Introduction
Licorice is a well-known herb that has been widely
used in traditional Chinese medicine. According to the Chinese
Pharmacopoeia (1), three original
plants from the family Leguminosae, Glycyrrhiza uralensis
Fisch, Glycyrrhiza inflata Bat. and Glycyrrhiza
glabra L. are prescribed as licorice. Numerous studies indicate
that the most pharmacologically important bioactive compounds in
licorice include ~20 types of triterpenoids, 300 types of
flavonoids and various polysaccharides (2,3).
Among these, the triterpenoid glycyrrhizic acid (GL) is
particularly notable. GL is a sweet-tasting compound extensively
used as a flavoring agent in the food industry. Upon hydrolysis, GL
yields two molecules of glucuronic acid and one molecule of
glycyrrhetinic acid (GA). In the intestines, GL undergoes enzymatic
conversion to GA-3-O-mono-β-d-glucuronide, a derivative with
enhanced bioavailability and metabolic potential (4).
GL-based therapies, including compound GL tablets
and compound GL injections, have been prescribed to treat a number
of inflammatory and immune diseases, including viral and autoimmune
hepatitis (5). The therapeutic
effects of GL have also been validated in diseases such as
cirrhosis, eczema, chronic urticaria, psoriasis, and ulcerative
colitis (6–8). Furthermore, as an inhibitor of
cellular signal transduction molecules that has been demonstrated
to suppress angiogenesis and the secretion of tumor-associated
cytokines, GL effectively inhibits the migration and invasion of
various types of cancer cells (9).
Due to its low toxicity and minimal side effects, GL exhibits a
wide range of potential therapeutic applications (10).
The present review discusses the effects of GL on
intracellular and extracellular inflammatory signaling pathways,
aiming to provide a theoretical foundation for the clinical
applications of GL and its formulations. Furthermore, the present
review highlights emerging therapeutic strategies involving GL,
underscoring its potential as a cornerstone for developing new
treatments across a spectrum of diseases.
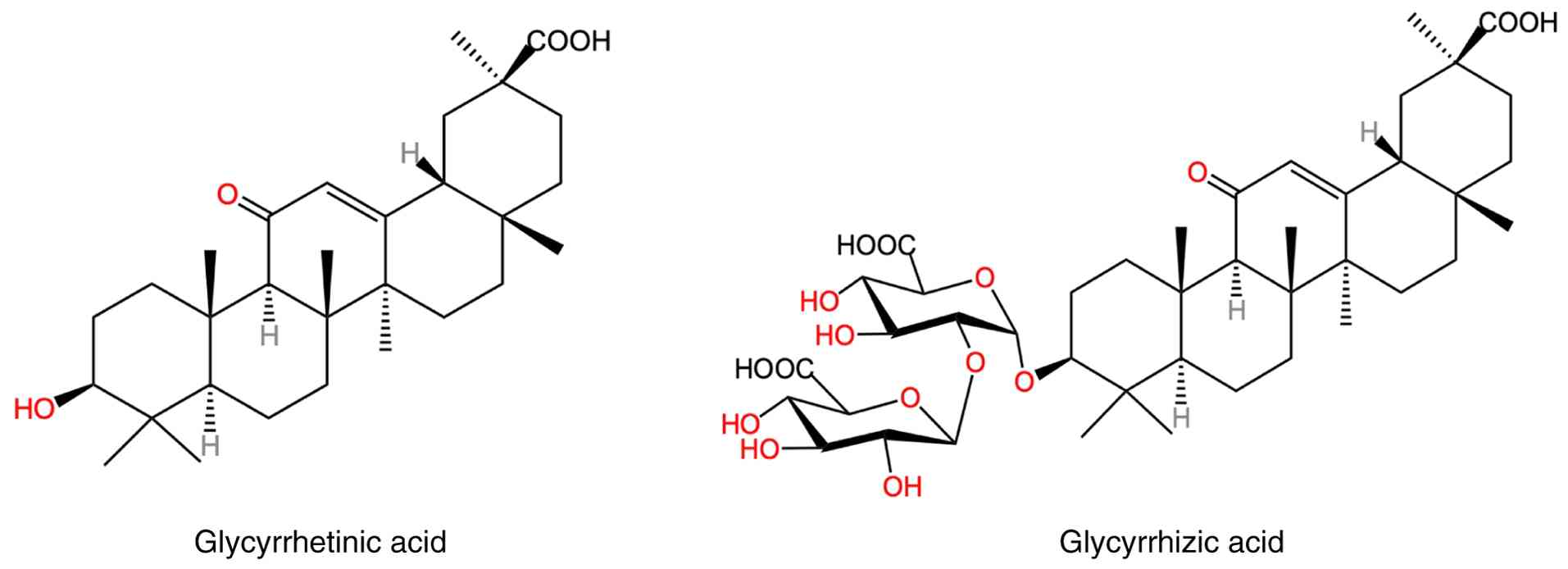
Chemical structure and pharmacokinetics of
GL
GL, also known as glycyrrhizin, is the principal
bioactive component of licorice. This compound is a pentacyclic
triterpenoid saponin composed of one molecule of GA conjugated with
two molecules of glucuronic acid (11). The molecular formula of GL is
C42H62O16, with a relative
molecular weight of 822.93 kDa, whereas its derivative GA, which
has a relative molecular weight of 470.69 kDa, has the molecular
formula C30H46O4 (Fig. 1). The structure of GL includes five
interconnected rings termed rings A-E, with functional groups
present on rings A, C and E; hydroxyl and carboxyl groups are
present at the 3rd and 30th carbon positions, respectively. These
positions are the primary sites for chemical modification of GL,
enabling the synthesis of various GA derivatives with potential
therapeutic applications (12).
Following its intake, the aglycone section of GL is
hydrolyzed by glucuronidase to form two isomers, 18α-GA and 18β-GA
(13). The α-form has higher
lipophilicity than the β-form, making it more likely to bind to
receptor proteins within the body. The conformation of the D and E
rings closely resemble that of prednisolone, allowing GA to easily
bind to target cells of steroid hormones, which confers its
antitoxic effects (14). 18β-GA,
with a structure similar to cortisol, can mimic the activity of
cortisol by inhibiting 11β-hydroxysteroid dehydrogenase activity
(11b-HSDs) (12). This isomer also
inhibits the activation of the classical complement pathway and
reduces reactive oxygen species (ROS) levels, exerting
anti-inflammatory and glucocorticoid hormone-like effects (6).
When administered orally, GL is broken down by
β-D-glucuronidase in the intestines to produce GA. Following
intravenous administration, GL is metabolized by β-D-glucuronidase
in liver lysosomes to produce 3-monoglucuronic GA, which is
subsequently distributed via the enterohepatic circulation. GL is
metabolized to GA under the action of gut bacteria and is
subsequently reabsorbed through the intestinal wall to exert its
pharmacological effects (15).
High-mobility group box-1 (HMGB1)
HMGB1 is a nuclear protein that functions as a
structural chromatin-binding factor, contributing to the
maintenance of nucleosome architecture and the regulation of gene
transcription. In response to various stimuli, HMGB1 can be
actively secreted by immune and non-immune cells or passively
released into the extracellular milieu (16). HMGB1 is a multi-functional
regulator: Intracellularly, it acts as an architectural
chromatin-binding factor. It can be passively released by damaged
or virus-infected cells, or actively secreted by innate immune
cells in response to exogenous bacterial products (e.g. endotoxin
or CpG-DNA) or endogenous inflammatory stimuli (17). Extracellular HMGB1 mediates the
inflammatory response upon binding to inflammation mediators, such
as lipopolysaccharide (LPS), DNA or the cytokine
interleukin-(IL-)1β. Once released, HMGB1 can bind to the receptor
for advanced glycation end-products (RAGE) in soluble or cell-bound
forms and to toll-like receptor-2 (TLR2) and TLR4 (16). This binding induces the production
of inflammatory cytokines, chemokines, adhesion molecules and ROS,
activating downstream signaling pathways such as the p38
mitogen-activated protein kinase (MAPK) kinase (MEK) (18), c-Jun N-terminal kinase (JNK)
(19) and nuclear factor-κB
(NF-κB) pathways. These pathways, in turn, stimulate the expression
and release of other pro-inflammatory cytokines, initiating a
positive feedback loop via the NF-κB signaling pathway, which leads
to cellular inflammation, damage and apoptosis. HMGB1 is therefore
implicated in the pathogenesis of a variety of chronic inflammatory
diseases, autoimmune disorders and malignant tumors (16,20).
In recent years, research on GL has primarily
focused on its role as a specific inhibitor of HMGB1 (21,22).
Experimental studies have demonstrated that GL can directly bind to
both high-mobility group boxes of HMGB1 with a dissociation
constant of ~150 µM, thereby suppressing its chemotactic and
mitogenic activities (18,23,24).
Previous studies have explored the mechanisms by which GL-mediated
inhibition of HMGB1 exerts protective effects in various disease
models (25–27). GL effectively suppresses the
pro-inflammatory cytokine activity of extracellular HMGB1 and
confers protection against ischemia-reperfusion (I/R)-induced
injury in the spinal cord, liver, brain and myocardium in animal
models (16). For instance, in a
rat liver model of I/R, GL administration was shown to reduce HMGB1
expression in Kupffer cells, demonstrating potential as a
preventive treatment for I/R injury, particularly with regards to
hepatobiliary surgery (28).
Furthermore, a study by Zhai et al (29) reported that GL alleviated I/R
injury in rat myocardium by directly inhibiting the cytokine
activity of extracellular HMGB1 and blocking the phosphorylation of
the JNK/apoptosis regulator BAX (Bax) pathway.
In spinal cord injury models, GA, the active
metabolite of GL, was found to reduce inflammation by inhibiting
HMGB1 activity via the p38/JNK signaling pathway (30). Additionally, GL has been shown to
reduce the HMGB1-induced apoptosis of hepatocytes via a
p38-dependent mitochondrial pathway, further supporting the
therapeutic potential of GL in alleviating HMGB1-mediated liver
injuries, such as viral hepatitis, liver I/R injury and
sepsis-associated liver injury (31).
The HMGB1 is also stimulates the proliferation of
cancer and endothelial cells, actives angiogenesis and induces
inflammation formation, which has a negative impact on tumor
progression and recurrence (32–34).
As a HMGB1 inhibitor, GL hinders tumor regeneration in mice by
blocking protein-stimulated cell proliferation and migration,
inhibiting HMGB1-mediated angiogenesis, and reducing inflammatory
conditions levels (16).
