Introduction
Due to their remarkable chemical diversity and
biological activity, natural products have long been recognized as
a notable source of lead compounds with substantial therapeutic
potential. According to published statistics, >70% of drugs
approved internationally between 1981 and 2014 were directly
derived from natural products, natural product derivatives or
synthetic compounds that recapitulate the pharmacological effects
of natural products. Collectively, these observations underscore
the central role of natural products in the development of modern
pharmaceuticals (1,2).
Tea (Camellia sinensis; Theaceae), with a
history of >5,000 years of consumption and medicinal use in
China, is one of the most widely consumed functional beverages
worldwide (3). Black tea, the
predominant tea product, is widely consumed in Europe and North
America, and has been recognized as ‘Generally Recognized as Safe’
(GRAS) by the U.S. Food and Drug Administration (FDA) (4). During black tea manufacture, catechin
polyphenols in tea leaves undergo extensive enzymatic oxidation,
yielding polymeric products such as theaflavins and thearubigins
(5). Among these, theaflavins are
characteristic dimers with a benzotropolone structure (5). In view of their distinctive color and
broad bioactivities, theaflavins are often referred to as the
‘golden molecules’ in black tea and comprise 2–6% of the soluble
solids in brewed black tea (6).
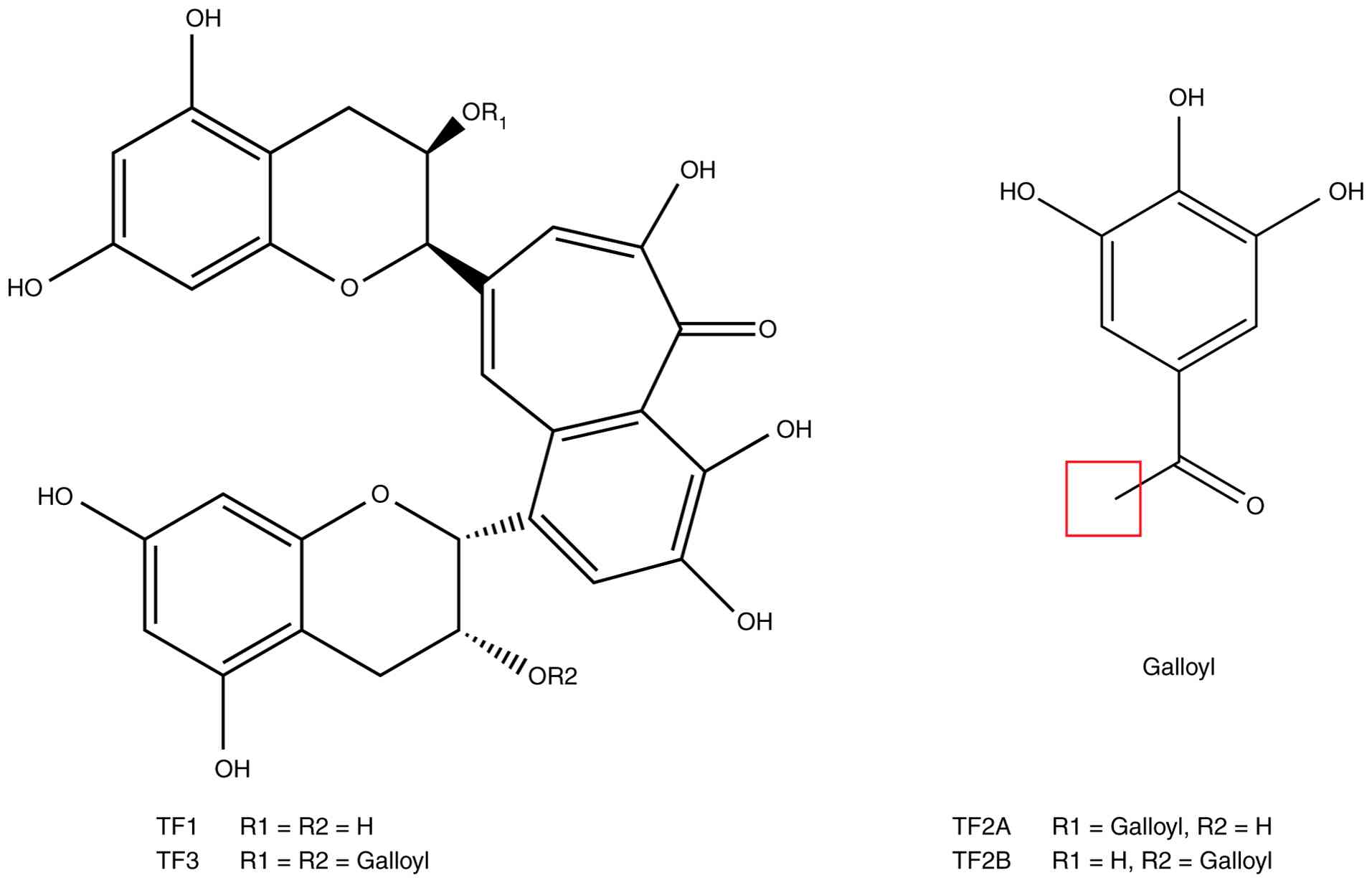
At present, four major theaflavin monomers have been
identified in black tea, including TF derivatives theaflavin (TF1),
theaflavin-3-gallate (TF2A), theaflavin-3′-gallate (TF2B) and
theaflavin-3,3′-digallate (TF3) (7) (Fig.
1). Among these, TF3 is not only the most abundant theaflavin
in black tea (~1.05%), which is markedly significantly surpassing
TF2A (0.34%), TF2B (0.11%) and TF1 (0.08%), but it is also
considered one of the most biologically active theaflavin
components (8,9). Structurally, TF3 is formed via
oxidative dimerization of (−)-epigallocatechin gallate (EGCG) and
(−)-epicatechin gallate (ECG), and contains a high number of
phenolic hydroxyl groups, thereby conferring strong reducing
capacity (9). This structural
basis may partially account for its reported antioxidant,
anti-inflammatory, antiviral, antimicrobial and anticancer
activities (8,10–13).
Studies have indicated that the biological effects
of TF3 are not confined to a single disease entity or signaling
pathway; rather, they span multiple systemic disease domains.
Accumulating evidence suggests that TF3 may exert protective or
interventional effects in tumorigenesis and progression, viral and
bacterial infections, diabetes and its complications, non-alcoholic
and alcoholic liver diseases, osteoporosis and arthritis,
atherosclerosis, and neurodegenerative disorders such as
Alzheimer's disease (14–22). These effects are typically mediated
by the modulation of multiple mechanisms, including cell cycle
regulation, apoptosis, oxidative stress responses, inflammatory
signaling, angiogenesis and metabolic homeostasis, supporting the
multitarget and network-regulatory properties of TF3 (19–22).
To the best of our knowledge, despite the increasing
number of studies on TF3, a comprehensive review integrating its
structural characteristics, biotransformation, mechanisms of action
and disease-related biological effects remains unavailable. In
particular, the understanding of the translational trajectory of
TF3 from a dietary bioactive constituent to applications in
functional foods and nutraceuticals is still fragmented. Therefore,
the present review aims to provide a systematic review focusing on
the chemical structure and metabolic characteristics of TF3, its
principal pharmacological activities and molecular mechanisms, as
well as key issues such as delivery strategies, safety and the
feasibility of human intake, thereby providing a theoretical basis
for further mechanistic research and translational development of
TF3.
Structure and metabolites of TF3
Structure and origin of TF3
As one of the most popular functional beverages
globally, black tea has attracted widespread attention due to its
unique flavor and putative health benefits in humans (23,24).
Numerous epidemiological studies have suggested that tea-derived
polyphenols are associated with a reduced risk of various chronic
diseases (25). The main phenolic
components of black tea are theaflavins and thearubigins, which are
primarily formed during the processing of tea leaves through
enzymatic oxidation and subsequent non-enzymatic oxidation
reactions of catechins (5).
Theaflavins are a class of dimeric polyphenols with
a characteristic benzotropolone skeleton, comprising a central
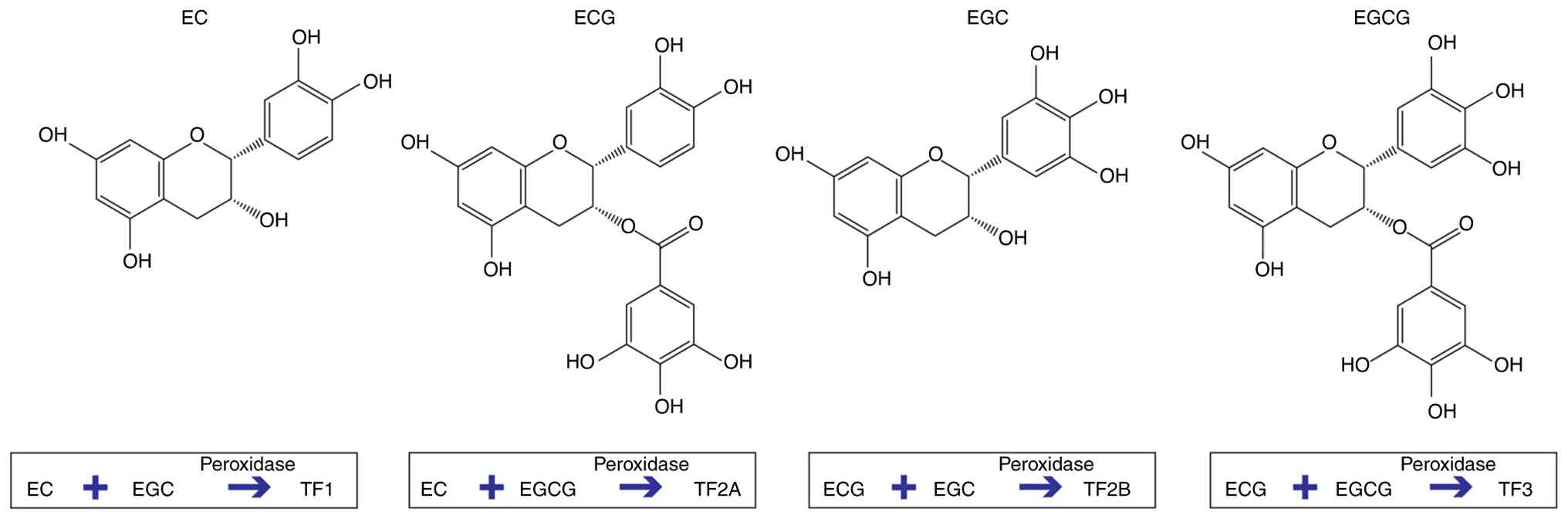
tricyclic structure and two phenolic side-chain rings (5). Biological and pharmacological study
have generally focused on the four major tea catechins, namely ECG,
EGCG, (−)-epicatechin (EC) and (−)-epigallocatechin (EGC) (26). During black tea
fermentation/manufacture, these catechins undergo oxidative
polymerization, catalyzed by polyphenol oxidase or peroxidase,
leading to the formation of different structural types of
theaflavin monomers through pathways such as C2-C2′ or C4-C8
coupling (5,27). EC and EGC combine to form TF1, EC
and EGCG combine to form TF2A, ECG and EGC combine to form TF2B,
and ECG and EGCG combine to form TF3 (9) (Fig.
2). Accordingly, structural differences among these theaflavins
are considered to contribute to variation in biological potency and
functional specificity (5).
TF3 is the most abundant (~1.05%) and is widely
regarded as one of the most biologically active theaflavin
constituents in black tea (28).
TF3 is formed through the enzyme-catalyzed oxidative dimerization
of EGCG and ECG, and its molecular structure is rich in eight
phenolic hydroxyl groups and multiple ester bonds, thereby
conferring strong free radical-scavenging capacity. Collectively,
these structural features provide a physicochemical basis for its
antioxidant, anti-inflammatory and anticancer effects (10,18,29).
Stability and degradation mechanisms
of TF3
Although TF3 exhibits strong biological activity,
its intrinsic structural lability contributes to rapid degradation
in complex environments, thereby limiting its further application
in food and medical contexts (30). The abundant phenolic hydroxyl
groups in TF3 are prone to redox reactions, and its stability is
influenced by multiple factors, including solution pH, temperature,
oxygen exposure, light and the presence of metal ions (31–33).
Available evidence indicates that TF3 is highly
susceptible to degradation under near-neutral or alkaline
conditions. In vitro experiments using high-performance
liquid chromatography (HPLC) have demonstrated that after
incubation for 35 min at 37°C in 50 mM Tris-HCl buffer, TF3 content
decreased by >50% (12).
Furthermore, TF3 exhibits pronounced thermal instability, as it is
prone to hydrolysis and/or degradation under high-temperature tea
brewing or pasteurization conditions (6). Oxygen is also a key determinant of
stability. Oxygen-rich environments can promote rapid oxidation,
resulting in the formation of pigmented byproducts (30). In addition, within food matrices,
TF3 may undergo non-enzymatic binding or complexation with
proteins, polysaccharides or minerals, further compromising
bioactivity retention and oral bioavailability (34). Accordingly, in the development of
tea-based functional foods, parameters such as storage-related pH
fluctuations, thermal processing conditions, oxygen control and
interactions with other food components should be systematically
considered to improve TF3 stability in complex matrices.
