Introduction
Cancer remains one of the most notable public health
challenges worldwide. Although the global disease burden persists,
overall cancer-related mortality has declined in recent decades,
largely due to the widespread implementation of early screening
programs, increased emphasis on preventive strategies and the
continuous development of innovative therapeutic modalities
(1). Contemporary oncology has
adopted a multidisciplinary and collaborative model of
comprehensive care, integrating surgery, radiotherapy,
chemotherapy, targeted therapy and immunotherapy. In clinical
settings, combination chemotherapy continues to represent the
standard first-line treatment for metastatic pancreatic cancer.
Notably, fluorouracil-based combination regimens have demonstrated
superior survival outcomes in patients with advanced pancreatic
disease when compared with gemcitabine combined with taxane-based
therapies (2). In addition,
multidisciplinary team-guided preoperative interventions serve a
pivotal role in optimizing perioperative management and improving
outcomes for patients with gastrointestinal malignancies (3). Beyond conventional treatments,
Traditional Chinese Medicine has been shown to exhibit diverse
biological activities, including immunomodulatory,
anti-inflammatory and direct antitumor effects, thereby providing
promising options for adjunctive cancer therapy (4,5). In
recent years, nanomedicine has emerged as a transformative approach
in oncology, exploiting nanoscale size effects and tunable surface
characteristics. Through mechanisms such as targeted drug delivery,
controlled release and enhanced immunogenicity, nanoparticle-based
systems can synergize with immunotherapy to improve therapeutic
efficacy in malignancies such as prostate cancer. In addition,
these platforms hold potential for overcoming immune evasion and
reshaping the tumor microenvironment (6). Furthermore, advances in smart
nanocarriers and stimulus-responsive drug delivery systems are
accelerating the transition of cancer therapy toward greater
precision and personalization. Nevertheless, conventional
therapeutic strategies continue to face substantial limitations,
including treatment-related toxicity, radio- and chemoresistance,
and pronounced heterogeneity in therapeutic efficacy across tumor
types. For example, survival rates for pancreatic cancer and
glioblastoma remain markedly lower than those observed in breast
and prostate cancer (7).
Consequently, the identification of novel therapeutic strategies
capable of reversing drug resistance, enhancing treatment
responsiveness and minimizing adverse effects remains a critical
priority and a central focus of contemporary oncology research.
MicroRNAs (miRNAs/miRs) are endogenous, non-coding
RNA molecules 20–24 nucleotides in length that function as critical
post-transcriptional regulators of gene expression. Their principal
mechanism involves binding to complementary sequences within the
3′-untranslated regions of target mRNAs, thereby inducing
transcriptional repression or translational inhibition (8). Through this mode of regulation,
miRNAs govern a wide spectrum of essential biological processes,
including cell proliferation, differentiation, apoptosis and
metabolic homeostasis. Notably, individual miRNAs are capable of
targeting hundreds of genes and a single gene may be simultaneously
regulated by multiple miRNAs, collectively forming highly complex
and interconnected regulatory networks (9). This extensive regulatory capacity
underscores the indispensable role of miRNAs in maintaining
cellular equilibrium. Accordingly, aberrant miRNA expression has
emerged as a defining feature of numerous pathological conditions,
including malignant tumors (10),
cardiovascular diseases (11),
neurodegenerative disorders (12)
and immune dysregulation (13).
Advances in high-throughput sequencing technologies and
bioinformatics analyses have further emphasized the clinical
potential of miRNAs as diagnostic biomarkers and therapeutic
targets, firmly establishing them as a central focus of epigenetic
research (14,15). In oncology, miRNAs are increasingly
recognized for their crucial involvement in maintaining cancer stem
cell properties and modulating therapeutic responses, particularly
in the development of drug resistance (16). Among these regulatory molecules,
miR-205 exhibits highly context-dependent expression patterns and
functional roles that vary substantially across different tissues
and cancer types. Its ability to act either as an oncogene or as a
tumor suppressor, a phenomenon commonly referred to as a ‘dual
role’ in cancer biology, has attracted considerable scientific
attention (17). Beyond
oncogenesis, miR-205 also participates in a range of non-malignant
pathological processes. The present review systematically
summarizes advances in the understanding of miR-205, encompassing
its biological characteristics, regulatory mechanisms, functional
roles and prognostic importance across diverse diseases.
Furthermore, it integrates current evidence on key target genes and
signaling pathways associated with miR-205, thereby providing a
foundation for the development of precise diagnostic tools and
targeted therapeutic strategies.
Expression patterns and physiological
functions of miR-205
Initially identified in mice and pufferfish, miR-205
is now recognized as an evolutionarily conserved miRNA, with
homologs present across species ranging from zebrafish to humans
(18,19). In humans, miR-205 is encoded on
chromosome 1q32.2. As research has progressed, it has become
evident that the biological functions of miR-205 in cancer are
highly context-dependent. Whether miR-205 exerts oncogenic or
tumor-suppressive effects is largely determined by the cellular
microenvironment and the specific gene networks under its
regulation (17). Similar to most
miRNAs, miR-205 does not induce cellular phenotypic changes, such
as alterations in proliferation, migration or invasion, through the
modulation of a single target gene. Instead, it concurrently
regulates multiple downstream targets, thereby initiating complex
and interconnected signaling cascades. For example, in lung cancer
models, downregulation of miR-205 results in the upregulation of
the transcription factor zinc finger E-box binding homeobox (ZEB)2,
which subsequently suppresses erbB3 expression. This multilayered
regulatory axis ultimately enhances tumor cell proliferation and
invasiveness, thereby accelerating disease progression. Notably,
the reduction in erbB3 expression represents an indirect downstream
consequence of miR-205 dysregulation in this context (20).
Beyond tumor biology, miR-205 serves an essential
role in maintaining epithelial homeostasis, and regulating tissue
repair and scar formation (Fig.
1). Loss of miR-205 disrupts PI3K/AKT signaling, leading to
impaired proliferation, adhesion and migration of skin progenitor
and stem cells. Such dysregulation can result in defective
epidermal barrier formation, abnormal hair follicle morphogenesis,
and, in severe cases, neonatal lethality (21). Supporting these observations, Jiang
et al (22) employed
Agilent miRNA microarrays combined with quantitative (q)PCR
validation to demonstrate marked downregulation of miR-205 in
hypertrophic scar tissue. Functional analyses revealed that miR-205
overexpression suppressed fibroblast migration, scar hyperplasia
and collagen synthesis by directly targeting thrombospondin-1.
Consistently, Qi et al (23) reported that miR-205 can inhibit
extracellular matrix production through direct targeting of Smad2,
thereby attenuating hypertrophic scar formation.
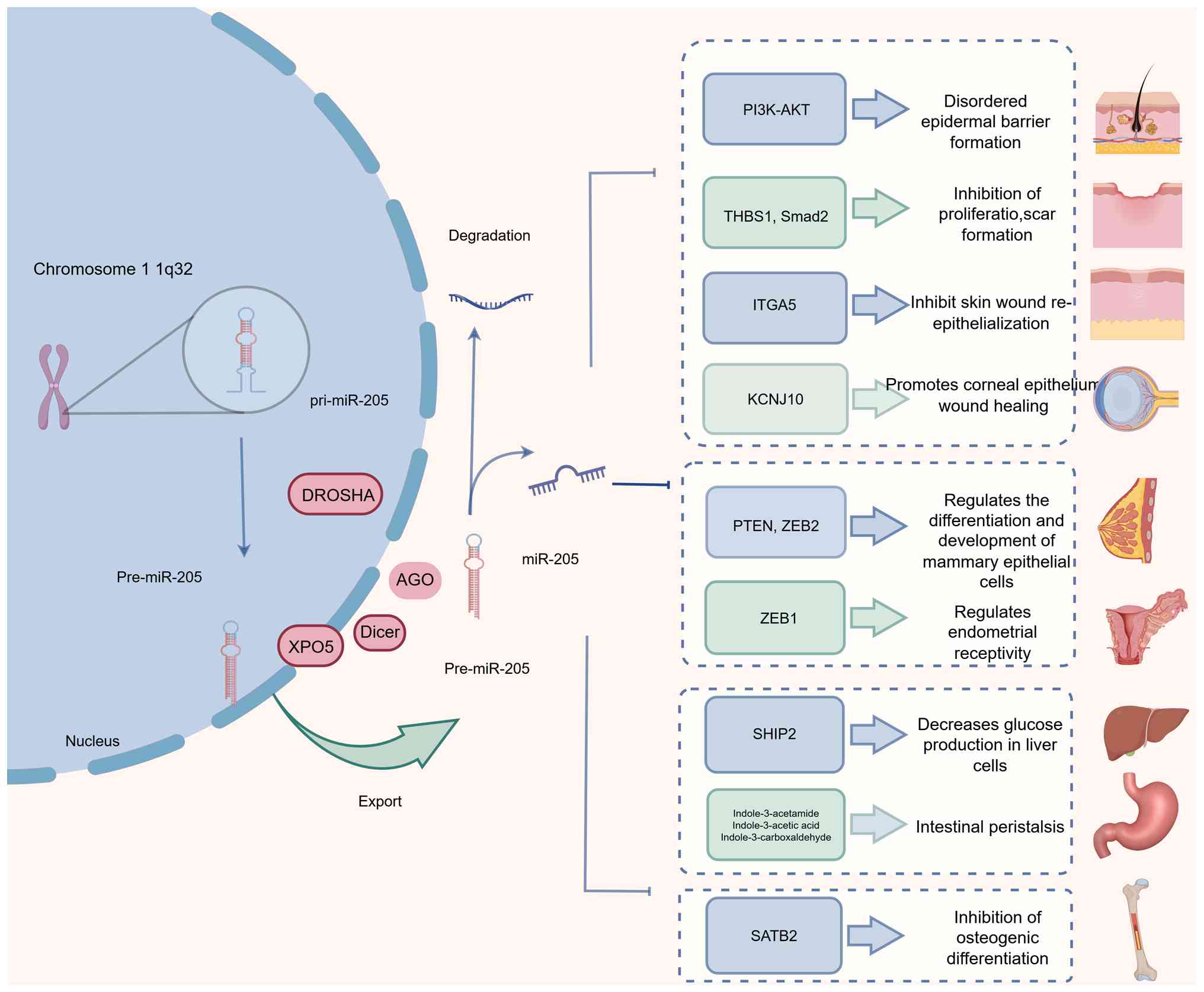 | Figure 1.Expression and physiological function
of miR-205 (normal arrows indicate promotion and T-shaped arrows
indicate inhibition). miR, microRNA; DROSHA, drosha ribonuclease
III; XPO5, exportin-5; DICER1, ribonuclease III gene; THBS1,
thrombospondin 1 gene; SMAD2, SMAD family member 2; ITGA5,
integrin, α5 (fibronectin receptor, alpha polypeptide); KCNJ10,
potassium inwardly rectifying channel subfamily J member 10; SHIP2,
Src homology 2 domain-containing inositol 5′-phosphatase 2; SATB2,
special AT-rich sequence-binding protein 2. |
Emerging evidence has further indicated that miR-205
participates in regulatory crosstalk involving long non-coding RNAs
(lncRNAs) and mRNAs via competition for shared miRNA response
elements. In this competitive endogenous RNA (ceRNA) network,
lncRNAs can function as molecular sponges for miRNAs, binding to
their complementary sequences and relieving repression of miRNA
target genes, thus adding an additional layer of
post-transcriptional regulation (24). In the context of keloid formation,
Su et al (25) demonstrated
that the lncRNA HOXA11-AS may act as a ceRNA for miR-205. Silencing
HOXA11-AS or restoring miR-205 expression significantly inhibited
fibroblast proliferation and extracellular matrix deposition while
promoting apoptosis, thereby suppressing pro-fibrotic processes.
Notably, the functional consequences of miR-205 regulation are
highly context-specific. In skin wound healing, Wang et al
(26) reported that miR-205
downregulation facilitated keratinocyte migration and accelerated
wound closure by suppressing integrin α5 expression. By contrast,
Lin et al (27) observed a
pro-healing role for miR-205 in the cornea, where its
overexpression promoted epithelial repair by targeting the
potassium channel KCNJ10 and enhancing cellular proliferation.