In addition, GL has previously been evaluated as a
potential therapeutic agent targeting sepsis. Promising results
indicate that GL modulates the serum level and gene expression of
HMGB1 and other pro-inflammatory cytokines, thereby maintaining
hemodynamic stability and protecting vital organs from LPS-induced
endotoxemia in a porcine model (35).
In ophthalmic applications, the topical
administration of glycyrrhizinate-genistein micelle-based eye drops
has been shown to markedly promote corneal epithelial and nerve
regeneration in diabetic mice. This therapeutic effect is likely
mediated through the inhibition of HMGB1 signaling via
downregulation of HMGB1 and its receptors RAGE and TLR4, as well as
the suppression of inflammatory cytokines such as IL-6 and IL-1β
(36). Furthermore, as an
inhibitor of HMGB1, GA has been shown to alleviate symptoms of
conjunctivitis, blepharitis and dry eye disease by reducing
pro-inflammatory protein levels in tear fluid (37).
Additionally, GL provides neuroprotection by
inhibiting HMGB1 activity in the nervous system, thereby improving
chronic stress-induced depressive behavior. This is achieved via
modulation of the kynurenine pathway, which has been linked to
stress-induced neuroinflammation and depressive symptoms (38).
Relevant signaling pathways
NF-κB and its signaling pathway
NF-κB is an important transcription factor
responsible for regulating inflammation and immune responses, and
is closely associated with immune cell activation, T- and
B-lymphocyte development, stress responses and apoptosis (39). Under normal conditions, NF-κB
remains bound to inhibitor of κB (IκB) and remains inactive in the
cytoplasm. However, when stimulated by upstream factors, IκB-α
undergoes ubiquitination and degradation. This process releases the
NF-κB p65 subunit from its inhibitory complex with IκB-α, allowing
it to translocate to the nucleus and activate the transcription of
various genes (40).
Once activated, NF-κB promotes the expression of
genes that facilitate cell proliferation, inhibit apoptosis and
support cancer cell proliferation. In human glioblastoma U251
cells, GL suppresses cell proliferation in a dose- and
time-dependent manner. This effect is mediated through the
downregulation of p65 expression and inhibition the NF-κB pathway,
demonstrating GL's anti-inflammatory and antitumor properties
(41). Additionally, dipotassium
glycyrrhizate, a potassium salt of GL, has been shown to exhibit
anti-proliferative effects in U251 and U138MG cells by inducing
apoptosis and upregulating microRNA (miR)-4443 and miR-3620, which
inhibit NF-κB post-transcriptionally (42). Given that overexpression of NF-κB
is a hallmark of malignant glioma, this transcription factor
remains a key target for GL in treating such cancers (43,44).
GL has also been found to reduce the ratio of M1-like macrophages
in colon. Furthermore, by inhibiting the LPS/HMGB1/NF-κB signaling
pathway, it suppresses the production of C-C motif chemokine 2 and
TNF-α in colonic macrophages (45). Furthermore, in hepatocellular
carcinoma (HCC) cells, GL induces DNA damage and inactivates NF-κB,
which collectively contribute to G1-phase arrest. This
arrest is mediated through the activation of ataxia-telangiectasia
mutated proteins, increased expression of cell cycle inhibitors p21
and p27, and the inhibition of NF-κB-mediated cyclin D1 expression
(46).
GL has also been shown to improve bone loss and
trabecular parameters in ovariectomized mice. Bone marrow stromal
cells isolated from these mice have been shown to exhibit enhanced
receptor activator of NF-κB-induced osteoclast formation
capabilities, a characteristic that GL notably reverses. NF-κB
plays an important role in osteoclastogenesis, yet glycyrrhizin
inhibits the NF-κB signaling pathway in ovariectomized mice, posing
GL administration as a potential adjunctive therapy for
postmenopausal osteoporosis (47,48).
Furthermore, GL has been shown to alleviate acute
lung injury induced by LPS by reducing the production of
inflammatory factors, such as IL-1β, monocyte chemoattractant
protein-1 and cyclooxygenase-2, HMGB1 and adhesion molecules. This
effect is mediated by the upregulation of angiotensin-converting
enzyme 2 and inhibition of the caveolin-1/NF-κB signaling pathway
(49). In human bronchial
epithelial cells treated with toluene diisocyanate-albumin
conjugate, GL-mediated inhibition of HMGB1 has been demonstrated to
lower nuclear factor erythroid 2-related factor 2 (Nrf2) expression
and reduce ROS production, resulting in increased matrix
metalloproteinase (MMP) levels and reduced NOD-like receptor pyrin
domain-containing 3 (NLRP3) inflammasome activation. As such, GL
has been shown to enhance the activation of the NLRP3 inflammasome
by modulating the HMGB1-regulated ROS/NF-κB pathway (50).
In a rat model of isoproterenol-induced myocardial
ischemia, GL has been shown to dose-dependently downregulate
phosphorylated-(p-)NF-κB p65 and p-IκBα levels, enhancing cardiac
antioxidant capacity and reducing cardiomyocyte apoptosis (51). Similarly, a mouse model of
myocardial fibrosis induced by isoproterenol has demonstrated that
inflammatory responses are amplified by NF-κB-mediated TLR4
activation. However, magnesium isoglycyrrhizinate (MgIG) has been
shown to protect against isoproterenol-induced myocardial fibrosis
by inhibiting the TLR4/NF-κB p65 signaling pathway (52).
Additionally, GL has demonstrated renal protective
effects in an insulin-resistant rat model of aluminum-induced renal
toxicity by inhibiting oxidative stress as well as the TLR4/NF-κB
pathway (53). Cisplatin (CP), a
commonly used anti-cancer drug, often causes nephrotoxicity
(54). Treatment with GL or 18β-GA
has been shown to restore oxidative homeostasis and reduce
inflammation in the kidneys of CP-treated mice to near-normal
levels, likely via upregulation of Nrf2 and downregulation of
activated NF-κB (55).
Endothelial dysfunction is an important factor in
the pathogenesis of diabetes and its vascular complications.
Pre-treatment with GL has been shown to markedly reduce human
umbilical vein endothelial cell apoptosis induced by advanced
glycation end-products, as well as exhibit protective effects
against endothelial dysfunction by inhibiting the RAGE/NF-κB
pathway. These anti-apoptotic, anti-inflammatory and antioxidant
activities enable GL to demonstrate potential therapeutic benefits
for diabetic vascular complications (56).
In a model of cerebral I/R injury, GL has been shown
to inhibit the secretion of inflammatory cytokines, including
IL-1β, IL-6 and TNF-α, in serum and brain tissue. Additionally, GL
been shown to protect against I/R-induced cerebral ischemic disease
by inhibiting the expression of the HMGB1-mediated TLR4/NF-κB
pathway (57).
Furthermore, MgIG, a magnesium salt of the 18α-GA
derivative of GL, has been shown to possess liver-protective,
anti-inflammatory, antioxidant and antiviral properties (52). These properties underlie its
clinical efficacy; for example, MgIG has been shown to markedly
ameliorate liver fibrosis by preventing the nucleus translocation
of NF-κB (58).
Phosphoinositide 3-kinase
(PI3K)/protein kinase B (Akt) signaling pathway
PI3Ks are enzymes that catalyze the conversion of
phosphatidylinositol 4,5-bisphosphate into phosphatidylinositol
3,4,5-trisphosphate. These enzymes play notable roles in a number
of cellular processes, such as proliferation, cancer progression
and inflammation. Akt is a key player in the PI3K/Akt signaling
pathway, which has been implicated in multiple diseases such as
cancer, diabetes, cardiovascular diseases and neurological
disorders (59–61).
The effects of GL on the PI3K/Akt pathway vary
depending on cellular context, tissue type and disease model. In
numerous pathological conditions, GL functions primarily as an
inhibitor of PI3K activity (62).
As PI3K/Akt signaling acts upstream of the NF-κB signaling pathway,
experimental evidence has demonstrated that GL suppresses
IL-1β-induced phosphorylation of PI3K/Akt and subsequent NF-κB
activation, thereby attenuating the inflammatory response (62) and subsequently mitigating liver
injury. Additionally, GL suppresses both inflammation and apoptosis
via the inhibition of HMGB1 and the PI3K/mammalian target of
rapamycin (mTOR) signaling pathway (63). In LPS-stimulated macrophages, both
GL and 18β-GA have been shown to inhibit the activity of the p110δ
and p110γ subunits of PI3K, therefore inhibiting subsequent NF-κB
activation. This inhibition has also been shown to result in a
dose-dependent reduction in LPS-induced TNF-α, IL-6 and IL-1β
production in RAW264.7 cells (64).
Furthermore, GL has been shown to induce excessive
autophagy in HCC cells both in vitro and in vivo, a
process that is regulated by the concurrent inhibition of the
Akt/mTOR and extracellular signal-regulated kinase (ERK)1/2
pathways. This has highlighted autophagy-mediated cell death as a
potential strategy for tumor suppression (65). A study reported by Tsai et
al (66) found that GL
markedly inhibited tumor cell growth, invasion and the
phosphorylation of ERK, Akt and epidermal growth factor receptor.
In HCC cells, GL treatment inhibited anti-apoptotic and metastatic
protein expression, thereby promoting caspase-8/9-mediated
apoptosis in both in vitro and in vivo models. In the
gastric cancer cell line MGC-803, GL has been shown to induce
apoptosis. This effect is accompanied by inhibition of PI3K/Akt,
downregulation of the expression of Bcl-1, survivin and p65
expression, upregulation of Bax and promotion of poly(ADP-ribose)
polymerase (PARP) (67). A study
reported by Niu et al (68)
supported the GL-mediated inhibition of HMGB1 both in vitro
and in vivo, which affected the protein brahma homolog 1 and
PI3K/Akt/mTOR pathways and suppressed the epithelial-mesenchymal
transition in pulmonary fibrosis in mice, ultimately slowing the
progression of silicosis.
Furthermore, GL and 18β-GA have been shown to reduce
mitochondrial bioenergetics and activate the PI3K/Akt pathway in
PC12 cells, therefore protecting these cells from ischemic injury
by modulating the intracellular antioxidant system and reducing
mitochondria-induced apoptosis (69).
MAPK signaling pathway
Numerous studies have shown that ROS can activate
the MAPK pathway. In mammals, there are three subgroups of MAPKs:
ERKs, JNKs and p38 MAPKs. All three MAPK subgroups are involved in
regulating cellular growth and apoptosis, which are important for
normal cellular function (70).
