Introduction
The global morbidity rate of preeclampsia (PE) is
3–7%. According to statistics, 10–20% of maternal deaths are
associated with PE, particularly early onset PE that occurs before
34 weeks of pregnancy, which is a serious threat to maternal and
child health (1). The etiology and
pathogenesis of PE have always been important research topics in
obstetrics. Over the years, through several studies (2–5) on PE,
several pathogenesis hypotheses have been formulated, including:
Vascular endothelial cell damage, insufficient remodeling of spiral
uterine arteries, excessive activation of inflammatory immunity,
genetic factors and insulin resistance. However, the mechanism of
its pathogenesis is yet to be completely understood.
The invasion of trophoblasts is regulated by various
intracellular and extracellular signaling pathways involving
multiple processes, such as recognition and digestion of the
extracellular matrix, and directional migration (6). Cytoskeleton dynamic remodeling is an
important target for regulating the invasion and migration of
trophoblasts (7).
The formation of placental blood vessels ensures
that the placenta has sufficient blood perfusion and meets the
growth requirements of the fetus. Abnormal formation of placental
blood vessels can lead to a decrease in placental blood perfusion,
ischemia and hypoxia in the uterine placenta, causing PE and growth
restriction (2,8). Therefore, the successful completion of
the placental vascular development process is the key to
maintaining the pregnancy process and pregnancy outcome. During a
normal pregnancy, the balance between pro-angiogenic factors and
anti-angiogenic factors is maintained to promote placental
vascularization and development (9). Moreover, during the process of normal
placental angiogenesis, the formation of blood vessel branches
gradually increases; however, in patients with PE, capillary
network formation is impaired (10).
MicroRNAs (miRNAs/miRs) are a class of small
non-coding RNAs with a length of ~21–25 nucleotides, that regulate
target gene expression by complementary binding to specific sites
in the 3′ untranslated region (3′UTR) of target gene transcripts,
which is an important mechanism of epigenetic regulation. It is now
known that the placenta has its specific miRNA family cluster, and
these miRNAs are involved in the functional regulation of the
placenta (11,12). Furthermore, the dysregulation of
miRNAs in the placenta of patients with PE during pregnancy is
closely associated with the occurrence and development of this
disease (13–15). Additionally, a variety of miRNAs
(such as miR-125a-5p and miR-215-5p) are expressed in trophoblasts,
and regulate cell proliferation, differentiation, invasion and
apoptosis by targeting mRNAs (15–17).
Numerous studies have also reported that miRNAs may be involved in
vascular remodeling and immune function regulation at the
maternal-fetal interface (18–21).
miR-302a is a member of the miR-302a/367 cluster,
and has been investigated in various types of cancer, such as
breast and cervical cancer and glioblastoma (22–27).
To the best of our knowledge, however, few studies have focused on
the role of miR-302a in the pathogenesis of PE. The present study
aimed to assess the expression levels of miR-302a in patients with
PE and matched normotensive women using reverse
transcription-quantitative (RT-q)PCR. The biological function of
miR-302a in the formation of PE was investigated by cell viability,
colony formation, migration and invasion assays. Furthermore, the
molecular regulation mechanism was also assessed by dual-luciferase
reporter, RT-qPCR and western blot assay.
Materials and methods
Tissues
The placental samples from patients with PE (n=35;
16 severe and 19 non-severe) and normal pregnancy controls (n=20;
age, 26–34 years) were collected from the Department of Obstetrics,
Jinan City People's Hospital (Jinan, China) from January 2017 to
December 2019.
Inclusion criteria were as follows: i) Patients at
28–40 weeks gestation; ii) blood pressure higher >140/90 mmHg
and iii) proteinuria >0.3 g/24 h. Exclusion criteria were as
follows: i) Patients in active labor and ii) patients with a
history other clinical disorders, such as chronic hypertension or
renal disease. Tissue blocks (~1 cm3) were dissected
from placental tissues, washed with sterile PBS and then
immediately stored at −80°C for further research. Written informed
consent was obtained from all participants. The present study was
approved by the Ethics Committee of Jinan City People's Hospital
(approval no. KYLL-2017-276).
Cell culture and transfection
HTR-8/SVneo cells were cultured in RPMI-1640 medium
(Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
(Wisent, Inc.) at 37°C in a humidified 5% CO2 incubator.
Cells were transfected using Lipofectamine® 2000
(Invitrogen; Thermo Fisher Scientific, Inc.) at 37°C for 4–6 h,
according to the manufacturer's protocols. Cells were transfected
with 200 nM miR-302a mimic or miR-302a inhibitor, or 200 nM
negative control (NC) (Shanghai GenePharma Co., Ltd.). Subsequent
experiments were performed 24 h after transfection. The miRNA
sequences were as follows: miR-302a mimic,
5′-UAAGUGCUUCCAUGUUUUGGUGA-3′; miR-NC 5′-UUCUCCGAACGUGUCACGUTT−3′;
miR-302a inhibitor 5′-CACCAAAACATGGAAGCACTT−3′; and miR inhibitor
control (5′-TAACACGTCTATACGCCCA-3′).
RT-qPCR. Total RNA was isolated using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.). The miRNA extraction procedures from exosomes were performed
as described previously (28). The
miRNA was reverse-transcribed into cDNA using One Step PrimeScript
miRNA cDNA Synthesis kit (Takara Biotechnology Co., Ltd.) at 37°C
for 60 min followed by 5 min incubation at 85°C. The mRNA was
reverse-transcribed with PrimeScript RT Reagent Kit (Takara
Biotechnology Co., Ltd.) at 37°C for 15 min and 85°C for 5 sec. The
relative expression levels of miRNAs and mRNA were assessed using
the SYBR Premix Ex Taq™ II kit (Takara Biotechnology Co., Ltd.).
The thermocycling conditions were as follows: Initial denaturation
at 95°C for 30 sec, 40 cycles of annealing at 95°C for 5 sec and
elongation and extension at 60°C for 30 sec. Small nuclear RNA U6
expression was used as an endogenous control for miRNA, and GAPDH
was used as an internal control for mRNAs. Relative expression was
analyzed using the 2−ΔΔCq method (29). The primers used were as follows:
VEGFA forward, 5′-AACTTTCTGCTGTCTTGGGT-3′ and reverse,
5′-TCTCGATTGGATGGCAGTA-3′; GAPDH forward,
5′-GTCTCCTCTGACTTCAACAGCG-3′ and reverse,
5′-ACCACCCTGTTGCTGTAGCCAA-3′; miR-302a forward,
5′-TAAGTGCTTCCATGTTTTGGTGA-3′ and reverse,
5′-GAACATGTCTGCGTATCTCAGACTTC-3′; and U6 forward,
5′-GCTTCGGCAGCACATATACTAAAAT-3′ and reverse,
5′-CGCTTCACGAATTTGCGTGTCAT-3′.
Western blotting
Tissues and cells were homogenized in RIPA lysis
bu-er (Beyotime Institute of Biotechnology) and the protein
concentration was determined using a BCA protein quantitation kit.
For each sample, 60 µg protein was separated by 10% SDS-PAGE and
transferred to a 0.45-µm nitrocellulose membrane. The membrane was
blocked with 5% skimmed milk in PBS for 1 h at room temperature.
Following three washes with PBS, the membrane was incubated with
the primary antibodies (dilution, 1:1,000-1:2,000) overnight at
4°C. Following washing with PBS, the membranes were incubated with
HRP-conjugated secondary antibodies (goat anti-rabbit IgG-HRP,
1:3,000, cat. no. M21002; goat anti-mouse IgG-HRP, 1:3,000, cat.
no. M21001; both Abmart Pharmaceutical Technology Co., Ltd.) at
room temperature for 1 h. The signal intensity was detected using
an enhanced chemiluminescence detection system (PerkinElmer, Inc.)
and Image Quant LAS 4000 (GE Healthcare Life Sciences). β-actin was
used as an internal control. ImageJ 1.52a (National Institutes of
Health) was used for densitometry.
