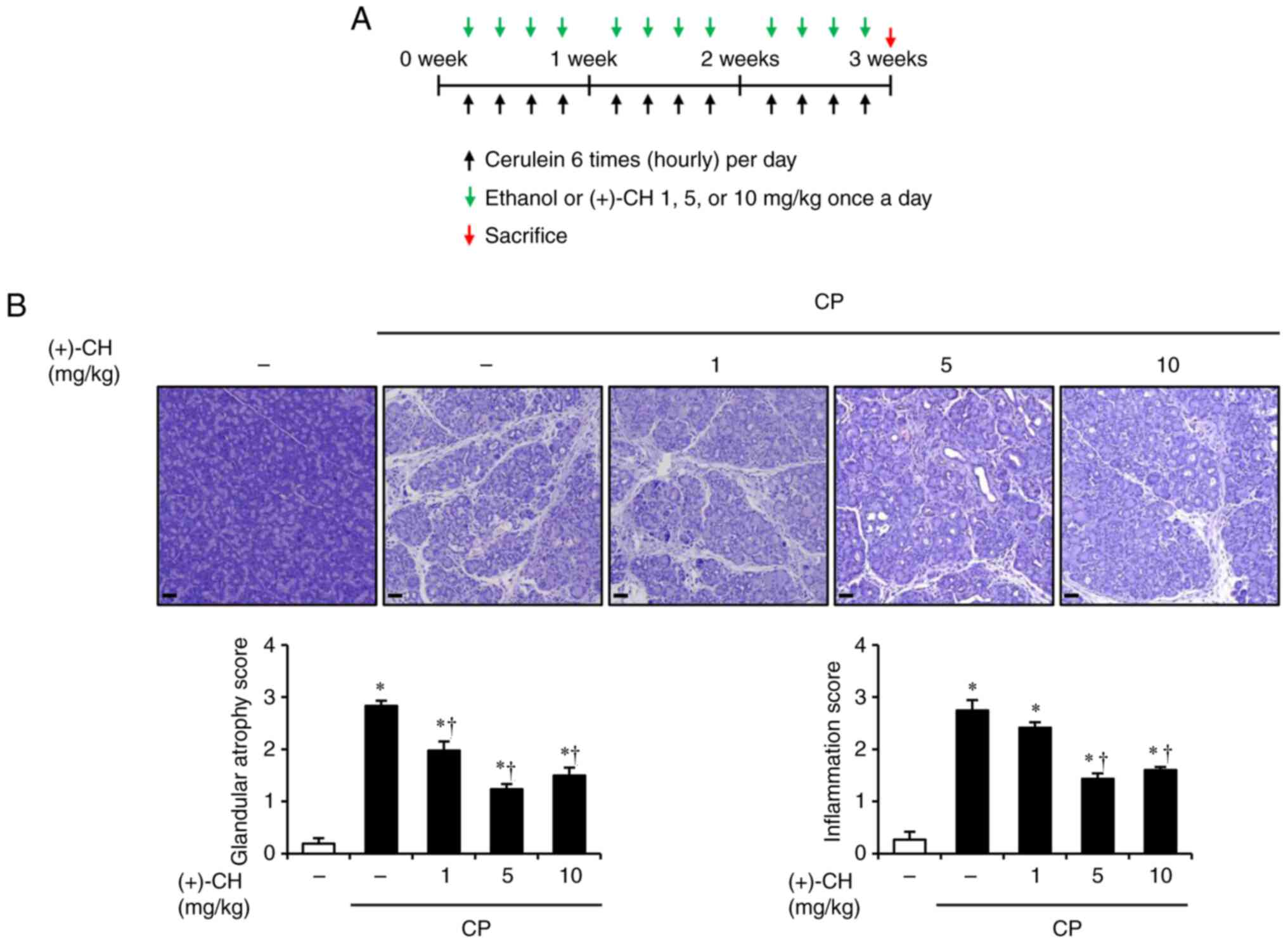
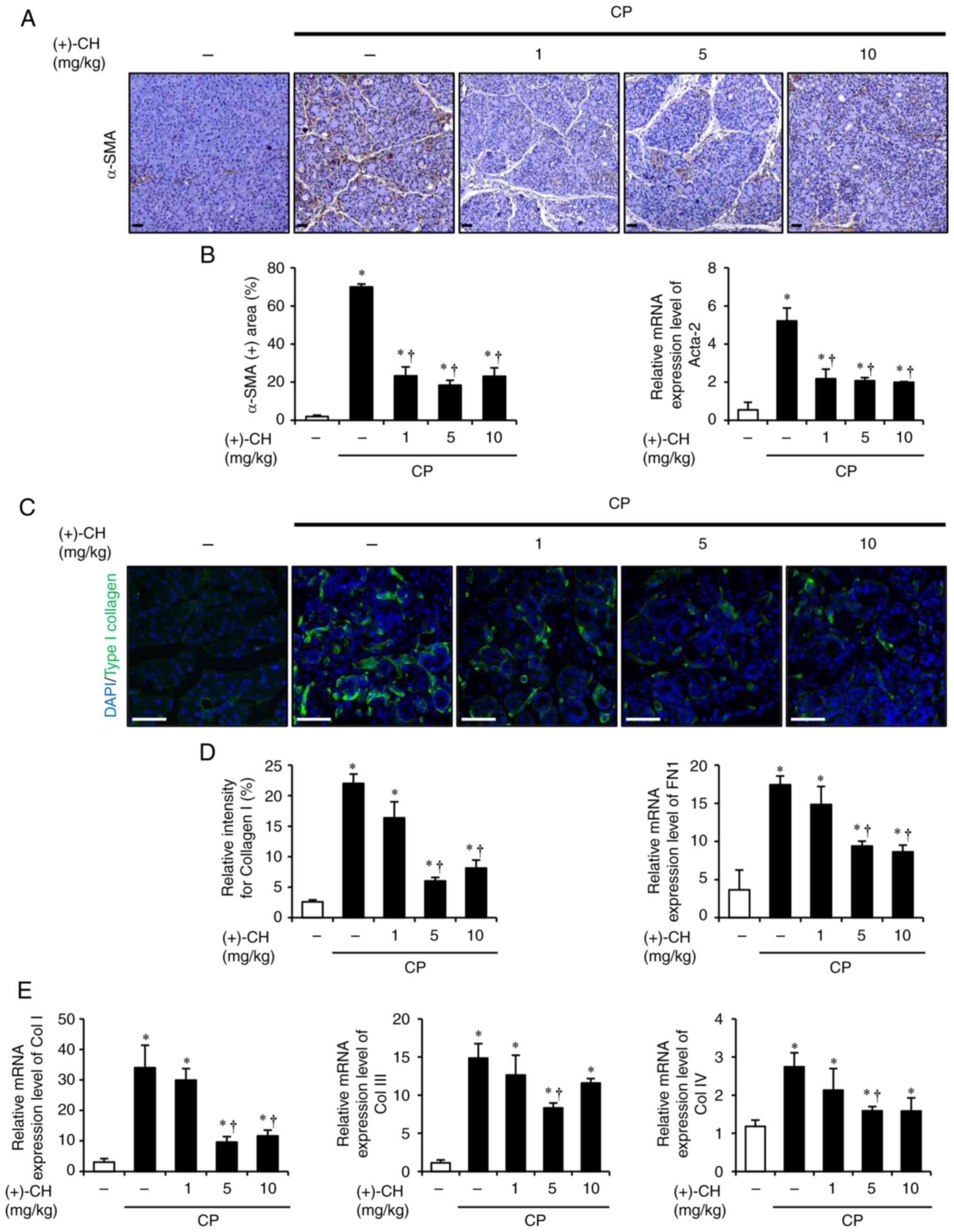
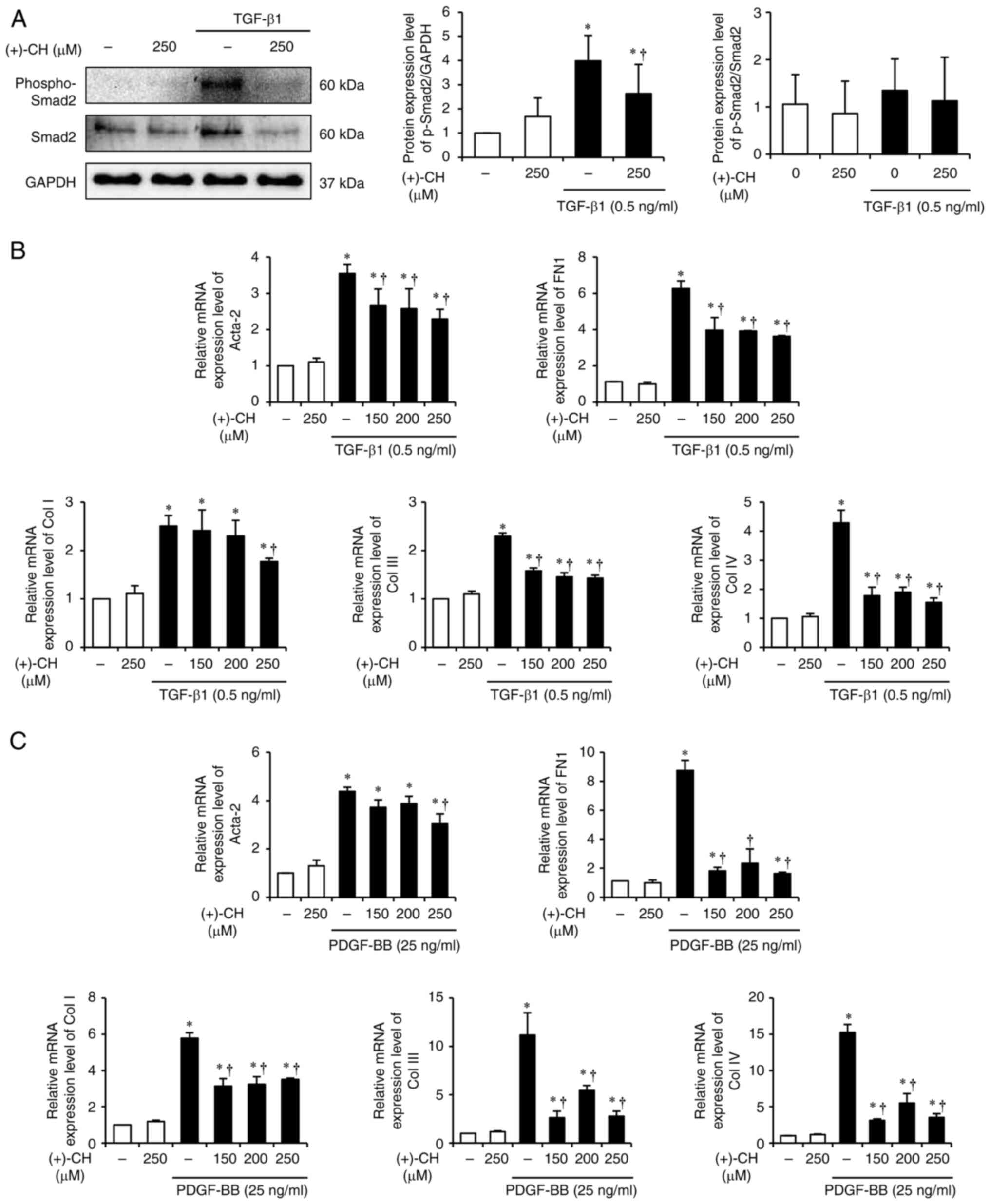
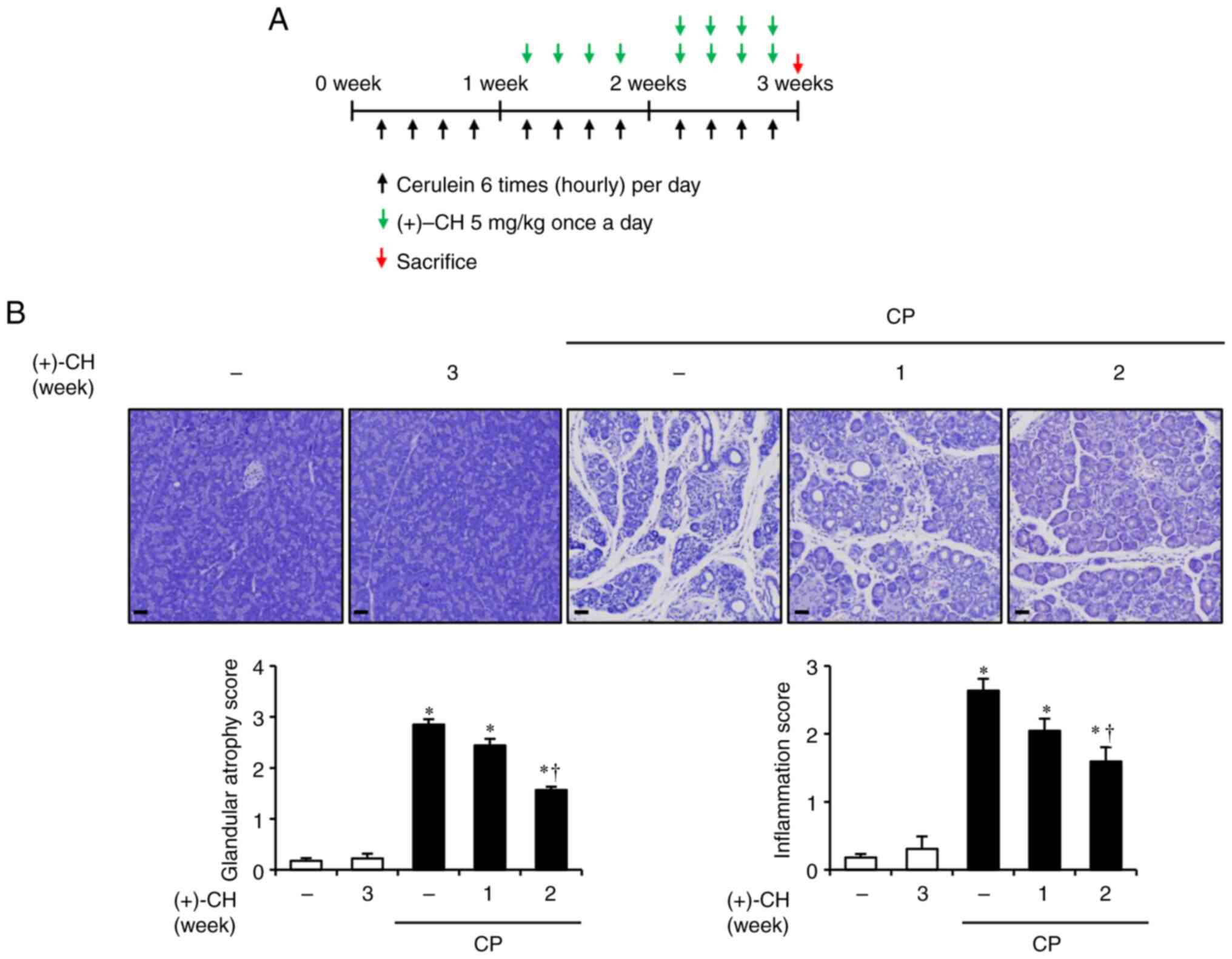
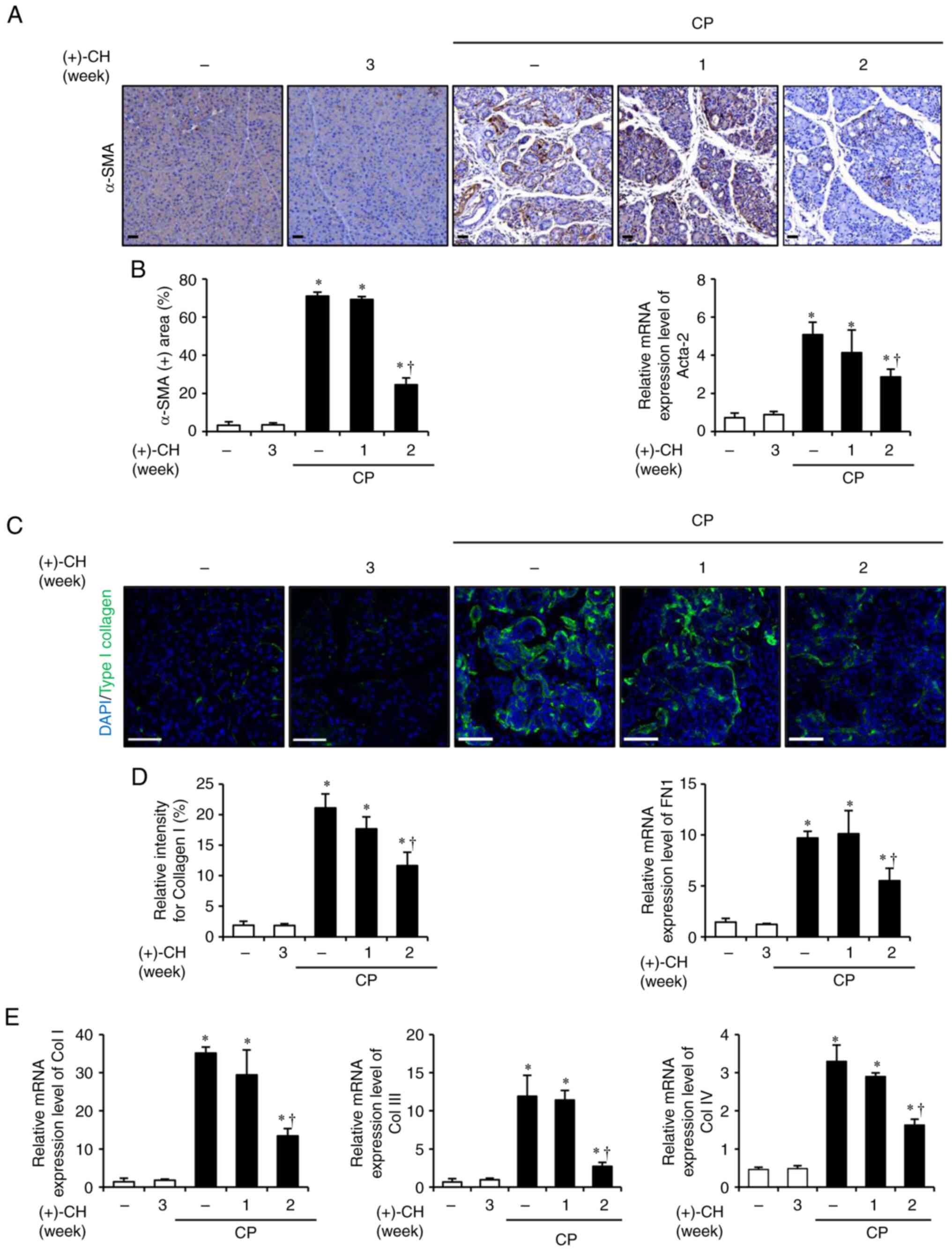
|
1
|
Yang HY, Wang D, Liu Y, Han C, Xin L, Li
ZS, Liao Z and Hu LH: Global status in chronic pancreatitis
research. Pancreas. 49:1283–1289. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Olesen SS, Mortensen LH, Zinck E, Becker
U, Drewes AM, Nøjgaard C, Novovic S, Yadav D and Tolstrup JS: Time
trends in incidence and prevalence of chronic pancreatitis: A
25-year population-based nationwide study. United European
Gastroenterol J. 9:82–90. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Etemad B and Whitcomb DC: Chronic
pancreatitis: Diagnosis, classification, and new genetic
developments. Gastroenterology. 120:682–707. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Olesen SS, Frandsen LK, Poulsen JL,
Vestergaard P, Rasmussen HH and Drewes AM: The prevalence of
underweight is increased in chronic pancreatitis outpatients and
associates with reduced life quality. Nutrition. 43–44. 1–7.
