Introduction
Hepatocellular carcinoma (HCC), which is the primary
subtype of liver cancer, is the sixth most frequent malignant tumor
and the second deadliest of all types of cancer globally (1). HCC is characterized by aggressive
behavior, high morbidity, easily occurring metastasis and
resistance to currently available chemotherapeutic drugs (2). In 2015, 466,000 new HCC cases were
diagnosed in China, of which 422,000 patients succumbed to fatal
tumors (3). Currently, surgical
excision is a treatment option for patients with early-stage HCC,
but the vast majority of cases are diagnosed at an advanced stage,
at which point the curative effect of radiochemotherapy is poor
(4). Over the last decade, the
clinical outcomes of HCC have substantially improved due to the
significant progress in diagnostic and therapeutic techniques
(5). However, even when receiving
first-line anticancer therapies, >50% of patients with HCC will
experience tumor relapse, including intrahepatic and distant
metastasis, which results in poor prognosis (6). Thus, research focusing on the
mechanisms modulating HCC metastasis and aggressiveness are
urgently needed for the development of attractive therapeutic
targets.
In the human genome, only 2% of the RNA can be
translated into protein and the residual RNAs lack protein-coding
ability and are thus termed noncoding RNAs (7). Long noncoding RNAs (lncRNAs) are
defined as a form of noncoding RNA transcripts that are >200
nucleotides in length (8). Studies
have focused on the important functions of lncRNAs in cancer
research (9–11). Differentially expressed lncRNAs have
been observed in human cancers and contribute to various aspects of
the biological activity of tumors (12,13).
In HCC, a number of lncRNAs are reported to be dysregulated and
regulate anti- or pro-oncogenic activities during
hepatocarcinogenesis and cancer progression (14,15).
MicroRNAs (miRNAs) are another type of noncoding RNA
transcript that also exerts nonprotein-coding capacity (16). miRNAs can bind directly to the
3′-untranslated region (UTR) of downstream target genes and
consequently inhibit translation or mRNA degradation, thereby
controlling gene expression at the posttranslational level
(17). Notably, lncRNAs are capable
of interacting with miRNAs and thus implementing their functions
(18). As gene regulators, lncRNAs
can work as competing endogenous RNAs (ceRNAs) by decoying or
competing with shared miRNAs, thereby lowering the miRNA-induced
inhibition of gene expression (19). Therefore, studying lncRNAs and
miRNAs may facilitate research on HCC diagnosis and anticancer
treatments.
The association of lncRNAs with HCC has been widely
reported (20–22), yet detailed functions of ZSCAN16-AS1
have seldom been unraveled in HCC. The present study examined the
expression pattern of ZSCAN16-AS1 in HCC. To address the functions
of ZSCAN16-AS1 in HCC, loss-of-function assays were implemented
in vitro and in vivo. In addition, the possible
molecular events through which ZSCAN16-AS1 exerts its tumorigenic
actions in HCC were demonstrated.
Materials and methods
Clinical specimens and cell lines
The present study was approved by the Ethics
Committee of The People's Hospital of Tongliang District (approval
no. EC.2015-39). All patients provided informed written consent for
the use of their tissue samples. A total of 47 pairs of HCC tissues
and adjacent normal tissues (distance, 2 cm) were obtained from
patients (31 males and 16 females; age range, 41–68 years) in The
People's Hospital of Tongliang District between May 2015 and
November 2016. Following tumor excision, the tumors were quickly
stored in liquid nitrogen. The patients had not received
chemotherapy, radiotherapy, or other anticancer treatments.
The transformed human liver epithelial-3 (THLE-3)
cell line and two HCC cell lines (Hep3B and HuH7) were acquired
from the Cell Bank of the Chinese Academy of Sciences. THLE-3 was
cultured in bronchial epithelial basal medium (BEGM; Clonetics
Corporation), to which 10% fetal bovine serum (FBS; Gibco; Thermo
Fisher Scientific, Inc.), 5 ng/ml EGF and 70 ng/ml
phosphoethanolamine were added. Hep3B and HuH7 cells were grown in
Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher
Scientific, Inc.) with 10% FBS, 1% GlutaMAX, 1% nonessential amino
acids and 1% penicillin/streptomycin. An additional 100 mM 1%
sodium pyruvate solution was added to the Hep3B cell culture.
In addition, two other HCC cell lines, SNU-398 and
SNU-182, were purchased from the ATCC. SNU-398 cells were
maintained in RPMI-1640 (Gibco; Thermo Fisher Scientific, Inc.)
containing 10% FBS and 1% penicillin/streptomycin. RPMI-1640 with
10% FBS, 1% GlutaMAX, 1% nonessential amino acids and 1%
penicillin/streptomycin were applied to SNU-182 cells. All cells
were kept in a humidified atmosphere equipped with 5%
CO2 at 37°C.
Cell transfection
Small interfering RNAs (siRNAs) targeted to silence
ZSCAN16-AS1 (si-ZSCAN16-AS1s) and nontargeted siRNA (si-NC) were
designed and synthesized by Shanghai GenePharma Co., Ltd. The
si-ZSCAN16-AS1#1 sequence was 5′-TTGTAAAATTGAAATATTTGAAT-3′; the
si-ZSCAN16-AS1#2 sequence was 5′-TACCAAAAAATAAAAATATGAAC-3′; the
si-ZSCAN16-AS1#3 sequence was 5′-GGCATACTTAGTTTTACATTTTT-3′; and
the si-NC sequence was 5′-CACGATAAGACAATGTATTT-3′. miR-451a mimic
and miR-451a inhibitors were obtained from Guangzhou RiboBio Co.,
Ltd., with miRNA mimic control (NC mimic) and miRNA inhibitor
control (NC inhibitor) as the controls. The miR-451a mimic sequence
was 5′-UUGAGUCAUUACCAUUGCCAAA-3′ and the NC mimic sequence was
5′-UUGUACUACACAAAAGUACUG-3′. The miR-451a inhibitor sequence was
5′-AACUCAGUAAUGGUAACGGUUU-3′ and the NC inhibitor sequence was
5′-ACUACUGAGUGACAGUAGA-3′. The cDNA encoding the ATF2 coding
sequence was amplified by Shanghai GenePharma Co., Ltd., and was
inserted into the pcDNA3.1 plasmid, thereby generating the
pcDNA3.1-ATF2 plasmid. The empty pcDNA3.1 plasmid was applied as
the control. Cells were transfected with siRNAs (100 pmol), miRNA
mimic (100 pmol), miRNA inhibitor (100 pmol) or plasmid (4 µg)
utilizing Lipofectamine® 2000 reagent (Invitrogen;
Thermo Fisher Scientific, Inc.). After 6 h incubation with
transfection reagent at 37°C, the medium was replaced with fresh
medium. Then, 48 h after transfection, reverse
transcription-quantitative (RT-q) PCR, cell apoptosis detection by
flow cytometry, Transwell cell migration and invasion assays and
western blotting were performed. A Cell Counting Kit-8 (CCK-8)
assay was implemented at 24 h post-transfection.
RT-qPCR
Tissues or cells (2×106) were immersed in
Beyozol (Beyotime Institute of Biotechnology) for total RNA
extraction. To quantify miRNA expression, reverse transcription was
performed according to the manufacturer's instructions, using a
miRcute miRNA First-Strand cDNA Synthesis kit followed by PCR
amplification using the miRcute miRNA qPCR Detection kit SYBR-Green
(both from Tiangen Biotech Co., Ltd.). U6 small nuclear RNA served
as an endogenous control to analyze miRNA expression. To analyze
ZSCAN16-AS1 and ATF2 expression, a PrimeScript RT Reagent kit with
gDNA Eraser (Takara Biotechnology Co., Ltd.) was used to carry out
reverse transcription. The obtained cDNA was subjected to TB Green
Premix Ex Taq™ II (Takara Biotechnology Co., Ltd.) to perform qPCR.
The thermocycling conditions were as follows: Initial denaturation
at 95°C for 30 sec, followed by 40 cycles at 95°C for 3 sec, 60°C
for 30 sec and 72°C for 30 sec. Relative ZSCAN16-AS1 and ATF2
expression was normalized to that of GAPDH. The 2−ΔΔCq
method (23) was used to process
all data. RNA extraction, cDNA synthesis, and qPCR were performed
according to the manufacturer's protocols. These experiments were
repeated three times.
