Introduction
Low back pain (LBP) is a prevalent worldwide health
issue, which not only impairs patient mobility, but also leads to a
loss of productivity and imposes a notable economic burden on
society (1). According to a
previous study, 84% of global population experience LBP in their
daily lives (2), and a 2019
systematic analysis covering 369 diseases across 204 countries
identified LBP as the fourth leading cause of disability among
individuals aged 25–74 years (3).
The etiology of LBP is multifactorial, with intervertebral disc
degeneration (IVDD) considered to be the main underlying cause of
LBP among the various etiological factors (4).
IVDD is characterized by structural deterioration of
the IVD, often resulting in premature disc aging (5). With the progression of IVDD, the
level of inflammatory cytokines increases, the degradation of
collagen increases and the phenotype of intervertebral disc cells
change (6). According to previous
evidence-based research, multiple factors including mechanical,
genetic, traumatic, inflammatory and biological factors cause or
accelerate the development of IVDD (7). However, genetic predisposition is
particularly notable, with increasing evidence highlighting the
involvement of long non-coding RNAs (lncRNAs) in IVDD
pathophysiology (8).
Circular RNAs (circRNAs), a prominent class of
ncRNAs, have emerged as pivotal regulators in IVDD pathogenesis,
with their complex mechanisms becoming increasingly elucidated
through advancing experimental investigations and in-depth research
(9). As circRNAs are substantiated
by robust statistical analyses and biochemical evidence, they have
been established as integral components of spliced transcripts
across hundreds of genes, underscoring their fundamental biological
significance (10). Despite their
demonstrated involvement in diverse pathological processes, the
precise molecular mechanisms and functional implications of
circRNAs in IVDD pathogenesis remain incompletely characterized.
The present comprehensive review systematically examined previous
and recent advances in understanding circRNA-mediated regulatory
networks in IVDD progression, particularly evaluating their
emerging potential as diagnostic biomarkers and therapeutic
targets, and identified current research limitations to inform
future investigative directions.
Normal anatomic structure of IVD and the
pathogenesis of IVDD
Structure and function of IVD
The IVD is a fibrocartilaginous structure positioned
between adjacent vertebrae, comprising three primary components: i)
The nucleus pulposus (NP); ii) the annulus fibrosus (AF); and iii)
the cartilaginous endplate (CEP) (11). The gelatinous NP resides at the
core of the disc and is primarily composed of water, a dense
network of type II collagen and proteoglycans (12). This composition creates a hydrated,
gel-like structure that generates substantial osmotic pressure,
which is essential for maintaining disc height, distributing
mechanical loads and ensuring overall disc integrity (13).
Encapsulating the NP is the organized AF, which
consists of 15–25 concentric lamellae of collagen fibers (14). This structure exhibits a
morphological gradient: The inner AF is rich in type II collagen,
providing elasticity and integration with the NP, whereas the outer
AF is composed of robust type I collagen fibers, offering tensile
strength and resistance to mechanical stress (15). This anisotropic, multi-lamellar
architecture effectively contains the NP and provides structural
stability to the disc (16).
The CEP are layers of hyaline cartilage situated
superiorly and inferiorly to the NP. In adults, these avascular
structures serve as the critical interface for nutrient diffusion
and metabolic waste exchange between the vascular-rich vertebral
bodies and the avascular disc interior (17).
The functional integrity of the IVD relies on the
precise structural and biochemical equilibrium among the NP, AF and
CEP. The disruption of this delicate homeostasis is a hallmark of
IVDD pathogenesis.
Pathophysiology of IVDD
IVDD is influenced by multiple risk factors,
including smoking, alcohol consumption, occupational standing
height, socioeconomic status, sleep disturbances, hypertension,
type II diabetes and obesity. Of note, metabolic dysregulation
appears to have a more profound impact on IVDD progression than
biomechanical stress (18). The
pathological process of IVDD is characterized by extracellular
matrix dysregulation, leading to progressive proteoglycan loss and
dehydration of the NP (19). In
addition, excessive mechanical loading and aging impair cartilage
endplate function, further exacerbating disc degeneration (20). Apoptosis and autophagy of nucleus
pulposus cells (NPCs) contribute markedly to IVDD pathophysiology,
whereas hypoxia and an acidic microenvironment deplete essential
nutrients, ultimately resulting in AF fissures and NP herniation
(21). Inflammatory cytokines,
including tumor necrosis factor-α (TNF-α) and interleukins-1α/β
(IL-1α/β), IL-6 and IL-17, play a critical role in promoting disc
degeneration by inducing metabolic imbalances within the IVD
(2).
In general, the molecular mechanisms affecting IVDD
are varied, but the main factors involved are apoptosis, the
release of inflammatory factors and the degradation of
extracellular matrix (ECM) (22).
The depletion of ECM components results from metabolic
disturbances, leading to a marked reduction in type II collagen,
proteoglycans and water content. Concurrently, the activity of
ECM-degrading enzymes, such as matrix metalloproteinases (MMPs),
aggrecanases and A disintegrin and MMP with thrombospondin motifs
(ADAMTS), is increased, further accelerating ECM degradation
(23). Along with ECM degradation,
the activation and infiltration of immune cells, including
CD4+ and CD8+ T cells, M1 macrophages, mast
cells and neutrophils, as well as pyroptosis and apoptosis of NP
cells, contribute to the release of pro-inflammatory cytokines and
chemokines, perpetuating a self-reinforcing inflammatory cascade.
These mediators, such as TNF-α, IL-1β, IL-6, IL-8 and prostaglandin
E2, further accelerate ECM breakdown, thus forming a vicious cycle
(24). At the same time, metabolic
imbalances in ECM and the production of inflammatory substances
promote the aging and death of NP cells (25). As a result, apoptosis, ECM
degradation and inflammatory cytokine production are interrelated
and interdependent and together lead to the degradation of IVD
(Fig. 1).
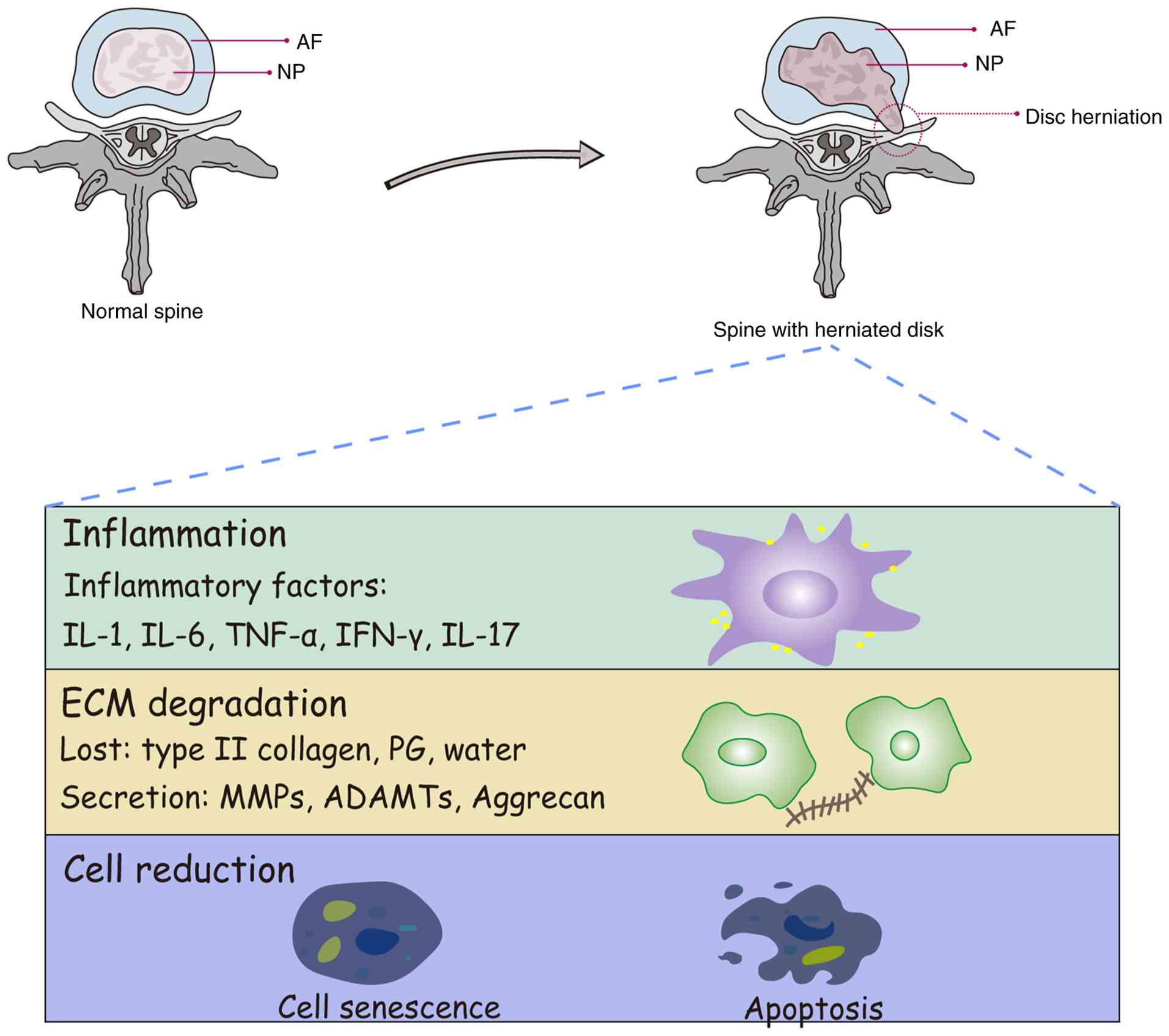 | Figure 1.Pathophysiology of IVDD. The IVD,
composed of the NP, AF and nutrient-supplying CEP, undergoes
degeneration through multifactorial etiologies. Risk modulators
including lifestyle factors, biomechanical stress, socioeconomic
parameters and metabolic disorders collectively drive pathological
cascades. These stimuli induce cellular apoptosis, ECM degradation
and inflammatory mediator release, ultimately progressing to disc
structural failure. Advanced degeneration manifests as disc height
loss, annular rupture with NP herniation and nerve root
compression, establishing a direct mechanistic basis for chronic
low back pain. IVDD, intervertebral disc degeneration; NP, nucleus
pulposus; AF, annulus fibrosus; CEP, cartilaginous endplates; ECM,
extracellular matrix; MMP, matrix metalloproteinase; ADAMTs, A
disintegrin and MMP with thrombospondin motifs. |
Characteristics, history, synthesis,
mechanism and function of circRNA
ncRNAs, comprising primarily microRNAs (miRNAs),
lncRNAs and circRNAs, have emerged as crucial regulators in various
cellular processes through extensive research over the past decades
(26). Among these ncRNA subtypes,
circRNAs have attracted particular scientific attention due to
their unique covalently closed circular structure and regulatory
potential (27). The investigation
of circRNAs has undergone notable evolution since their initial
discovery, marking important milestones in the understanding of RNA
biology.
