Introduction
Myocardial ischemia-reperfusion injury (MIRI) is a
notable complication associated with interventional therapies for
cardiovascular diseases, such as acute myocardial infarction. MIRI
can impair both cardiac systolic and diastolic functions, leading
to arrhythmias and deteriorated cardiac function (1). The pathophysiological mechanism of
MIRI is complex and may involve multiple factors, including
intracellular calcium overload, production of oxygen free radicals,
inflammatory responses and apoptosis, all of which contribute to
myocardial injury and are associated with adverse clinical outcomes
(2,3). Therefore, it is important to further
elucidate the underlying mechanisms of MIRI and explore new
therapeutic strategies. Pyroptosis, a form of regulated cell death
characterized by cellular lysis, exacerbates inflammatory cascades
by releasing pro-inflammatory cytokines (4,5).
During MIRI, the release of inflammatory mediators, including
nucleotide-binding domain leucine-rich repeat (NLR) family
proteins, caspases-1/4/5/11 and interleukin (IL)-1β, trigger
pyroptosis, resulting in cell death (6). Consequently, targeting the inhibition
of pyroptosis represents a promising therapeutic strategy for
MIRI.
Puerarin (Pue) is a natural plant-derived medicine
extracted from the root of Pueraria lobata, which exhibits
multiple pharmacological effects, such as lactate reduction,
anti-inflammatory action and pro-angiogenic activity. Pue also
demonstrates therapeutic potential for cardiovascular disease,
osteoporosis, neurological diseases and intestinal fat absorption
(7–9). A previous study has demonstrated that
targeted delivery of the Pue herbal monomer exerts anti-pyroptotic
effects by suppressing the nucleotide-binding oligomerization
domain-like receptor family pyrin domain-containing 3
(NLRP3)/caspase-1/IL-1β/18 signaling pathway in a rat model of
myocardial infarction, promoting macrophage polarization to the
anti-inflammatory M2 phenotype and thereby alleviating the
inflammatory microenvironment (10). Pue has been shown to exhibit
notable suppression of pyroptosis in cardiomyocytes (11), but its role in regulating
pyroptosis in MIRI requires further investigation.
The nuclear factor E2-related factor 2 (NRF2)/heme
oxygenase-1 (HO-1) signaling pathway is an important intracellular
antioxidant stress signaling pathway that maintains the redox
balance in cells and protects them from oxidative damage (12). NRF2 binds to antioxidant response
elements (AREs), for which HO-1 is a downstream target gene. Thus,
the NRF2/HO-1 axis mediates cell responses to oxidative stress.
Oxidative stress results in the notable generation of reactive
oxygen species (ROS), triggering NLRP3 inflammasome-mediated
pyroptosis and aggravating cell damage (13). Oxidative stress can be ameliorated
and ROS can be suppressed by activating NRF2 (14). Furthermore, in vitro
experiments have demonstrated that Pue activates NRF2 and reduces
ROS levels in cardiomyocytes (15). Therefore, based on current
findings, the present study hypothesized that NRF2 activation may
have modulated pyroptosis, while Pue was hypothesized to display a
dual regulatory effect on NRF2 signaling and pyroptosis. However,
the mechanism by which Pue regulates pyroptosis requires
exploration.
Materials and methods
Animal model establishment
A total of 32 male Sprague-Dawley rats (age, 8–10
weeks; weight, 250–300 g) were purchased from Changzhou Kavens
Experimental Animal Co., Ltd. All protocols in the present study
strictly complied with ethical guidelines for animal research
(16), and the animal experiment
procedures were reviewed and approved by the Ethics Committee of
Chongqing Medical University (Chongqing, China; approval no.
IACUC-CQMU-2024-0618). The rats were supplied with food and water
ad libitum and were housed in conditions maintained at a
temperature range of 22–25°C and a relative humidity of 50–60%
under a 12-h light/dark cycle.
Following 1 week of acclimation, the rats were
anesthetized with 50 mg/kg pentobarbital sodium intraperitoneally.
An incision was made on the neck of each rat, after which the
trachea was exposed by separating the muscle. A transverse incision
of 1 mm was made in the cricoid cartilage and a tracheal tube was
subsequently inserted and connected to a small animal ventilator
for positive pressure ventilation. The ventilator parameters were
adjusted in order to maintain the respiratory rate at 80
breaths/per min. The heart was exposed and the pericardium was
carefully dissected. A needle was inserted 1–2 mm below the lower
edge of the left atrial appendage and out of the pulmonary artery
cone. The myocardium gradually became pale and ST-segment elevation
was measured via electrocardiogram. After 30 min of left anterior
descending artery ligation, the sutures were loosened and
reperfusion was performed for 120 min. The sham operation group
only underwent thoracotomies without suture ligation of arteries. A
total of 100 mg/kg Pue (cat. no. P111270; Shanghai Aladdin
Biochemical Technology Co., Ltd.) and 10 mg/kg pyroptosis inhibitor
MCC950 (cat. no. S7809; Selleck Chemicals) were intraperitoneally
injected 24, 12 and 2 h before ischemia/reperfusion (I/R),
respectively. The rats were randomly divided into sham, I/R, Pue +
I/R and MCC950 + I/R groups (n=8 per group). An equal volume of
0.9% saline solution was administered intraperitoneally to both the
sham and I/R groups.
The health and behavior of rats were monitored every
4 h during the first 24 h after operation and twice daily
thereafter. Myocardial tissues were harvested immediately after the
rats were euthanized at 72 h after myocardial I/R. The human
endpoints in the current study were as follows: i) Weight loss
reached ≥20% and a reduction in food and water intake by ≥40%
within 3 days following MIRI; ii) the occurrence of severe
breathing difficulties, paralysis or continuous convulsions; iii)
rats showed an inability to obtain food or water independently; or
iv) extensive tissue necrosis or uncontrollable infection occurred.
The rats were sacrificed by intraperitoneal injection of 250 mg/kg
pentobarbital sodium. Following euthanasia, respiratory and cardiac
arrest were observed, and monitoring of these parameters was
sustained for an additional 5 min to verify the successful
sacrifice of rats. During surgical procedures, buprenorphine (0.05
mg/kg) was subcutaneously administered twice daily to relieve pain.
The total duration of the experiment was 10 days. A total of two
rats were euthanized within 24 h of reperfusion due to meeting the
aforementioned endpoints and were replaced with new rats.
Echocardiography
Small animal echocardiography was performed using
the Vevo2100 imaging system (FUJIFILM VisualSonics; FUJIFILM
Corporation). Anesthesia was administered by intraperitoneal
injection of 50 mg/kg pentobarbital sodium. The rats were shaved,
and a 30 MHz probe was placed on the left sternal border between
the 4 and 5th intercostal spaces of the left sternum. The angle was
adjusted to obtain the best view, which displayed the maximum
anatomical detail of the cardiac structures. Two-dimensional,
M-mode and color Doppler ultrasound images were collected
successively, and heart rate, left ventricular end-diastolic
diameter and left ventricular end-systolic diameter were recorded.
The left ventricular ejection fraction (LVEF) value was calculated.
The mean value for each parameter was derived from five consecutive
cardiac cycles.
Triphenyltetrazolium chloride
(TTC)/Evans blue staining
A thoracotomy was performed on the rats after they
were anesthetized with 50 mg/kg pentobarbital sodium
intraperitoneally. The anterior descending coronary artery was
ligated quickly, and 1 ml 0.5% Evans blue dye (cat. no. E2129;
Sigma-Aldrich Co. LLC) was injected through the thoracic aorta. The
heart was then isolated and preserved. The slices (1 mm) were made
after the heart had been collected, stained with 1% TTC (cat. no.
D025-1-3; Nanjing Jiancheng Bioengineering Institute) at 37°C for
15–20 min in the dark, fixed in 4% formalin at room temperature for
24 h and photographed using a light microscope (cat. no. VHX–X1,
Keyence Co., Ltd.). ImageJ (version 1.8.0; National Institutes of
Health) was used to analyze the myocardial infarctions.
Hematoxylin-eosin staining (H&E)
staining
Myocardial tissue samples were fixed in 4% neutral
formaldehyde at room temperature for 48 h. The tissue was
dehydrated with an ethanol gradient (75% for 4 h, 85% for 2 h, 90%
for 1.5 h, 95% for 1 h and absolute ethanol I/II for 0.5 h), then
stained with the transparent agent xylene for transparency and
placed in a paraffin tank to complete the wax immersion and
embedding procedure. The wax blocks were sectioned into slices of 4
µm, which were baked at 60°C for 3 h. The sections were dewaxed
after baking. Hematoxylin staining was performed at room
temperature for 5 min, before sections were washed with water for 5
min to achieve bluing. The sections were stained with 1% eosin
staining solution at room temperature for 5 min and subsequently
washed with water for 30 sec to complete H&E staining. After
dehydration, the sections were made transparent, mounted, observed
and images captured using a light microscope (cat. no. IX71;
Olympus Corporation).
Masson's trichrome staining
The cardiac tissue was fixed, embedded and
sectioned, following the aforementioned H&E staining protocol.
The section thickness was 5 µm. The sections were treated with
hematoxylin solution (5 min), ponceau-acid fuchsin solution (3
min), 1% phosphomolybdic acid solution (5 min) and aniline blue
staining solution (all room temperature; 5 min) successively. After
rinse in 1% acetic acid (1 min) and running water (30 sec),
sections were dehydrated and mounted. Myocardial fibers were
stained red, collagen fibers were stained blue and cell nuclei were
stained dark blue. Images were captured using a light microscope
(cat. no. BX53; Olympus Corporation).
Immunohistochemistry (IHC)
Tissues were fixed with 4% paraformaldehyde at room
temperature for 24 h. Paraffin sections (4 µm) were deparaffinized
in xylene (cat. no. X112054; Aladdin, Inc.), rehydration through a
graded ethanol and rinsed with distilled water for 5 min. The
sections were placed in a beaker containing citrate buffer as the
antigen retrieval solution (cat. no. P0086; Beyotime Institute of
Biotechnology), heated in a microwave at 700 W (95±3°C) for 15 min,
and then cooled naturally. Antigen retrieval was performed by
rinsing with PBS three times for 5 min each. Endogenous peroxidase
activity was blocked by incubating samples with 3% hydrogen
peroxide at room temperature for 15 min. Then, 5% goat serum (cat.
no. C0265; Beyotime Institute of Biotechnology) was added at room
temperature for 30 min to prevent non-specific staining.