The primary antibodies used were as follows:
N-cadherin (cat. no. 13116; Cell Signaling Technology, Inc.),
E-cadherin (cat. no. 3195; Cell Signaling Technology, Inc.),
Vimentin (cat. no. 5741; Cell Signaling Technology, Inc.), snail
family transcriptional repressor 1 (Snail; cat. no. 3879; Cell
Signaling Technology, Inc.), β-actin (cat. no. 4970; Cell Signaling
Technology, Inc.), VEGFA (cat. no. ab1316; Abcam), CD63 (cat. no.
55051; Cell Signaling Technology, Inc.), tumor susceptibility 101
(TSG101; cat. no. ab125011; Abcam), notch receptor 1 (Notch 1; cat.
no. 3608; Cell Signaling Technology, Inc.), Notch 2 (cat. no. 5732;
Cell Signaling Technology, Inc.), δ like canonical Notch ligand 4
(Dll4; cat. no. 2589; Cell Signaling Technology, Inc.),
phosphorylated (P-)ERK1/2 (cat. no. 4370; Cell Signaling
Technology, Inc.), ERK1/2 (cat. no. 4695; Cell Signaling
Technology, Inc.), P-AKT (cat. no. 4060; Cell Signaling Technology,
Inc.) and AKT (cat. no. 4691; Cell Signaling Technology, Inc.).
Dual-luciferase reporter assay
The TargetScan online tool (targetscan.org/vert_71/) was used to predict the
potential target genes of miR-302a. The 3′UTR of VEGFA was cloned
into the pGL3 vector (Promega Corporation). The mutation in the
miRNA binding site was generated using RCR-based mutagenesis
(Takara Biotechnology Co., Ltd.). The wild-type or mutant
luciferase reporters (50 ng) and 0.5 pmol miR-302a mimics or
negative control were co-transfected into HTR-8/SVneo cells using
Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) at 37°C. Following 36 h transfection, the
luciferase activities were measured using a dual-luciferase
reporter system (Promega Corporation). Renilla luciferase
activity was normalized to firefly luciferase activity.
Cell viability and colony formation
assays
Cell viability was detected using a Cell Counting
Kit-8 (CCK-8; Beyotime Institute of Biotechnology) assay according
to the manufacturer's instructions. Cells were seeded into 96-well
plates at a density of 2×103 cells/well and transfected
with miR-302a mimics, inhibitor or NC for 0, 24, 48, 72 or 96 h. At
the specific time points, 10 µl CCK-8 solution was added into each
well and incubated for 2 h at 37°C. Subsequently, the optical
density value at 490 nm was detected using a Varioskan Flash
microplate reader (Thermo Fisher Scientific, Inc.).
For the colony formation assays, cells were seeded
into 6-well plates at a density of 500 cells/well and incubated at
37°C for 14 days. Subsequently, the cells were washed, fixed with
100% methanol at room temperature for 15 min, then stained with 1%
crystal violet solution at room temperature for 15 min. Images were
captured by camera without magnification.
Cell migration and invasion
assays
Motility was assessed using a wound healing assay.
Cells were seeded onto 12-well plates and incubated with normal
medium with 10% FBS. A wound was created on the surface of the
plates using a 10-100-µl sterile micropipette tip. Subsequently,
cells were washed with PBS and incubated in serum-free medium at
37°C for 48 h. Images were captured by light microscope
(magnification, ×200; Olympus Corporation). The distances were
measured using ImageJ 1.52a software (National Institutes of
Health).
Cell migratory and invasive abilities were measured
using Transwell assays. Transwell chambers (8-µm pores; BD
Biosciences) were used. Cells were added to the top chamber of the
inserts, and 600 µl medium with 20% FBS was added to the lower
chamber. For migration assay, the chambers were not coated with
Matrigel (BD Biosciences). However, the chambers were coated with
Matrigel at 37°C for 30 min in invasion assays. Following
incubation at 37°C for 24 h, cells in the upper chamber were
removed with a cotton swab, and cells on the lower surface of the
chamber were fixed with 100% methanol at room temperature for 15
min and stained with 1% crystal violet solution at room temperature
for 15 min. The cells were counted under a light microscope
(magnification, ×200; Olympus Corporation).
Vascular ring formation
Matrigel (BD Biosciences) was added to each well of
a 24-well plate and allowed to solidify at 37°C for 30 min.
Subsequently, HUVECs (American Type Culture Collection) were
transferred to the wells at a density of 1×105
cells/well. Following cell incubation at 37°C for 24 h in a
humidified chamber with 5% CO2 in air, the formation of
capillary-like structures was examined under a light microscope
(magnification, ×200). For each well, three fields in the central
area were selected randomly.
Isolation of exosomes and
purification
The isolation and purification of exosomes were
performed as described previously by Thery et al (30). Exosomes were isolated from
supernatant of the HTR-8/SVneo cells via differential
centrifugation. The cells were initially removed by centrifugation
at 300 × g, and other debris were removed at 3,000 × g. The
supernatant was centrifuged at 10,000 × g to remove shedding
vesicles and other vesicles with larger sizes. Finally, the
supernatant was centrifuged at 110,000 × g for 70 min. All
ultracentrifugation steps were performed at 4°C. Exosomes were
collected from the pellet and resuspended in PBS. The exosomes were
visualized by transmission electron microscopy (magnification,
×400; JEOL, Ltd.) as previously reported (31).
Isolation of exosomes was performed using exosome
isolation reagent for plasma or serum (cat. no. C10110-2; Guangzhou
RiboBio Co., Ltd.), according to the manufacturer's protocol.
Chemical assays
For RNase assay, the exosome pellet was suspended
with PBS (500 µl) and 10 µg/ml RNase A (Omega Bio-Tek, Inc.) was
added. The mixture was incubated at 37°C for 1 h.
The exosome inhibitor GW4869 (10 µM; Sigma-Aldrich;
Merck KGaA) was used to inhibit exosome secretion. GW4869 were
pre-incubated with trophoblast cells at 37°C for 1 h before the
secretion assay.
Statistical analysis
All data are presented as the mean ± SD of at least
three independent experiments. All statistical analyses were
performed using GraphPad Prism 6 (GraphPad Software, Inc.).
Differences were analyzed using a paired Student's t-test for two
group comparisons. The association between miR-302a expression and
VEGFA expression was analyzed via Pearson's correlation analysis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
miR-302a expression is increased in
patients with PE
Placental samples were collected from 35 patients
with PE and 20 matched normotensive women. The basic clinical
characteristics of the patients in the PE and control groups are
presented in Table I. Patients with
PE exhibited a significant increase in the systolic pressure and
diastolic blood pressure compared with patients in the control
group. Proteinuria was also identified in the PE group. Moreover,
the gestational age at delivery of patients with PE was
significantly lower compared with that of patients in the control
group.
 | Table I.Clinical characteristics of
preeclamptic and normal pregnancies. |
Table I.