The Ras/Raf/MAPK/MEK/ERK pathway is activated by receptor tyrosine
kinase signals from growth factors and cytokines. Once activated,
Ras activates Raf, which subsequently activates MEK1/2, ultimately
leading to ERK1/2 activation; continuous ERK pathway activation
promotes cellular proliferation (71). Furthermore, the JNK pathway is
activated by MEK7 and MEK4, leading to the translocation of
activated JNK to the nucleus, where it further activates various
transcription factors to regulate cellular proliferation (72).
As an inhibitor of HMGB1, GL reduces inflammation
and fibrosis by inhibiting the MAPK and SMAD family member (Smad)3
signaling pathways, respectively (73); this has been shown to alleviate
pulmonary toxicity induced by bleomycin, a drug used for treating
various tumors (74).
Additionally, GL markedly inhibits ROS production, blocking a
cascade of events comprising endoplasmic reticulum calcium release,
endoplasmic reticulum stress (ERS), MAPK activation and cell death
(75). This GL-mediated ROS
suppression provides effective photoprotection, offering potential
for cosmetic or therapeutic purposes. In human skin fibroblasts
(Hs68 cell line), GL protects against UV-B-induced damage by
mitigating both Ca2+ imbalance, ERS, as well as by
suppressing MAPK activation and subsequent apoptosis (70). Furthermore, GL has been shown to
exhibit cytotoxic effects, induce apoptosis and promote
G0/G1 phase cell cycle arrest in the rat
pituitary adenoma-derived MMQ and GH3 cell lines. These results
indicate that GL promotes cell cycle arrest and apoptosis through a
ROS-dependent activation of the MAPK pathway (76).
The p38 MAPK pathway is activated by
pro-inflammatory cytokines such as IL-1, IL-6 and TNF-α. Activated
p38 MAPK influences downstream transcription factors, including
NF-κB and transcription factor (TF)-1, −2 and −6, in order to
regulate cellular proliferation, differentiation and growth
processes (77). An experimental
study has shown that GL may prevent colitis by reducing the
expression of NF-κB p65 and p38 MAPK (40). Additionally, GL has been shown to
modulate the T helper 1/T helper 2 cell balance by suppressing OX40
(CD134)-OX40 ligand signaling and p38 MAPK activity, thus reducing
disease severity in ovalbumin-induced asthma models (78). GL also mitigates inflammation
following spinal cord injury by regulating the p38/JNK pathway to
inhibit HMGB1 expression (30).
Furthermore, GL has previously been used as an anti-apoptotic agent
due to its inhibition of JNK1/2 and p38 MAPK phosphorylation, as
well as its suppression of CCAAT/enhancer-binding protein (C/EBP)
homologous protein, resulting in the reduction of ERS (79).
The ERK and JNK signaling pathways are both
associated with learning and memory functions (80). A study has demonstrated that GL
improves short-term memory by reducing the phosphorylation of ERK
and JNK, both of which are important for regulating neuroplasticity
and inflammatory responses (81).
In vascular endothelial cells, GL has demonstrated the potential to
inhibit angiogenesis by suppressing the ROS/ERK signaling axis,
potentially slowing the progression of angiogenesis-dependent
diseases such as various types of cancer (82).
In a chicken model of Mycoplasma
gallisepticum infection, GL has been found to suppress the
infection-induced expression of MMP2, MMP9 and inflammatory
cytokines via the p38 and JNK signaling pathways. In vivo
histopathological analysis has revealed that GL treatment markedly
alleviates tracheal and lung injuries resulting from M.
gallisepticum infection (83).
In a rat model of sepsis-induced kidney injury, GL has been shown
to markedly suppress LPS-induced oxidative stress by activating the
ERK pathway (84). Furthermore,
the C/EBP family plays a core role in regulating adipogenesis
within the transcriptional network that controls this process. GL
has been shown to inhibit early-stage adipogenesis in the 3T3-L1
cell line by inhibiting the MEK/ERK-mediated expression of C/EBPβ
and C/EBPδ (85).
Janus kinase (JAK)/signal transducer
and activator of transcription (STAT)3 signaling pathway
JAKs, such as JAK1, JAK2, JAK3 and non-receptor
tyrosine-protein kinase TYK2, are transmembrane tyrosine kinases
that activate STAT proteins (86).
Upon cytokine receptor binding, JAKs become activated and
phosphorylate tyrosine residues on the receptor tail. This
phosphorylation facilitates the binding of STAT3. The
phosphorylated STAT3 then undergoes dimerization and translocates
from the cytoplasm to the nucleus, where it regulates target gene
expression, including NF-κB, cyclin D1, survivin, apoptosis
regulator Bcl-2 and vascular endothelial growth factor, in order to
increase cell proliferation. JAK1 in particular plays a notable
role in cytokine signaling pathways that regulate inflammatory
cytokine expression (87).
Several studies have shown that phosphorylated
non-receptor tyrosine kinases, such as Akt and mTOR, can activate
STAT3 proteins (88–90). GL has been shown to suppress the
Akt/mTOR/STAT3 signaling pathway, downregulate cyclin D1 and
survivin, promote the cleavage of caspase-3 and PARP, and inhibit
TF-1 cell proliferation in vitro; this inhibition of cell
proliferation has resulted in reductions in TF-1 tumor volume in
vivo (91). A previous study
on the non-small cell lung cancer cell line HCC827 has shown that
GL inhibits cancer cell migration and invasion by targeting the
JAK/STAT/HMGB1 pathway (92).
Additionally, a study reported by Tian et al
(93) demonstrated that GL
ammonium salt reduced hepatocyte apoptosis by suppressing the
JAK1/STAT1/interferon regulatory factor 1 signaling pathway,
inhibiting oxidative stress, downregulating p-JNK expression and
modulating apoptosis-related protein expression, therefore
alleviating liver injury and restoring T helper cell balance in the
liver. Another study reported by Guo et al (94) observed that GL promoted CYG-binding
protein 1-mediated activation of the interferon-γ
(IFN-γ)/STAT1/Smad7 signaling pathway, which reduced liver fibrosis
and suppressed hepatic stellate cell (HSC) activation.
Pyroptosis signaling pathway
Pyroptosis is a regulated form of programmed cell
death that is characterized by inflammatory responses. Pyroptotic
signaling pathways are primarily categorized into canonical
pathways and non-canonical pathways, which are mediated by
caspase-1 and caspase-11, respectively. Pyroptotic cell death is
marked by the activation of inflammatory caspases, predominantly
caspase-1, −4, −5 and −11, and the cleavage of gasdermin family
proteins. Cleaved gasdermin proteins subsequently form membrane
pores, resulting in cell membrane rupture, the release of
inflammatory mediators and cell death (95). The NLRP3 inflammasome is a key
regulator of pyroptosis. The inflammasome recruits
apoptosis-associated speck-like protein to form an active
inflammasome complex that responds to various exogenous and
endogenous stressors by secreting inflammatory factors such as
IL-1β or IL-18, leading to inflammation (96).
GL enhances the expression of the tumor suppressor
protein p53 and upregulates the levels of caspase-9 and cleaved
caspase-3 (97). These results
indicated that GL treatment induces apoptosis, which is consistent
with previous reports (98–100).
Additionally, caspase-11 acts as an LPS receptor, mediating
ferroptosis, coagulopathy and lethality in endotoxemia and
bacterial sepsis. GL treatment has been shown to markedly suppress
caspase-11-dependent immune responses in endotoxemia and
experimental sepsis models, resulting in reduced coagulopathy,
organ damage and mortality (101). Furthermore, GL treatment has been
shown to inhibit liver I/R injury and promote pyroptosis in Kupffer
cells through gasdermin D-mediated cell death (102).
Other related signaling pathways
In HSCs, GL exerts anti-fibrotic effects by
inhibiting the expression of Smad2, Smad3 and Smad7, all of which
are activated by transforming growth factor-β (TGF-β)1-actived
signaling pathway (103). A
previous study in a rat model of liver fibrosis found that combined
administration of GL and aspartate aminotransferase notably reduced
Smad3 mRNA levels and the protein levels of p-Smad2/3, Smad3 and
TGF-β1. These findings further support the inhibitory effect of GL
on the TGF-β1/Smad signaling pathway (104). GL has also been shown to
alleviate gefitinib-induced liver injury by inhibiting the p53/p21
pathway, thereby promoting cell-cycle progression (105).
Furthermore, GL has been shown to regulate the
Hippo/yes-associated protein (YAP) pathway, a key modulator of cell
proliferation and apoptosis, by inhibiting YAP nuclear
translocation, thus preventing myocardial I/R injury (106). Additionally, GL alleviate
steroid-induced femoral head necrosis in both in vivo and
in vitro models by activating the Wnt/β-catenin pathway. The
activation of this pathway reduces oxidative stress, enhances
osteogenic differentiation and suppresses the adipogenic
differentiation of mesenchymal stem cells. These combined effects
restore osteogenic homeostasis in the femoral head, thereby
mitigating necrosis (107). A
study reported by Lai et al (108) demonstrated that GL protected
against myocardial I/R injury by mitigating inflammation and cell
death, potentially through the inhibition of ERS. Furthermore, GL
administration has been demonstrated to alleviate fibrosis and
inflammation caused by high glucose levels in glomerular podocytes
by upregulating the AMP-activated protein kinase pathway and its
associated regulatory factors (109). In both in vitro and in
vivo models of acute liver failure, GL treatment has been shown
to markedly inhibit ferroptosis by reducing oxidative stress
(110). Co-administration of
glycyrrhizin mitigates triptolide (TPL)-induced nephrotoxicity.
This protection is achieved though the repair of TPL-damaged tight
junction structures in renal tubules, mediated via the
RhoA/Rho-associated kinase-1/myosin light chain signaling pathway
(111) (Fig. 2).
