H2O2 for 6 h at 37°C. For immunoblotting,
COV434 cells were harvested and lysed using lysis buffer containing
0.5% Triton X-100, 1 mM EDTA, 50 mM Tris-HCl, pH 7.4 and 40 mM
NaCl. Cell lysates were diluted at 1:1,000 in protein assay dye
reagent (Bio-Rad Laboratories, Inc.) and densitometry was measured
at 595 nm by SPECTROstar®Nano. Briefly, 30 µg cell
lysate was separated via 10, 12 or 16% SDS-PAGE and transferred to
nitrocellulose membranes (Bio-Rad Laboratories, Inc.). The
membranes were blocked with 5% skim milk in TBS with 1% Tween 20
(TBST) for 1 h at room temperature, and then incubated with
anti-p53, acetyl p53, cleaved PARP, cleaved caspase-3, SIRT1, PUMA,
PMAIP1 or β-actin antibodies at 4°C overnight. The membranes were
washed three times using TBST and then incubated with secondary
antibodies, including goat anti-rabbit polyclonal antibody and goat
anti-mouse polyclonal antibody at room temperature for 1 h. After
washing, the blot was detected with a western detection kit
(AbClon, Inc.), according to the manufacturer's instructions, and
X-ray film. Protein expression was analyzed using ImageJ 1.52t
(National Institutes of Health).
RT-qPCR
To quantify p53, BAX, BIM, PMAIP1, PUMA and
GAPDH mRNA expression, cells were lysed with
TRIzol® reagent (Ambion, Inc.). Total RNA (2 µg) was
synthesized into cDNA using the Transcriptor First Strand cDNA
Synthesis kit (Roche Diagnostics) according to the manufacturer's
instructions. qPCR was performed using FastStart Essential DNA
Green Master mix (Roche Diagnostics). Data were obtained using a
LightCycler® 96 Instrument (Roche Diagnostics) and
consisted of three programs: Preincubation, 3-step amplification
and melting. The ‘preincubation’ program was set to a temperature
of 95°, ramp of 4.4°C/sec, duration of 600 sec and no acquisition
mode. The ‘3-step amplification’ program was set to a temperature
of 95°C for 10 sec, 60°C for 10 sec and 72°C for 10 sec, and the
number of cycles was 40. The ‘melting’ program was set to 95°C for
10 sec, 65°C for 60 sec and 97°C for 1 sec. Data analysis was
performed using LightCycler® 96 SW 1.1 (Roche
Diagnostics). Relative gene expression data was analyzed with the
2−∆∆Cq method (25). All
primers were purchased from Bioneer Corporation, and their
sequences were as follows: p53 forward,
5′-AGGCCTTGGAACTCAAGGAT-3′ and reverse, 5′-CCCTTTTTGGACTTCAGGTG-3′;
BAX forward, 5′-TTGCTTCAGGGTTTCATCCA-3′ and reverse,
5′-AGACACTCGCTCAGCTTCTTG−3′; BIM forward,
5′-CAAGTTCAAGCGGTTCTCCT-3′ and reverse, 5′-CAGCCTGCCTCATGGAAG−3′;
PUMA forward, 5′-GACGACCTCAACGCACAGTA-3′ and reverse,
5′-AGGAGTCCCATGATGAGATTGT-3′; PMAIP1 forward,
5′-GGAGATGCCTGGGAAGAAG-3′ and reverse, 5′-CCTGAGTTGAGTAGCACACTCG-3;
and GAPDH forward, 5′-AGCCACATCGCTCAGACAC-3′ and reverse,
5′-GCCCAATACGACCAAATCC-3′.
Statistical analysis
Data are presented as the mean ± SD of ≥3
independent experiments in duplicate. Statistical analysis was
performed using one-way ANOVA with Tukey's test or two-way ANOVA
followed by Bonferroni's correction. P<0.05 were considered to
indicate a statistically significant difference.
Results
H2O2-induced ROS
production results in COV434 granulosa cell apoptosis and decreases
SIRT1 expression
The effect of H2O2 on COV434
granulosa cell apoptosis was examined, and it was identified that
H2O2 treatment induced granulosa cell
apoptosis dose-dependently (Fig.
1A). Next, it was investigated whether endogenous ROS were
generated by H2O2 treatment in granulosa
cells. COV434 cells were treated with or without
H2O2 for 2, 4 or 6 h. The results indicated
that endogenous ROS levels were slightly increased following 2 h of
H2O2 treatment, but elevated ~1.5- or 2-fold
after 4 or 6 h of H2O2 treatment,
respectively (Fig. 1B).
