Introduction
Pancreatic carcinoma (PC) is one of the most
aggressive solid malignant tumors, seriously threatening human
health and life. The incidence of PC has risen in recent years, and
60,430 new PC cases and 48,220 PC-related deaths were estimated in
2021 worldwide (1). Despite
considerable improvements in surgical techniques, chemotherapeutic
regimens and neoadjuvant chemoradiotherapies in recent years, the
5-year survival rate of patients with PC remains <5% on account
of early local invasion and distant metastasis (2). Thus, to improve clinical diagnosis and
treatment, identification of the molecular mechanisms associated PC
tumorigenesis is of great importance.
Zinc-finger protein 217 (ZNF217), a member of the
Krüppel-like family of transcriptional factors, is an oncogene that
coordinates complex cellular progresses, controls the early and
final stages of tumor development, and is considered to be a
powerful biomarker of tumorigenesis (3). ZNF217 expression is frequently
amplified in malignant tumors, such as breast cancer,
hepatocellular carcinoma and colorectal cancer; high expression
levels of ZNF217 are associated with a poor clinical prognosis and
the development of metastases (4–6).
However, whether ZNF217 is involved in the progression of PC
remains unclear.
Transcription factor interferon regulatory factor 5
(IRF5), a member of the IRF family, has been widely recognized as a
key regulator of inflammation, providing novel avenues for the
development of therapeutic agents for inflammatory diseases
(7). Accumulating evidence has
demonstrated that IRF5 is involved in the regulation of genes
induced by oncogenesis. For example, IRF5 was reported to be highly
expressed in human thyroid cancer cells and to promote cancer cell
proliferation, accelerating the development of thyroid cancer
(8). Conversely, IRF5 also acts as
a tumor suppressor in both gastric and renal cancer, where its
expression is downregulated (9,10).
Thus, the true role of IRF5 appears to depend on the cancer type in
question. Until now, the role of IRF5 in PC has not been
investigated.
According to predictions from the AnimalTFDB3.0
website (http://bioinfo.life.hust.edu.cn/AnimalTFDB/), an
association was observed between IRF5 and ZNF217, suggesting that
IRF5 may be a transcription factor of ZNF217. However, until now,
there is no evidence demonstrating the link between IRF5 and ZNF217
expression in PC. Therefore, the aim of the present study was to
determine the role of ZNF217 and its connection with IRF5 in PC,
and to investigate its effects on cellular proliferation, migration
and invasiveness, as well as its potential molecular mechanism.
Materials and methods
Bioinformatics analysis
Gene Expression Profiling Interactive Analysis
(GEPIA; http://gepia.cancer-pku.cn) is an
interactive web server for the analysis of RNA sequencing
expression data from The Cancer Genome Atlas and the Genotype
Tissue Expression projects (11).
The expression of ZNF217 and IRF5 was analyzed using the GEPIA
database.
Cell culture
The HPDE6c7 normal human pancreatic ductal
epithelial cell line and human PC cell lines (BxPC-3, SW1990,
PANC-1 and CFPAC-1) were obtained from the Shanghai Advanced
Research Institute, Chinese Academy of Sciences. Cells were
cultured in DMEM (Thermo Fisher Scientific, Inc.) supplemented with
10% FBS (Thermo Fisher Scientific, Inc.) and 1%
penicillin/streptomycin, and maintained at 37°C (5% CO2)
in a humidified atmosphere.
Transfection
cDNA encoding ZNF217 or IRF5 was amplified and
inserted into the pcDNA3.1 vector (Shanghai GenePharma Co., Ltd) to
overexpress ZNF217 (Oe-ZNF217) or IRF5 (Oe-IRF5), respectively. An
empty vector was used as the negative control (Oe-NC) for Oe-ZNF217
and Oe-IRF5. Short hairpin (sh)RNAs (pGPU6) targeting ZNF217
(shRNA-ZNF217#1 and shRNA-ZNF217#2), shRNA-IRF5#1, shRNA-IRF5#2
were obtained from Shanghai GenePharma Co., Ltd., and an empty
pGPU6 vector was considered as the negative control (shRNA-NC). The
shRNA sequences were as follows: shRNA-ZNF217#1 forward,
5′-CCGGCAAGGTATACTCTTCAAATAACTCGAGTTATTTGAAGAGTATACCTTGTTTTTG-3′
and reverse,
5′-AATTCAAAAACAAGGTATACTCTTCAAATAACTCGAGTTATTTGAAGAGTATACCTTG-3′;
shRNA-ZNF217#2 forward,
5′-CCGGCATGGTGATGAGGGCATTTAACTCGAGTTAAATGCCCTCATCACCATGTTTTTG-3′
and reverse,
5′-AATTCAAAAACATGGTGATGAGGGCATTTAACTCGAGTTAAATGCCCTCATCACCATG-3′;
shRNA-IRF5#1 forward,
5′-CCGGGGGTGCACACCCATGTTATAACTCGAGTTATAACATGGGTGTGCACCCTTTTTG-3′
and reverse,
5′-AATTCAAAAAGGGTGCACACCCATGTTATAACTCGAGTTATAACATGGGTGTGCACCC-3′;
and shRNA-IRF5#2 forward,
5′-CCGGATGCTAGATATCTGCATATTTCTCGAGAAATATGCAGATATCTAGCATTTTTTG-3′
and reverse,
5′-AATTCAAAAAATGCTAGATATCTGCATATTTCTCGAGAAATATGCAGATATCTAGCAT-3′.
Cells were seeded into 6-well plates (2×105 cells/well)
24 h before transfection, and then transfected with a final
concentration of 50 nM shRNA and/or 15 nM overexpression vectors
using Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's instructions at
37°C for 48 h. Subsequently, cells were harvested for subsequent
experiments.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from cells using
TRIzol® reagent (Invitrogen; Thermo Fisher Scientific,
Inc.) according to the manufacturer's instructions. A UV
spectrophotometer was used to determine the RNA A280/A260 value.