Subsequently, diluted primary antibodies against IL-1β (1:200, cat.
no. ab9722; Abcam), NLRP3 (1:5,000, cat. no. MA5-32255; Thermo
Fisher Scientific, Inc.) and NRF2 (1:500, cat. no. PA5-27882;
Thermo Fisher Scientific, Inc.) were added at 4°C overnight. An
HRP-labeled secondary antibody (1:500, cat. no. 7071; Cell
Signaling Technology) were incubated at 37°C for 1 h. The sections
were developed using a DAB chromogenic agent (cat. no. ZLI-9018;
Beijing Zhongshan Jinqiao Biotechnology Co., Ltd.), counterstained
with hematoxylin at room temperature for 3 min. The sections were
dehydrated, cleared with xylene and mounted with neutral balsam
(cat. no. ZLI-9555; Beijing Zhongshan Jinqiao Biotechnology Co.,
Ltd.). Finally, sections were observed under a light microscope
(cat. no. XDS-1A, Shanghai Cany Precision Instrument Co., Ltd.).
Quantitative analysis was performed using ImageJ (version 1.8.0;
National Institutes of Health). Brown-yellow staining indicated the
positive expression of inflammasome markers.
Cell culture and model
establishment
H9C2 rat embryonic cardiomyocytes (cat. no.
ACL-0143; China Center for Type Culture Collection) were cultured
in high-glucose Dulbecco's modified Eagle's medium (cat. no.
SH30243.01; HyClone™; Cytiva), supplemented with 10% fetal bovine
serum (cat. no. A511-001; Lonsera; Shanghai Shuangru Biotechnology
Co., Ltd.) and 1% penicillin-streptomycin (cat. no. BL505A;
Biosharp). The cells were cultured in a 5% CO2 incubator
at 37°C. The oxidative stress model was established by treating the
cells with different concentrations of H2O2
(0, 100, 200, 300 and 400 µM) for different durations (0, 15, 30,
60, 90 and 120 min) in order to screen for the optimal time and
concentration for the purposes of the study. These cells were
randomly divided into 3 groups: i) The control group, which
consisted of normal cultured cells; ii) the
H2O2 group, containing cells treated with
H2O2; and iii) the Pue +
H2O2 group, in which cells were pre-treated
with Pue at the selected concentration of 100 mM for 2 h prior to
the addition of H2O2.
Cardiomyocyte transfection
To overexpress NRF2 in H9C2 cardiomyocytes, 2.5 µg
of pcDNA3.1-NRF2 plasmid (Gencefe) was mixed with
Lipofectamine® 8000 transfection reagent (cat. no.
C0533; Beyotime Institute of Biotechnology) according to the
manufacturer's instructions and the empty pcDNA3.1 plasmid was used
as a negative control. Cardiomyocytes were transfected for 48 h.
Transfection efficiency was assessed via western blotting. The
cells were then randomly divided into 5 groups (n=3 wells/group):
i) The negative control overexpression (OE-NC) group, as
aforementioned; ii) the H2O2 + OE-NC group,
in which were transfected with empty pcDNA3.1 plasmids as a
negative control and then treated with H2O2;
iii) the H2O2 + NRF2 overexpression (OE-NRF2)
group, comprising cells transfected with the pcDNA3.1-NRF2 plasmid
and treated with H2O2; iv) the
H2O2 + OE-NC + Pue group, consisting of cells
transfected with the empty pcDNA3.1 plasmid, pretreated with Pue
for 2 h and treated with H2O2; and v) the
H2O2 + OE-NRF2 + Pue group, containing cells
that were transfected with the pcDNA3.1-NRF2 plasmid, pretreated
with Pue for 2 h and treated with H2O2.
Temperature of transfection was 37°C. The cells were cultured for
48 h after transfection, seeded into 96-well plates and cultured at
37°C for an additional 24 h before subsequent experimentation.
Cell counting kit-8 (CCK-8) assay
A CCK-8 kit (cat. no. C0037; Beyotime Institute of
Biotechnology) was used to evaluate the viability of H9C2 cells.
According to the manufacturer's instructions, H9C2 cells were
incubated in 96-well plates. For H2O2
toxicity curve, cells were exposed 0, 100, 200, 300 and 400 µmol/l)
of H2O2 for different durations (0, 15, 30,
60, 90, 120 min). For the Pue toxicity curve, cells were incubated
at 37°C with Pue (0, 20, 40, 60, 80, 100, 120 µmol/l) for 2 h. 20
µl of CCK-8 solution was added to each well, after which samples
were incubated at 37°C in a 5% CO2 incubator for 3 h in
the dark. The absorbance at a 450 nm wavelength was measured using
an enzyme detector (cat. no. MK3; Thermo Fisher Scientific,
Inc.).
Examination by transmission electron
microscopy (TEM)
H9C2 cardiomyocytes were fixed with 2.5%
glutaraldehyde at 4°C for 2 h, centrifuged and sedimented at 90 × g
for 5 min at 4°C and subsequently fixed as aforementioned. Cells
were washed 3 times with 0.1 M phosphate buffer solution (pH 7.4),
each time for 15 min. Then, 1% osmium tetroxide was added to
suspend the cell clusters at room temperature for 2 h. After
dehydrating the tissue blocks, these were embedded in a
resin-embedding agent (cat. no. 90529-77-4; Structure Probe, Inc.)
at 60°C for 48 h and sliced into ultra-thin sections of 60–80 nm.
The sections were sequentially stained with 2% uranium acetate (15
min) and lead citrate solutions (both 15 min) at room temperature.
The ultrastructure of the cells was observed using TEM (cat. no.
HT7700; Hitachi High-Technologies Corporation).
Measurement of lactate dehydrogenase
(LDH)
The supernatant of H9C2 cardiomyocytes was collected
from cells in each group. The supernatant was analyzed for LDH
activity according an LDH activity kit (cat. no. BC0680; Beijing
Solarbio Science & Technology Co., Ltd.). The supernatant was
mixed with the LDH detection working solution at a ratio of 1:1 and
incubated at room temperature for 30 min in the dark. Once the
reaction terminated, the absorbance was measured immediately at a
wavelength of 450 nm using an enzyme detector (cat. no. MK3; Thermo
Fisher Scientific, Inc.) to calculate LDH activity in the
supernatant.
Hoechst 33342/PI staining
The Hoechst 33342/PI double fluorescence staining
kit (cat. no. CA1120; Beijing Solarbio Science & Technology
Co., Ltd.) was used to detect pyroptosis in H9C2 cardiomyocytes
from each group. The Hoechst 33342/PI staining solution (5 µl) were
added to each group, mixed well and incubated for 30 min at 4°C in
the dark. After washing the cells three times with PBS, samples
were observed under a fluorescence microscope and images were
captured. Hoechst 33342 resulted in blue fluorescence, marking all
cells, while PI emitted red fluorescence, which marked dead cells.
The results were analyzed using ImageJ software (version 1.8.0;
National Institutes of Health).
Immunofluorescence detection
H9C2 cells were fixed with 4% paraformaldehyde at
room temperature for 15 min, followed by permeabilization with 0.1%
Triton X-100 for 10 min. Samples were blocked with 5% BSA (cat. no.
ST023; Beyotime Institute of Biotechnology) at room temperature for
1 h, before the cells were incubated with NRF2 (1:200; cat. no.
16396-1-AP; Proteintech Group, Inc.), HO-1 (1:200; cat. no.
10701-1-AP; Proteintech Group, Inc.) primary antibodies at 4°C
overnight. Cells were subsequently incubated with the secondary
fluorescent antibody (goat anti-rabbit cyanine 3-conjugated; 1:500;
cat. no. SA00009-2; Proteintech Group, Inc.) at room temperature
for 1 h in the dark. Following this, cells were incubated with
Hoechst 33258 (1:1,000; cat. no. C1011; Beyotime Institute of
Biotechnology) at room temperature for 15 min to stain the nuclei
of the cells. Fluorescence microscopy (cat. no. BZ-X800E; Keyence
Co., Ltd.) was used to observe and photograph the samples and
fluorescence intensity was evaluated using ImageJ (version 1.8.0;
National Institutes of Health). NRF2 and HO-1 showed positive
signals in the form of red fluorescence, while the Hoechst
33258-stained nuclei of the cells exhibited blue fluorescence.
Mitochondrial membrane potential
assay
The mitochondrial membrane potential of H9c2
cardiomyocytes was detected using the JC-1 Staining kit (cat. no.
H9C2; Beijing Solarbio). Following the manufacturer's instructions,
cells were incubated with JC-1 at concentration of 2 µM for 30 min
at 37°C in the dark, ensuring complete coverage of the cells. Then
the cells were washed three times with pre-warmed (37°C) PBS.
Nuclei were stained with 1 ml Hoechst 33342 (cat. no. C1022;
Beyotime Institute of Biotechnology) for 5 min at room temperature
in the dark. Red (JC-1 aggregates) and green (JC-1 monomers)
fluorescence images were observed under fluorescence microscope
(cat. no. BZ-X800E; Keyence Co., Ltd.), and the red/green
fluorescence intensity ratio was quantified using ImageJ (version
1.8.0; National Institutes of Health).
RT-qPCR
Using the TRIzol Extraction Kit (Thermo Fisher
Scientific, Inc.), total RNA was extracted from rat myocardial
tissues and reverse transcribed into cDNA using the RevertAid First
Strand cDNA Synthesis Kit (cat. no. K1622; Thermo Fisher
Scientific, Inc.). According to the manufacturer's instructions,
qPCR was performed using the SYBR Green PCR kit (cat. no. F-415XL;
Thermo Fisher Scientific, Inc.) on the 7500 Real-Time PCR System
(Applied Biosystems; Thermo Fisher Scientific, Inc.) to evaluate
mRNA expression of the target genes. Primer Premier 5.0 software
(Sangon Biotech Co., Ltd.) was used for the design and synthesizing
of all primers, which are shown in Table I.
 | Table I.Primer sequences for reverse
transcription-quantitative PCR. |
Table I.