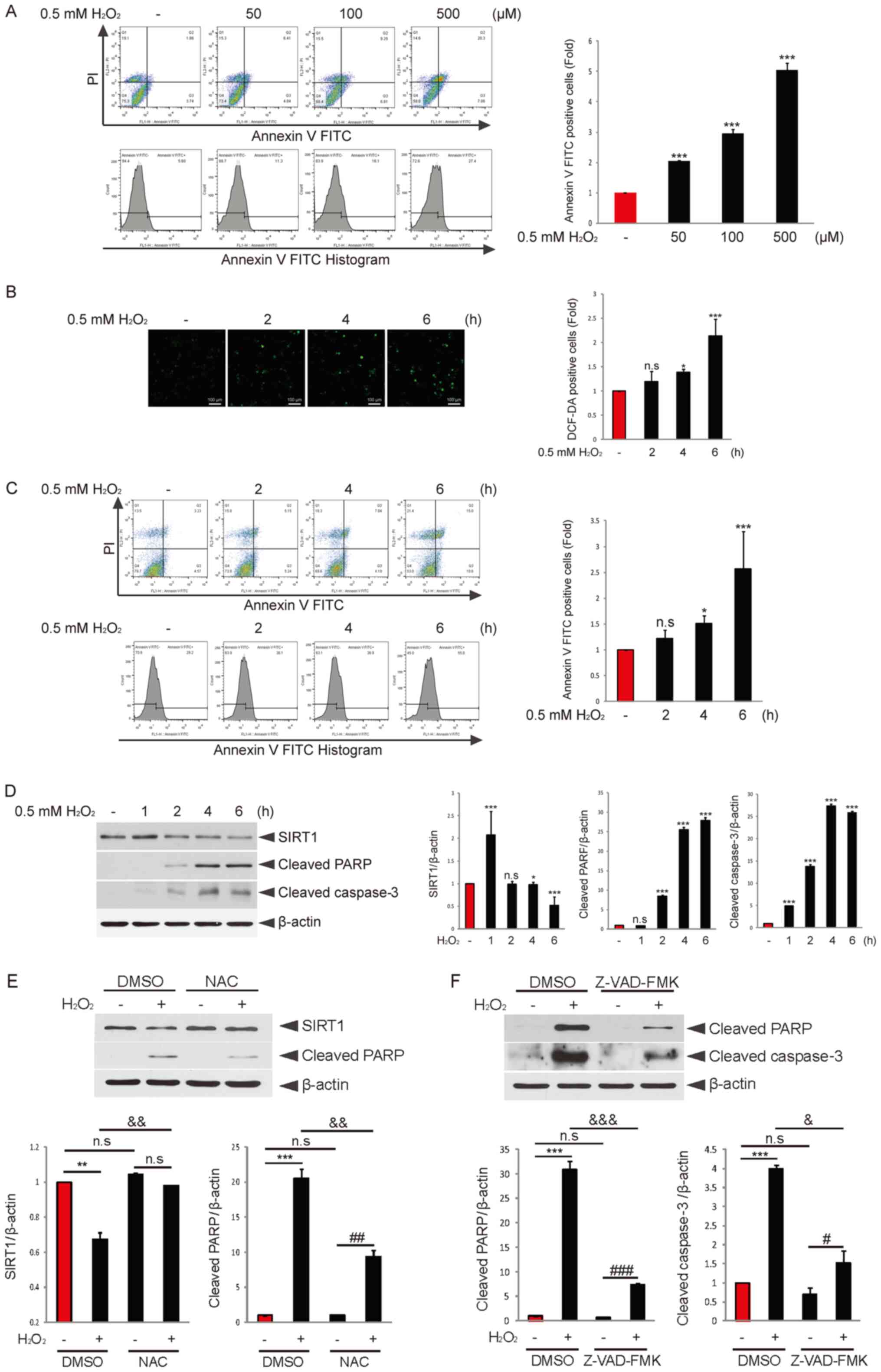 | Figure 1.Granulosa cell apoptosis is induced
time-dependently, and endogenous SIRT1 expression is decreased upon
H2O2 treatment. (A) COV434 cells were
incubated with or without H2O2 for 6 h at the
indicated concentrations. Apoptosis analysis was performed using
flow cytometry. Quantitation of Annexin V-positive cells is
presented. (B) COV434 cells were incubated with or without
H2O2 for the indicated times. DCF-DA (10 mM)
was added and incubated for 20 min. DCF fluorescence was determined
using flow cytometry. Quantitation of DCF-DA-positive cells was
conducted (scale bar, 100 µm). (C) COV434 cells were treated with
or without H2O2 for the indicated times.
Apoptosis analysis was performed using flow cytometry. (D) COV434
cells were incubated with or without H2O2 for
the indicated times. Immunoblotting using anti-SIRT, anti-cleaved
PARP, anti-cleaved caspase-3 and-actin antibodies was performed.
*P<0.05, ***P<0.001 vs. without H2O2
group. (E) COV434 cells were pretreated with DMSO or NAC (5 mM) for
2 h and then incubated with or without H2O2
for 6 h. Immunoblotting using SIRT1 or cleaved PARP antibody was
performed. (F) COV434 cells were pretreated with DMSO or Z-VAD-FMK
for 2 h and then incubated with or without
H2O2 for 6 h. Immunoblotting using cleaved
PARP or caspase-3 antibody was conducted. Representative images of
three independent experiments are shown. **P<0.01,
***P<0.001; #P<0.05, ##P<0.01,
###P<0.001; &P<0.05,
&&P<0.01,
&&&P<0.001 (two-way ANOVA with
Bonferroni's correction). n.s. non-significant; NAC,
N-acetyl-L-cysteine; SIRT1, sirtuin 1; PARP, poly(ADP-ribose)
polymerase; H2DCFDA, 2′,7′-Dichlorofluorescein diacetate. |
Subsequently, the effect of oxidative stress on
COV434 cell apoptosis was evaluated. COV434 cells were incubated
with or without H2O2 for 2, 4 or 6 h, and
apoptotic cells were increased ~2-fold after 6 h of
H2O2 treatment (Fig. 1C). Furthermore, the expression
levels of cleaved PARP or caspase-3 were significantly increased by
H2O2 treatment in a time-dependent manner
(Fig. 1D). It has been suggested
that SIRT1 provides resistance to apoptosis induced by oxidative
stress (26). As expected, SIRT1
expression was decreased by H2O2 treatment
(Fig. 1D), which may affect
H2O2-induced granulosa cell apoptosis.
The effective antioxidant NAC was used to block
H2O2 action. To investigate the effect of NAC
on cleaved PARP or SIRT1 expression levels regulated by
H2O2, COV434 cells were pretreated with NAC
for 2 h and then incubated with or without
H2O2 for 6 h. Cotreatment with NAC
effectively restored H2O2-downregulated SIRT1
expression and attenuated H2O2-upregulated
PARP expression compared with the H2O2 only
group (Fig. 1E). Moreover, it was
evaluated whether H2O2-induced cell death was
implicated in the caspase-dependent apoptotic pathway. Cotreatment
with Z-VAD-FMK, a well-known pan caspase inhibitor, significantly
decreased the expression levels of cleaved PARP and caspase-3
upregulated by H2O2 compared with the
H2O2 only group (Fig. 1F). These findings suggested that
H2O2-induced ROS production resulted in
granulosa cell apoptosis in a caspase-dependent manner, and that
SIRT1 may serve a key role in oxidative stress-induced granulosa
cell apoptosis.