Total RNA was reverse transcribed into cDNA using the RT Reagent
Kit (Takara Biotechnology Co., Ltd.) in accordance with the
manufacturer's protocols. qPCR was performed using the Real-time
PCR Sequence Detection system (Bio-Rad Laboratories, Inc.) with the
SYBR Green PCR Master mix (Thermo Fisher Scientific, Inc.) per the
manufacturer's protocols. The thermocycling conditions were as
follows: 50°C for 2 min and 95°C for 10 min, followed by 40 cycles
of 95°C for 10 sec and 60°C for 30 sec. The primer sequences were
as follows: ZNF217 forward, 5′-GAGAAGCGAATGGTGAAAGC-3′ and reverse,
5′-CAGCGCTCAAGTATGCAAAA-3′; IRF5 forward,
5′-CATTACTGTACAGGTGGTGC-3′ and reverse, 5′-AGATGTGATGGAGCTCCTTG-3′;
and β-actin forward, 5′-CTCCATCCTGGCCTCGCTGT-3′ and reverse,
5′-GCTGTCACCTTCACCGTTCC-3′. β-actin was used as the internal
reference, and the relative expression level of each gene was
calculated using the 2−ΔΔCq method (12).
Cell Counting Kit-8 (CCK-8) assay
Cell viability was assessed using the CCK-8 assay
(Beyotime Institute of Biotechnology) according to the
manufacturer's protocol. Briefly, cells were incubated in 96-well
plates (5×103 cells/well) at 37°C. CCK-8 solution was
then added to each well at 24, 48 and 72 h, and the cells were
incubated for another 3 h at 37°C. The absorption value of each
well at 450 nm was detected using an ELx808 microplate reader
(BioTek Instruments, Inc.).
Colony formation assay
Cells were resuspended in DMEM and seeded into
6-well plates (500 cells/well) and incubated for 2 weeks. During
this period, the culture media were replaced every 2–3 days.
Finally, the cells were washed with PBS, fixed with 100% methanol
for 10 min at room temperature and stained with 0.5% crystal violet
for 5 min at room temperature. The number of cell colonies (>50
cells) was observed and counted using an inverted microscope
(Olympus Corporation).
Immunofluorescence assay
Cells were seeded in 24-well plates
(2×104 cells/well). Following a 24-h incubation at 37°C,
cells were fixed in 4% paraformaldehyde at 4°C for 30 min, washed
three times with PBS and then permeabilized with 0.5% Triton X-100
for 15 min at room temperature. Subsequently, the cells were
blocked with 5% bovine serum albumin (Thermo Fisher Scientific,
Inc.) at room temperature for 60 min, followed by incubation with a
primary antibody against Ki67 (1:200; cat. no. ab15580; Abcam)
overnight at 4°C. A mouse anti-rabbit immunoglobulin G
(IgG)-fluorescein isothiocyanate (FITC) secondary antibody (1:100;
cat. no. sc-2359; Santa Cruz Biotechnology, Inc.) was added and the
cells were incubated at room temperature for 1 h in the dark,
followed by the addition of DAPI at room temperature for 3 min
(also in the dark). Immunofluorescence images were acquired using a
confocal laser microscope (magnification, ×100; Leica Microsystems
GmbH).
Wound-healing assay
Cells were cultured to 100% confluence and the
monolayers were scratched using a 200-µl pipette tip to create a
wound. Non-adherent cells were removed by washing three times with
PBS, and cells were then cultured in fresh serum-free DMEM for 48
h. Images were captured at 0 and 48 h using an inverted light
microscope (magnification, ×100; Olympus Corporation).
Transwell assay
Cells were resuspended in 200 µl serum-free DMEM and
then seeded into the upper chambers of 24-well Transwell inserts
(5×104 cells/well), which were pre-coated with Matrigel
(BD Biosciences) overnight at 37°C; 600 µl DMEM supplemented with
10% FBS was added to the lower chambers. After a 24-h incubation
period, the cells on the upper surface of the chamber were removed
using a cotton swab, and the invasive cells were fixed with 100%
methanol at room temperature for 10 min and stained with 0.1%
crystal violet for 5 min at room temperature. The images were
captured using an inverted light microscope (magnification, ×100;
Olympus Corporation).
Chromatin immunoprecipitation (ChIP)
assay
It was predicted from JASPAR database (https://jaspar.genereg.net/) that there were three
potential IRF5 responsive elements binding to the ZNF217 promoter
region. To verify this binding relationship at the E2 region, ChIP
assay was conducted. Cells were cross-linked by incubation with 1%
formaldehyde for 10 min at 37°C, followed by quenching at room
temperature for 5 min with glycine (125 mM; Sigma-Aldrich; Merck
KGaA). Subsequently, cells were harvested by centrifugation at 300
× g for 3 min at room temperature, washed with PBS, and lysed in
SDS lysis buffer (Upstate Biotechnology, Inc.), and the chromatin
from the cell lysates was sonicated with a 10-sec on and 10-sec off
mode for 12 cycles on ice to shear the DNA into fragments at 20
kHz. Following sonication, the samples were centrifuged at 13,000 ×
g for 10 min at 4°C, and the supernatant was pre-adsorbed with 80
µl salmon sperm DNA/protein A-agarose (MilliporeSigma). The 100 µl
lysates were incubated with 5 µg anti-IgG (cat. no. ab109489;
Abcam) and anti-IRF5 (cat. no. ab181553; Abcam) antibodies for 2 h
at 4°C. The immune complexes were recovered using a salmon sperm
DNA-saturated protein A agarose gel (MilliporeSigma) according to
the manufacturer's protocol. Following immunoprecipitation and
elution, the eluent was heated to 65°C to reverse the cross-link.
The immunoprecipitated DNA was purified using the ChIP DNA Clean
& Concentrator Kits (Zymo Research) according to the
manufacturer's instructions and was quantified by RT-qPCR as
aforementioned. The ChIP primer sequences used for the detection of
ZNF217 enrichment on E2 promoter were as follows: Forward,
5′-TCTTCATGCCTCTACCCATCC-3′, and reverse,
5′-ATGGCTCTGCCCTAATCCTCT-3′.