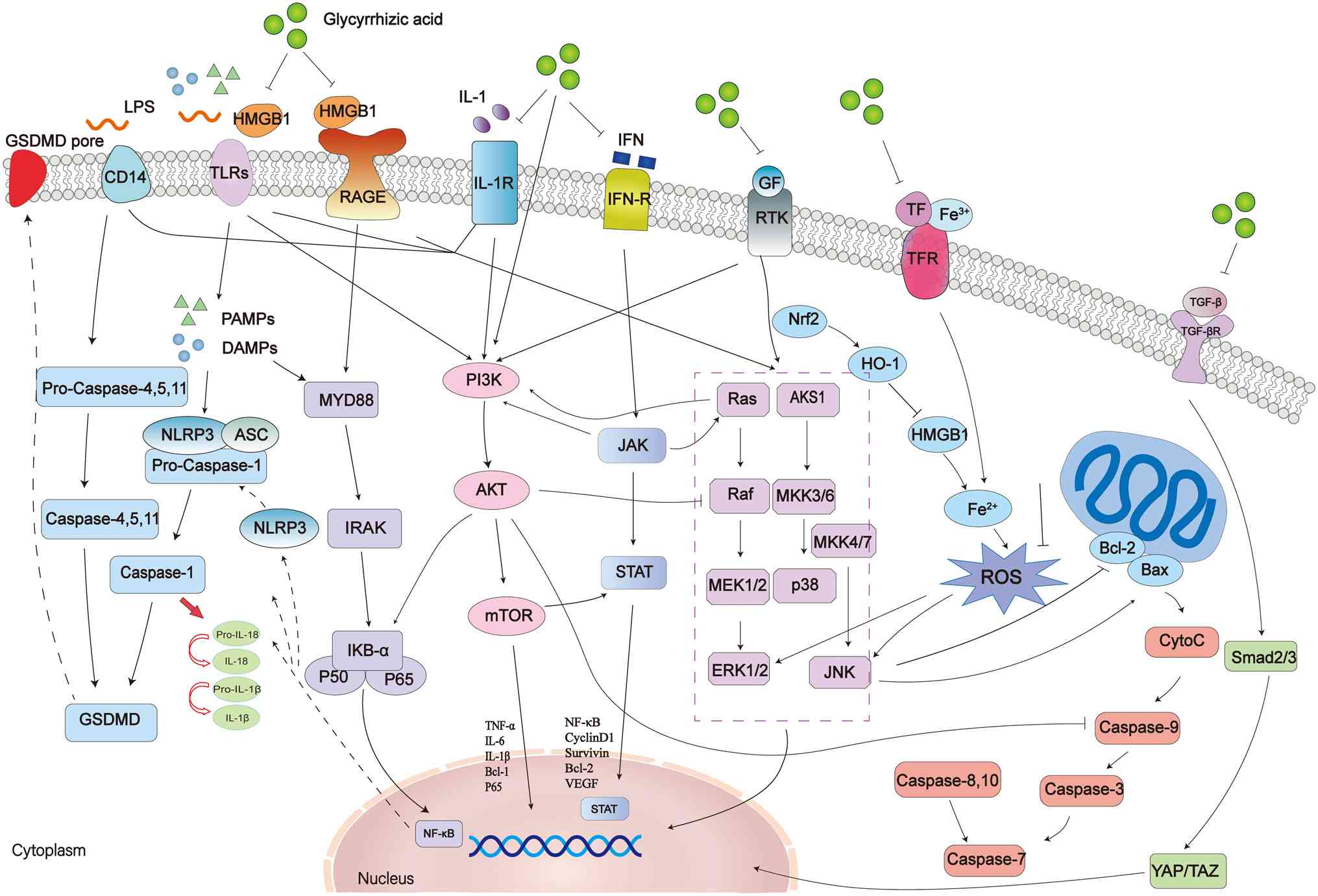 | Figure 2.Mechanisms and signaling pathways of
GL. GL, a bioactive compound derived from licorice roots, exhibits
various pharmacological effects by modulating multiple signaling
pathways. GL plays an important role in anti-inflammatory,
antioxidant and antitumor processes by targeting key molecular
pathways such as the PI3K/Akt/mTOR, MAPK and NF-κB signaling
pathways. GL inhibits inflammation by suppressing HMGB1, resulting
in reduced NF-κB activation and cytokine release. GL also regulates
apoptosis via modulation of caspase activation, the Bcl-2/Bax ratio
and mitochondrial oxidative stress. Additionally, GL has been shown
to induce autophagy by inhibiting the Akt/mTOR pathway. The
protective effects of GL extend to various conditions, including
neurodegenerative diseases, cardiovascular disorders and
immune-related conditions, primarily through the reduction of
oxidative stress and inflammation. This schematic provides a
comprehensive overview of the role of GL in disease modulation
through its interactions with cellular signaling pathways. The
dashed box indicates the three major branches of the MAPK signaling
pathway. GL, glycyrrhizic acid; PI3K, phosphoinositide 3-kinase;
Akt, protein kinase B; mTOR, mechanistic target of rapamycin; MAPK,
mitogen-activated protein kinase; NF-κB, nuclear factor κB; HMGB1,
high mobility group box 1; Bcl-2, B-cell lymphoma 2; Bax,
Bcl-2-associated X protein. |
Discussion
GL, a natural triterpenoid compound, exhibits a
range of biological activities, including anti-inflammatory,
antioxidant and antitumor effects. These pharmacological properties
are mediated through multiple signaling pathways, including the
NF-κB, PI3K/Akt, MAPK, JAK/STAT3 and pyroptotic pathways (Table I).
 | Table I.Summary of the major mechanisms and
therapeutic applications of GL. |
Table I.
Summary of the major mechanisms and
therapeutic applications of GL.
| Mechanistic
category | Key molecular
actions of GL | Representative
therapeutic effects | (Refs.) |
|---|
| HMGB1
inhibition | Directly binds
HMGB1 and blocks its interaction with the RAGE/TLR2/TLR4 receptor
axis, thereby suppressing the downstream release of inflammatory
mediators. | Reduces
inflammation and tissue injury; protects the liver, cornea and
neural tissues. | (28,30,31,36,38) |
| NF-κB pathway
suppression | Inhibits NF-κB p65
activation and decreases TNF-α, IL-1β and IL-6 levels. | Exerts
anti-inflammatory and antioxidant effects; inhibits tumor
proliferation; confers cardiopulmonary and renal protection. | (41–46,49–55) |
| PI3K/Akt/mTOR
pathway regulation | Primarily inhibits
PI3K/Akt signaling; downregulates cyclin D1 and survivin; promotes
autophagy and apoptosis. Promotion the PI3K/Akt pathway in PC12
cells. | Suppresses tumor
growth and metastasis; ameliorates inflammatory and fibrotic
diseases. Protecting these cells from ischemic injury by modulating
the intracellular antioxidant system and reducing
mitochondria-induced apoptosis. | (66–69) |
| MAPK pathway
modulation | Inhibits ERK, JNK
and p38 activation; reduces oxidative and ER stress. | Exerts
anti-fibrotic and cytoprotective effects; protects lung, skin and
neural tissues. | (70,74,76) |
| JAK/STAT3 pathway
inhibition | Blocks the
phosphorylation JAK1 and STAT3; downregulates proliferation- and
survival-related genes. | Suppresses tumor
cell proliferation and invasion; reduces immune-mediated liver
injury. | (91–94) |
| Regulation of
pyroptosis activity | Inhibits caspase;
reduces gasdermin D-mediated pyroptosis. | Protects against
liver injury; Inhibits tumor cell proliferation. | (97–100,102) |
| TGF-β/Smad pathway
inhibition | Blocks Smad2/3
activation. | Exerts
anti-fibrotic effects in liver tissues. | (104) |
| Hippo/YAP pathway
modulation | Inhibits YAP
nuclear translocation. | Reduces myocardial
ischemia-reperfusion injury. | (106) |
| Wnt/β-catenin
pathway activation | Enhances osteogenic
differentiation via the Wnt/β-catenin pathway. | Prevents
osteonecrosis and maintains bone homeostasis. | (107) |
GL has also been shown to attenuate oxidative stress
and mediate the activation of inflammatory signaling cascades. For
example, GL inhibits the ERK, JNK and p38 MAPK pathways, resulting
in the reduced secretion of pro-inflammatory cytokines.
Furthermore, GL negatively regulates the JAK/STAT3 pathway by
suppressing STAT3 phosphorylation (112), leading to reduced cellular
proliferation and inflammatory responses. Through inhibition of the
PI3K/Akt/mTOR pathway, GL modulates cell survival and apoptosis,
downregulates the expression of cell cycle-related proteins, such
as cyclin D1 and survivin, and promotes tumor cell apoptosis
(9). In fibrosis-associated
disorders, GL has been shown to exert anti-fibrotic effects
primarily through inhibition of the TGF-β/Smad signaling axis,
resulting in reduced HSC activation. Additionally, GL has been
shown to modulate the Hippo/YAP pathway, thus mitigating myocardial
I/R injury, and to activate the Wnt/β-catenin pathway, which
contributes to the maintenance of osteogenic homeostasis and
ameliorates steroid-induced femoral head necrosis. The
antioxidative and anti-inflammatory actions of GL have also been
demonstrated in models of high glucose-induced podocyte injury,
acute liver failure and neuroinflammation. Notably, GL has been
shown to suppress NLRP3 inflammasome-mediated pyroptosis by
inhibiting the HMGB1/TLR4/NF-κB signaling pathway, reducing
inflammatory damage and demonstrating notable neuroprotective and
anticonvulsant effects in kainic acid-induced status epilepticus
mouse model (113). In models of
endotoxemia and sepsis, GL has been shown to alleviate immune
dysregulation by suppressing caspase-11-dependent coagulation
abnormalities and mitigating organ damage, demonstrating notable
systemic protective effects.
Recent studies support the expanding pharmacological
relevance of GL and related triterpenoids. Network pharmacology and
molecular docking analyses have revealed that GA exerts its effects
in diabetic nephropathy by targeting multiple key pathways involved
in inflammation and fibrosis, providing additional mechanistic
evidence for the renoprotective potential of licorice-derived
compounds (114). A recent review
highlighted the therapeutic potential of glycyrrhizin in
neuroinflammatory and neurodegenerative diseases, emphasizing its
ability to modulate oxidative stress, apoptosis and HMGB1-related
signaling within the central nervous system (115). Furthermore, a recent study has
examined the efficacy of traditional and nanotechnology-based
delivery approaches for GL, underscoring the importance of advanced
formulations, such as improved nanotechnology, for enhancing drug
bioavailability and clinical utility (116). These findings have complemented
the discussion of the present review by demonstrating that
GL-associated compounds modulate broader pathological process (such
as inflammation) across organ systems, and that the clinical
administration may benefit from innovative delivery strategies.