Inhibition of SIRT1 expression or
activity promotes H2O2-induced granulosa cell
apoptosis and p53 acetylation
It has been reported that SIRT1 deacetylates p53,
thereby suppressing stimuli-induced apoptosis (27,28).
Based on these studies, it was examined whether regulation of SIRT1
activity or expression affects p53 activity to contribute to
H2O2-induced granulosa cell apoptosis. COV434
cells were cotreated with or without H2O2
plus the SIRT1 inhibitor EX527. H2O2
treatment decreased SIRT1 expression, and increased cleaved PARP
and acetylated p53 expression levels (Fig. 2A). In addition, cotreatment with
EX527 resulted in a higher increase in PARP and acetylated p53
expression levels, but only affected SIRT1 expression slightly.
Knockdown of SIRT1 expression via lentivirus-mediated SIRT1 shRNA
abolished SIRT1 expression and enhanced the expression levels of
cleaved PARP and acetylated p53 induced by
H2O2 treatment compared with the control
shRNA group (Fig. 2B).
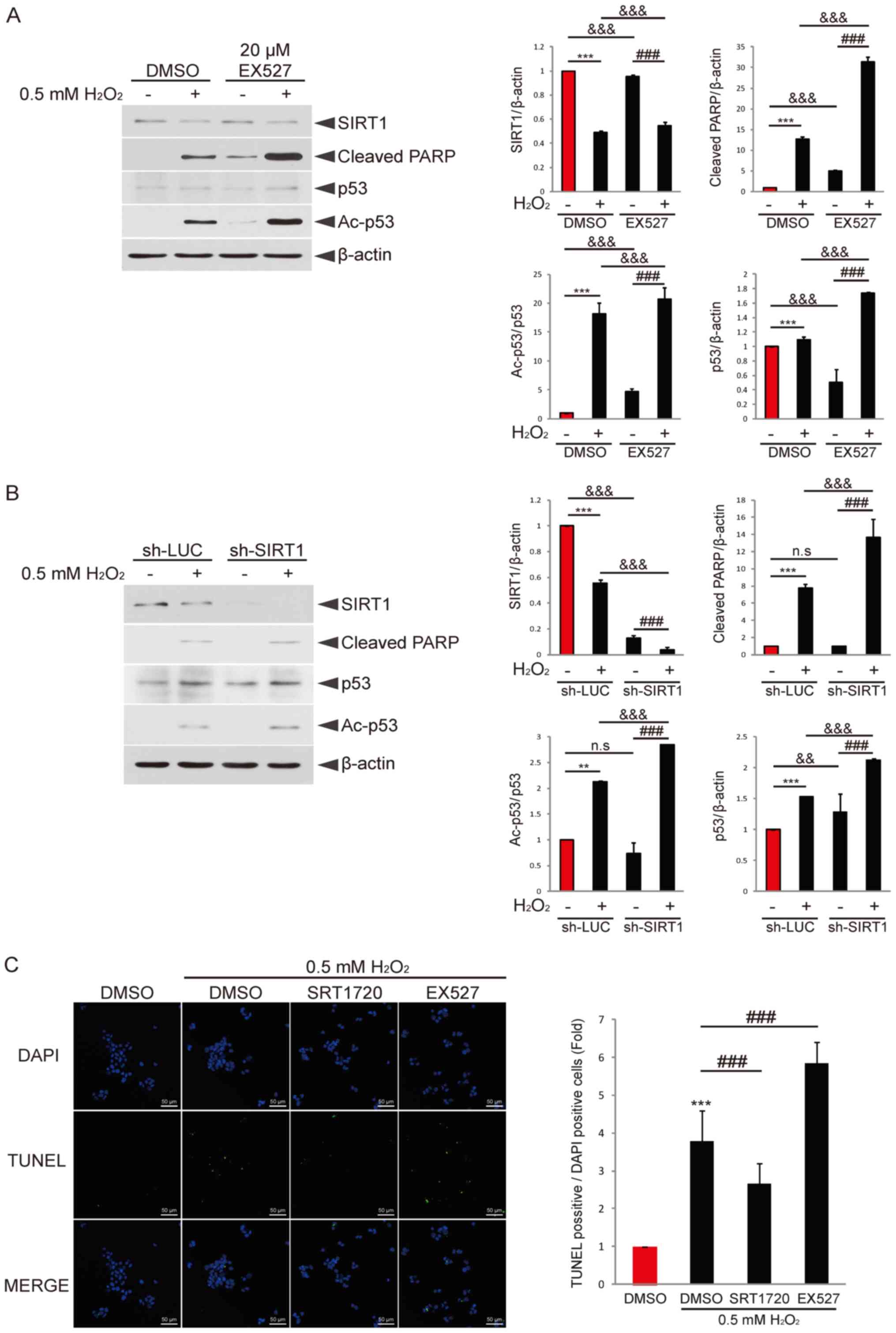 | Figure 2.Regulation of SIRT1 activity affects
H2O2-induced p53 acetylation and granulosa
cell apoptosis. (A) COV434 cells were pretreated with DMSO or EX527
for 2 h and then incubated with or without
H2O2 for 6 h. Immunoblotting using
anti-SIRT1, anti-cleaved PARP, anti-p53, anti-Ac-p53 and β-actin
antibodies was performed. ***P<0.001 vs. DMSO;
###P<0.001 vs. EX527;
&&&P<0.001 (two-way ANOVA with
Bonferroni's correction). (B) COV434 cells were transduced with
sh-LUC or sh-SIRT1 for 48 h and then treated with or without
H2O2 for 6 h. Immunoblotting was performed
using the indicated antibody. **P<0.01, ***P<0.001;
###P<0.001; &&&P<0.001
(two-way ANOVA with Bonferroni's correction). (C) COV434 cells were
pretreated with DMSO, EX527 or SRT1720 for 2 h and then incubated
with or without H2O2 for 6 h. Cells were
fixed and then analyzed for apoptosis via TUNEL assay. Nuclei were
stained with DAPI. The ratio of TUNEL-positive cells to
DAPI-positive cells is presented. Representative images of three
independent experiments are provided (scale bar, 50 µm).