From the beginning, in 1976, Sanger et al
(28) discovered viroids
containing single-stranded and covalently closed circular RNA
molecules in higher plants. Then in the 1990s, there was a
noticeable decline in interest and enthusiasm for circRNA. The
discovery of the human estrogen sulfotransferase 1 gene and the
deletion of the colon cancer gene were attributed to atypical
splicing events during transcription. In addition, the human
cytochrome P450, rat androgen binding protein and rat cytochrome
P450 2C 24 genes were found to generate other circRNAs. Although
the existence of circRNA has been verified, the value of circRNA
has been markedly underestimated (28). It was only discovered in 2012 that
circRNAs exist in a variety of human cell types, and that they can
be derived from the reverse splicing of precursor mRNAs (pre-mRNAs)
(29); since then, the research on
circRNA has been markedly increased. Due to the circRNA ring
structure and long half-life, circRNA has become a biological
marker for the examination of cancer cells, and provides a new
target and direction for future cancer treatment (30). In addition, the success of circRNA
synthesis in vitro has made it possible for the stability,
low cytotoxicity and long antigen-producing ability of circRNA to
play a role in vaccine research (31). As RNA technology continues to
advance, it is highly likely that the considerable potential of
circRNAs in therapeutic interventions for various diseases will be
further substantiated.
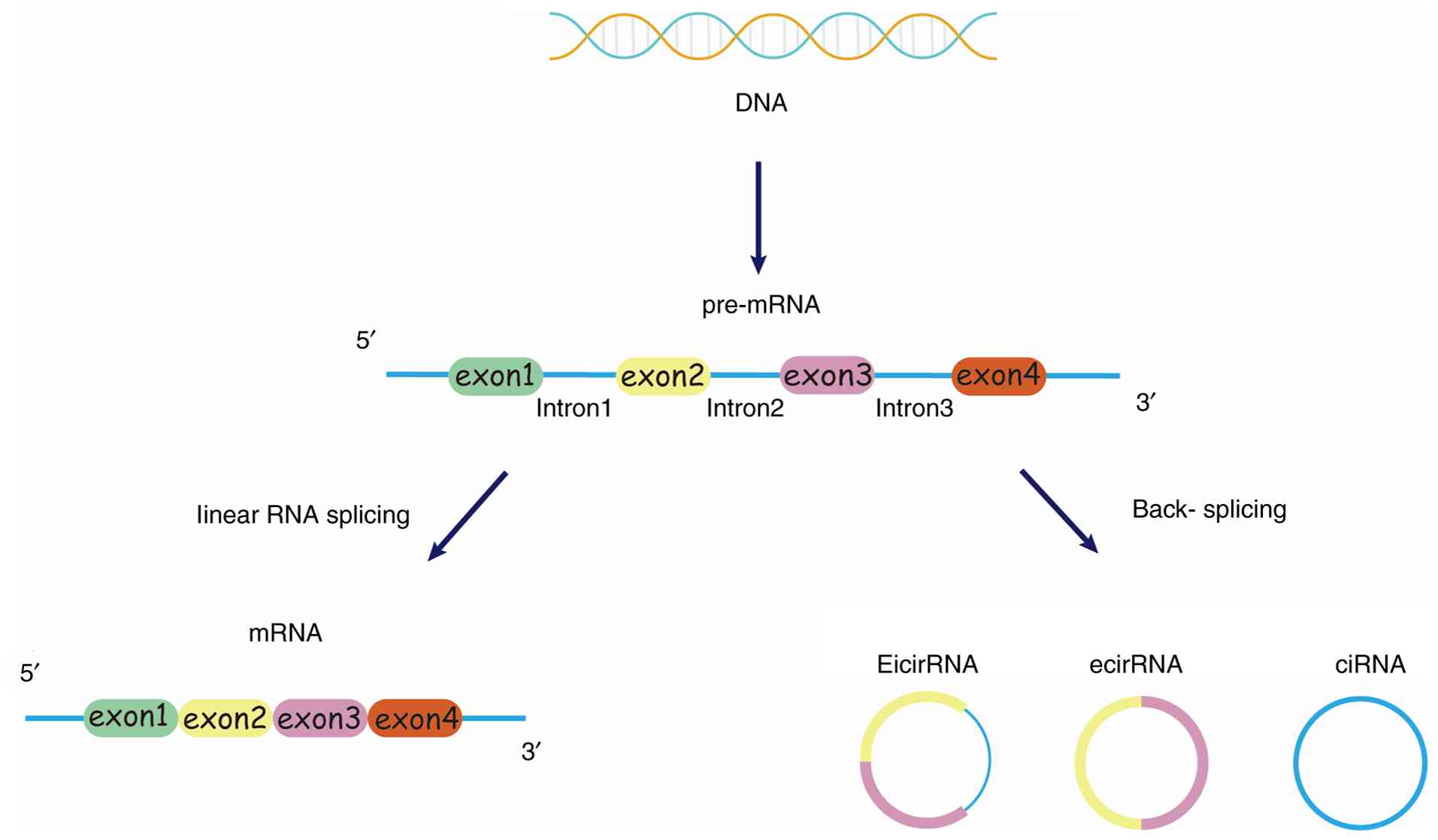
CircRNAs, as a subset of ncRNAs, are formed through
the back-splicing of pre-mRNA (32). This results in a closed-loop
structure that lacks 3′ and 5′ termini, rendering circRNA resistant
to exonuclease degradation and conferring remarkable stability
(33); however, circRNAs are
generally synthesized at the expense of their linear equivalents
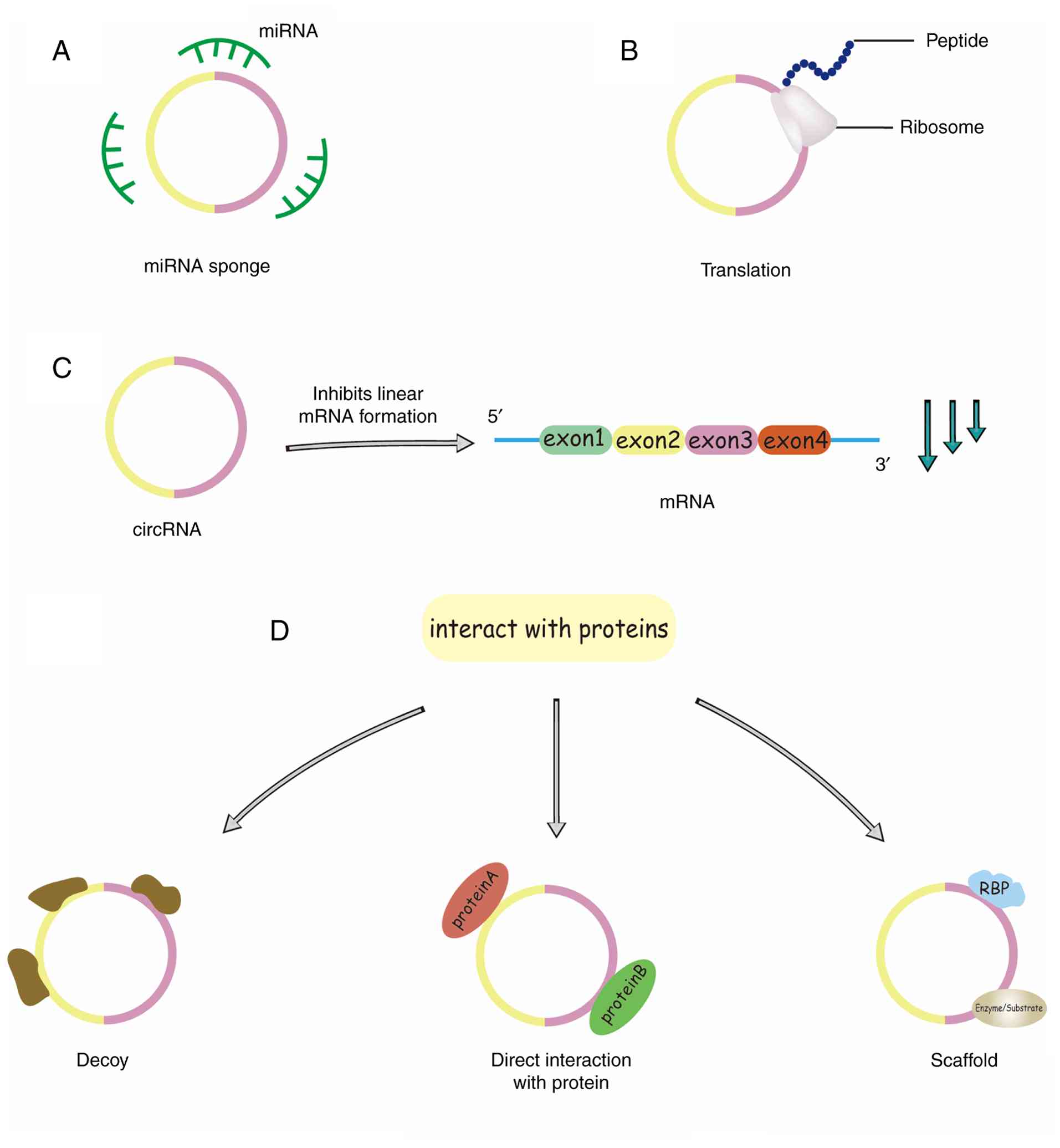
(Fig. 2C) (34). CircRNA has been reported to be
involved in physiological and pathological processes in a variety
of ways. circRNAs can act as miRNA sponges (Fig. 2A) or decoys to enhance the activity
of target RNA translation. Furthermore, circRNAs can interact with
RNA-binding proteins (RBPs) through distinct mechanisms: they can
act as protein ‘sponges’ to sequester RBPs and indirectly regulate
their function, or they can bind to specific proteins directly to
enhance their activity. Some circRNAs serve as molecular scaffolds,
facilitating interactions between enzymes and substrates to
modulate reaction kinetics (Fig.
