Introduction
Prostate-specific membrane antigen (PSMA, also known
as FOLH1/GCPII/NAALADase) is a type II transmembrane glycoprotein
that is highly expressed in prostate cancer cells and now widely
exploited as a molecular imaging and therapeutic target. PSMA
PET/CT has transformed prostate cancer staging and restaging,
enabling the detection of micrometastatic disease and guiding
metastasis-directed therapies. In particular, radioligand therapy
(RLT) has emerged as a life-prolonging treatment for advanced
prostate cancer (1). Despite these
clinical advances, the biological mechanisms regulating PSMA
expression, its heterogeneity across metastatic sites and its
functional consequences remain poorly understood (2). Therefore, a comprehensive overview of
PSMA PET imaging or RLT is beyond the scope of this review.
Instead, it focuses on the molecular functions of PSMA that
underlie its biological and therapeutic relevance. A unified
conceptual framework integrating the metabolic enzymatic activity
of PSMA, non-enzymatic scaffold signaling, tumor-associated
angiogenesis and immunological consequences within tumor biology is
the focus of this review.
Recent studies have revealed that PSMA is not merely
a passive marker but a multifunctional protein involved in
glutamate and folate metabolism, clathrin-mediated internalization,
potentially in signaling processes that influence tumor
progression. Importantly, PSMA expression is not restricted to
prostate epithelial cells but can also be found in tumor-associated
endothelium across a diverse range of malignancies, including renal
cell carcinoma, colorectal cancer and glioblastoma (3,4).
This endothelial expression profile implicates PSMA in angiogenesis
and tumor microenvironment (TME) remodeling. Consistent with this,
paracrine mechanisms have been proposed in which PSMA-positive
extracellular vesicles (EVs) or membrane fragments induce PSMA
expression in endothelial cells to enhance angiogenesis, suggesting
a positive feedback loop for reinforcing tumor vascularization
(5). Additional evidence from
renal cell carcinoma and other urological malignancies further
supports the concept of PSMA as a pan-tumor neovascular marker
(6).
This review will provide a comprehensive summary of
current knowledge on PSMA, covering its structural and enzymatic
features, transcriptional and epigenetic regulatory profile,
endothelial expression and roles in angiogenesis, interactions with
immune and metabolic pathways, and clinical applications and future
therapeutic opportunities (Fig.
1). Our goal is to shift the perspective on PSMA from a mere
diagnostic biomarker to a potential driver of tumor biology and a
versatile pan-cancer therapeutic target.
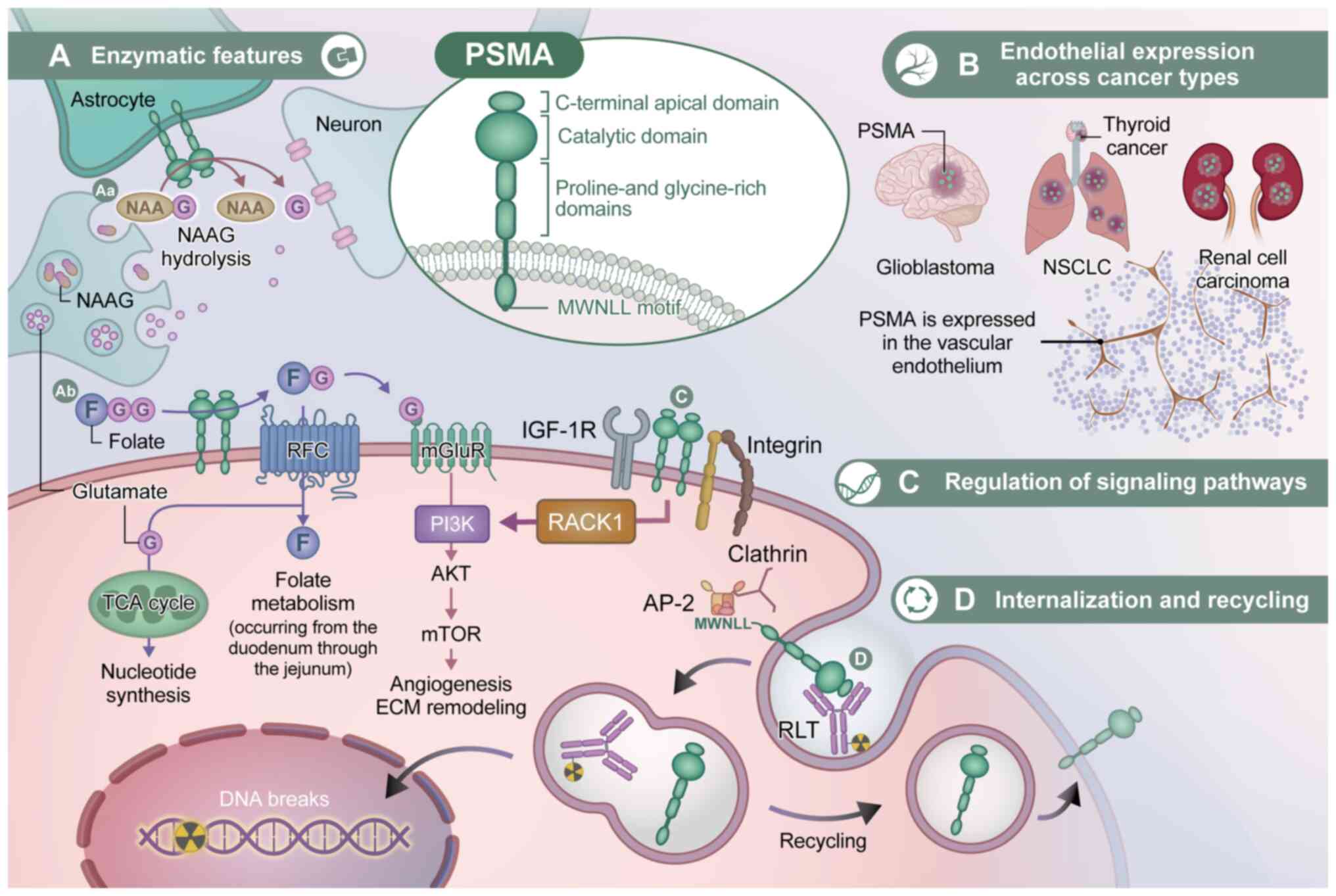 | Figure 1.Integrated schematic of PSMA
molecular functions and therapeutic implications. (A) Enzymatic
features. PSMA structure and enzymatic activities in the (Aa) brain
and (Ab) intestine, linking glutamate signaling and folate
metabolism. (B) Endothelial expression across cancer types. PSMA
expression in tumor-associated vascular endothelium and its role in
tumor angiogenesis. (C) Regulation of signaling pathways. PSMA
functions as a signaling hub through interactions with integrins
and other membrane proteins. (D) Internalization and recycling.
Ligand-induced internalization and recycling of PSMA, enabling
targeted therapy and sustained surface signaling. This schematic
illustration was created with the support of Medical Fig
(https://medicalfig.medicaleducation.co.jp/). AP-2,
adaptor protein 2; ECM, extracellular matrix; F, folate; G,
glutamate; IGF-1R, insulin-like growth factor 1 receptor; mGluR,
metabotropic glutamate receptor; NAAG, N-acetylaspartylglutamate;
NSCLC, non-small cell lung cancer; PSMA, prostate-specific membrane
antigen; RACK1, receptor for activated C kinase 1; RFC, reduced
folate carrier; RLT, radioligand therapy; TCA cycle, tricarboxylic
acid cycle. |
Structure and basic molecular functions of
PSMA
Features as a membrane protein
PSMA is encoded by the FOLH1 gene on chromosome
11p11.2 and was first cloned in the early 1990s as a prostate
cancer-associated surface antigen (7). Shortly thereafter, Bzdega et
al (8) and Pinto et al
(9) identified its enzymatic
identity to be a glutamate carboxypeptidase II (GCPII; NAALADase
I).
Structurally, PSMA is a type II transmembrane
glycoprotein with a short N-terminal cytoplasmic tail [~19 (amino
acids)] containing a canonical MWNLL internalization motif, a
single transmembrane helix (~24 aa) and a large extracellular
ectodomain (~700 aa) that harbors the catalytic machinery (10). The ectodomain is organized into
protease, apical and dimerization domains, where the protein exists
predominantly as a non-covalent homodimer, which is essential for
optimal substrate binding and catalytic efficiency (10).
PSMA is heavily N-glycosylated at up to 10 sites,
where proper glycosylation is required for stability, surface
expression and enzymatic activity. By contrast deglycosylation
leads to ER retention and loss of function (11). Internalization is constitutive and
clathrin-dependent, which is further accelerated by ligand or
antibody binding (12).
Interaction with filamin A (FLNa) restrains constitutive
endocytosis and has been reported to attenuate NAALADase activity
(13). These properties underpin
the use of PSMA as an entry point for antibody-drug conjugates
(ADCs) and RLT.
Enzymatic activities: General
view
PSMA is a member of the M28 metallopeptidase family,
functioning as a glutamate-preferring carboxypeptidase. Its
binuclear zinc active site is located within the protease domain,
coordinated by a number of key residues, such as His377, Asp387,
Asp453, Glu425 and His553. A positively-charged substrate-binding
cavity (Arg210, Arg463, Arg534 and Arg536) stabilizes the
γ-carboxylate group of glutamate (10). Pharmacologically, PSMA can be
inhibited by phosphonate analogs [including
2-(Phosphonomethyl)pentanedioic acid] and can be clinically
targeted by a range of urea-based ligands, such as PSMA-617 and
DCFPyL, for imaging and therapy (11).