In a normal mammary epithelial cell culture model,
miR-205 was found to be highly expressed in progenitor-like cells,
where it promoted mammary epithelial differentiation and
development through repression of ZEB2 (28). Beyond epithelial tissues, miR-205
has also been implicated in reproductive biology. Yu et al
(29) reported a marked
upregulation of miR-205 in endometrial epithelial cells following
ovulation, whereas reduced miR-205 expression was observed in the
endometrial tissues of women with infertility. Notably,
intrauterine administration of a miR-205 inhibitor during the
mid-secretory phase, corresponding to the window of implantation,
significantly decreased embryo implantation rates and was
associated with altered regulation of ZEB1. These findings suggest
a critical role for miR-205 in modulating endometrial receptivity
and embryo implantation (29).
Beyond pathological conditions, miR-205 also serves
as an important regulator of fundamental physiological processes,
including systemic metabolism and cellular differentiation. Its
role in metabolic regulation has been highlighted by two
independent studies. In hepatocytes, miR-205 suppresses the
SHIP2/FOXO signaling axis, thereby reducing hepatic glucose
production (30). In parallel,
emerging evidence has suggested that miR-205 enhances intestinal
motility by modulating gut microbiota composition and tryptophan
metabolism (31). In the context
of cell differentiation, Hu et al (32) demonstrated that miR-205
overexpression significantly inhibited the osteogenic
differentiation potential of bone marrow-derived mesenchymal stem
cells (BMSCs). Mechanistically, this effect was mediated through
direct targeting of the SATB2/Runx2 signaling pathway, as reflected
by the downregulation of key osteogenic markers, including bone
sialoprotein, osteopontin, alkaline phosphatase (ALP) and
osteocalcin.
Transcriptional regulation of miR-205
Regulation by transcription
factors
The transcriptional regulation of miR-205 is
orchestrated by multiple transcription factors, among which p53
serves a central role (33).
Specifically, p53 directly activates miR-205 transcription by
binding to a canonical p53 response element (p53RE) located within
the upstream promoter region of the miR-205 gene. Through this
regulatory axis, miR-205 contributes to the control of critical
cellular processes, including cell cycle arrest, apoptosis and the
maintenance of genomic stability. Consequently, loss-of-function
mutations in TP53 can disrupt this transcriptional activation,
representing a key mechanism underlying miR-205 downregulation in
several malignancies, such as triple-negative breast cancer (BC)
(34). In addition to p53, the
closely related family members p63 and p73 also recognize and bind
to p53REs owing to their structural homology with p53, thereby
positively regulating miR-205 expression (35). However, in specific tumor
microenvironmental contexts, mutant p53 has been shown to
indirectly repress miR-205 transcription by destabilizing p63 and
impairing its transcriptional activity (36). Other transcription factors further
contribute to the fine-tuned regulation of miR-205. The
transcription factor Sp1 can bind to regulatory regions upstream of
the miR-205 host gene under conditions of DNA damage, thereby
promoting the co-transcription of miR-205 and its host gene. By
contrast, the epithelial-mesenchymal transition (EMT)-associated
transcription factor Twist1 directly interacts with the miR-205
promoter to suppress its transcription, linking miR-205 regulation
to EMT and tumor progression pathways (37).
Epigenetic regulation
DNA methylation is a fundamental epigenetic
modification characterized by the addition of a methyl group to the
fifth carbon of cytosine residues, resulting in the formation of
5-methylcytosine. Genomic regions enriched in cytosine-guanine
dinucleotides, known as CpG islands, are typically located within
upstream regulatory elements, particularly gene promoter regions.
Aberrant hypermethylation of promoter-associated CpG islands in
tumor suppressor genes can lead to transcriptional silencing and
functional inactivation, representing a common and critical
mechanism driving cancer initiation and progression (38). Epigenetic modifications, including
DNA and histone methylation, serve pivotal roles in a wide range of
pathophysiological processes, such as oncogenesis and aging, by
regulating gene expression, modulating chromatin architecture,
influencing protein function and affecting RNA processing. Among
these mechanisms, DNA methylation is essential for maintaining
genomic stability, organizing higher-order chromatin structure and
precisely controlling transcriptional activity (39).
Accumulating evidence has indicated that the
expression of specific miRNAs, including miR-205, is frequently
regulated by aberrant DNA methylation during disease development,
particularly in cancer, where CpG island hypermethylation commonly
leads to the silencing of tumor-associated miRNAs (40). For miR-205, CpG sites are densely
distributed within both the promoter region of its host gene
(MIR205HG) and the miRNA coding locus itself (34). In HER2-positive BC, activation of
the HER2/Ras/Raf/MEK/ERK signaling cascade induces the upregulation
of DNA methyltransferases, resulting in hypermethylation of the
MIR205HG promoter and consequent transcriptional repression of
miR-205 (41). This epigenetic
silencing contributes to BC progression, as reduced miR-205
expression is associated with the enhanced proliferation, invasion
and migratory capacity of cancer cells. Thus, elucidating the
methylation-dependent regulation of miR-205 not only expands the
understanding of miRNA-mediated oncogenic signaling in BC but also
highlights potential avenues for epigenetically targeted
therapeutic intervention.
Supporting this regulatory paradigm, Mancini et
al (42) reported coordinated
histone modifications at the miR-205 locus in prostate cancer
cells, including increased levels of the repressive mark H3K27me3
and reduced enrichment of the activating mark H3K4me3. Together,
these chromatin alterations may synergize with DNA methylation to
enforce stable silencing of miR-205 (42). Similarly, epigenetic mechanisms
have been implicated in EMT during lung cancer progression. Tellez
et al (43) demonstrated
that EMT initiation was preceded by chromatin remodeling
characterized by H3K27me3 accumulation, which was subsequently
consolidated by DNA methylation, leading to permanent repression of
miR-200b, miR-200c and miR-205. Downregulation of these miRNAs has
been shown to be strongly associated with a dedifferentiated
phenotype in both immortalized human bronchial epithelial cells and
primary lung tumors.
Beyond oncology, aberrant methylation of the miR-205
promoter has also been observed in non-malignant disorders. In a
cellular model of Parkinson's disease (PD), Wang et al
(44) identified hypermethylation
of the miR-205 promoter region. Notably, pharmacological
demethylation restored miR-205 expression and suppressed
leucine-rich repeat kinase 2 (LRRK2), implicating miR-205
methylation as a critical regulatory mechanism in PD pathogenesis
and a potential therapeutic target (44). Moreover, emerging evidence has
suggested that epigenetic regulation of miR-205 may influence
immune-related disease outcomes. In the context of COVID-19, Vaz
et al (45) reported that
elevated circulating miR-205 levels at hospital admission were
associated with an increased risk of adverse clinical outcomes,
proposing peripheral blood miR-205 as a potential prognostic
biomarker for predicting progression to severe or critical disease.
Collectively, these findings underscore the central role of
epigenetic dysregulation of miR-205 across a broad spectrum of
pathological conditions, and highlight its considerable promise as
both a prognostic indicator and a target for epigenetically
informed therapeutic strategies.
Role in non-cancerous diseases
Cardiovascular diseases
The role of miR-205 in cardiovascular and
hypertensive disorders is multifaceted and highly
context-dependent, with evidence supporting both protective and
disease-modifying functions. In the setting of environmental
stress-induced cardiotoxicity, Feng et al (46) demonstrated that PM2.5
exposure-mediated upregulation of miR-205 attenuated myocardial
injury by targeting IL-1 receptor-associated kinase-like 2 and
activating the TNF receptor-associated factor 6/NF-κB signaling
pathway. Consistent with these cardioprotective effects, Xiao et
al (47) reported that
enhanced miR-205 expression in atrial myocytes from an atrial
fibrosis rat model markedly suppressed atrial fibroblast
proliferation and migration through downregulation of
fibrosis-associated markers, including collagen I, α-smooth muscle
actin (α-SMA) and prolyl 4-hydroxylase subunit α3. A similar
protective role of miR-205 has been observed in hypertensive
disorders of pregnancy. Liu et al (48) showed that IL-32-mediated
downregulation of miR-205 promoted trophoblast invasiveness via
activation of the MMP2/MMP9/NF-κB axis, suggesting that elevated
miR-205 expression may suppress disease initiation and progression
in pregnancy-induced hypertension. The protective effects of
miR-205 extend to multiple cardiovascular pathologies through
distinct molecular mechanisms. In vascular disease, Huang et
al (49) demonstrated that the
natural compound icariin alleviated atherosclerosis by upregulating
miR-205, thereby inhibiting ERBB4 signaling in vascular smooth
muscle cells. By contrast, homocysteine (Hcy)-induced pulmonary
vascular dysfunction has been linked to hypermethylation-mediated
silencing of miR-205. Under physiological conditions, miR-205
supports microvascular angiogenesis by targeting FOXO1; thus, its
epigenetic repression contributes to vascular impairment (50). In the context of cardiac fibrosis,
Xiao et al demonstrated that miR-205 overexpression can
directly target euchromatic histone lysine methyltransferase 2,
thereby attenuating atrial fibrosis, and improving associated
mitochondrial and metabolic dysfunctions (51).
Diabetes
Accumulating evidence has indicated that miR-205
serves diverse and context-dependent roles in the pathogenesis of
diabetic complications. In the setting of diabetic wound healing,
Liu et al (52) treated
human umbilical vein endothelial cells with diabetic foot
ulcer-derived extracellular vesicles (DF-EVs; 5 µg/ml) and control
vesicles. Functional assays revealed that DF-EV exposure markedly
impaired endothelial cell migration and angiogenic capacity.
Consistently, wounds treated with DF-EVs exhibited markedly reduced
neovascularization within local granulation tissue by day 7,
indicating that DF-EVs negatively regulated angiogenesis in
diabetic foot ulcers. Mechanistically, miR-205 was found to be
enriched in EVs isolated from diabetic wound fluid, where it
inhibited angiogenesis and delayed wound healing by suppressing
vascular endothelial growth factor A (VEGFA) at both the mRNA and
protein levels (52,53). By contrast, miR-205 has been
implicated in the progression of diabetic nephropathy through a
distinct regulatory mechanism. Zheng et al (54) identified a double-negative feedback
loop in which miR-205 directly targeted histone deacetylase
(HDAC)2, whereas HDAC2 reciprocally repressed miR-205 transcription
via specificity protein 1 (SP1) binding sites. This
HDAC2/SP1/miR-205 regulatory circuit can promote extracellular
matrix accumulation in renal tubular epithelial cells, thereby
accelerating the progression of diabetic nephropathy (54). Beyond vascular and renal
complications, miR-205 also contributes to pancreatic islet
dysfunction. Ouni et al (55) reported notable upregulation of
miR-205 in the pancreatic islets of diabetes-prone mice, where it
impaired islet function by directly targeting the
diabetes-associated transcription factor Tcf7l2.
Neurological diseases
Beyond its established roles in cancer and
metabolism, miR-205 is increasingly recognized as a critical
regulator in diverse neurological disorders. In PD, a
neurodegenerative condition marked by progressive motor
dysfunction, Wang et al (44) demonstrated that
hypermethylation-induced silencing of the miR-205 promoter
contributed to disease pathogenesis via dysregulation of LRRK2
expression. In models of depression, He et al (56) reported that Mahonia alkaloids
ameliorated depressive-like behaviors by downregulating miR-205,
which led to increased expression of key neuroprotective factors
such as connexin 43, brain-derived neurotrophic factor and cAMP
response element-binding protein. Furthermore, neurotoxicity
induced by sevoflurane (Sev), a commonly used volatile anesthetic,
can be mitigated through modulation of miR-205 (57,58).
Zhang et al (58) reported
that knockdown of the lncRNA NKILA alleviated Sev-induced
neurotoxicity by downregulating miR-205 and subsequently
upregulating ELAVL1. Furthermore, in cerebral ischemia models, Yang
et al (59) demonstrated
that dexmedetomidine treatment upregulated miR-205, which inhibited
high-mobility group box 1 (HMGB1), thereby reducing oxidative
stress and inflammation, and improving ischemia/reperfusion injury
outcomes in rats.