Clinical characteristics of
preeclamptic and normal pregnancies.
|
Characteristics | Normal | Preeclampsia | P-value |
|---|
| Maternal age,
years | 29±2.67 | 30±3.22 | 0.245 |
| Systolic blood
pressure, mmHg | 117±10 | 134±13 | <0.0001 |
| Diastolic blood
pressure, mmHg | 68±6 | 89±8 | <0.0001 |
| Proteinuria | 0 | 1.94±0.42 | <0.0001 |
| BMI,
kg/m2 | 29.1±4.3 | 29.6±5.2 | 0.7171 |
| Birth weight,
g | 3564±537 | 3018±452 | 0.002 |
| Gestational age at
delivery, weeks | 39.2±1.8 | 37.1±1.5 | <0.0001 |
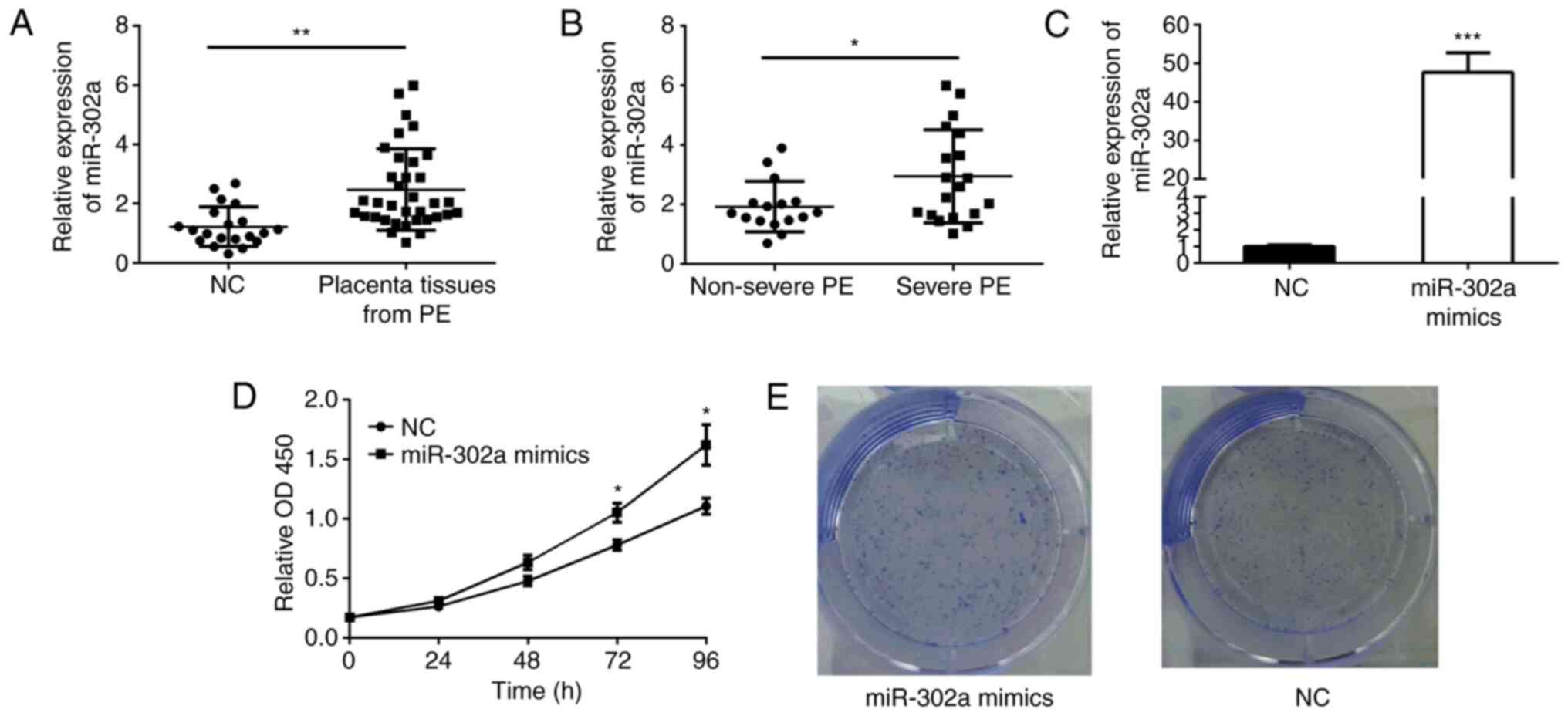
The relative expression levels of miR-302a in
placental tissues from patients with PE (n=35) and normal controls
(n=20) were detected using RT-qPCR. The results demonstrated that
miR-302a expression was upregulated in PE (Fig. 1A). Subsequently, the present study
compared the relative expression levels of miR-302a in placental
tissues from patients with severe PE (n=16) and non-severe PE
(n=19), and revealed that the expression of miR-302a was higher in
patients with severe compared with non-severe PE (Fig. 1B).
miR-302a overexpression promotes cell
proliferation
The miR-302a mimics were stably transfected into
HTR-8/SVneo cells. The overexpression of miR-302a was confirmed by
RT-qPCR, and the relative expression levels of miR-302a were
increased in the miR-302a mimics group compared with in the NC
group (Fig. 1C). The CCK-8 assay
demonstrated that the relative cell viability in the miR-302a
mimics group was higher than that in the NC group (Fig. 1D). Similarly, in a colony formation
assay, the overexpression of miR-302a markedly promoted colony
formation (Fig. 1E).
miR-302a overexpression promotes cell
migration and invasion
A Transwell assay was used to detect the effect of
miR-302a on cell migration and invasion. The results revealed that
overexpression of miR-302a increased the invasive and migratory
capacity compared with that in the NC group (Fig. 2A and B). A wound healing assay was
used to investigate the effect of miR-302a on the motility of
cells. An increased migratory rate was observed in the miR-302a
overexpression group (Fig. 2C).
Additionally, these results could be confirmed by investigating the
expression levels of epithelial-mesenchymal transition markers.
Western blot analysis results demonstrated that overexpression of
miR-302a could downregulate the expression levels of the epithelial
marker E-cadherin (0.602-fold change), and upregulate the
expression levels of mesenchymal markers, including N-cadherin
(1.675-fold change), Vimentin (1.634-fold change), Snail