***P<0.001 vs. DMSO; ###P<0.001. N.S.,
non-significant; sh-LUC, lentivirus-mediated LUC shRNA; sh-SIRT1,
SIRT1 shRNA; SIRT1, sirtuin 1; PARP, poly(ADP-ribose) polymerase;
Ac, acetylated. |
Next, the effect of a SIRT1 inhibitor or activator
on H2O2-induced granulosa cell apoptosis was
investigated. The results demonstrated that cotreatment with the
SIRT1 activator SRT1720 diminished
H2O2-induced granulosa cell apoptosis
(Fig. 2C). By contrast, cotreatment
with EX527 enhanced apoptosis. These results indicated that the
expression level or activity of SIRT1 may be a key factor that
determines H2O2-induced granulosa cell
apoptosis. Moreover, it was suggested that p53 activity regulated
by SIRT1 activity or expression may contribute to apoptosis.
Knockdown of p53 decreases PUMA and
PMAIP1 expression levels and granulosa cell apoptosis induced by
H2O2
To examine whether p53 serves a key role in
H2O2-induced granulosa cell apoptosis, each
control siRNA or p53 siRNA was expressed in COV434 cells. The
results indicated that knockdown of p53 attenuated granulosa cell
apoptosis induced by H2O2 treatment (Fig. 3A). Next, to investigate the mRNA
expression levels of p53 and its target genes BAX, BIM, PUMA and
PMAIP1, COV434 cells were transfected with control siRNA or p53
siRNA. Following 48 h of transfection, cells were incubated with or
without H2O2 for 6 h. RT-qPCR analysis
demonstrated that the mRNA expression levels of PUMA, PMAIP1 and
BAX induced by H2O2 were significantly
decreased by knockdown of p53 compared with the control siRNA +
H2O2 group. Furthermore, p53 expression was
slightly decreased by p53 knockdown compared with the control siRNA
+ H2O2 group. Whereas, BIM expression was
slightly increased by p53 knockdown compared with the control siRNA
+ H2O2 group (Fig. 3B). To confirm these findings,
immunoblotting analysis was performed. The results indicated that
knockdown of p53 significantly decreased the expression levels of
p53, PUMA, PMAIP1 and cleaved PARP, and reduced SIRT1 expression
slightly, compared with the control siRNA group (Fig. 3C). Thus, it was suggested that p53
may be a critical effector of H2O2-induced
granulosa cell apoptosis, and that PUMA and PMAIP1 may be
implicated in the regulation of apoptosis.
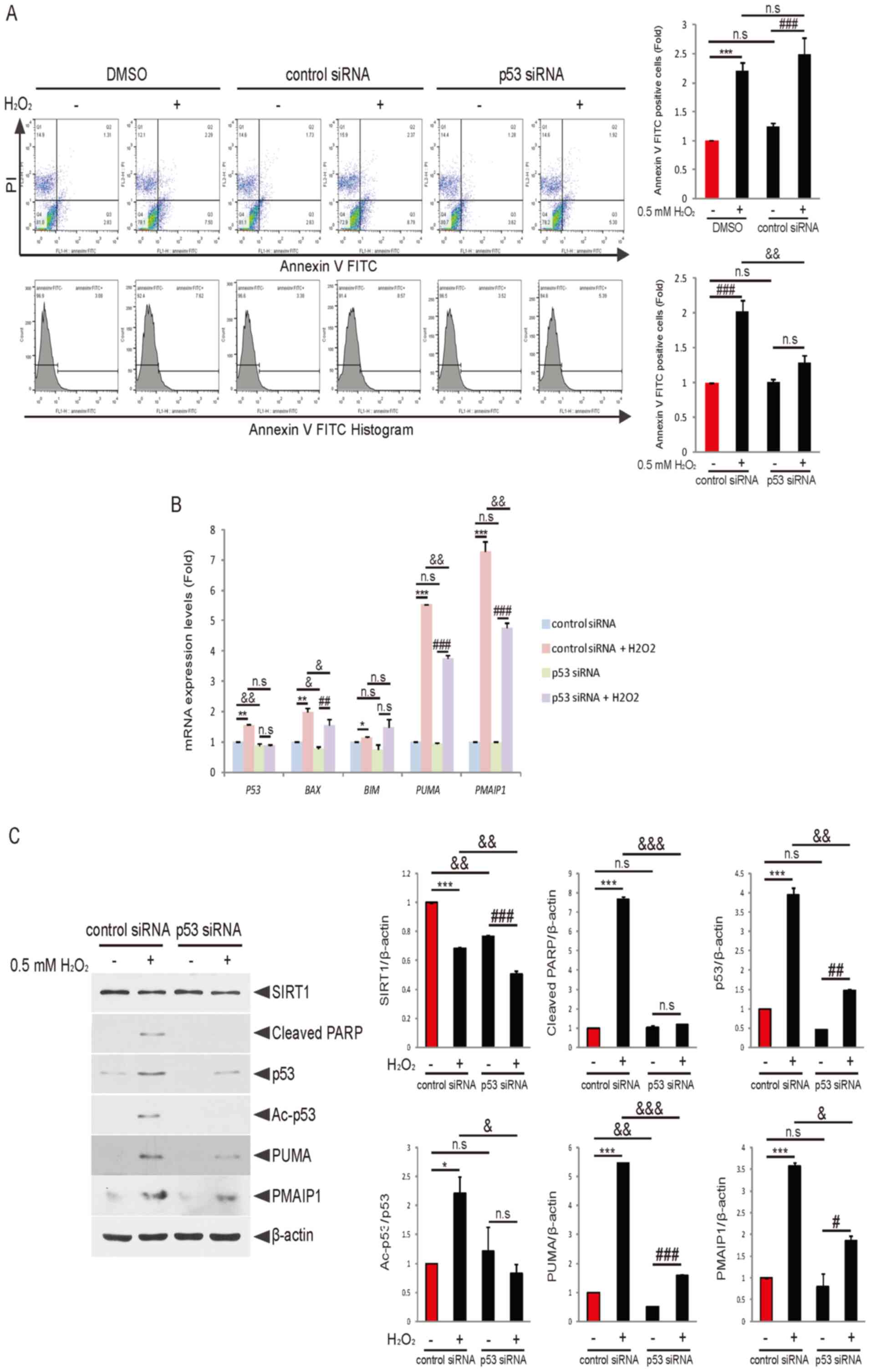 | Figure 3.Knockdown of p53 attenuates
H2O2-induced PUMA and PMAIP1 expression
levels and granulosa cell apoptosis. (A) COV434 cells were treated
with DMSO or transfected with control siRNA or p53 siRNA. Following
48 h of transfection, cells were incubated with or without
H2O2 for 6 h and then analyzed for apoptosis
using flow cytometry. Quantification of Annexin V-positive cells is
presented. ***P<0.001; ###P<0.001;
&&P<0.01 (two-way ANOVA with Bonferroni's
correction). (B) COV434 cells were transfected with control siRNA
or p53 siRNA for 48 h, and then cells were treated with or without
H2O2 for 6 h. Reverse
transcription-quantitative PCR analysis was performed using the
indicated primers. The relative mRNA expression normalized to GAPDH