Primer sequences for reverse
transcription-quantitative PCR.
| Gene | Sequence
(5′-3′) |
|---|
| GSDMD | F:
CAGCATCCTTGAGTGTCT |
|
| R:
GAGCCTTAGTAGCCAGTAG |
| NLRP3 | F:
TTGAAGAGGAGTGGATAGGT |
|
| R:
GGTGTAGCGTCTGTTGAG |
| ASC | F:
ACAGCCAGAACAGAACATT |
|
| R:
GTCCTTCAGTCAGCACATT |
| IL-1β | F:
GACAGAACATAAGCCAACAA |
|
| R:
ACACAGGACAGGTATAGA |
|
| TTC |
| NRF2 | F:
CCATTTACGGAGACCC |
|
| R:
CACTGTGCCCTTGAGC |
| GAPDH | F:
ATCGGACGCCTGGTTA |
|
| R:
CGCTCCTGGAAGATGG |
Western blotting
Myocardial tissue and cardiomyocytes were lysed in
RIPA lysis buffer (cat. no. P0013B; Beyotime Institute of
Biotechnology) at 4°C for 30 min; the supernatant was subsequently
collected after centrifugation. As NRF2 is a nuclear transcription
factor, its level in the nucleus was primarily detected in the
present study. GAPDH was used as a loading control for cytoplasmic
protein, while histone H3 was used as a loading control for the
detection of nuclear NRF2. Nuclear proteins were extracted using
the Nuclear and Cytoplasmic Protein Extraction Kit (cat. no. P0027;
Beyotime Institute of Biotechnology) in strict accordance with
manufacturer's instructions. Sample protein concentration was
determined using a BCA protein concentration determination kit
(cat. no. BL521A; Biosharp Life Sciences). A total of 30 µg
protein/lane was loaded for electrophoresis. Proteins were
separated by SDS-PAGE using a 5% stacking gel at 80 V for 30 min
and 10% separating gel at 120 V for 60 min, and were transferred to
PVDF membranes. To prevent non-specific binding, membranes were
incubated with 5% BSA (cat. no. ST023; Beyotime Institute of
Biotechnology) at 37°C for 2 h. Subsequently, samples were
incubated with NRF2 (1:1,000; cat. no. 16396-1-AP; Proteintech
Group, Inc.), HO-1 (1:1,000; cat. no. 10701-1-AP; Proteintech
Group, Inc.), NLRP3 (1:1,000; cat. no. 68102-1-Ig; Proteintech
Group, Inc.), apoptosis-associated speck-like protein (ASC;
1:1,000; cat. no. BD-PT0365; Biodragon), gasdermin D (GSDMD)
(1:1,000; cat. no. BD-PT7991; Biodragon), caspase-1 (1:1,000; cat.
no. BD-PC0003; Biodragon), IL-1β (1:1,000; cat. no. BD-PT5201;
Biodragon), histone H3 (1:1,000; cat. no. abs145120; Absin) and
glyceraldehyde-3-phosphate dehydrogenase (GAPDH; 1:1,000; cat. no.
60004-1-Ig; Proteintech Group, Inc.) primary antibodies overnight
at 4°C. After incubation with primary antibodies, PVDF membranes
were washed three times with TBST (0.1% Tween-20) for 5 min each.
The PVDF membranes were then placed in a secondary antibody
solution of dilution 1:5,000 containing HRP-labeled goat
anti-rabbit IgG (cat. no. ZB-2301; Beijing Zhongshan Jinqiao
Biotechnology Co., Ltd.) and HRP-labeled goat anti-mouse IgG (cat.
no. ZB-2305; Beijing Zhongshan Jinqiao Biotechnology Co., Ltd.) and
incubated at 37°C for 1 h. After adding ECL visualization reagent
(cat. no. P0018S; Beyotime Institute of Biotechnology), the samples
were placed in a chemiluminescence imaging system (cat. no. Tanon
5200; Tanon Science & Technology Co., Ltd.) for detection. The
intensities of protein bands were semi-quantified using ImageJ
software (version 1.8.0; National Institutes of Health).
Statistical analysis
Statistical analyses were performed using GraphPad
Prism software (version 9.1; Dotmatics), and data were presented as
mean ± standard deviation of at least 3 times. Unpaired Student's
t-tests and one-way ANOVA with Bonferroni post-hoc test were used,
while for two factors, two-way ANOVA with the Bonferroni post-hoc
test was used instead. P<0.05 was considered to indicate a
statistically significant difference.
Results
Pue preserves cardiac function in
myocardial I/R rats
To evaluate the protective effects of Pue
preconditioning on MIRI, the present study established a rat model
of MIRI (Fig. 1A). There were no
significant differences in cardiac function parameters observed
among the four experimental groups during pre-operative
echocardiography. The I/R group showed significantly increases in
left ventricular internal diastolic diameter and left ventricular
internal diameter at systole along with significantly decreased
LVEF values compared with the sham group post-operation. Therefore,
Pue administration improved cardiac function in I/R model rats
(Fig. 1B). TTC staining revealed
that all three experimental groups showed higher myocardial
infarction areas, represented by lighter reddish areas, than the
sham group, with the most obvious infarction observed in the I/R
group. Compared with the I/R group, both Pue and MCC950 treatments
significantly reduced infarction area, suggesting that Pue
intervention alleviated myocardial infarction (Fig. 1C and D).
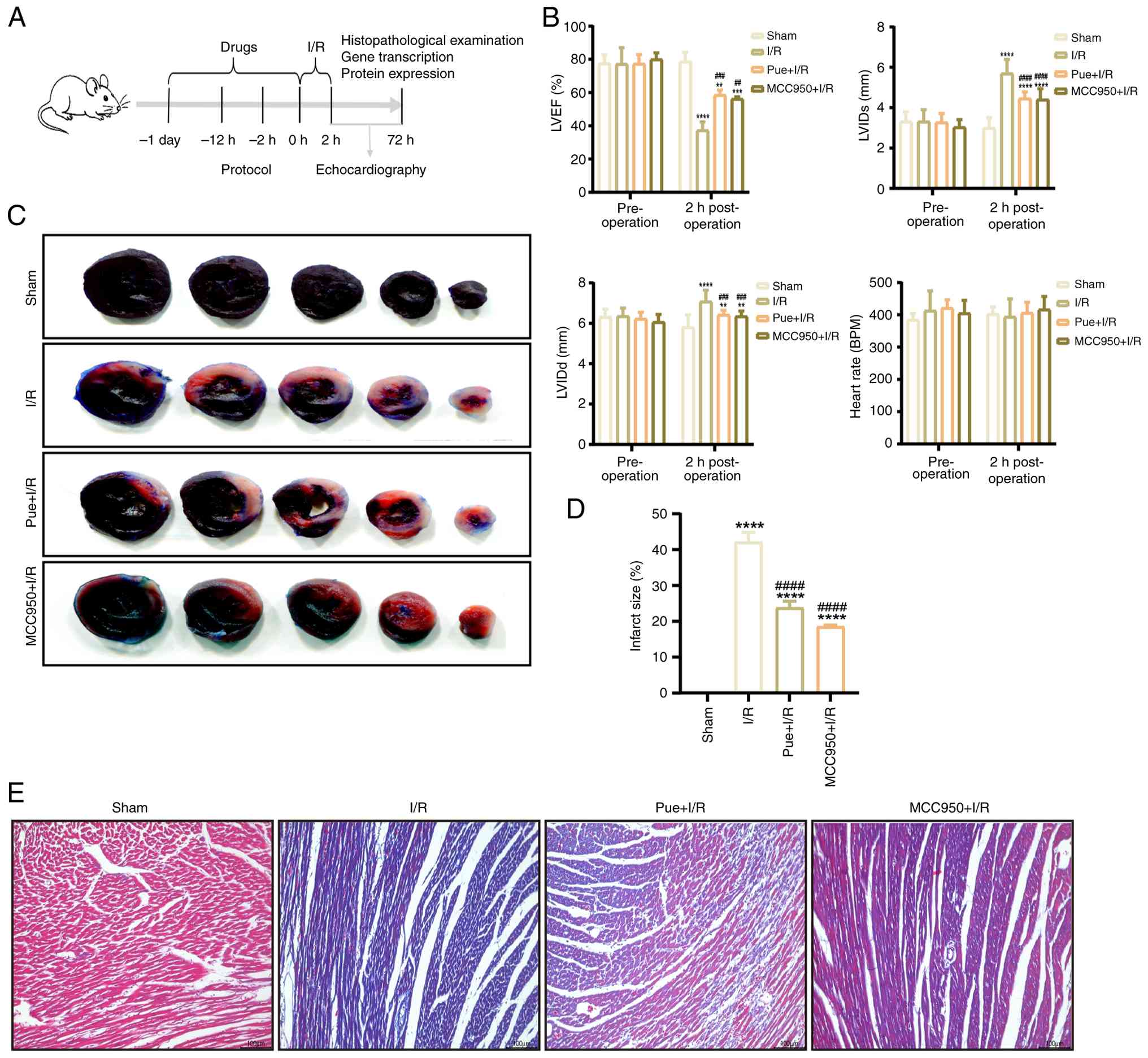 | Figure 1.Pue displays cardioprotective effects
against myocardial ischemia-reperfusion injury in vivo. (A)
Timeline of Pue administration and schematic of the rat I/R injury
model. (B) Changes in cardiac function (LVIDd, LVIDs, LVEF) and
heart rate were observed in each group at 2 h after myocardial I/R.
(C) Triphenyltetrazolium chloride staining of myocardial tissues.
(D) Quantitative comparison of infarction areas among each group
(n=8). (E) Masson's trichrome staining of myocardial tissues (scale
bar, 100 µm). **P<0.01, ***P<0.001 and ****P<0.0001 vs.
sham group; ##P<0.01, ###P<0.001 and
####P<0.0001 vs. I/R group. Pue, puerarin; LVIDd,
left ventricular internal dimension at end-diastole; LVIDs, left
ventricular internal dimension at end-systole; LVEF, left
ventricular ejection fraction; I/R, ischemia/reperfusion. |
Masson's trichrome staining of myocardial sections
was performed in each group to evaluate myocardial fibrosis. In the
I/R group, myocardial tissue was notably damaged. The arrangement
of myocardial fibers was uneven, a number of fibers were broken or
dissolved and these characteristics were also accompanied by
fibrous connective tissue hyperplasia. Pue and MCC950 treatments
markedly reduced the area of myocardial infarcts and inhibited
myocardial fibrosis (Fig. 1E).
These findings demonstrated that Pue alleviated myocardial ischemic
injury by reducing the myocardial infarction area and inhibiting
fibrosis, thereby restoring cardiac function after I/R injury.
Pue inhibits inflammation and
pyroptosis during myocardial I/R
Following myocardial I/R, cardiomyocytes undergo
various forms of cell death, leading to the expansion of the
damaged area and the exacerbation of ischemic injury. Recent
studies have demonstrated that pyroptosis is associated with
myocardial I/R and depends on inflammatory mediators (4,17,18).