Rheumatoid arthritis (RA),
osteoporosis (OP) and other orthopedic diseases
miR-205 exhibits dynamic expression during the
chondrogenic differentiation of BMSCs and serves important roles in
multiple orthopedic diseases, including RA and OP (60). Ma et al (61) demonstrated that exosomal miR-205
(exo-miR-205) derived from BMSCs attenuated RA progression in
vitro by targeting MDM2, which in turn modulated the MAPK and
NF-κB signaling pathways, key mediators of inflammation and joint
degradation. In OP, Huang et al (60) identified miR-205 as upregulated via
microarray and bioinformatics analyses, findings subsequently
validated by reverse transcription (RT)-qPCR in clinical samples
from 30 patients with OP. Functional studies revealed that miR-205
expression was reduced during osteogenic differentiation, and its
overexpression inhibited this process by targeting RUNX2, resulting
in reduced expression of osteogenic markers such as collagen type I
α1 and ALP. Conversely, inhibition of miR-205 promoted osteogenic
differentiation (60). In
intervertebral disc degeneration (IDD), Zhu et al (62) reported that the lncRNA LINC00284
was upregulated in degenerative disc tissues and IL-1β-stimulated
nucleus pulposus (NP) cells. Knockdown of LINC00284 mitigated NP
cell degeneration and extracellular matrix degradation by
functioning as a molecular sponge for miR-205, thereby relieving
suppression of the Wnt/β-catenin pathway. This led to enhanced cell
proliferation, reduced apoptosis and decreased MMP3 expression,
ultimately alleviating IDD progression (62).
Chronic periodontitis
Jiang et al (63) identified a potential role for
miR-205 in the resolution of chronic periodontitis. This previous
study detected elevated serum levels of miR-205 in patients
following treatment, which were associated with downregulation of
its target, HMGB1. Clinically, increased miR-205 expression was
associated with improvements in key periodontal parameters,
including probing depth, attachment loss, plaque index and gingival
index, whereas HMGB1 expression was positively associated with
disease severity. These findings position miR-205 and HMGB1 as
critical modulators of chronic periodontitis pathogenesis and as
promising therapeutic targets (63). Building on the recognized
importance of the T helper cell (Th)17/regulator T cell (Treg)
balance in inflammatory regulation (64), Kang et al (65) explored the therapeutic potential of
exo-miR-205 derived from periodontal ligament stem cells. In a rat
model of periodontitis, administration of exo-miR-205 targeted
X-box binding protein 1, shifted immune homeostasis towards Tregs,
attenuated pro-inflammatory cytokine production, including TNF-α,
IL-6 and IL-1β, and ultimately suppressed disease progression
(65).
Allergic rhinitis (AR)
AR is an immunoglobulin E (IgE)-mediated
inflammatory disorder of the upper airways. Clinical analysis by
Suojalehto et al (66)
detected upregulation of miR-205 in the nasal mucosa of patients
with symptomatic AR. Complementary experimental data from Zhang
et al (67) using an
ovalbumin (OVA)-sensitized murine model of AR showed notable
increases in miR-205 expression. Mechanistically, knockdown of
miR-205 ameliorated allergic responses by reducing serum levels of
total and OVA-specific IgE, suppressing local production of Th2
cytokines (IL-4, IL-5 and IL-13) in the nasal mucosa, thereby
inhibiting AR development (67).
In a related study, the circular RNA (circRNA) circARF3 has been
shown to alleviate AR symptoms in mice by acting as a molecular
sponge for miR-205, resulting in upregulation of Sirtuin 5 and
consequent attenuation of allergic inflammation (68). Collectively, these findings
underscore a pathogenic role for miR-205 in AR and highlight its
potential as a therapeutic target.
Other inflammatory conditions
The role of miR-205 in inflammation is highly
context-dependent, exhibiting both pathogenic and protective
effects depending on the disease model. In abdominal aortic
aneurysm (AAA), Kim et al (69) identified a pathogenic role for
miR-205, which can target the protective factors TIMP3 and RECK.
This targeting led to unchecked MMP activity and exacerbated
inflammation, thereby driving AAA progression. Conversely, in
models of sepsis and post-traumatic lung injury, miR-205 exerts a
protective effect by targeting HMGB1, mitigating excessive
inflammatory responses and reducing tissue damage (70).
Respiratory diseases
In respiratory diseases, miR-205 serves diverse
roles through the regulation of distinct molecular targets. Zhao
et al (71) demonstrated
that under hypoxic conditions, proline-rich protein VII induced the
upregulation of miR-205 in rat pulmonary vascular smooth muscle
cells. Elevated miR-205 attenuated pulmonary hypertension by
targeting β-catenin, leading to inhibition of cell proliferation
and promotion of apoptosis (70).
In acute respiratory distress syndrome (ARDS), the serum levels of
miR-205 were shown to be elevated and to exert protective effects
by targeting COMM domain-containing protein 1. This interaction
antagonizes the pro-survival function of the lncRNA SNHG5 in A549
cells, thereby attenuating ARDS pathogenesis (72). In pulmonary fibrosis (PF), Sun
et al (73) demonstrated
that miR-205 overexpression improved fibrotic pathology by
targeting GATA-binding protein 3 and suppressing endoplasmic
reticulum stress in a murine model. Similarly, in silicosis,
miR-205 expression has been reported to be downregulated in
alveolar macrophages. Mechanistic experiments have revealed that
miR-205 targets E2F1 to reduce S-phase kinase-associated protein
2-mediated ubiquitination of Beclin1, promoting autophagy and
inhibiting the progression of silicosis-associated PF (74).
Urinary system disorders
Renal interstitial fibrosis (RIF), a progressive and
irreversible pathological hallmark of chronic kidney disease,
ultimately leads to end-stage renal disease. In a unilateral
ureteral obstruction mouse model of RIF, miR-205 expression has
been shown to be markedly downregulated. By contrast, restoration
of miR-205 levels suppresses the progression of fibrosis by
inhibiting HDAC5, resulting in reduced expression of key fibrotic
markers, including α-SMA, collagen IV and fibronectin (75). In acute kidney injury, Zhang et
al (76) demonstrated that
miR-205 overexpression ameliorated sepsis-induced renal damage in
rats by concurrently targeting HMGB1 and phosphatase and tensin
homolog (PTEN). Furthermore, Zhou et al (77) identified a novel regulatory axis in
sepsis-associated acute kidney injury (SA-AKI), wherein the circRNA
circ_0006944, upregulated in patients with SA-AKI, acted as a
molecular sponge for miR-205. Functional upregulation of miR-205
alleviated SA-AKI by targeting ubiquitin-like protein 4A (UBL4A),
highlighting the circ_0006944/miR-205/UBL4A pathway as a key
mechanism in this condition.
Psoriasis
In psoriasis, Xue et al (78) observed notable downregulation of
miR-205 in patient skin lesions. Functional assays showed that
miR-205 overexpression ameliorated the psoriatic phenotype in a
mouse model by targeting angiopoietin-2, VEGFA, bone morphogenetic
protein and activin membrane-bound inhibitor, thereby inactivating
the MAPK and Wnt/β-catenin signaling pathways (78).
Digestive system diseases
The role of miR-205 in liver injury is complex and
context-dependent. In trichloroethylene (TCE)-induced liver injury,
Wang et al (79) reported
that miR-205 overexpression exacerbated hepatic damage by targeting
retinoic acid receptor-related orphan receptor α (RORα), promoting
M1 macrophage polarization and inflammation. Similarly, Hu et
al (80) revealed that miR-205
was upregulated in non-alcoholic fatty liver disease models and
contributed to disease progression by targeting neuraminidase 1.
Conversely, Fang et al (81) revealed a protective role in
alcohol-related liver disease, where miR-205 targeted importin α5
to suppress NF-κB pathway activation and mitigate liver pathology.
Beyond hepatic diseases, Smith et al (82) identified downregulation of miR-205
in ulcerative esophagitis. This previous functional study indicated
that miR-205 upregulation may promote epithelial repair in response
to reflux injury by inhibiting cytokeratin 14 expression, inducing
apoptosis and suppressing proliferation in esophageal epithelial
cells (82).
Influenza A
Bao et al (83) confirmed that both oseltamivir and
Jin Chai Kangbingdu Capsule can inhibit influenza A virus
replication by upregulating miR-205, which directly targets the
viral nucleoprotein gene and suppresses its expression.
Sepsis
In a septic model using lipopolysaccharide-induced
HK-2 cells, the lncRNA TapSAKI has been reported to be upregulated,
whereas miR-205 is downregulated. Mechanistically, miR-205
overexpression can alleviate cytotoxic injury and suppress sepsis
progression by targeting interferon regulatory factor 3 (84).
Myelodysplastic syndrome (MDS)
Jang et al (85) identified a marked 12.5-fold
increase in serum miR-205 levels in 65 patients with MDS compared
with in 11 controls. Subsequent validation demonstrated that
miR-205 promoted MDS pathogenesis by targeting the tumor suppressor
PTEN (85).
It is evident that miR-205 serves a crucial role in
various non-malignant diseases, holding profound implications for
the treatment of diverse conditions (Fig. 2).
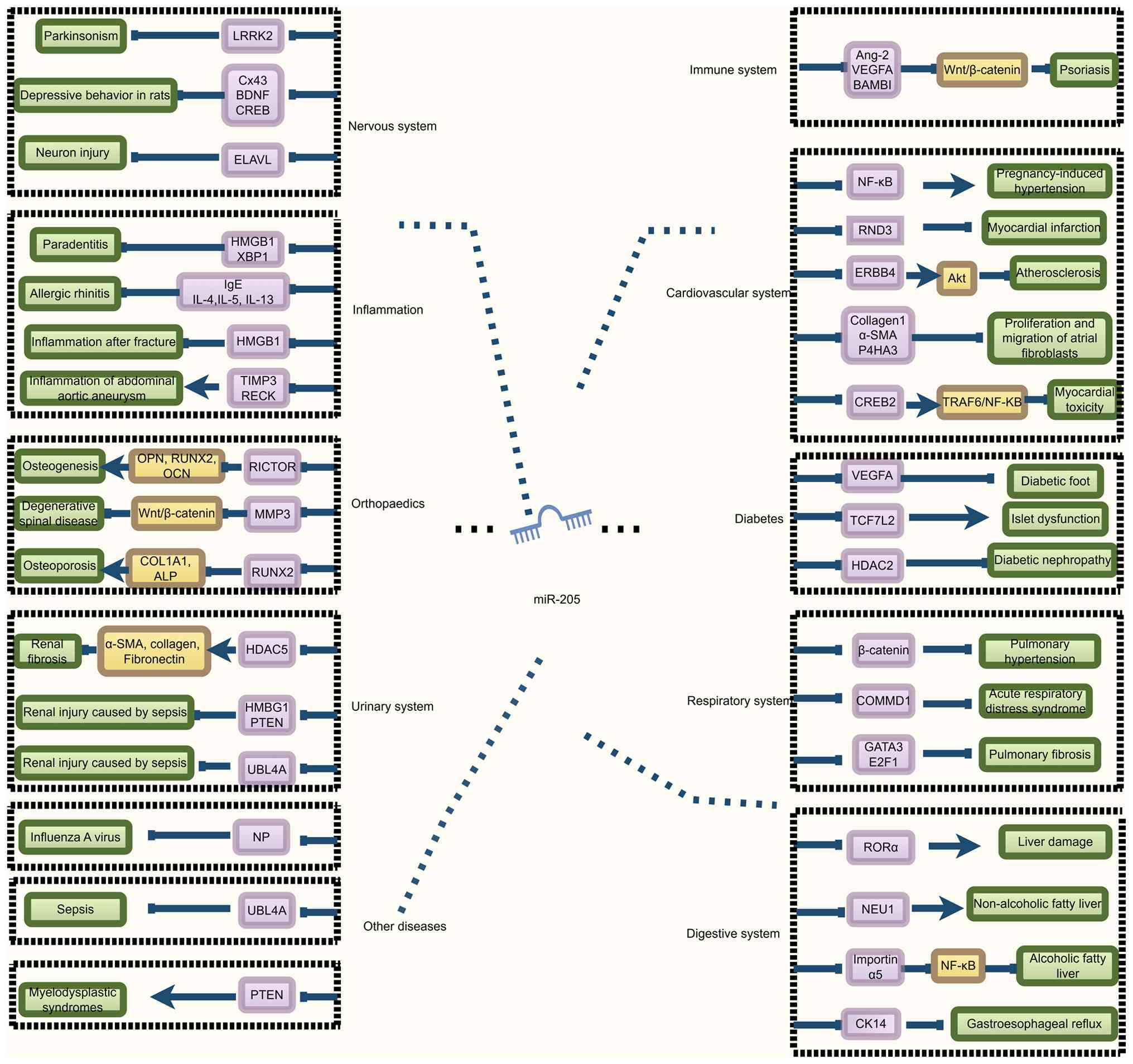 | Figure 2.Role of miR-205 in non-cancerous
diseases (normal arrows indicate promotion and T-shaped arrows
indicate inhibition. MiR-205 regulates diseases (green) by
inhibiting the downstream target gene (purple). LRKK2, leucine-rich
repeat kinase 2; Cx43, connexin 43; BDNF, brain-derived
neurotrophic factor; CREB, cAMP-response element binding protein;
ELVAL, embryonic lethal abnormal vision like 1; HMGB1, high
mobility group box 1; XBP1, X-box binding protein 1; IL,
interleukin; TIMP3, TIMP metallopeptidase inhibitor 3; RECK,
reversion-inducing cysteine-rich protein with Kazal motifs; RICTOR,
RPTOR independent companion of MTOR complex 2; MMP3, matrix
metalloproteinase 3; RUNX2, recombinant human Runt-related
transcription factor 2; HDAC5, histone deacetylase 5; UBL4A,
ubiquitin-like 4A; OPN, osteopontin; OCN, bone
gamma-carboxyglutamate protein; ALP, alkaline phosphatase; α-SMA,
α-smooth muscle actin; Ang-2, angiopoietin-2; BAMBI, BMP and
activin membrane bound inhibitor; RND3, Rho family GTPase 3; ERBB4,
Erb-B2 receptor tyrosine kinase 4; P4HA3, prolyl 4-hydroxylase
subunit α3; CREB2, cyclic AMP-responsive element-binding protein 2;
TCF7L2, transcription factor 7 Like 2; HDAC2, histone deacetylase
2; COMMD1, copper metabolism MURR1 domain 1; GATA3, GATA binding
protein 3; E2F1, E2F transcription factor 1; RORα, RAR-related
orphan receptor α; NEU1, neuraminidase 1; CK14, cytokeratin 14;
TRAF6, TNF receptor-associated factor 6. |
Role of miR-205 in malignant tumors
Expression characteristics in
cancer
miR-205 exhibits distinct, tissue-specific
expression patterns across various malignancies, which underlie its
context-dependent dual role in tumorigenesis and cancer progression
(86). It can function either as a
tumor suppressor, commonly downregulated in cancers such as renal
cell carcinoma (87), prostate
cancer (88), BC, colorectal
cancer (CRC) and melanoma (89),
or as an oncogene, promoting tumor development in malignancies
including non-small cell lung cancer (NSCLC) (90), bladder cancer (91), ovarian cancer (92), nasopharyngeal carcinoma (NPC), head
and neck cancer (93) and
esophageal adenocarcinoma (94).
