Introduction
Advanced oral squamous cell carcinoma (OSCC) is
characterized by invasion of surrounding tissues, metastasis to
lymph nodes, resistance to chemotherapy, and poor prognosis
(1,2). Most OSCC cases develop from oral
epithelial dysplasia (OED) and carcinoma in situ (CIS)
(2,3). Clinically, the mass or ulcer is
obtained from the tongue, gingiva, oral floor and buccal mucosa of
the patients (1–4). Histologically, sampled tissue is
diagnosed as atypical squamous epithelium (ASE), OED, CIS, or SCC,
depending on the grade of nuclear atypia and stromal invasion
(2,3,5).
However, it is often difficult to determine ASE, OED, or CIS based
on hematoxylin and eosin (H&E) findings alone (4–6).
Therefore, immunohistochemistry may be helpful in such cases
(4–6).
Casein kinase 1ε (CK-1ε) is an enzyme of the casein
kinase family and regulates cell proliferation, migration, and
circadian rhythm (7–9). The level of CK-1ε in ovarian cancer
cells is higher than that in non-cancerous cells (10). Differentiated embryonic
chondrocyte gene 1 (DEC1) is a basic helix-loop-helix transcription
factor, which regulates inflammation, apoptosis, migration
epithelial mesenchymal transition (EMT), cell differentiation, and
circadian rhythm (11–16). It has been revealed that DEC1 is
highly expressed in tumor cells compared with non-tumor cells of
the cervical, pancreatic and oral lineages (12,13,15). Proliferating cell nuclear antigen
(PCNA) is a nuclear marker of G1/S phase cell cycle regulation
(17). It was recently
demonstrated that differences in the localization of positive PCNA
staining may distinguish between conventional squamous cell
carcinoma and basaloid squamous cell carcinoma (4). CD44 is a known cancer stem cell
marker that regulates cell proliferation and migration and is
highly expressed in dysplasia and OSCC compared with non-tumor
cells (18–20). However, this has not been revealed
in ASE and CIS. The present study aimed to immunohistochemically
examine the expression levels of CK-1ε, DEC1, PCNA and CD44 in
biopsy samples of ASE and CIS.
Materials and methods
Tissue preparation
Histological biopsy specimens between December 2012
and December 2018 were retrieved from the archives of the
Department of Diagnostic Pathology of Wakayama Medical University
(Wakayama, Japan) according to the guidelines of the Japanese
Society of Pathology. The present study was approved (approval no.
1715) by the Research Ethics Committee of Wakayama Medical
University and histological specimens were retrieved from Wakayama
Medical University hospital archives. Oral informed consents were
provided by all patients for the use of their tissues.
A total of 20 cases of ASE and 12 cases of CIS were
selected. Diagnoses were performed by at least two pathologists. In
the present study, ASE tissues with or without inflammatory and
benign lesions were used. CIS with obvious nuclear atypia was used
but invasive OSCC was not included. Clinical and histological
information are presented in Table
I. The percentage of intensity score was calculated using
Microsoft excel.
 | Table I.Immunohistochemical detection of
CK-1ε, DEC1, PCNA and CD44 in ASE and CIS. |
Table I.
Immunohistochemical detection of
CK-1ε, DEC1, PCNA and CD44 in ASE and CIS.
| Case | Age/Sex | Lesions | Diagnosis | CK-1ε | DEC1 | PCNA | CD44 |
|---|
| 1 | 60/F | Tongue | ASE | 1 | 3 | 2 | 1 |
| 2 | 55/M | Tongue | ASE | 1 | 3 | 2 | 2 |
| 3 | 75/F | Tongue | ASE | 1 | 2 | 1 | 1 |
| 4 | 62/M | Tongue | ASE | 1 | 3 | 1 | 1 |
| 5 | 70/M | Tongue | ASE | 1 | 2 | 2 | 1 |
| 6 | 67/F | Tongue | ASE | 1 | 3 | 3 | 3 |
| 7 | 76/F | Tongue | ASE | 1 | 3 | 3 | 1 |
| 8 | 28/F |
Gingiva | ASE | 1 | 2 | 1 | 1 |
| 9 | 69/F |
Gingiva | ASE | 1 | 2 | 1 | 1 |
| 10 | 65/F |
Gingiva | ASE | 1 | 3 | 1 | 1 |
| 11 | 58/F |
Gingiva | ASE | 1 | 3 | 2 | 1 |
| 12 | 59/F |
Gingiva | ASE | 1 | 3 | 2 | 1 |
| 13 | 77/F |
Gingiva | ASE | 1 | 3 | 2 | 1 |
| 14 | 84/F |
Gingiva | ASE | 2 | 2 | 2 | 3 |
| 15 | 74/M | Buccal | ASE | 1 | 3 | 2 | 1 |
| 16 | 68/F | Buccal | ASE | 1 | 2 | 2 | 2 |
| 17 | 74/M | Buccal | ASE | 1 | 3 | 3 | 1 |
| 18 | 76/F | Buccal | ASE | 1 | 3 | 2 | 2 |
| 19 | 77/F |
Oral floor | ASE | 1 | 3 | 2 | 1 |
| 20 | 54/F |
Palate | ASE | 2 | 3 | 2 | 1 |
| 21 | 67/M | Tongue | CIS | 3 | 1 | 3 | 2 |
| 22 | 78/M | Tongue | CIS | 3 | 3 | 2 | 2 |
| 23 | 68/M | Tongue | CIS | 2 | 1 | 3 | 2 |
| 24 | 60/M | Tongue | CIS | 3 | 1 | 3 | 3 |
| 25 | 55/F | Tongue | CIS | 3 | 2 | 3 | 2 |
| 26 | 74/M |
Gingiva | CIS | 3 | 1 | 2 | 1 |
| 27 | 69/F |
Gingiva | CIS | 2 | 1 | 3 | 2 |
| 28 | 71/M |
Gingiva | CIS | 2 | 1 | 3 | 3 |
| 29 | 80/F | Buccal | CIS | 2 | 1 | 2 | 3 |
| 30 | 69/F | Buccal | CIS | 3 | 3 | 2 | 3 |
| 31 | 84/F |
Palate | CIS | 3 | 1 | 2 | 3 |
| 32 | 78/F |
Palate | CIS | 2 | 1 | 2 | 1 |
Immunohistochemistry
CK-1ε, DEC1, PCNA and CD44 expression levels in ASE
and CIS tissues were profiled using a Discovery Auto-Stainer with
automated protocols (software Nex-ES v10.6) (Ventana Medical
Systems, Inc.; Roche Diagnostics) as previously described (4).