2017.
|
|
5
|
Lin CH, Yeh NC, Wang JJ, Ho CH, Her SH,
Tsay WI and Chien CC: Effect of chronic pancreatitis on
complications and mortality in DM patients: A 10-year nationwide
cohort study. J Clin Endocrinol Metab. 105:e739–e745. 2020.
View Article : Google Scholar
|
|
6
|
Drewes AM, Krarup AL, Detlefsen S,
Malmstrøm ML, Dimcevski G and Funch-Jensen P: Pain in chronic
pancreatitis: The role of neuropathic pain mechanisms. Gut.
57:1616–1627. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Madro A: Malnutrition in chronic
pancreatitis: Causes, assessment methods, and therapeutic
management. Can J Gastroenterol Hepatol.
2020:88754872020.PubMed/NCBI
|
|
8
|
Kirkegård J, Mortensen FV and
Cronin-Fenton D: Chronic pancreatitis and pancreatic cancer risk: A
systematic review and meta-analysis. Am J Gastroenterol.
112:1366–1372. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Malka D, Hammel P, Sauvanet A, Rufat P,
O'Toole D, Bardet P, Belghiti J, Bernades P, Ruszniewski P and Lévy
P: Risk factors for diabetes mellitus in chronic pancreatitis.
Gastroenterology. 119:1324–1332. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Han M, Tran TPT and Oh JK: Chronic
pancreatitis and cancer risk in a matched cohort study using
national claims data in South Korea. Sci Rep. 12:55452022.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Muniraj T, Aslanian HR, Farrell J and
Jamidar PA: Chronic pancreatitis, a comprehensive review and
update. Part I: Epidemiology, etiology, risk factors, genetics,
pathophysiology, and clinical features. Dis Mon. 60:530–550. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Dufour MC and Adamson MD: The epidemiology
of alcohol-induced pancreatitis. Pancreas. 27:286–290. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Koehn FE and Carter GT: The evolving role
of natural products in drug discovery. Nat Rev Drug Discov.
4:206–220. 2005. View
Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chacko SM, Thambi PT, Kuttan R and
Nishigaki I: Beneficial effects of green tea: A literature review.
Chin Med. 5:132010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Khan N and Mukhtar H: Tea polyphenols for
health promotion. Life Sci. 81:519–533. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kim HK, Yang TH and Cho HY: Antifibrotic
effects of green tea on in vitro and in vivo models of liver
fibrosis. World J Gastroenterol. 15:5200–5205. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Higdon JV and Frei B: Tea catechins and
polyphenols: Health effects, metabolism, and antioxidant functions.
Crit Rev Food Sci Nutr. 43:89–143. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Isemura M: Catechin in human health and
disease. Molecules. 24:5282019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Asaumi H, Watanabe S, Taguchi M, Tashiro
M, Nagashio Y, Nomiyama Y, Nakamura H and Otsuki M: Green tea
polyphenol (−)-epigallocatechin-3-gallate inhibits ethanol-induced
activation of pancreatic stellate cells. Eur J Clin Invest.
36:113–122. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cao Y, Bao S, Yang W, Zhang J, Li L, Shan
Z and Teng W: Epigallocatechin gallate prevents inflammation by
reducing macrophage infiltration and inhibiting tumor necrosis
factor-α signaling in the pancreas of rats on a high-fat diet. Nutr
Res. 34:1066–1074. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wei R, Cortez Penso NE, Hackman RM, Wang Y
and Mackenzie GG: Epigallocatechin-3-gallate (EGCG) suppresses
pancreatic cancer cell growth, invasion, and migration partly
through the inhibition of Akt pathway and epithelial-mesenchymal
transition: Enhanced efficacy when combined with gemcitabine.
Nutrients. 11:18562019. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim JM and Heo HJ: The roles of catechins
in regulation of systemic inflammation. Food Sci Biotechnol.