The primers were as follows: ZSCAN16-AS1,
5′-GGGCTGCAATAAAACAGCAAA-3′ (forward) and
5′-CAATTTCCTATCCCGACCCTCT-3′ (reverse); ATF2,
5′-CAGGAACTGTTCTAGCACCAGC-3′ (forward) and
5′-CAGGAGTTTCAGGCTGCAGTAA-3′ (reverse); GAPDH,
5′-CGGAGTCAACGGATTTGGTCGTAT-3′ (forward) and
5′-AGCCTTCTCCATGGTGGTGAAGAC-3′ (forward); miR-23c,
5′-TCGGCAGGGGGTAATCACTGG-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-23b-3p,
5′-TCGGCAGGGGAAATCCCTGG-3′ (forward) and 5′-CACTCAACTGGTGTCGTGGA-3′
(reverse); miR-130a-5p, 5′-TCGGCAGGGCUCUUUUCACAUU-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-181a-5p,
5′-TCGGCAGGAACAUUCAACGCUG-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-181c-5p,
5′-TCGGCAGGAACAUUCAACCUG-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-4524a-5p,
5′-TCGGCAGGAUAGCAGCAUGAAC-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-451a,
5′-TCGGCAGGAAACCGUUACCAUU-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-146a-5p,
5′-TCGGCAGGUGAGAACUGAAUUC-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); miR-22-3p,
5′-TCGGCAGGACAGTTCTTCAACT-3′ (forward) and
5′-CACTCAACTGGTGTCGTGGA-3′ (reverse); and U6,
5′-CTCGCTTCGGCAGCACA-3′ (forward) and 5′-AACGCTTCACGAATTTGCGT-3′
(reverse).
CCK-8 assay
Transfected cells were harvested to prepare a cell
suspension. A 100 µl volume of cell suspension carrying
2×103 cells was then transferred to 96-well plates,
followed by cultivation for different time points (0, 24, 48 and 72
h) in an incubator. At different time points (0, 24, 48 and 72 h),
10 µl of CCK-8 reagent (Dojindo Molecular Technologies, Inc.) was
employed to treat the cells at 37°C for another 2 h. Finally, the
absorbance at 450 nm was measured using a microplate reader.
Cell apoptosis detection by flow
cytometry
The collected cells were transferred to flow tubes.
Following centrifugation at 4°C at 1,000 × g for 5 min, the
supernatant fluid was removed and transfected cells were
resuspended in 195 µl of Annexin V-FITC binding buffer from the
Annexin V-FITC Apoptosis Detection kit (Beyotime Institute of
Biotechnology). Immediately, 5 µl of Annexin V-FITC and 10 µl of PI
were added to the cell suspension, followed by 15 min of
cultivation without light. A flow cytometer (FACScan, BD
Biosciences) was used to analyze cell apoptosis. Data were analyzed
using CellQuest software v.2.9 (BD Biosciences). The percentage of
early + late apoptotic cells was calculated.
Transwell cell migration and invasion
assays
The harvested cells were centrifuged at room
temperature at 1,000 × g for 15 min and resuspended in culture
medium without serum. The cell concentration was adjusted to
2.5×105 cells/ml. Transwell inserts (8-µm filter; BD
Biosciences) that were precoated with Matrigel (BD Biosciences)
were used for the invasion test, whereas the migration test was
performed without Matrigel. Precoating was performed at 37°C for 2
h. A total of 200 µl of cell suspension was seeded into the upper
chambers, while 20% FBS-contained 600 µl of culture medium was
applied to the lower chambers as a chemoattractant. One day later,
the cells remaining in the upper chambers were cleaned with a
cotton bud. The cells that passed through the pores were fixed in
4% paraformaldehyde at room temperature for 30 min and stained with
0.1% crystal violet at room temperature for 30 min. Images of
migrated or invaded cells were obtained and quantified under an
optical light microscope (magnification, ×200). A total of five
fields were randomly selected and the number of migrated/invaded
cells was counted.
Tumor xenograft model
Experiments involving animals were implemented under
the approval of the Institutional Animal Care and Use Committee of
The People's Hospital of Tongliang District. A 2nd lentiviral
system was used in the production of lentiviruses. Short hairpin
RNA (shRNA) targeting ZSCAN16-AS1 (sh-ZSCAN16-AS1) and the negative
control shRNA (sh-NC) were acquired from GenePharma Inc. The
sh-ZSCAN16-AS1 sequence was
5′-CCGGTTGTAAAATTGAAATATTTGAATCTCGAGATTCAAATATTTCAATTTTACAATTTTTG-3′
and sh-NC sequence was
5′-CCGGCACGATAAGACAATGTATTTCTCGAGAAATACATTGTCTTATCGTGTTTTTG-3′.
Following insertion into the pLKO.1 vector (Addgene Inc.), they
were transfected into 239T cells (Cell Bank of the Chinese Academy
of Sciences) in the presence of lentiviral packaging plasmid psPAX2
and envelope expression plasmid pMD2.G (both from Addgene Inc.).
The transfection was implemented using Lipofectamine®
2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.). The
proportion of lentiviral plasmid: pLKO.1: psPAX2: pMD2.G was 2:1:1
and 30 µg plasmids were employed in lentivirus packaging. Following
5 h incubation at 37°C with 5% CO2, the culture medium
was discarded and cells were maintained in fresh Dulbecco's
modified Eagle medium that was supplemented with 10% FBS, 1%
glutamax, 1% non-essential amino acids and 1% sodium pyruvate
solution (all from Gibco; Thermo Fisher Scientific, Inc.).
Lentiviruses expressing sh-ZSCAN16-AS1 or sh-NC were harvested via
ultracentrifugation at 4°C at 1,000 × g for 2 h and then mixed with
polybrene (5 µg/ml; Sigma-Aldrich; Merck KGaA) and Dulbecco's
modified Eagle's medium. Following injection into HuH7 cells with a
multiplicity of infection 5, puromycin was applied to select HuH7
cells with stable ZSCAN16-AS1 ablation.
For subcutaneous injection, a total of
2×106 sh-ZSCAN16-AS1- or sh-NC-transfected HuH7 cells
were resuspended in 100 µl phosphate buffer saline and implanted
into the flank of 4- to 5-week-old male BALB/c nude mice (n=6; mean
weight, 20.4 g), which were obtained from the Shanghai SLAC
Laboratory Animal Co., Ltd. All mice were housed under specific
pathogen-free conditions at 25°C and 50% humidity, with a 10:14
light/dark cycle and ad libitum access to food and water.
The tumor size was examined weekly. After four weeks, all mice were
euthanized by cervical dislocation and the tumor xenografts were
excised, imaged and weighed. The volume of the tumor xenografts was
calculated using the following formula: volume=(length ×
width2)/2.
Subcellular fractionation assay
Cytoplasmic and nuclear fractions of HCC cells were
separated via a Cytoplasmic and Nuclear RNA Purification kit
(Norgen Biotek Corp.). RNA was then analyzed with RT-qPCR to
calculate the relative distribution of ZSCAN16-AS1 in HCC
cells.
Bioinformatics prediction
StarBase 3.0 (http://starbase.sysu.edu.cn/) was applied for the
prediction of direct binding between ZSCAN16-AS1 and miR-451a.
TargetScan (http://www.targetscan.org), miRDB (http://mirdb.org/) and StarBase 3.0 were used to
determine the target genes of miR-451a.
Luciferase reporter assay
ZSCAN16-AS1 and ATF2 fragments carrying the miR-451a
binding site were amplified before they were inserted into the
pMIR-luciferase reporter plasmid (Promega Corporation). The
resulting luciferase reporter plasmids were labeled as
ZSCAN16-AS1-wild-type (wt) and ATF2-wt. The luciferase reporter
plasmids that contained a mutant (mut) miR-451a binding site were
synthesized following the same experimental steps and the resulting
plasmids were labeled ZSCAN16-AS1-mut and ATF2-mut. HCC cells were
transfected with miR-451a mimic or NC mimic in parallel with the
luciferase reporter plasmids utilizing Lipofectamine®
2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.). After 48
h of cultivation, a dual-luciferase reporter assay system (Promega
Corporation) was adopted for the assessment of luciferase
activity.
RNA immunoprecipitation (RIP)
The Magna RIP RNA-Binding Protein
Immunoprecipitation kit (EMD Millipore) was applied in the assay.
In brief, 1×106 HCC cells were harvested and cultivated
in complete RIP lysis buffer to obtain whole-cell extracts. Then,
RIP buffer supplemented with magnetic beads that were conjugated
with human anti-Ago2 or anti-IgG antibodies (Millipore) was
utilized to incubate the cell extract overnight at 4°C. An input
control was assayed simultaneously to function as the positive
control, while IgG served as the negative control. Magnetic beads
were collected via centrifugation at 4°C at 1,000 × g for 5 min and
probed with proteinase K to remove the protein. After extracting
the immunoprecipitated RNA, the relative enrichment of ZSCAN16-AS1,
miR-451a and ATF2 was detected by RT-qPCR.