Dual-luciferase reporter assay
Three deletion mutants (E1 Del, E2 Del and E3 Del)
from potential IRF5 responsive elements in the ZNF217 promoter
region (E1, E2, and E3) were obtained from Genloci Biotechnologies,
Inc. The IRF5-binding motif and ZNF217 promoter full length or the
deletion mutants were ligated into pGL3-based plasmids (Promega
Corporation). When cell growth reached 70%, cells were transfected
with 15 nM pGL3-based reporter constructs and pRL-SV40 (which was
used as the internal reference) using Lipofectamine 2000. After 48
h incubation at 37°C, the relative luciferase activity of each
group was determined using a Dual-Luciferase Reporter Assay kit
(Promega Corporation); luciferase activity was normalized to
Renilla.
Statistical analysis
All data were analyzed using SPSS software (version
20; IBM Corp.) and are presented as the mean ± SD from at least
three repeats. A paired Student's t-test (comparisons between two
groups) or one-way ANOVA with Tukey's post hoc test (comparisons
among ≥3 groups) were used to compare differences. For the
Kaplan-Meier survival curve analysis, Mantel-Cox (log-rank) test
was performed. To determine the correlation between ZNF217 and
IRF5, the Pearson's correlation coefficient was calculated using
the non-log scale and visualized using the log-scale axis.
P<0.05 was considered to indicate a statistically significant
difference.
Results
ZNF217 expression is upregulated in
patients with PC and is associated with a poor outcome
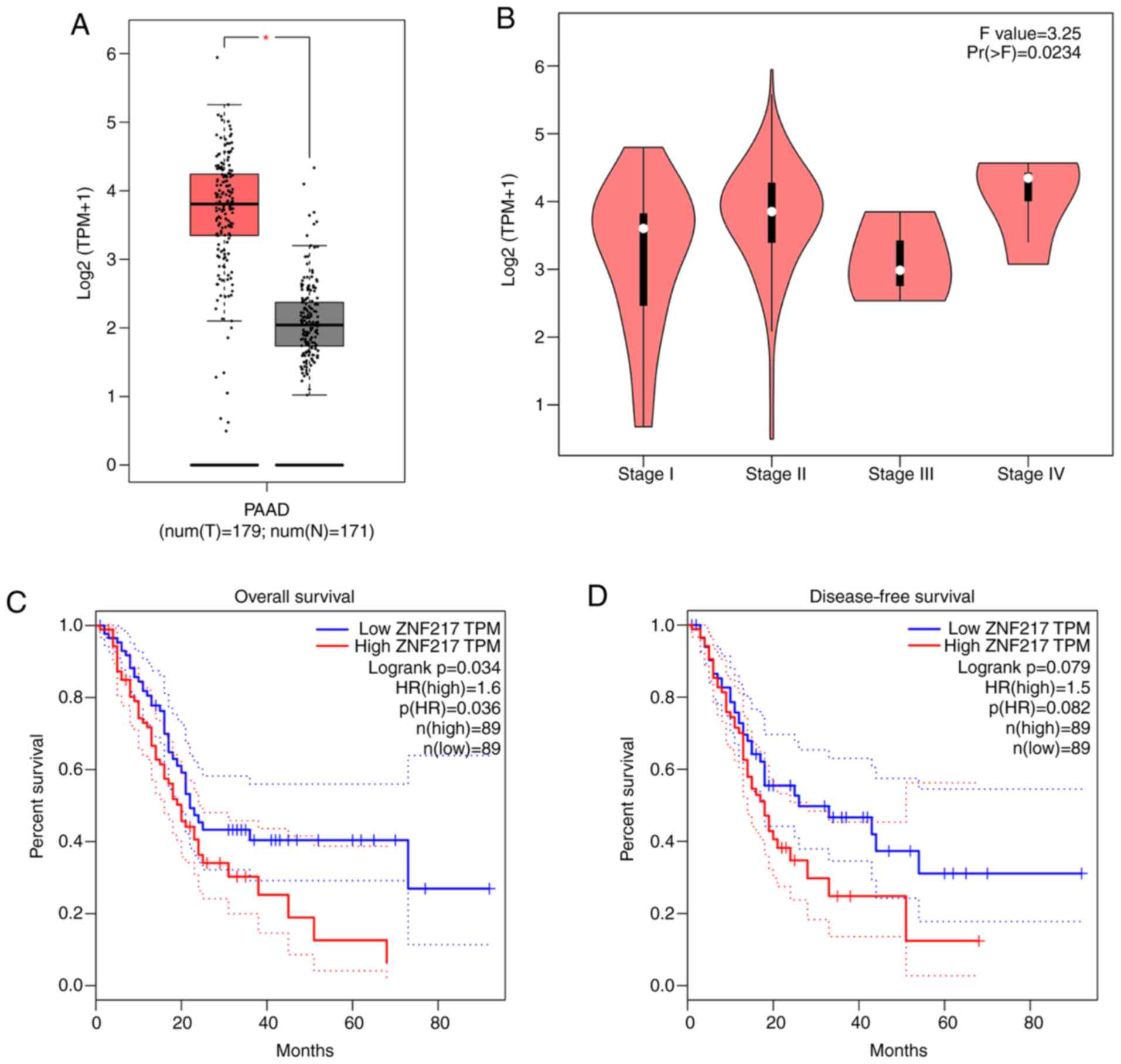
Data from GEPIA were analyzed to investigate ZNF217
expression in PC. Significantly higher expression levels of ZNF217
were observed in the tumor tissues of patients with PC compared
with the normal tissues (Fig. 1A).
The expression of ZNF217 was also found to vary with PC tumor stage
(Fig. 1B). The overall trend of
ZNF217 expression was upward from stage I to IV, with the exception
of at stage III. The abnormally low expression of ZNF217 expression
at stage III may be because relatively few samples were used. In
addition, Kaplan-Meier curve analysis revealed that high ZNF217
expression was significantly associated with poor overall survival
rate (P<0.05), but was not significantly associated with
disease-free survival rate (P=0.079) (cut-off value=50% used to
separate high and low groups) (Fig. 1C
and D). These findings demonstrated that ZNF217 is highly
expressed in the tumor tissues of patients with PC and suggested
that ZNF217 may be associated with poor survival rate.