The present review systematically summarized the
signaling networks involved in the pharmacological actions of
glycyrrhizin across diverse disease models (Table II) (30,40,41,42,44–53,55–57,58,63,65–70,73,79,82–85,94,95,97,103–107). A thorough understanding of the
complex crosstalk among signaling pathways modulated by GL is
important for informing the design of rigorous, multicenter,
large-scale randomized controlled trials.
 | Table II.Signaling pathways of glycyrrhizic
acid in disease as organized by disease or organ. |
Table II.
Signaling pathways of glycyrrhizic
acid in disease as organized by disease or organ.
| A,
Glioblastoma |
|---|
|
|---|
| Signaling
pathways | Model | (Refs.) |
|---|
| NF-κB | U251 glioblastoma
cell line. | (41,42,44) |
|
| B,
Liver |
|
| NF-κB | HCC cells. | (46) |
|
| Rat hepatic
stellate cells. | (58) |
| PI3K/Akt | Human liver
macrophages; LPS-induced acute liver injury in C57/B6J mice. | (63) |
|
| HCC cells. | (65) |
|
| SK-Hep1 and Hep3B
cells; SK-Hep1/luc2 bearing mice. | (66) |
|
JAK1/STAT1/IRF1 | Concanavalin
A-induced liver injury in BALB/c mice. | (94) |
|
IFN-γ/STAT1/Smad7 |
CCl4-induced liver fibrosis in
C57/B6J mice; human LX-2 cells and primary hepatic stellate
cells. | (95) |
| TGF-β/Smad | Rat hepatic
stellate cells. | (103) |
| TGF-β1/Smad | Ligation-induced
(BDL) and dimethylnitrosamine-induced liver fibrosis in Wistar
rats; mice hepatic stellate cells. | (104) |
| p53/p21 | AML-12 normal mouse
liver cells, CRL-2254; male Institute of Cancer Research mice. | (105) |
|
| C, Bone |
|
| NF-κB | Bone marrow
monocytes or macrophages; ovariectomized mice. | (47,48) |
| Wnt/β-catenin | LPS with
methylprednisolone-induced osteonecrosis of the femoral head in SD
rats; C3H10T1/2 murine mesenchymal stem cells. | (107) |
|
| D,
Colon |
|
| NF-κB | Colonic
macrophages. | (45) |
| Pyroptosis |
1,2-dimethyhydrazine-induced precancerous
lesions in Wistar rats. | (97) |
|
| E, Lung |
|
| NF-κB | HUVECs; LPS-induced
acute lung injury in mice. | (49) |
|
| Human bronchial
epithelial cells. | (50) |
| PI3K/Akt | Human non-small
cell lung cancer cells, A549 cells; C57BL/6N mice intratracheally
instilled with a sterile SiO2 dust suspension. | (68) |
| MAPK | Bleomycin-induced
pulmonary toxicity in BALB/c mice. | (73) |
|
| OVA-induced asthma
in 48 BALB/c mice. | (79) |
|
| Chicken primary
alveolar type II epithelial cells. | (84) |
|
| F,
Cardiac |
|
| NF-κB | ISO-induced
myocardial cardiotoxicity in SD rats. | (51) |
|
| ISO-induced
myocardial cardiotoxicity in Kunming mice. | (52) |
| Hippo/YAP | H9c2 cells; I/R rat
model. | (106) |
|
| G,
Kidney |
|
| NF-κB | Aluminum and
fructose-induced renal injury in Wistar rats. | (53) |
|
| Cisplatin-induced
nephrotoxicity in BALB/c mice. | (55) |
| MAPK | LPS stimulated rat
mesangial cells (HBZY-1); septic rat models. | (85) |
|
| H, Vascular
system |
|
| Signaling
pathways | Model | (Refs.) |
| NF-κB | HUVECs. | (56) |
| MAPK | BALB/c mice. | (83) |
|
| I,
Cerebral |
|
| NF-κB | I/R injury in SD
rats. | (57) |
| MAPK | Scopolamine-induced
cognitive impairment in mice. | (82) |
|
| J, Gastric
cancer |
|
| PI3K/Akt | Gastric cancer cell
lines, MGC-803 cells, BGC-823 cells and SGC-7901) cells. | (67) |
|
| K,
Neurodegenerative disease |
|
| PI3K/Akt | Rat
pheochromocytoma cell line PC12 | (68) |
|
| L, Skin |
|
| MAPK | Human skin
fibroblast Hs68 cells. | (70) |
|
| M,
Intestine |
|
| NF-κB and MAPK |
Methotrexate-induced enteritis in SD
rats. | (40) |
|
| N,
Spine |
|
| p38/JNK | Highly aggressively
proliferating immortalized rat microglia cells. | (30) |
Notably, the biological activity of GL exhibits
context-dependent variability, which is influenced by cell type,
tissue environment and disease state. Elucidating the primary
signaling mechanisms regulated by GL in each pathological context
is important for optimizing targeted therapeutic strategies. Given
the limited oral bioavailability of GL, previous research have
focused on the development of novel strategies, such as combination
therapies and nanocarrier-based delivery systems, to enhance its
pharmacokinetic properties and reduce adverse effects in patients
(117–119). Mechanistic insights into
GL-mediated signaling regulation will facilitate the rational
design of synergistic drug combinations and advanced delivery
systems for GL-based therapies, therefore maximizing their
therapeutic efficacy.
The present review extended the existing literature
regarding GL activity by adopting a pathway-oriented perspective
that integrated molecular mechanisms with disease relevance. While
a previous review by Semwal et al (120) comprehensively summarized the
pharmacological activities of glycyrrhizin, the present review
placed greater emphasis on the organization of available evidence
according to key signaling axes. HMGB1 inhibition was highlighted
as a central upstream event linking inflammatory, oncological and
fibrotic processes. Additionally, downstream pathways, such as the
NF-κB, PI3K/Akt, MAPK, JAK/STAT3 and pyroptotic pathways, were
discussed in a unified framework. To facilitate the interpretation
of these complex interactions, concise mechanistic summary tables
have been provided with the aim of improving accessibility for
readers from diverse scientific backgrounds (Tables I and II).
In conclusion, GL exerts multifaceted biological
effects in various pathologies through the modulation of diverse
signaling pathways, highlighting its potential as a promising
therapeutic agent for inflammatory diseases, oxidative
stress-related disorders, tumor progression and fibrosis. Continued
mechanistic exploration and pathway-specific studies are required
to provide a solid theoretical foundation for the clinical
translation of GL.
Acknowledgements
Not applicable.
Availability of data and materials
Not applicable.
Authors' contributions
JY drafted and reviewed the manuscript. WX and YF
independently performed literature searches. HQ and NW reviewed the
information of the selected literature. WS was responsible for
supervision and conceptualization, as well as writing, reviewing
and editing the manuscript. JY, WX, YF, NW and WS contributed to
manuscript revision. Data authentication is not applicable. All
authors read and approved the final version of the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
11b-HSDs
|
11β-hydroxysteroid dehydrogenase
activity
|
|
Akt
|
protein kinase B, PKB
|
|
ASC
|
apoptosis-associated speck-like
protein containing a CARD
|
|
C/EBP
|
CCAAT/enhancer-binding protein
|
|
CD134
|
OX40
|
|
CP
|
cisplatin
|
|
CUGBP1
|
CYG-binding protein1
|
|
ERS
|
endoplasmic reticulum stress
|
|
ERK
|
extracellular signal-regulated
kinase
|
|
GSDMD
|
Gasdermin D
|
|
GL
|
glycyrrhizic acid
|
|
HCC
|
hepatocellular carcinoma
|
|
HMGB1
|
high-mobility group box 1
|
|
IL-1β
|
interleukin1β
|
|
I/R
|
ischemia-reperfusion
|
|
JAK
|
Janus kinase
|
|
JNK
|
c-Jun N-terminal kinase
|
|
LPS
|
lipopolysaccharide
|
|
MAPK
|
mitogen-activated protein kinase
|
|
MgIG
|
magnesium isoglycyrrhizinate
|
|
MMP
|
matrix metalloproteinase
|
|
NF-κB
|
nuclear factor
kappa-light-chain-enhancer of activated B cells
|
|
NLRP3
|
NOD-LRR- and pyrin domain-containing
protein 3
|
|
Nrf2
|
nuclear factor erythroid 2-related
factor 2
|
|
p38
|
p38 mitogen-activated protein
kinase
|
|
PARP
|
poly(ADP-ribose) polymerase
|
|
PI3K
|
phosphoinositide 3-kinase
|
|
RAGE
|
receptor for advanced glycation
end-products
|
|
Smad
|
SMAD family member
|
|
STAT3
|
signal transducer and activator of
transcription 3
|
|
TGF-β
|
transforming growth factor-β
|
|
TLR2
|
toll-like receptor-2
|
|
TLR4
|
toll-like receptor-4
|
|
TNF-α
|
tumor necrosis factor-α
|
|
YAP
|
yes-associated protein
|
References
|
1
|
Chinese Pharmacopoeia Commission, .
Pharmacopoeia of the People's Republic of China. China Medical
Science Press; https://ydz.chp.org.cn/#/mainFebruary 14–2026
|
|
2
|
Seki H, Sawai S, Ohyama K, Mizutani M,
Ohnishi T, Sudo H, Fukushima EO, Akashi T, Aoki T, Saito K and
Muranaka T: Triterpene functional genomics in licorice for
identification of CYP72A154 involved in the biosynthesis of
glycyrrhizin. Plant Cell. 23:4112–4123. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zhu Z, Tao W, Li J, Guo S, Qian D, Shang
E, Su S and Duan JA: Rapid determination of flavonoids in licorice
and comparison of three licorice species. J Sep Sci. 39:473–482.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zuo J, Meng T, Wang Y and Tang W: A review
of the antiviral activities of glycyrrhizic acid, glycyrrhetinic
acid and glycyrrhetinic acid monoglucuronide. Pharmaceuticals
(Basel). 16:6412023. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gao W, Zhao Y, Guo L, Wang Y, Gong H,
Zhang B and Yan M: Comparative effectiveness of glycyrrhizic acid
preparations aimed at improving liver function of patients with
chronic hepatitis B: A network meta-analysis of 53 randomized
controlled trials. Phytomedicine. 116:1548832023. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bi X, Yang L, Lin Y, Deng W, Jiang T,
Zhang L, Lu Y, Yi W, Xie Y and Li M: Efficacy and safety of
glycyrrhizic acid in treatment of autoimmune hepatitis. Am J Chin
Med. 51:391–405. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Chen S, Cao W, Xiao X, Wang L, Wan R, Zou
Z, Yang Q and Li Y: A systematic review and meta-analysis of
efficacy and safety of compound glycyrrhizin combined with
second-generation non-sedated antihistamine for the treatment of
chronic urticaria. J Dermatolog Treat. 35:22995972024. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li M, Xiang L and Li Y: Efficacy and
safety of compound glycyrrhizin in patients with alopecia areata: A
systematic review and meta-analysis. Ann Med. 57:24916592025.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Zhang Y, Sheng Z, Xiao J, Li Y, Huang J,
Jia J, Zeng X and Li L: Advances in the roles of glycyrrhizic acid
in cancer therapy. Front Pharmacol. 14:12651722023. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Su X, Wu L, Hu M, Dong W, Xu M and Zhang
P: Glycyrrhizic acid: A promising carrier material for anticancer
therapy. Biomed Pharmacother. 95:670–678. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mohammed EAH, Peng Y, Wang Z, Qiang X and
Zhao Q: Synthesis, antiviral, and antibacterial activity of the
glycyrrhizic acid and glycyrrhetinic acid derivatives. Russ J
Bioorg Chem. 48:906–918. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen K, Yang R, Shen FQ and Zhu HL:
Advances in pharmacological activities and mechanisms of
glycyrrhizic acid. Curr Med Chem. 27:6219–6243. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ploeger B, Mensinga T, Sips A, Seinen W,
Meulenbelt J and DeJongh J: The pharmacokinetics of glycyrrhizic
acid evaluated by physiologically based pharmacokinetic modeling.