expression is displayed. *P<0.05, **P<0.01, ***P<0.001;
##P<0.01, ###P<0.001;
&P<0.05, &&P<0.01 (two-way
ANOVA with Bonferroni's correction). (C) COV434 cells were
transfected with control siRNA or p53 siRNA. Following 48 h of
transfection, cells were treated with or without
H2O2 for 6 h. Protein expression levels of
SIRT1, cleaved PARP, p53, Ac-p53, PUMA and PMAIP1 were estimated
using immunoblotting. Representative results of three independent
experiments are shown. *P<0.05, ***P<0.001;
#P<0.05, ##P<0.01,
###P<0.001; &P<0.05,
&&P<0.01,
&&&P<0.001 (two-way ANOVA with
Bonferroni's correction). N.S., non-significant; Ac, acetylated;
SIRT1 shRNA; SIRT1, sirtuin 1; PARP, poly(ADP-ribose) polymerase;
siRNA, small interfering RNA; BIM, Bcl2 like 11; PUMA,
p53-upregulated modulator of apoptosis; PMAIP1,
phorbol-12-myristate-13-acetate-induced protein 1. |
Knockdown of PUMA or PMAIP1 attenuates
H2O2-induced granulosa cell apoptosis
As indicated by the results, PUMA and PMAIP1 are
potential targets for p53-mediated granulosa cell apoptosis upon
H2O2 treatment. To examine whether knockdown
of PUMA or PMAIP1 affects H2O2-induced
granulosa cell apoptosis, COV434 cells were transfected with PUMA
or PMAIP1 siRNA for 48 h and then treated with
H2O2 for 6 h. Knockdown of PUMA decreased
both cleaved PARP expression (Fig.
4A) and the apoptosis (Fig. 4B)
induced by H2O2 compared with each control
siRNA group. Additionally, the transfection of PMAIP1 siRNA
significantly decreased H2O2-induced cleavage
PARP expression (Fig. 4C) and
apoptosis (Fig. 4D). Collectively,
the results suggested that PUMA and PMAIP1 mediated
H2O2-induced granulosa cell apoptosis via the
SIRT1/p53 axis.
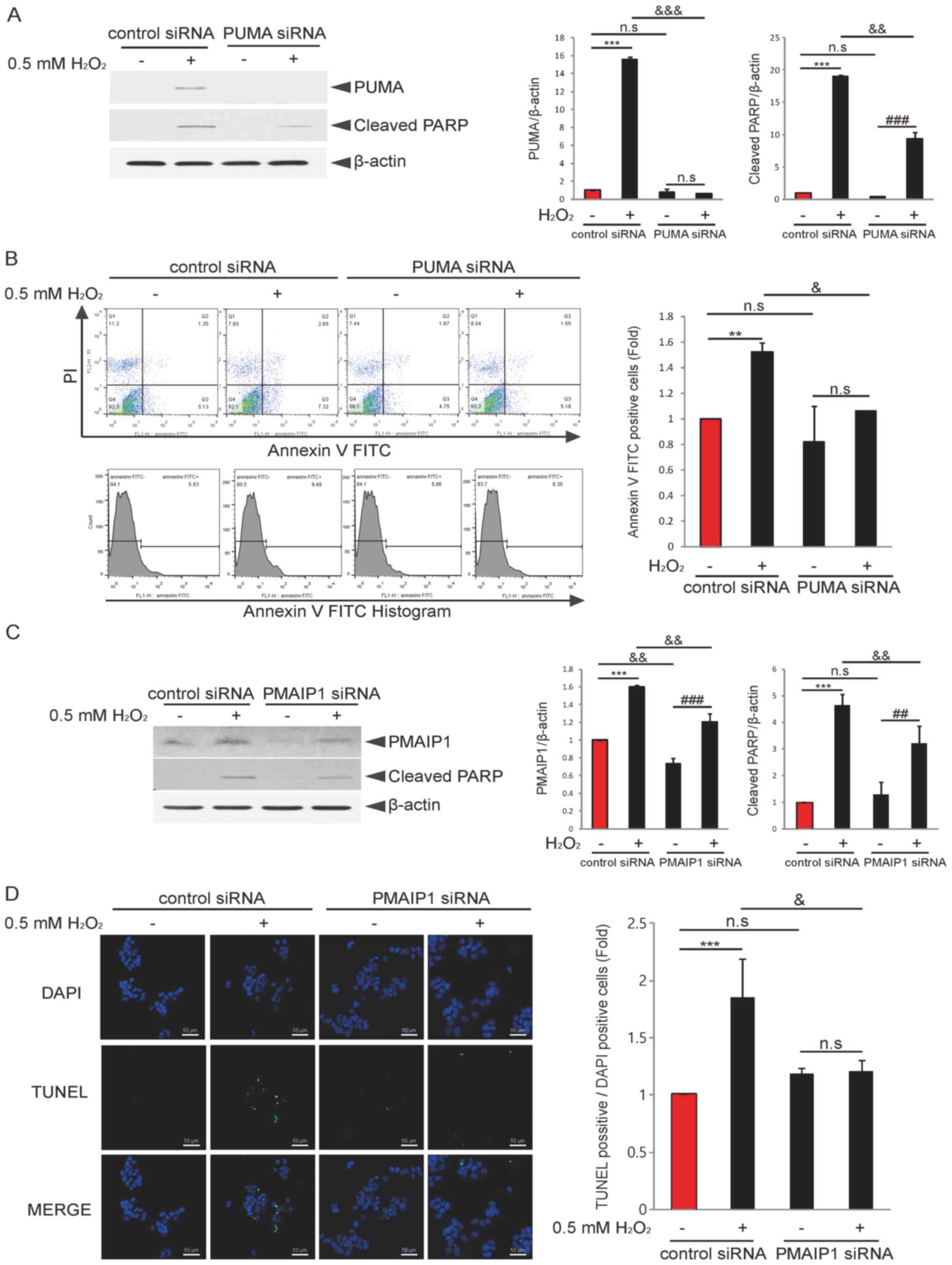 | Figure 4.Knockdown of PUMA or PMAIP1 decreases
H2O2-induced granulosa cell apoptosis. (A)
COV434 cells were transfected with control siRNA or PUMA siRNA for
48 h and then treated with or without H2O2
for 6 h. Immunoblotting using PUMA or cleaved PARP antibodies was
performed. ***P<0.001; ###P<0.001;
&&P<0.01,
&&&P<0.001 (two-way ANOVA with
Bonferroni's correction). (B) COV434 cells were transfected with
control siRNA or PUMA siRNA for 48 h and then incubated with or
without H2O2 for 6 h. Cells were subjected to
flow cytometry for apoptosis analysis. Quantitation of Annexin
V-positive cells is presented. **P<0.01;
&P<0.05 (two-way ANOVA with Bonferroni's
correction). (C) COV434 cells were transfected with control siRNA
or PMAIP1 siRNA for 48 h and then treated with or without
H2O2 for 6 h. Immunoblotting using PMAIP1 or
cleaved PARP antibodies was performed. ***P<0.001;
##P<0.01, ###P<0.001;
&&P<0.01 (two-way ANOVA with Bonferroni's
correction). (D) COV434 cells were transfected with control siRNA
or PMAIP1 siRNA for 48 h and then incubated with or without
H2O2 for 6 h. Cells were subjected to TUNEL
assay for apoptosis analysis. Nuclei staining was performed with
DAPI. The ratio of TUNEL-positive cells to DAPI-positive cells is
indicated, as well as representative results of three independent
experiments (scale bar, 50 µm). ***P<0.001;