2D). Furthermore, circRNAs can recruit specific proteins to
distinct cellular compartments, thereby influencing various
physiological and pathological processes (35). Emerging evidence reveals that
circRNAs exhibit protein-coding potential, even though
protein-coding genes constitute <2% of the entire genome
(Fig. 2B) (36).
CircRNA is similar to linear mRNA in that circRNA is
derived from the linear pre-mRNA transcribed by RNA polymerase II
and can be produced by reverse splicing (37). Based on their composition, circRNAs
can be divided into three types: Exonic circRNAs (ecircRNAs),
exon-intron circRNA (eicircRNA) and circular intronic RNA (ciRNA)
(Fig. 3). EcircRNAs are composed
of ≥1 exons, and represent ~85% of all circRNAs (38); they predominantly localize to the
cytoplasm, where they play crucial roles in post-transcriptional
regulation. By contrast, eicircRNAs and ciRNAs are typically
retained in the nucleus, where they modulate gene transcription
(39). In addition, ecircRNAs
localized in the cytoplasm are very stable in the cell, with a
half-life four times longer than the average half-life of mRNA
(10). Due to their cytoplasmic
localization and structural stability, ecircRNAs have been the most
extensively studied to date (40).
At present, >1 million reliable circRNAs have been identified
across various species (41), with
numerous studies having demonstrated that circRNAs exhibit tissue
and developmental stage-specific expression patterns and are
implicated in diverse cellular processes and disease pathogenesis
(42,43). Of note, despite the canonical miRNA
sponge theory, recent studies have revealed a spectrum of
alternative mechanisms for circRNAs, including their role as
dynamic molecular scaffolds that facilitate enzyme-substrate
interactions, their capacity to modulate transcription in the
nucleus and their potential for cap-independent translation to
generate functional peptides (44). This expanded functional repertoire
markedly enhances their regulatory complexity in cellular processes
and disease pathogenesis (45).
However, despite their recognized roles in various diseases, the
precise functions of circRNAs in IVDD remain incompletely
understood.
Regulatory role of circRNA in IVDD
As previously discussed, circRNAs modulate cellular
functions through diverse mechanisms. To better elucidate their
role in IVDD, a comprehensive review of existing literature was
conducted, and circRNA functions were categorized into four
distinct mechanisms based on their influence on IVDD. This
classification not only facilitates a deeper understanding of
individual circRNA-mediated regulatory pathways but also provides
insights for future research directions and potential clinical
applications.
CircRNA can act as miRNA sponges
Due to their closed-loop structure, circRNAs lack
free 3′ and 5′ ends, rendering them stable and evolutionarily
conserved in eukaryotic cells (28). As a direct target of circRNAs and
as a member of ncRNAs, miRNAs function as key post-transcriptional
regulators by binding to the 3′-untranslated region of target
mRNAs, leading to translation inhibition or transcript degradation
(46). Since miRNA and its target
RNA have complementary binding sites, and both circRNAs and miRNAs
are mainly located in the cytoplasm, circRNAs can regulate the
translation rate of target mRNA and affect its stability by pairing
with the complementary sequence of miRNA (47).
With the increased understanding of and research on
ncRNAs, the intricate interactions between circRNAs and miRNAs,
along with their biological implications, have become progressively
clearer. The ability of circRNAs to act as miRNA sponges has been
implicated in various disease processes, including IVDD. CircRNAs
can modulate disc degeneration by sequestering miRNA regulatory
factors, thereby suppressing the translation of target mRNAs
(48).
For instance, circEYA3 has been identified as a key
regulator in IVDD, promoting ECM degradation, inflammatory
responses and apoptosis in NP cells. By integrating
circRNA/miRNA/mRNA interactions, researchers established a
circRNA/miRNA/mRNA regulatory network centered on
circEYA3/miR-196a-5p/early B-cell factor 1 (EBF1). Experimental
validation demonstrated that miR-196a-5p expression was inversely
associated with circEYA3 and EBF1 expression levels (49). Further in vivo studies
confirmed that circEYA3 acts as a sponge for miR-196a-5p,
indirectly regulating EBF1 expression. In addition, Wang et
al (49) demonstrated that
EBF1 binds to the promoter region of inhibitor of nuclear factor
kappa B kinase β, thereby enhancing its transcription. The encoded
protein I-κ-B kinase 1 facilitates the phosphorylation of inhibitor
of κBα, disrupting NF-κB pathway homeostasis, leading to the
activation of NF-κB target genes and subsequent pathological
changes that drive IVDD progression.
Through systematic screening, hsa_circ_0083756 was
identified as a key regulator in NP cells (50). Experimental evidence demonstrated
its role in promoting NP cell apoptosis and IL-1β production, while
modulating ECM synthesis and degradation. Mechanistic studies
revealed that circ_0083756 functions as a molecular sponge for
miR-558, which in turn regulates triggering receptor expressed on
myeloid cells 1 (TREM1) expression. This circ_0083756/miR-558/TREM1
axis was shown to notably influence NP cell proliferation,
apoptosis, ECM homeostasis and inflammatory responses. The findings
were further validated in animal models, confirming the relevance
of the pathway in disc degeneration (50).
Inflammatory cytokines such as IL-1β and TNF-α have
been shown to upregulate circ_0005918 expression in a
time-dependent manner, contributing to IVDD. By designing a
controlled study, research findings established a link between
circ_0005918 upregulation and IVDD progression. This circRNA was
found to promote NP cell proliferation, ECM degradation and
inflammatory cytokine secretion, including IL-1β, IL-6 and TNF-α,
through the sponging of miR-622. Compared with the control group,
the degree of disc degeneration was associated with miR-622
expression levels but exhibited an inverse relationship with
circ_0005918 expression (51).
Further investigation into circ_0134111 identified a
binding site for miR-578, which was found to counteract the effects
of circ_0134111 on NP cell proliferation and inflammatory factor
expression, including IL-6, IL-8, MMP-9 and ADAMTS-5. In addition,
miR-578 reversed the circ_0134111-induced downregulation of type II
collagen and aggrecan (ACAN). These findings indicated that
circ_0134111 contributes to disc degeneration by modulating miR-578
expression; however, whether VEGF, a gene associated with miR-578,
plays a role in this pathway remains to be explored (52).
The functional role of circ-FAM169A in IVDD
progression was investigated, demonstrating its activity as a
competing endogenous RNA (ceRNA) targeting miR-583. Considering
that key regulatory genes in IVDD affect ECM composition through
SRY-box transcription factor 9 (SOX9) regulation, the study further
confirmed SOX9 as a direct target of miR-583, showing an inverse
association with IVDD severity. Pathway analysis suggested that
miR-583 potentially mediates its effects through epidermal growth
factor receptor, phosphatidylinositol 3-kinase-protein kinase B and
bone morphogenetic protein pathways, although the specific
mechanism of the circ-FAM169A-miR-583 pathway in IVDD requires
further investigation (53).
CEP, an integral component of the IVD, facilitates
nutrient diffusion, and its degradation accelerates IVDD
progression (54). The study on
intermittent cyclic mechanical tension revealed its association
with cytoskeletal morphology and ECM protein secretion in CEP cells
(55). The selection of
circ_0022382 as a research target was based on its 4-fold higher
expression in experimental groups compared with the controls.
Further functional analysis indicated that circRNA_0022382
mitigates pressure-induced degeneration in CEP chondrocytes, as
demonstrated through control group comparisons and cell staining
assays. Additional findings established an antagonistic
relationship between circ_0022382 and miR-4726-5p, confirming the
role of circ_0022382 as a miRNA sponge that regulates TGF-β3
expression. Finally, in vivo experiments using a rat model
provided evidence that the circRNA_0022382/miR-4726-5p/TGF-β3 axis
alleviates disc degeneration by enhancing ECM synthesis in CEP
chondrocytes (56).