This functional dichotomy highlights the complex,
microenvironment-dependent regulatory networks that govern miR-205
activity (Table I) (17,93,95–112).
 | Table I.Roles of microRNA-205 in malignant
tumor. |
Table I.
Roles of microRNA-205 in malignant
tumor.
| A, Tumor
suppressive role |
|---|
|
|---|
| First author,
year | Target gene | Cancer type | Mechanism | (Refs.) |
|---|
| Kalinkova et
al, 2021 | ZEB1 | Breast cancer | Restrains invasion
and migration | (97) |
| Shen et al,
2021 | E-cadherin | Breast cancer | Restrains invasion
and migration | (98) |
| Ma et al,
2021 | CXCL11 | Gastric cancer | Inhibition of
proliferation and invasion | (99) |
| Wang et al,
2010 | CCNB2 | Thyroid cancer | Inhibition of
proliferation and migration | (100) |
| Wang et al,
2016 | PTEN | Renal
carcinoma | Inhibition of
proliferation, invasion and migration | (101) |
| Qiao et al,
2010 | MAPK10 | Ovarian cancer | Inhibition of
proliferation and migration | (102) |
| Wang et al,
2025 | VEGFA | Pancreatic ductal
carcinoma | Inhibition of lymph
node metastasis | (104) |
|
| B, Oncogenic
role |
|
| First author,
year | Target
gene | Cancer
type |
Mechanism | (Refs.) |
|
| Zhang et al,
2020 | ANKRD2 | Head and neck
squamous cell carcinoma | Ability to promote
proliferation, invasion and migration | (93) |
| Ma et al,
2014; Xie et al, 2012; Witten et al, 2010 Liu et
al, 2020 | CHN1 | Cervical
cancer | Promotes
proliferation, invasion and migration | (105–108) |
| Yang et al,
2022 | DSC2 | Nasal pharyngeal
cancer | Promotes
proliferation, invasion and migration | (110) |
| Guo et al,
2025 | CALM1 | Nasal pharyngeal
cancer | Promotes
proliferation, invasion and migration | (109) |
| Yi et al,
2022 | DNAJA1 | Liver cancer | Ability to promote
proliferation and migration | (111) |
| Xu et al,
2021 | APBB2 | Non-small cell lung
cancer | Ability to promote
proliferation and migration | (112) |
Tumor suppressive role
Evidence suggests that the loss of miR-205
expression may facilitate mammary gland development (95). Consistent with its
tumor-suppressive role, miR-205 is frequently downregulated in BC,
with expression levels inversely associated with malignancy grade
(96). Functionally, Kalinkova
et al (97) demonstrated
that restoring miR-205 expression in BC cells inhibited invasion
and migration by targeting ZEB1. This anti-invasive effect has been
further supported by findings that miR-205 upregulation promotes
the epithelial marker E-cadherin while suppressing the mesenchymal
marker vimentin (98). The
tumor-suppressive role of miR-205 extends beyond BC. For example,
in gastric cancer, Ma et al (99) reported that miR-205 impeded cell
proliferation and invasion by downregulating CXCL11 and inhibiting
AKT signaling. Similarly, Wang et al (100) reported that miR-205 can target
cyclin B2 to inhibit proliferation and migration in thyroid cancer
cells, where Wang et al (101) demonstrated that suppression of
the PTEN/AKT pathway by miR-205 curbed malignant phenotypes in
renal cell carcinoma.
The function of miR-205 shows notable context
dependence, even within the same cancer type. In ovarian cancer,
some studies have described a tumor-suppressive role: miR-205
expression has been reported to be downregulated in ovarian cancer
cells, with its overexpression inhibiting proliferation and
migration by targeting MAPK10 (102). Conversely, Cai et al
(103) reported an oncogenic
function, showing that miR-205 enhanced invasion in ovarian cancer
cell lines (OVCAR-5, OVCAR-8 and SKOV-3) and promoted tumorigenesis
by downregulating TCF21, which led to upregulation of MMP2 and
MMP10 (92,103). This suggests that the role of
miR-205 may vary across different molecular subtypes or stages of
ovarian cancer. Similarly, in pancreatic ductal adenocarcinoma
(PDAC), evidence points to a dual role: Wang et al (104) identified elevated miR-205 in
plasma-derived EVs and PDAC tissues, whereas EV-miR-205 appeared to
suppress metastasis by targeting VEGFA and was negatively
associated with lymph node metastasis.
Oncogenic role
miR-205 acts as an oncogene in several malignancies,
as demonstrated by its upregulated expression in both tumor tissue
and patient serum. In cervical cancer, miR-205 overexpression has
been shown to promote proliferation, invasion and migration by
directly targeting and downregulating chimerin 1 (105–108). Similarly, in NPC, miR-205
enhances tumorigenesis and progression through multiple mechanisms:
It drives cell proliferation and invasion by suppressing
calmodulin-1 (109), and
facilitates metastasis via exosome-mediated downregulation of
desmocollin-2, which activates the EGFR/ERK pathway and upregulates
MMP2 and MMP9 (110). This
oncogenic role is also evident in liver cancer, where miR-205
targets DNAJA1 to promote proliferation and metastasis (111), and in NSCLC, where it fosters
tumor progression by downregulating APBB2 (112). The functional importance of
miR-205 in these types of cancer is further emphasized by
regulatory interactions with tumor-suppressive lncRNAs such as
VENTXP1, which inhibits head and neck squamous cell carcinoma
tumorigenesis by repressing miR-205 and consequently upregulating
its target ankyrin repeat domain 2 (93).
Clinical application prospects
Diagnostic value
miRNAs are highly stable molecules that can be
reliably detected in various physiological fluids, such as serum
and plasma, as well as tissue specimens, using multiple robust
technical platforms (113). Due
to their notable potential for elucidating disease mechanisms, and
advancing diagnostic and therapeutic strategies, miRNAs have become
a major focus of biomedical research (114). Notably, their application as
non-invasive biomarkers for cancer diagnosis and prognosis has
garnered substantial interest (115).
Evidence supports the substantial diagnostic and
prognostic value of miR-205 in several types of cancer. Re et
al (116) conducted a miRNome
analysis using next-generation sequencing on 43 patients with
intestinal-type sinonasal adenocarcinoma (ITAC) following surgical
resection. This previous study revealed downregulation of miR-205
in tumor tissues, with low miR-205 expression serving as an
independent predictor of poorer disease-free survival (DFS) and
overall survival (OS), highlighting its potential as a prognostic
biomarker for ITAC (116).
Complementing these findings, Li et al (117) performed a meta-analysis
evaluating the diagnostic efficacy of miR-205 in lung cancer,
primarily squamous cell carcinoma, involving 564 patients and 667
controls. The analysis demonstrated high diagnostic performance,
with a sensitivity of 0.88 (95% CI: 0.78–0.94), specificity of 0.78
(95% CI: 0.66–0.86) and an area under the receiver operating
characteristic curve (AUC) of 0.90 (95% CI: 0.87–0.92). The
diagnostic odds ratio of 25.86 further underscored its strong
potential for lung cancer screening and clinical application
(117). The diagnostic utility of
miR-205 is also supported by its detectability in circulating
exosomes and EVs. Zhao et al (118), analyzing data from The Cancer
Genome Atlas, reported notable downregulation of exo-miR-205 in
patients with CRC, including those with early-stage disease.
Although sensitivity (53.6%) and specificity (71.9%) for
early-stage CRC were moderate, the notable postoperative increase
in miR-205 levels suggested its potential as a dynamic marker for
disease monitoring (118). This
aligns with growing interest in EV-derived miRNAs as biomarkers for
cancer diagnosis and prognosis (119). Supporting this, a clinical study
by Bang et al (120)
involving 220 subjects found that EV-derived miR-205 was elevated
in patients with cancer-associated stroke compared with those with
cancer alone, indicating its utility in predicting cancer-related
coagulopathies. Similarly, Wang et al (121) demonstrated markedly elevated
serum exo-miR-205 levels in patients with NSCLC relative to
controls, further reinforcing its diagnostic relevance.
Zhao et al (122) investigated the diagnostic
potential of miR-205 in thyroid nodules by analyzing patient serum
samples. This previous study revealed that both miR-205 and thyroid
stimulating hormone receptor (TSHR) mRNA expression levels were
notably elevated in patients with benign or malignant thyroid
nodules compared with those in the controls. For diagnosing thyroid
nodules (vs. controls), miR-205 alone yielded an AUC of 0.867,
outperforming TSHR mRNA (AUC=0.760). Notably, combining both
markers improved diagnostic accuracy, achieving an AUC of 0.896
with a sensitivity of 96.43% and specificity of 76.81% at the
optimal cut-off. Additionally, miR-205 and TSHR mRNA maintained
moderate diagnostic efficacy in distinguishing malignant from
benign nodules, with AUC values of 0.738 and 0.729, respectively.
These findings collectively underscored the clinical utility of
miR-205, especially when combined with TSHR mRNA, as a valuable
biomarker panel for thyroid nodule diagnosis and risk
stratification (122).
Prognostic value
miR-205 serves a pivotal regulatory role in
gynecological malignancies and demonstrates prognostic potential
across various types of cancer. A comprehensive meta-analysis by Wu
et al (123), encompassing
data from 5,835 patients, revealed that the prognostic impact of
miR-205 was highly cancer-type-specific. In BC, elevated miR-205
expression was significantly associated with improved OS [hazard
ratio (HR)=0.84, 95% confidence interval (CI): 0.72–0.98, P=0.022].