Drug Metab Rev. 33:125–147. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wagle A, Seong SH, Zhao BT, Woo MH, Jung
HA and Choi JS: Comparative study of selective in vitro and in
silico BACE1 inhibitory potential of glycyrrhizin together with its
metabolites, 18α- and 18β-glycyrrhetinic acid, isolated from
Hizikia fusiformis. Arch Pharm Res. 41:409–418. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shetty AV, Thirugnanam S, Dakshinamoorthy
G, Samykutty A, Zheng G, Chen A, Bosland MC, Kajdacsy-Balla A and
Gnanasekar M: 18α-glycyrrhetinic acid targets prostate cancer cells
by down-regulating inflammation-related genes. Int J Oncol.
39:635–640. 2011.PubMed/NCBI
|
|
16
|
Musumeci D, Roviello GN and Montesarchio
D: An overview on HMGB1 inhibitors as potential therapeutic agents
in HMGB1-related pathologies. Pharmacol Ther. 141:347–357. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang H, Antoine DJ, Andersson U and Tracey
KJ: The many faces of HMGB1: Molecular structure-functional
activity in inflammation, apoptosis, and chemotaxis. J Leukoc Biol.
93:865–873. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Mollica L, De Marchis F, Spitaleri A,
Dallacosta C, Pennacchini D, Zamai M, Agresti A, Trisciuoglio L,
Musco G and Bianchi ME: Glycyrrhizin binds to high-mobility group
box 1 protein and inhibits its cytokine activities. Chem Biol.
14:431–441. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Fu Y, Zhou E, Wei Z, Song X, Liu Z, Wang
T, Wang W, Zhang N, Liu G and Yang Z: Glycyrrhizin inhibits
lipopolysaccharide-induced inflammatory response by reducing TLR4
recruitment into lipid rafts in RAW264.7 cells. Biochim Biophys
Acta. 1840:1755–1764. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Arumugam T, Ramachandran V, Gomez SB,
Schmidt AM and Logsdon CD: S100P-derived RAGE antagonistic peptide
reduces tumor growth and metastasis. Clin Cancer Res. 18:4356–4364.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Su L, Zhu Y, Li X, Wang D, Chen X, Liu Z,
Li J, Zhang C and Zhang J: Topical adhesive spatio-temporal
nanosystem co-delivering chlorin e6 and HMGB1 inhibitor
glycyrrhizic acid for in situ psoriasis chemo-phototherapy. Acta
Pharm Sin B. 15:1126–1142. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Gadanec LK, Andersson U, Apostolopoulos V
and Zulli A: Glycyrrhizic acid inhibits high-mobility group box-1
and homocysteine-induced vascular dysfunction. Nutrients.
15:31862023. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sha X, Wang C, Liu Y, Zhong N, Lu Y, Zhang
Q, Lu S, He D, Jin Y, Tang Y and Wang S: Multifunctional
glycyrrhizic acid-loaded nanoplatform combining ferroptosis
induction and HMGB1 blockade for enhanced tumor immunotherapy. J
Nanobiotechnology. 23:2242025. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Li Y, Wu J, Du F, Tang T, Lim JCW,
Karuppiah T, Liu J and Sun Z: Neuroprotective potential of
glycyrrhizic acid in ischemic stroke: Mechanisms and therapeutic
prospects. Pharmaceuticals (Basel). 17:14932024. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lin YT, Ho CY, Sun GC, Wong TY, Hsiao M,
Tseng CJ and Cheng PW: Targeting acetylated high mobility group box
1 protein (HMGB1) and toll-like receptor (TLR4) interaction to
alleviate hypertension and neuroinflammation in fructose-fed rats.
Br J Pharmacol. 183:171–188. 2026. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yu C, Xiang Y, Zhang M, Wen J, Duan X,
Wang L, Deng G and Fang P: Glycyrrhizic acid alleviates semen
strychni-induced neurotoxicity through the inhibition of HMGB1
phosphorylation and inflammatory responses. J Neuroimmune
Pharmacol. 19:212024. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sun Q, Li L, Li J, Li J, Li SY, Zhang Y,
Chen XS, Liu SS and Hua ZY: Glycyrrhizin alleviates brain injury in
necrotizing enterocolitis model mice by suppressing HMGB1/TLR4
pathway. Int Immunopharmacol. 150:1142942025. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ogiku M, Kono H, Hara M, Tsuchiya M and
Fujii H: Glycyrrhizin prevents liver injury by inhibition of
high-mobility group box 1 production by Kupffer cells after
ischemia-reperfusion in rats. J Pharmacol Exp Ther. 339:93–98.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhai CL, Zhang MQ, Zhang Y, Xu HX, Wang
JM, An GP, Wang YY and Li L: Glycyrrhizin protects rat heart
against ischemia-reperfusion injury through blockade of
HMGB1-dependent phospho-JNK/Bax pathway. Acta Pharmacol Sin.
33:1477–1487. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wu Z, Wang Z, Xie Z, Zhu H, Li C, Xie S,
Zhou W, Zhang Z and Li M: Glycyrrhizic acid attenuates the
inflammatory response after spinal cord injury by inhibiting high
mobility group box-1 protein through the p38/Jun N-terminal kinase
signaling pathway. World Neurosurg. 158:e856–e864. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Gwak GY, Moon TG, Lee DH and Yoo BC:
Glycyrrhizin attenuates HMGB1-induced hepatocyte apoptosis by
inhibiting the p38-dependent mitochondrial pathway. World J
Gastroenterol. 18:679–684. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wang S and Zhang Y: HMGB1 in inflammation
and cancer. J Hematol Oncol. 13:1162020. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chen R, Zou J, Zhong X, Li J, Kang R and
Tang D: HMGB1 in the interplay between autophagy and apoptosis in
cancer. Cancer Lett. 581:2164942024. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Sims GP, Rowe DC, Rietdijk ST, Herbst R
and Coyle AJ: HMGB1 and RAGE in inflammation and cancer. Annu Rev
Immunol. 28:367–388. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Wang W, Zhao F, Fang Y, Li X, Shen L, Cao
T and Zhu H: Glycyrrhizin protects against porcine endotoxemia
through modulation of systemic inflammatory response. Crit Care.
17:R442013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hou Y, Xin M, Li Q and Wu X: Glycyrrhizin
micelle as a genistein nanocarrier: Synergistically promoting
corneal epithelial wound healing through blockage of the HMGB1
signaling pathway in diabetic mice. Exp Eye Res. 204:1084542021.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Burillon C, Chiambaretta F and Pisella PJ:
Efficacy and safety of glycyrrhizin 2.5% eye drops in the treatment
of moderate dry eye disease: Results from a prospective, open-label
pilot study. Clin Ophthalmol. 12:2629–2636. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Wang B, Lian YJ, Dong X, Peng W, Liu LL,
Su WJ, Gong H, Zhang T, Jiang CL, Li JS and Wang YX: Glycyrrhizic
acid ameliorates the kynurenine pathway in association with its
antidepressant effect. Behav Brain Res. 353:250–257. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Yu HJ, Park HJ, Lee B and Hahm DH: The
bidirectional interaction between NF-κ B and glucocorticoid
receptor: Underlying mechanisms of chronic stress-induced
pathology. J Immunol Res. 2025:55178402025. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang YM and Du GQ: Glycyrrhizic acid
prevents enteritis through reduction of NF-κB p65 and p38MAPK
expression in rat. Mol Med Rep. 13:3639–3646. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Li S, Zhu JH, Cao LP, Sun Q, Liu HD, Li
WD, Li JS and Hang CH: Growth inhibitory in vitro effects of
glycyrrhizic acid in U251 glioblastoma cell line. Neurol Sci.
35:1115–1120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Bonafé GA, Dos Santos JS, Fernandes AMAP,
Ziegler JV, Marson FAL, Rocha T, Carvalho PO and Ortega MM:
Anti-migratory effect of dipotassium glycyrrhizinate on
glioblastoma cell lines: Microarray data for the identification of
key MicroRNA signatures. Front Oncol. 12:8195992022. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Staudt LM: Oncogenic activation of
NF-kappaB. Cold Spring Harb Perspect Biol. 2:a0001092010.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Li W, Liu X, Zhang B, Qi D, Zhang L, Jin Y
and Yang H: Over-expression of candidate tumor suppressor ECRG4
inhibits glioma proliferation and invasion. J Exp Clin Cancer Res.
29:892010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Qiu M, Huang K, Liu Y, Yang Y, Tang H, Liu
X, Wang C, Chen H, Xiong Y, Zhang J and Yang J: Modulation of
intestinal microbiota by glycyrrhizic acid prevents high-fat
diet-enhanced pre-metastatic niche formation and metastasis.
Mucosal Immunol. 12:945–957. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Wang WS, Chen YS, Kuo CY, Tsai JJ, Hsu FT,
Chung JG and Pan PJ: DNA damage and NF-κB inactivation implicate
glycyrrhizic acid-induced G1 phase arrest in
hepatocellular carcinoma cells. J Food Biochem.