Xiang et al (8) identified Hsa_circ_0044722 (circCIDN)
as a target gene through heat map and microarray analyses, and
showed that it had a protective effect on NP cells exposed to
stress load by transfecting them. Subsequently, it was verified
that circRNA-CIDN directly binds to miR-34a-5p through
complementary target sites, suggesting that it may act as a miRNA
sponge for miR-34a-5p in NP cells, in which sirtuin (SIRT1) is the
target mRNA of miR-34a-5p. Finally, reverse
transcription-quantitative PCR (RT-qPCR) was used to verify that
circRNA-CIDN overexpression inhibited the expression of miR-34a-5p
and weakened the inhibitory effect of miR-34a-5p on target mRNA,
thereby inhibiting the compression-induced apoptosis of NP cells
and ECM degradation to delay the process of disc degeneration.
The expression of circ-4099 in NP cells is
influenced by TNF-α in a dose- and time-dependent manner through
the MAPK and NF-κB pathways. Experimental validation indicated that
circ-4099 enhances collagen II and ACAN levels, thereby restoring
ECM synthesis in NP cells. Concurrently, investigations into
miR-616-5p revealed its opposing role in modulating inflammatory
gene expression and ECM synthesis, supporting the hypothesis that
circ-4099 functions as a competitive endogenous RNA. Further
findings demonstrated that circ-4099 exerts its regulatory effects
by counteracting miR-616-5p's suppression of SOX9, a pivotal NP
cell regulator known to mitigate IVDD by promoting matrix protein
expression and reducing inflammatory responses. This highlighted
circ-4099 as a promising research avenue and therapeutic target
(57).
Investigations into circSNHG5 identified its
downregulation in IVDD, with functional studies showing that
silencing this circRNA led to decreased chondrocyte proliferation
and increased ECM degradation. Fluorescence in situ
hybridization and luciferase reporter assays confirmed that
circSNHG5 acts as an miR-495-3p sponge, inhibiting its activity.
CBP/p300-Interacting Transactivator with Glu/Asp-rich
carboxy-terminal domain 2 (CITED2), a downstream target of
miR-495-3p, was found to play a role in IVDD attenuation by
reducing MMP13 levels while enhancing collagen II and ACAN
expression. Ultimately, findings suggested that the
circSNHG5/miR-495-3p/CITED2 axis serves as a protective mechanism
against CEP degradation (58).
CircGLCE was stably present in the cytoplasm of NP
cells, but circGLCE was found by microarray analysis and RT-qPCR to
be notably downregulated in IVDD. CircGLCE silencing led to the
increased apoptosis and ECM degradation of NP cells, which was
verified by immunofluorescence and flow cytometry, suggesting that
circGLCE exerts a regulatory effect on NP cells. Subsequently, it
was determined that circGLCE served as an miR-587 sponge in NP
cells, and signal transducing adaptor family member 1 (STAP1) was
the target mRNA of miR-587. Silencing miR-587 could reverse the
circGLCE downregulation-induced IVDD, whereas STAP1 could promote
the expression of collagen II and ACAN. Finally, animal IVDD models
were established to verify that circGLCE alleviates IVDD by
targeting miR-587/STAP1 (59).
Emerging evidence underscores the pivotal role of
circRNAs as miRNA sponges, regulating target mRNAs and influencing
IVDD pathogenesis. These insights offer valuable guidance for
exploring the molecular mechanisms of IVDD and developing novel
therapeutic strategies.
Through the study of the sponge mechanism, circRNAs
have been identified as key regulators in IVDD by acting as ceRNAs.
By sequestering specific miRNAs, circRNAs modulate the expression
of target genes, thereby influencing apoptosis, inflammation and
ECM metabolism in NP cells and chondrocytes. However, existing
studies focus on different circRNAs, each associated with distinct
signaling pathways and target molecules, highlighting the
complexity and heterogeneity of their regulatory roles in IVDD.
Given the pivotal role of circRNAs in IVDD
pathogenesis, further research is required to elucidate their
molecular mechanisms in greater depth. Expanding investigations
into the circRNA-miRNA-mRNA regulatory network, particularly in
relation to inflammation and mechanical stress pathways, is
crucial. Furthermore, exploring the upstream and downstream
regulatory mechanisms of specific circRNAs will provide deeper
insights into their functional significance in IVDD.
Due to their stability and tissue specificity,
circRNAs hold great promise as non-invasive diagnostic biomarkers
for IVDD. Future research should focus on developing targeted
therapeutic strategies, such as circRNA inhibitors or miRNA mimics,
to modulate their expression effectively. There are currently a
large number of ongoing research projects in this field, and
significant progress has been made. Furthermore, the application of
nanotechnology for the precise and stable delivery of RNA molecules
presents an innovative avenue to enhancing therapeutic
efficacy.
Collectively, the molecular sponge mechanism remains
the most extensively studied and characterized function of
circRNAs. Current evidence demonstrates that circRNA-mediated miRNA
sponging exerts regulatory effects on multiple pathological
processes in IVDD, including ECM metabolism, inflammatory factor
expression and NP cell apoptosis, with analogous regulatory
functions observed in the cartilage endplate. Nevertheless, the
majority of circRNAs remain uncharacterized, and their potential
involvement in IVDD pathogenesis, particularly through
sponge-mediated mechanisms, requires systematic investigation.
These unresolved questions represent critical directions for future
research in this field.
CircRNAs can be transported by
exosomes to influence the normal structure of IVD
Exosomes are extracellular vesicles with diameters
ranging from 40–160 nm. They encapsulate various cellular
components, including DNA, RNA, metabolic byproducts and proteins,
and facilitate intercellular communication through diverse
mechanisms, thereby triggering complex biological responses
(60). Studies have demonstrated
that circRNAs exhibit exceptional stability and are abundantly
expressed in exosomes. As a novel class of genetic information
molecules, exosomal circRNAs have gained considerable research
attention due to their intrinsic stability, high abundance and
widespread distribution. Their unique characteristics position them
as promising candidates for further investigation in intercellular
communication and disease biomarker discovery (61,62).
Extensive research has demonstrated that exosomes
play a pivotal role in disease pathogenesis and offer novel
therapeutic avenues. For instance, exosomal miRNAs are implicated
in oxidative stress and immune dysregulation, serving as potential
biomarkers for assessing vitiligo activity (63). In addition, exosomes contribute to
the progression of cancer and cardiovascular diseases (64). In the musculoskeletal system, they
have also been demonstrated to be involved in IVDD.
Chen et al (65) isolated and cultured primary human
NP cells from degenerated IVDs. RT-qPCR analysis revealed a marked
downregulation of circ_0036763 and U2 small nuclear RNA auxiliary
factor 2 (U2AF2), whereas miR-583 was upregulated. Transfection
experiments and western blotting confirmed that circ_0036763
promotes the expression of ACAN. The study further identified the
U2AF2/circ_0036763/miR-583/ACAN axis as a key regulatory pathway in
IVDD, wherein U2AF2 facilitates circ_0036763 maturation, and
circ_0036763 sponges miR-583 to enhance ACAN expression. In
addition, exosomal U2AF2 derived from bone marrow mesenchymal stem
cells was shown to mitigate IVDD, highlighting the therapeutic
potential of exosome-mediated RNA delivery.
Investigations have demonstrated notable findings
through comparative analysis of exosomes derived from normal and
degenerative disc cells, employing electron microscopy and western
blotting for morphological characterization and molecular marker
identification. Through comprehensive circRNA microarray profiling
and subsequent PCR validation, circRNA_0000253 was identified as
markedly upregulated in degenerative disc cells, and computational
bioinformatics analysis established miR-141-5p as a putative target
of circRNA_0000253. Mechanistic studies revealed that
circRNA_0000253 regulates cellular proliferation and apoptotic
processes through the modulation of SIRT1 and ECM-associated
components, including collagen II and ACAN. In vivo
validation using a rat IVDD model, incorporating MRI, X-ray imaging
and histological staining techniques, provided compelling evidence
that circRNA_0000253 promotes disc degeneration through its
miR-141-5p sponge function and subsequent downregulation of SIRT1
(66).
These findings suggest that the exosome-mediated
delivery of specific circRNAs holds promise as a targeted therapy
for IVDD. However, challenges remain in translating laboratory
findings into clinical applications. Of note, studies lack in-depth
analysis of the stability and delivery efficiency of exosomes in
vivo. In addition, the limited sample sizes may affect the
generalizability of their conclusions, underscoring the need for
further research (Table I).
 | Table I.CircRNAs act as miRNA sponges in
IVDD. |
Table I.