GCPII activity and glutamate
signaling
As NAALADase I/GCPII, PSMA hydrolyzes
N-acetylaspartylglutamate (NAAG) into N-acetylaspartate (NAA) and
glutamate. In the central nervous system, this reaction regulates
extracellular glutamate levels to modulate metabotropic and
ionotropic glutamate receptor signaling, thereby serving a key role
in neuronal excitability (14).
Consistent with this, genetic knockout or pharmacologic inhibition
of GCPII reduces susceptibility to seizures and excitotoxic injury
by limiting glutamate release, supporting a physiological role of
NAAG metabolism in neuroprotection (8,15).
In prostate cancer cells, PSMA-derived glutamate
exerts a distinct biological role beyond neurotransmitter
regulation. Increased pericellular glutamate generated by GCPII
activity can activate metabotropic glutamate receptor 1 (mGluR1),
leading to the downstream stimulation of PI3Kβ-AKT signaling and
subsequent phosphorylation of mTOR effectors, such as ribosomal
protein S6 kinase β1 (S6K) and eukaryotic initiation factor
4E-binding protein 1 (4EBP1), ultimately promoting cell
proliferation and survival (16,17).
In particular, this aforementioned signaling cascade is initiated
by enzymatic substrate hydrolysis and represents a
metabolically-mediated mechanism by which PSMA indirectly engages
oncogenic signaling pathways, rather than a direct
receptor-scaffolding function.
Folate hydrolase activity
PSMA also exhibits folate hydrolase activity,
cleaving poly-γ-glutamylated folates into monoglutamate forms that
are competent for cellular uptake through the proton-coupled folate
transporter (PCFT) and the reduced folate carrier (RFC). Whilst
this function is more physiologically relevant in the jejunum, it
is markedly upregulated in prostate cancer, where the increased
folate availability supports nucleotide synthesis and one-carbon
metabolism (18).
Previous transcriptomic analyses have demonstrated
that FOLH1 expression is positively correlated with that of genes
involved in one-carbon metabolism, including MTHFD1L and SHMT2,
suggesting a coordinated metabolic program in PSMA-high tumors
(17). Through this enzymatic
activity, PSMA increases the intracellular pool of folate-derived
one-carbon units, thereby supporting DNA synthesis, redox balance
and epigenetic regulation through S-adenosylmethionine-dependent
methylation reactions.
The contribution of folate hydrolase activity to
tumor progression is however primarily metabolic in nature and does
not require the direct participation of PSMA in receptor-associated
signaling complexes. Together with GCPII-mediated glutamate
production, this function positions PSMA as a metabolic gatekeeper
that aligns nutrient availability with the biosynthetic and
epigenetic demands of proliferating cancer cells.
Substrate specificity and signaling
scaffolding
Beyond its enzymatic activities, PSMA also confers
non-enzymatic functions as a signaling scaffold that reorganizes
membrane-associated receptor complexes. Structural and biochemical
studies have shown that PSMA can interact with β1-integrin and
insulin-like growth factor-1 receptor through various adaptor
proteins, such as receptor for activated C kinase 1, thereby
forming macromolecular signaling assemblies at the plasma membrane
(16).
Through these interactions, PSMA redirects
downstream signaling from the MAPK pathway towards the PI3K-AKT
axis, a process referred to as ‘signaling rewiring’. In particular,
this shift in signaling balance promotes cell survival, migration
and angiogenic potential, which has been associated with more
aggressive tumor phenotypes (16).
However, scaffold-mediated PSMA signaling is
independent of its catalytic activity. Pharmacologic inhibition of
GCPII enzymatic function does not abrogate PSMA-driven PI3K-AKT
activation mediated through integrin-associated complexes,
underscoring that this pathway operates separately from its role in
substrate hydrolysis-dependent metabolic signaling. This
distinction highlights PSMA's dual identity as both an ectoenzyme
and a non-enzymatic signaling adaptor.
In addition to integrin and growth factor receptor
signaling, emerging proteomic studies have suggested that PSMA can
associate with broader cytoskeletal and membrane-organizing
proteins, potentially expanding its scaffold repertoire beyond its
currently characterized partners (19). Defining the full interactome of
PSMA will be essential for understanding how its non-enzymatic
scaffolding functions can contribute to therapy resistance,
cellular plasticity and tumor progression.
Scaffold functions and unexplored
protease activity
Collectively, PSMA functions as a dual-mode
regulator of tumor biology by integrating catalytic metabolic
inputs and non-enzymatic signaling rewiring through distinct but
convergent mechanisms. Its enzymatic activities can generate
glutamate and monoglutamyl folates, thereby indirectly activating
oncogenic signaling pathways through metabolic reprogramming. In
parallel, PSMA can also serve as a signaling scaffold to reorganize
receptor-associated complexes at the plasma membrane, redirecting
downstream signaling towards PI3K-AKT dominance independently of
its substrate hydrolysis capabilities.
Although both axes tend to converge on PI3K-AKT
signaling, their biological origins and regulatory contexts are
fundamentally different. Metabolically-driven signaling depends on
local substrate availability and enzymatic activity, whereas
scaffold-mediated signaling is governed by membrane localization,
protein-protein interactions and cytoskeletal organization. The
relative contribution of these two mechanisms is therefore
context-dependent, varying across cell types, disease stages and
therapeutic pressures.
This dual functional framework provides a conceptual
basis to understand several unresolved features of PSMA biology,
including intratumoral heterogeneity, treatment-induced PSMA
modulation and differential therapeutic responses. In particular,
it offers an explanation into how PSMA can sustain oncogenic
signaling even under conditions of enzymatic inhibition or
metabolic stress.
In endothelial cells, PSMA-driven angiogenesis
appears to rely predominantly on scaffold-mediated signaling rather
than its metabolic enzyme activity, whereas in prostate cancer
cells both axes may operate in parallel. Recognizing this
functional duality is critical for the rational design of
PSMA-targeted therapies and combination strategies.
Intracellular trafficking and endocytosis of
PSMA
Clathrin-dependent endocytosis
triggered by ligand binding
PSMA undergoes constitutive internalization, which
is markedly accelerated upon ligand or antibody engagement. The
short N-terminal cytoplasmic tail contains the MWNLL motif, both
necessary and sufficient for clathrin- and adaptor
protein-2-dependent endocytosis (12,20).
Imaging and mutagenesis studies have reported its localization to
clathrin-coated pits, whilst disruption of the MWNLL motif has been
shown to abrogate its internalization and delays surface clearance
(20). Ligand-triggered
endocytosis has been observed with monoclonal antibody (such J591),
Glu-urea-based inhibitor and ADC treatment, resulting in rapid
trafficking to early endosomes. From there, PSMA can either recycle
back to the plasma membrane or proceed to late endosomes and
lysosomes for ligand degradation or payload release (1).
Filamin A and endocytic
regulation
The cytoplasmic tail of PSMA also bind FLNa, linking
it to the actin cytoskeleton. FLNa binding restrains constitutive
endocytosis and reduces NAALADase activity (12). Disruption of this interaction has
been observed to enhance internalization, indicating that
cytoskeletal context and extracellular cues can dynamically
regulate PSMA trafficking (2).
Implications for targeted drug
delivery
Ligand-induced internalization provides the
mechanistic basis for using PSMA as a therapeutic gateway. ADCs
exploit this process for lysosomal trafficking and payload release,
resulting in potent cytotoxicity in PSMA-positive cells (1). Similarly, RLT benefits from
intracellular trapping of radionuclides, such as 177Lu
and 225Ac, which increase the tumor absorbed dose
(2). Beyond these, PSMA-targeted
nanoparticle platforms, including supramolecular systems using
cucurbit-[8]-uril host-guest chemistry, can achieve selective
uptake and therapeutic efficacy in prostate cancer models (21). Taken together, these approaches
highlight PSMA as a versatile entry point for the precise delivery
of drugs, nucleic acids and imaging agents.
Clinical and translational
implications
PSMA's trafficking behavior has direct clinical
relevance. Its saturable, time-dependent uptake informs dosing
strategies to maximize tumor targeting whilst avoiding receptor
downregulation (12). Recycling of
internalized PSMA to the cell surface allows for repeated ligand
binding and payload delivery, a principle leveraged in fractionated
RLT to enhance the cumulative tumor dose (1). However, resistance may arise from
altered endosomal trafficking, lysosomal dysfunction or the
transcriptional downregulation of PSMA, all of which reduce payload
release. Co-targeting trafficking pathways or pharmacologically
enhancing PSMA surface expression may therefore improve therapeutic
efficacy and durability (17).
Regulation of PSMA expression and
transcriptional control
Androgen receptor (AR) signaling and
PSMA expression dynamics
FOLH1 expression is strongly regulated by AR
signaling and generally shows an inverse correlation with AR
activity. Under androgen stimulation, AR represses PSMA
transcription, reducing PSMA protein expression at the cell surface
(22). Conversely, androgen
deprivation therapy (ADT) or potent AR antagonists, such as
enzalutamide or apalutamide, induce a rapid ‘PSMA flare’,
characterized by increased transcription and surface expression
(23). Clinically, this enhances
lesion detectability on PSMA PET/CT and may improve the uptake of
PSMA-targeted radioligands (24).
Mechanistic studies have suggested that AR may
directly repress FOLH1 through intronic androgen response elements
and indirectly through MYC and homeobox B13 signaling (17). In castration-resistant prostate
cancer (CRPC), PSMA expression typically remains high or even
increases, reflecting altered AR dependency and partial escape from
AR-mediated repression.