As a result, the anti-pyroptotic effects of Pue can be observed by
detecting the release of inflammatory factors. H&E staining
showed notable inflammatory cell infiltration, nuclear displacement
and cardiomyocyte necrosis in the I/R group compared with the sham
group, which were markedly reduced in the Pue treatment group
(Fig. 2A). IHC was used to assess
inflammasome protein expression and localization in myocardial
tissue. Compared with the sham group, the expression of NLRP3 and
IL-1β in the I/R group were significantly increased, whereas these
expression significantly decreased after treatments with Pue and
MCC950. Since the NRF2/HO-1 pathway serves an important role in
regulating the antioxidant responses of cell, IHC also demonstrated
a significant increase in the expression of NRF2 in the Pue and
MCC950 groups compared with the I/R group (Fig. 2B and C).
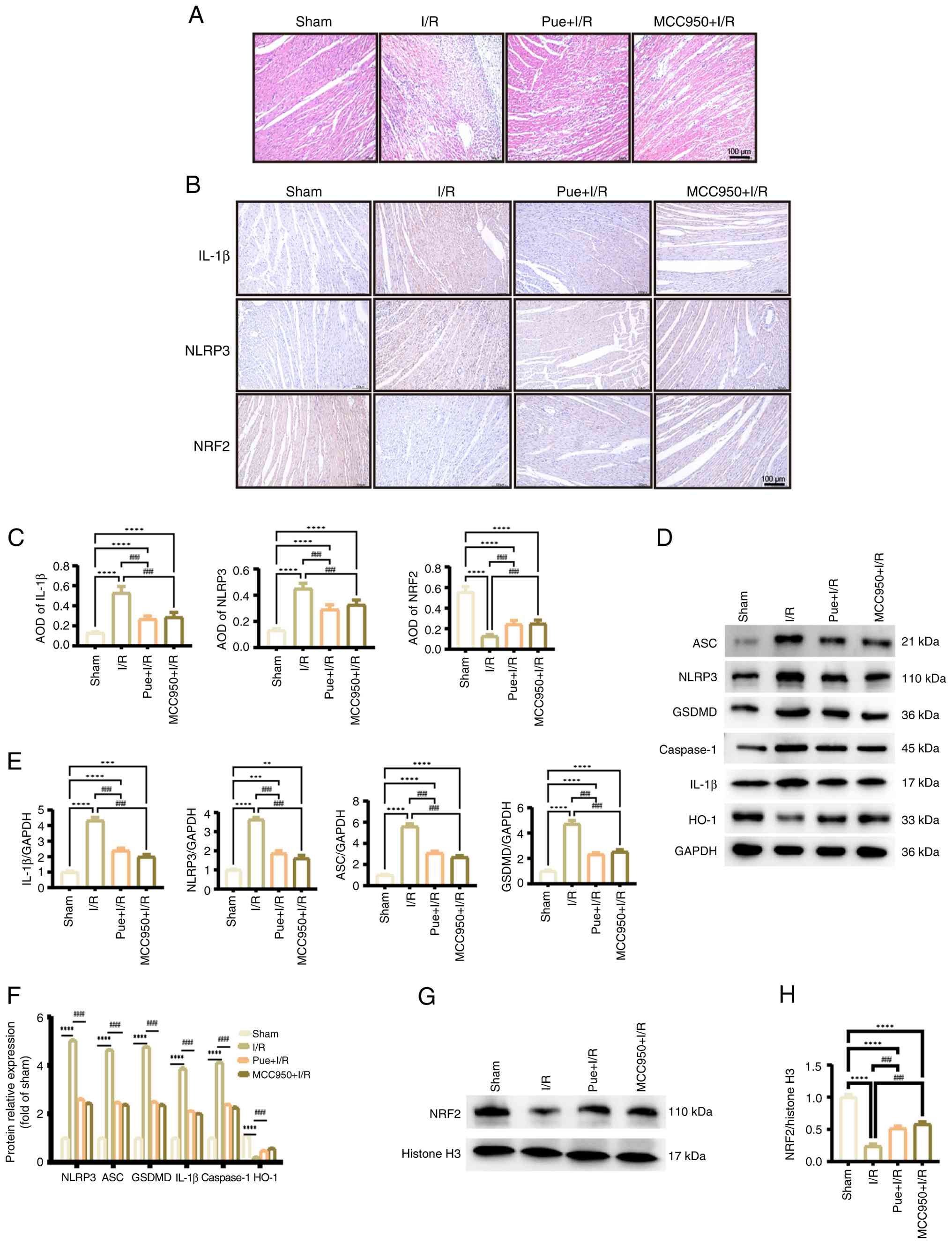 | Figure 2.Pue inhibits the key proteins of the
inflammasome and pyroptosis in myocardial ischemia-reperfusion
injury. (A) Hematoxylin-eosin staining showed the inflammatory
condition of myocardial tissue (n=8; scale bar, 100 µm). (B)
Immunohistochemistry detected the positive expression of
inflammasome markers in myocardial tissues of each group (scale
bar, 100 µm). (C) The AOD values of inflammasome markers obtained
by immunohistochemical staining. (D) Expression of pyroptotic
proteins and HO-1. (E) Quantification of relative mRNA levels of
key pyroptotic markers. (F) Semi-quantification of relative
expression levels of pyroptotic proteins and HO-1. (G) Protein
expression of nuclear NRF2 was assessed by western blotting. (H)
Semi-quantification of relative expression levels of NRF2 obtained
by western blotting. **P<0.01, ***P<0.001 and ****P<0.0001
vs. sham group; ####P<0.0001 vs. I/R group.. AOD,
average optical density; HO-1, heme oxygenase-1; ASC,
apoptosis-associated speck-like protein; NLRP3, nucleotide-binding
oligomerization domain-like receptor family pyrin domain-containing
3; GSDMD, gasdermin D; Pue, puerarin; I/R,
ischemia/reperfusion. |
To further study the effect of Pue on inhibiting
pyroptosis in MIRI, RT-qPCR and western blotting were performed to
detect the expression of key pyroptosis-related markers in
cardiomyocytes. Compared with the sham group, the expression levels
of pyroptosis-related markers such as ASC, IL-1β, NLRP3 and GSDMD
were significantly elevated in the I/R group. These expression
levels were significantly attenuated after treatment with Pue and
MCC950 (Fig. 2E). Western blot
analysis showed that, compared with the sham group, the expression
levels of NLRP3, ASC, GSDMD, IL-1β and caspase-1 significantly
increased in the I/R group, while the expression of NRF2 and HO-1
proteins were significantly reduced in the I/R group. In the Pue +
I/R group, the expression of key pyroptotic proteins was
significantly decreased compared with the I/R group, whereas the
expression levels of NRF2 and HO-1 were significantly increased
(Fig. 2D, F-H). Based on these
findings, it was possible to conclude that Pue inhibited pyroptosis
and reduced MIRI, and that its mechanism may have involved the
NRF2/HO-1 signaling pathway. Thus, Pue showed the potential to
alleviate MIRI by regulating pyroptosis.
Pue alleviates H2O2-induced
cardiomyocyte pyroptosis. To investigate the protective effect
of Pue on cardiomyocytes under oxidative stress, H9C2 cells were
exposed to H2O2 to establish an in
vitro model of oxidative stress. Cell viability decreased in a
time- and dose-dependent manner following
H2O2 treatment. To ensure that the inhibition
rate was within the range of 20 to 30%, H2O2
(100 µM; 120 min) was selected for subsequent experiments by CCK-8
assays (Fig. S1A and B). The
present study evaluated the impact of Pue on the viability of H9C2
cells before further experiments. Pretreatment with different
concentrations (20–120 µmol/l) of Pue for 2 h significantly
improved cell viability in a dose-dependent manner. Finally, 100 µM
Pue was selected for the following experiments (Fig. S1C). The morphology of
cardiomyocytes was observed by microscope. The morphology of cells
in the control group was normal, while cells shrinkage and the
number decreased in the H2O2 group. By
contrast, the number of cells in the Pue +
H2O2 group increased significantly (Fig. S1D).