CircRNAs act as miRNA sponges in
IVDD.
| CircRNA | Pathway | Function | Expression | (Refs.) |
|---|
| EYA3 |
circEYA3/miR-196a-5p/EBF1/IKKβ | ECM degradation in
NP cells↑ | Upregulated | (49) |
|
|
| Secretion of
inflammatory cytokines↑ |
|
|
|
|
| Apoptosis in NP
cells↑ |
|
|
| 0083756 |
circ_0083756/miR-558/TREM1 | NP cells
proliferation↓ | Upregulated | (50) |
|
|
| Secretion of
inflammatory cytokines↑ |
|
|
|
|
| ECM degradation in
NP cells↑ |
|
|
| 0005918 |
circ_005918/miR-622 | NP cells
growth↑ | Upregulated | (51) |
|
|
| ECM degradation in
NP cells↑ |
|
|
|
|
| Secretion of
inflammatory cytokines↑ |
|
|
| 0134111 |
circ_0134111/miR-578 | NP cells
proliferation↑ | Upregulated | (52) |
|
|
| Pro-inflammatory
cytokines secretion↑ |
|
|
|
|
| ECM degradation in
NP cells↑ |
|
|
| FAM169A |
circ-FAM169A-miR-583 | ECM synthesis in NP
cells↑ | Upregulated | (53) |
|
|
| MMP
expression↓ |
|
|
| 0022382 |
circ_0022382/miR-4726-5p/TGF-β3 | Degeneration of
chondrocytes↓ | Downregulated | (56) |
|
|
| Endplate
chondrocyte ECM synthesis↑ |
|
|
| CIDN (0044722) |
circ-CIDN/miR-34a-5p/SIRT1 | Compression-induced
NP cells apoptosis↓ | Downregulated | (8) |
|
|
| ECM degradation in
NP cells↓ |
|
|
| 4099 |
circ-4099/miR-616-5p/SOX9 | ECM degradation in
NP cells↓ | Upregulated | (57) |
|
|
| Inflammation↓ |
|
|
| SNHG5
(0077254) |
circSNHG5/miR-495-3p/CITED2 | Chondrocytes
proliferation↑ | Downregulated | (58) |
|
|
| ECM degradation in
chondrocytes↓ |
|
|
| GLCE |
circGLCE/miR-587/STAP1 | Apoptosis in NP
cells↓ | Downregulated | (59) |
|
|
| ECM degradation in
NP cells↓ |
|
|
CircRNAs interact with proteins
As discussed earlier, circRNAs regulate DD primarily
by acting as ceRNAs to sponge miRNAs; however, studies on the
direct interaction between circRNAs and proteins in IVDD remain
limited. This section provides an overview of the mechanisms
through which circRNAs interact with proteins and their potential
implications in IVDD.
CircRNAs can function in multiple ways: As protein
sponges, altering the physiological functions of proteins; as
protein scaffolds, facilitating interactions between proteins; or
as molecular recruiters, directing proteins to specific cellular
locations (67). These
interactions play a critical role in regulating cellular processes
such as proliferation, apoptosis, angiogenesis, mRNA translation,
energy metabolism and differentiation (68). Of note, RBPs, which recognize and
bind RNA through specific structural motifs, form intricate
regulatory networks with circRNAs (69). RBPs orchestrate various aspects of
circRNA biology, including biogenesis, degradation,
nucleocytoplasmic transport and translational regulation, thereby
fine-tuning circRNA function (70). In the context of IVDD, emerging
evidence underscores the significance of circRNA-protein
interactions in disease pathogenesis, particularly through their
modulation of specific cellular processes.
CircFUNDC1 ameliorates IVDD by promoting
PTEN-induced kinase 1 (PINK1)-dependent mitophagy in NPCs under
oxidative stress. It executes this function not as a miRNA sponge,
but by directly interacting with and stabilizing the cyclin
dependent kinase (CDK)9 protein. This interaction enhances the
phosphorylation of RNA polymerase II, thereby facilitating the
transcription of the key mitophagy gene PINK1. This mechanism
establishes a novel paradigm wherein a circRNA directly regulates a
kinase to control transcriptional activation in disc homeostasis
(71).
The reduced m6A methylation and elevated expression
of circGPATCH2L in degenerated human IVD tissues have garnered
notable attention from researchers. Overexpression and knockdown
experiments revealed that circGPATCH2L promotes the degeneration of
NPCs by inducing apoptosis. Furthermore, circGPATCH2L interacts
with tripartite motif containing 28 to inhibit its phosphorylation,
resulting in P53 accumulation and DNA damage. Furthermore, it was
found that circGPATCH2L is recognized and degraded by the YTH
N6-methyladenosine RNA binding protein F2 (YTHDF2)-ribosomal
protein L10 (RPL10)-ribonuclease P/RNases P/mitochondrial RNA
processing ribonuclease complex, which helps maintain homeostasis
in normal NPCs. Finally, a mouse model study confirmed that the
knockdown of circGPATCH2L can alleviate IVDD. However, the small
sample size of patients in the study (4 patients in the
degenerative group and 4 in the control group) may affect its
statistical power. The dynamic regulation of m6A modification has
not been deeply discussed in this study. The potential interactions
of circGPATCH2L with other DNA damage-related proteins have also
not been fully analyzed (9).
Another study identified circATXN1 as notably
upregulated in NPCs from aged individuals (>70 years old) of
degenerated intervertebral discs. To investigate its role,
researchers constructed a circATXN1 overexpression vector and
confirmed its activation of aging markers CDK inhibitor 2A,
specifically the P16 isoform and P21. Further analysis revealed
that circATXN1 downregulated collagen type II α1 chain and SOX9,
impacting ECM homeostasis and mitochondrial function. Using RNA
immunoprecipitation (RIP), RNA affinity purification and mass
spectrometry, heterogeneous nuclear ribonucleoprotein A2/B1
(HNRNPA2B1) was identified as a direct binding partner of
circATXN1. HNRNPA2B1 facilitates the biogenesis of circATXN1 by
regulating the splicing of its precursor transcript, while
concurrently reducing ATXN1 mRNA levels. In a therapeutic approach,
researchers developed a small interfering RNA-embedded DNA
tetrahedral nanogel to efficiently silence circATXN1, leading to
improvements in tissue structure and ECM content in an aged mouse
IVDD model. However, while the interaction between circATXN1 and
HNRNPA2B1 has been characterized, the precise mechanism through
which it regulates protein mislocalization remains unclear and
warrants further investigation (72).
In the musculoskeletal system, Chen et al
(73) revealed that the
overexpression of circTMEFF1 aggravated muscle atrophy, while the
inhibition of its expression alleviated the atrophy. circTMEFF1
mainly promotes muscular atrophy through two mechanisms; the first
is by binding to TAR DNA-binding protein 43 protein, inducing
mitochondrial DNA release and activating cyclic GMP-AMP
synthase-stimulator of interferon gene signaling pathway, thus
promoting atrophy. The other mechanism is by encoding a new
protein, TMEFF1-339aa, which has a pro-atrophy effect. It was
revealed that this works through a dual mechanism: Binding proteins
and translation proteins. In addition, circ-Foxo3 blocks the cell
cycle by binding to CDK2 and cyclin dependent kinase inhibitor 1 to
form a ternary complex (74). In
oncology research, circZKSCAN1 has been found to suppress cancer
stemness by acting as an RBP sponge, modulating the function of
fragile X mental retardation protein. The characterization of
circZKSCAN1 provided a new perspective on the role of circRNAs in
tumor biology (75).
Although the above studies have indicated that
circRNAs interact with proteins to influence IVDD, the precise
molecular mechanisms remain largely unexplored. This knowledge gap
may be attributed to the complexity of protein-circRNA
interactions, the technical challenges of in vitro and in
vivo studies, and the early stage of research in this area. In
addition, the experimental validation of circRNA-protein
interactions is hindered by notable technical challenges and high
costs. Furthermore, the limited availability of specialized
databases and analytical tools for circRNA-protein interactions
poses a substantial barrier to advancing research in this field.
These objective factors collectively impede the progress of related
experimental studies. Most of the existing research on
circRNA-protein interactions focuses on cancer, while their role in
musculoskeletal disorders, including IVDD, requires further
investigation.
CircRNA may influence IVDD through its
translation function
To date, there has been no direct evidence linking
the protein-coding function of circRNAs to IVDD. However, the
emerging mechanisms through which circRNAs encode proteins are
noteworthy and may represent a promising new research direction in
understanding the pathophysiology of disc degeneration.
Initially regarded as mere splicing byproducts,
circRNAs have been increasingly recognized for their diverse
physiological functions (29);
among them, their capacity to encode proteins has garnered marked
attention (76). CircRNAs contain
≥1 open reading frames (ORFs), which, due to their circular
structure, can facilitate the translation of short peptides or even
proteins >100 amino acids in length (77).
This translational mechanism has been explored in
the musculoskeletal system. For example, Yin et al (78) identified an ORF in circFAM188B and
demonstrated its ability to encode a novel protein,
circFam188B-103AA, which regulates the proliferation and
differentiation of chicken skeletal muscle satellite cells. Legnini
et al (79) discovered that
circ-ZNF609, containing an ORF, plays a role in myoblast
proliferation and protein translation. Despite these advances, the
molecular activities of such proteins and their interactions with
linear mRNA counterparts remain poorly understood. Similarly, Zhu
et al (80) identified a
circRNA, circDdb1, that promotes muscle atrophy; their pivotal
finding was that circDdb1 is translated into a novel protein,
circDdb1-867aa. This protein executes the function by entering the
nucleus to inhibit Pax7, a master transcription factor for muscle
regeneration. This work crucially establishes a paradigm where a
coding circRNA directly disrupts myogenic transcription.
While current research on circRNA-encoded proteins
primarily focuses on cancer and tumor biology, this field is still
in its infancy (30,81,82).
Numerous circRNAs with protein-coding potential remain
undiscovered, and the underlying translation mechanisms require
further exploration (83). To
date, no studies have directly implicated circRNA-mediated protein
translation in IVDD; this gap may be attributed to the rarity of
circRNAs with functional ORFs, the small molecular weight of the
encoded proteins and the technical challenges associated with their
detection. Nevertheless, given the established role of circRNAs in
the musculoskeletal system, it is plausible that they may also
influence disc degeneration, although this hypothesis awaits
experimental validation.
Potential clinical application
Currently, the management of IVDD relies on two
traditional approaches: Non-surgical conservative treatment and
surgical intervention (84).