Transcriptional and epigenetic
regulation
Beyond AR, FOLH1 expression is shaped by
prostate-specific cis-regulatory elements and inflammatory
pathways. Chronic NF-κB activation is a known driver of prostate
cancer progression (25), where
crosstalk between AR and NF-κB represents a key axis influencing
FOLH1 transcription (26). A
well-characterized intragenic enhancer within intron 3 of FOLH1,
known as the PSMA enhancer element, augments promoter activity in
PSMA-expressing cells (27,28).
Although direct NF-κB-driven activation of the FOLH1 promoter has
not been conclusively shown, PSMA can itself activate NF-κB through
integrin-dependent complexes, linking it to angiogenesis and
survival pathways (29,30).
Splice variants and functional
implications
PSMA undergoes alternative splicing, generating
isoforms with distinct subcellular localization and functional
properties.
However, the canonical full-length protein is
membrane-anchored and enzymatically active, enabling ligand
binding, internalization and downstream signaling. The splice
variant lacking exon 13 (PSMAΔ13) is predominantly retained in the
cytoplasm and fails to localize to the cell surface, resulting in
reduced availability of functional PSMA at the plasma membrane
(31).
Functionally, PSMAΔ13 has been proposed to act as a
dominant-negative isoform by interfering with membrane trafficking
or stability of the full-length protein, thereby decreasing
effective surface PSMA levels. Such a reduction is expected to
directly impair ligand binding, internalization and payload
delivery, providing a mechanistic basis for reduced sensitivity to
PSMA-targeted approaches, including RLT and antibody-drug
conjugates (ADCs).
Experimental studies have previously demonstrated
that PSMA protein expression alone is insufficient to confer its
biological effects. In prostate cancer models, expression of
catalytically inactive or structurally-altered PSMA mutants
abolished PSMA-mediated functional effects despite preserved
protein expression, indicating that intact enzymatic activity and
proper membrane localization are critical for PSMA function
(31). These findings support the
notion that PSMAΔ13 represents a functionally deficient PSMA state
rather than a simple quantitative reduction in expression.
Additional truncated PSMA isoforms have also been
reported, including variants that may be secreted and detectable in
circulation (1). Whilst the
biological and clinical significance of PSMAΔ13 and related
truncated isoforms remains to be fully elucidated, further studies
are required to define their prevalence, regulation and direct
impact on PSMA-targeted diagnostics and therapies.
Upregulation during progression and
spatial heterogeneity
PSMA expression generally increases with disease
progression, showing higher expression levels in tumors with higher
Gleason scores, advanced stage, and CRPC (32). However, marked spatial
heterogeneity complicates its clinical application. Studies have
shown site-specific variation in PSMA expression across metastases,
with major therapeutic implications (33).
Liver metastases frequently display PSMA-low or
PSMA-negative phenotypes, making them difficult to detect on
PSMA-PET (34,35). This ‘cold’ phenotype is associated
with the reduced uptake of RLT and inferior responses, possibly
reflecting lineage plasticity, neuroendocrine differentiation or
the epigenetic silencing of FOLH1 (36). Importantly, PSMA-low or
PSMA-negative phenotypes do not necessarily indicate the complete
loss of FOLH1 transcription, since alternative splicing, impaired
membrane trafficking or intracellular retention can all result in
functionally inactive PSMA despite preserved gene expression.
By contrast, bone and nodal metastases typically
maintain high expression levels, at time exceeding that of primary
tumors. Hope et al (37)
reported the high specificity of 68Ga-PSMA-11 PET/CT for
nodal disease, although sensitivity was reduced for lesions <5
mm. These findings highlight the need to integrate PSMA-PET with
histopathology and complementary diagnostics, such as dual-tracer
imaging, image-guided biopsy or circulating tumor DNA profiling.
Elucidating the molecular drivers of PSMA-low states will be
essential to optimize patient selection and develop strategies to
restore expression before therapy.
Tumor angiogenesis and TME
PSMA expression in the tumor
endothelium across cancer types
PSMA expression is not restricted to prostate
epithelium but is also strongly expressed in the endothelium of the
tumor-associated neovasculature across a number of other solid
tumors. Haffner et al (3)
reported vascular PSMA positivity in 66% of gastric and 85% of
colorectal adenocarcinomas, as well as in most metastases derived
from these primary tumors. Subsequent studies confirmed vascular
PSMA expression in renal cell carcinoma, glioblastoma,
non-small-cell lung cancer, pancreatic, breast and thyroid cancers
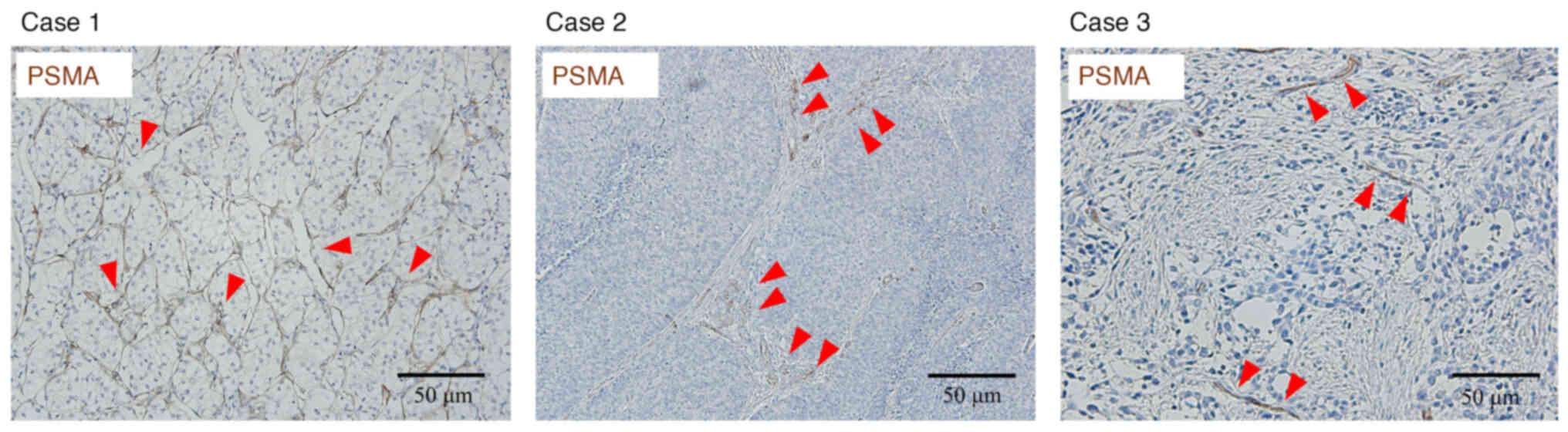
(38–44). Representative immunohistochemistry
images from three genitourinary cancer cases analyzed in the
present study (Fig. 2)
demonstrated that PSMA expression was consistently restricted to
the endothelium of tumor-associated neovessels across renal cell
carcinoma, upper tract urothelial carcinoma and bladder cancer. As
summarized in Table I, vascular
PSMA expression has also been reported across a wide range of solid
tumors, with substantial variability in reported frequencies (n/N)
depending on cancer type and study. However, in other urological
malignancies, such as testicular germ cell tumors or penile
carcinoma, PSMA expression has not been detected in
tumor-associated vasculature, indicating that endothelial PSMA
expression is not universally present across all cancer types.
 | Table I.Tumor-associated vascular PSMA
expression and clinical implications across cancer types. |
Table I.
Tumor-associated vascular PSMA
expression and clinical implications across cancer types.
| Tumor type | Vascular PSMA
expression, n/N (%) (ref.) | Detection method
(as reported) | Clinical
implications (ref.) |
|---|
| Prostate
cancer | 4/33 (12.1)
(5) | IHC (PSMA clone
3E6; CD31) | High diagnostic
performance of PSMA-PET for primary and metastatic disease, with
reduced sensitivity for liver metastases (34,35,65);
transient PSMA flare after ADT (70,71);
efficacy of PSMA-targeted RLT in mCRPC (67–69). |
| Bladder UC | 84/124 (67.7)
(48); present (n/N not reported)
(49) | IHC/IF (PSMA;
CD31) | PSMA-PET signal
largely vascular; vascular PSMA associated with prognosis and
therapeutic targeting potential (48,49,66). |
| UTUC | Detectable (n/N not
reported) (4) | IHC | Variable PSMA-PET
uptake compared with FDG; vascular PSMA expression may enable
targeting in selected cases (50,66). |
| Renal cell
carcinoma | 16/21 (76.2)
(23); 24/30 (80.0) (24); 45/45 (100.0) (6) | IHC (various
antibodies; CD31) | Non-prostate PSMA
uptake predominantly reflects vascular PSMA expression; rationale
for vasculature-directed strategies rather than tumor-cell imaging
(39,53,66). |
| Non-small cell lung
cancer | 135/275 (49.1)
(44) | IHC (various
antibodies; CD31/CD34) | PSMA-PET uptake
largely reflects vascular PSMA expression in tumor-associated
vasculature; potential angiogenesis-related imaging/targeting
(39,44,66). |
| Pancreatic
cancer | Reported (n/N not
reported) (4,38) | IHC (reported in
reviews) | Generally weak
PSMA-PET uptake; vascular PSMA expression suggests possible
targeting in selected settings (39,66). |
| Breast cancer | Vascular PSMA
expression reported (n/N not reported) (17,43) | IHC | PSMA-PET uptake may
be observed, including in brain metastases; vascular PSMA
expression represents a potential therapeutic target (39,66). |
| Thyroid cancer | Distant mets: 9/9
(100.0) (40); LN mets: 8/12
(66.7) (40); 120/267 (44.9)
(42) | IHC (PSMA;
CD31/CD34) | PSMA-PET uptake
predominantly vascular; therapeutic relevance particularly in RAIR
disease and metastases (39,40,42,66). |
| Glioblastoma | 32/32 (100.0)
(38); 5/5 (100.0) (41); 53/69 (76.8) (54) | IHC (3E6 or other
clones; CD31/VWF) | PSMA-PET and
therapeutic approaches primarily target vascular PSMA expression in
tumor-associated vasculature; neuro-oncological vascular targeting
rationale (38,39,63). |
| Gastric/colorectal
cancer | Gastric: 79/119
(66.4) (3); CRC: 110/130 (84.6)
(3) | IHC (PSMA clone
3E6; CD31) | Frequent vascular
PSMA expression supports feasibility of vascular imaging/targeting;
association with tumor grade in CRC (3,39,66). |
It should however be noted that vascular PSMA
expression is not uniformly observed across all tumor types, where
reported positivity rates vary substantially among studies and
malignancies. These observations underscore the need for further
stringent tumor-type-specific validation rather than assuming the
universal applicability of vascular PSMA-targeted imaging or
therapeutic strategies.