(1.473-fold change), p-AKT/AKT (0.868-fold change; Fig. 2D).
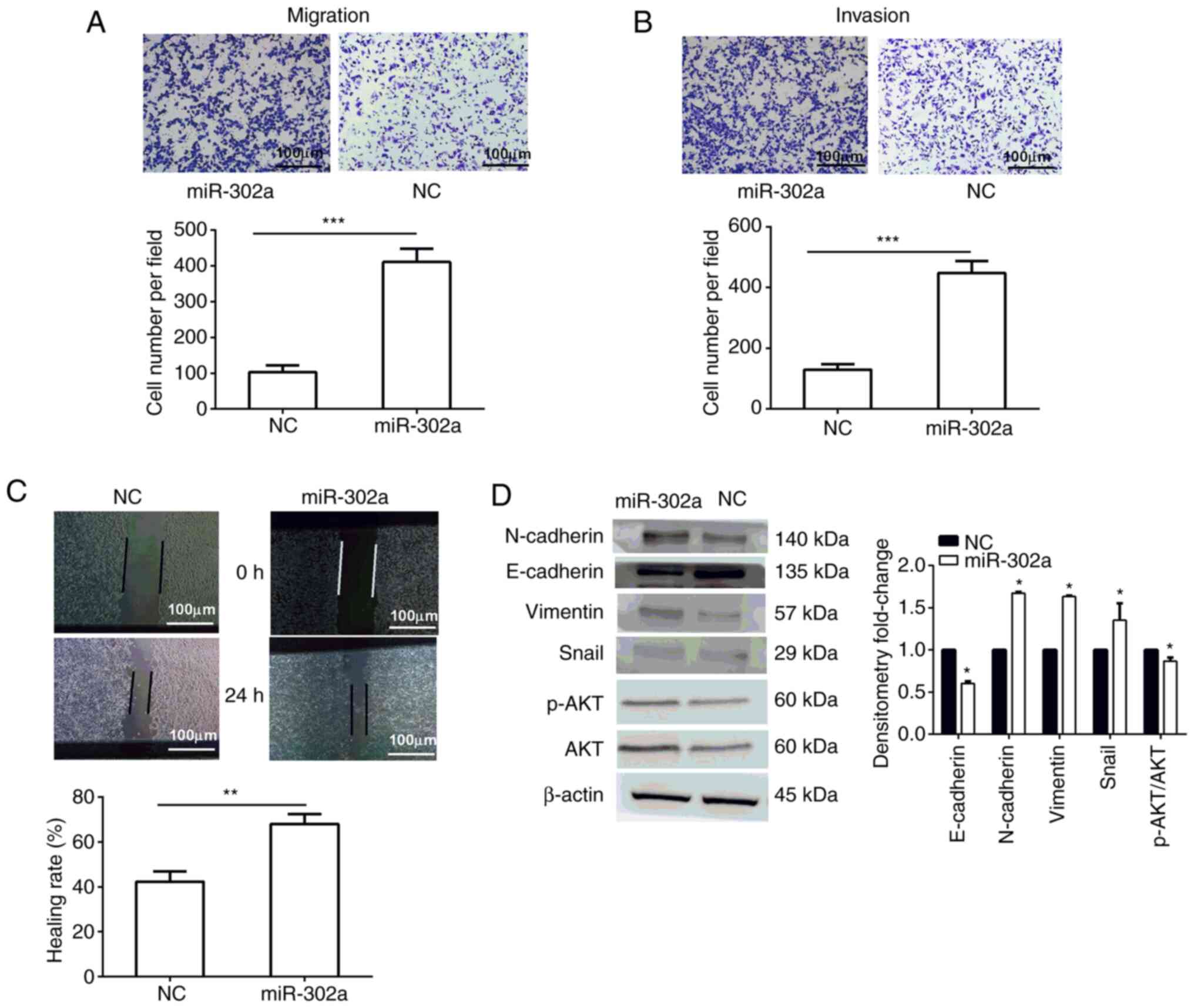 | Figure 2.Role of miR-302a in cell migration
and invasion. (A) Effect of miR-302a on invasion, as determined by
a Transwell assay. (B) Effect of miR-302a on migration, as
determined by a Transwell assay. (C) Cell healing rate detected by
a wound healing assay. Magnification, ×200; scale bar, 100 µm. (D)
Western blot analysis of epithelial-mesenchymal transition markers
and AKT signaling pathway markers. Data are presented as the mean ±
SD (n=3). *P<0.05, **P<0.01, ***P<0.001 vs. NC. miR-302a,
microRNA-302a; NC, negative control; P-, phosphorylated; Snail,
snail family transcriptional repressor 1. |
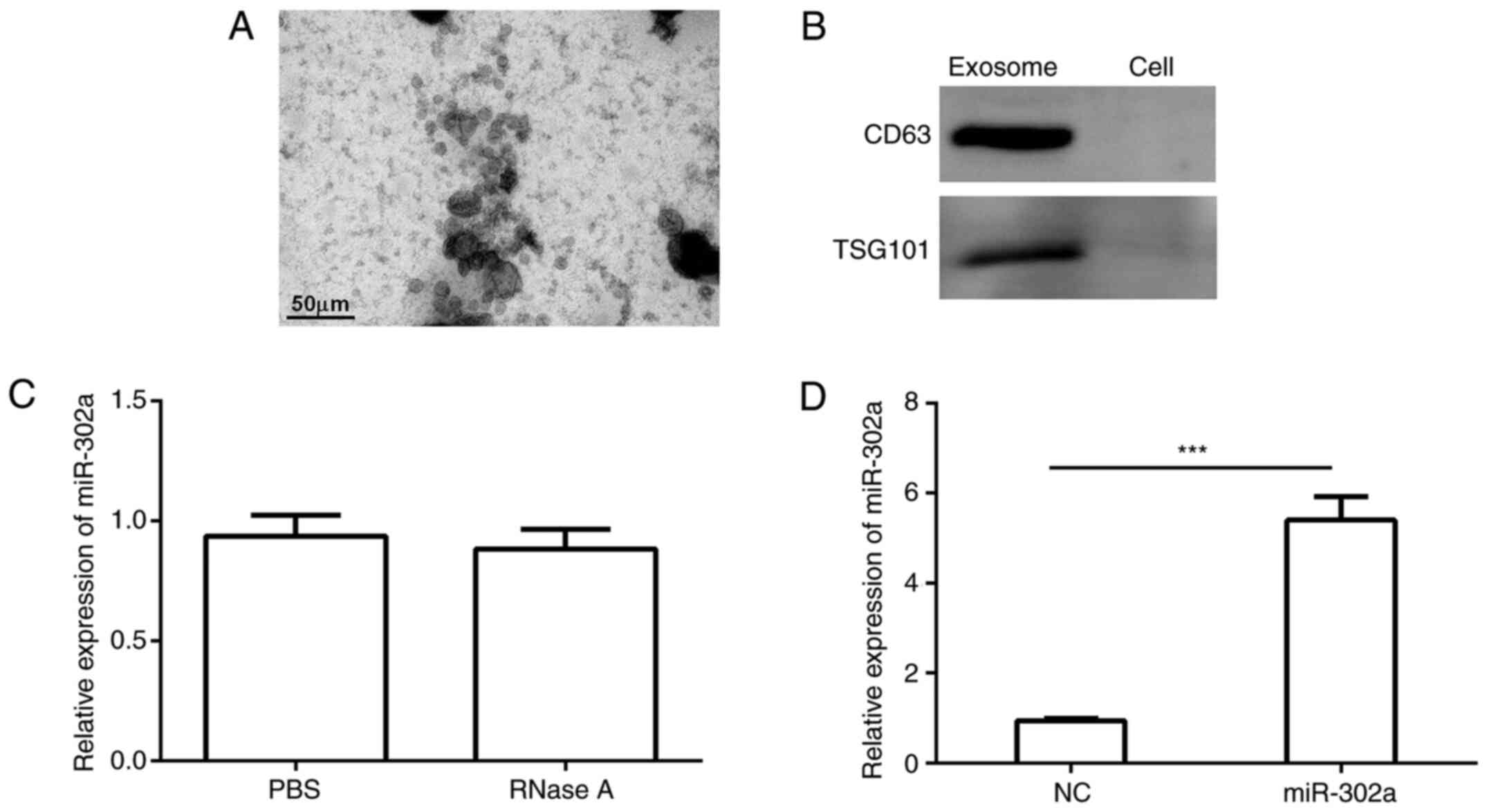
miR-302a is carried by exosomes
Exosomes were isolated from serum via
ultracentrifugation. The exosomes were identified by electron
microscopy according to their typical morphology (Fig. 3A). Additionally, specific protein
markers (TSG101 and CD63) were used to verify that these vesicles
were exosomes (Fig. 3B). To
determine whether miR-302a was indeed present in exosomes, RNase A
(10 µg/ml) or PBS buffer was used to treat exosomes. As shown in
Fig. 3C, no significant difference
was identified between the RNase A and PBS groups. Subsequently,
the present study detected the relative expression levels of
miR-302a in exosomes. The results of RT-qPCR demonstrated that the
relative expression levels of miR-302a in exosomes from the PE
group were significantly higher compared with those in exosomes
from the NC group (Fig. 3D).
The exosome inhibitor GW4869 (10 µM) was applied to
trophoblast cells to inhibit exosome secretion. The results
indicated that the expression level of miR-302a in trophoblast
cells was increased, but not significantly (P>0.05).
Furthermore, the migratory and proliferative abilities were not
markedly promoted (Fig. S1).
Role of exosomal miR-302a in
angiogenesis
Exosomes were obtained from the medium of
HTR-8/SVneo cells transfected with miR-302a or control. The HUVECs
were cultured using these exosomes and their biological function
was evaluated. It was identified that labeled miR-302a could be
transferred via exosomes to medium, and then transferred to HUVECs
via exosomes (Fig. 4A). Compared
with the control group, exosomes with miR-302a overexpression
decreased the invasive ability of HUVECs, as evaluated by a
Transwell assay (Fig. 4B), and
decreased the ring formation ability, as determined by an
endothelial tube formation assay (Fig.