ZNF217 silencing suppresses PC cell
proliferation
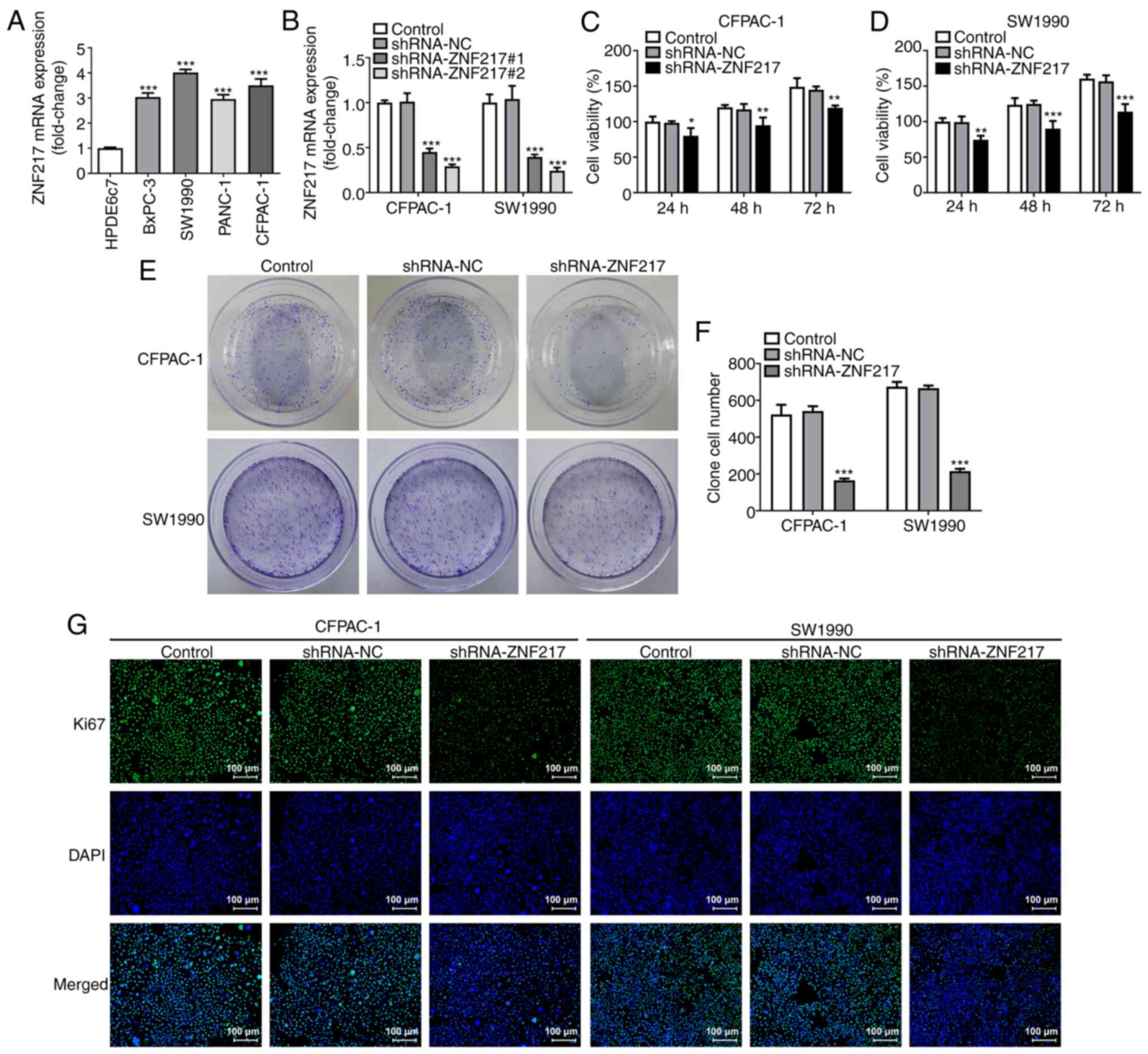
RT-qPCR was performed to detect the mRNA expression
levels of ZNF217 in different PC cell lines (BxPC-3, SW1990, PANC-1
and CFPAC-1) compared with the normal human pancreatic ductal
epithelial cell line (HPDE6c7). The results revealed that the
ZNF217 mRNA expression was significantly increased in PC cell
lines, but particularly in SW1990 and CFPAC-1 cells (Fig. 2A), thus SW1990 and CFPAC-1 cells
were used for subsequent experiments. To investigate the specific
role of ZNF217 in PC, ZNF217 expression was silenced by
transfection with shRNA-ZNF217#1 or shRNA-ZNF217#2 (Fig. 2B). Owing to a higher transfection
efficacy, shRNA-ZNF217#2 was used for further investigation. CCK-8
assays revealed that cellular viability was inhibited by silencing
ZNF217 expression, and that the inhibitory effects became more
significant with prolonged incubation times (Fig. 2C and D). The colony formation assay
results showed that ZNF217 knockdown significantly decreased the
number of SW1990 and CFPAC-1 cell colonies formed (Fig. 2E and F). In addition,
immunofluorescence detection revealed markedly decreased expression
of Ki67 upon ZNF217 silencing in both SW1990 and CFPAC-1 cells
(Fig. 2G). These findings suggested
that ZNF217 silencing suppressed cellular proliferation in PC.