Functional models also support a causal role of
PSMA: in mice with transgenic adenocarcinoma of prostate, PSMA
knockout (Folh1-/-) reduced microvessel density, increased hypoxia
and apoptosis, impairing tumor growth (16). Altogether, all of the
aforementioned findings proposed PSMA as an active contributor to
pathological angiogenesis and a robust, pan-cancer vascular marker
with translational potential for imaging and therapy.
Paracrine and epigenetic regulation of
endothelial PSMA
Endothelial PSMA is actively regulated rather than
passively expressed. EVs shed from PSMA-positive tumor cells can
induce de novo PSMA expression in HUVECs, promoting
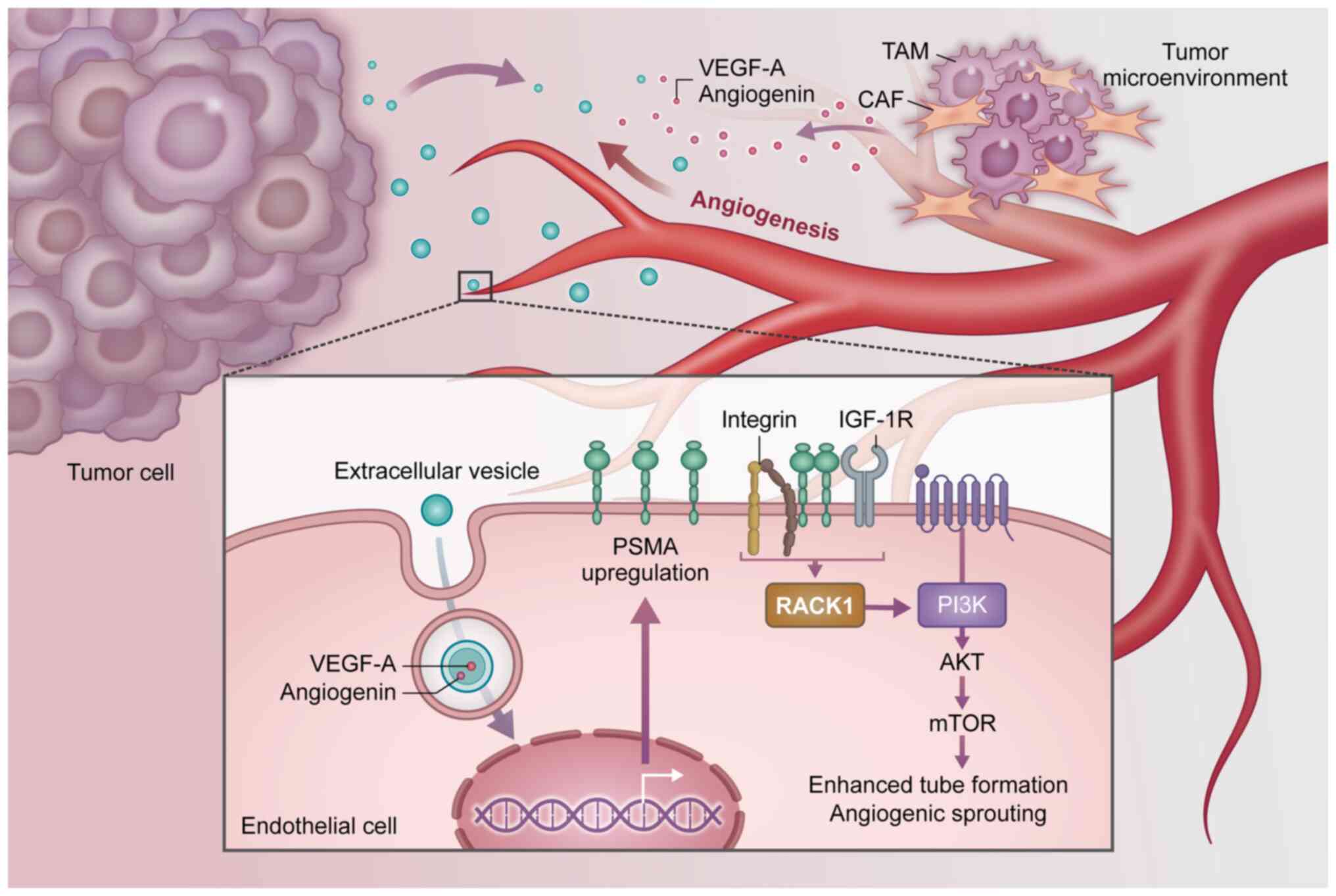
angiogenesis through NF-κB signaling (5). Machado et al (45) showed that PSMA-positive particles
can stimulate VEGF-A and angiogenin release with 4EBP1
phosphorylation, linking PSMA to mTOR-dependent translational
control. Similar findings were observed with renal cell
carcinoma-derived vesicles carrying growth differentiation factor
15 and myeloid-derived growth factor (6). Although these findings support a
paracrine regulatory model, the precise molecular mechanisms and
their relevance in human tumors remain to be fully elucidated.
These data support a paracrine model in which
tumor-derived vesicles provide both inductive signals and
pro-angiogenic cargo. Importantly, vascular PSMA expression
persists even in metastases lacking epithelial PSMA (36). However, epigenetic regulation also
contributes. Promoter hypermethylation or histone deacetylation can
silence FOLH1, whereas histone deacetylase (HDAC) inhibition can
restore endothelial PSMA (37).
Therefore, endothelial PSMA is likely dynamically regulated through
a paracrine-epigenetic feedback loop.
Downstream signaling pathways
PSMA can function not only as a peptidase but also
as a signaling hub (Fig. 3).
Conway et al (46)
previously showed that PSMA can regulates laminin-specific
β1-integrin-p21-activated kinase 1 signaling, which is essential
for endothelial invasion. Loss of PSMA was found to impair
extracellular matrix (ECM) invasion and angiogenesis (46). Caromile et al (16) demonstrated that PSMA can redirect
signaling from MAPK to PI3K-AKT-mTOR, enhancing endothelial
survival, proliferation and sprouting. In another study, Grant
et al (47) further showed
that PSMA can regulate angiogenesis in experimental models,
including in vitro endothelial assays and in vivo mouse
neovascularization models, at least in part independently of
canonical VEGF signaling pathways (5). Although the precise relationship
between PSMA-driven angiogenic signaling and the canonical
VEGF/VEGFR pathway remains incompletely understood, PSMA is
currently hypothesized to regulate angiogenesis through mechanisms
that are at least partially independent of VEGF signaling. These
studies position PSMA as a central integrator of ECM cues,
cytoskeletal remodeling and pro-survival pathways, reinforcing its
therapeutic relevance.
Emerging pan-cancer implications
Recognition of vascular PSMA across cancers broadens
its biological and clinical significance. Immunohistochemistry and
PET studies showed positivity in bladder cancer, upper tract
urothelial carcinoma (UTUC), glioblastoma, thyroid, pancreatic,
lung and breast tumors (38,39,47).
In urothelial carcinoma, vascular PSMA expression has been shown to
be positively correlated with increased microvessel density and
poor prognosis (48,49). These findings suggest that
prostate-derived therapies can be repurposed for vascular targeting
across tumor types. Bladder and UTUC show heterogeneous but
detectable vascular PSMA (4,50),
where vascular expression persists even in tumors with low
epithelial PSMA expression (such as in neuroendocrine variants)
(33). Site-specific heterogeneity
is likely critical. In prostate cancer, liver metastases frequently
lack PSMA expression, causing false-negative PET and poor RLT
responses (34,35), whereas bone and nodal metastases
maintain robust expression and remain excellent therapeutic targets
for exploiting PSMA. Knockout models have previously confirm that
the loss of PSMA can profoundly impair angiogenesis and tumor
growth (16).
Collectively, these data suggest that vascular PSMA
can serve as both a biomarker and therapeutic entry point. Future
strategies may combine vascular-directed RLT, ADCs or epigenetic
modulators (36). Integrating
PSMA-PET with complementary imaging and liquid biopsy may more
efficiently capture heterogeneity and refine patient selection.