Cell culture and treatment
CA9-22 cells (cat. no. JCRB0625) were obtained from
the Japanese Cancer Research Resources Bank and used until passage
five. CA9-22 cells stably overexpressing DEC1 (CA9-22DEC1) and
control empty vector-transfected cells (CA9-22vector) were
evaluated for stable cells as previously described (21). These cells were cultured in
Dulbecco's modified Eagle's medium (Sigma-Aldrich; Merck KGaA) as
previously described (4,21). Transient plasmid transfection of a
FLAG-tagged CK-1ε was performed using FuGENE HD (Promega
Corporation) as previously described (22).
Western blotting
CA9-22 cells were lysed using M-PER lysis buffer
(Thermo Fisher Scientific, Inc.) and protein concentration was
determined by bicinchoninic acid (BCA) assay. 40 µg of protein was
loaded on 12.5% SDS-polyacrylamide gels. The separated proteins
were subsequently transferred onto PVDF membranes, which were
incubated with primary antibodies overnight at 4°C. The incubation
with secondary antibodies was performed for 1 h at room
temperature. Western blotting was performed as previously described
(4). AE-9300 Ez capture MG (ATTO)
was used to capture images. Three independent biological replicates
were performed.
Antibodies
The following commercial antibodies were used: CK-1ε
(1:200, mouse monoclonal; cat. no. sc-373912; Santa Cruz
Biotechnology, Inc.), DEC1 (1:400; rabbit polyclonal; cat. no.
NB100-1800; Novus Biologicals, LLC), PCNA (1:1,000; mouse
monoclonal; cat. no. sc-56), HCAM (CD44; 1:200; mouse monoclonal;
sc-7297; both from Santa Cruz Biotechnology, Inc.), FLAG (1:1,000;
mouse monoclonal; cat. no. F3165) and actin (1:10,000; mouse
monoclonal, A5441; both from Sigma-Aldrich; Merck KGaA).
Anti-rabbit and mouse HRP IgG secondary antibodies (1:5,000; cat.
nos. 17502 and 17601, respectively) were purchased from
Immuno-Biological Laboratories, Co., Ltd.
Results
Histological features of ASE and
CIS
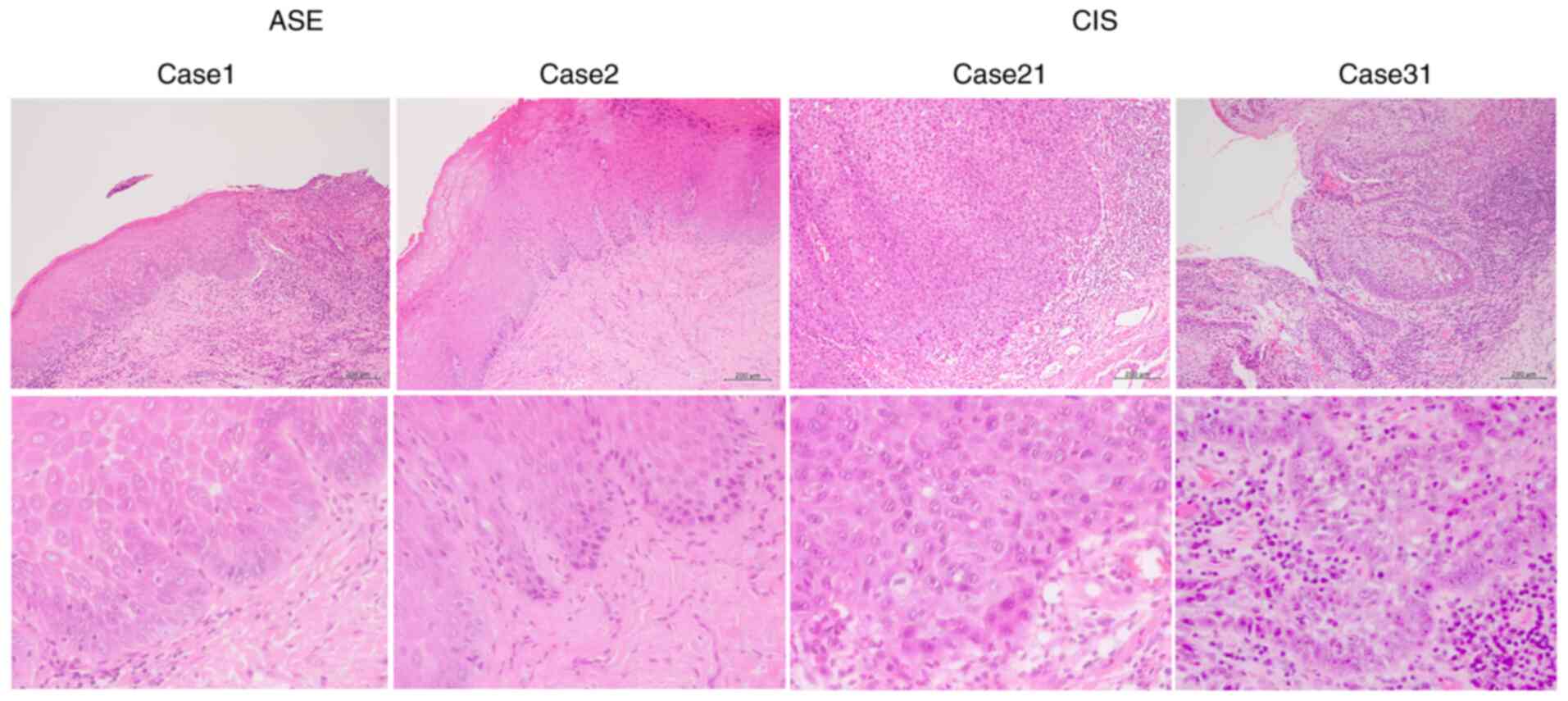
Representative histological H&E-stained images
for cases 1 and 2 of ASE, and 21 and 31 of CIS, respectively, are
revealed in Fig. 1. Mild nuclear
atypia was observed in the ASE samples, whereas severe nuclear
atypia was observed in the CIS samples.
Immunohistochemical detection of
CK-1ε, DEC1, PCNA, and CD44 in ASE and CIS
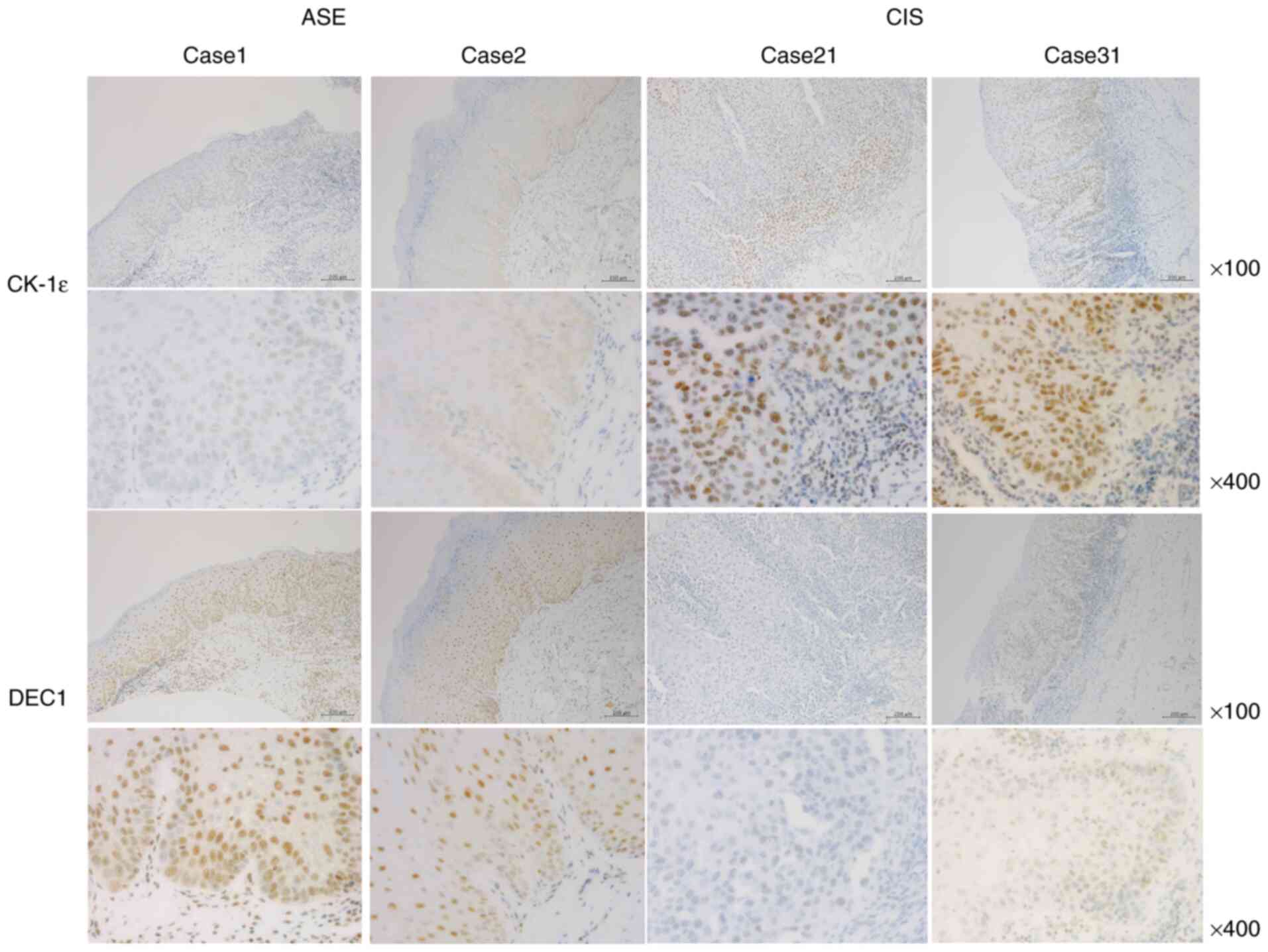
Representative images of CK-1ε and DEC1
immunoreactivities for cases 1 and 2 of ASE and 21 and 31 of CIS