4C). Furthermore, several protein markers of angiogenesis,
including VEGFA (0.302-fold change), Notch 1 (0.693-fold change),
Notch 2 (0.697-fold change), Dll4 (0.574-fold change), P-ERK1/2
(0.439-fold change) and ERK1/2 (0.51-fold change), were detected
using western blotting (Fig. 4D).
The results demonstrated that overexpression of miR-302a
downregulated the expression levels of these protein markers. These
results indicated that miR-302a carried by exosomes inhibited
angiogenesis.
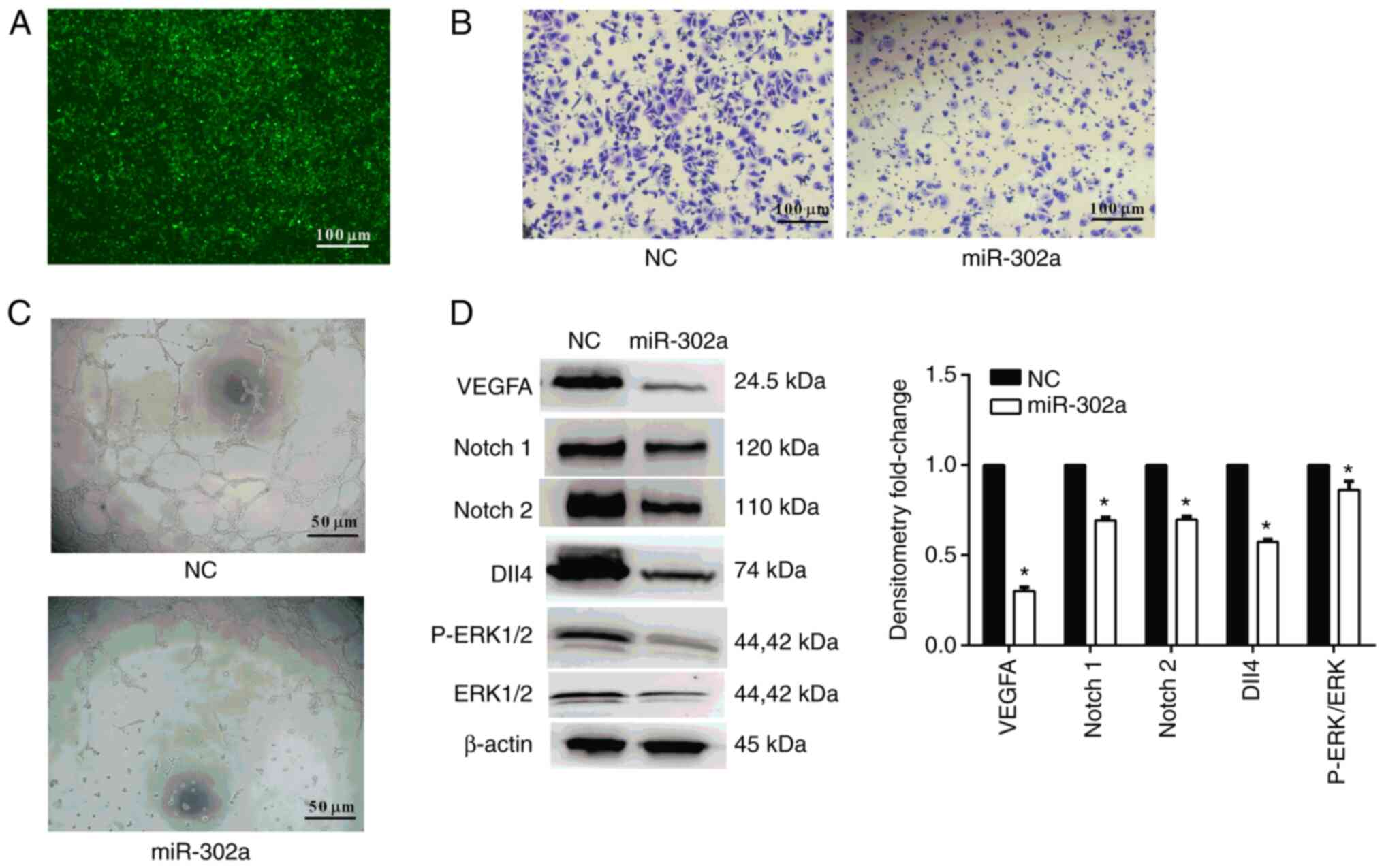 | Figure 4.Effect of exosomal miR-302a on
angiogenesis. (A) Fluorescence microscopy was used to detect the
signals of miR-302a in HUVECs. HTR-8/SVneo cells were transfected
with labeled miR-302a, and then the HUVECs were cultured with Exos
isolated from HTR-8/SVneo medium (magnification, ×100). Scale bar,
100 µm. (B) Effect of exosomal miR-302a on HUVEC invasion detected
by a Transwell assay (magnification, ×200). Scale bar, 100 µm. (C)
Effect of exosomal miR-302a on angiogenesis detected by a ring
formation assay (magnification, ×400). Scale bar, 50 µm. (D)
Western blot analysis of protein markers of angiogenesis. Data are
presented as the mean ± SD (n=3). *P<0.05 vs. NC. miR-302a,
microRNA-302a; NC, negative control; Notch, notch receptor; Dll4, δ
like canonical Notch ligand 4; P-, phosphorylated; Exo,
exosome. |
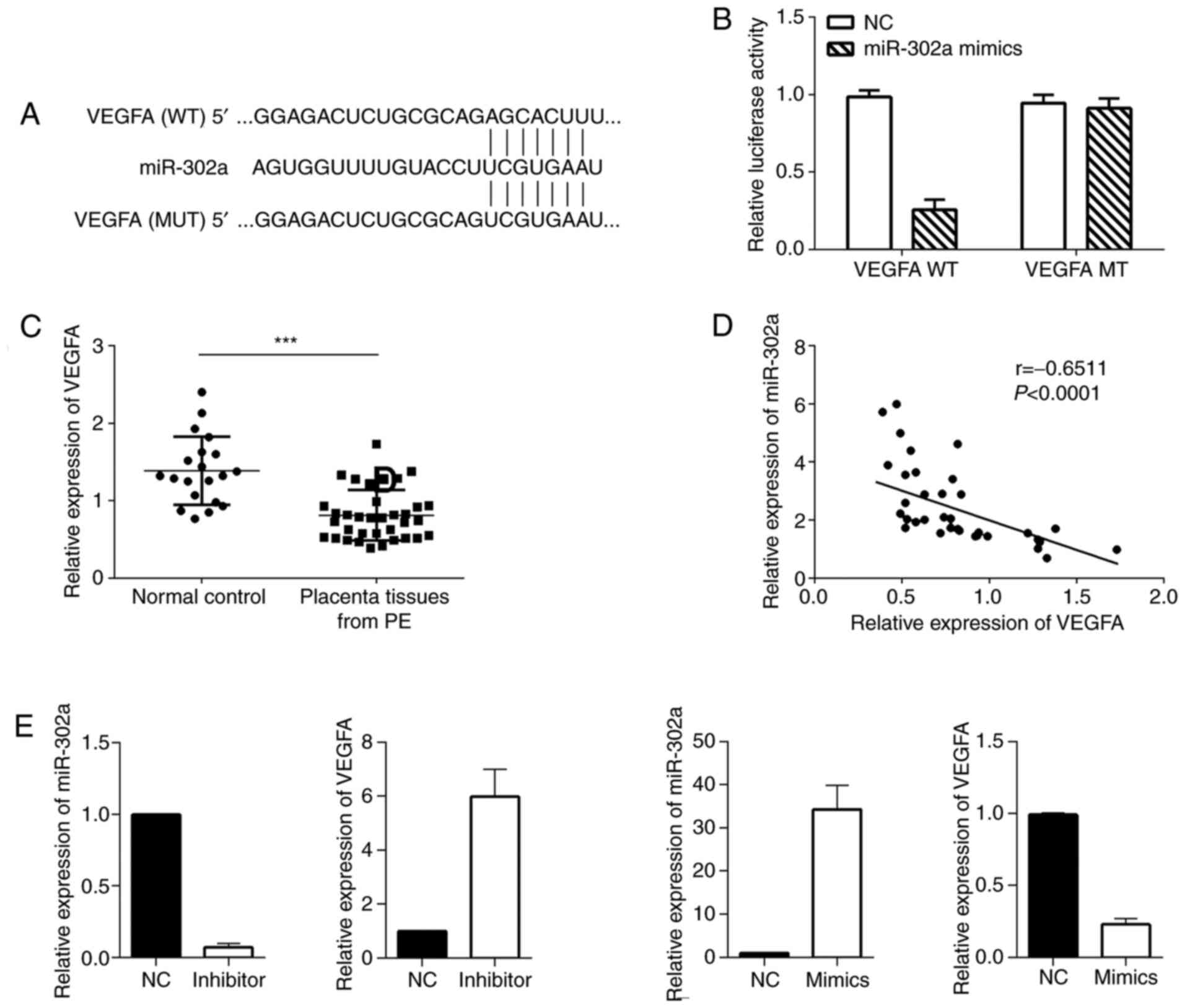
miR-302a directly targets and inhibits
VEGFA expression
The putative targets of miR-302a were predicted
using bioinformatics tools, including TargetScan 7.1. VEGFA was
found to be a putative target of miR-302a, and the binding site was
identified in the 3′UTR of VEGFA mRNA (Fig. 5A). Subsequently, a luciferase assay
was used to determine the interaction between miR-302a and VEGFA
3′UTR. The 3′UTR of VEGFA or the corresponding mutant segments were
cloned into pGL3 vectors and then transfected into cells to detect
the differences in luciferase values. The results demonstrated that
the luciferase activity was markedly reduced in cells transfected
with wild-type 3′UTR of VEGFA, and no obvious reduction was
observed in cells transfected with mutant 3′UTR of VEGFA (Fig. 5B). These results suggested that
miR-302a could directly bind to the 3′UTR of VEGFA.