Bioavailability, metabolites and
conversion of TF3
The oral bioavailability of TF3 under physiological
conditions is generally low, primarily due to its high polarity,
large molecular weight (~868.7 Da), low lipophilicity and intrinsic
structural lability (35). An
animal study showed that after continuous consumption of black tea
containing TF3 (50 mg/g diet) for 2 weeks, TF3 concentrations in
mice remained markedly low (~1 nmol/g) (36). In a human study, volunteers who
ingested 700 mg of a pure theaflavin mixture (equivalent to
drinking ~30 cups of black tea) had a maximum plasma concentration
of TF3 of 1 ng/ml, with a urinary concentration of 4.2 ng/ml
(37). Collectively, these
findings indicate that the in vivo absorption efficiency of
TF3 is low, with bioavailability constrained by gastrointestinal
instability (such as pH-induced degradation), limited transmembrane
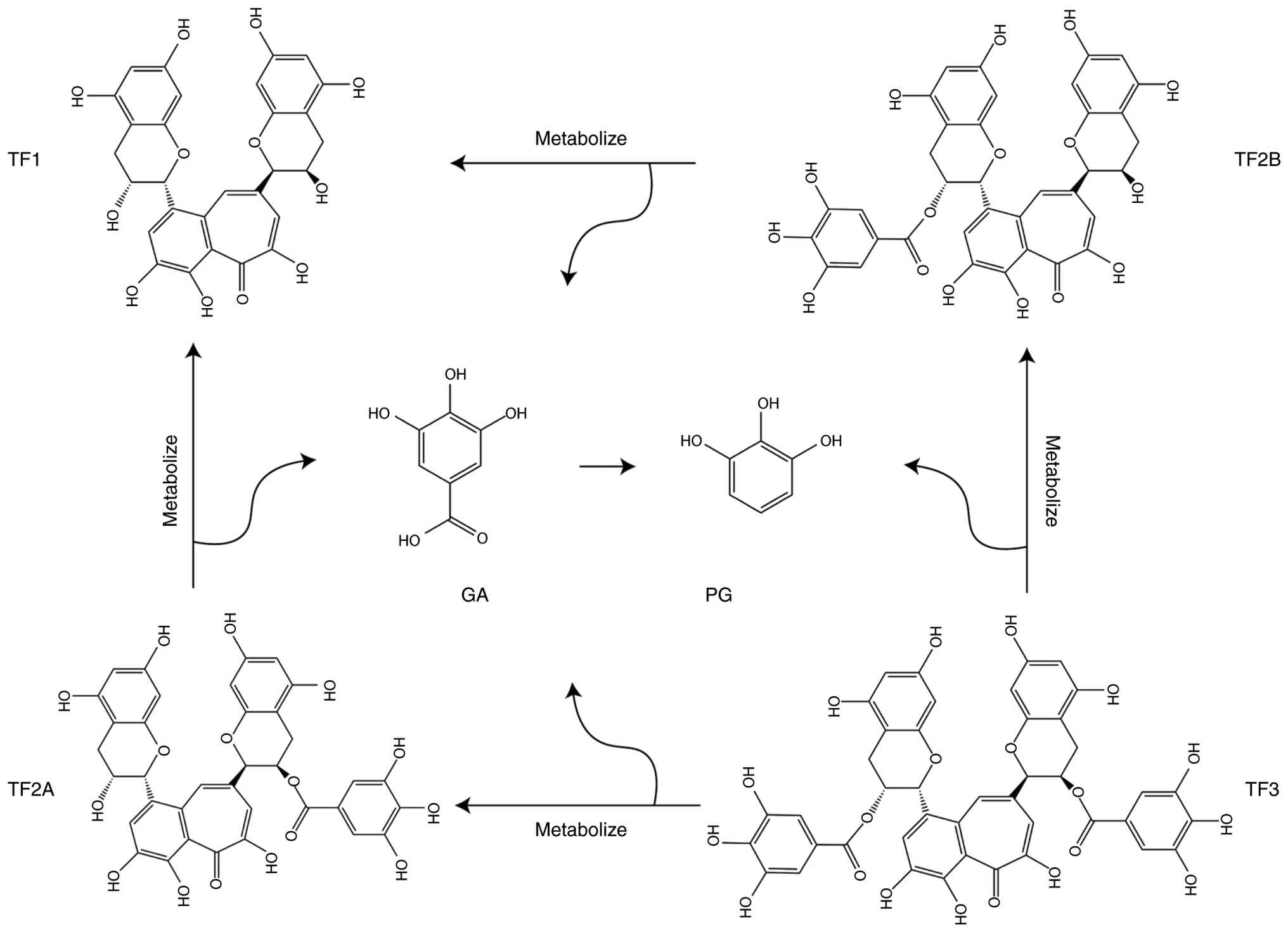
transport and substantial first-pass metabolism (38). During digestion, unabsorbed TF3 can
enter the colon and undergo gradual transformation into a series of
small-molecule metabolites under the action of the gut microbiota,
including theaflavin, TF2A, TF2B, gallic acid (GA) and
protocatechuic acid (PG) (39)
(Fig. 3). Probiotic strains such
as Lactobacillus plantarum 299v and Bacillus subtilis
have been reported to participate in the metabolic transformation
of TF3, suggesting that the gut microbiota may serve a notable role
in shaping TF3-associated pharmacological effects (40,41).
Although the parent compound of TF3 appears to exert
limited biological effects in vivo, studies have
increasingly suggested that its metabolites possess pharmacological
activities and may synergistically contribute to the overall in
vivo activity attributed to TF3. The partially de-esterified
dimers, TF2A and TF2B, retain antioxidant and antimicrobial
activities, and may continue to exert effects as colon-delivered
active forms (41–43). GA, a degradation product of TF3,
has been reported to inhibit NF-κB signaling and reduce
inflammatory cytokine expression in multiple disease models,
indicating anti-inflammatory and anticancer potential (44,45).
Similarly, PG has been reported to induce apoptosis in A549 lung
cancer cells, inhibit tumor growth in vivo and exhibit
antioxidant activity (46–48).
The microbiota-driven conversion of TF3 into GA and
PG may shift the bioactivity profile of TF3 toward enhanced
anticancer and anti-inflammatory effects (49); however, to the best of our
knowledge, the extent to which this metabolic process contributes
to specific disease outcomes remains unclear. Addressing this gap
will require integrated pharmacokinetic-pharmacodynamic
investigations that compare TF3 and its metabolites within the same
disease models to delineate metabolite contributions to overall
efficacy. In this context, TF3 may function as a precursor or
prodrug, which could broaden its application as a functional food
ingredient or dietary supplement. Overall, these observations
suggest that TF3-related biological effects may not be solely
dependent on the parent structure but may instead reflect the
combined actions of metabolites such as GA, PG, TF2A and TF2B.
Therefore, elucidating the mechanisms of action of TF3-derived
metabolites and systematically comparing their pharmacological
profiles with the parent compound will help define the in
vivo functional positioning of TF3 and provide a theoretical
rationale for developing more targeted and accessible delivery
strategies.
Biological activities of TF3
TF3 has been reported to exhibit a variety of
biological activities, including antioxidant, anti-inflammatory,
antimicrobial, antiviral and anticancer properties. These
characteristics suggest that TF3 may contribute to the modulation
of multiple disease processes (12,13,50–52).
Numerous studies have indicated that the pleiotropic activities of
TF3 support its potential relevance to neurodegenerative disorders,
periodontal diseases, viral pathogenesis and cancer. Furthermore,
available evidence suggests that TF3 influences key
disease-associated pathways, including the canonical inflammatory
and stress signaling axes (NF-κB and MAPK), antioxidant defense
systems [nuclear factor erythroid 2-related factor 2
(Nrf2)/Kelch-like ECH-associated protein 1 (Keap1)], virus
replication-associated proteases [such as 3C-like protease (3CLpro)
and non-structural protein 2b-non-structural protein 3 (NS2B-NS3)],
and multiple receptor tyrosine kinases and their downstream
signaling cascades [such as EGFR and platelet-derived growth factor
receptor (PDGFR)] (50,53–55).
Antioxidant properties of TF3
Tea is widely regarded as a natural antioxidant,
largely attributable to the redox-active properties of its
polyphenols (56). Studies have
shown that catechins possess substantial antioxidant capacity,
enabling the inhibition of free-radical generation, direct
scavenging of reactive species and chelation of transition metal
ions, thereby limiting lipid peroxidation in vitro and in
vivo (57,58). Theaflavins, formed through the
oxidation of epicatechin, are also considered to exhibit
antioxidant potential, as they can reduce intracellular reactive
oxygen species (ROS) and mitigate hydroxyl radical-induced DNA
damage (59). Among the four major
theaflavins in black tea, TF3 exhibits the highest scavenging
activity against hydrogen peroxide and
2,2-diphenyl-1-picrylhydrazyl (DPPH) radicals. TF1 scavenges
superoxide anion radicals, whereas TF2B can scavenge singlet oxygen
and hydrogen peroxide, and attenuate hydroxyl radical-induced DNA
damage (9). These findings suggest
that galloylated theaflavins display stronger scavenging activity
against hydroxyl and DPPH radicals than their non-galloylated
counterparts.
Oxidative stress can induce neuronal apoptosis via
excessive generation of ROS, such as superoxide anions and hydroxyl
radicals and is widely implicated in the pathogenesis of
neurodegenerative diseases (60).
TF3 has been reported to suppress oxidative stress by modulating
oxidase activity and to exert neuroprotective effects in
pheochromocytoma PC12 cells, primarily through the inhibition of
oxidase activity, which reduces cell apoptosis (60). Additionally, TF3 promotes the
dissociation of Nrf2 from Keap1, induces Nrf2 phosphorylation and
facilitates its nuclear translocation, where Nrf2 binds to the
antioxidant response element and initiates transcription of heme
oxygenase-1, superoxide dismutases, glutathione (GSH) peroxidase
and catalases (61).
However, it has been indicated that TF3 exhibits
cell type-dependent effects, yielding divergent outcomes in
malignant tumor cells compared with normal cells. For instance, in
normal GN46 fibroblasts, TF3 exerts antioxidant effects; at 250 µM,
it promotes intracellular GSH synthesis by activating
δ-glutamylcysteine synthetase and GSH synthetase. By contrast, in
oral squamous cell carcinoma HSC-2 cells, TF3 (250–500 µM) induces
apoptosis by disrupting redox homeostasis and antioxidant defenses,
resulting in decreased GSH levels and caspase-3 activation, which
subsequently leads to poly (ADP-ribose) polymerase cleavage
(56). Concurrently, TF3 induces
apoptosis primarily via activation of the intrinsic mitochondrial
pathway, integrating oxidative stress signaling, Bcl-2 family
regulation, and caspase cascade activation (5,62).
The detailed process is shown in Fig.
4A.
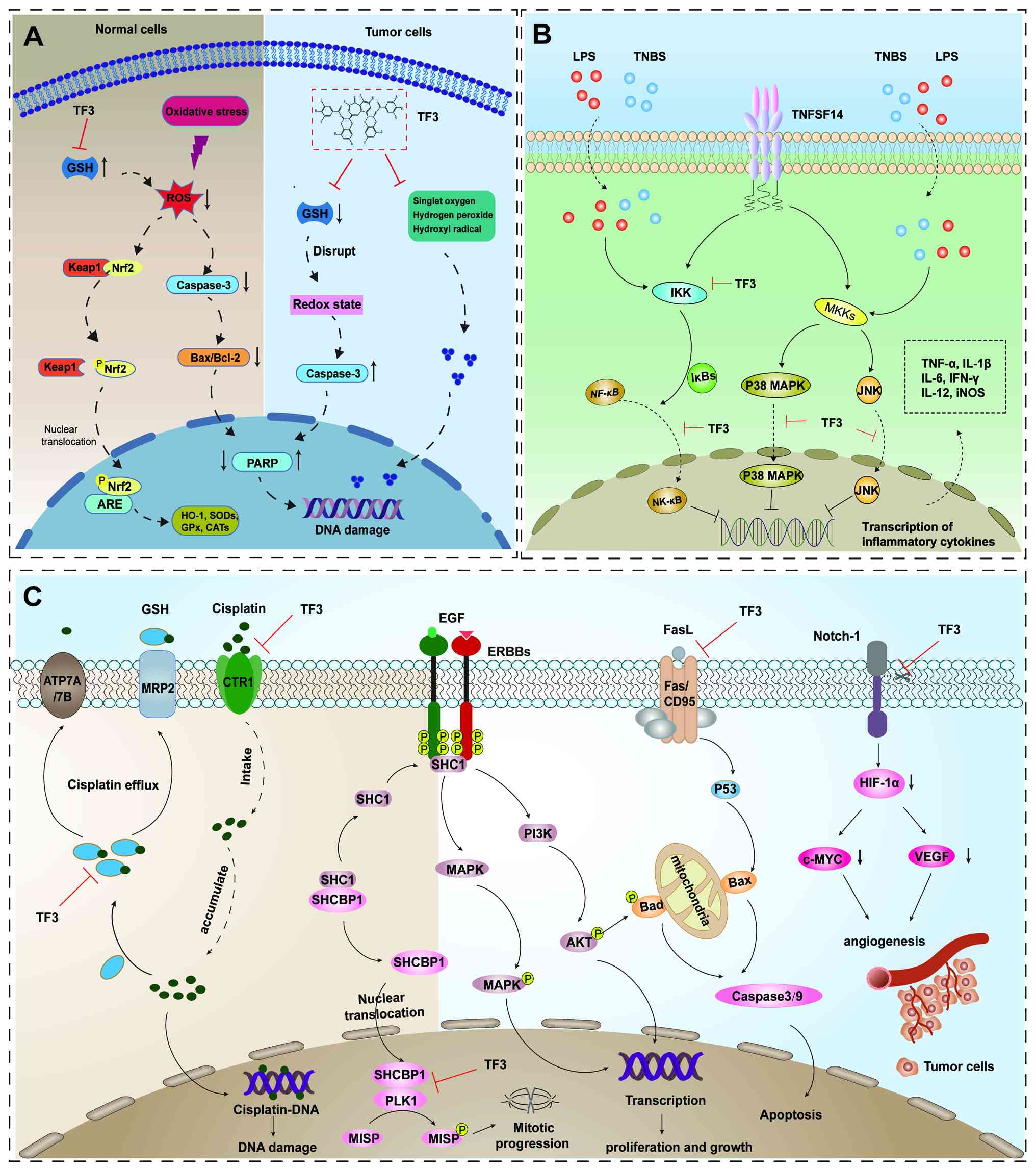 | Figure 4.Schematic illustration of the
molecular mechanisms of TF3 in antioxidant defense, inflammation
suppression and tumor inhibition. (A) Antioxidant mechanism of TF3.