are shown in Fig. 2. A total of
three grades (1, weak; 2, moderate; and 3, strong) of intensity
were defined by immunohistochemistry. CK-1ε was weakly expressed in
the nuclei of ASE samples (100% with intensity levels 1–2), whereas
it was strongly expressed in the nuclei of CIS samples (100% with
intensity levels 2–3) (Tables I
and II). By contrast, DEC1 was
strongly expressed in ASE and weakly expressed in CIS. All ASE
samples stained with DEC1 had an intensity of 2 or 3, while 88% of
CIS samples stained with DEC1 had intensities of 1 or 2.
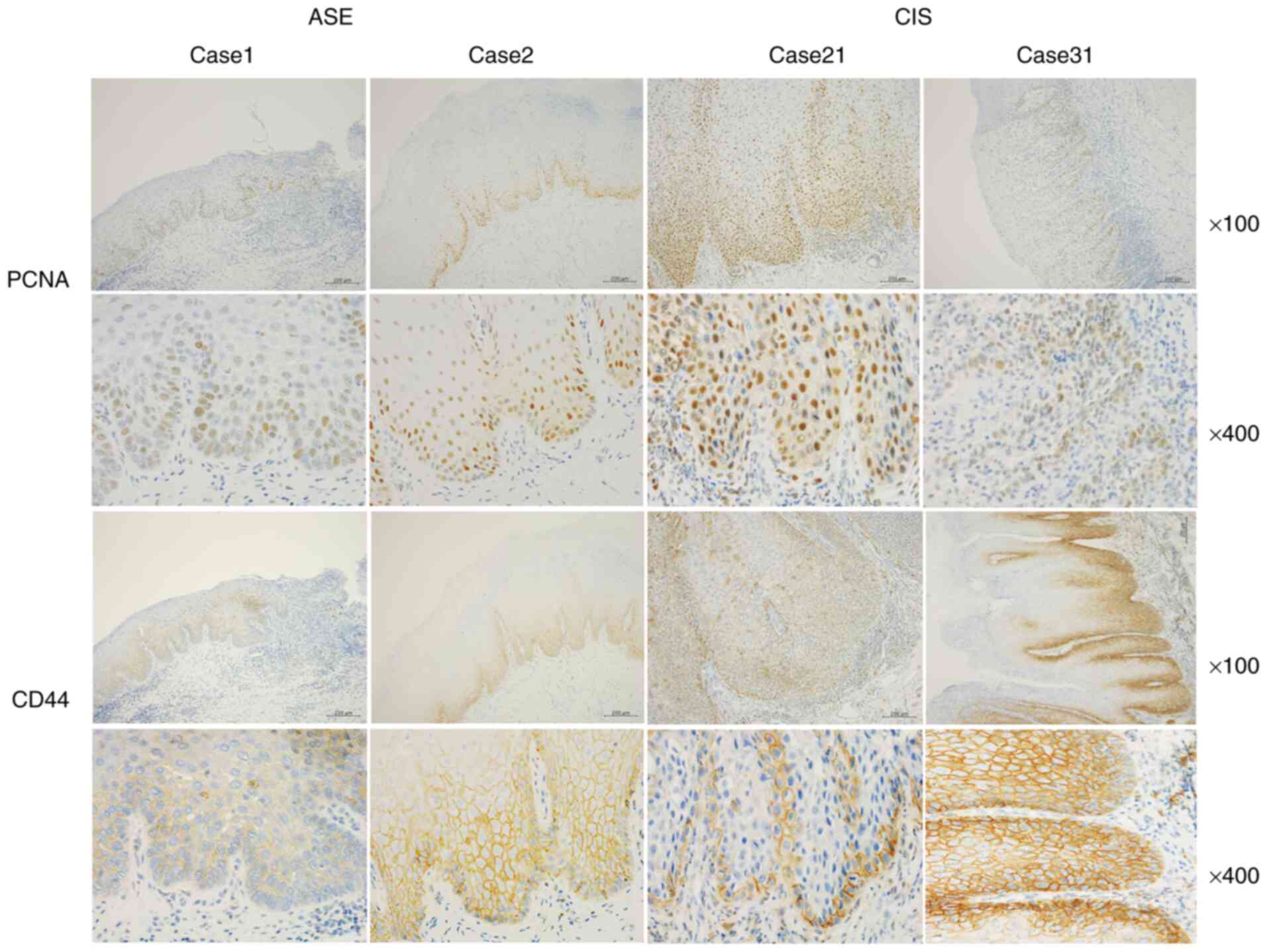
Representative images of PCNA and CD44 immunoreactivities for cases
1 and 2 of ASE and 21 and 31 of CIS are presented in Fig. 3. PCNA was weakly and moderately
expressed in the nucleus of ASE, and moderately and strongly
expressed in the nucleus of CIS. Overall, 85% of the ASE samples
stained with PCNA had intensities of 1 or 2, while 60% had an
intensity of 2. Collectively, 100% of the CIS samples stained with
PCNA had intensities of 2 and 3. Furthermore, 90% of ASE samples
stained with CD44 had intensities of 1 and 2, while 84% of the CIS
samples had intensities of 2 and 3.