46:e141282022.PubMed/NCBI
|
|
47
|
Yin Z, Zhu W, Wu Q, Zhang Q, Guo S, Liu T,
Li S, Chen X, Peng D and Ouyang Z: Glycyrrhizic acid suppresses
osteoclast differentiation and postmenopausal osteoporosis by
modulating the NF-κB, ERK, and JNK signaling pathways. Eur J
Pharmacol. 859:1725502019. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Tang Y, Lv XL, Bao YZ and Wang JR:
Glycyrrhizin improves bone metabolism in ovariectomized mice via
inactivating NF-κB signaling. Climacteric. 24:253–260. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Chen Y, Qu L, Li Y, Chen C, He W, Shen L
and Zhang R: Glycyrrhizic acid alleviates lipopolysaccharide
(LPS)-induced acute lung injury by regulating
angiotensin-converting enzyme-2 (ACE2) and caveolin-1 signaling
pathway. Inflammation. 45:253–266. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Jiao B, Guo S, Yang X, Sun L, Sai L, Yu G,
Bo C, Zhang Y, Peng C, Jia Q and Dai Y: The role of HMGB1 on
TDI-induced NLPR3 inflammasome activation via ROS/NF-κB pathway in
HBE cells. Int Immunopharmacol. 98:1078592021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Yang J, Shi Y, Chen H, Wang X, Chen Y and
Yang B: Glycyrrhizic acid attenuates myocardial injury: Involvement
of RIP140/NF-kB pathway. Biomed Pharmacother. 95:62–67. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Ma D, Zhang J, Zhang Y, Zhang X, Han X,
Song T, Zhang Y and Chu L: Inhibition of myocardial hypertrophy by
magnesium isoglycyrrhizinate through the TLR4/NF-κB signaling
pathway in mice. Int Immunopharmacol. 55:237–244. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Emara NA, Mahmoud MF, El Fayoumi HM and
Mahmoud AAA: The renoprotective effect of glycyrrhizic acid in
insulin-resistant rats exposed to aluminum involves the inhibition
of TLR4/NF-κB signaling pathway. Naunyn Schmiedebergs Arch
Pharmacol. 394:863–872. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Arany I and Safirstein RL: Cisplatin
nephrotoxicity. Semin Nephrol. 23:460–464. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Wu CH, Chen AZ and Yen GC: Protective
effects of glycyrrhizic acid and 18β-glycyrrhetinic acid against
cisplatin-induced nephrotoxicity in BALB/c mice. J Agric Food Chem.
63:1200–1209. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Feng L, Zhu MM, Zhang MH, Wang RS, Tan XB,
Song J, Ding SM, Jia XB and Hu SY: Protection of glycyrrhizic acid
against AGEs-induced endothelial dysfunction through inhibiting
RAGE/NF-κB pathway activation in human umbilical vein endothelial
cells. J Ethnopharmacol. 148:27–36. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Yan S, Fang C, Cao L, Wang L, Du J, Sun Y,
Tong X, Lu Y and Wu X: Protective effect of glycyrrhizic acid on
cerebral ischemia/reperfusion injury via inhibiting HMGB1-mediated
TLR4/NF-κB pathway. Biotechnol Appl Biochem. 66:1024–1030. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Qu Y, Chen WH, Zong L, Xu MY and Lu LG:
18α-Glycyrrhizin induces apoptosis and suppresses activation of rat
hepatic stellate cells. Med Sci Monit. 18:BR24–BR32. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Leng T, Zhao J, Xiao Z, Hao S, Xie W and
Nie Q: The impact of the PI3K/AKT pathway on arrhythmia: Mechanisms
of action and therapeutic potential. J Mol Histol. 57:602026.
View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Rana N, Deval Singh H, Garg A, Monika
Tiwari R and Sharma S: Current landscape and recent developments of
PI3K/AKT/mTOR targeted inhibitors for breast carcinoma. Bioorg
Chem. 170:1094302026. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Haque MA, Poullikkas T, Kaisar FMA, Khan
A, Haque S, Mollik M, Mowa MJ, Khatun MH, Mamun A and Bithy MBA:
Targeting tumorigenic coactivators in the PI3K/AKT signaling
pathway: A novel approach for cancer treatment. Cancer Med.
14:e713042025. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Jiang RH, Xu JJ, Zhu DC, Li JF, Zhang CX,
Lin N and Gao WY: Glycyrrhizin inhibits osteoarthritis development
through suppressing the PI3K/AKT/NF-κB signaling pathway in vivo
and in vitro. Food Funct. 11:2126–2136. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Shen CH, Ma ZY, Li JH, Li RD, Tao YF,
Zhang QB and Wang ZX: Glycyrrhizin improves inflammation and
apoptosis via suppressing HMGB1 and PI3K/mTOR pathway in
lipopolysaccharide-induced acute liver injury. Eur Rev Med
Pharmacol Sci. 24:7122–7130. 2020.PubMed/NCBI
|
|
64
|
Wang CY, Kao TC, Lo WH and Yen GC:
Glycyrrhizic acid and 18β-glycyrrhetinic acid modulate
lipopolysaccharide-induced inflammatory response by suppression of
NF-κB through PI3K p110δ and p110γ inhibitions. J Agric Food Chem.
59:7726–7733. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Zhang X, Yang H, Yue S, He G, Qu S, Zhang
Z, Ma B, Ding R, Peng W, Zhang H, et al: The mTOR inhibition in
concurrence with ERK1/2 activation is involved in excessive
autophagy induced by glycyrrhizin in hepatocellular carcinoma.
Cancer Med. 6:1941–1951. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Tsai JJ, Pan PJ, Hsu FT, Chung JG and
Chiang IT: Glycyrrhizic acid modulates apoptosis through
extrinsic/intrinsic pathways and inhibits protein kinase B- and
extracellular signal-regulated kinase-mediated metastatic potential
in hepatocellular carcinoma in vitro and in vivo. Am J Chin Med.
48:223–244. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Wang H, Ge X, Qu H, Wang N, Zhou J, Xu W,
Xie J, Zhou Y, Shi L, Qin Z, et al: Glycyrrhizic acid inhibits
proliferation of gastric cancer cells by inducing cell cycle arrest
and apoptosis. Cancer Manag Res. 12:2853–2861. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Niu Z, Lin J, Hao C, Xu X, Wang C, Dai K,
Deng X, Deng M, Guo Y and Yao W: Glycyrrhizic acid attenuates
pulmonary fibrosis of silicosis by inhibiting the interaction
between HMGB1 and BRG1 through PI3K/Akt/mTOR pathway. Int J Environ
Res Public Health. 19:87432022. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Kao TC, Shyu MH and Yen GC:
Neuroprotective effects of glycyrrhizic acid and
18beta-glycyrrhetinic acid in PC12 cells via modulation of the
PI3K/Akt pathway. J Agric Food Chem. 57:754–761. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Farrukh MR, Nissar UA, Kaiser PJ, Afnan Q,
Sharma PR, Bhushan S and Tasduq SA: Glycyrrhizic acid (GA) inhibits
reactive oxygen species mediated photodamage by blocking ER stress
and MAPK pathway in UV-B irradiated human skin fibroblasts. J
Photochem Photobiol B. 148:351–357. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Parker MI, Nikonova AS, Sun D and Golemis
EA: Proliferative signaling by ERBB proteins and RAF/MEK/ERK
effectors in polycystic kidney disease. Cell Signal. 67:1094972020.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Meng Q and Xia Y: c-Jun, at the crossroad
of the signaling network. Protein Cell. 2:889–898. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Zhu ZH, Li X, He LF, Cai HF, Ye B and Wu
ZM: Glycyrrhizic acid, as an inhibitor of HMGB1, alleviates
bleomycin-induced pulmonary toxicity in mice through the MAPK and
Smad3 pathways. Immunopharmacol Immunotoxicol. 43:461–470. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Gederaas OA, Sharma A, Mbarak S, Sporsheim
B, Høgset A, Bogoeva V, Slupphaug G and Hagen L: Proteomic analysis
reveals mechanisms underlying increased efficacy of bleomycin by
photochemical internalization in bladder cancer cells. Mol Omics.
19:585–597. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Wang D, Wong HK, Feng YB and Zhang ZJ:
18beta-glycyrrhetinic acid induces apoptosis in pituitary adenoma
cells via ROS/MAPKs-mediated pathway. J Neurooncol. 116:221–230.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Jain R, Hussein MA, Pierce S, Martens C,
Shahagadkar P and Munirathinam G: Oncopreventive and
oncotherapeutic potential of licorice triterpenoid compound
glycyrrhizin and its derivatives: Molecular insights. Pharmacol
Res. 178:1061382022. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Prickett TD and Brautigan DL: Cytokine
activation of p38 mitogen-activated protein kinase and apoptosis is
opposed by alpha-4 targeting of protein phosphatase 2A for
site-specific dephosphorylation of MEK3. Mol Cell Biol.
27:4217–4227. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Wu Q, Tang Y, Hu X, Wang Q, Lei W, Zhou L
and Huang J: Regulation of Th1/Th2 balance through OX40/OX40L
signalling by glycyrrhizic acid in a murine model of asthma.
Respirology. 21:102–111. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Tsai JJ, Kuo HC, Lee KF and Tsai TH:
Glycyrrhizin represses total parenteral nutrition-associated acute
liver injury in rats by suppressing endoplasmic reticulum stress.
Int J Mol Sci. 14:12563–12580. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Peng S, Zhang Y, Zhang J, Wang H and Ren
B: ERK in learning and memory: A review of recent research. Int J
Mol Sci. 11:222–232. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Ban JY, Park HK and Kim SK: Effect of
glycyrrhizic acid on scopolamine-induced cognitive impairment in
mice. Int Neurourol J. 24 (Suppl 1):S48–S55. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Kim KJ, Choi JS, Kim KW and Jeong JW: The
anti-angiogenic activities of glycyrrhizic acid in tumor
progression. Phytother Res. 27:841–846. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Wang Y, Wang L, Luo R, Sun Y, Zou M, Wang
T, Guo Q and Peng X: Glycyrrhizic acid against Mycoplasma
gallisepticum-induced inflammation and apoptosis through
suppressing the MAPK pathway in chickens. J Agric Food Chem.