By contrast, in endometrial cancer, upregulated miR-205 predicted
poorer disease-specific survival (HR=2.19, 95% CI: 1.45–3.32,
P<0.001), highlighting its dual prognostic role and potential as
a therapeutic target in both malignancies (123). Beyond gynecological cancer,
miR-205 also exhibits tumor-suppressive properties in other
malignancies. Lu et al (124) reported marked downregulation of
miR-205 in hepatocellular carcinoma (HCC), especially in aggressive
tumor subtypes. Lower miR-205 expression was associated with
unfavorable clinicopathological characteristics, and shorter DFS
and OS, supporting its role as a tumor suppressor and promising
prognostic biomarker in HCC (124).
Therapeutic implications and overcoming
chemoresistance
Therapeutic potential in cancer
Beyond its prognostic importance, miR-205 has
emerged as a critical modulator of tumor chemoresistance,
demonstrating the ability to reverse drug resistance across
multiple cancer types (125). The
mechanisms underlying this effect frequently involve the inhibition
of key resistance-related pathways. For example, in
doxorubicin-resistant liver cancer cells (HepG2/DOX), miR-205
overexpression has been shown to restore sensitivity by
upregulating PTEN, which suppresses the PI3K/AKT/P-glycoprotein
(P-gp) axis and reduces P-gp expression, ultimately inhibiting
proliferation and inducing apoptosis (126). Similarly, in BC, miR-205
overcomes tamoxifen resistance by targeting mediator complex
subunit 1 (MED1) and HER3, disrupting the HER3-PI3K/AKT-MED1
signaling cascade (127). This
chemosensitizing role extends to other agents as well; for example,
miR-205 enhances cisplatin sensitivity in glioma by targeting E2F1
(128), and in gallbladder cancer
stem cells, its upregulation suppresses both the mRNA and protein
levels of PRKCE, thereby inhibiting gemcitabine resistance
(129).
Despite their therapeutic potential, the clinical
translation of miRNA-based therapies such as miR-205 faces notable
challenges, particularly in achieving tumor-specific targeting and
efficient in vivo delivery. Conventional targeted therapies
often encounter issues including drug resistance, adverse side
effects and limited efficacy against metastatic disease,
underscoring the urgent need for innovative delivery strategies
(130). Advances in nanomedicine
and natural delivery systems offer promising solutions.
Nanoparticle platforms, including zinc oxide and selenium
nanoparticles, exhibit intrinsic antitumor properties while serving
as carriers for therapeutic agents (131). Additionally, certain
plant-derived compounds, such as artemisinin and curcumin, can
modulate miRNA expression. Razzaq et al (132) demonstrated that miR-205, which is
typically downregulated in BC, can be effectively restored using
plant extracts, nanoparticles or hybrid plant-nano materials,
highlighting the potential of these hybrid strategies to sensitize
cancer cells and inhibit tumor progression. Lin et al
(133) developed a miRNA delivery
system utilizing PLGA nanoparticles conjugated with dual
cell-penetrating peptides (CPPs, R9 and p28) to target cutaneous
squamous cell carcinoma (cSCC). This previous study demonstrated
that CPP-conjugated nanoparticles loaded with miR-205 effectively
induced tumor regression in a mouse cSCC model and inhibited the
migratory ability of cSCC cells. Flow cytometric analysis further
revealed that miR-205 upregulation promoted apoptosis in cSCC
cells, thereby suppressing tumor growth. These results suggested
that such nanoparticle-based miRNA delivery systems represent a
promising therapeutic strategy for tumor treatment (133).
Therapeutic potential in non-tumor diseases
Exosomes have emerged as natural delivery vehicles
with superior biocompatibility and targeting abilities compared
with traditional biomarkers. For instance, Zhang et al
(134) successfully encapsulated
miR-205 within mesenchymal stem cell-derived exosomes, markedly
enhancing endothelial barrier function and reducing vascular
leakage in an alloxan-induced diabetic mouse model of retinopathy,
illustrating a novel, efficient therapeutic approach for vascular
diseases. Ybarra et al (135) first administered miR-205 mimics
to diabetic mice via vitreous cavity injection, observing a notable
reduction in VEGF levels. This decrease may be associated with
improved erythropoietic processes in abnormal blood vessels. Based
on these findings, it was proposed that intravitreal injection of
miR-205 may be used as a potential therapy for treating
angiogenesis (135). Rubini et
al (136) employed an
exosome-based delivery system to administer miRNAs, including
miR-205, as a therapeutic intervention for feline idiopathic
cystitis. Following treatment, a substantial proportion of cats
exhibited symptom relief within a relatively short period, with
~70% showing clinical improvement by day 15. Ultrasonographic
evaluations conducted before and after treatment revealed marked
morphological changes in the bladder, characterized by the
resolution of intraluminal material and restoration of mucosal
integrity. These findings indicated a successful therapeutic
outcome following the intervention (136).
Baicalin has been shown to upregulate miR-205
expression in hepatocytes, which subsequently targets and inhibits
importin α5, leading to inactivation of the NF-κB signaling
pathway. This cascade reduces the release of pro-inflammatory
cytokines, alleviates oxidative stress and suppresses hepatocyte
apoptosis, thereby attenuating the progression of
alcohol-associated liver disease. These findings have demonstrated
that certain herbal components can exert hepatoprotective effects
by upregulating miR-205, highlighting the therapeutic potential of
miR-205 in clinical applications (81). Huang et al (50) demonstrated that methylation levels
in the miR-205 promoter region were increased in methionine-fed
mice and Hcy-treated pulmonary microvascular endothelial cells
(PMVECs), leading to reduced miR-205 expression. RT-qPCR results
indicated that miR-205 reduction aggravate pulmonary vascular
dysfunction. These findings indicated that hypermethylation of the
miR-205 promoter may represent a key pathogenic mechanism in
Hcy-induced PMVEC dysfunction. Overexpression of miR-205 could
serve as a potential therapeutic target for protecting against
Hcy-induced pulmonary microvascular dysfunction (50). Zhou et al (137) used miRNA microarray and RT-qPCR
experiments to determine that miR-205 expression levels were
downregulated in fresh endometriotic tissue. Subsequently, miR-205
was upregulated in endometrial cells, and was shown to target and
suppress angiopoietin-2 (Ang2) expression, thereby activating the
ERK/AKT pathway in ectopic endometrial cells. Ang2 is a growth
factor belonging to the Ang/tyrosine kinase with Ig and EGF
homology domains signaling pathway, one of the main pathways
involved in angiogenesis. This inhibition reduced the migration and
invasion of ectopic endometrial stromal cells while promoting
apoptosis. These findings demonstrated that miR-205 serves as a
novel diagnostic biomarker and therapeutic target for endometriosis
treatment (137). Huang et
al (49) demonstrated that
upregulation of miR-205 expression reduced lipid accumulation and
plaque formation in mouse blood vessels, promoted apoptosis and
inhibited cell migration in an in vitro atherosclerotic cell
model constructed from human aortic vascular smooth muscle cells
induced by oxidized low-density lipoprotein. These experimental
results indicated that miR-205 may be used to mitigate the
progression of atherosclerosis (49). Wang et al (79) discovered that miR-205 expression
was elevated in serum exosomes enriched from patients with
occupational dermatitis caused by TCE, and it exhibited a notable
positive association with liver function injury markers.
Furthermore, in mouse models, miR-205 was shown to target and
promote RORα protein expression, thereby exacerbating TCE-induced
liver injury. Consequently, therapeutic downregulation of miR-205
expression could potentially mitigate liver damage in these mouse
(79). Zhang et al
(67) established a nasal mucosa
of OVA-sensitized mouse model of AR and demonstrated via RT-qPCR
that miR-205 expression was upregulated in mice with AR. Notably,
miR-205 knockdown reduced nasal rubbing and sneezing frequency
while alleviating pathological changes in the nasal mucosa,
indicating that miR-205 may serve as a potential therapeutic target
for AR (67).
Conclusion
miR-205 is a multifunctional regulator involved in
a wide range of malignant and non-malignant diseases. It modulates
critical signaling pathways that control fundamental cellular
processes such as apoptosis, proliferation, migration, angiogenesis
and inflammation. The biological effects of miR-205 are highly
context-dependent, influenced by its target genes and the specific
cellular environment.
In non-malignant conditions, miR-205 serves
essential roles in tissue repair, immune-inflammatory regulation,
and maintaining epithelial and metabolic homeostasis. Its functions
can be paradoxical, for example, it alleviates fibrosis in diabetic
nephropathy but may exacerbate inflammation in psoriasis. In
cancer, miR-205 dysregulation is closely linked to disease
progression, highlighting its potential as a diagnostic and
prognostic biomarker, especially via liquid biopsy, as well as a
therapeutic target. Targeted delivery of miR-205 mimics or
inhibitors using advanced nanocarriers, including exosomes and
liposomes, offers promising therapeutic strategies. Additionally,
combining miR-205 modulation with conventional treatments may
improve therapeutic outcomes.
Overall, research on miR-205 enriches the
understanding of disease mechanisms and identifies novel
therapeutic options. Translating these findings into clinical
practice will require sustained efforts to bridge rigorous basic
research with thorough clinical validation.
Acknowledgements
Figures were generated using FigDraw (https://www.figdraw.com/).
Funding
Funding was provided by the National Natural Science Foundation
of China Regional Project (grant no. 82560297), the Inner Mongolia
Science and Technology Research Project (grant no. 2024MS08069) the
Natural Science Foundation of Inner Mongolia Autonomous Region
(grant no.2025MS08117), the Science and Technology Program of the
Joint Fund of Scientific Research for the Public Hospitals of Inner
Mongolia Academy of Medical Sciences (grant no. 2024GLLH0323), the
14th Five-Year Plan of Science and Technology Innovation in Inner
Mongolia Autonomous Region (grant no. 2022YFSH0078), the Key
Project of Inner Mongolia Medical University (grant no.
YKD2021ZD007), the Zhiyuan Talent Program of Inner Mongolia Medical
University (grant nos. ZY0202020 and ZY20242107), the Doctoral
Start-up Foundation Project of Inner Mongolia Medical University
(grant no. YKD2024BSQD026), the Undergraduate Teaching Reform
Research and Practice Project of Inner Mongolia Medical University
in 2024 (grant no. NYJXGGSJ20244046), the Inner Mongolia Medical
University 2024 Maker Cultivation Project (grant no. 101322024038),
the Inner Mongolia Medical University 2025 Cultivation of
Excellence Program in Science and Technology Innovation for
Undergraduates (grant no. YCPY2025057), the Joint Project of Inner
Mongolia Medical University (grant no. YKD2022LH049) and the
Project of the Inner Mongolia Autonomous Region Educational Science
Research ‘14th Five-Year Plan’ (grant no. NGJGH2025307).
Availability of data and materials
Not applicable.
Authors' contributions
DC was responsible for writing the first draft and
generating the figures. HD and YS are responsible for reviewing and
editing the manuscript. Data authentication is not applicable. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kumar SH: Alternative endpoints to
mortality in cancer screening trials. Mol Oncol. 18:1817–1820.
2024. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Haggstrom L, Chan WY, Nagrial A, Chantrill
LA, Sim HW, Yip D and Chin V: Chemotherapy and radiotherapy for
advanced pancreatic cancer. Cochrane Database Syst Rev.
12:CD0110442024.PubMed/NCBI
|
|
3
|
Girnyi S, Marano L, Skokowski J, Mocarski
P, Kycler W, Gallo G, Dyzmann-Sroka A, Kazmierczak-Siedlecka K,
Kalinowski L, Banasiewicz T and Polom K: Prehabilitation approaches
for gastrointestinal cancer surgery: A narrative review. Rep Pract
Oncol Radiother. 29:614–626. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gao S, Zhang Z, Ye K, Wang W, Li J, Xu T
and Tan H: Comprehensive characterization of Rubus idaeus L.
Polysaccharides: Extraction, purification, structural diversity,
biological efficacy, and structure-activity relationships. J
Ethnopharmacol. 355:1206772026. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gao S, Ye K, Zhang Z, Wang W, Li J, Xu T
and Tan H: Polysaccharides of Pseudostellaria heterophylla (Miq.)
Pax ex Pax et Hoffm: Extraction, purification, structural
characteristics, pharmacological activities, and structure-activity
relationships: A review. Int J Biol Macromol. 330:1480822025.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Jiang Y, Wang C, Zu C, Rong X, Yu Q and
Jiang J: Synergistic potential of nanomedicine in prostate cancer
immunotherapy: Breakthroughs and prospects. Int J Nanomedicine.