ZNF217 silencing suppresses PC cell
migration and invasion
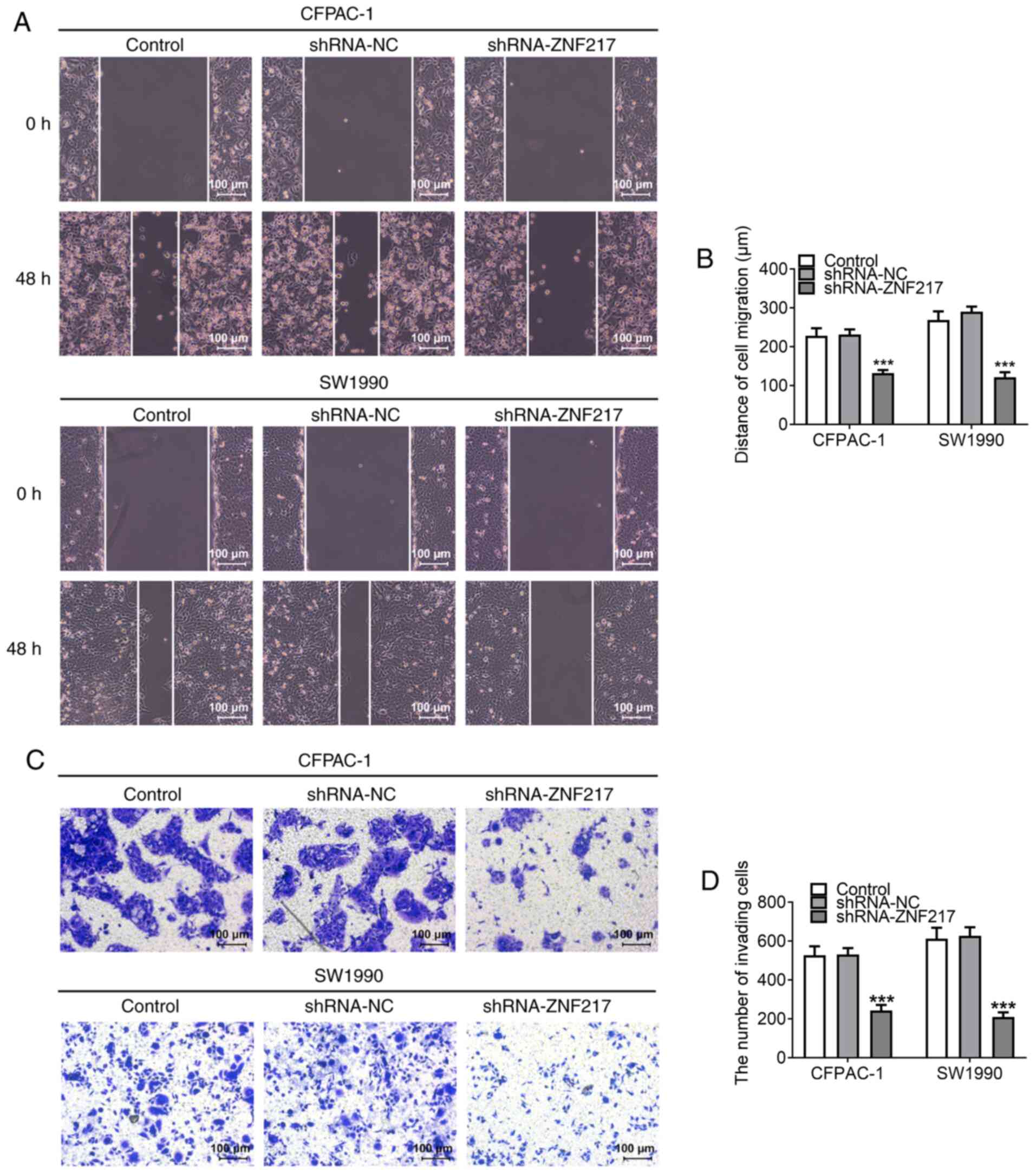
Wound-healing and Matrigel assays were performed to
evaluate PC cell migratory and invasive abilities, respectively. As
shown in Fig. 3A and B, the
distance of cell migration was reduced in CFPAC-1 and SW1990 cells
when ZNF217 expression was knocked down, indicating that ZNF217
silencing inhibited cellular migratory ability. In addition, the
number of invasive cells was decreased when ZNF217 was silenced
(Fig. 3C and D), indicating that
ZNF217 silencing inhibited cellular invasive ability.
IRF5 is upregulated in PC and
regulates ZNF217 expression by promoting ZNF217 transcription
To further verify the aforementioned results, the
expression of IRF5 in PC was assessed using bioinformatics
analysis. The GEPIA database revealed high expression levels of
IRF5 in PC tumor tissues compared with normal tissue (Fig. 4A). In addition, ZNF217 expression
showed a moderate positive linear correlation with that of IRF5 in
PC tissue samples (Fig. 4B).
Notably, three putative IRF5 responsive elements (E1, E2 and E3)
were identified in the ZNF217 promoter region (Fig. 4C). Through gain- and
loss-of-expression experiments, the effects of IRF5 on ZNF217 in PC
were investigated. RT-qPCR analysis demonstrated successful
transfection of the shRNA-IRF5#2 plasmid used for IRF5 silencing
and the Oe-IRF5 plasmid used for IRF5 overexpression (Fig. 4D). IRF5 knockdown significantly
decreased ZNF217 mRNA levels and transcriptional activity (Fig. 4E and F). To determine which element
was primarily responsible for the regulatory association between
IFR5 and ZNF217, the three putative IRF5 binding sites were then
individually deleted, and named E1-Del, E2-Del and E3-Del. Upon
IRF5 overexpression in CFPAC-1 and SW1990 cells, a limited increase
in ZNF217 transcriptional activity was observed when the E2 element
was deleted (Fig. 4G and H),
indicating that this element may be the primary responsible agent
for IRF5-induced activation of ZNF217 transcription. ChIP analysis
subsequently revealed that IRF5 was enriched at the ZNF217 promoter
within the E2 region (Fig. 4I),
demonstrating the binding association between IRF5 and the ZNF217
promoter.