In normal cells, TF3 markedly decreases ROS levels by promoting the
dissociation of Nrf2 from Keap1 inducing Nrf2 phosphorylation, and
facilitating its translocation into the nucleus to interact with
ARE, which initiates transcription of HO-1, SODs, GPx and CATs. In
cancer cells, TF3 decreases GSH levels, disrupts redox homeostasis
and activates caspase-3, resulting in increased PARP cleavage
(cleaved PARP ↑) and apoptosis. By contrast, in normal cells, TF3
maintains redox balance and reduces PARP cleavage (cleaved PARP ↓).
(B) Anti-inflammatory mechanism of TF3. TF3 reduces the levels of
pro-inflammatory cytokines TNF-α, IL-1β and IL-6 by suppressing the
activation of MAPK, JNK and NF-κB. (C) Antitumor mechanism of TF3.
TF3 decreases GSH levels to reduce efflux relaxation of cisplatin
and improve the expression of copper CTR1, which heightens the
sensitivity of ovarian cancer cells to cisplatin. When EGF
activates HER2, SHC1 dissociates from SHCBP1 and is recruited by
HER2 to activate the classical MAPK and PI3K signaling pathways.
Dissociated SHCBP1 enters the nucleus and interacts with PLK1,
which promotes phosphorylation of the mitotic protein MISP to
regulate mitosis. TF3 attenuates tumor cell-induced angiogenesis by
suppressing the cleavage of Notch-1 and subsequently decreasing
HIF-1α, c-Myc and VEGF expression. TF3, theaflavin-3,3′-digallate;
ROS, reactive oxygen species; GSH, glutathione; ARE, antioxidant
response element; HO-1, heme oxygenase-1; SODs, superoxide
dismutases; GPx, GSH peroxidase; CATs, catalases; PARP,
poly(ADP-ribose) polymerase; CTR1, copper transporter 1; SHC1, Src
homology 2 domain-containing transforming protein 1; SHCBP1, Shc
SH2-domain binding protein 1; PLK1, polo-like kinase 1; MISP,
mitotic spindle positioning protein; HIF-1α, hypoxia-inducible
factor 1α; TNBS, 2,4,6-trinitrobenzenesulfonic acid; MKK,
mitogen-activated protein kinase kinase; Keap1, Kelch-like
ECH-associated protein 1; Nrf2, nuclear factor erythroid 2-related
factor 2; LPS, lipopolysaccharide; TNFSF14, TNF superfamily member
14; ATP7A/7B, ATPase copper transporting α/β; FasL, Fas ligand;
MRP2, resistance-associated protein 2; P, phosphorylated. |
In summary, TF3 may function as an antioxidant that
contributes to the prevention and management of neurodegenerative
diseases and osteoporosis through regulation of cellular oxidative
stress (60,63). Conversely, in cancer models, TF3
can disrupt redox homeostasis by promoting excessive ROS generation
and activating apoptosis signaling pathways, thereby acting as an
inducer of oxidative stress-mediated apoptosis (64). Taken together, these observations
underscore the potential of TF3 in modulating redox-related
pathways relevant to human health.
Anti-inflammatory properties of
TF3
Inflammation is a protective response of biological
systems to external stimuli and serves as an essential mediator of
host defense. However, dysregulated or uncontrolled inflammation
can result in chronic, low-grade inflammatory states, ultimately
contributing to the development of multisystem diseases (65). Lipopolysaccharide (LPS), a
component of the outer membrane of Gram-negative bacteria, can
stimulate macrophages and circulating monocytes to release
pro-inflammatory cytokines, leading to transient immune activation
characterized by elevated levels of TNF-α, IL-1β and IL-6 (66,67).
TF3 reduces the levels of these cytokines by inhibiting
phosphorylation of p38 MAPK and JNK, as well as by suppressing the
nuclear translocation of NF-κB (p65) in LPS-treated RAW 264.7
macrophages (68). TF3 also
alleviates LPS-induced acute lung injury in mice (69).
Another study has demonstrated that oral
administration of TF3 (5 mg/kg; oral gavage) notably improved
trinitrobenzene sulfonic acid (TNBS)-mediated colitis by reducing
the mRNA and protein levels of IL-12, IFN-γ, TNF-α and inducible
nitric oxide synthase in the colonic mucosa. Additionally, TF3
markedly inhibited TNBS-induced NF-κB nuclear localization and
cytoplasmic IκB kinase activity, while preserving the stability of
the NF-κB inhibitor IκBα in colonic tissues and activated
macrophages (10,70). IL-6 contributes to bone resorption
and is positively associated with periodontal disease progression.
TF3 suppresses TNF superfamily member 14 (TNFSF14)-induced IL-6
production, attenuates TNFSF14 receptor expression, and blocks
activation of ERK and NF-κB in human gingival fibroblasts (71) (Fig.
4B).
In summary, these findings suggest that TF3 exerts
anti-inflammatory effects predominantly through the inhibition of
both NF-κB and MAPK activation thereby supporting its potential as
a therapeutic candidate for inflammation-related disorders.
Antimicrobial properties of TF3
TF3 has been reported to exhibit bactericidal
activity. The 2-methyl-D-erythritol-4-phosphate terpene
biosynthesis pathway is needed for the survival of most bacteria
and numerous human pathogens, rendering it a potential target for
the identification of novel antimicrobial agent (72). Deoxy-D-xylulose-5-phosphate
reductoisomerase (DXR) is a validated antimicrobial target within
this pathway (73). Among the four
major theaflavins in black tea, TF1, which lacks a GA ester side
chain, exhibits the lowest inhibitory activity against DXR
(IC50 >100 mM), whereas the other three theaflavins,
each containing at least one GA ester side chain, show stronger
inhibition (IC50=14.9–29.2 mM). Docking simulations
further indicate that TF3 interacts more strongly with DXR than
other theaflavins, owing to additional hydrogen bonding and close
contact at the binding interface. This observation not only helps
explain the stronger DXR inhibitory activity of TF3 relative to TF1
but also underscores the contribution of organic acid ester side
chains to the antimicrobial activity of theaflavins (12). Furthermore, TF3, as a natural
antimicrobial agent, exhibits antibacterial and sporicidal
activities, including disruption of bacterial membranes, inhibition
of metabolic activity and suppression of spore germination via
binding to key spore-associated proteins. Accordingly, TF3 may have
utility not only against common bacterial pathogens but also as a
complementary strategy for infections involving
antibiotic-resistant bacteria (74).
Metallo-β-lactamases (MBLs) are β-lactam resistance
determinants produced by a range of Gram-negative bacteria and are
major contributors to the increasing resistance to β-lactam
antibiotics (75). It has been
shown that certain pathogens, such as Staphylococcus aureus,
can acquire resistance to β-lactam antibiotics by producing
β-lactamases that hydrolyze the amide bond of the β-lactam ring.
Various β-lactam anti-infective regimens have been developed for
methicillin-resistant S. aureus, including
penicillin-β-lactamase inhibitor combinations, and first-, second-
and third-generation cephalosporins. TF3 reduces the minimum
inhibitory concentration of β-lactam antibiotics against the S.
aureus BAA1717 strain by 4- or 8-fold (fractional inhibitory
concentration index=0.313 or 0.188, respectively), whereas TF3
alone exhibits weak antibacterial activity. Additionally, TF3
provides marked protection against MBL-mediated hydrolysis of
nitrocefin (76). Its inhibitory
effects on three distinct MBL variants suggest broad-spectrum
inhibitory potential (76). TF3
also inhibits the production, secretion and activity of α-hemolysin
(Hla), alleviates Hla-associated immune responses and skin injury,
and protects the skin barrier, thereby supporting its future
development as a potential MBL-targeting adjunct (77).
TF3 also exhibits potential oral health benefits.
Research has suggested that it enhances the antibacterial effects
of antimicrobial agents against periodontal disease by inhibiting
the secretion of IL-8 and while increasing the secretion of
β-defensins in oral epithelial cells (78). Furthermore, theaflavins inhibit the
proliferation of Streptococcus mutans, showing protective
effects against dental caries in vitro. The underlying
mechanism involves reducing the formation of biofilm matrices
containing glucans and extracellular DNA (eDNA) (79). TF3 also attenuates the expression
of genes encoding glucosyltransferases [including
Glucosyltransferase B (gtfB), gtfC and gtfD], thereby potentially
lowering S. mutans-associated cariogenicity. Furthermore,
TF3 reduces eDNA formation in S. mutans biofilms by
negatively regulating genes involved in cell autolysis and membrane
vesicle-associated components, such as lysis-related gene A (lrgA),
lrgB and packaging enzyme A (80,81).
Another study has indicated that TF3 inhibits S. mutans
biofilm formation by suppressing enolase, lactate dehydrogenase,
F-type ATPase and proline dehydrogenase activity. Additionally, TF3
exerts antimicrobial activity against Listeria monocytogenes
by disrupting the cell membrane, altering the membrane potential
and fluidity, and inhibiting biofilm maturation. These actions
position TF3 as a natural antimicrobial candidate with potential
applications in food-surface disinfection and prevention of
bacterial contamination (51).
Furthermore, TF3 specifically interferes with quorum sensing
pathways, including the gelatinase (GelE) and serine protease
(SprE) system, as well as the protein translocation channel SecY,
and membrane protein functions, effectively inhibiting biofilm
formation by Enterococcus faecalis rather than relying
solely on bactericidal activity. Accordingly, TF3 may represent a
safe and feasible natural strategy for preventing or adjunctively
managing E. faecalis-associated root canal infections,
endocarditis and urinary tract infections (82).
In conclusion, TF3 exerts antimicrobial effects by
targeting enzymes related to bacterial survival and resistance
(such as DXR, MBLs, enolase, lactate dehydrogenase, F-type ATPase
and Hla), disrupting cell membranes and inhibiting spore
germination. Furthermore, when combined with antibiotics, TF3 can
enhance antibacterial efficacy. Taken together, TF3 may have
translational potential as a natural antimicrobial agent or
adjunct.
Antiviral properties of TF3
Human coronaviruses are major causes of upper
respiratory tract diseases in both animals and humans. Severe acute
respiratory syndrome coronavirus (SARS-CoV) is the etiological
agent of SARS (83,84); in the absence of widely effective,
specific therapeutics for SARS-CoV and given the continuing need
for antiviral drug development, identifying inhibitors targeting
the SARS-CoV main protease has become particularly important.
3CLpro of SARS-CoV has been identified as a promising drug target
(54). Among 720 natural product
compounds, 3-iso-theaflavin-3-gallate, tannic acid and TF3 were
identified as potential inhibitors of 3CLpro, exhibiting potent
inhibitory effects at concentrations <10 mM (16). Another study investigating
FDA-approved antiviral drugs for coronavirus disease 2019
(COVID-19) and SARS-CoV-2 proteases (covering 1,129 antiviral drug
ligands, 459 antimalarial drug ligands and 110 plant-specific
drugs) demonstrated that lopinavir, amoxicillin and TF3 exhibited
the highest docking scores. The predicted protease-inhibitor
complexes were then validated through 20-nsec molecular dynamics
simulations, which showed plausible conformational changes during
binding and favorable affinity for the main protease binding site
(85).
The RNA-dependent RNA polymerase (RdRp) involved in
SARS-CoV-2 replication provides another potential antiviral target
(86,87). Multiple studies have shown that
plant-derived polyphenols can inhibit the RdRp of RNA viruses.
Using a polyphenol database, several polyphenols were screened and
evaluated for their potential in treating COVID-19 by binding to
RdRp (88). Molecular docking
experiments indicated that EGCG, TF1, TF2A, TF2B, TF3, hesperidin,
quercetin and kaempferol can bind to the dynamic catalytic pocket
of SARS-CoV-2 RdRp. Furthermore, binding free energy estimates
derived from 150-nsec molecular dynamics simulations suggested that
EGCG, TF2A, TF2B and TF3 adopt relatively stable binding
conformations with RdRp (84).
Additionally, TF3 has been reported to bind to the SARS-CoV-2
SARS-Unique Domain, blocking its interactions with guanine
quadruplex RNA and host proteins, thereby inhibiting viral
replication and protein translation (89). Overall, these bioactive compounds
exhibit broad antiviral activity against COVID-19 and have been
proposed to display favorable pharmacokinetic characteristics
(90).
Regarding other viruses, theaflavins can block HIV-1
envelope glycoprotein-mediated membrane fusion, preventing HIV-1
entry into target cells and thereby exerting anti-HIV-1 activity
(91). TF3 may block the
interaction between the HIV-1 envelope glycoprotein and the target
cell membrane by binding to a highly conserved hydrophobic pocket
within the central trimeric helical structure formed by the
N-terminal heptad repeat of glycoprotein 41 (91). Furthermore, research has indicated
that TF3 is a potent inhibitor of the flavivirus NS2B-NS3
heterodimeric serine protease. TF3 binds to the Zika virus (ZIKV)
protease, thereby impairing viral maturation and replication. TF3
can also prevent viral polyprotein cleavage in ZIKV-infected cells
or in cells expressing only the NS2B-NS3 protease. Docking
simulations suggest that TF3 interacts with several key residues
within the ZIKV protease cleavage cavity (11). Finally, TF3 inhibits herpes simplex
virus (HSV) activity under neutral or acidic conditions. HSV is a
major cause of genital ulcers. When combined with lactic acid, TF3
inactivates HSV in low-pH environments, such as liquid semen and
cervicovaginal fluids (92).
In summary, these studies suggest that TF3 interacts
with multiple viral enzymes and domains, and can inhibit the
activity of SARS-CoV-2, HIV-1, ZIKV and HSV, supporting its
potential as a candidate antiviral agent.