31:957–970. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
EFSA Panel on Food Additives and Nutrient
Sources added to Food (ANS), . Younes M, Aggett P, Aguilar F,
Crebelli R, Dusemund B, Filipič M, Frutos MJ, Galtier P, Gott D, et
al: Scientific opinion on the safety of green tea catechins. EFSA
J. 16:e052392018.PubMed/NCBI
|
|
24
|
Alshatwi AA, Hasan TN, Alqahtani AM, Syed
NA, Shafi G, Al-Assaf AH and Al-Khalifa AS: Delineating the
anti-cytotoxic and anti-genotoxic potentials of catechin hydrate
against cadmium toxicity in human peripheral blood lymphocytes.
Environ Toxicol Pharmacol. 38:653–662. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Babich H, Krupka M, Nissim HA and
Zuckerbraun HL: Differential in vitro cytotoxicity of
(−)-epicatechin gallate (ECG) to cancer and normal cells from the
human oral cavity. Toxicol In Vitro. 19:231–242. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Alshatwi AA: Catechin hydrate suppresses
MCF-7 proliferation through TP53/Caspase-mediated apoptosis. J Exp
Clin Cancer Res. 29:1672010. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kweon B, Kim DU, Oh JY, Oh H, Kim YC, Mun
YJ, Bae GS and Park SJ: Arecae pericarpium water extract alleviates
chronic pancreatitis by deactivating pancreatic stellate cells.
Front Pharmacol. 13:9419552022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rai A, Das S, Chamallamudi MR, Nandakumar
K, Shetty R, Gill M, Sumalatha S, Devkar R, Gourishetti K and Kumar
N: Evaluation of the aphrodisiac potential of a chemically
characterized aqueous extract of Tamarindus indica pulp. J
Ethnopharmacol. 210:118–124. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Shahid A, Ali R, Ali N, Hasan SK, Bernwal
P, Afzal SM, Vafa A and Sultana S: Modulatory effects of catechin
hydrate against genotoxicity, oxidative stress, inflammation and
apoptosis induced by benzo(a)pyrene in mice. Food Chem Toxicol.
92:64–74. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Bragança de Moraes CM, Bitencourt S, de
Mesquita FC, Mello D, de Oliveira LP, da Silva GV, Lorini V,
Caberlon E, de Souza Basso B, Schmid J, et al: (+)-Catechin
attenuates activation of hepatic stellate cells. Cell Biol Int.
38:526–530. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zang G, Sandberg M, Carlsson PO, Welsh N,
Jansson L and Barbu A: Activated pancreatic stellate cells can
impair pancreatic islet function in mice. Ups J Med Sci.
120:169–180. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Steer ML, Waxman I and Freedman S: Chronic
pancreatitis. N Engl J Med. 332:1482–1490. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zimmermann A, Gloor B, Kappeler A, Uhl W,
Friess H and Büchler MW: Pancreatic stellate cells contribute to
regeneration early after acute necrotising pancreatitis in humans.
Gut. 51:574–578. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gress T, Menke A, Bachem M,
Müller-Pillasch F, Ellenrieder V, Weidenbach H, Wagner M and Adler
G: Role of extracellular matrix in pancreatic diseases. Digestion.
59:625–637. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Kennedy RH, Bockman DE, Uscanga L, Choux
R, Grimaud JA and Sarles H: Pancreatic extracellular matrix
alterations in chronic pancreatitis. Pancreas. 2:61–72. 1987.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Omary MB, Lugea A, Lowe AW and Pandol SJ:
The pancreatic stellate cell: A star on the rise in pancreatic
diseases. J Clin Invest. 117:50–59. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Haber PS, Keogh GW, Apte MV, Moran CS,
Stewart NL, Crawford DH, Pirola RC, McCaughan GW, Ramm GA and
Wilson JS: Activation of pancreatic stellate cells in human and
experimental pancreatic fibrosis. Am J Pathol. 155:1087–1095. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Ohnishi H, Miyata T, Yasuda H, Satoh Y,
Hanatsuka K, Kita H, Ohashi A, Tamada K, Makita N, Iiri T, et al:
Distinct roles of Smad2-, Smad3-, and ERK-dependent pathways in
transforming growth factor-beta1 regulation of pancreatic stellate
cellular functions. J Biol Chem. 279:8873–8878. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Apte MV, Haber PS, Darby SJ, Rodgers SC,
McCaughan GW, Korsten MA, Pirola RC and Wilson JS: Pancreatic
stellate cells are activated by proinflammatory cytokines:
Implications for pancreatic fibrogenesis. Gut. 44:534–541. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Schwarzenberg SJ and Whitcomb DC: Causes
and contributing risk factors for chronic pancreatitis in children
and adolescents. UpToDate in Medicine.