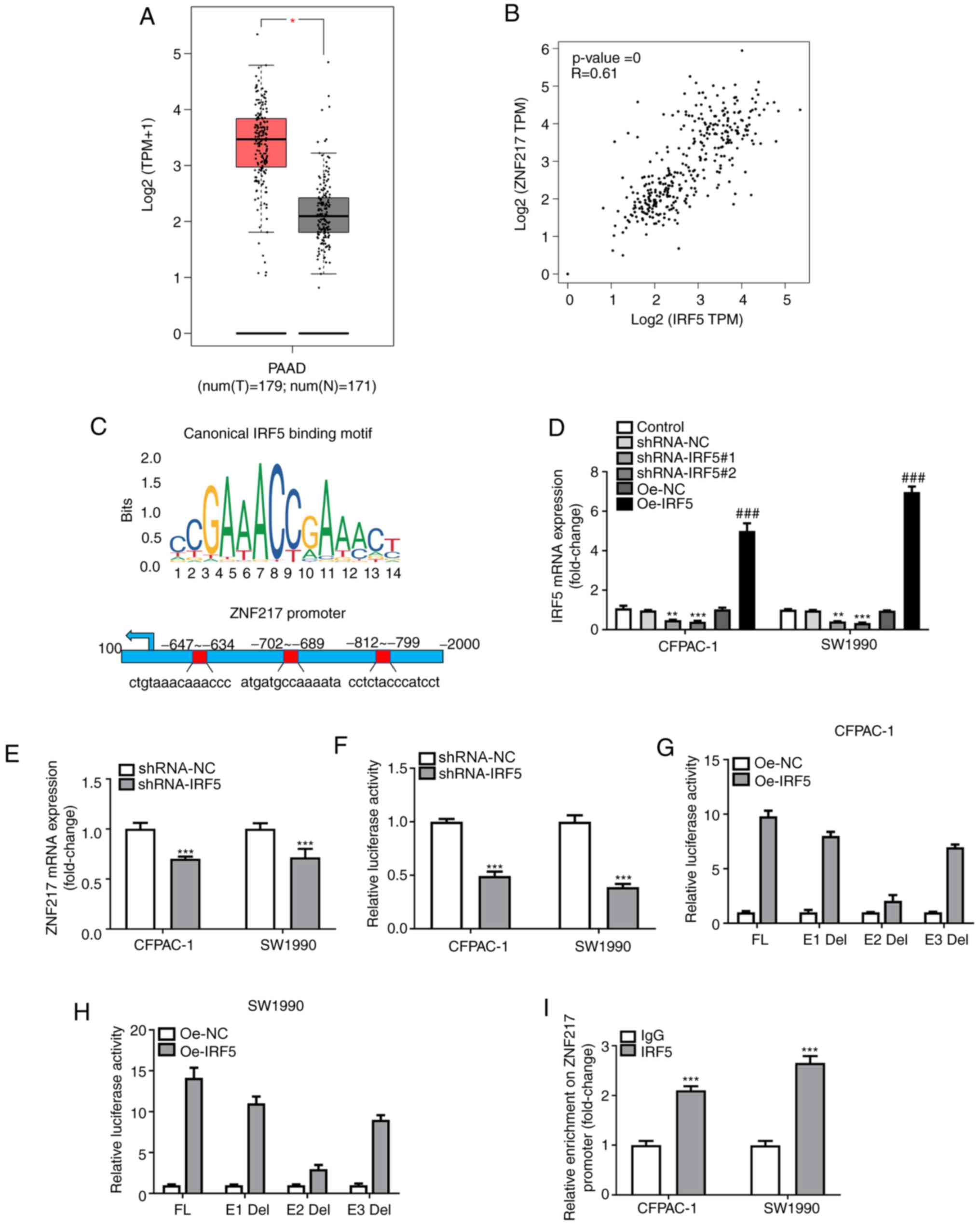 | Figure 4.IRF5 is increased in pancreatic
carcinoma and regulates ZNF217 expression. (A) Gene Expression
Profiling Interactive Analysis was used for IRF5 expression
analysis. The red box indicates the tumor samples, and the grey box
indicates the normal samples. *P<0.05. (B) Correlation between
ZNF217 and IRF5 expression. (C) Schematic diagram of the IRF5
binding motif (JASPAR database) and three potential IRF5 responsive
elements (E1, E2 and E3) in the ZNF217 promoter region. (D) IRF5
mRNA expression following knockdown or overexpression of IRF5 was
determined by RT-qPCR. **P<0.01, ***P<0.001 vs. shRNA-NC;
###P<0.001 vs. Oe-NC. (E) ZNF217 mRNA expression
following knockdown of overexpression of IRF5 was determined by
RT-qPCR. (F) Luciferase readout of ZNF217 promoter transcriptional
activity upon IFR5 silencing. ***P<0.001 vs. control. Luciferase
assay of three ZNF217 promoter deletion mutants upon IRF5
overexpression in (G) CFPAC-1 and (H) SW1990 cells. (I)
Immunoprecipitated chromatin fragments obtained from the chromatin
immunoprecipitation assay were analyzed by RT-qPCR. ***P<0.001
vs. IgG. Del, deletion; E, element; FL, full-length; HR, hazard
ratio; IRF5, interferon regulatory factor 5; N, normal; NC,
negative control; Oe, overexpression; PAAD, pancreatic
adenocarcinoma; RT-qPCR, reverse transcription-quantitative PCR;
shRNA, short hairpin RNA; T, tumor; TPM, transcripts per million;
TSS, transcriptional start; ZNF217, zinc-finger protein 217. |
IRF5 silencing suppresses PC cell
proliferation, migration and invasiveness by regulating ZNF217
The role of IRF5 in PC was evaluated. Compared with
HPDE6c7 cells, the mRNA expression levels of IRF5 were
significantly upregulated in SW1990 and CFPAC-1 PC cells (Fig. 5A). Subsequently, SW1990 cells were
transfected with Oe-NC or Oe-ZNF217, and the mRNA expression of
ZNF217 was significantly increased in the Oe-ZNF217 group compared
with the control (Fig. 5B). SW1990
cells were transfected with shRNA-IRF5 or co-transfected with
either Oe-NC or Oe-ZNF217, and a series of functional experiments
including CCK-8, colony formation, wound-healing and Matrigel
assays were conducted to assess cellular proliferation, migration
and invasiveness upon IRF5 silencing. IRF5 silencing significantly
decreased cell viability (Fig. 5C),
clone cell number (Fig. 5D and E),
migration (Fig. 5F and G) and
invasiveness (Fig. 5H and I),
indicating a potential antitumor activity of IRF5 knockdown.
Furthermore, the inhibitory effects of IRF5 silencing on cellular
proliferation, migration and invasiveness were partly abolished by
ZNF217 overexpression, suggesting that silencing IRF5 expression
had effects on PC cell proliferation, migration and invasiveness
through the regulation of ZNF217.