Subsequently, the relative expression levels of
VEGFA were detected in placental tissues from patients with PE.
Compared with that in the control group, VEGFA expression was
significantly decreased in the PE group (Fig. 5C). Next, the relationship between
the expression levels of VEGFA and miR-302a was analyzed. The
results revealed that VEGFA expression was moderately correlated
with miR-302a expression (Pearson r=−0.6511; P<0.0001; Fig. 5D). To verify the negative regulatory
effect of miR-302a on VEGFA, miR-302a inhibitor or its control were
co-transfected with wild-type or mutant luciferase reporters;
similarly, miR-302a mimics or its control were co-transfected into
cells with the wild-type or mutant luciferase reporters; It was
found that VEGFA expression was increased in the miR-302a-3p
inhibitor group but decreased in the miR-302a-3p mimic group
(Fig. 5E).
Discussion
Placenta formation and development are important
processes for maintaining normal pregnancy, and normal placental
blood vessel formation is a key link between them (32). Abnormal formation of placental blood
vessels can lead to a variety of pregnancy complications, including
PE (33). Blood vessel formation at
the maternal-fetal interface is an important process in normal
pregnancy (34). The interaction of
various regulatory factors and trophoblasts induces angiogenesis of
the placenta, and then completes the remodeling of the uterine
circumflex artery, increases placental blood flow perfusion and
finally forms a highly vascularized organ to ensure the oxygen
supply of the maternal fetus (35,36).
The placenta is rich in angiogenesis-related factors and receptors,
and the main regulatory factors include VEGFs, fibroblast growth
factors and angiopoietin, which interact with cells via paracrine
and autocrine mechanisms to jointly regulate vascular development
and recasting (37–39).
VEGF is a disulfide homodimeric glycoprotein, 40–45
kDa in weight, which has a certain chemotactic effect on
endothelial cells, and is involved in the process of angiogenesis
(40,41). Moreover, its expression is regulated
by multiple cytokines, such as IFN-γ, TNF-α and IL-1β (42,43).
The VEGF family includes VEGF-A, VEGF-B, VEGF-C, VEGF-D, VEGF-E and
placental growth factor. According to alternative splicing, VEGF-A
can be divided into several subtypes, such as VEGF121, VEGF165 and
VEGF189 (44). VEGF is a
pro-angiogenic factor that induces angiogenesis. The mechanism by
which VEGF induces angiogenesis is accomplished by acting on VEGF
receptors on the surface of vascular endothelial cells, promoting
the proliferation and migration of endothelial cells and increasing
vascular permeability (45).
Therefore, VEGF is also referred to as vascular permeability
factor. VEGF is highly expressed in villous trophoblasts and
interstitial villi during the early stages of pregnancy, and its
expression levels are relatively low in syncytiotrophoblast and no
obvious expression is observed in villous endothelial cells
(46,47). The receptors of VEGF include
VEGFR-1, VEGFR-2 and VEGFR-3, the first two of which are tyrosine
kinase receptors expressed in vascular endothelial cells and
trophoblasts (48). VEGF interacts
with VEGFR-1 to promote the proliferation of endothelial cells,
whereas it mainly interacts with VEGFR-2 to mediate most of the
other endothelial cell functions. VEGF has anti-apoptotic effects
and is an important protein in VEGF signaling. VEGF promotes
angiogenesis mainly by activating MEK1/2-ERK1/2 signaling pathway
phosphorylation and mediating cell proliferation (49). Alterations in the levels of the
angiogenesis-related factor VEGF in the maternal blood circulation
lead to angiogenesis dysfunction, which is an important mechanism
for the onset of PE (50).
miR-302a is a member of the miR-302a/367 cluster,
which is highly expressed in embryonic stem cells (51). miR-302a has been identified as a
tumor suppressor in several types of cancer, including breast
cancer, glioblastoma and cervical cancer. For example,
Ahmadalizadeh Khanehsar et al (22) reported that overexpression of the
miR-302a/367 cluster inhibited the proliferation of breast cancer
cells by suppressing the S-phase of the cell cycle. Yang et
al (23) also revealed that the
expression of miR-302a/367 cluster suppressed tumorigenic gene
expression patterns and abolished transformation-related phenotypes
in glioblastoma cells. In cervical cancer, miR-302a has been
identified to inhibit cell migration and invasion by targeting
defective in cullin neddylation 1 domain containing 1 (24). However, in several types of cancer,
miR-302a has been identified as an oncogene. Kim et al
(25) reported that miR-302a
promotes proliferation of human mesenchymal stem cells, while Liu
et al (52) proposed that
miR-302b promoted the proliferation of gastric cancer cells by
targeting CDK2, thereby inhibiting the ERK signaling pathway. In
prostate cancer, miR-302a expression is upregulated, and its forced
expression accelerates the proliferation of prostate cancer cells
(26). In the present study,
miR-302a expression was found to be upregulated in placenta cells
in PE, and overexpression of miR-302a markedly promoted cell
proliferation, migration and invasion.
Exosomes are a class of microvesicles, 30–150 nm in
diameter, and can be involved in intercellular communication by
releasing intracellular cargos, such as miRNAs, mRNAs, long
non-coding RNAs and proteins, into the extracellular environment
(27). Almost all cancer cells can
generate exosomes (53). Emerging
evidence has suggested that exosome-mediated miRNAs are involved in
regulating cancer development. It has been shown that exosomal
transfer of miR-126 promotes the antitumor response in malignant
mesothelioma (54). In gastric
cancer, exosomal transfer of miR-501 confers doxorubicin resistance
and tumorigenesis via targeting of BH3-like motif containing, cell
death inducer (55). Li et
al (56) observed that seven
miRNAs (including miR-153-3p and miR-325-3p) derived from exosomes
were differentially expressed in women with PE. Additionally,
exosomal encapsulation of miR-125a-5p has been identified to
inhibit trophoblast cell migration and proliferation by regulating
VEGFA expression (15). The present
study identified that miR-302a overexpression in HTR-8/SVneo cells
contributed to exosomal miR-302a upregulation. Additionally,
exosomal miR-302a promoted the angiopoiesis of endothelial cells
(HUVECs). The present study successfully constructed HTR-8/SVneo
cells stably overexpressing miR-302a and obtained exosomes that
were rich in miR-302a. Exosomes could be released from HTR-8/SVneo
cells stably overexpressing miR-302a to serum, which further
influenced the biological function of HUVECs. The present study
demonstrated that exosomes with miR-302a overexpression decreased
the invasive ability and inhibited angiogenesis in HUVECs.