Immunological roles of PSMA
PSMA expression and immunosuppressive
TME
PSMA expression has been associated with an
immunosuppressive TME. In prostate cancer and other solid tumors,
PSMA-positive lesions frequently show infiltration by
immunosuppressive myeloid populations and increased levels of
inhibitory cytokines, such as IL-10 and TGF-β (51,52).
These features suggest that PSMA-positive tumors create conditions
favoring immune evasion, by attenuating cytotoxic T cell activity.
Reviews further emphasized that PSMA targeting could reshape the
TME by reducing immunosuppressive signaling and altering myeloid
balance, although direct mechanistic evidence in patients remains
limited (51,53). There is also conceptual interest in
whether the enzymatic activity of PSMA in folate and glutamate
metabolism can influence T cell function, an area requiring further
study.
Correlations with tumor-infiltrating
lymphocytes (TILs), tumor-associated macrophages and immune
checkpoints
Transcriptomic and immunohistochemical studies have
indicated that PSMA-positive tumors tend to harbor low levels of
functional CD8+ TILs and an enrichment of exhausted or suppressive
phenotypes (51,52). In gliomas, endothelial PSMA
correlated with aberrant vascular morphology (53). Such vessels exhibit reduced
adhesion molecule expression and a disorganized structure,
impairing T cell trafficking and contributing to ‘vascular immune
exclusion’ (54). These
observations support a model in which both epithelial and
endothelial PSMA indirectly reinforce an immunologically cold
phenotype by altering the immune composition and restricting
lymphocyte access. However, direct causal links between PSMA
activity and immune modulation in patients remain largely
inferential and require further experimental validation.
Potential for vascular normalization
and immune checkpoint inhibitor (ICI) combination therapy
Given its vascular localization, PSMA is currently
being explored as a target for vascular normalization to improve
perfusion and immune infiltration, analogous to VEGF/VEGFR blockade
(52). Preclinical studies have
suggested that PSMA inhibition may synergize with ICIs, by
reversing immune exclusion and enhancing T cell recruitment
(55,56). In addition, PSMA-targeted
radiopharmaceutical therapy (RPT) can exert immunomodulatory
effects beyond cytotoxicity, including induction of immunogenic
cell death, antigen release and activation of innate pathways (such
as cyclic GMP-AMP synthase-stimulator of interferon genes)
(55–57). These mechanisms provide the
rationale for ongoing trials combining RPT with ICIs, as well as
the development of PSMA-targeted bispecific T-cell engagers (BiTEs)
and other immunotherapies that can convert PSMA-positive tumors
from immunologically ‘cold’ to ‘hot’ states (56,57).
Novel and exploratory functions of PSMA
Metabolic reprogramming
The enzymatic products of PSMA, glutamate and
monoglutamyl folate, connect it to central metabolic pathways.
Hydrolysis of NAAG elevates pericellular glutamate levels, which
fuels the TCA cycle through glutaminolysis and activates
mGluR1-PI3Kβ-AKT signaling, driving downstream phosphorylation of
mTOR effectors S6K and 4EBP1 (58,59).
Simultaneous parallel folate hydrolase activity generates
monoglutamyl folate for uptake by PCFT and RFC, augmenting
one-carbon metabolism and nucleotide synthesis whilst supplying
methyl donors required for epigenetic regulation (60).
Importantly, PSMA expression is highest in CRPC, a
stage marked by metabolic plasticity, suggesting that PSMA
functions as a metabolic gatekeeper aligning nutrient availability
with proliferative demand (18).
This dual contribution to energy and biosynthetic flux positions
PSMA at the intersection of metabolism, epigenetics and oncogenic
signaling.
Crosstalk with cancer-associated
fibroblasts (CAFs) and the ECM
Although direct evidence for PSMA-CAF interactions
remains limited, the role of CAFs in ECM remodeling and tumor
progression is well established (61,62).
Within this context, PSMA expression has been correlated with
enhanced MMP-2/9 activity and altered integrin signaling to
facilitate ECM degradation and invasion (63,64).
Furthermore, EVs carrying PSMA can be internalized by fibroblasts,
triggering the secretion of VEGF-A and FGF2 to promote a CAF-like
phenotype that is characterized by α-smooth muscle actin expression
and contractility (19). These
changes stiffen the stroma and generate invasive tracks, suggesting
that PSMA-mediated stromal reprogramming contributes to both
angiogenesis and metastatic potential.
Neuro-oncological significance
In the central nervous system (CNS), PSMA can
regulate synaptic NAAG turnover, generating glutamate to activate
mGluR and inotropic GluR signaling to modulate neuronal
excitability (8,15). Human biochemical studies further
support the neurophysiological importance of PSMA (14). These functions explain the low
PSMA-tracer uptake in normal brain but increased signals under
neuroinflammatory or blood-brain barrier-compromised
conditions.
Beyond physiological roles, PSMA expression can be
detected in both glioma cells and the tumor vasculature (41). Elevated PSMA activity may increase
extracellular glutamate levels, contributing to excitotoxic
neuronal injury and seizures. PSMA-derived glutamate can also
promote neuron-glioma synapse formation by
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid and
N-methyl-D-aspartate receptor activation, now recognized to be a
driver of glioma progression (63). Collectively, these findings link
PSMA-mediated neurotransmitter metabolism to neuro-oncological
processes and suggest therapeutic potential for PSMA inhibition in
brain tumors.
Clinical applications and therapeutic
implications
The major PSMA-targeted therapeutic strategies,
including radioligand therapy, antibody-drug conjugates, bispecific
antibodies, CAR-T cells and combination approaches, are summarized
in Table II.
 | Table II.Therapeutic strategies targeting
PSMA: Modalities, mechanisms and clinical development. |
Table II.
Therapeutic strategies targeting
PSMA: Modalities, mechanisms and clinical development.
| Modality | Examples | Mechanism | Clinical
status/notes |
|---|
| RLT |
177Lu-PSMA-617,
225Ac-PSMA-617 | Internalization →
delivery of β- or α-particles | VISION trial
demonstrated OS and PFS benefit; 225Ac-PSMA-617 was
effective in disease refractory to 177Lu-PSMA-617
therapy but was associated with xerostomia and cytopenia. |
| ADC | MLN2704, novel
cleavable-linker ADCs | PSMA-mediated
endocytosis → cytotoxic payload release | Early-phase trials;
toxicity (neuropathy and myelosuppression) improving with
next-generation designs |
| BiTEs | Pasotuxizumab | T-cell recruitment
to PSMA+ cells | Early trials: PSA
declines; CRS major AE |
| CAR-T cells | PSMA CAR-T | Engineered T cells
targeting PSMA | Antitumor activity
shown; on-target/off-tumor effects in salivary glands and
kidney |
| Nanoparticles | PSMA-targeted
liposomes, supra-molecular nanoparticles | Targeted drug/siRNA
delivery | Preclinical and
early translational studies |
| Combination
strategies | RLT + ICI; RLT +
PARPi; epigenetic priming | Synergistic effects
mediated by vascular normalization, radiosensitization and PSMA
upregulation | Trials ongoing |
PSMA-PET/CT: Current status and
limitations
PSMA- PET/CT with tracers, such as
68Ga-PSMA-11, 18F-DCFPyL and
18F-rhPSMA-7.3, have transformed prostate cancer staging
and restaging. The proPSMA trial demonstrated superior sensitivity
and accuracy over conventional imaging, detecting lesions even at
low PSA levels (65). This has
enabled metastasis-directed treatment and improved systemic therapy
selection. In this section, we focus on the current clinical
performance and limitations of PSMA-PET/CT.
Key limitations of PSMA tracker include
heterogeneous PSMA expression, particularly in liver metastases,
physiologic uptake in salivary glands, kidneys and ganglia and
restricted accessibility due to cost. Dual-tracer PET (PSMA +
fludeoxyglucose F-18) and quantitative PET metrics are currently
being explored to overcome these challenges and provide prognostic
information.
PSMA-PET uptake in non-prostate
malignancies and the role of vascular PSMA
PSMA uptake has been observed in multiple
non-prostate cancers, including renal, liver, brain and thyroid
malignancies (66). While
initially attributed to tumor-cell expression, immunohistochemistry
showed widespread PSMA in the tumor-associated vasculature
(3). Epigenetic regulation and
tumor-derived EVs also contribute to endothelial induction
(36,5). Therefore, a substantial fraction of
PSMA-PET signals in non-prostate cancers likely reflects vascular,
rather than epithelial, expression. This reframes PSMA-PET as a
tool for imaging angiogenesis and supports its use in selecting
patients for vascular-targeted therapies.
RLT: Efficacy and resistance
177Lu-PSMA-617 (Pluvicto™) has been
approved for metastatic CRPC following AR-pathway inhibition and
taxanes, based on results from the VISION trial, which showed
survival and progression-free survival benefits (67). The TheraP trial demonstrated higher
prostate-specific antigen (PSA) response rates and lower toxicity
compared with cabazitaxel (68).
α-emitter 225Ac-PSMA-617 has shown efficacy in
177Lu-refractory patients, with high PSA90 responses
(69), though xerostomia,
cytopenias and nephrotoxicity remain limiting. This toxicity
profile reflects physiological PSMA expression in non-malignant
tissues, most prominently in salivary and lacrimal glands, in
addition to in renal proximal tubules, where off-target uptake can
contribute to salivary gland dysfunction and renal toxicity.
Resistance arises from PSMA downregulation, altered
trafficking or lineage plasticity. Combination strategies with poly
(ADP-ribose) polymerase inhibitors, AR blockade, DNA repair
modulators and radiosensitizers are currently being investigated to
overcome resistance.