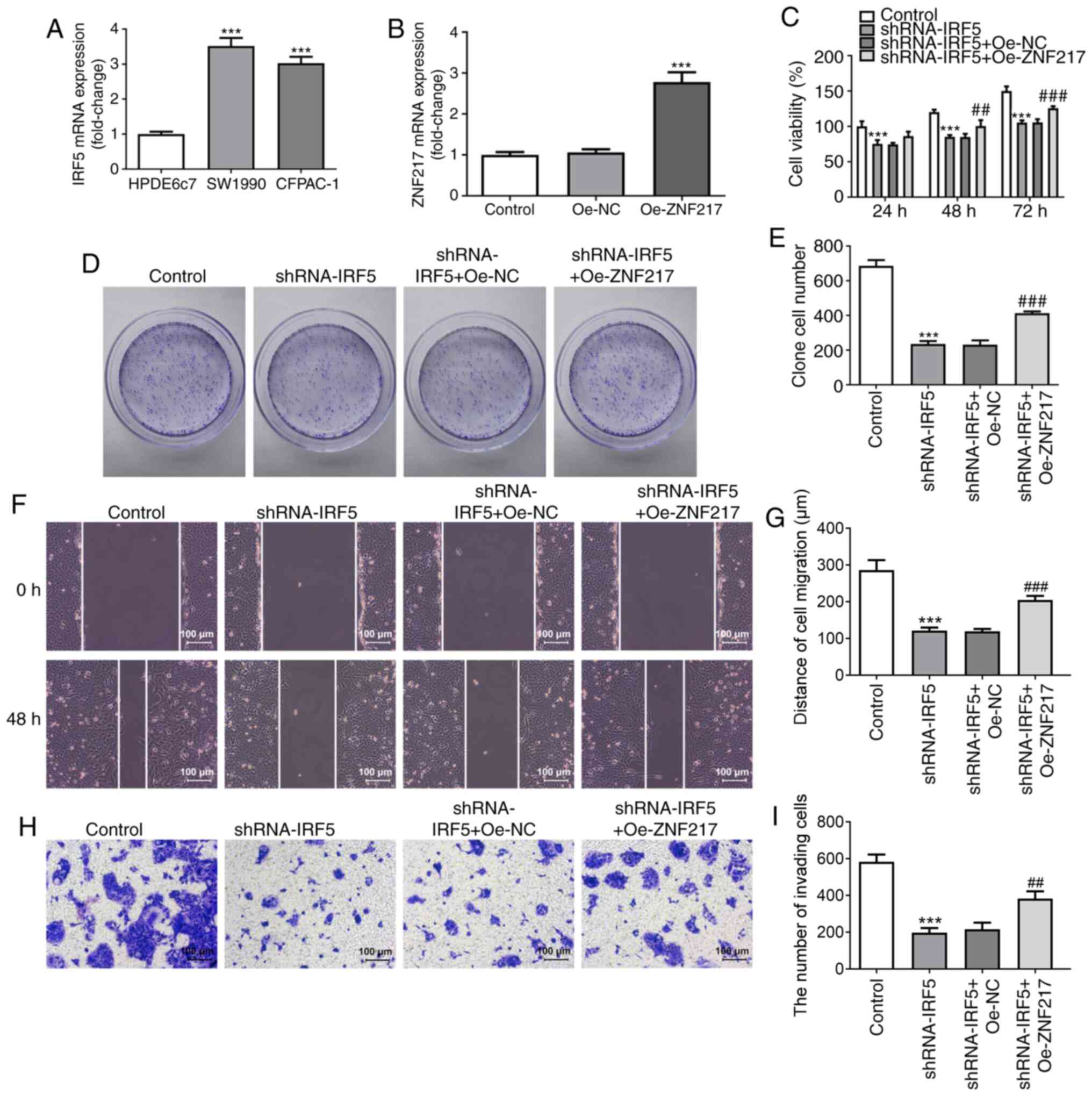 | Figure 5.IRF5 silencing suppresses PC cell
proliferation, migration and invasion by regulating ZNF217. (A)
IRF5 mRNA expression levels were detected in PC cell lines (SW1990
and CFPAC-1) and normal human pancreatic ductal epithelial cells
(HPDE6c7). ***P<0.001 vs. HPDE6c7 cells. (B) SW1990 cells were
transfected with Oe-NC or Oe-ZNF217, and ZNF217 mRNA expression was
detected. (C) SW1990 cells were transfected with shRNA-IRF5 alone
or co-transfected with either Oe-NC or Oe-ZNF217, and cell
viability at 24, 48 and 72 h post-transfection was detected using a
Cell Counting Kit-8 assay. (D) Colony formation and (E) the number
of colonies formed following transfection. (F) Wound-healing assays
were performed to assess SW1990 cell migration. (G) Quantification
of distance of cell migration. (H) Matrigel assay was performed to
assess SW1990 cell invasiveness. (I) Quantification of the number
of invading cells. ***P<0.001 vs. control;
##P<0.01, ###P<0.001 vs. shRNA-IRF5 +
Oe-NC. IRF5, interferon regulatory factor 5; NC, negative control;
Oe, overexpression; PC, pancreatic carcinoma; shRNA, short hairpin
RNA; ZNF217, zinc-finger protein 217. |
Discussion
In the past decades, several risk factors associated
with the occurrence of PC have been identified (13); however, owing to its unsatisfactory
5-year survival rate, there is an urgent requirement for the
identification of novel biomarkers to accurately monitor PC
development and progression. To the best of our knowledge, the
present study was the first to investigate the role and potential
regulatory mechanisms of ZNF217 in PC, the findings of which may
contribute to the early diagnosis, effective treatment and
prognosis of patients with PC.
ZNF217 has been found to be a crucial oncogene in
several types of human cancer. For example, ZNF217 was reported to
promote cellular migration, invasiveness and chemotaxis towards the
bone environment in breast cancer cells, accelerating breast cancer
metastasis to the bone (5). In
addition, elevated expression of ZNF217 promoted prostate cancer
growth by restraining ferroportin-mediated iron egress (14). Li et al (15) revealed that ZNF217 knockdown
attenuated the proliferation, migration and invasiveness of
colorectal cancer cells, confirming the oncogenic role of ZNF217 in
this malignancy. As the oncogenic activity of ZNF217 has been
reported, the inhibition of ZNF217 may present an important
approach for the treatment of malignant tumors. In the present
study, the GEPIA database revealed high expression levels of ZNF217
in the tumoral tissues compared with the normal tissues of patients
with PC, and this high expression level was associated with a poor
survival rate. A series of in vitro experiments revealed
that ZNF217 silencing significantly suppressed cell proliferation,
as well as migratory and invasive abilities, which was consistent
with the aforementioned previous reports, thus demonstrating an
important regulatory role for ZNF217 in PC, and a potential target
for PC diagnosis, treatment and prognosis.