19:9459–9486. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Siu LL: Cancer in 2025. Cancer Discov.
15:2408–2413. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hussen BM, Hidayat HJ, Salihi A, Sabir DK,
Taheri M and Ghafouri-Fard S: MicroRNA: A signature for cancer
progression. Biomed Pharmacother. 138:1115282021. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Xiao M, Li J, Li W, Wang Y, Wu F, Xi Y,
Zhang L, Ding C, Luo H, Li Y, et al: MicroRNAs activate gene
transcription epigenetically as an enhancer trigger. RNA Biol.
14:1326–1334. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Kwan JY, Psarianos P, Bruce JP, Yip KW and
Liu FF: The complexity of microRNAs in human cancer. J Radiat Res.
57 (Suppl 1):i106–i111. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wojciechowska A, Braniewska A and
Kozar-Kamińska K: MicroRNA in cardiovascular biology and disease.
Adv Clin Exp Med. 26:865–874. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li S, Lei Z and Sun T: The role of
microRNAs in neurodegenerative diseases: A review. Cell Biol
Toxicol. 39:53–83. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Khalaf K, Hana D, Chou JT, Singh C,
Mackiewicz A and Kaczmarek M: Aspects of the tumor microenvironment
involved in immune resistance and drug resistance. Front Immunol.
12:6563642021. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Zhu Z, Liang L, Zhang R, Wei Y, Su L,
Tejera P, Guo Y, Wang Z, Lu Q, Baccarelli AA, et al: Whole blood
microRNA markers are associated with acute respiratory distress
syndrome. Intensive Care Med Exp. 5:382017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Schultz NA, Dehlendorff C, Jensen BV,
Bjerregaard JK, Nielsen KR, Bojesen SE, Calatayud D, Nielsen SE,
Yilmaz M, Holländer NH, et al: MicroRNA biomarkers in whole blood
for detection of pancreatic cancer. JAMA. 311:392–404. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Plantamura I, Cataldo A, Cosentino G and
Iorio MV: miR-205 in breast cancer: State of the Art. Int J Mol
Sci. 22:272020. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Qin AY, Zhang XW, Liu L, Yu JP, Li H, Wang
SZ, Ren XB and Cao S: MiR-205 in cancer: An angel or a devil? Eur J
Cell Biol. 92:54–60. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wienholds E, Kloosterman WP, Miska E,
Alvarez-Saavedra E, Berezikov E, de Bruijn E, Horvitz HR, Kauppinen
S and Plasterk RH: MicroRNA expression in zebrafish embryonic
development. Science. 309:310–311. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Landgraf P, Rusu M, Sheridan R, Sewer A,
Iovino N, Aravin A, Pfeffer S, Rice A, Kamphorst AO, Landthaler M,
et al: A mammalian microRNA expression atlas based on small RNA
library sequencing. Cell. 129:1401–1414. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Jiang M, Zhong T, Zhang W, Xiao Z, Hu G,
Zhou H and Kuang H: Reduced expression of miR-205-5p promotes
apoptosis and inhibits proliferation and invasion in lung cancer
A549 cells by upregulation of ZEB2 and downregulation of erbB3. Mol
Med Rep. 15:3231–3238. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang D, Zhang Z, O'Loughlin E, Wang L, Fan
X, Lai EC and Yi R: MicroRNA-205 controls neonatal expansion of
skin stem cells by modulating the PI(3)K pathway. Nat Cell Biol.
15:1153–1163. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jiang D, Guo B, Lin F, Lin S and Tao K:
miR-205 inhibits the development of hypertrophic scars by targeting
THBS1. Aging (Albany NY). 12:22046–22058. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Qi J, Liu Y, Hu K, Zhang Y, Wu Y and Zhang
X: MicroRNA-205-5p regulates extracellular matrix production in
hyperplastic scars by targeting Smad2. Exp Ther Med. 17:2284–2290.
2019.PubMed/NCBI
|
|
24
|
Salmena L, Poliseno L, Tay Y, Kats L and
Pandolfi PP: A ceRNA hypothesis: The Rosetta stone of a hidden RNA
language? Cell. 146:353–358. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Su X, Ma Y, Wang Q and Gao Y: LncRNA
HOXA11-AS aggravates keloid progression by the regulation of
HOXA11-AS-miR-205-5p-FOXM1 pathway. J Surg Res. 259:284–295. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang T, Zhao N, Long S, Ge L, Wang A, Sun
H, Ran X, Zou Z, Wang J and Su Y: Downregulation of miR-205 in
migrating epithelial tongue facilitates skin wound
re-epithelialization by derepressing ITGA5. Biochim Biophys Acta.
1862:1443–1452. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin D, Halilovic A, Yue P, Bellner L, Wang
K, Wang L and Zhang C: Inhibition of miR-205 impairs the
wound-healing process in human corneal epithelial cells by
targeting KIR4.1 (KCNJ10). Invest Ophthalmol Vis Sci. 54:6167–6178.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Greene SB, Gunaratne PH, Hammond SM and
Rosen JM: A putative role for microRNA-205 in mammary epithelial
cell progenitors. J Cell Sci. 123:606–618. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Yu SL, Jeong DU, Noh EJ, Jeon HJ, Lee DC,
Kang M, Kim TH, Lee SK, Han AR, Kang J and Park SR: Exosomal
miR-205-5p improves endometrial receptivity by upregulating
E-cadherin expression through ZEB1 inhibition. Int J Mol Sci.
24:151492023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Langlet F, Tarbier M, Haeusler RA,
Camastra S, Ferrannini E, Friedländer MR and Accili D:
microRNA-205-5p is a modulator of insulin sensitivity that inhibits
FOXO function. Mol Metab. 17:49–60. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang L, Xi M, Cao W, Qin H, Qin D, Chen S,
Zhou S, Hou Y, Chen Y, Xiao X, et al: Electroacupuncture alleviates
functional constipation by upregulating host-derived miR-205-5p to
modulate gut microbiota and tryptophan metabolism. Front Microbiol.
16:15170182025. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Hu N, Feng C, Jiang Y, Miao Q and Liu H:
regulative effect of Mir-205 on osteogenic differentiation of bone
mesenchymal stem cells (BMSCs): Possible role of SATB2/Runx2 and
ERK/MAPK pathway. Int J Mol Sci. 16:10491–10506. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Ferrari E and Gandellini P: Unveiling the
ups and downs of miR-205 in physiology and cancer: Transcriptional
and post-transcriptional mechanisms. Cell Death Dis. 11:9802020.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Piovan C, Palmieri D, Di Leva G, Braccioli
L, Casalini P, Nuovo G, Tortoreto M, Sasso M, Plantamura I, Triulzi
T, et al: Oncosuppressive role of p53-induced miR-205 in triple
negative breast cancer. Mol Oncol. 6:458–472. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Tran MN, Choi W, Wszolek MF, Navai N, Lee
IL, Nitti G, Wen S, Flores ER, Siefker-Radtke A, Czerniak B, et al:
The p63 protein isoform ΔNp63α inhibits epithelial-mesenchymal
transition in human bladder cancer cells: Role of MIR-205. J Biol
Chem. 288:3275–3288. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Pan F, Mao H, Bu F, Tong X, Li J, Zhang S,
Liu X, Wang L, Wu L, Chen R, et al: Sp1-mediated transcriptional
activation of miR-205 promotes radioresistance in esophageal
squamous cell carcinoma. Oncotarget. 8:5735–5752. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wiklund ED, Bramsen JB, Hulf T, Dyrskjøt
L, Ramanathan R, Hansen TB, Villadsen SB, Gao S, Ostenfeld MS,
Borre M, et al: Coordinated epigenetic repression of the miR-200
family and miR-205 in invasive bladder cancer. Int J Cancer.
128:1327–1334. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Esteller M: CpG island hypermethylation
and tumor suppressor genes: A booming present, a brighter future.
Oncogene. 21:5427–5440. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lövkvist C, Dodd IB, Sneppen K and Haerter
JO: DNA methylation in human epigenomes depends on local topology
of CpG sites. Nucleic Acids Res. 44:5123–5132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wang Z, Wu J, Zhang G, Cao Y, Jiang C and
Ding Y: Associations of miR-499 and miR-34b/c polymorphisms with
susceptibility to hepatocellular carcinoma: An Evidence-based
evaluation. Gastroenterol Res Pract. 2013:7192022013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Hasegawa T, Adachi R, Iwakata H, Takeno T,
Sato K and Sakamaki T: ErbB2 signaling epigenetically suppresses
microRNA-205 transcription via Ras/Raf/MEK/ERK pathway in breast
cancer. FEBS Open Bio. 7:1154–1165. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Mancini M, Grasso M, Muccillo L, Babbio F,
Precazzini F, Castiglioni I, Zanetti V, Rizzo F, Pistore C, De
Marino MG, et al: DNMT3A epigenetically regulates key microRNAs
involved in epithelial-to-mesenchymal transition in prostate
cancer. Carcinogenesis. 42:1449–1460. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Tellez CS, Juri DE, Do K, Bernauer AM,
Thomas CL, Damiani LA, Tessema M, Leng S and Belinsky SA: EMT and
stem cell-like properties associated with miR-205 and miR-200
epigenetic silencing are early manifestations during
carcinogen-induced transformation of human lung epithelial cells.
Cancer Res. 71:3087–3097. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Wang H, Li J, Tao L, Lv L, Sun J, Zhang T,
Wang H and Wang J: MiR-205 regulates LRRK2 expression in dopamine
neurons in Parkinson's disease through methylation modification.
Iran J Public Health. 51:1637–1647. 2022.PubMed/NCBI
|
|
45
|
Vaz CO, Hounkpe BW, Oliveira JD, Mazetto
B, Cardoso Jacintho B, Aparecida Locachevic G, Henrique De Oliveira
Soares K, Carlos Silva Mariolano J, Castilho de Mesquita G,
Colombera Peres K, et al: MicroRNA 205-5p and COVID-19 adverse
outcomes: Potential molecular biomarker and regulator of the immune
response. Exp Biol Med (Maywood). 248:1024–1033. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Feng L, Wei J, Liang S, Sun Z and Duan J:
miR-205/IRAK2 signaling pathway is associated with urban airborne
PM(2.5)-induced myocardial toxicity. Nanotoxicology. 14:1198–1212.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Xiao Z, Reddy DPK, Xue C, Liu X, Chen X,
Li J, Ling X and Zheng S: Profiling of miR-205/P4HA3 following
angiotensin II-induced atrial fibrosis: Implications for atrial
fibrillation. Front Cardiovasc Med. 8:6093002021. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Liu J, Li W, Wang J, Bai L, Xu J, Chen X,
Wang S, Li L and Xu X: IL-32 regulates trophoblast invasion through
miR-205-NFκB-MMP2/9 axis contributing to the pregnancy-induced
hypertension. Biol Reprod. 111:780–799. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Huang P, Wang F, Zhang Y, Zhang Y, Qin M,
Ji J, Wei D and Ren L: Icariin alleviates atherosclerosis by
regulating the miR-205-5p/ERBB4/AKT signaling pathway. Int
Immunopharmacol. 114:1096112023. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Huang X, Li Z, Zhang L, Yang Y, Wang Y, Li
S, Li G, Feng H and Yang X: miR-205-5p inhibits
homocysteine-induced pulmonary microvascular endothelium
dysfunction by targeting FOXO1. Acta Biochim Biophys Sin
(Shanghai). 55:1456–1466. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Xiao Z, Xie Y, Huang F, Yang J, Liu X, Lin
X, Zhu P and Zheng S: MicroRNA-205-5p plays a suppressive role in
the high-fat diet-induced atrial fibrosis through regulation of the
EHMT2/IGFBP3 axis. Genes Nutr. 17:112022. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Liu J, Wang J, Fu W, Wang X, Chen H, Wu X,
Lao G, Wu Y, Hu M, Yang C, et al: MiR-195-5p and miR-205-5p in
extracellular vesicles isolated from diabetic foot ulcer wound
fluid decrease angiogenesis by inhibiting VEGFA expression. Aging.
13:19805–19821. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Zhu L, Wang G, Fischbach S and Xiao X:
Suppression of microRNA-205-5p in human mesenchymal stem cells
improves their therapeutic potential in treating diabetic foot
disease. Oncotarget. 8:52294–52303. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zheng Z, Zhang S, Chen J, Zou M, Yang Y,
Lu W, Ren S, Wang X, Dong W, Zhang Z, et al: The HDAC2/SP1/miR-205
feedback loop contributes to tubular epithelial cell extracellular
matrix production in diabetic kidney disease. Clin Sci (Lond).