Western blot analysis
Total protein from the cultured cells was extracted
by lysis in cell lysis buffer for western blotting and
immunoprecipitation (IP; Beyotime Institute of Biotechnology) and
quantified using an Enhanced BCA Protein Assay kit (Beyotime
Institute of Biotechnology). Protein was added to the loading
buffer and each well was loaded with equal amounts of protein (30
µg/lane). The separated proteins were transferred to PVDF membranes
and then subjected to 10% SDS-PAGE electrophoresis. After that, the
membranes were sealed with 5% defatted milk at room temperature for
2 h, after which the membranes underwent 12 h incubation at 4°C
with primary antibodies specifically binding to ATF2 (cat. no.
ab239361; 1:1,000) or GAPDH (ab181602; 1:1,000; Abcam). After 2 h
of cultivation at room temperature with horseradish
peroxidase-labelled secondary antibody (cat. no. ab205718; 1:5,000;
Abcam), the signals were detected with a BeyoECL Plus kit (Beyotime
Institute of Biotechnology). Quantity One software version 4.62
(Bio-Rad Laboratories, Inc.) was adopted for densitometry.
Statistical analysis
Data were calculated from three biological repeats
of each assay and presented as the mean ± standard deviation.
Paired Student's t-test was used to compare ZSCAN16-AS1, miR-451a
and ATF2 expression between HCC tumor tissues and normal tissues.
Significant differences between other two groups were detected by
unpaired Student's t-test. One-way analysis of variance followed by
Tukey's test was used to compare the differences among multiple
groups. Pearson's correlation test was used to determine the
expression correlations among ZSCAN16-AS1, miR-451a and ATF2. The
overall survival curves were plotted utilizing the Kaplan-Meier
method and were compared using the log-rank test. P<0.05 was
considered to indicate a statistically significant difference.
Results
The upregulation of ZSCAN16-AS1
indicates a poor prognosis for HCC
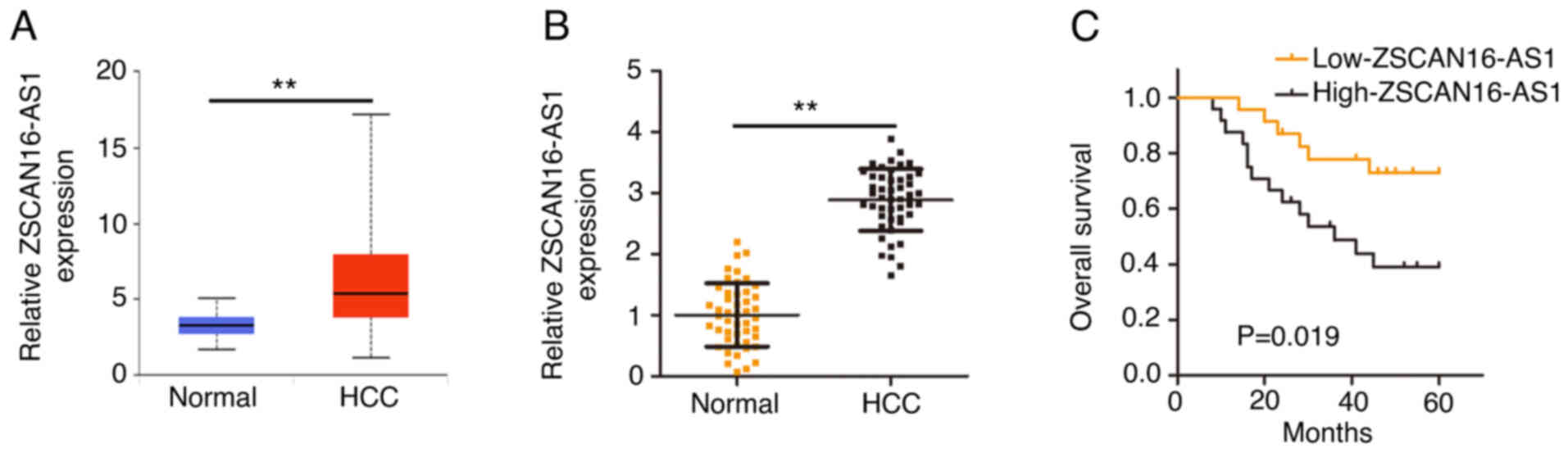
To comprehensively disclose the expression of
ZSCAN16-AS1, its expression in HCC was first analyzed using The
Cancer Genome Atlas (TCGA) database. ZSCAN16-AS1 level in HCC tumor
tissues was clearly increased compared with normal tissues
(Fig. 1A). RT-qPCR was implemented
to further confirm ZSCAN16-AS1 expression in HCC tissues and
matched adjacent normal tissues (n=47). Notably high ZSCAN16-AS1
level was validated in HCC tissues (Fig. 1B). In addition, all individuals were
divided into low-ZSCAN16-AS1 or high-ZSCAN16-AS1 expression groups
according to the median value of ZSCAN16-AS1 expression in the 47
HCC tissues. Patients in the high-ZSCAN16-AS1 expression group
displayed shorter overall survival rates than patients in the
low-ZSCAN16-AS1 expression group (Fig.
1C).
ZSCAN16-AS1 knockdown exerts
inhibitory effects on the malignant processes of HCC cells
To explore the biological actions of ZSCAN16-AS1,
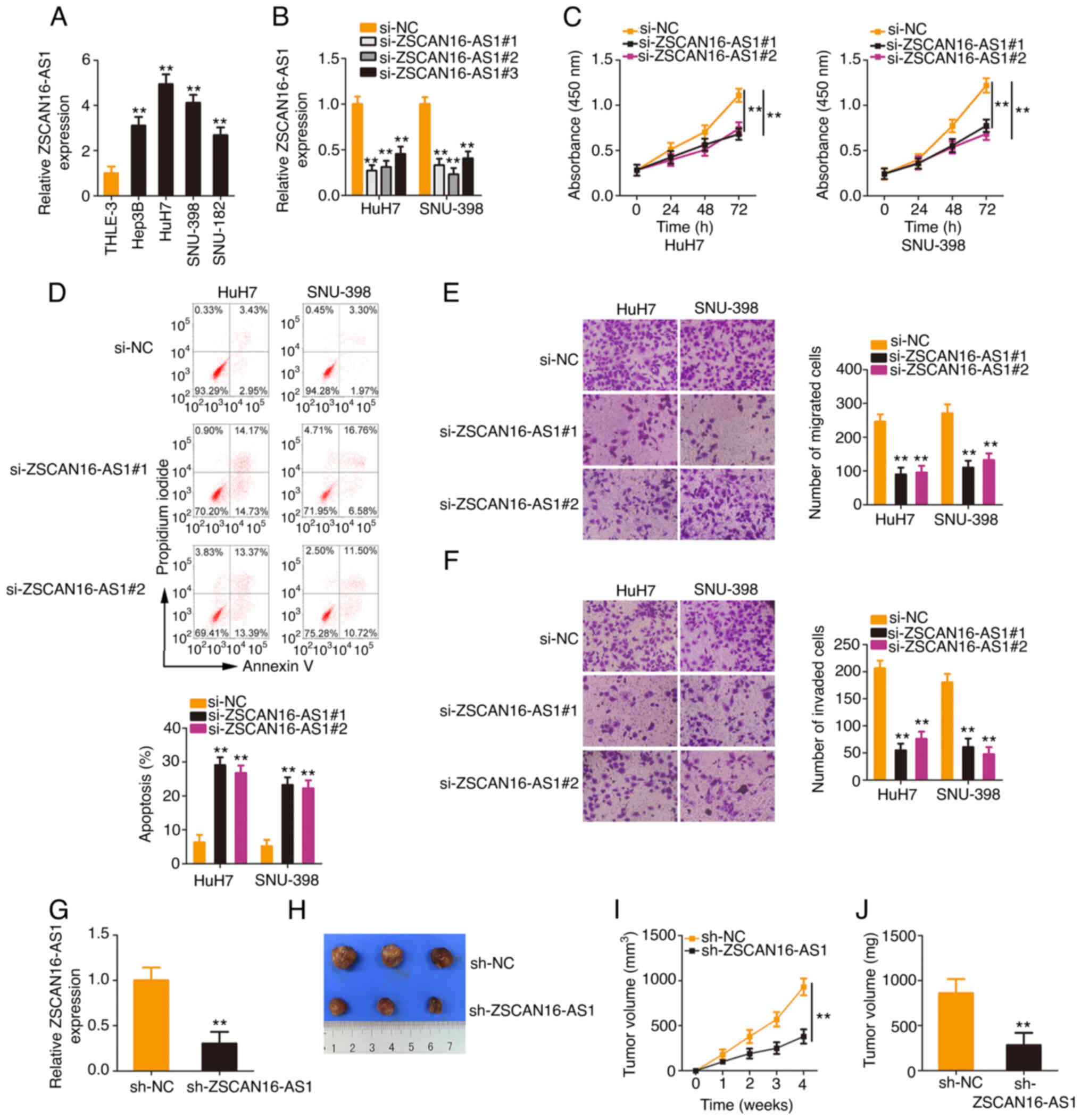
ZSCAN16-AS1 expression in HCC cell lines was measured. All four HCC
cell lines presented higher ZSCAN16-AS1 expression levels,
especially in the HuH7 and SNU-398 cell lines (Fig. 2A). Therefore, they were selected for
use in following experiments. ZSCAN16-AS1 expression was
effectively silenced in HuH7 and SNU-398 cells after transfection
with si-ZSCAN16-AS1 (Fig. 2B). To
avoid off-target effects, two siRNAs, si-ZSCAN16-AS1#1 and
si-ZSCAN16-AS1#2, were employed in the loss-of-function assays.