 | Table II.Percentage of intensity score of
CK-1ε, DEC1, PCNA and CD44 in ASE and CIS. |
Table II.
Percentage of intensity score of
CK-1ε, DEC1, PCNA and CD44 in ASE and CIS.
| Intensity | CK-1ε (%) | DEC1 (%) | PCNA (%) | CD44 (%) |
|---|
| ASE |
|
|
|
|
| 1 | 90 | 0 | 25 | 75 |
| 2 | 10 | 30 | 60 | 15 |
| 3 | 0 | 70 | 15 | 10 |
| CIS |
|
|
|
|
| 1 | 0 | 75 | 0 | 16 |
| 2 | 42 | 8 | 50 | 42 |
| 3 | 58 | 17 | 50 | 42 |
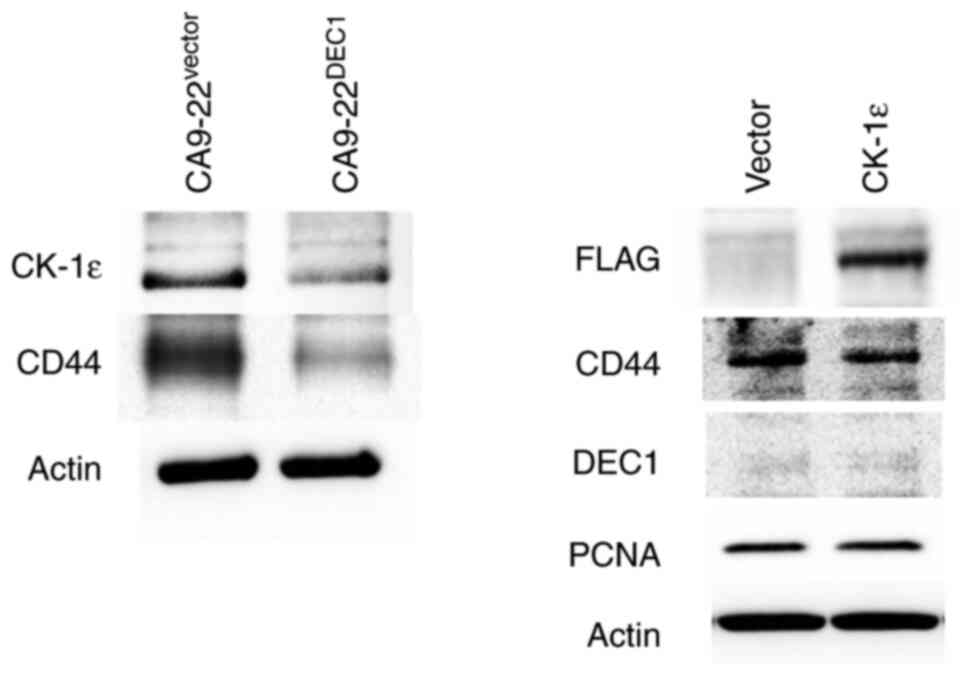
DEC1 overexpression decreases the
expression of CK-1ε and CD44 in oral cancer cells
It was further examined whether DEC1 overexpression
in CA9-22 cells (CA9-22DEC1) affected the levels of CK-1ε and CD44.
CA9-22-DEC1-overexpressing cells exhibited decreased expression of
CK-1ε and CD44 (Fig. 4). In
addition, it was previously reported that DEC1 overexpression
decreases the expression of PCNA (21). The transient overexpression of
CK-1ε was effective, but it had little effect on the expression of
CD44, DEC1 and PCNA. The present study suggested
immunohistochemical strategies for distinguishing between ASE and
CIS in Fig. 5.