|
|
42
|
Duggan SN, Ni Chonchubhair HM, Lawal O,
O'Connor DB and Conlon KC: Chronic pancreatitis: A diagnostic
dilemma. World J Gastroenterol. 22:2304–2313. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Klöppel G and Maillet B: Pathology of
acute and chronic pancreatitis. Pancreas. 8:659–670. 1993.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Aghdassi AA, Mayerle J, Christochowitz S,
Weiss FU, Sendler M and Lerch MM: Animal models for investigating
chronic pancreatitis. Fibrogenesis Tissue Repair. 4:262011.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zheng L, Xue J, Jaffee EM and Habtezion A:
Role of immune cells and immune-based therapies in pancreatitis and
pancreatic ductal adenocarcinoma. Gastroenterology. 144:1230–1240.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Saotome T, Inoue H, Fujimiya M, Fujiyama Y
and Bamba T: Morphological and immunocytochemical identification of
periacinar fibroblast-like cells derived from human pancreatic
acini. Pancreas. 14:373–382. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Bachem MG, Schneider E, Gross H,
Weidenbach H, Schmid RM, Menke A, Siech M, Beger H, Grünert A and
Adler G: Identification, culture, and characterization of
pancreatic stellate cells in rats and humans. Gastroenterology.
115:421–432. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Apte MV, Haber PS, Applegate TL, Norton
ID, McCaughan GW, Korsten MA, Pirola RC and Wilson JS: Periacinar
stellate shaped cells in rat pancreas: Identification, isolation,
and culture. Gut. 43:128–133. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
To WS and Midwood KS: Plasma and cellular
fibronectin: Distinct and independent functions during tissue
repair. Fibrogenesis Tissue Repair. 4:212011. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yamaguchi T, Kihara Y, Taguchi M, Nagashio
Y, Tashiro M, Nakamura H and Otsuki M: Persistent destruction of
the basement membrane of the pancreatic duct contributes to
progressive acinar atrophy in rats with experimentally induced
pancreatitis. Pancreas. 31:365–372. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Liu B, Ding Y, Li P, Wang T, He S, Jia Z
and Yang J: MicroRNA-219c-5p regulates bladder fibrosis by
targeting FN1. BMC Urol. 20:1932020. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Sand J, Genovese F and Karsdal M: Type IV
collagen. Biochemistry of Collagens, Laminins and Elastin.
Elsevier; Amsterdam: pp. 31–41. 2016, View Article : Google Scholar
|
|
53
|
Hu LH, Ji JT and Li ZS: Potential
application of miRNAs as diagnostic and therapeutic tools in
chronic pancreatitis. J Cell Mol Med. 19:2049–2057. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Attisano L and Wrana JL: Signal
transduction by the TGF-beta superfamily. Science. 296:1646–1647.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Gurusamy KS, Lusuku C and Davidson BR:
Pregabalin for decreasing pancreatic pain in chronic pancreatitis.
Cochrane Database Syst Rev. 2:CD0115222016.PubMed/NCBI
|
|
56
|
Kwon CI, Cho JH, Choi SH, Ko KH, Tirkes T,
Gromski MA and Lehman GA: Recent advances in the diagnosis and
management of chronic pancreatitis. Korean J Intern Med.
34:242–260. 2019. View Article : Google Scholar : PubMed/NCBI
|