Functionally, the CCK-8 assay confirmed that the proliferation of
HCC cells was clearly hindered after ZSCAN16-AS1 depletion
(Fig. 2C). Additionally,
ZSCAN16-AS1-silenced HCC cells showed more apoptosis than the si-NC
group (Fig. 2D). Furthermore, the
number of migrated (Fig. 2E) and
invaded (Fig. 2F) cells in
si-ZSCAN16-AS1-transfected HCC cells was clearly less than that in
the si-NC-transfected cells, which indicated that ZSCAN16-AS1
depletion impaired the migratory and invasive capacities of HCC
cells. In addition, data obtained from the tumor xenograft model
revealed that the tumor xenografts originating from HuH7 cells with
stable ZSCAN16-AS1 depletion (Fig.
2G) were smaller (Fig. 2H and
I) and lighter (Fig. 2J)
compared with those of the sh-NC group. Altogether, ZSCAN16-AS1
exhibited oncogenic properties that promote malignant behaviors in
HCC cells.
ZSCAN16-AS1 is a ceRNA and operates as
a molecular sponge for miR-451a in HCC
To investigate the mechanisms underlying the
oncogenic actions of ZSCAN16-AS1, a subcellular fractionation assay
was applied to separate the cytoplasmic and nuclear fractions of
HCC cells and RT-qPCR was then performed to assess the distribution
characteristics of ZSCAN16-AS1, which was validated to be located
mostly in the cytoplasm of HCC cells (Fig. 3A). Given the localization of
ZSCAN16-AS1 in HCC, it was inferred that ZSCAN16-AS1 may perform
tumor-promoting roles via a ceRNA. Using StarBase 3.0, 23 miRNAs
(Fig. 3B) were predicted to be
targets of ZSCAN16-AS1. Notably, by comparing the 23 miRNAs with
those dysregulated miRNAs in the TCGA-HCC database, miR-23c,
miR-23b-3p, miR-130a-5p, miR-181a-5p, miR-181c-5p, miR-4524a-5p,
miR-451a, miR-146a-5p and miR-22-3p were found to be decreased in
HCC. RT-qPCR was then performed to examine the regulatory actions
of ZSCAN16-AS1 on these candidates in HCC cells. As Fig. 3C shows, downregulation of
ZSCAN16-AS1 evidently increased miR-451a expression, whereas the
other miRNAs were unaffected. Decreased miR-451a expression was
identified in HCC (Fig. 3D) and
presented an inverse correlation with ZSCAN16-AS1 expression
(Fig. 3E). Using TCGA database, an
inverse correlation was observed between ZSCAN16-AS1 and miR-451a
in HCC (Fig. 3F). Furthermore, the
direct binding between ZSCAN16-AS1 and miR-451a (Fig. 3G) was corroborated by RIP and
luciferase reporter assays. Overexpressed miR-451a reduced the
luciferase activity of the ZSCAN16-AS1-wt reporter plasmid;
nevertheless, mutation of the binding site abolished the inhibitory
action (Fig. 3H). The outcomes of
the RIP assay showed that ZSCAN16-AS1 and miR-451a were enriched in
Ago2-containing immunoprecipitated RNA in contrast to that of the
IgG control (Fig. 3I). These
results suggest that ZSCAN16-AS1 worked as a miR-451a sponge in HCC
cells.
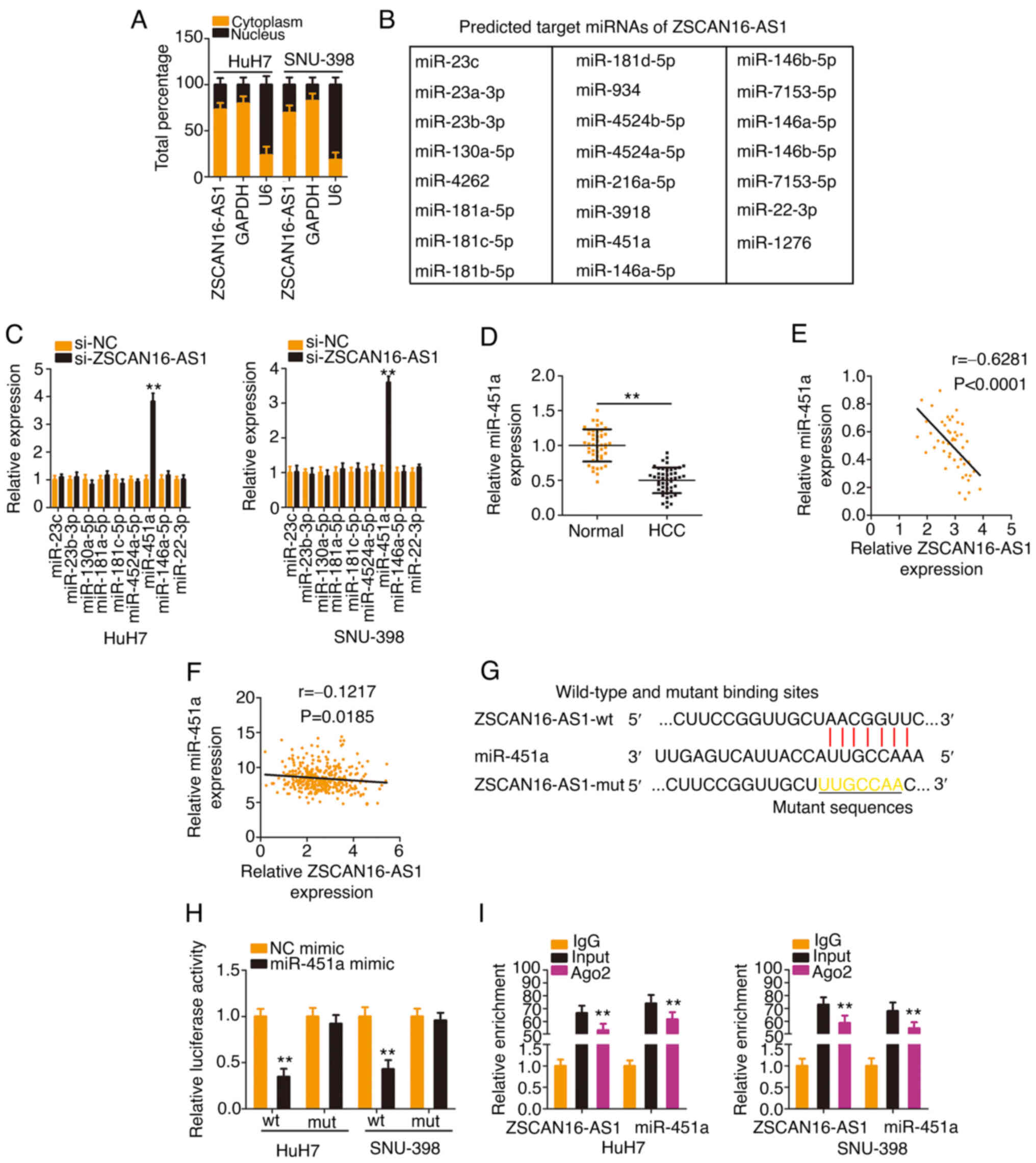 | Figure 3.ZSCAN16-AS1 acts as a natural
miR-451a sponge in HCC. (A) Relative abundances of ZSCAN16-AS1 in
the cytoplasmic and nuclear fractions of HCC cells. (B) The
putative targets of ZSCAN16-AS1 predicted by StarBase 3.0. (C) The
levels of miR-23c, miR-23b-3p, miR-130a-5p, miR-181a-5p,
miR-181c-5p, miR-4524a-5p, miR-451a, miR-146a-5p and miR-22-3p were