In human cancers, epigenetic changes represent a
vital mechanism for the activation of oncogenes or the repression
of tumor suppressor genes (6).
Accumulating evidence has suggested that IRF5 works as a
transcription factor to influence and regulate gene transcription
in various diseases. For example, Guo et al (16) reported that IRF5 was able to enhance
the transcription of matrix metalloproteinase 3 by binding to its
promoter, thus regulating its expression in human chondrocytes.
Pimenta et al (17) reported
that IRF5 regulated mammary epithelial cell migration by binding to
α6-tubulin, thus altering filamentous actin bundling and promoting
breast cancer cell migration. In the present study, an interaction
between ZNF217 promoter (E2 region) and IRF5 was identified. IRF5
was found to regulate the transcriptional activity and, thus, the
mRNA expression of ZNF217 by directly occupying its promoter
region. Therefore, high expression levels of IRF5 may account for
the upregulation of ZNF217 in PC.
There are, however, some limitations to the present
study. This study only focused on the roles of ZNF217 and IRF5 at
the cellular level in vitro; thus, further validation of the
roles of ZNF217 and IRF5 in vivo and at the clinical level
is required in future work.
In conclusion, the findings of the present study
revealed that ZNF217 was upregulated in PC and was associated with
poor patient survival rates. Furthermore, ZNF217 silencing exerted
notable inhibitory effects on the proliferation, migration and
invasion of PC cells. Additionally, IRF5 was demonstrated to
positively regulate ZNF217 expression by binding to the
promoter-specific region in PC. Thus, ZNF217 may be a potential
target for developing novel therapeutic strategies for PC.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
XQ and SL wrote the paper. XQ, SL, YQ and FW
performed the experiments. XQ, SL and YQ analyzed the data. LM
designed the experiments and revised the manuscript. All authors
read and approved the final manuscript and agree to be accountable
for all aspects of the research in ensuring that the accuracy or
integrity of any part of the work are appropriately investigated
and resolved. XQ and LM confirm the authenticity of all the raw
data.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2019. CA Cancer J Clin. 69:7–34. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Cheng Y, Wang K, Geng L, Sun J, Xu W, Liu
D, Gong S and Zhu Y: Identification of candidate diagnostic and
prognostic biomarkers for pancreatic carcinoma. EBioMedicine.
40:382–393. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cohen PA, Donini CF, Nguyen NT, Lincet H
and Vendrell JA: The dark side of ZNF217, a key regulator of
tumorigenesis with powerful biomarker value. Oncotarget.
6:41566–41581. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhang ZC, Zheng LQ, Pan LJ, Guo JX and
Yang GS: ZNF217 is overexpressed and enhances cell migration and
invasion in colorectal carcinoma. Asian Pac J Cancer Prev.
16:2459–2463. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bellanger A, Donini CF, Vendrell JA,
Lavaud J, Machuca-Gayet I, Ruel M, Vollaire J, Grisard E, Győrffy
B, Bièche I, et al: The critical role of the ZNF217 oncogene in
promoting breast cancer metastasis to the bone. J Pathol.
242:73–89. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Si W, Zhao Y, Zhou J, Zhang Q and Zhang Y:
The coordination between ZNF217 and LSD1 contributes to
hepatocellular carcinoma progress and is negatively regulated by
miR-101. Exp Cell Res. 379:1–10. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Almuttaqi H and Udalova IA: Advances and
challenges in targeting IRF5, a key regulator of inflammation. FEBS
J. 286:1624–1637. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Massimino M, Vigneri P, Fallica M, Fidilio
A, Aloisi A, Frasca F and Manzella L: IRF5 promotes the
proliferation of human thyroid cancer cells. Mol Cancer. 11:212012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bai Q, Liu L, Xia Y, Wang J, Xi W, Qu Y,
Xiong Y, Long Q, Xu J, Guo J, et al: IRF5 is associated with
adverse postoperative prognosis of patients with non-metastatic
clear cell renal cell carcinoma. Oncotarget. 8:44186–44194. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dong SM, Lee HG, Cho SG, Kwon SH, Yoon H,
Kwon HJ, Lee JH, Kim H, Park PG, Kim H, et al: Hypermethylation of
the interferon regulatory factor 5 promoter in Epstein-Barr
virus-associated gastric carcinoma. J Microbiol. 53:70–76. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Tang Z, Li C, Kang B, Gao G, Li C and
Zhang Z: GEPIA: A web server for cancer and normal gene expression
profiling and interactive analyses. Nucleic Acids Res. 45:W98–W102.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Rawla P, Sunkara T and Gaduputi V:
Epidemiology of pancreatic cancer: Global trends, etiology and risk
factors. World J Oncol. 10:10–27. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Jiang X, Zhang C, Qi S, Guo S, Chen Y, Du
E, Zhang H, Wang X, Liu R, Qiao B, et al: Elevated expression of
ZNF217 promotes prostate cancer growth by restraining
ferroportin-conducted iron egress. Oncotarget. 7:84893–84906. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Li Z, Du L, Dong Z, Yang Y, Zhang X, Wang
L, Li J, Zheng G, Qu A and Wang C: MiR-203 suppresses ZNF217
upregulation in colorectal cancer and its oncogenicity. PLoS One.
10:e01161702015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Guo L, Hao R, Tian F, An N and Wang K:
Interferon regulatory factor 5 (IRF5) regulates the expression of
matrix metalloproteinase-3 (MMP-3) in human chondrocytes. Int
Immunopharmacol. 55:231–236. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Pimenta EM and Barnes BJ: A conserved
region within interferon regulatory factor 5 controls breast cancer
cell migration through a cytoplasmic and transcription-independent
mechanism. Mol Cancer. 14:322015. View Article : Google Scholar : PubMed/NCBI
|