Anticancer properties of TF3
Over the past five decades, the understanding of the
bioactive effects of phytochemicals in suppressing tumor initiation
and progression has progressively expanded (93). A substantial body of evidence
indicates that polyphenolic compounds derived from black tea
exhibit anticancer activity against lung, liver, gastric, breast
and ovarian cancer (13,94–96).
Ovarian cancer is one of the most common and lethal
gynecological malignancies (97).
Due to low rates of early detection and the development of
resistance to first-line chemotherapeutic agents, the prognosis
remains poor, with a global 5-year survival rate of ~30% around the
year 2000 (97). Ovarian cancer
stem cells, characterized by differentiation, self-renewal and
metastatic capacity, are key contributors to recurrence and drug
resistance. Notably, TF3 exhibits lower cytotoxicity toward normal
ovarian cells than toward cancer cells, and suppresses the
viability of the ovarian cancer cell lines A2780/CP70 and OVCAR3by
modulating the Wnt/β-catenin signaling pathway (98). Another study has reported that TF3
reduced c-Myc, hypoxia-inducible factor 1α and VEGF expression by
inhibiting Notch-1 cleavage, thereby attenuating OVCAR-3-induced
angiogenesis in human umbilical vein endothelial cells and chick
chorioallantoic membrane models (13,99)
(Fig. 4C). Several studies have
also shown that TF3 suppresses tumor progression by activating
apoptosis-related signaling. TF3 in combination with ascorbic acid
activates caspase-3 and caspase-9 via the MAPK pathway, inducing
apoptosis in esophageal cancer Eca-109 cells and lung
adenocarcinoma SPC-A-1 cells (98,100,101). Furthermore, TF3 regulates the
ratio of pro- and anti-apoptotic proteins through Chk2
phosphorylation, initiating intrinsic apoptosis in a
p53-independent manner and inducing extrinsic apoptosis in ovarian
cancer by upregulating death receptor expression. TF3 also promotes
G0/G1 cell cycle arrest by enhancing p27
expression, thereby modulating ovarian cancer cell cycle
progression (102).
These studies suggest that TF3 inhibits ovarian
cancer-related malignant phenotypes and may represent a candidate
therapeutic agent. Notably, TF3 has been investigated as an
adjuvant to cisplatin in advanced ovarian cancer and exhibits
synergistic cytotoxicity in A2780/CP70 and OVCAR3 cells (103). Subsequent study has further
indicated that TF3 enhances cisplatin-induced DNA damage by
promoting intracellular accumulation of platinum (Pt) and formation
of DNA-Pt adducts, thereby attenuating cisplatin resistance.
Additionally, TF3 decreases GSH levels to reduce cisplatin efflux
and increases copper transporter 1 expression, consequently
enhancing ovarian cancer cell sensitivity to cisplatin (104). Multidrug resistance-associated
protein 2 [MRP2; gene name ATP-binding cassette (ABC)C2], a member
of the ABC transporter family, plays a critical role in the
development of cisplatin resistance. Accumulating in vitro
and in vivo evidence has demonstrated that MRP2 promotes
tumor cell resistance through multiple mechanisms, including the
regulation of cisplatin efflux, glutathione (GSH) conjugation
metabolism, and intracellular platinum accumulation (68).
Mutations in the p53 tumor suppressor gene
contribute to tumorigenesis and progression, with ~50% of malignant
tumors harboring p53 mutations (105). Theaflavins activate the death
receptor FAS, a member of the TNF receptor family involved in
programmed cell death, and inhibit the phosphorylated (p-)Akt/p-Bad
cell survival pathway, thereby promoting apoptosis in p53-mutant
breast cancer cells. Furthermore, theaflavins enhance the
p53/ROS/p38 MAPK positive feedback loop and suppress the
pro-migratory proteases MMP-2 and MMP-9, thereby inhibiting
migration in human breast cancer cells (106,107). Additionally, TF3 inhibits the
proliferation of androgen-responsive prostate cancer LNCaP cells by
suppressing androgen receptor expression, thereby reducing
androgen-mediated prostate-specific antigen and fatty acid synthase
levels (108). Furthermore, TF3
induces apoptosis and cell cycle arrest in prostate cancer cells by
activating the protein kinase Cδ/acid sphingomyelinase signaling
pathway, which is dependent on 67 kDa laminin receptor expression,
thereby inhibiting cancer cell proliferation. Notably, TF3 exhibits
low cytotoxicity in normal prostate cells and selectivity toward
cancer cells, supporting its potential as a candidate for
anti-prostate cancer therapy (109).
In addition, the study has suggested that TF3 can
inhibit receptor tyrosine kinase activity and downstream signaling.
In NIH3T3 fibroblasts and human squamous carcinoma A431 cells, TF3
notably inhibits the autophosphorylation of EGFR and PDGFR,
mediated by EGF and PDGF, respectively (55). Accordingly, TF3 can attenuate EGFR
interactions and downstream mitogenic signaling (110). Overexpression of fatty acid
synthase in human breast cancer MCF-7 cells has been reported to
further amplify EGF stimulation. TF3 restricts fatty acid synthase
activity at both the protein and mRNA level, thereby limiting lipid
synthesis and proliferation in MCF-7 cells (111).
Furthermore, overactivation of the Src homology 2
domain-containing transforming protein 1 (SHC1)/Shc SH2-domain
binding protein 1 (SHCBP1)/polo-like kinase 1 (PLK1) signaling axis
is implicated in gastric cancer resistance to HER2-targeted agents
such as trastuzumab, and has been described as a non-canonical
downstream pathway of HER2. Upon EGF-driven activation of HER2,
SHC1 dissociates from SHCBP1 and binds to HER2, thereby activating
the canonical MAPK and PI3K pathways. Released SHCBP1 translocates
into the nucleus and interacts with PLK1, and thereby facilitating
mitotic progression in gastric cancer cells. Notably, TF3 inhibits
formation of the SHCBP1-PLK1 complex, promotes its dissociation and
suppresses this signaling cascade (112). Combination treatment with TF3 and
trastuzumab has been reported to enhance therapeutic efficacy
against gastric cancer in vitro and in vivo (112).
TF3 inhibits osteosarcoma cell proliferation and
promotes apoptosis by increasing ROS levels, inducing DNA damage,
activating caspase signaling and perturbing cell-cycle progression.
These mechanisms support TF3 as a candidate anticancer compound for
osteosarcoma and provide a theoretical rationale for its further
evaluation in osteosarcoma therapy (14). TF3 also inhibits nasopharyngeal
carcinoma (NPC) cell proliferation, migration and invasion through
multiple mechanisms, including the induction of apoptosis,
suppression of metastasis-related signaling pathways, and
metabolomics analyses indicate that TF3 modulates metabolic
pathways in NPC cells. TF3-associated apoptosis, metabolic
reprogramming and regulation of key pathways support further
investigation as a natural compound for NPC intervention (113).
In conclusion, TF3 exerts anticancer effects across
multiple tumor types, including ovarian cancer, esophageal cancer,
lung cancer, gastric cancer, breast cancer, prostate cancer,
osteosarcoma and NPC, by promoting apoptosis, inducing cell cycle
arrest, and modulating receptor tyrosine kinase signaling and
downstream pathways (93–96,110,112). Additionally, TF3 may serve as an
adjuvant or sensitizer in combination with conventional
chemotherapeutic agents to enhance anticancer efficacy.
Other biological functions of TF3
Antidiabetic and cardiovascular protective
effects
Diabetes is a major global health burden and is
frequently accompanied by cardiovascular complications, which
represent the leading cause of death among patients with diabetes
(114). Increasing evidence
suggests that TF3 exerts multitarget antidiabetic effects by
improving insulin sensitivity, regulating hepatic glucose
metabolism and promoting pancreatic β-cell regeneration (19,115). By coordinately modulating glucose
homeostasis and insulin signaling pathways, TF3 has been proposed
to have potential as a natural antidiabetic agent (19).
Chronic hyperglycemia is associated with impaired
angiogenesis, persistent inflammation and defects in collagen
synthesis, resulting in delayed wound healing and an increased risk
of diabetic foot ulcers (116).
Notably, TF3, particularly when formulated as a nanoparticles
hydrogels (TFDG NPS@hydrogels),
has been reported to accelerate wound healing in diabetic models by
suppressing chronic inflammation, enhancing extracellular matrix
remodeling, promoting angiogenesis and activating the TGF-β1/SMAD3
signaling pathway (20).
In diabetic cardiomyopathy, hyperglycemic conditions
are associated with downregulation of connexin 43 (Cx43) expression
and impairment of cardiomyocyte autophagy, thereby increasing
susceptibility to arrhythmias (117). TF3 has been reported to mitigate
these pathological alterations by activating AMP-activated protein
kinase (AMPK) signaling, restoring Cx43 expression and improving
autophagic flux (118,119). Additionally, TF3 alleviates
diabetes-associated liver and kidney dysfunction by modulating
oxidative stress and regulating the circ-ITCH (circular RNA itchy
E3 ubiquitin protein ligase)/Nrf2 pathway, and enhances the
therapeutic effects of metformin, thereby supporting its potential
as an adjunctive strategy for diabetic complications (120).
Protection against osteoporosis and
arthritis
Osteoporosis is characterized by excessive bone
resorption and impaired bone formation (121). Evidence indicates that TF3
inhibits osteoclast differentiation, polarization and
bone-resorptive activity by suppressing receptor activator of NF-κΒ
ligand-induced ERK signaling (17). Furthermore, TF3 inhibits the
migration and differentiation of osteoclast precursor cells by
reducing the expression and activity of matrix metalloproteinases
(MMP-2 and MMP-9) (122).
In an inflammatory bone loss model, TF3 has been
reported to alleviate titanium particle-induced bone resorption and
to maintain bone integrity (17).
Under conditions of estrogen deficiency and inflammation, TF3 has
been shown to enhance osteoblast function, increase bone mass and
suppress inflammatory responses by activating osteogenic signaling
pathways such as the MAPK, Wnt/β-catenin and bone morphogenetic
protein/Smad signaling pathways (61).
In addition to osteoporosis, TF3 modulates immune
responses in arthritis by promoting M2 macrophage polarization and
inhibiting M1 polarization, while also regulating autophagy.
Collectively, these actions reduce inflammation and ameliorate
joint damage, suggesting that TF3 may represent a candidate for the
management of chronic inflammatory bone diseases such as rheumatoid
arthritis (18).
Hepatoprotective effects in fatty
liver diseases
Non-alcoholic fatty liver disease is driven by
dysregulated lipid metabolism, oxidative stress, inflammation and
disruption of the gut-liver axis, which collectively promote
progression toward fibrosis and cirrhosis (123). TF3 has been reported to attenuate
hepatic lipid accumulation and liver injury by regulating the fatty
acid desaturase 1/peroxisome proliferator-activated receptor
δ/fatty acid binding protein 4 axis and modulating gut microbiota
composition, thereby restoring lipid metabolic homeostasis in
non-alcoholic fatty liver disease models (15).
Similarly, alcoholic liver disease is associated
with acetaldehyde toxicity, oxidative stress, inflammation,
activation of hepatic stellate cells and lipid metabolic
disturbances (124). TF3 has been
reported to exert hepatoprotective effects by coordinately engaging
antioxidant, anti-inflammatory, anti-fibrotic mechanisms, as well
as modulating the gut-liver axis through suppression of hepatic
TLR4/NF-κB signaling, thereby alleviating liver injury and limiting
fibrotic progression (53). Taken
together, these findings support TF3 as a candidate natural agent
for the prevention and management of fatty liver diseases.
Other biological functions of TF3
In addition to metabolic and bone-related disorders,
TF3 has been reported to exhibit a range of other biological
activities. TF3 demonstrates anti-allergic effects by inhibiting
the expression of pro-inflammatory cytokines (such as IL-12, IFN-γ
and TNF-α) and preserving systemic antioxidant capacity in allergy
models (125). TF3 also exerts
anti-obesity effects by inhibiting fatty acid synthesis, enhancing
fatty acid oxidation and regulating lipid metabolism-related
signaling pathways (such as the AMPK, sterol regulatory
element-binding protein-1c, acetyl-CoA carboxylase and carnitine
palmitoyltransferase 1 signaling pathways) (58,126–128).
TF3 also exhibits protective effects against
atherosclerosis by improving lipid profiles, reducing inflammation
and oxidative stress, and reprogramming metabolic homeostasis
associated with plaque stabilization (21). In neurodegenerative disease models,
TF3 improves learning and memory deficits through antioxidant
mechanisms, by enhancing cholinergic neurotransmission (including
increased acetylcholine levels and acetylcholinesterase inhibition)
and regulating glutamatergic signaling, as well as activation of
the Nrf2 pathway, thereby supporting a potential role in delaying
brain aging and cognitive decline (22). A summary of these biological
functions is shown in Table I.
 | Table I.Possible biofunctions of
theaflavin-3,3′-digallate in cell and animal models. |
Table I.