detected in HCC cells following ZSCAN16-AS1 knockdown. (D) miR-451a
expression was measured by reverse transcription-quantitative PCR
in HCC tissues. (E) The relation between ZSCAN16-AS1 and miR-451a
levels in 47 HCC tissues. (F) The expression correlation between
ZSCAN16-AS1 and miR-451a in HCC was examined using The Cancer
Genome Atlas database. (G) The predicted complementary sequence
between ZSCAN16-AS1 and miR-451a. (H) Luciferase activities of
ZSCAN16-AS1-wt or ZSCAN16-AS1-mut were determined in the presence
of miR-451a mimic or NC mimic. (I) RNA immunoprecipitation assay
was implemented to evaluate ZSCAN16-AS1 and miR-451a enrichment in
immunoprecipitants of HCC cells. **P<0.01. miR, microRNA; HCC,
hepatocellular carcinoma; wt, wild-type; mut, mutant; NC, negative
control. |
miR-451a exerts anti-oncogenic actions
in HCC
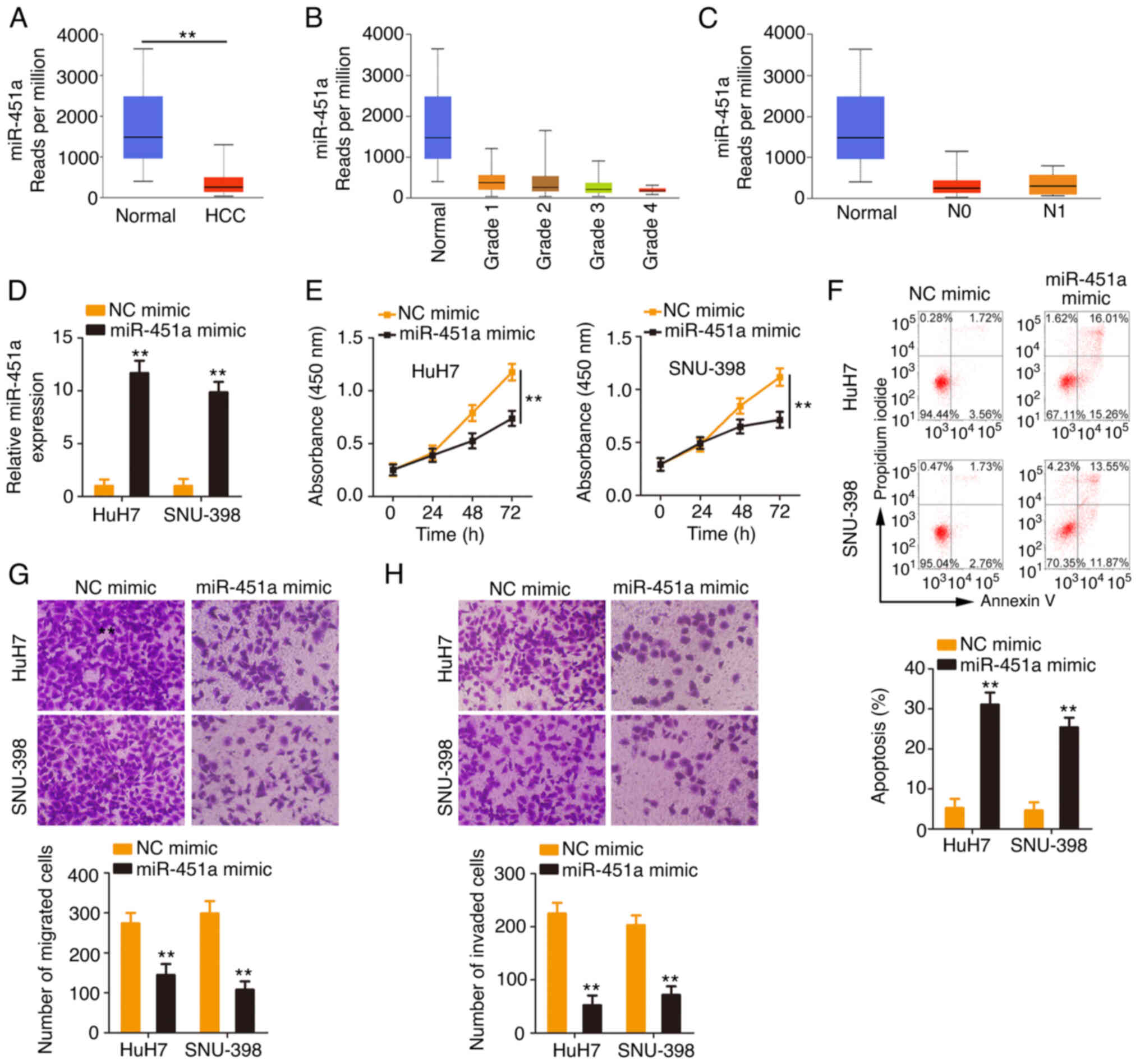
Considering the downregulation of miR-451a in HCC,
the present study next addressed its clinical value and detailed
its roles in regulating the malignant behaviors of HCC cells. As
shown in the TCGA database, low miR-451a expression (Fig. 4A) was closely correlated with the
tumor grade and lymph node metastasis (Fig. 4B and C) in HCC. miR-451a was
overexpressed in HCC cells after miR-451a mimic transfection
(Fig. 4D). Ectopic miR-451a
expression inhibited the proliferative ability of HCC cells
(Fig. 4E). In addition, the
percentage of apoptotic HCC cells was significantly increased by
miR-451a mimic transfection (Fig.
4F). Furthermore, HCC cells transfected with miR-451a mimic
manifested lower migratory and invasive (Fig. 4G and H) abilities than the NC mimic
control group. Overall, miR-451a exerted anti-oncogenic roles in
HCC.
ATF2 is under the regulation of the
ZSCAN16-AS1/miR-451a axis in HCC
Using bioinformatics analysis, a potential binding
site of miR-451a was identified in the ATF2 3′-UTR (Fig. 5A). The results of the luciferase
reporter assay ascertained that transfection with the miR-451a
mimic weakened the activity of the ZSCAN16-AS1-wt reporter plasmid
but not that of the reporter plasmid ZSCAN16-AS1-mut in HCC cells
(Fig. 5B). In addition, enforced
miR-451a expression appeared to lower ATF2 levels (Fig. 5C and D) in HCC cells. After
identifying ATF2 as a direct target of miR-451a, subsequent
experiments were implemented to explore whether a ceRNA pathway
consisting of ZSCAN16-AS1, miR-451a and ATF2 exists in HCC. The
data revealed that ATF2 expression (Fig. 5E and F) was decreased in
ZSCAN16-AS1-deficient HCC cells but was restored by cotransfection
with the miR-451a inhibitor (Fig. 5G
and H). Furthermore, ZSCAN16-AS1, miR-451a and ATF2 were all
enriched in Ago2-containing immunoprecipitated RNA compared with
those of the IgG control (Fig. 5I).
A higher ATF2 level was detected in HCC tissues compared with
adjacent normal tissues (Fig. 5J).
In addition, a negative correlation between ATF2 and miR-451a was
confirmed in the cohort of the present study (Fig. 5K) and TCGA database (Fig. 5L). A positive correlation between
ATF2 and ZSCAN16-AS1 was identified in the 47 HCC tissues (Fig. 5M) and TCGA database (Fig. 5N). Thus, ZSCAN16-AS1 is a ceRNA that
sequesters miR-451a and consequently positively regulates ATF2
expression in HCC.