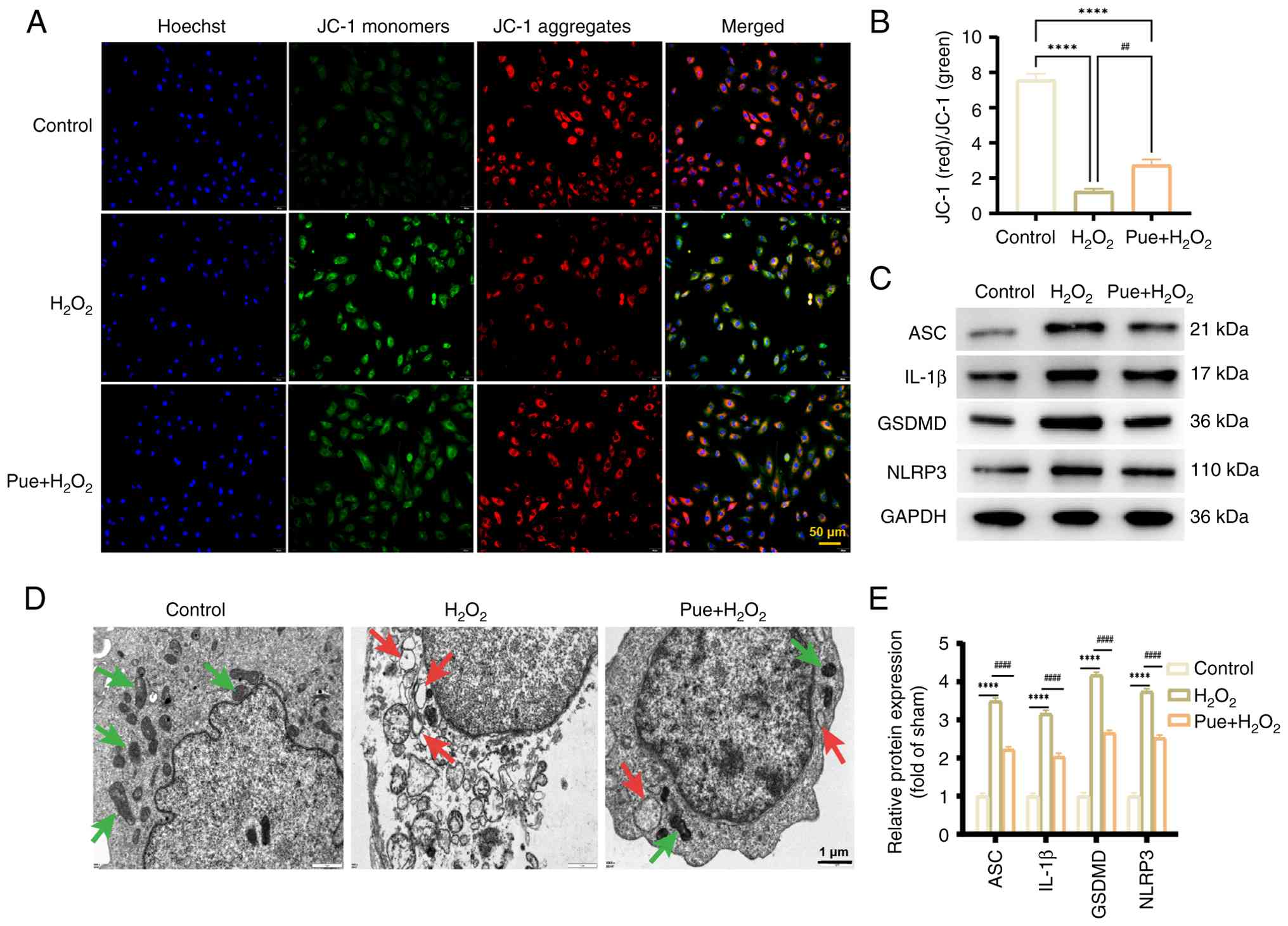
JC-1 staining was used to assess the mitochondrial
membrane potential of rat cardiomyocytes. Red fluorescence
indicated a high mitochondrial membrane potential, while green
fluorescence represented the low mitochondrial membrane potential
of cardiomyocytes. Compared with controls, cells in the
H2O2 group exhibited significantly reduced
red fluorescence and increased green fluorescence, suggesting a
notable decrease in mitochondrial membrane potential. Pue
pretreatment was shown to significantly increase the red/green
fluorescence ratio compared with the H2O2
group (Fig. 3A and B). The
ultrastructural changes in cells were observed by TEM. In the
control group, the morphology and size of mitochondria were normal
and mitochondrial cristae were continuous, complete and clearly
visible. By contrast, in the H2O2 group,
mitochondria became swollen, with notable loss of cristae, severe
vacuolization and nuclear alterations observed, indicating a
tendency towards pyroptosis. However, Pue treatment mitigated
mitochondrial swelling, disruption of cristae and membrane rupture
while restoring nuclear integrity (Fig. 3D). To determine whether the
pyroptosis pathway was activated, the expression of key proteins
was detected via western blotting. Compared with the control group,
the expression levels of NLRP3, ASC, GSDMD and IL-1β were
significantly increased in the H2O2 group.
However, the expression of the aforementioned proteins was shown to
decrease after Pue treatment. These results indicated that Pue
protected H9C2 cells from pyroptosis (Fig. 3C and E).
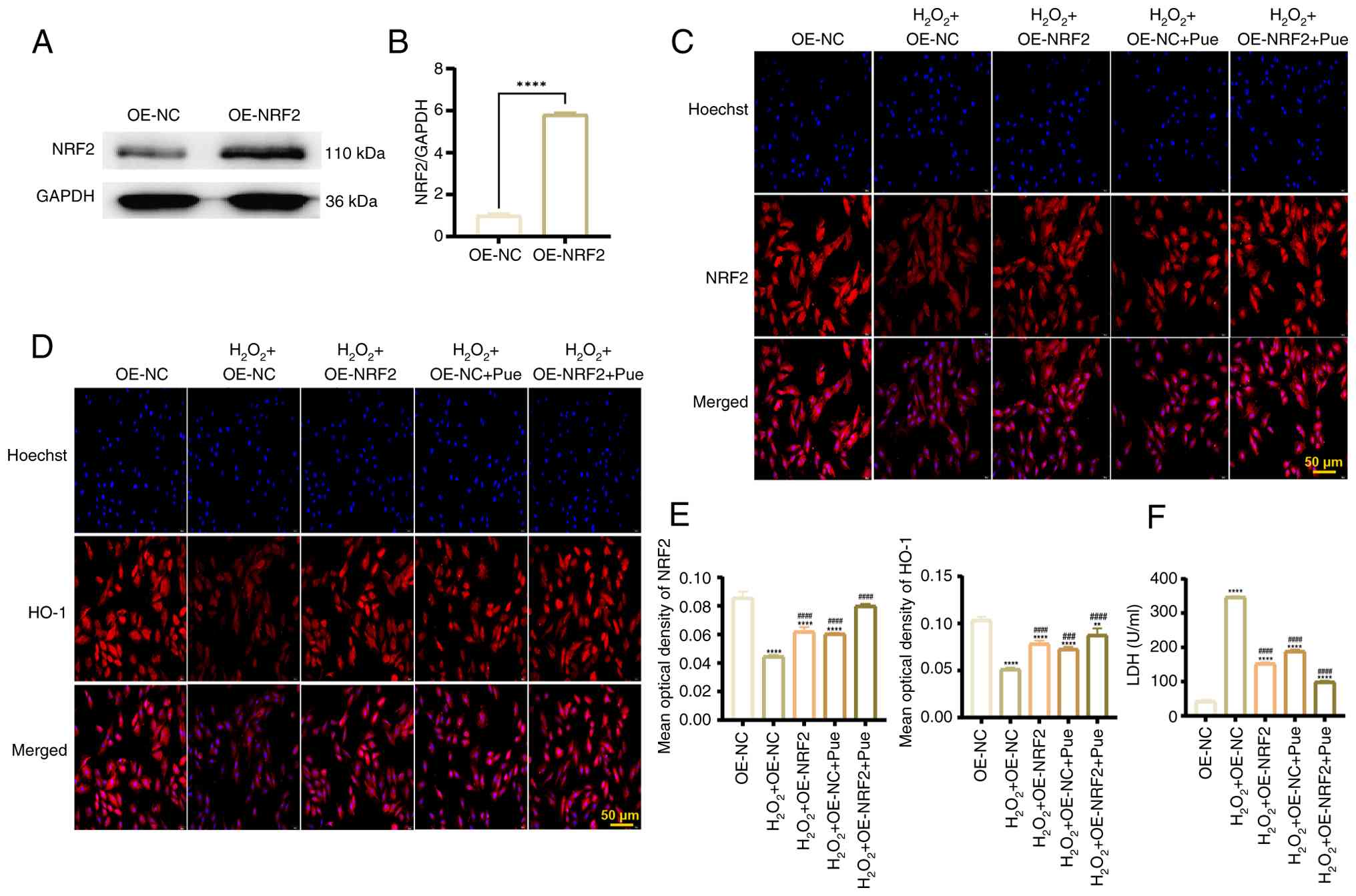
Pue activates the NRF2 pathway to
protect cardiomyocytes from pyroptosis
To clarify the possible mechanism by which Pue
inhibited cardiomyocyte pyroptosis, the present study constructed
an NRF2 overexpression plasmid and verified its expression after
transfection via western blotting. The expression of NRF2 in the
overexpression plasmid group was significantly higher compared with
that in the control plasmid group, indicating a successful
construction of the NRF2 overexpression plasmid (Fig. 4A and B). To observe the regulatory
effect of Pue on the NRF2/HO-1 signaling pathway, cardiomyocytes
were treated with Pue and reclassified into five groups.
Immunofluorescence results showed that NRF2 and HO-1 expression
levels decreased significantly in control group cells after
H2O2 treatment. However, compared with the
H2O2 + OE-NC group, NRF2 was significantly
activated in the H2O2 + OE-NRF2,
H2O2 + OE-NC + Pue and
H2O2 + OE-NRF2 + Pue group. Furthermore, the
significantly increased expression of HO-1 in the aforementioned
groups suggested that Pue enhanced the nuclear translocation of
NRF2 (Fig. 4C-E). During
myocardial infarction, the integrity of the cell membrane is
damaged, resulting in the release of LDH into the supernatant,
which may act as an indirect indication of pyroptosis (19). Based on the results of the LDH
release test, the concentration of LDH increased significantly
after H2O2 treatment compared with the OE-NC
group. Compared with the H2O2 + OE-NC group,
overexpression of NRF2 or Pue treatment, both individually and in
combination, significantly decreased the release of LDH that was
influenced by H2O2 treatment, suggesting that
Pue activated NRF2 to exert protective effects on cardiomyocytes
(Fig. 4F).