Possible biofunctions of
theaflavin-3,3′-digallate in cell and animal models.
| A, Antioxidant |
|---|
|
|---|
| First author/s,
year | Study type | Experimental models
and techniques | Isolation methods
of TFs | Target or signaling
pathway | (Refs.) |
|---|
| Schuck et
al, 2008 | In
vitro | HSC-2 cells and
GN46 fibroblasts under H2O2-induced oxidative
damage | Solvent
extraction | GSH↓, oxidative
stress↑ and activation of caspase-3 to cleave PARP | (56) |
| Lin et al,
2000 | In
vitro | HL-60 cells;
oxidative stress assay; intracellular ROS detection using
DCF-DA | Solvent extraction
after decaffeination by chloroform | Xanthine oxidase
↓ | (59) |
|
| B,
Anti-inflammatory |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Hosokawa et
al, 2010 | In
vitro | HGFs and tissue
samples from patients with inflamed gingiva | Purchased from
Nagara Science Co., Ltd | TNFSF14-induced
ERK, JNK and NF-κB activation ↓ | (71) |
| Ukil et al,
2006; Pan et al, 2000 | In vivo | Mice with
TNBS-induced colitis | Solvent
extraction | Reduced levels of
TNF-α, IL-12, IFN-γ and iNOS; decreased nuclear translocation of
NF-κB and cytosolic IKK activity, with preservation of IκBα | (10,70) |
| Wu et al,
2017 | In vitro/in
vivo | U937 human leukemia
cells, LO-2 hepatocytes, and RAW 264.7 macrophages; mouse model of
acute lung injury | Purchased from
Chinese Academy of Sciences | p-c-JNK and p-p38
MAPK ↓, and TNF-α, IL-1β and IL-6 ↓ | (69) |
|
| C,
Antibacterial |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Hui et al,
2016 | In
vitro | HPLC and docking
experiment | Purchased from | DXR ↓ | (12) |
|
|
|
| Chengdu
Biopurify |
|
|
|
|
|
| Phytochemicals,
Ltd. |
|
|
| Teng et al,
2019 | In
vitro | Nitrocefin assay;
checkerboard and time-kill assays to evaluate antibacterial
synergy; molecular docking analysis | Purchased from
Dalian Meilun Biology Technology Co., Ltd | Metallo-β-lactamase
activity ↓ | (76) |
| Lombardo Bedran, et
al, 2015 | In vitro
cell line and checkerboard technique | OBA-9 human oral
epithelial Niagen Bioscience | Purchased from | IL-8 ↓ and hBD
secretion ↑ | (78) |
| Wang et al,
2019 | In
vitro | Streptococcus
mutans UA159 and biofilm formation and biofilm dispersion
assays | Purchased from
Chengdu Biopurify, Ltd. | gtfB, gtfC and gtfD
encoding glucosyltransferases ↓, and holing-like proteins lrgA,
lrgB and srtA ↓ | (81) |
|
| D,
Antiviral |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Cui et al,
2020 | In vitro
(Zika virus) | Fluorescence-based
screening assay and molecular docking studies | Obtained from the
TargetMol Chemicals Inc. | NS2B-NS3 | (11) |
| Chen et al,
2005 | In vitro
(SARS-CoV) | HPLC proteolytic
assay; fluorogenic substrate peptide assay | Gifted from Dr
Yu-Chih Liang, School of Medical Technology, Taipei Medical
University | 3C-like protease
↓ | (16) |
| Singh et al,
2021 | In vitro
(SARS-CoV) | Molecular docking
studies, molecular dynamics simulations and ADMET studies | Not described | RNA-dependent RNA
polymerase | (90) |
| Liu et al,
2005 | In vitro
(HIV-1) | MT-2 cells, CEM
cells and normal diploid human lung fibroblast cells (MRC-5);
computer-aided molecular docking | Obtained from
MicroSource Discovery Systems, Inc. | Gp41 | (91) |
| Isaacs and Xu,
2013 | In vitro
(HSV) | Vero and CV-1
cells | Not described | Inactivation of
HSV | (92) |
|
| E,
Anticancer |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Gao et al,
2016 | In vitro
(ovarian cancer) | HUVEC tube
formation assay, chick chorioallantoic membrane assay and
ELISA | Purified by
semi-preparative HPLC | Inhibiting Notch-1
cleavage, c-Myc, HIF-1α and VEGF ↓, and Akt/mTOR/p70S6K/4E-BP1
↓ | (13) |
| Pan et al,
2018 | In vitro
(ovarian cancer) | Cell viability
assay, tumor sphere formation assay, ALDH assay and ALDH-based
sorting | Purified by
semi-preparative HPLC | Caspase-3 and −7 ↑,
and Wnt/β-catenin ↓ | (98) |
| Gao et al,
2013 | In vitro
(lung and esophageal cancer) | SPC-A-1 human lung
adenocarcinoma cells and Eca-109 human esophageal carcinoma
cells | Purified by
semi-preparative HPLC | Caspases-3/9 ↑ and
MAPK ↑ | (101) |
| Gao et al,
2019 | In vitro
(ovarian cancer) | Hoechst 33342
staining assay and caspase-glo assay | Purified by
semi-preparative HPLC | Phosphorylation of
Chk2 ↑ and p27 ↑ | (102) |
| Pan et al,
2018 | In vitro
(ovarian cancer) | Determined by
inductively coupled plasma mass spectrometry and GSH assay | Purified by
semi-preparative HPLC | GSH ↓ and CTR1
↑ | (104) |
| Lahiry et
al, 2010 | In vitro
(breast cancer) | Flow cytometry and
co-immunoprecipitation | Purchased from
MilliporeSigma | Fas death
receptor/caspase-8 ↑ and p-Akt/p-Bad ↓ | (106) |
| Adhikary et
al, 2010 | In vitro
(breast cancer) | Assessment of ROS
and coimmunoprecipitation | Purchased from
MilliporeSigma | p53/ROS/p38MAPK ↑,
and MMP-2 and MMP-9 ↓ | (107) |
| Lee et al,
2004 | In vitro
(prostate cancer) | 5a-reductase assay
and ELISA | Not described | Androgen receptor ↓
and fatty acid synthase ↓ | (108) |
| Shi et al,
2021 | In vitro and
in vivo (gastric cancer) | Molecular docking
and SPR technology | Purchased from
Shandong Topscience Biotech Co., Ltd. | SHCBP1 ↓ | (112) |
|
| F, Anti-high
glucose |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Shen et al,
2019 | In
vitro | Tandem
mCherry-GFP-LC3 fluorescence detection and dye transfer assay | Purchased from
Shanghai Aladdin Biochemical Technology Co., Ltd. | Cx43 ↑ and AMPK
↑ | (119) |
|
| G,
Anti-osteoporosis |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Hu et al,
2017 | In
vitro/in vivo | Micro-computed
tomography scanning and resorption pit assay | Purchased from
MilliporeSigma | Bone destruction ↓
and ERK ↓ | (17) |
| Oka et al,
2012 | In
vitro/in vivo | Osteoclast
formation assay and gelatin zymography | Purchased from Wako
Pure Chemical Industries, Ltd. | MMP-2 and MMP-9
↓ | (122) |
|
| H,
Antiallergic |
|
| First author/s,
year | Study
type | Experimental
models | Isolation
methods of TFs | Target or
signaling pathway | (Refs.) |
|
| Yoshino et
al, 2010 | In
vitro/in vivo | Mouse type IV
allergic model; determination of antioxidant activities | Supplied by
Unilever Japan KK | Antioxidant
activity ↑, and IL-12, IFN-γ and TNF-α ↓ | (125) |
Delivery, dosage and safety research
progress of TF3
Application of delivery systems in
enhancing TF3 bioavailability
Despite TF3 having been reported to exhibit
pharmacological activities, including antioxidant,
anti-inflammatory and anticancer effects, its low bioavailability
and limited stability in practical settings restrict broader
application in functional foods and dietary supplements (129,130). Accordingly, several advanced
delivery systems have been developed to improve the physicochemical
properties and pharmacokinetic behavior of TF3, thereby enhancing
its biological performance and expanding its potential utility in
food and medical contexts.
Common delivery systems include nanoemulsions,
liposomes, protein complex co-precipitates and polysaccharide-based
microgels (131–133). Among these, nanoemulsions, due to
their small droplet size, favorable dispersibility and thermal
stability, have been reported to improve TF3 solubility and
stability in aqueous media. By forming a protective interfacial
layer that limits oxidation and enzymatic degradation,
nanoemulsions can prolong the gastrointestinal residence time of
TF3 and thereby improve oral bioavailability (134). Liposomes, with a membrane-mimetic
architecture, can protect TF3 from acidic conditions and enzymatic
degradation in the gastrointestinal tract, facilitate
trans-epithelial transport, and enhance stability and systemic
exposure. In addition, liposomes can modulate TF3 release kinetics,
enabling sustained-release or targeted-release profiles. Protein
complex co-precipitation systems generate stable complexes by
associating TF3 with casein or whey protein via electrostatic,
hydrophobic and hydrogen-bond interactions. This approach not only
improves TF3 dispersibility in liquid foods but also leverages
gastric proteolysis to delay release, thereby reducing premature
degradation and facilitating absorption in the small intestine.
Polysaccharide-based microgels (such as chitosan, sodium alginate
and pectin microgels) exhibit pH responsiveness, biodegradability
and mucoadhesive properties, enabling controlled TF3 release under
varying gastrointestinal conditions and potentially enhancing
tissue targeting (132).
Furthermore, alginate can form gel shells through
Ca2+-mediated crosslinking to encapsulate TF3 for
colon-targeted release, whereas chitosan can increase adhesion to
the intestinal mucosa via electrostatic interactions, thereby
improving transepithelial transport efficiency (135).
Through the design of these delivery systems, TF3
degradation in the gastrointestinal environment can be attenuated,
its solubility and absorption enhanced, and its release profile and
targeting behavior optimized. Notably, TF3-loaded nanoparticles and
hydrogel formulations have been reported to improve therapeutic
outcomes in preclinical models, thereby supporting the feasibility
of delivery-driven enhancement (134,135). Furthermore, nanocarrier systems
enable TF3 to be delivered in a controlled manner to specific
tissues, such as tumors, inflammatory sites or metabolically active
organs (136,137). Functionalized nanoparticles can
further improve cellular uptake and reduce off-target effects,
thereby creating opportunities for the translational development of
TF3 in cancer, inflammation and metabolic diseases (29).
In summary, well-designed delivery systems not only
improve the stability and bioavailability of TF3 in the
gastrointestinal tract but also enhance therapeutic performance
through targeted delivery. Future research should focus on the
safety and biocompatibility of carrier materials, rigorous
validation of delivery efficiency and industrial scalability to
facilitate the translation of TF3 into nutritional medicine and
personalized nutrition.
Safety evaluation and human dosage
conversion of TF3
In advancing TF3 as an active ingredient in
functional foods or dietary supplements, systematic evaluation of
its safety profile and feasible human intake levels is essential.
Existing evidence suggests that TF3, as a constituent derived from
traditional tea, is supported by a favorable safety background
(138). The US FDA has classified
tea and its major polyphenolic constituents as ‘GRAS’, providing
preliminary assurance for food-related applications of TF3
(139). However, systematic
toxicological studies specifically focused on TF3 remain limited,
particularly regarding chronic toxicity and target organ safety
under long-term, high-dose exposure.
An in vivo study reported no overt adverse
effects within commonly used experimental dose ranges. In a single
preclinical study comprising several animal experiments, TF3 was
administered orally at ~ 20 and 40 mg/kg body weight for several
weeks in C57BL/6 mice (6–8 weeks old, 20–25 g), no notable changes
in body weight, abnormal behavior or pathological damage to major
organs were observed (69).
Although TF3 appears to be well tolerated in short-term animal
studies, most available investigations have primarily focused on
pharmacodynamic outcomes, and comprehensive toxicological
evaluations remain limited.
Converting efficacious doses from animal
experiments to feasible human intake levels is a key step in
assessing the nutritional applicability of TF3. Using the commonly
applied body surface area-based conversion approach, a 5 mg/kg dose
in mice corresponds to ~0.4 mg/kg in humans; thus, an adult
weighing 60 kg would require an estimated intake of ~24 mg TF3 per
day to achieve comparable exposure (140,141). By contrast, TF3 typically
accounts for 0.20–0.54% (1.99 to 5.39 mg/g) of the dry weight of
black tea, and a single serving (~2 g tea leaves) provides <11
mg TF3 (134). Therefore, routine
tea consumption alone is unlikely to reach intake levels associated
with bioactivity in animal models. This gap suggests that TF3 may
be more suitably delivered via functional food fortification,
nutritional supplements or optimized delivery systems rather than
relying exclusively on conventional tea consumption. In this
context, incorporation of the aforementioned delivery strategies
may improve TF3 stability and bioavailability, potentially enabling
biological effects at lower practical intake levels and thereby
enhancing feasibility and safety for population-level use.
In conclusion, although existing studies support an
acceptable short-term safety profile for TF3, the long-term safety
threshold and generalizability to broader populations require
further systematic investigation. Future studies should include
multi-dose, extended-duration and multi-species in vivo
toxicology assessments, complemented by human intervention studies,
to define safe intake ranges and functional dose intervals for TF3.
This evidence will be essential for standardized application in
functional foods and nutritional medicine.
Metabolic mechanisms and clinical
translation of TF3
Efficacy comparison and mechanistic
differences between TF3 and its metabolites
A key issue in the clinical translation of TF3 is
whether its biological effects are primarily mediated by the intact
parent compound or by degradation products and gut
microbiota-derived metabolites, such as GA and PG. This question is
particularly relevant given the chemical instability of TF3 and
limited systemic exposure following oral administration.
A pharmacokinetic study indicated that TF3 exhibits
low oral absorption and undergoes extensive gastrointestinal
biotransformation, resulting in markedly low concentrations in
plasma and peripheral tissues (43). By contrast, GA and PG are
low-molecular-weight phenolic compounds that are more readily
absorbed and can achieve higher systemic concentrations (142,143). Accordingly, GA and PG may
constitute principal bioactive species downstream of TF3, notably
contributing to its in vivo effects.
Mechanistically, TF3 and its metabolites display
both overlapping and distinct modes of action. TF3 has been
described primarily as a pleiotropic modulator of oxidative stress,
inflammatory signaling pathways (such as the NF-κB, MAPK and Nrf2
signaling pathways) and receptor tyrosine kinases (63,70).
By contrast, GA and PG have been reported to exert more consistent
cytotoxic and pro-oxidant effects in tumor cells, inducing
apoptosis through mitochondrial dysfunction, caspase activation and
suppression of NF-κB signaling (144,145).
Despite these observations, direct head-to-head
comparisons of TF3 and its metabolites under standardized
experimental conditions remain scarce. The absence of such
comparative analyses constitutes a major knowledge gap and
complicates interpretation of the relative contributions of TF3 and
its metabolites to in vivo efficacy.