&P<0.05 (two-way ANOVA with Bonferroni's
correction). N.S., non-significant; PARP, poly(ADP-ribose)
polymerase; siRNA, small interfering RNA; BIM, BCL2 like 11; PUMA,
p53-upregulated modulator of apoptosis; PMAIP1,
phorbol-12-myristate-13-acetate-induced protein 1. |
Discussion
A previous study suggested that the apoptotic rate
of granulosa cells tended to be higher in patient who are infertile
compared with a control group (29). Additionally, the incidence of
granulosa cell apoptosis was higher in patients with <6 oocytes
compared with patients with ≥6 oocytes, and was also increased in
patients who are not pregnant compared with patients who are
pregnant (30). Therefore, the
regulation of granulosa cell apoptosis appears to be critical, and
accumulating evidence indicates that factors associated with
granulosa cell apoptosis can act as targets for clinical
applications (2,31). It has also been reported that
granulosa cell apoptosis is increased in a SIRT1 knockdown group
(18,32). Additionally, SIRT1 has been proposed
to protect oocytes, and the activity of SIRT1 can preserve oocyte
capacity in oxidative stress-mediated ovarian dysfunction,
including aging (33). Consistent
with these studies, the present results indicated that SIRT1
knockdown accelerated oxidative stress-induced COV434 granulosa
cell apoptosis. The human COV434 granulosa cell line was used in
the current study. It has been suggested that COV434 cells have
three distinct properties that are essential in granulosa cells,
including: Production of 17β-estradiol in response to follicle
stimulating hormone, the presence of specific molecular markers of
apoptosis, such as bcl-2/adenovirus E1B 19 kDa interacting protein
(Nip)1, Nip2, bcl-2 homologous antagonist/killer and Bax, enabling
the induction of follicular atresia and the capacity to form
intercellular connections with cells surrounding an oocyte
(34).
It has been reported that p53 mediates granulosa
cell apoptosis and that p53 induction in granulosa cells is harmful
to ovarian function by disrupting oocyte quality (10,35).
In addition, PUMA and PMAIP1 are downstream targets of p53 and
BH3-only proteins that inhibit the pro-survival Bcl2 family
(36). PUMA knockout mice retain
primordial follicles and preserve their reproductive function in
DNA-damaging conditions compared with wild-type mice, and
primordial follicular oocytes from PUMA or PUMA/PMAIP1
double-knockout mice are protected from DNA damage-induced
apoptosis and can produce healthy offspring (37,38).
Collectively, these studies demonstrate that p53 and its targets,
PUMA and PMAIP1, are critical effectors regulating granulosa cell
apoptosis.
ROS-mediated oxidative stress can induce apoptosis
and is associated with several diseases, including cancer, diabetes
and liver fibrosis (39). It has
been suggested that under oxidative stress conditions, SIRT1
regulates the activity of the transcription factor p53, a tumor
suppressor and inducer of apoptosis, and the FOXO family to affect
cell survival or apoptosis (19).