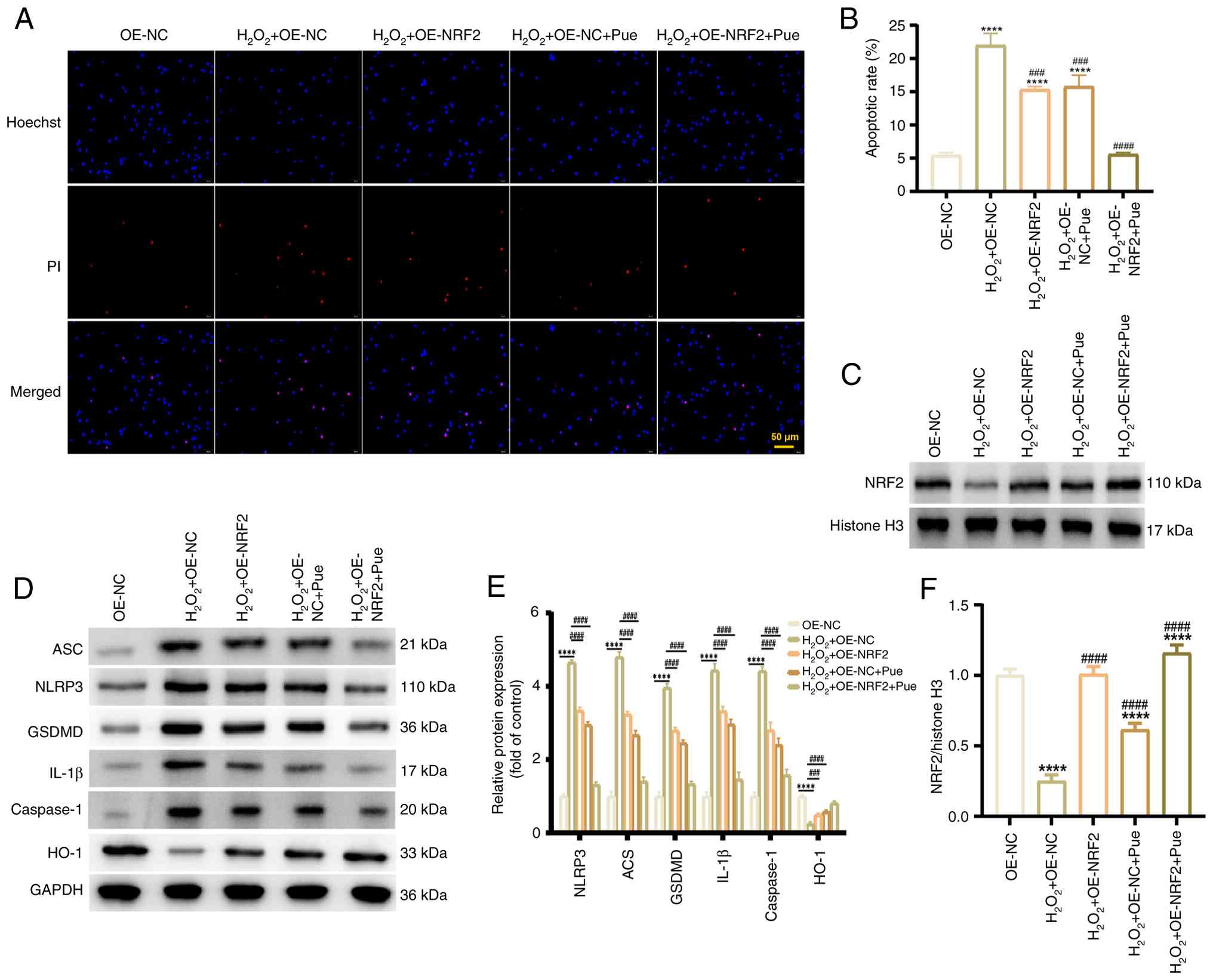
Pue inhibits H2O2-induced
cardiomyocyte pyroptosis through the NRF2/HO-1 pathway. To
further clarify whether Pue affected pyroptosis via the NRF2/HO-1
pathway, Hoechst 33342/PI staining was used to detect pyroptotic
cells. Compared with the OE-NC group, the red fluorescence of cells
in the H2O2 + OE-NC group was significantly
enhanced, indicating an increased degree of pyroptosis. However,
the red fluorescence of cardiomyocytes significantly decreased
after an overexpression of NRF2 or treatment with Pue, suggesting a
notable reduction in the proportion of pyroptotic cells following
these treatments compared with the H2O2 +
OE-NC group (Fig. 5A and B).
Western blot analysis demonstrated that the protein expression of
pyroptosis-related proteins NLRP3, ASC, GSDMD, caspase-1 and IL-1β
was lower in the H2O2 + OE-NC + Pue and
H2O2 + OE-NRF2 + Pue groups compared with
that in the H2O2 + OE-NC group. Meanwhile,
the protein levels of NRF2 and HO-1 were higher in the
aforementioned groups compared with the H2O2
+ OE-NC group (Fig. 5C-F). Under
conditions of oxidative stress, Pue appears to have increased NRF2
expression and decreased cardiomyocyte pyroptosis. Thus, Pue was
shown to activate the NRF2/HO-1 signaling pathway, thereby
inhibiting NLRP3/caspase-1/GSDMD pathway-mediated pyroptosis and
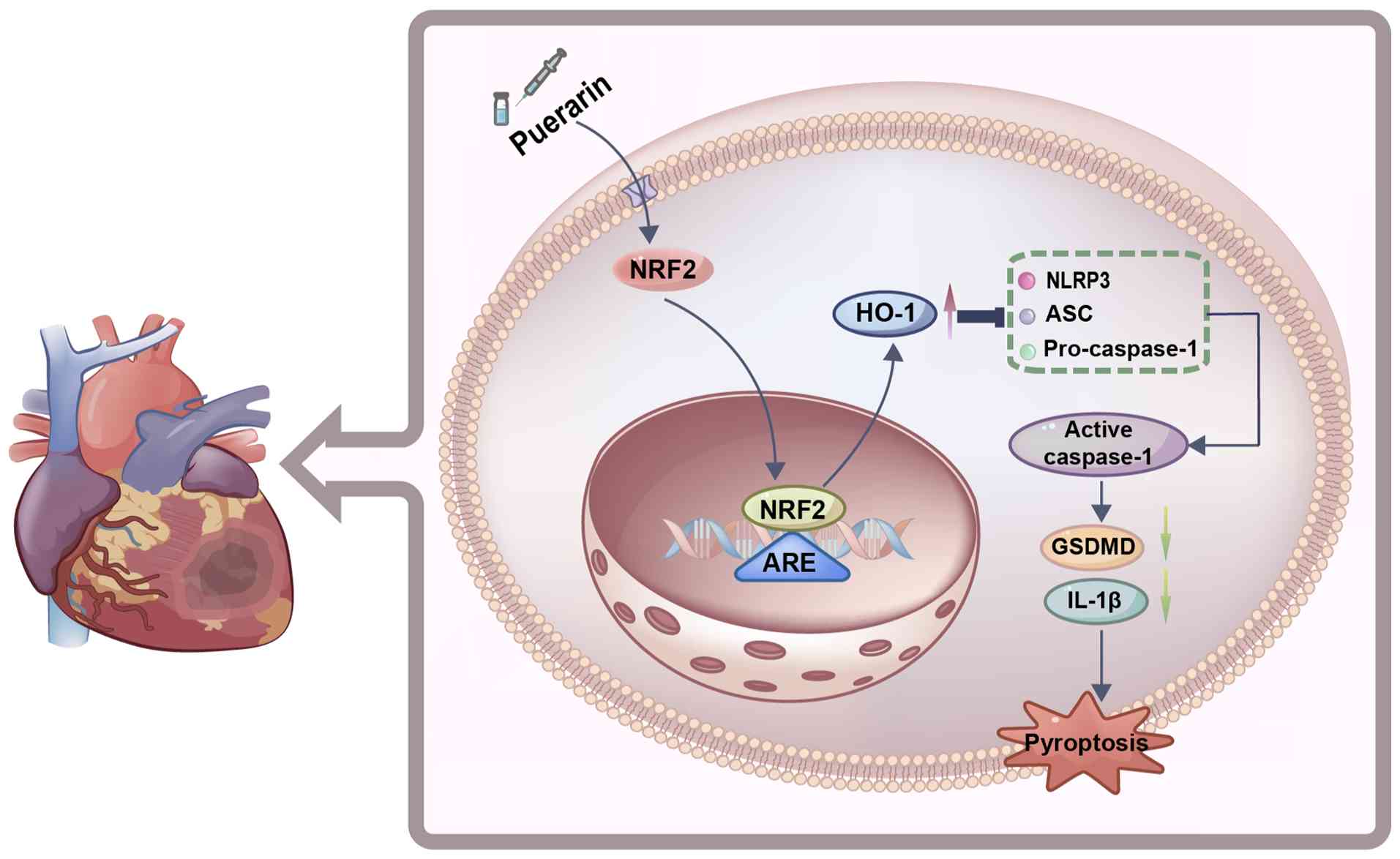
protecting cardiomyocytes from injury during MIRI (Fig. 6).
Discussion
Pue, as the main active component of the traditional
Chinese medicine Pueraria lobata, has been demonstrated to
display multiple protective effects on the cardiovascular system
(7). The present study aimed to
verify the anti-pyroptotic effect of Pue, exploring its influence
on the molecular mechanisms involved in MIRI. Primarily, the
present study observed that Pue exhibited a cardioprotective effect
on MIRI. Pretreatment with Pue effectively mitigated MIRI-induced
myocardial injury, improved cardiac function and partially
decreased myocardial fibrosis Additionally, MIRI has been shown to
result in elevated oxidative stress and an inflammatory response,
accompanied by pyroptosis of cardiomyocytes (20). In the present study, both in
vivo and in vitro experiments demonstrated that Pue
reduced the expression of pyroptosis-related proteins and relieved
cardiomyocyte pyroptosis, ultimately improving the inflammatory
microenvironment. Furthermore, NRF2 activation exhibited
cardioprotective effects by regulating HO-1, which is its
downstream antioxidant enzyme. The overexpression of NRF2
significantly enhanced the inhibitory effect of Pue on pyroptosis,
further validating the importance of this pathway. Therefore, the
present study provided a novel theoretical basis for the protective
mechanism of Pue during MIRI.
Pyroptosis is a form of programmed cell death that
occurs via two pathways: The classical and non-classical pathways.
In the classical pyroptotic pathway, NLRP3 serves as a component of
the inflammasome, activating caspase-1 via ASC and producing and
releasing IL-1β and IL-18. Meanwhile, GSDMD is cleaved into the
GSDMD-N terminal domain, which forms a membrane pore that results
in cell membrane rupture and pyroptosis (21,22).
Inflammasome inhibitors exhibit the potential to reverse the
activation of NLRP3 inflammasome-mediated pyroptosis and therefore
ameliorate MIRI (23). Inhibition
of caspase-1 can reduce myocardial infarct size and cardiac
dysfunction caused by MIRI (24).
Kawaguchi et al (25) also
found that in comparison with the control group, ASC- and
caspase-1-knockout mice showed marked improvements in infarct area
formation, myocardial fibrosis and the development of cardiac
dysfunction in the setting of MIRI. Consequently, inhibition of
pyroptosis has been shown to alleviate myocardial injury caused by
I/R.
Pue is a natural flavonoid that exhibits a variety
of biological and pharmacological properties (7). In a rat model of sepsis-associated
encephalopathy, Pue can inhibit the classical pathway of pyroptosis
mediated by NLRP3/caspase-1/GSDMD and alleviate damage to the
blood-brain barrier (26).
Furthermore, Pue inhibits inflammatory responses and pyroptosis,
reduces serum levels of inflammatory cytokines, enhances the
antioxidant capacity of the kidneys and improves renal function
(27). These protective effects of
Pue are associated with the suppression of pyroptosis, mediated
through the activation of the AMPK/NAD-dependent protein
deacetylase sirtuin-1 (SIRT1) and SIRT1/NLRP3/caspase-1 pathways
(28,29). Both the animal and cell models of
MIRI displayed increased levels of pyroptotic proteins. In the rat
model of myocardial infarction, RT-qPCR showed that the expression
levels of the pyroptosis-related proteins ASC, Il-1β, NLRP3 and
GSDMD significantly decreased after Pue intervention. Furthermore,
western blot analysis also provided evidence that the protein
expression levels of NLRP3, ASC, GSDMD, IL-1β and caspase-1
decreased in the Pue group. These results suggested that Pue
inhibited cardiomyocyte pyroptosis during MIRI.