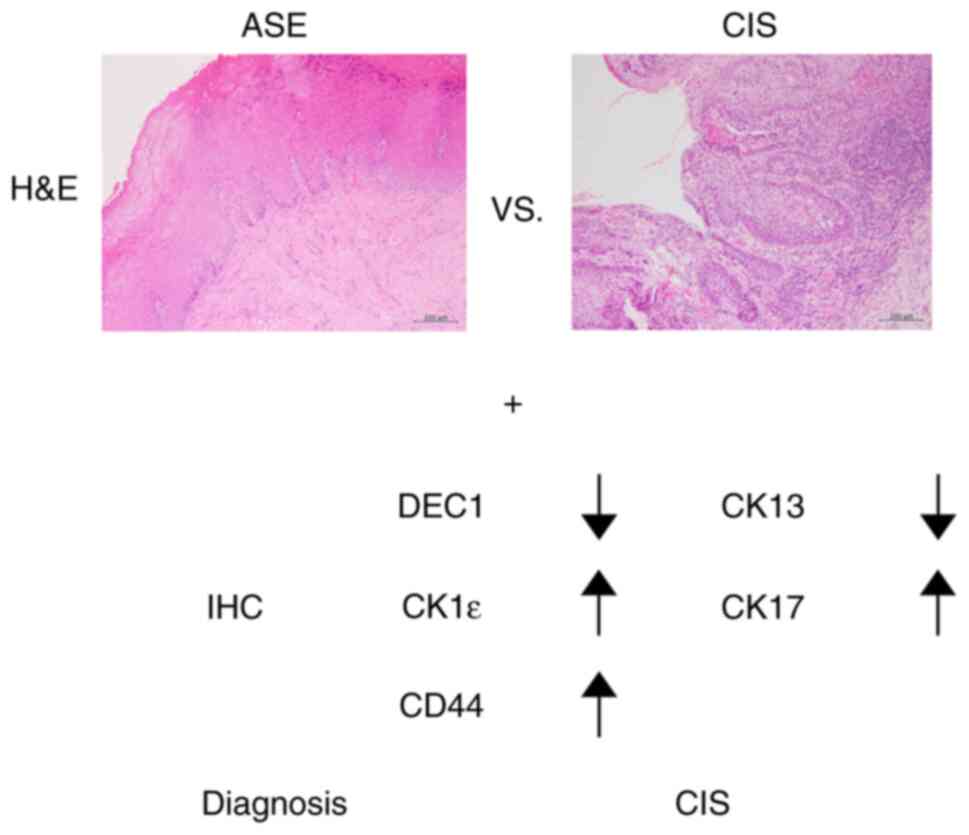 | Figure 5.Pathological differentiation of ASE
and CIS. Immunohistochemical findings of CK13, CK17, DEC1, CK-1ε
and CD44 may help distinguish between ASE and CIS. A diagnosis of
CIS using IHC is indicated by decreased expression of CK13 and DEC1
and increased expression of CK17, CK-1ε and CD44. ASE, atypical
squamous epithelium; CIS, carcinoma in situ; DEC1,
differentiated embryonic chondrocyte gene 1; CK-1ε, casein kinase
1ε; IHC, immunohistochemistry; H&E, hematoxylin and eosin
staining. |
Discussion
In the present study, the expression of CK-1ε, DEC1,
PCNA, and CD44 was examined in ASE and CIS biopsy samples. It was
previously reported that DEC1 expression was increased in invasive
cancer cells compared with that in non-cancerous cells (13). Notably, DEC1 expression was lower
in CIS than in ASE samples. In addition, DEC1 was reported to be
more highly expressed in spindle lesions of EMT than in
conventional invasive lesions (12,15). Although DEC1 expression was lower
in CIS compared with ASE samples, invasive processes such as EMT
from CIS may induce DEC1 expression. It is well known that
transforming growth factor beta (TGFβ) induces EMT and DEC1
expression in various cancer cells (15,23–27). Consistent with this finding, DEC1
expression was higher in ASE and invasive OSCC samples than that in
CIS samples (12,13,15).
It has been reported that DEC1 promotes tumor
progression, EMT, metastasis, and anti-apoptotic activity via
hypoxia-inducible factor-1α, TGFβ and SMAD3 (14,21). CD44 promotes tumor proliferation,
invasion and anti-apoptotic functions (28). DEC1 negatively regulates the
expression of stem cell markers SOX2 and c-MYC in cervical cancer
cells (12). Consistent with
this, it was observed that the cancer stem cell marker CD44 was
decreased in CA9-22-DEC1-overexpressing cells. In addition,
opposite expression patterns of DEC1 and CD44 were observed in ASE
and CIS samples. These results suggested that DEC1 negatively
regulated CD44 expression, although both the expression levels of
DEC1 and CD44 were higher in invasive cancer cells. DEC1 regulates
target genes by binding E-boxes and sp1 sites (14,16,22). Notably, overexpression of
circadian clock gene Period2 (PER2) decreased CD44
expression in lung cancer cells (29). Since DEC1 negatively regulates
PER2 through E-boxes (14,16),
it is possible that DEC1 regulates CD44 via E-boxes. It was
hypothesized that DEC1 suppresses CD44 expression to slow the
invasive progression of OSCC. Future studies are required to
clarify how DEC1 regulates CD44. It was revealed that CK-1ε
expression is similar to that of CD44. Low expression levels of
CK-1ε have been associated with poor prognosis in advanced OSCC
(30). These previous results
suggested that CK-1ε expression is inversely correlated with DEC1
in CIS and in invasive OSCC. Therefore, DEC1 negatively regulates
CK-1ε. In the present study, no marked changes in PCNA were
identified in ASE and CIS. However, it was observed that DEC1
overexpression decreased the PCNA expression in CA9-22 cells, as
previously described (21). It
was hypothesized that DEC1 negatively regulates PCNA in invasive
OSCC but not in CIS. Clarification on how these molecules
functionally regulate ASE and CIS, using a mouse model, is
warranted in future studies.