136:223–238. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Ouni M, Gottmann P, Westholm E, Schwerbel
K, Jähnert M, Stadion M, Rittig K, Vogel H and Schürmann A: MiR-205
is up-regulated in islets of diabetes-susceptible mice and targets
the diabetes gene Tcf7l2. Acta Physiol (Oxf). 232:e136932021.
View Article : Google Scholar : PubMed/NCBI
|
|
56
|
He J, Li D, Wei J, Wang S, Chu S, Zhang Z,
He F, Wei D, Li Y, Xie J, et al: Mahonia Alkaloids (MA) Ameliorate
depression induced gap junction dysfunction by miR-205/Cx43 axis.
Neurochem Res. 47:3761–3776. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Cascella M and Bimonte S: The role of
general anesthetics and the mechanisms of hippocampal and
extra-hippocampal dysfunctions in the genesis of postoperative
cognitive dysfunction. Neural Regen Res. 12:1780–1785. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Zhang Y and Chen C: Knockdown of lncRNA
NKILA suppresses sevoflurane-induced neuronal cell injury partially
by targeting miR-205-5p/ELAVL1 axis. Gen Physiol Biophys.
42:285–295. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Yang JJ, Zhao YH, Yin KW, Zhang XQ and Liu
J: Dexmedetomidine inhibits inflammatory response and oxidative
stress through regulating miR-205-5p by targeting HMGB1 in cerebral
ischemic/reperfusion. Immunopharmacol Immunotoxicol. 43:478–486.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Huang M, Li X, Zhou C, Si M, Zheng H, Chen
L and Ding H: Noncoding RNA miR-205-5p mediates osteoporosis
pathogenesis and osteoblast differentiation by regulating RUNX2. J
Cell Biochem. 121:4196–4203. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Ma W, Tang F, Xiao L, Han S, Yao X, Zhang
Q and Zhou J, Wang Y and Zhou J: miR-205-5p in exosomes divided
from chondrogenic mesenchymal stem cells alleviated rheumatoid
arthritis via regulating MDM2 in fibroblast-like synoviocytes. J
Musculoskelet Neuronal Interact. 22:132–141. 2022.PubMed/NCBI
|
|
62
|
Zhu M, Yan X, Zhao Y, Xue H, Wang Z, Wu B,
Li X and Shen Y: lncRNA LINC00284 promotes nucleus pulposus cell
proliferation and ECM synthesis via regulation of the
miR-205-3p/Wnt/β-catenin axis. Mol Med Rep. 25:1792022. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Jiang F, Zhou Y, Zhang R and Wen Y:
miR-205 and HMGB1 expressions in chronic periodontitis patients and
their associations with the inflammatory factors. Am J Transl Res.
13:9224–9232. 2021.PubMed/NCBI
|
|
64
|
Su X, Zhang J and Qin X: CD40
up-regulation on dendritic cells correlates with Th17/Treg
imbalance in chronic periodontitis in young population. Innate
Immun. 26:482–489. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Kang L, Miao Y, Jin Y, Shen S and Lin X:
Exosomal miR-205-5p derived from periodontal ligament stem cells
attenuates the inflammation of chronic periodontitis via targeting
XBP1. Immun Inflamm Dis. 11:e7432023. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Suojalehto H, Toskala E, Kilpeläinen M,
Majuri ML, Mitts C, Lindström I, Puustinen A, Plosila T, Sipilä J,
Wolff H, et al: MicroRNA profiles in nasal mucosa of patients with
allergic and nonallergic rhinitis and asthma. Int Forum Allergy
Rhinol. 3:612–620. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Zhang S, Lin S, Tang Q and Yan Z:
Knockdown of miR-205-5p alleviates the inflammatory response in
allergic rhinitis by targeting B-cell lymphoma 6. Mol Med Rep.
24:8182021. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Zheng J, Chen X, Zhan JB, Li CW, Wei X and
Jiang HY: CircARF3 mitigates allergic rhinitis through targeting
microRNA-205-5p/Sirtuin 5 Axis. Int Arch Allergy Immunol.
184:1056–1070. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Kim CW, Kumar S, Son DJ, Jang IH,
Griendling KK and Jo H: Prevention of abdominal aortic aneurysm by
anti-microRNA-712 or anti-microRNA-205 in angiotensin II-infused
mice. Arterioscler Thromb Vasc Biol. 34:1412–1421. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Zhou W, Wang J, Li Z, Li J and Sang M:
MicroRNA-205-5b inhibits HMGB1 expression in LPS-induced sepsis.
Int J Mol Med. 38:312–318. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Zhao F, Pan C, Zhang Y, Yang J and Xing X:
Polyphyllin VII alleviates pulmonary hypertension by inducing
miR-205-5p to target the β-catenin pathway. Biomed Pharmacother.
167:1155162023. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Wang J, Zhang Y and Zhang L: Long
non-coding RNA SNHG5 suppresses the development of acute
respiratory distress syndrome by targeting miR-205/COMMD1 axis. Mol
Cell Biochem. 476:1063–1074. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
73
|
Sun B, Xu S, Yan Y, Li Y, Li H, Zheng G,
Dong T and Bai J: miR-205 suppresses pulmonary fibrosis by
targeting GATA3 through inhibition of endoplasmic reticulum stress.
Curr Pharm Biotechnol. 21:720–726. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Qian Q, Ma Q, Wang B, Qian Q, Zhao C, Feng
F and Dong X: MicroRNA-205-5p targets E2F1 to promote autophagy and
inhibit pulmonary fibrosis in silicosis through impairing
SKP2-mediated Beclin1 ubiquitination. J Cell Mol Med. 25:9214–9227.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Zhao Y, Wang H, Tang Y, Wang J, Wu X, He
Z, He Y and Tang Z: SNHG16/miR-205/HDAC5 is involved in the
progression of renal fibrosis. J Biochem Mol Toxicol.
38:e236172024. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Zhang Y, Xia F, Wu J, Yang AX, Zhang YY,
Zhao H and Tao WY: MiR-205 influences renal injury in sepsis rats
through HMGB1-PTEN signaling pathway. Eur Rev Med Pharmacol Sci.
23:10950–10956. 2019.PubMed/NCBI
|
|
77
|
Zhou F, Liu D, Ye J and Li B: Circ_0006944
aggravates LPS-induced HK2 cell injury via modulating
miR-205-5p/UBL4A pathway. Autoimmunity. 56:22760662023. View Article : Google Scholar : PubMed/NCBI
|
|
78
|
Xue Y, Liu Y, Bian X, Zhang Y, Li Y, Zhang
Q and Yin M: miR-205-5p inhibits psoriasis-associated proliferation
and angiogenesis: Wnt/β-catenin and mitogen-activated protein
kinase signaling pathway are involved. J Dermatol. 47:882–892.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Wang H, Wang F, Li Y, Zhou P, Cai S, Wu Q,
Ding T, Wu C and Zhu Q: Exosomal miR-205-5p contributes to the
immune liver injury induced by trichloroethylene: Pivotal role of
RORα mediating M1 Kupffer cell polarization. Ecotoxicol Environ
Saf. 285:1170502024. View Article : Google Scholar : PubMed/NCBI
|
|
80
|
Hu Y, Ye H and Shi LX: MicroRNA-205
ameliorates lipid accumulation in non-alcoholic fatty liver disease
through targeting NEU1. Eur Rev Med Pharmacol Sci. 23:10072–10082.
2019.PubMed/NCBI
|
|
81
|
Fang L, Wang HF, Chen YM, Bai RX and Du
SY: Baicalin confers hepatoprotective effect against
alcohol-associated liver disease by upregulating microRNA-205. Int
Immunopharmacol. 107:1085532022. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Smith CM, Michael MZ, Watson DI, Tan G,
Astill DS, Hummel R and Hussey DJ: Impact of gastro-oesophageal
reflux on microRNA expression, location and function. BMC
Gastroenterol. 13:42013. View Article : Google Scholar : PubMed/NCBI
|
|
83
|
Bao Y, Shi Y, Zhou L, Gao S, Yao R, Guo S,
Geng Z, Bao L, Zhao R and Cui X: MicroRNA-205-5p: A potential
therapeutic target for influenza A. J Cell Mol Med. 26:5917–5928.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
84
|
Han X, Yuan Z, Jing Y, Zhou W, Sun Y and
Xing J: Knockdown of lncRNA TapSAKI alleviates LPS-induced injury
in HK-2 cells through the miR-205/IRF3 pathway. Open Med (Wars).
16:581–590. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Jang SJ, Choi IS, Park G, Moon DS, Choi
JS, Nam MH, Yoon SY, Choi CH and Kang SH: MicroRNA-205-5p is
upregulated in myelodysplastic syndromes and induces cell
proliferation via PTEN suppression. Leuk Res. 47:172–177. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
86
|
Chauhan N, Dhasmana A, Jaggi M, Chauhan SC
and Yallapu MM: miR-205: A potential biomedicine for cancer
therapy. Cells. 9:19572020. View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Majid S, Saini S, Dar AA, Hirata H,
Shahryari V, Tanaka Y, Yamamura S, Ueno K, Zaman MS, Singh K, et
al: MicroRNA-205 inhibits Src-mediated oncogenic pathways in renal
cancer. Cancer Res. 71:2611–2621. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Kalogirou C, Linxweiler J, Schmucker P,
Snaebjornsson MT, Schmitz W, Wach S, Krebs M, Hartmann E, Puhr M,
Müller A, et al: MiR-205-driven downregulation of cholesterol
biosynthesis through SQLE-inhibition identifies therapeutic
vulnerability in aggressive prostate cancer. Nat Commun.
12:50662021. View Article : Google Scholar : PubMed/NCBI
|
|
89
|
Xu Y, Brenn T, Brown ER, Doherty V and
Melton DW: Differential expression of microRNAs during melanoma
progression: miR-200c, miR-205 and miR-211 are downregulated in
melanoma and act as tumour suppressors. Br J Cancer. 106:553–561.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
90
|
Lu LG and Zhang GM: Serum miR-205-5p level
for non-small-cell lung cancer diagnosis. Thorac Cancer.
13:1102–1103. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
91
|
Ganji SM, Saidijam M, Amini R,
Mousavi-Bahar SH, Shabab N, Seyedabadi S and Mahdavinezhad A:
Evaluation of MicroRNA-99a and MicroRNA-205 expression levels in
bladder cancer. Int J Mol Cell Med. 6:87–95. 2017.PubMed/NCBI
|
|
92
|
Wei J, Zhang L, Li J, Zhu S, Tai M, Mason
CW, Chapman JA, Reynolds EA, Weiner CP and Zhou HH: MicroRNA-205
promotes cell invasion by repressing TCF21 in human ovarian cancer.
J Ovarian Res. 10:332017. View Article : Google Scholar : PubMed/NCBI
|
|
93
|
Zhang LM, Su LX, Hu JZ, Wang M, Ju HY, Li
X, Han YF, Xia WY, Guo W, Ren GX and Fan XD: Epigenetic regulation
of VENTXP1 suppresses tumor proliferation via
miR-205-5p/ANKRD2/NF-kB signaling in head and neck squamous cell
carcinoma. Cell Death Dis. 11:8382020. View Article : Google Scholar : PubMed/NCBI
|
|
94
|
Hezova R, Kovarikova A, Srovnal J,
Zemanova M, Harustiak T, Ehrmann J, Hajduch M, Sachlova M, Svoboda
M and Slaby O: MiR-205 functions as a tumor suppressor in
adenocarcinoma and an oncogene in squamous cell carcinoma of
esophagus. Tumour Biol. 37:8007–8018. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
95
|
Cataldo A, Cheung DG, Hagan JP, Fassan M,
Sandhu-Deol S, Croce CM, Di Leva G and Iorio MV: Genetic loss of
miR-205 causes increased mammary gland development. Noncoding RNA.