In MIRI, mitochondrial dysfunction can result in
oxidative stress, apoptosis and pyroptosis (30,31).
The relevant literature has reported that Pue recovers impaired
mitochondrial membrane potential and ROS levels, thus protecting
mitochondrial function (32,33).
In the present study, in vitro experiments were conducted to
establish an oxidative stress model. Here, JC-1 staining showed a
significant increase of mitochondrial membrane potential following
Pue intervention. Additionally, TEM revealed that Pue could
alleviate mitochondrial swelling and the disintegration of inner
cristae compared with the H2O2 control group.
This supported the protective effect of Pue on mitochondria.
Therefore, the regulatory effect of Pue on mitochondrial function
may have also been an important mechanism of pyroptotic inhibition.
The increase in HO-1 and NRF2 expression observed in MIRI rats
after Pue administration suggested a possible link between Pue,
pyroptosis and NRF2 activity, although the underlying mechanism for
this requires further exploration.
A large amount of oxygen free radicals, such as
superoxide anions and hydroxyl radicals, are produced during MIRI,
leading to oxidative stress injury (34,35).
The NRF2/HO-1 signaling pathway is an important intracellular
antioxidant stress signaling pathway. It is predominantly composed
of NRF2 and its target genes, playing a key role in maintaining
redox balance within cells and protecting cells from oxidative
damage (36,37). In the nucleus, NRF2 binds to the
ARE and initiates the transcriptional expression of a series of
downstream antioxidant genes. Amongst these, HO-1 is one of the
important target genes of NRF2 (38). HO-1 is an inducible enzyme with
multiple biological functions. Notably, HO-1 is capable of
catalyzing the decomposition of heme into biliverdin, carbon
monoxide and free iron. These metabolites have strong antioxidant
effects (39,40). Furthermore, research has found that
the expression of the inflammatory factors TNF-α, IL-1 and IL-6
significantly decrease in myocardial tissues after activation of
the NRF2/HO-1 signaling pathway, and the infiltration of
inflammatory cells is similarly reduced. This shows that the
NRF2/HO-1 signaling pathway is capable of inhibiting the
inflammatory response in MIRI (41).
LDH is released from cells following pyroptosis due
to perforation of the membrane when pyroptosis occurs. Therefore,
LDH concentration can be used as an indicator to assess the
occurrence of cell membrane damage due to pyroptosis (42). The results of the present study
showed that the LDH concentration in the H2O2
+ OE-NRF2 cell group was lower than in the
H2O2 + OE-NC treatment group. Additionally,
Hoechst 33342/PI staining showed that the proportion of pyroptotic
cells was significantly lower in the H2O2 +
OE-NRF2 than in the H2O2 + OE-NC group,
indicating that NRF2 activation inhibited pyroptosis. Oxidative
stress, inflammatory response and pyroptosis appear to exhibit
complex interactions. Notably the accumulation of ROS can activate
the NLRP3 inflammasome to promote the occurrence of pyroptosis,
while the inflammatory factors released during pyroptosis, such as
IL-1β, are capable of further exacerbating oxidative stress and the
inflammatory response, resulting in the formation of a cycle.
Therefore, the NRF2/HO-1 signaling pathway was not only responsible
for alleviating oxidative stress injury and inflammation in
cardiomyocytes, but was also responsible for inhibiting
cardiomyocyte pyroptosis. These results suggested that regulation
of the NRF2/HO-1 pathway may become a suitable strategy for
preventing pyroptosis.
It has previously been shown that Pue is capable of
enhancing the activity of antioxidant enzymes, such as superoxide
dismutase and glutathione peroxidase, as well as inhibiting the
generation of lipid peroxidation products, eliminating oxygen free
radicals and alleviating damage to cardiac muscle cells caused by
oxidative stress (43,44). Pue has been shown to regulate the
NRF2 pathway and antioxidant enzyme expression in a mouse model of
colitis, as well as exert antioxidant properties (45). Pue can also induce the expression
of SIRT1, increase the expression of NRF2 and promote the
translocation of NRF2 into the nucleus, displaying potential in
treating metabolic dysfunction-related fatty liver diseases
(46). In a renal I/R model, the
expression of NRF2 and HO-1 proteins are increased following
injection of Pue, revealing that Pue may mediate its renal
protective effects through the NRF2/HO-1 signaling pathway
(47). In the present study, NRF2
demonstrated a significant increase in activation and nuclear
translocation in the H2O2 + OE-NC + Pue group
compared with the H2O2 + OE-NC group,
suggesting that administration of Pue enhanced NRF2 nuclear
translocation. Furthermore, HO-1 expression also increased,
indicating that Pue activated the NRF2/HO-1 signaling pathway and
enhanced the antioxidant capacity of cardiomyocytes. The expression
of pyroptosis-associated proteins was significantly reduced in the
Pue-treated groups (H2O2 + OE-NC + Pue and
H2O2 + OE-NRF2 + Pue) compared with the
H2O2 + OE-NC, suggesting an anti-pyroptotic
effect of Pue. This also provides evidence that Pue exerts its
anti-pyroptotic effects via the NRF2/HO-1 signaling.
Summarily, the present study, to the best of our
knowledge, revealed for the first time the molecular mechanism by
which Pue inhibited cardiomyocyte pyroptosis and alleviated MIRI
via NRF2/HO-1 pathway regulation. These results not only enrich
theories concerning the cardioprotective effect of Pue but also
provide a new potential strategy for clinical treatment of MIRI. As
a natural drug, Pue has the advantages of low toxicity and wide
availability compared with synthetic drugs, such as MCC950. As Pue
also interacts with other signaling pathways, such as the PI3K/AKT
and MAPK pathways (48,49). Hence, it is necessary to explore
the effects produced by these interactions on MIRI. Although the
present study did not directly detect cell death pathways, such as
apoptosis or necroptosis, Pue has been shown to improve
mitochondrial swelling and activate the NRF2/HO-1 pathway. Thus, it
was speculated that Pue may indirectly regulate apoptosis by
preserving mitochondrial function. Furthermore, understanding the
effects of Pue and other therapeutic approaches on different modes
of cell death may help to optimize treatment strategies. In the
future, Pue can be combined with other existing reperfusion
therapies to evaluate drug interactions and ensure the safety of
clinical medication.
In conclusion, the present study revealed that Pue
inhibited NLRP3/caspase-1/GSDMD-mediated pyroptosis by regulating
the NRF2/HO-1 signaling pathway, thereby alleviating myocardial
injury during MIRI. In vivo and in vitro experiments
demonstrated that Pue exerted protective effects on cardiac
function during MIRI and inhibited pyroptosis of cardiomyocytes,
providing evidence for the anti-pyroptotic function of Pue exerted
through the NRF2/HO-1 pathway. The findings of the present study
provided a promising approach for the treatment of MIRI and offered
new insights into the interaction between oxidative stress and
pyroptosis.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present work was supported by Youth project of science and
technology research program of Chongqing Education Commission of
China (grant no. KJQN202302833),the Yunnan Fundamental Research
Kunming Medical University Projects (grant no. 202501AY070001-136)
and the Natural Science Research Projects of Chongqing Medical and
Pharmaceutical College (grant no. YGZZK2025104).
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
XZ and YD designed the study. XZ performed the main
experiments. JL, TH, LT and DL were responsible for data analysis.
XZ and YD confirm the authenticity of all the raw data. All authors
read and approved the final version of the manuscript.
Ethics approval and consent to
participate
All protocols in the present study strictly complied
with the ethical guidelines for animal research, and the animal
experiment procedures were reviewed and approved by the Ethics
Committee of Chongqing Medical University (Chongqing, China;
approval no. IACUC-CQMU-2024-0618).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Heusch G: Myocardial ischemia/reperfusion:
Translational pathophysiology of ischemic heart disease. Med.
5:10–31. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Sánchez-Hernández CD, Torres-Alarcón LA,
González-Cortés A and Peón AN: Ischemia/reperfusion injury:
Pathophysiology, current clinical management, and potential
preventive approaches. Med Inflamm. 2020:84053702020. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhang S, Yan F, Luan F, Chai Y, Li N, Wang
YW, Chen ZL, Xu DQ and Tang YP: The pathological mechanisms and
potential therapeutic drugs for myocardial ischemia reperfusion
injury. Phytomedicine. 129:1556492024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Toldo S and Abbate A: The role of the
NLRP3 inflammasome and pyroptosis in cardiovascular diseases. Nat
Rev Cardiol. 21:219–237. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Vasudevan SO, Behl B and Rathinam VA:
Pyroptosis-induced inflammation and tissue damage. Semin Immunol.
69:1017812023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
He J, Liu D, Zhao L, Zhou D, Rong J, Zhang
L and Xia Z: Myocardial ischemia/reperfusion injury: Mechanisms of
injury and implications for management (review). Exp Ther Med.
23:4302022. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Jiang Z, Cui X, Qu P, Shang C, Xiang M and
Wang J: Roles and mechanisms of puerarin on cardiovascular disease:
A review. Biomed Pharmacother. 147:1126552022. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gao M, Zhang Z, Lai K, Deng Y, Zhao C, Lu
Z and Geng Q: Puerarin: A protective drug against
ischemia-reperfusion injury. Front Pharmacol. 13:9276112022.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lyu Q, Xue W, Liu R, Ma Q, Kasaragod VB,
Sun S, Li Q, Chen Y, Yuan M, Yang Y, et al: A brain-to-gut signal
controls intestinal fat absorption. Nature. 634:936–943. 2024.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang K, Sun Y, Zhu K, Liu Y, Zheng X, Yang
Z, Man F, Huang L, Zhu Z, Huang Q, et al: Anti-pyroptosis
biomimetic nanoplatform loading puerarin for myocardial infarction
repair: From drug discovery to drug delivery. Biomaterials.