CK13 and CK17 have been previously used to
distinguish between ASE and CIS (1,6).
In CIS, the expression of CK13 is decreased whereas that of CK17 is
increased. Τhe use of additional immunohistochemical markers such
as CK-1ε, DEC1, CD44, CK13 and CK17 is suggested to distinguish ASE
and CIS. However, changes in these immunohistochemical markers are
often observed upon severe inflammation, such as in active ulcers
and in ASE. Furthermore, the usefulness of biopsies to distinguish
between ASE and CIS is limited since ASE occasionally exhibits
severe nuclear atypia. Therefore, the difference between ASE and
CIS should be clarified using resected samples. Another limitation
was the small sample size and the absence of statistical analysis
to quantify results in the present study. As a result, a larger
number of samples needs to be examined in future studies.
Acknowledgements
Not applicable.
Funding
The present study was supported by JSPS KAKENHI (grant no.
16K09624).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
FS performed experiments, pathological diagnosis and
wrote the draft of the manuscript. SO and NS performed
immunohistochemistry and western blotting. FS, SO and NS confirmed
the authenticity of all the raw data. YM checked pathological
diagnosis. UKB and KO helped acquisition of data and interpretation
of data. UKB, KO and YM revised the manuscript. All authors read
and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved (approval no. 1715)
by the Research Ethics Committee of Wakayama Medical University
(Wakayama, Japan) and histological specimens were retrieved from
Wakayama Medical University hospital archives. Oral informed
consents were provided by all patients for the use of their
tissues.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ASE
|
atypical squamous epithelium
|
|
CIS
|
carcinoma in situ
|
|
EMT
|
epithelial to mesenchymal
transition
|
|
H&E
|
hematoxylin and eosin
|
|
OED
|
oral epithelial dysplasia
|
|
OSCC
|
oral squamous cell carcinoma
|
|
DEC1
|
differentiated embryonic chondrocyte
gene 1
|
|
CK-1ε
|
casein kinase 1ε
|
|
PCNA
|
proliferating cell nuclear antigen
|
References
|
1
|
Ikeda M, Shima K, Kondo T and Semba I:
Atypical immunohistochemical patterns can complement the
histopathological diagnosis of oral premalignant lesions. J Oral
Biosci. 62:93–98. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Speight PM and Farthing PM: The pathology
of oral cancer. Br Dent J. 225:841–847. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Brouns E, Baart J, Karagozoglu Kh, Aartman
I, Bloemena E and van der Waal I: Malignant transformation of oral
leukoplakia in a well-defined cohort of 144 patients. Oral Dis.
20:e19–e24. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sato F, Bhawal UK, Tojyo I, Fujita S,
Murata SI and Muragaki Y: Differential expression of claudin-4,
occludin, SOX2 and proliferating cell nuclear antigen between
basaloid squamous cell carcinoma and squamous cell carcinoma. Mol
Med Rep. 20:1977–1985. 2019.PubMed/NCBI
|
|
5
|
Odell E, Kujan O, Warnakulasuriya S and
Sloan P: Oral epithelial dysplasia: Recognition, grading and
clinical significance. Oral Dis. 27:947–976. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nobusawa A, Sano T, Negishi A, Yokoo S and
Oyama T: Immunohistochemical staining patterns of cytokeratins 13,
14, and 17 in oral epithelial dysplasia including orthokeratotic
dysplasia. Pathol Int. 64:20–27. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fish KJ, Cegielska A, Getman ME, Landes GM
and Virshup DM: Isolation and characterization of human casein
kinase I epsilon (CKI), a novel member of the CKI gene family. J
Biol Chem. 270:14875–14883. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Lee C, Etchegaray JP, Cagampang FR, Loudon
AS and Reppert SM: Posttranslational mechanisms regulate the
mammalian circadian clock. Cell. 107:855–867. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang Z, Zhou L, Wang Y, Peng Q, Li H,
Zhang X, Su Z, Song J, Sun Q, Sayed S, et al: The CK1δ/ε-AES axis
regulates tumorigenesis and metastasis in colorectal cancer.
Theranostics. 11:4421–4435. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Rodriguez N, Yang J, Hasselblatt K, Liu S,
Zhou Y, Rauh-Hain JA, Ng SK, Choi PW, Fong WP, Agar NY, et al:
Casein kinase I epsilon interacts with mitochondrial proteins for
the growth and survival of human ovarian cancer cells. EMBO Mol
Med. 4:952–963. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang X, Sato F, Tanimoto K, Rajeshwaran N,
Thangavelu L, Makishima M and Bhawal UK: The potential roles of
Dec1 and Dec2 in Periodontal inflammation. Int J Mol Sci.