Based on these reports, the present study aimed to elucidate the
mechanism linking SIRT1 with p53 in oxidative stress-induced
granulosa cell apoptosis. The current results indicated that p53
acetylation upregulated by SIRT1 suppression upon
H2O2 treatment promoted granulosa cell
apoptosis. Moreover, it has been reported that cytoplasmic p53
deacetylation by ROS-activated SIRT1 in mouse embryonic stem cells
suppresses p53 nuclear translocation, leading to p53 translocation
into mitochondria, its binding to Bcl2 and cytochrome c
release, suggesting a role for SIRT1 in p53
transcription-independent apoptosis (40). Taken together, these findings
demonstrate that oxidative stress may induce cell type-specific
apoptosis by activating or inactivating SIRT1 in a p53
transcription-dependent or p53 transcription-independent manner,
indicating the significance of SIRT1 activity against p53.
In conclusion, the present study demonstrated that
SIRT1 downregulation by H2O2 treatment
promoted p53 activity, thereby accelerating granulosa cell
apoptosis. The findings of this study improve the understanding of
the mechanistic role of the SIRT1/p53 axis in the regulation of
oxidative stress-induced granulosa cell apoptosis, and suggest the
potential use of SIRT1 activators as a strategy to inhibit
granulosa cell apoptosis. However, the present study did not
identify the detailed underlying molecular mechanism via which the
SIRT1/p53 regulatory axis regulates granulosa cell apoptosis. Thus,
future studies will aim to investigate novel target proteins
regulated by SIRT1 in the H2O2 response, as
well as examine the effect of p53 post-translational modification
on oxidative stress-induced granulosa cell apoptosis.
Acknowledgements
Not applicable.
Funding
This work was supported by National Research
Foundation of Korea (NRF) grants funded by the Korean government
(grant no. NRF-2019R1I1A3A01063191). This work was also supported
by the Priority Research Centers Program through the NRF (grant no.
NRF-2017R1A6A1A03015713).
Availability of data and materials
All data generated or analyzed during the present
study are included in this published article.
Authors' contributions
SAP and NRJ conducted the experiments, collected the
data and analyzed the data. JHP analyzed the data. SMO designed and
supervised the project. SAP, NRJ and SMO wrote the manuscript. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Matsuda F, Inoue N, Manabe N and Ohkura S:
Follicular growth and atresia in mammalian ovaries: Regulation by
survival and death of granulosa cells. J Reprod Dev. 58:44–50.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Zhao Z, Shi H, Li J, Zhang Y, Chen C and
Guo Y: Cumulative live birth rates according to the number of
oocytes retrieved following the ‘freeze-all’ strategy. Reprod Biol
Endocrinol. 18:142020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vabre P, Gatimel N, Moreau J, Gayrard V,
Picard-Hagen N, Parinaud J and Leandri RD: Environmental
pollutants, a possible etiology for premature ovarian
insufficiency: A narrative review of animal and human data. Environ
Health. 16:372017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Boone DL, Carnegie JA, Rippstein PU and
Tsang BK: Induction of apoptosis in equine chorionic gonadotropin
(eCG)-primed rat ovaries by anti-eCG antibody. Biol Reprod.
57:420–427. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Boone DL and Tsang BK: Caspase-3 in the
rat ovary: Localization and possible role in follicular atresia and
luteal regression. Biol Reprod. 58:1533–1539. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Cecconi S, Ciccarelli C, Barberi M,
Macchiarelli G and Canipari R: Granulosa cell-oocyte interactions.
Eur J Obstet Gynecol Reprod Biol. 115 (Suppl 1):S19–S22. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Clarke HJ: Regulation of germ cell
development by intercellular signaling in the mammalian ovarian
follicle. Wiley Interdiscip Rev Dev Biol. 7:10.1002/wdev.294. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Zhang JQ, Shen M, Zhu CC, Yu FX, Liu ZQ,
Ally N, Sun SC, Li K and Liu HL: 3-Nitropropionic acid induces
ovarian oxidative stress and impairs follicle in mouse. PLoS One.
9:e865892014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang M, Zhang Q, Hu Y, Xu L, Jiang Y,
Zhang C, Ding L, Jiang R, Sun J, Sun H and Yan G: miR-181a
increases FoxO1 acetylation and promotes granulosa cell apoptosis
via SIRT1 downregulation. Cell Death Dis. 8:e30882017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Haraguchi H, Hirota Y, Saito-Fujita T,
Tanaka T, Shimizu-Hirota R, Harada M, Akaeda S, Hiraoka T, Matsuo
M, Matsumoto L, et al: Mdm2-p53-SF1 pathway in ovarian granulosa
cells directs ovulation and fertilization by conditioning oocyte
quality. FASEB J. 33:2610–2620. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Livera G, Uzbekov R, Jarrier P,
Fouchécourt S, Duquenne C, Parent AS, Marine JC and Monget P: Loss
of oocytes due to conditional ablation of Murine double minute 2
(Mdm2) gene is p53-dependent and results in female sterility. FEBS
Lett. 590:2566–2574. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
He M, Tan B, Vasan K, Yuan H, Cheng F,
Ramos da Silva S, Lu C and Gao SJ: SIRT1 and AMPK pathways are
essential for the proliferation and survival of primary effusion
lymphoma cells. J Pathol. 242:309–321. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kume S, Haneda M, Kanasaki K, Sugimoto T,
Araki S, Isono M, Isshiki K, Uzu T, Kashiwagi A and Koya D: Silent
information regulator 2 (SIRT1) attenuates oxidative stress-induced
mesangial cell apoptosis via p53 deacetylation. Free Radic Biol
Med. 40:2175–2182. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Li Y, Yang G, Yang X, Wang W, Zhang J, He
Y, Zhang W, Jing T and Lin R: Nicotinic acid inhibits NLRP3
inflammasome activation via SIRT1 in vascular endothelial cells.
Int Immunopharmacol. 40:211–218. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mendelsohn AR and Larrick JW: The
NAD+/PARP1/SIRT1 Axis in aging. Rejuvenation Res. 20:244–247. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zha S, Li Z, Cao Q, Wang F and Liu F:
PARP1 inhibitor (PJ34) improves the function of aging-induced
endothelial progenitor cells by preserving intracellular NAD(+)
levels and increasing SIRT1 activity. Stem Cell Res Ther.