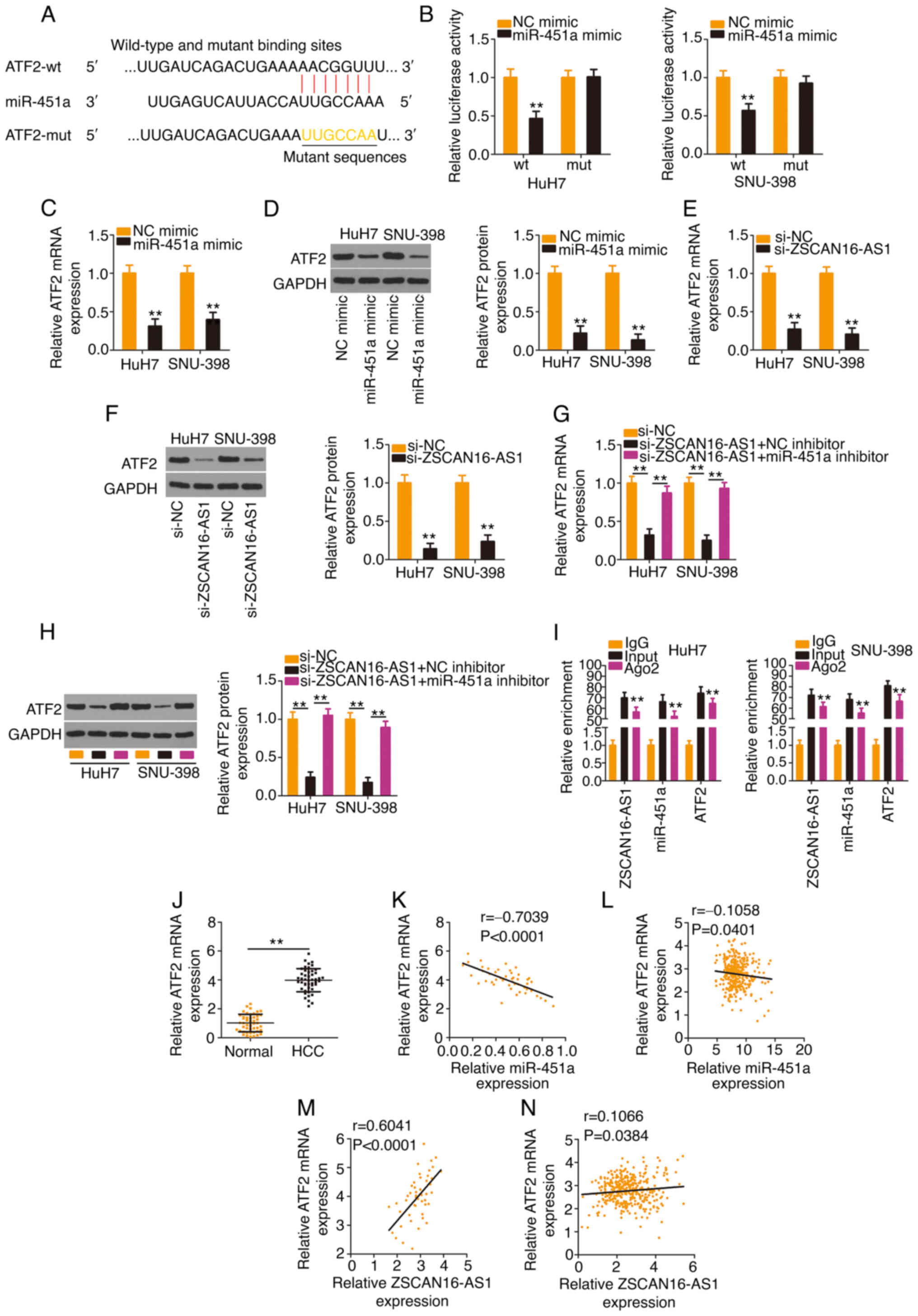 | Figure 5.The ZSCAN16-AS1/miR-451a axis
regulates ATF2 expression in HCC cells. (A) The predicted binding
site of miR-451a within the 3′-UTR of ATF2. The mutated binding
site is also shown. (B) HCC cells were transfected with ATF2-wt or
ATF2-mut in combination with miR-451a mimic or NC mimic; 48 h
later, the luciferase activity was quantified. (C and D) ATF2
expression was determined in HCC cells when miR-451a was
overexpressed. (E) mRNA and (F) protein expression of ATF2 in HCC
cells following ZSCAN16-AS1 ablation. si-ZSCAN16-AS1 together with
miR-451a inhibitor or NC inhibitor was transfected into HCC cells,
followed by quantification of ATF2 (G) mRNA and (H) protein levels.
(I) RNA immunoprecipitation was used to assess ZSCAN16-AS1,
miR-451a and ATF2 enrichment in immunoprecipitants in HCC cells.
(J) ATF2 level was determined in HCC tissues using reverse
transcription-quantitative PCR. (K) The correlation of ATF2 mRNA
and miR-451a expression in 47 HCC tissues. (L) The expression
correlation between ATF2 and miR-451a in HCC was examined using
TCGA database. (M) The relation between ATF2 mRNA and ZSCAN16-AS1
in 47 HCC tissues. (N) The expression correlation between
ZSCAN16-AS1 and ATF2 in HCC was examined using TCGA database.
**P<0.01. miR, microRNA; ATF2, activating transcription factor
2; HCC, hepatocellular carcinoma; UTR, untranslated region; wt,
wild-type; mut, mutant; NC, negative control; TCGA, The Cancer
Genome Atlas; si, small interfering. |
miR-451a/ATF2 axis mediates the
pro-oncogenic actions of ZSCAN16-AS1 in HCC
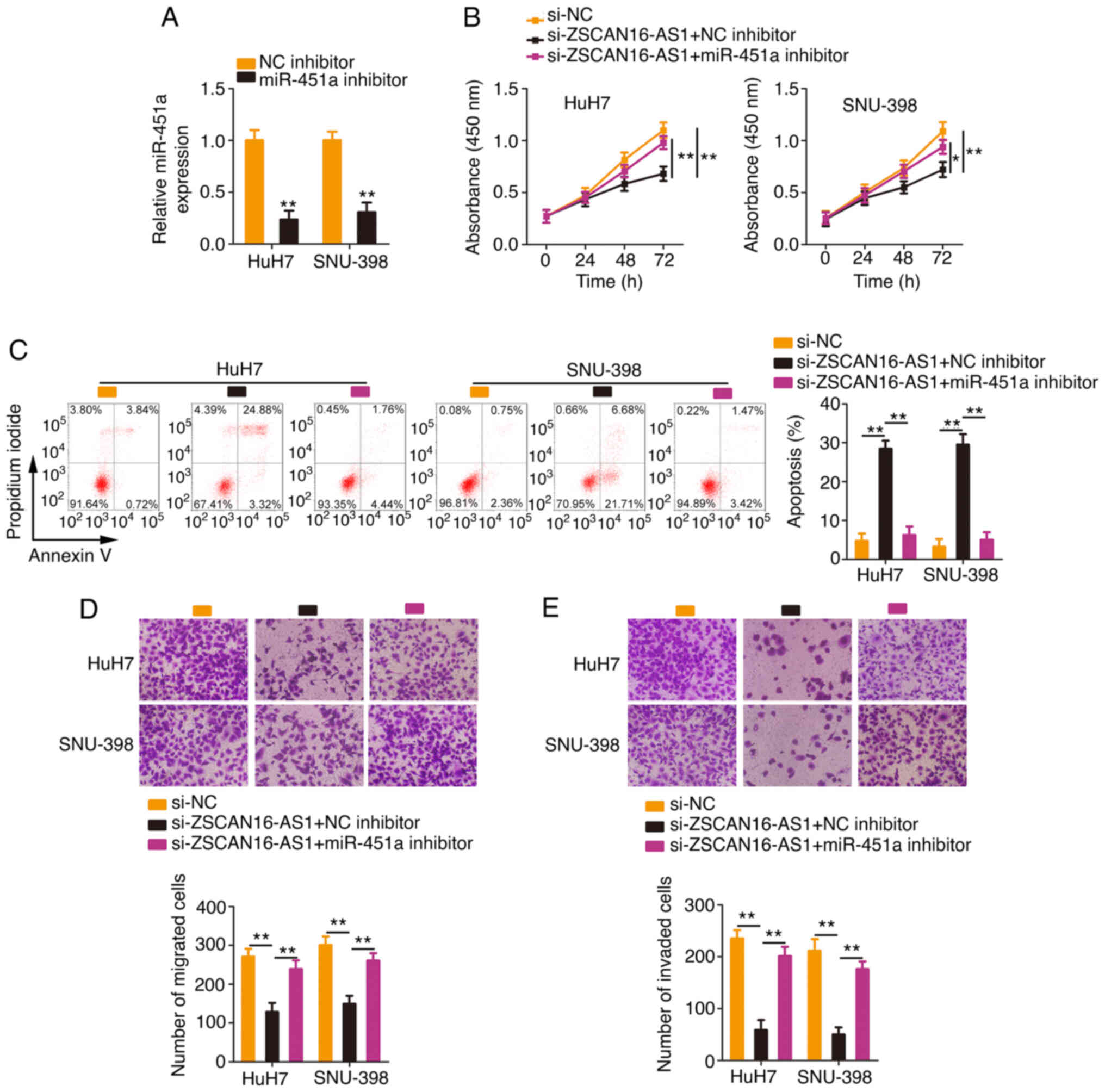
Rescue experiments were conducted with the aim of
illustrating whether the miR-451a/ATF2 axis is required for the
cancer-repressing actions of si-ZSCAN16-AS1 in HCC cells. The
efficiency of the miR-451a inhibitor transfection was tested via
RT-qPCR (Fig. 6A). The miR-451a
inhibitor or NC inhibitor together with si-ZSCAN16-AS1 were
introduced into HCC cells. The inhibition of miR-451a ameliorated
the antiproliferative roles of si-ZSCAN16-AS1 in HCC cells
(Fig. 6B). Flow cytometry
demonstrated that miR-451a inhibitor cotransfection eliminated the
promotive effect of ZSCAN16-AS1 depletion on HCC cell apoptosis
(Fig. 6C). Similarly, Transwell
cell migration and invasion assays showed the same tendency
(Fig. 6D and E).