314:1228902025. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Sun S, Gong D, Liu R, Wang R, Chen D, Yuan
T, Wang S, Xing C, Lv Y, Du G and Fang L: Puerarin Inhibits
NLRP3-Caspase-1-GSDMD-Mediated Pyroptosis via P2X7 receptor in
cardiomyocytes and macrophages. Int J Mol Sci. 24:131692023.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhang Q, Liu J, Duan H, Li R, Peng W and
Wu C: Activation of Nrf2/HO-1 signaling: An important molecular
mechanism of herbal medicine in the treatment of atherosclerosis
via the protection of vascular endothelial cells from oxidative
stress. J Adv Res. 34:43–63. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Qiu Z, He Y, Ming H, Lei S, Leng Y and Xia
ZY: Lipopolysaccharide (LPS) aggravates high Glucose- and
Hypoxia/reoxygenation-induced injury through activating
ROS-Dependent NLRP3 Inflammasome-mediated pyroptosis in H9C2
cardiomyocytes. J Diabetes Res. 2019:81518362019. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhang J, Pan W, Zhang Y, Tan M, Yin Y, Li
Y, Zhang L, Han L, Bai J, Jiang T and Li H: Comprehensive overview
of Nrf2-related epigenetic regulations involved in
ischemia-reperfusion injury. Theranostics. 12:6626–6645. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jiang S, Qiu S, Mu Y, Liu C, Han Y, Jiang
J and Wang Y: Puerarin reduces susceptibility to ventricular
arrhythmias and inhibits ferroptosis via Sirt1/Nrf2 signaling in
high-fat-diet rats. Free Radic Biol Med. 227:472–484. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
MacArthur Clark JA and Sun D: Guidelines
for the ethical review of laboratory animal welfare People's
Republic of China National Standard GB/T 35892-2018 [Issued 6
February 2018 Effective from 1 September 2018]. Animal Model Exp
Med. 3:103–113. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chen L, Mao LS, Xue JY, Jian YH, Deng ZW,
Mazhar M, Zou Y, Liu P, Chen MT, Luo G and Liu MN: Myocardial
ischemia-reperfusion injury: The balance mechanism between
mitophagy and NLRP3 inflammasome. Life Sci. 355:1229982024.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang AY, Su JB, Sun HT, Liu Q, Li R,
Zhang Y, Wang Y, Wang MY, Ji LM, Gao SQ, et al: Stachyose
ameliorates myocardial ischemia-reperfusion injury by inhibiting
cardiomyocyte ferroptosis and macrophage pyroptosis. Int
Immunopharmacol. 143:1133342024. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Luo T, Jia X, Feng WD, Wang JY, Xie F,
Kong LD, Wang XJ, Lian R, Liu X, Chu YJ, et al: Bergapten inhibits
NLRP3 inflammasome activation and pyroptosis via promoting
mitophagy. Acta Pharmacol Sin. 44:1867–1878. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Pagliaro P and Penna C: Inhibitors of
NLRP3 inflammasome in ischemic heart disease: Focus on functional
and redox aspects. Antioxidants (Basel). 12:13962023. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Coll RC, Schroder K and Pelegrín P: NLRP3
and pyroptosis blockers for treating inflammatory diseases. Trends
Pharmacol Sci. 43:653–668. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Broz P and Dixit VM: Inflammasomes:
Mechanism of assembly, regulation and signalling. Nat Rev Immunol.
16:407–420. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang L, Jiang YH, Fan C, Zhang Q, Jiang
YH, Li Y and Xue YT: MCC950 attenuates doxorubicin-induced
myocardial injury in vivo and in vitro by inhibiting NLRP3-mediated
pyroptosis. Biomed Pharmacother. 143:1121332021. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liao Y, Liu K and Zhu L: Emerging roles of
inflammasomes in cardiovascular diseases. Front Immunol.
13:8342892022. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kawaguchi M, Takahashi M, Hata T, Kashima
Y, Usui F, Morimoto H, Izawa A, Takahashi Y, Masumoto J, Koyama J,
et al: Inflammasome activation of cardiac fibroblasts is essential
for myocardial ischemia/reperfusion injury. Circulation.
123:594–604. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhou S, Li Y, Hong Y, Zhong Z and Zhao M:
Puerarin protects against sepsis-associated encephalopathy by
inhibiting NLRP3/caspase-1/GSDMD pyroptosis pathway and reducing
blood-brain barrier damage. Eur J Pharmacol. 945:1756162023.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang K, Tang Z, Liu S, Liu Y, Zhang H and
Zhan H: Puerarin protects renal ischemia-reperfusion injury in rats
through NLRP3/Caspase-1/GSDMD pathway. Acta Cir Bras.
38:e3873232023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang L, Xie X, Chen Q, Chen Y, Xu X and
Liang T: Puerarin reduces diabetic nephropathy-induced podocyte
pyroptosis by modulating the SIRT1/NLRP3/caspase-1 pathway. Mol
Cell Endocrinol. 595:1124092025. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Peng ZT and Liu H: Puerarin attenuates
LPS-induced inflammatory injury in gastric epithelial cells by
repressing NLRP3 inflammasome-mediated apoptosis. Toxicol In Vitro.
81:1053502022. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Chang X, Liu J, Wang Y, Guan X and Liu R:
Mitochondrial disorder and treatment of ischemic cardiomyopathy:
Potential and advantages of Chinese herbal medicine. Biomed
Pharmacother. 159:1141712023. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Peng JF, Salami OM, Lei C, Ni D, Habimana
O and Yi GH: Targeted mitochondrial drugs for treatment of
myocardial ischaemia-reperfusion injury. J Drug Target. 30:833–844.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Niu P, Sun Y, Wang S, Li G, Tang X and Sun
J, Pan C and Sun J: Puerarin alleviates the ototoxicity of
gentamicin by inhibiting the mitochondria-dependent apoptosis
pathway. Mol Med Rep. 24:8512021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Sheng G, Wu Y, Yao L, Liu H, Zhang P, Song
C, Wu G and Zhu H: Puerarin improves the comorbidity of chronic
pain and depression by binding with Bax and reducing mitochondrial
dysfunction. Mol Pain. 21:174480692513352302025. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Dhalla NS, Shah AK, Adameova A and
Bartekova M: Role of oxidative stress in cardiac dysfunction and
subcellular defects due to ischemia-reperfusion injury.
Biomedicines. 10:14732022. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Xiang M, Lu Y, Xin L, Gao J, Shang C,
Jiang Z, Lin H, Fang X, Qu Y and Wang Y: Role of oxidative stress
in reperfusion following myocardial ischemia and its treatments.
Oxid Med Cell Longev. 2021:66140092021. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Geng J and Zhang C: Liensinine attenuates
inflammatory response and oxidative stress by activation of
Nrf2/HO-1 signaling in L-NAME-induced gestational hypertension.
Naunyn Schmiedebergs Arch Pharmacol. 398:14089–14098. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
El-Emam SZ, Soubh AA, Al-Mokaddem AK and
Abo El-Ella DM: Geraniol activates Nrf-2/HO-1 signaling pathway
mediating protection against oxidative stress-induced apoptosis in
hepatic ischemia-reperfusion injury. Naunyn Schmiedebergs Arch
Pharmacol. 393:1849–1858. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yang X, Liu Y, Cao J, Wu C, Tang L, Bian
W, Chen Y, Yu L, Wu Y, Li S, et al: Targeting epigenetic and
post-translational modifications of NRF2: Key regulatory factors in
disease treatment. Cell Death Discov. 11:1892025. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Kuo PC, Weng WT, Scofield BA, Paraiso HC,
Yu II and Yen JJ: Ischemia-induced endogenous Nrf2/HO-1 axis
activation modulates microglial polarization and restrains ischemic
brain injury. Front Immunol. 15:14405922024. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Manavi MA, Mohammad Jafari R, Shafaroodi H
and Dehpour AR: The Keap1/Nrf2/ARE/HO-1 axis in epilepsy: Crosstalk
between oxidative stress and neuroinflammation. Int
Immunopharmacol. 153:1143042025. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Sun YY, Zhu HJ, Zhao RY, Zhou SY, Wang MQ,
Yang Y and Guo ZN: Remote ischemic conditioning attenuates
oxidative stress and inflammation via the Nrf2/HO-1 pathway in MCAO
mice. Redox Biol. 66:1028522023. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Shi Y, Yang Y, Xu W, Shi D, Xu W, Fu X, Lv
Q, Xia J and Shi F: E3 ubiquitin ligase SYVN1 is a key positive
regulator for GSDMD-mediated pyroptosis. Cell Death Dis.
13:1062022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Huang Y, Wu H, Hu Y, Zhou C, Wu J, Wu Y,
Wang H, Lenahan C, Huang L, Nie S, et al: Puerarin attenuates
oxidative stress and ferroptosis via AMPK/PGC1α/Nrf2 pathway after
subarachnoid hemorrhage in rats. Antioxidants (Basel). 11:12592022.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Hou BY, Zhao YR, Ma P, Xu CY, He P, Yang
XY, Zhang L, Qiang GF and DU GH: Hypoglycemic activity of puerarin
through modulation of oxidative stress and mitochondrial function
via AMPK. Chin J Nat Med. 18:818–826. 2020.PubMed/NCBI
|
|
45
|
Jeon YD, Lee JH, Lee YM and Kim DK:
Puerarin inhibits inflammation and oxidative stress in dextran
sulfate sodium-induced colitis mice model. Biomed Pharmacother.
124:1098472020. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Yang M, Xia L, Song J, Hu H, Zang N, Yang
J, Zou Y, Wang L, Zheng X, He Q, et al: Puerarin ameliorates
metabolic dysfunction-associated fatty liver disease by inhibiting
ferroptosis and inflammation. Lipids Health Dis. 22:2022023.
View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang J, Zheng Q, Chen Z, Liu X, Wan S and
Wang L: Puerarin alleviates renal ischemia/reperfusion injury by
inhibiting apoptosis and endoplasmic reticulum stress via Nrf2/HO-1
pathway. Iran J Basic Med Sci. 28:187–193. 2025.PubMed/NCBI
|
|
48
|
Chen ZQ, Zhou Y, Huang JW, Chen F, Zheng
J, Li HL, Li T and Li L: Puerarin pretreatment attenuates
cardiomyocyte apoptosis induced by coronary microembolization in
rats by activating the PI3K/Akt/GSK-3β signaling pathway. Korean J
Physiol Pharmacol. 25:147–157. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Wang Z, Yu Y, Shao W, Zhao Y, Li Z, Han J,
Wen J, Meng Y, Lin Y and Wang S: Puerarin ameliorates alcoholic
liver disease by regulating intestinal flora and MAPK/Nrf2
signalling pathways. Ecotoxicol Environ Saf. 309:1196992026.
View Article : Google Scholar : PubMed/NCBI
|