The current study performed the aforementioned
experiments only in one cell line and the findings were not
verified in vivo. Moreover, the differential expression of
miR-302a was not verified in the peripheral blood between patients
with PE and normal pregnant women. Moreover, proliferation and
migration belong to different biological functions compared with
angiogenesis. The current study detected that miR-302a promoted the
migration and proliferation of trophoblast cells but suppressed
angiogenesis in HUVECs. The present study focused on the effect of
VEGFA on the angiogenesis of HUVECs. Future studies will further
examine the potential pathways of how miR-302 targeted VEGFA to
affect PE and to identify other potential pathways via which
miR-302 promotes the migration and proliferation of trophoblasts
in vitro and in vivo.
In conclusion, the results of the present study
demonstrated that miR-302a functioned as an oncogene. It was found
that miR-302a promoted the proliferation, migration and invasion of
placenta cells. Furthermore, it was demonstrated that miR-302a
could be released by cells via exosomes, and exosomal miR-302a
repressed the angiogenesis of HUVECs. A luciferase reporter assay
indicated that VEGFA was a direct target of miR-302a. Additionally,
miR-302a expression was negatively correlated with VEGFA
expression. Therefore, it was suggested that miR-302a may regulate
the pathogenesis of PE via VEGFA.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
This study was conceived and designed by LX. The
data was collected, analyzed and interpreted by MW, YZ, LL and GW.
MW, YZ and GW wrote the manuscript. LX and MW confirm the
authenticity of all the raw data. LX inspected the data. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from all
participants. The present study was approved by the Ethics
Committee of Jinan City People's Hospital (approval no.
KYLL-2017-276).
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Ding Z, Liang J, Lu Y, Yu Q, Songyang Z,
Lin SY and Mills GB: A retrovirus-based protein complementation
assay screen reveals functional AKT1-binding partners. Proc Natl
Acad Sci USA. 103:15014–15019. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Peng M, Yang M, Ding Y, Yu L, Deng Y, Lai
W and Hu Y: Mechanism of endogenous digitalis-like factorinduced
vascular endothelial cell damage in patients with severe
preeclampsia. Int J Mol Med. 41:985–994. 2018.PubMed/NCBI
|
|
3
|
Varberg KM and Soares MJ: Paradigms for
investigating invasive trophoblast cell development and
contributions to uterine spiral artery remodeling. Placenta. May
3–2021.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Miko E, Meggyes M, Bogar B, Schmitz N,
Barakonyi A, Varnagy A, Farkas B, Tamas P, Bodis J, Szekeres-Bartho
J, et al: Involvement of Galectin-9/TIM-3 pathway in the systemic
inflammatory response in early-onset preeclampsia. PLoS One.
8:e718112013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Anim-Nyame N, Gamble J, Sooranna SR,
Johnson MR and Steer PJ: Relationship between insulin resistance
and tissue blood flow in preeclampsia. J Hypertens. 33:1057–1063.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Knofler M and Pollheimer J: IFPA award in
placentology lecture: Molecular regulation of human trophoblast
invasion. Placenta. 33 (Suppl 2):S55–S62. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Burke SD, Zsengeller ZK, Khankin EV, Lo
AS, Rajakumar A, DuPont JJ, McCurley A, Moss ME, Zhang D, Clark CD,
et al: Soluble fms-like tyrosine kinase 1 promotes angiotensin II
sensitivity in preeclampsia. J Clin Invest. 126:2561–2574. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Merviel P, Carbillon L, Challier JC,
Rabreau M, Beaufils M and Uzan S: Pathophysiology of preeclampsia:
Links with implantation disorders. Eur J Obstet Gynecol Reprod
Biol. 115:134–147. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hong K, Park HJ and Cha D: Clinical
implications of placenta-derived angiogenic/anti-angiogenic
biomarkers in pre-eclampsia. Biomark Med. 15:523–536. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Grisaru-Granovsky S, Maoz M, Barzilay O,
Yin YJ, Prus D and Bar-Shavit R: Protease activated receptor-1,
PAR1, promotes placenta trophoblast invasion and beta-catenin
stabilization. J Cell Physiol. 218:512–521. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Vaiman D: Genes, epigenetics and miRNA
regulation in the placenta. Placenta. 52:127–133. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Morales-Prieto DM, Ospina-Prieto S,
Chaiwangyen W, Schoenleben M and Markert UR: Pregnancy-associated
miRNA-clusters. J Reprod Immunol. 97:51–61. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jairajpuri DS, Malalla ZH, Mahmood N and
Almawi WY: Circulating microRNA expression as predictor of
preeclampsia and its severity. Gene. 627:543–548. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ding J, Huang F, Wu G, Han T, Xu F, Weng
D, Wu C, Zhang X, Yao Y and Zhu X: MiR-519d-3p suppresses invasion
and migration of trophoblast cells via targeting MMP-2. PLoS One.
10:e01203212015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xueya Z, Yamei L, Sha C, Dan C, Hong S,
Xingyu Y and Weiwei C: Exosomal encapsulation of miR-125a-5p
inhibited trophoblast cell migration and proliferation by
regulating the expression of VEGFA in preeclampsia. Biochem Biophys
Res Commun. 525:646–653. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Brooks SA, Martin E, Smeester L, Grace MR,
Boggess K and Fry RC: miRNAs as common regulators of the
transforming growth factor (TGF)-β pathway in the preeclamptic
placenta and cadmium-treated trophoblasts: Links between the
environment, the epigenome and preeclampsia. Food Chem Toxicol.
98:50–57. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang X and Meng T: miR-215-5p decreases
migration and invasion of trophoblast cells through regulating CDC6
in preeclampsia. Cell Biochem Funct. 38:472–479. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Tamaru S, Mizuno Y, Tochigi H, Kajihara T,
Okazaki Y, Okagaki R, Kamei Y, Ishihara O and Itakura A:
MicroRNA-135b suppresses extravillous trophoblast-derived
HTR-8/SVneo cell invasion by directly down regulating CXCL12 under
low oxygen conditions. Biochem Biophys Res Commun. 461:421–426.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Pankiewicz K, Fijalkowska A, Issat T and
Maciejewski TM: Insight into the key points of preeclampsia
pathophysiology: Uterine artery remodeling and the role of
microRNAs. Int J Mol Sci. 22:31322021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Lv Y, Lu C, Ji X, Miao Z, Long W, Ding H
and Lv M: Roles of microRNAs in preeclampsia. J Cell Physiol.
234:1052–1061. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Chen J, Zhao L, Wang D, Xu Y, Gao H, Tan W
and Wang C: Contribution of regulatory T cells to immune tolerance
and association of microRNA210 and Foxp3 in preeclampsia. Mol Med
Rep. 19:1150–1158. 2019.PubMed/NCBI
|
|
22
|
Ahmadalizadeh Khanehsar M, Hoseinbeyki M,
Fakhr Taha M and Javeri A: Repression of TGF-β signaling in breast
cancer cells by miR-302/367 cluster. Cell J. 21:444–450.