70:1996–2009. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Zhao H, Liu Z, Shen H, Jin S and Zhang S:
Glycyrrhizic acid pretreatment prevents sepsis-induced acute kidney
injury via suppressing inflammation, apoptosis and oxidative
stress. Eur J Pharmacol. 781:92–99. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Yamamoto M, Nagasawa Y and Fujimori K:
Glycyrrhizic acid suppresses early stage of adipogenesis through
repression of MEK/ERK-mediated C/EBPβ and C/EBPδ expression in
3T3-L1 cells. Chem Biol Interact. 346:1095952021. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Yin Q, Wang L, Yu H, Chen D, Zhu W and Sun
C: Pharmacological effects of polyphenol phytochemicals on the
JAK-STAT signaling pathway. Front Pharmacol. 12:7166722021.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Spinelli FR, Colbert RA and Gadina M:
JAK1: Number one in the family; number one in inflammation?
Rheumatology (Oxford). 60 (Suppl 2):ii3–ii10. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Li L, Zhou D, Zheng Y and Xie W:
Expression and functions of the STAT3-SCLIP pathway in chronic
myeloid leukemia cells. Exp Ther Med. 12:3381–3386. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Chen B, Chen Q, Lu M, Zou E, Lin G, Yao J,
Wang L, Gan Y, Chen B, Chen G and Wu L: Hypocrellin A against
intrahepatic Cholangiocarcinoma via multi-target inhibition of the
PI3K-AKT-mTOR, MAPK, and STAT3 signaling pathways. Phytomedicine.
135:1560222024. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
He Y, Wang H, Lin S, Chen T, Chang D, Sun
Y, Wang C, Liu Y, Lu Y, Song J, et al: Advanced effect of curcumin
and resveratrol on mitigating hepatic steatosis in metabolic
associated fatty liver disease via the PI3K/AKT/mTOR and HIF-1/VEGF
cascade. Biomed Pharmacother. 165:1152792023. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
He SQ, Gao M, Fu YF and Zhang YN:
Glycyrrhizic acid inhibits leukemia cell growth and migration via
blocking AKT/mTOR/STAT3 signaling. Int J Clin Exp Pathol.
8:5175–5181. 2015.PubMed/NCBI
|
|
92
|
Wu X, Wang W, Chen Y, Liu X, Wang J, Qin
X, Yuan D, Yu T, Chen G, Mi Y, et al: Glycyrrhizin suppresses the
growth of human NSCLC cell line HCC827 by downregulating HMGB1
level. Biomed Res Int. 2018:69167972018.PubMed/NCBI
|
|
93
|
Tian X, Liu Y, Liu X, Gao S and Sun X:
Glycyrrhizic acid ammonium salt alleviates Concanavalin A-induced
immunological liver injury in mice through the regulation of the
balance of immune cells and the inhibition of hepatocyte apoptosis.
Biomed Pharmacother. 120:1094812019. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Guo M, Wang Z, Dai J, Fan H, Yuan N, Gao
L, Peng H and Cheng X: Glycyrrhizic acid alleviates liver fibrosis
in vitro and in vivo via activating CUGBP1-mediated
IFN-γ/STAT1/Smad7 pathway. Phytomedicine. 112:1545872023.
View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Wei S, Feng M and Zhang S: Molecular
characteristics of cell pyroptosis and its inhibitors: A review of
activation, regulation, and inhibitors. Int J Mol Sci.
23:161152022. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Coll RC, Schroder K and Pelegrín P: NLRP3
and pyroptosis blockers for treating inflammatory diseases. Trends
Pharmacol Sci. 43:653–668. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Khan R, Khan AQ, Lateef A, Rehman MU,
Tahir M, Ali F, Hamiza OO and Sultana S: Glycyrrhizic acid
suppresses the development of precancerous lesions via regulating
the hyperproliferation, inflammation, angiogenesis and apoptosis in
the colon of Wistar rats. PLoS One. 8:e560202013. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Hibasami H, Iwase H, Yoshioka K and
Takahashi H: Glycyrrhizin induces apoptosis in human stomach cancer
KATO III and human promyelotic leukemia HL-60 cells. Int J Mol Med.
16:233–236. 2005.PubMed/NCBI
|
|
99
|
Han Y, Sheng W, Liu X, Liu H, Jia X, Li H,
Wang C, Wang B, Hu T and Ma Y: Glycyrrhizin ameliorates colorectal
cancer progression by regulating NHEJ pathway through inhibiting
HMGB1-induced DNA damage response. Sci Rep. 14:249482024.
View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Bian F, Niu FH, Qu PY, Gong F and Yan JZ:
Glycyrrhizic acid inhibits DNA damage repair and enhances
cisplatin-induced apoptosis of melanoma cells. Chem Biol Drug Des.
103:e145362024. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Wang Z, Yang X, Wang X, Liang F and Tang
Y: Glycyrrhizin attenuates caspase-11-dependent immune responses
and coagulopathy by targeting high mobility group box 1. Int
Immunopharmacol. 107:1087132022. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Hua S, Ma M, Fei X, Zhang Y, Gong F and
Fang M: Glycyrrhizin attenuates hepatic ischemia-reperfusion injury
by suppressing HMGB1-dependent GSDMD-mediated kupffer cells
pyroptosis. Int Immunopharmacol. 68:145–155. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Dong L, Sun JY, Fang GT, Jiang LD and Wang
JY: Effects of glycyrrhizin on TGFbeta1 stimulated hepatic stellate
cell signaling transduction. Zhonghua Gan Zang Bing Za Zhi.
13:828–831. 2005.(In Chinese). PubMed/NCBI
|
|
104
|
Zhou Y, Tong X, Ren S, Wang X, Chen J, Mu
Y, Sun M, Chen G, Zhang H and Liu P: Synergistic anti-liver
fibrosis actions of total astragalus saponins and glycyrrhizic acid
via TGF-β1/Smads signaling pathway modulation. J Ethnopharmacol.
190:83–90. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Li M, Peng S, Bu J, Quan S, Liu L, Yue Z,
Wang L and Li Y: Glycyrrhizic acid alleviates gefitinib-induced
liver injury by regulating the p53/p21 pathway and releasing cell
cycle arrest. Food Chem Toxicol. 200:1154052025. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Cheng X, Liu Y, Qi B, Wang Y, Zheng Y,
Liang X, Chang Y, Ning M, Gao W and Li T: Glycyrrhizic acid
alleviated MI/R-induced injuries by inhibiting Hippo/YAP signaling
pathways. Cell Signal. 115:1110362024. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Xu H, Fang L, Zeng Q, Chen J, Ling H, Xia
H, Ge Q, Wu C, Zou K, Wang X, et al: Glycyrrhizic acid alters the
hyperoxidative stress-induced differentiation commitment of MSCs by
activating the Wnt/β-catenin pathway to prevent SONFH. Food Funct.
14:946–960. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Lai T, Shen Y, Chen C, Huang B, Deng T,
Zhao Z, Zhang Z, Huang Z and Pan X: Glycyrrhizic acid ameliorates
myocardial ischemia-reperfusion injury in rats through inhibiting
endoplasmic reticulum stress. Eur J Pharmacol. 908:1743532021.
View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Zhao TQ, Li Y, Zhang M, Zhao MC, Cao X and
Hou SZ: Glycyrrhizic acid protects glomerular podocytes induced by
high glucose by modulating SNARK/AMPK signaling pathway. Curr Med
Sci. 43:696–707. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Wang Y, Chen Q, Shi C, Jiao F and Gong Z:
Mechanism of glycyrrhizin on ferroptosis during acute liver failure
by inhibiting oxidative stress. Mol Med Rep. 20:4081–4090.
2019.PubMed/NCBI
|
|
111
|
Zhou L, Yang Y, Fu X, Xia B, Li C, Lu C,
Qi Y, Zhang H and Liu T: The protective effect and molecular
mechanism of glycyrrhizic acid glycosides against Tripterygium
glycosides induced nephrotoxicity based on the RhoA/ROCK1
signalling pathway. J Ethnopharmacol. 319:1171002024. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Fang L, Wang X and Guan W: Glycyrrhizic
acid ameliorates sepsis-induced acute lung injury through
suppression of endoplasmic reticulum stress. Discov Med.
37:841–849. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wei L, Ou S, Meng Y, Sun L, Zhang L, Lu Y
and Wu Y: Glycyrrhizin as a potential disease-modifying therapy for
epilepsy: Insights into targeting pyroptosis to exert
neuroprotective and anticonvulsant effects. Front Pharmacol.
15:15307352025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Meng FD, Yuan L, Xu DJ, Che MY, Hou SZ, Lu
DD, Liu WJ and Nan Y: Exploring the targets and molecular mechanism
of glycyrrhetinic acid against diabetic nephropathy based on
network pharmacology and molecular docking. World J Diabetes.
14:1672–1692. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
An J, Han M, Chen P, Wang L, Gao L, Zhou
Z, Huang S, Du J and Peng F: Therapeutic potential of glycyrrhizin
in nervous system diseases. Chin Med J (Engl). 139:457–459. 2026.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Gupta J, Kumar D, Gupta R, Kumar S and
Kumar M: Emerging trends in the pharmacological and therapeutic
potential of ginger: From traditional medicine to nanotechnological
innovations. Curr Pharm Des. 31:2995–3016. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li Q, Deng Y, Han X, Dong Y, Wu X, Zhu W
and Yan M: Naringenin nanosuspensions embedded glycyrrhizin-based
hydrogel ameliorates cholestatic liver injury in mice by inhibiting
oxidative stress and HMGB1-mediated inflammation. Colloids Surf B
Biointerfaces. 261:1154662026. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Cui Q, Song X, Zhou L, Dong J, Wei Y, Liu
Z and Wu X: Fabrication of resveratrol-loaded soy protein
isolate-glycyrrhizin nanocomplex for improving bioavailability via
pH-responsive hydrogel properties. Int J Biol Macromol.
258:1289502024. View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Sun Z, Zhang M, Wei Y, Li M, Wu X and Xin
M: A simple but novel glycymicelle ophthalmic solution based on two
approved drugs empagliflozin and glycyrrhizin: In vitro/in vivo
experimental evaluation for the treatment of corneal alkali burns.
Biomater Sci. 11:2531–2542. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Semwal DK, Kumar A, Semwal RB, Dadhich NK,
Chauhan A and Kumar V: Glycyrrhizin (glycyrrhizic
acid)-pharmacological applications and associated molecular
mechanisms. Drugs Drug Candidates. 4:442025. View Article : Google Scholar : PubMed/NCBI
|