22:103492021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Sato F, Bhawal UK, Sugiyama N, Osaki S,
Oikawa K and Muragaki Y: Potential role of DEC1 in cervical cancer
cells involving overexpression and apoptosis. Clocks Sleep.
2:26–38. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Bhawal UK, Sato F, Arakawa Y, Fujimoto K,
Kawamoto T, Tanimoto K, Ito Y, Sasahira T, Sakurai T, Kobayashi M,
et al: Basic helix-loop-helix transcription factor DEC1 negatively
regulates cyclin D1. J Pathol. 224:420–429. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Sato F, Bhawal UK, Yoshimura T and
Muragaki Y: DEC1 and DEC2 crosstalk between circadian rhythm and
tumor progression. J Cancer. 7:153–159. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Wu Y, Sato F, Yamada T, Bhawal UK,
Kawamoto T, Fujimoto K, Noshiro M, Seino H, Morohashi S, Hakamada
K, et al: The BHLH transcription factor DEC1 plays an important
role in the epithelial-mesenchymal transition of pancreatic cancer.
Int J Oncol. 41:1337–1346. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Honma S, Kawamoto T, Takagi Y, Fujimoto K,
Sato F, Noshiro M, Kato Y and Honma K: Dec1 and Dec2 are regulators
of the mammalian molecular clock. Nature. 419:841–844. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tsai ST and Jin YT: Proliferating cell
nuclear antigen (PCNA) expression in oral squamous cell carcinomas.
J Oral Pathol Med. 24:313–315. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Yu SS and Cirillo N: The molecular markers
of cancer stem cells in head and neck tumors. J Cell Physiol.
235:65–73. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bourguignon LYW, Earle C and Shiina M:
Activation of matrix hyaluronan-mediated CD44 signaling, epigenetic
regulation and chemoresistance in head and neck cancer stem cells.
Int J Mol Sci. 18:18492017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ghazi N, Saghravanian N, Taghi Shakeri M
and Jamali M: Evaluation of CD44 and TGF-B expression in oral
carcinogenesis. J Dent (Shiraz). 22:33–40. 2021.PubMed/NCBI
|
|
21
|
Sato F, Otsuka T, Kohsaka A, Le HT, Bhawal
UK and Muragaki Y: Smad3 suppresses epithelial cell migration and
proliferation via the clock gene Dec1, which negatively regulates
the expression of clock genes Dec2 and Per1. Am J Pathol.
189:773–783. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sato F, Muragaki Y and Zhang Y: DEC1
negatively regulates AMPK activity via LKB1. Biochem Biophys Res
Commun. 467:711–716. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Ooshima A, Park J and Kim SJ:
Phosphorylation status at Smad3 linker region modulates
transforming growth factor-β-induced epithelial-mesenchymal
transition and cancer progression. Cancer Sci. 110:481–488. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Roberts AB, Tian F, Byfield SD, Stuelten
C, Ooshima A, Saika S and Flanders KC: Smad3 is key to
TGF-beta-mediated epithelial-to-mesenchymal transition, fibrosis,
tumor suppression and metastasis. Cytokine Growth Factor Rev.
17:19–27. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kon N, Hirota T, Kawamoto T, Kato Y,
Tsubota T and Fukada Y: Activation of TGF-beta/activin signalling
resets the circadian clock through rapid induction of Dec1
transcripts. Nat Cell Biol. 10:1463–1469. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ehata S, Hanyu A, Hayashi M, Aburatani H,
Kato Y, Fujime M, Saitoh M, Miyazawa K, Imamura T and Miyazono K:
Transforming growth factor-beta promotes survival of mammary
carcinoma cells through induction of antiapoptotic transcription
factor DEC1. Cancer Res. 67:9694–9703. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zawel L, Yu J, Torrance CJ, Markowitz S,
Kinzler KW, Vogelstein B and Zhou S: DEC1 is a downstream target of
TGF-beta with sequence-specific transcriptional repressor
activities. Proc Natl Acad Sci USA. 99:2848–2853. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Guo Q, Yang C and Gao F: The state of CD44
activation in cancer progression and therapeutic targeting. FEBS J.
Sep 3–2021.(Epub ahead of print). View Article : Google Scholar
|
|
29
|
Xiang R, Cui Y, Wang Y, Xie T, Yang X,
Wang Z, Li J and Li Q: Circadian clock gene Per2 downregulation in
non-small cell lung cancer is associated with tumour progression
and metastasis. Oncol Rep. 40:3040–3048. 2018.PubMed/NCBI
|
|
30
|
Lin SH, Lin YM, Yeh CM, Chen CJ, Chen MW,
Hung HF, Yeh KT and Yang SF: Casein kinase 1 epsilon expression
predicts poorer prognosis in low T-stage oral cancer patients. Int
J Mol Sci. 15:2876–2891. 2014. View Article : Google Scholar : PubMed/NCBI
|