2020.PubMed/NCBI
|
|
23
|
Yang CM, Chiba T, Brill B, Delis N, von
Manstein V, Vafaizadeh V, Oellerich T and Groner B: Expression of
the miR-302/367 cluster in glioblastoma cells suppresses
tumorigenic gene expression patterns and abolishes transformation
related phenotypes. Int J Cancer. 137:2296–2309. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jiang Y, Hou R, Li S, Li S and Dang G:
MicroRNA-302 inhibits cell migration and invasion in cervical
cancer by targeting DCUN1D1. Exp Ther Med. 16:1000–1008.
2018.PubMed/NCBI
|
|
25
|
Kim JY, Shin KK, Lee AL, Kim YS, Park HJ,
Park YK, Bae YC and Jung JS: MicroRNA-302 induces proliferation and
inhibits oxidant-induced cell death in human adipose tissue-derived
mesenchymal stem cells. Cell Death Dis. 5:e13852014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Guo Y, Cui J, Ji Z, Cheng C, Zhang K,
Zhang C, Chu M, Zhao Q, Yu Z, Zhang Y, et al: miR-302/367/LATS2/YAP
pathway is essential for prostate tumor-propagating cells and
promotes the development of castration resistance. Oncogene.
36:6336–6347. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Cheng L, Sharples RA, Scicluna BJ and Hill
AF: Exosomes provide a protective and enriched source of miRNA for
biomarker profiling compared to intracellular and cell-free blood.
J Extracell Vesicles. 32014.doi: 10.3402/jev.v3.23743. PubMed/NCBI
|
|
28
|
Melo SA, Sugimoto H, O'connell JT, Kato N,
Villanueva A, Vidal A, Qiu L, Vitkin E, Perelman LT, Melo CA, et
al: Cancer exosomes perform cell-independent microRNA biogenesis
and promote tumorigenesis. Cancer Cell. 26:707–721. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Thery C, Amigorena S, Raposo G and Clayton
A: Isolation and characterization of exosomes from cell culture
supernatants and biological fluids. Curr Protoc Cell Biol.
3:222006.PubMed/NCBI
|
|
31
|
Zhu Q, Li Q, Niu X, Zhang G, Ling X, Zhang
J, Wang Y and Deng Z: Extracellular vesicles secreted by human
urine-derived stem cells promote ischemia repair in a mouse model
of hind-limb ischemia. Cell Physiol Biochem. 47:1181–1192. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu F, Wu W, Wu K, Chen Y, Wu H, Wang H
and Zhang W: MiR-203 participates in human placental angiogenesis
by inhibiting VEGFA and VEGFR2 expression. Reprod Sci. 25:358–365.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rada CC, Murray G and England SK: The SK3
channel promotes placental vascularization by enhancing secretion
of angiogenic factors. Am J Physiol Endocrinol Metab.
307:E935–E943. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Moser G, Guettler J, Forstner D and
Gauster M: Maternal platelets-friend or foe of the human placenta?
Int J Mol Sci. 20:56392019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Pereira RD, De Long NE, Wang RC, Yazdi FT,
Holloway AC and Raha S: Angiogenesis in the placenta: The role of
reactive oxygen species signaling. Biomed Res Int. 2015:8145432015.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Reynolds LP and Redmer DA: Angiogenesis in
the placenta. Biol Reprod. 64:1033–1040. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bogic LV, Brace RA and Cheung CY:
Developmental expression of vascular endothelial growth factor
(VEGF) receptors and VEGF binding in ovine placenta and fetal
membranes. Placenta. 22:265–275. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Devi HL, Kumar S, Konyak YY, Bharati J,
Bhimte A, Pandey Y, Kumar K, Paul A, Kala A, Samad HA, et al:
Expression and functional role of fibroblast growth factors (FGF)
in placenta during different stages of pregnancy in water buffalo
(Bubalus bubalis). Theriogenology. 143:98–112. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Tian KW, Zhang YY, Jiang H and Han S:
Author correction: Intravenous C16 and angiopoietin-1 improve the
efficacy of placenta-derived mesenchymal stem cell therapy for EAE.
Sci Rep. 10:80972020. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ferrara N, Gerber HP and Lecouter J: The
biology of VEGF and its receptors. Nat Med. 9:669–676. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jussila L and Alitalo K: Vascular growth
factors and lymphangiogenesis. Physiol Rev. 82:673–700. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
George J, Shmilovich H, Deutsch V, Miller
H, Keren G and Roth A: Comparative analysis of methods for
assessment of circulating endothelial progenitor cells. Tissue Eng.
12:331–335. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Nagineni CN, William A, Cherukuri A,
Samuel W, Hooks JJ and Detrick B: Inflammatory cytokines regulate
secretion of VEGF and chemokines by human conjunctival fibroblasts:
Role in dysfunctional tear syndrome. Cytokine. 78:16–19. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Holmes DI and Zachary I: The vascular
endothelial growth factor (VEGF) family: Angiogenic factors in
health and disease. Genome Biol. 6:2092005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Ferrara N: Vascular endothelial growth
factor: Basic science and clinical progress. Endocr Rev.
25:581–611. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
An H J, Kim JH, Ahn EH, Kim YR, Kim JO,
Park HS, Ryu CS, Kim EG, Cho SH, Lee WS and Kim NK: 3′-UTR
polymorphisms in the vascular endothelial growth factor gene (VEGF)
contribute to susceptibility to recurrent pregnancy loss (RPL). Int
J Mol Sci. 20:33192019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Coultas L, Chawengsaksophak K and Rossant
J: Endothelial cells and VEGF in vascular development. Nature.
438:937–945. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Nevo O, Lee DK and Caniggia I: Attenuation
of VEGFR-2 expression by sFlt-1 and low oxygen in human placenta.
PLoS One. 8:e811762013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang K, Jiang YZ, Chen DB and Zheng J:
Hypoxia enhances FGF2- and VEGF-stimulated human placental artery
endothelial cell proliferation: Roles of MEK1/2/ERK1/2 and
PI3K/AKT1 pathways. Placenta. 30:1045–1051. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Song C, Xie S, Wang J, Lian J, Diao B and
Tang Y: Association of angiotensinogen gene polymorphisms and
angiogenic factors with preeclampsia in Chinese women. Gynecol
Obstet Invest. 76:64–68. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Zhang Z, Hong Y, Xiang D, Zhu P, Wu E, Li
W, Mosenson J and Wu WS: MicroRNA-302/367 cluster governs hESC
self-renewal by dually regulating cell cycle and apoptosis
pathways. Stem Cell Rep. 4:645–657. 2015. View Article : Google Scholar
|
|
52
|
Liu FY, Wang LP, Wang Q, Han P, Zhuang WP,
Li MJ and Yuan H: miR-302b regulates cell cycles by targeting CDK2
via ERK signaling pathway in gastric cancer. Cancer Med.
5:2302–2313. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Thery C, Zitvogel L and Amigorena S:
Exosomes: Composition, biogenesis and function. Nat Rev Immunol.
2:569–579. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
54
|
Monaco F, Gaetani S, Alessandrini F,
Tagliabracci A, Bracci M, Valentino M, Neuzil J, Amati M, Bovenzi
M, Tomasetti M and Santarelli L: Exosomal transfer of miR-126
promotes the anti-tumour response in malignant mesothelioma: Role
of miR-126 in cancer-stroma communication. Cancer Lett. 463:27–36.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Liu X, Lu Y, Xu Y, Hou S, Huang J, Wang B,
Zhao J, Xia S, Fan S, Yu X, et al: Exosomal transfer of miR-501
confers doxorubicin resistance and tumorigenesis via targeting of
BLID in gastric cancer. Cancer Lett. 459:122–134. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Li H, Ouyang Y, Sadovsky E, Parks WT, Chu
T and Sadovsky Y: Unique microRNA signals in plasma exosomes from
pregnancies complicated by preeclampsia. Hypertension. 75:762–771.
2020. View Article : Google Scholar : PubMed/NCBI
|