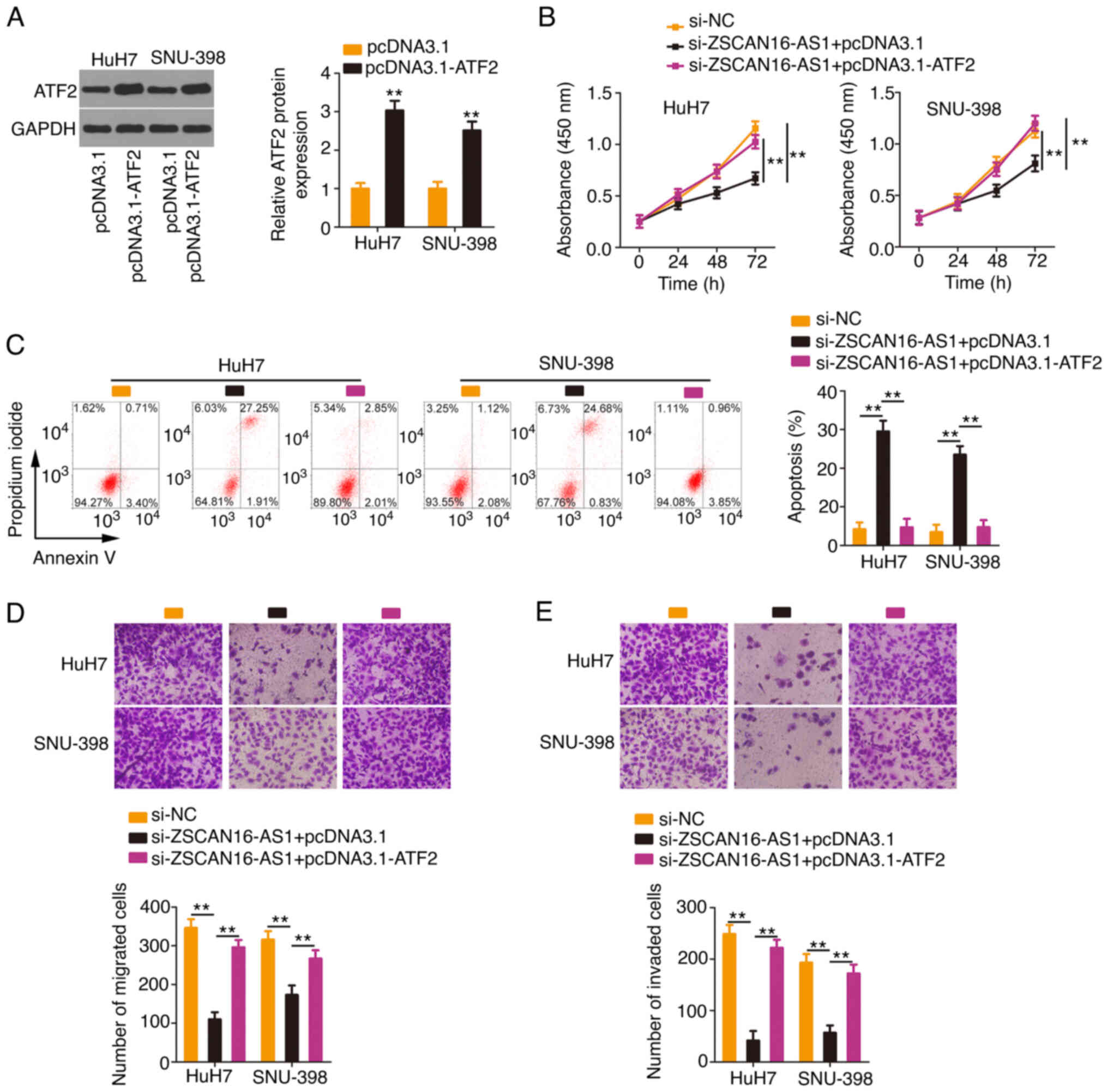
HCC cells were transfected with the
ATF2-overexpressing plasmid pcDNA 3.1-ATF2 (Fig. 7A) or empty pcDNA 3.1 plasmid in the
presence of si-ZSCAN16-AS1. Proliferation inhibition and apoptosis
promotion (Fig. 7B and C) by
ZSCAN16-AS1 silencing could be rescued in HCC cells after
cotransfection with pcDNA3.1-ATF2. In addition, ATF2 overexpression
restored the motility (Fig. 7D and
E) of HCC cells, which were hindered by si-ZSCAN16-AS1. The
above results affirmed that ZSCAN16-AS1 exerted tumorigenic roles
in HCC via the miR-451a/ATF2 axis.
Discussion
The importance of lncRNAs in the oncogenicity of HCC
has been widely studied (15,24).
Studies have demonstrated the abnormal expression of lncRNAs in HCC
and confirm it to be an important driving force for HCC malignancy
(25–27). Thus, lncRNAs may be effective
targets for HCC diagnosis, prognosis and therapy. At present,
numerous lncRNAs have been validated in the human genome by
transcriptome sequencing, but the detailed roles of various lncRNAs
in HCC genesis and development have not been thoroughly
characterized and remain to be determined. The present study
clarified whether ZSCAN16-AS1 was implicated in the aggressive
properties of HCC and the possible mechanisms involved were
explored.
A number of lncRNAs are reported to be involved in
HCC etiology and progression. For example, ANCR (28), LINC02580 (29) and MT1JP (30) are underexpressed in HCC and are
confirmed to be tumor suppressors. By contrast, TMPO-AS1 (31), ST8SIA6-AS1 (32) and ADAMTS9-AS1 (33) are overexpressed in HCC and aggravate
malignant characteristics. However, until now, there has been no
relevant study regarding the relationship between ZSCAN16-AS1 and
HCC progression. In the present study, the data confirmed the high
expression of ZSCAN16-AS1 in HCC by the TCGA database and the
cohort of the present study. Analysis of the survival data revealed
that overexpressed ZSCAN16-AS1 exhibited a prominent correlation
with poor clinical outcomes of HCC patients. Following ZSCAN16-AS1
silencing, HCC cell proliferation, migration and invasion were
inhibited, whereas cell apoptosis was promoted in vitro.
Furthermore, the absence of ZSCAN16-AS1 restricted the tumor growth
of HCC cells in vivo. Accordingly, the present study
identified a novel oncogenic lncRNA, ZSCAN16-AS1, in HCC.
An understanding of the detailed mechanisms by which
ZSCAN16-AS1 regulates HCC malignancy may offer novel insights into
gene regulatory networks, which may lead to important implications
in cancer. As gene regulators, lncRNAs take part in gene regulation
by employing different mechanisms. Generally, lncRNAs located in
the nucleus directly bind to proteins and control gene expression
at the transcriptional level (34).
By contrast, cytoplasmic lncRNAs serve as endogenous decoys for
miRNAs through sequence complementarity, thereby influencing gene
expression and signal transduction pathways at the
posttranscriptional level (35).
Consequently, the actions of lncRNAs are determined by their unique
subcellular localizations. The present study demonstrated that
ZSCAN16-AS1 was prominently distributed in the cytoplasm of HCC
cells, which provided evidence for ZSCAN16-AS1 as a ceRNA.
Using StarBase 3.0, a potential interaction between
miR-451a and ZSCAN16-AS1 was predicted. RT-qPCR analysis
demonstrated that the downregulation of ZSCAN16-AS1 increased
miR-451a expression in HCC cells. A luciferase reporter assay
combined with RIP and correlation analysis identified ZSCAN16-AS1
as an endogenous molecular sponge for miR-451a. miR-451a was
further demonstrated to directly target ATF2 in HCC cells through
mechanistic experiments. In addition, ATF2 was shown to be
positively controlled by ZSCAN16-AS1 in HCC cells, via miR-451a
sequestration. Together these results established a new ceRNA
pathway in HCC that consists of ZSCAN16-AS1, miR-451a and ATF2.
Low expression of miR-451a was previously reported
in HCC (36,37), which was in accordance with the
present study. In addition, the data revealed that miR-451a showed
a significant correlation with the tumor grade and lymph node
metastasis of HCC patients and served a cancer-inhibiting role in
HCC cells. Mechanistically, ATF2 is a downstream effector of
miR-451a in HCC, as verified by bioinformatics prediction, the
luciferase reporter assay and molecular analysis. ATF2, which is a
member of the cAMP response element-binding family, performs
important regulatory activities in the genesis and progression of
various human cancers (38,39). In the present study, ATF2 was found
to be in control of the ZSCAN16-AS1/miR-451a axis. Notably, rescue
experiments further confirmed that miR-451a knockdown or ATF2
resumption rescued the proliferation suppression, apoptosis
promotion and migration and invasion inhibition caused by
ZSCAN16-AS1 depletion. Therefore, the oncogenicity of HCC was
weakened by ZSCAN16-AS1 due to its nature as a ceRNA and sponge of
miR-451a, which thus alleviated miR-451a-mediated ATF2
downregulation.