Next-generation immuno- and cell-based
therapeutics
PSMA is an appealing target for ADCs due to its
tumor selectivity and internalization. First-generation constructs,
such as MLN2704, were however limited by toxicity, but newer
designs with optimized linkers and payloads are in development
(1). BiTEs, such as pasotuxizumab,
have achieved durable PSA declines in early trials, though cytokine
release syndrome remains a challenge (69). Chimeric antigen receptor (CAR)
T-cell therapy cells targeting PSMA showed antitumor activity but
raised concerns of on-target/off-tumor effects in salivary glands
and kidneys. Logic-gated or transient CAR approaches are also
currently being explored to mitigate risk.
Vascular PSMA as a therapeutic
target
Consistent vascular expression of PSMA enables
anti-angiogenic therapy across various tumor types. Unlike
VEGF/VEGFR inhibitors, which frequently face obstructions of
adaptive resistance, PSMA-targeted approaches can ablate
PSMA-positive vessels or deliver drugs directly to the vasculature.
Preclinical studies with PSMA-targeted liposomal doxorubicin and
vascular-directed RLT have revealed their abilities of vessel
ablation and tumor necrosis (45).
Combinations with ICIs, VEGF blockade or CAR-T therapies may
further normalize vasculature, enhance immune infiltration and
improve tumor control.
Linking molecular function to patient
selection and therapy design
PSMA expression is dynamic and can be
pharmacologically modulated. Short-term ADT induces a transient
‘PSMA flare’, enhancing PET detectability and radioligand uptake
(70,71). Epigenetic interventions, such as
HDAC inhibition, can restore PSMA expression in low-expressing
tumors (36). These approaches
expand patient eligibility and support personalized strategies in
which PSMA expression is actively enhanced before imaging or
therapy.
Toward a universal anti-angiogenic
strategy
Because vascular PSMA expression is pan-cancer, it
represents a potential universal target for angiogenesis-directed
therapy. Selective ablation of abnormal PSMA-positive vessels could
reduce hypoxia-driven aggressiveness whilst providing a route for
vascular-specific drug delivery. In combination with immunotherapy,
PSMA vascular targeting may normalize the vasculature, promote
immune infiltration and produce durable antitumor responses,
positioning PSMA as a candidate for broad-spectrum anti-angiogenic
strategies.
Conclusions
In conclusion, this review revisits PSMA through a
unified conceptual framework in which its metabolic enzyme
activity, non-enzymatic scaffold signaling, tumor-associated
angiogenesis and immunological consequences are functionally
interconnected, providing a coherent biological basis for its
diverse clinical manifestations and therapeutic potential.
PSMA has progressed from being considered only as a
biomarker of prostate cancer to being recognized as an active
driver of tumor biology. Its dual role as a
glutamate/folate-processing ectoenzyme and as a signaling scaffold
situates PSMA at the crossroads of tumor metabolism, angiogenesis,
immune modulation and therapeutic response. Evidence across
preclinical and clinical studies shows that PSMA can contribute to
remodeling the TME, enhancing neovascularization, supporting
stromal activation and shaping immune cell infiltration, thereby
exerting a functional influence beyond its diagnostic utility.
Looking forward, several priorities emerge for
advancing PSMA biology toward clinical translation. First, a deeper
mechanistic understanding of PSMA heterogeneity, including
alternative splicing, functional loss of surface expression and
context-dependent regulation, will be essential to explain
discordant imaging findings and variable therapeutic responses.
Second, clarifying how PSMA-driven angiogenic signaling interfaces
with canonical VEGF/VEGFR pathways will inform rational
combinations of PSMA-targeted and anti-angiogenic therapies. Third,
strategies to mitigate off-target toxicities, particularly salivary
gland and renal uptake in RLT, remain critical for improving
treatment durability and patient quality of life.
At the same time, advances in precision medicine are
reshaping the therapeutic landscape of prostate cancer.
PSMA-targeted RLT now joins PARP inhibition, immune checkpoint
blockade and AKT inhibitors as components of biomarker-driven
treatment, whilst novel strategies for molecular subsets, such as
CDK12-altered or neuroendocrine tumors are emerging. The
integration of liquid biopsy, multi-omics approaches and
AI-assisted imaging-genomics fusion promises to refine patient
selection and expand the reach of personalized medicine.
Taken together, PSMA should be viewed not only as a
diagnostic tracer target, but as a versatile biological switch and
therapeutic entry point capable of influencing multiple hallmarks
of cancer. The convergence of PSMA-targeted modalities with
systemic therapies and precision medicine strategies offers a
realistic path to improve outcomes in prostate cancer and
potentially other solid tumors. Whether PSMA ultimately fulfills
its promise as a pan-cancer anti-angiogenic and immunomodulatory
target will depend on sustained translational efforts in the coming
decade, but its central role in tumor biology is now firmly
established.
Acknowledgements
Not applicable.
Funding
This work was supported by JSPS KAKENHI (grant nos. 22K09449 and
22KK0135), Nippon Shinyaku Research Grant, and Sumitomo Foundation
Grant for Basic Science Research Projects.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
RW wrote the manuscript. NM and TK revised the
manuscript. ToS and TaS contributed to the conceptualization of the
study and critically revised the manuscript. RW and NM confirm the
authenticity of all the raw data. All authors have read and
approved the final version of the manuscript.
Ethics approval and consent to
participate
This study was approved by the Institutional Review
Board of Ehime University Hospital (IRB no. 2201011; Toon, Japan).
Informed consent was obtained from all participants using an
opt-out approach in accordance with institutional guidelines.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ADC
|
antibody-drug conjugate
|
|
ADT
|
androgen deprivation therapy
|
|
AR
|
androgen receptor
|
|
BiTE
|
bispecific T-cell engager
|
|
CAF
|
cancer-associated fibroblast
|
|
CAR-T
|
chimeric antigen receptor T cell
|
|
CRPC
|
castration-resistant prostate
cancer
|
|
ECM
|
extracellular matrix
|
|
EV
|
extracellular vesicle
|
|
FDG
|
fluorodeoxyglucose
|
|
FOLH1
|
folate hydrolase 1
|
|
GCPII
|
glutamate carboxypeptidase II
|
|
HDAC
|
histone deacetylase
|
|
ICI
|
immune checkpoint inhibitor
|
|
mGluR1
|
metabotropic glutamate receptor 1
|
|
NAAG
|
N-acetylaspartylglutamate
|
|
NAALADase I
|
N-acetylated-α-linked acidic
dipeptidase
|
|
PARP
|
poly(ADP-ribose) polymerase
|
|
PCFT
|
proton-coupled folate transporter
|
|
PET/CT
|
positron emission tomography/computed
tomography
|
|
PSA
|
prostate-specific antigen
|
|
PSMA
|
prostate-specific membrane
antigen
|
|
RFC
|
reduced folate carrier
|
|
RLT
|
radioligand therapy
|
|
RPT
|
radiopharmaceutical therapy
|
|
TAM
|
tumor-associated macrophage
|
|
TCA cycle
|
tricarboxylic acid cycle
|
|
TIL
|
tumor-infiltrating lymphocyte
|
|
TME
|
tumor microenvironment
|
|
UTUC
|
upper tract urothelial carcinoma
|
References
|
1
|
Maes J, Gesquière S, De Spiegeleer A, Maes
A and Van de Wiele C: Prostate-specific membrane antigen biology
and pathophysiology in prostate carcinoma, an update: Potential
implications for targeted imaging and therapy. Int J Mol Sci.
25:97552024. View Article : Google Scholar
|
|
2
|
Hyväkkä A, Virtanen V, Kemppainen J,
Grönroos TJ, Minn H and Sundvall M: More than meets the eye:
Scientific rationale behind molecular imaging and therapeutic
targeting of prostate-specific membrane antigen (PSMA) in
metastatic prostate cancer and beyond. Cancers (Basel).
13:22442021. View Article : Google Scholar
|
|
3
|
Haffner MC, Kronberger IE, Ross JS,
Sheehan CE, Zitt M, Mühlmann G, Ofner D, Zelger B, Ensinger C, Yang
XJ, et al: Prostate-specific membrane antigen expression in the
neovasculature of gastric and colorectal cancers. Hum Pathol.
40:1754–1761. 2009. View Article : Google Scholar
|
|
4
|
Van de Wiele C, Sathekge M, de Spiegeleer
B, De Jonghe PJ, Debruyne PR, Borms M, Beels L and Maes A: PSMA
expression on neovasculature of solid tumors. Histol Histopathol.
35:919–927. 2020.
|
|
5
|
Watanabe R, Maekawa M, Kiyoi T, Kurata M,
Miura N, Kikugawa T, Higashiyama S and Saika T: PSMA-positive
membranes secreted from prostate cancer cells have potency to
transform vascular endothelial cells into an angiogenic state.
Prostate. 81:1390–1401. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Watanabe R, Kagimoto K, Chosei M, Sakaue
T, Kurata M, Miura N, Kitazawa R, Kikugawa T, Higashiyama S and
Saika T: Vesicles secreted by renal cell carcinoma cells cause
vascular endothelial cells to express PSMA and drive tumor
progression. Cells. 14:1652025. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Israeli RS, Powell CT, Fair WR and Heston
WD: Molecular cloning of a complementary DNA encoding a
prostate-specific membrane antigen. Cancer Res. 53:227–230.