Potential for combination therapy and
clinical translation pathways of TF3
Given the multitarget regulatory properties of TF3,
it appears well suited for combination-based therapeutic
strategies. Preclinical studies have reported that TF3 enhances the
efficacy of chemotherapeutic agents (such as cisplatin) by
promoting intracellular drug accumulation and reducing GSH-mediated
drug efflux, thereby increasing tumor cell susceptibility to
apoptosis (103,104). In the antimicrobial field, TF3
has been reported to restore β-lactam antibiotic activity by
inhibiting MBLs, supporting its potential role as an adjunct to
antibiotic therapy (76).
Collectively, these findings suggest that TF3 may be more effective
as a synergistic modulator than as a standalone intervention.
Future translational research should prioritize
standardized comparisons of TF3 and its metabolites at
physiologically relevant doses. Key steps include
formulation-dependent pharmacokinetic characterization, systematic
safety evaluation and disease-specific efficacy validation. Given
the long history of TF3 dietary exposure, it may be particularly
amenable to development as a nutraceutical, functional food
ingredient or adjunct therapeutic agent. Accordingly, a
metabolism-informed translational roadmap that integrates delivery
strategies with combination regimens may facilitate the progression
of TF3 from experimental studies to clinical application.
Conclusion and future prospects
TF3 has been proposed to serve a role in the
prevention and management of human diseases, particularly in
cancer-related contexts. As a major theaflavin component in black
tea, TF3 exhibits a broad spectrum of reported bioactivities,
including antioxidant, anti-inflammatory, antiviral, anticancer and
anti-allergic effects. With respect to antitumor activity, TF3 has
been reported to display comparatively low cytotoxicity toward
normal cells while inhibiting tumor cells, supporting its potential
translational relevance. In addition, TF3 may act synergistically
with conventional chemotherapeutic agents, such as cisplatin and
trastuzumab, thereby enhancing therapeutic responsiveness.
However, despite numerous in vitro and in
vivo studies describing multiple biological functions of TF3,
translational development continues to face several challenges.
First, most available evidence is derived from cellular and animal
models, whereas systematic clinical validation remains limited,
precluding robust assessment of efficacy and long-term safety in
human populations. Second, the experimental doses used in numerous
studies exceed those achievable through routine tea consumption,
and feasible intake levels, safety thresholds and human metabolic
characteristics remain insufficiently defined, thereby constraining
the practical development of TF3 as a functional food ingredient or
dietary supplement. Notably, some studies rely on specific disease
models or target overexpression systems, and stable therapeutic
effects under complex pathological conditions have not been
confirmed, limiting generalizability. Furthermore, as a natural
product, TF3 is chemically labile, readily degradable and
challenging to obtain at high purity, posing challenges for product
standardization, scalability and industrial translation. Although
delivery strategies (such as nanoemulsions, liposomes and
microcapsules) have been explored to improve stability and
bioavailability, most remain at an early stage and lack clinical
validation and large-scale implementation.
In light of these considerations, future research
should prioritize the following areas: i) Detailed characterization
of TF3 metabolism and degradation mechanisms, including stability
testing across pHs, solvents and temperatures, together with
delineation of its metabolic fate in the human gastrointestinal
tract; ii) clarification of pharmacological differences between TF3
and its major metabolites across disease models, with
identification of core active species and key molecular targets to
inform rational delivery optimization; iii) systematic toxicology
and pharmacokinetic investigations using multi-dose,
extended-duration and multi-species designs, complemented by human
intervention trials to define safe intake ranges and functional
dose intervals; and iv) development of standardized, scalable and
cost-efficient TF3 formulations to improve accessibility and
sustainability in functional foods and clinical nutrition.
In conclusion, TF3 is a natural bioactive molecule
with multitarget regulatory potential and an apparently favorable
short-term tolerability profile, and it may have value for
therapeutic development. Future efforts should emphasize
mechanistic elucidation, formulation optimization and
evidence-based clinical research to support translation from
experimental studies to real-world disease intervention, thereby
contributing to the development of natural product-derived
therapeutics and precision nutrition strategies.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Key Research and
Development Program of China (grant no. 2022YFA1305000 to J.G.),
the Fundamental Research Funds for the Central Universities (grant
no. lzujbky-2023-ey11 to J.G.), the National Natural Science
Foundation of China (grant no. 32200426 to J.G.), the Major Science
and Technology Projects of Gansu Province (grant no. 25ZDFA003 to
J.G.), the Talents Program of the Lanzhou University Second
Hospital (grant no. yjrckyqdj-2022-02 to J.G.).
Availability of data and materials
Not applicable.
Authors' contributions
TM and SZ contributed to the conception and design
of the review and drafted the main text of the manuscript. SZ and
MX were involved in data collection, literature analysis and
interpretation of the data, and prepared the figures and table. JG
and ZM made substantial contributions to the conceptual framework
of the review, critically revised the manuscript for important
intellectual content and provided expert input on data
interpretation. All authors participated in manuscript revision,
read and approved the final version to be published and agreed to
be accountable for all aspects of the work.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Newman DJ and Cragg GM: Natural products
as sources of new drugs from 1981 to 2014. J Nat Prod. 79:629–661.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Varghese R and Dalvi YB: Natural products
as anticancer agents. Curr Drug Targets. 22:1272–1287. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen ZM and Lin Z: Tea and human health:
Biomedical functions of tea active components and current issues. J
Zhejiang Univ Sci B. 16:87–102. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Burdock GA and Carabin IG: Generally
recognized as safe (GRAS): History and description. Toxicol Lett.
150:3–18. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Abudureheman B, Yu X, Fang D and Zhang H:
Enzymatic oxidation of tea catechins and its mechanism. Molecules.
27:9422022. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
He HF: Research progress on theaflavins:
Efficacy, formation, and preparation. Food Nutr Res.
61:13445212017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Murakami S, Takahashi S, Muguruma H,
Osakabe N, Inoue H and Ohsawa T: Polyphenol analysis in black tea
with a carbon nanotube electrode. Anal Sci. 35:529–534. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Wu YY, Li W, Xu Y, Jin EH and Tu YY:
Evaluation of the antioxidant effects of four main theaflavin
derivatives through chemiluminescence and DNA damage analyses. J
Zhejiang Univ Sci B. 12:744–751. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sang S, Lambert JD, Ho CT and Yang CS: The
chemistry and biotransformation of tea constituents. Pharmacol Res.
64:87–99. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ukil A, Maity S and Das PK: Protection
from experimental colitis by theaflavin-3,3′-digallate correlates
with inhibition of IKK and NF-kappaB activation. Br J Pharmacol.
149:121–131. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Cui X, Zhou R, Huang C, Zhang R, Wang J,
Zhang Y, Ding J, Li X, Zhou J and Cen S: Identification of
theaflavin-3,3′-digallate as a novel zika virus protease inhibitor.
Front Pharmacol. 11:5143132020. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Hui X, Yue Q, Zhang DD, Li H, Yang SQ and
Gao WY: Antimicrobial mechanism of theaflavins: They target
1-deoxy-D-xylulose 5-phosphate reductoisomerase, the key enzyme of
the MEP terpenoid biosynthetic pathway. Sci Rep. 6:389452016.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Gao Y, Rankin GO, Tu Y and Chen YC:
Theaflavin-3, 3′-digallate decreases human ovarian carcinoma
OVCAR-3 cell-induced angiogenesis via Akt and Notch-1 pathways, not
via MAPK pathways. Int J Oncol. 48:281–292. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Law YY, Hsieh YH, Hsieh YS, Lee YH, Tang
CL, Lee CY, Yang SF, Chu SC and Chen PN: Theaflavin-3,3′-digallate
triggers apoptosis in osteosarcoma cells via the caspase pathway. J
Cancer. 16:2567–2577. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Zhou C, Zhang W, Lin H, Zhang L, Wu F,
Wang Y, Yu S, Peng X, Cheng W, Li M, et al: Effect of
theaflavin-3,3′-digallate on leptin-deficient induced nonalcoholic
fatty liver disease might be related to lipid metabolism regulated
by the Fads1/PPARδ/Fabp4 axis and gut microbiota. Front Pharmacol.
13:9252642022. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chen CN, Lin CP, Huang KK, Chen WC, Hsieh
HP and Liang PH: Hsujt inhibition of sars-cov 3c-like protease
activity by theaflavin-3,3′-digallate (TF3). Evid Based Complement
Alternat Med. 2:209–215. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hu X, Ping Z, Gan M, Tao Y, Wang L, Shi J,
Wu X, Zhang W, Yang H, Xu Y, et al: Theaflavin-3,3′-digallate
represses osteoclastogenesis and prevents wear debris-induced
osteolysis via suppression of ERK pathway. Acta Biomater.
48:479–488. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang L, Li W, Hou Z, Wang Z, Zhang W,
Liang X, Wu Z, Wang T, Liu X, Peng X, et al:
Theaflavin-3,3′-digallate ameliorates collagen-induced arthritis
through regulation of autophagy and macrophage polarization. J
Inflamm Res. 16:109–126. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhou H, Wu Y, Kim E, Pan H, He P, Li B,
Chen YC and Tu Y: Simultaneous tests of theaflavin-3,3′-digallate
as an anti-diabetic drug in human hepatoma G2 cells and Zebrafish
(Danio rerio). Nutrients. 13:43792021. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Dong X, Miao J, Wu L, Kong Z, Liu Z, Jia
D, Zhai Q, Zhang D and Xu Y: Diabetic wound healing breakthrough:
theaflavin-3, 3′-digallate simplenanoparticles@hydrogel
activates the TGF-β1/SMAD3 pathway. Phytomedicine. 141:1566172025.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Cen K, Huang Y, Xie Y, Zhang R, Cai Q, Zou
L, Xiang Q, Yang C and Liu Y: Theaflavin-3,3′-digallate stabilizes
vulnerable plaques by reprogramming metabolic homeostasis in
neovascularization via HK2/TIGAR. Phytomedicine. 145:1570142025.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cao Y, Zhang Y, Jia Z, Jia H, Sun Y, Yuan
H, Bian Y, Xu B, Fu J and Qin F: Theaflavin-3,3′-digallate
ameliorates learning and memory impairments in mice with premature
brain aging induced by D-galactose. Physiol Behav. 261:1140772023.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pan SY, Nie Q, Tai HC, Song XL, Tong YF,
Zhang LJ, Wu XW, Lin ZH, Zhang YY, Ye DY, et al: Tea and tea
drinking: China's outstanding contributions to the mankind. Chin
Med. 17:272022. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cabrera C, Artacho R and Gimenez R:
Beneficial effects of green tea-a review. J Am Coll Nutr. 25:79–99.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lin JK: Cancer chemoprevention by tea
polyphenols through modulating signal transduction pathways. Arch
Pharm Res. 25:561–571. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tanaka T and Matsuo Y: Production
mechanisms of black tea polyphenols. Chem Pharm Bull (Tokyo).
68:1131–1142. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Takemoto M and Takemoto H: Synthesis of
theaflavins and their functions. Molecules. 23:9182018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Leung LK, Su Y, Chen R, Zhang Z, Huang Y
and Chen ZY: Theaflavins in black tea and catechins in green tea
are equally effective antioxidants. J Nutr. 131:2248–2251. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Lei S, Xie M, Hu B, Zhou L, Sun Y,
Saeeduddin M, Zhang H and Zeng X: Effective synthesis of
theaflavin-3,3′-digallate with epigallocatechin-3-O-gallate and
epicatechin gallate as substrates by using immobilized pear
polyphenol oxidase. Int J Biol Macromol. 94((Pt A)): 709–718. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xiao J and Högger P: Stability of dietary
polyphenols under the cell culture conditions: Avoiding erroneous
conclusions. J Agric Food Chem. 63:1547–1557. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Jhoo JW, Lo CY, Li SM, Sang SM, Ang CYW,
Heinze TM and Ho CT: Stability of black tea polyphenol, theaflavin,
and identification of theanaphthoquinone as its major radical
reaction product. J Agric Food Chem. 53:6146–6150. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Krupkova O, Ferguson SJ and Wuertz-Kozak
K: Stability of (−)-epigallocatechin gallate and its activity in
liquid formulations and delivery systems. J Nutr Biochem. 37:1–12.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Shingai Y, Fujimoto A, Nakamura M and
Masuda T: Structure and function of the oxidation products of
polyphenols and identification of potent lipoxygenase inhibitors
from Fe-catalyzed oxidation of resveratrol. J Agric Food Chem.
59:8180–8186. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Wang M, Xu J, Han T and Tang L: Effects of
theaflavins on the structure and function of bovine lactoferrin.
Food Chem. 338:1280482021. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Lipinski CA, Lombardo F, Dominy BW and
Feeney PJ: Experimental and computational approaches to estimate
solubility and permeability in drug discovery and development
settings. Adv Drug Deliv Rev. 46:3–26. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Henning SM, Aronson W, Niu Y, Conde F, Lee
NH, Seeram NP, Lee RP, Lu J, Harris DM, Moro A, et al: Tea
polyphenols and theaflavins are present in prostate tissue of
humans and mice after green and black tea consumption. J Nutr.
136:1839–1843. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Mulder TP, van Platerink CJ, Wijnand
Schuyl PJ and van Amelsvoort JM: Analysis of theaflavins in
biological fluids using liquid chromatography-electrospray mass
spectrometry. J Chromatogr B Biomed Sci Appl. 760:271–279. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fan Y, Lei Z, Huang J, Su D, Ni D and Chen
Y: Effect and mechanism of theaflavins on fluoride transport and
absorption in caco-2 cells. Foods. 12:14872023. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Selma MV, Espín JC and Tomás-Barberán FA:
Interaction between phenolics and gut microbiota: Role in human
health. J Agric Food Chem. 57:6485–6501. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Chen H, Parks TA, Chen X, Gillitt ND,
Jobin C and Sang S: Structural identification of mouse fecal
metabolites of theaflavin 3,3′-digallate using liquid
chromatography tandem mass spectrometry. J Chromatogr A.