9:2242018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zheng T and Lu Y: SIRT1 protects human
lens epithelial cells against oxidative stress by inhibiting
p53-dependent apoptosis. Curr Eye Res. 41:1068–1075. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Han Y, Luo H, Wang H, Cai J and Zhang Y:
SIRT1 induces resistance to apoptosis in human granulosa cells by
activating the ERK pathway and inhibiting NF-κB signaling with
anti-inflammatory functions. Apoptosis. 22:1260–1272. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hori YS, Kuno A, Hosoda R and Horio Y:
Regulation of FOXOs and p53 by SIRT1 modulators under oxidative
stress. PLoS One. 8:e738752013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yamakuchi M and Lowenstein CJ: miR-34,
SIRT1 and p53: The feedback loop. Cell Cycle. 8:712–715. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Vousden KH and Prives C: Blinded by the
light: The growing complexity of p53. Cell. 137:413–431. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Aubrey BJ, Kelly GL, Janic A, Herold MJ
and Strasser A: How does p53 induce apoptosis and how does this
relate to p53-mediated tumour suppression? Cell Death Differ.
25:104–113. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Hosokawa K, Aharoni D, Dantes A, Shaulian
E, Schere-Levy C, Atzmon R, Kotsuji F, Oren M, Vlodavsky I and
Amsterdam A: Modulation of Mdm2 expression and p53-induced
apoptosis in immortalized human ovarian granulosa cells.
Endocrinology. 139:4688–4700. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Yang H, Xie Y, Yang D and Ren D: Oxidative
stress-induced apoptosis in granulosa cells involves JNK, p53 and
Puma. Oncotarget. 8:25310–25322. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tatone C, Di Emidio G, Vitti M, Di Carlo
M, Santini S Jr, D'Alessandro AM, Falone S and Amicarelli F:
Sirtuin functions in female fertility: Possible role in oxidative
stress and aging. Oxid Med Cell Longev. 2015:6596872015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ghosh A, Sengupta A, Seerapu GPK, Nakhi A,
Shivaji Ramarao EVV, Bung N, Bulusu G, Pal M and Haldar D: A novel
SIRT1 inhibitor, 4bb induces apoptosis in HCT116 human colon
carcinoma cells partially by activating p53. Biochem Biophys Res
Commun. 488:562–569. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gu X, Wang Z, Gao J, Han D, Zhang L, Chen
P, Luo G and Han B: SIRT1 suppresses p53-dependent apoptosis by
modulation of p21 in osteoblast-like MC3T3-E1 cells exposed to
fluoride. Toxicol In Vitro. 57:28–38. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Idil M, Cepni I, Demirsoy G, Ocal P,
Salihoğlu F, Senol H, Elibol F and Irez T: Does granulosa cell
apoptosis have a role in the etiology of unexplained infertility?
Eur J Obstet Gynecol Reprod Biol. 112:182–184. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nakahara K, Saito H, Saito T, Ito M, Ohta
N, Sakai N, Tezuka N, Hiroi M and Watanabe H: Incidence of
apoptotic bodies in membrana granulosa of the patients
participating in an in vitro fertilization program. Fertil Steril.
67:302–308. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Almeida CP, Ferreira MCF, Silveira CO,
Campos JR, Borges IT, Baeta PG, Silva FHS, Reis FM and Del Puerto
HL: Clinical correlation of apoptosis in human granulosa cells-A
review. Cell Biol Int. 42:1276–1281. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Xiong F, Hu L, Zhang Y, Xiao X and Xiao J:
miR-22 inhibits mouse ovarian granulosa cell apoptosis by targeting
SIRT1. Biol Open. 5:367–371. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Di Emidio G, Falone S, Vitti M,
D'Alessandro AM, Vento M, Di Pietro C, Amicarelli F and Tatone C:
SIRT1 signalling protects mouse oocytes against oxidative stress
and is deregulated during aging. Hum Reprod. 29:2006–2017. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang H, Vollmer M, De Geyter M,
Litzistorf Y, Ladewig A, Dürrenberger M, Guggenheim R, Miny P,
Holzgreve W and De Geyter C: Characterization of an immortalized
human granulosa cell line (COV434). Mol Hum Reprod. 6:146–153.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kim JM, Yoon YD and Tsang BK: Involvement
of the Fas/Fas ligand system in p53-mediated granulosa cell
apoptosis during follicular development and atresia. Endocrinology.
140:2307–2317. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Chen L, Willis SN, Wei A, Smith BJ,
Fletcher JI, Hinds MG, Colman PM, Day CL, Adams JM and Huang DC:
Differential targeting of prosurvival Bcl-2 proteins by their
BH3-only ligands allows complementary apoptotic function. Mol Cell.
17:393–403. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Kerr JB, Hutt KJ, Michalak EM, Cook M,
Vandenberg CJ, Liew SH, Bouillet P, Mills A, Scott CL, Findlay JK
and Strasser A: DNA damage-induced primordial follicle oocyte
apoptosis and loss of fertility require TAp63-mediated induction of
Puma and Noxa. Mol Cell. 48:343–352. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Nguyen QN, Zerafa N, Liew SH, Morgan FH,
Strasser A, Scott CL, Findlay JK, Hickey M and Hutt KJ: Loss of
PUMA protects the ovarian reserve during DNA-damaging chemotherapy
and preserves fertility. Cell Death Dis. 9:6182018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Brieger K, Schiavone S, Miller FJ Jr and
Krause KH: Reactive oxygen species: From health to disease. Swiss
Med Wkly. 142:w136592012.PubMed/NCBI
|
|
40
|
Yi J and Luo J: SIRT1 and p53, effect on
cancer, senescence and beyond. Biochim Biophys Acta.
1804:1684–1689. 2010. View Article : Google Scholar : PubMed/NCBI
|


