1993.PubMed/NCBI
|
|
8
|
Bzdega T, Turi T, Wroblewska B, She D,
Chung HS, Kim H and Neale JH: Molecular cloning of a peptidase
against N-acetylaspartylglutamate from a rat hippocampal cDNA
library. J Neurochem. 69:2270–2277. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Pinto JT, Suffoletto BP, Berzin TM, Qiao
CH, Lin S, Tong WP, May F, Mukherjee B and Heston WD:
Prostate-specific membrane antigen: A novel folate hydrolase in
human prostatic carcinoma cells. Clin Cancer Res. 2:1445–1451.
1996.PubMed/NCBI
|
|
10
|
Davis MI, Bennett MJ, Thomas LM and
Bjorkman PJ: Crystal structure of prostate-specific membrane
antigen, a tumor marker and peptidase. Proc Natl Acad Sci USA.
102:5981–5986. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Barinka C, Rovenská M, Mlcochová P,
Hlouchová K, Plechanovová A, Majer P, Tsukamoto T, Slusher BS,
Konvalinka J and Lubkowski J: Structural insight into the
pharmacophore pocket of human glutamate carboxypeptidase II. J Med
Chem. 50:3267–3273. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Liu H, Rajasekaran AK, Moy P, Xia Y, Kim
S, Navarro V, Rahmati R and Bander NH: Constitutive and
antibody-induced internalization of prostate-specific membrane
antigen. Cancer Res. 58:4055–4060. 1998.PubMed/NCBI
|
|
13
|
Anilkumar G, Rajasekaran SA, Wang S,
Hankinson O, Bander NH and Rajasekaran AK: Prostate-specific
membrane antigen association with filamin A modulates its
internalization and NAALADase activity. Cancer Res. 63:2645–2648.
2003.PubMed/NCBI
|
|
14
|
Tsai G, Passani LA, Slusher BS, Carter R,
Baer L, Kleinman JE and Coyle JT: Abnormal excitatory
neurotransmitter metabolism in schizophrenic brains. Arch Gen
Psychiatry. 52:829–836. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Slusher BS, Vornov JJ, Thomas AG, Hurn PD,
Harukuni I, Bhardwaj A, Traystman RJ, Robinson MB, Britton P, Lu
XC, et al: Selective inhibition of NAALADase, which converts NAAG
to glutamate, reduces ischemic brain injury. Nat Med. 5:1396–1402.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Caromile LA, Dortche K, Rahman MM, Grant
CL, Stoddard C, Ferrer FA and Shapiro LH: PSMA redirects cell
survival signaling from the MAPK to the PI3K-AKT pathways to
promote the progression of prostate cancer. Sci Signal.
10:eaag33262017. View Article : Google Scholar
|
|
17
|
Boixareu C, Taha T, Venkadakrishnan VB, de
Bono J and Beltran H: Targeting the tumour cell surface in advanced
prostate cancer. Nat Rev Urol. 22:569–589. 2025. View Article : Google Scholar
|
|
18
|
Sedlák F, Kvasnička A, Marešová B,
Brumarová R, Dobešová D, Dostálová K, Šrámková K, Pehr M, Šácha P,
Friedecký D and Konvalinka J: Parallel metabolomics and lipidomics
of a PSMA/GCPII deficient mouse model reveal alteration of NAAG
levels and brain lipid composition. ACS Chem Neurosci.
15:1342–1355. 2024. View Article : Google Scholar
|
|
19
|
Pellegrino S and Fonti R: A look into the
future: The role of PSMA beyond prostate cancer. Eur J Nucl Med Mol
Imaging. 51:278–280. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Rajasekaran SA, Anilkumar G, Oshima E,
Bowie JU, Liu H, Heston W, Bander NH and Rajasekaran AK: A novel
cytoplasmic tail MXXXL motif mediates the internalization of
prostate-specific membrane antigen. Mol Biol Cell. 14:4835–4845.
2003. View Article : Google Scholar
|
|
21
|
Zhang X, Qi S, Liu D, Du J and Jin J:
PSMA-Targeted supramolecular nanoparticles prepared from
cucurbit[8]uril-based ternary host-guest recognition for prostate
cancer therapy. Front Chem. 10:8475232022. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Minner S, Wittmer C, Graefen M, Salomon G,
Steuber T, Haese A, Huland H, Bokemeyer C, Yekebas E, Dierlamm J,
et al: High level PSMA expression is associated with early PSA
recurrence in surgically treated prostate cancer. Prostate.
71:281–288. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Meller B, Bremmer F, Sahlmann CO, Hijazi
S, Bouter C, Trojan L, Meller J and Thelen P: Alterations in
androgen deprivation enhanced prostate-specific membrane antigen
(PSMA) expression in prostate cancer cells as a target for
diagnostics and therapy. EJNMMI Res. 5:662015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Calais J, Kishan AU, Cao M, Fendler WP,
Eiber M, Herrmann K, Ceci F, Reiter RE, Rettig MB, Hegde JV, et al:
Potential impact of 68Ga-PSMA-11 PET/CT on the planning of
definitive radiation therapy for prostate cancer. J Nucl Med.
59:1714–1721. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Staal J and Beyaert R: Inflammation and
NF-κB signaling in prostate cancer: Mechanisms and clinical
implications. Cells. 7:1222018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Basílio J, Hochreiter B, Hoesel B,
Sheshori E, Mussbacher M, Hanel R and Schmid JA: Antagonistic
functions of androgen receptor and NF-κB in prostate
cancer-experimental and computational analyses. Cancers (Basel).
14:61642022. View Article : Google Scholar
|
|
27
|
Watt F, Martorana A, Brookes DE, Ho T,
Kingsley E, O'Keefe DS, Russell PJ, Heston WD and Molloy PL: A
tissue-specific enhancer of the prostate-specific membrane antigen
gene, FOLH1. Genomics. 73:243–254. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Noss KR, Wolfe SA and Grimes SR:
Upregulation of prostate specific membrane antigen/folate hydrolase
transcription by an enhancer. Gene. 285:247–256. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Gao Y, Zheng H, Li L, Feng M, Chen X, Hao
B, Lv Z, Zhou X and Cao Y: Prostate-specific membrane antigen
(PSMA) promotes angiogenesis of glioblastoma through interacting
with ITGB4 and regulating NF-κB signaling pathway. Front Cell Dev
Biol. 9:5983772021. View Article : Google Scholar
|
|
30
|
Perico ME, Grasso S, Brunelli M,
Martignoni G, Munari E, Moiso E, Fracasso G, Cestari T, Naim HY,
Bronte V, et al: Prostate-specific membrane antigen (PSMA)
assembles a macromolecular complex regulating growth and survival
of prostate cancer cells ‘in vitro’ and correlating with
progression ‘in vivo’. Oncotarget. 7:74189–74202. 2016. View Article : Google Scholar
|
|
31
|
Ghosh A, Wang X, Klein E and Heston WD:
Novel role of prostate-specific membrane antigen in suppressing
prostate cancer invasiveness. Cancer Res. 65:727–731. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Silver DA, Pellicer I, Fair WR, Heston WD
and Cordon-Cardo C: Prostate-specific membrane antigen expression
in normal and malignant human tissues. Clin Cancer Res. 3:81–85.
1997.PubMed/NCBI
|
|
33
|
Bakht MK, Yamada Y, Ku SY, Venkadakrishnan
VB, Korsen JA, Kalidindi TM, Mizuno K, Ahn SH, Seo JH, Garcia MM,
et al: Landscape of prostate-specific membrane antigen
heterogeneity and regulation in AR-positive and AR-negative
metastatic prostate cancer. Nat Cancer. 4:699–715. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Damjanovic J, Janssen JC, Prasad V,
Diederichs G, Walter T, Brenner W and Makowski MR: 68Ga-PSMA-PET/CT
for the evaluation of liver metastases in patients with prostate
cancer. Cancer Imaging. 19:372019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Mattoni S, Farolfi A, Formaggio F, Bruno
G, Caroli P, Cerci JJ, Eiber M, Fendler WP, Golfieri R, Herrmann K,
et al: PSMA PET for the evaluation of liver metastases in
castration-resistant prostate cancer patients: A multicenter
retrospective study. Cancers (Basel). 14:56802022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Sayar E, Patel RA, Coleman IM, Roudier MP,
Zhang A, Mustafi P, Low JY, Hanratty B, Ang LS, Bhatia V, et al:
Reversible epigenetic alterations mediate PSMA expression
heterogeneity in advanced metastatic prostate cancer. JCI Insight.
8:e1629072023. View Article : Google Scholar
|
|
37
|
Hope TA, Eiber M, Armstrong WR, Juarez R,
Murthy V, Lawhn-Heath C, Behr SC, Zhang L, Barbato F, Ceci F, et
al: Diagnostic accuracy of 68Ga-PSMA-11 PET for pelvic nodal
metastasis detection prior to radical prostatectomy and pelvic
lymph node dissection: A multicenter prospective phase 3 imaging
trial. JAMA Oncol. 7:1635–1642. 2021. View Article : Google Scholar
|
|
38
|
Wernicke AG, Edgar MA, Lavi E, Liu H,
Salerno P, Bander NH and Gutin PH: Prostate-specific membrane
antigen as a potential novel vascular target for treatment of
glioblastoma multiforme. Arch Pathol Lab Med. 135:1486–1489. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Fragomeni RA, Amir T, Sheikhbahaei S,
Harvey SC, Javadi MS, Solnes LB, Kiess AP, Allaf ME, Pomper MG,
Gorin MA and Rowe SP: Imaging of nonprostate cancers using
PSMA-targeted radiotracers: Rationale, current state of the field,
and a call to arms. J Nucl Med. 59:871–877. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Moore M, Panjwani S, Mathew R, Crowley M,
Liu YF, Aronova A, Finnerty B, Zarnegar R, Fahey TJ III and
Scognamiglio T: Well-differentiated thyroid cancer neovasculature
expresses prostate-specific membrane antigen-a possible novel
therapeutic target. Endocr Pathol. 28:339–344. 2017. View Article : Google Scholar
|
|
41
|
Nomura N, Pastorino S, Jiang P, Lambert G,
Crawford JR, Gymnopoulos M, Piccioni D, Juarez T, Pingle SC, Makale
M and Kesari S: Prostate specific membrane antigen (PSMA)
expression in primary gliomas and breast cancer brain metastases.