However, neither strategy can fully restore degenerated disc tissue
or reverse the pathological progression of IVDD (85). The development of biomaterials has
opened new avenues for addressing these limitations. Researchers
have increasingly focused on leveraging biomaterials to mitigate
oxidative stress, reduce inflammatory factors, regulate ECM
homeostasis and modulate cellular functions in animal models;
however, evidence from human studies remains limited (23). In the following section,
advancements in biomaterial-based therapies for IVDD are
summarized, their mechanisms of action were elucidated and future
research directions and therapeutic strategies were
highlighted.
Gao et al (86) developed an injectable
kaempferol-fibrin glue, combining kaempferol with fibrinogen to
create a biomaterial with low immunogenicity and high stability.
Animal studies demonstrated its efficacy in promoting NP cell
proliferation and reducing cellular inflammation. Lu et al
(87) investigated Physalin A, a
natural bioactive withanolide extracted from Physalis
alkekengi var, which enhances NP cell autophagy by inhibiting
the PI3K/AKT/mTOR pathway and mitigates tissue fibrosis by
suppressing the SMAD2/3 pathway. Chen et al (88) designed a zinc-oxidized sodium
alginate-gelatin (ZOG) hydrogel loaded with antagomir-204-3p (AM).
This composite material leverages the anti-apoptotic properties of
AM, the antibacterial effects of zinc ions and the mechanical
strength of ZOG to restore disc height and correct ECM metabolic
imbalances. Wang et al (89) developed oxymatrine-loaded liposomes
(OMT-LIP), where oxymatrine exerts anti-fibrotic and
anti-inflammatory effects, and liposomes enable sustained drug
release. Animal experiments confirmed that OMT-LIP reduces ECM
degradation in NP cells and suppresses the expression of MMP-319
and IL-6, thereby alleviating IVDD. Zhang et al (90) constructed MnO2-based
nanoassemblies that decompose H2O2, alleviate
oxidative stress and improve the hypoxic and acidic
microenvironment of NPCs, while also enabling controlled drug
release to slow IVDD progression.
In summary, emerging biomaterials, including
nanomaterials, inorganic compounds and hydrogels, have shown
promise in mitigating IVDD. Antioxidant and anti-inflammatory
agents play a pivotal role in preserving ECM integrity and NP cell
function; however, the specific molecular pathways through which
these materials exert their effects remain incompletely understood.
Furthermore, it is unclear whether combining multiple materials and
drugs to target different pathways could yield superior therapeutic
outcomes compared with single-pathway interventions. These
questions represent critical areas for future research and
exploration. At the same time, there is a scarcity of studies on
the integration of circRNA with biomaterials, particularly for IVDD
therapy. This limited research can be attributed to several key
factors. First, the gene regulatory mechanisms of circRNA in IVDD
remain unclear, complicating its therapeutic application. Secondly,
developing biomaterials for circRNA delivery requires balancing
biocompatibility, stability and targeting, which poses notable
technical challenges. Despite these challenges, biomaterial
research remains one of the fields most closely tied to clinical
applications. The combination of circRNA with advanced biomaterials
continues to hold great promise, making it a potential research
hotspot for future IVDD therapies.
Conclusions and future prospects
The present review provided a comprehensive overview
of the historical context, biogenesis and functional roles of
circRNAs, with a particular focus on their involvement in IVDD. The
current understanding of circRNA-mediated mechanisms in IVDD was
elucidated, highlighting their regulatory roles as miRNA sponges,
their interactions with proteins and their potential involvement in
exosome-mediated signaling pathways. These findings underscore the
critical role of circRNAs in modulating key cellular processes,
including apoptosis, inflammation and ECM metabolism, which are
central to the pathogenesis of IVDD.
The predominant mechanism through which circRNAs
influence IVDD is through their function as ceRNAs, where they
sequester specific miRNAs to regulate the expression of target
genes. This miRNA sponge mechanism has been extensively studied,
with several circRNAs identified as either promoters or protectors
of disc degeneration. For instance, circRNAs such as circEYA3 and
circ_0083756 have been shown to exacerbate IVDD by promoting ECM
degradation and inflammatory responses, while others, such as
circRNA-CIDN and circGLCE exhibit protective effects by mitigating
apoptosis and ECM disruption. However, current research on circRNAs
in IVDD exhibits several methodological limitations that warrant
critical appraisal. The field is heavily reliant on in vitro
models utilizing cytokine-induced NP cells, an approach that fails
to recapitulate the complex pathophysiology of the human disc
environment. Consequently, findings from these simplified systems
may lack in vivo relevance. Furthermore, the mechanistic
exploration is predominantly confined to the competitive ceRNA
network paradigm. While valuable, this focus often overlooks
alternative functions of circRNAs, such as interactions with RBPs
or protein translation, and is frequently unsupported by robust
in vivo functional validation. Finally, clinical translation
is hampered by studies with limited human sample sizes and the
frequent use of suboptimal control tissues. Future investigations
should prioritize the use of sophisticated in vivo models,
larger and well-defined clinical cohorts and more specific genetic
tools to unequivocally establish causal relationships and explore
the full mechanistic spectrum of circRNAs in IVDD.
Furthermore, the role of circRNAs in protein
interactions and their potential to encode functional peptides
remains an underexplored area. While preliminary evidence suggests
that circRNAs can interact with RBPs and even encode small
peptides, the implications of these interactions in IVDD are yet to
be fully elucidated. The development of advanced techniques, such
as RNA RIP and mass spectrometry, will be crucial in uncovering the
molecular mechanisms underlying circRNA-protein interactions. In
addition, the exploration of circRNA-encoded peptides and their
functional roles in disc degeneration represents a promising avenue
for future research, potentially opening new therapeutic
avenues.
Another critical area for future investigation is
the application of circRNAs as non-invasive diagnostic biomarkers
for IVDD. Given their stability and tissue-specific expression
patterns, circRNAs hold notable promise for early detection and
monitoring of disc degeneration. Furthermore, the development of
targeted therapeutic strategies, such as circRNA inhibitors or
miRNA mimics, could offer novel approaches to modulate circRNA
expression and function. The integration of nanotechnology for the
precise delivery of RNA-based therapeutics may further enhance the
efficacy of these interventions.
In conclusion, while marked progress has been made
in understanding the role of circRNAs in IVDD, several knowledge
gaps remain. Current research predominantly focuses on the
mechanism of circRNA as miRNA sponges, while investigations into
circRNA-protein interactions and the translation potential of
circRNAs remain in their nascent stages. Furthermore, research
exploring the integration of circRNAs with biomaterials is
particularly limited. These underexplored areas represent critical
directions for future research breakthroughs. In the present
review, emerging trends were highlight and promising research
directions were outlined, which may help bridge the current
knowledge gap regarding the role of circRNAs in IVDD and facilitate
the development of improved therapeutic strategies to alleviate the
associated socioeconomic burden. By addressing these challenges,
new innovative diagnostic tools and therapeutic strategies can be
developed, ultimately improving the management of IVDD and
enhancing patient outcomes.
Acknowledgements
Not applicable.
Funding
The present review was funded by the National Natural Science
Foundation of China (grant no. 82202753) and the Shandong
Provincial Natural Science Foundation (grant no. ZR2022QH282).
Availability of data and materials
Not applicable.
Authors' contributions
XFD conceived the study. LYW and KY designed the
study and wrote the manuscript. HJZ, DCW and FW contributed to the
critical revision of the manuscript for important intellectual
content. All authors contributed to perform the literature search.
All authors read and approved the final manuscript. Data
authentication is not applicable.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Knezevic NN, Candido KD, Vlaeyen JWS, Van
Zundert J and Cohen SP: Low back pain. Lancet. 398:78–92. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Risbud MV and Shapiro IM: Role of
cytokines in intervertebral disc degeneration: Pain and disc
content. Nat Rev Rheumatol. 10:44–56. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dieleman JL, Cao J, Chapin A, Chen C, Li
Z, Liu A, Horst C, Kaldjian A, Matyasz T, Scott KW, et al: US
health care spending by payer and health condition, 1996–2016.
JAMA. 323:863–884. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Jöud A, Petersson IF and Englund M: Low
back pain: Epidemiology of consultations. Arthritis Care Res
(Hoboken). 64:1084–1088. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Xu D, Ma X, Sun C, Han J, Zhou C, Wong SH,
Chan MTV and Wu WKK: Circular RNAs in intervertebral disc
degeneration: An updated review. Front Mol Biosci. 8:7814242022.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Roberts S, Evans H, Trivedi J and Menage
J: Histology and pathology of the human intervertebral disc. J Bone
Joint Surg Am. 88 (Suppl 2):S10–S14. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xie G, Wu T, Ji G, Wu H, Lai Y, Wei B and
Huang W: Circular RNA and intervertebral disc degeneration:
Unravelling mechanisms and implications. Front Mol Biosci.
10:13020172023. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Xiang Q, Kang L, Wang J, Liao Z, Song Y,
Zhao K, Wang K, Yang C and Zhang Y: CircRNA-CIDN mitigated
compression loading-induced damage in human nucleus pulposus cells
via miR-34a-5p/SIRT1 axis. EBioMedicine. 53:1026792020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen Z, Song J, Xie L, Xu G, Zheng C, Xia
X, Lu F, Ma X, Zou F, Jiang J and Wang H: N6-methyladenosine
hypomethylation of circGPATCH2L regulates DNA damage and apoptosis
through TRIM28 in intervertebral disc degeneration. Cell Death
Differ. 30:1957–1972. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Jeck WR and Sharpless NE: Detecting and
characterizing circular RNAs. Nat Biotechnol. 32:453–461. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Leão Monteiro R: Future of low back pain:
Unravelling IVD components and MSCs' potential. Cell Regen.