1218:7297–7306. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Chen H, Hayek S, Rivera Guzman J, Gillitt
ND, Ibrahim SA, Jobin C and Sang S: The microbiota is essential for
the generation of black tea theaflavins-derived metabolites. PLoS
One. 7:e510012012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Sun L, Su Y, Hu K, Li D, Guo H and Xie Z:
Microbial-transferred metabolites of black tea theaflavins by human
gut microbiota and their impact on antioxidant capacity. Molecules.
28:58712023. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu Z, de Bruijn WJC, Bruins ME and
Vincken JP: Microbial metabolism of theaflavin-3,3′-digallate and
its gut microbiota composition modulatory effects. J Agric Food
Chem. 69:232–245. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Choi KC, Lee YH, Jung MG, Kwon SH, Kim MJ,
Jun WJ, Lee J, Lee JM and Yoon HG: Gallic acid suppresses
lipopolysaccharide-induced nuclear factor-kappaB signaling by
preventing RelA acetylation in A549 lung cancer cells. Mol Cancer
Res. 7:2011–2021. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Lo C, Lai TY, Yang JH, Yang JS, Ma YS,
Weng SW, Chen YY, Lin JG and Chung JG: Gallic acid induces
apoptosis in A375.S2 human melanoma cells through caspase-dependent
and -independent pathways. Int J Oncol. 37:377–385. 2010.PubMed/NCBI
|
|
46
|
Giftson Senapathy J, Jayanthi S,
Viswanathan P, Umadevi P and Nalini N: Effect of gallic acid on
xenobiotic metabolizing enzymes in 1,2-dimethyl hydrazine induced
colon carcinogenesis in Wistar rats-a chemopreventive approach.
Food Chem Toxicol. 49:887–892. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Han YH, Kim SZ, Kim SH and Park WH:
Pyrogallol inhibits the growth of lung cancer Calu-6 cells via
caspase-dependent apoptosis. Chem Biol Interact. 177:107–114. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kawada M, Ohno Y, Ri Y, Ikoma T, Yuugetu
H, Asai T, Watanabe M, Yasuda N, Akao S, Takemura G, et al:
Anti-tumor effect of gallic acid on LL-2 lung cancer cells
transplanted in mice. Anticancer Drugs. 12:847–852. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Ding Y, Chen B, Gao Z, Suo H and Xiao H:
Pre-treated theaflavin-3,3′-digallate has a higher inhibitory
effect on the HCT116 cell line. Food Nutr Res. 61:14003402017.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Xu C, Ni S, Xu N, Yin G, Yu Y, Zhou B,
Zhao G, Wang L, Zhu R, Jiang S and Wang Y:
Theaflavin-3,3′-digallate inhibits erastin-induced chondrocytes
ferroptosis via the Nrf2/GPX4 signaling pathway in osteoarthritis.
Oxid Med Cell Longev. 2022:35319952022. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Lin Y, Shen C, Zhao J, Wang C, Obara M,
Maung AT, Morita M, Abdelaziz MNS, Masuda Y, Honjoh KI and Miyamoto
T: Antibacterial effect and mechanism of theaflavin against
Listeria monocytogenes and its application on apple skins. J Food
Sci. 89:6653–6663. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Mhatre S, Srivastava T, Naik S and
Patravale V: Antiviral activity of green tea and black tea
polyphenols in prophylaxis and treatment of COVID-19: A review.
Phytomedicine. 85:1532862021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Tang MG, Xiong LG, Huang JA, Hara Y, Zhang
S, Liu ZH and Liu AL: Theaflavin-3,3′-digallate prevents alcoholic
liver injury by suppressing hepatic TLR4/NF-κB signaling and
modulating the gut-liver axis in mice. J Nutr Biochem.
145:1100312025. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Anand K, Ziebuhr J, Wadhwani P, Mesters JR
and Hilgenfeld R: Coronavirus main proteinase (3CLpro) structure:
Basis for design of anti-SARS drugs. Science. 300:1763–1767. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Liang YC, Chen YC, Lin YL, Lin-Shiau SY,
Ho CT and Lin JK: Suppression of extracellular signals and cell
proliferation by the black tea polyphenol,
theaflavin-3,3′-digallate. Carcinogenesis. 20:733–736. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Schuck AG, Ausubel MB, Zuckerbraun HL and
Babich H: Theaflavin-3,3′-digallate, a component of black tea: An
inducer of oxidative stress and apoptosis. Toxicol In Vitro.
22:598–609. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Yang Z, Jie G, Dong F, Xu Y, Watanabe N
and Tu Y: Radical-scavenging abilities and antioxidant properties
of theaflavins and their gallate esters in H2O2-mediated oxidative
damage system in the HPF-1 cells. Toxicol In Vitro. 22:1250–1256.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Luo XY, Takahara T, Hou J, Kawai K,
Sugiyama T, Tsukada K, Takemoto M, Takeuchi M, Zhong L and Li XK:
Theaflavin attenuates ischemia-reperfusion injury in a mouse fatty
liver model. Biochem Biophys Res Commun. 417:287–293. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Lin JK, Chen PC, Ho CT and Lin-Shiau SY:
Inhibition of xanthine oxidase and suppression of intracellular
reactive oxygen species in HL-60 cells by
theaflavin-3,3′-digallate, (−)-epigallocatechin-3-gallate, and
propyl gallate. J Agric Food Chem. 48:2736–2743. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Zhang J, Cai S, Li J, Xiong L, Tian L, Liu
J, Huang J and Liu Z: Neuroprotective effects of theaflavins
against oxidative stress-induced apoptosis in PC12 Cells. Neurochem
Res. 41:3364–3372. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ge G, Yang S, Hou Z, Gan M, Tao H, Zhang
W, Li W, Wang Z, Hao Y, Gu Y and Geng D: Theaflavin-3,3′-digallate
promotes the formation of osteoblasts under inflammatory
environment and increases the bone mass of ovariectomized mice.
Front Pharmacol. 12:6489692021. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Pan MH, Liang YC, Lin-Shiau SY, Zhu NQ, Ho
CT and Lin JK: Induction of apoptosis by the oolong tea polyphenol
theasinensin A through cytochrome c release and activation of
caspase-9 and caspase-3 in human U937 cells. J Agric Food Chem.
48:6337–6346. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Ai Z, Wu Y, Yu M, Li J and Li S:
Theaflavin-3, 3′-Digallate Suppresses RANKL-induced
osteoclastogenesis and attenuates ovariectomy-induced bone loss in
mice. Front Pharmacol. 11:8032020. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
He T, Lin X, Yang C, Chen Z, Wang L, Li Q,
Ma J, Zhan F, Wang Y, Yan J and Quan Z: Theaflavin-3,3′-digallate
plays a ROS-mediated dual role in ferroptosis and apoptosis via the
MAPK pathway in human osteosarcoma cell lines and xenografts. Oxid
Med Cell Longev. 2022:89663682022. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Mantovani A, Allavena P, Sica A and
Balkwill F: Cancer-related inflammation. Nature. 454:436–444. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Goodman RB, Pugin J, Lee JS and Matthay
MA: Cytokine-mediated inflammation in acute lung injury. Cytokine
Growth Factor Rev. 14:523–535. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Tiwari MM, Brock RW, Megyesi JK, Kaushal
GP and Mayeux PR: Disruption of renal peritubular blood flow in
lipopolysaccharide-induced renal failure: Role of nitric oxide and
caspases. Am J Physiol Renal Physiol. 289:F1324–F1332. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Wen X, Buckley B, McCandlish E, Goedken
MJ, Syed S, Pelis R, Manautou JE and Aleksunes LM: Transgenic
expression of the human MRP2 transporter reduces cisplatin
accumulation and nephrotoxicity in Mrp2-null mice. Am J Pathol.
184:1299–1308. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Wu Y, Jin F and Wang Y, Li F, Wang L, Wang
Q, Ren Z and Wang Y: In vitro and in vivo anti-inflammatory effects
of theaflavin-3,3′-digallate on lipopolysaccharide-induced
inflammation. Eur J Pharmacol. 794:52–60. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Pan MH, Lin-Shiau SY, Ho CT, Lin JH and
Lin JK: Suppression of lipopolysaccharide-induced nuclear
factor-kappaB activity by theaflavin-3,3′-digallate from black tea
and other polyphenols through down-regulation of IkappaB kinase
activity in macrophages. Biochem Pharmacol. 59:357–367. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Hosokawa Y, Hosokawa I, Ozaki K, Nakanishi
T, Nakae H and Matsuo T: Tea polyphenols inhibit IL-6 production in
tumor necrosis factor superfamily 14-stimulated human gingival
fibroblasts. Mol Nutr Food Res. 54 (Suppl 2):S151–S158. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Keylor MH, Matsuura BS and Stephenson CR:
Chemistry and biology of resveratrol-derived natural products. Chem
Rev. 115:8976–9027. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Singh N, Chevé G, Avery MA and McCurdy CR:
Targeting the methyl erythritol phosphate (MEP) pathway for novel
antimalarial, antibacterial and herbicidal drug discovery:
Inhibition of 1-deoxy-D-xylulose-5-phosphate reductoisomerase (DXR)
enzyme. Curr Pharm Des. 13:1161–1177. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Yussof A, Cammalleri B, Fayemiwo O, Lopez
S and Chu T: Antibacterial and sporicidal activity evaluation of
theaflavin-3,3′-digallate. Int J Mol Sci. 23:21532022. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Boyd SE, Livermore DM, Hooper DC and Hope
WW: Metallo-β-Lactamases: Structure, function, epidemiology,
treatment options, and the development pipeline. Antimicrob Agents
Chemother. 64:e003972020. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Teng Z, Guo Y, Liu X, Zhang J, Niu X, Yu
Q, Deng X and Wang J: Theaflavin-3,3´-digallate increases the
antibacterial activity of β-lactam antibiotics by inhibiting
metallo-β-lactamase activity. J Cell Mol Med. 23:6955–6964. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Goc A, Sumera W, Rath M and Niedzwiecki A:
Inhibition of α-hemolysin activity of Staphylococcus aureus by
theaflavin 3,3′-digallate. PLoS One. 18:e02909042023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Lombardo Bedran TB, Morin MP, Palomari
Spolidorio D and Grenier D: Black tea extract and its theaflavin
derivatives inhibit the growth of periodontopathogens and modulate
interleukin-8 and β-defensin secretion in oral epithelial cells.
PLoS One. 10:e01431582015. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Kong J, Xia K, Su X, Zheng X, Diao C, Yang
X, Zuo X, Xu J and Liang X: Mechanistic insights into the
inhibitory effect of theaflavins on virulence factors production in
Streptococcus mutans. AMB Express. 11:1022021. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Nakahara K, Kawabata S, Ono H, Ogura K,
Tanaka T, Ooshima T and Hamada S: Inhibitory effect of oolong tea
polyphenols on glycosyltransferases of mutans Streptococci. Appl
Environ Microbiol. 59:968–973. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
81
|
Wang S, Wang Y, Wang Y, Duan Z, Ling Z, Wu
W, Tong S, Wang H and Deng S: Theaflavin-3,3′-digallate suppresses
biofilm formation, acid production, and acid tolerance in
streptococcus mutans by targeting virulence factors. Front
Microbiol. 10:17052019. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Wang S, Deng S and Wang Y:
Theaflavin-3,3′-digallate effectively attenuates biofilm formation
by Enterococcus faecalis via the targeting of specific quorum
sensing pathways. Microb Pathog. 193:1067392024. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Latif AA and Mukaratirwa S: Zoonotic
origins and animal hosts of coronaviruses causing human disease
pandemics: A review. Onderstepoort J Vet Res. 87:e1–e9. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Gogoi M, Borkotoky M, Borchetia S,
Chowdhury P, Mahanta S and Barooah AK: Black tea bioactives as
inhibitors of multiple targets of SARS-CoV-2 (3CLpro, PLpro and
RdRp): A virtual screening and molecular dynamic simulation study.
J Biomol Struct Dyn. 40:7143–7166. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Peele KA, Potla Durthi C, Srihansa T,
Krupanidhi S, Ayyagari VS, Babu DJ, Indira M, Reddy AR and
Venkateswarulu TC: Molecular docking and dynamic simulations for
antiviral compounds against SARS-CoV-2: A computational study.
Inform Med Unlocked. 19:1003452020. View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Elfiky AA: Novel guanosine derivatives
against Zika virus polymerase in silico. J Med Virol. 92:11–16.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Elfiky AA and Azzam EB: Novel guanosine
derivatives against MERS CoV polymerase: An in silico perspective.
J Biomol Struct Dyn. 39:2923–2931. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Ahmed-Belkacem A, Guichou JF, Brillet R,
Ahnou N, Hernandez E, Pallier C and Pawlotsky JM: Inhibition of RNA
binding to hepatitis C virus RNA-dependent RNA polymerase: A new
mechanism for antiviral intervention. Nucleic Acids Res.
42:9399–9409. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Qin B, Li Z, Tang K, Wang T, Xie Y,
Aumonier S, Wang M, Yuan S and Cui S: Identification of the
SARS-unique domain of SARS-CoV-2 as an antiviral target. Nat
Commun. 14:39992023. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Singh S, Sk MF, Sonawane A, Kar P and
Sadhukhan S: Plant-derived natural polyphenols as potential
antiviral drugs against SARS-CoV-2 via RNA-dependent RNA polymerase
(RdRp) inhibition: An in-silico analysis. J Biomol Struct Dyn.
39:6249–6264. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Liu S, Lu H, Zhao Q, He Y, Niu J, Debnath
AK, Wu S and Jiang S: Theaflavin derivatives in black tea and
catechin derivatives in green tea inhibit HIV-1 entry by targeting
gp41. Biochim Biophys Acta. 1723:270–281. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
92
|
Isaacs CE and Xu W:
Theaflavin-3,3′-digallate and lactic acid combinations reduce
herpes simplex virus infectivity. Antimicrob Agents Chemother.