Cancer Cell Int. 14:262014. View Article : Google Scholar
|
|
42
|
Bychkov A, Vutrapongwatana U, Tepmongkol S
and Keelawat S: PSMA expression by microvasculature of thyroid
tumors-Potential implications for PSMA theranostics. Sci Rep.
7:52022017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Kasoha M, Unger C, Solomayer EF, Bohle RM,
Zaharia C, Khreich F, Wagenpfeil S and Juhasz-Böss I:
Prostate-specific membrane antigen (PSMA) expression in breast
cancer and its metastases. Clin Exp Metastasis. 34:479–490. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Schmidt LH, Heitkötter B, Schulze AB,
Schliemann C, Steinestel K, Trautmann M, Marra A, Hillejan L, Mohr
M, Evers G, et al: Prostate specific membrane antigen (PSMA)
expression in non-small cell lung cancer. PLoS One.
12:e01862802017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Machado CML, Skubal M, Haedicke K, Silva
FP, Stater EP, Silva TLAO, Costa ET, Masotti C, Otake AH, Andrade
LNS, et al: Membrane-derived particles shed by PSMA-positive cells
function as pro-angiogenic stimuli in tumors. J Control Release.
364:312–325. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Conway RE, Petrovic N, Li Z, Heston W, Wu
D and Shapiro LH: Prostate-specific membrane antigen regulates
angiogenesis by modulating integrin signal transduction. Mol Cell
Biol. 26:5310–5324. 2006. View Article : Google Scholar
|
|
47
|
Grant CL, Caromile LA, Ho V, Durrani K,
Rahman MM, Claffey KP, Fong GH and Shapiro LH: Prostate specific
membrane antigen (PSMA) regulates angiogenesis independently of
VEGF during ocular neovascularization. PLoS One. 7:e412852012.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Li Y, Zhang K, Yang F, Jiao D, Li M, Zhao
X, Xu C, Liu S, Li H, Shi S, et al: Prognostic value of
vascular-expressed PSMA and CD248 in urothelial carcinoma of the
bladder. Front Oncol. 11:7710362021. View Article : Google Scholar
|
|
49
|
Schreiber H, Hänze J, Nimphius W, Verburg
FA, Luster M, Hofmann R and Hegele A: Prostate specific membrane
antigen (PSMA) in urothelial cell carcinoma (UCC) is associated
with tumor grading and staging. J Cancer Res Clin Oncol.
146:305–313. 2020. View Article : Google Scholar
|
|
50
|
Lin BH, Chen SH, Chen SM, Qiu QR, Gao RC,
Wei Y, Zheng QS, Miao WB and Xu N: Head-to-head comparisons of
68Ga-PSMA-11 and 18F-FDG PET/CT in evaluating patients with upper
tract urothelial carcinoma: A prospective pilot study. Int Urol
Nephrol. 55:2753–2764. 2023. View Article : Google Scholar
|
|
51
|
Liu D, Wang L and Guo Y: Advances in and
prospects of immunotherapy for prostate cancer. Cancer Lett.
601:2171552024. View Article : Google Scholar
|
|
52
|
Ge R, Wang Z and Cheng L: Tumor
microenvironment heterogeneity an important mediator of prostate
cancer progression and therapeutic resistance. NPJ Precis Oncol.
6:312022. View Article : Google Scholar
|
|
53
|
Puik JR, Le C, Kazemier G, Oprea-Lager DE,
Swijnenburg RJ, Giovannetti E, Griffioen AW and Huijbers EJ:
Prostate-specific membrane antigen as target for
vasculature-directed therapeutic strategies in solid tumors. Crit
Rev Oncol Hematol. 205:1045562025. View Article : Google Scholar
|
|
54
|
Maguid MS, Saad El Dine MG, Gabal SM and
Fandoud SM: Prostate-specific membrane antigen (PSMA) expression in
the neovasculature of high grade gliomas (Histopathological and
Immunohistochemical Study). Asian Pac J Cancer Prev. 24:1797–1808.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Bellavia MC, Patel RB and Anderson CJ:
Combined targeted radiopharmaceutical therapy and immune checkpoint
blockade: From preclinical advances to the clinic. J Nucl Med.
63:1636–1641. 2022.PubMed/NCBI
|
|
56
|
Altunay B, Schäfer L, Morgenroth A, Peña
Q, Lammers T, Saar M, Mottaghy FM and Lütje S: Combining
PSMA-targeted radiopharmaceutical therapy with immunotherapy. J
Nucl Med. 28:1522–1527. 2025. View Article : Google Scholar
|
|
57
|
Jiao R and Dadachova E: Combination of
radioligand therapy and immunotherapy: How to make it work in
clinic? Immunotargets Ther. 14:755–759. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Sheehan B, Guo C, Neeb A, Paschalis A,
Sandhu S and de Bono JS: Prostate-specific membrane antigen biology
in lethal prostate cancer and its therapeutic implications. Eur
Urol Focus. 8:1157–1168. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Palamiuc L and Emerling BM: PSMA brings
new flavors to PI3K signaling: A role for glutamate in prostate
cancer. J Exp Med. 215:17–19. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
O'Keefe DS, Bacich DJ, Huang SS and Heston
WDW: A perspective on the evolving story of PSMA biology,
PSMA-based imaging, and endoradiotherapeutic strategies. J Nucl
Med. 59:1007–1013. 2018. View Article : Google Scholar
|
|
61
|
Wright K, Ly T, Kriet M, Czirok A and
Thomas SM: Cancer-associated fibroblasts: Master tumor
microenvironment modifiers. Cancers (Basel). 15:18992023.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Bonollo F, Thalmann GN, Kruithof-de Julio
M and Karkampouna S: The role of cancer-associated fibroblasts in
prostate cancer tumorigenesis. Cancers (Basel). 12:18872020.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
McBriar JD, Shafiian N, Scharf S, Boockvar
JA and Wernicke AG: Prostate-specific membrane antigen use in
glioma management: Past, present, and future. Clin Nucl Med.
49:806–816. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Hameed MY, Gul M and Chaudhry A, Muzaffar
H, Sheikh M, Chee W, Ayyash S, Ayyash J, Al-Hindi M, Shahare H and
Chaudhry A: From oncogenesis to theranostics: The transformative
role of PSMA in prostate cancer. Cancers (Basel). 16:30392024.
View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Hofman MS, Lawrentschuk N, Francis RJ,
Tang C, Vela I, Thomas P, Rutherford N, Martin JM, Frydenberg M,
Shakher R, et al: Prostate-specific membrane antigen PET-CT in
patients with high-risk prostate cancer before curative-intent
surgery or radiotherapy (proPSMA): A prospective, randomised,
multicentre study. Lancet. 395:1208–1216. 2020. View Article : Google Scholar
|
|
66
|
de Galiza Barbosa F, Queiroz MA, Nunes RF,
Costa LB, Zaniboni EC, Marin JFG, Cerri GG and Buchpiguel CA:
Nonprostatic diseases on PSMA PET imaging: A spectrum of benign and
malignant findings. Cancer Imaging. 20:232020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Sartor O, de Bono J, Chi KN, Fizazi K,
Herrmann K, Rahbar K, Tagawa ST, Nordquist LT, Vaishampayan N,
El-Haddad G, et al: Lutetium-177-PSMA-617 for metastatic
castration-resistant prostate cancer. N Engl J Med. 385:1091–1103.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Kratochwil C, Bruchertseifer F, Rathke H,
Bronzel M, Apostolidis C, Weichert W, Haberkorn U, Giesel FL and
Morgenstern A: Targeted α-therapy of metastatic
castration-resistant prostate cancer with 225Ac-PSMA-617: Dosimetry
estimate and empiric dose finding. J Nucl Med. 58:1624–1631. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Sridaran D, Bradshaw E, DeSelm C,
Pachynski R, Mahajan K and Mahajan NP: Prostate cancer
immunotherapy: Improving clinical outcomes with a multi-pronged
approach. Cell Rep Med. 4:1011992023. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Ettala O, Malaspina S, Tuokkola T, Luoto
P, Löyttyniemi E, Boström PJ and Kemppainen J: Prospective study on
the effect of short-term androgen deprivation therapy on PSMA
uptake evaluated with 68Ga-PSMA-11 PET/MRI in men with
treatment-naïve prostate cancer. Eur J Nucl Med Mol Imaging.
47:665–673. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Vaz S, Hadaschik B, Gabriel M, Herrmann K,
Eiber M and Costa D: Influence of androgen deprivation therapy on
PSMA expression and PSMA-ligand PET imaging of prostate cancer
patients. Eur J Nucl Med Mol Imaging. 47:9–15. 2020. View Article : Google Scholar : PubMed/NCBI
|