13:12024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhou D, Liu H, Zheng Z and Wu D: Design
principles in mechanically adaptable biomaterials for repairing
annulus fibrosus rupture: A review. Bioact Mater. 31:422–439.
2023.PubMed/NCBI
|
|
13
|
Zehra U, Tryfonidou M, Iatridis JC,
Illien-Jünger S, Mwale F and Samartzis D: Mechanisms and clinical
implications of intervertebral disc calcification. Nat Rev
Rheumatol. 18:352–362. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang A, Cheng Z, Chen Y, Shi P, Gan W and
Zhang Y: Emerging tissue engineering strategies for annulus
fibrosus therapy. Acta Biomater. 167:1–15. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Mohd Isa IL, Teoh SL, Mohd Nor NH and
Mokhtar SA: Discogenic low back pain: Anatomy, pathophysiology and
treatments of intervertebral disc degeneration. Int J Mol Sci.
24:2082022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kirnaz S, Capadona C, Wong T, Goldberg JL,
Medary B, Sommer F, McGrath LB Jr and Härtl R: Fundamentals of
intervertebral disc degeneration. World Neurosurg. 157:264–273.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Ma Z, Liu X, Zhang M, Wu Z, Zhang X, Li S,
Li S, An J and Luo Z: Research progress on the role of cartilage
endplate in intervertebral disc degeneration. Cell Biochem Funct.
42:e41182024. View
Article : Google Scholar : PubMed/NCBI
|
|
18
|
Guo W, Li BL, Zhao JY, Li XM and Wang LF:
Causal associations between modifiable risk factors and
intervertebral disc degeneration. Spine J. 24:195–209. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xia Q, Zhao Y, Dong H, Mao Q, Zhu L, Xia
J, Weng Z, Liao W, Hu Z, Yi J, et al: Progress in the study of
molecular mechanisms of intervertebral disc degeneration. Biomed
Pharmacother. 174:1165932024. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Adams MA and Roughley PJ: What is
intervertebral disc degeneration, and what causes it? Spine (Phila
Pa 1976). 31:2151–2161. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Roh EJ, Darai A, Kyung JW, Choi H, Kwon
SY, Bhujel B, Kim KT and Han I: Genetic therapy for intervertebral
disc degeneration. Int J Mol Sci. 22:15792021. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Clouet J, Vinatier C, Merceron C,
Pot-Vaucel M, Hamel O, Weiss P, Grimandi G and Guicheux J: The
intervertebral disc: From pathophysiology to tissue engineering.
Joint Bone Spine. 76:614–618. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Mohd Isa IL, Mokhtar SA, Abbah SA, Fauzi
MB, Devitt A and Pandit A: Intervertebral disc degeneration:
Biomaterials and tissue engineering strategies toward precision
medicine. Adv Healthc Mater. 11:e21025302022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang J, Markova D, Anderson DG, Zheng Z,
Shapiro IM and Risbud MV: TNF-α and IL-1β promote a
disintegrin-like and metalloprotease with thrombospondin type I
motif-5-mediated aggrecan degradation through syndecan-4 in
intervertebral disc. J Biol Chem. 286:39738–39749. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang F, Cai F, Shi R, Wang XH and Wu XT:
Aging and age related stresses: A senescence mechanism of
intervertebral disc degeneration. Osteoarthritis Cartilage.
24:398–408. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhu J, Zhang X, Gao W, Hu H, Wang X and
Hao D: lncRNA/circRNA-miRNA-mRNA ceRNA network in lumbar
intervertebral disc degeneration. Mol Med Rep. 20:3160–3174.
2019.PubMed/NCBI
|
|
27
|
Yin X, Lin H, Lin L, Miao L, He J and Zhuo
Z: LncRNAs and CircRNAs in cancer. MedComm (2020). 3:e1412022.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sanger HL, Klotz G, Riesner D, Gross HJ
and Kleinschmidt AK: Viroids are single-stranded covalently closed
circular RNA molecules existing as highly base-paired rod-like
structures. Proc Natl Acad Sci U S A. 73:3852–3856. 1976.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhao X, Zhong Y, Wang X, Shen J and An W:
Advances in circular RNA and its applications. Int J Med Sci.
19:975–985. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Conn VM, Chinnaiyan AM and Conn SJ:
Circular RNA in cancer. Nat Rev Cancer. 24:597–613. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Niu D, Wu Y and Lian J: Circular RNA
vaccine in disease prevention and treatment. Signal Transduct
Target Ther. 8:3412023. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Yang Y, Yujiao W, Fang W, Linhui Y, Ziqi
G, Zhichen W, Zirui W and Shengwang W: The roles of miRNA, lncRNA
and circRNA in the development of osteoporosis. Biol Res.
53:402020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Li Q, Ren X, Wang Y and Xin X: CircRNA: A
rising star in leukemia. PeerJ. 11:e155772023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kelly S, Greenman C, Cook PR and
Papantonis A: Exon skipping is correlated with exon
circularization. J Mol Biol. 427:2414–2417. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Kristensen LS, Andersen MS, Stagsted LVW,
Ebbesen KK, Hansen TB and Kjems J: The biogenesis, biology and
characterization of circular RNAs. Nat Rev Genet. 20:675–691. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wang M, Yu F, Wu W, Zhang Y, Chang W,
Ponnusamy M, Wang K and Li P: Circular RNAs: A novel type of
non-coding RNA and their potential implications in antiviral
immunity. Int J Biol Sci. 13:1497–1506. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Chen L, Wang C, Sun H, Wang J, Liang Y,
Wang Y and Wong G: The bioinformatics toolbox for circRNA discovery
and analysis. Brief Bioinform. 22:1706–1728. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Di Agostino S, Riccioli A, De Cesaris P,
Fontemaggi G, Blandino G, Filippini A and Fazi F: Circular RNAs in
embryogenesis and cell differentiation with a focus on cancer
development. Front Cell Dev Biol. 8:3892020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hsiao KY, Lin YC, Gupta SK, Chang N, Yen
L, Sun HS and Tsai SJ: Noncoding effects of circular RNA CCDC66
promote colon cancer growth and metastasis. Cancer Res.
77:2339–2350. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Altesha MA, Ni T, Khan A, Liu K and Zheng
X: Circular RNA in cardiovascular disease. J Cell Physiol.
234:5588–5600. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Wu W, Ji P and Zhao F: CircAtlas: An
integrated resource of one million highly accurate circular RNAs
from 1070 vertebrate transcriptomes. Genome Boil. 21:1012020.
View Article : Google Scholar
|
|
42
|
Nguyen DT: An integrative pipeline for
circular RNA quantitative trait locus discovery with application in
human T cells. Bioinformatics. 39:btad6672023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Wang Q, Yang D, Zuo Y, Wang D and Li W:
Emerging roles of circular RNAs in tuberculosis. Front Immunol.
13:9957012022. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Fischer JW and Leung AKL: CircRNAs: A
regulator of cellular stress. Crit Rev Biochem Mol Biol.
52:220–233. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Xu F, Xiao Q, Du WW, Wang S and Yang BB:
CircRNA: Functions, applications and prospects. Biomolecules.
14:15032024. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Hausser J and Zavolan M: Identification
and consequences of miRNA-target interactions-beyond repression of
gene expression. Nat Rev Genet. 15:599–612. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Liu Y, Xue M, Du S, Feng W, Zhang K, Zhang
L, Liu H, Jia G, Wu L, Hu X, et al: Competitive endogenous RNA is
an intrinsic component of EMT regulatory circuits and modulates
EMT. Nat Commun. 10:16372019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Militello G, Weirick T, John D, Döring C,
Dimmeler S and Uchida S: Screening and validation of lncRNAs and
circRNAs as miRNA sponges. Brief Bioinform. 18:780–788.
2017.PubMed/NCBI
|
|
49
|
Wang T, Yan X, Song D, Li Y, Li Z and Feng
D: CircEYA3 aggravates intervertebral disc degeneration through the
miR-196a-5p/EBF1 axis and NF-κB signaling. Commun Boil. 7:3902024.
View Article : Google Scholar
|
|
50
|
Du X, Chen S, Cui H, Huang Y, Wang J, Liu
H, Li Z, Liang C, Zheng Z and Wang H: Circular RNA hsa_circ_0083756
promotes intervertebral disc degeneration by sponging miR-558 and
regulating TREM1 expression. Cell Prolif. 55:e132052022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Cui Y, Zhao X and Wu Y: Circ_0005918
sponges miR-622 to aggravate intervertebral disc degeneration.
Front Cell Dev Biol. 10:9052132022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Yan P, Sun C, Luan L, Han J, Qu Y, Zhou C
and Xu D: Hsa_circ_0134111 promotes intervertebral disc
degeneration via sponging miR-578. Cell Death Discov. 8:552022.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Li Y, Pan D, Liu S, Xing X, Zhou H, Zhang
B, Zhang D, Li B, Li G, Tao B, et al: Identification of
circ-FAM169A sponges miR-583 involved in the regulation of
intervertebral disc degeneration. J Orthop Translat. 26:121–131.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Urban JP, Smith S and Fairbank JCT:
Nutrition of the intervertebral disc. Spine (Phila Pa 1976).