The present study identified a novel lncRNA,
ZSCAN16-AS1, which was overexpressed in HCC and related to poor
prognosis in HCC patients. ZSCAN16-AS1 aggravated the malignancy of
HCC cells by regulating the miR-451a/ATF2 axis. An in-depth
understanding of the ceRNA network of ZSCAN16-AS1/miR-451a/ATF2 in
HCC might be instrumental in the development of attractive targets
for molecular therapy.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author upon reasonable
request.
Authors' contributions
WW and CL designed the study and wrote the
manuscript. CL, QW, CS, HW and BZ conducted all experiments. QW
analyzed the data. CL and WW confirm the authenticity of all the
raw data. All authors have read and approved the final draft.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of The People's Hospital of Tongliang District and was
conducted in accordance with the principles of the Declaration of
Helsinki. All patients provided informed written consent for the
use of their tissue samples. Experiments involving animals were
implemented under the approval of the Institutional Animal Care and
Use Committee of The People's Hospital of Tongliang District.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2020. CA Cancer J Clin. 70:7–30. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Helal Tel A, Radwan NA and Shaker M:
Extrahepatic metastases as initial manifestations of hepatocellular
carcinoma: An Egyptian experience. Diagn Pathol. 10:822015.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Inchingolo R, Posa A, Mariappan M and
Spiliopoulos S: Locoregional treatments for hepatocellular
carcinoma: Current evidence and future directions. World J
Gastroenterol. 25:4614–4628. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Kulik L and El-Serag HB: Epidemiology and
management of hepatocellular carcinoma. Gastroenterology.
156:477–491.e1. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gelli M, Sebagh M, Porcher R, Romanelli E,
Vibert E, Sa Cunha A, Castaing D, Rosmorduc O, Samuel D, Adam R and
Cherqui D: Liver resection for early hepatocellular carcinoma:
Preoperative predictors of non transplantable recurrence and
implications for treatment allocation. Ann Surg. 272:820–826. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Djebali S, Davis CA, Merkel A, Dobin A,
Lassmann T, Mortazavi A, Tanzer A, Lagarde J, Lin W, Schlesinger F,
et al: Landscape of transcription in human cells. Nature.
489:101–108. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Karlsson O and Baccarelli AA:
Environmental health and long non-coding RNAs. Curr Environ Health
Rep. 3:178–187. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Peng WX, Koirala P and Mo YY:
LncRNA-mediated regulation of cell signaling in cancer. Oncogene.
36:5661–5667. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bhan A, Soleimani M and Mandal SS: Long
noncoding RNA and cancer: A new paradigm. Cancer Res. 77:3965–3981.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li J, Meng H, Bai Y and Wang K: Regulation
of lncRNA and its role in cancer metastasis. Oncol Res. 23:205–217.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bolha L, Ravnik-Glavac M and Glavac D:
Long noncoding RNAs as biomarkers in cancer. Dis Markers.
2017:72439682017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Botti G, Marra L, Malzone MG, Anniciello
A, Botti C, Franco R and Cantile M: LncRNA HOTAIR as prognostic
circulating marker and potential therapeutic target in patients
with tumor diseases. Curr Drug Targets. 18:27–34. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Heo MJ, Yun J and Kim SG: Role of
non-coding RNAs in liver disease progression to hepatocellular
carcinoma. Arch Pharm Res. 42:48–62. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Abbastabar M, Sarfi M, Golestani A and
Khalili E: lncRNA involvement in hepatocellular carcinoma
metastasis and prognosis. EXCLI J. 17:900–913. 2018.PubMed/NCBI
|
|
16
|
Chen X, Fan S and Song E: Noncoding RNAs:
New players in cancers. Adv Exp Med Biol. 927:1–47. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Abba M, Mudduluru G and Allgayer H:
MicroRNAs in cancer: Small molecules, big chances. Anticancer
Agents Med Chem. 12:733–743. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yang C, Wang Y, Xue W, Xie Y, Dong Q and
Zhu C: Competing endogenous RNA (ceRNA) network analysis of
autophagy-related genes in hepatocellular carcinoma. Pharmgenomics
Pers Med. 13:445–462. 2020.PubMed/NCBI
|
|
19
|
Niu ZS, Wang WH, Dong XN and Tian LM: Role
of long noncoding RNA-mediated competing endogenous RNA regulatory
network in hepatocellular carcinoma. World J Gastroenterol.
26:4240–4260. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Vaes E, Khan M and Mombaerts P:
Statistical analysis of differential gene expression relative to a
fold change threshold on NanoString data of mouse odorant receptor
genes. BMC bioinformatics. 15:392014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Ballester M, Cordon R and Folch JM: DAG
expression: High-throughput gene expression analysis of real-time
PCR data using standard curves for relative quantification. PLoS
One. 8:e803852013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
van Iterson M, t Hoen PA, Pedotti P,
Hooiveld GJ, den Dunnen JT, van Ommen GJ, Boer JM and Menezes RX:
Relative power and sample size analysis on gene expression
profiling data. BMC Genom. 10:4392009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Peng L, Yuan XQ, Zhang CY, Peng JY, Zhang
YQ, Pan X and Li GC: The emergence of long non-coding RNAs in
hepatocellular carcinoma: An update. J Cancer. 9:2549–2558. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Mai H, Zhou B, Liu L, Yang F, Conran C, Ji
Y, Hou J and Jiang D: Molecular pattern of lncRNAs in
hepatocellular carcinoma. J Exp Clin Cancer Res. 38:1982019.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lim LJ, Wong SYS, Huang F, Lim S, Chong
SS, Ooi LL, Kon OL and Lee CG: Roles and regulation of long
noncoding RNAs in hepatocellular carcinoma. Cancer Res.
79:5131–5139. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wei L, Wang X, Lv L, Liu J, Xing H, Song
Y, Xie M, Lei T, Zhang N and Yang M: The emerging role of microRNAs
and long noncoding RNAs in drug resistance of hepatocellular
carcinoma. Mol Cancer. 18:1472019. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Song XZ, Xu XJ, Ren XN, Ruan XX, Wang YL
and Yao TT: LncRNA ANCR suppresses the progression of
hepatocellular carcinoma through the inhibition of Wnt/β-catenin
signaling pathway. OncoTargets Ther. 13:8907–8917. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Xu L, Wang Z, Yin C, Pan F, Shi T and Tian
Y: Long noncoding RNA LINC02580 suppresses the invasion-metastasis
cascade in hepatocellular carcinoma by targeting SRSF1. Biochem
Biophys Res Commun. 533:685–691. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mo W, Dai Y, Chen J, Liang L, Xu S and Xu
X: Long Noncoding RNA (lncRNA) MT1JP Suppresses Hepatocellular
Carcinoma (HCC) in vitro. Cancer Manag Res. 12:7949–7960. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu X and Shen Z: LncRNA TMPO-AS1
aggravates the development of hepatocellular carcinoma via
miR-429/GOT1 axis. Am J Med Sci. 360:711–720. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang X, Xu S, Hu C, Fang K, Zhou J, Guo
Z, Zhu G and Li L: LncRNA ST8SIA6-AS1 promotes hepatocellular
carcinoma progression by regulating MAGEA3 and DCAF4L2 expression.
Biochem Biophys Res Commun. 533:1039–1047. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Zhang Z, Li H, Hu Y and Wang F: Long
non-coding RNA ADAMTS9-AS1 exacerbates cell proliferation,
migration, and invasion via triggering of the PI3K/AKT/mTOR pathway
in hepatocellular carcinoma cells. Am J Transl Res. 12:5696–5707.
2020.PubMed/NCBI
|
|
34
|
Zhang XZ, Liu H and Chen SR: Mechanisms of
long non-coding RNAs in cancers and their dynamic regulations.
Cancers (Basel). 12:12452020. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang H and Lu B: The roles of
ceRNAs-mediated autophagy in cancer chemoresistance and metastasis.
Cancers (Basel). 12:29262020. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wei GY, Hu M, Zhao L and Guo WS: miR-451a
suppresses cell proliferation, metastasis and EMT via targeting
YWHAZ in hepatocellular carcinoma. Eur Rev Med Pharmacol Sci.
23:5158–5167. 2019.PubMed/NCBI
|
|
37
|
Zhao S, Li J, Zhang G, Wang Q, Wu C, Zhang
Q, Wang H, Sun P, Xiang R and Yang S: Exosomal miR-451a functions
as a tumor suppressor in hepatocellular carcinoma by targeting
LPIN1. Cell Physiol Biochem. 53:19–35. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Huebner K, Procházka J, Monteiro AC,
Mahadevan V and Schneider-Stock R: The activating transcription
factor 2: An influencer of cancer progression. Mutagenesis.
34:375–389. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Watson G, Ronai ZA and Lau E: ATF2, a
paradigm of the multifaceted regulation of transcription factors in
biology and disease. Pharmacol Res. 119:347–357. 2017. View Article : Google Scholar : PubMed/NCBI
|