10:42023.PubMed/NCBI
|
|
96
|
Xiao Y, Humphries B, Yang C and Wang Z:
MiR-205 Dysregulations in breast cancer: The complexity and
opportunities. Noncoding RNA. 5:532019.PubMed/NCBI
|
|
97
|
Kalinkova L, Nikolaieva N, Smolkova B,
Ciernikova S, Kajo K, Bella V, Kajabova VH, Kosnacova H, Minarik G
and Fridrichova I: miR-205-5p Downregulation and ZEB1 upregulation
characterize the disseminated tumor cells in patients with invasive
ductal breast cancer. Int J Mol Sci. 23:1032021. View Article : Google Scholar : PubMed/NCBI
|
|
98
|
Shen Y, Xu Y, Huang L, Chi Y and Meng L:
MiR-205 suppressed the malignant behaviors of breast cancer cells
by targeting CLDN11 via modulation of the epithelial-to-mesenchymal
transition. Aging. 13:13073–13086. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
99
|
Ma X, Wang N, Chen K and Zhang C:
Oncosuppressive role of MicroRNA-205-3p in gastric cancer through
inhibition of proliferation and induction of senescence:
Oncosuppressive role of MicroRNA-205 in gastric cancer. Transl
Oncol. 14:1011992021. View Article : Google Scholar : PubMed/NCBI
|
|
100
|
Wang X, Zhang H, Jiao K, Zhao C, Liu H,
Meng Q, Wang Z, Feng C and Li Y: Effect of miR-205 on proliferation
and migration of thyroid cancer cells by targeting CCNB2 and the
mechanism. Oncol Lett. 19:2568–2574. 2020.PubMed/NCBI
|
|
101
|
Wang H, Chen B, Duan B, Zheng J and Wu X:
miR-205 suppresses cell proliferation, invasion, and metastasis via
regulation of the PTEN/AKT pathway in renal cell carcinoma. Mol Med
Rep. 14:3343–3349. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
102
|
Qiao B, Wang Q, Zhao Y and Wu J:
miR-205-3p functions as a tumor suppressor in ovarian carcinoma.
Reprod Sci. 27:380–388. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
103
|
Cai KQ, Yang WL, Capo-Chichi CD,
Vanderveer L, Wu H, Godwin AK and Xu XX: Prominent expression of
metalloproteinases in early stages of ovarian tumorigenesis. Mol
Carcinog. 46:130–143. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
104
|
Wang Y, Qin C, Zhao Y, Zhao B, Li Z, Li T,
Zhang X and Wang W: Extracellular vesicles-miR-205-5p inhibits
lymphatic metastasis in pancreatic cancer through diffusely
downregulating VEGFA. J Cancer. 16:2197–2211. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
105
|
Ma Q, Wan G, Wang S, Yang W, Zhang J and
Yao X: Serum microRNA-205 as a novel biomarker for cervical cancer
patients. Cancer Cell Int. 14:812014. View Article : Google Scholar : PubMed/NCBI
|
|
106
|
Xie H, Zhao Y, Caramuta S, Larsson C and
Lui WO: miR-205 expression promotes cell proliferation and
migration of human cervical cancer cells. PLoS One. 7:e469902012.
View Article : Google Scholar : PubMed/NCBI
|
|
107
|
Witten D, Tibshirani R, Gu SG, Fire A and
Lui WO: Ultra-high throughput sequencing-based small RNA discovery
and discrete statistical biomarker analysis in a collection of
cervical tumours and matched controls. BMC Biol. 8:582010.
View Article : Google Scholar : PubMed/NCBI
|
|
108
|
Liu J, Li Y, Chen X, Xu X, Zhao H, Wang S,
Hao J, He B, Liu S and Wang J: Upregulation of miR-205 induces CHN1
expression, which is associated with the aggressive behaviour of
cervical cancer cells and correlated with lymph node metastasis.
BMC Cancer. 20:10292020. View Article : Google Scholar : PubMed/NCBI
|
|
109
|
Guo Z, Zhu H, Zhang R, Shan Q and Wang Y:
miR-205-5p promotes the proliferation, migration, and invasion of
nasopharyngeal carcinoma cells by regulating CALM1. Crit Rev
Immunol. 45:25–38. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
110
|
Yang W, Tan S, Yang L, Chen X, Yang R,
Oyang L, Lin J, Xia L, Wu N, Han Y, et al: Exosomal miR-205-5p
enhances angiogenesis and nasopharyngeal carcinoma metastasis by
targeting desmocollin-2. Mol Ther Oncolytics. 24:612–623. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
111
|
Yi L, He S, Cheng Z, Chen X, Ren X and Bai
Y: DNAJA1 stabilizes EF1A1 to promote cell proliferation and
metastasis of liver cancer mediated by miR-205-5p. J Oncol.
2022:22924812022. View Article : Google Scholar : PubMed/NCBI
|
|
112
|
Xu LB, Xiong J, Zhang YH, Dai Y, Ren XP,
Ren YJ, Han D, Wei SH and Qi M: miR-205-3p promotes lung cancer
progression by targeting APBB2. Mol Med Rep. 24:5882021. View Article : Google Scholar : PubMed/NCBI
|
|
113
|
Gautam A, Kumar R, Dimitrov G, Hoke A,
Hammamieh R and Jett M: Identification of extracellular miRNA in
archived serum samples by next-generation sequencing from RNA
extracted using multiple methods. Mol Biol Rep. 43:1165–1178. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
114
|
Brandenburger T and Lorenzen JM:
Diagnostic and therapeutic potential of microRNAs in acute kidney
injury. Front Pharmacol. 11:6572020. View Article : Google Scholar : PubMed/NCBI
|
|
115
|
Backes C, Meese E and Keller A: Specific
miRNA disease biomarkers in blood, serum and plasma: Challenges and
prospects. Mol Diagn Ther. 20:509–518. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
116
|
Re M, Tomasetti M, Monaco F, Amati M,
Rubini C, Sollini G, Bajraktari A, Gioacchini FM, Santarelli L and
Pasquini E: MiRNome analysis identifying miR-205 and miR-449a as
biomarkers of disease progression in intestinal-type sinonasal
adenocarcinoma. Head Neck. 44:18–33. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
117
|
Li JH, Sun SS, Li N, Lv P, Xie SY and Wang
PY: MiR-205 as a promising biomarker in the diagnosis and prognosis
of lung cancer. Oncotarget. 8:91938–91949. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
118
|
Zhao Y, Zhao Y, Liu L, Li G, Wu Y, Cui Y
and Xie L: Tumor-exosomal miR-205-5p as a diagnostic biomarker for
colorectal cancer. Clin Transl Oncol. 27:1185–1197. 2025.
View Article : Google Scholar : PubMed/NCBI
|
|
119
|
Mills J, Capece M, Cocucci E, Tessari A
and Palmieri D: Cancer-derived extracellular vesicle-associated
MicroRNAs in intercellular communication: One Cell's trash is
another Cell's treasure. Int J Mol Sci. 20:61092019. View Article : Google Scholar : PubMed/NCBI
|
|
120
|
Bang OY, Kim EH, Oh MJ, Yoo J, Oh GS,
Chung JW, Seo WK, Kim GM, Ahn MJ and Yang SW; Investigators of the
OASIS-CANCER Study, : Circulating
extracellular-vesicle-incorporated MicroRNAs as potential
biomarkers for ischemic stroke in patients with cancer. J Stroke.
25:251–265. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
121
|
Wang X, Jiang X, Li J, Wang J, Binang H,
Shi S, Duan W, Zhao Y and Zhang Y: Serum exosomal miR-1269a serves
as a diagnostic marker and plays an oncogenic role in non-small
cell lung cancer. Thorac Cancer. 11:3436–3447. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
122
|
Zhou J, Cao L and Chen Z: Differentiation
of benign thyroid nodules from malignant thyroid nodules through
miR-205-5p and thyroid-stimulating hormone receptor mRNA. Hormones
(Athens). 20:571–580. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
123
|
Wu Z, Tang H, Xiong Q, Liu D, Xia T, Liang
H and Ye Q: Prognostic role of microRNA-205 in human gynecological
cancer: A meta-analysis of fourteen studies. DNA Cell Biol.
39:875–889. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
124
|
Lu J, Lin Y, Li F, Ye H, Zhou R, Jin Y, Li
B, Xiong X and Cheng N: MiR-205 suppresses tumor growth, invasion,
and epithelial-mesenchymal transition by targeting SEMA4C in
hepatocellular carcinoma. FASEB J. fj201800113R2018.doi:
10.1096/fj.201800113R (Epub ahead of print). PubMed/NCBI
|
|
125
|
Lin LF, Li YT, Han H and Lin SG:
MicroRNA-205-5p targets the HOXD9-Snail1 axis to inhibit triple
negative breast cancer cell proliferation and chemoresistance.
Aging. 13:3945–3956. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
126
|
Li M, Li ZH, Song J, Li X, Zhai P, Mu X,
Qiu F and Yao L: miR-205 reverses MDR-1 mediated doxorubicin
resistance via PTEN in human liver cancer HepG2 cells. Cell J.
24:112–119. 2022.PubMed/NCBI
|
|
127
|
Ouyang B, Bi M, Jadhao M, Bick G and Zhang
X: miR-205 regulates tamoxifen resistance by targeting estrogen
receptor coactivator MED1 in human breast cancer. Cancers.
16:39922024. View Article : Google Scholar : PubMed/NCBI
|
|
128
|
Li FF, Xing C, Wu LL and Xue F: MiR-205
enhances cisplatin sensitivity of glioma cells by targeting E2F1.
Eur Rev Med Pharmacol Sci. 22:299–306. 2018.PubMed/NCBI
|
|
129
|
Zhang GF, Wu JC, Wang HY, Jiang WD and Qiu
L: Overexpression of microRNA-205-5p exerts suppressive effects on
stem cell drug resistance in gallbladder cancer by down-regulating
PRKCE. Biosci Rep. 40:BSR201945092020. View Article : Google Scholar : PubMed/NCBI
|
|
130
|
Sadhukhan P, Kundu M, Chatterjee S, Ghosh
N, Manna P, Das J and Sil PC: Targeted delivery of quercetin via
pH-responsive zinc oxide nanoparticles for breast cancer therapy.
Mater Sci Eng C Mater Biol Appl. 100:129–140. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
131
|
Vekariya KK, Kaur J and Tikoo K: ERα
signaling imparts chemotherapeutic selectivity to selenium
nanoparticles in breast cancer. Nanomedicine. 8:1125–1132. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
132
|
Razzaq F, Shahid S and Shahid W:
Modulation of miR-205 expression using a Cheiranthus cheiri
phyto-nano hybrid as a potential therapeutic agent against breast
cancer. RSC Adv. 14:37286–37298. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
133
|
Lin CY, Fang JY, Hsiao CY, Lee CW,
Alshetaili A and Lin ZC: Dual cell-penetrating peptide-conjugated
polymeric nanocarriers for miRNA-205-5p delivery in gene therapy of
cutaneous squamous cell carcinoma. Acta Biomater. 196:332–349.
2025. View Article : Google Scholar : PubMed/NCBI
|
|
134
|
Zhang HY, Zhang QY, Liu Q, Feng SG, Ma Y,
Wang FS, Zhu Y, Yao J and Yan B: Exosome-loading miR-205: A
two-pronged approach to ocular neovascularization therapy. J
Nanobiotechnol. 23:362025. View Article : Google Scholar
|
|
135
|
Ybarra M, Martínez-Santos M, Oltra M,
Muriach M, Pires ME, Ceresoni C, Sancho-Pelluz J and Barcia JM:
miR-205-5p modulates high glucose-induced VEGFA levels in diabetic
mice and ARPE-19 cells. Antioxidants (Basel). 14:2182025.
View Article : Google Scholar : PubMed/NCBI
|
|
136
|
Rubini A, Zanotti F, Licastro D, Calogero
G, Bettini G, Piccoli C, Rubini G, Lovatti L and Zavan B:
Therapeutic potential of feline adipose-derived stem cell exosomes
in the treatment of feline idiopathic cystitis: A characterization
and functional analysis of miRNA content. Nanotheranostics.
9:38–51. 2025. View Article : Google Scholar : PubMed/NCBI
|
|
137
|
Zhou CF, Liu MJ, Wang W, Wu S, Huang YX,
Chen GB, Liu LM, Peng DX, Wang XF, Cai XZ, et al: miR-205-5p
inhibits human endometriosis progression by targeting ANGPT2 in
endometrial stromal cells. Stem Cell Res Ther. 10:2872019.
View Article : Google Scholar : PubMed/NCBI
|
