29:2700–2709. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Feng C, Liu M, Fan X, Yang M, Liu H and
Zhou Y: Intermittent cyclic mechanical tension altered the microRNA
expression profile of human cartilage endplate chondrocytes. Mol
Med Rep. 17:5238–5246. 2018.PubMed/NCBI
|
|
56
|
Hu B, Xiao L, Wang C, Liu C, Zhang Y, Ding
B, Gao D, Lu Y and Xu H: Circ_0022382 ameliorated intervertebral
disc degeneration by regulating TGF-β3 expression through sponge
adsorption of miR-4726-5p. Bone. 154:1161852022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Wang H, He P, Pan H, Long J, Wang J, Li Z,
Liu H, Jiang W and Zheng Z: Circular RNA circ-4099 is induced by
TNF-α and regulates ECM synthesis by blocking miR-616-5p inhibition
of Sox9 in intervertebral disc degeneration. Exp Mol Med. 50:1–14.
2018. View Article : Google Scholar
|
|
58
|
Zhang J, Hu S, Ding R, Yuan J, Jia J, Wu T
and Cheng X: CircSNHG5 sponges Mir-495-3p and modulates CITED2 to
protect cartilage endplate from degradation. Front Cell Dev Biol.
9:6687152021. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Chen Z, Zhang W, Deng M, Li Y and Zhou Y:
CircGLCE alleviates intervertebral disc degeneration by regulating
apoptosis and matrix degradation through the targeting of
miR-587/STAP1. Aging (Albany NY). 12:21971–21991. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Kalluri R and LeBleu VS: The biology,
function, and biomedical applications of exosomes. Science.
367:eaau69772020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang F, Jiang J, Qian H, Yan Y and Xu W:
Exosomal circRNA: Emerging insights into cancer progression and
clinical application potential. J Hematol Oncol. 16:672023.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Xue Q, Huang Y, Chang J, Cheng C, Wang Y,
Wang X and Miao C: CircRNA-mediated ceRNA mechanism in
osteoarthritis: Special emphasis on circRNAs in exosomes and the
crosstalk of circRNAs and RNA methylation. Biochem Pharmacol.
212:1155802023. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Li W, Pang Y, He Q, Song Z, Xie X, Zeng J
and Guo J: Exosome-derived microRNAs: Emerging players in vitiligo.
Front Immunol. 15:14196602024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
He C, Zheng S, Luo Y and Wang B: Exosome
theranostics: Biology and translational medicine. Theranostics.
8:237–255. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Chen X, Cai D, Li H, Wei Q, Li X, Han Z,
Liang J, Xie J, Ruan J, Liu J, et al: Exosomal U2AF2 derived from
human bone marrow mesenchymal stem cells attenuates the
intervertebral disc degeneration through circ_0036763/miR-583/ACAN
axis. Regen Ther. 25:344–354. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Song J, Chen ZH, Zheng CJ, Song KH, Xu GY,
Xu S, Zou F, Ma XS, Wang HL and Jiang JY: Exosome-transported
circRNA_0000253 competitively adsorbs MicroRNA-141-5p and increases
IDD. Mol Ther Nucleic Acids. 21:1087–1099. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Huang A, Zheng H, Wu Z, Chen M and Huang
Y: Circular RNA-protein interactions: Functions, mechanisms, and
identification. Theranostics. 10:3503–3517. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Das A, Sinha T, Shyamal S and Panda AC:
Emerging role of circular RNA-protein interactions. Noncoding RNA.
7:482021.PubMed/NCBI
|
|
69
|
Shaath H, Vishnubalaji R, Elango R,
Kardousha A, Islam Z, Qureshi R, Alam T, Kolatkar PR and Alajez NM:
Long non-coding RNA and RNA-binding protein interactions in cancer:
Experimental and machine learning approaches. Semin Cancer Biol.
86:325–345. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zheng S, Zhang X, Odame E, Xu X, Chen Y,
Ye J, Zhou H, Dai D, Kyei B, Zhan S, et al: CircRNA-protein
interactions in muscle development and diseases. Int J Mol Sci.
22:32622021. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Gu T, He Y, Zhou J, Qiu X, Yang W, Zhu Q,
Liang Y, Zheng Y, Yik JHN, Haudenschild DR, et al: CircFUNDC1
interacts with CDK9 to promote mitophagy in nucleus pulposus cells
under oxidative stress and ameliorates intervertebral disc
degeneration. Cell Death Dis. 16:942025. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Yu C, Zhao J, Cheng F, Chen J, Chen J, Xu
H, Shi K, Xia K, Ding S, Wang K, et al: Silencing circATXN1 in
aging nucleus pulposus cell alleviates intervertebral disc
degeneration via correcting progerin mislocalization. Research
(Wash D C). 7:03362024.PubMed/NCBI
|
|
73
|
Chen R, Yang T, Jin B, Xu W, Yan Y, Wood
N, Lehmann HI, Wang S, Zhu X, Yuan W, et al: CircTmeff1 promotes
muscle atrophy by interacting with TDP-43 and encoding A novel
TMEFF1-339aa protein. Adv Sci (Weinh). 10:e22067322023. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Du WW, Yang W, Liu E, Yang Z, Dhaliwal P
and Yang BB: Foxo3 circular RNA retards cell cycle progression via
forming ternary complexes with p21 and CDK2. Nucleic Acids Res.
44:2846–2858. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhu YJ, Zheng B, Luo GJ, Ma XK, Lu XY, Lin
XM, Yang S, Zhao Q, Wu T, Li ZX, et al: Circular RNAs negatively
regulate cancer stem cells by physically binding FMRP against CCAR1
complex in hepatocellular carcinoma. Theranostics. 9:3526–3540.
2019. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Cheng J, Li G, Wang W, Stovall DB, Sui G
and Li D: Circular RNAs with protein-coding ability in oncogenesis.
Biochim Biophys Acta Rev Cancer. 1878:1889092023. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Wu P, Mo Y, Peng M, Tang T, Zhong Y, Deng
X, Xiong F, Guo C, Wu X, Li Y, et al: Emerging role of
tumor-related functional peptides encoded by lncRNA and circRNA.
Mol Cancer. 19:222020. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Yin H, Shen X, Zhao J, Cao X, He H, Han S,
Chen Y, Cui C, Wei Y, Wang Y, et al: Circular RNA CircFAM188B
encodes a protein that regulates proliferation and differentiation
of chicken skeletal muscle satellite cells. Front Cell Dev Biol.
8:5225882020. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Legnini I, Di Timoteo G, Rossi F, Morlando
M, Briganti F, Sthandier O, Fatica A, Santini T, Andronache A, Wade
M, et al: Circ-ZNF609 Is a circular RNA that can be translated and
functions in myogenesis. Mol Cell. 66:22–37.e9. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Zhu X, Yang T, Zheng Y, Nie Q, Chen J, Li
Q, Ren X, Yin X, Wang S, Yan Y, et al: EIF4A3-induced circular RNA
CircDdb1 promotes muscle atrophy through encoding a novel protein
CircDdb1-867aa. Adv Sci (Weinh). 11:e24069862024. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Chen S, Cao X, Zhang J, Wu W, Zhang B and
Zhao F: circVAMP3 drives CAPRIN1 phase separation and inhibits
hepatocellular carcinoma by suppressing c-Myc translation. Adv Sci
(Weinh). 9:e21038172022. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Margvelani G, Maquera KAA, Welden JR,
Rodgers DW and Stamm S: Translation of circular RNAs. Nucleic Acids
Res. 53:gkae11672025. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Yi Q, Feng J, Lan W, Shi H and Sun W and
Sun W: CircRNA and lncRNA-encoded peptide in diseases, an update
review. Mol Cancer. 23:2142024. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Kim MW, Kang CN and Choi SH: Update of the
natural history, pathophysiology, and treatment strategies of
degenerative cervical myelopathy: A narrative review. Asian Spine
J. 17:213–221. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Chen X, Zhang A, Zhao K, Gao H, Shi P,
Chen Y, Cheng Z, Zhou W and Zhang Y: The role of oxidative stress
in intervertebral disc degeneration: Mechanisms and therapeutic
implications. Ageing Res Rev. 98:1023232024. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Gao W, Bao J, Zhang Y, He D, Zhang L,
Zhang J, Pan H and Wang D: Injectable kaempferol-loaded fibrin glue
regulates the metabolic balance and inhibits inflammation in
intervertebral disc degeneration. Sci Rep. 13:200012023. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Lu R, Xu H, Deng X, Wang Y, He Z, Xu S,
Liang S, Huang X, You H, Guo F, et al: Physalin A alleviates
intervertebral disc degeneration via anti-inflammatory and
anti-fibrotic effects. J Orthop Translat. 39:74–87. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Chen T, Qian Q, Makvandi P, Zare EN, Chen
Q, Chen L, Zhang Z, Zhou H, Zhou W, Wang H, et al: Engineered
high-strength biohydrogel as a multifunctional platform to deliver
nucleic acid for ameliorating intervertebral disc degeneration.
Bioact Mater. 25:107–121. 2023.PubMed/NCBI
|
|
89
|
Wang H, Ding Y, Zhang W, Wei K, Pei Y, Zou
C, Zhang C, Ding J, Fang H and Tan S: Oxymatrine liposomes for
intervertebral disc treatment: Formulation, in vitro and vivo
assessments. Drug Des Devel Ther. 14:921–931. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Zhang W, Yang M, Sun T, Zhang J, Zhao Y,
Li J and Li Z: Can manganese dioxide microspheres be used as
intermediaries to alleviate intervertebral disc degeneration with
strengthening drugs? Front Bioeng Biotechnol. 10:8662902022.
View Article : Google Scholar : PubMed/NCBI
|