57:3806–3814. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Barboza JR, Pereira FAN, Vasconcelos CC,
de Sousa Ribeiro MN and Lopes AJO: Molecular mechanisms of action
and chemosensitization of tumor cells in ovarian cancer by
phytochemicals: A narrative review on pre-clinical and clinical
studies. Phytother Res. 37:2484–2512. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Chung FL: The prevention of lung cancer
induced by a tobacco-specific carcinogen in rodents by green and
black tea. Proc Soc Exp Biol Med. 220:244–248. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Wen S, Sun L, An R, Zhang W, Xiang L, Li
Q, Lai X, Huo M, Li D and Sun S: A combination of Citrus reticulata
peel and black tea inhibits migration and invasion of liver cancer
via PI3K/AKT and MMPs signaling pathway. Mol Biol Rep. 47:507–519.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
96
|
Kim TL, Jeong GH, Yang JW, Lee KH,
Kronbichler A, van der Vliet HJ, Grosso G, Galvano F, Aune D, Kim
JY, et al: Tea consumption and risk of cancer: An umbrella review
and meta-analysis of observational studies. Adv Nutr. 11:1437–1452.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
97
|
Mizuno T, Suzuki N, Makino H, Furui T,
Morii E, Aoki H, Kunisada T, Yano M, Kuji S, Hirashima Y, et al:
Cancer stem-like cells of ovarian clear cell carcinoma are enriched
in the ALDH-high population associated with an accelerated
scavenging system in reactive oxygen species. Gynecol Oncol.
137:299–305. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Pan H, Kim E, Rankin GO, Rojanasakul Y, Tu
Y and Chen YC: Theaflavin-3, 3′-digallate inhibits ovarian cancer
stem cells via suppressing Wnt/β-Catenin signaling pathway. J Funct
Foods. 50:1–7. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Gao Y, Rankin GO, Tu Y and Chen YC:
Inhibitory effects of the four main theaflavin derivatives found in
black tea on ovarian cancer cells. Anticancer Res. 36:643–651.
2016.PubMed/NCBI
|
|
100
|
Li W, Wu JX and Tu YY: Synergistic effects
of tea polyphenols and ascorbic acid on human lung adenocarcinoma
SPC-A-1 cells. J Zhejiang Univ Sci B. 11:458–464. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
101
|
Gao Y, Li W, Jia L, Li B, Chen YC and Tu
Y: Enhancement of (−)-epigallocatechin-3-gallate and
theaflavin-3-3′-digallate induced apoptosis by ascorbic acid in
human lung adenocarcinoma SPC-A-1 cells and esophageal carcinoma
Eca-109 cells via MAPK pathways. Biochem Biophys Res Commun.
438:370–374. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Gao Y, Yin J, Tu Y and Chen YC:
Theaflavin-3,3′-digallate suppresses human ovarian carcinoma
OVCAR-3 cells by regulating the checkpoint kinase 2 and p27 kip1
pathways. Molecules. 24:6732019. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Pan H, Li J, Rankin GO, Rojanasakul Y, Tu
Y and Chen YC: Synergistic effect of black tea polyphenol,
theaflavin-3,3′-digallate with cisplatin against cisplatin
resistant human ovarian cancer cells. J Funct Foods. 46:1–11. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Pan H, Kim E, Rankin GO, Rojanasakul Y, Tu
Y and Chen YC: Theaflavin-3,3′-digallate enhances the inhibitory
effect of cisplatin by regulating the copper transporter 1 and
glutathione in human ovarian cancer cells. Int J Mol Sci.
19:1172018. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Hollstein M, Sidransky D, Vogelstein B and
Harris CC: p53 mutations in human cancers. Science. 253:49–53.
1991. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Lahiry L, Saha B, Chakraborty J, Adhikary
A, Mohanty S, Hossain DM, Banerjee S, Das K, Sa G and Das T:
Theaflavins target Fas/caspase-8 and Akt/pBad pathways to induce
apoptosis in p53-mutated human breast cancer cells. Carcinogenesis.
31:259–268. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Adhikary A, Mohanty S, Lahiry L, Hossain
DM, Chakraborty S and Das T: Theaflavins retard human breast cancer
cell migration by inhibiting NF-kappaB via p53-ROS cross-talk. FEBS
Lett. 584:7–14. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Lee HH, Ho CT and Lin JK:
Theaflavin-3,3′-digallate and penta-O-galloyl-beta-D-glucose
inhibit rat liver microsomal 5alpha-reductase activity and the
expression of androgen receptor in LNCaP prostate cancer cells.
Carcinogenesis. 25:1109–1118. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Sun L, Wen S, Li Q, Lai X, Chen R, Zhang
Z, Cao J and Sun S: Theaflavin-3,3′-di-gallate represses prostate
cancer by activating the PKCδ/aSMase signaling pathway through a 67
kDa laminin receptor. Food Funct. 13:4421–4431. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Mizuno H, Cho YY, Zhu F, Ma WY, Bode AM,
Yang CS, Ho CT and Dong Z: Theaflavin-3, 3′-digallate induces
epidermal growth factor receptor downregulation. Mol Carcinog.
45:204–212. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Yeh CW, Chen WJ, Chiang CT, Lin-Shiau SY
and Lin JK: Suppression of fatty acid synthase in MCF-7 breast
cancer cells by tea and tea polyphenols: A possible mechanism for
their hypolipidemic effects. Pharmacogenomics J. 3:267–276. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Shi W, Zhang G, Ma Z, Li L, Liu M, Qin L,
Yu Z, Zhao L, Liu Y, Zhang X, et al: Hyperactivation of
HER2-SHCBP1-PLK1 axis promotes tumor cell mitosis and impairs
trastuzumab sensitivity to gastric cancer. Nat Commun. 12:28122021.
View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Wan C, Hu X, Yang P, Li M, Nisar MF, Zhou
Z, Gan Y, Cai Y and Liu Z: Screening of theaflavins that inhibit
proliferation of nasopharyngeal carcinoma cells through
metabolomics approaches. Food Sci Nutr. 13:e706422025. View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Macpherson H, Formica M, Harris E and Daly
RM: Brain functional alterations in type 2 Diabetes-A systematic
review of fMRI studies. Front Neuroendocrinol. 47:34–46. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Cai X, Liu Z, Dong X, Wang Y, Zhu L, Li M
and Xu Y: Hypoglycemic and lipid lowering effects of theaflavins in
high-fat diet-induced obese mice. Food Funct. 12:9922–9931. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Burgess JL, Wyant WA, Abdo Abujamra B,
Kirsner RS and Jozic I: Diabetic wound-healing science. Medicina
(Kaunas). 57:10722021. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Desplantez T: Cardiac Cx43, Cx40 and Cx45
co-assembling: involvement of connexins epitopes in formation of
hemichannels and Gap junction channels. BMC Cell Biol. 18 (Suppl
1):32017. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Li X, Yu L, Gao J, Bi X, Zhang J, Xu S,
Wang M, Chen M, Qiu F and Fu G: Apelin ameliorates high
glucose-induced downregulation of connexin 43 via AMPK-Dependent
pathway in neonatal rat cardiomyocytes. Aging Dis. 9:66–76. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Shen Z, Chen Q, Jin T, Wang M, Ying H, Lu
J, Wang M, Zhang W, Qiu F, Jin C, et al: Theaflavin 3,3′-digallate
reverses the downregulation of connexin 43 and autophagy induced by
high glucose via AMPK activation in cardiomyocytes. J Cell Physiol.
234:17999–18016. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Fu H, He J, Li C and Chang H:
Theaflavin-3,3′-digallate protects liver and kidney functions in
diabetic rats by up-regulating Circ-ITCH and Nrf2 signaling
pathway. J Agric Food Chem. 72:14630–14639. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Rachner TD, Khosla S and Hofbauer LC:
Osteoporosis: Now and the future. Lancet. 377:1276–1287. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Oka Y, Iwai S, Amano H, Irie Y, Yatomi K,
Ryu K, Yamada S, Inagaki K and Oguchi K: Tea polyphenols inhibit
rat osteoclast formation and differentiation. J Pharmacol Sci.
118:55–64. 2012. View Article : Google Scholar
|
|
123
|
Qu Z, Liu C, Li P, Xiong W, Zeng Z, Liu A,
Xiao W, Huang J, Liu Z and Zhang S: Theaflavin promotes myogenic
differentiation by regulating the cell cycle and surface mechanical
properties of C2C12 cells. J Agric Food Chem. 68:9978–9992. 2020.
View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Sussman NL and Lucey MR: Alcohol and
alcoholic liver disease. Clin Liver Dis. 23:xiii–xiv. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
125
|
Yoshino K, Yamazaki K and Sano M:
Preventive effects of black tea theaflavins against mouse type IV
allergy. J Sci Food Agric. 90:1983–1987. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Lin CL, Huang HC and Lin JK: Theaflavins
attenuate hepatic lipid accumulation through activating AMPK in
human HepG2 cells. J Lipid Res. 48:2334–2343. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
127
|
Zhang W, An R, Li Q, Sun L, Lai X, Chen R,
Li D and Sun S: Theaflavin TF3 relieves hepatocyte lipid deposition
through activating an AMPK signaling pathway by targeting Plasma
Kallikrein. J Agric Food Chem. 68:2673–2683. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Chen G, Yuan H, Zhang L, Zhang J, Li K and
Wang X: Pancreatic lipase immobilization on cellulose filter paper
for inhibitors screening and network pharmacology study of
anti-obesity mechanism. Talanta. 280:1267502024. View Article : Google Scholar : PubMed/NCBI
|
|
129
|
Yang CS, Wang X, Lu G and Picinich SC:
Cancer prevention by tea: Animal studies, molecular mechanisms and
human relevance. Nat Rev Cancer. 9:429–439. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Chen D, Milacic V, Chen MS, Wan SB, Lam
WH, Huo C, Landis-Piwowar KR, Cui QC, Wali A, Chan TH and Dou QP:
Tea polyphenols, their biological effects and potential molecular
targets. Histol Histopathol. 23:487–496. 2008.PubMed/NCBI
|
|
131
|
Mozafari MR, Johnson C, Hatziantoniou S
and Demetzos C: Nanoliposomes and their applications in food
nanotechnology. J Liposome Res. 18:309–327. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Rashidinejad A, Birch EJ, Sun-Waterhouse D
and Everett DW: Delivery of green tea catechin and epigallocatechin
gallate in liposomes incorporated into low-fat hard cheese. Food
Chem. 156:176–183. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Han X, Niu X, Xu M, Feng R, Han Q, Liu B,
Cheng Y, Yun S, Cheng F, Feng C and Cao J: Flammulina velutipes
protein-Flammulina velutipes soluble polysaccharide-tea polyphenols
particles stabilized Pickering emulsions for the delivery of
β-carotene. Int J Biol Macromol. 285:1382992025. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Paiva L, Lima E, Motta M, Marcone M and
Baptista J: Investigation of the azorean camellia sinensis
processing conditions to maximize the theaflavin 3,3′-di-O-Gallate
content as a potential antiviral compound. Antioxidants (Basel).
11:10662022. View Article : Google Scholar : PubMed/NCBI
|
|
135
|
Li H, Shen S, Yu K, Wang H and Fu J:
Construction of porous structure-based carboxymethyl
chitosan/sodium alginate/tea polyphenols for wound dressing. Int J
Biol Macromol. 233:1234042023. View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Davis ME, Chen ZG and Shin DM:
Nanoparticle therapeutics: An emerging treatment modality for
cancer. Nat Rev Drug Discov. 7:771–782. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Kenchegowda M, Rahamathulla M, Hani U,
Begum MY, Guruswamy S, Osmani RAM, Gowrav MP, Alshehri S, Ghoneim
MM, Alshlowi A and Gowda DV: Smart nanocarriers as an emerging
platform for cancer therapy: A review. Molecules. 27:1462021.
View Article : Google Scholar : PubMed/NCBI
|
|
138
|
Chacko SM, Thambi PT, Kuttan R and
Nishigaki I: Beneficial effects of green tea: A literature review.
Chin Med. 5:132010. View Article : Google Scholar : PubMed/NCBI
|
|
139
|
Hara Y: Tea catechins and their
applications as supplements and pharmaceutics. Pharmacol Res.
64:100–104. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
140
|
Reagan-Shaw S, Nihal M and Ahmad N: Dose
translation from animal to human studies revisited. FASEB J.
22:659–661. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
141
|
Nair AB and Jacob S: A simple practice
guide for dose conversion between animals and human. J Basic Clin
Pharm. 7:27–31. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
142
|
Pimpão RC, Ventura MR, Ferreira RB,
Williamson G and Santos CN: Phenolic sulfates as new and highly
abundant metabolites in human plasma after ingestion of a mixed
berry fruit purée. Br J Nutr. 113:454–463. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
143
|
Shahrzad S, Aoyagi K, Winter A, Koyama A
and Bitsch I: Pharmacokinetics of gallic acid and its relative
bioavailability from tea in healthy humans. J Nutr. 131:1207–1210.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
144
|
Ji BC, Hsu WH, Yang JS, Hsia TC, Lu CC,
Chiang JH, Yang JL, Lin CH, Lin JJ, Suen LJ, et al: Gallic acid
induces apoptosis via caspase-3 and mitochondrion-dependent
pathways in vitro and suppresses lung xenograft tumor growth in
vivo. J Agric Food Chem. 57:7596–7604. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
145
|
Han YH, Moon HJ, You BR, Kim SZ, Kim SH
and Park WH: Propyl gallate inhibits the growth of HeLa cells via
caspase-dependent apoptosis as well as a G1 phase arrest of the
cell cycle. Oncol Rep. 23:1153–1158. 2010.PubMed/NCBI
|